1. ZOLLINGER - ELLISON SYNDROME
|
|
|
- Kory Griffith
- 6 years ago
- Views:
Transcription
1 1. ZOLLINGER - ELLISON SYNDROME Zollinger Ellison syndrome is characterized by gastric hypersecretion, ulceration (ulceration) and gastrin-producing tumor. Gastrin is a peptide hormone synthesized by the G-cells of the stomach (and δ-cells of the pancreas) and has a stimulating effect on the production of hydrochloric acid. Gastrinoma often occurs in the pancreas. It is a malignant tumor and has metastatic potential. Most patients suffering from gastric ulcers and they are refractory to treatment. Diagnosis is based on anamnestic data, laboratory of gastrin levels (<95pmol/L) and the investigation of gastric acid secretion. Casuistics: 32 year old man comes to the clinic due to the chronic diarrhea. Recently patient experiences black stools. He has been treated with bisoprolol 5 mg daily due to high blood pressure for several years. Six months ago, he completed treatment for Helicobacter pylori eradication due to the duodenal ulcer. Sometimes he suffers burning feelings in the stomach area and overall discomfort. Recently, he feels tired, and he observed weight loss (about 8 kg in 2 months). He is not a smoker, occasionally alcohol drinker (red wine). He is allergic to ampicilin. He works as bank clerk. Clinical findings: Height 178 cm, weight 65 kg, BP 145/80, RR 16/min, T=36,8ºC Patient is conscious, orientated, actively cooperating. Skin is pale. Respiration alveolar, without side-phenomena. Cardiac action regular, 66 per minute, no murmur. Normal abdomen, Fig.1 Endoscopic picture of duodenal ulcer 1 palpation without pain and resistance. Liver of normal size. Peristalsis +. Endoscopic scan of upper gastrointestinal tract has been indicated. Multiple ulceration has been confirmed in duodenum with patches of blood trickle. Basic biochemistry can be found in table. Blood count and leukocyte differential without pathological observation. Zollinger-Ellison s syndrome has been confirmed by abnormal gastrin concentration in plasma 480 pmol/l. Gastric acid secretion test has been indicated. The gastric secretion samples are taken by nasogastric tube during 60 minutes (BAO basic acid output). Analyte Proteins S-Na + S-K + S-Ca S-Cl - HCO3 - gastrin Concentration 58 g/ L 138 mmol/l 4,2mmol/L 2,75 mmol/l 90 mmol/l 30 mmol/l 480 pmol/ L After the first sampling, the patient receives dose of i.v. pentagastrin (gastrin analogous) and the gastric secretion samples are taken again in 60 min time course. Samples are taken every 15 minutes - 4 times all together (MAO maximal acid output). On the basis of the measured amounts of hydrochloric acid the level of hypersecretion can be estimated. 1 F.R. SCHILLER, Klaus. ET AL. Atlas of Gastrointestinal Endoscopy and Related Pathology. 2. ed. UK: Blackwell Science Ltd, ISBN /6
2 Physiological values: BAO: 3-5 mmol/h; MAO: mmol/h Our laboratory has received 5 samples of one patient. The first one is labelled BAO (see above). Four samples with labels MAO 1 MAO 4 do represent the amount of secret taken in 15 minutes interval. Your task is to analyze the samples and estimate the level of HCl secretion disorder. 2. INSTRUCTIONS FOR ANALYSIS 2.1. Standardization of sodium hydroxide solution with oxalic acid Sodium hydroxide is water soluble hygroscopic compound. Solutions prepared by dissolving of NaOH in water are not exactly defined, because NaOH is unstable. NaOH reacts with atmospheric water and carbon dioxide. It is necessary to find exact concentration by standardization. For alcalimetric titrations we use oxalic acid dihydrate as primary standard. On your lab desk you can find prepared oxalic acid solution with precise defined concentration. Sodium hydroxide is titrated with oxalic acid solution as follows: HOOC-COOH + 2 NaOH NaOOC-COONa + 2 H2O The endpoint is visualized with acid-base indicator - Tashiro (mixed indicator contains methyl red and methylene blue). Colour change is from violet in acid to green in basic solution (Fig. 2). Oxalic acid is moderately strong acid. For correct determination of equivalence point, add just before the point calcium chloride solution. Fig. 2 HOOC-COOH + CaCl2 Ca(-OOC-COO-) + 2HCl Calcium chloride displaces hydrogen ions in the molecule of oxalic acid. This will convert titration to the titration of strong acid HCl with NaOH with a sharp colour change. Procedure: To make the NaOH solution, weigh 2.20 g of the solid sodium hydroxide. Add solid NaOH to approximately 50 ml of water (use beaker). Mix the solution with glass stirrer gently until NaOH is dissolved. Then adjust the volume in volumetric flask to 250 ml. Mix the solution well by turning upside down. 1. Rinse a burette with 10 ml of prepared sodium hydroxide solution. Discard the rinse solution. Repeat the rinsing on more time. Adjust the volume to the zero mark. 2. Measure 10 ml of oxalix acid standard solution to a 250 ml titration flask. Dilute with 30 ml of distilled water. Add 2 drops of Tashiro indicator solution. Concentration of standard oxalic acid solution is mol/l. 2/6
3 3. Titrate the sample to the grey colour. Then add 10 ml of 20 % calcium chloride solution. Mix it well and rinse the inner walls with distilled water. Carefully titrate to green colour (1-2 drops). 4. Record a volume of NaOH used. Repeat the titration procedure, above, 2 more times. From average used volume calculate the concentration of NaOH solution The BAO and MAO estimation On every desk there are samples of gastric secretion taken by nasogastric tube. The samples are marked BAO and MAO 1 MAO 4. Volumes are in the table: BAO ml MAO ml MAO MAO ml MAO 3 80 ml MAO 4 80 ml BAO estimation: On your desk there are samples of gastric secretion taken by nasogastric tube. Your task is to analyze and compute BAO. For titration use the earlier standardized NaOH solution. 1. Fill the burette with NaOH solution up to the marked level. 2. Into the rinsed titration flask add 10 ml of the gastric secretion sample. 3. Sample in the titration flask dilute by approximately 20 ml of distilled water and titrate using Tashiro indicator to the equivalence. 4. Estimation has to be carried out twice. Using the estimated NaOH consumption compute the HCl amount of substance, and subsequently BAO. MAO estimation: On your desk there are samples of gastric secretion labelled MAO 1- MAO MAO estimation carries out using identical way as in previous case (BAO). Each sample has to be analyzed twice. All together there will be carried out 8 estimations. 2. When finished, please, do not forget to rinse the burette by distilled water and make up the desk. 3/6
4 3. Results 3.1. Standardization of the NaOH solution c(naoh)=.. Calculation: 3.2. BAO a MAO estimation A/ BAO calculation: B/ MAO calculation: 4/6
5 Write your results into the following table: BAO MAO Sample - MAO 1 MAO 2 MAO 3 MAO 4 Volume (cm 3 ) NaOH usage (cm 3 ) Average NaOH usage (cm 3 ) n(hcl) in analyzed sample volume (mmol) n(hcl) whole sample volume (mmol) Σn(HCl) n=amount of substance Discussion 1. How would you explain estimated patient s BAO and MAO values? 2. Describe, how would change the experiment precision, in case of not using CaCl2 in the titration. 3. Try to explain the occurrence of diarrhoea. Try to explain the cause. 4. Find the abnormal laboratory results and try to outline the reasons of theirs changes. 5. Why is ulcer dangerous and describe possible complications. Conclusion 5/6
6 1. Hydrochloric acid APPENDIX 1: MATERIAL SAFETY DATA SHEET Risk phrases: H314 Causes burns. H335 Irritating to respiratory system. Safety phrases: H314 Causes burns. P280 Wear suitable protective clothing, gloves and eye/face protection. 2. Sodium hydroxide Risk phrases: H314 Causes burns. Safety phrases: P280 Wear suitable protective clothing, gloves and eye/face protection. 3. Oxalic acid Xn Risk phrases: H302/312 Harmful if swallowed or in contact with skin Safety phrases: P262 Do not get in eyes, on skin, or on clothing P302/352 Wash with plenty of soap and water 6/6
EXPERIMENT 2: ACID/BASE TITRATION. Each person will do this laboratory individually. Individual written reports are required.
