VIDRL & WHO Measles IgM Proficiency Panel Final Report Prepared by Jennie Leydon, Michaela Riddell & Dr Mike Catton Director VIDRL
|
|
|
- Griselda Lorin Johnston
- 5 years ago
- Views:
Transcription
1 VIDRL & WHO Measles IgM Proficiency Panel Final Report Prepared by Jennie Leydon, Michaela Riddell & Dr Mike Catton Director VIDRL
2 Table of Contents Tables and Figures... 3 Abbreviations... 4 Introduction... 5 Aim... 5 Methods... 5 Panel composition... 5 Validation of panel... 6 Distribution of panel... 7 Analysis... 7 Results (measles)... 8 Reporting of kit details... 8 Results analysed by kit... 9 Results analysed by panel number... 2 Results (rubella)... 8 Reporting of kit in use... 3 Discrepant results... 4 Discussion
3 Tables and Figures Table : Table 2: Table 3: Table 4: Table 5: Table 6: Panel composition detailing IgM status of panel number The assays used versus the number of correct results Percentage of laboratories correctly identifying all positive and negatives by assay used Scores achieved by participating laboratories The percent of laboratories that reported the correct result for each individual panel number Result classification for those panel samples which were not correctly identified for measles IgM Table 7: Details of result classification for those panel samples which were not correctly identified for rubella IgM Figure : Figure 2: Figure 3: Methods in use for measles IgM OD values for each positive panel sample for laboratories using the Dade Behring kit OD values for each negative panel sample for laboratories using the Dade Behring assay Figure 4: OD value of all panel samples for laboratories using a selection of commercial assays other than DadeBehring Figure 5: Methods in use for rubella IgM 3
4 Abbreviations ANOVA CDC EMRO N OD P QA S/CO VIDRL WHO Analysis of variance Centers for Disease Control and Prevention Eastern Mediterranean Regional Office Negative Optical density Positive Quality assurance Sample/cut-off ratio Victorian Infectious Diseases Reference Laboratory World Health Organization 4
5 Measles IgM proficiency panel 2002 Panel number: Introduction As the world moves towards control of measles, confirmation of clinically diagnosed measles by IgM serology will become increasingly important. Proficiency testing is an important part of measles laboratory programs as both false positive and false negative results can occur with some of the commonly used measles IgM enzyme immunoassays (EIA). Aim The aims of this panel are to. Assess the proficiency of laboratories with-in the WHO laboratory network 2. Identify problems with any assays routinely used in these laboratories 3. Check the accuracy of data reporting. Methods Panel composition This panel was a renumbered version of the 0080 panel distributed between promulgation of the correct results of the 0080 panel and distribution of the next annual panel (0002) All samples were undiluted serum samples, comprising 0 Measles IgM positive (sourced from 999 measles outbreak in Victoria, Australia) 5 Measles IgM negative (VIDRL staff volunteers) 2 Parvovirus IgM positive (Diagnostic sera) 2 Rubella IgM positive (Diagnostic sera) Dengue IgM positive (supplied by the WHO Arbovirus Reference Lab, Qld. Australia) All samples were negative for HIV, Hepatitis BsAg & Hepatitis C. Table details the composition of the panel by sample number and validated result of testing. 5
6 WHO Panel Results PANEL 0080 Sample Measles IgM Rubella IgM Status Positive Negative MEASLES IgM POSITIVE Positive Negative Positive Negative Positive Negative Positive Negative Negative Negative PARVO IgM POSITIVE Negative Positive RUBELLA IgM POSITIVE Negative Negative DENGUE IgM POSITIVE Negative Negative PARVO IgM POSITIVE Negative Negative MEASLES IgM NEGATIVE Negative Negative Negative Negative Positive Negative MEASLES IgM POSITIVE Positive Negative Negative Positive* MEASLES IgM NEGATIVE Positive Negative MEASLES IgM POSITIVE Positive Negative Negative Negative MEASLES IgM NEGATIVE Negative Positive RUBELLA IgM POSITIVE Positive Negative MEASLES IgM POSITIVE *False positive result Table : Panel composition detailing IgM status of panel number. Validation of panel The panel was tested at VIDRL using two methods for Measles IgM: and two methods for Rubella IgM. DadeBehring Enzygnost anti- measles virus IgM Chemicon Light Diagnostics Measles IgM Capture Enzyme Immunoassay 6
7 Beckman Access Chemiluminescent Rubella IgM DiaSorin ETI-RUBEK-M reverse PLUS capture assay Before general distribution the panel was tested for measles IgM by CDC, Atlanta USA and the Central Public Health Laboratory, Colindale UK. The results obtained by the other two laboratories confirmed those obtained by VIDRL. Distribution of panel Results were returned from 7 laboratories from WHO EMRO region. Each laboratory has been assigned a dedicated number. This number is known only to VIDRL and that laboratory. The countries that returned results were: Bahrain Cyprus Egypt Iran Iraq Jordan Lebanon Morocco Oman Pakistan Qatar Saudi Arabia Syria Tunisia United Arab Emirates Yemen 7
8 Analysis Analysis was performed by laboratory and panel number. A range of kits was used by participating laboratories. The group using Dade Behring kits was the only one large enough to compare results as there were insufficient numbers for comparison in the other groups. The proportion of correct results, based on the positive/negative interpretation reported by the laboratory, was calculated by laboratory and according to kit used. The Dade Behring group The laboratory assigned optical density (OD) values and interpretation (positive/negative) were recorded for each of the panel numbers. The positive/negative cut off was assumed to be 0.2 unless stated otherwise. OD values for all positive samples were graphed for all laboratories and inspected for normality. This inspection was repeated separately for all negative samples. 0Other kit groups Three laboratories used the Chemicon assay, two laboratories used the Virion assay. The remaining five laboratories used a variety of assay methods. No comparative analysis was attempted since there were insufficient numbers of laboratories using the same kit for meaningful analysis. The OD values obtained by laboratories using commercial EIA kits was plotted but no further comparisons could be made. Reporting of kit details Lot numbers All laboratories supplied lot or reagent details Expiry dates laboratory did not supply an expiry date. 8
9 Results analysed by kit Kit Details Chemicon DadeBehring Diagnostic Automation Radim Virion UDI Biotest Human Figure : Methods in use for measles IgM testing Table 2 demonstrates the number of correct results by kit. Assay Number Number of samples correctly identified of labs using kit Chemicon 3 2 Dade Behring 7 4 2* Other 7 6 * laboratory only tested 9 samples Table 2: The assay used versus the number of correct results that each laboratory achieved. Assay Number of laboratories using assay Proportion of laboratories with all positives correct Proportion of laboratories with all negatives correct Dade Behring 7 7% 7% Other commercial kits 0 00% 80% Table 3: The proportion of laboratories correctly identifying all positive and negatives by assay. 9
10 Dade Behring results- IgM positive samples O. D. val ue Laboratory Number Figure 2: OD values for each positive panel sample by laboratory for laboratories using the DadeBehring kit. The cut-off is 0.2 OD units. Dade Behring- IgM negative samples O.D. value Laboratory Number Figure 3: OD values for each negative panel sample by laboratory for laboratories using the Dade Behring assay. The cut-off is 0.2 OD units, a true negative is <0.OD units and equivocal range OD units. Other commercial assays
11 Figure 4: OD value of all panel samples for laboratories using a selection of commercial assays other than DadeBehring. Laboratories 5 & 0 used the Chemicon assay and laboratories 37 & 74 used the Virion assay. Results analysed by panel number Twelve (2) laboratories achieved a perfect score (20/20) SCORE NUMBER OF LABS 20/20 2 9/20 2 8/20 4/20 /20 TOTAL 7 Table 4: Scores achieved by participating laboratories assessed by the P/N interpretations as returned to VIDRL.
