Pain from copper intrauterine device insertion: Randomized trial of prophylactic ibuprofen
|
|
|
- Anabel Haynes
- 6 years ago
- Views:
Transcription
1 American Journal of Obstetrics and Gynecology (2006) 195, Pain from copper intrauterine device insertion: Randomized trial of prophylactic ibuprofen David Hubacher, PhD, a Veronica Reyes, RN, b Sonia Lillo, RN, b Ana Zepeda, PhD, b Pai-Lien Chen, PhD, a Horacio Croxatto, MD b Family Health International, a Research Triangle Park, NC; Instituto Chileno de Medicina Reproductiva (ICMER), b Santiago, Chile Received for publication April 25, 2006; revised July 7, 2006; accepted August 31, 2006 KEY WORDS Intrauterine device Insertion Pain Ibuprofen Prevention Objectives: This study was undertaken to determine whether 400 mg of prophylactic ibuprofen can alleviate pain from insertion of an intrauterine device (IUD) and to measure level of pain with improved techniques. Study design: We conducted a randomized, double-blind, placebo-controlled trial of 2019 firsttime IUD users: 1008 women received placebo and 1011 women received 400 mg of ibuprofen. Participants took the single tablet at least 45 minutes before IUD insertion. Immediately after insertion, participants recorded level of pain by using a 10-cm visual analog scale, with the value of 10 meaning worst imaginable pain. Results: Median level of pain was 1.0 for both ibuprofen and placebo participants; rank test statistics confirmed no difference. Some subgroups of women experienced higher pain (eg, nulliparous women), but ibuprofen still had no important impact on level of pain. Conclusion: Even among first-time users, pain from IUD insertion is generally low. Prophylactic ibuprofen as used in this protocol does not reduce IUD insertion pain. Ó 2006 Mosby, Inc. All rights reserved. The intrauterine device (IUD) is used by approximately 145 million women worldwide 1 and is the most common form of reversible contraception. The insertion procedure, experienced by about 40 million women each year, can cause pain and discomfort in several ways: use of the tenaculum to grasp the cervix and straighten the uterus for proper insertion, transcervical actions (including measuring uterine depth, inserting the IUD insertion Supported by a grant from the National Institutes of Health (HD39717 to D.H.). The views expressed in this article do not necessarily reflect those of the authors institutions or the funding entity. Reprints not available from the authors. tube, and removing the tube), and placement of the device in the uterus. 2 Though IUD use is apparently on the rise in the United States, yet less than 2% of US women of reproductive age 3 use it, the method is more widely used in many European countries: United Kingdom (5%), Switzerland (6%), Germany (6%), Austria (9%), Finland (18%) and France (21%). 4 New research confirming safety, 5-7 recent changes in the Food and Drug Administration approved prescribing information 8 (dropping a recommendation that only parous women use an IUD), and Food and Drug Administration approval of a new levonorgestrel system in year 2000 may broaden US access to this form of birth control /$ - see front matter Ó 2006 Mosby, Inc. All rights reserved. doi: /j.ajog
2 Hubacher et al 1273 Figure 1 Distribution of level of pain from IUD insertion (n = 2018) measured by a 10 cm visual analog scale. Pain from the IUD insertion procedure (or fear of it) may limit the appeal of this contraceptive method, yet today s clinicians have no facts to share with potential acceptors because the existing information is outdated, inaccessible, not applicable, or somewhat meaningless. For example, in large contraceptive efficacy trials, pain at insertion (using a 4-point categorical scale) was not recorded from the patients perspective, but instead was recorded from the clinicians subjective assessment of that experienced by the patient; some level of pain was registered in 13% to 24% of insertions. 9,10 The scales and manner in which pain information was collected in a Brazilian study are unknown, though 9% experienced severe pain from the IUD insertion procedure. 11 In 1, small, well-done study of 58 IUD insertions, researchers used a scale ranging from 0 (no pain) to 10, but the devices used in that study are unavailable today and the results are not applicable. 12 From a different study of only 23 insertions, investigators reported that 4 patients described the process as painful, 16 as uncomfortable, and 3 as painless. 13 Some research has shown that nulliparity, nonlactation, and lengthier time since last pregnancy are associated with more insertion pain 11,14,15 but because the aforementioned shortcomings are common to these studies as well, the findings and ways to prevent pain deserve additional study. To prevent or alleviate pain at insertion, both oral analgesics and cervical anesthetics have been recommended by some 16 though neither medication have undergone rigorous testing to measure effectiveness. A recent American College of Obstetricians and Gynecologists publication 17 on the IUD notes that many clinicians use ibuprofen or nonsteroidal anti-inflammatory drugs for pain control on insertion, but there are limited data of their efficacy. A randomized trial of 50 participants comparing 300 mg of naproxen to placebo found that when taken 1.5 hours before IUD insertion, pain at insertion (using a 5-point scale) was equivalent in the 2 groups; however, use of paracervical blocks may have prevented detecting an impact on pain at insertion. 18 Table I Level of pain at insertion using a 10 cm visual analog scale, by treatment Level of pain Treatment Placebo (N = 1008) Jensen et al 19 conducted a trial of 55 participants and showed that 600 mg of ibuprofen taken at least 1 hour before IUD insertion did not alleviate insertion pain, compared with placebo; median level of pain (on a scale of 0-10) was 3.0. A publication from the Royal College of Obstetricians and Gynaecologists 20 states that Pain relief prior to, and during, IUD insertion should be discussed with women and administered appropriately though there is a lack of randomized controlled trials investigating the use of oral analgesia. Pain relief has been demonstrated with the use of anesthetic gel; in 1 trial of 102 IUD insertions, researchers found that 2% lidocaine gel when applied to the intracervix significantly lowered pain. 21 Because ibuprofen has been shown to reduce IUDinduced side effects, including pain, 22,23 its prophylactic use may alleviate insertion pain; however, existing clinical research provides little guidance on the topic. Only a large trial may be able to determine whether particular subgroups of women (who are likely to experience pain) respond differently to ibuprofen. This article reports on a randomized controlled trial that addresses previous weaknesses in attempts to prevent and simply measure pain from IUD insertion. Materials and methods Ibuprofen* (N = 1010) Mean (95% CI) 2.0 ( ) 1.8 ( ) Median Distribution of pain scores N % N % * Datum on level of pain was missing for 1 ibuprofen participant. We conducted a randomized trial of first-time IUD users to test the possible benefit of prophylactic use of ibuprofen; the main study outcome (early removal caused by bleeding and pain) and more detail on the methodology have been reported previously. 24 We chose first-time IUD users to ensure fair capture/reporting of novel experiences. Briefly, eligible women were aged 18 to 49 years,
3 1274 Hubacher et al Table II Level of pain at insertion: median (n), mean (95% CI), by background characteristics and treatment Total Mean levels (95% CI) by treatment Background characteristics Median (n) Mean (95% CI) Placebo (N = 1008) Ibuprofen (N = 1010) Age (y)* % (327) 1.5 ( ) 1.6 ( ) 1.3 ( ) (794) 1.8 ( ) 1.9 ( ) 1.8 ( ) (464) 2.1 ( ) 2.2 ( ) 2.0 ( ) 30C 1.2 (433) 2.2 ( ) 2.3 ( ) 2.1 ( ) Parity y Nulliparous 2.0 (102) 2.7 ( ) 2.8 ( ) 2.6 ( ) Parous 1.0 (1916) 1.9 ( ) 2.0 ( ) 1.8 ( ) Days since start of last menstrual period*!6 1.0 (1928) 1.9 ( ) 2.0 ( ) 1.8 ( ) (33) 1.0 ( ) 0.9 ( ) 1.1 ( ) 11C 0.9 (57) 1.8 ( ) 1.5 ( ) 2.1 ( ) Time since last pregnancy ended among previously pregnant women (mo)*!3 0.3 (133) 1.0 ( ) 1.1 ( ) 1.0 ( ) (200) 1.6 ( ) 1.6 ( ) 1.5 ( ) O6 1.2 (1625) 2.0 ( ) 2.1 ( ) 1.9 ( ) Currently breastfeeding y Yes 0.8 (538) 1.6 ( ) 1.6 ( ) 1.6 ( ) No 1.1 (1480) 2.0 ( ) 2.1 ( ) 1.9 ( ) * P!.05 using nonparametric Kruskal-Wallis test. y P!.05 using nonparametric Wilcoxon rank test. literate, had menstruated in the last 6 weeks, had never used an IUD, were more than 6 weeks postpartum if recently pregnant, and had no medical contraindications to IUDs or ibuprofen. Participants were recruited at 42 Ministry of Health facilities and 1 private clinic (Instituto Chileno de Medicina Reproductiva, ICMER) in Santiago, Chile. The ethics committees of Family Health International (FHI), the Chilean Ministry of Health, and ICMER approved the protocol and data collection. Research staff obtained written consent from all participants before enrollment. We complied fully with CONSORT guidelines for the conduct and reporting of this trial. 25 On the day of enrollment, participants were randomly assigned either ibuprofen or placebo tablets (identical in appearance); participants took 1 tablet immediately. Clinicians waited at least 45 minutes before inserting the copper IUD (Cu-T380A) and completed other enrollment/preparation tasks in the interim. Participants, clinicians, and the research team were blinded to the true identify of the tablets. At enrollment, clinicians recorded basic sociodemographic, obstetric, and menstrual histories as reported by participants. After IUD insertion, the clinicians taught participants how to record level of pain by using the visual analog scale; we used a 10-cm horizontal line with the left hash mark denoting no pain and the right hash mark denoting worst pain imaginable. Participants wrote an X on the line to characterize their pain; study clinicians measured the distance from the left hash mark to the X and recorded the measurement in centimeters (to 1 decimal place). In pain research, visual analog scales that use a line to represent the continuum of no pain to worst pain imaginable are thought to be superior to other techniques. 26 This study was designed to investigate the possible effect of ibuprofen on IUD continuation rates; thus, the power calculations performed for that purpose are not applicable here. For the present analysis on level of pain at insertion, we discovered that the data were not normally distributed. In light of this, our statistical tests and comparisons use the rank distributions; the Wilcoxon and Kruskal-Wallis nonparametric tests were used to evaluate and report differences. However, because of the large study size and familiarity that most readers have with parametric measures, we also computed means (and 95% CIs) and used them in some graphics 27 to highlight statistically significant nonparametric differences (nonparametric graphics do not convey these differences). We examined a priori participant characteristics associated with higher insertion pain to confirm earlier reports and examined new factors as well. In addition, we did subgroup analyses to determine whether ibuprofen might provide some benefit. To better isolate the possible impact of ibuprofen, we used multivariable regression techniques to assess numerous factors simultaneously. SAS version 9.0 for Windows was used for all statistical analyses (SAS Institute Inc, Cary, NC).
