Test Report No.: GZHG OT Date: Oct 11, 2011 Page 1 of 7 PERSONAL SAFETY CORPORATION 1655 PROGRESS DRIVE, HIAWATHA, LOWA 52233, USA
|
|
|
- Eugene Wilcox
- 5 years ago
- Views:
Transcription
1 Test Report No.: GZHG824985OT Date: Oct, 2 Page of 7 PERSONAL SAFETY CORPORATION 655 PROGRESS DRIVE, HIAWATHA, LOWA 52233, USA The following sample was submitted and identified on behalf of the client as Sample Description : PVC SHEET Sample Receiving Date : Sep 2, 2 Testing Period : Sep 2, 2to Oct 4, 2 Test Requested : ) Skin Irritant 2) Skin Sensitization Test Method : -2) ISO 993-:2 Biological evaluation of medical devices Part : Tests for irritation and delayed-type hypersensitivity Test Summary : ) The primary Irritation Index (PII) of the submitted sample is, no erythema and oedema. 2) The hypersensitivity scale of the submitted sample is, no visible change. Signed for and on behalf of SGS-CSTC Ltd. Yan Lau Approved Signatory
2 Test Report No.: GZHG824985OT Date: Oct, 2 Page 2 of 7 ) Skin irritant test Species: Rabbit Strain: New Zealand Rabbit (Certificate No. 94) Source: GUANGDONG MEDICAL LABORATORY ANIMAL CENTER Sex: 3 female Body weight range: 2.kg-3.kg Number of animals: Three Sample preparation: The submitted thin sheet sample was cut into 2.5cm 2.5cm and used in the experiment directly. (Negative control:.5 ml.9% saline solution per site) Experimental procedure Summary: ) Primary skin irritant Fur was generally clipped within 24h before testing on the backs of the animals on both sides (about cm 5cm respectively). The submitted sample and the negative control were applied to the test sites and control sites respectively shown in Figure. The application sites were covered with two layers of gauze patch and a layer of cellophane, and then were wrapped with a semi-occlusive bandage for 4h. At the end of the contact time, remove all the patches, mark the sites followed by water washing. At h, 24h, 48h and 72h following removal of the samples, describe and score the skin reactions for erythema and oedema, determine the Primary Irritation Index (PII) and the response categories according to ISO 993- :2. Results are shown in Table Figure. Applied sites on the skin Note: -Head; 2-test site; 3-control site; 4-clipped region on the back; 5-control site; 6-test site; 7-tail.
3 Test Report No.: GZHG824985OT Date: Oct, 2 Page 3 of 7 2) Cumulative skin irritant 72 hours after removal of the primary skin irritant samples, cumulative skin irritant test was directly conducted on the same sites as shown in Figure. The sample sheet and the negative control were applied on the test and the control sites respectively. The application sites were covered with two layers of gauze patch and a layer of cellophane, and then the application sites were wrapped with a semi-occlusive bandage for 4h. At the end of the contact time, removed all the patches, marked the sites followed by water washing. Recorded the appearances of the application site within h after removal of the patches and immediately prior to the next application. The number of exposures was 4 days. From the second exposure, fur was clipped before every exposure. After the last exposure, describe the appearance of each application site at h, 24 h, 48 h and 72 h following removal of the samples. For each animal, add together the Primary Irritation s for both erythema and oedema at each time specified. Divide this total by the total figure of observations to obtain the Irritation per animal. Add together the Irritation s of all animals and divide by the total number of animals. This value is the Cumulative Irritation Index. The Cumulative Irritation Index is compared to the categories of Irritation Response defined in Table 2 according to ISO 993-:2 to obtain the Response Category. Table Scoring system for skin reaction Skin reaction and eschar formation No erythema Very slight erythema (barely perceptible) Well-defined erythema Moderate erythema Severe erythema (beet-redness) to eschar formation preventing grading of erythema formation No oedema Very slight oedema (barely perceptible) Well-defined oedema (edges of area well-defined by definite raising) Moderate oedema (raised approximately mm) Severe oedema (raised more than mm and extending beyond exposure area) Value Table 2 Irritation Response categories in rabbit Mean score Response category ~<.4 Negligible.5~<.9 Slight 2~<4.9 Moderate 5~8. Severe
4 Test Report No.: GZHG824985OT Date: Oct, 2 Page 4 of 7 Test result: Table 3 Scoring board for primary dermal irritation Animal No. Sex Body Sample Reference sample weight (g) 24h 48h 72h 24h 48h 72h Before test After test primary dermal irritation Category Table 4 Scoring board for Cumulative Skin irritation Date Animal Sample Negative Sample Amount Average Sore for 4 days per animal Average Sore for per day per animal Sample: Sample: Negative Sample: Negative Sample: Cumulative Irritation Response categories Negligible Negligible
5 Test Report No.: GZHG824985OT Date: Oct, 2 Page 5 of 7 3) Delayed-type hypersensitivity Species: Guinea Pigs Strain: SPF Grade Hartley Guinea Pig (Certificate No. 9286) Source: GUANGDONG MEDICAL LABORATORY ANIMAL CENTER Sex: 5 Male Body weight range: 25~3g Number of animals: Fifteen Sample preparation: The sample was washed with distilled water and dried with filter paper, then cut into pieces of.5cm cm, extracted by.9% saline solution (6 cm 2 sample / ml saline solution), 37±, 24h. (Negative control:.9% saline solution). ml sample extract solution and ml negative control solution were prepared. Experimental procedure Summary: 5 Guinea Pigs were randomly divided into two groups: 5 for control group and for testing group. Induction: Clip furs from the back of guinea pig s cervix before test. Clean the fur-clipped area with 75% ethanol. Make intradermal injections into each animal at the injection sites as shown in Figure 2. Figure 2 Intradermal injection sites Note: Head; 2. ml intradermal injection sites A For testing group:.5 ml FCA (Freund's complete adjuvant) +.5 ml.9% saline solution; For control group:. ml FCA; B For testing group:. ml sample extract solution; For control group:. ml negative control solution; C For testing group:.5 ml sample extraction solution +.25 ml FCA +.25 ml saline solution; For control group:.5 ml FCA +.5 ml negative control solution. (Note: Use high-speed dispersion machine to facilitate liquid mixing.) 3 Fur-clipped inner part of scapulas 4 Tail Seven days after injection, clip furs from the injection area again, clean with 75% ethanol. If no irritation produced, pretreat the area with % sodium dodecyl sulfate massaged into the skin 24h before the patch was applied. 2cm 4cm filter paper patches immerged in the sample extract solution till saturated were applied on the injection sites and secured with an occlusive dressing. Remove the dressings and patches after 48h. Treat the control animals similarly, using the negative control solution.
