FEP Medical Policy Manual
|
|
|
- Jonas Bertram Gilbert
- 5 years ago
- Views:
Transcription
1 FEP Medical Policy Manual Effective Date: October 15, 2018 Related Policies: None Three-Dimensional Printed Orthopedic Implants Description This evidence review addresses orthopedic implants that are constructed by additive manufacturing, commonly known as 3-dimensional (3D) printing. Three situations are considered: 3D printing of standard-sized implants, 3D printing of patient-matched implants for individuals who have typical bone and joint anatomy, and custom 3D printed implants for patients who have bone or joint deformity. OBJECTIVE The objective of this evidence review is to determine whether 3-dimensional printed orthopedic implants have a similar net health benefit compared with traditionally manufactured orthopedic implants. POLICY STATEMENT Three-dimensional (3D) printed orthopedic implants that have a design that is approved or cleared by the Food and Drug Administration (FDA) and produced in standard sizes for patients with typical bone and joint anatomy are investigational. Patient-matched 3D printed implants that are based on non-standard shapes and sizes for patients with typical bone and joint anatomy and do not qualify as custom devices according to FDA custom device exemption requirements are investigational. Custom 3D printed implants for patients with bone or joint deformity may be considered medically necessary when the devices are produced at a central manufacturing facility and meet FDA custom device exemption requirements. Three-dimensional printed orthopedic implants produced outside of FDA-regulated manufacturing facilities are investigational. POLICY GUIDELINES This policy does not address custom mandible or maxillofacial implants. BENEFIT APPLICATION Experimental or investigational procedures, treatments, drugs, or devices are not covered (See General Exclusion Section of brochure). Original Policy Date: October 2018 Page: 1
2 Effective Policy Date: October 15, 2018 Page: 2 of 5 FDA REGULATORY STATUS In 2017, UFDA published guidance for industry and technical considerations for 3D printed medical devices. 1 The recommendations in this guidance are intended to supplement any device-specific recommendations and represent FDA s initial thinking and recommendations. The guidance does not apply to 3D printing at the point-of-care. FDA expects that AM [additive manufacturing] devices will follow the same regulatory requirements and submission expectations as the classification and/or regulation to which a non-am device of the same type is subject. The required information, characterization, and testing will depend on a variety of factors, such as whether it is an implant or instrument, and whether it is available in standard sizes or is patientmatched. The FDA has noted that although patient-matched devices are sometimes referred to as customized devices, they are not custom devices meeting custom device exemption requirements under the U.S. Federal Food, Drug, and Cosmetic Act unless they comply with all of the criteria of section 520(b). FDA published guidance for industry and on the custom device exemption act in Custom devices are those created or modified to comply with the order of an individual physician or dentist, do not exceed 5 units per year, and are reported by the manufacturer to FDA for devices manufactured and distributed under section 520(b) of the Food, Drug, and Cosmetic Act. Under Section 520(b) of the Food, Drug, and Cosmetic Act, custom devices are exempt from premarket approval (PMA) requirements and conformance to mandatory performance standards. A device not covered by an existing marketing approval would require either a PMA or a valid exemption from the requirements to obtain PMA approval in order to be introduced into interstate commerce. Examples of potential valid exemptions or alternatives from the PMA requirement include: (1) establishing the substantial equivalence of the new device to a valid predicate device, (2) approval of an Investigational Device Exemption (IDE) or (3) meeting all custom device exemption requirements. Custom Devices are not exempt from any other requirements, including, but not limited to, the Quality System Regulation, including Design Controls (21 CFR Part 820); Medical Device Reporting (21 CFR Part 803); Labeling (21 CFR Part 801); Corrections and Removals (21 CFR Part 806); and Registration and Listing (21 CFR Part 807). A custom device may not be marketed to the general public. FDA has also noted that most patient-matched devices will fall within the existing regulatory pathway for that device type. In addition to standard labeling, specific labeling information is recommended for AM devices that are patient-matched. FDA has stated that modifications to a 510(k)-cleared device that maintain its original intended use and could be clinically studied do not appropriately qualify as a custom device. A number of titanium spinal interbody implants with increased roughness and porosity than traditional designs have received marketing clearance by FDA through the 510(k) process. They have a biomechanical stiffness similar to polyetheretherketone cages and less than solid titanium. They include: Cascadia Cervical and Cascadia AN Lordotic Oblique Interbody Systems (K2M) EIT (Emerging Implant Technologies) IB3D (Medicrea) Modulus XLIF (NuVasive) NanoHive interbodies (HD Lifesciences).
3 Effective Policy Date: October 15, 2018 Page: 3 of 5 Porous 3D printed titanium implants for minimally invasive sacroiliac joint fusion have received 510(k) clearances. ifuse 3D (SI Bone). Custom knee implants include: ConforMIS itotal Cruciate Retaining Knee Replacement System (ConforMIS) ConforMIS itotal Posterior Stabilized Knee Replacement System (ConforMIS) ConforMIS iuni Unicondylar Knee Replacement System (ConforMIS) ConforMIS itotal Hip system (ConforMIS). RATIONALE Summary of Evidence For individuals who have typical bone and joint anatomy and are undergoing standard orthopedic procedures who receive a standard-sized 3D printed implant, the evidence includes an RCT and systematic review. Relevant outcomes include symptoms, functional outcomes, and quality of life. Threedimensional printed implants are often manufactured with titanium and allow greater porosity than can be achieved with traditional manufacturing techniques. Greater porosity is believed to facilitate bony ingrowth and theoretically improve the stability of the implant. However, the effect of these devices on the adjacent bone, particularly subsidence and resorption, is unknown. Studies are needed that compare these newer devices with the established alternatives. The evidence is insufficient to determine the effects of the technology on health outcomes. For individuals who have typical bone and joint anatomy and are undergoing standard orthopedic procedures who receive a patient-matched 3D printed implant, the evidence includes no comparative studies. Relevant outcomes include symptoms, functional outcomes, and quality of life. Studies are needed to determine whether patient-matched implants improve outcomes compared with conventional implants. It is noted that other methods for the customization of orthopedic procedures, specifically patient-specific cutting guides and sex-specific implants, have failed to demonstrate improvements in health outcomes. Demonstration of improvement in key outcome measures is needed to justify the greater resource utilization (eg, time, imaging) of patient-matched 3D printed devices. The evidence is insufficient to determine the effects of the technology on health outcomes. For individuals who have bone or joint deformity requiring a custom orthopedic implant who receive a custom 3D printed implant, the evidence includes case series. Relevant outcomes include symptoms, functional outcomes, and quality of life. The largest case series with the longest follow-up is from outside of the United States. The most commonly reported indications are for revision total hip arthroplasty with severe acetabular defects, reconstruction following orthopedic tumor resection, and spinal abnormalities. These cases would require a custom process for design and manufacturing, even with traditional manufacturing methods. Therefore, the design and manufacturing of a single implant with 3D printing is an advantage of this technology. The evidence is sufficient to determine that the technology results in a meaningful improvement in the net health outcome. SUPPLEMENTAL INFORMATION Practice Guidelines and Position Statements American Society for Testing and Material The American Society for Testing and Material has drafted standards for additive manufacturing. 15 The specification on Titanium-6 Aluminum-4 Vanadium with Powder Bed Fusion covers additively manufactured titanium-6aluminum-4vanadium components using full-melt powder bed fusion such as electron beam melting and laser melting. The Society states that the components produced by these processes are used typically in applications that require mechanical properties similar to machined