12 Results by panel number Panel no. Measles IgM status Number correct Panel no. Measles IgM status Number correct P P P P P N N N N N 7 00% 7 00% 7 00% 7 00% 7 00% 5 88% 7 00% 5 88% 5 88% 6 94% N N P P N P P N N P 5 88% 5 88% 7 00% 5 88% 4 82% 7 00% 7 00% 6 94% 5 88% 7 00% Table 5: Percentage of laboratories reporting correct result for each individual panel number. Analysis of discrepant results Panel no Measles N N N N N N P N N N IgM status Positive Negative Equivocal Not tested Total Table 6: Details of result classification for those panel samples which were not correctly identified by reporting laboratories. 2
13 Rubella serology Fifteen laboratories returned results for rubella IgM. Nine laboratories tested the whole panel for Rubella IgM, and six laboratories tested between 3 and samples. Kit Details Biokit Biochem Biorad DadeBehring Human Organon Tecknika Radim UDI Diagnostics Roche Figure 5: Methods in use for rubella IgM testing Analysis of discrepant results Panel No Diagnosis Measles Measles Healthy adult Measles Rubella IgM POS 2 Rubella IgM EQUIV Table 7: Details of result classification for those panel samples which were not correctly identified by reporting laboratories for rubella IgM. 3
14 Discussion Measles The panel was distributed to National and Regional Measles reference laboratories within the WHO EMRO measles network and 7 laboratories returned results for analysis. The results overall were very encouraging. Eighty-two percent of laboratories achieved a score of 90% or greater. A score of 00%was achieved by 7% of laboratories. The Dade Behring Enzygnost IgM measles kit was used by 4% of laboratories for investigating measles so we were able to compare the reactivity of samples for these laboratories. The number of users of other kits was too small for any comparisons. Among users of Dade Behring EIAs there were 3 samples where discrepant results were reported.. Sample 02, reported positive by lab 03 (healthy volunteer) 2. Sample 04, reported negative by lab 09 (actually in equivocal range) and equivocal by lab 03(this sample was from a young adult with laboratory confirmed measles by serology &PCR) 3. Sample 05, reported equivocal by lab 025 (healthy volunteer) Laboratory 025 had O.D. values noticeably higher than the other Dade Behring users and may wish to review their protocols. There were 0 sera where discrepant results were reported. One was a measles IgM positive sample the other nine were measles IgM negative samples (5 healthy volunteers, 2 parvovirus IgM positive, dengue IgM positive and rubella IgM positive). Two laboratories reported the majority of discrepant results. These two laboratories (00 and 075) were using Chemicon and Biotest assays respectively. Laboratory 075 did not give a batch number or expiry date for the kit used. 4
15 Most users of alternative assays to DadeBehring had perfect scores. The exceptions were laboratories 00 and 075 using Chemicon & Biotest assays respectively. Laboratory 00 used the same kit and lot number as laboratory 0 who achieved a perfect score. These laboratories obtained false positive results on nine samples (00, 0, 02, 05, 08, 006, 009, 008, & 09, 5 healthy volunteers, 2 parvovirus IgM positives, dengue IgM positive and rubella IgM positive respectively)..the discrepant results were very close to the cut-off value in contrast to the true positives which gave significant O.D. values. Good data reporting is just as important as obtaining the correct result. Three laboratories did not supply a cut-off value and one laboratory did not supply any values for the samples tested. Rubella Although not designed as a rubella QAP panel,testing this measles QAP for rubella gave an opportunity for some comparative rubella testing on well characterised samples. Sample 05 was an unsatisfactory sample for Rubella IgM testing as it is known to be IgM positive by a number of commercial kits but is not from a patient with a clinical illness. Excluding sample 05 from the analysis 67% of laboratories reported all samples that they tested correctly, 2 laboratories reported only one incorrect result and another 2 laboratories had 2 incorrect results. One laboratory tested 0 samples and reported them all positive as they diluted all samples :00 (due to insufficient volumes) and then multiplied the result by 00. Excluding the results of the laboratory that altered the kit protocol and reported all samples as positive there was 4 false positive results reported :3 were measles IgM positive samples and one was a healthy volunteer. 5
UKNEQAS Toxoplasma Serology. It is the fault of the kits or is it?
 UKNEQAS Toxoplasma Serology It is the fault of the kits or is it? Purpose of the Schemes The Toxoplasma subschemes consist of IgG and IgM and are designed to: Provide information allowing participants
UKNEQAS Toxoplasma Serology It is the fault of the kits or is it? Purpose of the Schemes The Toxoplasma subschemes consist of IgG and IgM and are designed to: Provide information allowing participants
FINAL REPORT HBV Serology External Quality Assessment Scheme
 FINAL REPORT HBV Serology External Quality Assessment Scheme Programme codes: HBVC4310, HBVC435 Panel ID: 2008/Mar/11 HBV Serology EQAS Panel ID 2008/Mar/11 Page 1 of 9 The NRL is a: NATA-accredited proficiency
FINAL REPORT HBV Serology External Quality Assessment Scheme Programme codes: HBVC4310, HBVC435 Panel ID: 2008/Mar/11 HBV Serology EQAS Panel ID 2008/Mar/11 Page 1 of 9 The NRL is a: NATA-accredited proficiency
your liver Care for Think about hepatitis
 your liver Care for Think about hepatitis World Hepatitis Day 2015 What is hepatitis? Hepatitis is the common name for all inflammatory diseases of the liver. Liver inflammation is most often caused by
your liver Care for Think about hepatitis World Hepatitis Day 2015 What is hepatitis? Hepatitis is the common name for all inflammatory diseases of the liver. Liver inflammation is most often caused by
