Molecular dipstick for sleeping sickness. A molecular dipstick test for the diagnosis of sleeping sickness
|
|
|
- Lesley McGee
- 5 years ago
- Views:
Transcription
1 1 Running title: Molecular dipstick for sleeping sickness A molecular dipstick test for the diagnosis of sleeping sickness S. Deborggraeve, 1,3 * F. Claes, 1 T. Laurent, 1 P. Mertens, 2 T. Leclipteux, 2 J. C. Dujardin, 1 P. Herdewijn, 3 and P. Büscher Department of Parasitology, Institute of Tropical Medicine, Antwerp, Belgium, 1 Coris BioConcept, Gembloux, Belgium, 2 and Laboratory of Medicinal Chemistry, Rega Institute for Medicinal Research, Leuven, Belgium * Corresponding author. Mailing address: Deborggraeve S, Department of Parasitology, Institute of Tropical Medicine, Nationalestraat 155, B-2000 Antwerpen, Belgium, Tel: , Fax: , sdeborggraeve@itg.be Page 1 of 23
2 Summary Human African trypanosomiasis (HAT) or sleeping sickness, is a neglected disease that affects poor rural populations across sub-saharan Africa. Confirmation of diagnosis is based on detection of parasites in either blood or lymph by microscopy. Here we present the development and the first phase evaluation of a simple and rapid test (HAT-PCR-OC) for detection of amplified Trypanosoma brucei DNA. PCR products are visualised on a dipstick through hybridisation with a gold conjugated probe (oligochromatography). Visualisation is straightforward and takes only 5 minutes. Controls for both the PCR reaction and for DNA migration are incorporated in the assay. The lower detection limit of the test is 5 fg of pure T. brucei DNA. One parasite in 180 µl of blood is still detectable. Sensitivity and specificity for T. brucei were calculated at 100% when tested on blood samples from 26 confirmed sleeping sickness patients, 18 negative controls (non-endemic region) and 50 negative control blood samples from endemic region. HAT-PCR-OC is a promising new tool for diagnosis of sleeping sickness in laboratory settings and the diagnostic format described here may have wider application for other infectious diseases. Page 2 of 23
3 Introduction Human African trypanosomiasis (HAT) is a complex of protozoan infections, fatal if untreated, which can be caused by infection with either Trypanosoma brucei (T. b.) gambiense (chronic HAT) or T. b. rhodesiense (acute HAT). Both human infective subspecies are cyclically transmitted by tsetse flies (genus Glossina). HAT is a severely neglected disease, typically affecting rural populations of poor people across sub-saharan Africa (23). Over 60 million people are at risk of contracting the disease with an estimated 50-70,000 persons newly infected annually (2). In the absence of treatment, HAT patients inevitably die after a more or less prolonged period of grave illness. Disease control for T. b. gambiense relies heavily on accurate diagnosis and effective treatment of patients and for T. b. rhodesiense on treatment of patients and of the animal reservoir (24). Classical diagnosis requires demonstration of parasites in blood or lymph which for T. b. gambiense infection is problematic due to extremely low parasitaemias in infected people. It is estimated that 20 to 30% of the patients remain undiagnosed by standard parasitological techniques (20). New molecular techniques based on the polymerase chain reaction (PCR) have been developed as surrogate for parasite detection. Applications of PCR for detecting T. brucei and its subspecies have been reported, which can be highly effective for detection of T. b. rhodesiense and for distinguishing T. b. rhodesiense from T. b. gambiense (7, 16, 17, 18, 22, 25). The use of these highly specific and sensitive PCR methods is however only suitable for screening for disease prevalence and for research purposes and are not commonly used to inform diagnosis of HAT (5). This is partly due to the cumbersome methods of PCR product detection. Amplicons are normally identified using UV trans-illumination after being electrophoresed in the presence of ethidium bromide (a carcinogen). Alternative methods for Page 3 of 23
4 PCR product detection such as real-time PCR, PCR-ELISA or mass spectrometry have been developed (3, 10, 21) but are complex, expensive, equipment, recourse and personnel hungry. There is a demand for a simplified method of amplification and product detection which would make these tools useable in the facilities available in regional sleeping sickness diagnostic laboratories. Oligochromatography (OC) provides a simple and rapid dipstick format for detection of amplified PCR products (Coris BioConcept, Gembloux, Belgium; Patent n WO 2004/099438A1) (12, 13, 15, 19). PCR products are visualised by hybridisation with a gold conjugated probe. This PCR product detection format takes only 5 minutes and no other equipment than a dry heating block and a pipette are needed. An internal control (IC) for the PCR reaction and a control for the chromatographic migration are incorporated in the assay. We present here the development and phase I evaluation of a T. brucei specific PCR- Oligochromatography test, called HAT-PCR-OC (Human African Trypanosomiasis- Polymerase Chain Reaction-Oligochromatography). We have chosen the 18S ribosomal RNA gene (rdna) as the target for the HAT-PCR-OC since it is a multi copy gene that contains sequences conserved within Trypanosomatids and species specific sequences (11) Methods Parasite DNA DNA was obtained from T. b. gambiense (LiTat 1.3) and T. b. rhodesiense (STIB 382) parasites amplified in HsdCpb:WU rats (Harlan, the Netherlands). Trypanosomes were separated from the blood using di-ethyl-amino-ethyl (DEAE) chromatography (9), followed by repeated centrifugation (20 minutes, 2000 g) and sediment washes with Phosphate Page 4 of 23
5 Buffered Saline Glucose (PSG) (38 mm Na 2 HPO 4.2H 2 0, 2 mm NaHPO 4, 80 mm glucose, 29 mm NaCl, ph 8.0). Trypanosome pellets were stored at 80 C. Twenty µl of trypanosome pellet (approximately cells) were resuspended in 200 µl of Phosphate Buffered Saline (PBS) (8.1 mm Na 2 HPO 4.2H 2 O, 1.4 mm NaHPO 4, 140 mm NaCl, ph 7.4) and the DNA was extracted with the QIAamp DNA mini kit (Qiagen, Hilden, Germany), resulting in pure DNA in 200 µl TE elution buffer. The typical yield of DNA extracted from a 20 µl pellet was 150 ng/µl or 30 µg of total DNA. The extracts obtained were diluted 200 times in water and divided into aliquots of 2 ml in microcentrifuge tubes for storage at 20 C. Purified DNA from other pathogens, i.e. Leishmania donovani, Trypanosoma cruzi, Mycobacterium tuberculosis, Plasmodium falciparum and Schistosoma mansoni was obtained from other research groups Blood samples Informed consent was obtained from patients, or their parents or guardians and from nondiseased persons. The human and animal experimentation guidelines of the Institute of Tropical Medicine (Antwerp, Belgium) were followed. Non endemic controls Venous blood on EDTA from 28 healthy human Belgian blood donors who had never visited a HAT endemic country (6). Endemic controls Venous blood on EDTA from 50 healthy human volunteers in Kinshasa, D.R. Congo. Sleeping sickness patients Page 5 of 23
6 Venous blood on heparin from 26 T. b. gambiense patients enrolled in a clinical study performed in D. R. Congo and with confirmed presence of parasites in the blood. Spiked blood Blood on EDTA spiked with T. b. gambiense (LiTat 1.3) parasites was used throughout the development of the assay and for the estimation of its lower detection limit. Bloodstream form trypanosomes were grown in rats. At day 3 post infection, tail blood was taken and diluted in PSG and the number of parasites per ml was counted in a KOVA cell counting chamber (Hycor Biomedical Inc., California, USA). A 10-fold dilution series of parasites was made in freshly taken naive human blood, ranging from 10,000 parasites/180 µl to 1 parasite/180 µl blood. Non-spiked blood was used as negative control DNA extraction from blood samples As recommended by Qiagen, 180 µl of blood were mixed with an equal volume of AS1 buffer (Qiagen, Hilden, Germany) and stored at ambient temperature until DNA extraction. Blood can be stored in this stabilising buffer for up to three months in the dark without loss of DNA quality. DNA was extracted with the QIAamp DNA mini kit (Qiagen, Hilden, Germany) according to the manufacturer s instructions and stored at 20 C. Briefly, DNA is adsorbed onto the silica-gel membrane in the QIAamp Spin Column, washed by two centrifugation steps and eluted in 50 µl elution buffer. 180 µl sample of whole blood will yield an estimated 3-12 µg of DNA. The internal control line in the oligochromatography (see below) showed the presence of inhibitory factors in the extracted DNA in some samples (1). In such cases, DNA was further purified as follows: 1/10 volume of 3 M sodium acetate ph 5.2 and 2 volumes of ice-cold 100% ethanol were added, followed by overnight DNA precipitation at -20 C. The suspension was centrifuged at 16,100 g for 15 minutes and the Page 6 of 23