6 Test Report No.: GZHG824985OT Date: Oct, 2 Page 6 of 7 Observation: At 4 days after the above induction phase, immerge 2cm 4cm patches into the sample solution till saturated. Clip furs from untested sites of both side of the stomach as challenge sites, clean with 75% ethanol, apply the patches on the challenge sites and secure with an occlusive dressing for 24 hours. Observe the appearance of the challenge sites of testing and control groups 24h and 48h after removal of the dressing and patches. Describe and grade the skin reaction for erythema and oedema according to the Magnusson and Kligman grading given in Table 5 for each challenge site and at each time interval. According to Magnusson and Kligman Grade Standard, provided the grade is less than for the animals in control group, while the grade is bigger than for the testing group, the result normally indicate sensitization. If grades of or greater are noted in control animals, then the reactions of test animals which exceed the most severe reaction in control animals are presumed to be sensitization. If the response is equivocal, rechallenge is recommended to confirm the results from the first challenge. The outcome of the test is presented as the frequency of positive challenge results in test and control animals. Table 5 Magnusson and Kligman scale Patch test reaction No visible change Discrete or patchy erythema Moderate and confluent erythema Intense erythema and swelling Grading scale 2 3 Test result: Group Test group Control group No. Table 6 Scoring board for delayed-type hypersensitivity 24h after remove test sample 48h after remove test sample erythema oedema erythema oedema Remark :This test was subcontracted to qualified subcontractor. Grade
7 Test Report No.: GZHG824985OT Date: Oct, 2 Page 7 of 7 Sample photo: *** End of Report ***
Azadirachtin Evaluation of Classification and Labelling Proposal with regard to Skin Sensitisation
 MITSUI AgriScience International page 1 of 9 Azadirachtin Evaluation of Classification and Labelling Proposal with regard to Skin Sensitisation Date: 17.11.2014 Report Number: 234379-A2-050206-01 Author:
MITSUI AgriScience International page 1 of 9 Azadirachtin Evaluation of Classification and Labelling Proposal with regard to Skin Sensitisation Date: 17.11.2014 Report Number: 234379-A2-050206-01 Author:
PacificBeam SHELLIII/PB-SHELL3: ACUTE DERMAL IRRITATION IN THE RABBIT PROJECT NUMBER: 2533/0014 AUTHOR:
 PAGE 1 OF 14 PAGES PacificBeam SHELLIII/PB-SHELL3: ACUTE DERMAL IRRITATION IN THE RABBIT PROJECT NUMBER: 2533/0014 AUTHOR: A Sanders STUDY SPONSOR: M.I.C.(Medical Intelligence Corporation) Co., Ltd. 2-2-15
PAGE 1 OF 14 PAGES PacificBeam SHELLIII/PB-SHELL3: ACUTE DERMAL IRRITATION IN THE RABBIT PROJECT NUMBER: 2533/0014 AUTHOR: A Sanders STUDY SPONSOR: M.I.C.(Medical Intelligence Corporation) Co., Ltd. 2-2-15
ISO INTERNATIONAL STANDARD. Biological evaluation of medical devices Part 10: Tests for irritation and skin sensitization
 INTERNATIONAL STANDARD ISO 10993-10 Third edition 2010-08-01 Biological evaluation of medical devices Part 10: Tests for irritation and skin sensitization Évaluation biologique des dispositifs médicaux
INTERNATIONAL STANDARD ISO 10993-10 Third edition 2010-08-01 Biological evaluation of medical devices Part 10: Tests for irritation and skin sensitization Évaluation biologique des dispositifs médicaux
ACTS Testing Labs, Inc. Buffalo, NY Patty Dick. Primary Dermal Irritation in Rabbits. 10/29/96 COMPLETED: H/l/96
 Leberco*Celsis Testing 123 Hawthorne Street Roselle Park, NJ 07204-0206 908.245.1933 / 800.523.LABS Fax 908.245.6253 Nov. 5, 1996 SUBMITTED TO: ACTS Testing Labs, Inc. Buffalo, NY Patty Dick ASSAY NUMBER:
Leberco*Celsis Testing 123 Hawthorne Street Roselle Park, NJ 07204-0206 908.245.1933 / 800.523.LABS Fax 908.245.6253 Nov. 5, 1996 SUBMITTED TO: ACTS Testing Labs, Inc. Buffalo, NY Patty Dick ASSAY NUMBER:
SCCP. Opinion on. Alkyl (C16, C18, C22) trimethylammonium chloride
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Public Health and Risk Assessment C7 - Risk assessment SCIENTIFIC COMMITTEE ON CONSUMER PRODUCTS SCCP Opinion on Alkyl
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Public Health and Risk Assessment C7 - Risk assessment SCIENTIFIC COMMITTEE ON CONSUMER PRODUCTS SCCP Opinion on Alkyl
Addendum 1 to the Draft Assessment Report
 Addendum 1 to the Draft Assessment Report of 22 November 2000 Benzoic acid Volume 1, Level 2 Volume 3, B.2 Physical and chemical properties B.4 Proposals for the classification and labelling B.6 Toxicology
Addendum 1 to the Draft Assessment Report of 22 November 2000 Benzoic acid Volume 1, Level 2 Volume 3, B.2 Physical and chemical properties B.4 Proposals for the classification and labelling B.6 Toxicology
OECD GUIDELINE FOR THE TESTING OF CHEMICALS
 Adopted: 24 th April 2002 OECD GUIDELINE FOR THE TESTING OF CHEMICALS Acute Dermal Irritation/Corrosion INTRODUCTION 1. OECD Guidelines for Testing of Chemicals are periodically reviewed to ensure that
Adopted: 24 th April 2002 OECD GUIDELINE FOR THE TESTING OF CHEMICALS Acute Dermal Irritation/Corrosion INTRODUCTION 1. OECD Guidelines for Testing of Chemicals are periodically reviewed to ensure that
Acute Oral Toxicity. Acute Toxic Class Method of Difenacoum in Rats.
 The Activa / PelGar Brodifacoum and Difenacoum Task Force RMS Finland Section A6.1.1 Annex Point IIA6.1 Acute Oral Toxicity Acute Toxic Class Method of Difenacoum in Rats. 1 REFERENCE Official use only
The Activa / PelGar Brodifacoum and Difenacoum Task Force RMS Finland Section A6.1.1 Annex Point IIA6.1 Acute Oral Toxicity Acute Toxic Class Method of Difenacoum in Rats. 1 REFERENCE Official use only
OPINION ON ALKYL (C16, C18, C22) TRIMETHYLAMMONIUM CHLORIDE. For other uses than as a preservative
 Scientific Committee on Consumer Products SCCP OPINION ON ALKYL (C16, C18, C22) TRIMETHYLAMMONIUM CHLORIDE For other uses than as a preservative COLIPA n P72 The SCCP adopted this opinion at its 11 th
Scientific Committee on Consumer Products SCCP OPINION ON ALKYL (C16, C18, C22) TRIMETHYLAMMONIUM CHLORIDE For other uses than as a preservative COLIPA n P72 The SCCP adopted this opinion at its 11 th
EXPERIMENTAL STUDY ON CROSS-CONTACT ALLERGY DUE TO DITHIOCARBAMATE FUNGICIDES
 Industrial Health, 1977, 15, 87. EXPERIMENTAL STUDY ON CROSS-CONTACT ALLERGY DUE TO DITHIOCARBAMATE FUNGICIDES Toshio MATSUSHITA õ, Mitsuki YOSHIOKA õ, Yoshiki ARIMATSU õ õ and Shigeru NOMURA õ õ õd epartment
Industrial Health, 1977, 15, 87. EXPERIMENTAL STUDY ON CROSS-CONTACT ALLERGY DUE TO DITHIOCARBAMATE FUNGICIDES Toshio MATSUSHITA õ, Mitsuki YOSHIOKA õ, Yoshiki ARIMATSU õ õ and Shigeru NOMURA õ õ õd epartment
Test Report No.: GZHL IP Date: Sep 01, 2016 Page 1 of 12
 Test Report No.: GZHL1604014433IP Date: Sep 01, 2016 Page 1 of 12 DAILI DND GLOBAL CO., LTD. NO.5 ALY.3, IN.79, XINXING ST., YONGKANG DIST., TAINAN CITY 71C, TAIWAN (R.O.C) The following sample(s) was/were
Test Report No.: GZHL1604014433IP Date: Sep 01, 2016 Page 1 of 12 DAILI DND GLOBAL CO., LTD. NO.5 ALY.3, IN.79, XINXING ST., YONGKANG DIST., TAINAN CITY 71C, TAIWAN (R.O.C) The following sample(s) was/were
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Antigenicity and Irritancy Tests of Leukocyte-Reduction Filters Using Animal Models
 Kobe J. Med. Sci., Vol. 52, No. 2, pp. 17-26, 2006 Antigenicity and Irritancy Tests of Leukocyte-Reduction Filters Using Animal Models AKIRA KOKUBUNJI 1, 3, MASARU NATSUAKI 2, KAZUTAKA KANO 3, SHUNRO KAI
Kobe J. Med. Sci., Vol. 52, No. 2, pp. 17-26, 2006 Antigenicity and Irritancy Tests of Leukocyte-Reduction Filters Using Animal Models AKIRA KOKUBUNJI 1, 3, MASARU NATSUAKI 2, KAZUTAKA KANO 3, SHUNRO KAI
Application Guide for Full-Thickness Wounds
 Application Guide for Full-Thickness Wounds PriMatrix Dermal Repair Scaffold PriMatrix Ag Antimicrobial Dermal Repair Scaffold Application Guide for Full Thickness Wounds PriMatrix is a unique dermal repair
Application Guide for Full-Thickness Wounds PriMatrix Dermal Repair Scaffold PriMatrix Ag Antimicrobial Dermal Repair Scaffold Application Guide for Full Thickness Wounds PriMatrix is a unique dermal repair
OPINION ON. For other uses than as a preservative
 Scientific Committee on Consumer Safety SCCS OPINION ON ALKYL (C 16, C 18, C 22 ) TRIMETHYLAMMONIUM CHLORIDE For other uses than as a preservative COLIPA n P72 The SCCS adopted this opinion at its 5 th
Scientific Committee on Consumer Safety SCCS OPINION ON ALKYL (C 16, C 18, C 22 ) TRIMETHYLAMMONIUM CHLORIDE For other uses than as a preservative COLIPA n P72 The SCCS adopted this opinion at its 5 th
Objective To determine the immediate irritation effects and the irritation power of a particular materials.