4 Effective Policy Date: October 15, 2018 Page: 4 of 5 forgings and wrought products. Components manufactured to this specification are often, but not necessarily, post processed via machining, grinding, electrical discharge machining, polishing, and so forth to achieve desired surface finish and critical dimensions. Not applicable. U.S. Preventive Services Task Force Recommendations Medicare National Coverage There is no national coverage determination (NCD). In the absence of an NCD, coverage decisions are left to the discretion of local Medicare carriers. REFERENCES 1. U.S. Food and Drug Administration. Technical considerations for additive manufactured medical devices: Guidance for industry and Food and Drug Administration staff. 2017; pdf. Accessed January 23, U.S. Food and Drug Administration. Custom device exemption: Guidance for industry and Food and Drug Administration Staff. 2014; 99.p df. Accessed January 23, Chen Y, Wang X, Lu X, et al. Comparison of titanium and polyetheretherketone (PEEK) cages in the surgical treatment of multilevel cervical spondylotic myelopathy: a prospective, randomized, control study with over 7- year follow-up. Eur Spine J. Jul 2013;22(7): PMID Seaman S, Kerezoudis P, Bydon M, et al. Titanium vs. polyetheretherketone (PEEK) interbody fusion: Metaanalysis and review of the literature. J Clin Neurosci. Oct 2017;44: PMID Maurer TB, Ochsner PE, Schwarzer G, et al. Increased loosening of cemented straight stem prostheses made from titanium alloys. An analysis and comparison with prostheses made of cobalt-chromium-nickel alloy. Int Orthop. Jun 2001;25(2): PMID Arabnejad S, Johnston B, Tanzer M, et al. Fully porous 3D printed titanium femoral stem to reduce stressshielding following total hip arthroplasty. J Orthop Res. Aug 2017;35(8): PMID Cheng T, Zhu C, Wang J, et al. No clinical benefit of gender-specific total knee arthroplasty. Acta Orthop. Aug 2014;85(4): PMID Citak M, Kochsiek L, Gehrke T, et al. Preliminary results of a 3D-printed acetabular component in the management of extensive defects. Hip Int. Dec :0. PMID Mao Y, Xu C, Xu J, et al. The use of customized cages in revision total hip arthroplasty for Paprosky type III acetabular bone defects. Int Orthop. Oct 2015;39(10): PMID Liang H, Ji T, Zhang Y, et al. Reconstruction with 3D-printed pelvic endoprostheses after resection of a pelvic tumour. Bone Joint J. Feb 2017;99-b(2): PMID Mobbs RJ, Coughlan M, Thompson R, et al. The utility of 3D printing for surgical planning and patientspecific implant design for complex spinal pathologies: case report. J Neurosurg Spine. Apr 2017;26(4): PMID Li H, Qu X, Mao Y, et al. Custom acetabular cages offer stable fixation and improved Hip Scores for revision THA with severe bone defects. Clin Orthop Relat Res. Mar 2016;474(3): PMID Ding HW, Yu GW, Tu Q, et al. Computer-aided resection and endoprosthesis design for the management of malignant bone tumors around the knee: outcomes of 12 cases. BMC Musculoskelet Disord. Nov ;14:331. PMID Luo W, Huang L, Liu H, et al. Customized knee prosthesis in treatment of giant cell tumors of the proximal tibia: application of 3-dimensional printing technology in surgical design. Med Sci Monit. Apr ;23: PMID American Society for Testing and Material. Additive manufacturing technology standards. n.d.; Accessed April 23, 2018.
5 Effective Policy Date: October 15, 2018 Page: 5 of 5 POLICY HISTORY Date Action Description September 2018 New Policy Policy created with literature review through March 20, Threedimensional printed implants are considered medically necessary for custom implants for patients with bone or joint deformity and investigational for standard and patient-matched implants.
Three-Dimensional Printed Orthopedic Implants
 Three-Dimensional Printed Orthopedic Implants Policy Number: 7.01.161 Last Review: 8/1/2018 Origination: 08/2018 Next Review: 8/1/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide
Three-Dimensional Printed Orthopedic Implants Policy Number: 7.01.161 Last Review: 8/1/2018 Origination: 08/2018 Next Review: 8/1/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide
Medical Policy. MP Three-Dimensional Printed Orthopedic Implants
 Medical Policy MP 7.01.161 BCBSA Ref. Policy: 7.01.161 Last Review: 05/30/2018 Effective Date: 08/30/2018 Section: Surgery Related Policies 7.01.144 Personalized Cutting Guides DISCLAIMER Our medical policies
Medical Policy MP 7.01.161 BCBSA Ref. Policy: 7.01.161 Last Review: 05/30/2018 Effective Date: 08/30/2018 Section: Surgery Related Policies 7.01.144 Personalized Cutting Guides DISCLAIMER Our medical policies
Facet Arthroplasty. Policy Number: Last Review: 9/2018 Origination: 9/2009 Next Review: 3/2019
 Facet Arthroplasty Policy Number: 7.01.120 Last Review: 9/2018 Origination: 9/2009 Next Review: 3/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for total facet
Facet Arthroplasty Policy Number: 7.01.120 Last Review: 9/2018 Origination: 9/2009 Next Review: 3/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for total facet
July 10, X-spine Systems, Incorporated David Kirschman, MD Chief Medical Officer 452 Alexandersville Road Miamisburg, Ohio 45342
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service July 10, 2015 Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 X-spine
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service July 10, 2015 Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 X-spine
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: July 15, 2017 Related Policies: 1.01.05 Ultrasound Accelerated Fracture Healing Device 7.01.07 Electrical Bone Growth Stimulation of the Appendicular Skeleton
FEP Medical Policy Manual Effective Date: July 15, 2017 Related Policies: 1.01.05 Ultrasound Accelerated Fracture Healing Device 7.01.07 Electrical Bone Growth Stimulation of the Appendicular Skeleton
OSSIS is an ISO accredited company.
 CUSTOM IMPLANTS Over the last 15 years Ossis orthopaedic surgeons and materials and design engineers have been at the leading-edge of innovation in patient-specific implants. Ossis combines the use of
CUSTOM IMPLANTS Over the last 15 years Ossis orthopaedic surgeons and materials and design engineers have been at the leading-edge of innovation in patient-specific implants. Ossis combines the use of
Specializing in Mechanical Testing for Medical Devices. Office:
 Specializing in Mechanical Testing for Medical Devices Office: 260.489.1444 Email: sales@jtlamerica.com www.jtlamerica.com Spine Spinal implant test standards can t keep up with the rapid pace of device
Specializing in Mechanical Testing for Medical Devices Office: 260.489.1444 Email: sales@jtlamerica.com www.jtlamerica.com Spine Spinal implant test standards can t keep up with the rapid pace of device
OSSIS is an ISO accredited company.