7 DNA resuspended in 500 µl of 70% ethanol. The suspension was again centrifuged for 10 minutes at 16,100 g and the ethanol was removed. DNA was dried in a vacuum chamber for at least 30 minutes and finally resuspended in 50 µl of water and stored at -20 C Primers and probes (Fig. 1) Primers, biotinylated primers, internal control DNA and probes were synthesised by Sigma (Bornem, Belgium). Primers Sequences of the 18S ribosomal RNA gene (rdna) of the Trypanosomatidae parasites T. brucei gambiense (GenBank accession number AJ009141), T. brucei rhodesiense (GenBank accession number AJ009142), T. cruzi (GenBank accession number AF303660) and L. donovani (GenBank accession number X07773) were aligned and primers (18S-F and 18S- R) were designed to amplify a sequence of the Trypanosomatidae 18S rdna using the DNAman software (Lynnon Corporation, Quebec, Canada). Primers are situated in Trypanosomatidae conserved sequences that surround a T. brucei conserved sequence. The reverse primer 18S-R was biotinylated at the 5 end. T. brucei gold probe The alignments described above were used to design a 17 bp DNA probe that can hybridise with a T. brucei specific sequence situated between the Trypanosomatidae conserved primer sequences. Absence of putative secondary structures within the expected PCR amplicon was checked via mfold version 3.1. (26). The probe was conjugated with gold particles using the procedure described in Patent WO 2004/099438A1 (19). Internal control DNA Page 7 of 23
8 The internal control DNA (IC DNA) was constructed to be of the same length as the T. brucei sequence (106 bp). Its sequence is identical to the T. brucei sequence except for the 17 bp central part. This central part was designed to have the same GC content as the T. brucei 17 bp sequence (Patent WO 2004/099438A1) (19). IC gold probe A DNA probe was designed that can hybridise with the 17 bp IC specific sequence within the IC DNA. This IC probe was conjugated with gold particles as described above. Migration control probe The migration control probe is identical to the 17 bp central sequence of the IC DNA PCR amplification An asymmetric PCR was designed using 4 fold more biotinylated reverse primer than forward primer. The 50 µl reaction mixture contained 10 mm Tris-HCl (ph 8.3 at 25 C), 50 mm KCl (PCR buffer, Sigma, Bornem, Belgium), 200 µm of each deoxynucleotide triphosphate (Roche, Mannheim, Germany), 2.5 mm MgCl 2 (Sigma, Bornem, Belgium), 0.2 µm forward primer 18S-F, 0.8 µm biotinylated reverse primer 18S-R, 0.1 mg/ml acetylated bovine serum albumine (Promega, Madison, USA), 3.2 am IC DNA, 1 U of HotStar Taq polymerase (Qiagen, Hilden, Germany) and 5 µl of sample DNA. An initial denaturation step of 94 C for 15 min to activate the HotStar Taq polymerase was followed by 40 cycles of 94 C for 30 s, 60 C for 30 s and 72 C for 30 s, with a final extension at 72 C for 5 minutes. Amplification was conducted in 200 µl thin-wall PCR tubes (Abgene, Epsom, UK) in a T3 thermocycler 48 (Biometra, Göttingen, Germany). 168 Page 8 of 23
9 Oligochromatography Preparation The Oligochromatography (OC) dipstick is constructed with polymer backing faced on each side with a lower absorbent pad which overlaps a nitrocellulose membrane (in the middle) and an upper absorbent pad (Fig. 2). The facing side represents the test for T. brucei and the back is the control. The lower absorbent pad at the test side is impregnated with the T. brucei gold probe and the lower absorbent pad at the control side is impregnated with the IC gold probe. On the membrane at the test side, a line of Neutralite avidin (Belovo SA, Bastogne, Belgium) is coated. On the membrane at the control side, two control lines are coated. A first line of Neutralite avidin (control for PCR) and a second line of a probe that is complementary to the IC gold probe (migration control). The OC conditions for the phase I evaluation were as follows. The PCR product was denaturated at 94 C for 30 seconds and kept on ice. Forty µl were mixed with an equal volume of migration buffer preheated at 55 C followed by dipping the OC dipstick into the solution. Principle After amplification of a sample containing T. brucei DNA, the PCR product solution contains both IC amplicons and T. brucei amplicons. During migration at 55 C through the lower absorbent pad, the solution takes up the impregnated gold probes. The T. brucei gold probe hybridises with the biotinylated strands of the T. brucei amplicons during migration. These complexes accumulate on the Neutralite avidin line at the test side resulting in a visible red line. On the back side the IC gold probe hybridises with the biotinylated strands of the IC amplicons and these complexes accumulate on the Neutralite avidin line. Unbound IC gold probe migrates further and is trapped by the complementary migration control probe Page 9 of 23
10 line. Both control lines determine whether the HAT-PCR-OC is valid or invalid (Fig. 3). The migration control line should always be visible to validate the test. A test is invalid when both the IC amplicon control line and the T. brucei test line are invisible (Fig. 3d) indicating that the PCR reaction failed possibly due to inhibitory factors in the extracted DNA. When a sample contains very high concentrations of Trypanosomatidae DNA, competition between this target DNA and the IC template DNA for the primers can result in an invisible IC amplicon control line combined with a strongly visible T. brucei test line (Fig. 3c). In the latter case, the test is considered valid Results Analytical sensitivity The detection limit of the HAT-PCR-OC was evaluated on a serial dilution of T. b. gambiense DNA in water. The lower detection limit was 5 fg of parasite DNA which is about 1/40 of the DNA content of one parasite (Fig. 4a). Moreover, the test was able to detect ten times less DNA compared to agarose gel electrophoresis (data not shown). The lower detection limit was also evaluated on DNA extracted from a blood sample series spiked with decreasing numbers of living T. b. gambiense parasites. HAT-PCR-OC was consistently able to detect one single parasite in 180 µl of blood (Fig. 4b). The assay remained negative when non-spiked control blood samples were tested Analytical specificity The species specificity of the HAT-PCR-OC was assessed on purified DNA from T. b. gambiense, T. b. rhodesiense, Leishmania donovani, Trypanosoma cruzi, Mycobacterium Page 10 of 23
11 tuberculosis, Plasmodium falciparum and Schistosoma mansoni. Positive results were only obtained with the two T. brucei subspecies (Fig. 4c) Diagnostic sensitivity and specificity (Fig. 5) HAT-PCR-OC was positive with the blood samples from all 26 T. b. gambiense sleeping sickness patients and negative with blood samples from all 28 healthy Belgian blood donors. With the 50 endemic negative control blood samples, 47 tests were valid and negative in HAT-PCR-OC. Three test results were invalid since the IC for PCR was negative, indicating the presence of PCR inhibitory factors in the extracted DNA. Therefore, DNA of these samples was ethanol precipitated and resuspended in water. When re-tested, valid negative test results were obtained. Thus, HAT-PCR-OC results corresponded fully with the infection status of the sampled persons indicating 100% sensitivity and specificity. Page 11 of 23
12 Discussion A major constraint for effective sleeping sickness control is imperfect diagnosis. Reliable and fast diagnostic tools are desperately needed. The HAT-PCR-OC test described here has been developed as a simplified molecular test for rapid and sensitive detection of T. brucei parasites in blood samples. HAT-PCR-OC combines both the sensitivity and specificity of PCR with the simplicity and speed of membrane chromatography (visible after 5 minutes by the naked eye). OC, in contrast to conventional amplicon detection techniques, doesn t require post-amplification preparation, neither sophisticated equipment. The data presented here show that the assay is able to detect one single parasite in a 180 µl volume of blood. When evaluated using both trypanosome positive patient and control trypanosome negative samples, HAT-PCR-OC results corresponded 100% with the infection status of the tested persons. HAT-PCR-OC shows clear potential for implementation as a reference diagnostic test in mid level equipped laboratory facilities (clean laboratories with power and cold storage) and will be of particular value for evaluation of cure assessment during clinical trials of new drugs or drug combinations. The simple detection methodology described here may facilitate PCR application in HAT endemic countries. Further refinement of this assay will widen its application. New simple nucleic acid extraction techniques together with novel isothermal nucleic acid amplification methods such as loop-mediated isothermal amplification (LAMP) (8, 14) and nucleic acid based amplification (NASBA) (4) will mean that the constraint to field PCR (namely the need for thermocycling reactions) could be eliminated. A further application of the HAT-PCR-OC test format would be for diagnosis of all Trypanozoon infections since the detection probe is specific for all Trypanozoon taxa Page 12 of 23
