CHY2026: General Biochemistry. Unit 4:Amino Acid Chemistry
|
|
|
- Jodie Sims
- 6 years ago
- Views:
Transcription
1 CHY2026: General Biochemistry Unit 4:Amino Acid Chemistry
2
3 Hydrogen Amino group Carboxyl Group Unique side chain (R-group) R Central Carbon
4 Amino Acid Structure The molecule is comprised of the elements carbon (C), hydrogen (H), oxygen (O), nitrogen (N) and small quantities of sulphur (S) CHONs The α- carbon in all amino acids is asymmetric (except glycine) Because of this two optically active forms exist The NH 2 is to the right of the COOH: D-form The NH 2 is to the left of the COOH: L-form Only L- forms are used to make proteins
5 Amino Acid Structure Glycine does not contain any asymmetric carbon This is the simplest amino acid
6 Properties of Amino Acid Amino acids are dipolar ions (zwitterions) in solution In the dipolar form amino group becomes positively charged (NH 3+ ) and the carboxylic acid becomes negatively charged (COO - ) on2.jpg The dipolar ion exists as a neutral ion and will have zero mobility in electrophoresis
7 Properties of Amino Acid Amino Acids are amphoteric There is no ph at which a.a have no ionic character The ph value at which the number of cations is equal that of anion is called the isoelectric point (pi) Every a.a have a different pi: The 3 basic a.a have higher pi and the 2 acidic a.a have lower pi At ph lower than the pi, the protein will have a net positive charge and as a cation will migrate towards the negative electrode (cathode) At ph higher than the pi, the protein will have a net negative charge and the anion will migrate towards the positive electrode (anode)
8 Properties of Amino Acid Notice that the isoelectric point depends on the amino acid structure in a predictable way acidic amino acids: aspartic acid (2.8), glutamic acid (3.2) neutral amino acids: (5.0 to 6.3) basic amino acids: lysine (9.7), arginine (10.8), histidine (7.6) Electrophoresis uses differences in isoelectric points to separate mixtures of amino acids. A streak of the amino acid mixture is placed in the center of a layer of acrylamide gel or a piece of filter paper wet with a buffer solution. Two electrodes are placed in contact with the edges of the gel or paper, and a potential of several thousand volts is applied across the electrodes. Positively charged (cationic) amino acids are attracted to the negative electrode (the cathode), and negatively charged (anionic) amino acids are attracted to the positive electrode (the anode). An amino acid at its isoelectric point has no net charge, so it does not move.
9 Amino Acids There are 20 amino acids that can make up proteins 9 essential amino acids 11 non essential amino acids Essential amino acids they are not synthesized in the body and are normally required in the diet Non essential amino acids they are synthesized de novo in humans
10 Essential Histidine (His) Isoleucine (Ile) Leucine (Leu) Lysine (Lys) Methionine (Met) Phenylalanine (Phe) Threonine (Thr) Tryptophan (Trp) Valine (Val) Non essential Alanine (Ala) Arginine (Arg) Asparagine (Asn) Aspartic acid (Asp) Cysteine (Cys) Glutamic acid (Glu) Glutamine (Gln) Glycine (Gly) Proline (Pro) Serine (Ser) Tyrosine (Tyr)
11 Amino Acids Amino acids can be grouped based on the properties of the R group non polar polar uncharged acidic (negatively charged) basic (positively charged)
12
13 Non polar Aromatic R group
14
15 Polar uncharged aromatic R group
16 Acidic R group (negatively charged R group)
17 Basic R group (positively charged R group)
18 Acid Dissociation Constant: pka The dissociable α-cooh and α-nh + 3 are responsible for the two characteristic pk values A third amino acid is indicative of a dissociation in the R group (pk R ) (ph and pk a are notations for proton concentration and equilibrium constant for ionization pk a is a measure of the tendency of a group to give up protons) Ionic properties of amino acids α-carboxyl group: pka s range from 2.0 to 2.4 α-amino group: pka s range from 9.0 to 9.8 Side chains Acidic pka s for asp, glu Basic pka s for his, arg, lys
19 Proteins Proteins are made up of amino acids linked together by peptide bonds (condensation reaction)
20 The peptide bonds in proteins are metastable, meaning that in the presence of water they will break spontaneously, releasing 2-4 kcal mol -1 of free energy, but this process is extremely slow In living organisms, the process is facilitated by enzymes Living organisms also employ enzymes to form peptide bonds; this process requires free energy
21 Proteins 2 amino acids react dipeptide 3 amino acids tripeptide Less than 10 amino acids - oligopeptide 4 or more amino acids polypeptide Polypeptide with a MW > 6000 or 100 amino acids protein Each amino acid in a chain is termed a residue The two ends of the chain are named amino acid terminal (N- terminal) and carboxylic terminal (C-terminal) respectively These terminals are the only ionizable groups (except for the side chain) in a protein
22 Hydrolysis Proteins can be hydrolyzed (using acid or a suitable enzyme) to form individual amino acids
23 Biological Roles of Peptides Intermediates for the formation of proteins Antibacterial properties e.g. penicillin Growth factors e.g. folic acid Hormones Control oxidation-reduction potential of cell e.g. glutathione Increased levels in urine may be indicative of psychological disorders such as depression and schizophrenia
24 Industrial Importance of Peptides Inhibitory peptides are used in clinical research to examine the effects of peptides on the inhibition of cancer proteins and other diseases Peptides are used in imaging where, specific peptides in the fluorescing material help with early detection of potentially life-threatening cancers Peptides are used in skin care to increase the amount of collagen being produced in the skin, stimulate fibroblast growth, and stimulate the production of keratinocytes The zero-calorie sweetener found in many diet beverages is a synthesized peptide
25 Proteins They are the most abundant intracellular macromolecule most of the protein mass is found in the skeletal muscle Proteins found in animal contains % sulphur except for insulin which contains 3.4%
26 Biological Role of Proteins They are biological catalysts enzymes They act as carrier molecules transporting small molecules and ions e.g. haemoglobin (protein) transports oxygen in the erythrocytes Responsible for the high tensile strength of the skin and bone Immunoregulation Receptor proteins help in the transmission of nerve impulses Major component of muscle
27 Protein Classification 1. Classification based on source (a) Animal protein higher quality protein (b) Plant protein low quality protein (does not mean it s a poor protein source) 2. Classification based on shape (a) Globular/Corpuscular proteins: spherical or ovoid shape, soluble in water or aqueous media. Tertiary and quaternary structures are usually associated with this class Further classified as: Cytochrome C Blood proteins: serum albumin, glycoproteins, antibodies, haemoglobin, hormones Enzymes Nutrient Proteins (b) Fibrous/Fibrillar proteins: serve in structural or protective roles They can stretch and later recoil e.g. collagens, elastins, keratins and fibroin
28 Protein Structure Protein structure can be grouped into four levels of organization 1. Primary structure linear sequence of amino acids. Main linkage peptide bonds How important is the primary sequence? Some small change in a.a can make a big difference Normal Hb has 2 alpha chains and 2 beta chains Normal Hb Sickle cell Hb -Thr-Pro-Glu-Glu-Lys-Ala -Thr-Pro-Val-Glu-Lys-Ala Red blood cells carrying HbS behave normally when there is an ample supply of oxygen. Reduced oxygen concentration results in the rbc becoming sickle-shaped Sickled cells may clog capillaries The body s defenses destroy the clogging cells and the loss of blood cells causes anaemia
