Precautionary Statement ( )
|
|
|
- Pauline Willis
- 5 years ago
- Views:
Transcription
1 Precautionary Statement ( ) Biomet Sports Medicine, Inc E. Airport Dr. Rev. A Ontario, CA Date: 06/07 Sleeve and Button Soft Tissue Devices Utilizing ZipLoop Technology ATTENTION OPERATING SURGEON DESCRIPTION The Sleeve and Button Soft Tissue Devices utilizing ZipLoop technology consist of a two-piece design. The first piece is either a button or sleeve, and the second piece is a ZipLoop construct. The ZipLoop construct is an adjustable loop created with a single piece of fiber material. The devices are available in various material and size combinations. When the loop is pulled tight, the Sleeve or Button locks against the bone, fixating the soft tissue. NOTE: The terms Ziploop construct and loop are used interchangeably throughout this document. Materials Loop Sleeve Button Ultra-High Molecular Weight Polyethylene (UHMWPE)/Polypropylene or Polyester UHMWPE/Polypropylene or Polyester Titanium alloy or Polyetheretherketone (PEEK) INDICATIONS The Sleeve and Button Soft Tissue Devices utilizing ZipLoop technology are intended for soft tissue to bone fixation for the following indications: Shoulder Bankart lesion repair SLAP lesion repair Acromio-clavicular repair Capsular shift / capsulolabral reconstruction Deltoid repair Rotator cuff tear repair Biceps tenodesis Foot and Ankle Medial / lateral repair and reconstruction Mid- and forefoot repair Hallux valgus reconstruction Metatarsal ligament/tendon repair or reconstruction Achilles Tendon Repair
2 Elbow Ulnar or radial collateral ligament reconstruction Lateral epicondylitis repair Biceps tendon reattachment Knee Extra-capsular repair: MCL, LCL, and posterior oblique ligament Iliotibial band tenodesis Patellar tendon repair VMO advancement Joint capsule closure Hand and Wrist Collateral ligament repair Scapholunate ligament reconstruction Tendon transfers in phalanx Volar plate reconstruction Hip Acetabular labral repair In addition to the aforementioned indications, the Button Soft Tissue Devices (all sizes) and size specific Sleeve Soft Tissue Devices (as shown in Table 1) are indicated for the following: Knee ACL/PCL repair/reconstruction ACL/PCL patellar bone-tendon-bone grafts Double-Tunnel ACL reconstruction Table 1 SLEEVE SOFT TISSUE DEVICES Specific Combinations for ACL/PCL Indications Loop Size Sleeve Size UHMWPE/ Polypropylene Polyester UHMWPE/ Polypropylene Polyester #2-0 or > -- #2 or > -- #2-0 or > #0 or > -- #2 or > -- #5 or > Example: Note: The following sleeve soft tissue device combination may be used for ACL/PCL indications: #2-0 UHMWPE/Polypropylene loop + #2 UHMWPE/polypropylene sleeve The loop and sleeve sizes denote the diameter of the original UHMWPE/Polypropylene or polyester braided fiber.
3 CONTRAINDICATIONS 1. Infection. 2. Patient conditions including blood supply limitations and insufficient quantity or quality of bone or soft tissue. 3. Patients with mental or neurologic conditions who are unwilling or incapable of following postoperative care instructions. 4. Foreign body sensitivity. Where material sensitivity is suspected, testing is to be completed prior to implantation of the device. WARNINGS Biomet Sports Medicine internal fixation devices provide the surgeon with a means to aid in the management of soft tissue to bone reattachment procedures. While these devices are generally successful in attaining these goals, they cannot be expected to replace normal healthy bone or withstand the stress placed upon the device by full or partial weight bearing or load bearing, particularly in the presence of nonunion, delayed union, or incomplete healing. Therefore, it is important that immobilization (use of external support, walking aids, braces, etc.) of the treatment site be maintained until healing has occurred. Surgical implants are subject to repeated stresses in use, which can result in fracture or damage to the implant. Factors such as the patient s weight, activity level, and adherence to weight bearing or load bearing instructions have an effect on the service life of the implant. The surgeon must be thoroughly knowledgeable not only in the medical and surgical aspects of the implant, but also must be aware of the mechanical and material aspects of the surgical implants. Patient selection factors to be considered include: 1) need for soft tissue to bone fixation, 2) ability and willingness of the patient to follow postoperative care instructions until healing is complete, and 3) a good nutritional state of the patient. 1. Correct selection of the implant is extremely important. The potential for success in soft tissue to bone fixation is increased by the selection of the proper type of implant. While proper selection can help minimize risks, neither the device nor grafts, when used, are designed to withstand the unsupported stress of full weight bearing, load bearing or excessive activity. 2. Improper selection, placement, positioning, and fixation of the device can lead to failure of the device or the procedure. The surgeon is to be familiar with the device, the method of application and the surgical procedure prior to performing surgery. The surgeon must select a type or types of internal fixation devices appropriate for treatment. 3. The implants can loosen or be damaged and the graft can fail when subjected to increased loading associated with nonunion or delayed union. If healing is delayed, or does not occur, the implant or the procedure may fail. Loads produced by weight bearing and activity levels may dictate the longevity of the implant. 4. Inadequate fixation at the time of surgery can increase the risk of loosening and migration of the device or tissue supported by the device. Sufficient bone quantity and quality are important to adequate fixation and success of the procedure. Bone quality must be assessed at the time of surgery. Adequate fixation in diseased bone may be more difficult. Patients with poor quality bone, such as osteoporotic bone, are at greater risk of device loosening and procedure failure. 5. Care is to be taken to ensure adequate soft tissue fixation at the time of surgery. Failure to achieve adequate fixation or improper positioning or placement of the device can contribute to a subsequent undesirable result. 6. The use of appropriate immobilization and postoperative management is indicated as part of the treatment until healing has occurred. 7. Do not modify implants. Do not notch or bend titanium or PEEK implants. Notches or scratches put in the implants during the course of surgery may contribute to breakage.
4 8. Correct handling of the device is extremely important. Avoid crushing or crimping damage due to application of surgical instruments such as forceps or needle holders. 9. Do not use excessive force when inserting the device. Excessive force may cause damage to the device and/or adversely affect its performance. 10. The device can break or be damaged due to excessive activity or trauma. This could lead to failure requiring additional surgery and device removal. 11. DO NOT USE if there is a loss of sterility of the device. 12. Discard and DO NOT USE opened or damaged devices. Use only devices that are package in unopened or undamaged containers. 13. Ensure contact of tissue to bone when implanting. 14. Adequately instruct the patient. Postoperative care is important. The patient s ability and willingness to follow instructions is one of the most important aspects of successful fracture management. Patients with senility, mental illness, alcoholism, or drug abuse may be at higher risk of device failure. These patients may ignore instructions and activity restrictions. The patient is to be instructed in the use of external supports (walking aids, slings, braces, etc.) that are intended to immobilize the treatment site and limit weight bearing or load bearing. The patient is to be made fully aware and warned that the device does not replace normal healthy bone, and that the device can break or be damaged as a result of stress, activity, load bearing, or weight bearing. The patient is to be made aware of the surgical risks and possible adverse effects prior to surgery, and warned that failure to follow postoperative care instructions can cause failure of the implant and the treatment. The patient is to be advised of the need for regular postoperative follow-up examination as long as the device remains implanted. Patients that engage in stressful physical activities are to be warned that injury at or near the implant site can lead to failure of the device and/or the treatment. 15. Noncompliance with postoperative instructions could lead to failure of the device, which could require additional surgery and device removal. 16. Implant materials are subject to corrosion. Implanting metals and alloys subjects them to constant changing environments of salts, acids, and alkalis that can cause corrosion. Putting dissimilar metals and alloys in contact with each other can accelerate the corrosion process, which may contribute to fracture of implants. Every effort should be made to use compatible metals and alloys when using them in combination, i.e. screws and plates. PRECAUTIONS Do not reuse implants. While an implant may appear undamaged, previous stress may have created imperfections that would reduce the service life of the implant. Do not treat with implants that have been, even momentarily, placed in a different patient. Instruments are available to aid in the accurate implantation of internal fixation devices. Intraoperative fracture or breaking of instruments has been reported. Surgical instruments are subject to wear with normal usage. Instruments that have experienced extensive use or excessive force are susceptible to fracture. Surgical instruments should only be used for their intended purpose. Biomet Sports Medicine recommends that all instruments be regularly inspected for wear and disfigurement. If device contains MaxBraid suture, refer to manufacturer package insert for further information. POSSIBLE ADVERSE EFFECTS 1. Nonunion or delayed union, which may lead to breakage of the implant. 2. Bending or fracture of the implant.