 CUSTOM IMPLANTS Over the last 15 years Ossis orthopaedic surgeons and materials and design engineers have been at the leading-edge of innovation in patient-specific implants. Ossis combines the use of
CUSTOM IMPLANTS Over the last 15 years Ossis orthopaedic surgeons and materials and design engineers have been at the leading-edge of innovation in patient-specific implants. Ossis combines the use of
MEDICAL POLICY SUBJECT: COMPUTER ASSISTED NAVIGATION FOR KNEE AND HIP ARTHROPLASTY
 MEDICAL POLICY PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
MEDICAL POLICY PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
MEDICAL POLICY SUBJECT: COMPUTER ASSISTED NAVIGATION FOR KNEE AND HIP ARTHROPLASTY
 MEDICAL POLICY SUBJECT: COMPUTER ASSISTED PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
MEDICAL POLICY SUBJECT: COMPUTER ASSISTED PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
Interbody Fusion Implants with PEEK core and Plasmapore XP titanium coating DEGENERATIVE SPINAL DISORDERS AESCULAP XP THE CHOICE OF EXPERTS
 Interbody Fusion Implants with PEEK core and Plasmapore XP titanium coating DEGENERATIVE SPINAL DISORDERS AESCULAP XP THE CHOICE OF EXPERTS P E E K + T I T A N I U M AESCULAP XP Built on experience Aesculap,
Interbody Fusion Implants with PEEK core and Plasmapore XP titanium coating DEGENERATIVE SPINAL DISORDERS AESCULAP XP THE CHOICE OF EXPERTS P E E K + T I T A N I U M AESCULAP XP Built on experience Aesculap,
LIMB COMPRESSION DEVICES FOR VENOUS THROMBOEMBOLISM PROPHYLAXIS
 PROPHYLAXIS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs are dependent
PROPHYLAXIS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs are dependent
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: December 2016 Effective Date: January 15, 2017 Related Policies: 7.01.87 Artificial Intervertebral Disc: Lumbar Spine Artificial Intervertebral Disc: Cervical Spine
FEP Medical Policy Manual Last Review: December 2016 Effective Date: January 15, 2017 Related Policies: 7.01.87 Artificial Intervertebral Disc: Lumbar Spine Artificial Intervertebral Disc: Cervical Spine
Protocol. Electrical Stimulation of the Spine as an Adjunct to Spinal Fusion Procedures
 Electrical Stimulation of the Spine as an Adjunct to Spinal Fusion (70185) Medical Benefit Effective Date: 07/01/12 Next Review Date: 01/19 Preauthorization Yes Review Dates: 09/07, 09/08, 09/09, 05/10,
Electrical Stimulation of the Spine as an Adjunct to Spinal Fusion (70185) Medical Benefit Effective Date: 07/01/12 Next Review Date: 01/19 Preauthorization Yes Review Dates: 09/07, 09/08, 09/09, 05/10,
Class II Special Controls Guidance Document: Intraoral Devices for Snoring and/or Obstructive Sleep Apnea; Guidance for Industry and FDA
 Class II Special Controls Guidance Document: Intraoral Devices for Snoring and/or Obstructive Sleep Apnea; Guidance for Industry and FDA Document issued on: November 12, 2002 This document supersedes the
Class II Special Controls Guidance Document: Intraoral Devices for Snoring and/or Obstructive Sleep Apnea; Guidance for Industry and FDA Document issued on: November 12, 2002 This document supersedes the
Electrical Stimulation of the Spine as an Adjunct to Spinal Fusion Procedures Corporate Medical Policy
 Electrical Stimulation of the Spine as an Adjunct to Spinal Fusion Procedures Corporate Medical Policy File name: Electrical Stimulation of the Spine as an Adjunct to Spinal Fusion Procedures File code:
Electrical Stimulation of the Spine as an Adjunct to Spinal Fusion Procedures Corporate Medical Policy File name: Electrical Stimulation of the Spine as an Adjunct to Spinal Fusion Procedures File code:
SNuVASivE: 51. 0(k) Premarket Notification Long Lateral Spinal System
 K111410 Page 1 of 2 SNuVASivE: 51 0(k) Premarket Notification Long Lateral Spinal System 510(k) SummaryJU 20201 In accordance with Title 21 of the Code of Federal Regulations, Part 807, and in particular
K111410 Page 1 of 2 SNuVASivE: 51 0(k) Premarket Notification Long Lateral Spinal System 510(k) SummaryJU 20201 In accordance with Title 21 of the Code of Federal Regulations, Part 807, and in particular
HOSPITAL "A" (2007) ORTHOPEDIC JOINT IMPLANT COMPONENT MATRIX
 HOSPITAL "A" (2007) ORTHOPEDIC JOINT IMPLANT COMPONENT MATRIX HIP IMPLANTS TOTAL HIPS Ultra High Demand (metal on metal) Femur, ingrowth Femoral head, metal Acetabular shell, ingrowth Acetabular liner,
HOSPITAL "A" (2007) ORTHOPEDIC JOINT IMPLANT COMPONENT MATRIX HIP IMPLANTS TOTAL HIPS Ultra High Demand (metal on metal) Femur, ingrowth Femoral head, metal Acetabular shell, ingrowth Acetabular liner,
MAR (k) SUMMARY. Stanmore Implants Worldwide Ltd JTS Extendible Implant Preparation Date: March 17, A ppl ica nt/spo0nsor:
 510(k) SUMMARY MAR 2 2 201 Stanmore Implants Worldwide Ltd JTS Extendible Implant Preparation Date: March 17, 2011 A ppl ica nt/spo0nsor: Contact Person: Proprietary name: Common or Usual Name: Classification
510(k) SUMMARY MAR 2 2 201 Stanmore Implants Worldwide Ltd JTS Extendible Implant Preparation Date: March 17, 2011 A ppl ica nt/spo0nsor: Contact Person: Proprietary name: Common or Usual Name: Classification
Donald W. Guthner Orgenix, LLC Ill Hill Road. Douglassville, PA (888) ORGENIX ( ) (484) (FAX) Intramedullary Nails, Nails
 K070741 ~, SANATMETAL Manufacturer of Orthopaedic and Traumatologic Products H-3301, Eger Faiskola ut 5. Phone: +(36)36-512-900 Fax: +(36)36-512-932 e-mail: metalasanatmetal.hu 510(k) Statement of Summary
K070741 ~, SANATMETAL Manufacturer of Orthopaedic and Traumatologic Products H-3301, Eger Faiskola ut 5. Phone: +(36)36-512-900 Fax: +(36)36-512-932 e-mail: metalasanatmetal.hu 510(k) Statement of Summary
MAY Page 1 of 2
 MAY 2 22012 Page 1 of 2 2. 5 10(k) SUMMARY Sponsor Name: 510(k) Contact: Consensus Orthopedics, Inc. 1115 Windfield Way, Suite 100 El Dorado Hills, CA 95762 Matthew M. Hull, RAC Phone: (916) 355-7156/
MAY 2 22012 Page 1 of 2 2. 5 10(k) SUMMARY Sponsor Name: 510(k) Contact: Consensus Orthopedics, Inc. 1115 Windfield Way, Suite 100 El Dorado Hills, CA 95762 Matthew M. Hull, RAC Phone: (916) 355-7156/
Dynamic Spinal Visualization and Vertebral Motion Analysis
 Dynamic Spinal Visualization and Vertebral Motion Analysis Policy Number: 6.01.46 Last Review: 2/2019 Origination: 2/2006 Next Review: 2/2020 Policy Blue Cross and Blue Shield of Kansas City (Blue KC)
Dynamic Spinal Visualization and Vertebral Motion Analysis Policy Number: 6.01.46 Last Review: 2/2019 Origination: 2/2006 Next Review: 2/2020 Policy Blue Cross and Blue Shield of Kansas City (Blue KC)
1115 Windfield Way, Suite 100 El Dorado Hills, CA Matthew M. Hull, RAC Phone: (916) / Fax: (916)
 Page 1 of 4 2. 510(k) SUMMARY JUN 2 7 2011 Sponsor Name: 510(k) Contact: Consensus Orthopedics, Inc. 1115 Windfield Way, Suite 100 El Dorado Hills, CA 95762 Matthew M. Hull, RAC Phone: (916) 355-7156/
Page 1 of 4 2. 510(k) SUMMARY JUN 2 7 2011 Sponsor Name: 510(k) Contact: Consensus Orthopedics, Inc. 1115 Windfield Way, Suite 100 El Dorado Hills, CA 95762 Matthew M. Hull, RAC Phone: (916) 355-7156/
Total Knee Replacement
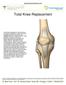 Total Knee Replacement A total knee replacement, also known as total knee arthroplasty, involves removing damaged portions of the knee, and capping the bony surfaces with man-made prosthetic implants.
