special interest since changes in the activity of these enzymes are known hypophysial hormones, notably those stimulating the thyroid gland and the
|
|
|
- Kristopher Cody Price
- 5 years ago
- Views:
Transcription
1 EFFECT OF HYPOPHYSECTOMY OR TREATMENT WITH THY- ROXINE OR TESTOSTERONE ON SECRETORY RESPONSES AND RESPIRATORY ENZYMES OF THE SUBMAXILLARY GLAND OF THE RAT. By P. OHLIN. From the Institute of Physiology, University of Lund, Sweden. (Received for publication 19th March 1965) The effect of hypophysectomy or of treatment with thyroxine or testosterone on the function of the submaxillary gland of rats was studied. (1) The dry weight of the submaxillary gland decreased by 46 per cent after hypophysectomy. The gland weight increased by 14 per cent after thyroxine and by 29 per cent after testosterone. (2) The maximal secretory response to pilocarpine was reduced after hypophysectomy. After hormonal treatment the maximal secretory response was increased when expressed per gland. It was also increased when calculated per unit weight after thyroxine, but not after testosterone. (3) The activity of succinic dehydrogenase, cytochrome oxidase and fumarase was not significantly changed after hormonal treatment when expressed per unit weight. The results are discussed and related to known morphological changes of the submaxillary gland of rats after hypophysectomy or after treatment with thyroxine or testosterone. THE relationship between hormones and salivary glands has attracted much attention since the discovery of the sexual dimorphism of the submaxillary gland of mice [Lacassagne, 194 a]. Most work in this field was done on mice and rats. The structure of the gland was found to be dependent on hypophysial hormones, notably those stimulating the thyroid gland and the gonads. Thus, the submaxillary gland of rats atrophied markedly after hypophysectomy [Gabe, 195; Sreebny, 1953], thyroidectomy [Leblond and Grad, 1948] or gonadectomy [Shafer and Muhler, 1953]. The atrophy concerned mainly the cells of the granular tubules. Treatment with thyroxine and testosterone prevented atrophic changes [Grad, 1949; Shafer et al., 1956]. Furthermore, the granular tubules increased in size when intact rats were treated with thyroid hormones or testosterone [Lacassagne, 194 b; Shafer and Muhler, 1953 and 1956]. To what extent these structural changes indicate modifications of the function of the submaxillary gland of rats is not known. In order to study functional aspects a method was used which made it possible to record the secretory responses to sialagogue drugs. As another indicator of the functional capacity, the activity of some respiratory enzymes, succinic dehydrogenase, cytochrome oxidase and fumarase, was studied. This seemed of special interest since changes in the activity of these enzymes are known to occur concomitantly with structural changes, e.g. after parasympathetic denervation [Nordenfelt et al., 196] and after hypophysectomy [Ohlin, 1962]. Since the parasympathetic nerve supply is important for the function of salivary glands some studies were also made in order to see whether parasympathetic denervation influenced the effects of hormonal treatment. 446
2 Hormonal Influences on -Rat's Submaxillary Gland 447 METHODS Female rats of a colony bred at this Institute were used. The breeding animals received a pelleted diet (Anticimex, 21); the experimental animals were given milk, wheat bread and barley ad libitum. Litters of four to six rats, when aged 4 to 5 months, were used; two rats of each litter were taken as controls. Three groups of animals were studied. In one group the rats were hypophysectomized and examined about 4 weeks later. Completeness of hypophysectomy was controlled as described by Jacobsohn [196]. The results after hypophysectomy refer to eleven rats. In these animals microscopic examination did not reveal any hypophysial cells other than small pars tuberalis cells lining the pituitary stalk. In two other groups intact rats were treated with either DL-thyroxine, 1,ug. daily subcutaneously, or testosterone propionate, -2 mg. in oil daily intramuscularly, for 3 weeks. Twenty-four rats were given thyroxine and thirty-two testosterone. Studies were performed on the submaxillary gland of different groups of rats as follows: (1) after hypophysectomy or hormonal treatment the secretory responses to sialagogue drugs were estimated; (2) after hormonal treatment the activity of respiratory enzymes was studied. The effect of hypophysectomy on enzymic activity had been investigated previously [Ohlin, 1962]; (3) the effect of hormonal treatment on parasympathetically denervated glands was studied. (1) To estimate the