N-Substituted amino acids as multifunctional additives used in cooling water. Part II: N-carboxymethyl and phosphonomethyl amino acids
|
|
|
- Gyles Fields
- 5 years ago
- Views:
Transcription
1 Int. J. Corros. Scale Inhib., 2016, 5, no. 2, N-Substituted amino acids as multifunctional additives used in cooling water. Part II: N-carboxymethyl and phosphonomethyl amino acids J. Telegdi 1,2 1 Óbuda University, Faculty of Light Industry and Environmental Engineering, Doberdó u. 6., 1034 Budapest, Hungary 2 Department of Interfaces and Surface Modification, Institute of Materials and Environmental Chemistry, Research Centre for Natural Sciences, Hungarian Academy of Sciences, Magyar tudósok körútja 2, 1117 Budapest, Hungary telegdi.judit@ttk.mta.hu Abstract The aim of research was to find additives with different capacities that can control the undesired corrosion and scale formation at the same time. Amino acids with different (carboxymethyl and/or phosphonomethyl) substituents were investigated to prove their multifunctional effectiveness. The most interesting parameters were the number of COOH and PO 3 H 2 groups as well as the structure of the side chain in the amino acids. The experimental data proved that both the alkyl side chains (with or without additional carboxy and phosphonic groups) as well as the number of the ionizable substituents in the molecules influence the both efficacies. Key words: inhibitor, corrosion, scale formation, N-carboxymethyl amino acid, N- phosphonomethyl amino acid. Received: April 19, Published: April 26, doi: / Introduction When both the corrosion and the scaling should be controlled in aqueous environment, it could happen via two procedures: a. Two chemicals are simultaneously added to the system that can control the corrosion and the scaling [1]. The problem one has to face is that the parallel addition of the two different chemicals has an unwanted effect, i.e. their efficiency could be decreased by each other. The most frequently applied compositions consist of phosphonic acids and polymers with anions, carboxy and sulfo groups [2]. b. When one molecule can control the metal corrosion and the scale deposition they are mainly polymers: polycarboxylic acids (naphthalene polycarboxylic acid, acrylic polycarboxylic acids [3], itaconic acid sodium allylsulfonate sodium hypophosphite copolymer [4], or poly(aspartic acid triptophane grafted polymer [5] as well as new phosphorous containing terpolymers [6]. Generally, the
2 Int. J. Corros. Scale Inhib., 2016, 5, no. 2, molecular weight of the polymers is between 200 and The efficiency of the inhibitors depends on the molecular weight, on the ph, ion concentration, temperature and on the presence of other additives [7, 8], which could be phosphonomethyl amine oxide [9], quaternary ammonium compounds [10] as well as aliphatic amino alcohols used together with diamino compounds [11]. The polymers that can efficiently control the scale formation are generally not biodegradable. This was the reason I have decided to work with small molecules with a natural basic part. In the case, when the chemicals from the treated industrial waters reach lakes and rivers, their biodegradation results in environmentally friendly molecules. The other deteriorating process is the corrosion of metals. There are inhibitor molecules in wide range, their application depends on the environment they are used (solid surface, aqueous or oily liquids, temperature, pressure, etc.). Some of the most effective chemicals are those, which contain phosphono group(s). In these molecules, there is a covalent bond between the phosphorous and carbon atoms and this bond is very stable. Some examples are the hydroxyethane-diphosphonic acid, nitrilotri-phosphonic acid, 1- phosphonobutane-tricarboxylic acid. Numerous papers describe their effectiveness [12 19]. When we have decided to solve the inhibition of the above mentioned double unwanted processes, first we have synthesized N-carboxymethyl and N-phosphonomethyl substituted natural amino acids (their syntheses are not part of this paper) and then with special scale and gravimetric corrosion tests the influence of the molecule structures (ionic groups, alkyl chains) and effect of the ph were elucidated. 2. Experimental 2.1 Materials Metal: Carbon steel was used in the corrosion experiments (composition: Fe 99.57%, C 0.05%, Si 0.05%, Mn 0.3%, S 0.01%, P 0.02%). Inhibitors used in these experiments were as follows: N-carboxymethyl derivatives of glycine, alanine, β-alanine, norvaline, valine, aspartic acid, glutamic acid, phenylalanine, and lysine. 2.2 Corrosion test The corrosion rates were determined by gravimetric technique and the efficiency (η %) calculated according to the equation: η m0 m i m 100[%], 0 where m 0 represents the weight loss of the coupon when inhibitor is absent, m i is the weight loss of the coupon measured in the presence of inhibitor.
3 Int. J. Corros. Scale Inhib., 2016, 5, no. 2, The corrosion tests were carried out in model solution ([0.47 g CaSO 4 2H 2 O g MgSO 4 H 2 O g NaHCO g CaCl 2 H 2 O] / 1 L distilled water, ph: 3.7 and 8.5). 2.3 Scale inhibition test 200 ml of the two model solutions (3.12 g NaHCO 3 / 1 L and 2.22 g CaCl 2 / 1 L in distilled water) were mixed, and after adding an aliquot of the N-substituted amino acid solutions (0.1%) the solution was heated to 60 C. Samples taken after 2, 4, and 24 hours were filtered and the calcium content of the clear filtrate was determined by titration with ethylenediamine tetraacetic acid. The scale inhibiting activity was calculated by comparison of the calcium content in the original solution and in the samples with inhibitor, removed from the test solution. 3. Results and discussion 3.1 Results of experiments measured in the presence of N-carboxymethyl amino acids As the Figure 1 demonstrates, this group of amino acid derivatives show antiscaling as well as anticorrosion activity, which depends on the side chain of amino acids. For the sake of comparison, the inhibitive activities of iminodiacetic acid and nitrilotriacetic acids (which have similar structures but are not derivatives of natural amino acids) are also involved in the figure. The efficiency data prove that most of the compounds studied can decrease the rate of corrosion and the scale deposition. Analyzing the structure-activity relationship, a branched side chain in the amino acid spoils the effect (see the case of valine norvalin and β-alanine alanine). The adverse effect could be explained by sterical hindrance. Longer alkyl chain is not favourale for scale inhibition, which is reflected in the antiscaling activity of alanine and norvaline derivatives. Data on anticorrosion effectiveness demonstrates the importance of the N-carboxymethyl aspartic acid. This activity is in the range of the efficacy of the nitrilotriacetic acid, which is an industrial scale deposition inhibitor. In the series of investigated N-carboxymethyl amino acids, the substituted aspartic acid and glutamic acid are apt for industrial application. 3.2 Results of experiments measured in the presence of of N-phosphonomethyl amino acids In the case of this group of compounds, not only the anticorrosion activity but the phdependence of these data, too, was in the focus. As Figure 2 presents, the anticorrosive efficiency of these amino acid derivatives depends to a high degree upon the acidity of the test solution.
