Examining the effects of sleep delay on depressed males and females and healthy controls
|
|
|
- Logan Gilbert
- 5 years ago
- Views:
Transcription
1 J Sleep Res. (2014) 23, Depression and sleep Examining the effects of sleep delay on depressed males and females and healthy controls JENNIFER R. GOLDSCHMIED, PHILIP CHENG, ROSEANNE ARMITAGE and PATRICIA J. DELDIN Department of Psychology, University of Michigan, Ann Arbor, MI, USA Keywords sex effects, slow wave activity, sleep regulation Correspondence Jennifer Goldschmied, MS, Department of Psychology, University of Michigan, 530 Church Street, Ann Arbor, MI 48109, USA. fax: ; Accepted in revised form 10 May 2014; received 21 February 2014 DOI: /jsr SUMMARY Individuals with major depressive disorder typically exhibit sleep electroencephalograpy abnormalities which have been shown to vary by sex. Recent research has shown that depressed males display deficits in slow wave sleep and delta electroencephalograph (EEG) activity that are not apparent in depressed females. This may suggest that males and females with depression vary with respect to their homeostatic regulation of sleep. Utilizing archival data, the present study examined the effects of a 3-h sleep delay, which represents a mild sleep challenge, on slow wave activity in healthy controls and individuals with depression. All participants slept in the laboratory for three sequential nights. On the third night in the laboratory, the participants bedtime was delayed by 3 h. Slow wave activity was calculated utilizing power spectral analysis and compared across groups. Following the sleep delay, males with depression exhibited the lowest slow wave activity compared to all other groups. These results may suggest that males with depression are at a greater risk for homeostatic dysregulation than females, and may require specialized intervention. INTRODUCTION Sleep disturbance is one of the central symptoms reported by most individuals with major depressive disorder (MDD; Mendlewicz, 2009). Years of research looking at differences in sleep between healthy subjects and those diagnosed with MDD have greatly increased our understanding of the qualitative difference in sleep in those with MDD. With regard to the most common differences in visually scored sleep variables, research has shown consistently that individuals with MDD have more proportionate time awake, a measure of sleep efficiency, shorter time to enter REM sleep (REM latency), greater amount of REM sleep, increased amounts of rapid eye movements (REM density) and decreases in slow wave sleep compared to healthy controls. Understanding these qualitative differences has been an essential first step; however, newer methodology, including quantitative electroencephalograph (EEG) analysis, may allow us to assess more clearly the differences in sleep regulation in MDD. Borbely and Wirz-Justice (1982) suggested that the sleep disturbances occurring in individuals with MDD, including delayed sleep onset and difficulty maintaining sleep, were a result of a reduced drive for sleep (process S). This model also suggests that those with MDD would present reduced 664 slow wave sleep during the first portion of the night, when slow wave activity is typically at its highest. Kupfer et al. (1986) demonstrated that there was indeed a reduction in slow wave EEG activity in NREM sleep (SWA) in those with MDD; however, they also noted that there was only a very small relationship between time awake and reduced delta activity in those with MDD. Utilizing quantitative EEG analyses, Armitage et al. (1992, 1995, 1997, 2000a,b) have also shown that individuals with MDD exhibit an abnormal distribution of SWA across the night. These findings suggest that those with MDD exhibit greater dysfunction than originally theorized with respect to physiological functioning, including homeostatic regulation. Research into the function of SWA has shown that SWA increases proportionately to the amount of prior wakefulness and dissipates rapidly over NREM sleep time (Borbely, 1981). Moreover, how quickly SWA asymptotes after sleep onset is presumed to reflect the basic sleep drive, whereas SWA dissipation has been taken as a proxy for neuronal recovery function. In order to explore SWA regulation and its relationship to homeostatic functioning it is necessary to utilize a sleep manipulation, such as sleep deprivation, to challenge the system. A 3-h sleep delay, as described in Armitage et al. (2007, 2012), has been utilized successfully
2 Sleep delay effects in males and females with MDD 665 to study SWA response in MDD, as it results in similar increases in SWA in the first NREM period as other sleep manipulations, indicative of the increase in the homeostatic sleep response (Armitage and Hoffmann, 2001; Dijk, 1991). This procedure also allows total sleep time to be held constant, in addition to allowing recovery sleep to occur during the same night. Thus far, the evidence supports both reduced sleep drive and impaired recovery in individuals with MDD. However, none of these studies took into account that sex differences may modify the results. The incidence of MDD is two times greater in females than males, and as a result, there has been much research into the sex differences in MDD. Somatic symptoms, including complaints of fatigue and insomnia, have been found to be twice as high in females as in males, in both lifetime and 12-month estimates of MDD (Silverstein et al., 2013). There are also substantial sex differences in the neurobiology of MDD between sexes, including the findings that there is a hyper-response in the HPA axis in females with MDD, and that females respond differently to antidepressants revealing a difference in the pharmacokinetics and dynamics of the female brain and metabolic system (Armitage, 2007). Additionally, while waking EEG asymmetry has typically been found in frontal regions in samples including female participants, Knott et al. (2001) found evidence of such asymmetry over both frontal and other regions in a solely male sample of depressed patients. They interpreted these findings to suggest that depression in males affects unique brain areas than those affected in females. Less research, however, has been conducted into the sex differences in MDD in sleep. Armitage et al. (2000a) have noted that the sex differences in individuals with MDD are twice as large as those in healthy controls. With respect to the regulation of SWA in MDD, Reynolds et al. (1990) demonstrated that males with MDD had lower delta wave counts during the first NREM period, and across the night, compared to females who exhibited significantly higher delta activity. Additionally, utilizing a subset of participants from the present sample (n = 30), Armitage (2007) noted that males with MDD demonstrate an impaired time course for delta activity, including slower dissipation, as indicated by a slower rate of decay from exponential regression analyses during the course of the night. In contrast, Armitage et al. (2000a) also demonstrated that females with MDD do not show impaired homeostatic regulation of SWA, but rather show more poorly synchronized sleep EEG rhythms and increased fast frequency EEG at baseline than males with MDD (2001; 1995) which, they suggest, is indicative of more global EEG frequency dysregulation (2007) and more dynamic organization of sleep microarchitecture (2010). Taken together, these findings suggest that MDD does not statically affect males and females, especially with respect to sleep, and perhaps other physiological regulatory mechanisms. Whereas females may display more generalized EEG dysregulation, the slower dissipation of SWA exhibited by males with MDD reflects a dysfunctional recovery process and may thus suggest that general homeostatic functioning of males is more impaired than females. In addition, very recent research has looked into the topography of SWA in both individuals with MDD and healthy controls using high-density EEG (Plante et al., 2012). Results of this work revealed that SWA was higher in females with MDD compared to age-matched healthy controls, found most significantly in the bilateral prefrontal regions during the first NREM period. Alternatively, they did not find any differences in SWA in males with depression compared to healthy males. These findings further substantiate the claim that there are sexspecific impairments in MDD, and may also suggest that females with MDD have developed mechanisms with regard to SWA regulation, specifically, to compensate for certain disease-related dysfunction. The purpose of the current study was to confirm sex differences in SWA, in MDD compared to healthy controls, and extend the work to include assessments of the accumulation, dissipation and topography of SWA in response to a mild sleep challenge. We predicted that men with MDD would show a blunted response to the challenge, whereas women with MDD would more resemble their healthy counterparts. METHODS Participants Sleep data were selected from our archival database of participants collected over the past 10 years from the Sleep and Chronophysiology laboratory at the University of Texas Southwestern Medical Center at Dallas (UTSW) and the University of Michigan (UM), under the same conditions. Participants were self-referred and responded to advertisements in the community, and habitually slept between 6 and 8 h per night. Inclusion criteria for the study required 2 consecutive nights of recording without any difficulties or deviations from protocol. As determined by medical history or polysomnogram, participants were free of sleep disorders including narcolepsy, sleep apnea, bruxism or periodic limb movements, and were not engaged in shiftwork. All participants were unmedicated, other than non-steroidal antiinflammatory drugs, prior to sleep study for 4 weeks or more. All subjects provided written informed consent, and the protocol was approved by the Institutional Review Boards at UTSW and UM. Individuals with MDD The sample included 40 men and 40 women, between the ages of 20 and 41 years, diagnosed with MDD. All diagnoses were based on the Structured Clinical Interview for DSM-III-R and IV (SCID; non-patient version, Spitzer et al., 1997). Participants met criteria for non-psychotic MDD, but no other current Axis I disorders, substance abuse or substance
