(VanDongen & Peart, 1974) have been attributed to an increase in intracellular [Ca]
|
|
|
- Bertha Fisher
- 5 years ago
- Views:
Transcription
1 J. Phygiol. (1981), 315, pp With 4 text-ftgures Printed in Great Britain CALCIUM DEPENDENCY OF THE INHIBITORY EFFECT OF ANTIDIURETIC HORMONE ON IN VITRO RENIN SECRETION IN RATS BY PAUL C. CHURCHILL From the Department of Physiology, Wayne State University School of Medicine, Detroit, Michigan 48201, U.S.A. (Received 1 July 1980) SUMMARY 1. Synthetic arginine-vasopressin (ADH or antidiuretic hormone) inhibited renin secretary rate of rat renal cortical slices. The response was concentration-dependent and was maximal at 041 U. ml.-'. 2. Ca depletion (incubation of slices in medium containing Na2EGTA and no added CaCl2) stimulated renin secretion and eventually abolished the inhibitory effect of ADH. Both these effects were reversible. 3. The Ca antagonist D-600, at 0-5 pm, reversed the inhibitory effect of K depolarization on secretary rate but had no effect on secretary rate of non-depolarized slices. In the presence of 0 5 /M-D-600, ADH inhibited the secretary rate of either depolarized or non-depolarized slices. 4. These results confirm and extend previous observations suggesting that Ca plays an inhibitory coupling role in the control of renin secretion. Moreover they suggest that although Ca influx through voltage-sensitive Ca channels influences the secretary activity of juxtaglomerular cells, ADH activates an independent pathway for Ca mobilization. INTRODUCTION The granulated juxtaglomerular cells (JG cells), one of the three components of the renin-secreting juxtaglomerular apparatus, are considered to be the site of renin synthesis, storage and release (Davis & Freeman, 1976). It is believed that these secretary cells are derived from vascular smooth muscle (Barajas, 1979), and independently of whether or not the JG cells themselves are contractile, many agents and manipulations that affect the contractile state of vascular smooth muscle cells also affect the secretary activity of JG cells (Peart, 1978). For instance, it is well known that angiotensin II elicits contraction of vascular smooth muscle and inhibits the secretion of renin. Both effects appear to be receptor-mediated in that they are dose-dependent, have a plateau at high concentrations, and can be inhibited by angiotensin analogues such as saralasin; other analogues are agonists with respect to vascular smooth muscle contraction and the same ones inhibit the secretion of renin (VanDongen, Peart & Boyd, 1974; Freer, 1975; Bumpus, 1977; Naftilan & Oparil, 1978). Both the contraction (Goodman, 1978) and the inhibition of renin secretion (VanDongen & Peart, 1974) have been attributed to an increase in intracellular [Ca] /81/ $07.50 D 1981 The Physiological Society
2 22 P. C. CHURCHILL resulting from an influx of extracellular Ca and/or a mobilization of membrane-bound Ca. Neither angiotensin-induced vasoconstriction (Freer, 1975) nor angiotensininduced inhibition of renin secretion (Churchill, 1980) is blocked by 'specific' Ca antagonists such as verapamil and D-600 (methoxy verapamil), suggesting (Fleckenstein, 1977) that angiotensin-receptor interaction does not lead to a depolarizationinduced influx of extracellular Ca. Like angiotensin II, antidiuretic hormone (ADH) is both a vasoconstrictor and an inhibitor of the secretion of renin (Vander, 1968; Goodman, 1978). In the experiments described below, the inhibitory effect, and the Ca dependency of the inhibitory effect, of ADH on renin secretion from rat kidney slices were studied. METHODS Experimental procedures for studying renin secretion from rat kidney slices have been described previously (Churchill, 1979). Briefly, for each ofseveral experiments five adult male Sprague-Dawley rats were anaesthetized with ether and nephrectomized. The renal capsule was removed and four thin cortical slices cut from each kidney. The resulting forty slices were randomized and kept in incubation medium at room temperature for no longer than 20 min before beginning the incubations. Slices were placed in flasks (two slices per flask) each of which contained 10 ml. of medium which has been equilibrated previously at 37 0C with a 95%/5 % mixture of 02 and CO2. The flasks were stoppered, placed in an oscillating incubator at 37 'C, and gassed with the mixture throughout the incubation. Periodically, as indicated in the Results section, 200,l. samples of incubation medium were withdrawn and centrifuged at 4 'C. The supernatants were frozen until determination of renin activity. Following the incubations, the slices were dried and weighed. The composition of the incubation medium was: 125 mm-nacl, 19 mm-nahco3, 4 mm-kc1, 2-65 mm-cacl2, 1M2 mm-nah2po4, 0-8 mm-mgso4, and 0-2 g per 100 ml. each of glucose and bovine albumin (United States Biochemical Corp). Deviations from this composition are described in the Results section. Na and Ca salts of EGTA (ethyleneglycol-bis[beta-aminoethyl ether]n,n'- tetraacetic acid) and ADH (synthetic arginine-vasopressin, grade VII) were obtained from the Sigma Chemical Co. D-600 (methoxy verapamil) was obtained from the Knoll Pharmaceutical Co. Methods for the determination of renin have been described previously (Churchill, 1979). Briefly, samples of supernatant were incubated with rat renin substrate at 37 'C for 30 min. Radioimmunoassay was used to measure the angiotensin I generated during the incubation. The renin activity of a sample was expressed in nanograms of angiotensin I generated per hour of incubation of the sample with renin substrate per millilitre of sample (ng hr-' ml.-'). Total renin secreted at a given time was calculated as the renin activity of the medium (ng hr-' ml.-') multiplied by the volume of the incubation medium (ml.) and divided by the tissue dry weight (mg), yielding the units ng hr-' mg-'. Renin secretary rate was calculated as the increment in the total amount of renin during a given interval of incubation of the slices, e.g. ng hr-' mg-' per 30 min. Results are presented as means +S.E.M.8. The paired and unpaired t tests were used to assess the statistical significance of observed changes and differences, respectively. RESULTS Dose-response relationship In the first series of experiments, slices were incubated in media containing 0-1 U. ml.-' ADH. The first 30 min of incubation were allowed for stabilization, and the increments in renin during the min period of incubation were taken as secretary rates. In Fig. 1 mean secretary rates are plotted versus the logarithm of the ADH concentration. The shaded area at the top represents the mean+ S.E.M. of the secretary rate in the absence of ADH (control rate). ADH concentrations from to 1 U. ml.-' suppressed secretion (P < 0-05 maximum). The response appeared
3 Ca, ADH AND RENIN SECRETION 23 to be maximal at 04 U. ml.-', as rates in media containing 0 1, 0 5 and 1 U. ml. were not significantly different from each other (p > 0-6). Rates in media containing these concentrations of ADH were , and % of the control rate, respectively. It is unlikely that this residual secretary rate can be accounted for by passive leak of renin from non-viable cells; as will be shown below, the release of renin from rat kidney slices can be completely suppressed by depolarization. 300 r > C') ED EI CD cn 0 oa I - Control~rate±SE. - TaI I TI I,I I ADH (U. ml-') Fig. 1. The effect of ADH on renin secretin from rat renal cortical slices. Secretory rates were measured during the min period of incubation. Means + s.e.m.s; n = 6 for each concentration ofadh, except n = 12 at 0 1 U. ml.-'. The shaded area at the top represents the mean control rate + 1 s.e.m. (rate in the absence of ADH). Effect of Ca depletion Slices were incubated in medium containing 2 mm-na2egta and no added CaCl2. So that any effect could be attributed specifically to a reduction in [Ca] and not to the presence of EGTA, other slices were incubated in medium containing 2 mm- CaEGTA plus the usual concentration of CaCl2 (2 65 mm). ADH (0-1 U. ml.-', final concentration) was added at either 30, 60 or 90 min after beginning the incubations. Secretory rates of each slice were determined during a 20 min period just before the addition of ADH (initial rate) and during a 20 min period beginning just after the addition of ADH (final rate). Percentage of final/initial was calculated for each slice, and mean values versus time of ADH addition are plotted in Fig. 2. In medium containing CaCl2, percentage final/initial was significantly less than 100 (P < ), irrespective of the time of addition of ADH. Thus, the presence of EGTA did not prevent the inhibitory effect. In contrast, buffering extracellular [Ca] to a low value (approximately 10-8 M: Caldwell, 1970) prevented the inhibitory effect. Although ADH did not inhibit secretion when added at 30 min (percentage final/initial < 100: P < 0-001), the degree of inhibition was somewhat less than that found in medium with 2-5 mm-cacl2. The addition of ADH at either 60 or 90 min failed to inhibit secretion at all; indeed, percentage final/initial exceeded 100 (P < 0-005) when ADH was added at these times. In order to determine whether the effects of Ca depletion were reversible, slices were incubated in medium containing 1 mm-na2egta and no added CaCl2. After
