ADVERSE EVENT REPORTING AN FDA REQUIREMENT WHAT, WHEN, WHO & HOW
|
|
|
- Baldwin Mason
- 6 years ago
- Views:
Transcription
1 ADVERSE EVENT REPORTING AN FDA REQUIREMENT WHAT, WHEN, WHO & HOW AMERICAN ACADEMY OF ORTHOPAEDIC SURGEONS 75TH ANNUAL MEETING MARCH 5-9, 2008 SAN FRANCISCO, CALIFORNIA COMMITTEE ON BIOMEDICAL ENGINEERING PREPARED BY: JOHN S. KIRKPATRICK, M.D. A. SETH GREENWALD, D.PHIL.(OXON) MICHAEL E. BEREND, M.D. GREGORY A. BROWN, M.D., PH.D. DENNIS MCGOWAN, M.D. WILLIAM MIHALKO, M.D., PH.D.
2 Most orthopaedic surgeons in the course of their practice will encounter and explant medical devices which have failed. Such failures may simply result from long-term in-vivo device usage, component material breakdown, technique at the time of implantation or patient noncompliance. Changes in the Safe Medical Devices Act of 1990 (SMDA), a Law enacted by the Congress, requires that medical device user facilities inclusive of hospitals, ambulatory surgical facilities and nursing homes report such events to the Food and Drug Administration (FDA) and manufacturers. These events are inclusive of device problems which cause or contribute to the death, serious illness or serious injury of a patient. The orthopaedic surgeon is involved in the identification and interpretation of these problems. In June 1993, the FDA introduced MedWatch, a medical products reporting program, to facilitate the reporting of adverse events and product problems that arise from medical device usage. What is an adverse event? An event whereby a death or serious injury was, or may have been caused by a medical device. Serious injury can be interpreted to mean requiring medical or surgical management to preclude impairment or damage to the body s function or structure with removal, observation, medical treatment, or hospitalization. This includes events resulting from: - Device malfunction - Improper / inadequate design - Labeling (problems) - Device failure - Manufacturing (problems) - Use error When should an adverse event be reported by an orthopaedic surgeon? Adverse events should be reported once the individual patient s safety is ensured. While hospitals and manufacturers have mandatory reporting, surgeons have no such requirement. Unfortunately, those best able to provide clinical context are removed from the process. Surgeon reporting of any and all device failures and adverse events will bring relevance to the report and improve the safety decisions made by the FDA. Who should report events? Surgeons provide the most relevant and useful information and provide insight into the cause and effect of the event. Perhaps the most important role is to differentiate whether the device failed or the disease process or patient biological issues caused the problem. Surgeons may participate in their facilities mandatory annual reporting process. Manufacturers are required to file adverse events. Surgeons may also file voluntary reports individually. How should an adverse event be reported? The surgeon can go to This web page has the instructions and the link to beginning the forms. Part 5 is the most important for surgeons to provide an interpretation of the event. Examples will include answers to Part 5 for the cases in this exhibit. What about HIPPAA? The [HIPAA] Privacy Rule permits covered entities to disclose protected health information, without authorization, to public health authorities who are legally authorized to receive such reports for the purpose of presenting or controlling disease, injury, or disability. This would include, for example, the reporting of a disease or injury; reporting vital events, such as births or deaths; and conducting health surveillance, investigations, or interventions. A public health authority is an agency or authority of the United States government, a State, a territory, a political subdivision of a State or territory, or Indian tribe that is responsible for public health matters as part of its official mandate, as well as a person or entity acting under a grant of authority from, or under a contract with, a public health agency. Examples of a public health authority include a State and local health departments, the Food and Drug Administration (FDA), the Centers for Disease Control and Prevention, and the Occupational Safety and Health Administration (OSHA). The following examples illustrate these points.
3 CASE 1 CASE 2 A 50-year-old obese female with multiple sclerosis had multiple failed thoracolumbar spinal fusions for low back pain. One of the fusion attempts had been complicated by a MRSA infection. She developed junctional kyphosis and neurological deficit superior to her construct and had evidence of pseudarthrosis also. She had decompression and re-fusion with replacement of her prior hardware and extension superiorly. Approximately 18 months later she presented with pain and crepitus in her low back and radiographs showed fracture of her rods bilaterally. She requested removal of her hardware and attempted fusion of her persistent pseudarthrosis. Was this a failure of the hardware? Technically yes, but this must be qualified by the failure of the patient s biology to heal her fusion while the hardware was in place. Patient had fusion with posterior fixation for failed prior fusion with a MRSA infection. The rods were noted to be broken and the patient had pain and crepitus from the broken rods rubbing together. Patient had removal and re-fusion of the non-union 18 months following implantation. In light of the patient s clinical setting this was interpreted as a failure of the patient s biology, not a failure of the device. This patient had a sudden onset of pain and difficulty ambulating 21 months after bilateral primary total knee arthroplasty surgery. He had no signs of infection and only had difficulty with his right knee. Radiographs revealed a catastrophic fatigue failure of the modular tibial baseplate and possible failure of the polyethylene insert as well. It is difficult to tell if excessive polyethylene wear or fatigue of the metal baseplate were the initial events in the implant failure. The patient underwent revision total knee surgery where failure of both the baseplate and polyethylene inserts were confirmed. Patient had catastrophic failure of the tibial base plate and polyethylene insert at less than two years after implantation. Unfortunately, pre-failure radiographs were not available. The potential contributing factors for this failure mechanism include: varus component positioning, excessive patient weight (BMI>40), and possible soft tissue imbalance adding stress to the medial aspect of the implant. There may also have been inadequate fixation of the medial aspect of the baseplate. While these factors may have contributed to the failure mechanism, the failure at such a short time period after implantation must also be a result of material or design failure as well.
4 CASE 3 CASE 4 A 68-year-old white female s/p placement of a Dorsal Column Stimulating generator in left buttock for intractable bilateral neuropathic leg pain refractory to all other treatments. The wound initially healed without complication--never any suggestion of an unstable or infected wound. After the wound was healed, the patient was taught the procedure for recharging the system. The patient was very satisfied with the system and had good control of the bilateral neuropathic leg pain. The patient continued to do well until approximately five months s/p system placement when she developed pain about the generator site when recharging. She went to the emergency room and was found to have a burn over the generator. The pocket was aspirated and no evidence for a deep infection. She was covered with antibiotics and the burn was followed. The burn broke down, the generator was exposed as shown in the picture, and required removal of the system. The wound closed with dressing changes. Risk factors included a history of alcoholism and poor nutrition. Patient with implantable stimulator with chronic wound, healed initially without incident. other risk factors or evidence for infection, cultures with cutaneuos organisms. Material of connector at wound was different than the rest of the device. It is unclear whether wound breakdown was from the primary infection, a reaction to the material, or the placement of the device in a relatively prominent area near the iliac crest. This patient had sudden onset pain and difficulty ambulating 15 years after a right primary total hip arthroplasty procedure. He reported no symptoms prior to the event where he described getting out of bed and feeling a pop and having difficulty bearing weight with a grinding sensation. A radiograph 6 months earlier confirmed some asymmetric wear but the new radiograph showed dissociation of the polyethylene liner from the metallic shell. Breakage of the locking mechanism was also evident. The patient underwent a revision with cementation of a new liner into the well fixed shell where confirmation of failure of the locking mechanism was determined. The liner had some asymmetric wear but was not catastrophic. Patient had a successful total hip arthroplasty for many years. For this amount of time wear of polyethylene was noted due to the eccentric position of the femoral head in the acetabular shell. On retrieval both wear of the polyethylene and failure of the locking mechanism were noted. Since the implant had been in place for 15 years, some wear was expected and this was not out of the ordinary for the time period. The failure of the locking mechanism was the problem that led to the need for revision surgery.