 EXPERIMENT 2: ACID/BASE TITRATION Each person will do this laboratory individually. Individual written reports are required. OVERVIEW. Acid/base titration, relying on a color change of the indicator, is
EXPERIMENT 2: ACID/BASE TITRATION Each person will do this laboratory individually. Individual written reports are required. OVERVIEW. Acid/base titration, relying on a color change of the indicator, is
TRATION: ANALYSIS OF SODIUM HYDROXIDE
 Experiment 8 Name: 22 Ti TRATION: ANALYSIS OF SODIUM HYDROXIDE In this experiment, you will learn the concept and technique of titration. You will use a chemical standard (potassium hydrogen phthalate)
Experiment 8 Name: 22 Ti TRATION: ANALYSIS OF SODIUM HYDROXIDE In this experiment, you will learn the concept and technique of titration. You will use a chemical standard (potassium hydrogen phthalate)
Experiment 6: STANDARDIZATION OF A BASE; MASS PERCENT OF AN ACID
 Experiment 6: STANDARDIZATION OF A BASE; MASS PERCENT OF AN ACID Introduction The reaction of an acid and a base to form a salt and water is known as neutralization. In this experiment; potassium acid
Experiment 6: STANDARDIZATION OF A BASE; MASS PERCENT OF AN ACID Introduction The reaction of an acid and a base to form a salt and water is known as neutralization. In this experiment; potassium acid
Chemistry 212. Experiment 3 ANALYSIS OF A SOLID MIXTURE LEARNING OBJECTIVES. - learn to analyze a solid unknown with volumetric techniques.
 Experiment 3 The objectives of this experiment are to LEARNING OBJECTIVES - learn to analyze a solid unknown with volumetric techniques. - use stoichiometry to determine the percentage of KHP in a solid
Experiment 3 The objectives of this experiment are to LEARNING OBJECTIVES - learn to analyze a solid unknown with volumetric techniques. - use stoichiometry to determine the percentage of KHP in a solid
Titration of Synthesized Aspirin A continuation of the aspirin synthesis lab
 Titration of Synthesized Aspirin A continuation of the aspirin synthesis lab In this lab, you will determine the percent purity of your product from the aspirin synthesis using an acid-base titration.
Titration of Synthesized Aspirin A continuation of the aspirin synthesis lab In this lab, you will determine the percent purity of your product from the aspirin synthesis using an acid-base titration.
Experiment 7, Analysis of KHP by titration with NaOH Wright College, Department of Physical Science and Engineering
 Name Date Experiment 7, Analysis of KHP by titration with NaOH Wright College, Department of Physical Science and Engineering In this experiment, you will determine the amount (percent) of potassium hydrogen
Name Date Experiment 7, Analysis of KHP by titration with NaOH Wright College, Department of Physical Science and Engineering In this experiment, you will determine the amount (percent) of potassium hydrogen
Experiment 10 Acid-base Titrations: Part A Analysis of vinegar and Part B Analysis of a Carbonate/Bicarbonate mixture
 Chemistry 112 Section 201 Dates of Experiment: March 8 and March 22 Noah McNally Acid-base Titrations: Part A Analysis of vinegar and Part B Analysis of a Carbonate/Bicarbonate mixture Unknown Number:
Chemistry 112 Section 201 Dates of Experiment: March 8 and March 22 Noah McNally Acid-base Titrations: Part A Analysis of vinegar and Part B Analysis of a Carbonate/Bicarbonate mixture Unknown Number:
CHAPTER 3: MATERIALS AND METHODS
 CHAPTER 3: MATERIALS AND METHODS Materials and Methods. The from different husks, fruits, and vegetables peels were estimated quantitatively by the following volumetric procedure s such as Bertrand s,
CHAPTER 3: MATERIALS AND METHODS Materials and Methods. The from different husks, fruits, and vegetables peels were estimated quantitatively by the following volumetric procedure s such as Bertrand s,
Strength of Vinegar by Acid-Base Titration
 Strength of Vinegar by Acid-Base Titration Test Exercise 100 points 1? QUESTIONS? How are acid/base titrations conducted? What is standardization? How do you standardize a solution of a base? How, and
Strength of Vinegar by Acid-Base Titration Test Exercise 100 points 1? QUESTIONS? How are acid/base titrations conducted? What is standardization? How do you standardize a solution of a base? How, and
EXPERIMENT. Titration of the Weak Acid Potassium Hydrogen Phthalate (KHP)
 INTRODUCTION EXPERIMENT Titration of the Weak Acid Potassium Hydrogen Phthalate (KHP) Materials generally considered to possess acidic and/or basic properties are widely distributed in nature and range
INTRODUCTION EXPERIMENT Titration of the Weak Acid Potassium Hydrogen Phthalate (KHP) Materials generally considered to possess acidic and/or basic properties are widely distributed in nature and range
Hardness by EDTA Titration
 SOP AMBL-104-A Page 1 of 5 Standard Operating Procedure AMBL-104-A Prepared: 7/2/2017 Revised: Prepared by: Terry E. Baxter Reviewed by: Hardness by EDTA Titration METHOD SUMMARY This SOP describes the
SOP AMBL-104-A Page 1 of 5 Standard Operating Procedure AMBL-104-A Prepared: 7/2/2017 Revised: Prepared by: Terry E. Baxter Reviewed by: Hardness by EDTA Titration METHOD SUMMARY This SOP describes the
Biodiesel Fundamentals for High School Chemistry Classes. Laboratory 3: Determination of the Acid Number of Vegetable Oils by Titration
 Laboratory 3: Determination of the Acid Number of Vegetable Oils by Titration Topics Covered ph vs. acid number Acidity and acid values in organic solutions Titration techniques How to obtain acid values
Laboratory 3: Determination of the Acid Number of Vegetable Oils by Titration Topics Covered ph vs. acid number Acidity and acid values in organic solutions Titration techniques How to obtain acid values
TRATION: ANALYSIS OF SODIUM HYDROXIDE
 Experiment 10 Name: 22 Ti TRATION: ANALYSIS OF SODIUM HYDROXIDE In this experiment, you will learn the concept and technique of titration. You will use a standard (potassium hydrogen phthalate) to determine
Experiment 10 Name: 22 Ti TRATION: ANALYSIS OF SODIUM HYDROXIDE In this experiment, you will learn the concept and technique of titration. You will use a standard (potassium hydrogen phthalate) to determine
MODULE TOPIC: Percent Composition of Elements using EDTA titration. LESSON PLAN 1: EDTA titration of Calcium in a Citracal Tablet
 MODULE TOPIC: Percent Composition of Elements using EDTA titration LESSON PLAN 1: EDTA titration of Calcium in a Citracal Tablet LESSON PLAN 2: EDTA titration of Magnesium in a sample of Epsom Salt STANDARD
MODULE TOPIC: Percent Composition of Elements using EDTA titration LESSON PLAN 1: EDTA titration of Calcium in a Citracal Tablet LESSON PLAN 2: EDTA titration of Magnesium in a sample of Epsom Salt STANDARD
Experiment The Thermodynamics of the Dissolution of Borax
 Experiment The Thermodynamics of the Dissolution of Borax Borax is a commonly added to (clothes) wash water to increase the ph for more effective cleansing. 0 B.J E C T I V E S = -RTln K R. X T K, standard
Experiment The Thermodynamics of the Dissolution of Borax Borax is a commonly added to (clothes) wash water to increase the ph for more effective cleansing. 0 B.J E C T I V E S = -RTln K R. X T K, standard
Lab #3 Potentiometric Titration of Soda Ash (after Christian, p , p ) (phenolphthalein)