29 Hydroxyurea (Droxia) is used to control symptoms of the disease It prompts the bone marrow to manufacture HbF which does not have the beta chains where mutation occurs Rbc containing HbF do not sickle. Even with hydoxyurea, the body still produces HbS but the presence of cells with HbF reduces the concentration of cell with HbS hence relieving the symptoms of the disease
30 2. Secondary structure spatial arrangement of a portion of the polypeptide. Main linkage- H bonding (a) α- helix e.g. α-keratin (b) β- pleated sheets e.g. silk fibroin (c) random coil α-keratin Silk fibroin
31 Protein Structure 3. Tertiary structure describe the shape of the entire polypeptide folding of the helices of globular proteins. All the chemical bonds are found in the structure e.g. myoglobin (cardiac and red skeletal muscle) Chaperones a protein that helps other proteins to fold into the biologically active conformation and enables partially denatured proteins to regain their biologically active conformation
32 4. Quaternary structure spatial arrangement of many polypeptide subunits all chemical bonds are found in the structure e.g. haemoglobin (tetrameric protein)
33 Haemoglobin In the adult human, Hb (tetramer) consists of 4 polypeptide chains (globins) 2 identical α chains (141 residues) and 2 identical β chains (146 residues) Each globin chain surrounds an iron-containing heme It is a conjugated proteins proteins that contains non-amino acid portion Prosthetic group the non-amino acid portion of a conjugated protein The globins are the amino acid portion and the prosthetic groups are the haem units Myoglobin Myoglobin is a small oxygen heme protein found in the muscle Myoglobin consist of 1 polypeptide chain, 153 residues long
34 Comparison between Haemoglobin and Myoglobin Haemoglobin Myoglobin 1 Hb can bind to 4 O 2 1 myoglobin binds to 1 O 2 The binding of oxygen to Hb exhibit positive cooperativity (when 1 oxygen molecule is bound it becomes easier for the other oxygen to bind Hb functions in oxygen transport. It must bind strongly to oxygen and be able to release it easily Oxygen binding to myoglobin does not exhibit positive cooperativity Myoglobin has the function of oxygen storage in muscles
35
36 Conformation of Proteins Protein folding is the physical process by which a polypeptide folds into its characteristic and functional three dimensional structure from random coil Each protein exists as an unfolded polypeptide when translated from a sequence of mrna to a linear chain of amino acids This polypeptide lacks any developed three dimensional structure Amino acids interact with each other to produce a well-defined three dimensional structure, the folded protein is known as the native state The resulting three dimensional structure is determined by the amino acid sequence The correct three dimensional structure is essential to function, although some parts of functional proteins may remain unfolded
37 Conformation of Proteins Failure to fold into native structure produces inactive proteins that are usually toxic Several neurodegenerative and other diseases are believed to result from the accumulation of amyloid fibrills formed by misfolded proteins Many allergies are caused by the folding of the proteins, for the immune system does not produce antibodies for certain protein structures
38 Chemical Bonds in Protein Structure 1. Primary Bond: Peptide bond backbone of the protein chain 2. Secondary Bonds: Hold the chain in its natural configuration (a) Disulphide Bond formed by the oxidation of the thiol or sulphurhydryl groups (-SH) of two cysteine residues to form cystine
39 Disulphide linkages can occur in two cysteine molecules that are apart in a polypeptide chain. This will cause the polypeptide chain to be folded back on itself e.g. oxytocin and insulin
40 (c) Hydrogen Bond: This occurs when a group containing a H atom is bonded to the electronegative atoms O or N. H-bond plays a role in stabilizing some conformations of the polypeptide chain e.g. silk fibroin 3. Nonpolar/ Hydrophobic Bond formed among amino acids with hydrophobic side groups 4. Ionic/Electrostatic Bond/Salt Bridge/Salt linkage: Ions possessing similar charge repel each other ions possessing dissimilar charge attract each other e.g. acidic and basic R groups Ionic bonds are responsible for maintaining the folded structure of globular proteins
41 Denaturation This refers to a change in the properties of a protein resulting in a loss of biological activity Denaturation can be brought about by physical agents (a) shaking (b) heat treatment (c) cooling or freezing (d) UV rays and chemical agents (a) ionizing radiation (b) organic solvents (c) detergents When proteins are denatured the molecule unfolds causing disorganization of internal structure... (bonds are broken) Some denatured proteins cannot be brought back to their original state...however generally its a reversible process
42 Methods of Protein Separation Electrophoresis - used to separate complex mixtures of proteins and to purify the protein for subsequent applications Western blotting is used to identify a specific protein in a complex mixture of proteins. The technique exploits both the efficiency of SDS-PAGE to separate a mixture of proteins into distinct protein bands, and the ability of immunochemical reagents to interact specifically with a given protein antigen Column Chromatography can be used to fractionate proteins. The mixture of proteins in solution is passed through a column containing a porous solid matrix The 3 types of matrices used are: (1) Ion-exchange chromatography, (2) gel-filtration chromatography and (3) affinity chromatography
43 Protein Quantification Knowing the quantity of a protein after each separation step is useful in checking the progress of purification and evaluating the technique's efficiency The concentration of a protein can be measured by immunological techniques such as ELISA or Western Blotting Activity can be measured using fluorescent techniques Mass spectrometry is another accurate analytical technique for determining protein mass. In this technique, atoms are ionized through a machine and passed through a vacuum into the detector
44 Industrial Importance of Proteins Denatured protein - used in the formation of gelatin, which is used in many foods, in the pharmaceutical and cosmetic industries Collagen has been widely used in cosmetic surgery, as a healing aid for burn patients for reconstruction of bone and a wide variety of dental, orthopedic and surgical purposes Collagens are also used in the construction of artificial skin substitutes used in the management of severe burns Corn gluten is used in feed concentrates and in small amounts as extenders for commercial resins and glues Corn gluten is sometimes destarched by acid or enzyme hydrolysis to yield a product that is almost entirely protein and is used in paints. Sometimes it is broken down by complete hydrolysis to yield monosodium glutamate, a seasoning for food
Biomolecules: amino acids
 Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Chemical Nature of the Amino Acids. Table of a-amino Acids Found in Proteins
 Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5
 Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
Chemistry 121 Winter 17
 Chemistry 121 Winter 17 Introduction to Organic Chemistry and Biochemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941;
Chemistry 121 Winter 17 Introduction to Organic Chemistry and Biochemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941;
Copyright 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings
 Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Molecular Biology. general transfer: occurs normally in cells. special transfer: occurs only in the laboratory in specific conditions.
