Key note presentation: Methods to improve the reproductive of camel Julian A. Skidmore Camel Reproduction Centre, Dubai, United Arab Emirates
|
|
|
- Deirdre Stone
- 5 years ago
- Views:
Transcription
1 Key note presentation: Methods to improve the reproductive of camel Julian A. Skidmore Camel Reproduction Centre, Dubai, United Arab Emirates ABSTRACT The reproductive performance of camels is generally regarded to be low due (40%). This could be due to the late age of reaching puberty (3-4 years), the long gestation period (13 months) and the relatively high incidence of abortions and non-conceptions possibly attributed to poor nutrition, poor management, limited breeding opportunities for the females due to seasonality of breeding and limited rutting potential of a single stud bull used to service too many females. Techniques such as artificial insemination and embryo transfer are therefore becoming increasingly important. Artificial insemination increases the productivity of male animals. It has been reported in camelids although insemination trials are rare, probably because of the difficulties involved in collecting and handling the semen due to the gelatinous nature of the seminal plasma. In addition, female camelids are induced ovulators, and therefore ovulation must be induced before insemination. This paper discusses different methods for collecting and handling camel semen and describes how to measure sperm concentration and morphology. It also examines the use of different buffers for liquid storage of fresh and chilled semen, the ideal number of live sperm to inseminate where to inseminate them and how to induce ovulation in the females. Embryo transfer offers the chance to greatly increase the productivity of female animals and thereby increase the rate of genetic improvement. This paper discusses the essential prerequisites of embryo transfer programmes, such as superovulation in donors and synchronising groups of recipient animals, as well as the recent advances in chilling and freezing of camel embryos. Key words: Camel, Improve, Reproduction INTRODUCTION Opportunities to improve reproductive performance in camels are limited by the long gestation period and the short breeding season, as well as the continuing use of traditional systems of reproductive management in most breeding herds. These age-old methods make it difficult to ensure an optimum number of females are pregnant at the end of the season and they can also lead to widespread venereal infections with consequent lowering of fertility. This low reproductive efficiency could be improved by better understanding of the reproductive cycle and increased use of assisted reproduction techniques such as artificial insemination and embryo transfer. This paper outlines methods that can be used to control the reproductive cycle and increase breeding efficiency. Mating at the correct time of the follicular wave cycle: Camels are induced ovulators and therefore normally only ovulate when mated. Thus follicles have a period of growth, maturity and regression, so it is more accurate to describe the changes in follicular development as a follicular wave pattern rather than 77
2 oestrous cycle. As oestrous behaviour in camels is highly variable (Musa and Abusineina, 1978), signs of oestrus are difficult to relate to follicular activity in the ovaries and cannot be used reliably to decide timing of breeding in camels. Ultrasonography of the ovaries is therefore regarded as the most reliable method to monitor follicular growth and determine the correct time for mating. It is very important that camels are mated at the correct stage of the follicular cycle to maximize the chances of ovulation and conception. Previous studies have shown that mating will induce ovulation in female camels when the dominant follicle measures between cm in diameter, but the ovulation rate is dramatically reduced to < 20% if the follicle diameter increases to cm and no follicle >3.0 cm ovulated (Skidmore et al., 1996). More recent studies indicate that the best time to mate or inseminate the camel to maximize the chances of conception is when the mature follicle measures between cm in diameter (Skidmore and Billah, 2006a). Artificial insemination Advantages Artificial insemination has several advantages. For example i) it allows more efficient use of genetically superior males by inseminating more than one female with a single ejaculate, ii) it eliminates the need to transport the male or female animal to the stud, as it is much easier to transport semen. This reduces the cost and risks of transporting valuable animals, iii) it reduces the risk of disease and infection, as all contact between male and female is prevented, and the semen can be treated with antibiotics before insemination and iv) it helps eliminate behavioural problems if, for example, a male camel refuses to mate a particular female. In addition, semen can be preserved for 24 h by chilling, which enables the semen to be shipped between farms or countries for insemination in other breeding herds, or it can be saved for many years by deep freezing, which extends the reproductive lifespan of the camel even beyond its death. Collection of semen Semen collection is camels presents many difficulties partly because they mate in sternal recumbancy, have a lengthy ejaculation throughout copulation, which lasts from 5 20 min, and the semen is highly viscous. Semen can be collected using a modified bull artificial vagina (AV: 30cm long, 5cm internal diameter) with a foam imitation cervix, of about 8cm in length, placed inside the AV (Bravo et al., 2000). The AV is prepared by filling with water at C to give an internal temperature of C, and air is pumped between the inner liner and outer rigid wall to create pressure inside the AV to stimulate ejaculation. A clear, glass waterjacketed (35-37 C) semen vessel is attached to the apex of the internal latex rubber liner to keep the semen warm during the lengthy ejaculation process and the entrance of the AV is lubricated with KY jelly before use. To collect the semen a sexually receptive female is used for the bull to mount but before he enters the female he is guided into the AV. The semen is usually ejaculated in fractions and this whole process can take between 5 and 10 min, although it may occasionally last for 20min or longer (Rai et al., 1988). As the collection procedure can be rather prolonged it can be advantageous to add about 1-2ml of extender to the collection vessel before the collection. 78
3 Semen Evaluation Once collected, the semen has to be evaluated and the following parameters noted: volume (usually between 2 10 ml), colour (creamy white), concentration (usual range between x 10 6 / ml, Anouassi et al., 1992) and motility. One of the main problems with camelid semen is its high viscosity which makes handling and estimation of the spermatozoa parameters difficult. This viscosity is usually attributed to the presence of mucopolysaccharides from secretions of the bulbourethral gland or the prostate, but the degree of viscosity depends on the individual male and on the quantity of the gel fraction in the ejaculate. According to some authors the semen will partially liquefy if stored at C for min (Abdel- Raouf and El Naggar, 1976; Musa et al., 1993) but other studies show it can take up to 8 h (Tibary and Anouassi, 1997a). Enzymatic methods using trypsin, collagenase and chymotrypsin as well as mechanical methods using centrifugation, density gradient centrifugation, vortexing and gentle pipetting have been investigated to try and reduce viscosity Gentle pipetting was the most successful as it did least damage to the spermatozoa. Short term semen preservation Several extenders have been used for dilution of freshly collected camel semen (Sieme et al., 1990) such as skimmed milk-glucose extender (Kenny et al., 1975), Dimitropolous 11, (Rasbech, 1984), Laiciphos (Cassou, 1959), Androhep (Waberski et al., 1989), glucose EDTA (Martin and Klug, 1979), sodium citrate-egg yolk (Kupper, 1954), lactose-egg yolk (Anouassi et al., 1992) and Green buffer- egg yolk (I.M.V. L Aigle, France; Skidmore et al., 2000). Most of these extenders contain an energy source (glucose or fructose), a protein for cold shock protection (lipoprotein from egg yolk or casein from milk), a buffering system and antibiotics. Once collected the semen is diluted with warmed (30-35 C) extender added slowly to the semen in a ratio of 1:1 3:1 (extender: semen) depending on the concentration of the ejaculate. It is better to let the semen liquefy before insemination to aid better mixing of the semen with the extender and to allow more accurate assessments of concentration and motility. Seime et al. (1990) assessed sperm motility to be better in Laiciphos (60%) and Androhep (68%) compared with Glucose EDTA (42%) but no pregnancy results were reported, whereas Anouassi et al. (1992) achieved pregnancy rates of 50% (5/10) with an extender containing 11% lactose and 20% egg yolk. In a subsequent study the best pregnancy rates were achieved with Laciphos + 20% egg yolk (53%; 7/13), although two subsequently aborted, and Green Buffer + 20% egg yolk (47%; 10/21) compared with other extenders such as skimmed milk extender (0%; 0/6; Skidmore et al., 2000). More recently, further studies have compared the extender INRA 96 (I.M.V.) with Green Buffer. Results showed that whereas motility was higher after dilution in Green Buffer (67%) compared with INRA-96 (59%) pregnancy rates were unaffected by diluent, Green Buffer (34%) and INRA-96 (34%; Morton et al., 2010). Optimum number of spermatozoa to inseminate and site of insemination Fibroscopic evaluation of the camel s cervix before and after mating has shown that semen is deposited partly intra-uterine and partly intra-cervical (Tibary and Anouassi, 1997b). Therefore in AI, semen is generally inseminated directly into the uterus using a bovine insemination catheter passed through the camel s cervix. Pregnancy rates of 50% have been achieved after insemination of 300 x 10 6 live spermatozoa (Bravo et al., 2000) or as few as 100 x 10 6 (Anouassi et al., 1992) directly into the uterine body. However, 79