5 3. Loosening or migration of the implant. 4. Allergic reaction to a foreign body. 5. Pain, discomfort, or abnormal sensation due to the presence of the device. 6. Nerve damage due to surgical trauma. 7. Necrosis of bone or tissue. 8. Inadequate healing. 9. Intraoperative or postoperative bone fracture and/or postoperative pain. STERILITY Biomet Sports Medicine internal fixation implants are supplied sterile by exposure to a minimum dose of 25kGy of gamma radiation or by Ethylene Oxide Gas (ETO) if device contains UHMWPE. Do not resterilize. Do not use past expiration date. Caution: Federal law (USA) restricts this device to sale, distribution or use by or on the order of a physician.
Midfoot Repair. Surgical Protocol by Stuart Miller, M.D.
 Midfoot Repair Surgical Protocol by Stuart Miller, M.D. It s small. It s strong. And it's all suture. The JuggerKnot Soft Anchor represents the next generation of suture anchor technology. The 1.4mm deployable
Midfoot Repair Surgical Protocol by Stuart Miller, M.D. It s small. It s strong. And it's all suture. The JuggerKnot Soft Anchor represents the next generation of suture anchor technology. The 1.4mm deployable
Features. The hollow core can be filled with autograft or allograft bone.
 Materials that resorb into the human body have been emerging over the past thirty years. Today, there are numerous implants available in the field of orthopedic surgery, the gold standard of ACL reconstruction
Materials that resorb into the human body have been emerging over the past thirty years. Today, there are numerous implants available in the field of orthopedic surgery, the gold standard of ACL reconstruction
Acute AC Joint Reconstruction Surgical Protocol by Eric McCarty, M.D.
 Featuring Acute AC Joint Reconstruction Surgical Protocol by Eric McCarty, M.D. ZipLoop Technology is a unique weave in which a single strand of braided polyethylene is woven through itself twice in opposite
Featuring Acute AC Joint Reconstruction Surgical Protocol by Eric McCarty, M.D. ZipLoop Technology is a unique weave in which a single strand of braided polyethylene is woven through itself twice in opposite
SureLock All-Suture Anchor System
 SureLock All-Suture Anchor System Guide for Bankart Repair Surgical Technique 2 SureLock All-Suture System Bankart Repair Surgical Technique Figure 1 *Curved and straight drill guides are available for
SureLock All-Suture Anchor System Guide for Bankart Repair Surgical Technique 2 SureLock All-Suture System Bankart Repair Surgical Technique Figure 1 *Curved and straight drill guides are available for
Femoral Fixation for ACL Reconstruction. Surgical Protocol by Mark Gittins, D.O.
 Femoral Fixation for ACL Reconstruction Surgical Protocol by Mark Gittins, D.O. Features A unique weave in which a single strand of braided polyethylene is woven through itself twice in opposite directions.
Femoral Fixation for ACL Reconstruction Surgical Protocol by Mark Gittins, D.O. Features A unique weave in which a single strand of braided polyethylene is woven through itself twice in opposite directions.
Surgical Technique. Figure 1 Figure 4. Figure 3 Figure 5. Figure 6
 Surgical Technique Figure 1 Figure 4 Figure 3 Figure 5 Drill through the femoral aimer with a graft passing pin until the pin penetrates the patient s skin. Using an acorn reamer, ream the desired length
Surgical Technique Figure 1 Figure 4 Figure 3 Figure 5 Drill through the femoral aimer with a graft passing pin until the pin penetrates the patient s skin. Using an acorn reamer, ream the desired length
Surgical Technique. Figure 1 Figure 4. Figure 3 Figure 5. Figure 6
 Surgical Technique Figure 1 Figure 4 Figure 3 Figure 5 Drill through the femoral aimer with a graft passing pin until the pin penetrates the patient s skin. Using an acorn reamer, ream the desired length
Surgical Technique Figure 1 Figure 4 Figure 3 Figure 5 Drill through the femoral aimer with a graft passing pin until the pin penetrates the patient s skin. Using an acorn reamer, ream the desired length
with ACL Reconstruction Single Tibial Tunnel Double Bundle Surgical Protocol by Eric McCarty, M.D.
 with ACL Reconstruction Single Tibial Tunnel Double Bundle Surgical Protocol by Eric McCarty, M.D. Features A unique weave in which a single strand of braided polyethylene is woven through itself twice
with ACL Reconstruction Single Tibial Tunnel Double Bundle Surgical Protocol by Eric McCarty, M.D. Features A unique weave in which a single strand of braided polyethylene is woven through itself twice
ToggleLoc. Fixation Device. Surgical Technique. Femoral Fixation for ACL Reconstruction SPORTS MEDICINE. Surgical Protocol by Mark Gittins, D.O.
 ToggleLoc Fixation Device Femoral Fixation for ACL Reconstruction Surgical Technique Surgical Protocol by Mark Gittins, D.O. SPORTS MEDICINE One Surgeon. One Patient. Over 1 million times per year, Biomet
ToggleLoc Fixation Device Femoral Fixation for ACL Reconstruction Surgical Technique Surgical Protocol by Mark Gittins, D.O. SPORTS MEDICINE One Surgeon. One Patient. Over 1 million times per year, Biomet
ToggleLoc Inline Device
 ToggleLoc Inline Device with ZipLoop Technology and TunneLoc Tibial Fixation for ACL Reconstruction Surgical Technique Surgical Protocol by Jeffrey M. Conrad, M.D. SPORTS MEDICINE One Surgeon. One Patient.
ToggleLoc Inline Device with ZipLoop Technology and TunneLoc Tibial Fixation for ACL Reconstruction Surgical Technique Surgical Protocol by Jeffrey M. Conrad, M.D. SPORTS MEDICINE One Surgeon. One Patient.
BTB ACL Reconstruction with the ToggleLoc Fixation Device with ZipLoop Technology. Surgical Technique by James R. Andrews, M.D.
 BTB ACL Reconstruction with the ToggleLoc Fixation Device with ZipLoop Technology Surgical Technique by James R. Andrews, M.D. Table of Contents Femoral Tunnel Preparation... 4 Prepare ToggleLoc Device...
BTB ACL Reconstruction with the ToggleLoc Fixation Device with ZipLoop Technology Surgical Technique by James R. Andrews, M.D. Table of Contents Femoral Tunnel Preparation... 4 Prepare ToggleLoc Device...