secretory responses of the submaxillary gland the following technique was developed. The rats were anaesthetized with ether and a thin polythene tube connected to an injection needle was inserted into an exposed femoral vein. Chloralose (1 mg./kg. body weight) was then given intravenously and the ether administration was discontinued. After intubation of the trachea the salivary ducts were exposed in the neck. After opening the submaxillary duct, medial to the sublingual duct, a fine glass cannula was inserted. Cannulse giving 8 drops out of 1 ml. of distilled water were used. Secretion appearing at the tip of the cannula was noted and registered on a smoked drum. The following drugs were injected intravenously to determine a threshold dose: acetylcholine chloride, -2-2 pg./kg.; methacholine chloride, -2-1,ug./kg.; and adrenaline chloride, -2-2,ug./kg., and the flow of saliva was recorded in each case for the doses above threshold. To record a maximal secretory response, pilocarpine was injected intravenously every 3-6 sec. in increasing doses from 5-1 and finally 2 jug./kg. The flow of saliva was recorded, using an ordinate recorder on a smoked drum. The maximal secretory response was usually reached within 1-15 min. after a total dose of about 2 mg. pilocarpine/kg. The maximal secretory response after pilocarpine was calculated and expressed in u1l. saliva per min. per gland or per mg. of glandular tissue (dry weight). To make this calculation possible the volume of one drop of saliva was determined. For this purpose saliva was collected when the gland was secreting at a maximal rate. The saliva was transferred as soon as possible from a pipette through the cannula used, the rate being kept equal to the maximal flow rate of the gland. At the end of the experiments on secretory responses to sialagogue drugs including pilocarpine, the submaxillary glands were removed, cleaned and weighed (wet weight). The dry weight of the glands was determined after heating to C. for 48 hr. The wet weight of the right submaxillary gland had previously been found to be the same as that of the left gland [Ohlin, 1962]. In six rats the left submaxillary gland was excised in advance, and the right gland was extirpated after estimation of secretory responses. The wet weight of the right gland exceeded that of the left one in all these animals, the average difference being 13 per cent. The dry weight of the right gland was, on the other hand, about the same as that of the left gland, 98±4-2 (6) * per cent. The difference observed in the wet weight was probably due to a * Mean ± standard error of mean (number of observations)
3 448 IOhlin slight cedema of the right gland after secretory activity. For this reason the gland weight is given as the dry weight after experiments on secretory responses. (2) For the enzymic estimations the animals were killed by cervical dislocation. The submaxillary glands were immediately dissected out, cleaned, dried between filter papers, weighed and homogenized in redistilled water. The activity of succinic dehydrogenase and cytochrome oxidase was estimated manometrically, and that of fumarase was determined spectrophotometrically [Nordenfelt et al., 196; Ohlin, 1962]. The activity of respiratory enzymes is expressed in units per gland, which indicates enzyme changes irrespective of weight changes, and in units per g. of glandular tissue (wet weight). For succinic dehydrogenase and cytochrome oxidase, 1 unit of activity corresponds to a utilization of 1,sl. oxygen in 3 min. at 37 C. For fumarase, 1 unit is defined as the amount of enzyme causing a change in optical density of.1/min. at 24/m,. (3) To study the effect of treatment with thyroxine or testosterone on parasympathetically denervated glands, the chorda-lingual nerve was cut under ether anawsthesia. The hormonal treatment was started on the day of denervation and continued for 3 weeks. The secretory responses to sialagogue drugs and the dry weight of the glands were estimated as described above. The effect of parasympathetic denervation will be described separately in detail [Ohlin, 1965]. RESULTS Gland Weight. - Within 4 weeks after hypophysectomy, the dry weight of the submaxillary gland decreased markedly. After treatment with thyroxine or testosterone, on the other hand, the dry weight increased (Table I). The decrease in gland weight after hypophysectomy and the increase after hormonal treatment were significant (P < -1). Similar changes of the wet weight were found after experiments on enzymic estimations. After treatment with thyroxine or testosterone the wet weight of the submaxillary gland increased from 153 h 4.2 (16) mg. to 175 i 3.9 (16) mg. after