5 CASE 5 CASE 6 Patient had a subtrochanteric femur fracture treated with a cephalomedullary nail. Patient was advised to partially weight bear, but progressed to weight bearing independently as their fracture pain improved. At 8 weeks postoperatively, patient was ambulating well, had minimal discomfort, and the radiograph revealed a broken distal screw, which was felt to be an incidental finding. Patient was treated for an unstable subtrochanteric femur fracture with an intramedullary hip screw and long nail. The patient was weight bearing as tolerated in immediate postoperative rehabilitation. The distal interlocking screw was noted to be broken at 8 weeks postoperatively. The fracture healed and the screw was asymptomatic. Fatigue failure of the screw may have allowed for dynamization and helped fracture healing. A 43-year-old female s/p patellofemoral arthroplasty with pain, mechanical symptoms, and patellar instability. The mobile bearing patellar component was noted to be dislocated medially as seen on the A/P and sunrise radiographs with the two metallic markers in the polyethylene displaced into the medial gutter. This was treated with revision to a non-modular fixed bearing cemented total knee arthroplasty with standard CR components. Patient with patellofemoral arthroplasty with mobile polyethylene patellar component. Polyethylene displaced from component with no trauma. Appears to be a failure of intended component interactions and motion limits.
6 PLEASE TYPE OR USE BLACK INK U.S. Department of Health and Human Services MEDWATCH The FDA Safety Information and Adverse Event Reporting Program A. PATIENT INFORMATION 1. Patient Identifier 2. AgeatTimeofEvent,or 3. Sex 4. Weight Date of Birth: Female Male or In confidence B. ADVERSE EVENT, PRODUCT PROBLEM OR ERROR Check all that apply: 1. Adverse Event Product Use Error 2. Outcomes Attributed to Adverse Event (Check all that apply) Death: (mm/dd/yyyy) Life-threatening Problem with Different Manufacturer of Same Medicine 3. Date of Event (mm/dd/yyyy) 4. Date of this Report (mm/dd/yyyy) 5. Describe Event, Problem or Product Use Error Product Problem (e.g., defects/malfunctions) Disability or Permanent Damage Congenital Anomaly/Birth Defect Hospitalization - initial or prolonged Other Serious (Important Medical Events) Required Intervention to Prevent Permanent Impairment/Damage (Devices) For VOLUNTARY reporting of adverse events, product problems and product use errors Page of lb kg D. SUSPECT PRODUCT(S) 1. Name, Strength, Manufacturer (from product label) Form Approved: OMB , Expires: 10/31/08 See OMB statement on reverse. FDA USE ONLY Triage unit sequence # 2. Dose or Amount Frequency Route 3. Dates of Use (If unknown, give duration) from/to (or best estimate) 4. Diagnosis or Reason for Use (Indication) 6. Lot # E. SUSPECT MEDICAL DEVICE 1. Brand Name 2. Common Device Name 3. Manufacturer Name, City and State 4. Model # Catalog # Serial # 7. Expiration Date Lot # 6. If Implanted, Give Date (mm/dd/yyyy) Expiration Date (mm/dd/yyyy) Other # 5. Event Abated After Use Stopped or Dose Reduced? 8. Event Reappeared After Reintroduction? 9. NDC # or Unique ID 5. Operator of Device Other: 7. If Explanted, Give Date (mm/dd/yyyy) 8. Is this a Single-use Device that was Reprocessed and Reused on a Patient? 9. If to Item. 8, Enter Name and Address of Reprocessor Health Professional Lay User/Patient 6. Relevant Tests/Laboratory Data, Including Dates F. OTHER (CONCOMITANT) MEDICAL PRODUCTS Product names and therapy dates (exclude treatment of event) 7. Other Relevant History, Including Preexisting Medical Conditions (e.g., allergies, race, pregnancy, smoking and alcohol use, liver/kidney problems, etc.) G. REPORTER (See confidentiality section on back) 1. Name and Address Phone # C. PRODUCT AVAILABILITY Product Available for Evaluation? (Do not send product to FDA) FORM FDA 3500 (10/05) Returned to Manufacturer on: (mm/dd/yyyy) 2. Health Professional? 3. Occupation 4. Also Reported to: Manufacturer 5. If you do NOT want your identity disclosed User Facility to the manufacturer, place an "X" in this box: Distributor/Importer Submission of a report does not constitute an admission that medical personnel or the product caused or contributed to the event.
FDA Safety Communication: Metal-on- Metal Hip Implants
 FDA Safety Communication: Metal-on- Metal Hip Implants Date Issued: Jan. 17, 2013 Audience: Orthopaedic surgeons Health care providers responsible for the ongoing care of patients with metal-onmetal hip
FDA Safety Communication: Metal-on- Metal Hip Implants Date Issued: Jan. 17, 2013 Audience: Orthopaedic surgeons Health care providers responsible for the ongoing care of patients with metal-onmetal hip
CASE REPORT. Bone transport utilizing the PRECICE Intramedullary Nail for an infected nonunion in the distal femur
 PRODUCTS CASE REPORT Bone transport utilizing the PRECICE Intramedullary Nail for an infected nonunion in the distal femur Robert D. Fitch, M.D. Duke University Health System 1 1 CONDITION Infected nonunion
PRODUCTS CASE REPORT Bone transport utilizing the PRECICE Intramedullary Nail for an infected nonunion in the distal femur Robert D. Fitch, M.D. Duke University Health System 1 1 CONDITION Infected nonunion
CLINICAL AND OPERATIVE APPROACH FOR TOTAL KNEE REPLACEMENT DR.VINMAIE ORTHOPAEDICS PG 2 ND YEAR
 CLINICAL AND OPERATIVE APPROACH FOR TOTAL KNEE REPLACEMENT DR.VINMAIE ORTHOPAEDICS PG 2 ND YEAR Evolution of TKR In 1860, Verneuil proposed interposition arthroplasty, involving the insertion of soft tissue
CLINICAL AND OPERATIVE APPROACH FOR TOTAL KNEE REPLACEMENT DR.VINMAIE ORTHOPAEDICS PG 2 ND YEAR Evolution of TKR In 1860, Verneuil proposed interposition arthroplasty, involving the insertion of soft tissue
ESC. Enhanced Stability Liners. Design Rationale & Surgical Technique
 ESC Enhanced Stability Liners Design Rationale & Surgical Technique Choice Without Compromise DePuy Synthes PINNACLE Hip Solutions are designed with a wide range of acetabular cup options, biological and
ESC Enhanced Stability Liners Design Rationale & Surgical Technique Choice Without Compromise DePuy Synthes PINNACLE Hip Solutions are designed with a wide range of acetabular cup options, biological and
2016 SIGN Conference: Treatment of Difficult Fractures Around The World
 2016 SIGN Conference: Treatment of Difficult Fractures Around The World September 21-24, 2016 Long Bone Osteosynthesis Failure DUONG BUNN Trauma-Orthopedique ward Preah Kossamak Preah Sihaknouk Ville Beach
2016 SIGN Conference: Treatment of Difficult Fractures Around The World September 21-24, 2016 Long Bone Osteosynthesis Failure DUONG BUNN Trauma-Orthopedique ward Preah Kossamak Preah Sihaknouk Ville Beach
Aesculap CeSpace TM PEEK and CeSpace TM XP Spinal Implant System Instructions for Use
 Aesculap CeSpace TM PEEK and CeSpace TM XP Spinal Implant System Instructions for Use Page 1 of 5 Indications for use: When used as a Vertebral Body Replacement Device: The CeSpace PEEK and CeSpace XP
Aesculap CeSpace TM PEEK and CeSpace TM XP Spinal Implant System Instructions for Use Page 1 of 5 Indications for use: When used as a Vertebral Body Replacement Device: The CeSpace PEEK and CeSpace XP
Osteoarthrosis, unspecified whether generalized or localized, lower leg. Osteoarthrosis, localized, not specified whether primary or secondary, pelvic
 Page 1 Appendix TABLE E-1 Codes (and Definitions) in Humana Database Used for Study Inclusion and Exclusion of Patients Who Underwent,, or 1 to 2-Level Inclusion ICD-9-P-8154 Total knee replacement ICD-9-D-71596
Page 1 Appendix TABLE E-1 Codes (and Definitions) in Humana Database Used for Study Inclusion and Exclusion of Patients Who Underwent,, or 1 to 2-Level Inclusion ICD-9-P-8154 Total knee replacement ICD-9-D-71596
CASE REPORT. Antegrade tibia lengthening with the PRECICE Limb Lengthening technology
 CASE REPORT Antegrade tibia lengthening with the PRECICE Limb Lengthening technology Austin T. Fragomen, M.D. Hospital for Special Surgery New York, NY 1 1 PR O D U CTS CONDITION Nonunion of an attempted
CASE REPORT Antegrade tibia lengthening with the PRECICE Limb Lengthening technology Austin T. Fragomen, M.D. Hospital for Special Surgery New York, NY 1 1 PR O D U CTS CONDITION Nonunion of an attempted
Duraloc CONSTRAINED LINER
 SURGICAL TECHNIQUE Duraloc CONSTRAINED LINER A COMPREHENSIVE ACETABULAR REVISION SYSTEM DURALOC CONSTRAINED LINER Introduction Dislocation is the most common postoperative complication in total hip reconstruction.