MCL Reconstruction Surgical Protocol by Tarek Fahl, M.D.
 MCL Reconstruction Surgical Protocol by Tarek Fahl, M.D. Features A unique weave in which a single strand of braided polyethylene is woven through itself twice in opposite directions This construct allows
MCL Reconstruction Surgical Protocol by Tarek Fahl, M.D. Features A unique weave in which a single strand of braided polyethylene is woven through itself twice in opposite directions This construct allows
Surgical Protocol written by Keith Lawhorn, M.D.
 Surgical Protocol written by Keith Lawhorn, M.D. The MaxFire Meniscal Repair System is an all-inside, all-suture meniscal repair system utilizing ZipLoop Technology. Anchors are preloaded on needle inserters,
Surgical Protocol written by Keith Lawhorn, M.D. The MaxFire Meniscal Repair System is an all-inside, all-suture meniscal repair system utilizing ZipLoop Technology. Anchors are preloaded on needle inserters,
HIP ARTHROSCOPY BROCHURE
 HIP ARTHROSCOPY BROCHURE Biomet Sports Medicine designed, developed, and launched the first all-suture anchor on the market, known as the JuggerKnot Soft Anchor. Once surgeons began utilizing this anchor
HIP ARTHROSCOPY BROCHURE Biomet Sports Medicine designed, developed, and launched the first all-suture anchor on the market, known as the JuggerKnot Soft Anchor. Once surgeons began utilizing this anchor
JuggerKnot and JuggerKnotless Soft Anchor. All-Inclusive Brochure
 JuggerKnot and JuggerKnotless Soft Anchor All-Inclusive Brochure 3 JuggerKnot and JuggerKnotless Soft Anchor All-Inclusive Brochure JuggerKnot Soft Anchor The Leader in All-Suture Anchors 1 Zimmer Biomet
JuggerKnot and JuggerKnotless Soft Anchor All-Inclusive Brochure 3 JuggerKnot and JuggerKnotless Soft Anchor All-Inclusive Brochure JuggerKnot Soft Anchor The Leader in All-Suture Anchors 1 Zimmer Biomet
with ZipLoop Technology For ACL Reconstruction ACL Reconstruction Medial Portal Surgical Protocol by Jefferey Michaelson, M.D.
 with ZipLoop Technology For ACL Reconstruction ACL Reconstruction Medial Portal Surgical Protocol by Jefferey Michaelson, M.D. Features A unique weave in which a single strand of braided polyethylene is
with ZipLoop Technology For ACL Reconstruction ACL Reconstruction Medial Portal Surgical Protocol by Jefferey Michaelson, M.D. Features A unique weave in which a single strand of braided polyethylene is
Technique Guide KISSloc Suture System
 Technique Guide KISSloc Suture System The KISSloc Suture System consists of a strong self-cinching suture assembly, a unique load-dispersing Arrow Plate and procedure specific instrumentation. Simple,
Technique Guide KISSloc Suture System The KISSloc Suture System consists of a strong self-cinching suture assembly, a unique load-dispersing Arrow Plate and procedure specific instrumentation. Simple,
JuggerKnot Soft Anchor 1.4/1.5 mm with Percutaneous Instrumentation for Low Profile/Trans-Cuff SLAP Repair
 JuggerKnot Soft Anchor 1.4/1.5 mm with Percutaneous Instrumentation for Low Profile/Trans-Cuff SLAP Repair Surgical Technique by Shabi Kahn, MD One Surgeon. One Patient. Over 1 million times per year,
JuggerKnot Soft Anchor 1.4/1.5 mm with Percutaneous Instrumentation for Low Profile/Trans-Cuff SLAP Repair Surgical Technique by Shabi Kahn, MD One Surgeon. One Patient. Over 1 million times per year,
Coracoid Bone Conserving Acromioclavicular Joint Reconstruction using ToggleLoc Device with ZipLoop Technology
 Coracoid Bone Conserving Acromioclavicular Joint Reconstruction using ToggleLoc Device with ZipLoop Technology Surgical Technique Surgical Protocol by Peter J. Evans, MD, PhD SPORTS MEDICINE One Surgeon.
Coracoid Bone Conserving Acromioclavicular Joint Reconstruction using ToggleLoc Device with ZipLoop Technology Surgical Technique Surgical Protocol by Peter J. Evans, MD, PhD SPORTS MEDICINE One Surgeon.
Medial Portal ACL Reconstruction
 Medial Portal ACL Reconstruction with Precision Flexible Reaming Instrumentation Surgical Technique by William Prickett, M.D. and Gregory J. Loren, M.D., FAAOS Table of Contents Introduction... 2 Portals...
Medial Portal ACL Reconstruction with Precision Flexible Reaming Instrumentation Surgical Technique by William Prickett, M.D. and Gregory J. Loren, M.D., FAAOS Table of Contents Introduction... 2 Portals...
ComposiTCP Anchor with BroadBand Tape
 ComposiTCP Anchor with BroadBand Tape featuring Medial Row Knot Tying Surgical Technique 2 ComposiTCP Anchor with BroadBand Tape Surgical Technique Figure 1 Figure 2 Step 1 Place the ComposiTCP punch/tap
ComposiTCP Anchor with BroadBand Tape featuring Medial Row Knot Tying Surgical Technique 2 ComposiTCP Anchor with BroadBand Tape Surgical Technique Figure 1 Figure 2 Step 1 Place the ComposiTCP punch/tap
ACL Reconstruction Single Tibial Tunnel Double Bundle
 ACL Reconstruction Single Tibial Tunnel Double Bundle with the ToggleLoc Fixation Device with ZipLoop Technology Surgical Technique by Eric McCarty, M.D. Table of Contents Portal Position and Sizing of
ACL Reconstruction Single Tibial Tunnel Double Bundle with the ToggleLoc Fixation Device with ZipLoop Technology Surgical Technique by Eric McCarty, M.D. Table of Contents Portal Position and Sizing of
Revision A Date:
 Biomet Trauma 56 East Bell Drive P.O. Box 587 Warsaw, Indiana 46581 USA 01-50-4053 Revision A Date: 2016-06 Biomet Unite3D Bridge with OsseoTi Technology ATTENTION OPERATING SURGEON DESCRIPTION Biomet
Biomet Trauma 56 East Bell Drive P.O. Box 587 Warsaw, Indiana 46581 USA 01-50-4053 Revision A Date: 2016-06 Biomet Unite3D Bridge with OsseoTi Technology ATTENTION OPERATING SURGEON DESCRIPTION Biomet
JuggerKnot Soft Anchor 1.5 mm with Percutaneous Instrumentation for Low Profile/Trans-Cuff PASTA Repair
 JuggerKnot Soft Anchor 1.5 mm with Percutaneous Instrumentation for Low Profile/Trans-Cuff PASTA Repair Surgical Technique by Shabi Kahn, MD One Surgeon. One Patient. Over 1 million times per year, Biomet
JuggerKnot Soft Anchor 1.5 mm with Percutaneous Instrumentation for Low Profile/Trans-Cuff PASTA Repair Surgical Technique by Shabi Kahn, MD One Surgeon. One Patient. Over 1 million times per year, Biomet
Surgical Technique. Suture. Insertion Rod. U-Guide. Grommet Swing Arm. Figure 1. Figure 2. Suture
 This brochure is presented to demonstrate the surgical technique utilized by Robert Frederick, M.D. Arthrotek, as the manufacturer of this device, does not practice medicine and does not recommend this
This brochure is presented to demonstrate the surgical technique utilized by Robert Frederick, M.D. Arthrotek, as the manufacturer of this device, does not practice medicine and does not recommend this
with ACL Reconstruction Single Tibial Tunnel Double Bundle Surgical Protocol by Eric McCarty, M.D.