 Chapter 9: Proteins Molecular Biology replication general transfer: occurs normally in cells transcription special transfer: occurs only in the laboratory in specific conditions translation unknown transfer:
Chapter 9: Proteins Molecular Biology replication general transfer: occurs normally in cells transcription special transfer: occurs only in the laboratory in specific conditions translation unknown transfer:
Human Biochemistry Option B
 Human Biochemistry Option B A look ahead... Your body has many functions to perform every day: Structural support, genetic information, communication, energy supply, metabolism Right now, thousands of
Human Biochemistry Option B A look ahead... Your body has many functions to perform every day: Structural support, genetic information, communication, energy supply, metabolism Right now, thousands of
AP Bio. Protiens Chapter 5 1
 Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
Amino acids. (Foundation Block) Dr. Essa Sabi
 Amino acids (Foundation Block) Dr. Essa Sabi Learning outcomes What are the amino acids? General structure. Classification of amino acids. Optical properties. Amino acid configuration. Non-standard amino
Amino acids (Foundation Block) Dr. Essa Sabi Learning outcomes What are the amino acids? General structure. Classification of amino acids. Optical properties. Amino acid configuration. Non-standard amino
1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids
 Amino acids 1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids 5-To understand amino acids synthesis Amino
Amino acids 1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids 5-To understand amino acids synthesis Amino
Objective: You will be able to explain how the subcomponents of
 Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Macromolecules of Life -3 Amino Acids & Proteins
 Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
CS612 - Algorithms in Bioinformatics
 Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Methionine (Met or M)
 Fig. 5-17 Nonpolar Fig. 5-17a Nonpolar Glycine (Gly or G) Alanine (Ala or A) Valine (Val or V) Leucine (Leu or L) Isoleucine (Ile or I) Methionine (Met or M) Phenylalanine (Phe or F) Polar Trypotphan (Trp
Fig. 5-17 Nonpolar Fig. 5-17a Nonpolar Glycine (Gly or G) Alanine (Ala or A) Valine (Val or V) Leucine (Leu or L) Isoleucine (Ile or I) Methionine (Met or M) Phenylalanine (Phe or F) Polar Trypotphan (Trp
Reactions and amino acids structure & properties
 Lecture 2: Reactions and amino acids structure & properties Dr. Sameh Sarray Hlaoui Common Functional Groups Common Biochemical Reactions AH + B A + BH Oxidation-Reduction A-H + B-OH + energy ª A-B + H
Lecture 2: Reactions and amino acids structure & properties Dr. Sameh Sarray Hlaoui Common Functional Groups Common Biochemical Reactions AH + B A + BH Oxidation-Reduction A-H + B-OH + energy ª A-B + H
Gentilucci, Amino Acids, Peptides, and Proteins. Peptides and proteins are polymers of amino acids linked together by amide bonds CH 3
 Amino Acids Peptides and proteins are polymers of amino acids linked together by amide bonds Aliphatic Side-Chain Amino Acids - - H CH glycine alanine 3 proline valine CH CH 3 - leucine - isoleucine CH
Amino Acids Peptides and proteins are polymers of amino acids linked together by amide bonds Aliphatic Side-Chain Amino Acids - - H CH glycine alanine 3 proline valine CH CH 3 - leucine - isoleucine CH
Proteins are sometimes only produced in one cell type or cell compartment (brain has 15,000 expressed proteins, gut has 2,000).