4 Hubacher et al 1275 Figure 2 Level of pain from IUD insertion showing significant differences between user groups based on the mean and 95% confidence interval (represented by the dot and extending arrows, respectively) from an overall scale of 0 to 10. Results Recruitment began in June 2002 and ended in August The details of recruitment and randomization (including a standardized flowchart diagram) have been reported previously. 24 A total of 2019 participants were enrolled; 1011 were assigned to ibuprofen and 1008 to placebo. All participants took the first tablet as instructed before IUD insertion. One ibuprofen participant did not have information on level of pain; thus, we analyzed data on 2018 participants in total. Overall, level of pain for the study population was low (Figure 1). Forty-eight percent reported a pain score of less than 1.0, and 15% experienced pain in the range of 1.0 to 1.9. On other extreme, 11% reported a score of 5.0 or higher and 4.4% reported a score of 7.0 or higher. Mean level of pain was 2.0 for placebo and 1.8 for ibuprofen; this difference was not significant (Table I). The median level of pain for both groups was 1.0 and the distribution of pain levels was similar; level of pain was equivalent in the 2 groups by using nonparametric rank tests. Age, parity, time since start of last menses, lactation status, and time since last pregnancy ended were associated with level of pain at insertion (Table II). Older women experienced higher pain than younger women. Nulliparous women and nonlactating women experienced significantly higher pain than parous and lactating women, respectively. Those whose last pregnancy ended within 3 months experienced significantly less pain than women whose last pregnancy ended at least 6 months ago. Within each subgroup, levels of pain did not differ significantly between placebo and ibuprofen participants. Thus, ignoring the placebo/ibuprofen breakdown and combining all data to improve precision on the estimates of mean level of pain, the primary subgroup differences are shown graphically (Figure 2). Multivariable regression analyses confirmed these results (data not shown). Namely, prophylactic ibuprofen did not alter level of pain even after controlling for numerous sociodemographic factors simultaneously.
5 1276 Hubacher et al Also in the regression analyses, statistically significant increases in pain (P value!.05) were associated with the following factors: increasing age, lower parity, lengthier time since last pregnancy, and nonlactation. Comment We measured IUD insertion pain in more than 2000 first-time users; with a 10-point visual analog scale, we found that the mean and median levels were 1.9 and 1.0, respectively. Prophylactic ibuprofen did not affect level of pain. In subgroup analyses of participants with known high likelihood of experiencing more discomfort, ibuprofen did not alleviate pain either. The primary conclusion from this trial is that the insertion procedure is not painful to the vast majority of women who are having an IUD inserted for the first time in their lives. Thus, given this finding, it is not surprising that prophylactic use of a test medication failed to alleviate already low pain. We also failed to identify a benefit of ibuprofen for subgroups in which higher pain was anticipated. Only 7 participants received paracervical block before insertion; this cannot explain lack of ibuprofen benefit. Fear of pain is often a barrier to seeking elective or even recommended medical procedures. The results of our study provide useful information to women who may be fearful of the IUD insertion procedure. Though the mean and median levels of pain from our study describe the experiences of 2018 women, individual tolerance, anatomic differences, genetics, subjective perceptions, as well as provider skills in inserting an IUD, may influence the level of pain that any 1 person experiences. We conducted our study in public facilities in Santiago, Chile; thus our study population is analogous to women seeking family planning services in Title X-supported clinics in the United States. We tested the minimal effective dosage (400 mg) at a reasonable time before insertion (at least 45 minutes); in most settings, a patient can be counseled, prepared, and receive an IUD within 45 minutes. We did not collect information on the time since last meal, which could affect absorption. A higher dose (such as 800 mg) would have increased the chances of ibuprofen-related side effects and perhaps jeopardized participant compliance with our primary study aim: to improve IUD continuation rates with repeated doses of ibuprofen taken during menses for 6 months. A higher preinsertion dose may not have produced different results; in a study on colposcopy, 800 mg of prophylactic ibuprofen had no impact on reducing pain. 28 Although early IUD removal rates vary slightly depending on which day of the menstrual cycle the IUD is inserted, 29 little has been done to study how level of pain at insertion varies by day of the cycle. In our study, 94% of participants had the IUD inserted within 5 days of the start of their last menstrual period; within that group, level of insertion pain did not vary by day. Outside of that period, we had too few cases to correlate levels of pain to different phases of the menstrual cycle that might alter pain (eg, endometrial build-up, increases in amount and consistency of cervical mucus that occur near ovulation). Clinicians should continue to provide or suggest analgesics such as ibuprofen for treating postinsertion related discomfort that may occur. There is no evidence, however, that prophylactic use of any analgesic is necessary. Acknowledgments We thank the 170 Ministry of Health clinicians for recruiting/enrolling participants and for completing our forms. We also thank Laboratorios Silesia S.A. of Chile for manufacturing and bottling the placebo and ibuprofen tablets. Finally, we thank Bosny Pierre-Louis, Sandra Cameron, and Monica Salvatierra for their dedicated effort. References 1. United Nations. Department of Economic and Social Affairs. World Contraceptive Use Available at: population/publications/contraceptive2003/wallchart_cp2003.pdf. Accessed on February 28, Hollingworth B. Pain control during insertion of an intrauterine device. Br J Fam Plann 1995;21: Mosher WD, Martinez GM, Chandra A, Abma JC, Willson SJ. Use of contraception and use of family planning services in the United States: Adv Data 2004; Population Reference Bureau. Family Planning Worldwide 2002 Data Sheet. Available at: Eng.pdf. Accessed on March 2, Farley TM, Rosenberg MJ, Rowe PJ, Chen JH, Meirik O. Intrauterine devices and pelvic inflammatory disease: an international perspective. Lancet 1992;339: Hubacher D, Lara-Ricalde R, Taylor DJ, Guerra-Infante F, Guzman-Rodriguez R. Use of copper intrauterine devices and the risk of tubal infertility among nulligravid women. N Engl J Med 2001;345: Grimes DA, Schulz KF. Prophylactic antibiotics for intrauterine device insertion: a metaanalysis of the randomized controlled trials. Contraception 1999;60: US Food and Drug Administration. Summary view: safety labeling changes approved by FDA Center for Drug Evaluation and Research (CDER); September Available at: gov/medwatch/safety/2005/sep05_quickview.htm. Accessed on March 12, Farr G, Amatya R. Contraceptive efficacy of the copper T380A and the multiload Cu250 IUD in three developing countries. Adv Contracept 1994;10: Farr G, Amatya R. Contraceptive efficacy of the copper T 380A and copper T 200 intrauterine devices: results from a comparative clinical trial in six developing countries. Contraception 1994;49: Lassner KJ, Chen CH, Kropsch LA, Oberle MW, Lopes IM, Morris L. Comparative study of safety and efficacy of IUD insertions by