 with ACL Reconstruction Single Tibial Tunnel Double Bundle Surgical Protocol by Eric McCarty, M.D. Features A unique weave in which a single strand of braided polyethylene is woven through itself twice
with ACL Reconstruction Single Tibial Tunnel Double Bundle Surgical Protocol by Eric McCarty, M.D. Features A unique weave in which a single strand of braided polyethylene is woven through itself twice
Surgical Protocol by E. Lyle Cain, M.D. and Jeffrey R. Dugas, M.D. of the American Sports Medicine Institute
 Surgical Protocol by E. Lyle Cain, M.D. and Jeffrey R. Dugas, M.D. of the American Sports Medicine Institute Indications Shoulder Indications Bankart repair SLAP lesion repair Acromioclavicular (AC) separation
Surgical Protocol by E. Lyle Cain, M.D. and Jeffrey R. Dugas, M.D. of the American Sports Medicine Institute Indications Shoulder Indications Bankart repair SLAP lesion repair Acromioclavicular (AC) separation
ACL Reconstruction Medial Portal
 ACL Reconstruction Medial Portal with ToggleLoc Fixation Device with ZipLoop Technology and ComposiTCP Interference Screw Surgical Technique by Jefferey Michaelson, M.D. Table of Contents Tunnel Preparation...
ACL Reconstruction Medial Portal with ToggleLoc Fixation Device with ZipLoop Technology and ComposiTCP Interference Screw Surgical Technique by Jefferey Michaelson, M.D. Table of Contents Tunnel Preparation...
Hip Arthroscopy. All Inclusive Brochure
 Hip Arthroscopy All Inclusive Brochure Zimmer Biomet Sports Medicine designed, developed, and launched the first allsuture anchor on the market, known as the JuggerKnot Soft Anchor. Once surgeons began
Hip Arthroscopy All Inclusive Brochure Zimmer Biomet Sports Medicine designed, developed, and launched the first allsuture anchor on the market, known as the JuggerKnot Soft Anchor. Once surgeons began
Improved access, ultra-low displacement 1 and the highest fixation strength 1. Q-FIX All-Suture Anchor
 Improved access, ultra-low displacement 1 and the highest fixation strength 1 All-Suture Anchor All-Suture Anchors The All-Suture Anchor is the next generation of all-suture anchor technology, providing
Improved access, ultra-low displacement 1 and the highest fixation strength 1 All-Suture Anchor All-Suture Anchors The All-Suture Anchor is the next generation of all-suture anchor technology, providing
Improved access, ultra-low displacement 1 and the highest fixation strength 1. Q-FIX All-Suture Anchor
 Improved access, ultra-low displacement 1 and the highest fixation strength 1 Q-FIX All-Suture Anchor Q-FIX All-Suture Anchors The Q-FIX All-Suture Anchor is the next generation of all-suture anchor technology,
Improved access, ultra-low displacement 1 and the highest fixation strength 1 Q-FIX All-Suture Anchor Q-FIX All-Suture Anchors The Q-FIX All-Suture Anchor is the next generation of all-suture anchor technology,
Lateral Ankle Ligamentous Instability. Surgical Technique by Jeffrey Nacht, M.D.
 Lateral Ankle Ligamentous Instability Surgical Technique by Jeffrey Nacht, M.D. It s small. It s strong. And it's all suture. The JuggerKnot Soft Anchor represents the next generation of suture anchor
Lateral Ankle Ligamentous Instability Surgical Technique by Jeffrey Nacht, M.D. It s small. It s strong. And it's all suture. The JuggerKnot Soft Anchor represents the next generation of suture anchor
Pinit Plate Small Bone Fusion System Bone Plate & Screw System
 Pinit Plate Small Bone Fusion System Bone Plate & Screw System Description The Pinit Plate Small Bone Fusion System consists of 2-hole bone plates made available in three length options and two thickness
Pinit Plate Small Bone Fusion System Bone Plate & Screw System Description The Pinit Plate Small Bone Fusion System consists of 2-hole bone plates made available in three length options and two thickness
Ankle Stabilization Surgical Technique using the Split Peroneus Longus Tendon. Surgical Protocol by Chad Brady, D.P.M.
 Ankle Stabilization Surgical Technique using the Split Peroneus Longus Tendon Surgical Protocol by Chad Brady, D.P.M. Bite does matter! The Rattler Interference Screw is designed specifically for hand
Ankle Stabilization Surgical Technique using the Split Peroneus Longus Tendon Surgical Protocol by Chad Brady, D.P.M. Bite does matter! The Rattler Interference Screw is designed specifically for hand
Peanut Growth Control Plating System
 Surgical Technique Peanut Growth Control Plating System An Innovative Approach To Hemi-Epiphysiodesis Provides controlled growth of healthy epiphysis, restoring proper anatomic alignment Advanced instrumentation
Surgical Technique Peanut Growth Control Plating System An Innovative Approach To Hemi-Epiphysiodesis Provides controlled growth of healthy epiphysis, restoring proper anatomic alignment Advanced instrumentation
Arthroscopic PCL Reconstruction
 Arthroscopic PCL Reconstruction Using Soft Tissue Graft and TunneLoc Tibial Fixation with ToggleLoc Fixation Device with ZipLoop Technology Surgical Technique by Mark Ganjianpour, M.D. Table of Contents
Arthroscopic PCL Reconstruction Using Soft Tissue Graft and TunneLoc Tibial Fixation with ToggleLoc Fixation Device with ZipLoop Technology Surgical Technique by Mark Ganjianpour, M.D. Table of Contents
ToggleLoc Fixation Device with ZipLoop Technology
 ToggleLoc Fixation Device with ZipLoop Technology Arthroscopic PCL Reconstruction Using Soft Tissue Graft and TunneLoc Tibial Fixation Surgical Technique Surgical Protocol by Mark Ganjianpour, M.D. One
ToggleLoc Fixation Device with ZipLoop Technology Arthroscopic PCL Reconstruction Using Soft Tissue Graft and TunneLoc Tibial Fixation Surgical Technique Surgical Protocol by Mark Ganjianpour, M.D. One
Technique Guide Small Bone Fusion System