 Lecture 2: Principles of Protein Structure: Amino Acids Why study proteins? Proteins underpin every aspect of biological activity and therefore are targets for drug design and medicinal therapy, and in
Lecture 2: Principles of Protein Structure: Amino Acids Why study proteins? Proteins underpin every aspect of biological activity and therefore are targets for drug design and medicinal therapy, and in
PHAR3316 Pharmacy biochemistry Exam #2 Fall 2010 KEY
 1. How many protons is(are) lost when the amino acid Asparagine is titrated from its fully protonated state to a fully deprotonated state? A. 0 B. 1 * C. 2 D. 3 E. none Correct Answer: C (this question
1. How many protons is(are) lost when the amino acid Asparagine is titrated from its fully protonated state to a fully deprotonated state? A. 0 B. 1 * C. 2 D. 3 E. none Correct Answer: C (this question
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A
 Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
PROTEINS. Building blocks, structure and function. Aim: You will have a clear picture of protein construction and their general properties
 PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
Page 8/6: The cell. Where to start: Proteins (control a cell) (start/end products)
 Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
The Structure and Function of Macromolecules
 The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
Lecture 4. Grouping Amino Acid 7/1/10. Proteins. Amino Acids. Where Are Proteins Located. Nonpolar Amino Acids
 Proteins Lecture 4 Proteins - Composition of Proteins (Amino Acids) Chapter 21 ection 1-6! Proteins are compounds of high molar mass consisting almost entirely of amino acid chain(s)! Molar masses range
Proteins Lecture 4 Proteins - Composition of Proteins (Amino Acids) Chapter 21 ection 1-6! Proteins are compounds of high molar mass consisting almost entirely of amino acid chain(s)! Molar masses range
Chemistry 20 Chapter 14 Proteins
 Chapter 14 Proteins Proteins: all proteins in humans are polymers made up from 20 different amino acids. Proteins provide structure in membranes, build cartilage, muscles, hair, nails, and connective tissue
Chapter 14 Proteins Proteins: all proteins in humans are polymers made up from 20 different amino acids. Proteins provide structure in membranes, build cartilage, muscles, hair, nails, and connective tissue
BIO 311C Spring Lecture 15 Friday 26 Feb. 1
 BIO 311C Spring 2010 Lecture 15 Friday 26 Feb. 1 Illustration of a Polypeptide amino acids peptide bonds Review Polypeptide (chain) See textbook, Fig 5.21, p. 82 for a more clear illustration Folding and
BIO 311C Spring 2010 Lecture 15 Friday 26 Feb. 1 Illustration of a Polypeptide amino acids peptide bonds Review Polypeptide (chain) See textbook, Fig 5.21, p. 82 for a more clear illustration Folding and
9/6/2011. Amino Acids. C α. Nonpolar, aliphatic R groups
 Amino Acids Side chains (R groups) vary in: size shape charge hydrogen-bonding capacity hydrophobic character chemical reactivity C α Nonpolar, aliphatic R groups Glycine (Gly, G) Alanine (Ala, A) Valine
Amino Acids Side chains (R groups) vary in: size shape charge hydrogen-bonding capacity hydrophobic character chemical reactivity C α Nonpolar, aliphatic R groups Glycine (Gly, G) Alanine (Ala, A) Valine
Chapter 20 and GHW#10 Questions. Proteins
 Chapter 20 and GHW#10 Questions Proteins Proteins Naturally occurring bioorganic polyamide polymers containing a sequence of various combinations of 20 amino acids. Amino acids contain the elements carbon,
Chapter 20 and GHW#10 Questions Proteins Proteins Naturally occurring bioorganic polyamide polymers containing a sequence of various combinations of 20 amino acids. Amino acids contain the elements carbon,
Amino Acids. Amino Acids. Fundamentals. While their name implies that amino acids are compounds that contain an NH. 3 and CO NH 3
 Fundamentals While their name implies that amino acids are compounds that contain an 2 group and a 2 group, these groups are actually present as 3 and 2 respectively. They are classified as α, β, γ, etc..
Fundamentals While their name implies that amino acids are compounds that contain an 2 group and a 2 group, these groups are actually present as 3 and 2 respectively. They are classified as α, β, γ, etc..
Chapter 21 Lecture Outline
 Chapter 21 Lecture Outline Amino Acids, Proteins, and Enzymes! Introduction! Proteins are biomolecules that contain many amide bonds, formed by joining amino acids. Prepared by Andrea D. Leonard University
Chapter 21 Lecture Outline Amino Acids, Proteins, and Enzymes! Introduction! Proteins are biomolecules that contain many amide bonds, formed by joining amino acids. Prepared by Andrea D. Leonard University
Introduction to Biochemistry Midterm exam )ومن أحياها(
 Introduction to Biochemistry Midterm exam 2016-2017 )ومن أحياها( 1. Which of the following amino (in a peptide chain) would probably be found at a beta bend or turn? a. lysine * b. Gly c. arg d. asn 2.
Introduction to Biochemistry Midterm exam 2016-2017 )ومن أحياها( 1. Which of the following amino (in a peptide chain) would probably be found at a beta bend or turn? a. lysine * b. Gly c. arg d. asn 2.
Structure of -amino acids. Stereoisomers of -amino acids. All amino acids in proteins are L-amino acids, except for glycine, which is achiral.
 amino acids Any of a large number of compounds found in living cells that contain carbon, oxygen, hydrogen, and nitrogen, and join together to form proteins. Amino acids contain a basic amino group (NH
amino acids Any of a large number of compounds found in living cells that contain carbon, oxygen, hydrogen, and nitrogen, and join together to form proteins. Amino acids contain a basic amino group (NH
Amino Acids. Review I: Protein Structure. Amino Acids: Structures. Amino Acids (contd.) Rajan Munshi
 Review I: Protein Structure Rajan Munshi BBSI @ Pitt 2005 Department of Computational Biology University of Pittsburgh School of Medicine May 24, 2005 Amino Acids Building blocks of proteins 20 amino acids
Review I: Protein Structure Rajan Munshi BBSI @ Pitt 2005 Department of Computational Biology University of Pittsburgh School of Medicine May 24, 2005 Amino Acids Building blocks of proteins 20 amino acids
I) Choose the best answer: 1- All of the following amino acids are neutral except: a) glycine. b) threonine. c) lysine. d) proline. e) leucine.
 1- All of the following amino acids are neutral except: a) glycine. b) threonine. c) lysine. d) proline. e) leucine. 2- The egg white protein, ovalbumin, is denatured in a hard-boiled egg. Which of the
1- All of the following amino acids are neutral except: a) glycine. b) threonine. c) lysine. d) proline. e) leucine. 2- The egg white protein, ovalbumin, is denatured in a hard-boiled egg. Which of the
Chapter 3: Amino Acids and Peptides
 Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
PROTEINS. Amino acids are the building blocks of proteins. Acid L-form * * Lecture 6 Macromolecules #2 O = N -C -C-O.