4 when it is deposited just into the body of the uterus there can be a considerable loss of spermatozoa, due to backflow of semen through the relatively short cervix. A further study was therefore carried out to investigate whether better results could be achieved if the semen was deposited at the tip of the horn, rather than the body of the uterus (Skidmore and Billah, 2006a). The advantage of deep uterine insemination is that the semen is deposited nearer the UTJ and therefore should reduce the number of spermatozoa needed for successful fertilization (Lopez Gatius, 2000). In this study by Skidmore and Billah (2006a) a total of 40, 80 or 150 x 10 6 motile spermatozoa were deposited either just through the cervix into the uterine body or at the tip of the uterine horn ipsilateral with the ovary containing the dominant follicle and the results are shown in Table 1. These results indicate that although a pregnancy rate of 53% or 43% could be achieved after insemination of 150 x 10 6 motile spermatozoa into the body of the uterus or at the tip of the horn respectively, a pregnancy rate of 40% could also be achieved when a reduced number of only 80 x 10 6 spermatozoa were deposited at the tip of the uterine horn. However, as only one camel inseminated with 40 x 10 6 spermatozoa at the tip of the horn conceived it would suggest that perhaps 80 x 10 6 is the minimum number of sperm needed to establish a pregnancy by deep intrauterine insemination. Table 1: Pregnancy rates following uterine body or deep insemination of 150, 80 or 40 million spermatozoa. Number of spermatozoa inseminated x 10 6 Method of insemination Pregnancy rate %; (no. pregnant / no. inseminated) Deep Uterine 43 a (6/14) 40 (6/15) 7 (1/14) Uterine body 53 a (8/15) 7 b (1/14) 0 c (0/14) Columns with same superscript are not significantly different ( aa p = 0.8); whereas rows with different superscripts are significantly different ( ab p = 0.014; ac p = 0.002). (From Skidmore and Billah 2006a) Timing of insemination in relation to ovulation As mentioned previously, mating induces ovulation in all camelids; however, when preparing animals for AI the camel cannot be mated so alternative hormonal methods have to be used to induce ovulation. A single injection of either 20µg of the GnRH analogue (Buserelin), or 3000 IU of hcg are most commonly used and will induce ovulation if injected when the dominant follicle measures between cm in diameter in >80% of cases (Skidmore and Billah, 2006a; Tibary and Anouassi, 1997c). One of the most important aspects of insemination in camelidae is its timing in relation to ovulation. The interval from mating to ovulation is not precisely known in camels although in a recent study where the ovaries of dromedary camels were scanned at regular 2 h intervals from h after induction of ovulation with 20 µg GnRH (i.v), the majority of camels ovulated between h after injection (Skidmore and Adams, unpublished data). To investigate the most appropriate time to inseminate the camels a study was carried out where females were inseminated with 150 x 10 6 live spermatozoa either at the same time or 24 h after the GnRH injection. The results indicated that whereas 53% of camels conceived if they were inseminated 24 h after GnRH injection, only 36% conceived if they were inseminated at the same time as the GnRH injection (Skidmore and Billah, 80
5 2006b). This would suggest that to maximize the chances of conception after AI the camel should be inseminated 24 h after GnRH injection. Embryo Transfer The technique of embryo transfer can also be used to increase the reproductive efficiency of camels as it can be used to produce multiple progeny from desirable genetic combinations of sire and dam. There are also other advantages as the female can continue in competitions without taking a year out carrying a pregnancy and conformational problems that may prevent her from carrying a calf to term or giving birth can be overcome. However, there are two essential prerequisites of successful embryo transfer in large domestic species; firstly induction of superovulation in donor animals by exogenous gonadotrophin therapy, and secondly simple methods for preparing groups of synchronised recipients. Superovulation Superovulation treatments, to stimulate the growth of multiple follicles, include the use of exogenous gonadotrophins such as equine chorionic gonadotrophin (ecg) or Follicle Stimulating Hormone (FSH) which may or may not, be given after a period of progesterone priming. This progesterone priming can be given either as a Progesterone Releasing Intravaginal Device (PRID) inserted into the vagina for a period of seven days, or as daily injections of mg progesterone - in - oil for up to 15 days (Cooper et al., 1990; Skidmore et al., 1992; McKinnon et al., 1992, 1994). i) Follicle stimulating hormone (FSH). Porcine or ovine FSH is generally used. In the dromedary a total dose of 18mg of ovine FSH (ofsh) or 400mg porcine FSH (pfsh) in 20ml is given over a period of 4 days in twice daily injections in gradually decreasing doses for example: Day 1: 2 x 4ml, Day 2: 2 x 3ml, Day 3: 2 x 2ml, Day 4: 2 x 1ml (Cooper et al., 1990; Skidmore et al., 1992). ii) Equine Chorionic Gonadotrophin (ecg). The dosage of ecg used varies from iu. and is injected in a single dose one day before, or on the day of completion of a 5-15 day progesterone regime (Skidmore et al., 1992; McKinnon et al., 1992) iii) Combined ecg and FSH. Previous studies by Skidmore et al., (2002) has shown that the best response is seen when a combination of both FSH and ecg are given. The ecg (2500iu) is given as a single injection on Day 1 of treatment together with pfsh injected over a period of 4 days, as described above, also starting on Day 1 of treatment (Skidmore et al., 2002). Following superovulation treatment, follicular growth is monitored by ultrasonography and the camel mated when the majority of follicles measure between cm in diameter. This is approximately 10 days after the start of treatment. Non-Surgical collection of embryos It is now well established that in camels the embryo does not reach the uterus until day 5.5 or 6 after ovulation (Day 0 = one day after mating). Therefore any attempt to collect embryos before day 6 post ovulation, results in low recovery rates. In practice the best recovery rates from dromedaries are achieved when the uterus is flushed on day 7 or 8 after ovulation. The most widely used method for the collection of embryos from camelidae remains the non-surgical technique. A flexible, two-way Gibbon balloon catheter (20 Gauge) is passed through the cervix, then the balloon inflated with 30-40mL of air to seal the internal os. The uterus is filled to moderate capacity with embryo flushing medium and 81
6 then recovered by gravity flow into sterile beakers. This procedure is repeated a total of three times before the recovered medium is passed through a sterile embryo filter and the residual filtrate searched for embryos using a stereoscopic binocular dissecting microscope. When embryos are located they are assessed morphologically and graded 1 to 5 (Grade 1 = excellent, Grade 2 = good/fair, Grade 3 = poor, yet expanded, Grade 4 = collapsed and degenerate, Grade 5 = fragmented and degenerate) before being individually washed 4 times by passing them through 4 drops of fresh flushing medium. As many as 20 or more embryos have been recovered in a single flush but because not all the follicles will ovulate at the same time these embryos can vary greatly in size and development (Skidmore et al., 2002). Non-surgical embryo transfer technique For transfer the embryos are individually aspirated into 0.25 ml straws which are placed in the embryo transfer gun. The embryo transfer gun is passed through the cervix and then manually guided into the left horn per rectum before the embryo is deposited. Pregnancy in the recipients is diagnosed by ultrasound scanning of the uterus initially between days 18 and 20, after ovulation followed by confirmation of a viable conceptus with a heart beat between days 25 and 30, as described by Skidmore et al. (1992). Synchronization with donors Synchronization of the