 SAFETY STUDIES 1.1 Skin Irritation through Open Patch Test 1.2 Skin Irritation through Patch Test. 1.3 Human Skin Irritation Test 1.4 Repeat Open Application Test 1.5 Test For Hypoallergenicity 1.6 Modified
SAFETY STUDIES 1.1 Skin Irritation through Open Patch Test 1.2 Skin Irritation through Patch Test. 1.3 Human Skin Irritation Test 1.4 Repeat Open Application Test 1.5 Test For Hypoallergenicity 1.6 Modified
Test Report. No. SHAEC Date: 12 Sep Page 1 of 6. (SVHC)
 Test Report. No. SHAEC1405830702 Date: 12 Sep 2014. Page 1 of 6. TOPDIODE MANUFACTURING COMPANY LIMITED. SHANGKENG INDUSTRIAL ROAD NO.3,SHANGKENG INDUSTRIAL PARK,CHANGPING TOWN,DONGGUAN,GUANGDONG,CHINA..
Test Report. No. SHAEC1405830702 Date: 12 Sep 2014. Page 1 of 6. TOPDIODE MANUFACTURING COMPANY LIMITED. SHANGKENG INDUSTRIAL ROAD NO.3,SHANGKENG INDUSTRIAL PARK,CHANGPING TOWN,DONGGUAN,GUANGDONG,CHINA..
Sumitomo Chemical Takeda Agro Co., Ltd. TI-435 February Acute Toxicity. Acute oral toxicity study in rats (LD 50 )
 Sumitomo Chemical Takeda Agro Co., Ltd. TI435 February 24 Section A6.1.1 Annex Point IIA VI.6.1.1/1 Acute Toxicity Oral Acute oral toxicity study in rats (LD 5 ) 1 REFERENCE 1.1 Reference Gardner, J. R.
Sumitomo Chemical Takeda Agro Co., Ltd. TI435 February 24 Section A6.1.1 Annex Point IIA VI.6.1.1/1 Acute Toxicity Oral Acute oral toxicity study in rats (LD 5 ) 1 REFERENCE 1.1 Reference Gardner, J. R.
Summary of Toxicity Studies on Imazapyr
 Summary of Toxicity Studies on Imazapyr Technical Department, Cyanamid (Japan) Ltd. (Received July 15, 1997 ; Accepted August 20, 1997) DESCRIPTIO OF THE TEST COMPOUD Imazapyr is a nonselective herbicide
Summary of Toxicity Studies on Imazapyr Technical Department, Cyanamid (Japan) Ltd. (Received July 15, 1997 ; Accepted August 20, 1997) DESCRIPTIO OF THE TEST COMPOUD Imazapyr is a nonselective herbicide
Information and Consent Form for SmartXide DOT Treatment PROCEDURE
 Information and Consent Form for SmartXide DOT Treatment This consent form includes general descriptions of various dermatological laser treatments, including possible benefits and risks that may occur
Information and Consent Form for SmartXide DOT Treatment This consent form includes general descriptions of various dermatological laser treatments, including possible benefits and risks that may occur
Test Report. No. CANEC Date: 07 Nov Page 1 of 7. (SVHC)
 Test Report. No. CANEC1316772101 Date: 07 Nov 2013. Page 1 of 7. HANGZHOU SILAN INTEGRATED CIRCUIT CO., LTD.. NO 308,NO.10 ROAD,EAST ZONE,ECONOMY&TECHNOLOGY DEVELOPMENT AREA,XIASHA,HANGZHOU CHINA. The
Test Report. No. CANEC1316772101 Date: 07 Nov 2013. Page 1 of 7. HANGZHOU SILAN INTEGRATED CIRCUIT CO., LTD.. NO 308,NO.10 ROAD,EAST ZONE,ECONOMY&TECHNOLOGY DEVELOPMENT AREA,XIASHA,HANGZHOU CHINA. The
skin corrosion factsheet animal test non-animal test give the animals 5 factsheet 1 / skin corrosion
 1 skin corrosion Corrosive agents are chemicals that cause irreversible damage and destruction of the skin, often burning through several layers of tissue. Corrosive reactions are typified by ulcers, bleeding,
1 skin corrosion Corrosive agents are chemicals that cause irreversible damage and destruction of the skin, often burning through several layers of tissue. Corrosive reactions are typified by ulcers, bleeding,
Nations, All rights reserved ANNEX 8 AN EXAMPLE OF CLASSIFICATION IN THE GLOBALLY HARMONIZED SYSTEM
 ANNEX AN EXAMPLE OF CLASSIFICATION IN THE GLOBALLY HARMONIZED SYSTEM Annex AN EXAMPLE OF CLASSIFICATION IN THE GLOBALLY HARMONIZED SYSTEM A.1 Classification proposal The following classification proposal
ANNEX AN EXAMPLE OF CLASSIFICATION IN THE GLOBALLY HARMONIZED SYSTEM Annex AN EXAMPLE OF CLASSIFICATION IN THE GLOBALLY HARMONIZED SYSTEM A.1 Classification proposal The following classification proposal
2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 NAME OF THE MEDICINAL PRODUCT Skinoren 20 % cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 g Skinoren cream contains 200 mg (20 %) azelaic acid. For the full list of excipients, see section 6.1 List
NAME OF THE MEDICINAL PRODUCT Skinoren 20 % cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 g Skinoren cream contains 200 mg (20 %) azelaic acid. For the full list of excipients, see section 6.1 List
Sensitization Properties of Propolis and Balsam of Peru in Guinea Pig Maximization Test (GPMT)
 Food and Nutrition Sciences, 2017, 8, 714-724 http://www.scirp.org/journal/fns ISSN Online: 2157-9458 ISSN Print: 2157-944X Sensitization Properties of Propolis and Balsam of Peru in Guinea Pig Maximization
Food and Nutrition Sciences, 2017, 8, 714-724 http://www.scirp.org/journal/fns ISSN Online: 2157-9458 ISSN Print: 2157-944X Sensitization Properties of Propolis and Balsam of Peru in Guinea Pig Maximization
ANNEX 8 AN EXAMPLE OF CLASSIFICATION IN THE
 ANNEX 8 AN EXAMPLE OF CLASSIFICATION IN THE GLOBALLY HARMONIZED SYSTEM - 427 - Annex 8 AN EXAMPLE OF CLASSIFICATION IN THE GLOBALLY HARMONIZED SYSTEM A8.1 Classification proposal The following classification
ANNEX 8 AN EXAMPLE OF CLASSIFICATION IN THE GLOBALLY HARMONIZED SYSTEM - 427 - Annex 8 AN EXAMPLE OF CLASSIFICATION IN THE GLOBALLY HARMONIZED SYSTEM A8.1 Classification proposal The following classification
Test Report No.: XMHL CW Date: Dec. 28, 2016 Page 1 of 8
 Test Report No.: XMHL1612008035CW Date: Dec. 28, 2016 Page 1 of 8 ANHUI GREEN PLANT FIBER PRODUCTION CO., LTD WEST XIQUAN ROAD, FEIDONG ECONOMIC DEVELOPMENT ZONE, HEFEI, CHINA The following sample(s) was/were
Test Report No.: XMHL1612008035CW Date: Dec. 28, 2016 Page 1 of 8 ANHUI GREEN PLANT FIBER PRODUCTION CO., LTD WEST XIQUAN ROAD, FEIDONG ECONOMIC DEVELOPMENT ZONE, HEFEI, CHINA The following sample(s) was/were
OPINION ON HYDROXYISOHEXYL 3-CYCLOHEXENE CARBOXALDEHYDE (HICC)
 SCCP/1456/11 Revision of 27 July 2012 Scientific Committee on Consumer Safety SCCS OPINION ON HYDROXYISOHEXYL 3-CYCLOHEXENE CARBOXALDEHYDE (HICC) The SCCP adopted this opinion at its 13 th plenary of 13-14
SCCP/1456/11 Revision of 27 July 2012 Scientific Committee on Consumer Safety SCCS OPINION ON HYDROXYISOHEXYL 3-CYCLOHEXENE CARBOXALDEHYDE (HICC) The SCCP adopted this opinion at its 13 th plenary of 13-14
Patient Self-Bandaging Leg and Individual Toe Application Guide
 3M Coban 2 for Lymphoedema Compression System Patient Self-Bandaging Leg and Individual Toe Application Guide Disclaimer: This guide is to be used under supervision by your healthcare practitioner following
3M Coban 2 for Lymphoedema Compression System Patient Self-Bandaging Leg and Individual Toe Application Guide Disclaimer: This guide is to be used under supervision by your healthcare practitioner following
Toxicology of Glutaraldehyde
 Toxicology of Glutaraldehyde TOXICOLOGY OF GLUTARALDEHYDE Review of Studies and Human Health Effects Prof. BRYAN BALLANTYNE MD., DSc., PhD. FFOM, FACOM, FAACT, FATS, FRCPath, FIBiol. Director of Applied
Toxicology of Glutaraldehyde TOXICOLOGY OF GLUTARALDEHYDE Review of Studies and Human Health Effects Prof. BRYAN BALLANTYNE MD., DSc., PhD. FFOM, FACOM, FAACT, FATS, FRCPath, FIBiol. Director of Applied
WOUND DRESSING Daily Dressing Packets
 AMERIGEL WOUND DRESSING Daily Dressing Packets P R O D U C T I N F O R M AT I O N MSDS APPLICATION PROTOCOLS AmeriGel WOUND DRESSING Daily Dressing Packets A HYDROGEL WITH A UNIQUE AUTOLYTIC DEBRIDER Diabetic
AMERIGEL WOUND DRESSING Daily Dressing Packets P R O D U C T I N F O R M AT I O N MSDS APPLICATION PROTOCOLS AmeriGel WOUND DRESSING Daily Dressing Packets A HYDROGEL WITH A UNIQUE AUTOLYTIC DEBRIDER Diabetic
Test Report. No. CANML Date: 06 Jun Page 1 of 6.