4 24 P. C. CHURCHILL ' 200 to E 100 C C~~~~~~ o L Time at which ADH was added (min) Fig. 2. The effect of Ca depletion on inhibition of renin secretion by ADH. ADH was added (0-1 U. ml.-', final concentration) either 30, 60 or 90 min after incubation had begun in medium with low [Ca] (Na2EGTA and no added CaCI2; 0) or normal [Ca] (CaEGTA plus 2-65 mm-cacl2; A). Initial and final secretary rates were determined during a 20 min period before ADH addition and during a 20 min period beginning at the time of ADH addition, respectively. Means +S.E.M.S of percentage final/initial are shown; n = 12 for each point in the figure CaC12 ADH + CaC12 Ca N200 CL - 00 Fig. 3. The reversibility of the effect on renin secretion of Ca depletion. Incubation of slices began in medium containing 1 mm-na2egta and no added CaCl2. At 40 min ADH and/or CaCJ2 were added (2 65 mm-cacl2, 0 1 U. ml.-' ADH, final concentrations). Each pair of columns represents secretary rate before and after these additions. Means + S.E.M.S; n = 6 for each experiment. 30 min either CaCl2 or CaCl2 plus ADH was added (2-65 mm-cacl2, 0.1 U. ml.-' ADH, final concentrations) and the incubations continued. Initial and final secretary rates were taken as the increments in renin during the and the min periods, allowing the first 10 min of incubation and the first 10 min after the additions of ADH and/or CaCl2 for stabilization. Means are shown in Fig. 3. The means of initial secretary rates in these two experiments were not significantly different from each other (P > 0 7). Combining the results, initial secretary rate averaged ng hr-' mg-' per 20 min. Comparison of this value with the control rate shown in
5 Ca, ADH AND RENIN SECRETION 25 Fig. 1 ( ng hr-1 mg-' per 30 min: note difference in units) suggests that Ca depletion increases renin secretion. Secretory rate declined following the addition of CaCl2 (P < 0-01) or CaCl2 plus ADH (P < 0-001). Final secretary rate was lower after the addition of CaCl2 plus ADH than after the addition of CaCl2 alone (P < 0 05). Thus, the addition of CaCl2 reverses the stimulatory effect on secretion of Ca depletion and restores the inhibitory effect of ADH. Previous studies have shown that secretary rate is stable if no changes are made in the composition of the medium during an incubation (Churchill, 1979; Churchill & Churchill, 1980a, b; Fig. 4 below). Therefore using each slice as its own control, as in the experiments shown in Figs. 2 and 3, is justified. Diluent 0-1 U. ml-' ADH 200 _ i -E 100 C I o WD K (mm) D-600 (pm) 9 E 0-5 pm -D pm-d -600 C r Diluent 10200T 100 T K (mm) ADH (U. ml.') Fig. 4. The effects on renin secretion of D-600 and ADH. Each pair of columns represents the initial and final secretary rates in a given experiment. Concentrations of substances present throughout the incubations are indicated under the various columns; substances added 40 min after the incubations had begun (between periods during which initial and final secretary rates were measured) are indicated above. Means +S.E.M.S; n = 6 for each experiment shown. Effects of ADH and D-600 Several experiments were performed to determine whether D-600 antagonized the effect of ADH. First, slices were incubated for 80 min either in medium with [K] = 4 mm or in medium with [K] = 60 mm (54 mm-kcl was added to medium of otherwise normal composition). Secretory rates were determined during the and min periods. Results are shown in the upper and lower left-hand panels of Fig. 4. In neither of these experiments was there a significant difference between initial and final secretary rates (P > 0 9). Comparisons of rates in the upper versus the lower panels demonstrate that K depolarization almost abolishes renin secretion, as has been reported previously (Churchill, 1980). In the third experiment the 80 min incubation was carried out in medium with [K] = 60 mm, and D-600 was added at 40 min. Initial and final secretary rates are presented in the lower middle panel. The addition of D-600 stimulated secretion significantly (P < 0 001), and the final rate in this experiment was not significantly different from that shown in the upper left-hand panel (P > 0 8), demonstrating that the inhibitory effect of K depolarization is completely antagonized by 0 5,tM-D-600. The fourth experiment, the results of
6 26 P. C. CHURCHILL which are shown in the lower right-hand panel, was carried out in medium with [K] = 60 mm and 0-1 U. ml.-' of ADH. The addition of D-600 at 40 min (0-5gM, final concentration) increased the final secretary rate in comparison with the initial rate (P < 0-01). However, the final rate in this experiment was suppressed in comparison with final rates shown either in the upper left-hand (P < 0 005) or in the lower middle (P < 0-01) panels. Therefore, ADH suppresses renin secretion even after all the effects of depolarization have been blocked with D-600. The fifth and sixth experiments were carried out using medium with [K] = 4 mm and 0-5 /SM-D-600. Either diluent or ADH was added at 40 min. Initial and final secretary rates are shown in the upper middle and right-hand panels. It can be seen that 0 5 #um-d-600 had no significant stimulatory or inhibitory effects in medium with [K] = 4 mm (P > 0-2, comparing upper left-hand and middle panels), and nor did this concentrationn of D-600 prevent a decrease in renin secretion when ADH was added (P < 0-001, comparing initial and final secretary rates in upper right-hand panel). DISCUSSION The intravenous or the intrarenal-arterial administration of ADH inhibits renin secretion in dogs and rats (Bunag, Page & McCubbin, 1967; Vander, 1968; Churchill, Churchill & McDonald, 1973). Although it has been suggested that this effect is mediated by a direct action of ADH on JG cells, several factors complicate the interpretation of ADH-induced changes in renin secretion in vivo. ADH may cause changes in arterial blood pressure, in intrarenal vascular pressures, in intrarenal distribution of blood flow, and in the filtration, transport and excretion of electrolytes, all of which are believed to affect renin secretion, either directly or indirectly (Davis & Freeman, 1976). Only some of these factors were excluded by using a non-filtering kidney, perfused at a constant pressure, to demonstrate an inhibitory effect (Shade, Davis, Johnson, Gotshall & Spielman, 1973). Many more complicating factors are excluded in the kidney slice preparation and, to our knowledge, this is the first report ofan inhibitory effect ofadh in such a preparation. Over the range U. ml.-i synthetic arginine-vasopressin, renin secretion was inhibited in a concentrationdependent manner. Both the 'saturation' of the response at high concentrations and the sigmoid nature of the dose-response relationship are consistent with a receptormediated event (Goodman & Gilman, 1975). In dogs, ADH inhibition of renin secretion is probably an important control mechanism. Unstimulated endogenous plasma levels of ADH are of the order of 1-2 1zU. ml.-' (Tagawa, Vander, Bonjour & Malvin, 1971), and infusions of pitressin (a mixture of arginine and lysine-vasopressin), in amounts calculated to raise plasma ADH by as little as 1-5uU. ml.-', produce detectable inhibitory effects on renin secretion (Bunag, et al. 1967; Vander, 1968; Tagawa et al. 1971; Shade et al. 1973). It should be noted, however, that in all these experiments the basal level of renin secretion was elevated (due variously to anaesthesia, prior Na deprivation or depletion, reduced renal perfusion pressure) in order to facilitate the detection of an inhibitory effect. Although ADH does decrease plasma renin and inhibit renin secretion in rats, the physiological relevance of the effect may be questioned. Based on the observation that plasma renin is elevated in rats lacking ADH, Gutman & Benzakein (1971, 1974) have suggested that endogenous ADH in normal rats may
7 Ca, ADH AND RENIN SECRETION 27 be sufficiently high tonically to inhibit renin secretion. However, a concentration of ADH of 1 mu. ml.-' (which is three orders of magnitude greater than endogenous plasma levels in dogs) failed to inhibit basal secretary rate of rat kidney slices (Fig. 1) or of the isolated-perfused rat kidney (VanDongen, 1975). Moreover, the doses of ADH found to decrease plasma renin or renin secretary rate in intact rats (5 mu. ml.-' i.v.: Churchill et al. 1973; 0A4-1'0 U. I.M. or s.c.: Gutman & Benzakein 1971,1974) are orders of magnitude greater, on a body weight basis, than those found to decrease plasma renin or renin secretary rate in dogs ( mu. min-' i.v.: Tagawa et al. 1971). Possibly only pharmacological levels of ADH inhibit renin secretion in rats. The results of the present study are in accord with previous reports that chelation of extracellular Ca with EGTA stimulates renin secretion from rat kidney slices (Churchill, 1979; Churchill & Churchill, 1980a, b). A stimulatory effect of Ca chelation has also been found on renin release from the isolated-perfused rat kidney (VanDongen & Peart, 1974; VanDongen, 1975), from isolated superfused rat glomeruli (Baumbach & Leyssac, 1977), and from pig kidney slices (Park & Malvin, 1978). It could be argued that Ca chelation enhances, not the secretion, but the passive leak, of renin from damaged cells. However, in previous experiments (Churchill, 1979; Churchill & Churchill, 1980 a, b) and in the experiments shown in Fig. 3, increasing extracellular concentration of free Ca from 10-8 to 10-3 M decreased renin _ secretion. Moreover, abruptly changing extracellular [Ca] from 10-8 to 10-3 M - or from 10-3 to 10-8 M produced opposite effects on renin secretion but did not - - alter the rate of accumulation of lactate dehydrogenase in the incubation medium (Churchill, 1979). Presumably, a generalized increase in the permeability of cell membranes would have been reflected by an increased leak of lactate dehydrogenase, an intracellular protein. Influx and/or mobilization of Ca appears to be a prerequisite for the inhibitory effect of ADH on renin secretion, as buffering extracellular [Ca] to a low value with EGTA not only stimulated the basal secretary rate but eventually abolished the inhibitory effect of ADH. The time course of this effect was similar to that found previously: Ca chelation only gradually blocked the inhibitory effect on renin secretion of angiotensin II (Churchill, 1980). Such a time course is not unexpected, as the addition of chelating agents to extracellular fluid only gradually reduces the amount of Ca which is available for influx and/or mobilization (Weiss, 1978). Smooth muscle cells and many secretary cells are stimulated to contract/secrete when depolarized by high extracellular [K], and it is well known that these responses are dependent upon an influx of extracellular Ca (Rubin, 1974; Fleckenstein, 1977; Rosenberger & Triggle, 1978). It is believed that K depolarization opens voltagesensitive Ca channels through which influx occurs; influx is blocked by drugs such as verapamil and D-600, and because of the apparent specificity of this effect the blocking of Ca-independent activities by verapamil or D-600 is taken as evidence for the existence of voltage-sensitive Ca channels (Fleckenstein, 1977; Rosenberger & Triggle, 1978). One might postulate the existence ofjg cell voltage-sensitive channels on the basis of the secretary activity of these cells and on the basis of their derivation from vascular smooth muscle (Barajas, 1979). As shown in Fig. 4 above, and as has been shown previously (Fray, 1978; Park & Malvin, 1978; Churchill, 1979), K depolarization blocks renin secretion. The inhibitory effect can in turn be blocked