reproductive cycle between the donor and the recipient is critical, and embryo transfer results in the dromedary suggest that the best recipients should ovulate hours after the donor. Transfer of embryos into recipients that have ovulated one day before the donor, or three of more days afterwards, result in very low pregnancy rates of <10% (McKinnon et al., 1994; Skidmore et al., 2002) To achieve this synchronization one of two methods can be used; either recipients can be selected from a random group. If using this technique a group of recipients at known stages of their reproductive cycles are examined 24 and 48 hours after the donor is bred and all females that have a mature follicle ( cm in diameter) are treated with GnRH or hcg (Anouassi and Ali, 1990; Skidmore et al., 2002). Alternatively better results can be obtained when recipients are induced to ovulate with hcg or GnRH following a treatment combining progesterone and ecg. The recipients are treated daily with progesterone-in-oil (100mg/day) for 10 to 15 days, to try and dampen the development of more follicles, and on the last day of progesterone treatment, i.u. ecg is injected to induce follicular development. Progesterone treatment is scheduled to end on the day of injection of gonadotrophin in the donor in an attempt to synchronize the recipient and donor. The ecg treatment guarantees the presence of mature follicles in the recipient at the same time or hours after the donor (McKinnon et al., 1994). CONCLUSIONS The reproductive performance of camels can be improved by mating at the correct stage of the cycle, using embryo transfer and transferring day 7 embryos in synchronized recipients on day 5 or 6 after ovulation or using artificial insemination and inseminating 150 x 10 6 live sperm in the uterine body or horn 24 h after GnRH injection. REFERENCES Abdel-Raouf, M. El-Naggar A., (1976). Studies on reproduction in camels (Camelus dromedarius). VI. Properties and constituents of ejaculated semen. Proc. VIIIth Int. Congr. Anim. Reprod. and A.I. (Cracow),
7 Anouassi, A. and Ali, A. (1990). Embryo transfer in the camel (Camelus dromedarius). In Proceedings: Is it Possible to Improve the Reproductive Performance of the Camel? Paris. pp Anouassi, A., Adnani, M., Raed, E.L., (1992). Artificial insemination in the camel requires induction of ovulation to achieve pregnancy. Proc. 1 st Int. Camel Conf., W.R. Allen, A.J. Higgins, I.G. Mayhew, D.H. Bravo, P.W., Skidmore, J.A., and Zhao, X.X., (2000). Reproductive aspects and storage of semen in Camelidae. in: Salamon, S., Maxwell, W.M.C. (Eds). Storage of semen in domestic animals. Anim. Reprod. Sci. 62: Cassou, M.R., cited by Krause D., (1961). Derzeitiger stand der verfahren zur konservierung von bullensperma. Dtsch. Tierarztl. Wschr. 68: Cooper, M.J., Skidmore, J.A., Ali, M., Billah, M., Wensvoort, S., Billah, A.M. and Allen, W.R. (1990). An attempt to induce and synchronise ovulation and superovulation in dromedary camels for embryo transfer. In Proceedings: Is it Possible to Improve the Reproductive Performance of the Camel? Paris.pp Kenney, R.M., Bergmann, R.V., Cooper, W.L., Morse, G.W., (1975). Minimal contamination techniques for breeding mares: Technique and preliminary findings. Proc. of the Ann. Conf. Am. Assoc. Equine Pract. 21: Kupper, H., (1954). Versuche zur Werdunnung des Hengstspermas. Hannover Tierarztl. Hochsch. Thesis. Lopez-Gatius F., (2000). Site of semen deposition in cattle: A review. Theriogenology 53: Martin, J.C., Klug, E., (1979). Zur Samenubertragung beim Pferd-Samenkonservierung in Kunststoffrohrchen. Prakt. Tierarzt 3: McKinnon, A.O. and Tinson, A.H. (1992). Embryo transfer in dromedary camels. In Proceedings: 1 st Int. Camel Conf., Dubai, U.A.E McKinnon, A.O., Tinson, A.H. and Nation, G. (1994). Embryo transfer in dromedary camels. Theriogenology. 41: Morton, K.M., Billah, M., Skidmore, J.A., Artificial insemination of dromedary camels with fresh and chilled semen: Effect of diluent and sperm dose, preliminary results. Reprod. Dom. Rum. VII. Abs Musa, B.E. and Abusineina, M.E. (1978). The oestrous cycle of the camel (Camelus dromedarius.) Vet. Rec. 102, Musa, B.H., Sieme, H., Merkt, B., Hago, M.J., Cooper, M.J., Allen, W.R., Jochle, W., (1993). Manipulation of reproductive functions in male and female camels. Anim. Reprod. Sci. 33: Rai A.K., Tandon, S.N., Khanna, N.D., (1988). Copulation time of Bikaneri male camels. Ind. J. Anim. Sci. 58: Rasbech, N.O., (1984). Instrumental inserinering i hesteavlen. Dansk. Vet. Tidsskr. 67: Sieme, H., Merkt, H., Musa, B., Badreldin, H., Willmen, T., (1990). Liquid and deep freeze preservation of camel semen using different extenders and methods. Proc. Unite de Coordination pour l'elevage Camelin. Workshop: Is it Possible to Improve the Reproductive Performance of the Camel? Paris, pp
8 Skidmore J.A., Billah M., Allen W.R., (1996). The ovarian follicular wave pattern and induction of ovulation in the mated and non-mated one - humped camel (Camelus dromedarius). J. Reprod. Fertil. 106: Skidmore, J.A., Allen, W.R., Cooper, M.J., Chaudhry, M.A., Billah, M. and Billah, A.M. (1992). The recovery and transfer of embryos in the dromedary camel: results of preliminary experiments. In Proceedings: 1 st Int. Camel Conf., Dubai, U.A.E. pp Skidmore, J.A., Billah, M. (2006b). Investigation of the most appropriate time for insemination of female camels (Camelus dromedarius) after GnRH injection and comparison of pregnancy rates after deep intra-uterine versus cervical insemination. Proceedings of First Conference of International Society of Camelids Research and Development (ISOCARD). pp. 54. Skidmore, J.A., Billah, M. and Allen, W.R. (2002). Investigation of factors affecting pregnancy rate after embryo transfer in the dromedary camel. Reproduction, Fertility and Development, 14: Skidmore, J.A., Billah, M., (2006a). Comparison of pregnancy rates in dromedary camels (Camelus dromedarius) after deep intra-uterine versus cervical insemination. Theriogenology 66: Skidmore, J.A., Billah, M., Allen, W.R., (2000). Using modern reproductive technologies such as embryo transfer and artificial insemination to improve the reproductive potential of dromedary camels. Revue Elev. Med. Vet. Pays. Trop. 53 (2): Snow, J.F. Wade, (Eds.). R & W Publications (Newmarket) Ltd. UK, pp Tibary, A., Anouassi, A., (1997a). Male breeding soundness examination, in: Institut Agronomique et Veterinarie Hassan II, Rabat, Maroc. (Eds.), Theriogenology in camelidae. Abu Dhabi Printing and Publishing Company. U.A.E. pp Tibary, A., Anouassi, A., (1997b). Artificial breeding and manipulation of reproduction in camelidae, in: Institut Agronomique et Veterinarie Hassan II, Rabat, Maroc. (Eds.), Theriogenology in camelidae. Abu Dhabi Printing and Publishing Company. U.A.E. pp Tibary, A., Anouassi, A., (1997c). Reproductive phusiology of the male, in: Institut Agronomique et Veterinarie Hassan II, Rabat, Maroc. (Eds.), Theriogenology in camelidae. Abu Dhabi Printing and Publishing Company. U.A.E. pp Waberski, D., Weitze, K.F., Rath, D., Sallmann, H.P., (1989). Wirkung von bovinem Serumalbumin und Zwitterionenpuffer auf flussigkonservierten Ebersamen. Zuchthygiene 24:
Management of recipients for an embryo transfer program in dromedary camels (Camelus dromedarius)
 Management of recipients for an embryo transfer program in dromedary camels (Camelus dromedarius) J.A. Skidmore* and M. Billah Camel Reproduction Centre, P.O. Box 79914, Dubai, United Arab Emirates Summary
Management of recipients for an embryo transfer program in dromedary camels (Camelus dromedarius) J.A. Skidmore* and M. Billah Camel Reproduction Centre, P.O. Box 79914, Dubai, United Arab Emirates Summary
WBC / ICAR 2008 Satellite Meeting on Camelid Reproduction
 Close window to return to IVIS www.ivis.org WBC / ICAR 2008 Satellite Meeting on Camelid Reproduction 12-13 July, 2008, Budapest, Hungary Program and Extended stracts Peter Nagy; Gyula Huszenicza; Judit
Close window to return to IVIS www.ivis.org WBC / ICAR 2008 Satellite Meeting on Camelid Reproduction 12-13 July, 2008, Budapest, Hungary Program and Extended stracts Peter Nagy; Gyula Huszenicza; Judit
Semen-induced ovulation in the bactrian camel (Camelus bactrianus)
 Semen-induced ovulation in the bactrian camel (Camelus bactrianus) B. X. Chen, Z. X. Yuen and G. W. Pan Department of Veterinary Medicine, Gansu Agricultural University, Wuwei, Gansu and *Haixi Institute
Semen-induced ovulation in the bactrian camel (Camelus bactrianus) B. X. Chen, Z. X. Yuen and G. W. Pan Department of Veterinary Medicine, Gansu Agricultural University, Wuwei, Gansu and *Haixi Institute
WBC / ICAR 2008 Satellite Meeting on Camelid Reproduction
 Close window to return to IVIS www.ivis.org WBC / ICAR 2008 Satellite Meeting on Camelid Reproduction 12-13 July, 2008, Budapest, Hungary Program and Extended Abstracts Peter Nagy; Gyula Huszenicza; Judit
Close window to return to IVIS www.ivis.org WBC / ICAR 2008 Satellite Meeting on Camelid Reproduction 12-13 July, 2008, Budapest, Hungary Program and Extended Abstracts Peter Nagy; Gyula Huszenicza; Judit
Embryo Transfer in the Camel, Can it be Applied to Field Conditions with Realistic Costs?