4 Int. J. Corros. Scale Inhib., 2016, 5, no. 2, Figure 1. Carboxymethyl amino acids investigated in scale and corrosion tests; IDE: iminodiacetic acid, NTE: nitrilotriacetic acid; KM: carboxymethyl; BAla: β-alanine; Ala: alanine; Asp: aspartic acid; Glu: glutamic acid; NVal: norvalin; Val: valine; Phe: phenylalanine. Figure 2. ph-dependent anticorrosion activities of N-phosphonomethyl amino acids; PM: phospohonomethyl; DPM: diphosphonomethyl; Gly: glycine; Asp: aspartic acid; Glu: glutamic acid; Phe: phenylalanine; Lys: lysine.
5 Int. J. Corros. Scale Inhib., 2016, 5, no. 2, Numerical data are summarized in Table 1. Table 1. The ph-dependent anticorrosion activities of N-phosphonomethyl amino acids. anticorr. activity anticorr. activity anticorr. activity N-phospohonomethyl amino acids η [%] η [%] η [%] ph 3 ph 7 ph 8.5 PMGly: H 2 O 3 P CH 2 NH CH 2 COOH DPMGly: (H 2 O 3 P CH 2 ) 2 NH CH 2 COOH PMPhe: H 2 O 3 P CH 2 NH CH(CH 2 C 6 H 5 ) COOH PMAsp: H 2 O 3 P CH 2 NH CH(CH 2 COOH) COOH PMGlu: H 2 O 3 P CH 2 NH CH(CH 2 CH 2 COOH) COOH DPMLys: H 2 O 3 P CH 2 NH CH((CH 2 ) 4 NH CH 2 H 2 O 3 P) COOH The presence of more ionizable groups in the amino acid side chain increases significantly the anticorrosion efficiency. When an inhibitive layer is formed on the metal surface, the carboxy and phosphono groups face the oxide layer covered metal surface and form a physical bond. When the number of these groups increases, the coverage of the surface with inhibitor molecules increases too; the aggressive components of the environment/solution cannot come near to the metal surface. The consequence is that the rate of corrosion decreases. The ph-dependence of the efficacy unequivocally proved that these molecules are not proper inhibitors for example in etching liquids as all of the investigated molecules do not decease but increase the metal dissolution under acidic condition. It shows that these molecules in protonated form cannot inhibit properly the metal corrosion. On the other hand, when the environment is neutral or mildly alkali, these additives decrease the rate of corrosion significantly and they are good inhibitors. When the molecular structure is under investigation, data demonstrate that the efficacy decreases when a phenyl ring is present in the side chain of the amino acid, as the case of the phenylalanine derivative shows. The most effective additives are the N,N-disubstituted amino acid, the activity does not depend on the length of the alkyl chain in the amino acid. Not only the anticorrosion property of these molecules was followed, but their inhibition of scale deposition was also measured. In order to compare these activities, Figure 3 summarizes their antiscaling and anticorrosion effectiveness. The same molecule i.e. N,N-disubstituted glycine was the most effective inhibitor of scale deposition as well as of corrosion.
6 Int. J. Corros. Scale Inhib., 2016, 5, no. 2, Figure 3. N-phosphonomethyl amino acids as inhibitors of scale formation and corrosion; PM: phospohonomethyl; DPM: diphosphonomethyl; Gly: glycine; Asp: aspartic acid; Glu: glutamic acid; Phe: phenylalanine. 4. Conclusion Summarizing results got in the presence on N-carboxymethyl and N-phosphonomethyl amino acids it unravels that the increase in number of phosphono groups in the substituted amino acids calls forth significant increase in effectiveness of both activities. The impact of the alkyl side chain in the amino acid is less important than the presence of additional carboxylic groups when already phosphono group is present in a molecule; this is explainable by the chelate formation at the metal/metal oxide surface via carboxylic and phosphono groups. This enlightens the increase in the efficiency on increase in the number of functional groups. The ph-dependent corrosion rates reveal on the importance of the molecular state. Comparing the negative effect of the acidic medium on the corrosion rate with those data got at neutral or slightly alkali ph, it is clear that in the adsorption processes at the metal/metal oxide interface mainly those molecules take part where the carboxylic and phosphonic groups are not protonated. From this group of compounds the N,N-di(phosphonomethyl)glycine can most effectively control both the corrosion and the scale deposition. The systematic survey confirms unequivocally that the equivalent number of the carboxylic groups does not show the same increase in effectiveness than that of the phosphono analogs.
7 Int. J. Corros. Scale Inhib., 2016, 5, no. 2, References 1. A. Al-Borno, M. Islam and R. Khraishi, Corrosion, 1989, 42, F. Moran, 1986, PCT Int. Appl. WO A1. 3. Ch. Carber, G. Kreh, R.P. Fan and L. Duinen, 1992, US A1. 4. Zh. Liu, H. Fu and L. Zhang, Desalination, 2016, 57, X. Sun, J. Zhang, Ch. Yin, J. Zhang and J. Han, J. Appl. Polym. Sci., 2015, 132, doi: /app Y. Chen, Y. Zhou, Q. Yao, Y. Bu and H. Wang, Desalin. Water Treat., 2015, 55, B. Clubley and G. Riddent, 1993, J. EP A1. 8. J.L. Fang, Y. Li and X.R. Ye, Corrosion (Houston, TX, U.S.), 1993, 49, Ch.M. Hwa, J.A. Kelly, J. Neton, P.M. Scanlon and R.R. Gaudette, 1994, EP B A.J. Swift, Microchim. Acta, 1995, 120, M. Duprat and F. Dabosi, Corrosion (Houston, TX, U.S.), 1981, 37, J. Telegdi, E. Kálmán and F.H. Kármán, Corros. Sci., 1992, 33, J. Telegdi, ACH Models Chem., 1995, 132, Yu.I. Kuznetsov, Progress in the Understanding and Prevention of Corrosion, in The Phosphonate inhibitors of Metal Corrosion, Wiley-VCH, 1995, pp E. Kálmán, I. Felhősi, F.H. Kármán, I. Lukovits, J. Telegdi and G. Pálinkás, Materials Science and Technology; A Comprehensive Treatment, Corrosion and Environmental Degradation, Eds. R.W. Chan, P. Haasen and E.J. Kramer, 2000, Vol. 1, pp , Wiley-VCH. 16. D.-J. Choi, S.-J. You and J.-G. Kim, Mater. Sci. Eng., A, 2002, 335, H. Amar, J. Benzakour, A. Derja, D. Villemin, B. Moreau and T. Braisaz, Appl. Surf. Sci., 2006, 252, M. Prabakaran, M. Venkatesh, S. Ramesh and V. Periasamy, Appl. Surf. Sci., 2013, 276, S. S. Rao, B.V. Appa Rao, S. Roopas Kiran and B. Sreedhar, J. Mater. Sci. Technol., 2014, 30, 77.