6 Hubacher et al 1277 physicians and nursing personnel in Brazil. Bull Pan Am Health Organ 1995;29: Goldstuck ND. A comparison of the initial pain response following insertion of the copper 7 and combined multiload Copper 250-short IUDs. Contracept Deliv Syst 1981;2: Seamark C. Is the fitting of an intrauterine contraceptive device a painful experience? Br J Fam Plann 1993;19: Chi IC, Galich LF, Tauber PF, Wilkens LR, Waszak CS, Siemens AJ, et al. Severe pain at interval IUD insertion: a case-control analysis of patient risk factors. Contraception 1986;34: Chi IC, Wilkens LR, Champion CB, Machemer RE, Rivera R. Insertional pain and other IUD insertion-related rare events for breastfeeding and non-breastfeeding womenda decade s experience in developing countries. Adv Contracept 1989;5: Grimes D. Intrauterine devices (IUDs). Contraceptive technology. 18th ed. New York: Ardent Medic, Inc; p American College of Obstetricians and Gynecologists. Intrauterine device. Obstet Gynecol 2005;105: ACOG practice bulletin no Massey SE, Varady JC, Henzl MR. Pain relief with naproxen following insertion of an intrauterine device. J Reprod Med 1974;13: Jensen HH, Blaabjerg J, Lyndrup J. Profylaktisk brug af prostaglandinsynthesehaemmere ved oplaegning af spiral. Ugeskr Laeger 1998;160: Penney G, Brechin S, de Souza A, Bankowska U, Belfield T, Gormley M, et al. FFPRHC Guidance (January 2004). The copper intrauterine device as long-term contraception. J Fam Plann Reprod Health Care 2004;30: Oloto EJ, Bromham DR, Murty JA. Pain and discomfort perception at IUD insertion-effect of short-duration, low-volume, intracervical application of two percent lignocain gel (Instillagel)-a preliminary study. Br J Fam Plann 1996;22: Roy S, Shaw ST Jr. Role of prostaglandins in IUD-associated uterine bleedingdeffect of a prostaglandin synthetase inhibitor (ibuprofen). Obstet Gynecol 1981;58: Makarainen L, Ylikorkala O. Ibuprofen prevents IUCD-induced increases in menstrual blood loss. BJOG 1986;93: Hubacher D, Reyes V, Lillo S, Pierre-Louis B, Zepeda A, Chen PL, et al. Preventing copper intrauterine device removals due to side effects among first-time users: randomized trial to study the effect of prophylactic ibuprofen. Hum Reprod 2006;21: Moher D, Schulz KF, Altman DG. The CONSORT statement: revised recommendations for improving the quality of reports of parallel-group randomised trials. Lancet 2001;357: Sriwatanakul K, Kelvie W, Lasagna L, Calimlim JF, Weis OF, Mehta G. Studies with different types of visual analog scales for measurement of pain. Clin Pharmacol Ther 1983;34: Mantha S, Thisted R, Foss J, Ellis JE, Roizen MF. A proposal to use confidence intervals for visual analog scale data for pain measurement to determine clinical significance. Anesth Analg 1993;77: Church L, Oliver L, Dobie S, Madigan D, Ellsworth A. Analgesia for colposcopy: double-masked, randomized comparison of ibuprofen and benzocaine gel. Obstet Gynecol 2001;97: White MK, Ory HW, Rooks JB, Rochat RW. Intrauterine device termination rates and the menstrual cycle day of insertion. Obstet Gynecol 1980;55:220-4.
Interventions for pain with intrauterine device insertion (Review)
 Interventions for pain with intrauterine device insertion (Review) Allen RH, Bartz D, Grimes DA, Hubacher D, O Brien P This is a reprint of a Cochrane review, prepared and maintained by The Cochrane Collaboration
Interventions for pain with intrauterine device insertion (Review) Allen RH, Bartz D, Grimes DA, Hubacher D, O Brien P This is a reprint of a Cochrane review, prepared and maintained by The Cochrane Collaboration
Dr. Russo reports no financial relationships relevant to this article. Dr Creinin is a senior clinical advisor for Medicines360.
 CONTRACEPTION Demand for long-acting reversible contraception is growing, including in adolescents and nulliparas. We need to challenge our historical reservations about the IUD and heed the call. Jennefer
CONTRACEPTION Demand for long-acting reversible contraception is growing, including in adolescents and nulliparas. We need to challenge our historical reservations about the IUD and heed the call. Jennefer
The use of long-acting reversible contraceptive
 Overcoming LARC complications: 7 case challenges The strings to your patient s intrauterine device (IUD) are missing. Clinical experience and ACOG direction guide the management plans for this and more
Overcoming LARC complications: 7 case challenges The strings to your patient s intrauterine device (IUD) are missing. Clinical experience and ACOG direction guide the management plans for this and more
Review of IUCD Complications: Lessons from CAT. Dr FG Mhlanga CAT Meeting 24 September 2016
 Review of IUCD Complications: Lessons from CAT Dr FG Mhlanga CAT Meeting 24 September 2016 INTRODUCTION The intrauterine device (IUD) is a reliable long term reversible, cost-effective,easy to use and
Review of IUCD Complications: Lessons from CAT Dr FG Mhlanga CAT Meeting 24 September 2016 INTRODUCTION The intrauterine device (IUD) is a reliable long term reversible, cost-effective,easy to use and
What s New in Adolescent Contraception?
 What s New in Adolescent Contraception? Abby Furukawa, MD Legacy Medical Group Portland Obstetrics and Gynecology April 29, 2017 Objectives Provide an update on contraception options for the adolescent
What s New in Adolescent Contraception? Abby Furukawa, MD Legacy Medical Group Portland Obstetrics and Gynecology April 29, 2017 Objectives Provide an update on contraception options for the adolescent
International Journal of Research in Pharmaceutical and Nano Sciences Journal homepage:
 Review Article ISSN: 2319 9563 International Journal of Research in Pharmaceutical and Nano Sciences Journal homepage: www.ijrpns.com A REVIEW ON INTRAUTERINE DEVICES Boddu Venkata Komali* 1, M. Kalyani
Review Article ISSN: 2319 9563 International Journal of Research in Pharmaceutical and Nano Sciences Journal homepage: www.ijrpns.com A REVIEW ON INTRAUTERINE DEVICES Boddu Venkata Komali* 1, M. Kalyani
Non-steroidal anti-inflammatory drugs for heavy bleeding or pain associated with intrauterine-device use(review)
 Cochrane Database of Systematic Reviews Non-steroidal anti-inflammatory drugs for heavy bleeding or pain associated with intrauterine-device use(review) GrimesDA,HubacherD,LopezLM,SchulzKF Grimes DA, Hubacher
Cochrane Database of Systematic Reviews Non-steroidal anti-inflammatory drugs for heavy bleeding or pain associated with intrauterine-device use(review) GrimesDA,HubacherD,LopezLM,SchulzKF Grimes DA, Hubacher
2
 1 2 3 1. Usinger KM et al. Intrauterine contraception continuation in adolescents and young women: a systematic review. J Pediatr Adolesc Gynecol 2016; 29: 659 67. 2. Kost K et al. Estimates of contraceptive
1 2 3 1. Usinger KM et al. Intrauterine contraception continuation in adolescents and young women: a systematic review. J Pediatr Adolesc Gynecol 2016; 29: 659 67. 2. Kost K et al. Estimates of contraceptive
1. Ortiz, M. E et al. Mechanisms of action of intrauterine devices. Obstet & Gynl Survey 1996; 51(12), 42S-51S.
 1 2 1. Ortiz, M. E et al. Mechanisms of action of intrauterine devices. Obstet & Gynl Survey 1996; 51(12), 42S-51S. The contraceptive action of all IUDs is mainly in the uterine cavity. The major effect
1 2 1. Ortiz, M. E et al. Mechanisms of action of intrauterine devices. Obstet & Gynl Survey 1996; 51(12), 42S-51S. The contraceptive action of all IUDs is mainly in the uterine cavity. The major effect
LEARNING OBJECTIVES. Beyond the Pill: Long Acting Contraception. Distribution Of Contraception Use By Women In The Us. Unintended Pregnancy is Common
 4:15 5 pm Beyond the Pill: Long Acting Contraceptives and IUDs Presenter Disclosure Information The following relationships exist related to this presentation: Christine L. Curry, MD, PhD: No financial
4:15 5 pm Beyond the Pill: Long Acting Contraceptives and IUDs Presenter Disclosure Information The following relationships exist related to this presentation: Christine L. Curry, MD, PhD: No financial
Instruction for the patient
 WS 4 Case 3 STI and IUD Your situation Instruction for the patient You are 32 years old, divorced and have one child; you have just started a new relationship You underwent surgical resection of the left
WS 4 Case 3 STI and IUD Your situation Instruction for the patient You are 32 years old, divorced and have one child; you have just started a new relationship You underwent surgical resection of the left
Unintended Pregnancy is Common LEARNING OBJECTIVES. Distribution Of Contraception Use By Women In The Us. Unintended Pregnancy And Contraceptive Use
 3:45 4:30 pm Beyond the Pill: Long Acting Contraceptives and IUDs Presenter Disclosure Information The following relationships exist related to this presentation: Christine L. Curry, MD, PhD: No financial
3:45 4:30 pm Beyond the Pill: Long Acting Contraceptives and IUDs Presenter Disclosure Information The following relationships exist related to this presentation: Christine L. Curry, MD, PhD: No financial
Simplifying Vide Contraception. University of Utah Department of Ob/Gyn Post Grad Course February 13, 2017 David Turok
 Simplifying Vide Contraception University of Utah Department of Ob/Gyn Post Grad Course February 13, 2017 David Turok Background Objectives At the conclusion of this presentation participants will be able
Simplifying Vide Contraception University of Utah Department of Ob/Gyn Post Grad Course February 13, 2017 David Turok Background Objectives At the conclusion of this presentation participants will be able
Lidocaine-Prilocaine Cream as Analgesia for IUD Insertion: A Prospective, Randomized, Controlled, Triple Blinded Study
 Global Journal of Health Science; Vol. 7, No. 4; 2015 ISSN 1916-9736 E-ISSN 1916-9744 Published by Canadian Center of Science and Education Lidocaine-Prilocaine Cream as Analgesia for IUD Insertion: A
Global Journal of Health Science; Vol. 7, No. 4; 2015 ISSN 1916-9736 E-ISSN 1916-9744 Published by Canadian Center of Science and Education Lidocaine-Prilocaine Cream as Analgesia for IUD Insertion: A
Intrauterine Devices (IUDs): Access for Women in the U.S.