 Proceedings of the Third Annual Meeting for Animal Production Under Arid Conditions, Vol.1:108-121 1998 United Arab Emirates University Embryo Transfer in the Camel, Can it be Applied to Field Conditions
Proceedings of the Third Annual Meeting for Animal Production Under Arid Conditions, Vol.1:108-121 1998 United Arab Emirates University Embryo Transfer in the Camel, Can it be Applied to Field Conditions
The effect of LH and GnRH analogues on induction of ovulation in Bactrian camel (Camelus bactrianus)
 The effect of LH and GnRH analogues on induction of ovulation in Bactrian camel (Camelus bactrianus) Moghiseh, A. 1 ; Niasari-Naslaji, A. 1* ; Nikjou, D. 1 ; Gerami, A. 2 ; Razavi, K. 3 and Mostafaey,
The effect of LH and GnRH analogues on induction of ovulation in Bactrian camel (Camelus bactrianus) Moghiseh, A. 1 ; Niasari-Naslaji, A. 1* ; Nikjou, D. 1 ; Gerami, A. 2 ; Razavi, K. 3 and Mostafaey,
Small Ruminant Reproductive Management Workshop
 Small Ruminant Reproductive Management Workshop Animal Nutrition and Physiology Center, North Dakota State University Sponsors: American Sheep and Goat Center, North Dakota State University, University
Small Ruminant Reproductive Management Workshop Animal Nutrition and Physiology Center, North Dakota State University Sponsors: American Sheep and Goat Center, North Dakota State University, University
UNDERSTANDING EMBRYO-TRANSFER (ET) A GUIDE TO THE BENEFIT OF ET IN YOUR HERD
 UNDERSTANDING EMBRYO-TRANSFER (ET) A GUIDE TO THE BENEFIT OF ET IN YOUR HERD Embryo Transfer allows one superior cow to produce a greater number of calves than normal in her lifetime TABLE OF CONTENTS
UNDERSTANDING EMBRYO-TRANSFER (ET) A GUIDE TO THE BENEFIT OF ET IN YOUR HERD Embryo Transfer allows one superior cow to produce a greater number of calves than normal in her lifetime TABLE OF CONTENTS
Ovarian Kinetics and Control of Ovulation (17 June 2000)
 In: Recent Advances in Camelid Reproduction, Skidmore J.A. and Adams G.P. (Eds.) Publisher: International Veterinary Information Service (www.ivis.org) Ovarian Kinetics and Control of Ovulation (17 June
In: Recent Advances in Camelid Reproduction, Skidmore J.A. and Adams G.P. (Eds.) Publisher: International Veterinary Information Service (www.ivis.org) Ovarian Kinetics and Control of Ovulation (17 June
Semen Characteristics and Artificial Insemination in the Bactrian Camel (25 Sep 2000)
 In: Recent Advances in Camelid Reproduction, Skidmore J.A. and Adams G.P. (Eds.) Publisher: International Veterinary Information Service (www.ivis.org) Semen Characteristics and Artificial Insemination
In: Recent Advances in Camelid Reproduction, Skidmore J.A. and Adams G.P. (Eds.) Publisher: International Veterinary Information Service (www.ivis.org) Semen Characteristics and Artificial Insemination
TECH EXTENSION. How low can we go? Finding a practical path to produce more pigs from fewer and better boars.
 TECH EXTENSION How low can we go? Finding a practical path to produce more pigs from fewer and better boars www.fastgenetics.com Artificial insemination (AI) has become the standard breeding method for
TECH EXTENSION How low can we go? Finding a practical path to produce more pigs from fewer and better boars www.fastgenetics.com Artificial insemination (AI) has become the standard breeding method for
XVII Congresso Internazionale SIVE
 SOCIETÀ ITALIANA VETERINARI PER EQUINI SOCIETÀ FEDERATA ANMVI XVII Congresso Internazionale SIVE XVII SIVE International Congress Palacongressi d Abruzzo Montesilvano (PE) - ITALY 4-6 Febbraio 2011 February
SOCIETÀ ITALIANA VETERINARI PER EQUINI SOCIETÀ FEDERATA ANMVI XVII Congresso Internazionale SIVE XVII SIVE International Congress Palacongressi d Abruzzo Montesilvano (PE) - ITALY 4-6 Febbraio 2011 February
The continued development of artificial insemination technologies in alpacas
 The continued development of artificial insemination technologies in alpacas Katherine M. Morton and W.M. Chis Maxwell 1 1 Centre for Advanced Technologies in Animal Genetics and Reproduction (ReproGen),
The continued development of artificial insemination technologies in alpacas Katherine M. Morton and W.M. Chis Maxwell 1 1 Centre for Advanced Technologies in Animal Genetics and Reproduction (ReproGen),
Advanced Non-Cycling Program. Health
 Advanced Non-Cycling Program Health Why Treat Non-Cycling Cows? Treating cows that have not been detected in oestrus ( non-cycling ) prior to the planned start of mating with DIB-Synch provides a return
Advanced Non-Cycling Program Health Why Treat Non-Cycling Cows? Treating cows that have not been detected in oestrus ( non-cycling ) prior to the planned start of mating with DIB-Synch provides a return
Artificial Insemination in Swine
 Artificial Insemination in Swine Artificial Insemination in Swine Routinely practiced on swine farms More efficient and effective use of boars AI centers distribute semen from genetically valuable boars
Artificial Insemination in Swine Artificial Insemination in Swine Routinely practiced on swine farms More efficient and effective use of boars AI centers distribute semen from genetically valuable boars
1 st International Symposium on Bison Health
 Faculty of Veterinary Medicine 1 st International Symposium on Bison Health Assisted Reproductive Technologies in Bison Robert McCorkell June 26, 2015 Artificial Insemination Frozen semen Estrus synchronization
Faculty of Veterinary Medicine 1 st International Symposium on Bison Health Assisted Reproductive Technologies in Bison Robert McCorkell June 26, 2015 Artificial Insemination Frozen semen Estrus synchronization
EMBRYO TRANSFER IN HORSES
 Vet Times The website for the veterinary profession https://www.vettimes.co.uk EMBRYO TRANSFER IN HORSES Author : Jonathan F Pycock, James R Crabtree Categories : Vets Date : March 14, 2011 Jonathan F
Vet Times The website for the veterinary profession https://www.vettimes.co.uk EMBRYO TRANSFER IN HORSES Author : Jonathan F Pycock, James R Crabtree Categories : Vets Date : March 14, 2011 Jonathan F
Syncrostim. The second chance for open cows. Presented for single cow treatment
 Syncrostim The second chance for open cows Presented for single cow treatment Syncrostim Equine chorionic gonadotrophin (ecg) for single cow treatment The active compound in Syncrostim is ecg, previously
Syncrostim The second chance for open cows Presented for single cow treatment Syncrostim Equine chorionic gonadotrophin (ecg) for single cow treatment The active compound in Syncrostim is ecg, previously
by Bergero and Cynthia Préfontaine photos by Shary B. Akers Introduction
 by Bergero and Cynthia Préfontaine photos by Shary B. Akers Introduction The embryo transfer (ET= embryo transfer) is a breeding method or reproductive technology, where an embryo is flushed from a donor
by Bergero and Cynthia Préfontaine photos by Shary B. Akers Introduction The embryo transfer (ET= embryo transfer) is a breeding method or reproductive technology, where an embryo is flushed from a donor
Animal Reproduction Science
 Animal Reproduction Science 136 (2013) 205 210 Contents lists available at SciVerse ScienceDirect Animal Reproduction Science jou rn al h om epa ge: www.elsevier.com/locate/anireprosci Use of assisted
Animal Reproduction Science 136 (2013) 205 210 Contents lists available at SciVerse ScienceDirect Animal Reproduction Science jou rn al h om epa ge: www.elsevier.com/locate/anireprosci Use of assisted
Reproductive Performance in Camel (Camelus dromedarius)
 Camel- International Journal of Veterinary Science : 1(1):13-27. January, 2013 Reproductive Performance in Camel (Camelus dromedarius) Aminu Deen Central Sheep and Wool Research Institute, Avikanagar,
Camel- International Journal of Veterinary Science : 1(1):13-27. January, 2013 Reproductive Performance in Camel (Camelus dromedarius) Aminu Deen Central Sheep and Wool Research Institute, Avikanagar,
Intrauterine Insemination - FAQs Q. How Does Pregnancy Occur?
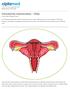 Published on: 8 Apr 2013 Intrauterine Insemination - FAQs Q. How Does Pregnancy Occur? A. The female reproductive system involves the uterus, ovaries, fallopian tubes, cervix and vagina. The female hormones,
Published on: 8 Apr 2013 Intrauterine Insemination - FAQs Q. How Does Pregnancy Occur? A. The female reproductive system involves the uterus, ovaries, fallopian tubes, cervix and vagina. The female hormones,
Biology of fertility control. Higher Human Biology
 Biology of fertility control Higher Human Biology Learning Intention Compare fertile periods in females and males What is infertility? Infertility is the inability of a sexually active, non-contracepting
Biology of fertility control Higher Human Biology Learning Intention Compare fertile periods in females and males What is infertility? Infertility is the inability of a sexually active, non-contracepting
Adoption and Foster Care
 GLOSSARY Family building via Adoption and Foster Care October 2018 www.familyequality.org/resources A Anonymous Donor: A person who donated sperm or eggs with the intention of never meeting resulting children.
GLOSSARY Family building via Adoption and Foster Care October 2018 www.familyequality.org/resources A Anonymous Donor: A person who donated sperm or eggs with the intention of never meeting resulting children.