N-Substituted unusual amino acids as corrosion inhibitors. Part IV: N-Acyl derivatives of unnatural amino acids with double bond
 Int. J. Corros. Scale Inhib., 2016, 5, no. 4, 360 366 N-Substituted unusual amino acids as corrosion inhibitors. Part IV: N-Acyl derivatives of unnatural amino acids with double bond J. Telegdi 1,2 1 Óbuda
Int. J. Corros. Scale Inhib., 2016, 5, no. 4, 360 366 N-Substituted unusual amino acids as corrosion inhibitors. Part IV: N-Acyl derivatives of unnatural amino acids with double bond J. Telegdi 1,2 1 Óbuda
Synergistic Inhibition of Corrosion of Carbon Steel by the Ternary Formulations containing Phosphonate, Zn (II) and Ascorbic Acid
 Abstract Research Journal of Recent Sciences ISSN 2277-2502. Synergistic Inhibition of Corrosion of Carbon Steel by the Ternary Formulations containing Phosphonate, Zn (II) and Ascorbic Acid Appa Rao B.
Abstract Research Journal of Recent Sciences ISSN 2277-2502. Synergistic Inhibition of Corrosion of Carbon Steel by the Ternary Formulations containing Phosphonate, Zn (II) and Ascorbic Acid Appa Rao B.
Chemical Nature of the Amino Acids. Table of a-amino Acids Found in Proteins
 Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
Nitrilotris(methylenephosphonic acid) Zinc Ions Uric Acid: A New Ternary Formulation as Corrosion Inhibitor for Carbon Steel
 Nitrilotris(methylenephosphonic acid) Zinc Ions Uric Acid: A New Ternary Formulation as Corrosion Inhibitor for Carbon Steel D. Sarada Kalyani 1,*, S. Srinivasa Rao 1 and M. Sarath Babu 2 1 Department
Nitrilotris(methylenephosphonic acid) Zinc Ions Uric Acid: A New Ternary Formulation as Corrosion Inhibitor for Carbon Steel D. Sarada Kalyani 1,*, S. Srinivasa Rao 1 and M. Sarath Babu 2 1 Department
Reactions and amino acids structure & properties
 Lecture 2: Reactions and amino acids structure & properties Dr. Sameh Sarray Hlaoui Common Functional Groups Common Biochemical Reactions AH + B A + BH Oxidation-Reduction A-H + B-OH + energy ª A-B + H
Lecture 2: Reactions and amino acids structure & properties Dr. Sameh Sarray Hlaoui Common Functional Groups Common Biochemical Reactions AH + B A + BH Oxidation-Reduction A-H + B-OH + energy ª A-B + H
Biomolecules: amino acids
 Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Molecular Biology. general transfer: occurs normally in cells. special transfer: occurs only in the laboratory in specific conditions.
 Chapter 9: Proteins Molecular Biology replication general transfer: occurs normally in cells transcription special transfer: occurs only in the laboratory in specific conditions translation unknown transfer:
Chapter 9: Proteins Molecular Biology replication general transfer: occurs normally in cells transcription special transfer: occurs only in the laboratory in specific conditions translation unknown transfer:
Page 8/6: The cell. Where to start: Proteins (control a cell) (start/end products)
 Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids
 Amino acids 1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids 5-To understand amino acids synthesis Amino
Amino acids 1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids 5-To understand amino acids synthesis Amino
Chapter 3: Amino Acids and Peptides
 Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
Model 2: Aldohexoses, aldopentoses, ketohexoses and ketopentoses
 Model 1: D and L Sugars CHEM1405 Worksheet 13: Sugars and Amino Acids The simplest sugar is glyceraldehyde. This is a chiral molecule and the two enantiomers are shown opposite with wedges and dashes and
Model 1: D and L Sugars CHEM1405 Worksheet 13: Sugars and Amino Acids The simplest sugar is glyceraldehyde. This is a chiral molecule and the two enantiomers are shown opposite with wedges and dashes and
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Biochemistry - I. Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I
 Biochemistry - I Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I Hello, welcome to the course Biochemistry 1 conducted by me Dr. S Dasgupta,
Biochemistry - I Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I Hello, welcome to the course Biochemistry 1 conducted by me Dr. S Dasgupta,
CS612 - Algorithms in Bioinformatics
 Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Chemistry 121 Winter 17
 Chemistry 121 Winter 17 Introduction to Organic Chemistry and Biochemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941;
Chemistry 121 Winter 17 Introduction to Organic Chemistry and Biochemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941;
Biomolecules Amino Acids & Protein Chemistry
 Biochemistry Department Date: 17/9/ 2017 Biomolecules Amino Acids & Protein Chemistry Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology Intended Learning Outcomes ILOs By the end
Biochemistry Department Date: 17/9/ 2017 Biomolecules Amino Acids & Protein Chemistry Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology Intended Learning Outcomes ILOs By the end
BIO 311C Spring Lecture 15 Friday 26 Feb. 1
 BIO 311C Spring 2010 Lecture 15 Friday 26 Feb. 1 Illustration of a Polypeptide amino acids peptide bonds Review Polypeptide (chain) See textbook, Fig 5.21, p. 82 for a more clear illustration Folding and
BIO 311C Spring 2010 Lecture 15 Friday 26 Feb. 1 Illustration of a Polypeptide amino acids peptide bonds Review Polypeptide (chain) See textbook, Fig 5.21, p. 82 for a more clear illustration Folding and
Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version]
![Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version] Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version]](/thumbs/84/90818314.jpg) Earth/matriX: SCIENCE TODAY Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version] By Charles William Johnson Earth/matriX Editions P.O.
Earth/matriX: SCIENCE TODAY Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version] By Charles William Johnson Earth/matriX Editions P.O.