Total Knee Replacement A total knee replacement, also known as total knee arthroplasty, involves removing damaged portions of the knee, and capping the bony surfaces with man-made prosthetic implants.
FDA 510(k) 101 The Basics
 FDA 510(k) 101 The Basics Floyd G. Larson President, PaxMed International San Diego, CA OMTEC June 17, 2010 Chicago Agenda History of 510(k) process FDA s risk based approach FDA guidance and standards
FDA 510(k) 101 The Basics Floyd G. Larson President, PaxMed International San Diego, CA OMTEC June 17, 2010 Chicago Agenda History of 510(k) process FDA s risk based approach FDA guidance and standards
Medical Policy. MP Dynamic Spinal Visualization and Vertebral Motion Analysis
 Medical Policy BCBSA Ref. Policy: 6.01.46 Last Review: 09/19/2018 Effective Date: 12/15/2018 Section: Radiology Related Policies 6.01.48 Positional Magnetic Resonance Imaging 9.01.502 Experimental / Investigational
Medical Policy BCBSA Ref. Policy: 6.01.46 Last Review: 09/19/2018 Effective Date: 12/15/2018 Section: Radiology Related Policies 6.01.48 Positional Magnetic Resonance Imaging 9.01.502 Experimental / Investigational
Topic: Percutaneous Axial Anterior Lumbar Fusion Date of Origin: June Section: Surgery Last Reviewed Date: June 2013
 Medical Policy Manual Topic: Percutaneous Axial Anterior Lumbar Fusion Date of Origin: June 2007 Section: Surgery Last Reviewed Date: June 2013 Policy No: 157 Effective Date: August 1, 2013 IMPORTANT REMINDER
Medical Policy Manual Topic: Percutaneous Axial Anterior Lumbar Fusion Date of Origin: June 2007 Section: Surgery Last Reviewed Date: June 2013 Policy No: 157 Effective Date: August 1, 2013 IMPORTANT REMINDER
BEST PRACTICE CHANGE CONTROL : DECIDING WHEN TO SUBMIT A 510(K) FOR A DEVICE CHANGE. Yuan Xu 18- JAN- 2018
 BEST PRACTICE CHANGE CONTROL : DECIDING WHEN TO SUBMIT A 510(K) FOR A DEVICE CHANGE Yuan Xu 18- JAN- 2018 Background AGENDA What is Medical Device? What is 510(k)? Principles of best practices of device
BEST PRACTICE CHANGE CONTROL : DECIDING WHEN TO SUBMIT A 510(K) FOR A DEVICE CHANGE Yuan Xu 18- JAN- 2018 Background AGENDA What is Medical Device? What is 510(k)? Principles of best practices of device
Corporate Medical Policy
 Corporate Medical Policy Computer Assisted Surgical Navigational Orthopedic Procedures File Name: Origination: Last CAP Review: Next CAP Review: Last Review: computer_assisted_surgical_navigational_orthopedic_procedures
Corporate Medical Policy Computer Assisted Surgical Navigational Orthopedic Procedures File Name: Origination: Last CAP Review: Next CAP Review: Last Review: computer_assisted_surgical_navigational_orthopedic_procedures
Optimum implant geometry
 Design Rationale Optimum implant geometry Extending proven Tri-Lock heritage The original Tri-Lock was introduced in 1981. This implant was the first proximally coated tapered-wedge hip stem available
Design Rationale Optimum implant geometry Extending proven Tri-Lock heritage The original Tri-Lock was introduced in 1981. This implant was the first proximally coated tapered-wedge hip stem available
Individual oncological implants
 Individual oncological implants Catalogue - individual oncological implants ONCOLOGICAL Materials for implants List of materials MATERIAL ISO ČSN DIN ASTM Stainless steel 5832-1 17350 výběr 17 443 W.Nr.
Individual oncological implants Catalogue - individual oncological implants ONCOLOGICAL Materials for implants List of materials MATERIAL ISO ČSN DIN ASTM Stainless steel 5832-1 17350 výběr 17 443 W.Nr.
USE OF PEEK IN SPINE SURGERY
 USE OF PEEK IN SPINE SURGERY Tracy Ma, MD William C. Welch, MD May 2017 Financial Disclosures Transcendental Spine stock Titanium Stress shielding: stress mismatch between titanium implant and adjacent
USE OF PEEK IN SPINE SURGERY Tracy Ma, MD William C. Welch, MD May 2017 Financial Disclosures Transcendental Spine stock Titanium Stress shielding: stress mismatch between titanium implant and adjacent
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: artificial_intervertebral_disc 4/2004 10/2017 10/2018 10/2017 Description of Procedure or Service During
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: artificial_intervertebral_disc 4/2004 10/2017 10/2018 10/2017 Description of Procedure or Service During
DEC (k) Summary. X-spine Systems, Inc Corporate Way #212 Centerville, OH Telephone (800) FAX (866)
 DEC 1 2 2005 Ksjq 5 1 0(k) Premarket Notification Capless' Pedicle Screw System 510(k) Summary ADMINISTRATIVE INFORMATION NManufacturcr Name: X-spine Systems, Inc. 7026 Corporate Way #212 Centerville,
DEC 1 2 2005 Ksjq 5 1 0(k) Premarket Notification Capless' Pedicle Screw System 510(k) Summary ADMINISTRATIVE INFORMATION NManufacturcr Name: X-spine Systems, Inc. 7026 Corporate Way #212 Centerville,
O(K) SUMMARY. Mega'Gen Co., Ltd , Eupchun-Ri, Jain-Myun. Gyeongsan, Gyeongbuk South Korea Phone: , Fax:
 EZ PLUS IMPLANT SYSTEM 13. 510O(K) SUMMARY Mega'Gen Co., Ltd. 114-8, Eupchun-Ri, Jain-Myun. Gyeongsan, Gyeongbuk South Korea Phone: 82-53-857-5770, Fax: 82-53-857-5432 510(K) Summary 510O(K) SUMMARY AND
EZ PLUS IMPLANT SYSTEM 13. 510O(K) SUMMARY Mega'Gen Co., Ltd. 114-8, Eupchun-Ri, Jain-Myun. Gyeongsan, Gyeongbuk South Korea Phone: 82-53-857-5770, Fax: 82-53-857-5432 510(K) Summary 510O(K) SUMMARY AND
Table of Contents: JULY TH EDITION
 Table of Contents: Table of Contents of the worldwide Orthopaedic Contract manufacturing Market 2016-2021 & Top 100 player profiles 275+ pages 450+ exhibits July 2017 5 th edition I- Worldwide orthopaedic
Table of Contents: Table of Contents of the worldwide Orthopaedic Contract manufacturing Market 2016-2021 & Top 100 player profiles 275+ pages 450+ exhibits July 2017 5 th edition I- Worldwide orthopaedic
MEDICAL POLICY SUBJECT: ARTIFICIAL LUMBAR INTERVERTEBRAL DISC
 MEDICAL POLICY SUBJECT: ARTIFICIAL LUMBAR PAGE: 1 OF: 7 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
MEDICAL POLICY SUBJECT: ARTIFICIAL LUMBAR PAGE: 1 OF: 7 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
Hip Joint Prostheses
 Translated English of Chinese Standard: YY0118-2005 www.chinesestandard.net Sales@ChineseStandard.net PHARMACEUTICAL INDUSTRY STANDARD YY ICS 11.040.40 OF THE PEOPLE S REPUBLIC OF CHINA C 35 YY 0118-2005
Translated English of Chinese Standard: YY0118-2005 www.chinesestandard.net Sales@ChineseStandard.net PHARMACEUTICAL INDUSTRY STANDARD YY ICS 11.040.40 OF THE PEOPLE S REPUBLIC OF CHINA C 35 YY 0118-2005
Handheld Radiofrequency Spectroscopy for Intraoperative Assessment of Surgical Margins During Breast-Conserving Surgery
 7.01.140 Handheld Radiofrequency Spectroscopy for Intraoperative Assessment of Surgical Margins During Breast-Conserving Surgery Section 7.0 Surgery Subsection Description Effective Date November 26, 2014
7.01.140 Handheld Radiofrequency Spectroscopy for Intraoperative Assessment of Surgical Margins During Breast-Conserving Surgery Section 7.0 Surgery Subsection Description Effective Date November 26, 2014
Continuing the Tradition. VerSys Heritage Hip System
 Continuing the Tradition VerSys Heritage Hip System Heritage Following the Tradition The low-friction hip prosthesis developed by Sir John Charnley has more than a 20-year history of outstanding results.