SURGICAL TECHNIQUE Duraloc CONSTRAINED LINER A COMPREHENSIVE ACETABULAR REVISION SYSTEM DURALOC CONSTRAINED LINER Introduction Dislocation is the most common postoperative complication in total hip reconstruction.
Precautionary Statement ( )
 Precautionary Statement (21282008) Biomet Sports Medicine, Inc. 21282008 4861 E. Airport Dr. Rev. A Ontario, CA 91761 Date: 06/07 Sleeve and Button Soft Tissue Devices Utilizing ZipLoop Technology ATTENTION
Precautionary Statement (21282008) Biomet Sports Medicine, Inc. 21282008 4861 E. Airport Dr. Rev. A Ontario, CA 91761 Date: 06/07 Sleeve and Button Soft Tissue Devices Utilizing ZipLoop Technology ATTENTION
American Joint Replacement Registry. Jeffrey P. Knezovich, CAE Executive Director ---- Caryn D. Etkin, PhD, MPH Director of Analytics
 American Joint Replacement Registry Jeffrey P. Knezovich, CAE Executive Director ---- Caryn D. Etkin, PhD, MPH Director of Analytics AJRR Mission and Vision AJRR is a multi stakeholder, independent, not
American Joint Replacement Registry Jeffrey P. Knezovich, CAE Executive Director ---- Caryn D. Etkin, PhD, MPH Director of Analytics AJRR Mission and Vision AJRR is a multi stakeholder, independent, not
Zimmer Anterior Buttress Plate System. Surgical Technique
 Zimmer Anterior Buttress Plate System Surgical Technique 2 Zimmer Anterior Buttress Plate System Surgical Technique Zimmer Anterior Buttress Plate System Surgical Technique Description, Indications & Contraindications...
Zimmer Anterior Buttress Plate System Surgical Technique 2 Zimmer Anterior Buttress Plate System Surgical Technique Zimmer Anterior Buttress Plate System Surgical Technique Description, Indications & Contraindications...
Knee Replacement Complications
 Knee Replacement Complications Knee replacements have become a routine surgery in the United States. Nearly 700,000 people each year receive this life-improving surgery and are able to enjoy richer, more
Knee Replacement Complications Knee replacements have become a routine surgery in the United States. Nearly 700,000 people each year receive this life-improving surgery and are able to enjoy richer, more
TOTAL KNEE ARTHROPLASTY (TKA)
 TOTAL KNEE ARTHROPLASTY (TKA) 1 Anatomy, Biomechanics, and Design 2 Femur Medial and lateral condyles Convex, asymmetric Medial larger than lateral 3 Tibia Tibial plateau Medial tibial condyle: concave
TOTAL KNEE ARTHROPLASTY (TKA) 1 Anatomy, Biomechanics, and Design 2 Femur Medial and lateral condyles Convex, asymmetric Medial larger than lateral 3 Tibia Tibial plateau Medial tibial condyle: concave
KASM Knee Articulating Spacer Mold. Surgical Summary
 KASM Knee Articulating Spacer Mold Surgical Summary KASM Designing Surgeons: Michael H. Bourne, M.D. Salt Lake Orthopedic Clinic E. Marc Mariani, M.D. Salt Lake Orthopedic Clinic U.S. Patent 6,942,475
KASM Knee Articulating Spacer Mold Surgical Summary KASM Designing Surgeons: Michael H. Bourne, M.D. Salt Lake Orthopedic Clinic E. Marc Mariani, M.D. Salt Lake Orthopedic Clinic U.S. Patent 6,942,475
Case report: Pain L THR [ post THR 2 years; with history of trivial fall] Your Diagnosis?
![Case report: Pain L THR [ post THR 2 years; with history of trivial fall] Your Diagnosis? Case report: Pain L THR [ post THR 2 years; with history of trivial fall] Your Diagnosis?](/thumbs/78/78456133.jpg) Case report: Pain L THR [ post THR 2 years; with history of trivial fall] Your Diagnosis? Diagnosis: Ceramic head fracture In the 1970 s, Boutin implemented ceramic in modern total hip arthroplasty (THA).
Case report: Pain L THR [ post THR 2 years; with history of trivial fall] Your Diagnosis? Diagnosis: Ceramic head fracture In the 1970 s, Boutin implemented ceramic in modern total hip arthroplasty (THA).
Enhanced Stability Constrained Liners. Design Rationale Surgical Technique
 Enhanced Stability Constrained Liners Design Rationale Surgical Technique The Pinnacle Acetabular Cup System was designed to maximize the number of options available to the surgeon, and provide those options
Enhanced Stability Constrained Liners Design Rationale Surgical Technique The Pinnacle Acetabular Cup System was designed to maximize the number of options available to the surgeon, and provide those options
Technique Guide Small Bone Fusion System
 Technique Guide Small Bone Fusion System The Pinit Plate Small Bone Fusion System is a super low profile, modular bone plate and screw system designed to stabilize a bunionectomy with a medial to lateral
Technique Guide Small Bone Fusion System The Pinit Plate Small Bone Fusion System is a super low profile, modular bone plate and screw system designed to stabilize a bunionectomy with a medial to lateral
Failed Subtrochanteric Fracture How I Decide What to Do?
 Failed Subtrochanteric Fracture How I Decide What to Do? Gerald E. Wozasek Thomas M. Tiefenboeck 5 October 2016, Washington Medical University of Vienna, Department of Trauma Surgery ordination @wozasek.at
Failed Subtrochanteric Fracture How I Decide What to Do? Gerald E. Wozasek Thomas M. Tiefenboeck 5 October 2016, Washington Medical University of Vienna, Department of Trauma Surgery ordination @wozasek.at
Fractures of the tibia shaft treated with locked intramedullary nail Retrospective clinical and radiographic assesment
 ARS Medica Tomitana - 2013; 4(75): 197-201 DOI: 10.2478/arsm-2013-0035 Șerban Al., Botnaru V., Turcu R., Obadă B., Anderlik St. Fractures of the tibia shaft treated with locked intramedullary nail Retrospective
ARS Medica Tomitana - 2013; 4(75): 197-201 DOI: 10.2478/arsm-2013-0035 Șerban Al., Botnaru V., Turcu R., Obadă B., Anderlik St. Fractures of the tibia shaft treated with locked intramedullary nail Retrospective
Case Study: David. Conditions Treated Femoral Neck Fracture with Avascular Necrosis of the Hip. Age Range During Treatment 16 Years
 Case Study: David Conditions Treated Femoral Neck Fracture with Avascular Necrosis of the Hip Age Range During Treatment 16 Years David S. Feldman, MD Chief of Pediatric Orthopedic Surgery Professor of
Case Study: David Conditions Treated Femoral Neck Fracture with Avascular Necrosis of the Hip Age Range During Treatment 16 Years David S. Feldman, MD Chief of Pediatric Orthopedic Surgery Professor of
.org. Tibia (Shinbone) Shaft Fractures. Anatomy. Types of Tibial Shaft Fractures
 Tibia (Shinbone) Shaft Fractures Page ( 1 ) The tibia, or shinbone, is the most common fractured long bone in your body. The long bones include the femur, humerus, tibia, and fibula. A tibial shaft fracture
Tibia (Shinbone) Shaft Fractures Page ( 1 ) The tibia, or shinbone, is the most common fractured long bone in your body. The long bones include the femur, humerus, tibia, and fibula. A tibial shaft fracture
REFLECTION Constrained Liner Introduction
 Surgical Technique REFLECTION Constrained Liner Introduction Surgical treatment of recurrent dislocation should be directed toward correction of any mechanical abnormalities. If dislocation is caused
Surgical Technique REFLECTION Constrained Liner Introduction Surgical treatment of recurrent dislocation should be directed toward correction of any mechanical abnormalities. If dislocation is caused
Case Report Anterior Subluxation after Total Hip Replacement Confirmed by Radiographs: Report of Two Cases
 SAGE-Hindawi Access to Research Advances in Orthopedics Volume 2011, Article ID 519254, 4 pages doi:10.4061/2011/519254 Case Report Anterior Subluxation after Total Hip Replacement Confirmed by Radiographs:
SAGE-Hindawi Access to Research Advances in Orthopedics Volume 2011, Article ID 519254, 4 pages doi:10.4061/2011/519254 Case Report Anterior Subluxation after Total Hip Replacement Confirmed by Radiographs:
TC-PLUS Primary Knee IMPORTANT MEDICAL INFORMATION SPECIAL NOTE. This Package Insert is for product distributed in the US only.