 Technique Guide Small Bone Fusion System The Pinit Plate Small Bone Fusion System is a super low profile, modular bone plate and screw system designed to stabilize a bunionectomy with a medial to lateral
Technique Guide Small Bone Fusion System The Pinit Plate Small Bone Fusion System is a super low profile, modular bone plate and screw system designed to stabilize a bunionectomy with a medial to lateral
Arthroscopic and Mini-Open Rotator Cuff Repair. using JuggerKnot Soft Anchor 2.9 mm with ALLthread Knotless PEEK-Optima Anchor Surgical Technique
 Arthroscopic and Mini-Open Rotator Cuff Repair using JuggerKnot Soft Anchor 2.9 mm with ALLthread Knotless PEEK-Optima Anchor Surgical Technique It s small. It s strong. And it's all suture. The JuggerKnot
Arthroscopic and Mini-Open Rotator Cuff Repair using JuggerKnot Soft Anchor 2.9 mm with ALLthread Knotless PEEK-Optima Anchor Surgical Technique It s small. It s strong. And it's all suture. The JuggerKnot
Arthroscopic and Mini-Open Rotator Cuff Repair
 Arthroscopic and Mini-Open Rotator Cuff Repair with the JuggerKnot Soft Anchor - 2.9 mm with ALLthread Knotless PEEK-Optima Anchor Surgical Technique Table of Contents Arthroscopic Repair... 4 Arthroscopic
Arthroscopic and Mini-Open Rotator Cuff Repair with the JuggerKnot Soft Anchor - 2.9 mm with ALLthread Knotless PEEK-Optima Anchor Surgical Technique Table of Contents Arthroscopic Repair... 4 Arthroscopic
Lapidus Arthrodesis System Instructions for Use
 Lapidus Arthrodesis System Instructions for Use Description The AlignMATE Lapidus Arthrodesis System consists of bone plates and bone screws (locking, non-locking and interfragmentary), which are intended
Lapidus Arthrodesis System Instructions for Use Description The AlignMATE Lapidus Arthrodesis System consists of bone plates and bone screws (locking, non-locking and interfragmentary), which are intended
JuggerKnot Soft Anchor 1.0 mm Mini. Scapholunate Ligament Repair/Reconstruction. Brochure and Surgical Technique
 JuggerKnot Soft Anchor 1.0 mm Mini Scapholunate Ligament Repair/Reconstruction Brochure and Surgical Technique One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide
JuggerKnot Soft Anchor 1.0 mm Mini Scapholunate Ligament Repair/Reconstruction Brochure and Surgical Technique One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide
JuggerKnot Soft Anchor 1.0 mm Mini
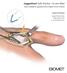 JuggerKnot Soft Anchor 1.0 mm Mini Ulnar Collateral Ligament (UCL) Repair of the Thumb Surgical Technique Surgical Protocols by Mark Rekant, M.D. A. Lee Osterman, M.D. One Surgeon. One Patient. Over 1
JuggerKnot Soft Anchor 1.0 mm Mini Ulnar Collateral Ligament (UCL) Repair of the Thumb Surgical Technique Surgical Protocols by Mark Rekant, M.D. A. Lee Osterman, M.D. One Surgeon. One Patient. Over 1
JuggerLoc Bone-to-Bone System for Ankle Syndesmosis Fixation. Surgical Technique
 JuggerLoc Bone-to-Bone System for Ankle Syndesmosis Fixation Surgical Technique Table of Contents Position and Preparation... 2 Incision... 2 Fracture Reduction... 2 Drill Fibula and Tibia... 3 JuggerLoc
JuggerLoc Bone-to-Bone System for Ankle Syndesmosis Fixation Surgical Technique Table of Contents Position and Preparation... 2 Incision... 2 Fracture Reduction... 2 Drill Fibula and Tibia... 3 JuggerLoc
For the Attention of the Operating Surgeon: IMPORTANT INFORMATION ON THE MATRIXRIB FIXATION SYSTEM
 For the Attention of the Operating Surgeon: IMPORTANT INFORMATION ON THE MATRIXRIB FIXATION SYSTEM 10/16 GP2685-E-CAN DESCRIPTION The MatrixRIB Fixation System consists of locking plates, locking screws,
For the Attention of the Operating Surgeon: IMPORTANT INFORMATION ON THE MATRIXRIB FIXATION SYSTEM 10/16 GP2685-E-CAN DESCRIPTION The MatrixRIB Fixation System consists of locking plates, locking screws,
Positioning Sleeve Surgical Technique
 Positioning Sleeve Surgical Technique Surgical Technique The Comprehensive Fracture System offers a unique tool to evaluate and re-establish humeral height, which can be critical to restoring proper biomechanics
Positioning Sleeve Surgical Technique Surgical Technique The Comprehensive Fracture System offers a unique tool to evaluate and re-establish humeral height, which can be critical to restoring proper biomechanics
WristMotion Wrist Hemiarthroplasty System Instructions for Use
 WristMotion Wrist Hemiarthroplasty System Instructions for Use Description The Arthrosurface WristMotion Wrist Hemiarthroplasty System consists of a contoured capitate articular implant designed to articulate
WristMotion Wrist Hemiarthroplasty System Instructions for Use Description The Arthrosurface WristMotion Wrist Hemiarthroplasty System consists of a contoured capitate articular implant designed to articulate
Flexible Fragment Fixation. Surgical Technique
 Flexible Fragment Fixation Surgical Technique 2 F 3 Flexible Fragment Fixation The F 3 Fragment Plating System offers low profile, yet strong fixation in a locked plating construct that can be contoured
Flexible Fragment Fixation Surgical Technique 2 F 3 Flexible Fragment Fixation The F 3 Fragment Plating System offers low profile, yet strong fixation in a locked plating construct that can be contoured
with Medial Patellofemoral Ligament (MPFL) Reconstruction Surgical Protocol by Ronald Navarro, M.D.
 with Medial Patellofemoral Ligament (MPFL) Reconstruction Surgical Protocol by Ronald Navarro, M.D. Features A unique weave in which a single strand of braided polyethylene is woven through itself twice
with Medial Patellofemoral Ligament (MPFL) Reconstruction Surgical Protocol by Ronald Navarro, M.D. Features A unique weave in which a single strand of braided polyethylene is woven through itself twice
ACP1 CERVICAL PLATE SPINAL SYSTEM SURGICAL TECHNIQUE GUIDE II.