13 including T. b. brucei, T. evansi and T. equiperdum that affect cattle, buffaloes, small ruminants, camel and horses and that are responsible for severe losses in the agricultural sector. This is of particular value for T. b. rhodesiense HAT in which case the disease is maintained in the domestic animal reservoir (24). A common drawback of PCR is the risk of sample contamination with PCR products leading to false positive results. When implementing HAT-PCR-OC, one should take measures to avoid this risk. The oligochromatography procedure should be performed in closed test tubes and pre- and post-pcr manipulations should be physically separated, preferably in separate lab spaces. Finally, used dipsticks should be thrown away after reading the result or should be archived in a post amplification room. To our knowledge this is the first nucleic acid based diagnostic test in dipstick format for vector-borne diseases. The proof of principal for HAT-PCR-OC presented in this study opens perspectives for its use in laboratories within countries where sleeping sickness is endemic. The same format can be applied for other infectious diseases such as malaria, tuberculosis, leishmaniasis, Chagas disease, schistosomiasis, etc. for which more simple and applicable molecular diagnostic tools are needed. Page 13 of 23
14 Acknowledgements This study received financial support from the International Atomic Energy Agency (IAEA, Vienna, Austria, contract number 12851/RBF) and from the International Livestock Research Institute (ILRI, Nairobi, Kenya). We kindly acknowledge Dr. C. Grevelding (Institüt für Genetik, Heinrich-Heine-Universität, Düsseldorf, Germany) for providing us with DNA from Schistosoma mansoni. We are grateful to Mrs. S. De Doncker, Dr. F. Portaels and Dr. U. D Alessandro (Institute of Tropical Medicine, Antwerp) for providing DNA from Leishmania donovani, Trypanosoma cruzi, Mycobacterium tuberculosis and Plasmodium falciparum. Special thanks to S.C. Welburn for critical reading of the manuscript. Page 14 of 23
15 References 1. Al-Soud, W. A., L. J. Jönsson, and P. Radström Identification and characterization of immunoglobulin G in blood as a major inhibitor of diagnostic PCR. J Clin Microbiol 38: Anonymus Human African trypanosomiasis (sleeping sickness): epidemiological update. Weekly Epidemiological Record 81: Becker, S., J. R. Franco, P. P. Simarro, A. Stich, P.M. Abel, and D. Steverding Real-time PCR for detection of Trypanosoma brucei in human blood samples. Diagn Microbiol Infect Dis 50: Chang, A. B. and J. D. Fox NASBA and other transcription based amplification methods for research and diagnostic microbiology. Rev Med Microbiol 10: Chappuis, F., L. Loutan, P. Simarro, V. Lejon, and P. Büscher Options for the field diagnosis of human African trypanosomiasis. Clin Microbiol Rev 18: De Doncker, S., V. Hutse, S. Abdellati, S. Rijal, B.M. Singh Karki, S. Decuypere, D. Jacquet, D. Le Ray, M. Boelaert, S. Koirala, and J. C. Dujardin A new PCR-ELISA for diagnosis of visceral leishmaniasis in blood of HIV-negative subjects. Trans R Soc Trop Med Hyg 99: Kabiri, M., J. R. Franco, P. P. Simarro, J. A. Ruiz, M. Sarsa, and D. Steverding Detection of Trypanosoma brucei gambiense in sleeping sickness suspects by Page 15 of 23
16 PCR amplification of expression-site-associated genes 6 and 7. Trop Med Int Health 4: Kuboki, N., N. Inoue, T. Sakurai, F. Di Cello, D. J. Grab, H. Suzuki, C. Sugimoto, and T. Igarashi Loop-mediated isothermal amplification for detection of African trypanosomes. J Clin Microbiol 41: Lanham, S. M. and D. G. Godfrey Isolation of salivarian trypanosomes from man and other mammals using DEAE-cellulose. Exp Parasitol 28: Martin-Sanchez, J., J. A. Pineda, M. Andreu-Lopez, J. Delgado, J. Macias, R. De La Rosa, and F. Morillas-Marquez The high sensitivity of a PCR-ELISA in the diagnosis of cutaneous and visceral leishmaniasis caused by Leishmania infantum. Ann Trop Med Parasitol 96: Maslov, D. A., J. Lukes, M. Jirku, and L. Simpson Phylogeny of trypanosomes as inferred from the small and large subunit rrnas: implications for the evolution of parasitism in the trypanosomatid protozoa. Mol Biochem Parasitol 75: Mertens, P., I. Renuart, R. Boreux, M.-P. Hayette, and T. Leclipteux. Detection of Toxoplasma gondii by oligochromatography of nested-pcr amplicons rd General Meeting of the American Society of Medicine, Washington Mertens, P., L. Vijgen, C. Olungu, S. Degallaix, I. Renuart, M. Van Ranst, and T. Leclipteux Detection of SARS-CoV by oligochromatography of RT-PCR amplicons. Clin Microbiol Inf 10 (supp. 3): Page 16 of 23
17 Notomi, T., H. Okayama, H. Masubuchi, T. Yonekawa, K. Watanabe, N. Amino, and T. Hase Loop-mediated isothermal amplification of DNA. Nucleic Acids Res 28: Olungu, C., P. Mertens, S. Degallaix, I. Renuart, O. Legeay, R. Laube, and T. Leclipteux. Detection of HAV by oligochromatography of RT-PCR amplicons th General Meeting of the American Society of Medicine, New Orleans, May Picozzi, K., E. M. Fèvre, M. Odiit, M. C. Eisler, I. Maudlin, and S. C. Welburn Sleeping sickness in Uganda: a thin line between two fatal diseases. BMJ 331: Radwanska, M., M. Chamekh, L. Vanhamme, F. Claes, S. Magez, E. Magnus, P. De Baetselier, P. Büscher, and E. Pays The serum resistance-associated gene as a diagnostic tool for the detection of Trypanosoma brucei rhodesiense. Am J Trop Med Hyg 67: Radwanska, M., F. Claes, S. Magez, E. Magnus, D. Perez-Morga, E. Pays, and P. Büscher Novel primer sequences for a polymerase chain reaction-based detection of Trypanosoma brucei gambiense. Am J Trop Med Hyg 67: Renuart, I., P. Mertens, and T. Leclipteux One step oligochromatographic device and method of use. EU Patent WO2004/099438:A Robays, J., M. M. C. Bilengue, P. Van der Stuyft, and M. Boelaert The effectiveness of active population screening and treatment from sleeping sickness control in the Democratic republic of Congo. Trop Med Int Health 9: Page 17 of 23
18 Sampath, R., S. A. Hofstadler, L. B. Blyn, M.W. Eshoo, T. A. Hall, C. Massire, H. M. Levene, J. C. Hannis, P. M. Harrell, B. Neuman, M. J. Buchmeier, Y. Jiang, R. Ranken, J. J. Drader, V. Samant, R. H. Griffey, J. M. McNeil, S. T. Crooke, and D. J. Ecker Rapid identification of emerging pathogens: coronavirus. Em Inf Dis 11: Schares, G. and D. Mehlitz Sleeping sickness in Zaire: a nested polymerase chain reaction improves the identification of Trypanosoma (Trypanozoon) brucei gambiense by specific kinetoplast DNA probes. Trop Med Int Health 1: Trouiller, P., P. Olliaro, E. Torreele, J. Orbinski, R. Laing, and N. Ford Drug development for neglected diseases: a different market and a public-health policy failure. Lancet 359: Welburn, S. C., P. G. Coleman, I. Maudlin, E. M. Fèvre, M. Odiit, and M. C. Eisler Crisis, what crisis? Control of Rhodesian sleeping sickness. Trends Parasitol 22: Welburn, S. C., K. Picozzi, E. M. Fèvre, P. G. Coleman, M. Odiit, M. Carrington, and I. Maudlin Identification of human-infective trypanosomes in animal reservoir of sleeping sickness in Uganda by means of serum-resistance-associated (SRA) gene. Lancet 358: Zuker, M Mfold web server for nucleic acid folding and hybridazation prediction. Nucleic Acids Res 31: Page 18 of 23
19 Figure 1. Alignment of the HAT-PCR-OC target sequence within the 18S rrna gene of the Trypanosomatidae parasites Trypanosoma brucei gambiense (T.b.g.) (GenBank accession number AJ009141), Trypanosoma brucei rhodesiense (T.b.r.) (GenBank accession number AJ009142), Trypanosoma cruzi (T.c.) (GenBank accession number AF303660) and Leishmania donovani (L.d.) (GenBank accession number X07773). Situation of the forward primer 18S-F, the biotin labelled reverse primer 18S-R, and the T. brucei (T.b.) detection probe. Gaps in the sequence are presented by dots T.b.g. T.b.r. T.c. L.d. CGCCAAGCTAATACATGAACCAA 18S-F GCAGGTCGCTTACTTACTTTAAT 18S-R TTGTGTTTACGCACTTG T.b. detection probe Page 19 of 23
20 Figure 2. HAT-PCR-OC test principle Test side. In case of a positive sample, during migration, the T. brucei gold probes hybridise with the T. brucei amplicons that will accumulate on the Neutralite avidin line on the test side of the stick. Control side. During migration the internal control (IC) gold probes hybridise with IC amplicons that will accumulate on the Neutralite avidin line on the control side of the stick (control for PCR). The unbound IC gold probes hybrise with the complementary migration control probes (migration control) Control side (back) Test side (front) upper absorbent pad supporting polymer backing nitrocellulose membrane Trypanosomatidae specific primers T. brucei specific sequence T. brucei gold probe Target sequence for IC gold probe IC gold probe Neutralite avidin Biotin lower absorbent pad impregnated with IC gold probes lower absorbent pad impregnated with T. brucei gold probes Page 20 of 23