 TC-PLUS Primary Knee IMPORTANT MEDICAL INFORMATION SPECIAL NOTE This Package Insert is for product distributed in the US only. The component material is provided on the outside carton label. Components
TC-PLUS Primary Knee IMPORTANT MEDICAL INFORMATION SPECIAL NOTE This Package Insert is for product distributed in the US only. The component material is provided on the outside carton label. Components
Comparitive Study between Proximal Femoral Nailing and Dynamic Hip Screw in Intertrochanteric Fracture of Femur *
 Open Journal of Orthopedics, 2013, 3, 291-295 Published Online November 2013 (http://www.scirp.org/journal/ojo) http://dx.doi.org/10.4236/ojo.2013.37053 291 Comparitive Study between Proximal Femoral Nailing
Open Journal of Orthopedics, 2013, 3, 291-295 Published Online November 2013 (http://www.scirp.org/journal/ojo) http://dx.doi.org/10.4236/ojo.2013.37053 291 Comparitive Study between Proximal Femoral Nailing
HOSPITAL "A" (2007) ORTHOPEDIC JOINT IMPLANT COMPONENT MATRIX
 HOSPITAL "A" (2007) ORTHOPEDIC JOINT IMPLANT COMPONENT MATRIX HIP IMPLANTS TOTAL HIPS Ultra High Demand (metal on metal) Femur, ingrowth Femoral head, metal Acetabular shell, ingrowth Acetabular liner,
HOSPITAL "A" (2007) ORTHOPEDIC JOINT IMPLANT COMPONENT MATRIX HIP IMPLANTS TOTAL HIPS Ultra High Demand (metal on metal) Femur, ingrowth Femoral head, metal Acetabular shell, ingrowth Acetabular liner,
YOU MAY NEED A KNEE REPLACEMENT
 Knee pain? YOU MAY NEED A KNEE REPLACEMENT Introduction If you re reading this brochure, you or a loved one is likely suffering from a severe knee problem. Serious knee joint conditions can cause pain,
Knee pain? YOU MAY NEED A KNEE REPLACEMENT Introduction If you re reading this brochure, you or a loved one is likely suffering from a severe knee problem. Serious knee joint conditions can cause pain,
Hip Fractures. Anatomy. Causes. Symptoms
 Hip Fractures A hip fracture is a break in the upper quarter of the femur (thigh) bone. The extent of the break depends on the forces that are involved. The type of surgery used to treat a hip fracture
Hip Fractures A hip fracture is a break in the upper quarter of the femur (thigh) bone. The extent of the break depends on the forces that are involved. The type of surgery used to treat a hip fracture
The Leader in Orthopaedic Innovation
 The Leader in Orthopaedic Innovation Wright is a leading international manufacturer and distributor of superior, easy to use, and innovative orthopaedic implants and instrumentation. For over 50 years,
The Leader in Orthopaedic Innovation Wright is a leading international manufacturer and distributor of superior, easy to use, and innovative orthopaedic implants and instrumentation. For over 50 years,
Total Hip Replacement
 Please contactmethroughthegoldcoasthospitaswityouhaveanyproblemsafteryoursurgery. Dr. Benjamin Hewitt Orthopaedic Surgeon Total Hip Replacement The hip joint is a ball and socket joint that connects the
Please contactmethroughthegoldcoasthospitaswityouhaveanyproblemsafteryoursurgery. Dr. Benjamin Hewitt Orthopaedic Surgeon Total Hip Replacement The hip joint is a ball and socket joint that connects the
CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician.
 CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician. ENGLISH Mpact 3D Metal Implants and Augments 3D Metal INSTRUCTION FOR USE Important notice: the device(s) can
CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician. ENGLISH Mpact 3D Metal Implants and Augments 3D Metal INSTRUCTION FOR USE Important notice: the device(s) can
Specializing in Mechanical Testing for Medical Devices. Office:
 Specializing in Mechanical Testing for Medical Devices Office: 260.489.1444 Email: sales@jtlamerica.com www.jtlamerica.com Spine Spinal implant test standards can t keep up with the rapid pace of device
Specializing in Mechanical Testing for Medical Devices Office: 260.489.1444 Email: sales@jtlamerica.com www.jtlamerica.com Spine Spinal implant test standards can t keep up with the rapid pace of device
JOINT RULER. Surgical Technique For Knee Joint JRReplacement
 JR JOINT RULER Surgical Technique For Knee Joint JRReplacement INTRODUCTION The Joint Ruler * is designed to help reduce the incidence of flexion, extension, and patellofemoral joint problems by allowing
JR JOINT RULER Surgical Technique For Knee Joint JRReplacement INTRODUCTION The Joint Ruler * is designed to help reduce the incidence of flexion, extension, and patellofemoral joint problems by allowing
Pedicle Subtraction Osteotomy. Case JB. Antonio Castellvi 5/19/2017
 Pedicle Subtraction Osteotomy John M. Small MD Florida Orthopedic Institute University South Florida Department Orthopedic Surgery Castellvi Spine May 11, 2017 Case JB 66 y/o male 74 235 lbs Retired police
Pedicle Subtraction Osteotomy John M. Small MD Florida Orthopedic Institute University South Florida Department Orthopedic Surgery Castellvi Spine May 11, 2017 Case JB 66 y/o male 74 235 lbs Retired police
UHMWPE LONGEVITY: INFLUENCES OF THE ORTHOPAEDIC TRIAD
 UHMWPE LONGEVITY: INFLUENCES OF THE ORTHOPAEDIC TRIAD AMERICAN ACADEMY OF ORTHOPAEDIC SURGEONS 66th Annual Meeting February 4-9, 1999 Anaheim, California COMMITTEE ON BIOMEDICAL ENGINEERING COMMITTEE ON
UHMWPE LONGEVITY: INFLUENCES OF THE ORTHOPAEDIC TRIAD AMERICAN ACADEMY OF ORTHOPAEDIC SURGEONS 66th Annual Meeting February 4-9, 1999 Anaheim, California COMMITTEE ON BIOMEDICAL ENGINEERING COMMITTEE ON
Automated Industry Report 823 Depuy Synthes Australia Attune CR Total Knee
 Automated Industry Report 823 Depuy Synthes Australia Total Knee Report Generated: 9 January 2019 This report has been prepared by the Australian Orthopaedic Association National Joint Replacement Registry
Automated Industry Report 823 Depuy Synthes Australia Total Knee Report Generated: 9 January 2019 This report has been prepared by the Australian Orthopaedic Association National Joint Replacement Registry
Patient Information for Consent
 Patient Information for Consent OS12 Femoral Shaft Fracture Surgery (Femoral Nailing) Expires end of February 2017 Issued June 2016 Local information If you need any more information please contact your
Patient Information for Consent OS12 Femoral Shaft Fracture Surgery (Femoral Nailing) Expires end of February 2017 Issued June 2016 Local information If you need any more information please contact your
The S.T.A.R. Scandinavian Total Ankle Replacement. Patient Information
 The S.T.A.R. Scandinavian Total Ankle Replacement Patient Information Patient Information This patient education brochure is presented by Small Bone Innovations, Inc. Patient results may vary. Please
The S.T.A.R. Scandinavian Total Ankle Replacement Patient Information Patient Information This patient education brochure is presented by Small Bone Innovations, Inc. Patient results may vary. Please
Total Hip Replacement. Find out why Total Hip Replacement may be right for you.