 I. ACP1 CERVICAL PLATE II. SPINAL SYSTEM SURGICAL TECHNIQUE GUIDE I. Introduction The Gold Standard Orthopaedics, LLC ACP1 Spinal System was designed with surgeons to incorporate strength, functionality,
I. ACP1 CERVICAL PLATE II. SPINAL SYSTEM SURGICAL TECHNIQUE GUIDE I. Introduction The Gold Standard Orthopaedics, LLC ACP1 Spinal System was designed with surgeons to incorporate strength, functionality,
ENDO-FUSE INTRA-OSSEOUS FUSION SYSTEM The following languages are included in this packet:
 ENDO-FUSE INTRA-OSSEOUS FUSION SYSTEM 137900-5 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) - Chinese
ENDO-FUSE INTRA-OSSEOUS FUSION SYSTEM 137900-5 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) - Chinese
BIOLOX delta Option Ceramic Femoral Head System
 BIOLOX delta Option Ceramic Femoral Head System Product Features and Instructions for Use Knees Hips Extremities Cement and Accessories PMI Technology Product Features BIOLOX delta ceramic heads are composed
BIOLOX delta Option Ceramic Femoral Head System Product Features and Instructions for Use Knees Hips Extremities Cement and Accessories PMI Technology Product Features BIOLOX delta ceramic heads are composed
The AperFix II System
 The AperFix II System A Complete Anatomic Solution Transtibial Surgical Technique 2 AperFix II System Transtibial Surgical Technique Figure 1 A Complete Anatomic Solution The Cayenne Medical AperFix and
The AperFix II System A Complete Anatomic Solution Transtibial Surgical Technique 2 AperFix II System Transtibial Surgical Technique Figure 1 A Complete Anatomic Solution The Cayenne Medical AperFix and
Visit Linvatec.com today to learn more. Surgical Technique: Sequential Meniscal Running Stitch
 ORDERING INFORMATION Sequent Meniscal Repair System MR004S MR007S MR004C MR007C SC047D Straight Needle, 4 Implants Straight Needle, 7 Implants Curved Needle, 4 Implants Curved Needle, 7 implants Sequent
ORDERING INFORMATION Sequent Meniscal Repair System MR004S MR007S MR004C MR007C SC047D Straight Needle, 4 Implants Straight Needle, 7 Implants Curved Needle, 4 Implants Curved Needle, 7 implants Sequent
Metallic Internal Fixation Devices The following languages are included in this packet:
 Metallic Internal Fixation Devices 150848-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese
Metallic Internal Fixation Devices 150848-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese
Document No Rev. B Pg. 1 of 5 Approvals / Date Title: INSTRUCTIONS FOR USE DJO SURGICAL MODULAR REVISION HIP SYSTEM AND ACETABULAR CAGE
 Document No. 0400-0205 Rev. B Pg. 1 of 5 Approvals / Date Title: INSTRUCTIONS FOR USE DJO SURGICAL MODULAR REVISION HIP SYSTEM AND ACETABULAR CAGE Revision ECO Date ECO Summary of Changes Deann Rector
Document No. 0400-0205 Rev. B Pg. 1 of 5 Approvals / Date Title: INSTRUCTIONS FOR USE DJO SURGICAL MODULAR REVISION HIP SYSTEM AND ACETABULAR CAGE Revision ECO Date ECO Summary of Changes Deann Rector
Talar Dome System Surgical Technique
 Talar Dome System Surgical Technique CAP TALAR DOME RESURFACING HEMIARTHROPLASTY IMPLANT Surgical Technique Guide Description The HemiCAP Contoured Articular Prosthetic incorporates an articular resurfacing
Talar Dome System Surgical Technique CAP TALAR DOME RESURFACING HEMIARTHROPLASTY IMPLANT Surgical Technique Guide Description The HemiCAP Contoured Articular Prosthetic incorporates an articular resurfacing
EVOLVE TRIAD BONE SCREWS
 EN EVOLVE TRIAD BONE SCREWS 146886-1 English (en) The following languages are included in this packet: For additional languages, visit our website www.wmt.com. Then click on the Prescribing Information
EN EVOLVE TRIAD BONE SCREWS 146886-1 English (en) The following languages are included in this packet: For additional languages, visit our website www.wmt.com. Then click on the Prescribing Information
BIOLOX delta Option Ceramic Femoral Head System. Product Features and Instructions for Use
 BIOLOX delta Option Ceramic Femoral Head System Product Features and Instructions for Use Product Features BIOLOX delta ceramic heads are composed of 75% aluminum oxide and approximately 25% zirconia.
BIOLOX delta Option Ceramic Femoral Head System Product Features and Instructions for Use Product Features BIOLOX delta ceramic heads are composed of 75% aluminum oxide and approximately 25% zirconia.
Scan-Anchor....Not just another soft tissue bone anchor, but a new reference! Presentation booklet
 Scan-Anchor...Not just another soft tissue bone anchor, but a new reference! Presentation booklet Pre-Loaded Soft Tissue Bone Anchors A little history about soft tissue anchors. We have come a long way
Scan-Anchor...Not just another soft tissue bone anchor, but a new reference! Presentation booklet Pre-Loaded Soft Tissue Bone Anchors A little history about soft tissue anchors. We have come a long way
Customized Cranial and Craniofacial Implants. Instructions For Use. 7000revD_Instructions_For_Use. Page 1 of 8
 Customized Cranial and Craniofacial Implants Instructions For Use Page 1 of 8 Table of Contents INDICATIONS... 3 DESCRIPTION OF DEVICE... 3 CONDITIONS OF USE... 3 CONTRAINDICATIONS... 3 KELYNIAM GLOBAL
Customized Cranial and Craniofacial Implants Instructions For Use Page 1 of 8 Table of Contents INDICATIONS... 3 DESCRIPTION OF DEVICE... 3 CONDITIONS OF USE... 3 CONTRAINDICATIONS... 3 KELYNIAM GLOBAL
MetaFix Ludloff Plate
 Merete MetaFix Ludloff Plate Low Profile Locking Bone Plate System Surgical Technique and Ordering Information - Content - Content 1. Description.................................................. 3 2.
Merete MetaFix Ludloff Plate Low Profile Locking Bone Plate System Surgical Technique and Ordering Information - Content - Content 1. Description.................................................. 3 2.
IMPORTANT MEDICAL INFORMATION Advanced Orthopaedic Solutions INTRAMEDULLARY NAILS Warnings and Precautions (SINGLE USE ONLY)
 IMPORTANT MEDICAL INFORMATION Advanced Orthopaedic Solutions INTRAMEDULLARY NAILS Warnings and Precautions (SINGLE USE ONLY) IMPORTANT NOTE Intramedullary nails provide an alternative to open reduction
IMPORTANT MEDICAL INFORMATION Advanced Orthopaedic Solutions INTRAMEDULLARY NAILS Warnings and Precautions (SINGLE USE ONLY) IMPORTANT NOTE Intramedullary nails provide an alternative to open reduction
QIN4432TRICREV01. IMPORTANT PRODUCT INFORMATION FOR TRITANIUM C Cervical Cage. Version 1 1 STERILE PRODUCT
 IMPORTANT PRODUCT INFORMATION FOR TRITANIUM C Cervical Cage STERILE PRODUCT DESCRIPTION The Stryker Spine Tritanium C Anterior Cervical Cage is a hollow, ring shaped titanium alloy (Ti-6Al- 4V) interbody
IMPORTANT PRODUCT INFORMATION FOR TRITANIUM C Cervical Cage STERILE PRODUCT DESCRIPTION The Stryker Spine Tritanium C Anterior Cervical Cage is a hollow, ring shaped titanium alloy (Ti-6Al- 4V) interbody
Apache Cervical Interbody Fusion Device. Surgical Technique. Page of 13. LC-005 Rev F
 LC-005 Rev F Apache Cervical Interbody Fusion Device Page of 13 Surgical Technique INDICATIONS: When used as an intervertebral body fusion device, the Genesys Spine Interbody Fusion System is indicated
LC-005 Rev F Apache Cervical Interbody Fusion Device Page of 13 Surgical Technique INDICATIONS: When used as an intervertebral body fusion device, the Genesys Spine Interbody Fusion System is indicated
For use by an Accredited Orthopaedic Surgeon only
 Page 1 of 5 For use by an Accredited Orthopaedic Surgeon only 1. Purpose: External fixators are intended to aid in surgical stabilization following operative procedures to treat fractures, enable correction
Page 1 of 5 For use by an Accredited Orthopaedic Surgeon only 1. Purpose: External fixators are intended to aid in surgical stabilization following operative procedures to treat fractures, enable correction
D. The following factors are of extreme importance to the eventual success of the procedure.
 Reprocessed by Stryker Sustainability Solutions 1810 W. Drake Drive Tempe, AZ 85283 sustainability.stryker.com phone: 888.888.3433 English REPROCESSED EXTERNAL FIXATION DEVICES ATTENTION OPERATING SURGEON
Reprocessed by Stryker Sustainability Solutions 1810 W. Drake Drive Tempe, AZ 85283 sustainability.stryker.com phone: 888.888.3433 English REPROCESSED EXTERNAL FIXATION DEVICES ATTENTION OPERATING SURGEON
Duraloc CONSTRAINED LINER
 SURGICAL TECHNIQUE Duraloc CONSTRAINED LINER A COMPREHENSIVE ACETABULAR REVISION SYSTEM DURALOC CONSTRAINED LINER Introduction Dislocation is the most common postoperative complication in total hip reconstruction.