2. Are data reliable and complete?
 2. Are data reliable and complete? Tom Hiatt WHO/HQ/Stop TB TB monitoring and evaluation unit hiattt@who.int Data quality: what is it? 1. Data Quality: The Accuracy Dimension Jack E. Olson "Data has quality
2. Are data reliable and complete? Tom Hiatt WHO/HQ/Stop TB TB monitoring and evaluation unit hiattt@who.int Data quality: what is it? 1. Data Quality: The Accuracy Dimension Jack E. Olson "Data has quality
HIV Serology Quality Assessment Program Summary for Panel HIVS Oct22
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Serology Quality Assessment Program Summary
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Serology Quality Assessment Program Summary
Parvovirus B19 Mixed Titer Performance Panel PVP201
 A SERACARE PANEL PRODUCT INTENDED USE The is a panel of naturally occurring plasma specimens. This panel is intended for use by diagnostic manufacturers and clinical laboratories to evaluate their Parvovirus
A SERACARE PANEL PRODUCT INTENDED USE The is a panel of naturally occurring plasma specimens. This panel is intended for use by diagnostic manufacturers and clinical laboratories to evaluate their Parvovirus
Programmatic Feasibility of Measles Elimination
 Programmatic Feasibility of Measles Elimination World Health Organization Eastern Mediterranean Region June 2010 Acronyms: EMR Eastern Mediterranean Region of the World Health Organization 1 EMRO EPI GAVI
Programmatic Feasibility of Measles Elimination World Health Organization Eastern Mediterranean Region June 2010 Acronyms: EMR Eastern Mediterranean Region of the World Health Organization 1 EMRO EPI GAVI
Serology and International units
 Serology and International units L. Grangeot-Keros, National Reference Laboratory for Rubella, Virology Department, A. Béclère Hospital, Clamart, France Detection of rubella-specific IgG antibody Assays
Serology and International units L. Grangeot-Keros, National Reference Laboratory for Rubella, Virology Department, A. Béclère Hospital, Clamart, France Detection of rubella-specific IgG antibody Assays
Brigitte Khoury, Ph.D. Director, Arab Regional Center for Research, Training and Policy Making in Mental Health Dept. of Psychiatry, American
 Brigitte Khoury, Ph.D. Director, Arab Regional Center for Research, Training and Policy Making in Mental Health Dept. of Psychiatry, American University of Beirut UNESCO s 2010 Science Report on Scientific
Brigitte Khoury, Ph.D. Director, Arab Regional Center for Research, Training and Policy Making in Mental Health Dept. of Psychiatry, American University of Beirut UNESCO s 2010 Science Report on Scientific
HIV Serology Quality Assessment Program Summary for Panel HIVSER 2017Apr19
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Serology Quality Assessment Program Summary
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Serology Quality Assessment Program Summary
12-14 October 2009, WHO, Geneva
 The 7 th WHO Global Measles and Rubella Laboratory Network Meeting 12-14 October 2009, WHO, Geneva Summary and Recommendations Global and Regional Update The measles and rubella laboratory network (LabNet)
The 7 th WHO Global Measles and Rubella Laboratory Network Meeting 12-14 October 2009, WHO, Geneva Summary and Recommendations Global and Regional Update The measles and rubella laboratory network (LabNet)
HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2017Apr19
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2017Apr19
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2017Apr19
HIV Serology Quality Assessment Program Summary for Panel HIVSER 2018Apr19
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Serology Quality Assessment Program Summary
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Serology Quality Assessment Program Summary
HTLV Serology Quality Assessment Program Summary for Panel HTLS Oct22
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLS425 2015Oct22
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLS425 2015Oct22
HIV Serology Quality Assessment Program Revised Summary for Panel HIVSER 2017Oct27
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Serology Quality Assessment Program Revised
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Serology Quality Assessment Program Revised
HIV Testing and Systems for Managing HIV Implementation in Resource Limited Settings
 HIV Testing and Systems for Managing HIV Implementation in Resource Limited Settings Elizabeth M. Dax with the staff of the National Serology Reference Laboratory, Australia My Background Worked in HIV
HIV Testing and Systems for Managing HIV Implementation in Resource Limited Settings Elizabeth M. Dax with the staff of the National Serology Reference Laboratory, Australia My Background Worked in HIV
The Jordan University Hospital, Amman, Jordan
 The Jordan University Hospital, Amman, Jordan Pneumococcal vaccine and overcoming the immunization rift Najwa Khuri-Bulos JUH Amman, Jordan February 13 2008 Pneumococcal disease burden 1.6 million deaths
The Jordan University Hospital, Amman, Jordan Pneumococcal vaccine and overcoming the immunization rift Najwa Khuri-Bulos JUH Amman, Jordan February 13 2008 Pneumococcal disease burden 1.6 million deaths
Foot and Mouth Disease Middle East situation Summary of Answers to the Questionnaire Beirut, Lebanon, 7 9 April 2009
 Foot and Mouth Disease Middle East situation Summary of Answers to the Questionnaire Beirut, Lebanon, 7 9 April 2009 Dr Pierre Primot OIE for the Middle East Purpose of the Questionnaire The purpose of
Foot and Mouth Disease Middle East situation Summary of Answers to the Questionnaire Beirut, Lebanon, 7 9 April 2009 Dr Pierre Primot OIE for the Middle East Purpose of the Questionnaire The purpose of
Dr Ghazi Yehia OIE Regional Representative for the Middle East
 Foot and mouth disease control strategies in North Africa and the Middle East The current situation Asuncion, Paraguay, 24-26 June 2009 Dr Ghazi Yehia OIE Regional Representative for the Middle East Acknowledgements
Foot and mouth disease control strategies in North Africa and the Middle East The current situation Asuncion, Paraguay, 24-26 June 2009 Dr Ghazi Yehia OIE Regional Representative for the Middle East Acknowledgements
Chapter 4. Antibody detection methods for laboratory confirmation of measles, rubella, and CRS
 Chapter 4. Antibody detection methods for laboratory confirmation of measles, rubella, and CRS In this chapter: 4.1 Selection and comparison of EIAs for IgM detection 4.2 Interpretation of IgM results
Chapter 4. Antibody detection methods for laboratory confirmation of measles, rubella, and CRS In this chapter: 4.1 Selection and comparison of EIAs for IgM detection 4.2 Interpretation of IgM results