Continuing the Tradition VerSys Heritage Hip System Heritage Following the Tradition The low-friction hip prosthesis developed by Sir John Charnley has more than a 20-year history of outstanding results.
Spinal Surgical Robots: Market Shares, Strategies, and Forecasts, Worldwide,
 Spinal Surgical Robots: Market Shares, Strategies, and Forecasts, Worldwide, 2016-2022 Table of Contents Spinal Surgical Robot: Executive Summary The study is designed to give a comprehensive overview
Spinal Surgical Robots: Market Shares, Strategies, and Forecasts, Worldwide, 2016-2022 Table of Contents Spinal Surgical Robot: Executive Summary The study is designed to give a comprehensive overview
Regenerex Porous Titanium Construct. Knees Hips Extremities Cement and Accessories PMI Trauma Technology
 Regenerex Porous Titanium Construct Knees Hips Extremities Cement and Accessories PMI Trauma Technology One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide personalized
Regenerex Porous Titanium Construct Knees Hips Extremities Cement and Accessories PMI Trauma Technology One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide personalized
Principles of acetabular fixation in primary and revision hip arthroplasty Piotr Wojciechowski, Damian Kusz, Anna WAGNER
 Principles of acetabular fixation in primary and revision hip arthroplasty Piotr Wojciechowski, Damian Kusz, Anna WAGNER Department of Orthopedics and Traumatology Medical University of Silesia Head of
Principles of acetabular fixation in primary and revision hip arthroplasty Piotr Wojciechowski, Damian Kusz, Anna WAGNER Department of Orthopedics and Traumatology Medical University of Silesia Head of
SPINE. 510(k) Summary. Anterior/anterolateral noncervical use (KWQ) (Prodct Cde):Noncervical
 k10 3?I13 SPINE 510(k) Summary FB-22f FB 24f This summary of 510(k) safety and effectiveness information is being submitted in accordance with the requirements of 21 CFR 807.92. Preparation Date: November
k10 3?I13 SPINE 510(k) Summary FB-22f FB 24f This summary of 510(k) safety and effectiveness information is being submitted in accordance with the requirements of 21 CFR 807.92. Preparation Date: November
Enhancing stability and increasing range of motion. Metasul LDH Large Diameter Head
 Enhancing stability and increasing range of motion Metasul LDH Large Diameter Head The Metasul large diameter head technology is the result of in-depth research, development and clinical experience that
Enhancing stability and increasing range of motion Metasul LDH Large Diameter Head The Metasul large diameter head technology is the result of in-depth research, development and clinical experience that
Plasmapore XP. Stability that Starts at the Surface
 Plasmapore XP Stability that Starts at the Surface The Difference The Plasmapore XP Difference The unique structure, properties and processing of the Plasmapore XP coating results in early bone apposition
Plasmapore XP Stability that Starts at the Surface The Difference The Plasmapore XP Difference The unique structure, properties and processing of the Plasmapore XP coating results in early bone apposition
LARS (Ligament Augmentation & Reconstruction System) Literature
 LARS-Related Studies and Papers ACL: 1. Level of Evidence: IV Li, H. et al (2011). Enhancement of the osseointegration of a polyethylene Terephthalate artificial ligament graft in a bone tunnel using 58S
LARS-Related Studies and Papers ACL: 1. Level of Evidence: IV Li, H. et al (2011). Enhancement of the osseointegration of a polyethylene Terephthalate artificial ligament graft in a bone tunnel using 58S
KASM Knee Articulating Spacer Mold. Surgical Summary
 KASM Knee Articulating Spacer Mold Surgical Summary KASM Designing Surgeons: Michael H. Bourne, M.D. Salt Lake Orthopedic Clinic E. Marc Mariani, M.D. Salt Lake Orthopedic Clinic U.S. Patent 6,942,475
KASM Knee Articulating Spacer Mold Surgical Summary KASM Designing Surgeons: Michael H. Bourne, M.D. Salt Lake Orthopedic Clinic E. Marc Mariani, M.D. Salt Lake Orthopedic Clinic U.S. Patent 6,942,475
Optimum implant geometry
 Design Rationale Optimum implant geometry Extending the proven Tri-Lock heritage The original Tri-Lock was introduced in 1981. This implant was the first proximally coated tapered-wedge hip stem available
Design Rationale Optimum implant geometry Extending the proven Tri-Lock heritage The original Tri-Lock was introduced in 1981. This implant was the first proximally coated tapered-wedge hip stem available
3D titanium interbody fusion cages sharx. White Paper
 3D titanium interbody fusion cages sharx (SLM selective laser melted) Goal of the study: Does the sharx intervertebral cage due to innovative material, new design, and lordotic shape solve some problems
3D titanium interbody fusion cages sharx (SLM selective laser melted) Goal of the study: Does the sharx intervertebral cage due to innovative material, new design, and lordotic shape solve some problems
Talos -C (HA) and the PEEK-OPTIMA HA Enhanced Technology
 v Talos -C (HA) and the HA Enhanced Technology The innovative Talos -C (HA) IBF Implant made with polymer is fully integrated with the well known material Hydroxyapatite (HA). The Talos -C (HA) IBF Implant
v Talos -C (HA) and the HA Enhanced Technology The innovative Talos -C (HA) IBF Implant made with polymer is fully integrated with the well known material Hydroxyapatite (HA). The Talos -C (HA) IBF Implant
510(k) Summary. Acumed, LLC 5885 NW Cornelius Pass Road Hillsboro, OR 97124
 K(102998 Page I of 2 510(k) Summary Device Trade Name: Congruent Bone Plate System J A.N 4 21 Manufacturer: Contact: Prepared by: Acumed, LLC 5885 NW Cornelius Pass Road Hillsboro, OR 97124 Mr. Ed Boehmer
K(102998 Page I of 2 510(k) Summary Device Trade Name: Congruent Bone Plate System J A.N 4 21 Manufacturer: Contact: Prepared by: Acumed, LLC 5885 NW Cornelius Pass Road Hillsboro, OR 97124 Mr. Ed Boehmer
U.S. MARKET FOR MINIMALLY INVASIVE SPINAL IMPLANTS
 U.S. MARKET FOR MINIMALLY INVASIVE SPINAL IMPLANTS idata_usmis15_rpt Published in December 2014 By idata Research Inc., 2014 idata Research Inc. Suite 308 4211 Kingsway Burnaby, British Columbia, Canada,
U.S. MARKET FOR MINIMALLY INVASIVE SPINAL IMPLANTS idata_usmis15_rpt Published in December 2014 By idata Research Inc., 2014 idata Research Inc. Suite 308 4211 Kingsway Burnaby, British Columbia, Canada,
PARADIGM SPINE. Anterior Cervical Fusion Cage. Cervical Interbody Fusion
 PARADIGM SPINE Anterior Cervical Fusion Cage Cervical Interbody Fusion DESIGN RATIONALE The OptiStrain TM C* interbody fusion cage follows well established biomechanical principles: The slot design of
PARADIGM SPINE Anterior Cervical Fusion Cage Cervical Interbody Fusion DESIGN RATIONALE The OptiStrain TM C* interbody fusion cage follows well established biomechanical principles: The slot design of
ko~ Tk (K) SUMMARY