21 Figure 3. Possible HAT-PCR-OC test result. a. valid test, positive result, b. valid test, negative result, c. valid test, positive result of a sample with high T. brucei DNA content, d. invalid test 406 Legend of test sides a. front a. back b. front b. back c. front c. back d. front d. back Legend of control sides Upper absorbent pad T. brucei test line Lower absorbent pad Upper absorbent pad migration control line PCR control line Lower absorbent pad Page 21 of 23
22 Figure 4. Analytical sensitivity and species specificity of the HAT-PCR-OC test a. Serial dilution of T. b. gambiense DNA in water. Dipstick 1-7: 500 pg, 50 pg, 5 pg, 500 fg, 50 fg, 5 fg, 0.5 fg per PCR reaction b. Serial dilution of living T. b. gambiense bloodstream form parasites in naïve human blood. Dipstick 1-6: 10,000, 1,000, 100, 10, 1, 0 parasites in 180 µl of blood. c. HAT-PCR-OC results obtained with DNA form T. b. gambiense (dipstick 1), T. b. rhodesiense (dipstick 2), Leishmania donovani (dipstick 3), Trypanosoma cruzi (dipstick 4), Mycobacterium tuberculosis (dipstick 5), Plasmodium falciparum (dipstick 6) and Schistosoma mansoni (dipstick 7) a. b c Page 22 of 23
23 Figure 5. Diagnostic sensitivity and specificity of the HAT-PCR-OC. a. HAT-PCR-OC results for 26 blood samples from confirmed T. b. gambiense sleeping sickness patients (dipstick 1 26) and one negative control (-). b. HAT-PCR-OC results for one positive control (+) and for 5 blood samples from endemic negative controls (dipstick 1 5) a. b Page 23 of 23
Molecular Dipstick Test for Diagnosis of Sleeping Sickness
 JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 2006, p. 2884 2889 Vol. 44, No. 8 0095-1137/06/$08.00 0 doi:10.1128/jcm.02594-05 Copyright 2006, American Society for Microbiology. All Rights Reserved. Molecular
JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 2006, p. 2884 2889 Vol. 44, No. 8 0095-1137/06/$08.00 0 doi:10.1128/jcm.02594-05 Copyright 2006, American Society for Microbiology. All Rights Reserved. Molecular
ISOTHERMAL AMPLIFICATION IN MOLECULAR DIAGNOSIS OF SLEEPING SICKNESS
 Molecular Diagnostics Symposium 2014 Zurich, 27 February 2014 ISOTHERMAL AMPLIFICATION IN MOLECULAR DIAGNOSIS OF SLEEPING SICKNESS Stijn Deborggraeve Parasite Diagnostics Unit, Institute of Tropical Medicine
Molecular Diagnostics Symposium 2014 Zurich, 27 February 2014 ISOTHERMAL AMPLIFICATION IN MOLECULAR DIAGNOSIS OF SLEEPING SICKNESS Stijn Deborggraeve Parasite Diagnostics Unit, Institute of Tropical Medicine
Nucleic Acid Sequence-Based Amplification with Oligochromatography for Detection of Trypanosoma brucei in Clinical Samples
 JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 2009, p. 630 635 Vol. 47, No. 3 0095-1137/09/$08.00 0 doi:10.1128/jcm.01430-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Nucleic Acid
JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 2009, p. 630 635 Vol. 47, No. 3 0095-1137/09/$08.00 0 doi:10.1128/jcm.01430-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Nucleic Acid
Molecular analysis of archived blood slides reveals an atypical human Trypanosoma. infection
 Molecular analysis of archived blood slides reveals an atypical human Trypanosoma infection Running head: Molecular analysis of Trypanosoma infection Stijn Deborggraeve a,f,*, Mathurin Koffi b, Vincent
Molecular analysis of archived blood slides reveals an atypical human Trypanosoma infection Running head: Molecular analysis of Trypanosoma infection Stijn Deborggraeve a,f,*, Mathurin Koffi b, Vincent
The development of new diagnostic tools for sleeping sickness
 The development of new diagnostic tools for sleeping sickness P. Büscher Institute of Tropical Medicine, Antwerp WHO CC for Research and Training on Human African Trypanosomiasis Diagnosis 1 1 Distribution
The development of new diagnostic tools for sleeping sickness P. Büscher Institute of Tropical Medicine, Antwerp WHO CC for Research and Training on Human African Trypanosomiasis Diagnosis 1 1 Distribution
Diagnostic Accuracy of Loopamp Trypanosoma brucei Detection Kit for Diagnosis of Human African Trypanosomiasis in Clinical Samples
 Diagnostic Accuracy of Loopamp Trypanosoma brucei Detection Kit for Diagnosis of Human African Trypanosomiasis in Clinical Samples Patrick Mitashi 1,2,3, Epco Hasker 1, Dieudonné Mumba Ngoyi 2,3, Pati
Diagnostic Accuracy of Loopamp Trypanosoma brucei Detection Kit for Diagnosis of Human African Trypanosomiasis in Clinical Samples Patrick Mitashi 1,2,3, Epco Hasker 1, Dieudonné Mumba Ngoyi 2,3, Pati
Trypanosomiasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course
 Trypanosomiasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course UNCLASSIFIED Disclaimer The views expressed in this presentation are those of the speaker and do not reflect the official policy
Trypanosomiasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course UNCLASSIFIED Disclaimer The views expressed in this presentation are those of the speaker and do not reflect the official policy
Blood Smears Only 6 October Sample Preparation and Quality Control 15B-K
 NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 6 October 5 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only is
NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 6 October 5 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only is
Norgen s HIV proviral DNA PCR Kit was developed and validated to be used with the following PCR instruments: Qiagen Rotor-Gene Q BioRad icycler
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com HIV Proviral DNA PCR Kit Product # 33840 Product Insert Background Information
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com HIV Proviral DNA PCR Kit Product # 33840 Product Insert Background Information
Hepatitis B Virus Genemer
 Product Manual Hepatitis B Virus Genemer Primer Pair for amplification of HBV Viral Specific Fragment Catalog No.: 60-2007-10 Store at 20 o C For research use only. Not for use in diagnostic procedures
Product Manual Hepatitis B Virus Genemer Primer Pair for amplification of HBV Viral Specific Fragment Catalog No.: 60-2007-10 Store at 20 o C For research use only. Not for use in diagnostic procedures
Chagas disease: Challenges in Diagnostics
 Chagas disease: Challenges in Diagnostics Emily Adams Royal Tropical Institute (KIT), KIT Biomedical Research The Netherlands e-mail: e.adams@kit.nl Aims of KIT Biomedical Research To contribute to the
Chagas disease: Challenges in Diagnostics Emily Adams Royal Tropical Institute (KIT), KIT Biomedical Research The Netherlands e-mail: e.adams@kit.nl Aims of KIT Biomedical Research To contribute to the
Product # Kit Components
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Pneumocystis jirovecii PCR Kit Product # 42820 Product Insert Background Information
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Pneumocystis jirovecii PCR Kit Product # 42820 Product Insert Background Information
Norgen s HIV Proviral DNA PCR Kit was developed and validated to be used with the following PCR instruments: Qiagen Rotor-Gene Q BioRad T1000 Cycler
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com HIV Proviral DNA PCR Kit Product# 33840 Product Insert Intended
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com HIV Proviral DNA PCR Kit Product# 33840 Product Insert Intended
Articles. Copyright Büscher et al. Open Access article distributed under the terms of CC BY-NC-ND.
 Sensitivity and specificity of HAT Sero-K-SeT, a rapid diagnostic test for serodiagnosis of sleeping sickness caused by Trypanosoma brucei gambiense: a case-control study Philippe Büscher, Pascal Mertens,
Sensitivity and specificity of HAT Sero-K-SeT, a rapid diagnostic test for serodiagnosis of sleeping sickness caused by Trypanosoma brucei gambiense: a case-control study Philippe Büscher, Pascal Mertens,
WRAIR- GEIS 'Operational Clinical Infectious Disease' Course
 Trypanosomiasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course The opinions or assertions contained herein are the private views of the author, and are not to be construed as official, or
Trypanosomiasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course The opinions or assertions contained herein are the private views of the author, and are not to be construed as official, or
A Novel Noninvasive Method for Diagnosis of Visceral Leishmaniasis by. rk39 Test in Sputum Samples
 JCM Accepts, published online ahead of print on 0 June 00 J. Clin. Microbiol. doi:0./jcm.00-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
JCM Accepts, published online ahead of print on 0 June 00 J. Clin. Microbiol. doi:0./jcm.00-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
Kit Components Product # EP42720 (24 preps) MDx 2X PCR Master Mix 350 µl Cryptococcus neoformans Primer Mix 70 µl Cryptococcus neoformans Positive
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Cryptococcus neoformans End-Point PCR Kit Product# EP42720 Product
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Cryptococcus neoformans End-Point PCR Kit Product# EP42720 Product
Protozoa from tissues. Leishmania spp. Naegleria fowleri Toxoplasma gondii Trichomonas vaginalis Trypanosoma spp.