True Status A B. HTLV I Positive. Indeterminate. HTLV I/II Negative HV15 HV16 HV18 HV21 HV22 HV44 HV55. Selected Legend: Flagged: Unresolved
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 206Apr2 Panel
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 206Apr2 Panel
Arbovirus Reports 2015
 Arbovirus Reports Arboviruses (Arthropod-borne) are a group of viral infections transmitted by the bite of arthropods, most commonly mosquitoes. Some of these infections are endemic; others may be imported
Arbovirus Reports Arboviruses (Arthropod-borne) are a group of viral infections transmitted by the bite of arthropods, most commonly mosquitoes. Some of these infections are endemic; others may be imported
HIV Viral Load Quality Assessment Program Summary for Panel HIVVL 2018Oct26
 1 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Viral Load Quality Assessment Program Summary
1 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Viral Load Quality Assessment Program Summary
HIV Viral Load Quality Assessment Program Summary for Panel HIVVL 2017Oct27
 1 The National Laboratory for HIV Reference Services is Accredited to ISO 15189 and ISO 17043 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology
1 The National Laboratory for HIV Reference Services is Accredited to ISO 15189 and ISO 17043 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology
Annex 3. Meetings held in the Eastern Mediterranean Region, 2014
 Annex 3. Meetings held in the Eastern Mediterranean Region, Statutory and advisory meetings Expanded meeting for members of the Advisory Committee on Health Research to discuss integrating research in
Annex 3. Meetings held in the Eastern Mediterranean Region, Statutory and advisory meetings Expanded meeting for members of the Advisory Committee on Health Research to discuss integrating research in
CEPHIA Consortium for the Evaluation and Performance of HIV Incidence Assays STANDARD OPERATING PROCEDURE
 CEPHIA Consortium for the Evaluation and Performance of HIV Incidence Assays STANDARD OPERATING PROCEDURE TITLE : SOP for (off board dilution) Less Sensitive Modified VITROS Enzyme Immunoassay CEPHIA DOCUMENT
CEPHIA Consortium for the Evaluation and Performance of HIV Incidence Assays STANDARD OPERATING PROCEDURE TITLE : SOP for (off board dilution) Less Sensitive Modified VITROS Enzyme Immunoassay CEPHIA DOCUMENT
Progress report on emerging and re-emerging diseases including dengue and dengue haemorrhagic fever
 Regional Committee for the Eastern Mediterranean Sixty-third session Provisional agenda item 3(c) EM/RC63/INF.DOC 2 Rev.2 September 2016 Introduction Progress report on emerging and re-emerging diseases
Regional Committee for the Eastern Mediterranean Sixty-third session Provisional agenda item 3(c) EM/RC63/INF.DOC 2 Rev.2 September 2016 Introduction Progress report on emerging and re-emerging diseases
Eastern Mediterranean Region Framework for health information systems and core indicators for monitoring health situation and health system
 Eastern Mediterranean Region Framework for health information systems and core indicators for monitoring health situation and health system performance 2017 Eastern Mediterranean Region Framework for
Eastern Mediterranean Region Framework for health information systems and core indicators for monitoring health situation and health system performance 2017 Eastern Mediterranean Region Framework for
Regional & National Partnerships to Stop TB in the Eastern Mediterranean Region-lessons learnt
 Regional & National Partnerships to Stop TB in the Eastern Mediterranean Region-lessons learnt Dr. Mohamed Awad Tag Eldin Chair, Coordinating Board of the Eastern Mediterranean Partnership to Stop TB Eastern
Regional & National Partnerships to Stop TB in the Eastern Mediterranean Region-lessons learnt Dr. Mohamed Awad Tag Eldin Chair, Coordinating Board of the Eastern Mediterranean Partnership to Stop TB Eastern
Maternal Health in Arab States
 Maternal Health in Arab States Improving Maternal Health Improving maternal health is MDG number 5 and it has two targets Target A that measures reduction in maternal mortality ratio (MMR) and attended
Maternal Health in Arab States Improving Maternal Health Improving maternal health is MDG number 5 and it has two targets Target A that measures reduction in maternal mortality ratio (MMR) and attended
The global progress towards poliomyelitis
 Communicable diseases Poliomyelitis eradication The global progress towards poliomyelitis eradication in 2014 was substantial. However, the disease remains endemic in the Region. Of the 215 polio globally
Communicable diseases Poliomyelitis eradication The global progress towards poliomyelitis eradication in 2014 was substantial. However, the disease remains endemic in the Region. Of the 215 polio globally
Global Measles and Rubella Update November 2018
 Global Measles and Rubella Update November 218 Measles Number of Reported Measles by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug Sep
Global Measles and Rubella Update November 218 Measles Number of Reported Measles by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug Sep
H. pylori IgM CLIA kit
 H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2016Oct28
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2016Oct28
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory HTLV Serology Quality Assessment Program Summary for Panel HTLVSER 2016Oct28
Rubella virus IgG ELISA Kit
 Rubella virus IgG ELISA Kit Catalog Number KA0223 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Rubella virus IgG ELISA Kit Catalog Number KA0223 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
HIV-1 AccuVert Seroconversion Panel (PRB966) / Batch #
 s/co c/ml DATA SHEET OVERVIEW 0600-0248 (PRB966) is a 10-member panel of undiluted, naturally occurring plasma samples. Panel members represent serial bleeds collected from a single patient over the course
s/co c/ml DATA SHEET OVERVIEW 0600-0248 (PRB966) is a 10-member panel of undiluted, naturally occurring plasma samples. Panel members represent serial bleeds collected from a single patient over the course
CHEMILUMINESCENCE ENZYME IMMUNOASSAY (CLIA) Toxoplasma IgG. Cat # (20-25 C Room temp.) Volume
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Acute parvovirus B19 infection frequently causes false positive results in the
 CVI Accepts, published online ahead of print on 0 December 00 Clin. Vaccine Immunol. doi:0./cvi.000-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
CVI Accepts, published online ahead of print on 0 December 00 Clin. Vaccine Immunol. doi:0./cvi.000-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
The study was cross-sectional, conducted during the academic year 2004/05.