 Lab #3 Potentiometric Titration of Soda Ash (after Christian, p.692-694, p.718-720) I: INTRODUCTION In this lab, an unknown sample of soda ash (a crude mixture of sodium carbonate) will be titrated with
Lab #3 Potentiometric Titration of Soda Ash (after Christian, p.692-694, p.718-720) I: INTRODUCTION In this lab, an unknown sample of soda ash (a crude mixture of sodium carbonate) will be titrated with
Date... Name... Group... Urine sample (Tube No 2)
 Date... Name... Group... Instructions for the practical lesson on biochemistry Topic: Non-protein nitrogen compounds Task 1: Estimation of creatinine in serum and urine 1. Trichloroacetic acid 1.22 mol/l
Date... Name... Group... Instructions for the practical lesson on biochemistry Topic: Non-protein nitrogen compounds Task 1: Estimation of creatinine in serum and urine 1. Trichloroacetic acid 1.22 mol/l
3 To gain experience monitoring a titration with a ph electrode and determining the equivalence point.
 Titrations PURPOSE To determine the concentration of acetic acid in vinegar. GOALS 1 To perform an acid-base titration. 2 To gain experience titrating carefully to a visible endpoint. 3 To gain experience
Titrations PURPOSE To determine the concentration of acetic acid in vinegar. GOALS 1 To perform an acid-base titration. 2 To gain experience titrating carefully to a visible endpoint. 3 To gain experience
ENZYME CONCENTRATIONS AND ENZYME ACTIVITY: PLANNING SHEET
 Activity 2.11 Student Sheet ENZYME CONCENTRATIONS AND ENZYME ACTIVITY: PLANNING SHEET To investigate how enzyme concentration can affect the initial rate of reaction. Wear eye protection, lab coats and
Activity 2.11 Student Sheet ENZYME CONCENTRATIONS AND ENZYME ACTIVITY: PLANNING SHEET To investigate how enzyme concentration can affect the initial rate of reaction. Wear eye protection, lab coats and
Method 7.6 Raw sugar: reducing sugars by the Luff Schoorl method
 Section 7: Raw sugar p 1/5 Method 7.6 Raw sugar: reducing sugars by the Luff Schoorl method 1. Rationale The method is applicable to all raw sugars and is used to determine the amount of reducing sugars
Section 7: Raw sugar p 1/5 Method 7.6 Raw sugar: reducing sugars by the Luff Schoorl method 1. Rationale The method is applicable to all raw sugars and is used to determine the amount of reducing sugars
SOUTH AFRICAN NATIONAL STANDARD
 ISBN 978-0-626-31165-0 SOUTH AFRICAN NATIONAL STANDARD Water Sulfide content Published by SABS Standards Division 1 Dr Lategan Road Groenkloof Private Bag X191 Pretoria 0001 Tel: +27 12 428 7911 Fax: +27
ISBN 978-0-626-31165-0 SOUTH AFRICAN NATIONAL STANDARD Water Sulfide content Published by SABS Standards Division 1 Dr Lategan Road Groenkloof Private Bag X191 Pretoria 0001 Tel: +27 12 428 7911 Fax: +27
EXPERIMENT 4 TITRATION OF AN UNKNOWN ACID
 EXPERIMENT 4 TITRATION OF AN UNKNOWN ACID The reaction of an acid and a base to form a salt and water is known as neutralization. In this experiment you will titrate an known amount of KHP with an unknown
EXPERIMENT 4 TITRATION OF AN UNKNOWN ACID The reaction of an acid and a base to form a salt and water is known as neutralization. In this experiment you will titrate an known amount of KHP with an unknown
Change to read: BRIEFING
 BRIEFING Dibasic Calcium Phosphate Dihydrate, USP 29 page 359. The Japanese Pharmacopoeia is the coordinating pharmacopeia for the international harmonization of the compendial standards for the Dibasic
BRIEFING Dibasic Calcium Phosphate Dihydrate, USP 29 page 359. The Japanese Pharmacopoeia is the coordinating pharmacopeia for the international harmonization of the compendial standards for the Dibasic
EXPERIMENT 4 DETERMINATION OF REDUCING SUGARS, TOTAL REDUCING SUGARS, SUCROSE AND STARCH
 Practical Manual Food Chemistry and Physiology EXPERIMENT 4 DETERMINATION OF REDUCING SUGARS, TOTAL REDUCING SUGARS, SUCROSE AND STARCH Structure 4.1 Introduction Objectives 4.2 Experiment 4a: Reducing
Practical Manual Food Chemistry and Physiology EXPERIMENT 4 DETERMINATION OF REDUCING SUGARS, TOTAL REDUCING SUGARS, SUCROSE AND STARCH Structure 4.1 Introduction Objectives 4.2 Experiment 4a: Reducing
NEW KJELDAHL CHEMICALS AND FLASKS FOR THE NITROGEN DETERMINATION
 NEW KJELDAHL CHEMICALS AND FLASKS FOR THE NITROGEN DETERMINATION KJELDAHL FLASKS The bulb-shaped flasks with a long neck are primarily used in the determination of organically bonded nitrogen. Before the
NEW KJELDAHL CHEMICALS AND FLASKS FOR THE NITROGEN DETERMINATION KJELDAHL FLASKS The bulb-shaped flasks with a long neck are primarily used in the determination of organically bonded nitrogen. Before the
EXPERIMENT 13 QUALITATIVE ANALYSIS
 PURPOSE: 1. To separate and identify each cation in a mixture of Ag +, Cu 2+, Zn 2+, and Ca 2+ cations. 2. To identify the cations present in three individually assigned unknown mixtures that may contain
PURPOSE: 1. To separate and identify each cation in a mixture of Ag +, Cu 2+, Zn 2+, and Ca 2+ cations. 2. To identify the cations present in three individually assigned unknown mixtures that may contain
Determination of Total Hardness in Water by Automatic Titration
 TECHNICAL NOTE Determination of Total Hardness in Water by Automatic Titration No. 7 Introduction Total hardness due to calcium and magnesium in water is determined using the preprogrammed method, T7 Total
TECHNICAL NOTE Determination of Total Hardness in Water by Automatic Titration No. 7 Introduction Total hardness due to calcium and magnesium in water is determined using the preprogrammed method, T7 Total
Safety Data Sheet. Product Name: DetectX Palladium Screening Fluorescent Detection Kit. Section 1: Identification. Section 2: Hazard(s) Identification
 Safety Data Sheet Revision Date: 17 October 2016 Product Name: DetectX Palladium Screening Fluorescent Detection Kit Section 1: Identification Product Name: DetectX Palladium Screening Fluorescent Detection
Safety Data Sheet Revision Date: 17 October 2016 Product Name: DetectX Palladium Screening Fluorescent Detection Kit Section 1: Identification Product Name: DetectX Palladium Screening Fluorescent Detection
Determining the Empirical Formula of Zn and I Minneapolis Community and Technical College v.9.09
 Determining the Empirical Formula of Zn and I Minneapolis Community and Technical College v.9.09 I. Introduction The purpose of this experiment is for you to react zinc with iodine in order to determine
Determining the Empirical Formula of Zn and I Minneapolis Community and Technical College v.9.09 I. Introduction The purpose of this experiment is for you to react zinc with iodine in order to determine
GB Translated English of Chinese Standard: GB NATIONAL STANDARD
 Translated English of Chinese Standard: GB5009.5-2016 www.chinesestandard.net Sales@ChineseStandard.net GB NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA GB 5009.5-2016 National food safety standard
Translated English of Chinese Standard: GB5009.5-2016 www.chinesestandard.net Sales@ChineseStandard.net GB NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA GB 5009.5-2016 National food safety standard
Pectins. Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016
 Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Pectins This monograph was also published in: Compendium of Food Additive Specifications.