REGULAR ARTICLE. Davide Monaco 1*, Giovanni M. Lacalandra 1 and Khalid A. El-Bahrawy 2
 doi: 10.9755/ejfa.v25i4.15498 REGULAR ARTICLE Ovarian monitoring and effects of Controlled Intravaginal Drug Releaser (CIDR) on vaginal environment and follicular activity in dromedary camels, during non-breeding
doi: 10.9755/ejfa.v25i4.15498 REGULAR ARTICLE Ovarian monitoring and effects of Controlled Intravaginal Drug Releaser (CIDR) on vaginal environment and follicular activity in dromedary camels, during non-breeding
Unit B Understanding Animal Body Systems. Lesson 7 Understanding Animal Reproduction
 Unit B Understanding Animal Body Systems Lesson 7 Understanding Animal Reproduction 1 Terms Anestrus Artificial insemination Castration Cervix Copulation Diestrus Egg Ejaculation Estrous cycle Estrus Fertilization
Unit B Understanding Animal Body Systems Lesson 7 Understanding Animal Reproduction 1 Terms Anestrus Artificial insemination Castration Cervix Copulation Diestrus Egg Ejaculation Estrous cycle Estrus Fertilization
ONTARIO PORK RESEARCH PROPOSAL FINAL REPORT
 PROPOSAL # 04/16 ONTARIO PORK RESEARCH PROPOSAL FINAL REPORT Project Leader: R.N. Kirkwood/ R.M. Friendship Project Title: Effect of addition of seminal plasma to thawed semen on sow fertility Objectives
PROPOSAL # 04/16 ONTARIO PORK RESEARCH PROPOSAL FINAL REPORT Project Leader: R.N. Kirkwood/ R.M. Friendship Project Title: Effect of addition of seminal plasma to thawed semen on sow fertility Objectives
Chapter 14 Reproduction Review Assignment
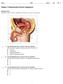 Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
The effect of time of insemination with fresh cooled semen relative to ovulation on pregnancy and embryo loss rates in the mare
 1 2 3 The effect of time of insemination with fresh cooled semen relative to ovulation on pregnancy and embryo loss rates in the mare 4 5 6 7 8 9 JR Newcombe 1 and J Cuervo-Arango 2 1 Equine Fertility
1 2 3 The effect of time of insemination with fresh cooled semen relative to ovulation on pregnancy and embryo loss rates in the mare 4 5 6 7 8 9 JR Newcombe 1 and J Cuervo-Arango 2 1 Equine Fertility
Reproductive Anatomy and Physiology of Cattle. Heat detection and Timing of Insemination s
 10/4/2009 Reproductive Anatomy and Physiology of Cattle AI Technique in Cattle 1930 s Handling of Frozen Semen 1950 s Heat detection and Timing of Insemination - 1970 s Sexed Semen 1980 s 10/4/2009 10/4/2009
10/4/2009 Reproductive Anatomy and Physiology of Cattle AI Technique in Cattle 1930 s Handling of Frozen Semen 1950 s Heat detection and Timing of Insemination - 1970 s Sexed Semen 1980 s 10/4/2009 10/4/2009
Information Booklet. Exploring the causes of infertility and treatment options.
 Information Booklet Exploring the causes of infertility and treatment options www.ptafertility.co.za info@ptafertility.co.za +27 12 998 8854 Faith is taking the first step even if you don t see the whole
Information Booklet Exploring the causes of infertility and treatment options www.ptafertility.co.za info@ptafertility.co.za +27 12 998 8854 Faith is taking the first step even if you don t see the whole
Animal Science 434. Semen Collection. Effect of Age on Sperm Output. Age When Semen Can Be Collected. Text: Ch. 10 and 11. Sexual Behavior (cont.
 Animal Science 434 Age When Semen Can Be Collected Sexual Behavior (cont.) B. Applied Reproductive Behavior of the Male: Semen Collection and Processing Text: Ch. 10 and 11 Bull Boar Ram Stallion Dog 12
Animal Science 434 Age When Semen Can Be Collected Sexual Behavior (cont.) B. Applied Reproductive Behavior of the Male: Semen Collection and Processing Text: Ch. 10 and 11 Bull Boar Ram Stallion Dog 12
Semen Preservation Dr Hany Lotfi Faculty of veterinary medicine zagazig uinversity
 Semen Preservation Dr Hany Lotfi Faculty of veterinary medicine zagazig uinversity Aim Prolonged the life span of the sperm cell without reducing its fertilizing capacity Idea Induce reversible controllable
Semen Preservation Dr Hany Lotfi Faculty of veterinary medicine zagazig uinversity Aim Prolonged the life span of the sperm cell without reducing its fertilizing capacity Idea Induce reversible controllable
GONADOTROPHIN (LUTEINISING)- RELEASING HORMONE AND ANALOGUES (GnRH OR LHRH)
 GONADOTROPHIN (LUTEINISING)- RELEASING HORMONE AND ANALOGUES (GnRH OR LHRH) Naturally occurring hormone, produced by the hypothalamus and transferred to the anterior pituitary gland in the hypophyseal
GONADOTROPHIN (LUTEINISING)- RELEASING HORMONE AND ANALOGUES (GnRH OR LHRH) Naturally occurring hormone, produced by the hypothalamus and transferred to the anterior pituitary gland in the hypophyseal
Breeding Management in the Bitch: Timing Really is Everything! Erin E. Runcan, DVM, DACT Fairfield Pet Hospital, Lancaster, OH
 Breeding Management in the Bitch: Timing Really is Everything! Erin E. Runcan, DVM, DACT Fairfield Pet Hospital, Lancaster, OH It is often surprising how many well-seasoned breeders will have little knowledge
Breeding Management in the Bitch: Timing Really is Everything! Erin E. Runcan, DVM, DACT Fairfield Pet Hospital, Lancaster, OH It is often surprising how many well-seasoned breeders will have little knowledge
Preservation of Liquid Boar Semen: Effect of Genotype, Boar and Sperm Parameters on Motility and Acrosome Integrity
 VETERINARY RESEARCH INTERNATIONAL Journal homepage: www.jakraya.com/journal/vri ORIGINAL ARTICLE Preservation of Liquid Boar Semen: Effect of Genotype, Boar and Sperm Parameters on Motility and Acrosome
VETERINARY RESEARCH INTERNATIONAL Journal homepage: www.jakraya.com/journal/vri ORIGINAL ARTICLE Preservation of Liquid Boar Semen: Effect of Genotype, Boar and Sperm Parameters on Motility and Acrosome
Animal Science 434" Semen Collection" Effect of Age on Sperm Output" Age When Semen Can Be Collected" Text: Ch. 10 and 11"
 Animal Science 434" Age When Semen Can Be Collected" Lecture 15b: Sexual Behavior (cont.) B. Applied Reproductive Behavior of the Male: Semen Collection and Processing Text: Ch. 10 and 11" "Bull "Boar
Animal Science 434" Age When Semen Can Be Collected" Lecture 15b: Sexual Behavior (cont.) B. Applied Reproductive Behavior of the Male: Semen Collection and Processing Text: Ch. 10 and 11" "Bull "Boar
AI and its Influence on Production Efficiency
 AI and its Influence on Production Efficiency Christianne E. Glossop - Malmesbury, Wiltshire, England 2007 Introduction AI in pigs is by no means a new technique. Methods for semen collection and processing,
AI and its Influence on Production Efficiency Christianne E. Glossop - Malmesbury, Wiltshire, England 2007 Introduction AI in pigs is by no means a new technique. Methods for semen collection and processing,
Proper steps for bull semen dilution and freezing. IMV Technologies France
 Proper steps for bull semen dilution and freezing IMV Technologies France Introduction Since Polge reported the first successful cryopreservation of spermatozoa in 1949, spermatozoa from many mammalian
Proper steps for bull semen dilution and freezing IMV Technologies France Introduction Since Polge reported the first successful cryopreservation of spermatozoa in 1949, spermatozoa from many mammalian
Pakistan Veterinary Journal
 RESEARCH ARTICLE Pakistan Veterinary Journal ISSN: 0253-8318 (PRINT), 2074-7764 (ONLINE) Accessible at: www.pvj.com.pk Hormonal Profiles in the Serum and Follicular Fluid of Female Camel (Camelus dromedarius)
RESEARCH ARTICLE Pakistan Veterinary Journal ISSN: 0253-8318 (PRINT), 2074-7764 (ONLINE) Accessible at: www.pvj.com.pk Hormonal Profiles in the Serum and Follicular Fluid of Female Camel (Camelus dromedarius)
INTRODUCTION TO EMBRYO TRANSFER
 INTRODUCTION TO EMBRYO TRANSFER Embryo transfer is widely practiced in cattle by animal scientists and veterinarians throughout the world. Its main purpose is to propagate valuable genetic potential as
INTRODUCTION TO EMBRYO TRANSFER Embryo transfer is widely practiced in cattle by animal scientists and veterinarians throughout the world. Its main purpose is to propagate valuable genetic potential as
Male Reproduction Organs. 1. Testes 2. Epididymis 3. Vas deferens 4. Urethra 5. Penis 6. Prostate 7. Seminal vesicles 8. Bulbourethral glands
 Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
OVARIAN RESPONSES AND CONCEPTION RATES IN RESPONSE TO GnRH, hcg, AND PROGESTERONE 1