 Tobacco use is one of the chief preventable risk factors of some 25 noncommunicable diseases, many of which are eventually fatal. The World Health Organization (WHO) attributes some 4 million deaths annually
Tobacco use is one of the chief preventable risk factors of some 25 noncommunicable diseases, many of which are eventually fatal. The World Health Organization (WHO) attributes some 4 million deaths annually
WORLD HEALTH ORGANIZATION REGIONAL OFFICE FOR THE WESTERN PACIFIC
 WORLD HEALTH ORGANIZATION REGIONAL OFFICE FOR THE WESTERN PACIFIC REPORT WORKSHOP ON LABORATORY SURVEILLANCE FOR MEASLES ELIMINATION IN THE WESTERN PACIFIC REGION Manila, Philippines 24-25 August 2004
WORLD HEALTH ORGANIZATION REGIONAL OFFICE FOR THE WESTERN PACIFIC REPORT WORKSHOP ON LABORATORY SURVEILLANCE FOR MEASLES ELIMINATION IN THE WESTERN PACIFIC REGION Manila, Philippines 24-25 August 2004
AccuVert HIV-1 Seroconversion Panel PRB955 ( ) / Batch #
 Signal to cutoff (s/co) Copies per ml (c/ml) OVERVIEW AccuVert is a 5-member validation panel of undiluted, naturally occurring plasma samples (1 vial per member, 1.0 ml per vial). Panel members represent
Signal to cutoff (s/co) Copies per ml (c/ml) OVERVIEW AccuVert is a 5-member validation panel of undiluted, naturally occurring plasma samples (1 vial per member, 1.0 ml per vial). Panel members represent
For Research Use Only
 Rubella IgM capture EIA An Enzyme Immunoassay for the detection of rubella virus specific IgM in human oral fluid, serum and plasma samples. Cat. No: RuVM014 For Research Use Only Designed & Developed
Rubella IgM capture EIA An Enzyme Immunoassay for the detection of rubella virus specific IgM in human oral fluid, serum and plasma samples. Cat. No: RuVM014 For Research Use Only Designed & Developed
Progress report on the achievement of the health-related Millennium Development Goals and global health goals after 2015
 Regional Committee for the Eastern Mediterranean Fifty-ninth session Provisional agenda item 2 (c) EM/RC59/INF.DOC.3 July 2012 Progress report on the achievement of the health-related Millennium Development
Regional Committee for the Eastern Mediterranean Fifty-ninth session Provisional agenda item 2 (c) EM/RC59/INF.DOC.3 July 2012 Progress report on the achievement of the health-related Millennium Development
Department of Defense Armed Forces Health Surveillance Branch Global MERS-CoV Surveillance Summary (14 DEC 2016)
 Department of Defense Armed Forces Health Surveillance Branch Global MERS-CoV Surveillance Summary () For questions or comments, please contact: dha.ncr.health-surv.list.afhs-ib-alert-response@mail.mil
Department of Defense Armed Forces Health Surveillance Branch Global MERS-CoV Surveillance Summary () For questions or comments, please contact: dha.ncr.health-surv.list.afhs-ib-alert-response@mail.mil
Global Measles and Rubella Update. April 2018
 Global Measles and Rubella Update April 218 Measles Number of Reported Measles by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug Sep Oct
Global Measles and Rubella Update April 218 Measles Number of Reported Measles by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug Sep Oct
AccuVert HBV Seroconversion Panel PHM941(M) ( )
 PACKAGE INSERT PHM941(M) (0605-0061) INTENDED USE PHM941(M) (0605-0061) is a group of serial bleeds from an individual plasma donor during HBV seroconversion. This panel is intended for use by diagnostics
PACKAGE INSERT PHM941(M) (0605-0061) INTENDED USE PHM941(M) (0605-0061) is a group of serial bleeds from an individual plasma donor during HBV seroconversion. This panel is intended for use by diagnostics
Breast Cancer in the Eastern Mediterranean Region A Burden with Potential. King Hussein Cancer Center
 Breast Cancer in the Eastern Mediterranean Region A Burden with Potential Presented by: Mahmoud M. Sarhan, MD, MMM, CPE CEO & Director General Presented at: International Symposium on Breast Cancer in
Breast Cancer in the Eastern Mediterranean Region A Burden with Potential Presented by: Mahmoud M. Sarhan, MD, MMM, CPE CEO & Director General Presented at: International Symposium on Breast Cancer in
Dietary adequacy of Egyptian children with autism
 Dietary adequacy of Egyptian children with autism Presented by Dr. Nagwa Abdel Meguid Prof. of Human Genetics and special Needs, NRC,Egypt Head of the CONEM Egypt Child Brain Research Group Council for
Dietary adequacy of Egyptian children with autism Presented by Dr. Nagwa Abdel Meguid Prof. of Human Genetics and special Needs, NRC,Egypt Head of the CONEM Egypt Child Brain Research Group Council for
Measles IgM Capture EIA
 Measles IgM Capture EIA An Enzyme Immunoassay for the detection of human IgM antibodies to measles virus in oral fluid, serum and plasma samples Cat. No: MeVM010 For in-vitro diagnostic use C V Designed
Measles IgM Capture EIA An Enzyme Immunoassay for the detection of human IgM antibodies to measles virus in oral fluid, serum and plasma samples Cat. No: MeVM010 For in-vitro diagnostic use C V Designed
Diabetes management: lessons from around the globe MENA. J. Belkhadir (Morocco)
 Diabetes management: lessons from around the globe MENA J. Belkhadir (Morocco) Managing Diabetes: challenges faced in the MENA Region and strategies developed Jamal Belkhadir Diabetologist, Endocrinologist
Diabetes management: lessons from around the globe MENA J. Belkhadir (Morocco) Managing Diabetes: challenges faced in the MENA Region and strategies developed Jamal Belkhadir Diabetologist, Endocrinologist
Global Measles and Rubella Update October 2018
 Global Measles and Rubella Update October 218 Measles Number of Reported Measles Cases by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug
Global Measles and Rubella Update October 218 Measles Number of Reported Measles Cases by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug
Australian National Influenza Centres Activity
 Australian National Influenza Centres Activity 2011-12 Professor Dominic Dwyer Medical Virologist Director ICPMR and WHO NIC Westmead Hospital Sydney, NSW 2145, Australia dominic.dwyer@sydney.edu.au WHO
Australian National Influenza Centres Activity 2011-12 Professor Dominic Dwyer Medical Virologist Director ICPMR and WHO NIC Westmead Hospital Sydney, NSW 2145, Australia dominic.dwyer@sydney.edu.au WHO
The FAO/OIE FMD Global Control Strategy Joseph Domenech, OIE on behalf of the joint FAO/OIE GF TADs PPR Working Group
 The FAO/OIE FMD Global Control Strategy Joseph Domenech, OIE on behalf of the joint FAO/OIE GF TADs PPR Working Group 5 th meeting of the GF TADs Regional Steering Committee for Europe October 8 th -9th
The FAO/OIE FMD Global Control Strategy Joseph Domenech, OIE on behalf of the joint FAO/OIE GF TADs PPR Working Group 5 th meeting of the GF TADs Regional Steering Committee for Europe October 8 th -9th
Influenza is a highly infectious disease caused
 review article Oman Medical Journal [2017], Vol. 32, No. 5: 359-364 Influenza Research in the Eastern Mediterranean Region: A Review Wasiq Khan 1, Amira S. El Rifay 2, Mamun Malik 1 and Ghazi Kayali 3,4
review article Oman Medical Journal [2017], Vol. 32, No. 5: 359-364 Influenza Research in the Eastern Mediterranean Region: A Review Wasiq Khan 1, Amira S. El Rifay 2, Mamun Malik 1 and Ghazi Kayali 3,4
Eastern Mediterranean Region
 Eastern Mediterranean Region Afghanistan 16 63 15 45 2 141 Adult (15+) 9 16 8 468 11 935 % Urban 15.6 18.3 2. % Rural 84.3 81.8 8. Life expectancy at birth, 199-1995 : 43 (males), 44 (females) Infant mortality