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Pectins This monograph was also published in: Compendium of Food Additive Specifications.
Chemistry Iodometric Determination of Vitamin C
 Chemistry 3200 Triiodide, I 3, is a mild oxidizing agent that is widely used in oxidation/reduction titrations. Triiodide is prepared by combining potassium iodide, KI, and potassium iodate, KI 3, in acidic
Chemistry 3200 Triiodide, I 3, is a mild oxidizing agent that is widely used in oxidation/reduction titrations. Triiodide is prepared by combining potassium iodide, KI, and potassium iodate, KI 3, in acidic
Determining the Molecular Mass of an Unknown Acid by Titration
 Determining the Molecular Mass of an Unknown Acid by Titration Objectives: To perform an analytical titration. To standardize a basic solution. To determine the equivalent mass of an unknown acid. Background:
Determining the Molecular Mass of an Unknown Acid by Titration Objectives: To perform an analytical titration. To standardize a basic solution. To determine the equivalent mass of an unknown acid. Background:
QUANTITATIVE TEST (CHEMICAL) FOR SUGARS IN SUGARCANE. Talha Saeed. Faisal Iftikhar. Mam AMMARA AINEE
 Assignment title QUANTITATIVE TEST (CHEMICAL) FOR SUGARS IN SUGARCANE Submitted by Subject Talha Saeed Roll # 37 Faisal Iftikhar Roll # 18 B.Sc. (Hons) Food Science and Technology 6 th Semester (Regular)
Assignment title QUANTITATIVE TEST (CHEMICAL) FOR SUGARS IN SUGARCANE Submitted by Subject Talha Saeed Roll # 37 Faisal Iftikhar Roll # 18 B.Sc. (Hons) Food Science and Technology 6 th Semester (Regular)
LAB: DIFFUSION ACROSS A SELECTIVELY PERMEABLE MEMBRANE
 LAB: DIFFUSION ACROSS A SELECTIVELY PERMEABLE MEMBRANE NAME: PERIOD: DATE: Building Background Knowledge: 1) SELECTIVELY PERMEABLE MEMBRANE: Every cell is surrounded by a selectively permeable membrane
LAB: DIFFUSION ACROSS A SELECTIVELY PERMEABLE MEMBRANE NAME: PERIOD: DATE: Building Background Knowledge: 1) SELECTIVELY PERMEABLE MEMBRANE: Every cell is surrounded by a selectively permeable membrane
Potentiometric determination. Application Bulletin 125/3 e. Branch General analytical laboratories; water analysis; beverages
 Simultaneous determination of calcium, magnesium, and photometric indication in water and beverage samples Branch General analytical laboratories; water analysis; beverages Keywords Titration; potentiometric
Simultaneous determination of calcium, magnesium, and photometric indication in water and beverage samples Branch General analytical laboratories; water analysis; beverages Keywords Titration; potentiometric
Standardization of a Base, Mass Percent of an Acid
 Experiment 7 Standardization of a Base, Mass Percent of an Acid Pre-Lab Assignment Before coming to lab: Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise.
Experiment 7 Standardization of a Base, Mass Percent of an Acid Pre-Lab Assignment Before coming to lab: Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise.
Determination of Langelier Index in Water
 Determination of Langelier Index in Water Based on Hach method 8073 ISO 10523: 2008 ISO 6059: 1984 ISO 9963: 1994 DOC316.52.93108 Total Alkalinity and Ca Hardness: 10-1000 mg/l CaCO3 1. Introduction Langelier
Determination of Langelier Index in Water Based on Hach method 8073 ISO 10523: 2008 ISO 6059: 1984 ISO 9963: 1994 DOC316.52.93108 Total Alkalinity and Ca Hardness: 10-1000 mg/l CaCO3 1. Introduction Langelier
IODOMETRIC TITRATION
 IODOMETRIC TITRATION Oxidizing agents In most iodometric titrations, when an excess of iodide ion is present, the tri-iodide ion is formed: I + I - I 3 - Since iodine is readily soluble in a solution of
IODOMETRIC TITRATION Oxidizing agents In most iodometric titrations, when an excess of iodide ion is present, the tri-iodide ion is formed: I + I - I 3 - Since iodine is readily soluble in a solution of
ANATOMY PHYSIOLOGY LAB 2-1 HOW POWERFUL IS YOUR ANTACID?