 Test Report. No. CANML1308006802 Date: 06 Jun 2013. Page 1 of 6. DONGGUAN YAOSHENG SILICON GEL MATERIAL TECHNOLOGY CO., LTD. NO 18 CHANGSHENG DISTRICT, TIANMEI CUN, HUANGJIANG TOWN, DONGGUAN CITY, CHINA..
Test Report. No. CANML1308006802 Date: 06 Jun 2013. Page 1 of 6. DONGGUAN YAOSHENG SILICON GEL MATERIAL TECHNOLOGY CO., LTD. NO 18 CHANGSHENG DISTRICT, TIANMEI CUN, HUANGJIANG TOWN, DONGGUAN CITY, CHINA..
Bromadiolone CAS Active substance in Biocidal Products, Product Type 14 (Rodenticides) The Bromadiolone Task Force
 Competent Authority Report According to Directive 98/8/EC CAS 28772-56-7 Active substance in Biocidal Products, Product Type 14 (Rodenticides) The Task Force DOCUMENT III-A Section 6: Toxicological and
Competent Authority Report According to Directive 98/8/EC CAS 28772-56-7 Active substance in Biocidal Products, Product Type 14 (Rodenticides) The Task Force DOCUMENT III-A Section 6: Toxicological and
Antimicrobial AlphaSan Test Report Summary Table 08/21/01
 AlphaSan RC 5000 Physical Chemical Properties Acute Oral Toxicity, Rat EPA FIFRA 81-1 Acute Dermal Toxicity, Rat EPA FIFRA 81-2 Primary Dermal Irritation Rabbit EPA FIFRA 81-5 Primary Eye Irritation Rabbit
AlphaSan RC 5000 Physical Chemical Properties Acute Oral Toxicity, Rat EPA FIFRA 81-1 Acute Dermal Toxicity, Rat EPA FIFRA 81-2 Primary Dermal Irritation Rabbit EPA FIFRA 81-5 Primary Eye Irritation Rabbit
Skin Photoallergy Test 皮肤光变态反应试验方法. China Food and Drug Administration. Translated by Chemlinked
 Skin Photoallergy Test 皮肤光变态反应试验方法 China Food and Drug Administration Translated by Chemlinked Date of Publication: Aug. 15, 2017 Date of Implementation:Aug. 15, 2017 Disclaimer This is an unofficial document
Skin Photoallergy Test 皮肤光变态反应试验方法 China Food and Drug Administration Translated by Chemlinked Date of Publication: Aug. 15, 2017 Date of Implementation:Aug. 15, 2017 Disclaimer This is an unofficial document
Test Report. No. CANHG A01 Date: 29 Aug Page 1 of 9.
 Test Report. No. Date: 29 Aug 2017. Page 1 of 9. FFG LIMITED. 27F MORRISON PLAZA,5-9 MORRISON HILL ROAD,CAUSEWAYBAY,HONG KONG. This report is to supersede test report CANHG1716962901. The following sample(s)
Test Report. No. Date: 29 Aug 2017. Page 1 of 9. FFG LIMITED. 27F MORRISON PLAZA,5-9 MORRISON HILL ROAD,CAUSEWAYBAY,HONG KONG. This report is to supersede test report CANHG1716962901. The following sample(s)
Surgical Gown. A disposable gown worn by medical staff during surgery. A disposable gown worn by medical staff during surgery
 Surgical Gown A disposable gown worn by medical staff during surgery Used by the surgeon during surgery; significant blood cover DOGS Surgical Gown A disposable gown worn by medical staff during surgery
Surgical Gown A disposable gown worn by medical staff during surgery Used by the surgeon during surgery; significant blood cover DOGS Surgical Gown A disposable gown worn by medical staff during surgery
CASE 1: TYPE-II DIABETIC FOOT ULCER
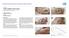 CASE 1: TYPE-II DIABETIC FOOT ULCER DIABETIC FOOT ULCER 48 YEAR-OLD MALE Mr. C., was a 48-year old man with a history of Type-II diabetes over the past 6 years. The current foot ulcer with corresponding
CASE 1: TYPE-II DIABETIC FOOT ULCER DIABETIC FOOT ULCER 48 YEAR-OLD MALE Mr. C., was a 48-year old man with a history of Type-II diabetes over the past 6 years. The current foot ulcer with corresponding
METRIGUARD. Technical Bulletin
 METRIGUARD Technical Bulletin MetriGuard is a multi-purpose disinfectant intended for use in cleaning, decontaminating and disinfecting hard non-porous, inanimate surfaces and non-critical instruments
METRIGUARD Technical Bulletin MetriGuard is a multi-purpose disinfectant intended for use in cleaning, decontaminating and disinfecting hard non-porous, inanimate surfaces and non-critical instruments
Test Report. No. CANEC Date: 25 Jul Page 1 of 7. (SVHC)
 Test Report. No. CANEC1411532127 Date: 25 Jul 2014. Page 1 of 7. KINGBOARD LAMINATES HOLDINGS LIMITED. 2/F.,HARBOUR VIEW 1,NO.12 SCIENCE PARK EAST AVENUE,PHASE II HONG KONG SCIENCE PARK,SHATIN,N.T. HONG
Test Report. No. CANEC1411532127 Date: 25 Jul 2014. Page 1 of 7. KINGBOARD LAMINATES HOLDINGS LIMITED. 2/F.,HARBOUR VIEW 1,NO.12 SCIENCE PARK EAST AVENUE,PHASE II HONG KONG SCIENCE PARK,SHATIN,N.T. HONG
Test Report No.: XMHL CW Date: Apr. 4, 2018 Page 1 of 7
 Test Report No.: XMHL1803001230CW Date: Apr. 4, 2018 Page 1 of 7 QUANZHOU YIQIANG PLASTIC CO., LTD DONGMEN INDUSTRIAL, FENGZHOU, NANAN, 362333 The following sample(s) was/were submitted and identified
Test Report No.: XMHL1803001230CW Date: Apr. 4, 2018 Page 1 of 7 QUANZHOU YIQIANG PLASTIC CO., LTD DONGMEN INDUSTRIAL, FENGZHOU, NANAN, 362333 The following sample(s) was/were submitted and identified
Margosa Extract Evaluation of Classification and Labelling Proposal with regard to Skin Sensitisation
 page 1 of 13 Margosa Extract Evaluation of Classification and Labelling Proposal with regard to Skin Sensitisation Date: 20.11.2014 Report Number: 126243-A2-050206-01 Author: Prof. Dr. Wolfgang Pfau GAB
page 1 of 13 Margosa Extract Evaluation of Classification and Labelling Proposal with regard to Skin Sensitisation Date: 20.11.2014 Report Number: 126243-A2-050206-01 Author: Prof. Dr. Wolfgang Pfau GAB
Test Report. No. CANEC Date: 05 Sep Page 1 of 7. (SVHC)
 Test Report. No. CANEC1414516501 Date: 05 Sep 2014. Page 1 of 7. SKYSILICON CO., LTD. NO.25, XI YONG AVE, SHA PING BA DISTRICT, CHONG QING, CHINA.. The following sample(s) was/were submitted and identified
Test Report. No. CANEC1414516501 Date: 05 Sep 2014. Page 1 of 7. SKYSILICON CO., LTD. NO.25, XI YONG AVE, SHA PING BA DISTRICT, CHONG QING, CHINA.. The following sample(s) was/were submitted and identified
The effect of jewel weed in preventing poison ivy dermatitis
 Journalof Wilderness Medicine 2, 178-182 (1991) ORIGINAL ARTICLE The effect of jewel weed in preventing poison ivy dermatitis B.J. ZINK, E.J. orren, M. ROSENTHAL and B. SINGAL Department of Emergency Medicine,
Journalof Wilderness Medicine 2, 178-182 (1991) ORIGINAL ARTICLE The effect of jewel weed in preventing poison ivy dermatitis B.J. ZINK, E.J. orren, M. ROSENTHAL and B. SINGAL Department of Emergency Medicine,
LESSON ASSIGNMENT. After completing this lesson, you should be able to:
 LESSON ASSIGNMENT LESSON 6 Roller Bandages. LESSON ASSIGNMENT Paragraphs 6-1 through 6-10. LESSON OBJECTIVES After completing this lesson, you should be able to: 6-1. Identify the functions of roller bandages.