 November 2016 Fact Sheet Intrauterine Devices (IUDs): Access for Women in the U.S. Intrauterine devices (IUDs) are one of the most effective forms of reversible contraception. IUDs, along with implants,
November 2016 Fact Sheet Intrauterine Devices (IUDs): Access for Women in the U.S. Intrauterine devices (IUDs) are one of the most effective forms of reversible contraception. IUDs, along with implants,
Intrauterine Devices and Pelvic Inflammatory Disease: Dispelling the Myth. Dr Amy Moten SHine SA Co-authors: Dr Richelle Douglas; Dr Alison Creagh SHQ
 Australasian Sexual Health Conference Adelaide, November 14-16 Intrauterine Devices and Pelvic Inflammatory Disease: Dispelling the Myth Dr Amy Moten SHine SA Co-authors: Dr Richelle Douglas; Dr Alison
Australasian Sexual Health Conference Adelaide, November 14-16 Intrauterine Devices and Pelvic Inflammatory Disease: Dispelling the Myth Dr Amy Moten SHine SA Co-authors: Dr Richelle Douglas; Dr Alison
The most commonly chosen methods of contraception
 Original Research Effects of Age, Parity, and Device Type on Complications and Discontinuation of Intrauterine Devices Joelle Aoun, MD, Virginia A. Dines, BS, Dale W. Stovall, MD, Mihriye Mete, PhD, Casey
Original Research Effects of Age, Parity, and Device Type on Complications and Discontinuation of Intrauterine Devices Joelle Aoun, MD, Virginia A. Dines, BS, Dale W. Stovall, MD, Mihriye Mete, PhD, Casey
LARC: Disclosures. Long Acting Reversible Contraception. Objectives 10/23/2013. I have no relevant financial disclosures
 LARC: Long Acting Reversible Contraception Disclosures I have no relevant financial disclosures Jennifer Kerns, MD, MPH Assistant Professor, UCSF Obstetrics, Gynecology and Reproductive Sciences San Francisco
LARC: Long Acting Reversible Contraception Disclosures I have no relevant financial disclosures Jennifer Kerns, MD, MPH Assistant Professor, UCSF Obstetrics, Gynecology and Reproductive Sciences San Francisco
LONG-ACTING REVERSIBLE CONTRACEPTION. Summary Tables
 LONG-ACTING REVERSIBLE CONTRACEPTION Summary Tables Bridging the Divide: A Project of the Jacobs Institute of Women s Health June 2016 Table 1. Summary of LARC Methods Available Years Since Effective Copper
LONG-ACTING REVERSIBLE CONTRACEPTION Summary Tables Bridging the Divide: A Project of the Jacobs Institute of Women s Health June 2016 Table 1. Summary of LARC Methods Available Years Since Effective Copper
A Comparative Study between the Side Effects of Copper Intrauterine Device in Women with Non-scarred and Scarred Uterus
 Iraqi JMS Published by Al-Nahrain College of Medicine ISSN 161-659 Email: Iraqi_jms_alnahrain@yahoo.com http://www. colmed-nahrain.edu.iq/ A Comparative Study between the Side Effects of Copper Intrauterine
Iraqi JMS Published by Al-Nahrain College of Medicine ISSN 161-659 Email: Iraqi_jms_alnahrain@yahoo.com http://www. colmed-nahrain.edu.iq/ A Comparative Study between the Side Effects of Copper Intrauterine
INTRAUTERINE DEVICES AND INFECTIONS. Tips for Evaluation and Management
 INTRAUTERINE DEVICES AND INFECTIONS Tips for Evaluation and Management Objectives At the end of this presentation, the participant should be able to: 1. Diagnose infection after IUD placement 2. Provide
INTRAUTERINE DEVICES AND INFECTIONS Tips for Evaluation and Management Objectives At the end of this presentation, the participant should be able to: 1. Diagnose infection after IUD placement 2. Provide
Same-Day Access to Highly Effective Reversible Contraception: Moving from Plan B to Happily Ever After
 Same-Day Access to Highly Effective Reversible Contraception: Moving from Plan B to Happily Ever After Eleanor Bimla Schwarz, MD, MS Director, Women s Health Services Research Unit Center for Research
Same-Day Access to Highly Effective Reversible Contraception: Moving from Plan B to Happily Ever After Eleanor Bimla Schwarz, MD, MS Director, Women s Health Services Research Unit Center for Research
Surgical Interruption of Pelvic Nerve Pathways for Primary and Secondary Dysmenorrhea
 Page: 1 of 7 Last Review Status/Date: June 2015 for Primary and Secondary Dysmenorrhea Description Two laparoscopic surgical approaches are proposed as adjuncts to conservative surgical therapy for the
Page: 1 of 7 Last Review Status/Date: June 2015 for Primary and Secondary Dysmenorrhea Description Two laparoscopic surgical approaches are proposed as adjuncts to conservative surgical therapy for the
The modern intrauterine device (IUD) is highly reliable and
 Intrauterine Device Knowledge and Practices: A National Survey of Obstetrics and Gynecology Residents Jennifer Tang, MD, MSCR, Rie Maurer, MA, and Deborah Bartz, MD, MPH Objectives: The primary objective
Intrauterine Device Knowledge and Practices: A National Survey of Obstetrics and Gynecology Residents Jennifer Tang, MD, MSCR, Rie Maurer, MA, and Deborah Bartz, MD, MPH Objectives: The primary objective
Application for inclusion of levonorgestrel - releasing IUD for contraception in the WHO Model List of Essential Medicines
 Application for inclusion of levonorgestrel - releasing IUD for contraception in the WHO Model List of Essential Medicines 1. Summary statement of the proposal for inclusion LNG-IUS is an effective contraceptive;
Application for inclusion of levonorgestrel - releasing IUD for contraception in the WHO Model List of Essential Medicines 1. Summary statement of the proposal for inclusion LNG-IUS is an effective contraceptive;
Dedicated postpartum IUD inserter. Dr. Anne Burke, Johns Hopkins Medicine Dr. Leo Han, OHSU Healthcare July 13, 2016
 Dedicated postpartum IUD inserter Dr. Anne Burke, Johns Hopkins Medicine Dr. Leo Han, OHSU Healthcare July 13, 2016 Unmet need for FP in postpartum period Across 27 countries, 65% of postpartum women had
Dedicated postpartum IUD inserter Dr. Anne Burke, Johns Hopkins Medicine Dr. Leo Han, OHSU Healthcare July 13, 2016 Unmet need for FP in postpartum period Across 27 countries, 65% of postpartum women had
Intrauterine devices (IUDs) are long-acting reversible
 Contraception: Original Research Paracervical Block for Intrauterine Device Placement Among Nulliparous Women A Randomized Controlled Trial Sheila K. Mody, MD, MPH, John Paul Farala, MD, Berenice Jimenez,
Contraception: Original Research Paracervical Block for Intrauterine Device Placement Among Nulliparous Women A Randomized Controlled Trial Sheila K. Mody, MD, MPH, John Paul Farala, MD, Berenice Jimenez,
Key words: Contraception, Copper T380A, Discontinuation.