 ANATOMY PHYSIOLOGY LAB 2-1 HOW POWERFUL IS YOUR ANTACID? Introduction: Explore the properties and uses of antacids. Watch an "antacid in action" as it neutralizes simulated stomach acid. Determine the
ANATOMY PHYSIOLOGY LAB 2-1 HOW POWERFUL IS YOUR ANTACID? Introduction: Explore the properties and uses of antacids. Watch an "antacid in action" as it neutralizes simulated stomach acid. Determine the
GASTROINTESTINAL AND ANTIEMETIC DRUGS. Submitted by: Shaema M. Ali
 GASTROINTESTINAL AND ANTIEMETIC DRUGS Submitted by: Shaema M. Ali GASTROINTESTINAL AND ANTIEMETIC DRUGS by: Shaema M. Ali There are four common medical conditions involving the GI system 1) peptic ulcers
GASTROINTESTINAL AND ANTIEMETIC DRUGS Submitted by: Shaema M. Ali GASTROINTESTINAL AND ANTIEMETIC DRUGS by: Shaema M. Ali There are four common medical conditions involving the GI system 1) peptic ulcers
Kirkcaldy High School - Chemistry Higher Assignment Pupil Guide. Antioxidants
 Kirkcaldy High School - Chemistry Higher Assignment Pupil Guide Name: Class: Antioxidants This assignment is worth 20% of the overall marks for the Higher course assessment. The course is graded A-D. In
Kirkcaldy High School - Chemistry Higher Assignment Pupil Guide Name: Class: Antioxidants This assignment is worth 20% of the overall marks for the Higher course assessment. The course is graded A-D. In
LAB: DIFFUSION ACROSS A SELECTIVELY PERMEABLE MEMBRANE
 LAB: DIFFUSION ACROSS A SELECTIVELY PERMEABLE MEMBRANE NAME: PERIOD: DATE: Building Background Knowledge: 1) SELECTIVELY PERMEABLE MEMBRANE: Every cell is surrounded by a selectively permeable membrane
LAB: DIFFUSION ACROSS A SELECTIVELY PERMEABLE MEMBRANE NAME: PERIOD: DATE: Building Background Knowledge: 1) SELECTIVELY PERMEABLE MEMBRANE: Every cell is surrounded by a selectively permeable membrane
Purity Tests for Modified Starches
 Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Purity Tests for Modified Starches This monograph was also published in: Compendium
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Purity Tests for Modified Starches This monograph was also published in: Compendium
You should wear eye protection throughout this practical. Amylase is harmful, avoid contact with eyes and skin.
 Practical 3 - The identification of biological chemicals present in solutions This practical focuses on making decisions about measurements and observations, recording and presenting data and observations,
Practical 3 - The identification of biological chemicals present in solutions This practical focuses on making decisions about measurements and observations, recording and presenting data and observations,
KIDNEY STONE ANALYSIS
 KIDNEY STONE ANALYSIS Lesson aim To familiarise students with the use of chemical analysis in a healthcare related context, and to give students an idea of how the results can be used in diagnosis. Lesson
KIDNEY STONE ANALYSIS Lesson aim To familiarise students with the use of chemical analysis in a healthcare related context, and to give students an idea of how the results can be used in diagnosis. Lesson
Testing the capacity of biological buffer systems to resist changes in ph By P. H. Meter
 Testing the capacity of biological buffer systems to resist changes in ph By P. H. Meter Personal Engagement and Exploration Research Question: How do plant and animal tissues respond to changes in ph?
Testing the capacity of biological buffer systems to resist changes in ph By P. H. Meter Personal Engagement and Exploration Research Question: How do plant and animal tissues respond to changes in ph?
» Croscarmellose Sodium is a cross linked polymer of carboxymethylcellulose sodium.
 BRIEFING Croscarmellose Sodium, NF 22 page 2856 and page 702 of PF 30(2) [Mar. Apr. 2004]. A modification is made in the test for Degree of substitution to correct the endpoint color to agree with the
BRIEFING Croscarmellose Sodium, NF 22 page 2856 and page 702 of PF 30(2) [Mar. Apr. 2004]. A modification is made in the test for Degree of substitution to correct the endpoint color to agree with the
Application Bulletin
 No. 119/2 e Application Bulletin Of interest to: General analytical laboratories, Water; Food A 1, 2, 7, 11 Potentiometric determination of trace bromide and iodide in chlorides Summary Bromide is removed
No. 119/2 e Application Bulletin Of interest to: General analytical laboratories, Water; Food A 1, 2, 7, 11 Potentiometric determination of trace bromide and iodide in chlorides Summary Bromide is removed
Aluminium No 36 Flux FO360050
 1/5 Aluminium No 6 Flux FO60050 [SU] Industrial uses: Uses of substances as such or in preparations at industrial sites; [PC8] Welding and soldering products (with flux coatings or flux cores.), flux products;
1/5 Aluminium No 6 Flux FO60050 [SU] Industrial uses: Uses of substances as such or in preparations at industrial sites; [PC8] Welding and soldering products (with flux coatings or flux cores.), flux products;
Experiment 3. Potentiometric Titration Using a ph Electrode. information necessary for both purposes by monitoring the ph of the solution as the
 Experiment 3 Potentiometric Titration Using a Electrode Introduction Titrations are most commonly performed either to find out how much analyte is present or to measure equilibrium constants of the analyte.
Experiment 3 Potentiometric Titration Using a Electrode Introduction Titrations are most commonly performed either to find out how much analyte is present or to measure equilibrium constants of the analyte.
Core practical 1: Investigate how enzyme concentration affects the initial rate of an enzyme-controlled reaction
 Core practical 1 Student sheet Core practical 1: Investigate how enzyme concentration affects the initial Objectives To be able to measure the initial rate of enzyme activity To understand why measuring
Core practical 1 Student sheet Core practical 1: Investigate how enzyme concentration affects the initial Objectives To be able to measure the initial rate of enzyme activity To understand why measuring
Experiment: Iodometric Titration Analysis of Ascorbic Acid Chem251 modified 09/2018
 Experiment: Iodometric Titration Analysis of Ascorbic Acid Chem251 modified 09/2018 Experiment. Iodometric Titration of Ascorbic Acid. Objective: The goal of this lab is to determine the concentration
Experiment: Iodometric Titration Analysis of Ascorbic Acid Chem251 modified 09/2018 Experiment. Iodometric Titration of Ascorbic Acid. Objective: The goal of this lab is to determine the concentration
Organic Molecule Composition of Milk: Lab Investigation
 Name: Organic Molecule Composition of Milk: Lab Investigation Introduction & Background Milk & milk products have been a major food source from earliest recorded history. Milk is a natural, nutritionally
Name: Organic Molecule Composition of Milk: Lab Investigation Introduction & Background Milk & milk products have been a major food source from earliest recorded history. Milk is a natural, nutritionally
Complexometric Titration of Calcium in Antacids SUSB-017 Prepared by M. J. Akhtar and R. C. Kerber, SUNY at Stony Brook (Rev 1/13, RFS)
 61 SUSB- 017 017 exercise Complexometric Titration of Calcium in Antacids SUSB-017 Prepared by M. J. Akhtar and R. C. Kerber, SUNY at Stony Brook (Rev 1/13, RFS) purpose To determine the amount of calcium
61 SUSB- 017 017 exercise Complexometric Titration of Calcium in Antacids SUSB-017 Prepared by M. J. Akhtar and R. C. Kerber, SUNY at Stony Brook (Rev 1/13, RFS) purpose To determine the amount of calcium
Safety Data Sheet. May cause stomach ache, diarrhoea, nausea and vomiting.