LESSON ASSIGNMENT LESSON 6 Roller Bandages. LESSON ASSIGNMENT Paragraphs 6-1 through 6-10. LESSON OBJECTIVES After completing this lesson, you should be able to: 6-1. Identify the functions of roller bandages.
Test Report No.: XMHL CW Date: Sep. 04, 2017 Page 1 of 5
 Test Report No.: XMHL1708005106CW Date: Sep. 04, 2017 Page 1 of 5 HENAN GX-TECH CO., LTD CHENGDONG INDUSTRIAL PARK, LINGBAO CITY, HENAN PROVINCE The following sample(s) was/were submitted and identified
Test Report No.: XMHL1708005106CW Date: Sep. 04, 2017 Page 1 of 5 HENAN GX-TECH CO., LTD CHENGDONG INDUSTRIAL PARK, LINGBAO CITY, HENAN PROVINCE The following sample(s) was/were submitted and identified
The Percutaneous Endoscopic Gastrostomy. Geoffrey Axiak Clinical Nutrition Nurse St. Luke s Hospital
 The Percutaneous Endoscopic Gastrostomy Geoffrey Axiak Clinical Nutrition Nurse St. Luke s Hospital What is a P.E.G.? Percutaneous Endoscopic Gastrostomy Indications for P.E.G. Insertion In cases of long-term
The Percutaneous Endoscopic Gastrostomy Geoffrey Axiak Clinical Nutrition Nurse St. Luke s Hospital What is a P.E.G.? Percutaneous Endoscopic Gastrostomy Indications for P.E.G. Insertion In cases of long-term
Test Report. No. NGBHG Date: 20 Jun Page 1 of 6.
 Test Report No NGBHG1401710301 Date: 20 Jun 2014 Page 1 of 6 NINGBO AYC BIOLOGICAL TECHNOLOGY CO,LTD ROOM 7-8,BUILDING #2,YINCHEN BUSINESS CENTER,JIANGDONG DISTRICT, NINGBO,CHINA The following sample(s)
Test Report No NGBHG1401710301 Date: 20 Jun 2014 Page 1 of 6 NINGBO AYC BIOLOGICAL TECHNOLOGY CO,LTD ROOM 7-8,BUILDING #2,YINCHEN BUSINESS CENTER,JIANGDONG DISTRICT, NINGBO,CHINA The following sample(s)
SCIENTIFIC COMMITTEE ON CONSUMER PRODUCTS SCCP. Memorandum Classification and categorization of skin sensitisers and grading of test reactions
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Public Health and Risk Assessment C7 - Risk assessment SCIENTIFIC COMMITTEE ON CONSUMER PRODUCTS SCCP Memorandum Classification
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Public Health and Risk Assessment C7 - Risk assessment SCIENTIFIC COMMITTEE ON CONSUMER PRODUCTS SCCP Memorandum Classification
Test Report. No. CANHG A01 Date: 23 Jun Page 1 of 7.
 Test Report. No. CANHG1610790608 A01 Date: 23 Jun 2016. Page 1 of 7. SHANGHAI RICH CHEMICAL NEW MATERIAL CO.,LTD. NO. 18 BUILDING,YUXIU ROAD 136,SONGJIANG,SHANGHAI,CHINA. This report is to supersede test
Test Report. No. CANHG1610790608 A01 Date: 23 Jun 2016. Page 1 of 7. SHANGHAI RICH CHEMICAL NEW MATERIAL CO.,LTD. NO. 18 BUILDING,YUXIU ROAD 136,SONGJIANG,SHANGHAI,CHINA. This report is to supersede test
Test Report. No. CANEC Date: 17 Apr Page 1 of 6.
 Test Report. No. CANEC1203982501 Date: 17 Apr 2012. Page 1 of 6. COOKLINE. PFÄLZER RING 5.D-68766 HOCKENHEIM, GERMANY... The following sample(s) was/were submitted and identified on behalf of the clients
Test Report. No. CANEC1203982501 Date: 17 Apr 2012. Page 1 of 6. COOKLINE. PFÄLZER RING 5.D-68766 HOCKENHEIM, GERMANY... The following sample(s) was/were submitted and identified on behalf of the clients
SKIN IRRITATION AND SKIN SENSITIZATION STUDY FOR POLYMERIC AND ADHESIVE MATRIX DIFFUSIONAL TRANSDERMAL DRUG DELIVERY DEVICE OF ATENOLOL
 ISSN: 223-7346 B. Venkateswara Reddy et al. / Journal of Global Trends in Pharmaceutical Sciences 5(2)-(214) 173 1736 (Research Article) Journal of Global Trends in Pharmaceutical Sciences Journal home
ISSN: 223-7346 B. Venkateswara Reddy et al. / Journal of Global Trends in Pharmaceutical Sciences 5(2)-(214) 173 1736 (Research Article) Journal of Global Trends in Pharmaceutical Sciences Journal home
CAVIWIPES and CAVIWIPES XL
 CAVIWIPES and CAVIWIPES XL Technical Bulletin CaviWipes and CaviWipes XL Disinfecting Towelettes are non-woven disposable towelettes pre-saturated with CaviCide. CaviWipes and CaviWipes XL are intended
CAVIWIPES and CAVIWIPES XL Technical Bulletin CaviWipes and CaviWipes XL Disinfecting Towelettes are non-woven disposable towelettes pre-saturated with CaviCide. CaviWipes and CaviWipes XL are intended
Guinea Pig Maximization Test for Trichloroethylene and Its Metabolites 1
 BIOMEDICAL AND ENVIRONMENTAL SCIENCES 15, 113-118 (2002) Guinea Pig Maximization Test for Trichloroethylene and Its Metabolites 1 TANG XIAO-JIANG #, +, LI LAI-YU #, HUANG JIAN-XUN #, AND DENG YING-YU #
BIOMEDICAL AND ENVIRONMENTAL SCIENCES 15, 113-118 (2002) Guinea Pig Maximization Test for Trichloroethylene and Its Metabolites 1 TANG XIAO-JIANG #, +, LI LAI-YU #, HUANG JIAN-XUN #, AND DENG YING-YU #
Test Report. No. SHAHG Date: 25 Aug Page 1 of 5.