3 666 J. R. Goldschmied et al. dependence (American Psychological Association, 1994) within 12 months prior to baseline study. The 17-item Hamilton Rating Scale for Depression (HRS-D; Hamilton, 1960) was used to assess symptom severity. Those with MDD were mild-to-moderately depressed (HRS-D mean score ), had an adult age of onset (mean age of onset ), and had experienced two prior episodes of depression (mean number of episodes ). Polysomnographic data were incomplete as a result of recording difficulties for three participants, and thus the final sample included 40 men and 37 women (mean age ). Healthy controls The healthy control (HC) group consisted of eighty healthy adults, years of age (40 men and 40 women; mean age ). The HCs also underwent SCID to confirm the absence of personal or family history of psychopathology. All healthy controls subjects had Hamilton Rating Scale for Depression scores 2. Subset analysis In addition to our a priori hypotheses, a secondary analysis examining topographical changes resulting from sleep delay was included. Only participants with complete data for electrode sites F3, F4, C3, C4, P3, P4, O3 and O4 were included from our original sample (n = 92). Procedures For 5 days prior to study, participants kept an 11:00 06:00 hours sleep schedule, verified by sleep diary and actigraphy. Participants spent three consecutive nights in the sleep laboratory, utilizing the same sleep schedule for the first two nights. The first night served as an adaptation to the laboratory environment and screening for independent sleep disorders, while the second served as the baseline. Bedtime and rise time were delayed by 3 h on the third night, the SWA regulation challenge. Total available sleep time was held constant at 7 h on all nights. Subjects refrained from napping, using alcohol and drugs, and limited caffeine use to one cup before noon for the 24 h before the study, confirmed by diary and random urine screening. Standard laboratory procedures were followed (Armitage et al., 2012). On the first overnight in the laboratory, leg leads, chest and abdomen respiration bands and nasal oral thermistors were used, in addition to a full EEG montage. On each successive night, the montage included C3, C4, F3, F4, P3, P4, O1 and O2 EEG, left and right electro-oculography (EOG) and a bipolar electromyography (EMG). The reference electrode was comprised of linked earlobes passed through a 10 KΩ resistor to minimize possible artefacts. All impedances remained below 2 kω, and EEG was monitored throughout the sleep delay period to verify that subjects did not fall asleep. Sleep EEG One hundred and twenty-seven subjects were collected on a GRASS TM P511 amplifier-based paperless polygraph, described in detail elsewhere (Armitage et al., 2002). Data from the remaining 30 subjects were collected on a Vitaport TM III digital data acquisition system, described in detail in Armitage et al., Data systems were cross-validated, simultaneously recording and analysing data from 10 subjects. EEG data were quantified at the equivalent of a sensitivity of 5 (50 lv, 0.5 s calibration), a gain of , filters set at 0.3 and 30 Hz, respectively, and a filter to attenuate electrical noise. Research personnel scored sleep records visually following standard criteria (Rechtschaffen and Kales, 1968), after training to a 90% agreement on an epoch-by-epoch basis. Thereafter, any epochs that contained movement, breathing or muscle artefact or recording difficulties were omitted from further analysis. Power spectral analysis (PSA) was performed on the EEG data in 2-s blocks using an algorithm based on a fast- Fourier transform (512 samples for each 2 s). The sampling rate was set to 256 Hz, with a Hanning window taper to reduce overlap between adjacent frequencies. The PSA generates power in all five frequencies, but the analysis for the present paper was restricted to the delta activity expressed as lv 2. Delta power was averaged in 30-s epochs to provide identical epoch lengths to the stage-score data and sorted by NREM period for each subject on each night in the laboratory. The NREM period was defined as the succession of stages 2, 3 or 4 of 15-min duration and terminated by stage REM or a period of wakefulness of 5 min. Stage 1 sleep epochs were excluded. No minimum REM duration was required for the first or last REM periods. Delta power was summed and then averaged relative to the number of epochs in each NREM period, for each subject, referred to henceforth as slow wave activity (SWA). For statistical purposes, only the first three NREM periods were included for analysis, as not all subjects had four or more NREM periods across the night. In addition to the raw SWA power, the latency to each NREM period and duration of each NREM period on each night were computed and contrasted across groups. Further, in order to normalize the data, a %SWA measure was included, expressing SWA on the delay night to SWA on the baseline night, controlling for any potential individual differences in power values between and within groups. Data analysis All data were coded for sex (male, female), group (HC, MDD), EEG system (Grass, Vitaport) and age (below and including 30 years of age, above 30 years) and entered into SAS TM for statistical analysis. One four-way, split-plot factorial,
4 Sleep delay effects in males and females with MDD 667 repeated-measures analysis of variance (ANOVA) was computed on SWA amplitude measures, using age and EEG system as statistical covariates. The NREM period was treated as a three-level within-subjects variable. Univariate statistics for each NREM period are only reported if a main effect or interaction was obtained. Least-squares multiple comparisons tested differences between individual means at an experiment-wise P < 0.05, to protect against type 1 errors. In each group, exponential regression analysis was also conducted to evaluate the accumulation and dissipation of SWA over the night, (Armitage et al., 2000b). For all analyses, the group 9 sex interaction was tested first, followed by simpler effects as appropriate statistically. RESULTS Healthy control participants The sleep delay manipulation had a significant impact on some PSG measures within the healthy control group. Table 1 displays means and standard deviations of key PSG variables. Table 2 displays the means and standard deviations of latency, duration and SWA power, for each group and sex, during baseline and delay nights in each NREM period. There were no significant main effects or interactions with respect to the latencies to each of the NREM periods, or the durations of each NREM period for the HC or MDD. Therefore, no further analyses were performed on these measures. As expected, the key dependent variable,%swa, indicated that SWA was higher following sleep delay than following baseline sleep, and was significantly different among males and females, group 9 sex (F 3,151 = 4.65, P < 0.005). Fig. 1 illustrates the SWA on the delay night expressed as a percentage of baseline sleep in each group. MDD participants Sleep delay also had a significant impact on PSG measures within the MDD group. Similar to the HC, SWA was also significantly higher following sleep delay in MDD females, group 9 sex (F 3,151 = 8.90, P < 0.001). MDD males, in contrast, showed less SWA after sleep delay during the first three NREM periods, as indicated by a significant NREM period 9 sex 9 group interaction (F 9,453 = 3.91, P < 0.001). In addition to the raw SWA power measures, males with MDD also showed a blunted SWA response to sleep delay compared to all other groups with regard to %SWA, sex 9 group, (F 3,151 = 4.65, P < 0.005). Analyses on asymptotic SWA confirmed that MDD males have a lower baseline accumulation of SWA during the night than both MDD females and HC females, falling outside the 95% confidence interval. The sleep delay manipulation, however, did not appear to significantly affect asymptotic SWA or rate of decay for either the raw or %SWA measures, for all groups, as evidenced by all values falling within the confidence intervals of the baseline SWA. Table 3 illustrates the means and confidence intervals of the factors, the rates of decay and asymptotes of the exponential regression equations that were calculated to demonstrate the differences in SWA during the course of the night. Secondary analysis In order to examine the effects of sleep delay on the topography of SWA distribution, a subset of the original Table 1 Means and standard deviations of polysomnographic variables, by group and sex HCM HCF MDDM MDDF Baseline Delay Baseline Delay Baseline Delay Baseline Delay Total sleep (10.5) (41.6) (12.8) (49.0) (44.4) (38.8) (24.8) (24.4) period (min) Sleep latency (min)* 7.0 (5.8) 4.5 (3.5) 7.7 (6.4) 4.0 (3.9) 10.4 (13.1) 8.3 (8.1) 18.6 (19.0) 8.6 (12.4) Sleep efficiency (%) 95.3 (2.0) 94.0 (4.6) 95.2 (2.9) 95.7 (3.3) 91.4 (8.5) 92.0 (5.6) 91.5 (6.3) 93.4 (5.9) Awake and 2.9 (2.1) 3.6 (2.6) 2.7 (1.9) 2.7 (2.5) 5.6 (6.5) 5.8 (5.0) 4.2 (4.8) 4.5 (5.1) movement (%) % Stage 1* 6.3 (6.1) 6.8 (5.8) 6.1 (5.1) 5.6 (4.4) 11.9 (8.0) 12.9 (9.2) 6.7 (6.2) 9.1 (8.6) % Stage 2* 57.2 (7.9) 54.6 (5.9) 54.8 (7.8) 51.9 (8.2) 53.4 (8.3) 54.1 (8.3) 51.7 (7.4) 50.5 (6.6) % SWS 10.1 (8.7) 10.7 (8.6) 13.1 (8.7) 13.5 (9.0) 7.5 (8.0) 7.5 (8.6) 14.7 (8.6) 14.1 (8.6) %REM 23.5 (5.7) 24.3 (7.9) 23.2 (5.4) 25.8 (6.3) 21.7 (5.9) 19.5 (7.6) 22.8 (7.0) 21.7 (8.6) REM latency 78.5 (26.5) 69.1 (26.3) 72.2 (24.6) 64.4 (28.0) 78.5 (27.5) 98.5 (76.9) 77.6 (38.0) 91.6 (67.1) Minute SWS in 1st NREM 22.6 (19.9) 19.5 (18.4) 28.2 (18.9) 25.8 (19.1) 15.3 (17.2) 15.1 (20.8) 28.2 (18.8) 29.1 (18.2) HCM, healthy controls male; HCF, healthy controls female; MDDM, major depressive disorder male; MDDF, major depressive disorder female SWS, slow wave sleep; REM, rapid eye movement; NREM, non-rem. *Denotes significant sleep delay main effect. Denotes significant group main effect.
5 668 J. R. Goldschmied et al. Table 2 Means and standard deviations of latency to SWA, duration of SWA and raw SWA power by group and sex HCM HCF MDDM MDDF Baseline Delay Baseline Delay Baseline Delay Baseline Delay NREM 1 Latency (min) 12.6 (9.7) 36.9 (64.5) 10.1 (6.6) 33.1 (63.8) 12.0 (7.7) 42.6 (66.0) 11.9 (9.5) 47.2 (74.1) Duration (min) 70.5 (24.1) 61.2 (13.7) 67.2 (20.5) 58.6 (16.5) 64.4 (24.9) 76.7 (38.1) 63.9 (22.3) 68.0 (26.1) SWA (110.7) (141.4) 574 (129) (140.2) (119.2) 451.9(100.5) (111.8) (119) power (lv2) NREM 2 Latency (min) (27.7) (69.4) 97.4 (27.5) 112 (68.1) 94.7 (40.5) (77.9) 90.4 (25.4) (73.9) Duration (min) 69.8 (17.4) 64.3 (19.5) 66.8 (16.2) 66.8 (18.5) 66.7 (25.2) 64.6 (20.5) 72.2 (21.6) 76.6 (23.5) SWA (97.5) (118.3) (117.9) (116.2) (91.9) (80.7) (99.4) (117) power (lv2) NREM 3 Latency (min) (49.7) (74.8) (36.1) (70.7) (66.4) (84.1) (46.4) (83.8) Duration (min) 66.5 (22.4) 56 (30.7) 66.0 (19.8) 54.3 (12.7) 65.4 (19.5) 57.1 (22.7) 67.4 (24.7) 62.3 (19.4) SWA (73.2) (85.4) (69.4) (86.5) (60.7) (79.9) (85.3) (86.9) power (lv2) NREM 4 Latency (min) (49.8) (83.9) (42.6) (73.6) (72.7) (84.6) (44.6) (96.1) Duration (min) 51.4 (20) 51.6 (25) 49.2 (15.9) 49.4 (10.9) 49.0 (17.3) 45.0 (17.0) 57.7 (16.2) 52.0 (15.7) SWA power (lv2) (83.8) (103.1) (69.6) (80.4) (52.8) (69.9) (64.0) (77.9) SWA, slow wave activity; HCM, healthy controls male; HCF, healthy controls female; MDDM, major depressive disorder male; MDDF, major depressive disorder female. Latencies defined as minutes from sleep onset to the beginning of each non-rapid eye movement (NREM) sleep period. Italic type denotes significant group effect at baseline; bold type denotes significant group effect after delay NREM 1 NREM 2 NREM 3 NREM 4 HC - M HC - F MDD - M MDD - F Baseline Figure 1. Percentage of slow wave activity (SWA) response to sleep delay relative to baseline night by non-rapid eye movement (NREM) sleep period, by group and sex. participants with complete data for electrode sites F3, F4, C3, C4, P3, P4, O3 and O4 was included for analysis of electrode sites and hemispheric asymmetry (n = 92). Results revealed that SWA distribution differed depending on the NREM period, NREM period 9 hemisphere 9 electrode site (F 9,792 = 4.49, P < 0.005). Delta activity was higher in the right hemisphere in the first three NREM periods, but equally high in the left and right hemispheres in the fourth NREM period. A similar pattern emerges where SWA appears to be highest in the frontal electrodes and lowest in the occipital electrodes in the first NREM period, suggesting that SWA is concentrated fronto-centrally and spreads anteroposteriorly. Nevertheless, the delta activity difference between electrode sites decreased across the four NREM periods. SWA activity was greater in the right than the left hemisphere in the fourth NREM period in all participants except females with depression, NREM period 9 hemisphere 9 sex 9 group interaction (F 9,264 = 2.38, P < 0.05). Additionally, the difference in SWA between groups decreases across NREM period. With respect to group and sex, the difference in SWA between the left and right hemispheres was greater in males than in females, and greater in healthy participants than those with depression. Analyses were also conducted on the relative SWA measures. A significant NREM period 9 electrode site interaction was obtained (F 9,792 = 22.70, P < 0.001), with the highest SWA in the frontal electrode site during the first NREM period, but in the occipital site during the fourth NREM period. DISCUSSION Following a mild sleep challenge, males with MDD showed impaired regulation of SWA, displaying both a blunted response in the first NREM period and slower dissipation of SWA across the night in response to the sleep delay. The sex