 Dairy Research 2006 OVARIAN RESPONSES AND CONCEPTION RATES IN RESPONSE TO GnRH, hcg, AND PROGESTERONE 1 J. S. Stevenson, M. A. Portaluppi, D. E. Tenhouse, A. Lloyd, D. R. Eborn, S. Kacuba 2 and J. M. DeJarnette
Dairy Research 2006 OVARIAN RESPONSES AND CONCEPTION RATES IN RESPONSE TO GnRH, hcg, AND PROGESTERONE 1 J. S. Stevenson, M. A. Portaluppi, D. E. Tenhouse, A. Lloyd, D. R. Eborn, S. Kacuba 2 and J. M. DeJarnette
EQUINE INTRACYTOPLASMIC SPERM INJECTION PROGRAM
 COLLEGE OF VETERINARY MEDICINE & BIOMEDICAL SCIENCES Department of Large Animal Clinical Sciences 4475 TAMU EQUINE INTRACYTOPLASMIC SPERM INJECTION PROGRAM Section of Theriogenology Veterinary Medical
COLLEGE OF VETERINARY MEDICINE & BIOMEDICAL SCIENCES Department of Large Animal Clinical Sciences 4475 TAMU EQUINE INTRACYTOPLASMIC SPERM INJECTION PROGRAM Section of Theriogenology Veterinary Medical
EMBRYO TRANSFER ANIMAL SCIENCE 8818-B INTRODUCTION
 ANIMAL SCIENCE 8818-B EMBRYO TRANSFER INTRODUCTION Embryo transfer* is a process by which an embryo is collected from a donor female and then transferred into a recipient female where the embryo completes
ANIMAL SCIENCE 8818-B EMBRYO TRANSFER INTRODUCTION Embryo transfer* is a process by which an embryo is collected from a donor female and then transferred into a recipient female where the embryo completes
REPRODUCTION & GENETICS. Hormones
 REPRODUCTION & GENETICS Hormones http://www.youtube.com/watch?v=np0wfu_mgzo Objectives 2 Define what hormones are; Compare and contrast the male and female hormones; Explain what each hormone in the mail
REPRODUCTION & GENETICS Hormones http://www.youtube.com/watch?v=np0wfu_mgzo Objectives 2 Define what hormones are; Compare and contrast the male and female hormones; Explain what each hormone in the mail
Chapter 28: REPRODUCTIVE SYSTEM: MALE
 Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Select the Sex of Your Next Calf Prior to Mating: Using Sexed Semen 1
 AN163 Select the Sex of Your Next Calf Prior to Mating: Using Sexed Semen 1 Gary R. Hansen 2 Introduction Through recent advances in reproductive technology, scientists have the ability to pre-select the
AN163 Select the Sex of Your Next Calf Prior to Mating: Using Sexed Semen 1 Gary R. Hansen 2 Introduction Through recent advances in reproductive technology, scientists have the ability to pre-select the
Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to Equine Chorionic Gonadotropin
 Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to quine Chorionic Gonadotropin S. L. Pulley, L. D. Wallace, H. I. Mellieon, and J. S. Stevenson
Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to quine Chorionic Gonadotropin S. L. Pulley, L. D. Wallace, H. I. Mellieon, and J. S. Stevenson
Hormonal management of ovarian activity in breeding camels two months ahead of the natural breeding season
 Hormonal management of ovarian activity in breeding camels two months ahead of the natural breeding season Quzy I 1, Suhel Anwar 2 and G N Purohit 3* 1 Bin Hamoodah Agricultural premises, Al-Ain, UAE 2
Hormonal management of ovarian activity in breeding camels two months ahead of the natural breeding season Quzy I 1, Suhel Anwar 2 and G N Purohit 3* 1 Bin Hamoodah Agricultural premises, Al-Ain, UAE 2
EQUINE INTRACYTOPLASMIC SPERM INJECTION PROGRAM
 COLLEGE OF VETERINARY MEDICINE & BIOMEDICAL SCIENCES Department of Large Animal Clinical Sciences 4475 TAMU January 2016 b EQUINE INTRACYTOPLASMIC SPERM INJECTION PROGRAM Section of Theriogenology & the
COLLEGE OF VETERINARY MEDICINE & BIOMEDICAL SCIENCES Department of Large Animal Clinical Sciences 4475 TAMU January 2016 b EQUINE INTRACYTOPLASMIC SPERM INJECTION PROGRAM Section of Theriogenology & the
Artificial Insemination with Canine Semen Stored at a Low Temperature
 FULL PAPER Theriogenology Artificial Insemination with Canine Semen Stored at a Low Temperature Toshihiko TSUTSUI 1), Tomoyuki TEZUKA 1), Yukiko MIKASA 1), Hirofumi SUGISAWA 1), Nobuyuki KIRIHARA 1), Tatsuya
FULL PAPER Theriogenology Artificial Insemination with Canine Semen Stored at a Low Temperature Toshihiko TSUTSUI 1), Tomoyuki TEZUKA 1), Yukiko MIKASA 1), Hirofumi SUGISAWA 1), Nobuyuki KIRIHARA 1), Tatsuya
Cryopreservation of epididymal alpaca (Vicugna pacos) sperm: a comparison of citrate-, Tris- and lactose-based diluents and pellets and straws
 CSIRO PUBLISHING Reproduction, Fertility and Development, 2007, 19, 792 796 www.publish.csiro.au/journals/rfd Cryopreservation of epididymal alpaca (Vicugna pacos) sperm: a comparison of citrate-, Tris-
CSIRO PUBLISHING Reproduction, Fertility and Development, 2007, 19, 792 796 www.publish.csiro.au/journals/rfd Cryopreservation of epididymal alpaca (Vicugna pacos) sperm: a comparison of citrate-, Tris-
1- Egg yolk sodium citrate:
 The main aim of semen dilution is to extension of the volume and creation of optimal media for living of the sperm. Preparation of the semen diluent: 1- Egg yolk sodium citrate: The buffer in this diluent
The main aim of semen dilution is to extension of the volume and creation of optimal media for living of the sperm. Preparation of the semen diluent: 1- Egg yolk sodium citrate: The buffer in this diluent
Male Reproductive System
 Male Reproductive System The male reproductive system consists of a number of sex organs that are part of the reproductive process. The following sections describe the function of each part of the male
Male Reproductive System The male reproductive system consists of a number of sex organs that are part of the reproductive process. The following sections describe the function of each part of the male
Animal Reproduction Chapter 46. Fission. Budding. Parthenogenesis. Fragmentation 11/27/2017
 Animal Reproduction Chapter 46 Both asexual and sexual reproduction occur in the animal kingdom Sexual reproduction is the creation of an offspring by fusion of a male gamete (sperm) and female gamete
Animal Reproduction Chapter 46 Both asexual and sexual reproduction occur in the animal kingdom Sexual reproduction is the creation of an offspring by fusion of a male gamete (sperm) and female gamete
Outline. Male Reproductive System Testes and Sperm Hormonal Regulation
 Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Small Ruminant Embryo Transfer. - Opportunities and Challenges A Practitioner s perspective Brian W. McOnie
 Small Ruminant Embryo Transfer - Opportunities and Challenges A Practitioner s perspective Brian W. McOnie Benefits of embryo transfer Embryo transfer (ET) technology gained commercial prominence in the
Small Ruminant Embryo Transfer - Opportunities and Challenges A Practitioner s perspective Brian W. McOnie Benefits of embryo transfer Embryo transfer (ET) technology gained commercial prominence in the
Tropical and Subtropical Agroecosystems
 REVIEW [REVISIÓN DE LITERATURA] Tropical and Subtropical Agroecosystems ARTIFICIAL INSEMINATION IN CAMELIDAE [INSEMINACIÓN ARTIFICIAL EN CAMELIDAE] I.F.M. Marai 1* and A.E.B. Zeidan 2 1 Department of Animal
REVIEW [REVISIÓN DE LITERATURA] Tropical and Subtropical Agroecosystems ARTIFICIAL INSEMINATION IN CAMELIDAE [INSEMINACIÓN ARTIFICIAL EN CAMELIDAE] I.F.M. Marai 1* and A.E.B. Zeidan 2 1 Department of Animal
Select Breeders Services
 Select Breeders Services MANAGEMENT OF MARES FOR FROZEN SEMEN INSEMINATION Verify that the mare is a suitable candidate for AI with frozen semen by performing a routine reproductive exam that may include
Select Breeders Services MANAGEMENT OF MARES FOR FROZEN SEMEN INSEMINATION Verify that the mare is a suitable candidate for AI with frozen semen by performing a routine reproductive exam that may include
Chapter 14 The Reproductive System
 Biology 12 Name: Reproductive System Per: Date: Chapter 14 The Reproductive System Complete using BC Biology 12, page 436-467 14. 1 Male Reproductive System pages 440-443 1. Distinguish between gametes
Biology 12 Name: Reproductive System Per: Date: Chapter 14 The Reproductive System Complete using BC Biology 12, page 436-467 14. 1 Male Reproductive System pages 440-443 1. Distinguish between gametes
Improving Fertility of Maghrabian She-Camels Treated with Different Hormonal Treatments during Breeding Season in Egypt
 IOSR Journal of Agriculture and Veterinary Science (IOSR-JAVS) e-issn: 2319-2380, p-issn: 2319-2372. Volume 9, Issue 10 Ver. I (October. 2016), PP 20-28 www.iosrjournals.org Improving Fertility of Maghrabian