8 28 P. C. CHURCHILL either by Ca chelation (Park & Malvin, 1978) or by D-600, in a concentrationdependent manner (Churchill, 1980). At 0-5,#M, D-600 restored the secretary rate of depolarized slices to the level found in non-depolarized slices. This same concentration had no effect on the secretary rate of non-depolarized slices. Since ADH decreased the secretary rate in the presence of 0 5/tM-D-600, whether or not the slices were depolarized, it is concluded that ADH does not act by depolarization of the cell membrane followed by Ca influx through voltage-sensitive Ca channels. The possibility that cyclic 3':5'-adenosine monophosphate (camp) plays a role should be mentioned. The second-messenger role of camp in the ADH responses of several transporting epithelia (including mammalian renal tubular cells) is well established: ADH-sensitive adenylate cyclase has been demonstrated, tissue levels of camp rise in response to ADH, and both exogenous camp and phosphodiesterase inhibitors mimic and/or potentiate the effect of ADH (reviewed by Dousa & Valtin, 1976). On the other hand, there are grounds for believing that camp does not mediate other effects ofadh, in particular the effects on vascular smooth muscle contractility and on the secretary activity of JG cells, which are derived from vascular smooth muscle (Barajas, 1979). The vascular effect is mediated by increased intracellular [Ca], whereas increased intracellular camp is usually associated with decreased intracellular [Ca] and relaxation of vascular smooth muscle. Similarly, the inhibitory effect on secretary activity appears to be mediated by increased intracellular [Ca] also, whereas increased intracellular camp is thought to stimulate renin secretion, although the evidence is less than compelling at the present time (reviewed by Oparil, 1976): beta-adrenergic agonists stimulate secretion, presumably by activating adenylate cyclase, although neither a hormone-sensitive adenylate cyclase associated with JG cell membranes nor an increase in JG levels of camp in response to catecholamines has been demonstrated. Exogenous camp and phosphodiesterase inhibitors mimicked or potentiated the stimulatory effects of beta-adrenergic agonists in some studies, but exactly the opposite effects were found in other studies. In conclusion, ADH may be added to a growing list of substances which inhibit the secretion of renin, most probably by increasing intracellular [Ca]. It has been shown that extracellular Ca is required for the inhibitory effects of angiotensin II (VanDongen & Peart, 1974; Churchill, 1980), of ouabain (Park & Malvin, 1978; Churchill, 1979), of vanadate (Churchill & Churchill, 1980b), of low extracellular [K] (Churchill & Churchill, 1980a) and of high extracellular [K] (Park & Malvin, 1978; Churchill, 1980). It is not yet clear whether all the substances which stimulate renin secretion act by decreasing intracellular [Ca], but there is evidence that stimulatory effects can be blocked by agents believed to increase intracellular [Ca]. For example, the stimulatory effect of beta-adrenergic agonists can be blocked by angiotensin II (VanDongen & Peart, 1974; Capponi, Gourjon & Vallotton, 1977), by ADH (VanDongen, 1975), by ouabain or vanadate (Churchill & Churchill, 1980b) and by low or high extracellular [K] (Churchill & Churchill, 1980c). Thus, it would appear that intracellular [Ca] plays an inhibitory coupling role in the control of renin secretion, which is opposite to its role in most other secretary cells (Rubin, 1974). The author wishes to thank M. C. Churchill for expert technical assistance and Professor Dr Oberdorf, of the Knoll Pharmaceutical Company, for a generous supply of D-600. This research was supported by the National Institutes of Health (grant HL ).
9 Ca, ADH AND RENIN SECRETION 29 REFERENCES BARAJAS, L. (1979). Anatomy of the juxtaglomerula apparatus. Am. J. Physiol. 237, F333-F343. BAUMBACH, L. & LEYSSAC, P. P. (1977). Studies on the mechanism of renin release from isolated superfused rat glomeruli: effects of calcium, calcium ionophore and lanthanum. J. Physiol. 273, BUMPUS, F. M. (1977). Mechanisms and sites ofaction ofnewer angiotensin agonists and antagonists in terms of activity and receptor. Fedn Proc. 36, BUNAG, R. D., PAGE, I. H. & MCCUBBIN, J. W. (1967). Inhibition of renin release by vasopressin and angiotensin. Cardiova8c. Res. 1, CALDWELL, P. C. (1970). Calcium chelation and buffers. In Calcium and Cellular Function, ed. CUTHBERT, A. W., pp New York: St Martins Press. CAPPONI, A. M., GOURJON, M. & VALLOTTON, M. D. (1977). Effect of f-blocking agents and angiotensin II on isoproterenol-stimulated renin release from rat kidney slices. Circulation Res. 40, CHURCHILL, M. C. & CHURCHILL, P. C. (1980a). Separate and combined effects of ouabain and extracellular potassium on renin secretion from rat renal cortical slices. J. Phy8iol. 300, CHURCHILL, P. C. (1979). Possible mechanism of the inhibitory effect of ouabain on renin secretion from rat renal cortical slices. J. Phy8iol. 294, CHURCHILL, P. C. (1980). Effect of D-600 on inhibition of in vitro renin release in the rat by high extracellular potassium and angiotensin II. J. Physiol. 304, CHURCHILL, P. C. & CHURCHILL, M. C. (1980b). Vanadate inhibits renin secretion from rat kidney slices. J. Pharmac. exp. Ther. 213, CHURCHILL, P. C. & CHURCHILL, M. C. (1980c). Biphasic effect of extracellular K on isoproterenolstimulated renin secretion from rat kidney slices. J. Pharmac. exp. Ther. 214, CHURCHILL, P. C., CHURCHILL, M. C. & MCDONALD, D. D. (1973). Renin release in anaesthetized rats. Kidney Int. 4, DAVIS, J. 0. & FREEMAN, R. H. (1976). Mechanisms regulating renin release. Physiol. Rev. 56, DOUSA, T. P. & VALTIN, H. (1976). Cellular actions of vasopressin in the mammalian kidney. Kidney Int. 10, FLECKENSTEIN, A. (1977). Specific pharmacology of calcium in myocardium, cardiac pacemakers, and vascular smooth muscle. A. Rev. Pharmac. Tox. 17, FRAY, J. C. S. (1978). Stretch receptor control of renin release in perfused rat kidney: effect of high perfusate potassium. J. Physiol. 282, FREER, R. J. (1975). Calcium and angiotensin tachyphylaxis in rat uterine smooth muscle. Am. J. Physiol. 228, GOODMAN, F. R. (1978). Calcium related basis of action of vascular agents: cellular approaches. In Calcium and Drug Action, ed. WEISS, G. B., pp New York: Plenum Press. GOODMAN, L. W. & GILMAN, A. (1975). The Pharmacological Basis of Therapeutics, 5th edn. New York: Macmillan. GUTMAN, Y. & BENZAKEIN, F. (1971). Effect of an increase and of lack of antidiuretic hormone on plasma renin activity in the rat. Life Sci. Oxford 10, GUTMAN, Y. & BENZAKEIN, F. (1974). Antidiuretic hormone and renin in rats with diabetes insipidus. Eur. J. Pharmac. 28, NAFTILAN, A. J. & OPARIL, S. (1978). Inhibition of renin release from rat kidney slices by the angiotensins. Am. J. Physiol 235, F62-F68. OPARIL, S. (1976). Renin 1976: Annual Research Reviews. Montreal: Eden Press. PARK, C. S. & MALVIN, R. L. (1978). Calcium in the control of renin release. Am. J. Physiol. 235, F22-F25. PEART, W. S. (1978). Intra-renal factors in renin release. Contr. Nephrol. 12, ROSENBERGER, L. & TRIGGLE, D. J. (1978). Calcium, calcium translocation, and specific calcium antagonists. In Calcium and Drug Action, ed. WEISS, G. B., pp New York: Plenum Press. RUBIN, R. P. (1974). Calcium and the Secretory Process. New York: Plenum Press. SHADE, R. E., DAVIS, J. O., JOHNSON, J. A., GOTSHALL, R. W. & SPIELMAN, W. S. (1973). Mechanism of action of angiotensin II and antidiuretic hormone on renin secretion. Am. J. Physiol. 224, TAGAWA, H., VANDER, A. J., BONJOUR, J.-P. & MALVIN, R. L. (1971). Inhibition of renin secretion by vasopressin in unanesthetized sodium-deprived dogs. Am. J. Physiol. 220,
10 30 P. C. CHURCHILL VANDER, A. J. (1968). Inhibition of renin release in the dog by vasopressin and vasotocin. Circulation Res. 23, VANDER, A. J. & GEELHOED, G. W. (1965). Inhibition of renin secretion by angiotensin II. Proc. Soc. exp. Biol. Med. 120, VANDoNGEN, R. (1975). Inhibition of renin secretion in the isolated rat kidney by antidiuretic hormone. Clin. Sci. 49, VANDONGEN, R. & PEART, W. S. (1974). Calcium dependence of the inhibitory effect of angiotensin on renin secretion in the isolated perfused kidney of the rat. Br. J. Pharmac. 50, VANDONGEN, R., PEART, W. S. & BOYD, G. W. (1974). Effect of angiotensin II and its nonpressor derivatives on renin secretion. Am. J. Phyaiol. 226, WEIss, G. B. (1978). Quantitative measurement of binding sites and washout components for calcium ion in vascular smooth muscle. In Calcium and Drug Action, ed. WEISs, G. E., pp New York: Plenum Press.