 Discontinuation Rates among Women Using either the Combined Oral Contraceptive Pills or an Intrauterine Contraceptive Device for Contraception: A Comparative Study Ehab Al-Rayyan MD*, Zakarya Bani Meri
Discontinuation Rates among Women Using either the Combined Oral Contraceptive Pills or an Intrauterine Contraceptive Device for Contraception: A Comparative Study Ehab Al-Rayyan MD*, Zakarya Bani Meri
Health Numeracy: Explaining risk in numbers patients can use. Kirtly Parker Jones MD
 Health Numeracy: Explaining risk in numbers patients can use Kirtly Parker Jones MD Learning Objectives List three different numerical means of presenting risk Describe three graphical methods of presenting
Health Numeracy: Explaining risk in numbers patients can use Kirtly Parker Jones MD Learning Objectives List three different numerical means of presenting risk Describe three graphical methods of presenting
SALSA: Self-Administered Lidocaine Gel for Pain Management with First Trimester Surgical Abortion A Randomized Trial. Paul D Blumenthal, MD, MPH
 SALSA: Self-Administered Lidocaine Gel for Pain Management with First Trimester Surgical Abortion A Randomized Trial Paul D Blumenthal, MD, MPH Background Pain is a limiting factor in where and how abortion
SALSA: Self-Administered Lidocaine Gel for Pain Management with First Trimester Surgical Abortion A Randomized Trial Paul D Blumenthal, MD, MPH Background Pain is a limiting factor in where and how abortion
BRIEF REPORTS. Providing Long-Acting Reversible Contraception in an Academic Family Medicine Center Jennifer Amico, MD, MPH; Justine Wu, MD, MPH
 Providing Long-Acting Reversible Contraception in an Academic Family Medicine Center Jennifer Amico, MD, MPH; Justine Wu, MD, MPH BACKGROUND AND OBJECTIVES: Providing long-acting reversible contraception
Providing Long-Acting Reversible Contraception in an Academic Family Medicine Center Jennifer Amico, MD, MPH; Justine Wu, MD, MPH BACKGROUND AND OBJECTIVES: Providing long-acting reversible contraception
Emergency Contraception THE FACTS
 Emergency Contraception Quick Facts What is it? Emergency contraception is birth control that you use after you have had unprotected sex--if you didn t use birth control or your regular birth control failed.
Emergency Contraception Quick Facts What is it? Emergency contraception is birth control that you use after you have had unprotected sex--if you didn t use birth control or your regular birth control failed.
Example Clinical Guideline for Immediate Postpartum LARC Insertion
 Example Clinical Guideline for Immediate Postpartum LARC Insertion RATIONALE Delay in contraceptive provision until the six week postpartum appointment can leave some women at risk for rapid repeat pregnancy.
Example Clinical Guideline for Immediate Postpartum LARC Insertion RATIONALE Delay in contraceptive provision until the six week postpartum appointment can leave some women at risk for rapid repeat pregnancy.
Infertility services reported by men in the United States: national survey data
 MALE FACTOR Infertility services reported by men in the United States: national survey data John E. Anderson, Ph.D., Sherry L. Farr, Ph.D., M.S.P.H., Denise J. Jamieson, M.D., M.P.H., Lee Warner, Ph.D.,
MALE FACTOR Infertility services reported by men in the United States: national survey data John E. Anderson, Ph.D., Sherry L. Farr, Ph.D., M.S.P.H., Denise J. Jamieson, M.D., M.P.H., Lee Warner, Ph.D.,
Levosert levonorgestrel 20mcg/24hour intrauterine device
 Levosert levonorgestrel 20mcg/24hour intrauterine device Verdict: Formulary inclusion: Formulary category: Restrictions: Reason for inclusion: Link to formulary: Link to medicine review summary: Levosert
Levosert levonorgestrel 20mcg/24hour intrauterine device Verdict: Formulary inclusion: Formulary category: Restrictions: Reason for inclusion: Link to formulary: Link to medicine review summary: Levosert
MEDICAL POLICY SUBJECT: FEMALE STERILIZATION. POLICY NUMBER: CATEGORY: Contract Clarification
 MEDICAL POLICY SUBJECT: FEMALE STERILIZATION PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: FEMALE STERILIZATION PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Disclosures. Contraceptive Method Use, U.S. Best Practices in Contraception: Advances, Tips, and Tricks
 Best Practices in Contraception: Advances, Tips, and Tricks Disclosures I have no disclosures I may discuss off-label use of some contraceptives Biftu Mengesha MD MAS Department of Obstetrics, Gynecology
Best Practices in Contraception: Advances, Tips, and Tricks Disclosures I have no disclosures I may discuss off-label use of some contraceptives Biftu Mengesha MD MAS Department of Obstetrics, Gynecology
Example CLINICAL GUIDELINES for Postpartum IUD insertion
 Example CLINICAL GUIDELINES for Postpartum IUD insertion Postpartum Intrauterine Device Insertion 1.0 Indications: 1.1 Insertion of an intrauterine device (IUD) for long-acting reversible contraception
Example CLINICAL GUIDELINES for Postpartum IUD insertion Postpartum Intrauterine Device Insertion 1.0 Indications: 1.1 Insertion of an intrauterine device (IUD) for long-acting reversible contraception
Patient Awareness and Understanding of Intrauterine Devices
 University of Vermont ScholarWorks @ UVM Family Medicine Block Clerkship, Student Projects College of Medicine 2016 Patient Awareness and Understanding of Intrauterine Devices Lindsey Marie Eastman University
University of Vermont ScholarWorks @ UVM Family Medicine Block Clerkship, Student Projects College of Medicine 2016 Patient Awareness and Understanding of Intrauterine Devices Lindsey Marie Eastman University
Health care providers' knowledge about contraceptive evidence: a barrier to quality family planning care?
 Contraception 81 (2010) 292 298 Original research article Health care providers' knowledge about contraceptive evidence: a barrier to quality family planning care? Christine Dehlendorf a,b, Kira Levy a,,
Contraception 81 (2010) 292 298 Original research article Health care providers' knowledge about contraceptive evidence: a barrier to quality family planning care? Christine Dehlendorf a,b, Kira Levy a,,
RESOLUTION NO. 301 (Co-Sponsored G) SUBSTITUTE ADOPTED See Below
 RESOLUTION NO. 301 (Co-Sponsored G) SUBSTITUTE ADOPTED See Below Support Placement and Coverage of Long-Acting Reversible Contraceptives (LARC) in the Early Postpartum Period Introduced by the California
RESOLUTION NO. 301 (Co-Sponsored G) SUBSTITUTE ADOPTED See Below Support Placement and Coverage of Long-Acting Reversible Contraceptives (LARC) in the Early Postpartum Period Introduced by the California
Disturbance of uterine bleeding patterns, often anecdotally
 Management of Initial Bleeding or Spotting After Levonorgestrel-Releasing Intrauterine System Placement A Randomized Controlled Trial Terje Sørdal, MD, Pirjo Inki, MD, PhD, John Draeby, MD, Mary O Flynn,
Management of Initial Bleeding or Spotting After Levonorgestrel-Releasing Intrauterine System Placement A Randomized Controlled Trial Terje Sørdal, MD, Pirjo Inki, MD, PhD, John Draeby, MD, Mary O Flynn,
Surgical Interruption of Pelvic Nerve Pathways for Primary and Secondary Dysmenorrhea. Original Policy Date
 MP 4.01.10 Surgical Interruption of Pelvic Nerve Pathways for Primary and Secondary Dysmenorrhea Medical Policy Section OB/Gyn/Reproduction Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date
MP 4.01.10 Surgical Interruption of Pelvic Nerve Pathways for Primary and Secondary Dysmenorrhea Medical Policy Section OB/Gyn/Reproduction Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date
Zurich Open Repository and Archive
 University of Zurich Zurich Open Repository and Archive Winterthurerstr. 190 CH-8057 Zurich http://www.zora.uzh.ch Year: 2008 Partial and complete expulsion of the Multiload 375 IUD and the levonorgestrel-releasing
University of Zurich Zurich Open Repository and Archive Winterthurerstr. 190 CH-8057 Zurich http://www.zora.uzh.ch Year: 2008 Partial and complete expulsion of the Multiload 375 IUD and the levonorgestrel-releasing
FDA-Approved Patient Labeling Patient Information Mirena (mur-ā-nah) (levonorgestrel-releasing intrauterine system)
 FDA-Approved Patient Labeling Patient Information Mirena (mur-ā-nah) (levonorgestrel-releasing intrauterine system) Mirena does not protect against HIV infection (AIDS) and other sexually transmitted infections
FDA-Approved Patient Labeling Patient Information Mirena (mur-ā-nah) (levonorgestrel-releasing intrauterine system) Mirena does not protect against HIV infection (AIDS) and other sexually transmitted infections