Proteins are sometimes only produced in one cell type or cell compartment (brain has 15,000 expressed proteins, gut has 2,000).
 Lecture 2: Principles of Protein Structure: Amino Acids Why study proteins? Proteins underpin every aspect of biological activity and therefore are targets for drug design and medicinal therapy, and in
Lecture 2: Principles of Protein Structure: Amino Acids Why study proteins? Proteins underpin every aspect of biological activity and therefore are targets for drug design and medicinal therapy, and in
Macromolecules of Life -3 Amino Acids & Proteins
 Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
9/6/2011. Amino Acids. C α. Nonpolar, aliphatic R groups
 Amino Acids Side chains (R groups) vary in: size shape charge hydrogen-bonding capacity hydrophobic character chemical reactivity C α Nonpolar, aliphatic R groups Glycine (Gly, G) Alanine (Ala, A) Valine
Amino Acids Side chains (R groups) vary in: size shape charge hydrogen-bonding capacity hydrophobic character chemical reactivity C α Nonpolar, aliphatic R groups Glycine (Gly, G) Alanine (Ala, A) Valine
Proteins consist in whole or large part of amino acids. Simple proteins consist only of amino acids.
 Today we begin our discussion of the structure and properties of proteins. Proteins consist in whole or large part of amino acids. Simple proteins consist only of amino acids. Conjugated proteins contain
Today we begin our discussion of the structure and properties of proteins. Proteins consist in whole or large part of amino acids. Simple proteins consist only of amino acids. Conjugated proteins contain
Introduction to Protein Structure Collection
 Introduction to Protein Structure Collection Teaching Points This collection is designed to introduce students to the concepts of protein structure and biochemistry. Different activities guide students
Introduction to Protein Structure Collection Teaching Points This collection is designed to introduce students to the concepts of protein structure and biochemistry. Different activities guide students
Lecture 3: 8/24. CHAPTER 3 Amino Acids
 Lecture 3: 8/24 CHAPTER 3 Amino Acids 1 Chapter 3 Outline 2 Amino Acid Are Biomolecules and their Atoms Can Be Visualized by Two Different Ways 1) Fischer projections: Two dimensional representation of
Lecture 3: 8/24 CHAPTER 3 Amino Acids 1 Chapter 3 Outline 2 Amino Acid Are Biomolecules and their Atoms Can Be Visualized by Two Different Ways 1) Fischer projections: Two dimensional representation of
Amino acids-incorporated nanoflowers with an
 Amino acids-incorporated nanoflowers with an intrinsic peroxidase-like activity Zhuo-Fu Wu 1,2,+, Zhi Wang 1,+, Ye Zhang 3, Ya-Li Ma 3, Cheng-Yan He 4, Heng Li 1, Lei Chen 1, Qi-Sheng Huo 3, Lei Wang 1,*
Amino acids-incorporated nanoflowers with an intrinsic peroxidase-like activity Zhuo-Fu Wu 1,2,+, Zhi Wang 1,+, Ye Zhang 3, Ya-Li Ma 3, Cheng-Yan He 4, Heng Li 1, Lei Chen 1, Qi-Sheng Huo 3, Lei Wang 1,*
CHAPTER 21: Amino Acids, Proteins, & Enzymes. General, Organic, & Biological Chemistry Janice Gorzynski Smith
 CHAPTER 21: Amino Acids, Proteins, & Enzymes General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 21: Amino Acids, Proteins, Enzymes Learning Objectives: q The 20 common, naturally occurring
CHAPTER 21: Amino Acids, Proteins, & Enzymes General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 21: Amino Acids, Proteins, Enzymes Learning Objectives: q The 20 common, naturally occurring
Amino acids. (Foundation Block) Dr. Essa Sabi
 Amino acids (Foundation Block) Dr. Essa Sabi Learning outcomes What are the amino acids? General structure. Classification of amino acids. Optical properties. Amino acid configuration. Non-standard amino
Amino acids (Foundation Block) Dr. Essa Sabi Learning outcomes What are the amino acids? General structure. Classification of amino acids. Optical properties. Amino acid configuration. Non-standard amino
Mutual Influence of HEDP and SDS - Zn 2+ System on Corrosion Inhibition of Carbon Steel
 Transactions of the SAEST 40 (2005) 35-39 Transactions of the SAEST Mutual Influence of and SDS - Zn 2+ System on Corrosion Inhibition of Carbon Steel Susai Rajendran 1*, A. John Amalraj 1, J. Wilson Sahayaraj
Transactions of the SAEST 40 (2005) 35-39 Transactions of the SAEST Mutual Influence of and SDS - Zn 2+ System on Corrosion Inhibition of Carbon Steel Susai Rajendran 1*, A. John Amalraj 1, J. Wilson Sahayaraj
Amino Acids. Amino Acids. Fundamentals. While their name implies that amino acids are compounds that contain an NH. 3 and CO NH 3
 Fundamentals While their name implies that amino acids are compounds that contain an 2 group and a 2 group, these groups are actually present as 3 and 2 respectively. They are classified as α, β, γ, etc..
Fundamentals While their name implies that amino acids are compounds that contain an 2 group and a 2 group, these groups are actually present as 3 and 2 respectively. They are classified as α, β, γ, etc..
CHM333 LECTURE 6: 1/25/12 SPRING 2012 Professor Christine Hrycyna AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID:
 AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID: - The R group side chains on amino acids are VERY important. o Determine the properties of the amino acid itself o Determine
AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID: - The R group side chains on amino acids are VERY important. o Determine the properties of the amino acid itself o Determine
Protiens and Amino Acids 2
 Protiens and Amino Acids 2 By Alaa J. Mahrath, Biochemistry Department / College of Medicine/ Babylon University 2 Amino Acid Stereoisomers Protiens and Amino Acids All the a-amino acids except for glycine
Protiens and Amino Acids 2 By Alaa J. Mahrath, Biochemistry Department / College of Medicine/ Babylon University 2 Amino Acid Stereoisomers Protiens and Amino Acids All the a-amino acids except for glycine
Amino Acids. Lecture 4: Margaret A. Daugherty. Fall Swiss-prot database: How many proteins? From where?