hyperalbuminaemia. However, other mechanisms may explain the hyperalbuminaemia-induced
 J. Physaol. (1980), 299, pp. 45-54 45 With 3 text-figurew Printed in Great Britain INFLUENCE OF RAISING ALBUMIN CONCENTRATION ON RENIN RELEASE IN ISOLATED PERFUSED RAT KIDNEYS BY JOHN C. S. FRAY AND ANTHONY
J. Physaol. (1980), 299, pp. 45-54 45 With 3 text-figurew Printed in Great Britain INFLUENCE OF RAISING ALBUMIN CONCENTRATION ON RENIN RELEASE IN ISOLATED PERFUSED RAT KIDNEYS BY JOHN C. S. FRAY AND ANTHONY
marked secretion ofcatecholamines and a subsequent inhibition ofsecretion although the basal secretion shows an initial rise.
 J. Physiol. (1969), 2, pp. 797-85 797 With 7 text-ftgurem Printed in Great Britain SODIUM IONS AND THE SECRETION OF CATECHOLAMINES By P. BANKS, ROSEMARY BIGGINS, R. BISHOP, B. CHRISTIAN AND N. CURRIE From
J. Physiol. (1969), 2, pp. 797-85 797 With 7 text-ftgurem Printed in Great Britain SODIUM IONS AND THE SECRETION OF CATECHOLAMINES By P. BANKS, ROSEMARY BIGGINS, R. BISHOP, B. CHRISTIAN AND N. CURRIE From
Relaxation responses of aortic rings from salt-loaded high calcium fed rats to potassium chloride, calcium chloride and magnesium sulphate
 Pathophysiology 4 (1998) 275 280 Relaxation responses of aortic rings from salt-loaded high calcium fed rats to potassium chloride, calcium chloride and magnesium sulphate B.J. Adegunloye, O.A. Sofola
Pathophysiology 4 (1998) 275 280 Relaxation responses of aortic rings from salt-loaded high calcium fed rats to potassium chloride, calcium chloride and magnesium sulphate B.J. Adegunloye, O.A. Sofola
High Ca Content of Pacemaker Tissues in the Frog Heart
 Short Communication Japanese Journal of Physiology, 34, 1117-1121,1984 High Ca Content of Pacemaker Tissues in the Frog Heart Yasuichiro FUKUDA Department of Physiology II, School of Medicine, Chiba University,
Short Communication Japanese Journal of Physiology, 34, 1117-1121,1984 High Ca Content of Pacemaker Tissues in the Frog Heart Yasuichiro FUKUDA Department of Physiology II, School of Medicine, Chiba University,
Regulation of fluid and electrolytes balance
 Regulation of fluid and electrolytes balance Three Compartment Fluid Compartments Intracellular = Cytoplasmic (inside cells) Extracellular compartment is subdivided into Interstitial = Intercellular +
Regulation of fluid and electrolytes balance Three Compartment Fluid Compartments Intracellular = Cytoplasmic (inside cells) Extracellular compartment is subdivided into Interstitial = Intercellular +
hyperpolarization (4-6 mv). The effect of isoprenaline, but not that of hyperpolarization of 4-8 mv.
 J. Physiol. (1974), 239, pp. 647-656 647 With 4 text-figures Printed in Great Britain THE EFFECT OF GLUCAGON ON THE LIVER CELL MEMBRANE POTENTIAL BY 0. H. PETERSEN From the Institute of Medical Physiology
J. Physiol. (1974), 239, pp. 647-656 647 With 4 text-figures Printed in Great Britain THE EFFECT OF GLUCAGON ON THE LIVER CELL MEMBRANE POTENTIAL BY 0. H. PETERSEN From the Institute of Medical Physiology
Urinary System Organization. Urinary System Organization. The Kidneys. The Components of the Urinary System
 Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
possibility that the kidney which is known to have a 'growth' inhibitor factor (Dicker,
 J. Physiol. (1980), 301, pp. 1-5 1 Printed in Great Britain ORIGIN OF THE HUMORAL FACTOR RESPONSIBLE FOR COMPENSATORY RENAL HYPERTROPHY BY S. E. DICKER AND CHRISTINE A. MORRIS From the Department of Chemistry,
J. Physiol. (1980), 301, pp. 1-5 1 Printed in Great Britain ORIGIN OF THE HUMORAL FACTOR RESPONSIBLE FOR COMPENSATORY RENAL HYPERTROPHY BY S. E. DICKER AND CHRISTINE A. MORRIS From the Department of Chemistry,
Diuretic Agents Part-2. Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia
 Diuretic Agents Part-2 Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Potassium-sparing diuretics The Ion transport pathways across the luminal and basolateral
Diuretic Agents Part-2 Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Potassium-sparing diuretics The Ion transport pathways across the luminal and basolateral
Renal Quiz - June 22, 21001
 Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
BIPN100 F15 Human Physiology (Kristan) Lecture 18: Endocrine control of renal function. p. 1
 BIPN100 F15 Human Physiology (Kristan) Lecture 18: Endocrine control of renal function. p. 1 Terms you should understand by the end of this section: diuresis, antidiuresis, osmoreceptors, atrial stretch
BIPN100 F15 Human Physiology (Kristan) Lecture 18: Endocrine control of renal function. p. 1 Terms you should understand by the end of this section: diuresis, antidiuresis, osmoreceptors, atrial stretch
Nephron Anatomy Nephron Anatomy
 Kidney Functions: (Eckert 14-17) Mammalian Kidney -Paired -1% body mass -20% blood flow (Eckert 14-17) -Osmoregulation -Blood volume regulation -Maintain proper ion concentrations -Dispose of metabolic
Kidney Functions: (Eckert 14-17) Mammalian Kidney -Paired -1% body mass -20% blood flow (Eckert 14-17) -Osmoregulation -Blood volume regulation -Maintain proper ion concentrations -Dispose of metabolic
014 Chapter 14 Created: 9:25:14 PM CST
 014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
BIOLOGY - CLUTCH CH.44 - OSMOREGULATION AND EXCRETION.