 ko~3367-3 13. 510(K) SUMMARY 7-Tk 1 92006 Mega'Gen Co., Ltd. 114-8, Eupchun-Ri, Jain-Myun, Gyeongsan, Gyeongbuk South Korea Phone: 82-53-857-5770, Fax: 82-53-857-5432 510(K) Summary 510O(K) SUMMARY AND
ko~3367-3 13. 510(K) SUMMARY 7-Tk 1 92006 Mega'Gen Co., Ltd. 114-8, Eupchun-Ri, Jain-Myun, Gyeongsan, Gyeongbuk South Korea Phone: 82-53-857-5770, Fax: 82-53-857-5432 510(K) Summary 510O(K) SUMMARY AND
Preparing a US FDA Medical Device 510(K) Submission
 Preparing a US FDA Medical Device 510(K) Submission If you want to introduce your medical device to the US market, you need to obtain clearance from the FDA. This clearance is obtained from the FDA via
Preparing a US FDA Medical Device 510(K) Submission If you want to introduce your medical device to the US market, you need to obtain clearance from the FDA. This clearance is obtained from the FDA via
PERCUTANEOUS BALLOON KYPHOPLASTY, RADIOFREQUENCY KYPHOPLASTY, AND MECHANICAL VERTEBRAL AUGMENTATION
 KYPHOPLASTY, AND MECHANICAL VERTEBRAL AUGMENTATION Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures,
KYPHOPLASTY, AND MECHANICAL VERTEBRAL AUGMENTATION Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures,
POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER CODING INFORMATION REFERENCES POLICY HISTORY
 Original Issue Date (Created): 3/1/2012 Most Recent Review Date (Revised): 9/6/2018 Effective Date: 11/1/2018 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER
Original Issue Date (Created): 3/1/2012 Most Recent Review Date (Revised): 9/6/2018 Effective Date: 11/1/2018 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER
March 19, Dear Ms. Strohkirch:
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service March 19, 2015 Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 Treace
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service March 19, 2015 Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 Treace
Arcos Modular Femoral Revision System
 Arcos Modular Femoral Revision System Arcos System Simplify the Complex The Arcos Modular Femoral Revision System meets the demands of complex hip revision surgery by offering surgeons and OR staff the
Arcos Modular Femoral Revision System Arcos System Simplify the Complex The Arcos Modular Femoral Revision System meets the demands of complex hip revision surgery by offering surgeons and OR staff the
Cervical corpectomy for sub-axial retro-vertebral body lesions
 Eshra Egyptian Journal of Neurosurgery (2019) 33:2 https://doi.org/10.1186/s41984-018-0004-9 Egyptian Journal of Neurosurgery RESEARCH Open Access Cervical corpectomy for sub-axial retro-vertebral body
Eshra Egyptian Journal of Neurosurgery (2019) 33:2 https://doi.org/10.1186/s41984-018-0004-9 Egyptian Journal of Neurosurgery RESEARCH Open Access Cervical corpectomy for sub-axial retro-vertebral body
PLR. Proximal Loading Revision Hip System
 PLR Proximal Loading Revision Hip System The PLR splined revision stem is designed to recreate the natural stresses in the revised femur, where proximal bone may be compromised. PLR Hip System Design Considerations
PLR Proximal Loading Revision Hip System The PLR splined revision stem is designed to recreate the natural stresses in the revised femur, where proximal bone may be compromised. PLR Hip System Design Considerations
Over 20 Years of Proven Clinical Success. Zimmer Natural-Knee II System
 Over 20 Years of Proven Clinical Success Zimmer Natural-Knee II System CSTi Porous Coating Structurally similar to human bone CSTi porous coating combines the excellent biocompatibility of titanium with
Over 20 Years of Proven Clinical Success Zimmer Natural-Knee II System CSTi Porous Coating Structurally similar to human bone CSTi porous coating combines the excellent biocompatibility of titanium with
The bi-metric XR series of Hip implants blends biomet s proven philosophy and tradition with Advanced technology. porous primary hip series
 The bi-metric XR series of Hip implants blends biomet s proven philosophy and tradition with Advanced technology porous primary hip series BI-METRIC XR SERIES implant technology The Bi-Metric XR Hip Series
The bi-metric XR series of Hip implants blends biomet s proven philosophy and tradition with Advanced technology porous primary hip series BI-METRIC XR SERIES implant technology The Bi-Metric XR Hip Series
OPERATIVE TECHNIQUE. CONSTRUX Mini PTC. Mini PTC Spacer System
 OPERATIVE TECHNIQUE CONSTRUX Mini PTC Mini PTC Spacer System TABLE OF CONTENTS Introduction 1 Operative Technique 2 Part Numbers 6 Indications For Use 7 INTRODUCTION 1 INTRODUCTION The CONSTRUX Mini PTC
OPERATIVE TECHNIQUE CONSTRUX Mini PTC Mini PTC Spacer System TABLE OF CONTENTS Introduction 1 Operative Technique 2 Part Numbers 6 Indications For Use 7 INTRODUCTION 1 INTRODUCTION The CONSTRUX Mini PTC
INTERSPINOUS FIXATION (FUSION) DEVICES
 INTERSPINOUS FIXATION (FUSION) DEVICES Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical
INTERSPINOUS FIXATION (FUSION) DEVICES Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical
The Fusion of. Strength, Design. Performance. and
 The Fusion of Strength, Design and Performance Interbody Fusion Platform Interbody Fusion Platform The DePuy Spine Interbody Fusion platform is designed to provide an open architecture implant in a load-sharing
The Fusion of Strength, Design and Performance Interbody Fusion Platform Interbody Fusion Platform The DePuy Spine Interbody Fusion platform is designed to provide an open architecture implant in a load-sharing
K Michael Kolber Regulatory Affairs
 510(k) Number Submitter Name and Address Name: Contact: Address: Consultant, K122105 for the SM I Cardiovascular Patch November 7, 2012 Appendix 4: 510(k) Summary per (21CFRSO7.92) DEC 1 202 K122105 Michael
510(k) Number Submitter Name and Address Name: Contact: Address: Consultant, K122105 for the SM I Cardiovascular Patch November 7, 2012 Appendix 4: 510(k) Summary per (21CFRSO7.92) DEC 1 202 K122105 Michael
Int J Clin Exp Med 2018;11(2): /ISSN: /IJCEM Yi Yang, Hao Liu, Yueming Song, Tao Li
 Int J Clin Exp Med 2018;11(2):1278-1284 www.ijcem.com /ISSN:1940-5901/IJCEM0063093 Case Report Dislocation and screws pull-out after application of an Isobar TTL dynamic stabilisation system at L2/3 in
Int J Clin Exp Med 2018;11(2):1278-1284 www.ijcem.com /ISSN:1940-5901/IJCEM0063093 Case Report Dislocation and screws pull-out after application of an Isobar TTL dynamic stabilisation system at L2/3 in
Application of Rapid Prototyping for Development of Custom Made Orthopedics Prostheses: dddddd An Investigative Study ddddddddd dddddddddd
 11 Application of Rapid Prototyping for Development of Custom Made Orthopedics Prostheses: dddddd An Investigative Study ddddddddd dddddddddd Sadegh Rahmati* Department of Mechanical Engineering, Islamic
11 Application of Rapid Prototyping for Development of Custom Made Orthopedics Prostheses: dddddd An Investigative Study ddddddddd dddddddddd Sadegh Rahmati* Department of Mechanical Engineering, Islamic
ProDisc-L Total Disc Replacement. IDE Clinical Study.