6 Sleep delay effects in males and females with MDD 669 Table 3 Exponential regression parameters and 95% confidence intervals (CI) on (a) baseline and delay nights, (b) %SWA, by group and sex Baseline Delay SWA 95% CI Decay 95% CI SWA 95% CI Decay 95% CI (a) HCM to to to to 10.7 HCF to to to to 14.8 MDDM to to to to 2.5 MDDF to to to to 27.5 %SWA 95% CI Decay 95% CI (b) HCM to to 2.6 HCF to to 2.5 MDDM to to 0.1 MDDF to to 6.5 HCM, healthy controls male; HCF, healthy controls female; MDDM, major depressive disorder male; MDDF, major depressive disorder female; SWA, slow wave activity; CI, confidence interval. Italic type denotes significant group difference; bold type denotes significant delay effect within group. differences found were also much greater than that of the healthy individuals, but were apparent only after being amplified by the sleep challenge. These results may demonstrate that males with MDD are at increased risk for more generalized homeostatic dysregulation than females and may require specialized intervention. Research has suggested that SWA, particularly that from the first part of the night, is a reflection of neuronal recovery, as it is associated directly with the amount of time spent awake prior to sleep (Dijk et al., 1990; Tononi and Cirelli, 2003). In keeping with this viewpoint, therefore, the results suggest that males with MDD, but not females, may show impaired neuronal recovery during sleep. Tononi and Cirelli (2003) further suggest that the amount of slow wave activity may be correlated directly with the amount of synaptic potentiation, described as the LTP-like changes in synaptic strength that occurs during prior wakefulness, which would ultimately balance the input on cortical neurons through a series of depolarizations and hyperpolarizations during sleep, throughout the brain. Therefore, our findings may reflect impairment in the ability to down-regulate the cortical circuits during sleep, which are strengthened throughout wake, in males with MDD. The functioning of SWA in males with MDD may also be more generally reflective of a sleep regulatory failure. We found that the patterns in dissipation of SWA during the course of the night were different between groups, which may suggest that males with MDD are not able to maintain more basic homeostatic functioning and recover sufficiently from a sleep challenge, in addition to having a reduced drive for sleep. If individuals with MDD are not able to maintain sleep homeostasis, it may also follow that other homeostatic systems may also be unable to regulate. Of particular interest are the metabolic abnormalities that have recently been noted in MDD. Germain et al. (2007) indicated that, compared to healthy subjects, individuals with MDD showed increased cerebral metabolic activity in limbic regions, while showing decreased metabolic activity in frontal and parietal regions. Additionally, their research has also found that subjects with MDD demonstrated a higher level of metabolic activity in several brain regions when transitioning from wakefulness to NREM sleep than did healthy subjects (Germain et al., 2004). They have pointed to these findings to suggest that thalamocortical dysfunction may be central to MDD. The results found here, with respect to the disturbance of SWA, could indeed be demonstrative of a problem in the generation of thalamocortical activity, or may indicate that thalamocortical activity is generated, but does not propagate throughout the entirety of the brain. Topographical analyses of SWA across different brain regions may be able to tease apart these distinct problems. The S-deficiency hypothesis, posited by Borbely (1982), suggests that those with MDD exhibit an impaired homeostatic drive for sleep, and as such should demonstrate a reduced SWA response to challenge. Furthermore, Gillin and colleagues (1981) have also suggested that, in this way, the sleep patterns in MDD may parallel typical ageing, as SWA has been shown to decrease in healthy older individuals. Frey et al. (2012) investigated this theory by comparing the response to a sleep challenge in both healthy older and depressed young females. Their results revealed that young depressed females exhibited increased SWA in response to sleep deprivation compared to either young healthy or older healthy females, analogous to the results found in the present study. They thus determined that the functioning of SWA in females with MDD did not reflect premature ageing. Conversely, our results demonstrate that males with MDD show an attenuated SWA response to a sleep challenge that might
7 670 J. R. Goldschmied et al. be expected of older healthy males. In this way, the premature ageing hypothesis in MDD may hold true only for males. Additionally, the impaired regulation of SWA in males with MDD found currently may also reflect a larger, unrecognized deficit, such as sleep-disordered breathing. Deldin and colleagues (2006) have identified a significant relationship between sleep-disordered breathing and major depressive disorder. The authors (Deldin et al., 2006) found that certain sleep-related breathing variables, such as average overnight oxygen saturation, predict membership accurately in a healthy control group versus MDD. Moreover, in a more recent study, they found that those with MDD displayed higher rates of respiratory events than healthy controls, despite not reaching threshold for clinically significant sleep apnea (Cheng et al., 2013). Taken together, these studies illustrate the importance of further understanding the relationship between major depression and sleep-disordered breathing and the potential physiological consequences which may include impaired SWA. As the prevalence of SDB is two times greater in males than females, our finding of impaired regulation of SWA in only males with MDD may further suggest that sleep-disordered breathing may be playing a role. Previous research in healthy male subjects has found that SWA is highest in the frontal regions compared to temporal and parietal regions, and have their highest amplitude during early sleep, or NREM periods 1 and 2 compared to late sleep, or NREM periods 3 and 4 (Riedner et al., 2007). Additionally, Ferrara et al. (2002) also found that, during a recovery night of sleep following selective slow wave sleep deprivation, the left hemisphere showed greater SWA power than the right hemisphere, which they suggest indicates that the left hemisphere has a greater sleep need following sleep deprivation. With respect to female subjects, Birchler-Pedross et al. (2011) found that females with MDD exhibited higher low-frequency EEG activity than healthy controls during night sleep and daytime naps, which they suggested could mean that females exhibit a generally higher homeostatic sleep pressure. Our results indicated that SWA was generally highest in the right hemisphere, regardless of the sleep delay, but was sex-, group- and NREM period-dependent. Contrary to previous findings (Plante et al., 2012), our results did not indicate any group or sex effects for electrode sites, including any significant differences for females with and without MDD, in the frontal site during the first NREM period, at baseline or following the sleep delay challenge. Our results also demonstrated that while SWA power was significantly different among groups, the latencies to each NREM period, or durations of each NREM period, were not. This suggests that enhanced SWA power is neither a result of significantly shorter latency to SWA nor due to prolonged duration of SWA in the first NREM period. Additionally, it has also been suggested that SWA and sleep latency may both be indices of sleep drive. Consistent with this view, we found that males with MDD had both lower SWA power and longer sleep latency in the first NREM period. However, while females with MDD exhibited higher SWA power following the sleep delay, they did not display shorter sleep latency. These results suggest that latency to SWA is not a proxy for SWA power. The results of this study, however, are not unique to those individuals with MDD. Recent findings from our laboratory (Armitage et al., 2012) showed that males with alcohol dependence (AD) had similarities to males with MDD, with regard to impaired homeostatic regulation of SWA. Males with AD demonstrated impaired generation of SWA following a challenge, in addition to the impaired speed of recovery of SWA during the course of the night. Females with AD, however, did not show this pattern, similar to the females with MDD in the current study. These findings, in addition to those found in the current study, suggest that the pathophysiology of MDD and other diseases are sex-dependent, and may shed light on the process of neuronal recovery. The clinical course of MDD has been shown to be affected by gender, with more disturbed sleep found in males with an earlier age of onset of MDD and in females with more lifetime episodes (Swanson et al., 2010). Taken together, these data suggest that clinicians and researchers should be sensitive to the possibility that the influence of gender on sleep, specifically, and depression, more generally, may not be consistent across groups. Our present findings should be interpreted in light of several limitations. In order to mimic the effects of total sleep deprivation while minimizing the time before recovery sleep, the present study utilized a 3-h sleep delay. As expected, our healthy control groups showed a response to the sleep delay in the predicted direction, exhibiting an increase in the percentage of SWA in the first NREM period. Males with MDD, also as predicted, exhibited a smaller response to the challenge compared to the other groups. This finding may point to the fact that males with MDD exhibit a blunted response to the challenge, as we have interpreted, or it could indicate that the homeostat of these individuals may be broken. It is possible that the 3-h sleep delay procedure is not strong enough to evoke a response, and that the system of the males with MDD only respond to a much larger threat in order to preserve resources. In order to tease apart these differences, future studies interested in exploring the homeostatic response in SWA may want to utilize full-night sleep deprivation. Our results are also somewhat at odds with an earlier study from our laboratory (Armitage et al., 2007), which showed that females with MDD had a greater response than healthy females to the sleep delay challenge. This difference, however, may be a result of clinical differences in the current sample, as the females with MDD reported a fewer number of lifetime episodes and a later age of onset than in our previous study. Although females with MDD did not demonstrate the same patterns as males with MDD with regard to SWA, previous studies have indicated that the disease still seems to have a substantial impact on the sleep system of females, such as biological rhythm abnormalities and dysregulation in the organization of sleep EEG (Armitage, 2007).