 Test Report No Date: 25 Aug 2016 Page 1 of 5 WUHAN XINYONGAN PAPER PLASTIC CO, LTD NO16 FENGHUANG ROAD, FENGHUANGSHAN INDUSTRIAL RAPK, CAIDIAN DISTRICT, WUHAN, CHINA The following sample(s) was/were submitted
Test Report No Date: 25 Aug 2016 Page 1 of 5 WUHAN XINYONGAN PAPER PLASTIC CO, LTD NO16 FENGHUANG ROAD, FENGHUANGSHAN INDUSTRIAL RAPK, CAIDIAN DISTRICT, WUHAN, CHINA The following sample(s) was/were submitted
Test Report. No. CANEC Date: 31 Oct Page 1 of 7. (SVHC)
 Test Report. No. CANEC1417334501 Date: 31 Oct 2014. Page 1 of 7. DONG GUAN BETTER ELECTRONICS TECHNOLOGY CO.,LTD. NAN MEN VILLAGE,HENGKEN ADMINISTRATIVE ZONE,LIAOBU TOWN,DONGGUAN,GUANGDONG CHINA.. The
Test Report. No. CANEC1417334501 Date: 31 Oct 2014. Page 1 of 7. DONG GUAN BETTER ELECTRONICS TECHNOLOGY CO.,LTD. NAN MEN VILLAGE,HENGKEN ADMINISTRATIVE ZONE,LIAOBU TOWN,DONGGUAN,GUANGDONG CHINA.. The
Monograph. Addendum 1. Ferric phosphate. Ecotoxicology. Rapporteur Member State: Germany. 26 May 2000
 11097/ECCO/BBA/00 Addendum 1 to draft assessment report (6613/ECCO/PSD/99) Monograph Addendum 1 26 May 2000 Ferric phosphate Ecotoxicology Rapporteur Member State: Germany 2/3 Addendum B.09.06 Effects
11097/ECCO/BBA/00 Addendum 1 to draft assessment report (6613/ECCO/PSD/99) Monograph Addendum 1 26 May 2000 Ferric phosphate Ecotoxicology Rapporteur Member State: Germany 2/3 Addendum B.09.06 Effects
Table of Contents. Dialysis Port Care Chemotherapy Port Care G-Tube Care Colostomy Bags Wound Dressings
 Table of Contents Dialysis Port Care Chemotherapy Port Care G-Tube Care Colostomy Bags Wound Dressings Dialysis Port Care Know What Type of Vascular Access You Have. Fistula: An artery in your forearm
Table of Contents Dialysis Port Care Chemotherapy Port Care G-Tube Care Colostomy Bags Wound Dressings Dialysis Port Care Know What Type of Vascular Access You Have. Fistula: An artery in your forearm
DISPERSE RED 17 OPINION OF THE SCIENTIFIC COMMITTEE ON COSMETIC PRODUCTS AND NON-FOOD PRODUCTS INTENDED FOR CONSUMERS. SCCNFP/0677/03, final
 OPINION OF THE SCIENTIFIC COMMITTEE ON COSMETIC PRODUCTS AND NON-FOOD PRODUCTS INTENDED FOR CONSUMERS CONCERNING DISPERSE RED 17 COLIPA n B5 adopted by the SCCNFP during the 24 th plenary meeting of 24-25
OPINION OF THE SCIENTIFIC COMMITTEE ON COSMETIC PRODUCTS AND NON-FOOD PRODUCTS INTENDED FOR CONSUMERS CONCERNING DISPERSE RED 17 COLIPA n B5 adopted by the SCCNFP during the 24 th plenary meeting of 24-25
Comparison of the Sensitivities of the Buehler Test and the Guinea Pig Maximization Test for Predictive Testing of Contact Allergy
 Acta Derm Venereol 2000; 80: 256±262 INVESTIGATIVE REPORT Comparison of the Sensitivities of the Buehler Test and the Guinea Pig Maximization Test for Predictive Testing of Contact Allergy SéREN FRANKILD
Acta Derm Venereol 2000; 80: 256±262 INVESTIGATIVE REPORT Comparison of the Sensitivities of the Buehler Test and the Guinea Pig Maximization Test for Predictive Testing of Contact Allergy SéREN FRANKILD
Test Report. No. NGBHG Date: 06 May Page 1 of 6.
 Test Report No NGBHG1300658901 Date: 06 May 2013 Page 1 of 6 NINGBO HAIHUAN RUBBER INDUSTRY INDUSTRY CO,LTD SOUTH ROAD NO183,XIDIAN TOWN,NINGHAI,NINGBO,CHINA The following sample(s) was/were submitted
Test Report No NGBHG1300658901 Date: 06 May 2013 Page 1 of 6 NINGBO HAIHUAN RUBBER INDUSTRY INDUSTRY CO,LTD SOUTH ROAD NO183,XIDIAN TOWN,NINGHAI,NINGBO,CHINA The following sample(s) was/were submitted
At the conclusion of this course the learner will be able to
 Objectives At the conclusion of this course the learner will be able to 1. Discuss basic anatomy and pathophysiology of burns 2. Describe burn injuries in terms of size, depth, coloration and characteristics
Objectives At the conclusion of this course the learner will be able to 1. Discuss basic anatomy and pathophysiology of burns 2. Describe burn injuries in terms of size, depth, coloration and characteristics
Alcohol Ethoxysulphates Human Health Risk Assessment
 Human & Environmental Risk Assessment on ingredients of European household cleaning products Alcohol Ethoxysulphates Human Health Risk Assessment January 2003 All rights reserved. No part of this publication
Human & Environmental Risk Assessment on ingredients of European household cleaning products Alcohol Ethoxysulphates Human Health Risk Assessment January 2003 All rights reserved. No part of this publication
Test Report. No. CANHG Date: 16 Apr Page 1 of 6.
 Test Report. No. CANHG1504584901 Date: 16 Apr 2015. Page 1 of 6. CHUANGXIN RUBBER PLASTIC & METAL CO., LTD.. NO.1 HUASHENG RD., XINGHUA INDUSTRIAL, ZONE, RONGGUI, SHUNDE FOSHAN GUANGDONG CHINA.. The following
Test Report. No. CANHG1504584901 Date: 16 Apr 2015. Page 1 of 6. CHUANGXIN RUBBER PLASTIC & METAL CO., LTD.. NO.1 HUASHENG RD., XINGHUA INDUSTRIAL, ZONE, RONGGUI, SHUNDE FOSHAN GUANGDONG CHINA.. The following
SAFETY DATA SHEET. NFPA Rating: Health: 1 Flammability: 3 Reactivity: 0 Special: 0
 åallergan SAFETY DATA SHEET NFPA Rating: Health: 1 Flammability: 3 Reactivity: 0 Special: 0 TELEPHONE CONTACTS: Product Technical and Medical Information: (800) 433-8871 Transportation Emergency 24-Hour
åallergan SAFETY DATA SHEET NFPA Rating: Health: 1 Flammability: 3 Reactivity: 0 Special: 0 TELEPHONE CONTACTS: Product Technical and Medical Information: (800) 433-8871 Transportation Emergency 24-Hour
WHEN THERE S A NEED FOR EPIDERMIS
 WHEN THERE S A NEED FOR EPIDERMIS PRESS START The automated CELLUTOME Epidermal Harvesting System The CELLUTOME System is an innovative epidermal harvesting device which can be easily integrated into your
WHEN THERE S A NEED FOR EPIDERMIS PRESS START The automated CELLUTOME Epidermal Harvesting System The CELLUTOME System is an innovative epidermal harvesting device which can be easily integrated into your
Professional Wound Management
 Professional Wound Management Bioteque Corporation is the first bio-technology company in Taiwan engaged in the intensive R&D activity and manufacturer of diversified high-end medical products. Founded
Professional Wound Management Bioteque Corporation is the first bio-technology company in Taiwan engaged in the intensive R&D activity and manufacturer of diversified high-end medical products. Founded
The Power of a Hydroconductive Wound Dressing with LevaFiber Technology
 The Power of a Hydroconductive Wound Dressing with LevaFiber Technology The first step in healing a chronic wound is to detoxify it by removing slough, necrotic tissue, exudate and bacteria, while keeping
The Power of a Hydroconductive Wound Dressing with LevaFiber Technology The first step in healing a chronic wound is to detoxify it by removing slough, necrotic tissue, exudate and bacteria, while keeping
ULTRASONIC DEVICE IN BONE CUTTING
 Acta orthop. scand. 52, 5-10, 1981 ULTRASONIC DEVICE IN BONE CUTTING A Histological and Scanning Electron Microscopical Study H. ARO*, H. KALLIONIEMI**, A. J. AHO* & P. KELLOKUMPU-LEHTINEN*** *Department
Acta orthop. scand. 52, 5-10, 1981 ULTRASONIC DEVICE IN BONE CUTTING A Histological and Scanning Electron Microscopical Study H. ARO*, H. KALLIONIEMI**, A. J. AHO* & P. KELLOKUMPU-LEHTINEN*** *Department
Chapter MEMBRANE TRANSPORT
 Chapter 3 I MEMBRANE TRANSPORT The cell membrane, or plasma membrane, is the outermost layer of the cell. It completely surrounds the protoplasm or living portion of the cell, separating the cell s interior