 Proteins: Linear polymers of amino acids workhorses of the cell tools, machines & scaffolds Lecture 6 Macromolecules #2 PRTEINS 1 Enzymes catalysts that mediate reactions, increase reaction rate Structural
Proteins: Linear polymers of amino acids workhorses of the cell tools, machines & scaffolds Lecture 6 Macromolecules #2 PRTEINS 1 Enzymes catalysts that mediate reactions, increase reaction rate Structural
Introduction to proteins and protein structure
 Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Q1: Circle the best correct answer: (15 marks)
 Q1: Circle the best correct answer: (15 marks) 1. Which one of the following incorrectly pairs an amino acid with a valid chemical characteristic a. Glycine, is chiral b. Tyrosine and tryptophan; at neutral
Q1: Circle the best correct answer: (15 marks) 1. Which one of the following incorrectly pairs an amino acid with a valid chemical characteristic a. Glycine, is chiral b. Tyrosine and tryptophan; at neutral
CHAPTER 21: Amino Acids, Proteins, & Enzymes. General, Organic, & Biological Chemistry Janice Gorzynski Smith
 CHAPTER 21: Amino Acids, Proteins, & Enzymes General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 21: Amino Acids, Proteins, Enzymes Learning Objectives: q The 20 common, naturally occurring
CHAPTER 21: Amino Acids, Proteins, & Enzymes General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 21: Amino Acids, Proteins, Enzymes Learning Objectives: q The 20 common, naturally occurring
Macromolecules Structure and Function
 Macromolecules Structure and Function Within cells, small organic molecules (monomers) are joined together to form larger molecules (polymers). Macromolecules are large molecules composed of thousands
Macromolecules Structure and Function Within cells, small organic molecules (monomers) are joined together to form larger molecules (polymers). Macromolecules are large molecules composed of thousands
Amino Acids. Lecture 4: Margaret A. Daugherty. Fall Swiss-prot database: How many proteins? From where?
 Lecture 4: Amino Acids Margaret A. Daugherty Fall 2004 Swiss-prot database: How many proteins? From where? 1986 Use http://us.expasy.org to get to swiss-prot database Proteins are the workhorses of the
Lecture 4: Amino Acids Margaret A. Daugherty Fall 2004 Swiss-prot database: How many proteins? From where? 1986 Use http://us.expasy.org to get to swiss-prot database Proteins are the workhorses of the
Lecture 3: 8/24. CHAPTER 3 Amino Acids
 Lecture 3: 8/24 CHAPTER 3 Amino Acids 1 Chapter 3 Outline 2 Amino Acid Are Biomolecules and their Atoms Can Be Visualized by Two Different Ways 1) Fischer projections: Two dimensional representation of
Lecture 3: 8/24 CHAPTER 3 Amino Acids 1 Chapter 3 Outline 2 Amino Acid Are Biomolecules and their Atoms Can Be Visualized by Two Different Ways 1) Fischer projections: Two dimensional representation of
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
Multiple-Choice Questions Answer ALL 20 multiple-choice questions on the Scantron Card in PENCIL
 Multiple-Choice Questions Answer ALL 20 multiple-choice questions on the Scantron Card in PENCIL For Questions 1-10 choose ONE INCORRECT answer. 1. Which ONE of the following statements concerning the
Multiple-Choice Questions Answer ALL 20 multiple-choice questions on the Scantron Card in PENCIL For Questions 1-10 choose ONE INCORRECT answer. 1. Which ONE of the following statements concerning the
Chapter 2 Biosynthesis of Enzymes
 Chapter 2 Biosynthesis of Enzymes 2.1 Basic Enzyme Chemistry 2.1.1 Amino Acids An amino acid is a molecule that has the following formula: The central carbon atom covalently bonded by amino, carboxyl,
Chapter 2 Biosynthesis of Enzymes 2.1 Basic Enzyme Chemistry 2.1.1 Amino Acids An amino acid is a molecule that has the following formula: The central carbon atom covalently bonded by amino, carboxyl,
Protein Structure and Function
 Protein Structure and Function Protein Structure Classification of Proteins Based on Components Simple proteins - Proteins containing only polypeptides Conjugated proteins - Proteins containing nonpolypeptide
Protein Structure and Function Protein Structure Classification of Proteins Based on Components Simple proteins - Proteins containing only polypeptides Conjugated proteins - Proteins containing nonpolypeptide
استاذ الكيمياءالحيوية
 قسم الكيمياء الحيوية د.دولت على سالمه استاذ الكيمياءالحيوية ٢٠١٥-٢٠١٤ الرمز الكودي : ٥١٢ المحاضرة األولى ١ Content : Definition of proteins Definition of amino acids Definition of peptide bond General
قسم الكيمياء الحيوية د.دولت على سالمه استاذ الكيمياءالحيوية ٢٠١٥-٢٠١٤ الرمز الكودي : ٥١٢ المحاضرة األولى ١ Content : Definition of proteins Definition of amino acids Definition of peptide bond General
Short polymer. Dehydration removes a water molecule, forming a new bond. Longer polymer (a) Dehydration reaction in the synthesis of a polymer
 HO 1 2 3 H HO H Short polymer Dehydration removes a water molecule, forming a new bond Unlinked monomer H 2 O HO 1 2 3 4 H Longer polymer (a) Dehydration reaction in the synthesis of a polymer HO 1 2 3
HO 1 2 3 H HO H Short polymer Dehydration removes a water molecule, forming a new bond Unlinked monomer H 2 O HO 1 2 3 4 H Longer polymer (a) Dehydration reaction in the synthesis of a polymer HO 1 2 3
The Structure and Func.on of Macromolecules Proteins GRU1L6
 The Structure and Func.on of Macromolecules Proteins GRU1L6 Proteins Proteins Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure
The Structure and Func.on of Macromolecules Proteins GRU1L6 Proteins Proteins Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure
3. AMINO ACID AND PEPTIDES
 3. AMINO ACID AND PEPTIDES 3.1 Amino Acids and Peptides General structure - Only 20 amino-acids are found in proteins - Amino group and carboxyl group - α-carbon and side chain group 3.1 Amino Acids and
3. AMINO ACID AND PEPTIDES 3.1 Amino Acids and Peptides General structure - Only 20 amino-acids are found in proteins - Amino group and carboxyl group - α-carbon and side chain group 3.1 Amino Acids and
Amino Acids and Proteins Hamad Ali Yaseen, PhD MLS Department, FAHS, HSC, KU Biochemistry 210 Chapter 22
 Amino Acids and Proteins Hamad Ali Yaseen, PhD MLS Department, FAHS, HSC, KU Hamad.ali@hsc.edu.kw Biochemistry 210 Chapter 22 Importance of Proteins Main catalysts in biochemistry: enzymes (involved in
Amino Acids and Proteins Hamad Ali Yaseen, PhD MLS Department, FAHS, HSC, KU Hamad.ali@hsc.edu.kw Biochemistry 210 Chapter 22 Importance of Proteins Main catalysts in biochemistry: enzymes (involved in
AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS
 AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine
AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine
BIOB111 - Tutorial activity for Session 14
 BIOB111 - Tutorial activity for Session 14 General topics for week 7 Session 14 Amino acids and proteins Students review the concepts learnt and answer the selected questions from the textbook. General
BIOB111 - Tutorial activity for Session 14 General topics for week 7 Session 14 Amino acids and proteins Students review the concepts learnt and answer the selected questions from the textbook. General
Biomolecules Amino Acids & Protein Chemistry
 Biochemistry Department Date: 17/9/ 2017 Biomolecules Amino Acids & Protein Chemistry Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology Intended Learning Outcomes ILOs By the end
Biochemistry Department Date: 17/9/ 2017 Biomolecules Amino Acids & Protein Chemistry Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology Intended Learning Outcomes ILOs By the end
Biochemistry - I. Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I
 Biochemistry - I Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I Hello, welcome to the course Biochemistry 1 conducted by me Dr. S Dasgupta,
Biochemistry - I Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I Hello, welcome to the course Biochemistry 1 conducted by me Dr. S Dasgupta,
Ionization of amino acids
 Amino Acids 20 common amino acids there are others found naturally but much less frequently Common structure for amino acid COOH, -NH 2, H and R functional groups all attached to the a carbon Ionization
Amino Acids 20 common amino acids there are others found naturally but much less frequently Common structure for amino acid COOH, -NH 2, H and R functional groups all attached to the a carbon Ionization
Lecture 11 AMINO ACIDS AND PROTEINS
 Lecture 11 AMINO ACIDS AND PROTEINS The word "Protein" was coined by J.J. Berzelius in 1838 and was derived from the Greek word "Proteios" meaning the first rank. Proteins are macromolecular polymers composed
Lecture 11 AMINO ACIDS AND PROTEINS The word "Protein" was coined by J.J. Berzelius in 1838 and was derived from the Greek word "Proteios" meaning the first rank. Proteins are macromolecular polymers composed
Bio Factsheet. Proteins and Proteomics. Number 340
 Number 340 Proteins and Proteomics Every living thing on the planet is composed of cells, and cells in turn are made of many types of molecules, including the biological molecules carbohydrates, lipids,
Number 340 Proteins and Proteomics Every living thing on the planet is composed of cells, and cells in turn are made of many types of molecules, including the biological molecules carbohydrates, lipids,
Amino Acids and Proteins (2) Professor Dr. Raid M. H. Al-Salih
 Amino Acids and Proteins (2) Professor Dr. Raid M. H. Al-Salih 1 Some important biologically active peptides 2 Proteins The word protein is derived from Greek word, proteios which means primary. As the
Amino Acids and Proteins (2) Professor Dr. Raid M. H. Al-Salih 1 Some important biologically active peptides 2 Proteins The word protein is derived from Greek word, proteios which means primary. As the
Proteins. Amino acids, structure and function. The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka
 Proteins Amino acids, structure and function The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka O O HO N N HN OH Ser65-Tyr66-Gly67 The Nobel prize in chemistry 2008 Osamu Shimomura,
Proteins Amino acids, structure and function The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka O O HO N N HN OH Ser65-Tyr66-Gly67 The Nobel prize in chemistry 2008 Osamu Shimomura,
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination # 5: Section Five May 7, Name: (print)
 Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
Introduction to Protein Structure Collection
 Introduction to Protein Structure Collection Teaching Points This collection is designed to introduce students to the concepts of protein structure and biochemistry. Different activities guide students
Introduction to Protein Structure Collection Teaching Points This collection is designed to introduce students to the concepts of protein structure and biochemistry. Different activities guide students
UNIT 2 Amino acids and Proteins
 UNIT 2 Amino acids and Proteins Significance of Proteins 1. Keep the cells and tissues growing, renewing and mending 2. Take part in some kinds of important physiological activities 3. Oxidation and supply
UNIT 2 Amino acids and Proteins Significance of Proteins 1. Keep the cells and tissues growing, renewing and mending 2. Take part in some kinds of important physiological activities 3. Oxidation and supply
OPTION GROUP: BIOLOGICAL MOLECULES 3 PROTEINS WORKBOOK. Tyrone R.L. John, Chartered Biologist
 NAME: OPTION GROUP: BIOLOGICAL MOLECULES 3 PROTEINS WORKBOOK Tyrone R.L. John, Chartered Biologist 1 Tyrone R.L. John, Chartered Biologist 2 Instructions REVISION CHECKLIST AND ASSESSMENT OBJECTIVES Regular
NAME: OPTION GROUP: BIOLOGICAL MOLECULES 3 PROTEINS WORKBOOK Tyrone R.L. John, Chartered Biologist 1 Tyrone R.L. John, Chartered Biologist 2 Instructions REVISION CHECKLIST AND ASSESSMENT OBJECTIVES Regular
Structure of proteins
 Structure of proteins Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Structure of proteins The 20 a.a commonly found
Structure of proteins Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Structure of proteins The 20 a.a commonly found
Biochemistry Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology Kharagpur. Lecture -02 Amino Acids II
 Biochemistry Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology Kharagpur Lecture -02 Amino Acids II Ok, we start off with the discussion on amino acids. (Refer Slide Time: 00:48)
Biochemistry Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology Kharagpur Lecture -02 Amino Acids II Ok, we start off with the discussion on amino acids. (Refer Slide Time: 00:48)
CHAPTER 29 HW: AMINO ACIDS + PROTEINS
 CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
Introduction to Proteomics Dr. Sanjeeva Srivastava Department of Biosciences and Bioengineering Indian Institute of Technology - Bombay
 Introduction to Proteomics Dr. Sanjeeva Srivastava Department of Biosciences and Bioengineering Indian Institute of Technology - Bombay Lecture 01 Introduction to Amino Acids Welcome to the proteomic course.
Introduction to Proteomics Dr. Sanjeeva Srivastava Department of Biosciences and Bioengineering Indian Institute of Technology - Bombay Lecture 01 Introduction to Amino Acids Welcome to the proteomic course.