8 Sleep delay effects in males and females with MDD 671 Additionally, although we were cognizant about our recruitment process, the generalizability of the sample is limited by our stringent inclusion criteria. We were interested in exploring the effects of sleep delay on a depressed sample, and as such we excluded any individuals who exhibited diagnostic criteria for any other Axis I disorder according to the Structured Clinical Interview for the DSM. Although this provided a relatively clean sample, research has shown that many individuals with MDD also have other comorbid Axis I disorders (Kessler et al., 1996), and therefore may limit how our results apply to the general population of those with MDD. In summary, this study utilized a 3-h sleep delay in order to evoke a SWA response in a sample of healthy controls and individuals with depression. Results revealed that males with MDD exhibit impaired regulation of SWA, with decreased accumulation of delta in the first NREM period, in addition to a slower rate of decay across NREM periods compared to healthy controls and females with MDD. AUTHOR CONTRIBUTIONS JRG completed the majority of data analyses and writing of manuscript; PC provided statistical and technical support for data analyses; RA is Director of UM Sleep and Chronophysiology Laboratory where the data were collected; PJD provided data analyses support and all edits of the manuscript. CONFLICTS OF INTEREST JG, PC and PD report no conflicts of interest. RA was previously a consultant for Eisai, Inc. REFERENCES American Psychological Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-IV. American Psychiatric Association, Washington, DC, Armitage, R. Microarchitectural findings in sleep EEG in depression: diagnostic implications. Biol. Psychiatry, 1995, 37: Armitage, R. Sleep and circadian rhythms in mood disorders. Acta Psychiatr. Scand., 2007, 115: Armitage, R. and Hoffmann, R. F. Sleep EEG, depression and gender. Sleep Med. Rev., 2001, 5: Armitage, R., Roffwarg, H. P., Rush, A. J., Calhoun, J. S., Purdy, D. G. and Giles, D. E. Digital period analysis of sleep EEG in depression. Biol. Psychiatry, 1992, 31: Armitage, R., Hudson, A., Trivedi, M. and Rush, A. J. Sex differences in the distribution of EEG frequencies during sleep: unipolar depressed outpatients. J. Affect. Disord., 1995, 34: Armitage, R., Trivedi, M., Hoffmann, R. and Rush, A. J. Relationship between objective and subjective sleep measures in depressed patients and healthy controls. Depress. Anxiety, 1997, 5: Armitage, R., Hoffmann, R., Fitch, T., Trivedi, M. and Rush, A. J. Temporal characteristics of delta activity during NREM sleep in depressed outpatients and healthy adults: group and sex effects. Sleep, 2000a, 23: Armitage, R., Hoffmann, R., Trivedi, M. and Rush, A. J. Slow-wave activity in NREM sleep: sex and age effects in depressed outpatients and healthy controls. Psychiatry Res., 2000b, 95: Armitage, R., Hoffmann, R. F., Emslie, G. J., Weinberg, W. A., Mayes, T. L. and Rush, A. J. Sleep microarchitecture as a predictor of recurrence in children and adolescents with depression. Int. J. Neuropsychopharmacol., 2002, 5: Armitage, R., Landis, C., Hoffmann, R. et al. The impact of a 4-hour sleep delay on slow wave activity in twins discordant for chronic fatigue syndrome. Sleep, 2007, 30: Armitage, R., Hoffmann, R., Conroy, D. A., Arnedt, J. T. and Brower, K. J. Effects of a 3-hour sleep delay on sleep homeostasis in alcohol dependent adults. Sleep, 2012, 35: Birchler-Pedross, A., Frey, S., Chellappa, S. L. et al. Higher frontal EEG synchronization in young women with major depression: a marker for increased homeostatic sleep pressure. Sleep, 2011, 34: Borbély, A. A., Baumann, F., Brandeis, D., Strauch, I. and Lehmann, D. Sleep deprivation: effect on sleep stages and EEG power density in man. Electroencephalogr Clin. Neurophysiol., 1981, 51: Borbely, A. A. and Wirz-Justice, A. Sleep, sleep deprivation and depression. A hypothesis derived from a model of sleep regulation. Hum. Neurobiol., 1982, 1: Cheng, P., Casement, M., Chen, C., Hoffmann, R. F., Armitage, R. and Deldin, P. J. Sleep-disordered breathing in major depressive disorder. J. Sleep Res., 2013, 22: Deldin, P. J., Phillips, L. K. and Thomas, R. J. A preliminary study of sleep-disordered breathing in major depressive disorder. Sleep Med., 2006, 7: Dijk, D., Brunner, D. P., Beersma, D. G. and Borbely, A. A. Electroencephalogram power density and slow wave sleep as a function of prior waking and circadian phase. Sleep, 1990, 13: Dijk, D. J., Cajochen, C., Tobler, I. and Borbely, A. A. Sleep extension in humans: sleep stages, EEG power spectra and body temperature. Sleep, 1991, 14: Ferrara, M., De Gennaro, L., Curcio, G., Cristiani, R., Corvasce, C. and Bertini, M. Regional differences of the human sleep electroencephalogram in response to selective slow-wave sleep deprivation. Cereb. Cortex, 2002, 12: Frey, S., Birchler-Pedross, A., Hofstetter, M. et al. Challenging the sleep homeostat: sleep in depression is not premature aging. Sleep Med., 2012, 13: Germain, A., Nofzinger, E. A., Kupfer, D. J. and Buysse, D. J. Neurobiology of non-rem sleep in depression: further evidence for hypofrontality and thalamic dysregulation. Am. J. Psychiatry, 2004, 161: Germain, A., Nofzinger, E. A., Meltzer, C. C. et al. Diurnal variation in regional brain glucose metabolism in depression. Biol. Psychiatry, 2007, 62: Gillin, J. C., Duncan, W. C., Murphy, D. L., et al. Age-related changes in sleep in depressed and normal subjects. Psychiatry Res., 1981, 4: Hamilton, M. A rating scale for depression. J. Neurol. Neurosurg. Psychiatr., 1960, 23: Kessler, R. C., Nelson, C. B., McGonagle, K. A. and Liu, J. Comorbidity of DSM-III-R major depressive disorder in the general population: results from the US National Comorbidity Survey. Br. J. Psychiatry, 1996, 30: Knott, V., Mahoney, C., Kennedy, S. and Evans, K. EEG power, frequency, asymmetry and coherence in male depression. Psychiatry Res. Neuroimaging, 2001, 106: Kupfer, D. J., Grochocinski, V. J. and McEachran, A. B. Relationship of awakening and delta sleep in depression. Psychiatry Res., 1986, 19: Mendlewicz, J. Sleep disturbances: core symptoms of major depressive disorder rather than associated or comorbid disorders. World J. Biol. Psychiatry, 2009, 10:
9 672 J. R. Goldschmied et al. Plante, D. T., Landsness, E. C., Peterson, M. J. et al. Sex-related differences in sleep slow wave activity in major depressive disorder: a high-density EEG investigation. BMC Psychiatry, 2012, 12: 146. Rechtschaffen, A. and Kales, A.. A Manual of Standardized Terminology, Techniques and Scoring System for Sleep Stages of Human Subjects. Brain Research Institute, Los Angeles, Reynolds, C. F. III, Kupfer, D. J., Thase, M. E. et al. Sleep, gender, and depression: an analysis of gender effects on the electroencephalographic sleep of 302 depressed outpatients. Biol. Psychiatry, 1990, 28: Riedner, B. A., Vyazovskiy, V. V., Huber, R. et al. Sleep homeostasis and cortical synchronization: III. A high-density EEG study of sleep slow waves in humans. Sleep, 2007, 30: Silverstein, B., Edwards, T., Gamma, A., Ajdacic-Gross, V., Rossler, W. and Angst, J. The role played by depression associated with somatic symptomatology in accounting for the gender difference in the prevalence of depression. Soc. Psychiatry Psychiatr. Epidemiol., 2013, 48: Spitzer, R. L., Gibbon, M. and Williams, J. B. User's Guide for the Structured Clinical Interview for DSM-IV Axis I Disorders SCID-I: Clinician Version. American Psychiatric Pub, New York, Swanson, L. M., Hoffmann, R. and Armitage, R. Sleep macroarchitecture in depression: sex differences. Open Sleep J., 2010, 3: Tononi, G. and Cirelli, C. Sleep and synaptic homeostasis: a hypothesis. Brain Res. Bull., 2003, 62:
Sleep Macroarchitecture in Depression: Sex Differences
 12 The Open Sleep Journal, 2010, 3, 12-18 Sleep Macroarchitecture in Depression: Sex Differences Open Access Leslie M. Swanson*, Robert Hoffmann and Roseanne Armitage Department of Psychiatry University
12 The Open Sleep Journal, 2010, 3, 12-18 Sleep Macroarchitecture in Depression: Sex Differences Open Access Leslie M. Swanson*, Robert Hoffmann and Roseanne Armitage Department of Psychiatry University
Effects of a 3-Hour Sleep Delay on Sleep Homeostasis in Alcohol Dependent Adults
 SLEEP HOMEOSTASIS IN ALCOHOL-DEPENDENT ADULTS http://dx.doi.org/10.5665/sleep.1638 Effects of a 3-Hour Sleep Delay on Sleep Homeostasis in Alcohol Dependent Adults Roseanne Armitage, PhD; Robert Hoffmann,
SLEEP HOMEOSTASIS IN ALCOHOL-DEPENDENT ADULTS http://dx.doi.org/10.5665/sleep.1638 Effects of a 3-Hour Sleep Delay on Sleep Homeostasis in Alcohol Dependent Adults Roseanne Armitage, PhD; Robert Hoffmann,
The role of fast and slow EEG activity during sleep in males and females with major depressive disorder
 Psychophysiology, 52 (2015), 1375 1381. Wiley Periodicals, Inc. Printed in the USA. Copyright VC 2015 Society for Psychophysiological Research DOI: 10.1111/psyp.12472 The role of fast and slow EEG activity
Psychophysiology, 52 (2015), 1375 1381. Wiley Periodicals, Inc. Printed in the USA. Copyright VC 2015 Society for Psychophysiological Research DOI: 10.1111/psyp.12472 The role of fast and slow EEG activity
Simplest method: Questionnaires. Retrospective: past week, month, year, lifetime Daily: Sleep diary What kinds of questions would you ask?
 Spencer Dawson Simplest method: Questionnaires Retrospective: past week, month, year, lifetime Daily: Sleep diary What kinds of questions would you ask? Did you nap during the day? Bed time and rise time
Spencer Dawson Simplest method: Questionnaires Retrospective: past week, month, year, lifetime Daily: Sleep diary What kinds of questions would you ask? Did you nap during the day? Bed time and rise time
Excessive Daytime Sleepiness Associated with Insufficient Sleep
 Sleep, 6(4):319-325 1983 Raven Press, New York Excessive Daytime Sleepiness Associated with Insufficient Sleep T. Roehrs, F. Zorick, J. Sicklesteel, R. Wittig, and T. Roth Sleep Disorders and Research
Sleep, 6(4):319-325 1983 Raven Press, New York Excessive Daytime Sleepiness Associated with Insufficient Sleep T. Roehrs, F. Zorick, J. Sicklesteel, R. Wittig, and T. Roth Sleep Disorders and Research
Patterns of Sleepiness in Various Disorders of Excessive Daytime Somnolence
 Sleep, 5:S165S174 1982 Raven Press, New York Patterns of Sleepiness in Various Disorders of Excessive Daytime Somnolence F. Zorick, T. Roehrs, G. Koshorek, J. Sicklesteel, *K. Hartse, R. Wittig, and T.
Sleep, 5:S165S174 1982 Raven Press, New York Patterns of Sleepiness in Various Disorders of Excessive Daytime Somnolence F. Zorick, T. Roehrs, G. Koshorek, J. Sicklesteel, *K. Hartse, R. Wittig, and T.
Sleep and circadian rhythms in mood disorders
 Acta Psychiatr Scand 2007: 115 (Suppl. 433): 104 115 All rights reserved Copyright Ó Blackwell Munksgaard 2007 ACTA PSYCHIATRICA SCANDINAVICA Sleep and circadian rhythms in mood disorders Armitage R. Sleep
Acta Psychiatr Scand 2007: 115 (Suppl. 433): 104 115 All rights reserved Copyright Ó Blackwell Munksgaard 2007 ACTA PSYCHIATRICA SCANDINAVICA Sleep and circadian rhythms in mood disorders Armitage R. Sleep
Slow oscillations in human non-rapid eye movement sleep electroencephalogram: effects of increased sleep pressure
 J. Sleep Res. () 9, 8 37 Slow oscillations in human EEG doi:./j.365-869.9.775.x Slow oscillations in human non-rapid eye movement sleep electroencephalogram: effects of increased sleep pressure ALESSIA
J. Sleep Res. () 9, 8 37 Slow oscillations in human EEG doi:./j.365-869.9.775.x Slow oscillations in human non-rapid eye movement sleep electroencephalogram: effects of increased sleep pressure ALESSIA
Selective Slow-Wave Sleep (SWS) Deprivation and SWS Rebound: Do We Need a Fixed SWS Amount per Night?