 !! www.clutchprep.com Osmoregulation regulation of solute balance and water loss to maintain homeostasis of water content Excretion process of eliminating waste from the body, like nitrogenous waste Kidney
!! www.clutchprep.com Osmoregulation regulation of solute balance and water loss to maintain homeostasis of water content Excretion process of eliminating waste from the body, like nitrogenous waste Kidney
P215 Spring 2018: Renal Physiology Chapter 18: pp , Chapter 19: pp ,
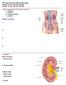 P215 Spring 2018: Renal Physiology Chapter 18: pp. 504-520, 525-527 Chapter 19: pp. 532-548, 553-560 I. Main Components of the Renal System 1. kidneys 2. ureters 3. urinary bladder 4. urethra 4 Major Functions
P215 Spring 2018: Renal Physiology Chapter 18: pp. 504-520, 525-527 Chapter 19: pp. 532-548, 553-560 I. Main Components of the Renal System 1. kidneys 2. ureters 3. urinary bladder 4. urethra 4 Major Functions
INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT
 Brit. J. Phawmacol. (1951), 6, 289. INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT BY From the Pharmacological Laboratory, University of St. Andrews, Medical School, Dundee (Received February 2, 1951)
Brit. J. Phawmacol. (1951), 6, 289. INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT BY From the Pharmacological Laboratory, University of St. Andrews, Medical School, Dundee (Received February 2, 1951)
Physio 12 -Summer 02 - Renal Physiology - Page 1
 Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
The principal functions of the kidneys
 Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Renal Physiology Part II. Bio 219 Napa Valley College Dr. Adam Ross
 Renal Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Fluid and Electrolyte balance As we know from our previous studies: Water and ions need to be balanced in order to maintain proper homeostatic
Renal Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Fluid and Electrolyte balance As we know from our previous studies: Water and ions need to be balanced in order to maintain proper homeostatic
Actions of prostaglandin F20 on the splenic vascular and capsular smooth muscle in the dog
 Br. J. Pharmac. (1971), 41, 1-7 Actions of prostaglandin F20 on the splenic vascular and capsular smooth muscle in the dog B. N. DAVIES ADi P. G. WITHRINGTON Department of Physiology, Medical College of
Br. J. Pharmac. (1971), 41, 1-7 Actions of prostaglandin F20 on the splenic vascular and capsular smooth muscle in the dog B. N. DAVIES ADi P. G. WITHRINGTON Department of Physiology, Medical College of
The Urinary System 15PART B. PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
THE EFFECTS OF ION CHANGES ON THE CONTRACTION OF THE RAT UTERUS STIMULATED BY OXYTOCIN
 Brit. J. Pharmacol. (1961), 16, 45-49. THE EFFECTS OF ION CHANGES ON THE CONTRACTION OF THE RAT UTERUS STIMULATED BY OXYTOCIN BY P. J. BENTLEY AND ELEANOR McEWEN From the Department of Physiology, The
Brit. J. Pharmacol. (1961), 16, 45-49. THE EFFECTS OF ION CHANGES ON THE CONTRACTION OF THE RAT UTERUS STIMULATED BY OXYTOCIN BY P. J. BENTLEY AND ELEANOR McEWEN From the Department of Physiology, The
Drug Receptor Interactions and Pharmacodynamics
 Drug Receptor Interactions and Pharmacodynamics Dr. Raz Mohammed MSc Pharmacology School of Pharmacy 22.10.2017 Lec 6 Pharmacodynamics definition Pharmacodynamics describes the actions of a drug on the
Drug Receptor Interactions and Pharmacodynamics Dr. Raz Mohammed MSc Pharmacology School of Pharmacy 22.10.2017 Lec 6 Pharmacodynamics definition Pharmacodynamics describes the actions of a drug on the
Urinary Physiology. Chapter 17 Outline. Kidney Function. Chapter 17
 Urinary Physiology Chapter 17 Chapter 17 Outline Structure and Function of the Kidney Glomerular Filtration Reabsorption of Salt and Water Renal Plasma Clearance Renal Control of Electrolyte and Acid-Base
Urinary Physiology Chapter 17 Chapter 17 Outline Structure and Function of the Kidney Glomerular Filtration Reabsorption of Salt and Water Renal Plasma Clearance Renal Control of Electrolyte and Acid-Base
Monday, 17 April 2017 BODY FLUID HOMEOSTASIS
 Monday, 17 April 2017 BODY FLUID HOMEOSTASIS Phenomenon: shipwrecked sailor on raft in ocean ("water, water everywhere but not a drop to drink") Why are the sailors thirsty? (What stimulated thirst?) Why
Monday, 17 April 2017 BODY FLUID HOMEOSTASIS Phenomenon: shipwrecked sailor on raft in ocean ("water, water everywhere but not a drop to drink") Why are the sailors thirsty? (What stimulated thirst?) Why
sought to obtain such evidence in an attempt to gain further insight into the secretary kidneys and isolated juxtaglomerular cells.
 J. Physiol. (1981), 320, pp. 3139 31 With 5 textfigures Printed in Great Britain MEHANISMS BY WHIH ALBUMIN STIMULATES RENIN SERETION IN ISOLATED KIDNEYS AND JUXTAGLOMERULAR ELLS BY J.. S. FRAY AND N. J.
J. Physiol. (1981), 320, pp. 3139 31 With 5 textfigures Printed in Great Britain MEHANISMS BY WHIH ALBUMIN STIMULATES RENIN SERETION IN ISOLATED KIDNEYS AND JUXTAGLOMERULAR ELLS BY J.. S. FRAY AND N. J.
BIPN 140 Problem Set 6
 BIPN 140 Problem Set 6 1) Hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
BIPN 140 Problem Set 6 1) Hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
(Received 23 January 1961) Crawford & Kennedy (1959) found the prolonged saluretic and diuretic
 454 J. Phyeiol. (1961), 157, pp. 454-461 With 3 text-figure Printed in Great Britain THE ACTION OF CHLOROTHIAZIDE IN THE PERFUSED CAT KIDNEY BY T. DE LIMA AND MARY F. LOCKETT From the Department of Physiology
454 J. Phyeiol. (1961), 157, pp. 454-461 With 3 text-figure Printed in Great Britain THE ACTION OF CHLOROTHIAZIDE IN THE PERFUSED CAT KIDNEY BY T. DE LIMA AND MARY F. LOCKETT From the Department of Physiology
Role of Ion Fluxes in Hydrogen Peroxide Pulmonary Vasoconstriction
 Physiol. Res. 44: 31-3 7, 1995 Role of Ion Fluxes in Hydrogen Peroxide Pulmonary Vasoconstriction J. W ILHELM, J. H E R G E T 1 Department of Medical Chemistry and Biochemistry and department of Physiology,
Physiol. Res. 44: 31-3 7, 1995 Role of Ion Fluxes in Hydrogen Peroxide Pulmonary Vasoconstriction J. W ILHELM, J. H E R G E T 1 Department of Medical Chemistry and Biochemistry and department of Physiology,
Receptors Families. Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia
 Receptors Families Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Receptor Families 1. Ligand-gated ion channels 2. G protein coupled receptors 3. Enzyme-linked
Receptors Families Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Receptor Families 1. Ligand-gated ion channels 2. G protein coupled receptors 3. Enzyme-linked
1. Urinary System, General
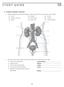 S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
Osmotic Regulation and the Urinary System. Chapter 50
 Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
One Minute Movies: Molecular Action at the Nephron Joy Killough / Westwood High School / Austin,TX
 One Minute Movies: Molecular Action at the Nephron Joy Killough / Westwood High School / Austin,TX To prepare your nephron model: ( A nephron is a tubule and the glomerulus. There are about a million of
One Minute Movies: Molecular Action at the Nephron Joy Killough / Westwood High School / Austin,TX To prepare your nephron model: ( A nephron is a tubule and the glomerulus. There are about a million of
(Snyder, Chang, Kuhar & Yamamura, 1975; Birdsall & Hulme, 1976). Thus, the
 J. Phyeiol. (1980), 299, pp. 521-531 521 With 5 text-figurea Printed in Great Britain THE RELATIONSHIP BETWEEN MUSCARINIC RECEPTOR BINDING AND ION MOVEMENTS IN RAT PAROTID CELLS BY JAMES W. PUTNEY, JR.*
J. Phyeiol. (1980), 299, pp. 521-531 521 With 5 text-figurea Printed in Great Britain THE RELATIONSHIP BETWEEN MUSCARINIC RECEPTOR BINDING AND ION MOVEMENTS IN RAT PAROTID CELLS BY JAMES W. PUTNEY, JR.*
Principles of Renal Physiology. 4th Edition
 Principles of Renal Physiology 4th Edition Principles of Renal Physiology 4th Edition Chris Lote Professor of Experimental Nephrology, University of Birmingham, UK SPRINGER SCIENCE+BUSINESS MEDIA, B.V.
Principles of Renal Physiology 4th Edition Principles of Renal Physiology 4th Edition Chris Lote Professor of Experimental Nephrology, University of Birmingham, UK SPRINGER SCIENCE+BUSINESS MEDIA, B.V.
A NEW TYPE OF DRUG ENHANCEMENT: INCREASED MAXIMUM RESPONSE TO CUMULATIVE NORADREN- ALINE IN THE ISOLATED RAT VAS DEFERENS
 Br. J. Pharmac. Chemother. (1968), 33, 171-176. A NEW TYPE OF DRUG ENHANCEMENT: NCREASED MAXMUM RESPONSE TO CUMULATVE NORADREN- ALNE N THE SOLATED RAT VAS DEFERENS BY A. BARNETT, D. D. GREENHOUSE AND R..
Br. J. Pharmac. Chemother. (1968), 33, 171-176. A NEW TYPE OF DRUG ENHANCEMENT: NCREASED MAXMUM RESPONSE TO CUMULATVE NORADREN- ALNE N THE SOLATED RAT VAS DEFERENS BY A. BARNETT, D. D. GREENHOUSE AND R..
Chapter 19 The Urinary System Fluid and Electrolyte Balance
 Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
MUSCLE TO ANGIOTENSIN 11 AFTER NEPHRECTOMY
 Br. J. Pharmac. (1974), 51, 435439 SPECIFIC SUPERSENSITIVITY OF SMOOTH MUSCLE TO ANGIOTENSIN 11 AFTER NEPHRECTOMY P. MEYER, A. PAPADIMITRIOU & M. WORCEL Physiologie et Pharmacologie, Inserm U7, Hopital
Br. J. Pharmac. (1974), 51, 435439 SPECIFIC SUPERSENSITIVITY OF SMOOTH MUSCLE TO ANGIOTENSIN 11 AFTER NEPHRECTOMY P. MEYER, A. PAPADIMITRIOU & M. WORCEL Physiologie et Pharmacologie, Inserm U7, Hopital
MAJOR FUNCTIONS OF THE KIDNEY
 MAJOR FUNCTIONS OF THE KIDNEY REGULATION OF BODY FLUID VOLUME REGULATION OF OSMOTIC BALANCE REGULATION OF ELECTROLYTE COMPOSITION REGULATION OF ACID-BASE BALANCE REGULATION OF BLOOD PRESSURE ERYTHROPOIESIS
MAJOR FUNCTIONS OF THE KIDNEY REGULATION OF BODY FLUID VOLUME REGULATION OF OSMOTIC BALANCE REGULATION OF ELECTROLYTE COMPOSITION REGULATION OF ACID-BASE BALANCE REGULATION OF BLOOD PRESSURE ERYTHROPOIESIS
BIPN 140 Problem Set 6
 BIPN 140 Problem Set 6 1) The hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
BIPN 140 Problem Set 6 1) The hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
2) This is a Point and Click question. You must click on the required structure.