Dr. Nafith Abu Tarboush
 11 Dr. Nafith Abu Tarboush July 3 rd 2013 Marah Dannoun Continuation of amino acids applications in life other than tryptophan and tyrosine: 1. Glutamate (glutamic acid): it s modified to give monosodium
11 Dr. Nafith Abu Tarboush July 3 rd 2013 Marah Dannoun Continuation of amino acids applications in life other than tryptophan and tyrosine: 1. Glutamate (glutamic acid): it s modified to give monosodium
2. Which of the following amino acids is most likely to be found on the outer surface of a properly folded protein?
 Name: WHITE Student Number: Answer the following questions on the computer scoring sheet. 1 mark each 1. Which of the following amino acids would have the highest relative mobility R f in normal thin layer
Name: WHITE Student Number: Answer the following questions on the computer scoring sheet. 1 mark each 1. Which of the following amino acids would have the highest relative mobility R f in normal thin layer
Practice Problems 3. a. What is the name of the bond formed between two amino acids? Are these bonds free to rotate?
 Life Sciences 1a Practice Problems 3 1. Draw the oligopeptide for Ala-Phe-Gly-Thr-Asp. You do not need to indicate the stereochemistry of the sidechains. Denote with arrows the bonds formed between the
Life Sciences 1a Practice Problems 3 1. Draw the oligopeptide for Ala-Phe-Gly-Thr-Asp. You do not need to indicate the stereochemistry of the sidechains. Denote with arrows the bonds formed between the
Amino acids. Dr. Mamoun Ahram Summer semester,
 Amino acids Dr. Mamoun Ahram Summer semester, 2017-2018 Resources This lecture Campbell and Farrell s Biochemistry, Chapters 3 (pp.66-76) General structure (Chiral carbon) The amino acids that occur in
Amino acids Dr. Mamoun Ahram Summer semester, 2017-2018 Resources This lecture Campbell and Farrell s Biochemistry, Chapters 3 (pp.66-76) General structure (Chiral carbon) The amino acids that occur in
Metabolic Classification of the Amino Acids
 Metabolic Classification of the Amino Acids *Essential and Non-essential * Glucogenic and Ketogenic 1 Essential Amino Acids Of the 20 amino acids that make up proteins 10 of them can be synthesized by
Metabolic Classification of the Amino Acids *Essential and Non-essential * Glucogenic and Ketogenic 1 Essential Amino Acids Of the 20 amino acids that make up proteins 10 of them can be synthesized by
Proteins: Structure and Function 2/8/2017 1
 Proteins: Structure and Function 2/8/2017 1 outline Protein functions hemistry of amino acids Protein Structure; Primary structure Secondary structure Tertiary structure Quaternary structure 2/8/2017 2
Proteins: Structure and Function 2/8/2017 1 outline Protein functions hemistry of amino acids Protein Structure; Primary structure Secondary structure Tertiary structure Quaternary structure 2/8/2017 2
Chapter 5: Structure and Function of Macromolecules AP Biology 2011
 Chapter 5: Structure and Function of Macromolecules AP Biology 2011 1 Macromolecules Fig. 5.1 Carbohydrates Lipids Proteins Nucleic Acids Polymer - large molecule consisting of many similar building blocks
Chapter 5: Structure and Function of Macromolecules AP Biology 2011 1 Macromolecules Fig. 5.1 Carbohydrates Lipids Proteins Nucleic Acids Polymer - large molecule consisting of many similar building blocks
COO - l. H 3 N C a H l R 1
 COO - l + H 3 N C a H l R 1 Amino acids There are 20 standard amino acids. All proteins are built from the same amino acids. The most important criteria for classification is affinity to water: hydrophilic
COO - l + H 3 N C a H l R 1 Amino acids There are 20 standard amino acids. All proteins are built from the same amino acids. The most important criteria for classification is affinity to water: hydrophilic
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Lipids: diverse group of hydrophobic molecules
 Lipids: diverse group of hydrophobic molecules Lipids only macromolecules that do not form polymers li3le or no affinity for water hydrophobic consist mostly of hydrocarbons nonpolar covalent bonds fats
Lipids: diverse group of hydrophobic molecules Lipids only macromolecules that do not form polymers li3le or no affinity for water hydrophobic consist mostly of hydrocarbons nonpolar covalent bonds fats
Amino acids & Protein Structure Chemwiki: Chapter , with most emphasis on 16.3, 16.4 and 16.6
 Amino acids & Protein Structure Chemwiki: Chapter 16. 16.1, 16.3-16.9 with most emphasis on 16.3, 16.4 and 16.6 1 1. Most jobs (except information storage) in cells are performed by proteins. 2. Proteins
Amino acids & Protein Structure Chemwiki: Chapter 16. 16.1, 16.3-16.9 with most emphasis on 16.3, 16.4 and 16.6 1 1. Most jobs (except information storage) in cells are performed by proteins. 2. Proteins
CHM333 LECTURE 6: 1/25/12 SPRING 2012 Professor Christine Hrycyna AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID:
 AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID: - The R group side chains on amino acids are VERY important. o Determine the properties of the amino acid itself o Determine
AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID: - The R group side chains on amino acids are VERY important. o Determine the properties of the amino acid itself o Determine
130327SCH4U_biochem April 09, 2013
 Option B: B1.1 ENERGY Human Biochemistry If more energy is taken in from food than is used up, weight gain will follow. Similarly if more energy is used than we supply our body with, weight loss will occur.
Option B: B1.1 ENERGY Human Biochemistry If more energy is taken in from food than is used up, weight gain will follow. Similarly if more energy is used than we supply our body with, weight loss will occur.
Date: EXERCISE 4. Figure 1. Amino acid structure.
 Student s name: Date: Points: Assistant s signature: Index numer: /6 EXERISE 4 AMIN AIDS AND PRTEINS. Amino acids are structural units (monomers) of proteins. There are 20 different amino acids coded for
Student s name: Date: Points: Assistant s signature: Index numer: /6 EXERISE 4 AMIN AIDS AND PRTEINS. Amino acids are structural units (monomers) of proteins. There are 20 different amino acids coded for
Peptides. The two amino acids are joined through a dehydration reaction.