 Sleep Research Online 2(1): 15-19, 1999 http://www.sro.org/1999/ferrara/15/ Printed in the USA. All rights reserved. 1096-214X 1999 WebSciences Selective Slow-Wave Sleep (SWS) Deprivation and SWS Rebound:
Sleep Research Online 2(1): 15-19, 1999 http://www.sro.org/1999/ferrara/15/ Printed in the USA. All rights reserved. 1096-214X 1999 WebSciences Selective Slow-Wave Sleep (SWS) Deprivation and SWS Rebound:
The Effects of Fluoxetine on Quantitative Sleep EEG in Depressed Outpatients
 The Effects of Fluoxetine on Quantitative Sleep EEG in Depressed Outpatients Robert F. Hoffmann, Ph.D., Roseanne Armitage, Ph.D., Madhukar H. Trivedi, M.D., and A. John Rush, M.D. In previous studies,
The Effects of Fluoxetine on Quantitative Sleep EEG in Depressed Outpatients Robert F. Hoffmann, Ph.D., Roseanne Armitage, Ph.D., Madhukar H. Trivedi, M.D., and A. John Rush, M.D. In previous studies,
Human sleep spindle characteristics after sleep deprivation
 Clinical Neurophysiology 114 (2003) 2258 2267 www.elsevier.com/locate/clinph Human sleep spindle characteristics after sleep deprivation Vera Knoblauch a, Wim L.J. Martens b, Anna Wirz-Justice a, Christian
Clinical Neurophysiology 114 (2003) 2258 2267 www.elsevier.com/locate/clinph Human sleep spindle characteristics after sleep deprivation Vera Knoblauch a, Wim L.J. Martens b, Anna Wirz-Justice a, Christian
EEG Arousals: Scoring Rules and Examples. A Preliminary Report from the Sleep Disorders Atlas Task Force of the American Sleep Disorders Association
 EEG Arousals: Scoring Rules and Examples A Preliminary Report from the Sleep Disorders Atlas Task Force of the American Sleep Disorders Association Sleep in patients with a number of sleep disorders and
EEG Arousals: Scoring Rules and Examples A Preliminary Report from the Sleep Disorders Atlas Task Force of the American Sleep Disorders Association Sleep in patients with a number of sleep disorders and
Hypersomnolence in Psychiatric Disorders
 Hypersomnolence in Psychiatric Disorders David T. Plante, M.D. Assistant Professor of Psychiatry Medical Director, Wisconsin Sleep Sleep Medicine Fellowship Director Duke Department of Psychiatry/Central
Hypersomnolence in Psychiatric Disorders David T. Plante, M.D. Assistant Professor of Psychiatry Medical Director, Wisconsin Sleep Sleep Medicine Fellowship Director Duke Department of Psychiatry/Central
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR
 Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR In Physiology Today What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR In Physiology Today What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR
 Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may have as many as 200,000
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may have as many as 200,000
Basics of Polysomnography. Chitra Lal, MD, FCCP, FAASM Assistant professor of Medicine, Pulmonary, Critical Care and Sleep, MUSC, Charleston, SC
 Basics of Polysomnography Chitra Lal, MD, FCCP, FAASM Assistant professor of Medicine, Pulmonary, Critical Care and Sleep, MUSC, Charleston, SC Basics of Polysomnography Continuous and simultaneous recording
Basics of Polysomnography Chitra Lal, MD, FCCP, FAASM Assistant professor of Medicine, Pulmonary, Critical Care and Sleep, MUSC, Charleston, SC Basics of Polysomnography Continuous and simultaneous recording
Daytime Alertness in Patients with Chronic Insomnia Compared with Asymptomatic Control Subjects
 Sleep I ():54-60, Raven Press, Ltd., New York 988 Association of Professional Sleep Societies Daytime Alertness in Patients with Chronic Insomnia Compared with Asymptomatic Control Subjects Edward Stepanski,
Sleep I ():54-60, Raven Press, Ltd., New York 988 Association of Professional Sleep Societies Daytime Alertness in Patients with Chronic Insomnia Compared with Asymptomatic Control Subjects Edward Stepanski,
SLEEP, SLEEP DEPRIVATION, AND PERFORMANCE
 30722 Sleep 5.qxd 6/27/2003 2:16 PM Page 567 SLEEP, SLEEP DEPRIVATION, AND PERFORMANCE Sleep Regulation in the Djungarian Hamster: Comparison of the Dynamics Leading to the Slow-Wave Activity Increase
30722 Sleep 5.qxd 6/27/2003 2:16 PM Page 567 SLEEP, SLEEP DEPRIVATION, AND PERFORMANCE Sleep Regulation in the Djungarian Hamster: Comparison of the Dynamics Leading to the Slow-Wave Activity Increase
Higher Frontal EEG Synchronization in Young Women with Major Depression: A Marker for Increased Homeostatic Sleep Pressure?
 FRONTAL EEG SYNCHRONIZATION IN YOUNG WOMEN WITH DEPRESSION http://dx.doi.org/10.5665/sleep.1440 Higher Frontal EEG Synchronization in Young Women with Major Depression: A Marker for Increased Homeostatic
FRONTAL EEG SYNCHRONIZATION IN YOUNG WOMEN WITH DEPRESSION http://dx.doi.org/10.5665/sleep.1440 Higher Frontal EEG Synchronization in Young Women with Major Depression: A Marker for Increased Homeostatic
NATIONAL COMPETENCY SKILL STANDARDS FOR PERFORMING POLYSOMNOGRAPHY/SLEEP TECHNOLOGY
 NATIONAL COMPETENCY SKILL STANDARDS FOR PERFORMING POLYSOMNOGRAPHY/SLEEP TECHNOLOGY Polysomnography/Sleep Technology providers practice in accordance with the facility policy and procedure manual which
NATIONAL COMPETENCY SKILL STANDARDS FOR PERFORMING POLYSOMNOGRAPHY/SLEEP TECHNOLOGY Polysomnography/Sleep Technology providers practice in accordance with the facility policy and procedure manual which
What Is the Moment of Sleep Onset for Insomniacs?
 Sleep, 6(1): 10-\5 1983 Raven Press, New York What Is the Moment of Sleep Onset for Insomniacs? Peter Rauri and Elaine Olmstead Dartmouth Medical School, Hanover, New Hampshire, U,S,A, Summary: Subjective
Sleep, 6(1): 10-\5 1983 Raven Press, New York What Is the Moment of Sleep Onset for Insomniacs? Peter Rauri and Elaine Olmstead Dartmouth Medical School, Hanover, New Hampshire, U,S,A, Summary: Subjective
Ultrashort Sleep-Wake Cycle: Timing of REM Sleep. Evidence for Sleep-Dependent and Sleep-Independent Components of the REM Cycle
 Sleep 10(1):62-68, Raven Press, New York 1987, Association of Professional Sleep Societies Ultrashort Sleep-Wake Cycle: Timing of REM Sleep. Evidence for Sleep-Dependent and Sleep-Independent Components
Sleep 10(1):62-68, Raven Press, New York 1987, Association of Professional Sleep Societies Ultrashort Sleep-Wake Cycle: Timing of REM Sleep. Evidence for Sleep-Dependent and Sleep-Independent Components
Western Hospital System. PSG in History. SENSORS in the field of SLEEP. PSG in History continued. Remember
 SENSORS in the field of SLEEP Mrs. Gaye Cherry: Scientist in Charge Department of Sleep and Respiratory Medicine Sleep Disorders Unit Western Hospital PSG in History 1875: Discovery of brain-wave activity
SENSORS in the field of SLEEP Mrs. Gaye Cherry: Scientist in Charge Department of Sleep and Respiratory Medicine Sleep Disorders Unit Western Hospital PSG in History 1875: Discovery of brain-wave activity
The AASM Manual for the Scoring of Sleep and Associated Events
 The AASM Manual for the Scoring of Sleep and Associated Events Summary of Updates in Version 2.1 July 1, 2014 The American Academy of Sleep Medicine (AASM) is committed to ensuring that The AASM Manual
The AASM Manual for the Scoring of Sleep and Associated Events Summary of Updates in Version 2.1 July 1, 2014 The American Academy of Sleep Medicine (AASM) is committed to ensuring that The AASM Manual
Clinical Trial Synopsis TL , NCT#
 Clinical Trial Synopsis, NCT#00492011 Title of Study: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Study to Evaluate the Ability of Ramelteon 1 mg, 4 mg, and 8 mg to Alleviate the Insomnia
Clinical Trial Synopsis, NCT#00492011 Title of Study: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Study to Evaluate the Ability of Ramelteon 1 mg, 4 mg, and 8 mg to Alleviate the Insomnia
Sleep Medicine. Maintenance of Certification Examination Blueprint. Purpose of the exam
 Sleep Medicine Maintenance of Certification Examination Blueprint Purpose of the exam The exam is designed to evaluate the knowledge, diagnostic reasoning, and clinical judgment skills expected of the
Sleep Medicine Maintenance of Certification Examination Blueprint Purpose of the exam The exam is designed to evaluate the knowledge, diagnostic reasoning, and clinical judgment skills expected of the
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: October 15, 2018 Related Policies: 2.01.18 Diagnosis and Medical Management of Obstructive Sleep Apnea Syndrome Polysomnography for Non-Respiratory Sleep Disorders
FEP Medical Policy Manual Effective Date: October 15, 2018 Related Policies: 2.01.18 Diagnosis and Medical Management of Obstructive Sleep Apnea Syndrome Polysomnography for Non-Respiratory Sleep Disorders
Sleep stages. Awake Stage 1 Stage 2 Stage 3 Stage 4 Rapid eye movement sleep (REM) Slow wave sleep (NREM)
 Sleep stages Awake Stage 1 Stage 2 Stage 3 Stage 4 Rapid eye movement sleep (REM) Slow wave sleep (NREM) EEG waves EEG Electrode Placement Classifying EEG brain waves Frequency: the number of oscillations/waves
Sleep stages Awake Stage 1 Stage 2 Stage 3 Stage 4 Rapid eye movement sleep (REM) Slow wave sleep (NREM) EEG waves EEG Electrode Placement Classifying EEG brain waves Frequency: the number of oscillations/waves
Index. sleep.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Actigraphy, 475, 485, 496 Adolescents, sleep disorders in, 576 578 Adults, sleep disorders in, 578 580 Advanced sleep phase disorder, 482 Age,
Note: Page numbers of article titles are in boldface type. A Actigraphy, 475, 485, 496 Adolescents, sleep disorders in, 576 578 Adults, sleep disorders in, 578 580 Advanced sleep phase disorder, 482 Age,
H-Reflex Suppression and Autonomic Activation During Lucid REM Sleep: A Case Study
 Sleep 12(4):374-378, Raven Press, Ltd., New York 1989 Association of Professional Sleep Societies Short Communication H-Reflex Suppression and Autonomic Activation During Lucid REM Sleep: A Case Study
Sleep 12(4):374-378, Raven Press, Ltd., New York 1989 Association of Professional Sleep Societies Short Communication H-Reflex Suppression and Autonomic Activation During Lucid REM Sleep: A Case Study
sleep latencies Arousal and sleepiness in insomnia patients the tendency to fall asleep
 Arousal and sleepiness in insomnia patients Sleepiness the tendency to fall asleep Elke De Valck, Raymond Cluydts, Sandra Pirrera Department of Cognitive and Physiological Psychology, Vrije Universiteit
Arousal and sleepiness in insomnia patients Sleepiness the tendency to fall asleep Elke De Valck, Raymond Cluydts, Sandra Pirrera Department of Cognitive and Physiological Psychology, Vrije Universiteit
Disclosures. Acknowledgements. Sleep in Autism Spectrum Disorders: Window to Treatment and Etiology NONE. Ruth O Hara, Ph.D.