 Protozoa from tissues Leishmania spp. Naegleria fowleri Toxoplasma gondii Trichomonas vaginalis Trypanosoma spp. Leishmaniasis Leishmania infantum, Leishmania donovani, in macrophages of man. Female sandflies:
Protozoa from tissues Leishmania spp. Naegleria fowleri Toxoplasma gondii Trichomonas vaginalis Trypanosoma spp. Leishmaniasis Leishmania infantum, Leishmania donovani, in macrophages of man. Female sandflies:
HAT. Platform EDITORIAL CONTENT P. 2 P. 2 P. 3 P. 3 P. 4 P. 4 P. 5 P. 5 P. 6 P. 6 TRAINING WORKSHOP FOR MEMBERS OF THE STEERING COMMITTEE MEETING
 HAT Platform EDITORIAL Dr Augustin Kadima Ebeja HAT Platform Coordinator We are proud to present our design for a bilingual logo that will help underscore our efforts to conduct clinical trials against
HAT Platform EDITORIAL Dr Augustin Kadima Ebeja HAT Platform Coordinator We are proud to present our design for a bilingual logo that will help underscore our efforts to conduct clinical trials against
Human Immunodeficiency Virus-1 (HIV-1) Genemer. Primer Pair for amplification of HIV-1 Specific DNA Fragment
 Product Manual Human Immunodeficiency Virus-1 (HIV-1) Genemer Primer Pair for amplification of HIV-1 Specific DNA Fragment Catalog No.: 60-2002-10 Store at 20 o C For research use only. Not for use in
Product Manual Human Immunodeficiency Virus-1 (HIV-1) Genemer Primer Pair for amplification of HIV-1 Specific DNA Fragment Catalog No.: 60-2002-10 Store at 20 o C For research use only. Not for use in
Confirmation of Trypanosome Parasitaemia in Previously Serologically Positive Individuals in the Abraka Area of Delta State, Nigeria
 JMBR: A Peer-review Journal of Biomedical Sciences December 2006 Vol. 5 No.2 pp-28-32 Confirmation of Trypanosome Parasitaemia in Previously Serologically Positive Individuals in the Abraka Area of Delta
JMBR: A Peer-review Journal of Biomedical Sciences December 2006 Vol. 5 No.2 pp-28-32 Confirmation of Trypanosome Parasitaemia in Previously Serologically Positive Individuals in the Abraka Area of Delta
Mitochondrial DNA Isolation Kit
 Mitochondrial DNA Isolation Kit Catalog Number KA0895 50 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4 Materials
Mitochondrial DNA Isolation Kit Catalog Number KA0895 50 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4 Materials
Evaluation of an EDTA version of CATT/Trypanosoma brucei gambiense. Prince Leopold Institute of Tropical Medicine, Department of Parasitology,
 Evaluation of an EDTA version of CATT/Trypanosoma brucei gambiense for serological screening of human blood samples Eddy Magnus 1 *, Veerle Lejon 1, Dominique Bayon 2, Dirk Buyse 3, Pere Simarro 4, Didier
Evaluation of an EDTA version of CATT/Trypanosoma brucei gambiense for serological screening of human blood samples Eddy Magnus 1 *, Veerle Lejon 1, Dominique Bayon 2, Dirk Buyse 3, Pere Simarro 4, Didier
Trypanosomiasis. Introduction. Epidemiology. Global Epidemiology. Trypanosomiasis Risk in UK Travellers
 Trypanosomiasis Introduction Epidemiology Risk for travellers Transmission Signs and symptoms Treatment Prevention References Reading list Links Introduction Trypanosomiasis is caused by parasitic protozoa
Trypanosomiasis Introduction Epidemiology Risk for travellers Transmission Signs and symptoms Treatment Prevention References Reading list Links Introduction Trypanosomiasis is caused by parasitic protozoa
Spotting Malaria reliably
 Spotting Malaria reliably Track down infections easily with highly sensitive Malaria-LAMP even in low-prevalent settings Molecular DX Microscopy and RDT s are not able to track down parasites in low-transmission
Spotting Malaria reliably Track down infections easily with highly sensitive Malaria-LAMP even in low-prevalent settings Molecular DX Microscopy and RDT s are not able to track down parasites in low-transmission
HIV-1 Genemer Detection Kit Ready to Use Amplification Kit for HIV-1 Specific DNA Fragment Analysis
 Product Manual HIV-1 Genemer Detection Kit Ready to Use Amplification Kit for HIV-1 Specific DNA Fragment Analysis For research use only. Not for use in diagnostic procedures for clinical purposes Catalog
Product Manual HIV-1 Genemer Detection Kit Ready to Use Amplification Kit for HIV-1 Specific DNA Fragment Analysis For research use only. Not for use in diagnostic procedures for clinical purposes Catalog
WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT. Product: Alere q HIV-1/2 Detect WHO reference number: PQDx
 WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT Product: Alere q HIV-1/2 Detect WHO reference number: PQDx 0226-032-00 Alere q HIV-1/2 Detect with product codes 270110050, 270110010 and 270300001,
WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT Product: Alere q HIV-1/2 Detect WHO reference number: PQDx 0226-032-00 Alere q HIV-1/2 Detect with product codes 270110050, 270110010 and 270300001,
Evaluation of (Rotat 1.2-PCR) Assays for Identifying Egyptian Trypanosoma evansi DNA
 ISSN 2079-2018 IDOSI Publications, 2012 DOI: 10.5829/idosi.apg.2012.3.1.6681 Evaluation of (Rotat 1.2-PCR) Assays for Identifying Egyptian Trypanosoma evansi DNA 1 1 2 Tarek R. Abou El-Naga, Safaa M. Barghash,
ISSN 2079-2018 IDOSI Publications, 2012 DOI: 10.5829/idosi.apg.2012.3.1.6681 Evaluation of (Rotat 1.2-PCR) Assays for Identifying Egyptian Trypanosoma evansi DNA 1 1 2 Tarek R. Abou El-Naga, Safaa M. Barghash,
Product Contents. 1 Specifications 1 Product Description. 2 Buffer Preparation... 3 Protocol. 3 Ordering Information 4
 INSTRUCTION MANUAL Quick-RNA Midiprep Kit Catalog No. R1056 Highlights 10 minute method for isolating RNA (up to 1 mg) from a wide range of cell types and tissue samples. Clean-Spin column technology allows
INSTRUCTION MANUAL Quick-RNA Midiprep Kit Catalog No. R1056 Highlights 10 minute method for isolating RNA (up to 1 mg) from a wide range of cell types and tissue samples. Clean-Spin column technology allows
Product Manual. Omni-Array Sense Strand mrna Amplification Kit, 2 ng to 100 ng Version Catalog No.: Reactions
 Genetic Tools and Reagents Universal mrna amplification, sense strand amplification, antisense amplification, cdna synthesis, micro arrays, gene expression, human, mouse, rat, guinea pig, cloning Omni-Array
Genetic Tools and Reagents Universal mrna amplification, sense strand amplification, antisense amplification, cdna synthesis, micro arrays, gene expression, human, mouse, rat, guinea pig, cloning Omni-Array
Blood Smears Only 3 February Sample Preparation and Quality Control
 NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 3 February 2015 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only
NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 3 February 2015 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only
E.Z.N.A. SQ Blood DNA Kit II. Table of Contents
 E.Z.N.A. SQ Blood DNA Kit II Table of Contents Introduction and Overview...2 Kit Contents/Storage and Stability...3 Blood Storage and DNA Yield...4 Preparing Reagents...5 100-500 μl Whole Blood Protocol...6
E.Z.N.A. SQ Blood DNA Kit II Table of Contents Introduction and Overview...2 Kit Contents/Storage and Stability...3 Blood Storage and DNA Yield...4 Preparing Reagents...5 100-500 μl Whole Blood Protocol...6
Comparison of light microscopy and nested PCR assay in detecting of malaria mixed species infections in an endemic area of Iran
 Comparison of light microscopy and nested PCR assay in detecting of malaria mixed species infections in an endemic area of Iran Aliehsan Heidari, Manizheh Nourian, Hossein Keshavarz Associate Prof. Dept.
Comparison of light microscopy and nested PCR assay in detecting of malaria mixed species infections in an endemic area of Iran Aliehsan Heidari, Manizheh Nourian, Hossein Keshavarz Associate Prof. Dept.
Laboratory diagnosis of Blood and tissue flagellates
 Laboratory diagnosis of Blood and tissue flagellates (Leishmania and trypanosma) Sarah Alharbi Clinical Laboratory department Collage of Applied Medical Sciences King Saud University Leishmania and trypanosma:
Laboratory diagnosis of Blood and tissue flagellates (Leishmania and trypanosma) Sarah Alharbi Clinical Laboratory department Collage of Applied Medical Sciences King Saud University Leishmania and trypanosma:
Cytomegalovirus (CMV) End-Point PCR Kit Product# EP36300
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Cytomegalovirus (CMV) End-Point PCR Kit Product# EP36300 Product
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Cytomegalovirus (CMV) End-Point PCR Kit Product# EP36300 Product
For Research Use Only Ver
 INSTRUCTION MANUAL Quick-cfDNA Serum & Plasma Kit Catalog No. D4076 Highlights High-quality DNA, including cell-free, is easily and robustly purified from up to 10 ml of serum/plasma, up to 1 ml amniotic
INSTRUCTION MANUAL Quick-cfDNA Serum & Plasma Kit Catalog No. D4076 Highlights High-quality DNA, including cell-free, is easily and robustly purified from up to 10 ml of serum/plasma, up to 1 ml amniotic
In vitro Parasite count and EC 50 in trypanosome cultures incubated with some selected iron chelators
 Global Advanced Research Journal of Medicine and Medical Sciences Vol. 1(1) pp. 017-026, March, 2012 Available online http://garj.org/garjmms/index.htm Copyright 2012 Global Advanced Research Journals
Global Advanced Research Journal of Medicine and Medical Sciences Vol. 1(1) pp. 017-026, March, 2012 Available online http://garj.org/garjmms/index.htm Copyright 2012 Global Advanced Research Journals
INSTRUCTION MANUAL. RNA Clean & Concentrator -5 Catalog Nos. R1015 & R1016. Highlights. Contents
 INSTRUCTION MANUAL Catalog Nos. R1015 & R1016 Highlights Quick (5 minute) method for cleaning and concentrating RNA. Ideal for purification of RNA from aqueous phase following an acid phenol extraction.
INSTRUCTION MANUAL Catalog Nos. R1015 & R1016 Highlights Quick (5 minute) method for cleaning and concentrating RNA. Ideal for purification of RNA from aqueous phase following an acid phenol extraction.