 Class: A&P2-1 Description: Test: Excretory Test Points: 144 Test Number: 28379 Printed: 31-March-10 12:03 1) This is a Point and Click question. You must click on the required structure. Click on the Bowman's
Class: A&P2-1 Description: Test: Excretory Test Points: 144 Test Number: 28379 Printed: 31-March-10 12:03 1) This is a Point and Click question. You must click on the required structure. Click on the Bowman's
EFFECT OF POTASSIUM ON PLASMA RENIN CONCENTRATION IN THE PRESENCE AND ABSENCE OF ADH (BRATTLEBORO RAT MODEL)
 EFFECT OF POTASSIUM ON PLASMA RENIN CONCENTRATION IN THE PRESENCE AND ABSENCE OF ADH (BRATTLEBORO RAT MODEL) Emma Fernandez-Repollet, Susan Opava-Stitzer, and Manuel Martinez-Maldonado Department of Physiology
EFFECT OF POTASSIUM ON PLASMA RENIN CONCENTRATION IN THE PRESENCE AND ABSENCE OF ADH (BRATTLEBORO RAT MODEL) Emma Fernandez-Repollet, Susan Opava-Stitzer, and Manuel Martinez-Maldonado Department of Physiology
Answers and Explanations
 Answers and Explanations 1. The answer is D [V B 4 b]. Distal K + secretion is decreased by factors that decrease the driving force for passive diffusion of K + across the luminal membrane. Because spironolactone
Answers and Explanations 1. The answer is D [V B 4 b]. Distal K + secretion is decreased by factors that decrease the driving force for passive diffusion of K + across the luminal membrane. Because spironolactone
Direct Effect of Beta-Adrenergic Stimulation on Renin Release by the Rat Kidney Slice In Vitro
 Direct Effect of Beta-Adrenergic Stimulation on Renin Release by the Rat Kidney Slice In Vitro By Myron H. Weinberger, Wataru Aoi, and David P. Henry ABSTRACT Controversy exists regarding the mechanism
Direct Effect of Beta-Adrenergic Stimulation on Renin Release by the Rat Kidney Slice In Vitro By Myron H. Weinberger, Wataru Aoi, and David P. Henry ABSTRACT Controversy exists regarding the mechanism
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance
 Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
modulating the tubuloglomerular feed-back mechanism in the canine kidney; Sciences Center, 4200 East Ninth Avenue, Denver, CO 80262, U.S.A.
 J. Physiol. (1986), 380, pp. 35-43 35 With 3 text-figures Printed in Great Britain RENAL VASOCONSTRICTOR RESPONSE TO HYPERTONIC SALINE IN THE DOG: EFFECTS OF PROSTAGLANDINS, INDOMETHACIN AND THEOPHYLLINE
J. Physiol. (1986), 380, pp. 35-43 35 With 3 text-figures Printed in Great Britain RENAL VASOCONSTRICTOR RESPONSE TO HYPERTONIC SALINE IN THE DOG: EFFECTS OF PROSTAGLANDINS, INDOMETHACIN AND THEOPHYLLINE
IONS ON THE ANTIDIURETIC ACTION OF VASOPRESSIN IN THE RAT
 Br. J. Pharmac. (1975), 55, 527-534 THE EFFECTS OF LITHIUM IONS ON THE ANTIDIURETIC ACTION OF VASOPRESSIN IN THE RAT F.A. JENNER & SHEILA MACNEIL Medical Research Council Unit for Metabolic Studies in
Br. J. Pharmac. (1975), 55, 527-534 THE EFFECTS OF LITHIUM IONS ON THE ANTIDIURETIC ACTION OF VASOPRESSIN IN THE RAT F.A. JENNER & SHEILA MACNEIL Medical Research Council Unit for Metabolic Studies in
Regulation of Body Fluids: Na + and Water Linda Costanzo, Ph.D.
 Regulation of Body Fluids: Na + and Water Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Why body sodium content determines ECF volume and the relationships
Regulation of Body Fluids: Na + and Water Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Why body sodium content determines ECF volume and the relationships
Human TRPC6 Ion Channel Cell Line
 TECHNICAL DATA SHEET ValiScreen Ion Channel Cell Line Caution: For Laboratory Use. A research product for research purposes only Human TRPC6 Ion Channel Cell Line Product No.: AX-012-C Lot No.: 512-548-A
TECHNICAL DATA SHEET ValiScreen Ion Channel Cell Line Caution: For Laboratory Use. A research product for research purposes only Human TRPC6 Ion Channel Cell Line Product No.: AX-012-C Lot No.: 512-548-A
EFFECT OF MUSCARINE ON RELEASE OF CATECHOLAMINES
 Br. J. Pharmac. (1982), 77, 455-46 EFFECT OF MUSCARINE ON RELEASE OF CATECHOLAMINES FROM THE PERFUSED ADRENAL GLAND OF THE CAT S.M. KIRPEKAR, J.C. PRAT & M.T. SCHIAVONE Department of Pharmacology, Downstate
Br. J. Pharmac. (1982), 77, 455-46 EFFECT OF MUSCARINE ON RELEASE OF CATECHOLAMINES FROM THE PERFUSED ADRENAL GLAND OF THE CAT S.M. KIRPEKAR, J.C. PRAT & M.T. SCHIAVONE Department of Pharmacology, Downstate
Plasma Renin Activity and Renin-Substrate Concentration in Patients with Liver Disease
 Plasma Renin Activity and Renin-Substrate Concentration in Patients with Liver Disease By Carlos R. Ayers, M.D. ABSTRACT Peripheral venous renin activity was determined by the method of Boucher in 15 patients
Plasma Renin Activity and Renin-Substrate Concentration in Patients with Liver Disease By Carlos R. Ayers, M.D. ABSTRACT Peripheral venous renin activity was determined by the method of Boucher in 15 patients
BIOL 2402 Fluid/Electrolyte Regulation
 Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Renal-Related Questions
 Renal-Related Questions 1) List the major segments of the nephron and for each segment describe in a single sentence what happens to sodium there. (10 points). 2) a) Describe the handling by the nephron
Renal-Related Questions 1) List the major segments of the nephron and for each segment describe in a single sentence what happens to sodium there. (10 points). 2) a) Describe the handling by the nephron
Regulation of Arterial Blood Pressure 2 George D. Ford, Ph.D.
 Regulation of Arterial Blood Pressure 2 George D. Ford, Ph.D. OBJECTIVES: 1. Describe the Central Nervous System Ischemic Response. 2. Describe chemical sensitivities of arterial and cardiopulmonary chemoreceptors,
Regulation of Arterial Blood Pressure 2 George D. Ford, Ph.D. OBJECTIVES: 1. Describe the Central Nervous System Ischemic Response. 2. Describe chemical sensitivities of arterial and cardiopulmonary chemoreceptors,
Copyright 2009 Pearson Education, Inc. Copyright 2009 Pearson Education, Inc. Figure 19-1c. Efferent arteriole. Juxtaglomerular apparatus
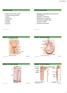 /6/0 About this Chapter Functions of the Kidneys Anatomy of the urinary system Overview of kidney function Secretion Micturition Regulation of extracellular fluid volume and blood pressure Regulation of
/6/0 About this Chapter Functions of the Kidneys Anatomy of the urinary system Overview of kidney function Secretion Micturition Regulation of extracellular fluid volume and blood pressure Regulation of
PHARMACOLOGICAL STUDY OF THE ANOCOCCYGEUS MUSCLE OF
 Br. J. Pharmac. (198). 71, 35-4 PHARMACOLOGICAL STUDY OF TH ANOCOCCYGUS MUSCL OF TH DOG A.R. DHPOUR, M.A. KHOYI, H. KOUTCHKI & M.R. ZARRINDAST Department of Pharmacology, Faculty of Medicine, University
Br. J. Pharmac. (198). 71, 35-4 PHARMACOLOGICAL STUDY OF TH ANOCOCCYGUS MUSCL OF TH DOG A.R. DHPOUR, M.A. KHOYI, H. KOUTCHKI & M.R. ZARRINDAST Department of Pharmacology, Faculty of Medicine, University
Blood Pressure Regulation 2. Faisal I. Mohammed, MD,PhD
 Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Blood Pressure Regulation 2. Faisal I. Mohammed, MD,PhD
 Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Blood Pressure Regulation 2 Faisal I. Mohammed, MD,PhD 1 Objectives Outline the intermediate term and long term regulators of ABP. Describe the role of Epinephrine, Antidiuretic hormone (ADH), Renin-Angiotensin-Aldosterone
Cellular Control of Renin Secretion
 Rev. Physiol. Biochem. Pharmacol., Vol. 113 Springer-Verlag 1989 Cellular Control of Renin Secretion ARMIN KURTZ Contents 1 Introduction 2 2 Renin Synthesis and Secretion 2.1 Experimental Models 4 2.2
Rev. Physiol. Biochem. Pharmacol., Vol. 113 Springer-Verlag 1989 Cellular Control of Renin Secretion ARMIN KURTZ Contents 1 Introduction 2 2 Renin Synthesis and Secretion 2.1 Experimental Models 4 2.2
Questions? Homework due in lab 6. PreLab #6 HW 15 & 16 (follow directions, 6 points!)
 Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
1. Antihypertensive agents 2. Vasodilators & treatment of angina 3. Drugs used in heart failure 4. Drugs used in arrhythmias
 1. Antihypertensive agents 2. Vasodilators & treatment of angina 3. Drugs used in heart failure 4. Drugs used in arrhythmias Only need to know drugs discussed in class At the end of this section you should
1. Antihypertensive agents 2. Vasodilators & treatment of angina 3. Drugs used in heart failure 4. Drugs used in arrhythmias Only need to know drugs discussed in class At the end of this section you should
Lactate and force production in skeletal muscle
 J Physiol 562.2 (2005) pp 521 526 521 Lactate and force production in skeletal muscle Michael Kristensen, Janni Albertsen, Maria Rentsch and Carsten Juel Copenhagen Muscle Research Centre, University of
J Physiol 562.2 (2005) pp 521 526 521 Lactate and force production in skeletal muscle Michael Kristensen, Janni Albertsen, Maria Rentsch and Carsten Juel Copenhagen Muscle Research Centre, University of
BODY FLUID. Outline. Functions of body fluid Water distribution in the body Maintenance of body fluid. Regulation of fluid homeostasis
 BODY FLUID Nutritional Biochemistry Yue-Hwa Chen Dec 13, 2007 Chen 1 Outline Functions of body fluid Water distribution in the body Maintenance of body fluid Intake vs output Regulation of body fluid Fluid
BODY FLUID Nutritional Biochemistry Yue-Hwa Chen Dec 13, 2007 Chen 1 Outline Functions of body fluid Water distribution in the body Maintenance of body fluid Intake vs output Regulation of body fluid Fluid
Collin County Community College RENAL PHYSIOLOGY
 Collin County Community College BIOL. 2402 Anatomy & Physiology WEEK 12 Urinary System 1 RENAL PHYSIOLOGY Glomerular Filtration Filtration process that occurs in Bowman s Capsule Blood is filtered and
Collin County Community College BIOL. 2402 Anatomy & Physiology WEEK 12 Urinary System 1 RENAL PHYSIOLOGY Glomerular Filtration Filtration process that occurs in Bowman s Capsule Blood is filtered and
Acid-Base Alterations and Renal Gluconeogenesis: Effect
 Journal of Clinical Investigation Vol. 46, No. 7, 1967 Acid-Base Alterations and Renal Gluconeogenesis: Effect of ph, Bicarbonate Concentration, and Pco2 * DONALD E. KAMM,t ROBERT E. Fuisz,4 A. DAVID GOODMAN,
Journal of Clinical Investigation Vol. 46, No. 7, 1967 Acid-Base Alterations and Renal Gluconeogenesis: Effect of ph, Bicarbonate Concentration, and Pco2 * DONALD E. KAMM,t ROBERT E. Fuisz,4 A. DAVID GOODMAN,
19. RENAL PHYSIOLOGY ROLE OF THE URINARY SYSTEM THE URINARY SYSTEM. Components and function. V BS 122 Physiology II 151 Class of 2011
 19. RENAL PHYSIOLOGY THE URINARY SYSTEM Components and function The urinary system is composed of two kidneys, the functionally filtering apparatus, which connect through two tubular structures called
19. RENAL PHYSIOLOGY THE URINARY SYSTEM Components and function The urinary system is composed of two kidneys, the functionally filtering apparatus, which connect through two tubular structures called
Cellular Messengers. Intracellular Communication
 Cellular Messengers Intracellular Communication Most common cellular communication is done through extracellular chemical messengers: Ligands Specific in function 1. Paracrines Local messengers (neighboring
Cellular Messengers Intracellular Communication Most common cellular communication is done through extracellular chemical messengers: Ligands Specific in function 1. Paracrines Local messengers (neighboring
Diagram of the inner portions of the kidney
 Excretory and Endocrine functions of the kidney The kidneys are the main excretory organs which eliminate in the urine, most metabolites primarily those containing nitrogen such as ammonia, urea and creatinine.
Excretory and Endocrine functions of the kidney The kidneys are the main excretory organs which eliminate in the urine, most metabolites primarily those containing nitrogen such as ammonia, urea and creatinine.
The ability of the kidneys to regulate extracellular fluid volume by altering sodium
 REGULATION OF EXTRACELLULAR FLUID VOLUME BY INTEGRATED CONTROL OF SODIUM EXCRETION Joey P. Granger Department of Physiology and Biophysics, University of Mississippi Medical Center, Jackson, Mississippi
REGULATION OF EXTRACELLULAR FLUID VOLUME BY INTEGRATED CONTROL OF SODIUM EXCRETION Joey P. Granger Department of Physiology and Biophysics, University of Mississippi Medical Center, Jackson, Mississippi
BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1
 BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1 1. a. Proximal tubule. b. Proximal tubule. c. Glomerular endothelial fenestrae, filtration slits between podocytes of Bowman's capsule.
BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1 1. a. Proximal tubule. b. Proximal tubule. c. Glomerular endothelial fenestrae, filtration slits between podocytes of Bowman's capsule.
In the name of GOD. Animal models of cardiovascular diseases: myocardial infarction & hypertension
 In the name of GOD Animal models of cardiovascular diseases: myocardial infarction & hypertension 44 Presentation outline: Cardiovascular diseases Acute myocardial infarction Animal models for myocardial
In the name of GOD Animal models of cardiovascular diseases: myocardial infarction & hypertension 44 Presentation outline: Cardiovascular diseases Acute myocardial infarction Animal models for myocardial
mid ihsan (Physiology ) GFR is increased when A -Renal blood flow is increased B -Sym. Ganglion activity is reduced C-A and B **
 (Physiology ) mid ihsan GFR is increased when A -Renal blood flow is increased B -Sym. Ganglion activity is reduced C-A and B ** Colloid pressure in the efferent arteriole is: A- More than that leaving
(Physiology ) mid ihsan GFR is increased when A -Renal blood flow is increased B -Sym. Ganglion activity is reduced C-A and B ** Colloid pressure in the efferent arteriole is: A- More than that leaving
RENAL PHYSIOLOGY WESTMEAD PRIMARY EXAM
 RENAL PHYSIOLOGY WESTMEAD PRIMARY EXAM RENAL PHYSIOLOGY - ANATOMY Glomerulus + renal tubule Each kidney has 1.3 million nephrons Cortical nephrons (85%) have shorter Loop of Henle than Juxtamedullary nephrons
RENAL PHYSIOLOGY WESTMEAD PRIMARY EXAM RENAL PHYSIOLOGY - ANATOMY Glomerulus + renal tubule Each kidney has 1.3 million nephrons Cortical nephrons (85%) have shorter Loop of Henle than Juxtamedullary nephrons
Effect of Sodium Loading and Depletion on Cyclic Nucleotides in Plasma and Aorta. Interaction between Prostacyclin and Cyclic Nucleotides
 Endocrinol. Japon. 1982, 29 (2), 245-250 Effect of Sodium Loading and Depletion on Cyclic Nucleotides in Plasma and Aorta. Interaction between Prostacyclin and Cyclic Nucleotides MANABU YOSHIMURA, TERUO
Endocrinol. Japon. 1982, 29 (2), 245-250 Effect of Sodium Loading and Depletion on Cyclic Nucleotides in Plasma and Aorta. Interaction between Prostacyclin and Cyclic Nucleotides MANABU YOSHIMURA, TERUO
Blood Pressure Regulation Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.
 Blood Pressure Regulation Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Introduction There are two basic mechanisms for regulating
Blood Pressure Regulation Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Introduction There are two basic mechanisms for regulating
Human Urogenital System 26-1
 Human Urogenital System 26-1 Urogenital System Functions Filtering of blood, Removal of wastes and metabolites Regulation of blood volume and composition concentration of blood solutes ph of extracellular
Human Urogenital System 26-1 Urogenital System Functions Filtering of blood, Removal of wastes and metabolites Regulation of blood volume and composition concentration of blood solutes ph of extracellular
ROLE OF CALCIUM IN DRUG ACTION ON SMOOTH MUSCLE 1, 2 NORIKO YUKISADA AND FUMIKO EBASHI
 Jap. J. Pharmacol. 11, 46-53 (1961) ROLE OF CALCIUM IN DRUG ACTION ON SMOOTH MUSCLE 1, 2 NORIKO YUKISADA AND FUMIKO EBASHI Department of Pharmacology, Faculty of Medicine, University of Tokyo, Tokyo Received
Jap. J. Pharmacol. 11, 46-53 (1961) ROLE OF CALCIUM IN DRUG ACTION ON SMOOTH MUSCLE 1, 2 NORIKO YUKISADA AND FUMIKO EBASHI Department of Pharmacology, Faculty of Medicine, University of Tokyo, Tokyo Received
Effects of adrenaline on nerve terminals in the superior cervical ganglion of the rabbit
 Br. J. Pharmac. (1971), 41, 331-338. Effects of adrenaline on nerve terminals in the superior cervical ganglion of the rabbit D. D. CHRIST AND S. NISHI Neurophysiology Laboratory, Department of Pharmacology,
Br. J. Pharmac. (1971), 41, 331-338. Effects of adrenaline on nerve terminals in the superior cervical ganglion of the rabbit D. D. CHRIST AND S. NISHI Neurophysiology Laboratory, Department of Pharmacology,
Renal Regulation of Sodium and Volume. Dr. Dave Johnson Associate Professor Dept. Physiology UNECOM
 Renal Regulation of Sodium and Volume Dr. Dave Johnson Associate Professor Dept. Physiology UNECOM Maintaining Volume Plasma water and sodium (Na + ) are regulated independently - you are already familiar
Renal Regulation of Sodium and Volume Dr. Dave Johnson Associate Professor Dept. Physiology UNECOM Maintaining Volume Plasma water and sodium (Na + ) are regulated independently - you are already familiar
Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion.