the IUD the IUD the IUD the IUD the IUD the IUD the IUD the IUD the IUD the IUD the IUD the IUD the IUD your guide to
 your guide to Helping you choose the method of contraception that s best for you IUD IUD the e IUD IU IUD the IUD 2 3 The intrauterine device (IUD) An IUD is a small plastic and copper device that s put
your guide to Helping you choose the method of contraception that s best for you IUD IUD the e IUD IU IUD the IUD 2 3 The intrauterine device (IUD) An IUD is a small plastic and copper device that s put
A Study of Physician Recommendations for Reversible Contraceptive Methods Using Standardized Patients
 A Study of Physician Recommendations for Reversible Contraceptive Methods Using Standardized Patients By Christine Dehlendorf, Kevin Grumbach, Eric Vittinghoff, Rachel Ruskin and Jody Steinauer Christine
A Study of Physician Recommendations for Reversible Contraceptive Methods Using Standardized Patients By Christine Dehlendorf, Kevin Grumbach, Eric Vittinghoff, Rachel Ruskin and Jody Steinauer Christine
Product Information. Confidence that lasts
 Confidence that lasts What is Mirena? Inhibition of sperm motility and function inside the uterus and the fallopian tubes, preventing fertilization (Videla-Rivero et al. 1987). Section of system Levonorgestrel
Confidence that lasts What is Mirena? Inhibition of sperm motility and function inside the uterus and the fallopian tubes, preventing fertilization (Videla-Rivero et al. 1987). Section of system Levonorgestrel
2/4/2011. What is your specialty? A. Family practice B. Internal medicine and subs C. OB/GYN D. Peds E. Surgery and subs
 Steve P. Buchanan D.O. FACOOG(Dist.) TOMA Mid Winter February 11, 2011 Dallas,TX Associate Professor OB/GYN UNTHSC/TCOM 1987- present Executive Vice President, American College of Osteopathic Obstetricians
Steve P. Buchanan D.O. FACOOG(Dist.) TOMA Mid Winter February 11, 2011 Dallas,TX Associate Professor OB/GYN UNTHSC/TCOM 1987- present Executive Vice President, American College of Osteopathic Obstetricians
Differences in Women Who Choose Subdermal Implants Versus Intrauterine Devices
 The Journal of Reproductive Medicine Differences in Women Who Choose Subdermal Implants Versus Intrauterine Devices Vien C. Lam, M.D., Emily E. Hadley, M.D., Abbey B. Berenson, M.D., Ph.D., Jacqueline
The Journal of Reproductive Medicine Differences in Women Who Choose Subdermal Implants Versus Intrauterine Devices Vien C. Lam, M.D., Emily E. Hadley, M.D., Abbey B. Berenson, M.D., Ph.D., Jacqueline
MENSTRUAL PATTERNS AND WOMEN'S ATTITUDES FOLLOWING STERILIZATION BY FA LOPE RINGS*
 FERTILITY AND STERILITY Copyright 1979 The American Fertility Society Vol. 31, No.6, June 1979 Printed in U.s.A. MENSTRUAL PATTERNS AND WOMEN'S ATTITUDES FOLLOWING STERILIZATION BY FA LOPE RINGS* LIDIA
FERTILITY AND STERILITY Copyright 1979 The American Fertility Society Vol. 31, No.6, June 1979 Printed in U.s.A. MENSTRUAL PATTERNS AND WOMEN'S ATTITUDES FOLLOWING STERILIZATION BY FA LOPE RINGS* LIDIA
INTERVENTIONAL PROCEDURES PROGRAMME
 NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of microwave endometrial ablation Introduction This overview has been prepared to assist
NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of microwave endometrial ablation Introduction This overview has been prepared to assist
VCHIP LARC Needs Assessment Survey
 VCHIP LARC Needs Assessment Survey Demographics 1. How many have you been in practice (post-training)? Choose one of the following answers 0-5 6-10 11-15 16-20 21 or more 2. What are your professional
VCHIP LARC Needs Assessment Survey Demographics 1. How many have you been in practice (post-training)? Choose one of the following answers 0-5 6-10 11-15 16-20 21 or more 2. What are your professional
Emergency contraception: Separating fact from fiction
 REVIEW CME CREDIT EDUCATIONAL OBJECTIVE: Readers will prescribe emergency contraception confidently PELIN BATUR, MD, FACP, NCMP Education Director, Primary Care Women s Health, Cleveland Clinic Independence
REVIEW CME CREDIT EDUCATIONAL OBJECTIVE: Readers will prescribe emergency contraception confidently PELIN BATUR, MD, FACP, NCMP Education Director, Primary Care Women s Health, Cleveland Clinic Independence
BLEEDING PATTERNS AND CONTRACEPTIVE DISCONTINUATION FG MHLANGA MTN ANNUAL MEETING 20 MARCH 2018
 BLEEDING PATTERNS AND CONTRACEPTIVE DISCONTINUATION FG MHLANGA MTN ANNUAL MEETING 20 MARCH 2018 Introduction Bleeding with contraception may lead to discontinuation and possible unintended pregnancy What
BLEEDING PATTERNS AND CONTRACEPTIVE DISCONTINUATION FG MHLANGA MTN ANNUAL MEETING 20 MARCH 2018 Introduction Bleeding with contraception may lead to discontinuation and possible unintended pregnancy What
38 OBG Management August 2012 Vol. 24 No. 8 obgmanagement.com
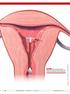 FIGURE 1 Copper intrauterine device displaced in the lower uterine segment with the left arm embedded in the myometrium. illustrations: craig zuckerman for obg management 38 OBG Management August 2012
FIGURE 1 Copper intrauterine device displaced in the lower uterine segment with the left arm embedded in the myometrium. illustrations: craig zuckerman for obg management 38 OBG Management August 2012
Primary dysmenorrhea, defined as painful menstruation
 Oral Contraceptives for Dysmenorrhea in Adolescent Girls A Randomized Trial Anne Rachel Davis, MD, Carolyn Westhoff, MD, Katharine O Connell, MD, and Nancy Gallagher, RN Objective: To assess whether a
Oral Contraceptives for Dysmenorrhea in Adolescent Girls A Randomized Trial Anne Rachel Davis, MD, Carolyn Westhoff, MD, Katharine O Connell, MD, and Nancy Gallagher, RN Objective: To assess whether a
Contraception. Objectives. Unintended Pregnancy. Unintended Pregnancy in the US. What s the Impact? 10/7/2014
 Contraception Tami Allen, RNC OB, MHA Robin Petersen, RN, MSN Perinatal Clinical Nurse Specialist Objectives Discuss the impact of unintended pregnancy in the United States Discuss the risks and benefits
Contraception Tami Allen, RNC OB, MHA Robin Petersen, RN, MSN Perinatal Clinical Nurse Specialist Objectives Discuss the impact of unintended pregnancy in the United States Discuss the risks and benefits
levonorgestrel 13.5mg intrauterine delivery system (Jaydess ) SMC No. (1036/15) Bayer
 levonorgestrel 13.5mg intrauterine delivery system (Jaydess ) SMC No. (1036/15) Bayer 6 March 2015 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
levonorgestrel 13.5mg intrauterine delivery system (Jaydess ) SMC No. (1036/15) Bayer 6 March 2015 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
Patient or Clinician: Duration of Use of Intrauterine Devices Based on Who Initiated Discussion of Placement
 ORIGINAL RESEARCH Patient or Clinician: Duration of Use of Intrauterine Devices Based on Who Initiated Discussion of Placement Tammy Chang, MD, MPH, MS, Michelle H. Moniz, MD, MSc, Melissa A. Plegue, MA,
ORIGINAL RESEARCH Patient or Clinician: Duration of Use of Intrauterine Devices Based on Who Initiated Discussion of Placement Tammy Chang, MD, MPH, MS, Michelle H. Moniz, MD, MSc, Melissa A. Plegue, MA,
SIMPLE PERMANENT CONTRACEPTION A HIGHLY EFFECTIVE AND COMPLICATION FREE BIRTH CONTROL DEVICE
 CONSIDER WHAT IS BEST FOR YOU... SIMPLE PERMANENT CONTRACEPTION A HIGHLY EFFECTIVE AND COMPLICATION FREE BIRTH CONTROL DEVICE Effective. Safe. Proven. FILSHIE - THE CLIP, A HIGHLY EFFECTIVE CHOICE FOR
CONSIDER WHAT IS BEST FOR YOU... SIMPLE PERMANENT CONTRACEPTION A HIGHLY EFFECTIVE AND COMPLICATION FREE BIRTH CONTROL DEVICE Effective. Safe. Proven. FILSHIE - THE CLIP, A HIGHLY EFFECTIVE CHOICE FOR
Intrauterine contraceptive devices (IUDs) are becoming
 Malpositioned Intrauterine Contraceptive Devices Risk Factors, Outcomes, and Future Pregnancies Kari P. Braaten, MD, MPH, Carol B. Benson, MD, Rie Maurer, MA, and Alisa B. Goldberg, MD, MPH OBJECTIVE:
Malpositioned Intrauterine Contraceptive Devices Risk Factors, Outcomes, and Future Pregnancies Kari P. Braaten, MD, MPH, Carol B. Benson, MD, Rie Maurer, MA, and Alisa B. Goldberg, MD, MPH OBJECTIVE:
Surgical Interruption of Pelvic Nerve Pathways for Primary and Secondary Dysmenorrhea
 Surgical Interruption of Pelvic Nerve Pathways for Primary and Secondary Dysmenorrhea Policy Number: 4.01.17 Last Review: 11/2013 Origination: 11/2007 Next Review: 11/2014 Policy Blue Cross and Blue Shield
Surgical Interruption of Pelvic Nerve Pathways for Primary and Secondary Dysmenorrhea Policy Number: 4.01.17 Last Review: 11/2013 Origination: 11/2007 Next Review: 11/2014 Policy Blue Cross and Blue Shield
ACCESS LARC INCREASING ACCESS TO IMMEDIATE POSTPARTUM LONG-ACTING REVERSIBLE CONTRACEPTION