 Page: 1 of 6 Product Code: BWP101, 1. IDENTIFICATION OF THE COMPANY AND PRODUCT Product Name: Product type: Usage: Company: E-mail: Website: Emergency Telephone: Detergent, cleaning agent Hand and machine
Page: 1 of 6 Product Code: BWP101, 1. IDENTIFICATION OF THE COMPANY AND PRODUCT Product Name: Product type: Usage: Company: E-mail: Website: Emergency Telephone: Detergent, cleaning agent Hand and machine
Feedstuffs Analysis G-22-1 PROTEIN
 Feedstuffs Analysis G-22-1 PROTEIN PRINCIPLE SCOPE Many modifications of the Kjeldahl method have been accepted for the estimation of protein in organic materials. It comprises sample oxidation and conversion
Feedstuffs Analysis G-22-1 PROTEIN PRINCIPLE SCOPE Many modifications of the Kjeldahl method have been accepted for the estimation of protein in organic materials. It comprises sample oxidation and conversion
Student Practical Guide (1) Milk of Magnesia
 School of Pharmacy Student Practical Guide (1b) Milk of Magnesia Facilitators Dr Mark Hewitt M.Hewitt@wlv.ac.uk Required Resources Pre-work: Read this guide Dr Rebecca Butler Rebecca.Butler@wlv.ac.uk Compulsory:
School of Pharmacy Student Practical Guide (1b) Milk of Magnesia Facilitators Dr Mark Hewitt M.Hewitt@wlv.ac.uk Required Resources Pre-work: Read this guide Dr Rebecca Butler Rebecca.Butler@wlv.ac.uk Compulsory:
Sodium Chloride Content in Ketchup by Precipitation Titration
 Background Sodiu Chloride Content in Ketchup by Precipitation Titration Sodiu chloride is one of the ost coon substances found in nature. Knowing the salt content in food products is iportant not only
Background Sodiu Chloride Content in Ketchup by Precipitation Titration Sodiu chloride is one of the ost coon substances found in nature. Knowing the salt content in food products is iportant not only
R & S Phrases. Introduction.
 R & S Phrases Introduction The so called R-Phrases give hints to special risks which may arise by the handling of hazardous substances or formulations. The letter R is the abbreviation for Risk. Accordin
R & S Phrases Introduction The so called R-Phrases give hints to special risks which may arise by the handling of hazardous substances or formulations. The letter R is the abbreviation for Risk. Accordin
CIPROXIFAN (cas ) MSDS
 Click http//www.guidechem.com/cas-184/184025-19-2.html for suppliers of this product CIPROXIFAN (cas 184025-19-2) MSDS 1. IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY/UNDERTAKING Product
Click http//www.guidechem.com/cas-184/184025-19-2.html for suppliers of this product CIPROXIFAN (cas 184025-19-2) MSDS 1. IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY/UNDERTAKING Product
Antacid therapy: Antacids side effects:
 Antacids Lec:5 When the general public ask why it takes antacids, the answer will include: 1.that uncomfortable feeling from overeating. 2. heart burn. 3. a growing hungry feeling between meals. Antacids
Antacids Lec:5 When the general public ask why it takes antacids, the answer will include: 1.that uncomfortable feeling from overeating. 2. heart burn. 3. a growing hungry feeling between meals. Antacids
MATERIAL SAFETY DATA SHEET
 Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
SAFETY DATA SHEET ARALDITE FUSION HARDENER
 1 / 4 REVISION DATE: September 2007 SDS NO.: 16447 SAFETY DATA SHEET 1 IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING PRODUCT NAME PRODUCT NO. 808300, 80840, 808416, 808423
1 / 4 REVISION DATE: September 2007 SDS NO.: 16447 SAFETY DATA SHEET 1 IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING PRODUCT NAME PRODUCT NO. 808300, 80840, 808416, 808423
Acetic Acid in Vinegar by Acid/Base Titration
 Acetic Acid in Vinegar by Acid/Base Titration Background Vinegar is typically ade fro the ferentation of alcoholic liquids such as wine. Its ain constituents are water and acetic acid, usually about 5
Acetic Acid in Vinegar by Acid/Base Titration Background Vinegar is typically ade fro the ferentation of alcoholic liquids such as wine. Its ain constituents are water and acetic acid, usually about 5
Take an initial volume reading and record it in your. 11/17/2014 ChemLab - Techniques - Titration
 Glassware Burets Flasks, Beakers, & Graduated Cylinders Pipets Repipets Volumetric Flasks Instruments Analytical Balance ph Meter: Analog ph Meter: Digital Spectrometer: Analog Spectrometer: Digital Spectrometer:
Glassware Burets Flasks, Beakers, & Graduated Cylinders Pipets Repipets Volumetric Flasks Instruments Analytical Balance ph Meter: Analog ph Meter: Digital Spectrometer: Analog Spectrometer: Digital Spectrometer:
Testing Protocol. Iodine Estimation of Salt
 Testing Protocol Iodine Estimation of Salt 1 Laboratory Procedure for Iodine Estimation of Salt Background material: Iodine is one of the first minerals recognized as essential for human health. Iodine
Testing Protocol Iodine Estimation of Salt 1 Laboratory Procedure for Iodine Estimation of Salt Background material: Iodine is one of the first minerals recognized as essential for human health. Iodine
6.02 Uniformity of Dosage Units
 6.02 Uniformity of Dosage Units Change 1. Content Uniformity, 3. Criteria and Table 6.02-2 as follows: 1. Content Uniformity Select not less than 30 units, and proceed as follows for the dosage form designated.
6.02 Uniformity of Dosage Units Change 1. Content Uniformity, 3. Criteria and Table 6.02-2 as follows: 1. Content Uniformity Select not less than 30 units, and proceed as follows for the dosage form designated.
Task: B Milk (20 marks for this task) Examination Rules:
 Task: B Milk (20 marks for this task) Examination Rules: 1. You are not allowed to bring any tools except any personal medicine or any personal medical equipment. 2. You must sit at your designated table.
Task: B Milk (20 marks for this task) Examination Rules: 1. You are not allowed to bring any tools except any personal medicine or any personal medical equipment. 2. You must sit at your designated table.
Chemical Pharmaceutical Quality Control. Prof.Dr.Joumaa Al- Zehouri Damascus university Faculty of Pharmacy
 Chemical Pharmaceutical Quality Control Prof.Dr.Joumaa Al- Zehouri Damascus university Faculty of Pharmacy COMPLEXOMETRIC REACTIONS AND TITRATIONS Prof.J.Al-Zehouri 1 ml of 0.1Msodium edetate is equivalent
Chemical Pharmaceutical Quality Control Prof.Dr.Joumaa Al- Zehouri Damascus university Faculty of Pharmacy COMPLEXOMETRIC REACTIONS AND TITRATIONS Prof.J.Al-Zehouri 1 ml of 0.1Msodium edetate is equivalent
SAC 17 Queen Mary University of London
 East Anglia Region of the RSC Analytical Division National Schools Analyst Competition Regional Heat Queen Mary University of London, Friday 24 th February 2017 School of Biological and Chemical Sciences,
East Anglia Region of the RSC Analytical Division National Schools Analyst Competition Regional Heat Queen Mary University of London, Friday 24 th February 2017 School of Biological and Chemical Sciences,
EXERCISE 3 Carbon Compounds
 LEARNING OBJECTIVES EXERCISE 3 Carbon Compounds Perform diagnostic tests to detect the presence of reducing sugars (Benedict s), starch (Lugol s), protein (Biuret), lipid (SudanIV) and sodium chloride
LEARNING OBJECTIVES EXERCISE 3 Carbon Compounds Perform diagnostic tests to detect the presence of reducing sugars (Benedict s), starch (Lugol s), protein (Biuret), lipid (SudanIV) and sodium chloride
SAFETY DATA SHEET according to 1907/2006/EC, Article 31
 CP148 v1.2 RS 56-8881,56-8890,56-8900,56-8904,56-890,800-646,800-685 RS review date: 01/12/15 SAFETY DATA SHEET according to 190/2006/EC, Article 31 SECTION 1: Identification of the substance/mixture and
CP148 v1.2 RS 56-8881,56-8890,56-8900,56-8904,56-890,800-646,800-685 RS review date: 01/12/15 SAFETY DATA SHEET according to 190/2006/EC, Article 31 SECTION 1: Identification of the substance/mixture and
SAFETY DATA SHEET FILDEK FLEXIBLE DECK COATING HARDENER
 SAFETY DATA SHEET 1 IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING PRODUCT NAME INTERNAL ID APPLICATION SUPPLIER FILDEK405H/4 Component of epoxy coating system Don Construction
SAFETY DATA SHEET 1 IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING PRODUCT NAME INTERNAL ID APPLICATION SUPPLIER FILDEK405H/4 Component of epoxy coating system Don Construction
This SDS adheres to the standards and regulatory requirements of Great Britain and may not meet the regulatory requirements in other countries.