 Lecture 4: Amino Acids Margaret A. Daugherty Fall 2004 Swiss-prot database: How many proteins? From where? 1986 Use http://us.expasy.org to get to swiss-prot database Proteins are the workhorses of the
Lecture 4: Amino Acids Margaret A. Daugherty Fall 2004 Swiss-prot database: How many proteins? From where? 1986 Use http://us.expasy.org to get to swiss-prot database Proteins are the workhorses of the
LAB#23: Biochemical Evidence of Evolution Name: Period Date :
 LAB#23: Biochemical Evidence of Name: Period Date : Laboratory Experience #23 Bridge Worth 80 Lab Minutes If two organisms have similar portions of DNA (genes), these organisms will probably make similar
LAB#23: Biochemical Evidence of Name: Period Date : Laboratory Experience #23 Bridge Worth 80 Lab Minutes If two organisms have similar portions of DNA (genes), these organisms will probably make similar
Practice Problems 3. a. What is the name of the bond formed between two amino acids? Are these bonds free to rotate?
 Life Sciences 1a Practice Problems 3 1. Draw the oligopeptide for Ala-Phe-Gly-Thr-Asp. You do not need to indicate the stereochemistry of the sidechains. Denote with arrows the bonds formed between the
Life Sciences 1a Practice Problems 3 1. Draw the oligopeptide for Ala-Phe-Gly-Thr-Asp. You do not need to indicate the stereochemistry of the sidechains. Denote with arrows the bonds formed between the
I) Choose the best answer: 1- All of the following amino acids are neutral except: a) glycine. b) threonine. c) lysine. d) proline. e) leucine.
 1- All of the following amino acids are neutral except: a) glycine. b) threonine. c) lysine. d) proline. e) leucine. 2- The egg white protein, ovalbumin, is denatured in a hard-boiled egg. Which of the
1- All of the following amino acids are neutral except: a) glycine. b) threonine. c) lysine. d) proline. e) leucine. 2- The egg white protein, ovalbumin, is denatured in a hard-boiled egg. Which of the
AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy
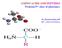 AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry MEDICAL AND BIOLOGICAL IMPORTANCE 1. building blocks of proteins. Some amino acids are found in
AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry MEDICAL AND BIOLOGICAL IMPORTANCE 1. building blocks of proteins. Some amino acids are found in
Ionization of amino acids
 Amino Acids 20 common amino acids there are others found naturally but much less frequently Common structure for amino acid COOH, -NH 2, H and R functional groups all attached to the a carbon Ionization
Amino Acids 20 common amino acids there are others found naturally but much less frequently Common structure for amino acid COOH, -NH 2, H and R functional groups all attached to the a carbon Ionization
The Structure and Function of Macromolecules
 The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
Chapter 2 Biosynthesis of Enzymes
 Chapter 2 Biosynthesis of Enzymes 2.1 Basic Enzyme Chemistry 2.1.1 Amino Acids An amino acid is a molecule that has the following formula: The central carbon atom covalently bonded by amino, carboxyl,
Chapter 2 Biosynthesis of Enzymes 2.1 Basic Enzyme Chemistry 2.1.1 Amino Acids An amino acid is a molecule that has the following formula: The central carbon atom covalently bonded by amino, carboxyl,
استاذ الكيمياءالحيوية
 قسم الكيمياء الحيوية د.دولت على سالمه استاذ الكيمياءالحيوية ٢٠١٥-٢٠١٤ الرمز الكودي : ٥١٢ المحاضرة األولى ١ Content : Definition of proteins Definition of amino acids Definition of peptide bond General
قسم الكيمياء الحيوية د.دولت على سالمه استاذ الكيمياءالحيوية ٢٠١٥-٢٠١٤ الرمز الكودي : ٥١٢ المحاضرة األولى ١ Content : Definition of proteins Definition of amino acids Definition of peptide bond General
Midterm 1 Last, First
 Midterm 1 BIS 105 Prof. T. Murphy April 23, 2014 There should be 6 pages in this exam. Exam instructions (1) Please write your name on the top of every page of the exam (2) Show all work for full credit
Midterm 1 BIS 105 Prof. T. Murphy April 23, 2014 There should be 6 pages in this exam. Exam instructions (1) Please write your name on the top of every page of the exam (2) Show all work for full credit
Amino Acids. Review I: Protein Structure. Amino Acids: Structures. Amino Acids (contd.) Rajan Munshi
 Review I: Protein Structure Rajan Munshi BBSI @ Pitt 2005 Department of Computational Biology University of Pittsburgh School of Medicine May 24, 2005 Amino Acids Building blocks of proteins 20 amino acids
Review I: Protein Structure Rajan Munshi BBSI @ Pitt 2005 Department of Computational Biology University of Pittsburgh School of Medicine May 24, 2005 Amino Acids Building blocks of proteins 20 amino acids
OPTIMIZING INHIBITOR BLENDS USING COMPUTER MODELING. Robert J. Ferguson French Creek Software Kimberton, PA ABSTRACT
 OPTIMIZING INHIBITOR BLENDS USING COMPUTER MODELING Robert J. Ferguson French Creek Software Kimberton, PA 19442-0684 ABSTRACT Multifunctional cooling water treatment programs address scale and corrosion
OPTIMIZING INHIBITOR BLENDS USING COMPUTER MODELING Robert J. Ferguson French Creek Software Kimberton, PA 19442-0684 ABSTRACT Multifunctional cooling water treatment programs address scale and corrosion
Chapter 20 and GHW#10 Questions. Proteins
 Chapter 20 and GHW#10 Questions Proteins Proteins Naturally occurring bioorganic polyamide polymers containing a sequence of various combinations of 20 amino acids. Amino acids contain the elements carbon,
Chapter 20 and GHW#10 Questions Proteins Proteins Naturally occurring bioorganic polyamide polymers containing a sequence of various combinations of 20 amino acids. Amino acids contain the elements carbon,
AA s are the building blocks of proteins
 Chamras Chemistry 106 Lecture otes Chapter 24: Amino Acids, Peptides, and Proteins General Formula: () n (') α-amino Acids: (n = 1) Example: Amino Acids and Proteins: Glycine Alanine Valine AA s are the
Chamras Chemistry 106 Lecture otes Chapter 24: Amino Acids, Peptides, and Proteins General Formula: () n (') α-amino Acids: (n = 1) Example: Amino Acids and Proteins: Glycine Alanine Valine AA s are the
Maha AbuAjamieh. Tamara Wahbeh. Mamoon Ahram
 12 Maha AbuAjamieh Tamara Wahbeh Mamoon Ahram - - Go to this sheet s last page for definitions of the words with an asterisk above them (*) - You should memorise the 3-letter abbreviations, of all the
12 Maha AbuAjamieh Tamara Wahbeh Mamoon Ahram - - Go to this sheet s last page for definitions of the words with an asterisk above them (*) - You should memorise the 3-letter abbreviations, of all the
INTRODUCTION TO BIOCHEMISTRY/POLYMERS. 3. With respect to amino acids, polypeptides, and proteins, know:
 INTRDUCTIN T BICEMISTRY/PLYMERS A STUDENT SULD BE ABLE T: 1. With respect to lipids, know: The characteristic common to members of the class (solubility in nonpolar solvents) The functional groups most
INTRDUCTIN T BICEMISTRY/PLYMERS A STUDENT SULD BE ABLE T: 1. With respect to lipids, know: The characteristic common to members of the class (solubility in nonpolar solvents) The functional groups most
Org/Biochem Final Lec Form, Spring 2012 Page 1 of 6
 Page 1 of 6 Missing Complete Protein and Question #45 Key Terms: Fill in the blank in the following 25 statements with one of the key terms in the table. Each key term may only be used once. Print legibly.