Eastern Mediterranean Region Afghanistan 16 63 15 45 2 141 Adult (15+) 9 16 8 468 11 935 % Urban 15.6 18.3 2. % Rural 84.3 81.8 8. Life expectancy at birth, 199-1995 : 43 (males), 44 (females) Infant mortality
Toxoplasma gondii IgM ELISA Kit
 Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
SEROLOGICAL DIAGNOSIS OF DENGUE INFECTIONS
 ECDC training Workshop on laboratory diagnosis of dengue virus infections Berlin, 23 27 January 2012 SEROLOGICAL DIAGNOSIS OF DENGUE INFECTIONS Cristina Domingo Carrasco Robert Koch Institut FACILITIES
ECDC training Workshop on laboratory diagnosis of dengue virus infections Berlin, 23 27 January 2012 SEROLOGICAL DIAGNOSIS OF DENGUE INFECTIONS Cristina Domingo Carrasco Robert Koch Institut FACILITIES
Development of a Regional Framework of Action in obesity Prevention in the Eastern Mediterranean Region
 Development of a Regional Framework of Action in obesity Prevention in the Eastern Mediterranean Region Ayoub Al Jawaldeh; Regional Adviser, WHO- EMRO 2011 Political declaration on NCDs, Sustainable Development
Development of a Regional Framework of Action in obesity Prevention in the Eastern Mediterranean Region Ayoub Al Jawaldeh; Regional Adviser, WHO- EMRO 2011 Political declaration on NCDs, Sustainable Development
Design Verification. Form: /5 Rev. 007 valid of
 Design Verification g 1 Determination of Diagnostic Sensitivity and Diagnostic Specificity... 2 Crossreactivity... 3 Potentially interfering substances... 3 2 Imprecision... 4 3 Standardisation... 4 4
Design Verification g 1 Determination of Diagnostic Sensitivity and Diagnostic Specificity... 2 Crossreactivity... 3 Potentially interfering substances... 3 2 Imprecision... 4 3 Standardisation... 4 4
WHO Prequalification of In Vitro Diagnostics Programme PUBLIC REPORT. Product: Murex HIV Ag/Ab Combination Number: PQDx
 WHO Prequalification of In Vitro Diagnostics Programme PUBLIC REPORT Product: Murex HIV Ag/Ab Combination Number: PQDx 0144-043-00 Abstract Murex HIV Ag/Ab Combination with product codes 7G79-09 (GE41,
WHO Prequalification of In Vitro Diagnostics Programme PUBLIC REPORT Product: Murex HIV Ag/Ab Combination Number: PQDx 0144-043-00 Abstract Murex HIV Ag/Ab Combination with product codes 7G79-09 (GE41,
G-B 2016 Kit Instructions CAP 2016
 G-B 2016 Kit Instructions CAP 2016 Syphilis Serology Survey Table of Contents Kit Contents Kit Contents... 1 Important: Before You Begin... 1 Detailed Testing Instructions... 1 Reporting Your Results...
G-B 2016 Kit Instructions CAP 2016 Syphilis Serology Survey Table of Contents Kit Contents Kit Contents... 1 Important: Before You Begin... 1 Detailed Testing Instructions... 1 Reporting Your Results...
Toxoplasma gondii IgM ELISA Kit
 Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
HIV-1 Seroconversion Panel PRB964
 A SERACARE PANEL PRODUCT Seroconversion INTENDED USE The is a group of serial bleeds from an individual plasma donor during seroconversion. This is intended for use by diagnostics manufacturers and clinical
A SERACARE PANEL PRODUCT Seroconversion INTENDED USE The is a group of serial bleeds from an individual plasma donor during seroconversion. This is intended for use by diagnostics manufacturers and clinical
Intercountry meeting on measles and rubella control/elimination
 Report on the Intercountry meeting on measles and rubella control/elimination Cairo, Egypt, 3 5 December 2007 Report on the Intercountry meeting on measles and rubella control/elimination Cairo, Egypt,
Report on the Intercountry meeting on measles and rubella control/elimination Cairo, Egypt, 3 5 December 2007 Report on the Intercountry meeting on measles and rubella control/elimination Cairo, Egypt,
Human Immunodeficiency Virus Serology
 Participant No: _ Human Immunodeficiency Virus Serology Survey No: RT :20 Please print the document labelled Clinical Notes & Test Instructions and enter your results via myqap. Kit Information Please
Participant No: _ Human Immunodeficiency Virus Serology Survey No: RT :20 Please print the document labelled Clinical Notes & Test Instructions and enter your results via myqap. Kit Information Please
SHOKI AL DOBAI, Ph.D. Pesticide Directorate, GDPP Ministry of Agriculture & Irrigation, Yemen
 SHOKI AL DOBAI, Ph.D. Pesticide Directorate, GDPP Ministry of Agriculture & Irrigation, Yemen Problems of vector borne diseases Malaria; Leishmaniasis; Rift Valley Fever; Dengue Fever; Other vectors/ Pest
SHOKI AL DOBAI, Ph.D. Pesticide Directorate, GDPP Ministry of Agriculture & Irrigation, Yemen Problems of vector borne diseases Malaria; Leishmaniasis; Rift Valley Fever; Dengue Fever; Other vectors/ Pest
The first and only fully-automated, multiplexed solution for Measles, Mumps, Rubella and Varicella-zoster virus antibody testing
 Bio-Rad Laboratories BioPlex 2200 System BioPlex 2200 MMRV IgG Kit The first and only fully-automated, multiplexed solution for Measles, Mumps, Rubella and Varicella-zoster virus antibody testing Bio-Rad
Bio-Rad Laboratories BioPlex 2200 System BioPlex 2200 MMRV IgG Kit The first and only fully-automated, multiplexed solution for Measles, Mumps, Rubella and Varicella-zoster virus antibody testing Bio-Rad
Report of the Seminar on FMD Progressive Control. Pathway. 18 th December Beirut, Lebanon
 1 Report of the Seminar on FMD Progressive Control. Pathway 18 th December 2012 Beirut, Lebanon 1. Introduction: i. The principal aim of the FMD Seminar was to discuss the preparation of national project
1 Report of the Seminar on FMD Progressive Control. Pathway 18 th December 2012 Beirut, Lebanon 1. Introduction: i. The principal aim of the FMD Seminar was to discuss the preparation of national project
Acquired immuno-deficiency syndrome (AIDS) in the Eastern Mediterranean Region
 WHO-EMlRC/INF.DOC. Acquired immuno-deficiency syndrome (AIDS) in the Eastern Mediterranean Region. Introduction By the end of 00, an estimated 0 million people in the world were living with HIV/AIDS, million
WHO-EMlRC/INF.DOC. Acquired immuno-deficiency syndrome (AIDS) in the Eastern Mediterranean Region. Introduction By the end of 00, an estimated 0 million people in the world were living with HIV/AIDS, million
WHO Prequalification of In Vitro Diagnostics Programme
 WHO Prequalification of In Vitro Diagnostics Programme International HIV/Viral Hepatitis Co-Infection Satellite Meeting 19 July 2014, Melbourne Anita Sands Prequalification Diagnostics Team Department
WHO Prequalification of In Vitro Diagnostics Programme International HIV/Viral Hepatitis Co-Infection Satellite Meeting 19 July 2014, Melbourne Anita Sands Prequalification Diagnostics Team Department
Foot and mouth disease control strategies in North Africa and the Middle East the current
 1. Title, names and addresses of authors Foot and mouth disease control strategies in North Africa and the Middle East the current situation G. Yehia (1), P. Primot (1) (1) OIE Regional Representation
1. Title, names and addresses of authors Foot and mouth disease control strategies in North Africa and the Middle East the current situation G. Yehia (1), P. Primot (1) (1) OIE Regional Representation
P0180 SeraQ LIAISON P0180
 0 P0180 The kit insert contains a detailed protocol and should be read carefully before testing the run control to ensure optimal performance Table of contents Intended Use... 3 Key to Symbols Used...