 Total Hip Replacement Find out why Total Hip Replacement may be right for you. UNDERSTANDING TOTAL HIP REPLACEMENT This brochure offers a brief overview of hip anatomy, arthritis and total hip arthroplasty.
Total Hip Replacement Find out why Total Hip Replacement may be right for you. UNDERSTANDING TOTAL HIP REPLACEMENT This brochure offers a brief overview of hip anatomy, arthritis and total hip arthroplasty.
Insert dissociation after fixed bearing PS constrained Genesis II total knee arthroplasty. A case series of nine patients
 Acta Orthop. Belg., 2015, 81, 747-751 ORIGINAL STUDY Insert dissociation after fixed bearing PS constrained Genesis II total knee arthroplasty. A case series of nine patients Timothy Voskuijl, Thijs A.
Acta Orthop. Belg., 2015, 81, 747-751 ORIGINAL STUDY Insert dissociation after fixed bearing PS constrained Genesis II total knee arthroplasty. A case series of nine patients Timothy Voskuijl, Thijs A.
JMSCR Vol. 03 Issue 08 Page August 2015
 www.jmscr.igmpublication.org Impact Factor 3.79 Index Copernicus Value: 5.88 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: http://dx.doi.org/10.18535/jmscr/v3i8.08 Study of Functional and Radiological Outcome
www.jmscr.igmpublication.org Impact Factor 3.79 Index Copernicus Value: 5.88 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: http://dx.doi.org/10.18535/jmscr/v3i8.08 Study of Functional and Radiological Outcome
A Structural Service Plan: Towards Better and Safer Spine Surgeries. Department of Orthopaedics & Traumatology Tuen Mun Hospital
 A Structural Service Plan: Towards Better and Safer Spine Surgeries Department of Orthopaedics & Traumatology Tuen Mun Hospital Cheung KK Wong CY Chan Andrew Tse Alfred Chow YY Department of Orthopaedics
A Structural Service Plan: Towards Better and Safer Spine Surgeries Department of Orthopaedics & Traumatology Tuen Mun Hospital Cheung KK Wong CY Chan Andrew Tse Alfred Chow YY Department of Orthopaedics
BRIDGE PLATING OF COMMINUTED SHAFT OF FEMUR FRACTURES
 BRIDGE PLATING OF COMMINUTED SHAFT OF FEMUR FRACTURES Mohammad Abul kalam, Pradeep Kumar, Mohammad Afzal Hussain and Iqbal Ahmad Abstract A prospective study of forty comminuted femoral shaft fractures,
BRIDGE PLATING OF COMMINUTED SHAFT OF FEMUR FRACTURES Mohammad Abul kalam, Pradeep Kumar, Mohammad Afzal Hussain and Iqbal Ahmad Abstract A prospective study of forty comminuted femoral shaft fractures,
Total Hip Replacement
 PATIENT EDUCATION Total Hip Replacement A guide to understanding total hip replacement surgery, featuring The George Archer Story. BACK TO MOBILITY George Archer s Personal Adventure On April16, 1996,
PATIENT EDUCATION Total Hip Replacement A guide to understanding total hip replacement surgery, featuring The George Archer Story. BACK TO MOBILITY George Archer s Personal Adventure On April16, 1996,
Locked plating constructs are creating a challenge for surgeons.
 Locked plating constructs are creating a challenge for surgeons. Three recent studies examining supracondylar femur fractures show concern for the high degree of stiffness of locked plating constructs
Locked plating constructs are creating a challenge for surgeons. Three recent studies examining supracondylar femur fractures show concern for the high degree of stiffness of locked plating constructs
Automated Industry Report 824 Depuy Synthes Australia Attune PS Total Knee
 Automated Industry Report 824 Depuy Synthes Australia Total Knee Report Generated: 9 January 2019 This report has been prepared by the Australian Orthopaedic Association National Joint Replacement Registry
Automated Industry Report 824 Depuy Synthes Australia Total Knee Report Generated: 9 January 2019 This report has been prepared by the Australian Orthopaedic Association National Joint Replacement Registry
AOANJRR Automated Industry Report Depuy Synthes Australia Attune PS Total Knee Data Period: 1 September August 2018
 AOANJRR Automated Industry Report 335 - Depuy Synthes Australia Total Knee Catalogue Numbers of Femoral Components included in this analysis Model Catalogue Range Range Description No. of Primary Procedures
AOANJRR Automated Industry Report 335 - Depuy Synthes Australia Total Knee Catalogue Numbers of Femoral Components included in this analysis Model Catalogue Range Range Description No. of Primary Procedures
Case Report Bilateral Distal Femoral Nailing in a Rare Symmetrical Periprosthetic Knee Fracture
 Case Reports in Orthopedics, Article ID 745083, 4 pages http://dx.doi.org/10.1155/2014/745083 Case Report Bilateral Distal Femoral Nailing in a Rare Symmetrical Periprosthetic Knee Fracture Marcos Carvalho,
Case Reports in Orthopedics, Article ID 745083, 4 pages http://dx.doi.org/10.1155/2014/745083 Case Report Bilateral Distal Femoral Nailing in a Rare Symmetrical Periprosthetic Knee Fracture Marcos Carvalho,
Osteosynthesis involving a joint Thomas P Rüedi
 Osteosynthesis involving a joint Thomas P Rüedi How to use this handout? The left column contains the information given during the lecture. The column at the right gives you space to make personal notes.
Osteosynthesis involving a joint Thomas P Rüedi How to use this handout? The left column contains the information given during the lecture. The column at the right gives you space to make personal notes.
Pinit Plate Small Bone Fusion System Bone Plate & Screw System
 Pinit Plate Small Bone Fusion System Bone Plate & Screw System Description The Pinit Plate Small Bone Fusion System consists of 2-hole bone plates made available in three length options and two thickness
Pinit Plate Small Bone Fusion System Bone Plate & Screw System Description The Pinit Plate Small Bone Fusion System consists of 2-hole bone plates made available in three length options and two thickness
EARLY CLINICAL RESULTS OF PRIMARY CEMENTLESS TOTAL KNEE ARTHROPLASTY
 EARLY CLINICAL RESULTS OF PRIMARY CEMENTLESS TOTAL KNEE ARTHROPLASTY Benkovich V. Perry T., Bunin A., Bilenko V., Unit for Joint Arthroplasty, Soroka Medical Center Ben Gurion University of Negev Beer
EARLY CLINICAL RESULTS OF PRIMARY CEMENTLESS TOTAL KNEE ARTHROPLASTY Benkovich V. Perry T., Bunin A., Bilenko V., Unit for Joint Arthroplasty, Soroka Medical Center Ben Gurion University of Negev Beer
Original Date: December 2015 Page 1 of 8 FOR CMS (MEDICARE) MEMBERS ONLY
 National Imaging Associates, Inc. Clinical guidelines TOTAL JOINT ARTHROPLASTY -Total Hip Arthroplasty -Total Knee Arthroplasty -Replacement/Revision Hip or Knee Arthroplasty CPT4 Codes: Please refer to
National Imaging Associates, Inc. Clinical guidelines TOTAL JOINT ARTHROPLASTY -Total Hip Arthroplasty -Total Knee Arthroplasty -Replacement/Revision Hip or Knee Arthroplasty CPT4 Codes: Please refer to
Robotic-Arm Assisted Surgery
 Mako TM Robotic-Arm Assisted Surgery for Total Hip Replacement A Patient s Guide Causes of Your Hip Pain Your joints are involved in almost every activity you do. Movements such as walking, bending and
Mako TM Robotic-Arm Assisted Surgery for Total Hip Replacement A Patient s Guide Causes of Your Hip Pain Your joints are involved in almost every activity you do. Movements such as walking, bending and
ProDisc-L Total Disc Replacement. IDE Clinical Study.