 Sleep in Autism Spectrum Disorders: Window to Treatment and Etiology Ruth O Hara, Ph.D. Associate Professor, Department of Psychiatry and Behavioral Sciences, Stanford University School of Medicine Disclosures
Sleep in Autism Spectrum Disorders: Window to Treatment and Etiology Ruth O Hara, Ph.D. Associate Professor, Department of Psychiatry and Behavioral Sciences, Stanford University School of Medicine Disclosures
Discrepancy between subjective and objective sleep in patients with depression
 PCN Psychiatric and Clinical Neurosciences 1323-13162003 Blackwell Science Pty Ltd 573June 2003 1114 Subjective and objective sleep K. Tsuchiyama et al. 10.1046/j.1323-1316.2003.01114.x Original Article259264BEES
PCN Psychiatric and Clinical Neurosciences 1323-13162003 Blackwell Science Pty Ltd 573June 2003 1114 Subjective and objective sleep K. Tsuchiyama et al. 10.1046/j.1323-1316.2003.01114.x Original Article259264BEES
Sleep Wake Cycle in Depression
 Sleep Wake Cycle in Depression Constantin R. Soldatos Professor of Psychiatry & Founder of Sleep Study Center Eginition Hospital University of Athens Lecture in Suzdal School, 20/04/2013 SLEEP WAKE CYCLE
Sleep Wake Cycle in Depression Constantin R. Soldatos Professor of Psychiatry & Founder of Sleep Study Center Eginition Hospital University of Athens Lecture in Suzdal School, 20/04/2013 SLEEP WAKE CYCLE
Sleep, Dreaming and Circadian Rhythms
 Sleep, Dreaming and Circadian Rhythms People typically sleep about 8 hours per day, and spend 16 hours awake. Most people sleep over 175,000 hours in their lifetime. The vast amount of time spent sleeping
Sleep, Dreaming and Circadian Rhythms People typically sleep about 8 hours per day, and spend 16 hours awake. Most people sleep over 175,000 hours in their lifetime. The vast amount of time spent sleeping
Supplemental Data. Inclusion/exclusion criteria for major depressive disorder group and healthy control group
 1 Supplemental Data Inclusion/exclusion criteria for major depressive disorder group and healthy control group Additional inclusion criteria for the major depressive disorder group were: age of onset of
1 Supplemental Data Inclusion/exclusion criteria for major depressive disorder group and healthy control group Additional inclusion criteria for the major depressive disorder group were: age of onset of
The REM Cycle is a Sleep-Dependent Rhythm
 Sleep, 2(3):299-307 1980 Raven Press, New York The REM Cycle is a Sleep-Dependent Rhythm L. C. Johnson Naval Health Research Center, San Diego, California Summary: Two studies, using data from fragmented
Sleep, 2(3):299-307 1980 Raven Press, New York The REM Cycle is a Sleep-Dependent Rhythm L. C. Johnson Naval Health Research Center, San Diego, California Summary: Two studies, using data from fragmented
Sleep Extension in Sleepy and Alert Normals
 Sleep 2(5):449-457, Raven Press, Ltd., New York 989 Association of Professional Sleep Societies Sleep Extension in Sleepy and Alert Normals Timothy Roehrs, Victoria Timms, Ardith Zwyghuizen-Doorenbos,
Sleep 2(5):449-457, Raven Press, Ltd., New York 989 Association of Professional Sleep Societies Sleep Extension in Sleepy and Alert Normals Timothy Roehrs, Victoria Timms, Ardith Zwyghuizen-Doorenbos,
Power Density in Theta/Alpha Frequencies of the Waking EEG Progressively Increases During Sustained Wakefulness
 Sleep, 18(10):890-894 1995 American Sleep Disorders Association and Sleep Research Society Power Density in Theta/Alpha Frequencies of the Waking EEG Progressively Increases During Sustained Wakefulness
Sleep, 18(10):890-894 1995 American Sleep Disorders Association and Sleep Research Society Power Density in Theta/Alpha Frequencies of the Waking EEG Progressively Increases During Sustained Wakefulness
Referenced EEG Offers a New Way to Prescribe the Right Medication
 SPEAKER NOTES Referenced EEG Offers a New Way to Prescribe the Right Medication Summarized by Thomas T. Thomas Prescribing medication to treat a brain disease is often a matter of trial and error, with
SPEAKER NOTES Referenced EEG Offers a New Way to Prescribe the Right Medication Summarized by Thomas T. Thomas Prescribing medication to treat a brain disease is often a matter of trial and error, with
EEG History. Where and why is EEG used? 8/2/2010
 EEG History Hans Berger 1873-1941 Edgar Douglas Adrian, an English physician, was one of the first scientists to record a single nerve fiber potential Although Adrian is credited with the discovery of
EEG History Hans Berger 1873-1941 Edgar Douglas Adrian, an English physician, was one of the first scientists to record a single nerve fiber potential Although Adrian is credited with the discovery of
LEARNING MANUAL OF PSG CHART
 LEARNING MANUAL OF PSG CHART POLYSOMNOGRAM, SLEEP STAGE SCORING, INTERPRETATION Sleep Computing Committee, Japanese Society of Sleep Research LEARNING MANUAL OF PSG CHART POLYSOMNOGRAM, SLEEP STAGE SCORING,
LEARNING MANUAL OF PSG CHART POLYSOMNOGRAM, SLEEP STAGE SCORING, INTERPRETATION Sleep Computing Committee, Japanese Society of Sleep Research LEARNING MANUAL OF PSG CHART POLYSOMNOGRAM, SLEEP STAGE SCORING,
Slow-wave sleep: do young adult men and women age
 J. Sleep Res. (1997) 6, 211 215 FAST TRACK PAPER Slow-wave sleep: do young adult men and women age differently? CINDY L. EHLERS 1,2 anddavid J. KUPFER 2 1 Department of Neuropharmacology, The Scripps Research
J. Sleep Res. (1997) 6, 211 215 FAST TRACK PAPER Slow-wave sleep: do young adult men and women age differently? CINDY L. EHLERS 1,2 anddavid J. KUPFER 2 1 Department of Neuropharmacology, The Scripps Research
A Modified Method for Scoring Slow Wave Sleep of Older Subjects
 Sleep, 5(2):195-199 1982 Raven Press, New York A Modified Method for Scoring Slow Wave Sleep of Older Subjects Wilse B. Webb and Lewis M. Dreblow Department of Psychology, University of Florida, Gainesville,
Sleep, 5(2):195-199 1982 Raven Press, New York A Modified Method for Scoring Slow Wave Sleep of Older Subjects Wilse B. Webb and Lewis M. Dreblow Department of Psychology, University of Florida, Gainesville,
TITLE: Homeostatic and Circadian Abnormalities in Sleep and Arousal in Gulf War Syndrome
 Award Number: W81XWH-10-2-0129 TITLE: Homeostatic and Circadian Abnormalities in Sleep and Arousal in Gulf War Syndrome PRINCIPAL INVESTIGATOR: Timothy M. Juergens, M.D. CONTRACTING ORGANIZATION: University
Award Number: W81XWH-10-2-0129 TITLE: Homeostatic and Circadian Abnormalities in Sleep and Arousal in Gulf War Syndrome PRINCIPAL INVESTIGATOR: Timothy M. Juergens, M.D. CONTRACTING ORGANIZATION: University
Patricia Carter, PhD, RN, CNS UT Austin School of
 Patricia Carter, PhD, RN, CNS UT Austin School of Nursing pcarter@mail.nur.utexas.edu @carter3236 Overview Normal sleep & Why we sleep Sleep architecture changes with aging Sleep disruptors Evidence based
Patricia Carter, PhD, RN, CNS UT Austin School of Nursing pcarter@mail.nur.utexas.edu @carter3236 Overview Normal sleep & Why we sleep Sleep architecture changes with aging Sleep disruptors Evidence based
Periodic Leg Movement, L-Dopa, 5-Hydroxytryptophan, and L-Tryptophan
 Sleep 10(4):393-397, Raven Press, New York 1987, Association of Professional Sleep Societies Short Report Periodic Leg Movement, L-Dopa, 5-Hydroxytryptophan, and L-Tryptophan C. Guilleminault, S. Mondini,
Sleep 10(4):393-397, Raven Press, New York 1987, Association of Professional Sleep Societies Short Report Periodic Leg Movement, L-Dopa, 5-Hydroxytryptophan, and L-Tryptophan C. Guilleminault, S. Mondini,
Sleep in the NCANDA cohort
 Sleep in the NCANDA cohort Fiona C. Baker, Ph.D. Center for Health Sciences, SRI International, California and Brain Function Research Group, University of the Witwatersrand, South Africa NCANDA FUNDING:
Sleep in the NCANDA cohort Fiona C. Baker, Ph.D. Center for Health Sciences, SRI International, California and Brain Function Research Group, University of the Witwatersrand, South Africa NCANDA FUNDING:
Circadian period and the timing of melatonin onset in men and women: predictors of sleep during the weekend and in the laboratory
 J Sleep Res. (213) 22, 155 159 Circadian rhythms Circadian period and the timing of melatonin onset in men and women: predictors of sleep during the weekend and in the laboratory ALPAR S. LAZAR 1 *, NAYANTARA
J Sleep Res. (213) 22, 155 159 Circadian rhythms Circadian period and the timing of melatonin onset in men and women: predictors of sleep during the weekend and in the laboratory ALPAR S. LAZAR 1 *, NAYANTARA
Interindividual differences in the dynamics of the homeostatic process are trait-like and distinct for sleep versus wakefulness
 J Sleep Res. (2017) 26, 171 178 Dynamics of sleep-wake homeostasis Interindividual differences in the dynamics of the homeostatic process are trait-like and distinct for sleep versus wakefulness THOMAS
J Sleep Res. (2017) 26, 171 178 Dynamics of sleep-wake homeostasis Interindividual differences in the dynamics of the homeostatic process are trait-like and distinct for sleep versus wakefulness THOMAS
Physiology of Normal Sleep: From Young to Old
 Physiology of Normal Sleep: From Young to Old V. Mohan Kumar Sree Chitra Tirunal Institute for Medical Sciences and Technology, Thiruvananthapuram 1 What is sleep? As per behavioral criteria: Reduced motor
Physiology of Normal Sleep: From Young to Old V. Mohan Kumar Sree Chitra Tirunal Institute for Medical Sciences and Technology, Thiruvananthapuram 1 What is sleep? As per behavioral criteria: Reduced motor
Contents Sleep stage scoring Sleep regulation Sleep stage scoring in infants Sleep spindles 11
 Contents. Summary. Introduction 5.. Sleep stage scoring 6.. Sleep regulation 6.. Sleep stage scoring in infants 8.. Sleep spindles... Definition of sleep spindles... Premature spindles... Sleep spindles