Page 1 of 6 Missing Complete Protein and Question #45 Key Terms: Fill in the blank in the following 25 statements with one of the key terms in the table. Each key term may only be used once. Print legibly.
Organic Chemistry - Problem Drill 23: Amino Acids, Peptides, and Proteins
 rganic Chemistry - Problem Drill 23: Amino Acids, Peptides, and Proteins No. 1 of 10 1. Which amino acid does not contain a chiral center? (A) Serine (B) Proline (C) Alanine (D) Phenylalanine (E) Glycine
rganic Chemistry - Problem Drill 23: Amino Acids, Peptides, and Proteins No. 1 of 10 1. Which amino acid does not contain a chiral center? (A) Serine (B) Proline (C) Alanine (D) Phenylalanine (E) Glycine
Copyright 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings
 Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
PHAR3316 Pharmacy biochemistry Exam #2 Fall 2010 KEY
 1. How many protons is(are) lost when the amino acid Asparagine is titrated from its fully protonated state to a fully deprotonated state? A. 0 B. 1 * C. 2 D. 3 E. none Correct Answer: C (this question
1. How many protons is(are) lost when the amino acid Asparagine is titrated from its fully protonated state to a fully deprotonated state? A. 0 B. 1 * C. 2 D. 3 E. none Correct Answer: C (this question
PROTEINS. Building blocks, structure and function. Aim: You will have a clear picture of protein construction and their general properties
 PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
Chapter 18. Carboxylic Acids and Their Derivatives. Nucleophilic Addition-Elimination at the Acyl Carbon
 Chapter 18 Carboxylic Acids and Their Derivatives. Nucleophilic Addition-Elimination at the Acyl Carbon Carboxylic Acids Organic compounds characterized by their acidity Contains COOH group (must be at
Chapter 18 Carboxylic Acids and Their Derivatives. Nucleophilic Addition-Elimination at the Acyl Carbon Carboxylic Acids Organic compounds characterized by their acidity Contains COOH group (must be at
AP Bio. Protiens Chapter 5 1
 Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
QUALITATIVE ANALYSIS OF AMINO ACIDS AND PROTEINS
 QUALITATIVE ANALYSIS OF AMINO ACIDS AND PROTEINS Amino acids are molecules containing an amine group, a carboxylic acid group and a side chain that varies between different amino acids. Amino acids of
QUALITATIVE ANALYSIS OF AMINO ACIDS AND PROTEINS Amino acids are molecules containing an amine group, a carboxylic acid group and a side chain that varies between different amino acids. Amino acids of
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination # 5: Section Five May 7, Name: (print)
 Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
Objective: You will be able to explain how the subcomponents of
 Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
CHAPTER 3 Amino Acids, Peptides, Proteins
 CHAPTER 3 Amino Acids, Peptides, Proteins Learning goals: Structure and naming of amino acids Structure and properties of peptides Ionization behavior of amino acids and peptides Methods to characterize
CHAPTER 3 Amino Acids, Peptides, Proteins Learning goals: Structure and naming of amino acids Structure and properties of peptides Ionization behavior of amino acids and peptides Methods to characterize
AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS
 AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine
AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine
Use of a pilot scale heat exchanger-cooling tower system for the evaluation of mineral scale inhibitors
 Abstract Int. J. Corros. Scale Inhib., 2013, 2, no. 4, 255 268 Use of a pilot scale heat exchanger-cooling tower system for the evaluation of mineral scale inhibitors E. Antonogiannakis, E. Tzagkaraki
Abstract Int. J. Corros. Scale Inhib., 2013, 2, no. 4, 255 268 Use of a pilot scale heat exchanger-cooling tower system for the evaluation of mineral scale inhibitors E. Antonogiannakis, E. Tzagkaraki
number Done by Corrected by Doctor Dr.Diala
 number 32 Done by Mousa Salah Corrected by Bahaa Najjar Doctor Dr.Diala 1 P a g e In the last lecture we talked about the common processes between all amino acids which are: transamination, deamination,
number 32 Done by Mousa Salah Corrected by Bahaa Najjar Doctor Dr.Diala 1 P a g e In the last lecture we talked about the common processes between all amino acids which are: transamination, deamination,
Lipids: diverse group of hydrophobic molecules
 Lipids: diverse group of hydrophobic molecules Lipids only macromolecules that do not form polymers li3le or no affinity for water hydrophobic consist mostly of hydrocarbons nonpolar covalent bonds fats
Lipids: diverse group of hydrophobic molecules Lipids only macromolecules that do not form polymers li3le or no affinity for water hydrophobic consist mostly of hydrocarbons nonpolar covalent bonds fats
Protein Structure Klemens Wild, BZH,
 Protein Structure recommended books Proteins protein definition From gr. proteios (superior, erstrangig) 1836 JJ Berzelius Functions: structural, enzymes, muscle, transport immune system, Linear polymer
Protein Structure recommended books Proteins protein definition From gr. proteios (superior, erstrangig) 1836 JJ Berzelius Functions: structural, enzymes, muscle, transport immune system, Linear polymer
PROTEINS. Amino acids are the building blocks of proteins. Acid L-form * * Lecture 6 Macromolecules #2 O = N -C -C-O.