 ProDisc-L Total Disc Replacement. IDE Clinical Study. A multi-center, prospective, randomized clinical trial. Instruments and implants approved by the AO Foundation Table of Contents Indications, Contraindications
ProDisc-L Total Disc Replacement. IDE Clinical Study. A multi-center, prospective, randomized clinical trial. Instruments and implants approved by the AO Foundation Table of Contents Indications, Contraindications
Case Report. Antegrade Femur Lengthening with the PRECICE Limb Lengthening Technology
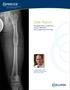 Case Report Antegrade Femur Lengthening with the PRECICE Limb Lengthening Technology S. Robert Rozbruch, MD Hospital for Special Surgery New York, NY, USA ABSTRACT This is a case illustrating a 4.5 cm
Case Report Antegrade Femur Lengthening with the PRECICE Limb Lengthening Technology S. Robert Rozbruch, MD Hospital for Special Surgery New York, NY, USA ABSTRACT This is a case illustrating a 4.5 cm
TM TM Surgical Technique
 TM TM Surgical Technique TABLE OF CONTENTS Reli SP Spinous Plating System Overview Device Description Implant Features Indications Instruments Access Instruments Preparation Instruments Insertion Instruments
TM TM Surgical Technique TABLE OF CONTENTS Reli SP Spinous Plating System Overview Device Description Implant Features Indications Instruments Access Instruments Preparation Instruments Insertion Instruments
VTI INTERLINK PEDICLE SCREW SYSTEM
 VTI INTERLINK PEDICLE SCREW SYSTEM SURGICAL TECHNIQUE FORWARD THINKING FOR THE BACK. DEVICE DESCRIPTION The VTI InterLink Pedicle Screw System is comprised of polyaxial pedicle screws in various diameters
VTI INTERLINK PEDICLE SCREW SYSTEM SURGICAL TECHNIQUE FORWARD THINKING FOR THE BACK. DEVICE DESCRIPTION The VTI InterLink Pedicle Screw System is comprised of polyaxial pedicle screws in various diameters
YOUR TOTAL HIP REPLACEMENT SURGERY STEPS TO RETURNING TO A LIFESTYLE YOU DESERVE
 YOUR TOTAL HIP REPLACEMENT SURGERY STEPS TO RETURNING TO A LIFESTYLE YOU DESERVE IMPORTANT. PLEASE NOTE. This brochure offers a brief overview of hip anatomy, arthritis and hip replacement surgery. The
YOUR TOTAL HIP REPLACEMENT SURGERY STEPS TO RETURNING TO A LIFESTYLE YOU DESERVE IMPORTANT. PLEASE NOTE. This brochure offers a brief overview of hip anatomy, arthritis and hip replacement surgery. The
The causes of OA of the knee are multiple and include aging (wear and tear), obesity, and previous knee trauma or surgery. OA affects usually the
 The Arthritic Knee The causes of OA of the knee are multiple and include aging (wear and tear), obesity, and previous knee trauma or surgery. OA affects usually the medial compartment of the knee, and
The Arthritic Knee The causes of OA of the knee are multiple and include aging (wear and tear), obesity, and previous knee trauma or surgery. OA affects usually the medial compartment of the knee, and
IMPORTANT MEDICAL INFORMATION Advanced Orthopaedic Solutions INTRAMEDULLARY NAILS Warnings and Precautions (SINGLE USE ONLY)
 IMPORTANT MEDICAL INFORMATION Advanced Orthopaedic Solutions INTRAMEDULLARY NAILS Warnings and Precautions (SINGLE USE ONLY) IMPORTANT NOTE Intramedullary nails provide an alternative to open reduction
IMPORTANT MEDICAL INFORMATION Advanced Orthopaedic Solutions INTRAMEDULLARY NAILS Warnings and Precautions (SINGLE USE ONLY) IMPORTANT NOTE Intramedullary nails provide an alternative to open reduction
Total Knee Original System Primary Surgical Technique
 Surgical Procedure Total Knee Original System Primary Surgical Technique Where as a total hip replacement is primarily a bony operation, a total knee replacement is primarily a soft tissue operation. Excellent
Surgical Procedure Total Knee Original System Primary Surgical Technique Where as a total hip replacement is primarily a bony operation, a total knee replacement is primarily a soft tissue operation. Excellent
DISTAL RADIUS. Instructions for Use
 DISTAL RADIUS Instructions for Use CAUTION: FEDERAL LAW RESTRICTS THESE DEVICES TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Table of Contents 1. INTENDED USE... 3 2. DEVICE DESCRIPTION... 3 3. METHOD OF
DISTAL RADIUS Instructions for Use CAUTION: FEDERAL LAW RESTRICTS THESE DEVICES TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Table of Contents 1. INTENDED USE... 3 2. DEVICE DESCRIPTION... 3 3. METHOD OF
TMJ Joint Replacement System
 TMJ Joint Replacement System Patient Information What is the Temporomandibular Joint (TMJ)? The Temporomandibular Joint is one of the body s most complex joints. It is similar to a ball and socket, but
TMJ Joint Replacement System Patient Information What is the Temporomandibular Joint (TMJ)? The Temporomandibular Joint is one of the body s most complex joints. It is similar to a ball and socket, but
RECOVERY. P r o t r u s i o
 RECOVERY P r o t r u s i o TM C a g e RECOVERY P r o t r u s i o TM C a g e Design Features Revision acetabular surgery is a major challenge facing today s total joint revision surgeon. Failed endo/bi-polars,
RECOVERY P r o t r u s i o TM C a g e RECOVERY P r o t r u s i o TM C a g e Design Features Revision acetabular surgery is a major challenge facing today s total joint revision surgeon. Failed endo/bi-polars,
YOUR TOTAL HIP REPLACEMENT SURGERY
 YOUR TOTAL HIP REPLACEMENT SURGERY STEPS TO RETURNING TO A LIFESTYLE YOU DESERVE Exactech_030H Rev A_Total Hip Replacement Surgery_PRINT.indd 1 IMPORTANT. PLEASE NOTE. This brochure offers a brief overview
YOUR TOTAL HIP REPLACEMENT SURGERY STEPS TO RETURNING TO A LIFESTYLE YOU DESERVE Exactech_030H Rev A_Total Hip Replacement Surgery_PRINT.indd 1 IMPORTANT. PLEASE NOTE. This brochure offers a brief overview
U2 PSA. Revision Knee. Surgical Protocol
 U2 PSA TM Revision Knee Surgical Protocol Table of Contents 1 Component Removal... 1 2 Tibial Preparation... 1 2.1 Tibial Canal Preparation... 1 2.2 Proximal Tibial Resection... 2 2.3 Non Offset Tibial
U2 PSA TM Revision Knee Surgical Protocol Table of Contents 1 Component Removal... 1 2 Tibial Preparation... 1 2.1 Tibial Canal Preparation... 1 2.2 Proximal Tibial Resection... 2 2.3 Non Offset Tibial
Periarticular knee osteotomy
 Periarticular knee osteotomy Turnberg Building Orthopaedics 0161 206 4803 All Rights Reserved 2018. Document for issue as handout. Knee joint The knee consists of two joints which allow flexion (bending)