 ProDisc-L Total Disc Replacement. IDE Clinical Study. A multi-center, prospective, randomized clinical trial. Instruments and implants approved by the AO Foundation Table of Contents Indications, Contraindications
ProDisc-L Total Disc Replacement. IDE Clinical Study. A multi-center, prospective, randomized clinical trial. Instruments and implants approved by the AO Foundation Table of Contents Indications, Contraindications
Salto Talaris Total Ankle Prosthesis
 Instructions for Use Salto Talaris Total Ankle Prosthesis Federal (USA) law restricts this device to sale by or on the order of a physician. Important The manufacturer recommends that all personnel responsible
Instructions for Use Salto Talaris Total Ankle Prosthesis Federal (USA) law restricts this device to sale by or on the order of a physician. Important The manufacturer recommends that all personnel responsible
MEDICAL POLICY SUBJECT: ARTIFICIAL CERVICAL INTERVERTEBRAL DISC
 MEDICAL POLICY SUBJECT: ARTIFICIAL CERVICAL PAGE: 1 OF: 13 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: ARTIFICIAL CERVICAL PAGE: 1 OF: 13 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
INTERNATIONAL JOURNAL OF PURE AND APPLIED RESEARCH IN ENGINEERING AND TECHNOLOGY
 INTERNATIONAL JOURNAL OF PURE AND APPLIED RESEARCH IN ENGINEERING AND TECHNOLOGY A PATH FOR HORIZING YOUR INNOVATIVE WORK STUDY AND ANALYSIS OF KNEE IMPLANT IN HUMAN BODY GAJANAN D. MANDAVGADE Assistant
INTERNATIONAL JOURNAL OF PURE AND APPLIED RESEARCH IN ENGINEERING AND TECHNOLOGY A PATH FOR HORIZING YOUR INNOVATIVE WORK STUDY AND ANALYSIS OF KNEE IMPLANT IN HUMAN BODY GAJANAN D. MANDAVGADE Assistant
U. AUG skeletal dynamics. 510(k) Summary of Safety and Effectiveness Skeletal Dynamics Geminus Volar Distal Radius Plate System
 U. AUG 23 2011 00 skeletal dynamics 510(k) Summary of Safety and Effectiveness Skeletal Dynamics Geminus Volar Distal Radius Plate System Submitter: Skeletal Dynamics, LLC 6905 SW 67 Avenue Suite 201 Miami,
U. AUG 23 2011 00 skeletal dynamics 510(k) Summary of Safety and Effectiveness Skeletal Dynamics Geminus Volar Distal Radius Plate System Submitter: Skeletal Dynamics, LLC 6905 SW 67 Avenue Suite 201 Miami,
NEURONETICS. 510(k) SUMMARY NEUROSTAR TMS THERAPY SYSTEM'
 NEURONETICS 510(k) SUMMARY NEUROSTAR TMS THERAPY SYSTEM' 510(k) Owner: Neuronetics, Inc. DEC 1 6 Z008 1 Great Valley Parkway, Suite 2 Malvern, PA 19355 Phone: 610-640-4202 Fax: 610-640-4206 Company Contact:
NEURONETICS 510(k) SUMMARY NEUROSTAR TMS THERAPY SYSTEM' 510(k) Owner: Neuronetics, Inc. DEC 1 6 Z008 1 Great Valley Parkway, Suite 2 Malvern, PA 19355 Phone: 610-640-4202 Fax: 610-640-4206 Company Contact:
MEDICAL POLICY. Proprietary Information of YourCare Health Plan
 MEDICAL POLICY SUBJECT: INTERVERTEBRAL DISC DECOMPRESSION: PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases,
MEDICAL POLICY SUBJECT: INTERVERTEBRAL DISC DECOMPRESSION: PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases,
Peripheral Subcutaneous Field Stimulation. Description
 Subject: Peripheral Subcutaneous Field Stimulation Page: 1 of 6 Last Review Status/Date: June 2016 Peripheral Subcutaneous Field Stimulation Description Peripheral subcutaneous field stimulation (PSFS,
Subject: Peripheral Subcutaneous Field Stimulation Page: 1 of 6 Last Review Status/Date: June 2016 Peripheral Subcutaneous Field Stimulation Description Peripheral subcutaneous field stimulation (PSFS,
THE US FDA S CUSTOM DEVICE EXEMPTION Practical Solutions for Handling the Sale of Patient-Specific Devices in the USA. August 2011 SPECIAL REPRINT
 August 2011 SPECIAL REPRINT UNDERSTANDING THE US FDA S CUSTOM DEVICE EXEMPTION Practical Solutions for Handling the Sale of Patient-Specific Devices in the USA By Danielle Woodlee Reproduced with the kind
August 2011 SPECIAL REPRINT UNDERSTANDING THE US FDA S CUSTOM DEVICE EXEMPTION Practical Solutions for Handling the Sale of Patient-Specific Devices in the USA By Danielle Woodlee Reproduced with the kind
The KineSpring Knee Implant System Product Information
 The KineSpring Knee Implant System Product Information The Treatment Gap Increasing numbers of young, active OA patients with longer life expectancy and higher activity demands. 1 Large increase in arthroplasty
The KineSpring Knee Implant System Product Information The Treatment Gap Increasing numbers of young, active OA patients with longer life expectancy and higher activity demands. 1 Large increase in arthroplasty
comprehensive Design Rationale shoulder system Knees Hips Extremities Cement and Accessories PMI TEchnology
 comprehensive shoulder system Design Rationale Knees Hips Extremities Cement and Accessories PMI TEchnology . INTRODUCTION The Comprehensive Shoulder System is an evolutionary design based on the successful
comprehensive shoulder system Design Rationale Knees Hips Extremities Cement and Accessories PMI TEchnology . INTRODUCTION The Comprehensive Shoulder System is an evolutionary design based on the successful
why bicompartmental? A REVOLUTIONARY ALTERNATIVE TO TOTAL KNEE REPLACEMENTS
 why bicompartmental? A REVOLUTIONARY ALTERNATIVE TO TOTAL KNEE REPLACEMENTS TKR is not always the answer Today, many patients with medial or lateral disease and patellofemoral involvement receive a Total
why bicompartmental? A REVOLUTIONARY ALTERNATIVE TO TOTAL KNEE REPLACEMENTS TKR is not always the answer Today, many patients with medial or lateral disease and patellofemoral involvement receive a Total
itotal Customized POSTERIOR-STABILIZED knee replacement system
 itotal PS Customized POSTERIOR-STABILIZED knee replacement system OUR VISION This is what we envision as the ideal total knee replacement (TKR) system: FOR THE SURGEON FOR THE PATIENT A REPRODUCIBLE PROCEDURE
itotal PS Customized POSTERIOR-STABILIZED knee replacement system OUR VISION This is what we envision as the ideal total knee replacement (TKR) system: FOR THE SURGEON FOR THE PATIENT A REPRODUCIBLE PROCEDURE
Protocol. Hip Resurfacing
 Protocol Hip Resurfacing (70180) Medical Benefit Effective Date: 10/01/14 Next Review Date: 11/19 Preauthorization No Review Dates: 07/12, 07/13, 07/14, 07/15, 11/15, 11/16, 11/17, 11/18 Preauthorization
Protocol Hip Resurfacing (70180) Medical Benefit Effective Date: 10/01/14 Next Review Date: 11/19 Preauthorization No Review Dates: 07/12, 07/13, 07/14, 07/15, 11/15, 11/16, 11/17, 11/18 Preauthorization
SURGICAL TECHNIQUE. Protrusio Cage A COMPREHENSIVE ACETABULAR REVISION SYSTEM
 SURGICAL TECHNIQUE Protrusio Cage A COMPREHENSIVE ACETABULAR REVISION SYSTEM Important: This essential product information does not include all of the information necessary for selection and use of a device.