thyroxine and to (21) mg. after testosterone. The changes in gland weight after hypophysectomy and hormonal treatment were marked even when related to body weight (Table I). At 4 weeks after hypophysectomy the dry weight of the submaxillary gland had decreased by 46 per cent, but the body weight was reduced by 14 per cent only. After treatment with thyroxine the dry weight of the glands was 14 per cent higher than in the controls; the body weight was increased by 6 per cent less than that of the controls. After treatment with testosterone the body weight increased by 5 per cent. The dry weight of the glands increased 29 per cent more than in the controls. Secretory Responses. - The threshold doses of acetylcholine, methacholine and adrenaline in the different control glands can be seen in Table II. When the doses of methacholine and adrenaline were increased above threshold a marked secretory response was evoked. The secretory response to methacholine, 1 pug./kg., was about one to three drops, and that to adrenaline, 2 pug./kg., was about one to two drops. Acetylcholine, on the other hand, evoked small secretory responses, usually less than one drop of saliva, even when large doses were given. Pilocarpine elicited a marked flow of saliva
4 Hormonal Influences on Rat's Submaxillary Gland 449 ceglo o, oq g- p4e4 Ei ' m WVtV 4-- cq tn Em E E'o. 1 Q ^ ]Weq eq e mxoo t4-1~ 6 eq c E-h4 Z11 e 4 Pr Ho eq H - m -I -X4 - e Meq - eq eo ~~o m -o m PEO i-4. Fi- eq eq - -
5 45 Ohlin, which usually started after 5 pug. pilocarpine/kg. The maximal secretory response of a control gland to pilocarpine was (24) jul. per min. or, expressed in,ul. per min. per mg. (dry weight),.46 E.31 (Table I). The effect of hypophysectomy on the threshold doses of the secretory drugs. studied can be seen in Table II. The threshold dose of acetylcholine seemed slightly increased, whereas the threshold doses of methacholine and adrenaline were not different from those of the controls. The effect of hypophysectomy on the secretory responses to doses above threshold was most clearly seen when the maximal secretory response to pilocarpine was determined. Under TABLE II. THRESHOLD DosEs (pg./kg.) OF ACETYCHOLINE (ACH), METHACHOLINE (MECH) AND ADRENALINE (ADR) IN THE SUBMAXILLARY GLAND; (1) OF UNTREATED RATS (=NTROL); (2) AFTER HYPOPHYSECTOMY; (3) OF INTACT RATS GIVEN THYROXINE OR; (4) TESTOSTERONE. THE FIGURES GIVE THL NUMBER OF RATS SHOWING THE INDICATED THRESHOLD DOSE. Control Hypophysectomy Thyroxine Testosterone ACh * *.. 4 MeCh * Adr 1* * these conditions a reduction by 56 per cent was found when the maximal secretory response was expressed per gland. When calculated per unit weight the decrease was 17 per cent, which was not significant (Table I). After treatment with thyroxine the threshold doses of acetylcholine and methacholine seemed slightly decreased (Table II). The threshold dose of acetylcholine was, for instance, in seven out of ten treated animals less than the lowest threshold dose in the controls. The threshold dose of adrenaline was not influenced however. The maximal secretory response to pilocarpinewas increased by 77 per cent when calculated per gland, and by 57 per cent when calculated per unit weight (Table I). After treatment with testosterone the threshold doses were influenced in. the same way as after thyroxine, though to a lesser degree (Table II). Ir. some rats lower threshold doses of acetylcholine and methacholine were found. The threshold dose of adrenaline did not differ from that of controls. After treatment with testosterone the maximal secretory response to pilocarpine was found increased by 41 per cent when expressed per gland
6 Hormonal Influences on Rat's Submaxillary Gland 451 Significant changes were not obtained after calculation per unit weight (Table I). The increase of the maximal secretory response to pilocarpine, expressed per gland, appeared to follow the increase of the gland weight. Respiratory Enzymes. - Succinic Dehydrogenase. The activity of this enzyme (and of cytochrome oxidase and fumarase) in the submaxillary gland of the two groups of controls to rats treated with thyroxine or testosterone was similar (Table III). On the other hand, the activity of succinic dehydrogenase was lower in these controls than found previously (Ohlin, 1962). The rats used in the present and previous studies did not belong to the same strain and they had received different diets. After treatment with thyroxine or testosterone the activity of succinic dehydrogenase per gland was slightly increased. When expressed per g. of glandular tissue (wet weight) the activity was found similar to that of the control glands (Table III). Cytochrome