0 P0180 The kit insert contains a detailed protocol and should be read carefully before testing the run control to ensure optimal performance Table of contents Intended Use... 3 Key to Symbols Used...
Strategy for cancer prevention and control in the Eastern Mediterranean Region
 Regional Committee for the EM/RC56/4 Eastern Mediterranean July 2009 Fifty-sixth Session Original: English Agenda item 6 (b) Technical paper Strategy for cancer prevention and control in the Eastern Mediterranean
Regional Committee for the EM/RC56/4 Eastern Mediterranean July 2009 Fifty-sixth Session Original: English Agenda item 6 (b) Technical paper Strategy for cancer prevention and control in the Eastern Mediterranean
United Kingdom National External Quality Assessment Service for Microbiology [Established 1971]
![United Kingdom National External Quality Assessment Service for Microbiology [Established 1971] United Kingdom National External Quality Assessment Service for Microbiology [Established 1971]](/thumbs/95/123501270.jpg) United Kingdom National External Quality Assessment Service for Microbiology [Established 97] UK NEQAS covers Andrology Chemistry Genetics Haematology Histopathology Immunology Leucocyte Immunophenotyping
United Kingdom National External Quality Assessment Service for Microbiology [Established 97] UK NEQAS covers Andrology Chemistry Genetics Haematology Histopathology Immunology Leucocyte Immunophenotyping
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005
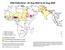 Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
FINAL REPORT HBV Serology External Quality Assessment Scheme
 FINAL REPORT HBV Serology External Quality Assessment Scheme Programme codes: HBVC4310, HBVC435 Panel ID: 2008/Oct/28 HBV Serology EQAS Panel ID 2008/Oct/28 Page 1 of 11 The NRL is a: NATA-accredited proficiency
FINAL REPORT HBV Serology External Quality Assessment Scheme Programme codes: HBVC4310, HBVC435 Panel ID: 2008/Oct/28 HBV Serology EQAS Panel ID 2008/Oct/28 Page 1 of 11 The NRL is a: NATA-accredited proficiency
The Test-Curriculum Matching Analysis: Science
 Appendix C The Test-Curriculum Matching Analysis: Science TIMSS went to great lengths to ensure that comparisons of student achievement across countries would be as fair and equitable as possible. The
Appendix C The Test-Curriculum Matching Analysis: Science TIMSS went to great lengths to ensure that comparisons of student achievement across countries would be as fair and equitable as possible. The
P0078 SeraQ ARCHITECT P0078
 P0078 SeraQ ARCHITECT P0078 The kit insert contains a detailed protocol and should be read carefully before testing the run control to ensure optimal performance Table of contents Intended Use... 3 Key
P0078 SeraQ ARCHITECT P0078 The kit insert contains a detailed protocol and should be read carefully before testing the run control to ensure optimal performance Table of contents Intended Use... 3 Key
Health Challenges and their Determinants in the EMR/MENA
 Health Challenges and their Determinants in the EMR/MENA Sameen Siddiqi, Regional Adviser, Health Policy and Planning, WHO EMRO The Middle East and North Africa Health Policy Forum Conference on Better
Health Challenges and their Determinants in the EMR/MENA Sameen Siddiqi, Regional Adviser, Health Policy and Planning, WHO EMRO The Middle East and North Africa Health Policy Forum Conference on Better
WHO Health Statistics : Applied through the lens of the Global Monitoring Framework for the Prevention and Control of Noncommunicable Diseases
 WHO Health Statistics : Applied through the lens of the Global Monitoring Framework for the Prevention and Control of Noncommunicable Diseases Dr E Tursan d'espaignet Prevention of Noncommunicable Diseases
WHO Health Statistics : Applied through the lens of the Global Monitoring Framework for the Prevention and Control of Noncommunicable Diseases Dr E Tursan d'espaignet Prevention of Noncommunicable Diseases
See external label 2 C-8 C 96 tests Chemiluminescence. CMV IgM. Cat # Diluted samples, controls & calibrator 100 µl 30 minutes
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Appendix F: Test-Curriculum Matching Analysis
 Appendix F: Test-Curriculum Matching Analysis TIMSS went to great lengths to ensure that comparisons of student achievement across countries would be as fair and equitable as possible. The TIMSS 2015 Assessment
Appendix F: Test-Curriculum Matching Analysis TIMSS went to great lengths to ensure that comparisons of student achievement across countries would be as fair and equitable as possible. The TIMSS 2015 Assessment
BDS, MSc, MSPH, MHPE, FFPH, ScD. Associate Prof. of Epidemiology and Biostatistics. Associate Prof. Medical Education
 Yusuf Al-Gau Gau d BDS, MSc, MSPH, MHPE, FFPH, ScD Associate Prof. of Epidemiology and Biostatistics Associate Prof. Medical Education Jordan University of Science & Technology Alarming high prevalence
Yusuf Al-Gau Gau d BDS, MSc, MSPH, MHPE, FFPH, ScD Associate Prof. of Epidemiology and Biostatistics Associate Prof. Medical Education Jordan University of Science & Technology Alarming high prevalence
Pharmaceuticals Import Summary Report. selected countries in MENA region
 Pharmaceuticals Import Summary Report selected countries in MENA region Contents.. 3. 3. 3. 3.3....3 5. 5. 5. 6. 6. 6. Disclaimer Global Imports. Imports in MENA Focus Pharmaceutical Categries Vaccines
Pharmaceuticals Import Summary Report selected countries in MENA region Contents.. 3. 3. 3. 3.3....3 5. 5. 5. 6. 6. 6. Disclaimer Global Imports. Imports in MENA Focus Pharmaceutical Categries Vaccines
See external label 2 C-8 C 96 tests CHEMILUMINESCENCE. CMV IgG. Cat # Step (20-25 C Room temp.) Volume
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Evaluation Report MHRA Four anti-hbc assays. 60 (Free to NHS) MHRA Report number MHRA Evaluation Report. Crown Copyright.