 Proteins: Linear polymers of amino acids workhorses of the cell tools, machines & scaffolds Lecture 6 Macromolecules #2 PRTEINS 1 Enzymes catalysts that mediate reactions, increase reaction rate Structural
Proteins: Linear polymers of amino acids workhorses of the cell tools, machines & scaffolds Lecture 6 Macromolecules #2 PRTEINS 1 Enzymes catalysts that mediate reactions, increase reaction rate Structural
Metabolic Classification of the Amino Acids
 Metabolic Classification of the Amino Acids *Essential and Non-essential * Glucogenic and Ketogenic 1 Essential Amino Acids Of the 20 amino acids that make up proteins 10 of them can be synthesized by
Metabolic Classification of the Amino Acids *Essential and Non-essential * Glucogenic and Ketogenic 1 Essential Amino Acids Of the 20 amino acids that make up proteins 10 of them can be synthesized by
The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5
 Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
Lecture 11 AMINO ACIDS AND PROTEINS
 Lecture 11 AMINO ACIDS AND PROTEINS The word "Protein" was coined by J.J. Berzelius in 1838 and was derived from the Greek word "Proteios" meaning the first rank. Proteins are macromolecular polymers composed
Lecture 11 AMINO ACIDS AND PROTEINS The word "Protein" was coined by J.J. Berzelius in 1838 and was derived from the Greek word "Proteios" meaning the first rank. Proteins are macromolecular polymers composed
COO - l. H 3 N C a H l R 1
 COO - l + H 3 N C a H l R 1 Amino acids There are 20 standard amino acids. All proteins are built from the same amino acids. The most important criteria for classification is affinity to water: hydrophilic
COO - l + H 3 N C a H l R 1 Amino acids There are 20 standard amino acids. All proteins are built from the same amino acids. The most important criteria for classification is affinity to water: hydrophilic
A Chemical Look at Proteins: Workhorses of the Cell
 A Chemical Look at Proteins: Workhorses of the Cell A A Life ciences 1a Lecture otes et 4 pring 2006 Prof. Daniel Kahne Life requires chemistry 2 amino acid monomer and it is proteins that make the chemistry
A Chemical Look at Proteins: Workhorses of the Cell A A Life ciences 1a Lecture otes et 4 pring 2006 Prof. Daniel Kahne Life requires chemistry 2 amino acid monomer and it is proteins that make the chemistry
Introduction to proteins and protein structure
 Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
ADSORPTION AND DESORPTION OF METAL IONS BY SYSTEMS BASED ON CELLULOSE DERIVATIVES THAT CONTAIN AMINO ACID RESIDUES"
 (41) Vol. 41, No.6 (1985) T-235 (Received May 24, 1984) ADSORPTION AND DESORPTION OF METAL IONS BY SYSTEMS BASED ON CELLULOSE DERIVATIVES THAT CONTAIN AMINO ACID RESIDUES" By Toshihiko Sato, Shigenori
(41) Vol. 41, No.6 (1985) T-235 (Received May 24, 1984) ADSORPTION AND DESORPTION OF METAL IONS BY SYSTEMS BASED ON CELLULOSE DERIVATIVES THAT CONTAIN AMINO ACID RESIDUES" By Toshihiko Sato, Shigenori
Human Biochemistry Option B
 Human Biochemistry Option B A look ahead... Your body has many functions to perform every day: Structural support, genetic information, communication, energy supply, metabolism Right now, thousands of
Human Biochemistry Option B A look ahead... Your body has many functions to perform every day: Structural support, genetic information, communication, energy supply, metabolism Right now, thousands of
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A
 Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
(65 pts.) 27. (10 pts.) 28. (15 pts.) 29. (10 pts.) TOTAL (100 points) Moorpark College Chemistry 11 Spring Instructor: Professor Gopal
 Moorpark College Chemistry 11 Spring 2012 Instructor: Professor Gopal Examination # 5: Section Five May 1, 2012 Name: (print) GOOD LUCK! Directions: Make sure your examination contains TWELVE total pages
Moorpark College Chemistry 11 Spring 2012 Instructor: Professor Gopal Examination # 5: Section Five May 1, 2012 Name: (print) GOOD LUCK! Directions: Make sure your examination contains TWELVE total pages
The Basics: A general review of molecular biology:
 The Basics: A general review of molecular biology: DNA Transcription RNA Translation Proteins DNA (deoxy-ribonucleic acid) is the genetic material It is an informational super polymer -think of it as the
The Basics: A general review of molecular biology: DNA Transcription RNA Translation Proteins DNA (deoxy-ribonucleic acid) is the genetic material It is an informational super polymer -think of it as the
AMINO ACIDS. Qualitative Tests
 AMINO ACIDS Qualitative Tests AMINO ACIDS Amino acid play A central role as building block of proteins. Amino acids also converted to specialized products. More than 300 different amino acids have been
AMINO ACIDS Qualitative Tests AMINO ACIDS Amino acid play A central role as building block of proteins. Amino acids also converted to specialized products. More than 300 different amino acids have been
If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out.
 Sign In Forgot Password Register username username password password Sign In If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki
Sign In Forgot Password Register username username password password Sign In If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki
(30 pts.) 16. (24 pts.) 17. (20 pts.) 18. (16 pts.) 19. (5 pts.) 20. (5 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
! Proteins are involved functionally in almost everything: " Receptor Proteins - Respond to external stimuli. " Storage Proteins - Storing amino acids
 Proteins Most structurally & functionally diverse group! Proteins are involved functionally in almost everything: Proteins Multi-purpose molecules 2007-2008 Enzymatic proteins - Speed up chemical reactions!
Proteins Most structurally & functionally diverse group! Proteins are involved functionally in almost everything: Proteins Multi-purpose molecules 2007-2008 Enzymatic proteins - Speed up chemical reactions!
Organic Molecules: Proteins
 Organic Molecules: Proteins Proteins Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport
Organic Molecules: Proteins Proteins Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2.
 BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
Structure of -amino acids. Stereoisomers of -amino acids. All amino acids in proteins are L-amino acids, except for glycine, which is achiral.
 amino acids Any of a large number of compounds found in living cells that contain carbon, oxygen, hydrogen, and nitrogen, and join together to form proteins. Amino acids contain a basic amino group (NH
amino acids Any of a large number of compounds found in living cells that contain carbon, oxygen, hydrogen, and nitrogen, and join together to form proteins. Amino acids contain a basic amino group (NH
Amino Acids االحماض األمينية
 Amino Acids االحماض األمينية Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Cell Structure Amino Acids The study of
Amino Acids االحماض األمينية Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Cell Structure Amino Acids The study of
Bio Factsheet. Proteins and Proteomics. Number 340
 Number 340 Proteins and Proteomics Every living thing on the planet is composed of cells, and cells in turn are made of many types of molecules, including the biological molecules carbohydrates, lipids,
Number 340 Proteins and Proteomics Every living thing on the planet is composed of cells, and cells in turn are made of many types of molecules, including the biological molecules carbohydrates, lipids,
Can wetting and dispersing additives improve the durability of coatings?