 Peptides Peptides The two amino acids are joined through a dehydration reaction. Peptides The Peptide Bond The peptide bond is usually drawn as a single bond, but actually has considerable double bond
Peptides Peptides The two amino acids are joined through a dehydration reaction. Peptides The Peptide Bond The peptide bond is usually drawn as a single bond, but actually has considerable double bond
A Chemical Look at Proteins: Workhorses of the Cell
 A Chemical Look at Proteins: Workhorses of the Cell A A Life ciences 1a Lecture otes et 4 pring 2006 Prof. Daniel Kahne Life requires chemistry 2 amino acid monomer and it is proteins that make the chemistry
A Chemical Look at Proteins: Workhorses of the Cell A A Life ciences 1a Lecture otes et 4 pring 2006 Prof. Daniel Kahne Life requires chemistry 2 amino acid monomer and it is proteins that make the chemistry
(30 pts.) 16. (24 pts.) 17. (20 pts.) 18. (16 pts.) 19. (5 pts.) 20. (5 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
االمتحان النهائي لعام 1122
 االمتحان النهائي لعام 1122 Amino Acids : 1- which of the following amino acid is unlikely to be found in an alpha-helix due to its cyclic structure : -phenylalanine -tryptophan -proline -lysine 2- : assuming
االمتحان النهائي لعام 1122 Amino Acids : 1- which of the following amino acid is unlikely to be found in an alpha-helix due to its cyclic structure : -phenylalanine -tryptophan -proline -lysine 2- : assuming
If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out.
 Sign In Forgot Password Register username username password password Sign In If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki
Sign In Forgot Password Register username username password password Sign In If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki
(65 pts.) 27. (10 pts.) 28. (15 pts.) 29. (10 pts.) TOTAL (100 points) Moorpark College Chemistry 11 Spring Instructor: Professor Gopal
 Moorpark College Chemistry 11 Spring 2012 Instructor: Professor Gopal Examination # 5: Section Five May 1, 2012 Name: (print) GOOD LUCK! Directions: Make sure your examination contains TWELVE total pages
Moorpark College Chemistry 11 Spring 2012 Instructor: Professor Gopal Examination # 5: Section Five May 1, 2012 Name: (print) GOOD LUCK! Directions: Make sure your examination contains TWELVE total pages
SRTUCTURE OF PROTEINS DR. A. TARAB DEPT. OF BIOCHEMISTRY HKMU
 SRTUCTURE OF PROTEINS DR. A. TARAB DEPT. OF BIOCHEMISTRY HKMU I. OVERVIEW The twenty amino acids commonly found in proteins are joined together by peptide bonds The linear sequence of the linked amino
SRTUCTURE OF PROTEINS DR. A. TARAB DEPT. OF BIOCHEMISTRY HKMU I. OVERVIEW The twenty amino acids commonly found in proteins are joined together by peptide bonds The linear sequence of the linked amino
Proteins. Bởi: OpenStaxCollege
 Proteins Bởi: OpenStaxCollege Proteins are one of the most abundant organic molecules in living systems and have the most diverse range of functions of all macromolecules. Proteins may be structural, regulatory,
Proteins Bởi: OpenStaxCollege Proteins are one of the most abundant organic molecules in living systems and have the most diverse range of functions of all macromolecules. Proteins may be structural, regulatory,
بسم هللا الرحمن الرحيم
 بسم هللا الرحمن الرحيم Q1: the overall folding of a single protein subunit is called : -tertiary structure -primary structure -secondary structure -quaternary structure -all of the above Q2 : disulfide
بسم هللا الرحمن الرحيم Q1: the overall folding of a single protein subunit is called : -tertiary structure -primary structure -secondary structure -quaternary structure -all of the above Q2 : disulfide
PROTEIN. By: Shamsul Azahari Zainal Badari Department of Resource Management and Consumer Studies Faculty of Human Ecology UPM
 PROTEIN By: Shamsul Azahari Zainal Badari Department of Resource Management and Consumer Studies Faculty of Human Ecology UPM OBJECTIVES OF THE LECTURE By the end of this lecture, student can: Define
PROTEIN By: Shamsul Azahari Zainal Badari Department of Resource Management and Consumer Studies Faculty of Human Ecology UPM OBJECTIVES OF THE LECTURE By the end of this lecture, student can: Define
1. (38 pts.) 2. (25 pts.) 3. (15 pts.) 4. (12 pts.) 5. (10 pts.) Bonus (12 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
Midterm 1 Last, First
 Midterm 1 BIS 105 Prof. T. Murphy April 23, 2014 There should be 6 pages in this exam. Exam instructions (1) Please write your name on the top of every page of the exam (2) Show all work for full credit
Midterm 1 BIS 105 Prof. T. Murphy April 23, 2014 There should be 6 pages in this exam. Exam instructions (1) Please write your name on the top of every page of the exam (2) Show all work for full credit
AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy
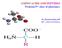 AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry MEDICAL AND BIOLOGICAL IMPORTANCE 1. building blocks of proteins. Some amino acids are found in
AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry MEDICAL AND BIOLOGICAL IMPORTANCE 1. building blocks of proteins. Some amino acids are found in
Bielkoviny, enzýmy. Július Cirák. Protein Structure Timothy G. Standish
 Bielkoviny, enzýmy Július irák Alanine Acid Different Amino Acid lasses 2 on-polar Aspartic acid 2 Amine Generic 2? R Acid Basic Polar istidine 2 S 2 + ysteine Levels f Protein rganization Primary Structure
Bielkoviny, enzýmy Július irák Alanine Acid Different Amino Acid lasses 2 on-polar Aspartic acid 2 Amine Generic 2? R Acid Basic Polar istidine 2 S 2 + ysteine Levels f Protein rganization Primary Structure
So where were we? But what does the order mean? OK, so what's a protein? 4/1/11
 So where were we? We know that DNA is responsible for heredity Chromosomes are long pieces of DNA DNA turned out to be the transforming principle We know that DNA is shaped like a long double helix, with
So where were we? We know that DNA is responsible for heredity Chromosomes are long pieces of DNA DNA turned out to be the transforming principle We know that DNA is shaped like a long double helix, with
Proteins. Dr. Basima Sadiq Jaff. /3 rd class of pharmacy. PhD. Clinical Biochemistry
 Proteins /3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry a Greek word that means of first importance. It is a very important class of food molecules that provide organisms not
Proteins /3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry a Greek word that means of first importance. It is a very important class of food molecules that provide organisms not