IOSR Journal of Agriculture and Veterinary Science (IOSR-JAVS) e-issn: 2319-2380, p-issn: 2319-2372. Volume 9, Issue 10 Ver. I (October. 2016), PP 20-28 www.iosrjournals.org Improving Fertility of Maghrabian
Effects of Buserelin acetate and semen extension on fertility in rabbit does under artificial insemination
 Nig. J. Anim. Prod. 2017 44(1):130-135 Nigerian Society for Animal Production Nigerian Journal of Animal Production Effects of Buserelin acetate and semen extension on fertility in rabbit does under artificial
Nig. J. Anim. Prod. 2017 44(1):130-135 Nigerian Society for Animal Production Nigerian Journal of Animal Production Effects of Buserelin acetate and semen extension on fertility in rabbit does under artificial
CAMEL: International Journal of Veterinary Science : 1(2): September, 2013
 CAMEL: International Journal of Veterinary Science : 1(2):97-113. September, 2013 Effect of gonadotrophin treatment on circulating estradiol and progesterone profiles, growth and maturation of follicles
CAMEL: International Journal of Veterinary Science : 1(2):97-113. September, 2013 Effect of gonadotrophin treatment on circulating estradiol and progesterone profiles, growth and maturation of follicles
Basic Reproduction & Genetics. Steve Pritchard UNL Extension Educator Boone-Nance Counties
 Basic Reproduction & Genetics Steve Pritchard UNL Extension Educator Boone-Nance Counties Hormonal Regulation of the Estrous Cycle Several hormones regulate the estrous cycle Changes in the concentrations
Basic Reproduction & Genetics Steve Pritchard UNL Extension Educator Boone-Nance Counties Hormonal Regulation of the Estrous Cycle Several hormones regulate the estrous cycle Changes in the concentrations
Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature
 REPRODUCTION Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature reduction -Testes wall made of fibrous connective
REPRODUCTION Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature reduction -Testes wall made of fibrous connective
Synchronization of Ovulation and Fixed-Time Insemination for Improvement of Conception Rate in Dairy Herds with Poor Estrus Detection Efficiency
 Journal of Reproduction and Development, Vol. 45, No. 1, 1999 Synchronization of Ovulation and Fixed-Time Insemination for Improvement of Conception Rate in Dairy Herds with Poor Estrus Detection Efficiency
Journal of Reproduction and Development, Vol. 45, No. 1, 1999 Synchronization of Ovulation and Fixed-Time Insemination for Improvement of Conception Rate in Dairy Herds with Poor Estrus Detection Efficiency
Buserelin animedica mg/ml Injektionslösung für Rinder, Pferde, Kaninchen
 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Buserelin animedica 0,004 mg/ml Injektionslösung für Rinder, Pferde, Kaninchen (Germany) Buserelin animedica 0.004 mg/ml Injektionslösung für Rinder, Pferde,
1. NAME OF THE VETERINARY MEDICINAL PRODUCT Buserelin animedica 0,004 mg/ml Injektionslösung für Rinder, Pferde, Kaninchen (Germany) Buserelin animedica 0.004 mg/ml Injektionslösung für Rinder, Pferde,
INDUCTIONS OF SUPEROVULATION USING SEVERAL FSH REGIMENS IN HOLSTEIN-FRIESIAN HEIFERS
 lpn. l. Vet. Res., 33, 45-50 (1985) INDUCTIONS OF SUPEROVULATION USING SEVERAL FSH REGIMENS IN HOLSTEIN-FRIESIAN HEIFERS Yoshiyuki TAKAHASHI and Hiroshi KANAGAWA (Received for publication, February 2,
lpn. l. Vet. Res., 33, 45-50 (1985) INDUCTIONS OF SUPEROVULATION USING SEVERAL FSH REGIMENS IN HOLSTEIN-FRIESIAN HEIFERS Yoshiyuki TAKAHASHI and Hiroshi KANAGAWA (Received for publication, February 2,
Fertility assessment and assisted conception
 Fertility assessment and assisted conception Dr Geetha Venkat MD FRCOG Director Pulse Learning Women s health 14 September 2016 Disclosure statement Dr Venkat is a director of Harley Street Fertility Clinic.
Fertility assessment and assisted conception Dr Geetha Venkat MD FRCOG Director Pulse Learning Women s health 14 September 2016 Disclosure statement Dr Venkat is a director of Harley Street Fertility Clinic.
Viability and Freezing Ability of Rabbit Collected in the Vagina after Prostaglandin Treatment
 Technical Note Japanese Journal of Physiology, 38, 585-589, 1988 Viability and Freezing Ability of Rabbit Collected in the Vagina after Prostaglandin Treatment Embryos Vlviane GARNIER, Jean Paul RENARD,
Technical Note Japanese Journal of Physiology, 38, 585-589, 1988 Viability and Freezing Ability of Rabbit Collected in the Vagina after Prostaglandin Treatment Embryos Vlviane GARNIER, Jean Paul RENARD,
Pregnancy rates of mares inseminated with semen cooled for 18 hours and then frozen 1
 Pregnancy rates of mares inseminated with semen cooled for 18 hours and then frozen 1 T. Backman, J. E. Bruemmer, J. K. Graham, and E. L. Squires 2 Animal Reproduction and Biotechnology Laboratory, Colorado
Pregnancy rates of mares inseminated with semen cooled for 18 hours and then frozen 1 T. Backman, J. E. Bruemmer, J. K. Graham, and E. L. Squires 2 Animal Reproduction and Biotechnology Laboratory, Colorado
Journal Of Agriculture and Social Research (JASR) Vol. 8, No.1, 2008
 THE EFFECTS OF DIFFERENT GONADOTROPHIN TREATMENTS ON THE EMBRYO GENERATION AND QUALITY OF EMBRYOS IN WEST AFRICAN DWARF GOATS IHEUKWUMERE, F. C. ABSTRACT The effects of Ovine FSH, Porcine FSH (FSH-P) and
THE EFFECTS OF DIFFERENT GONADOTROPHIN TREATMENTS ON THE EMBRYO GENERATION AND QUALITY OF EMBRYOS IN WEST AFRICAN DWARF GOATS IHEUKWUMERE, F. C. ABSTRACT The effects of Ovine FSH, Porcine FSH (FSH-P) and
Effect of diluents on storage of ram semen
 Journal of Agri-Food and Applied Sciences Available online at jaas.blue-ap.org JAAS Journal. Vol. (), pp. 9-, June, E-ISSN: - Effect of diluents on storage of ram semen Fatemeh Soltanpour * and Golamali
Journal of Agri-Food and Applied Sciences Available online at jaas.blue-ap.org JAAS Journal. Vol. (), pp. 9-, June, E-ISSN: - Effect of diluents on storage of ram semen Fatemeh Soltanpour * and Golamali
Why Cycle Control?" Manipulating Ovulation and Estrous Synchronization" Manipulating Ovulation" Cattle" Principle of PGF 2α Use"
 Why Cycle Control?" Manipulating Ovulation and Estrous Synchronization" John Parrish 1. Group females for parturition: " a) Decrease labor, calving period Reduce calving season" b) More uniform weaning
Why Cycle Control?" Manipulating Ovulation and Estrous Synchronization" John Parrish 1. Group females for parturition: " a) Decrease labor, calving period Reduce calving season" b) More uniform weaning
Testosterone Therapy-Male Infertility
 Testosterone Therapy-Male Infertility Testosterone Therapy-Male Infertility Many men are prescribed testosterone for a variety of reasons. Low testosterone levels (Low T) with no symptoms, general symptoms
Testosterone Therapy-Male Infertility Testosterone Therapy-Male Infertility Many men are prescribed testosterone for a variety of reasons. Low testosterone levels (Low T) with no symptoms, general symptoms
Clinical ICSI in the horse:
 Clinical ICSI in the horse: differences and similarities to human in an in vitro maturation-based system Katrin Hinrichs College of Veterinary Medicine & Biomedical Sciences Texas A&M University Standard
Clinical ICSI in the horse: differences and similarities to human in an in vitro maturation-based system Katrin Hinrichs College of Veterinary Medicine & Biomedical Sciences Texas A&M University Standard
Effect of repeated laparoscopic ovum pick-up on yield and quality of oocytes in goats
 Effect of repeated laparoscopic ovum pick-up on yield and quality of oocytes in goats Nor Farizah, A.H., Rahman, M.M., Wan Khadijah, W.E. and Abdullah, R.B.* Animal Biotechnology-Embryo Laboratory (ABEL),
Effect of repeated laparoscopic ovum pick-up on yield and quality of oocytes in goats Nor Farizah, A.H., Rahman, M.M., Wan Khadijah, W.E. and Abdullah, R.B.* Animal Biotechnology-Embryo Laboratory (ABEL),
Artificial Insemination
 Artificial Insemination Insemination of horses This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained
Artificial Insemination Insemination of horses This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained
Female Reproductive System. Lesson 10
 Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Embryo Transfer. Objectives. Performing Embryo Transfer. History of Embryo Transfer. Benefits for Embryo Transfer