 Can wetting and dispersing additives improve the durability of coatings? János Hajas BYK-Chemie, Wesel, Germany Topics: WETTING AND DISPERSING ADDITIVE, DURABILITY, CORROSION RESISTANCE 1. Abstracts There
Can wetting and dispersing additives improve the durability of coatings? János Hajas BYK-Chemie, Wesel, Germany Topics: WETTING AND DISPERSING ADDITIVE, DURABILITY, CORROSION RESISTANCE 1. Abstracts There
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination #5: Section Five December 7, Name: (print) Section:
 Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal Examination #5: Section Five December 7, 2011 Name: (print) Section: alkene < alkyne < amine < alcohol < ketone < aldehyde < amide
Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal Examination #5: Section Five December 7, 2011 Name: (print) Section: alkene < alkyne < amine < alcohol < ketone < aldehyde < amide
Amino Acids and Proteins Hamad Ali Yaseen, PhD MLS Department, FAHS, HSC, KU Biochemistry 210 Chapter 22
 Amino Acids and Proteins Hamad Ali Yaseen, PhD MLS Department, FAHS, HSC, KU Hamad.ali@hsc.edu.kw Biochemistry 210 Chapter 22 Importance of Proteins Main catalysts in biochemistry: enzymes (involved in
Amino Acids and Proteins Hamad Ali Yaseen, PhD MLS Department, FAHS, HSC, KU Hamad.ali@hsc.edu.kw Biochemistry 210 Chapter 22 Importance of Proteins Main catalysts in biochemistry: enzymes (involved in
2. Which of the following is NOT true about carbohydrates
 Chemistry 11 Fall 2011 Examination #5 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response questions
Chemistry 11 Fall 2011 Examination #5 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response questions
Arginine side chain interactions and the role of arginine as a mobile charge carrier in voltage sensitive ion channels. Supplementary Information
 Arginine side chain interactions and the role of arginine as a mobile charge carrier in voltage sensitive ion channels Craig T. Armstrong, Philip E. Mason, J. L. Ross Anderson and Christopher E. Dempsey
Arginine side chain interactions and the role of arginine as a mobile charge carrier in voltage sensitive ion channels Craig T. Armstrong, Philip E. Mason, J. L. Ross Anderson and Christopher E. Dempsey
Methionine (Met or M)
 Fig. 5-17 Nonpolar Fig. 5-17a Nonpolar Glycine (Gly or G) Alanine (Ala or A) Valine (Val or V) Leucine (Leu or L) Isoleucine (Ile or I) Methionine (Met or M) Phenylalanine (Phe or F) Polar Trypotphan (Trp
Fig. 5-17 Nonpolar Fig. 5-17a Nonpolar Glycine (Gly or G) Alanine (Ala or A) Valine (Val or V) Leucine (Leu or L) Isoleucine (Ile or I) Methionine (Met or M) Phenylalanine (Phe or F) Polar Trypotphan (Trp
1. (38 pts.) 2. (25 pts.) 3. (15 pts.) 4. (12 pts.) 5. (10 pts.) Bonus (12 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
Raghad Abu Jebbeh. Amani Nofal. Mamoon Ahram
 ... 14 Raghad Abu Jebbeh Amani Nofal Mamoon Ahram This sheet includes part of lec.13 + lec.14. Amino acid peptide protein Terminology: 1- Residue: a subunit that is a part of a large molecule. 2- Dipeptide:
... 14 Raghad Abu Jebbeh Amani Nofal Mamoon Ahram This sheet includes part of lec.13 + lec.14. Amino acid peptide protein Terminology: 1- Residue: a subunit that is a part of a large molecule. 2- Dipeptide:
Gentilucci, Amino Acids, Peptides, and Proteins. Peptides and proteins are polymers of amino acids linked together by amide bonds CH 3
 Amino Acids Peptides and proteins are polymers of amino acids linked together by amide bonds Aliphatic Side-Chain Amino Acids - - H CH glycine alanine 3 proline valine CH CH 3 - leucine - isoleucine CH
Amino Acids Peptides and proteins are polymers of amino acids linked together by amide bonds Aliphatic Side-Chain Amino Acids - - H CH glycine alanine 3 proline valine CH CH 3 - leucine - isoleucine CH
Amino acids. You are required to know and identify the 20 amino acids : their names, 3 letter abbreviations and their structures.
 Amino acids You are required to know and identify the 20 amino acids : their names, 3 letter abbreviations and their structures. If you wanna make any classification in the world, you have to find what
Amino acids You are required to know and identify the 20 amino acids : their names, 3 letter abbreviations and their structures. If you wanna make any classification in the world, you have to find what
Amino acids. Dr. Mamoun Ahram Summer semester,
 Amino acids Dr. Mamoun Ahram Summer semester, 2017-2018 Resources This lecture Campbell and Farrell s Biochemistry, Chapters 3 (pp.66-76) General structure (Chiral carbon) The amino acids that occur in
Amino acids Dr. Mamoun Ahram Summer semester, 2017-2018 Resources This lecture Campbell and Farrell s Biochemistry, Chapters 3 (pp.66-76) General structure (Chiral carbon) The amino acids that occur in
Carboxylic Acids. The Importance of Carboxylic Acids (RCO 2 H)
 Carboxylic Acids The Importance of Carboxylic Acids (RCO 2 H) Starting materials for acyl derivatives (esters, amides, and acid chlorides) Abundant in nature from oxidation of aldehydes and alcohols in
Carboxylic Acids The Importance of Carboxylic Acids (RCO 2 H) Starting materials for acyl derivatives (esters, amides, and acid chlorides) Abundant in nature from oxidation of aldehydes and alcohols in
Prelab 6: Carboxylic Acids
 The Structure of Carboxylic Acids Prelab 6: Carboxylic Acids Carboxylic acids contain a carboxyl functional group attached to a hydrocarbon (alkyl group) part. Carboxyl groups contain both a carbonyl group,
The Structure of Carboxylic Acids Prelab 6: Carboxylic Acids Carboxylic acids contain a carboxyl functional group attached to a hydrocarbon (alkyl group) part. Carboxyl groups contain both a carbonyl group,