Purification of viral nucleic acid from serum, plasma, cell-free biological fluids MACHEREY- NAGEL
 Purification of viral nucleic acid from serum, plasma, cell-free biological fluids Purification of viral nucleic acid from serum, plasma, cell-free biological fluids viral RNA: viral DNA: NucleoSpin RNA
Purification of viral nucleic acid from serum, plasma, cell-free biological fluids Purification of viral nucleic acid from serum, plasma, cell-free biological fluids viral RNA: viral DNA: NucleoSpin RNA
ABIOpure TM Viral (version 2.0)
 ABIOpure TM Viral (version 2.0) DNA/RNA Extraction Handbook Cat No: M561VT50 FOR RESEARCH USE ONLY Table of Contents Contents Page Kit Components 3 Precautions 3 Stability & Storage 4 General Description
ABIOpure TM Viral (version 2.0) DNA/RNA Extraction Handbook Cat No: M561VT50 FOR RESEARCH USE ONLY Table of Contents Contents Page Kit Components 3 Precautions 3 Stability & Storage 4 General Description
Stage determination in sleeping sickness: comparison of two cell counting and two parasite detection techniques
 Tropical Medicine and International Health doi:1.1111/tmi.11 volume 1 no pp 77 7 june 13 Stage determination in sleeping sickness: comparison of two cell counting and two parasite detection techniques
Tropical Medicine and International Health doi:1.1111/tmi.11 volume 1 no pp 77 7 june 13 Stage determination in sleeping sickness: comparison of two cell counting and two parasite detection techniques
Pinpoint Slide RNA Isolation System II Catalog No. R1007
 INSTRUCTION MANUAL Pinpoint Slide RNA Isolation System II Catalog No. R1007 Highlights Allows for the isolation of total RNA from paraffin-embedded tissue sections on glass slides Simple procedure combines
INSTRUCTION MANUAL Pinpoint Slide RNA Isolation System II Catalog No. R1007 Highlights Allows for the isolation of total RNA from paraffin-embedded tissue sections on glass slides Simple procedure combines
PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature
 PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature PRODUCT DESCRIPTION. RNAzol BD is a reagent for isolation of total RNA from whole blood, plasma or serum of human
PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature PRODUCT DESCRIPTION. RNAzol BD is a reagent for isolation of total RNA from whole blood, plasma or serum of human
For the 5 GATC-overhang two-oligo adaptors set up the following reactions in 96-well plate format:
 Supplementary Protocol 1. Adaptor preparation: For the 5 GATC-overhang two-oligo adaptors set up the following reactions in 96-well plate format: Per reaction X96 10X NEBuffer 2 10 µl 10 µl x 96 5 -GATC
Supplementary Protocol 1. Adaptor preparation: For the 5 GATC-overhang two-oligo adaptors set up the following reactions in 96-well plate format: Per reaction X96 10X NEBuffer 2 10 µl 10 µl x 96 5 -GATC
Received 1 July 2004/Returned for modification 3 August 2004/Accepted 12 August 2004
 INFECTION AND IMMUNITY, Dec. 2004, p. 7040 7044 Vol. 72, No. 12 0019-9567/04/$08.00 0 DOI: 10.1128/IAI.72.12.7040 7044.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Severity
INFECTION AND IMMUNITY, Dec. 2004, p. 7040 7044 Vol. 72, No. 12 0019-9567/04/$08.00 0 DOI: 10.1128/IAI.72.12.7040 7044.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Severity
Instructions for Use. RealStar Influenza S&T RT-PCR Kit /2017 EN
 Instructions for Use RealStar Influenza S&T RT-PCR Kit 3.0 01/2017 EN RealStar Influenza S&T RT-PCR Kit 3.0 For research use only! (RUO) 163003 INS-163000-EN-S02 96 01 2017 altona Diagnostics GmbH Mörkenstr.
Instructions for Use RealStar Influenza S&T RT-PCR Kit 3.0 01/2017 EN RealStar Influenza S&T RT-PCR Kit 3.0 For research use only! (RUO) 163003 INS-163000-EN-S02 96 01 2017 altona Diagnostics GmbH Mörkenstr.
Instructions for Use. APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests
 3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
For in vitro Veterinary Diagnostics only. Kylt Rotavirus A. Real-Time RT-PCR Detection.
 For in vitro Veterinary Diagnostics only. Kylt Rotavirus A Real-Time RT-PCR Detection www.kylt.eu DIRECTION FOR USE Kylt Rotavirus A Real-Time RT-PCR Detection A. General Kylt Rotavirus A products are
For in vitro Veterinary Diagnostics only. Kylt Rotavirus A Real-Time RT-PCR Detection www.kylt.eu DIRECTION FOR USE Kylt Rotavirus A Real-Time RT-PCR Detection A. General Kylt Rotavirus A products are
HIV-1 Viral Load Real Time (RG)
 -1 Viral Load Real Time (RG) Real Time RT-PCR type 1 RNA quantification assay MSP Reg. pending Valdense 3616. 11700. Montevideo. Uruguay. phone (598) 2 336 83 01. Fax (598) 2 336 71 60. Info@atgen.com.uy
-1 Viral Load Real Time (RG) Real Time RT-PCR type 1 RNA quantification assay MSP Reg. pending Valdense 3616. 11700. Montevideo. Uruguay. phone (598) 2 336 83 01. Fax (598) 2 336 71 60. Info@atgen.com.uy
SensoLyte pnpp Alkaline Phosphatase Assay Kit *Colorimetric*
 SensoLyte pnpp Alkaline Phosphatase Assay Kit *Colorimetric* Catalog # 72146 Kit Size 500 Assays (96-well plate) Optimized Performance: This kit is optimized to detect alkaline phosphatase activity Enhanced
SensoLyte pnpp Alkaline Phosphatase Assay Kit *Colorimetric* Catalog # 72146 Kit Size 500 Assays (96-well plate) Optimized Performance: This kit is optimized to detect alkaline phosphatase activity Enhanced
AIDS - Knowledge and Dogma. Conditions for the Emergence and Decline of Scientific Theories Congress, July 16/ , Vienna, Austria
 AIDS - Knowledge and Dogma Conditions for the Emergence and Decline of Scientific Theories Congress, July 16/17 2010, Vienna, Austria Reliability of PCR to detect genetic sequences from HIV Juan Manuel
AIDS - Knowledge and Dogma Conditions for the Emergence and Decline of Scientific Theories Congress, July 16/17 2010, Vienna, Austria Reliability of PCR to detect genetic sequences from HIV Juan Manuel
Product Contents. 1 Specifications 1 Product Description. 2 Buffer Preparation... 3 Protocol. 3 Ordering Information 4 Related Products..
 INSTRUCTION MANUAL Quick-RNA MidiPrep Catalog No. R1056 Highlights 10 minute method for isolating RNA (up to 1 mg) from a wide range of cell types and tissue samples. Clean-Spin column technology allows
INSTRUCTION MANUAL Quick-RNA MidiPrep Catalog No. R1056 Highlights 10 minute method for isolating RNA (up to 1 mg) from a wide range of cell types and tissue samples. Clean-Spin column technology allows
Kinetoplastids Handout
 Kinetoplastids Handout 1 Kinetoplastids widespread group of flagellated protozoa parasitize virtually all animal groups as well as plants and insects 3 distinct kinetoplastid species cause human disease
Kinetoplastids Handout 1 Kinetoplastids widespread group of flagellated protozoa parasitize virtually all animal groups as well as plants and insects 3 distinct kinetoplastid species cause human disease
For purification of viral DNA and RNA from a wide range of sample materials
 QIAamp virus kits For purification of viral DNA and RNA from a wide range of sample materials Automatable on QIAGEN s proven QIAamp Kits set the standard for purification of viral DNA and RNA. QIAamp virus
QIAamp virus kits For purification of viral DNA and RNA from a wide range of sample materials Automatable on QIAGEN s proven QIAamp Kits set the standard for purification of viral DNA and RNA. QIAamp virus
Internal Validation Guide of Y-STR Systems for Forensic Laboratories Printed in USA. 11/12 Part# GE713
 REFERENCE MANUAL Internal Validation Guide of Y-STR Systems for Forensic Laboratories 11/12 Internal Validation Guide of Y-STR Systems for Forensic Laboratories PLEASE DISCARD PREVIOUS VERSIONS. All technical
REFERENCE MANUAL Internal Validation Guide of Y-STR Systems for Forensic Laboratories 11/12 Internal Validation Guide of Y-STR Systems for Forensic Laboratories PLEASE DISCARD PREVIOUS VERSIONS. All technical
Instructions for Use. RealStar Influenza Screen & Type RT-PCR Kit /2017 EN
 Instructions for Use RealStar Influenza Screen & Type RT-PCR Kit 4.0 05/2017 EN RealStar Influenza Screen & Type RT-PCR Kit 4.0 For research use only! (RUO) 164003 INS-164000-EN-S01 96 05 2017 altona
Instructions for Use RealStar Influenza Screen & Type RT-PCR Kit 4.0 05/2017 EN RealStar Influenza Screen & Type RT-PCR Kit 4.0 For research use only! (RUO) 164003 INS-164000-EN-S01 96 05 2017 altona
Europium Labeling Kit
 Europium Labeling Kit Catalog Number KA2096 100ug *1 Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
Europium Labeling Kit Catalog Number KA2096 100ug *1 Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
Global Histone H3 Acetylation Assay Kit
 Global Histone H3 Acetylation Assay Kit Catalog Number KA0633 96 assays Version: 06 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Global Histone H3 Acetylation Assay Kit Catalog Number KA0633 96 assays Version: 06 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Identification of Trypanosoma brucei rhodesiense in wild Tsetse flies in Nkhotakota Wild Life Reserve by PCR
 Identification of Trypanosoma brucei rhodesiense in wild Tsetse flies in Nkhotakota Wild Life Reserve by PCR 32 nd International Scientific Council for Trypanosomiasis Research and Control (ISCTRC), September
Identification of Trypanosoma brucei rhodesiense in wild Tsetse flies in Nkhotakota Wild Life Reserve by PCR 32 nd International Scientific Council for Trypanosomiasis Research and Control (ISCTRC), September
Role of Paired Box9 (PAX9) (rs ) and Muscle Segment Homeobox1 (MSX1) (581C>T) Gene Polymorphisms in Tooth Agenesis
 EC Dental Science Special Issue - 2017 Role of Paired Box9 (PAX9) (rs2073245) and Muscle Segment Homeobox1 (MSX1) (581C>T) Gene Polymorphisms in Tooth Agenesis Research Article Dr. Sonam Sethi 1, Dr. Anmol
EC Dental Science Special Issue - 2017 Role of Paired Box9 (PAX9) (rs2073245) and Muscle Segment Homeobox1 (MSX1) (581C>T) Gene Polymorphisms in Tooth Agenesis Research Article Dr. Sonam Sethi 1, Dr. Anmol
Pelagia Research Library. European Journal of Experimental Biology, 2015, 5(10):1-5
 Available online at www.pelagiaresearchlibrary.com European Journal of Experimental Biology, 2015, 5(10):1-5 ISSN: 2248 9215 CODEN (USA): EJEBAU Molecular diagnosis of human immuno deficiency virus (HIV)
Available online at www.pelagiaresearchlibrary.com European Journal of Experimental Biology, 2015, 5(10):1-5 ISSN: 2248 9215 CODEN (USA): EJEBAU Molecular diagnosis of human immuno deficiency virus (HIV)
EXO-DNAc Circulating and EV-associated DNA extraction kit
 Datasheet EXO-DNAc Circulating and EV-associated DNA extraction kit This product is for research use only. It is highly recommended to read this users guide in its entirety prior to using this product.