 This SDS adheres to the standards and regulatory requirements of Great Britain and may not meet the regulatory requirements in other countries. 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING
This SDS adheres to the standards and regulatory requirements of Great Britain and may not meet the regulatory requirements in other countries. 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING
Material Safety Data Sheet
 PIONEER FOODS (PTY) LTD. t/a BOKOMO FOODS Reg. No. / Nr. 1957/000634/07 P.O Box 43028, Industria, 2042, Tel: 011-474 2402, Fax: 011-474 4450 Material Safety Data Sheet HYM-LURE - PASTE 940001 --- Effective
PIONEER FOODS (PTY) LTD. t/a BOKOMO FOODS Reg. No. / Nr. 1957/000634/07 P.O Box 43028, Industria, 2042, Tel: 011-474 2402, Fax: 011-474 4450 Material Safety Data Sheet HYM-LURE - PASTE 940001 --- Effective
E55A GELATIN, GELLING GRADE Gelatina
 00-0PDG.pdf 0 0 0 0 EA GELATIN, GELLING GRADE Gelatina DEFINITION Purified protein obtained from collagen of animals (including fish and poultry) by partial alkaline and/or acid hydrolysis, by enzymatic
00-0PDG.pdf 0 0 0 0 EA GELATIN, GELLING GRADE Gelatina DEFINITION Purified protein obtained from collagen of animals (including fish and poultry) by partial alkaline and/or acid hydrolysis, by enzymatic
Peptic ulcer disease Disorders of the esophagus
 Peptic ulcer disease Disorders of the esophagus Peptic ulcer disease Burning epigastric pain Exacerbated by fasting Improved with meals Ulcer: disruption of mucosal integrity >5 mm in size, with depth
Peptic ulcer disease Disorders of the esophagus Peptic ulcer disease Burning epigastric pain Exacerbated by fasting Improved with meals Ulcer: disruption of mucosal integrity >5 mm in size, with depth
Sodium Reference Electrode Filling Solution 2M NH 4 Cl Material Safety Data Sheet
 900010 Sodium Reference Electrode Filling Solution 2M NH 4 Cl I. PRODUCT IDENTIFICATION: Sodium Reference Electrode Filling Solution 2M NH 4 Cl 900010 NFPA RATINGS: HEALTH: 1 FLAMMABILITY: 0 REACTIVITY:
900010 Sodium Reference Electrode Filling Solution 2M NH 4 Cl I. PRODUCT IDENTIFICATION: Sodium Reference Electrode Filling Solution 2M NH 4 Cl 900010 NFPA RATINGS: HEALTH: 1 FLAMMABILITY: 0 REACTIVITY:
Material Safety Data Sheet Version 4.2
 Material Safety Data Sheet Version 4.2 Preparation / Revision Date 8/25/2011 1. PRODUCT AND COMPANY IDENTIFICATION Chemical Name Montelukast Dicyclohexylamine Salt Expiry Date 8/23/2014 Catalogue # M567990
Material Safety Data Sheet Version 4.2 Preparation / Revision Date 8/25/2011 1. PRODUCT AND COMPANY IDENTIFICATION Chemical Name Montelukast Dicyclohexylamine Salt Expiry Date 8/23/2014 Catalogue # M567990
Properties of Alcohols and Phenols Experiment #3
 Properties of Alcohols and Phenols Experiment #3 Objectives: To observe the solubility of alcohols relative to their chemical structure, to perform chemical tests to distinguish primary, secondary and
Properties of Alcohols and Phenols Experiment #3 Objectives: To observe the solubility of alcohols relative to their chemical structure, to perform chemical tests to distinguish primary, secondary and
Material Safety Data Sheet SunClean Concrete Tile Cleaner
 1. Production Description Product Name: Concrete Tile Cleaner Application of the substance: Cleaner Acid Based Manufacturer/Supplier: Sun Clean Cleaning Ind. LLC P.O. Box 41475, SHARJAH UAE Tel: +971 (6)
1. Production Description Product Name: Concrete Tile Cleaner Application of the substance: Cleaner Acid Based Manufacturer/Supplier: Sun Clean Cleaning Ind. LLC P.O. Box 41475, SHARJAH UAE Tel: +971 (6)
Station 1. Identify (= name) the spaces or structures labeled 1 9.
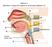 Station 1. Identify (= name) the spaces or structures labeled 1 9. 1 9 8 2 7 3 4 5 6 Station 2. Identify/name the parts or structures labeled 10 20. 10. Is this the right or left lung? 15 13 14 16 11 12
Station 1. Identify (= name) the spaces or structures labeled 1 9. 1 9 8 2 7 3 4 5 6 Station 2. Identify/name the parts or structures labeled 10 20. 10. Is this the right or left lung? 15 13 14 16 11 12
R- a S- věty v angličtině používané v bezpečnostních listech Single R-Phrases
 Single R-Phrases R1: Explosive when dry. R2: Risk of explosion by shock, friction, fire or other sources of ignition. R3: Extreme risk of explosion by shock, friction, fire or other sources of ignition.
Single R-Phrases R1: Explosive when dry. R2: Risk of explosion by shock, friction, fire or other sources of ignition. R3: Extreme risk of explosion by shock, friction, fire or other sources of ignition.