SURGICAL TECHNIQUE Duraloc CONSTRAINED LINER A COMPREHENSIVE ACETABULAR REVISION SYSTEM DURALOC CONSTRAINED LINER Introduction Dislocation is the most common postoperative complication in total hip reconstruction.
METAL HEMI SYSTEM
 METAL HEMI SYSTEM 152206-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese (sch) Türkçe
METAL HEMI SYSTEM 152206-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese (sch) Türkçe
VTI INTERLINK PEDICLE SCREW SYSTEM
 VTI INTERLINK PEDICLE SCREW SYSTEM SURGICAL TECHNIQUE FORWARD THINKING FOR THE BACK. DEVICE DESCRIPTION The VTI InterLink Pedicle Screw System is comprised of polyaxial pedicle screws in various diameters
VTI INTERLINK PEDICLE SCREW SYSTEM SURGICAL TECHNIQUE FORWARD THINKING FOR THE BACK. DEVICE DESCRIPTION The VTI InterLink Pedicle Screw System is comprised of polyaxial pedicle screws in various diameters
OPERATIVE TECHNIQUE. CONSTRUX Mini PTC. Mini PTC Spacer System
 OPERATIVE TECHNIQUE CONSTRUX Mini PTC Mini PTC Spacer System TABLE OF CONTENTS Introduction 1 Operative Technique 2 Part Numbers 6 Indications For Use 7 INTRODUCTION 1 INTRODUCTION The CONSTRUX Mini PTC
OPERATIVE TECHNIQUE CONSTRUX Mini PTC Mini PTC Spacer System TABLE OF CONTENTS Introduction 1 Operative Technique 2 Part Numbers 6 Indications For Use 7 INTRODUCTION 1 INTRODUCTION The CONSTRUX Mini PTC
Gallery Laminoplasty Spine System
 Surgical Technique Gallery Laminoplasty Spine System A smart, simple to use system with intuitive design features. Smart Plate Design Three hole, cobra head design Plates with hook or standard plates available
Surgical Technique Gallery Laminoplasty Spine System A smart, simple to use system with intuitive design features. Smart Plate Design Three hole, cobra head design Plates with hook or standard plates available
Zimmer Anterior Buttress Plate System. Surgical Technique
 Zimmer Anterior Buttress Plate System Surgical Technique 2 Zimmer Anterior Buttress Plate System Surgical Technique Zimmer Anterior Buttress Plate System Surgical Technique Description, Indications & Contraindications...
Zimmer Anterior Buttress Plate System Surgical Technique 2 Zimmer Anterior Buttress Plate System Surgical Technique Zimmer Anterior Buttress Plate System Surgical Technique Description, Indications & Contraindications...
BONE GRAFT WASHER _Rev A IMPORTANT INFORMATION ON THE BONE GRAFT WASHER
 0381080_Rev A BONE GRAFT WASHER IMPORTANT INFORMATION ON THE BONE GRAFT WASHER 03/2008 Medtronic Sofamor Danek USA, Inc. 1800 Pyramid Place Memphis, Tennessee 38132 Telephone: 800-933-2625 (in U.S.A.)
0381080_Rev A BONE GRAFT WASHER IMPORTANT INFORMATION ON THE BONE GRAFT WASHER 03/2008 Medtronic Sofamor Danek USA, Inc. 1800 Pyramid Place Memphis, Tennessee 38132 Telephone: 800-933-2625 (in U.S.A.)
Technique Guide Lapidus Arthrodesis System
 Technique Guide Lapidus Arthrodesis System HVA Angle IMA Angle The AlignMate Lapidus Arthrodesis System features low-profile, anatomically pre-contoured Bone Plates with either a combination of locking
Technique Guide Lapidus Arthrodesis System HVA Angle IMA Angle The AlignMate Lapidus Arthrodesis System features low-profile, anatomically pre-contoured Bone Plates with either a combination of locking
EVOLVE TRIAD SYSTEM
 EN EVOLVE TRIAD SYSTEM 146884-2 English (en) The following languages are included in this packet: For additional languages, visit our website www.wmt.com. Then click on the Prescribing Information option.
EN EVOLVE TRIAD SYSTEM 146884-2 English (en) The following languages are included in this packet: For additional languages, visit our website www.wmt.com. Then click on the Prescribing Information option.
TM TM Surgical Technique
 TM TM Surgical Technique TABLE OF CONTENTS Reli SP Spinous Plating System Overview Device Description Implant Features Indications Instruments Access Instruments Preparation Instruments Insertion Instruments
TM TM Surgical Technique TABLE OF CONTENTS Reli SP Spinous Plating System Overview Device Description Implant Features Indications Instruments Access Instruments Preparation Instruments Insertion Instruments
Tibial Fixation. with TunneLoc Device. Surgical Technique by Mark J. Albritton, M.D. and Sherwin Ho, M.D.
 Tibial Fixation with TunneLoc Device Surgical Technique by Mark J. Albritton, M.D. and Sherwin Ho, M.D. Table of Contents Surgical Technique... 4 Ordering Information... 11 Indications For Use... 12 Contraindications...
Tibial Fixation with TunneLoc Device Surgical Technique by Mark J. Albritton, M.D. and Sherwin Ho, M.D. Table of Contents Surgical Technique... 4 Ordering Information... 11 Indications For Use... 12 Contraindications...