Contents. Summary. Introduction 5.. Sleep stage scoring 6.. Sleep regulation 6.. Sleep stage scoring in infants 8.. Sleep spindles... Definition of sleep spindles... Premature spindles... Sleep spindles
The value of sleep deprivation as a diagnostic tool in adult sleepwalkers
 CME The value of sleep deprivation as a diagnostic tool in adult sleepwalkers Steve Joncas, BSc; Antonio Zadra, PhD; Jean Paquet, PhD; and Jacques Montplaisir, MD, PhD, CRCPc Abstract Objective: Adult
CME The value of sleep deprivation as a diagnostic tool in adult sleepwalkers Steve Joncas, BSc; Antonio Zadra, PhD; Jean Paquet, PhD; and Jacques Montplaisir, MD, PhD, CRCPc Abstract Objective: Adult
Circadian rhythm and Sleep. Radwan Banimustafa MD
 Circadian rhythm and Sleep Radwan Banimustafa MD Homeostasis Maintenance of equilibrium by active regulation of internal states: Cardiovascular function (blood pressure, heart rate) Body temperature Food
Circadian rhythm and Sleep Radwan Banimustafa MD Homeostasis Maintenance of equilibrium by active regulation of internal states: Cardiovascular function (blood pressure, heart rate) Body temperature Food
Circadian variation of EEG power spectra in NREM and REM sleep in humans: Dissociation from body temperature
 J. Sleep Res. (1999) 8, 189 195 Circadian variation of EEG power spectra in NREM and REM sleep in humans: Dissociation from body temperature DERK-JAN DIJK Circadian, Neuroendocrine and Sleep Disorders
J. Sleep Res. (1999) 8, 189 195 Circadian variation of EEG power spectra in NREM and REM sleep in humans: Dissociation from body temperature DERK-JAN DIJK Circadian, Neuroendocrine and Sleep Disorders
REGRESSION IN PMS AND SLEEP DISTURBANCES IN PMS
 REGRESSION IN PMS AND SLEEP DISTURBANCES IN PMS RUTH O HARA, JOACHIM HALLMAYER, JON BERNSTEIN DELLA BRO, GILLIAN REIERSON, WENDY FROEHLICH-SANTINO, ALEXANDER URBAN, CAROLIN PURMANN, SEAN BERQUIST, JOSH
REGRESSION IN PMS AND SLEEP DISTURBANCES IN PMS RUTH O HARA, JOACHIM HALLMAYER, JON BERNSTEIN DELLA BRO, GILLIAN REIERSON, WENDY FROEHLICH-SANTINO, ALEXANDER URBAN, CAROLIN PURMANN, SEAN BERQUIST, JOSH
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: January 15, 2018 Related Policies: 2.01.18 Diagnosis and Medical Management of Obstructive Sleep Apnea Syndrome Diagnosis and Medical Management of Obstructive
FEP Medical Policy Manual Effective Date: January 15, 2018 Related Policies: 2.01.18 Diagnosis and Medical Management of Obstructive Sleep Apnea Syndrome Diagnosis and Medical Management of Obstructive
EEG Electrode Placement
 EEG Electrode Placement Classifying EEG brain waves Frequency: the number of oscillations/waves per second, measured in Hertz (Hz) reflects the firing rate of neurons alpha, beta, theta, delta Amplitude:
EEG Electrode Placement Classifying EEG brain waves Frequency: the number of oscillations/waves per second, measured in Hertz (Hz) reflects the firing rate of neurons alpha, beta, theta, delta Amplitude:
Periodic Leg Movements in Narcolepsy
 In: Nacrolepsy: Symptoms, Causes... ISBN: 978-1-60876-645-1 Editor: Guillermo Santos, et al. 2009 Nova Science Publishers, Inc. Chapter 7 Periodic Leg Movements in Narcolepsy Ahmed Bahammam * Sleep Disorders
In: Nacrolepsy: Symptoms, Causes... ISBN: 978-1-60876-645-1 Editor: Guillermo Santos, et al. 2009 Nova Science Publishers, Inc. Chapter 7 Periodic Leg Movements in Narcolepsy Ahmed Bahammam * Sleep Disorders
Actigraphic Motor Asymmetries During Sleep
 INSTRUMENTATION AND METHODOLOGY Actigraphic Motor Asymmetries During Sleep Cristiano Violani, Paola Testa, and Maria Casagrande Dipartimento di Psicologia - Università di Roma La Sapienza Summary: Much
INSTRUMENTATION AND METHODOLOGY Actigraphic Motor Asymmetries During Sleep Cristiano Violani, Paola Testa, and Maria Casagrande Dipartimento di Psicologia - Università di Roma La Sapienza Summary: Much
EFFICACY OF MODAFINIL IN 10 TAIWANESE PATIENTS WITH NARCOLEPSY: FINDINGS USING THE MULTIPLE SLEEP LATENCY TEST AND EPWORTH SLEEPINESS SCALE
 EFFICACY OF MODAFINIL IN 10 TAIWANESE PATIENTS WITH NARCOLEPSY: FINDINGS USING THE MULTIPLE SLEEP LATENCY TEST AND EPWORTH SLEEPINESS SCALE Shih-Bin Yeh 1 and Carlos Hugh Schenck 2,3 1 Department of Neurology
EFFICACY OF MODAFINIL IN 10 TAIWANESE PATIENTS WITH NARCOLEPSY: FINDINGS USING THE MULTIPLE SLEEP LATENCY TEST AND EPWORTH SLEEPINESS SCALE Shih-Bin Yeh 1 and Carlos Hugh Schenck 2,3 1 Department of Neurology
Chapter 6. Consciousness
 Consciousness Psychology, Fifth Edition, James S. Nairne What s It For? The Value of Consciousness Setting Priorities for Mental Functioning Sleeping and Dreaming Altering Awareness: Psychoactive Drugs
Consciousness Psychology, Fifth Edition, James S. Nairne What s It For? The Value of Consciousness Setting Priorities for Mental Functioning Sleeping and Dreaming Altering Awareness: Psychoactive Drugs
Insomnia. Learning Objectives. Disclosure 6/7/11. Research funding: NIH, Respironics, Embla Consulting: Elsevier
 Insomnia Teofilo Lee-Chiong MD Professor of Medicine National Jewish Health University of Colorado Denver School of Medicine Learning Objectives Learn about the causes of transient and chronic Learn how
Insomnia Teofilo Lee-Chiong MD Professor of Medicine National Jewish Health University of Colorado Denver School of Medicine Learning Objectives Learn about the causes of transient and chronic Learn how
The frontal predominance in human EEG delta activity after sleep loss decreases with age
 European Journal of Neuroscience, Vol. 20, pp. 1402 1410, 2004 ª Federation of European Neuroscience Societies The frontal predominance in human EEG delta activity after sleep loss decreases with age Mirjam
European Journal of Neuroscience, Vol. 20, pp. 1402 1410, 2004 ª Federation of European Neuroscience Societies The frontal predominance in human EEG delta activity after sleep loss decreases with age Mirjam
Participant ID: If you had no responsibilities, what time would your body tell you to go to sleep and wake up?
 What does your sleep look like on a typical week? Total Sleep Time: Bedtime:, Sleep onset latency:, Number of Awakenings:, Wake time after sleep onset:, Rise time:, Out of bed:, Naps:? Notes: Is your sleep
What does your sleep look like on a typical week? Total Sleep Time: Bedtime:, Sleep onset latency:, Number of Awakenings:, Wake time after sleep onset:, Rise time:, Out of bed:, Naps:? Notes: Is your sleep
Diagnosis and treatment of sleep disorders
 Diagnosis and treatment of sleep disorders Normal human sleep Sleep cycle occurs about every 90 minutes, approximately 4-6 cycles occur per major sleep episode NREM (70-80%) slow wave sleep heart rate,
Diagnosis and treatment of sleep disorders Normal human sleep Sleep cycle occurs about every 90 minutes, approximately 4-6 cycles occur per major sleep episode NREM (70-80%) slow wave sleep heart rate,
Students will be able to determine what stage of sleep someone is in by analyzing their EEG.
 Outline 2Lesson Unit1.2 OVERVIEW Rationale: This lesson is intended to engage students with the concept of the neural circuit. The lesson and unit as a whole use sleep, a behavior everyone is familiar
Outline 2Lesson Unit1.2 OVERVIEW Rationale: This lesson is intended to engage students with the concept of the neural circuit. The lesson and unit as a whole use sleep, a behavior everyone is familiar
Assessment of Sleep Disorders DR HUGH SELSICK
 Assessment of Sleep Disorders DR HUGH SELSICK Goals Understand the importance of history taking Be able to take a basic sleep history Be aware the technology used to assess sleep disorders. Understand
Assessment of Sleep Disorders DR HUGH SELSICK Goals Understand the importance of history taking Be able to take a basic sleep history Be aware the technology used to assess sleep disorders. Understand
Milena Pavlova, M.D., FAASM Department of Neurology, Brigham and Women's Hospital Assistant Professor of Neurology, Harvard Medical School Medical
 Milena Pavlova, M.D., FAASM Department of Neurology, Brigham and Women's Hospital Assistant Professor of Neurology, Harvard Medical School Medical Director, Faulkner EEG and Sleep Testing Center Course
Milena Pavlova, M.D., FAASM Department of Neurology, Brigham and Women's Hospital Assistant Professor of Neurology, Harvard Medical School Medical Director, Faulkner EEG and Sleep Testing Center Course
Office Practice Coding Assistance - Overview
 Office Practice Coding Assistance - Overview Three office coding assistance resources are provided in the STABLE Resource Toolkit. Depression & Bipolar Coding Reference: n Provides ICD9CM and DSM-IV-TR
Office Practice Coding Assistance - Overview Three office coding assistance resources are provided in the STABLE Resource Toolkit. Depression & Bipolar Coding Reference: n Provides ICD9CM and DSM-IV-TR
MWT PROTOCOL PURPOSE POLICY
 PURPOSE A standard MWT protocol that is consistent with AASM practice parameters promotes consistency, allows comparisons between tests, and ensures accurate interpretations that are consistent with published
PURPOSE A standard MWT protocol that is consistent with AASM practice parameters promotes consistency, allows comparisons between tests, and ensures accurate interpretations that are consistent with published
The Distribution of EEG Frequencies in REM and NREM Sleep Stages in Healthy Young Adults
 Sleep, 18(5):334-341 1995 American Sleep Disorders Association and Sleep Research Society The Distribution of G Frequencies in RM and NRM Sleep Stages in Healthy Young Adults Roseanne Armitage The University
Sleep, 18(5):334-341 1995 American Sleep Disorders Association and Sleep Research Society The Distribution of G Frequencies in RM and NRM Sleep Stages in Healthy Young Adults Roseanne Armitage The University
NIH Public Access Author Manuscript Psychiatry Clin Neurosci. Author manuscript; available in PMC 2010 November 1.