SURGICAL TECHNIQUE Protrusio Cage A COMPREHENSIVE ACETABULAR REVISION SYSTEM Important: This essential product information does not include all of the information necessary for selection and use of a device.
Ultrafiltration in Decompensated Heart Failure. Description
 Subject: Ultrafiltration in Decompensated Heart Failure Page: 1 of 7 Last Review Status/Date: September 2016 Ultrafiltration in Decompensated Heart Failure Description Ultrafiltration is a technique being
Subject: Ultrafiltration in Decompensated Heart Failure Page: 1 of 7 Last Review Status/Date: September 2016 Ultrafiltration in Decompensated Heart Failure Description Ultrafiltration is a technique being
Biomechanics of. Knee Replacement. Mujda Hakime, Paul Malcolm
 Biomechanics of Knee Replacement Mujda Hakime, Paul Malcolm 1 Table of contents Knee Anatomy Movements of the Knee Knee conditions leading to knee replacement Materials Alignment and Joint Loading Knee
Biomechanics of Knee Replacement Mujda Hakime, Paul Malcolm 1 Table of contents Knee Anatomy Movements of the Knee Knee conditions leading to knee replacement Materials Alignment and Joint Loading Knee
SSSR HIP PROSTHESIS MODELS AND MATERIALS SURGICAL TECHNIQUES APPROACHES. Patrick Zingg Department of Orthopaedics University of Zurich, Balgrist
 HIP PROSTHESIS MODELS AND MATERIALS SURGICAL TECHNIQUES APPROACHES Patrick Zingg Department of Orthopaedics University of Zurich, Balgrist Zürich, Switzerland www.balgrist.ch IMPLANT TO BONE INTERFACE
HIP PROSTHESIS MODELS AND MATERIALS SURGICAL TECHNIQUES APPROACHES Patrick Zingg Department of Orthopaedics University of Zurich, Balgrist Zürich, Switzerland www.balgrist.ch IMPLANT TO BONE INTERFACE
Medical Policy. MP Dynamic Spinal Visualization
 Medical Policy MP 6.01.46 BCBSA Ref. Policy: 6.01.46 Last Review: 03/29/2018 Effective Date: 03/29/2018 Section: Radiology End Date: 12/14/2018 Related Policies 6.01.48 Positional Magnetic Resonance Imaging
Medical Policy MP 6.01.46 BCBSA Ref. Policy: 6.01.46 Last Review: 03/29/2018 Effective Date: 03/29/2018 Section: Radiology End Date: 12/14/2018 Related Policies 6.01.48 Positional Magnetic Resonance Imaging
 2nd International Workshop on Clinical Spine and Orthopedics Biomechanics 13th to 15th April, 2018 Auditorium, ISIC Program 13 th April, 2018: Clinical Orthopaedic biomechanics Time Topic 0900-1015 Session
2nd International Workshop on Clinical Spine and Orthopedics Biomechanics 13th to 15th April, 2018 Auditorium, ISIC Program 13 th April, 2018: Clinical Orthopaedic biomechanics Time Topic 0900-1015 Session
Answer. Hip System Design Features:
 Table of Contents Alliance Revision Balance Stanmore Bio-Moore II Taperloc Mallory-Head P.O. Box 587, Warsaw, IN 46581-0587 574.267.6639 2003 Biomet, Inc. All Rights Reserved web site: www.biomet.com email:
Table of Contents Alliance Revision Balance Stanmore Bio-Moore II Taperloc Mallory-Head P.O. Box 587, Warsaw, IN 46581-0587 574.267.6639 2003 Biomet, Inc. All Rights Reserved web site: www.biomet.com email:
Metha Short Hip Stem System
 Metha Short Hip Stem System Accuracy That Stands Alone Aesculap Orthopaedics Metha Short Hip Stem System Designed For Anatomic Accuracy The Metha Short Hip Stem is designed for anatomic accuracy to restore
Metha Short Hip Stem System Accuracy That Stands Alone Aesculap Orthopaedics Metha Short Hip Stem System Designed For Anatomic Accuracy The Metha Short Hip Stem is designed for anatomic accuracy to restore
CAUTION: Ceramic liners are not approved for use in the United States.
 Total Hip Prostheses, Self-Centering Hip Prostheses and Hemi-Hip Prostheses IMPORTANT: This essential product information sheet does not include all of the information necessary for selection and use of
Total Hip Prostheses, Self-Centering Hip Prostheses and Hemi-Hip Prostheses IMPORTANT: This essential product information sheet does not include all of the information necessary for selection and use of
SURGICAL TECHNIQUE CEMENTED & PRESS-FIT UNIFIED INSTRUMENTATION INTRAOPERATIVE FLEXIBILITY PROVEN BIOMECHANICS
 SURGICAL TECHNIQUE CEMENTED & PRESS-FIT UNIFIED INSTRUMENTATION INTRAOPERATIVE FLEXIBILITY PROVEN BIOMECHANICS INTRODUCTION The Summit Tapered Hip System s comprehensive set of implants and instruments
SURGICAL TECHNIQUE CEMENTED & PRESS-FIT UNIFIED INSTRUMENTATION INTRAOPERATIVE FLEXIBILITY PROVEN BIOMECHANICS INTRODUCTION The Summit Tapered Hip System s comprehensive set of implants and instruments
Sasaki E 1,2, Otsuka H 2, Sasaki N 2, and Ishibashi Y 1
 Influence of osteophyte resection of the posterior femoral condyle on extension range of motion and gap balance in cruciate retaining type total knee arthroplasty. - Intraoperative evaluation using navigation
Influence of osteophyte resection of the posterior femoral condyle on extension range of motion and gap balance in cruciate retaining type total knee arthroplasty. - Intraoperative evaluation using navigation
February 15, AtriCure, Inc. Melissa Smallwood Regulatory Affairs Specialist 7555 Innovation Way Mason, Ohio 45040
 February 15, 2018 AtriCure, Inc. Melissa Smallwood Regulatory Affairs Specialist 7555 Innovation Way Mason, Ohio 45040 Re: K180137 Trade/Device Name: AtriCure cryoice cryo-ablation probe (CRYO3), AtriCure
February 15, 2018 AtriCure, Inc. Melissa Smallwood Regulatory Affairs Specialist 7555 Innovation Way Mason, Ohio 45040 Re: K180137 Trade/Device Name: AtriCure cryoice cryo-ablation probe (CRYO3), AtriCure
Mini-Incision THA. What Qualifies? A Rose by Any Other Name? Todd V. Swanson, MD Las Vegas, Nevada, USA
 2 nd nd Annual Impact of Emerging Technology on Hip and Knee Reconstruction November 16, 2003 Mini-Incision THA Todd V. Swanson, MD Las Vegas, Nevada, USA A Rose by Any Other Name? Mini-Incision Surgery?
2 nd nd Annual Impact of Emerging Technology on Hip and Knee Reconstruction November 16, 2003 Mini-Incision THA Todd V. Swanson, MD Las Vegas, Nevada, USA A Rose by Any Other Name? Mini-Incision Surgery?
Zimmer Segmental System
 Zimmer Segmental System Simple solutions for solving complex salvage cases A Step Forward The Zimmer Segmental System is designed to address patients with severe bone loss associated with disease, trauma
Zimmer Segmental System Simple solutions for solving complex salvage cases A Step Forward The Zimmer Segmental System is designed to address patients with severe bone loss associated with disease, trauma