SURGICAL TECHNIQUE THE TENDON ANCHOR SYSTEM
 SURGICAL TECHNIQUE THE TENDON ANCHOR SYSTEM TABLE OF CONTENTS Introduction PAGE 1 Features and Benefits PAGE 1-2 Applications PAGE 3 Ordering Information PAGE 4 Indications & Contra-indications PAGE 5
SURGICAL TECHNIQUE THE TENDON ANCHOR SYSTEM TABLE OF CONTENTS Introduction PAGE 1 Features and Benefits PAGE 1-2 Applications PAGE 3 Ordering Information PAGE 4 Indications & Contra-indications PAGE 5
Surgical Technique. Apache Anterior Lumbar Interbody Fusion
 Surgical Technique Apache Anterior Lumbar Interbody Fusion 2 Table of Contents Page Preoperative Planning 4 Patient Positioning 4 Disc and Endplate Preparation 4 Distraction/Size Selection 5 Implantation
Surgical Technique Apache Anterior Lumbar Interbody Fusion 2 Table of Contents Page Preoperative Planning 4 Patient Positioning 4 Disc and Endplate Preparation 4 Distraction/Size Selection 5 Implantation
Aesculap CeSpace TM PEEK and CeSpace TM XP Spinal Implant System Instructions for Use
 Aesculap CeSpace TM PEEK and CeSpace TM XP Spinal Implant System Instructions for Use Page 1 of 5 Indications for use: When used as a Vertebral Body Replacement Device: The CeSpace PEEK and CeSpace XP
Aesculap CeSpace TM PEEK and CeSpace TM XP Spinal Implant System Instructions for Use Page 1 of 5 Indications for use: When used as a Vertebral Body Replacement Device: The CeSpace PEEK and CeSpace XP
DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY
 0086 Instructions for Use RCS Anterior Buttress Plate System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY There
0086 Instructions for Use RCS Anterior Buttress Plate System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY There
EXTERNAL FIXATION SYSTEMS The following languages are included in this packet:
 EXTERNAL FIXATION SYSTEMS 150860-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese (sch)
EXTERNAL FIXATION SYSTEMS 150860-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese (sch)
Technique Guide Wrist Hemiarthroplasty System
 Technique Guide Wrist Hemiarthroplasty System The WristMotion TM Wrist Hemiarthroplasty System restores mobility and maintains native biomechanics using a dual curvature HemiCAPITATE implant that locks
Technique Guide Wrist Hemiarthroplasty System The WristMotion TM Wrist Hemiarthroplasty System restores mobility and maintains native biomechanics using a dual curvature HemiCAPITATE implant that locks
SIDEKICK EZ FRAME EXTERNAL FIXATION SYSTEM The following languages are included in this packet:
 EN SIDEKICK EZ FRAME EXTERNAL FIXATION SYSTEM 149369-0 English (en) The following languages are included in this packet: For additional languages, visit our website www.wmt.com. Then click on the Prescribing
EN SIDEKICK EZ FRAME EXTERNAL FIXATION SYSTEM 149369-0 English (en) The following languages are included in this packet: For additional languages, visit our website www.wmt.com. Then click on the Prescribing
Description Materials Indications Patient selection factors to be considered include: Contraindications Absolute contraindications include:
 Description The Patello-Femoral Wave Arthroplasty Systems incorporate a distal femoral trochlear surface articular component that mates to a taper post via a taper interlock, and an all-polyethylene patella
Description The Patello-Femoral Wave Arthroplasty Systems incorporate a distal femoral trochlear surface articular component that mates to a taper post via a taper interlock, and an all-polyethylene patella
DARCO MIS FOREFOOT SCREW
 DARCO MIS FOREFOOT SCREW 140418-1 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) - Chinese (sch)
DARCO MIS FOREFOOT SCREW 140418-1 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) - Chinese (sch)
3.0 mm Cannulated Screw System. Surgical Technique
 3.0 mm Cannulated Screw System Surgical Technique 3 3.0 mm Cannulated Screw System Surgical Technique 3.0 mm Cannulated Screw System The 3.0 mm Cannulated Screw System is part of a series of cannulated
3.0 mm Cannulated Screw System Surgical Technique 3 3.0 mm Cannulated Screw System Surgical Technique 3.0 mm Cannulated Screw System The 3.0 mm Cannulated Screw System is part of a series of cannulated
TC-PLUS Primary Knee IMPORTANT MEDICAL INFORMATION SPECIAL NOTE. This Package Insert is for product distributed in the US only.
 TC-PLUS Primary Knee IMPORTANT MEDICAL INFORMATION SPECIAL NOTE This Package Insert is for product distributed in the US only. The component material is provided on the outside carton label. Components
TC-PLUS Primary Knee IMPORTANT MEDICAL INFORMATION SPECIAL NOTE This Package Insert is for product distributed in the US only. The component material is provided on the outside carton label. Components
RAPIDSORB RAPID ReSORBABLe CRANIAL CLAMP
 RAPIDSORB RAPID ReSORBABLe CRANIAL CLAMP Resorbable fixation of cranial bone flaps SURGICAL TeCHNIQUe RAPIDSORB Rapid Resorbable CRANIAL CLAMP Indication DePuy Synthes Companies of Johnson & Johnson RAPIDSORB
RAPIDSORB RAPID ReSORBABLe CRANIAL CLAMP Resorbable fixation of cranial bone flaps SURGICAL TeCHNIQUe RAPIDSORB Rapid Resorbable CRANIAL CLAMP Indication DePuy Synthes Companies of Johnson & Johnson RAPIDSORB
SYNPOR POROUS POLYETHYLENE IMPLANTS. For craniofacial and orbital augmentation and reconstruction
 SYNPOR POROUS POLYETHYLENE IMPLANTS For craniofacial and orbital augmentation and reconstruction SURGICAL TECHNIQUE TABLE OF CONTENTS INTRODUCTION SYNPOR Porous Polyethylene Implants 2 Indications and
SYNPOR POROUS POLYETHYLENE IMPLANTS For craniofacial and orbital augmentation and reconstruction SURGICAL TECHNIQUE TABLE OF CONTENTS INTRODUCTION SYNPOR Porous Polyethylene Implants 2 Indications and
nvp Posterior Lumbar Interbody Fusion System
 nvp Posterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
nvp Posterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
nvt Transforaminal Lumbar Interbody Fusion System
 nvt Transforaminal Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine
nvt Transforaminal Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine
nva Anterior Lumbar Interbody Fusion System
 nva Anterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
nva Anterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
GENERAL TECHNIQUE GUIDE
 GENERAL TECHNIQUE GUIDE PRODUCT OVERVIEW The Eclipse Soft Tissue Anchor employs MedShape s shape memory PEEK Altera technology for soft tissue fixation procedures in the shoulder, elbow, knee, hand & wrist,
GENERAL TECHNIQUE GUIDE PRODUCT OVERVIEW The Eclipse Soft Tissue Anchor employs MedShape s shape memory PEEK Altera technology for soft tissue fixation procedures in the shoulder, elbow, knee, hand & wrist,
Headless Compession Screw 2.5 / 3.0
 SURGICAL TECHNIQUE Headless Compession Screw 2.5 / 3.0 Titanium or Stainless Steel Cannulated Headless Design Multiple Thread Options Torx Driver Sterile and Non-Sterile Options Simple Instrumentation
SURGICAL TECHNIQUE Headless Compession Screw 2.5 / 3.0 Titanium or Stainless Steel Cannulated Headless Design Multiple Thread Options Torx Driver Sterile and Non-Sterile Options Simple Instrumentation
DISTAL RADIUS. Instructions for Use
 DISTAL RADIUS Instructions for Use CAUTION: FEDERAL LAW RESTRICTS THESE DEVICES TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Table of Contents 1. INTENDED USE... 3 2. DEVICE DESCRIPTION... 3 3. METHOD OF
DISTAL RADIUS Instructions for Use CAUTION: FEDERAL LAW RESTRICTS THESE DEVICES TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Table of Contents 1. INTENDED USE... 3 2. DEVICE DESCRIPTION... 3 3. METHOD OF
Headless Compession Screw 4.5 / 6.5 SURGICAL TECHNIQUE. Titanium or Stainless Steel. Cannulated Headless Design. Multiple Thread Options.
 Headless Compession Screw SURGICAL TECHNIQUE 4.5 / 6.5 Titanium or Stainless Steel Cannulated Headless Design Multiple Thread Options Torx Driver Sterile and Non-Sterile Options Simple Instrumentation
Headless Compession Screw SURGICAL TECHNIQUE 4.5 / 6.5 Titanium or Stainless Steel Cannulated Headless Design Multiple Thread Options Torx Driver Sterile and Non-Sterile Options Simple Instrumentation
BIOARCH SUBTALAR IMPLANT SYSTEM
 BIOARCH SUBTALAR IMPLANT SYSTEM 137308-4 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) - Chinese
BIOARCH SUBTALAR IMPLANT SYSTEM 137308-4 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) - Chinese
Royal Oak Cervical Plate System
 Royal Oak Cervical Plate System Manufactured by Nexxt Spine, Inc. Royal Oak Cervical Plate System INTRODUCTION FEATURES AND BENEFITS Table of Contents SURGICAL TECHNIQUE Step 1. Patient Positioning Step
Royal Oak Cervical Plate System Manufactured by Nexxt Spine, Inc. Royal Oak Cervical Plate System INTRODUCTION FEATURES AND BENEFITS Table of Contents SURGICAL TECHNIQUE Step 1. Patient Positioning Step