Periarticular knee osteotomy Turnberg Building Orthopaedics 0161 206 4803 All Rights Reserved 2018. Document for issue as handout. Knee joint The knee consists of two joints which allow flexion (bending)
JOURNALOF ORTHOPAEDIC TRAUMA
 JOT Special Case Report Series CASE REPORTS www.jorthotrauma.com JOURNALOF ORTHOPAEDIC TRAUMA OFFICIAL JOURNAL OF Orthopaedic Trauma Association Belgian Orthopaedic Trauma Association Canadian Orthopaedic
JOT Special Case Report Series CASE REPORTS www.jorthotrauma.com JOURNALOF ORTHOPAEDIC TRAUMA OFFICIAL JOURNAL OF Orthopaedic Trauma Association Belgian Orthopaedic Trauma Association Canadian Orthopaedic
Hemiarthroplasty (half hip replacement)
 Hemiarthroplasty (half hip replacement) Trauma and Orthopaedics Patient Information Leaflet Introduction This leaflet is about an operation called a half hip replacement. It gives information about the
Hemiarthroplasty (half hip replacement) Trauma and Orthopaedics Patient Information Leaflet Introduction This leaflet is about an operation called a half hip replacement. It gives information about the
For Commercial products, please refer to the following policy: Preauthorization via Web-Based Tool for Procedures
 Medical Coverage Policy Total Joint Arthroplasty Hip and Knee EFFECTIVE DATE: 08/01/2017 POLICY LAST UPDATED: 06/06/2017 OVERVIEW Joint replacement surgery, also known as arthroplasty, has proved to be
Medical Coverage Policy Total Joint Arthroplasty Hip and Knee EFFECTIVE DATE: 08/01/2017 POLICY LAST UPDATED: 06/06/2017 OVERVIEW Joint replacement surgery, also known as arthroplasty, has proved to be
CC TRIO VERSAFITCUP. Surgical Technique. each to their own. Hip Knee Spine Navigation
 VERSAFITCUP CC TRIO each to their own Surgical Technique Hip Knee Spine Navigation Versafitcup CC TRIO Surgical Technique Hip Knee Spine Navigation EACH TO THEIR OWN The Versafitcup CC Trio is a range
VERSAFITCUP CC TRIO each to their own Surgical Technique Hip Knee Spine Navigation Versafitcup CC TRIO Surgical Technique Hip Knee Spine Navigation EACH TO THEIR OWN The Versafitcup CC Trio is a range
National Joint Replacement Registry. Lay Summary 2015 Annual Report Hip and Knee Replacement
 National Joint Replacement Registry Lay Summary 2015 Annual Report Hip and Knee Replacement SUPPLEMENTARY REPORT 2015 TABLE OF CONTENTS Introduction... 1 A brief history of the Registry origins... 1 The
National Joint Replacement Registry Lay Summary 2015 Annual Report Hip and Knee Replacement SUPPLEMENTARY REPORT 2015 TABLE OF CONTENTS Introduction... 1 A brief history of the Registry origins... 1 The
Bone Bangalore
 Dr Suresh Annamalai MBBS, MRCS(Edn), FRCS( Tr & Orth)(Edn), FEBOT(European Board), Young Hip and Knee Fellowship(Harrogate, UK) Consultant Arthroplasty and Arthroscopic Surgeon Manipal Hospital, Whitefield,
Dr Suresh Annamalai MBBS, MRCS(Edn), FRCS( Tr & Orth)(Edn), FEBOT(European Board), Young Hip and Knee Fellowship(Harrogate, UK) Consultant Arthroplasty and Arthroscopic Surgeon Manipal Hospital, Whitefield,
VLIFT System Overview. Vertebral Body Replacement System
 VLIFT System Overview Vertebral Body Replacement System VLIFT System System Description The VLIFT Vertebral Body Replacement System consists of a Distractible In Situ (DIS) implant, which enables the surgeon
VLIFT System Overview Vertebral Body Replacement System VLIFT System System Description The VLIFT Vertebral Body Replacement System consists of a Distractible In Situ (DIS) implant, which enables the surgeon
Mr Aslam Mohammed FRCS, FRCS (Orth) Consultant Orthopaedic Surgeon Specialising in Lower Limb Arthroplasty and Sports Injury
 Mr Aslam Mohammed FRCS, FRCS (Orth) Consultant Orthopaedic Surgeon Specialising in Lower Limb Arthroplasty and Sports Injury I qualified from the Welsh National School of Medicine in Cardiff in 1984. I
Mr Aslam Mohammed FRCS, FRCS (Orth) Consultant Orthopaedic Surgeon Specialising in Lower Limb Arthroplasty and Sports Injury I qualified from the Welsh National School of Medicine in Cardiff in 1984. I
A U X I L I A R Y C O N N E C T O R S Surgical Technique
 A U X I L I A R Y C O N N E C T O R S Surgical Technique AUXILIARY CONNECTORS ISSYS LP Auxiliary Connectors The ISSYS LP auxiliary connectors were designed to provide medial-lateral variability for the
A U X I L I A R Y C O N N E C T O R S Surgical Technique AUXILIARY CONNECTORS ISSYS LP Auxiliary Connectors The ISSYS LP auxiliary connectors were designed to provide medial-lateral variability for the
EVOS MINI with IM Nailing
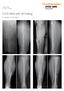 Case Series Dr. John A. Scolaro EVOS MINI with IM Nailing A series of studies Introduction Intramedullary nailing has become the standard for many long bone fractures. Fracture reduction prior to nail
Case Series Dr. John A. Scolaro EVOS MINI with IM Nailing A series of studies Introduction Intramedullary nailing has become the standard for many long bone fractures. Fracture reduction prior to nail
Anterior Approach to Hip Replacement Surgery
 Anterior Approach to Hip Replacement Surgery Introduction When debilitating pain and stiffness in your hip limits your daily activities, you may need a total hip replacement. The development of total hip
Anterior Approach to Hip Replacement Surgery Introduction When debilitating pain and stiffness in your hip limits your daily activities, you may need a total hip replacement. The development of total hip
If you have a condition that compresses your nerves, causing debilitating back pain or numbness along the back of your leg.
 Below, we have provided some basic information for your benefit. Please use this information is an educational aid only. It is not intended as medical advice for individual conditions or treatments. Talk
Below, we have provided some basic information for your benefit. Please use this information is an educational aid only. It is not intended as medical advice for individual conditions or treatments. Talk
Understanding Hip Implant Options
 Understanding Hip Implant Options Cup (Socket) Bearing (Liner) Head Stem (Femur) Modern Hip Implants (Ball) Metal Femoral Head on Marathon Cross-linked Polyethylene Liner Proven materials Polyethylene
Understanding Hip Implant Options Cup (Socket) Bearing (Liner) Head Stem (Femur) Modern Hip Implants (Ball) Metal Femoral Head on Marathon Cross-linked Polyethylene Liner Proven materials Polyethylene
Table of Contents.