Chapter 3 I MEMBRANE TRANSPORT The cell membrane, or plasma membrane, is the outermost layer of the cell. It completely surrounds the protoplasm or living portion of the cell, separating the cell s interior
Cambridge International Examinations Cambridge International General Certificate of Secondary Education
 www.xtremepapers.com Cambridge International Examinations Cambridge International General Certificate of Secondary Education *6508111556* BIOLOGY 0610/21 Paper 2 Core May/June 2014 1 hour 15 minutes Candidates
www.xtremepapers.com Cambridge International Examinations Cambridge International General Certificate of Secondary Education *6508111556* BIOLOGY 0610/21 Paper 2 Core May/June 2014 1 hour 15 minutes Candidates
PART 2. Toxicological Studies and Exposure Data and Information
 - A8/19 - Appendi 8 Format for the Compilation of Tier II Summaries Formulated product PART 2 Section 3 Toicological Studies and Eposure The eample of a summary and assessment of data which follows is
- A8/19 - Appendi 8 Format for the Compilation of Tier II Summaries Formulated product PART 2 Section 3 Toicological Studies and Eposure The eample of a summary and assessment of data which follows is
Risk Assessment Report on Tris (nonylphenyl)phosphite (TNPP)
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Public Health and Risk Assessment C7 - Risk assessment SCIENTIFIC COMMITTEE ON HEALTH AND ENVIRONMENTAL RISKS SCHER
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Public Health and Risk Assessment C7 - Risk assessment SCIENTIFIC COMMITTEE ON HEALTH AND ENVIRONMENTAL RISKS SCHER
Chemical Entity Cas No Proportion
 1. Identification of the Substance/Mixture and Supplier. Product name: Application: ERMA approval: Company: Telephone: Facsimile: Emergency telephone: Profender Spot on treatment for worms in cats HSR176
1. Identification of the Substance/Mixture and Supplier. Product name: Application: ERMA approval: Company: Telephone: Facsimile: Emergency telephone: Profender Spot on treatment for worms in cats HSR176
Test Report. No. SHAHG Date: 05 May Page 1 of 5.
 Test Report No Date: 05 May 2016 Page 1 of 5 RTCO (HK) CO,LIMITED ROOM 20A,KIU FU COMMERCIAL BUILDING,300 LOCKHART ROAD,WAN CHAI,HONG KONG The following sample(s) was/were submitted and identified on behalf
Test Report No Date: 05 May 2016 Page 1 of 5 RTCO (HK) CO,LIMITED ROOM 20A,KIU FU COMMERCIAL BUILDING,300 LOCKHART ROAD,WAN CHAI,HONG KONG The following sample(s) was/were submitted and identified on behalf
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL. Directorate C - Public Health and Risk Assessment C7 - Risk assessment SCCP
 SCCP098406 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Public Health and Risk Assessment C7 - Risk assessment SCIENTIFIC COMMITTEE ON CONSUMER PRODUCTS SCCP Opinion
SCCP098406 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Public Health and Risk Assessment C7 - Risk assessment SCIENTIFIC COMMITTEE ON CONSUMER PRODUCTS SCCP Opinion
Test Report. No. CANEC Date: 05 Jan Page 1 of 7. (SVHC)
 Test Report. No. CANEC1522020801 Date: 05 Jan 2016. Page 1 of 7. YUNNAN TIN CO.,LTD.. 49#MIDDLE OF CHANGYUAN ROAD,KUNMING NATIONAL HIGH&NEW TECH INDUSTRY DEVELOPMENT ZONE,KUNMING,YUNNAN CHINA.. The following
Test Report. No. CANEC1522020801 Date: 05 Jan 2016. Page 1 of 7. YUNNAN TIN CO.,LTD.. 49#MIDDLE OF CHANGYUAN ROAD,KUNMING NATIONAL HIGH&NEW TECH INDUSTRY DEVELOPMENT ZONE,KUNMING,YUNNAN CHINA.. The following
PART 3. Toxicological and Metabolism Studies on the Active Substance. Absorption, distribution, metabolism and excretion (ADME) in the rat
 - A7/31 - Appendix 7 Format for the Compilation of Tier II Summaries Active substance PART 3 Section 3 Toxicological and Metabolism Studies The example of a summary and assessment of data which follows
- A7/31 - Appendix 7 Format for the Compilation of Tier II Summaries Active substance PART 3 Section 3 Toxicological and Metabolism Studies The example of a summary and assessment of data which follows
Final Report. Acute dermal toxicity study in Wistar rats. Mr. M. Vasanthan, M Tech (Biotech)
 Study Title Test Item Study Director Sponsor Study Monitor Test Facility Acute dermal toxicity study in Wistar rats Nualgi Nano Nutrients Mr. M. Vasanthan, M Tech (Biotech) Nualgi Nano Biotech Co 651,
Study Title Test Item Study Director Sponsor Study Monitor Test Facility Acute dermal toxicity study in Wistar rats Nualgi Nano Nutrients Mr. M. Vasanthan, M Tech (Biotech) Nualgi Nano Biotech Co 651,
CAVIWIPES. Technical Bulletin
 CAVIWIPES Technical Bulletin CaviWipes Disinfecting Towelettes are non-woven disposable towelettes pre-saturated with CaviCide. CaviWipes are intended for use in health care settings such as hospitals,
CAVIWIPES Technical Bulletin CaviWipes Disinfecting Towelettes are non-woven disposable towelettes pre-saturated with CaviCide. CaviWipes are intended for use in health care settings such as hospitals,
Why You Should Choose In Light Wellness Systems. In Light Wellness Systems. Shining light on:
 Shining light on: CLEARED brilliant light therapy Why You Should Choose What is it? Who is it for? Why is it different? Why should you be interested? In Light W E L L N E S S S Y S T E M S facebook.com/ilwsystems
Shining light on: CLEARED brilliant light therapy Why You Should Choose What is it? Who is it for? Why is it different? Why should you be interested? In Light W E L L N E S S S Y S T E M S facebook.com/ilwsystems
Tel. : / Fax :
 Service Best service for best customers Management BRT Corporation cares for you through reliable sales management Service Management Satisfaction Being confident of its products, BRT corporation Quality
Service Best service for best customers Management BRT Corporation cares for you through reliable sales management Service Management Satisfaction Being confident of its products, BRT corporation Quality
DIMETHENAMID-P/RACEMIC DIMETHENAMID
 189 DIMETHENAMID-P/RACEMIC DIMETHENAMID First draft prepared by D.B. McGregor 1 and Roland Solecki 2 1 Toxicity Evaluation Consultants, Aberdour, Scotland; and 2 Safety of Substances and Preparations Coordination
189 DIMETHENAMID-P/RACEMIC DIMETHENAMID First draft prepared by D.B. McGregor 1 and Roland Solecki 2 1 Toxicity Evaluation Consultants, Aberdour, Scotland; and 2 Safety of Substances and Preparations Coordination
METRICIDE OPA PLUS SOLUTION. Technical Bulletin
 METRICIDE OPA PLUS SOLUTION Technical Bulletin MetriCide OPA Plus is a 0.60% ortho-phthalaldehyde solution that can be used for reprocessing heat sensitive semi-critical medical devices for which sterilization
METRICIDE OPA PLUS SOLUTION Technical Bulletin MetriCide OPA Plus is a 0.60% ortho-phthalaldehyde solution that can be used for reprocessing heat sensitive semi-critical medical devices for which sterilization
January Adult Burn Injured patients
 Guideline Full Title of Guideline: Author (include email and role): Division & Speciality: Scope (Target audience, state if Trust wide): Review date (when this version goes out of date): Explicit definition
Guideline Full Title of Guideline: Author (include email and role): Division & Speciality: Scope (Target audience, state if Trust wide): Review date (when this version goes out of date): Explicit definition
Protocol for protein SDS PAGE and Transfer
 Protocol for protein SDS PAGE and Transfer According to Laemmli, (1970) Alaa El -Din Hamid Sayed, Alaa_h254@yahoo.com Serum Selection of a protein source cell cultures (bacteria, yeast, mammalian, etc.)