Identification and qualitative Analysis. of Renal Calculi
 Identification and qualitative Analysis of Renal Calculi 1 -Renal Calculi: Kidney stones, renal calculi or renal lithiasis (stone formation) are small, hard deposits that form inside your kidneys. The
Identification and qualitative Analysis of Renal Calculi 1 -Renal Calculi: Kidney stones, renal calculi or renal lithiasis (stone formation) are small, hard deposits that form inside your kidneys. The
Class 10 Science - Sample Paper Set I
 Time allowed: 3 hours; Maximum marks: 90 General Instructions: a) All Questions are compulsory b) The Question Paper consists of 42 Questions divided in to four sections A, B, C and D c) Section- A comprises
Time allowed: 3 hours; Maximum marks: 90 General Instructions: a) All Questions are compulsory b) The Question Paper consists of 42 Questions divided in to four sections A, B, C and D c) Section- A comprises
Instruction Manual Updated 8/27/2013 Ver. 1.1
 Water Analysis Kit Part No. 144-95 Instruction Manual Updated 8/27/2013 Ver. 1.1 OFI Testing Equipment, Inc. 11302 Steeplecrest Dr. Houston, Texas 77065 U.S.A. Tele: 832.320.7300 Fax: 713.880.9886 www.ofite.com
Water Analysis Kit Part No. 144-95 Instruction Manual Updated 8/27/2013 Ver. 1.1 OFI Testing Equipment, Inc. 11302 Steeplecrest Dr. Houston, Texas 77065 U.S.A. Tele: 832.320.7300 Fax: 713.880.9886 www.ofite.com
SAFETY DATA SHEET Floorline Dry Foam Shampoo
 SAFETY DATA SHEET SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product name Product number FL1002, 531022, 059401, 059401 1.2. Relevant identified
SAFETY DATA SHEET SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product name Product number FL1002, 531022, 059401, 059401 1.2. Relevant identified
Genomic DNA Isolation Reagent Kit (Blood/Cultured Cell/Tissue)
 Genomic DNA Isolation Reagent Kit (Blood/Cultured Cell/Tissue) 1. IDENTIFICATION OF THE PREPARATION/SUBSTANCE AND OF THE COMPANY Preparation Name: Genomic DNA Isolation Reagent Kit (Blood/Cultured Cell/Tissue)
Genomic DNA Isolation Reagent Kit (Blood/Cultured Cell/Tissue) 1. IDENTIFICATION OF THE PREPARATION/SUBSTANCE AND OF THE COMPANY Preparation Name: Genomic DNA Isolation Reagent Kit (Blood/Cultured Cell/Tissue)
Cross-linking polymers alginate worms
 68 Inspirational chemistry Cross-linking polymers alginate worms Index 3.1.9 3 sheets This practical provides a fun look at cross-linking and a chance to explore some of the many and varied uses of sodium
68 Inspirational chemistry Cross-linking polymers alginate worms Index 3.1.9 3 sheets This practical provides a fun look at cross-linking and a chance to explore some of the many and varied uses of sodium
--> Buy True-PDF --> Auto-delivered in 0~10 minutes. GB Translated English of Chinese Standard: GB1886.
 Translated English of Chinese Standard: GB1886.235-2016 www.chinesestandard.net Buy True-PDF Auto-delivery. Sales@ChineseStandard.net NATIONAL STANDARD OF THE GB PEOPLE S REPUBLIC OF CHINA National Food
Translated English of Chinese Standard: GB1886.235-2016 www.chinesestandard.net Buy True-PDF Auto-delivery. Sales@ChineseStandard.net NATIONAL STANDARD OF THE GB PEOPLE S REPUBLIC OF CHINA National Food
Product Data For Research Use Only. Not For Use In Diagnostic Procedures
 Product Data For Research Use Only. Not For Use In Diagnostic Procedures Cultrex Cell Staining Kit Related product available from Trevigen. Catalog# Description Size Cultrex In Vitro Angiogenesis Assay
Product Data For Research Use Only. Not For Use In Diagnostic Procedures Cultrex Cell Staining Kit Related product available from Trevigen. Catalog# Description Size Cultrex In Vitro Angiogenesis Assay
This SDS adheres to the standards and regulatory requirements of Great Britain and may not meet the regulatory requirements in other countries.
 This SDS adheres to the standards and regulatory requirements of Great Britain and may not meet the regulatory requirements in other countries. 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE
This SDS adheres to the standards and regulatory requirements of Great Britain and may not meet the regulatory requirements in other countries. 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE
Analytical Method for 2, 4, 5-T (Targeted to Agricultural, Animal and Fishery Products)
 Analytical Method for 2, 4, 5-T (Targeted to Agricultural, Animal and Fishery Products) The target compound to be determined is 2, 4, 5-T. 1. Instrument Liquid Chromatograph-tandem mass spectrometer (LC-MS/MS)
Analytical Method for 2, 4, 5-T (Targeted to Agricultural, Animal and Fishery Products) The target compound to be determined is 2, 4, 5-T. 1. Instrument Liquid Chromatograph-tandem mass spectrometer (LC-MS/MS)
1 out of 8. Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 86th Meeting 2018 ERYTHROSINE
 1 out of 8 Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 86th Meeting 2018 ERYTHROSINE This monograph was also published in: Compendium of Food
1 out of 8 Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 86th Meeting 2018 ERYTHROSINE This monograph was also published in: Compendium of Food
COLE-PARMER LABORATORY SURFACTANT ION ELECTRODE INSTRUCTION MANUAL
 COLE-PARMER LABORATORY SURFACTANT ION ELECTRODE INSTRUCTION MANUAL Cole-Parmer Instrument Company (800)323-4340 Fax:(847)247-2929 625 East Bunker Court, Vernon Hills, Illinois 60061 http://www.coleparmer.com
COLE-PARMER LABORATORY SURFACTANT ION ELECTRODE INSTRUCTION MANUAL Cole-Parmer Instrument Company (800)323-4340 Fax:(847)247-2929 625 East Bunker Court, Vernon Hills, Illinois 60061 http://www.coleparmer.com
University of Malta Department of Chemistry GUIDE TO RISK ASSESSMENT
 University of Malta Department of Chemistry GUIDE TO RISK ASSESSMENT Obtain the Material Safety Data Sheets (MSDS s) for all chemicals used in the practical. These are readily available online. Determine
University of Malta Department of Chemistry GUIDE TO RISK ASSESSMENT Obtain the Material Safety Data Sheets (MSDS s) for all chemicals used in the practical. These are readily available online. Determine
BRIEFING. Pharmacopeial Discussion Group Sign Off Document Attributes EP JP USP Definition Identification B Identification C + + +
 BRIEFING Edetate Calcium Disodium, USP 29 page 779. The Japanese Pharmacopoeia is the coordinating pharmacopeia for the international harmonization of the compendial standards for the Edetate Calcium Disodium
BRIEFING Edetate Calcium Disodium, USP 29 page 779. The Japanese Pharmacopoeia is the coordinating pharmacopeia for the international harmonization of the compendial standards for the Edetate Calcium Disodium
This SDS adheres to the standards and regulatory requirements of Great Britain and may not meet the regulatory requirements in other countries.
 This SDS adheres to the standards and regulatory requirements of Great Britain and may not meet the regulatory requirements in other countries. 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE
This SDS adheres to the standards and regulatory requirements of Great Britain and may not meet the regulatory requirements in other countries. 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE
Dri-Sate DC, DR and DF Series Dry Acid Concentrate for Hemodialysis 45X, 36.83X and 35X Dilution
 Section 1. Product and Company Identification Product Name: Company Identification: Dri-Sate DC, DR and DF Series Dry Acid Concentrate for Hemodialysis 45X, 36.83X and 35X Dilution Rockwell Medical 30142
Section 1. Product and Company Identification Product Name: Company Identification: Dri-Sate DC, DR and DF Series Dry Acid Concentrate for Hemodialysis 45X, 36.83X and 35X Dilution Rockwell Medical 30142