 surgical technique The Ambassador TM Anterior Cervical Plate System is a versatile system of implants and instruments with a variety of sizes to provide optimal anatomic compatibility. The integrated cam
surgical technique The Ambassador TM Anterior Cervical Plate System is a versatile system of implants and instruments with a variety of sizes to provide optimal anatomic compatibility. The integrated cam
Tibial Nailing for Tibial Shaft Fracture
 Tibial Nailing for Tibial Shaft Fracture Patient Information Trauma and Orthopaedics Department Author ID: KC Leaflet Number: Musc 015 Name of Leaflet: Tibial Nailing for Nail Shaft Fracture Version: 4
Tibial Nailing for Tibial Shaft Fracture Patient Information Trauma and Orthopaedics Department Author ID: KC Leaflet Number: Musc 015 Name of Leaflet: Tibial Nailing for Nail Shaft Fracture Version: 4
Institute for Bone, Joint Replacement, Orthopaedics Spine and Sports Medicine
 24-Hour Helpline: 011-30403040 BLK Super Speciality Hospital Pusa Road, New Delhi - 110005 (India). www.blkhospital.com Institute for Bone, Joint Replacement, Orthopaedics Spine and Sports Medicine Introduction
24-Hour Helpline: 011-30403040 BLK Super Speciality Hospital Pusa Road, New Delhi - 110005 (India). www.blkhospital.com Institute for Bone, Joint Replacement, Orthopaedics Spine and Sports Medicine Introduction
A Patient s Guide to Partial Knee Resurfacing
 A Patient s Guide to Partial Knee Resurfacing Surgical Outcomes System (SOS ) www.orthoillustrated.com OrthoIllustrated is a leading Internet-based resource for patient education. Please visit this website
A Patient s Guide to Partial Knee Resurfacing Surgical Outcomes System (SOS ) www.orthoillustrated.com OrthoIllustrated is a leading Internet-based resource for patient education. Please visit this website
Life. Uncompromised. The KineSpring Knee Implant System Surgeon Handout
 Life Uncompromised The KineSpring Knee Implant System Surgeon Handout 2 Patient Selection Criteria Patient Selection Criteria Medial compartment degeneration must be confirmed radiographically or arthroscopically
Life Uncompromised The KineSpring Knee Implant System Surgeon Handout 2 Patient Selection Criteria Patient Selection Criteria Medial compartment degeneration must be confirmed radiographically or arthroscopically
Important notice: the device(s) can be prescribed and implanted only by a doctor legally authorized to perform this type of surgery.
 rev.10 CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician. ENGLISH - EVOLIS/GMK KNEE PROSTHESIS - INSTRUCTIONS FOR USE Important notice: the device(s) can be prescribed
rev.10 CAUTION Federal law (USA) restricts this device to sale, by or on the order of a physician. ENGLISH - EVOLIS/GMK KNEE PROSTHESIS - INSTRUCTIONS FOR USE Important notice: the device(s) can be prescribed
Crossed Steinmann Pin Fixation In Supracondylar Femur Fractures In Adults A Case Series
 Article ID: WMC005027 ISSN 2046-1690 Crossed Steinmann Pin Fixation In Supracondylar Femur Fractures In Adults A Case Series Peer review status: No Corresponding Author: Dr. Mohit K Jindal, Senior Resident,
Article ID: WMC005027 ISSN 2046-1690 Crossed Steinmann Pin Fixation In Supracondylar Femur Fractures In Adults A Case Series Peer review status: No Corresponding Author: Dr. Mohit K Jindal, Senior Resident,
The Treatment of Pelvic Discontinuity During Acetabular Revision
 The Journal of Arthroplasty Vol. 20 No. 4 Suppl. 2 2005 The Treatment of Pelvic Discontinuity During Acetabular Revision Scott M. Sporer, MD, MS,*y Michael O Rourke, MD,z and Wayne G. Paprosky, MD, FACS*y
The Journal of Arthroplasty Vol. 20 No. 4 Suppl. 2 2005 The Treatment of Pelvic Discontinuity During Acetabular Revision Scott M. Sporer, MD, MS,*y Michael O Rourke, MD,z and Wayne G. Paprosky, MD, FACS*y
Peritroch Hip Fractures. Robert M Harris MD. Hip Fractures. Factors Influencing Construct Strength: Uncontrolled factors 4/28/2016
 Peritroch Hip Fractures Should be treated with an IMHS Robert M Harris MD Hip Fractures General principles Approximately 250,000 hip fractures/ year Cost approximately $8.7 billion annually The number
Peritroch Hip Fractures Should be treated with an IMHS Robert M Harris MD Hip Fractures General principles Approximately 250,000 hip fractures/ year Cost approximately $8.7 billion annually The number
Surgical Technique. Apache Anterior Lumbar Interbody Fusion
 Surgical Technique Apache Anterior Lumbar Interbody Fusion 2 Table of Contents Page Preoperative Planning 4 Patient Positioning 4 Disc and Endplate Preparation 4 Distraction/Size Selection 5 Implantation
Surgical Technique Apache Anterior Lumbar Interbody Fusion 2 Table of Contents Page Preoperative Planning 4 Patient Positioning 4 Disc and Endplate Preparation 4 Distraction/Size Selection 5 Implantation
PATIENT INFORMATION REMEDY ACETABULAR CUP. Overview
 PATIENT INFORMATION REMEDY ACETABULAR CUP 1 Overview The REMEDY Hip Spacer is a temporary hip implant that is used as part of a two-stage procedure. If you are receiving the REMEDY Hip Spacer, your surgeon
PATIENT INFORMATION REMEDY ACETABULAR CUP 1 Overview The REMEDY Hip Spacer is a temporary hip implant that is used as part of a two-stage procedure. If you are receiving the REMEDY Hip Spacer, your surgeon
5/31/2018. Ipsilateral Femoral Neck And Shaft Fractures. Ipsilateral Neck-Shaft Fractures Introduction. Ipsilateral Neck-Shaft Fractures Introduction
 Ipsilateral Femoral Neck And Shaft Fractures Exchange Nailing For Non- Union Donald Wiss MD Cedars-Sinai Medical Center Los Angeles, California Introduction Uncommon Injury Invariably High Energy Trauma
Ipsilateral Femoral Neck And Shaft Fractures Exchange Nailing For Non- Union Donald Wiss MD Cedars-Sinai Medical Center Los Angeles, California Introduction Uncommon Injury Invariably High Energy Trauma
Subtrochanteric Femur Fracture. 70 yo female 7/22/2013
 Subtrochanteric Femur Fracture Philip J. Kregor, MD 70 yo female Mild hypertension, Type II Diabetes Fell from 20 feet while at a party when the porch collapsed Mild head injury, Rib fractures Had been
Subtrochanteric Femur Fracture Philip J. Kregor, MD 70 yo female Mild hypertension, Type II Diabetes Fell from 20 feet while at a party when the porch collapsed Mild head injury, Rib fractures Had been
Technique Guide KISSloc Suture System
 Technique Guide KISSloc Suture System The KISSloc Suture System consists of a strong self-cinching suture assembly, a unique load-dispersing Arrow Plate and procedure specific instrumentation. Simple,
Technique Guide KISSloc Suture System The KISSloc Suture System consists of a strong self-cinching suture assembly, a unique load-dispersing Arrow Plate and procedure specific instrumentation. Simple,
New Implantable Lengthening Nail: Preliminary Results. Matthew Harris MD, MBA Dror Paley MD, FRCS(C) Daniel Prince MD, MPH
 New Implantable Lengthening Nail: Preliminary Results Matthew Harris MD, MBA Dror Paley MD, FRCS(C) Daniel Prince MD, MPH LLRS New York, NY July 19, 2013 Disclosures Dror Paley MD, FRCS(C) Paid Consultant
New Implantable Lengthening Nail: Preliminary Results Matthew Harris MD, MBA Dror Paley MD, FRCS(C) Daniel Prince MD, MPH LLRS New York, NY July 19, 2013 Disclosures Dror Paley MD, FRCS(C) Paid Consultant
INTELLIGENT SPINAL SYSTEM
 INTELLIGENT SPINAL SYSTEM I. Introduction II. Product Specification III. Surgical Technique IV. Ordering Information V. IFU for Lospa IS SPINAL SYSTEM The LOSPA IS spinal system consists of
INTELLIGENT SPINAL SYSTEM I. Introduction II. Product Specification III. Surgical Technique IV. Ordering Information V. IFU for Lospa IS SPINAL SYSTEM The LOSPA IS spinal system consists of
Integra. Salto Talaris Total Ankle Prosthesis PATIENT INFORMATION
 Integra Salto Talaris Total Ankle Prosthesis PATIENT INFORMATION Fibula Articular Surface Lateral Malleolus Tibia Medial Malleolus Talus Anterior view of the right ankle region Talo-fibular Ligament Calcaneal
Integra Salto Talaris Total Ankle Prosthesis PATIENT INFORMATION Fibula Articular Surface Lateral Malleolus Tibia Medial Malleolus Talus Anterior view of the right ankle region Talo-fibular Ligament Calcaneal