 33 Embryo Transfer 305 Daniel Stein and Glenn Selk Objectives Discuss the history of embryo transfer. Learn the benefits of embryo transfer. Learn how to transfer embryos. Discuss the potential costs of
33 Embryo Transfer 305 Daniel Stein and Glenn Selk Objectives Discuss the history of embryo transfer. Learn the benefits of embryo transfer. Learn how to transfer embryos. Discuss the potential costs of
Sample Provincial exam Q s: Reproduction
 Sample Provincial exam Q s: Reproduction 11. Functions Testosterone Makes the male sex organs function normally, and also inhibits hypothalamus s release of GnRH and thus LH & FSH and thus testosterone
Sample Provincial exam Q s: Reproduction 11. Functions Testosterone Makes the male sex organs function normally, and also inhibits hypothalamus s release of GnRH and thus LH & FSH and thus testosterone
In Vitro Fertilization What to expect
 Patient Education In Vitro Fertilization What to expect This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this process,
Patient Education In Vitro Fertilization What to expect This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this process,
Proceedings of the 12th International Congress of the World Equine Veterinary Association WEVA
 www.ivis.org Proceedings of the 12th International Congress of the World Equine Veterinary Association WEVA November 2-5, 2011 Hyderabad, India Reprinted in IVIS with the Permission of WEVA Organizers
www.ivis.org Proceedings of the 12th International Congress of the World Equine Veterinary Association WEVA November 2-5, 2011 Hyderabad, India Reprinted in IVIS with the Permission of WEVA Organizers
In Vitro Fertilization
 Patient Education In Vitro Fertilization About the treatment This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this
Patient Education In Vitro Fertilization About the treatment This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this
In vitro Embryo Production in Calves
 In vitro Embryo Production in Calves Reuben J. Mapletoft 1, Ana Rita Tavares Krause 1 and Pietro S. Baruselli 2 1 WCVM, University of Saskatchewan, Saskatoon, SK S7N 5B4 CANADA 2 Departamento de Reprodução
In vitro Embryo Production in Calves Reuben J. Mapletoft 1, Ana Rita Tavares Krause 1 and Pietro S. Baruselli 2 1 WCVM, University of Saskatchewan, Saskatoon, SK S7N 5B4 CANADA 2 Departamento de Reprodução
STUDY ON FACTORS AFFECTING THE CONCEPTION RATE IN RED CHITTAGONG COWS. Abstract
 Bang. J. Anim. Sci. 2010, 39(1&2) : 52 57 ISSN 0003-3588 STUDY ON FACTORS AFFECTING THE CONCEPTION RATE IN RED CHITTAGONG COWS M. M. R. Mufti, M. K. Alam, M. S. Sarker, A. B. M. R. Bostami and N. G. Das
Bang. J. Anim. Sci. 2010, 39(1&2) : 52 57 ISSN 0003-3588 STUDY ON FACTORS AFFECTING THE CONCEPTION RATE IN RED CHITTAGONG COWS M. M. R. Mufti, M. K. Alam, M. S. Sarker, A. B. M. R. Bostami and N. G. Das
ANOTHER LOOK AT TIMING OF A I
 ANOTHER LOOK AT TIMING OF A I Ray L. Nebe l Department of Dairy Scienc e Virginia Polytechnic Institute and State Universit y Blacksburg, Virgini a Adoption by dairy producers of Al has made it one of
ANOTHER LOOK AT TIMING OF A I Ray L. Nebe l Department of Dairy Scienc e Virginia Polytechnic Institute and State Universit y Blacksburg, Virgini a Adoption by dairy producers of Al has made it one of
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion
 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Why Cycle Control? Manipulating Ovulation and Estrous Synchronization. Manipulating Ovulation. Cattle. Principle of PGF 2a Use
 Why Cycle Control? Manipulating and Estrous Synchronization John Parrish 1. Group females for parturition: a) Decrease labor, calving period Reduce calving season b) More uniform weaning weights. 2. Reduce
Why Cycle Control? Manipulating and Estrous Synchronization John Parrish 1. Group females for parturition: a) Decrease labor, calving period Reduce calving season b) More uniform weaning weights. 2. Reduce
17. Preventing pregnancy
 17. Preventing pregnancy Objectives By the end of this session, group members will be able to: Define contraception. List ways young people can prevent pregnancy. Background notes What is contraception?
17. Preventing pregnancy Objectives By the end of this session, group members will be able to: Define contraception. List ways young people can prevent pregnancy. Background notes What is contraception?
PUBVET, Publicações em Medicina Veterinária e Zootecnia.
 PUBVET, Publicações em Medicina Veterinária e Zootecnia. Disponível em: . Embryo recovery following artificial insemination of mares enrolled in a small AI breeding
PUBVET, Publicações em Medicina Veterinária e Zootecnia. Disponível em: . Embryo recovery following artificial insemination of mares enrolled in a small AI breeding
NICE fertility guidelines. Hemlata Thackare MPhil MSc MRCOG Deputy Medical Director London Women s Clinic
 NICE fertility guidelines Hemlata Thackare MPhil MSc MRCOG Deputy Medical Director London Women s Clinic About the LWC 4 centres around the UK London Cardiff Swansea Darlington The largest sperm bank in
NICE fertility guidelines Hemlata Thackare MPhil MSc MRCOG Deputy Medical Director London Women s Clinic About the LWC 4 centres around the UK London Cardiff Swansea Darlington The largest sperm bank in
Unit B Understanding Animal Body Systems. Lesson 6 Anatomy and Physiology of Animal Reproduction Systems
 Unit B Understanding Animal Body Systems Lesson 6 Anatomy and Physiology of Animal Reproduction Systems 1 Terms Alimentary canal Bladder Cervix Clitoris Cloaca Copulation Cowper s gland Epididymis Fallopian
Unit B Understanding Animal Body Systems Lesson 6 Anatomy and Physiology of Animal Reproduction Systems 1 Terms Alimentary canal Bladder Cervix Clitoris Cloaca Copulation Cowper s gland Epididymis Fallopian
Unit B: Anatomy and Physiology of Poultry. Lesson 4: Artificial Poultry Reproduction
 Unit B: Anatomy and Physiology of Poultry Lesson 4: Artificial Poultry Reproduction 1 1 Terms Artificial insemination Sexed semen Standing heat 2 2 I. Artificial insemination is the placing of semen in
Unit B: Anatomy and Physiology of Poultry Lesson 4: Artificial Poultry Reproduction 1 1 Terms Artificial insemination Sexed semen Standing heat 2 2 I. Artificial insemination is the placing of semen in
Sperm production. Sperm production. Meiosis. Mitosis. The cells of Leydig in testes secrete
 Sperm production Ductus deferens Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Sperm production Ductus deferens Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Sperm production. Sperm production. Controlling sperm production. Meiosis. Mitosis. The cells of Leydig in testes secrete
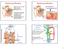 Ductus deferens Sperm production Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Ductus deferens Sperm production Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Infertility treatment
 In the name of God Infertility treatment Treatment options The optimal treatment is one that provide an acceptable success rate, has minimal risk and is costeffective. The treatment options are: 1- Ovulation
In the name of God Infertility treatment Treatment options The optimal treatment is one that provide an acceptable success rate, has minimal risk and is costeffective. The treatment options are: 1- Ovulation
Study Guide Answer Key Reproductive System
 Biology 12 Human Biology Textbook: BC Biology 12 Study Guide Answer Key Reproductive System 1. Distinguish between a gamete and a gonad using specific examples from the male and female systems. Gonads
Biology 12 Human Biology Textbook: BC Biology 12 Study Guide Answer Key Reproductive System 1. Distinguish between a gamete and a gonad using specific examples from the male and female systems. Gonads
Age and Fertility. A Guide for Patients Revised 2012 Copyright 2012 by the American Society for Reproductive Medicine
 1 Age and Fertility A Guide for Patients Revised 2012 Copyright 2012 by the American Society for Reproductive Medicine INTRODUCTION Fertility changes with age. Both males and females become fertile in
1 Age and Fertility A Guide for Patients Revised 2012 Copyright 2012 by the American Society for Reproductive Medicine INTRODUCTION Fertility changes with age. Both males and females become fertile in