 The Kidney Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion. The kidney has 6 roles in the maintenance of homeostasis. 6 Main Functions 1. Ion Balance
The Kidney Vertebrates possess kidneys: internal organs which are vital to ion and water balance and excretion. The kidney has 6 roles in the maintenance of homeostasis. 6 Main Functions 1. Ion Balance
This laboratory exercise uses a simple preparation and a straightforward
 LABORATORY DEMONSTRATION OF VASCULAR SMOOTH MUSCLE FUNCTION USING RAT AORTIC RING SEGMENTS Rayna J. Gonzales, Rebecca W. Carter, and Nancy L. Kanagy Vascular Physiology Group, Department of Cell Biology
LABORATORY DEMONSTRATION OF VASCULAR SMOOTH MUSCLE FUNCTION USING RAT AORTIC RING SEGMENTS Rayna J. Gonzales, Rebecca W. Carter, and Nancy L. Kanagy Vascular Physiology Group, Department of Cell Biology
A&P 2 CANALE T H E U R I N A R Y S Y S T E M
 A&P 2 CANALE T H E U R I N A R Y S Y S T E M URINARY SYSTEM CONTRIBUTION TO HOMEOSTASIS Regulates body water levels Excess water taken in is excreted Output varies from 2-1/2 liter/day to 1 liter/hour
A&P 2 CANALE T H E U R I N A R Y S Y S T E M URINARY SYSTEM CONTRIBUTION TO HOMEOSTASIS Regulates body water levels Excess water taken in is excreted Output varies from 2-1/2 liter/day to 1 liter/hour
Effects of Calcium Antagonist Drugs on Acetylcholine and High K Responses of a Molluscan Muscle Neptunea Antiqua
 Gen. Physiol. Biophys. (1995), 14, 419 426 419 Effects of Calcium Antagonist Drugs on Acetylcholine and High K Responses of a Molluscan Muscle Neptunea Antiqua F. I. ALOHAN Division of Biological Sciences,
Gen. Physiol. Biophys. (1995), 14, 419 426 419 Effects of Calcium Antagonist Drugs on Acetylcholine and High K Responses of a Molluscan Muscle Neptunea Antiqua F. I. ALOHAN Division of Biological Sciences,
THE EFFECTS OF AN ANGIOTENSIN BLOCKER (SARALASIN) ON KIDNEY FUNCTION IN DEHYDRATED SHEEP
 Quarterly Journal of Experimental Physiology (1982) 67, 97-103 Printed in Great Britain THE EFFECTS OF AN ANGIOTENSIN BLOCKER (SARALASIN) ON KIDNEY FUNCTION IN DEHYDRATED SHEEP NANCY E. YESBERG, MYRNA
Quarterly Journal of Experimental Physiology (1982) 67, 97-103 Printed in Great Britain THE EFFECTS OF AN ANGIOTENSIN BLOCKER (SARALASIN) ON KIDNEY FUNCTION IN DEHYDRATED SHEEP NANCY E. YESBERG, MYRNA
Effects of Saline and Mannitol on Renin and Distal Tubule Na in Rats
 786 Effects of Saline and Mannitol on Renin and Distal Tubule Na in Rats PAUL C. CHURCHILL, MONIQUE C. CHURCHILL, AND FRANKLIN D. MCDONALD SUMMARY The purpose of these experiments was to investigate the
786 Effects of Saline and Mannitol on Renin and Distal Tubule Na in Rats PAUL C. CHURCHILL, MONIQUE C. CHURCHILL, AND FRANKLIN D. MCDONALD SUMMARY The purpose of these experiments was to investigate the
Fluid and electrolyte balance, imbalance
 Fluid and electrolyte balance, imbalance Body fluid The fluids are distributed throughout the body in various compartments. Body fluid is composed primarily of water Water is the solvent in which all solutes
Fluid and electrolyte balance, imbalance Body fluid The fluids are distributed throughout the body in various compartments. Body fluid is composed primarily of water Water is the solvent in which all solutes
GENERAL CHARACTERISTICS OF THE ENDOCRINE SYSTEM FIGURE 17.1
 GENERAL CHARACTERISTICS OF THE ENDOCRINE SYSTEM FIGURE 17.1 1. The endocrine system consists of glands that secrete chemical signals, called hormones, into the blood. In addition, other organs and cells
GENERAL CHARACTERISTICS OF THE ENDOCRINE SYSTEM FIGURE 17.1 1. The endocrine system consists of glands that secrete chemical signals, called hormones, into the blood. In addition, other organs and cells
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D.
 RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
Renal physiology D.HAMMOUDI.MD
 Renal physiology D.HAMMOUDI.MD Functions Regulating blood ionic composition Regulating blood ph Regulating blood volume Regulating blood pressure Produce calcitrol and erythropoietin Regulating blood glucose
Renal physiology D.HAMMOUDI.MD Functions Regulating blood ionic composition Regulating blood ph Regulating blood volume Regulating blood pressure Produce calcitrol and erythropoietin Regulating blood glucose
Heart Failure (HF) Treatment
 Heart Failure (HF) Treatment Heart Failure (HF) Complex, progressive disorder. The heart is unable to pump sufficient blood to meet the needs of the body. Its cardinal symptoms are dyspnea, fatigue, and
Heart Failure (HF) Treatment Heart Failure (HF) Complex, progressive disorder. The heart is unable to pump sufficient blood to meet the needs of the body. Its cardinal symptoms are dyspnea, fatigue, and
CALCIUM EFFLUX AND SECRETION
 Br. J. Pharmac. (1974), 52, 387-392 CALCIUM EFFLUX AND SECRETION OF a-amylase FROM RAT PANCREAS S. HEISLER Department of Physiology and Pharmacology, Centre Hospitalier Universitaire, Universite de Sherbrooke,
Br. J. Pharmac. (1974), 52, 387-392 CALCIUM EFFLUX AND SECRETION OF a-amylase FROM RAT PANCREAS S. HEISLER Department of Physiology and Pharmacology, Centre Hospitalier Universitaire, Universite de Sherbrooke,
The Effects of Extracellular Calcium Removal on Sino-atrial Node Cells Treated with Potassium-depleted Solutions
 Short Communication Japanese Journal of Physiology, 36, 403-409, 1986 The Effects of Extracellular Calcium Removal on Sino-atrial Node Cells Treated with Potassium-depleted Solutions Shun-ichi MIYAMAE
Short Communication Japanese Journal of Physiology, 36, 403-409, 1986 The Effects of Extracellular Calcium Removal on Sino-atrial Node Cells Treated with Potassium-depleted Solutions Shun-ichi MIYAMAE
8. URINE CONCENTRATION
 8. URINE CONCENTRATION The final concentration of the urine is very dependent on the amount of liquid ingested, the losses through respiration, faeces and skin, including sweating. When the intake far
8. URINE CONCENTRATION The final concentration of the urine is very dependent on the amount of liquid ingested, the losses through respiration, faeces and skin, including sweating. When the intake far
STEIN IN-TERM EXAM -- BIOLOGY FEBRUARY 18, PAGE
 STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 18, 2016 -- PAGE 1 of 8 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 18, 2016 -- PAGE 1 of 8 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
STEIN IN-TERM EXAM -- BIOLOGY FEBRUARY 16, PAGE
 STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 16, 2017 -- PAGE 1 of 9 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 16, 2017 -- PAGE 1 of 9 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
RENAL PHYSIOLOGY. Physiology Unit 4
 RENAL PHYSIOLOGY Physiology Unit 4 Renal Functions Primary Function is to regulate the chemistry of plasma through urine formation Additional Functions Regulate concentration of waste products Regulate
RENAL PHYSIOLOGY Physiology Unit 4 Renal Functions Primary Function is to regulate the chemistry of plasma through urine formation Additional Functions Regulate concentration of waste products Regulate
Glomerular Capillary Blood Pressure
 Glomerular Capillary Blood Pressure Fluid pressure exerted by blood within glomerular capillaries Depends on Contraction of the heart Resistance to blood flow offered by afferent and efferent arterioles
Glomerular Capillary Blood Pressure Fluid pressure exerted by blood within glomerular capillaries Depends on Contraction of the heart Resistance to blood flow offered by afferent and efferent arterioles
Body fluid volume is small (~5L (blood + serum)) Composition can change rapidly e.g. due to increase in metabolic rate
 Renal physiology The kidneys Allow us to live on dry land. Body fluid volume is small (~5L (blood + serum)) Composition can change rapidly e.g. due to increase in metabolic rate Kidneys maintain composition
Renal physiology The kidneys Allow us to live on dry land. Body fluid volume is small (~5L (blood + serum)) Composition can change rapidly e.g. due to increase in metabolic rate Kidneys maintain composition
Physiology Unit 1 CELL SIGNALING: CHEMICAL MESSENGERS AND SIGNAL TRANSDUCTION PATHWAYS
 Physiology Unit 1 CELL SIGNALING: CHEMICAL MESSENGERS AND SIGNAL TRANSDUCTION PATHWAYS In Physiology Today Cell Communication Homeostatic mechanisms maintain a normal balance of the body s internal environment
Physiology Unit 1 CELL SIGNALING: CHEMICAL MESSENGERS AND SIGNAL TRANSDUCTION PATHWAYS In Physiology Today Cell Communication Homeostatic mechanisms maintain a normal balance of the body s internal environment