 November 2003 NUMBER Evaluation Report MHRA 03136 Four anti-hbc assays MHRA Evaluation Report MHRA Report number 03136 Crown Copyright 60 (Free to NHS) WHAT YOU CAN EXPECT FROM MHRA EVALUATION REPORTS
November 2003 NUMBER Evaluation Report MHRA 03136 Four anti-hbc assays MHRA Evaluation Report MHRA Report number 03136 Crown Copyright 60 (Free to NHS) WHAT YOU CAN EXPECT FROM MHRA EVALUATION REPORTS
NCCID RAPID REVIEW. 1. What are the case definitions and guidelines for surveillance and reporting purposes?
 NCCID RAPID REVIEW 1. What are the case definitions and guidelines for surveillance and reporting purposes? Middle East Respiratory Syndrome Coronavirus: Ten Questions and Answers for Canadian Public Health
NCCID RAPID REVIEW 1. What are the case definitions and guidelines for surveillance and reporting purposes? Middle East Respiratory Syndrome Coronavirus: Ten Questions and Answers for Canadian Public Health
Training Workshop Report
 Training Workshop Report The 4th Regional Hands-on Training Workshop on the Laboratory Diagnosis of Measles and Rubella focusing on Molecular Diagnosis Hong Kong (China) 22 27 November 2010 (WP)/ICP/IVD/1.1/001-A
Training Workshop Report The 4th Regional Hands-on Training Workshop on the Laboratory Diagnosis of Measles and Rubella focusing on Molecular Diagnosis Hong Kong (China) 22 27 November 2010 (WP)/ICP/IVD/1.1/001-A
LIAISON Measles IgG The fully automated solution for quantitative antibody detection
 LIAISON Measles IgG The fully automated solution for quantitative antibody detection FOR OUTSIDE THE US AND CANADA ONLY LIAISON Measles IgG Number of tests 100 Key assay features Method Assay range Solid
LIAISON Measles IgG The fully automated solution for quantitative antibody detection FOR OUTSIDE THE US AND CANADA ONLY LIAISON Measles IgG Number of tests 100 Key assay features Method Assay range Solid
AccuSet HBV Worldwide Performance Panel
 PACKAGE INSERT 0805-033 INTENDED USE The is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot HBV test methods. Characterized samples
PACKAGE INSERT 0805-033 INTENDED USE The is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot HBV test methods. Characterized samples
The Need for More and Better Palliative Care in Muslim-Majority Countries
 The Need for More and Better Palliative Care in Muslim-Majority Countries Joe Harford, Ph.D. Center for Global Health National Cancer Institute Email: harfordj@nih.gov Deena M. Aljawi King Faisal Specialist
The Need for More and Better Palliative Care in Muslim-Majority Countries Joe Harford, Ph.D. Center for Global Health National Cancer Institute Email: harfordj@nih.gov Deena M. Aljawi King Faisal Specialist
MICROWELL ELISA Measles IgM Catalog #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
B19 Virus EQA Programme Final Report QAV (B19DNA14)
 B19 Virus 2014 EQA Programme Final Report QAV034116 (B19DNA14) Prof. Hubert GM Niesters Scientific Expert on behalf of QCMD Report authorised by the QCMD Executive in July 2014 A UKAS accredited proficiency
B19 Virus 2014 EQA Programme Final Report QAV034116 (B19DNA14) Prof. Hubert GM Niesters Scientific Expert on behalf of QCMD Report authorised by the QCMD Executive in July 2014 A UKAS accredited proficiency
WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT. Product: Alere HIV Combo WHO reference number: PQDx
 WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT Product: Alere HIV Combo WHO reference number: PQDx 0243-013-00 Alere HIV Combo with product codes 7D2842, 7D2843, 7D2843SET manufactured by Alere
WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT Product: Alere HIV Combo WHO reference number: PQDx 0243-013-00 Alere HIV Combo with product codes 7D2842, 7D2843, 7D2843SET manufactured by Alere
Innovation in Diagnostics. ToRCH. A complete line of kits for an accurate diagnosis INFECTIOUS ID DISEASES
 Innovation in Diagnostics ToRCH A complete line of kits for an accurate diagnosis INFECTIOUS ID DISEASES EN TOXOPLASMOSIS Toxoplasmosis is a parasitic disease caused by with the obligate intracellular
Innovation in Diagnostics ToRCH A complete line of kits for an accurate diagnosis INFECTIOUS ID DISEASES EN TOXOPLASMOSIS Toxoplasmosis is a parasitic disease caused by with the obligate intracellular
Evaluation of Asthma Management in Middle EAst North Africa Adult population
 STUDY REPORT SUMMARY Evaluation of Asthma Management in Middle EAst North Africa Adult population Descriptive study on the management of asthma in an asthmatic Middle East Africa adult population Background/Rationale:
STUDY REPORT SUMMARY Evaluation of Asthma Management in Middle EAst North Africa Adult population Descriptive study on the management of asthma in an asthmatic Middle East Africa adult population Background/Rationale:
AccuSet HCV Performance Panel
 Signal to cutoff (s/co) DATA SHEET OVERVIEW AccuSet HCV (0810-0204) is a 25-member validation panel of undiluted, naturally occurring plasma samples (1 vial per member, 1.0 ml per vial). Panel members
Signal to cutoff (s/co) DATA SHEET OVERVIEW AccuSet HCV (0810-0204) is a 25-member validation panel of undiluted, naturally occurring plasma samples (1 vial per member, 1.0 ml per vial). Panel members
Mumps IgG Capture EIA
 Mumps IgG Capture EIA An Enzyme Immunoassay for the detection of human IgG antibodies to mumps virus in oral fluid, serum and plasma samples Cat. No: MuVG012 For in-vitro diagnostic use C V Designed &
Mumps IgG Capture EIA An Enzyme Immunoassay for the detection of human IgG antibodies to mumps virus in oral fluid, serum and plasma samples Cat. No: MuVG012 For in-vitro diagnostic use C V Designed &
Department of Defense Armed Forces Health Surveillance Branch Global MERS-CoV Surveillance Summary (16 NOV 2016)
 Department of Defense Armed Forces Health Surveillance Branch Global MERS-CoV Surveillance Summary (16 NOV 2016) For questions or comments, please contact: dha.ncr.health-surv.list.afhs-ib-alert-response@mail.mil
Department of Defense Armed Forces Health Surveillance Branch Global MERS-CoV Surveillance Summary (16 NOV 2016) For questions or comments, please contact: dha.ncr.health-surv.list.afhs-ib-alert-response@mail.mil
Downloaded from:
 Saba, HF; Kouyoumjian, SP; Mumtaz, GR; Abu-Raddad, LJ (2013) Characterising the progress in HIV/AIDS research in the Middle East and North Africa. Sexually transmitted infections, 89 Sup. iii5-9. ISSN
Saba, HF; Kouyoumjian, SP; Mumtaz, GR; Abu-Raddad, LJ (2013) Characterising the progress in HIV/AIDS research in the Middle East and North Africa. Sexually transmitted infections, 89 Sup. iii5-9. ISSN