 ACCESS LARC INCREASING ACCESS TO IMMEDIATE POSTPARTUM LONG-ACTING REVERSIBLE CONTRACEPTION Chapter Four: Policies and Procedures Overview This procedure outline is a pre-cursor to detailed procedures related
ACCESS LARC INCREASING ACCESS TO IMMEDIATE POSTPARTUM LONG-ACTING REVERSIBLE CONTRACEPTION Chapter Four: Policies and Procedures Overview This procedure outline is a pre-cursor to detailed procedures related
Long Acting Reversible Contraception: First Line Care for Adolescents. David A. Levine, MD, FAAP Melissa Kottke, MD, MPH, FACOG
 Long Acting Reversible Contraception: First Line Care for Adolescents David A. Levine, MD, FAAP Melissa Kottke, MD, MPH, FACOG Disclosures Melissa Kottke is a Nexplanon trainer for Merck Objectives Describe
Long Acting Reversible Contraception: First Line Care for Adolescents David A. Levine, MD, FAAP Melissa Kottke, MD, MPH, FACOG Disclosures Melissa Kottke is a Nexplanon trainer for Merck Objectives Describe
1. Ng M et a l. Global, regional, and national prevalence of overweight and obesity in children and adults during : A systematic analysis
 1 2 3 1. Ng M et a l. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980 2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet
1 2 3 1. Ng M et a l. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980 2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet
Characteristics of users of intrauterine devices and other reversible contraceptive methods in the United States
 CONTRACEPTION Characteristics of users of intrauterine devices and other reversible contraceptive methods in the United States Xin Xu, Ph.D., a Maurizio Macaluso, M.D., Dr.P.H., a Jennifer Frost, Ph.D.,
CONTRACEPTION Characteristics of users of intrauterine devices and other reversible contraceptive methods in the United States Xin Xu, Ph.D., a Maurizio Macaluso, M.D., Dr.P.H., a Jennifer Frost, Ph.D.,
The Doctor Is In. Brent N Davidson MD Vice Chair Women s Health Henry Ford Health System Medical Director Family Planning MDCH
 The Doctor Is In Brent N Davidson MD Vice Chair Women s Health Henry Ford Health System Medical Director Family Planning MDCH Contraception Resources from the CDC: 2016 U.S. Medical Eligibility Criteria
The Doctor Is In Brent N Davidson MD Vice Chair Women s Health Henry Ford Health System Medical Director Family Planning MDCH Contraception Resources from the CDC: 2016 U.S. Medical Eligibility Criteria
Chapter 7 Infertility, Contraception, and Abortion
 Chapter 7 Infertility, Contraception, and Abortion Infertility Incidence Affects about 10% to 15% of reproductive-age population Subfertility: prolonged time to conceive Sterility: inability to conceive
Chapter 7 Infertility, Contraception, and Abortion Infertility Incidence Affects about 10% to 15% of reproductive-age population Subfertility: prolonged time to conceive Sterility: inability to conceive
Research. Breast cancer represents a major
 Research GENERAL GYNECOLOGY Gynecologic conditions in participants in the NSABP breast cancer prevention study of tamoxifen and raloxifene (STAR) Carolyn D. Runowicz, MD; Joseph P. Costantino, DrPH; D.
Research GENERAL GYNECOLOGY Gynecologic conditions in participants in the NSABP breast cancer prevention study of tamoxifen and raloxifene (STAR) Carolyn D. Runowicz, MD; Joseph P. Costantino, DrPH; D.
An Illustrative Communication Strategy for Contraceptive Implants
 An Illustrative Communication Strategy for Contraceptive Implants: Step 1 (Analyze the Situation) 1 An Illustrative Communication Strategy for Contraceptive Implants Step 1: Analyze the Situation Health
An Illustrative Communication Strategy for Contraceptive Implants: Step 1 (Analyze the Situation) 1 An Illustrative Communication Strategy for Contraceptive Implants Step 1: Analyze the Situation Health
2
 1 2 3 1. Usinger KM et al. Intrauterine contraception continuation in adolescents and young women: a systematic review. J Pediatr Adolesc Gynecol 2016; 29: 659 67. 2. Kost K et al. Estimates of contraceptive
1 2 3 1. Usinger KM et al. Intrauterine contraception continuation in adolescents and young women: a systematic review. J Pediatr Adolesc Gynecol 2016; 29: 659 67. 2. Kost K et al. Estimates of contraceptive
Pap Smears Pelvic Examinations Well Woman Examinations. When should you have them performed???
 Pap Smears Pelvic Examinations Well Woman Examinations. When should you have them performed??? Arlene Evans-DeBeverly, PA-C Copyright 2012 There are always ongoing changes in gynecology, including the
Pap Smears Pelvic Examinations Well Woman Examinations. When should you have them performed??? Arlene Evans-DeBeverly, PA-C Copyright 2012 There are always ongoing changes in gynecology, including the
Extended use of intrauterine devices: How long can we go?
 Extended use of intrauterine devices: How long can we go? Justine P. Wu, MD, MPH Sarah Pickle, MD Rutgers Robert Wood Johnson Medical School Department of Family Medicine & Community Health Disclosures
Extended use of intrauterine devices: How long can we go? Justine P. Wu, MD, MPH Sarah Pickle, MD Rutgers Robert Wood Johnson Medical School Department of Family Medicine & Community Health Disclosures
Click to edit Master title style. Unintended Pregnancy: Prevalence. Unintended Pregnancy: Risk Groups. Unintended Pregnancy: Consequences 9/23/2015
 The Role of Childhood Violence, Self-esteem and Depressive Symptoms on Inconsistent Contraception Use among Young, Sexually Active Women Deborah B. Nelson, PhD Associate Professor Unintended Pregnancy:
The Role of Childhood Violence, Self-esteem and Depressive Symptoms on Inconsistent Contraception Use among Young, Sexually Active Women Deborah B. Nelson, PhD Associate Professor Unintended Pregnancy:
Welcome to Mirena. The Mirena Handbook: A Personal Guide to Your New Mirena. mirena.com. Mirena is the #1 prescribed IUD * in the U.S.
 Mirena is the #1 prescribed IUD * in the U.S. Welcome to Mirena The Mirena Handbook: A Personal Guide to Your New Mirena *Intrauterine Device Supported by 2015-2016 SHS data INDICATIONS FOR MIRENA Mirena
Mirena is the #1 prescribed IUD * in the U.S. Welcome to Mirena The Mirena Handbook: A Personal Guide to Your New Mirena *Intrauterine Device Supported by 2015-2016 SHS data INDICATIONS FOR MIRENA Mirena
Transcervical Sterilization
 Q UESTIONS & ANSWERS A BOUT Transcervical Sterilization A New Choice in Permanent Birth Control Choosing a Birth Control Method Women and their partners now have more birth control choices than ever. How
Q UESTIONS & ANSWERS A BOUT Transcervical Sterilization A New Choice in Permanent Birth Control Choosing a Birth Control Method Women and their partners now have more birth control choices than ever. How
Work Loss Associated With Increased Menstrual Loss in the United States
 Work Loss Associated With Increased Menstrual Loss in the United States Isabelle Côté, PhD, Philip Jacobs, DPhil, CMA, and David Cumming, MBChB OBJECTIVE: To estimate the effect of increased menstrual
Work Loss Associated With Increased Menstrual Loss in the United States Isabelle Côté, PhD, Philip Jacobs, DPhil, CMA, and David Cumming, MBChB OBJECTIVE: To estimate the effect of increased menstrual
An Overview of Long Acting Reversible Contraception Methods
 An Overview of Long Acting Reversible Contraception Methods Unintended Pregnancy All pregnancies should be intended; that is, they should be consciously and clearly desired at the time of conception. -
An Overview of Long Acting Reversible Contraception Methods Unintended Pregnancy All pregnancies should be intended; that is, they should be consciously and clearly desired at the time of conception. -
Lindsey Tingen, MD Department of Obstetrics and Gynecology, Greenville Health System Greenville, SC
 Postpartum IUD Insertion: Continued Usage at Six Months Based on Expulsion and Removal Rates at Greenville Memorial Hospital in the First Year After Adoption of the Practice Lindsey Tingen, MD Department
Postpartum IUD Insertion: Continued Usage at Six Months Based on Expulsion and Removal Rates at Greenville Memorial Hospital in the First Year After Adoption of the Practice Lindsey Tingen, MD Department
The number of women using long-acting reversible
 Long-acting reversible contraception: Who, what, when, and how This review provides practical tips and dispels some common misconceptions about these devices, which have higher rates of patient satisfaction
Long-acting reversible contraception: Who, what, when, and how This review provides practical tips and dispels some common misconceptions about these devices, which have higher rates of patient satisfaction