Datasheet EXO-DNAc Circulating and EV-associated DNA extraction kit This product is for research use only. It is highly recommended to read this users guide in its entirety prior to using this product.
Urine - Based XMRV RT-PCR Detection Kit Product # 34700
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Urine - Based XMRV RT-PCR Detection Kit Product # 34700 Product
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Urine - Based XMRV RT-PCR Detection Kit Product # 34700 Product
Parasitic Protozoa, Helminths, and Arthropod Vectors
 PowerPoint Lecture Slides for MICROBIOLOGY ROBERT W. BAUMAN Chapter 23 Parasitic Protozoa, Helminths, and Arthropod Vectors Parasitic Diseases Protozoan and helminthic parasites are emerging as serious
PowerPoint Lecture Slides for MICROBIOLOGY ROBERT W. BAUMAN Chapter 23 Parasitic Protozoa, Helminths, and Arthropod Vectors Parasitic Diseases Protozoan and helminthic parasites are emerging as serious
THE ROLE OF LIVESTOCK IN THE EPIDEMIOLOGY OF SLEEPING SICKNESS IN TANZANIA
 THE ROLE OF LIVESTOCK IN THE EPIDEMIOLOGY OF SLEEPING SICKNESS IN TANZANIA Imna Malele, Hamisi Nyingilili, Eugen Lyaruu, Winston Kitwika Tsetse & Trypanosomiasis Research Institute Majani Mapana, Off Korogwe
THE ROLE OF LIVESTOCK IN THE EPIDEMIOLOGY OF SLEEPING SICKNESS IN TANZANIA Imna Malele, Hamisi Nyingilili, Eugen Lyaruu, Winston Kitwika Tsetse & Trypanosomiasis Research Institute Majani Mapana, Off Korogwe
African Trypanosomiasis Detection Using Dempster-Shafer Theory Andino Maseleno, 2 Md. Mahmud Hasan
 African Trypanosomiasis Detection Using Dempster-Shafer Theory 1 Andino Maseleno, 2 Md. Mahmud Hasan 1 Department of Computer Science, Faculty of Science, Universiti Brunei Darussalam 2 Jalan Tungku Link,
African Trypanosomiasis Detection Using Dempster-Shafer Theory 1 Andino Maseleno, 2 Md. Mahmud Hasan 1 Department of Computer Science, Faculty of Science, Universiti Brunei Darussalam 2 Jalan Tungku Link,
Confirmation of antibodies against L-tryptophan-like epitope in human African trypanosomosis serological diagnostic
 Vol. 15(36), pp. 1986-1990, 7 September, 2016 DOI: 10.5897/AJB2016.15575 Article Number: 273864A60390 ISSN 1684-5315 Copyright 2016 Author(s) retain the copyright of this article http://www.academicjournals.org/ajb
Vol. 15(36), pp. 1986-1990, 7 September, 2016 DOI: 10.5897/AJB2016.15575 Article Number: 273864A60390 ISSN 1684-5315 Copyright 2016 Author(s) retain the copyright of this article http://www.academicjournals.org/ajb
Performance of Parasitological and Molecular Techniques for the Diagnosis and Surveillance of Gambiense Sleeping Sickness
 Performance of Parasitological and Molecular Techniques for the Diagnosis and Surveillance of Gambiense Sleeping Sickness Dieudonné Mumba Ngoyi 1,2, Rosine Ali Ekangu 1, Marie France Mumvemba Kodi 1, Patient
Performance of Parasitological and Molecular Techniques for the Diagnosis and Surveillance of Gambiense Sleeping Sickness Dieudonné Mumba Ngoyi 1,2, Rosine Ali Ekangu 1, Marie France Mumvemba Kodi 1, Patient
Mouse Cathepsin B ELISA Kit
 GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Mouse Cathepsin B ELISA Kit Catalog No. GWB-ZZD154
GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Mouse Cathepsin B ELISA Kit Catalog No. GWB-ZZD154
For Research Use Only Ver
 INSTRUCTION MANUAL Quick-RNA Miniprep Kit Catalog Nos. R1054 & R1055 Highlights High-quality total RNA (including small RNAs) from a wide range of samples. You can opt to isolate small and large RNAs in
INSTRUCTION MANUAL Quick-RNA Miniprep Kit Catalog Nos. R1054 & R1055 Highlights High-quality total RNA (including small RNAs) from a wide range of samples. You can opt to isolate small and large RNAs in
For Research Use Only Ver
 INSTRUCTION MANUAL Quick-RNA Miniprep Kit Catalog Nos. R1054 & R1055 Highlights High-quality total RNA (including small RNAs) from a wide range of samples. You can opt to isolate small and large RNAs in
INSTRUCTION MANUAL Quick-RNA Miniprep Kit Catalog Nos. R1054 & R1055 Highlights High-quality total RNA (including small RNAs) from a wide range of samples. You can opt to isolate small and large RNAs in
EPIGENTEK. EpiQuik Global Histone H3 Acetylation Assay Kit. Base Catalog # P-4008 PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE
 EpiQuik Global Histone H3 Acetylation Assay Kit Base Catalog # PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE The EpiQuik Global Histone H3 Acetylation Assay Kit is suitable for specifically measuring global
EpiQuik Global Histone H3 Acetylation Assay Kit Base Catalog # PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE The EpiQuik Global Histone H3 Acetylation Assay Kit is suitable for specifically measuring global
EPIGENTEK. EpiQuik Global Histone H4 Acetylation Assay Kit. Base Catalog # P-4009 PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE
 EpiQuik Global Histone H4 Acetylation Assay Kit Base Catalog # PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE The EpiQuik Global Histone H4 Acetylation Assay Kit is suitable for specifically measuring global
EpiQuik Global Histone H4 Acetylation Assay Kit Base Catalog # PLEASE READ THIS ENTIRE USER GUIDE BEFORE USE The EpiQuik Global Histone H4 Acetylation Assay Kit is suitable for specifically measuring global
MALARIA P. FALCIPARUM / P. VIVAX
 MALARIA P. FALCIPARUM / P. VIVAX 1. EXPLANATION OF THE TEST: Malaria is a serious, sometimes fatal, parasitic disease characterized by fever, chills, and anemia and is caused by a parasite that is transmitted
MALARIA P. FALCIPARUM / P. VIVAX 1. EXPLANATION OF THE TEST: Malaria is a serious, sometimes fatal, parasitic disease characterized by fever, chills, and anemia and is caused by a parasite that is transmitted
For Research Use Only Ver
 INSTRUCTION MANUAL ZR Viral RNA Kit Catalog Nos. R1034 & R1035 Highlights Quick, 5-minute recovery of viral RNA from plasma, serum and other samples. Omits the use of organic denaturants and proteases.
INSTRUCTION MANUAL ZR Viral RNA Kit Catalog Nos. R1034 & R1035 Highlights Quick, 5-minute recovery of viral RNA from plasma, serum and other samples. Omits the use of organic denaturants and proteases.
Loop-Mediated Isothermal Amplification (LAMP) Method for Rapid Detection of Trypanosoma brucei rhodesiense
 Loop-Mediated Isothermal Amplification (LAMP) Method for Rapid Detection of Trypanosoma brucei rhodesiense Zablon Kithinji Njiru 1,2 *, Andrew Stanislaw John Mikosza 3, Tanya Armstrong 3, John Charles
Loop-Mediated Isothermal Amplification (LAMP) Method for Rapid Detection of Trypanosoma brucei rhodesiense Zablon Kithinji Njiru 1,2 *, Andrew Stanislaw John Mikosza 3, Tanya Armstrong 3, John Charles
M E M O R A N D U M. No new drug is in clinical development for second stage HAT treatment.