 NIH Public Access Author Manuscript Published in final edited form as: Psychiatry Clin Neurosci. 2003 October ; 57(5): 542 544. An Open Pilot Study of Gabapentin vs. Trazodone to Treat Insomnia in Alcoholic
NIH Public Access Author Manuscript Published in final edited form as: Psychiatry Clin Neurosci. 2003 October ; 57(5): 542 544. An Open Pilot Study of Gabapentin vs. Trazodone to Treat Insomnia in Alcoholic
Theta waves in children s waking electroencephalogram resemble local aspects of sleep during wakefulness
 Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2017 Theta waves in children s waking electroencephalogram resemble local aspects
Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2017 Theta waves in children s waking electroencephalogram resemble local aspects
Index SLEEP MEDICINE CLINICS. Note: Page numbers of article titles are in boldface type. Cerebrospinal fluid analysis, for Kleine-Levin syndrome,
 165 SLEEP MEDICINE CLINICS Index Sleep Med Clin 1 (2006) 165 170 Note: Page numbers of article titles are in boldface type. A Academic performance, effects of sleepiness in children on, 112 Accidents,
165 SLEEP MEDICINE CLINICS Index Sleep Med Clin 1 (2006) 165 170 Note: Page numbers of article titles are in boldface type. A Academic performance, effects of sleepiness in children on, 112 Accidents,
INSOMNIAS. Stephan Eisenschenk, MD Department of Neurology
 INSOMNIAS INSOMNIAS General criteria for insomnia A. Repeated difficulty with sleep initiation, duration, consolidation or quality. B. Adequate sleep opportunity, persistent sleep difficulty and associated
INSOMNIAS INSOMNIAS General criteria for insomnia A. Repeated difficulty with sleep initiation, duration, consolidation or quality. B. Adequate sleep opportunity, persistent sleep difficulty and associated
Medications that are not FDA approved for children will be discussed. NAPNAP National Conference 2018
 Medications that are not FDA approved for children will be discussed NAPNAP National Conference 2018 (Honaker & Meltzer, 2016; Keyes, Maslowsky, Hamilton & Schulenberg, 2015) Chronically disrupted sleep
Medications that are not FDA approved for children will be discussed NAPNAP National Conference 2018 (Honaker & Meltzer, 2016; Keyes, Maslowsky, Hamilton & Schulenberg, 2015) Chronically disrupted sleep
Sleep Disorders. Sleep. Circadian Rhythms
 Sleep Disorders Sleep The Sleep Wakefulness Cycle: Circadian Rhythms Internally generated patterns of bodily functions that vary over a ~24-hour period Function even in the absence of normal cues 2 Circadian
Sleep Disorders Sleep The Sleep Wakefulness Cycle: Circadian Rhythms Internally generated patterns of bodily functions that vary over a ~24-hour period Function even in the absence of normal cues 2 Circadian
Correlation Dimension versus Fractal Exponent During Sleep Onset
 Correlation Dimension versus Fractal Exponent During Sleep Onset K. Šušmáková Institute of Measurement Science, Slovak Academy of Sciences Dúbravská cesta 9, 84 19 Bratislava, Slovak Republic E-mail: umersusm@savba.sk
Correlation Dimension versus Fractal Exponent During Sleep Onset K. Šušmáková Institute of Measurement Science, Slovak Academy of Sciences Dúbravská cesta 9, 84 19 Bratislava, Slovak Republic E-mail: umersusm@savba.sk
Modelling the relation of body temperature and sleep: importance of the circadian rhythm in skin temperature
 Modelling the relation of body temperature and sleep: importance of the circadian rhythm in skin temperature EUS J.W. VAN SOMEREN NETHERLANDS INSTITUTE FOR BRAIN RESEARCH, AMSTERDAM A close relation between
Modelling the relation of body temperature and sleep: importance of the circadian rhythm in skin temperature EUS J.W. VAN SOMEREN NETHERLANDS INSTITUTE FOR BRAIN RESEARCH, AMSTERDAM A close relation between
Sleep Across the Life Cycle
 SECTION II Anatomy and Physiology CHAPTER 3 Sleep Across the Life Cycle IOURI KREININ L E A R N I N G O B J E C T I V E S On completion of this chapter, the reader should be able to 1. Describe the elements
SECTION II Anatomy and Physiology CHAPTER 3 Sleep Across the Life Cycle IOURI KREININ L E A R N I N G O B J E C T I V E S On completion of this chapter, the reader should be able to 1. Describe the elements
Sleep and Sleep Hygiene in an Occupational Health & Safety Context
 Sleep and Sleep Hygiene in an Occupational Health & Safety Context Glenn Legault Ph.D. Center for Research in Occupational Safety and Health, Laurentian University Nov. 12, 2014 Overview: Sleep what is
Sleep and Sleep Hygiene in an Occupational Health & Safety Context Glenn Legault Ph.D. Center for Research in Occupational Safety and Health, Laurentian University Nov. 12, 2014 Overview: Sleep what is
Noise, nuance and nuisance: an introduction to sound and sleep. Kevin Morgan Clinical Sleep Research Unit Loughborough University
 Noise, nuance and nuisance: an introduction to sound and sleep Kevin Morgan Clinical Sleep Research Unit Loughborough University Why do we Sleep? Some proximate answers Without sufficient sleep we experience:
Noise, nuance and nuisance: an introduction to sound and sleep Kevin Morgan Clinical Sleep Research Unit Loughborough University Why do we Sleep? Some proximate answers Without sufficient sleep we experience:
NORAH Sleep Study External Comment Mathias Basner, MD, PhD, MSc
 NORAH Sleep Study External Comment Mathias Basner, MD, PhD, MSc University of Pennsylvania Perelman School of Medicine Page 1 > Mathias Basner Disclaimer The University of Pennsylvania and the German Aerospace
NORAH Sleep Study External Comment Mathias Basner, MD, PhD, MSc University of Pennsylvania Perelman School of Medicine Page 1 > Mathias Basner Disclaimer The University of Pennsylvania and the German Aerospace
Polysomnography Course Session: Sept 2017
 Polysomnography Course Session: Sept 2017 General Information Polysomnography course will be held at SLEEP AND ALERTNESS CLINIC Med-West Medical centre 750 Dundas St. W., Suite 2-259 (Conference Room)
Polysomnography Course Session: Sept 2017 General Information Polysomnography course will be held at SLEEP AND ALERTNESS CLINIC Med-West Medical centre 750 Dundas St. W., Suite 2-259 (Conference Room)
Chronotype and Sex Effects on Sleep Architecture and Quantitative Sleep EEG in Healthy Young Adults
 CIRCADIAN RHYTHMS Chronotype and Sex Effects on Sleep Architecture and Quantitative Sleep EEG in Healthy Young Adults Valérie Mongrain, MSc 1,2 ; Julie Carrier, PhD 1-3 ; Marie Dumont, PhD 1,2,4 1 Chronobiology
CIRCADIAN RHYTHMS Chronotype and Sex Effects on Sleep Architecture and Quantitative Sleep EEG in Healthy Young Adults Valérie Mongrain, MSc 1,2 ; Julie Carrier, PhD 1-3 ; Marie Dumont, PhD 1,2,4 1 Chronobiology
Recognition of Sleep Dependent Memory Consolidation with Multi-modal Sensor Data
 Recognition of Sleep Dependent Memory Consolidation with Multi-modal Sensor Data The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation
Recognition of Sleep Dependent Memory Consolidation with Multi-modal Sensor Data The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation
Circadian Rhythms in Children and Adolescents
 Circadian Rhythms in Children and Adolescents Sarah Morsbach Honaker, Ph.D., CBSM Assistant Professor of Pediatrics IU School of Medicine Society for Behavioral Sleep Medicine Practice and Consultation
Circadian Rhythms in Children and Adolescents Sarah Morsbach Honaker, Ph.D., CBSM Assistant Professor of Pediatrics IU School of Medicine Society for Behavioral Sleep Medicine Practice and Consultation
SOMNAMBULISM: CLINICAL ASPECTS AND PATHOPHYSIOLOGICAL HYPOTHESES. Zadra, A., Desautels, A., Petit, D., Montplaisir, J. (2013) The Lancet Neurology
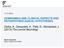 11.11.2015 SOMNAMBULISM: CLINICAL ASPECTS AND PATHOPHYSIOLOGICAL HYPOTHESES Zadra, A., Desautels, A., Petit, D., Montplaisir, J. (2013) The Lancet Neurology Marion Charmillot Sleep, Cognition and Health
11.11.2015 SOMNAMBULISM: CLINICAL ASPECTS AND PATHOPHYSIOLOGICAL HYPOTHESES Zadra, A., Desautels, A., Petit, D., Montplaisir, J. (2013) The Lancet Neurology Marion Charmillot Sleep, Cognition and Health
States of Consciousness
 States of Consciousness Sleep, Dreams, and Body Rhythms Introduction Consciousness Awareness of oneself and one s environment Body Rhythms Biological Rhythms Periodic physiological fluctuations Can affect
States of Consciousness Sleep, Dreams, and Body Rhythms Introduction Consciousness Awareness of oneself and one s environment Body Rhythms Biological Rhythms Periodic physiological fluctuations Can affect
The AASM Manual for the Scoring of Sleep and Associated Events
 The AASM Manual for the Scoring of Sleep and Associated Events The 2007 AASM Scoring Manual vs. the AASM Scoring Manual v2.0 October 2012 The American Academy of Sleep Medicine (AASM) is committed to ensuring
The AASM Manual for the Scoring of Sleep and Associated Events The 2007 AASM Scoring Manual vs. the AASM Scoring Manual v2.0 October 2012 The American Academy of Sleep Medicine (AASM) is committed to ensuring
Individual Planning: A Treatment Plan Overview for Individuals Sleep Disorder Problems.
 COURSES ARTICLE - THERAPYTOOLS.US Individual Planning: A Treatment Plan Overview for Individuals Sleep Disorder Problems. Individual Planning: A Treatment Plan Overview for Individuals Sleep Disorder Problems.
COURSES ARTICLE - THERAPYTOOLS.US Individual Planning: A Treatment Plan Overview for Individuals Sleep Disorder Problems. Individual Planning: A Treatment Plan Overview for Individuals Sleep Disorder Problems.
Exclusion criteria and outlier detection
 1 Exclusion criteria and outlier detection 1 2 Supplementary Fig. 1 31 subjects complied with the inclusion criteria as tested during the familiarization session. The upper part of the figure (ovals) indicates
1 Exclusion criteria and outlier detection 1 2 Supplementary Fig. 1 31 subjects complied with the inclusion criteria as tested during the familiarization session. The upper part of the figure (ovals) indicates
Introduction to Electrophysiology
 Introduction to Electrophysiology Dr. Kwangyeol Baek Martinos Center for Biomedical Imaging Massachusetts General Hospital Harvard Medical School 2018-05-31s Contents Principles in Electrophysiology Techniques
Introduction to Electrophysiology Dr. Kwangyeol Baek Martinos Center for Biomedical Imaging Massachusetts General Hospital Harvard Medical School 2018-05-31s Contents Principles in Electrophysiology Techniques
states of brain activity sleep, brain waves DR. S. GOLABI PH.D. IN MEDICAL PHYSIOLOGY
 states of brain activity sleep, brain waves DR. S. GOLABI PH.D. IN MEDICAL PHYSIOLOGY introduction all of us are aware of the many different states of brain activity, including sleep, wakefulness, extreme
states of brain activity sleep, brain waves DR. S. GOLABI PH.D. IN MEDICAL PHYSIOLOGY introduction all of us are aware of the many different states of brain activity, including sleep, wakefulness, extreme
INTRODUCTION BACKGROUND
 Use of Actigraphy for the Evaluation of Sleep Disorders and Circadian Rhythm Sleep-Wake Disorders An American Academy of Sleep Medicine Systematic Review Introduction: The purpose of this systematic review
Use of Actigraphy for the Evaluation of Sleep Disorders and Circadian Rhythm Sleep-Wake Disorders An American Academy of Sleep Medicine Systematic Review Introduction: The purpose of this systematic review
EEG and some applications (seizures and sleep)
 EEG and some applications (seizures and sleep) EEG: stands for electroencephalography and is a graphed representation of the electrical activity of the brain. EEG is the recording of electrical activity
EEG and some applications (seizures and sleep) EEG: stands for electroencephalography and is a graphed representation of the electrical activity of the brain. EEG is the recording of electrical activity
Index. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Abuse sleep physiology effects of, 880 882 substance, in adolescents, sleep problems and, 929 946. See also Substance use and abuse, in adolescents,
Note: Page numbers of article titles are in boldface type. A Abuse sleep physiology effects of, 880 882 substance, in adolescents, sleep problems and, 929 946. See also Substance use and abuse, in adolescents,
Sleep Complaints and Disorders in Epileptic Patients 순천향의대천안병원순천향의대천안병원신경과양광익
 Sleep Complaints and Disorders in Epileptic Patients 순천향의대천안병원순천향의대천안병원신경과양광익 Introduction The global physical, social and economic consequence of epilepsy are high. WHO 2000 study Improving QoL is increasingly
Sleep Complaints and Disorders in Epileptic Patients 순천향의대천안병원순천향의대천안병원신경과양광익 Introduction The global physical, social and economic consequence of epilepsy are high. WHO 2000 study Improving QoL is increasingly