Protocol for protein SDS PAGE and Transfer According to Laemmli, (1970) Alaa El -Din Hamid Sayed, Alaa_h254@yahoo.com Serum Selection of a protein source cell cultures (bacteria, yeast, mammalian, etc.)
Technical Report No. 78. Skin Sensitisation Testing: Methodological Considerations
 ECETOC Technical Report No. 78 Skin Sensitisation Testing: Methodological Considerations European Centre for Ecotoxicology Avenue E. Van Nieuwenhuyse 4, (Bte 6) and Toxicology of Chemicals B-1160 Brussels,
ECETOC Technical Report No. 78 Skin Sensitisation Testing: Methodological Considerations European Centre for Ecotoxicology Avenue E. Van Nieuwenhuyse 4, (Bte 6) and Toxicology of Chemicals B-1160 Brussels,
BASIC VIOLET 2 OPINION OF THE SCIENTIFIC COMMITTEE ON COSMETIC PRODUCTS AND NON-FOOD PRODUCTS INTENDED FOR CONSUMERS SCCNFP/0784/04.
 OPINION OF THE SCIENTIFIC COMMITTEE ON COSMETIC PRODUCTS AND NONFOOD PRODUCTS INTENDED FOR CONSUMERS CONCERNING BASIC VIOLET 2 COLIPA n B115 adopted by the SCCNFP on 23 April 2004 by means of the written
OPINION OF THE SCIENTIFIC COMMITTEE ON COSMETIC PRODUCTS AND NONFOOD PRODUCTS INTENDED FOR CONSUMERS CONCERNING BASIC VIOLET 2 COLIPA n B115 adopted by the SCCNFP on 23 April 2004 by means of the written
Nephrostomy Tube Care
 Nephrostomy Tube Care CEAC 0585 January 2012 Problems to report to the doctor If your nephrostomy tube falls out, call your urologist or go to a hospital Emergency Department immediately to have the tube
Nephrostomy Tube Care CEAC 0585 January 2012 Problems to report to the doctor If your nephrostomy tube falls out, call your urologist or go to a hospital Emergency Department immediately to have the tube
Wound Management for Nurses/Technicians What do we need to know?
 Wound Management for Nurses/Technicians What do we need to know? Laura Owen European Specialist in Small Animal Surgery Lecturer in Small Animal Surgery, University of Cambridge The Acute Open Wound PPE
Wound Management for Nurses/Technicians What do we need to know? Laura Owen European Specialist in Small Animal Surgery Lecturer in Small Animal Surgery, University of Cambridge The Acute Open Wound PPE
TRIS (NONYLPHENYL) PHOSPHITE SUMMARY RISK ASSESSMENT REPORT
 TRIS (NONYLPHENYL) PHOSPHITE CAS-No.: 26523-78-4 EINECS-No.: 247-759-6 SUMMARY RISK ASSESSMENT REPORT Final report, 2007 France Rapporteur for the risk assessment of tris(nonylphenyl) phosphite is the
TRIS (NONYLPHENYL) PHOSPHITE CAS-No.: 26523-78-4 EINECS-No.: 247-759-6 SUMMARY RISK ASSESSMENT REPORT Final report, 2007 France Rapporteur for the risk assessment of tris(nonylphenyl) phosphite is the
Skin Integrity and Wound Care
 Skin Integrity and Wound Care By Dr. Amer Hasanien & Dr. Ali Saleh Skin Integrity and Wound Care Skin integrity: the presence of normal Skin & Uninterrupted skin layers by wounds. Factors affecting appearance
Skin Integrity and Wound Care By Dr. Amer Hasanien & Dr. Ali Saleh Skin Integrity and Wound Care Skin integrity: the presence of normal Skin & Uninterrupted skin layers by wounds. Factors affecting appearance
Home Care for Your Nephrostomy Catheter - The James
 PATIENT EDUCATION patienteducation.osumc.edu Home Care for Your Nephrostomy Catheter - The James This handout tells you how to care for your nephrostomy catheter. If you have any questions about this information,
PATIENT EDUCATION patienteducation.osumc.edu Home Care for Your Nephrostomy Catheter - The James This handout tells you how to care for your nephrostomy catheter. If you have any questions about this information,
Inspirational chemistry 97. Index sheets. Rhubarb contains oxalic acid, which has the formula C H 3. O + 2 Mn CO 2
 Inspirational chemistry 97 Rates and rhubarb Index 4.1.3 3 sheets Rhubarb contains oxalic acid, which has the formula C 2 H 2 O 4 : O OH C C HO Figure 1 Structural formula of oxalic acid Oxalic acid reacts
Inspirational chemistry 97 Rates and rhubarb Index 4.1.3 3 sheets Rhubarb contains oxalic acid, which has the formula C 2 H 2 O 4 : O OH C C HO Figure 1 Structural formula of oxalic acid Oxalic acid reacts
PROPAMOCARB. First draft prepared by G. Wolterink 1 and M. Tasheva 2
 417 PROPAMOCARB First draft prepared by G. Wolterink 1 and M. Tasheva 2 1 Centre for Substances and Integrated Risk Assessment, National Institute for Public Health and the Environment, Bilthoven, Netherlands;
417 PROPAMOCARB First draft prepared by G. Wolterink 1 and M. Tasheva 2 1 Centre for Substances and Integrated Risk Assessment, National Institute for Public Health and the Environment, Bilthoven, Netherlands;
ISOXAFLUTOLE. First draft prepared by P.V. Shah 1 and Roland Alfred Solecki 2. America (USA) Federal Institute for Risk Assessment, Berlin, Germany
 ISOXAFLUTOLE First draft prepared by P.V. Shah 1 and Roland Alfred Solecki 2 1 Office of Pesticide Programs, Environmental Protection Agency, Washington, DC, United States of America (USA) 2 Federal Institute
ISOXAFLUTOLE First draft prepared by P.V. Shah 1 and Roland Alfred Solecki 2 1 Office of Pesticide Programs, Environmental Protection Agency, Washington, DC, United States of America (USA) 2 Federal Institute
TECHNICAL DATASHEET POUR TYPE ACRYLIC RESIN PDFTPT-082
 TCHNICAL DATASHT POUR TYP ACRYLIC RSIN 1 PRODUCT OVRVIW Methacrylate polymers have enjoyed great popularity in dentistry because they are easily processed by using relatively simple techniques; they have
TCHNICAL DATASHT POUR TYP ACRYLIC RSIN 1 PRODUCT OVRVIW Methacrylate polymers have enjoyed great popularity in dentistry because they are easily processed by using relatively simple techniques; they have
Section 1: Identification of the Substance/Mixture and of the Supplier
 SAFETY DATA SHEET Section 1: Identification of the Substance/Mixture and of the Supplier Product Name: SOLDERING FLUX Proper Shipping Name Zinc chloride solution Recommended use: Soldering Flux Company
SAFETY DATA SHEET Section 1: Identification of the Substance/Mixture and of the Supplier Product Name: SOLDERING FLUX Proper Shipping Name Zinc chloride solution Recommended use: Soldering Flux Company
5.15 HEXYTHIAZOX (176)
 Hexythiazox 225 5.15 HEXYTHIAZOX (176) TOXICOLOGY Hexythiazox is the ISO approved name for (trans-5-(4-chlorophenyl)-n-cyclohexyl-4-methyl-2-oxo- 3-thiazolidine-carboxamide (CAS No. 78587-05-0). Hexythiazox
Hexythiazox 225 5.15 HEXYTHIAZOX (176) TOXICOLOGY Hexythiazox is the ISO approved name for (trans-5-(4-chlorophenyl)-n-cyclohexyl-4-methyl-2-oxo- 3-thiazolidine-carboxamide (CAS No. 78587-05-0). Hexythiazox