 M E M O R A N D U M From: Director, NTD To: Secretary, Expert Committee on the Selection and Use of Essential Medicines Our ref: EML 2009 Attention: Dr Suzanne Hill Date: 13 February 2009 Your ref: E19/81/17
M E M O R A N D U M From: Director, NTD To: Secretary, Expert Committee on the Selection and Use of Essential Medicines Our ref: EML 2009 Attention: Dr Suzanne Hill Date: 13 February 2009 Your ref: E19/81/17
KINETOPLASTIDS. Kinetoplast. Nucleus
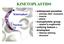 KINETOPLASTIDS Kinetoplast Nucleus widespread parasites animals (fish humans) insects plants monophyletic group related to euglenoids unifying feature = kinetoplast Giemsa staining structure KINETOPLAST
KINETOPLASTIDS Kinetoplast Nucleus widespread parasites animals (fish humans) insects plants monophyletic group related to euglenoids unifying feature = kinetoplast Giemsa staining structure KINETOPLAST
Phosphate buffered saline (PBS) for washing the cells TE buffer (nuclease-free) ph 7.5 for use with the PrimePCR Reverse Transcription Control Assay
 Catalog # Description 172-5080 SingleShot Cell Lysis Kit, 100 x 50 µl reactions 172-5081 SingleShot Cell Lysis Kit, 500 x 50 µl reactions For research purposes only. Introduction The SingleShot Cell Lysis
Catalog # Description 172-5080 SingleShot Cell Lysis Kit, 100 x 50 µl reactions 172-5081 SingleShot Cell Lysis Kit, 500 x 50 µl reactions For research purposes only. Introduction The SingleShot Cell Lysis
altona RealStar Instructions for Use RealStar CMV PCR Kit /2017 EN DIAGNOSTICS
 altona DIAGNOSTICS Instructions for Use RealStar CMV PCR Kit 1.2 08/2017 EN RealStar RealStar CMV PCR Kit 1.2 For research use only! (RUO) 021202 INS-021200-EN-S01 48 08 2017 altona Diagnostics GmbH Mörkenstr.
altona DIAGNOSTICS Instructions for Use RealStar CMV PCR Kit 1.2 08/2017 EN RealStar RealStar CMV PCR Kit 1.2 For research use only! (RUO) 021202 INS-021200-EN-S01 48 08 2017 altona Diagnostics GmbH Mörkenstr.
EXO-DNA Circulating and EV-associated DNA extraction kit
 Datasheet EXO-DNA Circulating and EV-associated DNA extraction kit This product is for research use only. It is highly recommended to read this users guide in its entirety prior to using this product.
Datasheet EXO-DNA Circulating and EV-associated DNA extraction kit This product is for research use only. It is highly recommended to read this users guide in its entirety prior to using this product.
HAT DNDi program progress and evolution in the path to elimination. Dr Antoine Tarral. HAT Platform, Kampala October 2018
 HAT DNDi program progress and evolution in the path to elimination Dr Antoine Tarral HAT Platform, Kampala October 2018 DNDi s Mission To develop new drugs or new formulations of existing drugs for people
HAT DNDi program progress and evolution in the path to elimination Dr Antoine Tarral HAT Platform, Kampala October 2018 DNDi s Mission To develop new drugs or new formulations of existing drugs for people
PRODUCT INFORMATION & MANUAL
 PRODUCT INFORMATION & MANUAL 0.4 micron for Overall Exosome Isolation (Cell Media) NBP2-49826 For research use only. Not for diagnostic or therapeutic procedures. www.novusbio.com - P: 303.730.1950 - P:
PRODUCT INFORMATION & MANUAL 0.4 micron for Overall Exosome Isolation (Cell Media) NBP2-49826 For research use only. Not for diagnostic or therapeutic procedures. www.novusbio.com - P: 303.730.1950 - P:
F. Chappuis 1, A. Pittet 1, P. A. Bovier 2, K. Adams 1, V. Godineau 1, S. Y. Hwang 1, E. Magnus 3 and P. Büscher 3
 Tropical Medicine and International Health volume 7 no 11 pp 942 948 november 2002 Field evaluation of the CATT/Trypanosoma brucei gambiense on blood-impregnated filter papers for diagnosis of human African
Tropical Medicine and International Health volume 7 no 11 pp 942 948 november 2002 Field evaluation of the CATT/Trypanosoma brucei gambiense on blood-impregnated filter papers for diagnosis of human African
اعداد رغداحمد رغد جمال الدين
 اعداد رغداحمد رغد جمال الدين Trypanosoma Causes Trypanosomiasis West African Trypanosomiasis T.brucei gambiense Sleeping sickness East African Trypanosomiasis T.brucei rhodesiense American Trypanosomiasis
اعداد رغداحمد رغد جمال الدين Trypanosoma Causes Trypanosomiasis West African Trypanosomiasis T.brucei gambiense Sleeping sickness East African Trypanosomiasis T.brucei rhodesiense American Trypanosomiasis
LANCE Eu-W1024 ITC Chelate & Europium Standard AD0013 Development grade
 AD0017P-4 (en) 1 LANCE Eu-W1024 ITC Chelate & Europium Standard AD0013 Development grade INTRODUCTION Fluorescent isothiocyanato-activated (ITC-activated) Eu-W1024 chelate is optimized for labelling proteins
AD0017P-4 (en) 1 LANCE Eu-W1024 ITC Chelate & Europium Standard AD0013 Development grade INTRODUCTION Fluorescent isothiocyanato-activated (ITC-activated) Eu-W1024 chelate is optimized for labelling proteins
Protocol for Gene Transfection & Western Blotting
 The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
Characterization of Trypanosoma brucei gambiense variant surface glycoprotein LiTat 1.5
 Short Communication Characterization of Trypanosoma brucei gambiense variant surface glycoprotein LiTat 1.5 L. Van Nieuwenhove 1 *, S. Rogé 1 *, V. Lejon 1, Y. Guisez 2 and P. Büscher 1 1 Department of
Short Communication Characterization of Trypanosoma brucei gambiense variant surface glycoprotein LiTat 1.5 L. Van Nieuwenhove 1 *, S. Rogé 1 *, V. Lejon 1, Y. Guisez 2 and P. Büscher 1 1 Department of
WHO Prequalification of Diagnostics Programme PUBLIC REPORT. Product: VERSANT HIV-1 RNA 1.0 Assay (kpcr) Number: PQDx
 WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: VERSANT HIV-1 RNA 1.0 Assay (kpcr) Number: PQDx 0115-041-00 Abstract The VERSANT HIV-1 RNA 1.0 Assay (kpcr) with product codes 10375763,
WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: VERSANT HIV-1 RNA 1.0 Assay (kpcr) Number: PQDx 0115-041-00 Abstract The VERSANT HIV-1 RNA 1.0 Assay (kpcr) with product codes 10375763,
Supplemental Materials and Methods Plasmids and viruses Quantitative Reverse Transcription PCR Generation of molecular standard for quantitative PCR
 Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
CHAPTER 5. Population genetic structure and cladistic analysis of Trypanosoma brucei isolates
 CHAPTER 5 Population genetic structure and cladistic analysis of Trypanosoma brucei isolates Eddy Chukwura Agbo 1, Peter-Henning Clausen 2, Philippe Büscher 3, Phelix A.O. Majiwa 4, Eric Claassen 5, and
CHAPTER 5 Population genetic structure and cladistic analysis of Trypanosoma brucei isolates Eddy Chukwura Agbo 1, Peter-Henning Clausen 2, Philippe Büscher 3, Phelix A.O. Majiwa 4, Eric Claassen 5, and
Urine - Based CMV PCR Detection Kit Product # 36300
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Urine - Based CMV PCR Detection Kit Product # 36300 Product Insert
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Urine - Based CMV PCR Detection Kit Product # 36300 Product Insert
Blood Smears Only 20 May Sample Preparation and Quality Control
 NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 20 May 2014 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only is
NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 20 May 2014 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only is
APPLICATIONS OF LATENT CLASS ANALYSIS IN DIAGNOSTIC VALIDATION, THE USER PERSPECTIVE
 APPLICATIONS OF LATENT CLASS ANALYSIS IN DIAGNOSTIC VALIDATION, THE USER PERSPECTIVE Author's name(s): Boelaert M 1 *, Verloo D. 2, Van der Stuyft P 1 Affiliation(s): 1 Prince Leopold Institute of Tropical
APPLICATIONS OF LATENT CLASS ANALYSIS IN DIAGNOSTIC VALIDATION, THE USER PERSPECTIVE Author's name(s): Boelaert M 1 *, Verloo D. 2, Van der Stuyft P 1 Affiliation(s): 1 Prince Leopold Institute of Tropical
Disease Transmission Methods
 Disease Transmission Methods In epidemiology, transmission simply means any method by which an infectious agent is spread from one host to another. Knowing the type of pathogen often, but not always, identifies
Disease Transmission Methods In epidemiology, transmission simply means any method by which an infectious agent is spread from one host to another. Knowing the type of pathogen often, but not always, identifies
Total Histone H3 Acetylation Detection Fast Kit (Colorimetric)
 Total Histone H3 Acetylation Detection Fast Kit (Colorimetric) Catalog Number KA1538 48 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use...
Total Histone H3 Acetylation Detection Fast Kit (Colorimetric) Catalog Number KA1538 48 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use...