CONSORT 2010 Statement Annals Internal Medicine, 24 March History of CONSORT. CONSORT-Statement. Ji-Qian Fang. Inadequate reporting damages RCT
 CONSORT-Statement Guideline for Reporting Clinical Trial Ji-Qian Fang School of Public Health Sun Yat-Sen University Inadequate reporting damages RCT The whole of medicine depends on the transparent reporting
CONSORT-Statement Guideline for Reporting Clinical Trial Ji-Qian Fang School of Public Health Sun Yat-Sen University Inadequate reporting damages RCT The whole of medicine depends on the transparent reporting
Women with epilepsy initiating a progestin IUD: A prospective pilot study of safety and acceptability
 FULL-LENGTH ORIGINAL RESEARCH Women with epilepsy initiating a progestin IUD: A prospective pilot study of safety and acceptability *Anne R. Davis, Heva J. Saadatmand, and Alison Pack SUMMARY Dr. Anne
FULL-LENGTH ORIGINAL RESEARCH Women with epilepsy initiating a progestin IUD: A prospective pilot study of safety and acceptability *Anne R. Davis, Heva J. Saadatmand, and Alison Pack SUMMARY Dr. Anne
Disclosures. Learning Objectives 4/18/2017 ADOLESCENT CONTRACEPTION UPDATE APRIL 28, Nexplanon trainer for Merck
 ADOLESCENT CONTRACEPTION UPDATE APRIL 28, 2017 Brandy Mitchell, MN, RN, ANP BC, WHNP BC University of Iowa Hospitals and Clinics Obstetrics and Gynecology Iowa Association of Nurse Practitioners Spring
ADOLESCENT CONTRACEPTION UPDATE APRIL 28, 2017 Brandy Mitchell, MN, RN, ANP BC, WHNP BC University of Iowa Hospitals and Clinics Obstetrics and Gynecology Iowa Association of Nurse Practitioners Spring
Management of Emergency Contraception (EC)
 DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) Management of Emergency Contraception (EC) The risks and benefits of an IUD or oral EC should be discussed and documented (see appendix). Reasonable measures
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) Management of Emergency Contraception (EC) The risks and benefits of an IUD or oral EC should be discussed and documented (see appendix). Reasonable measures
1. Attia AM et al. Role of the levonorgestrel intrauterine system in effective contraception. Patient Prefer Adherence 2013; 7:
 1 2 1. Attia AM et al. Role of the levonorgestrel intrauterine system in effective contraception. Patient Prefer Adherence 2013; 7: 777 85. 3 1. Wu JP et al. Extended use of the intrauterine device: a
1 2 1. Attia AM et al. Role of the levonorgestrel intrauterine system in effective contraception. Patient Prefer Adherence 2013; 7: 777 85. 3 1. Wu JP et al. Extended use of the intrauterine device: a
Relative contributions of anovulation and luteal phase defect to the reduced pregnancy rate of breastfeeding women*
 - FERTILITY AND STERILITY Copyright c() 1992 The American Fertility Society Printed on acid-free paper in U.S.A. Relative contributions of anovulation and luteal phase defect to the reduced pregnancy rate
- FERTILITY AND STERILITY Copyright c() 1992 The American Fertility Society Printed on acid-free paper in U.S.A. Relative contributions of anovulation and luteal phase defect to the reduced pregnancy rate
Family Planning UNMET NEED. The Nurse Mildred Radio Talk Shows
 Family Planning UNMET NEED The Nurse Mildred Radio Talk Shows TOPIC 9: IUD/COIL Guests FP counsellor from MSU, RHU& UHMG Nurse Mildred Nurse Betty Objectives of the programme: To inform listeners about
Family Planning UNMET NEED The Nurse Mildred Radio Talk Shows TOPIC 9: IUD/COIL Guests FP counsellor from MSU, RHU& UHMG Nurse Mildred Nurse Betty Objectives of the programme: To inform listeners about
Postpartum intrauterine device placement: a patient-friendly option
 Cwiak and Cordes Contraception and Reproductive Medicine (2018) 3:3 https://doi.org/10.1186/s40834-018-0057-x Contraception and Reproductive Medicine REVIEW Postpartum intrauterine device placement: a
Cwiak and Cordes Contraception and Reproductive Medicine (2018) 3:3 https://doi.org/10.1186/s40834-018-0057-x Contraception and Reproductive Medicine REVIEW Postpartum intrauterine device placement: a
Contraceptive Updates and Recommendations
 Contraceptive Updates and Recommendations Emily M. Godfrey, MD MPH Associate Professor, Departments of Family Medicine and Obstetrics and Gynecology, University of Washington, Seattle WA Guest Researcher,
Contraceptive Updates and Recommendations Emily M. Godfrey, MD MPH Associate Professor, Departments of Family Medicine and Obstetrics and Gynecology, University of Washington, Seattle WA Guest Researcher,
Contraceptive Updates and Recommendations
 Contraceptive Updates and Recommendations Emily M. Godfrey, MD MPH Associate Professor, Departments of Family Medicine and Obstetrics and Gynecology, University of Washington, Seattle WA Guest Researcher,
Contraceptive Updates and Recommendations Emily M. Godfrey, MD MPH Associate Professor, Departments of Family Medicine and Obstetrics and Gynecology, University of Washington, Seattle WA Guest Researcher,
Unintended pregnancies are a difficult public health problem
 The new england journal of medicine Clinical Practice Caren G. Solomon, M.D., M.P.H., Editor Long-Acting Reversible Contraception Kathryn M. Curtis, Ph.D., and Jeffrey F. Peipert, M.D., Ph.D. This Journal
The new england journal of medicine Clinical Practice Caren G. Solomon, M.D., M.P.H., Editor Long-Acting Reversible Contraception Kathryn M. Curtis, Ph.D., and Jeffrey F. Peipert, M.D., Ph.D. This Journal
Medical Eligibility for Contraception Use
 Medical Eligibility for Contraception Use DIVISION OF REPRODUCTIVE HEALTH CENTERS FOR DISEASE CONTROL AND PREVENTION 2016 US Medical Eligibility Criteria for Contraceptive Use (US MEC) Purpose To assist
Medical Eligibility for Contraception Use DIVISION OF REPRODUCTIVE HEALTH CENTERS FOR DISEASE CONTROL AND PREVENTION 2016 US Medical Eligibility Criteria for Contraceptive Use (US MEC) Purpose To assist
Notes to Teacher continued Contraceptive Considerations
 Abstinence a conscious decision to refrain from sexual intercourse 100% pregnancy will not occur if close contact between the penis and vagina does not take place. The risk of a number of STDs, including
Abstinence a conscious decision to refrain from sexual intercourse 100% pregnancy will not occur if close contact between the penis and vagina does not take place. The risk of a number of STDs, including
Immediate Postpartum Long-Term Reversible Contraception (LARC) Bethany Berry, CNM, MSN and Alyssa Givens, MSN, RN
 Immediate Postpartum Long-Term Reversible Contraception (LARC) Bethany Berry, CNM, MSN and Alyssa Givens, MSN, RN Disclosures O Bethany Berry CNM is a Nexplanon trainer with Merck O Alyssa Givens, RN has
Immediate Postpartum Long-Term Reversible Contraception (LARC) Bethany Berry, CNM, MSN and Alyssa Givens, MSN, RN Disclosures O Bethany Berry CNM is a Nexplanon trainer with Merck O Alyssa Givens, RN has
FACT SHEET: Copper IUD
 FACT SHEET: Copper IUD How does the copper IUD help me? The copper IUD is a hormone-free option. If you don t like the copper IUD or you decide to get pregnant, your health care provider can remove the
FACT SHEET: Copper IUD How does the copper IUD help me? The copper IUD is a hormone-free option. If you don t like the copper IUD or you decide to get pregnant, your health care provider can remove the
Cancer Risks of Ovulation Induction
 Cancer Risks of Ovulation Induction 5th World Congress on Ovulation Induction September 13-15, 2007 Louise A. Brinton, Ph.D. National Cancer Institute Rockville, Maryland, USA Ovulation Induction and Cancer
Cancer Risks of Ovulation Induction 5th World Congress on Ovulation Induction September 13-15, 2007 Louise A. Brinton, Ph.D. National Cancer Institute Rockville, Maryland, USA Ovulation Induction and Cancer
THE INTRA--UTERINE DEVICE
 CANADIAN CONSENSUS CONFERENCE THE INTRA--UTERINE DEVICE WHY CONSIDER THIS METHOD? Use of an IUD should be considered for all women who seek a reversible, effective, coitally-independent method of contraception.
CANADIAN CONSENSUS CONFERENCE THE INTRA--UTERINE DEVICE WHY CONSIDER THIS METHOD? Use of an IUD should be considered for all women who seek a reversible, effective, coitally-independent method of contraception.
NAVY AND MARINE CORPS PUBLIC HEALTH CENTER HEALTH ANALYSIS DEPARTMENT
 NAVY AND MARINE CORPS PUBLIC HEALTH CENTER HEALTH ANALYSIS DEPARTMENT 620 John Paul Jones Circle, Portsmouth, VA 23708 Phone: 757.953.0700 E-mail: health-analysis@nehc.mar.med.navy.mil Analysis of Long-Acting
NAVY AND MARINE CORPS PUBLIC HEALTH CENTER HEALTH ANALYSIS DEPARTMENT 620 John Paul Jones Circle, Portsmouth, VA 23708 Phone: 757.953.0700 E-mail: health-analysis@nehc.mar.med.navy.mil Analysis of Long-Acting