Oxidase and Fumarase. - The activity of cytochrome oxidase and fumarase seemed increased after treatment with thyroxine or testosterone when expressed per gland. This increase corresponded to the enlargement of the submaxillary gland after hormonal treatment. The activity was similar to that of the controls when expressed per g. of glandular tissue. Denervated Glands. - The dry weight of denervated glands was found to be 27 h 1-2 (13) mg. After treatment with thyroxine or testosterone the dry weight increased to 33 i 1-7 (7) mg., and 36 : 1.2 (12) mg., respectively. After section of the chorda-lingual nerve the threshold doses of acetylcholine, methacholine and adrenaline were lowered. Treatment with thyroxine or testosterone did not affect the threshold doses of these secretory drugs in the denervated glands. The maximal secretory response to pilocarpine increased, when calculated per gland, from 24 i 1.7 (12) 1l. per min. to (6) after thyroxine and to 31 i 4.5 (7) after testosterone. When expressed per mg. dry weight, the maximal secretory response was practically unchanged both after thyroxine and testosterone, which is presumably due to the increase in gland weight. DIscuSSION The sexual dimorphism of the submaxillary gland is morphologically not very marked in rats. The cells of the granular tubules are slightly more developed in male than in female rats [Grad and Leblond, 1949; Sreebny, 1953; Jacoby and Leeson, 1959]. This and other differences are believed to be due to actions of testicular hormones. Since it is not known whether the sexual dimorphism affects the parameters studied in the present investigation, females only were used. Most workers have investigated the effect of hormonal treatment on salivary glands after at least 2 weeks of treatment. The duration of the present experiments was 3 weeks. The amounts of hormones given in the present study were for thyroxine about double the physiological dose [Griesbach et al., 1949] and for testosterone about equal to the physiological dose. It should be mentioned here that bigger doses, thirty times for
7 452 Ohlin o v * ze p p r a I D..z L ] 4Q ueo ~ cq O r O C5> 4 1o C) m m (^ O (a) aq to m P r-4 r eq aq 1 O xo A 2e. o^ o O 2 e t72 o o cq ci P caq I:- ered r- co cs 1 F-- z E-I Z * PI E-q o c:; xo 2Q >4 Q bo m E L- a) *S - O E C~4E- V
8 Hormonal Influences on Rat's Submaxillary Gland 453 thyroxine [Wells and Munson, 1,958] and five times for testosterone [Argonz and Corral Saleta, 1961], did not seem further to increase the glandular enlargement. The weight of the submaxillary gland of. rats decreased after hypophysectomy but increased after treatment with thyroxine or testosterone. The structural changes after hypophysectomy and hormonal treatment are mainly found in the cells of the granular tubules [Lacassagne, 194 b; Gabe, 195; Shafer and Muhler, 1956]. The maximal secretory response to pilocarpine, expressed per gland, decreased after hypophysectomy and increased after hormonal treatment. Though some changes occur in glandular cells other than those of the granular tubules, e.g. after hypophysectomy in the acinar cells [Bixler et al., 1959], it seems reasonable to assume that the cells of the granular tubules are more important to the maximal secretory response to pilocarpine than the acinar cells. This would seem to agree with the finding in rats that pilocarpine.elicits a considerable flow of saliva from the cut ducts of salivary glands lacking acinar cells [Schneyer and Schneyer, 1961]. The maximal secretory response to pilocarpine, expressed per unit weight, was reduced after hypophysectomy and enhanced after treatment with thyroxine but unchanged after testosterone. It seems likely that the secretory capacity of the granular tubules is under the control of thyroxine and thus of thyrotropin; this does not exclude hypophysial factors other than thyrotropin. The present findings agree with the observations that the total amount of saliva appearing in the mouth after stimulation with pilocarpine is decreased after radiothyroidectomy [Shafer' et al., 1958]. Histochemically the activity of suepinlic dehydrogenase is observed to be high in the cells of the granular tubules and in the duct cells, but low in the acinar cells of the submaxillary gland of rats [Padykula, 1952]. Cytochrome oxidase has been studied only in rabbits. It is found to be rather evenly distributed in the cells of the submaxillary gland, though the content appears lower in the acinar cells than in the duet cells [Schneider and Person, 196]. After hypophysectomy the weight 9f the submaxillary gland of rats and the activity of succinic dehydrogenase, cytochrome oxidase and fumarase, when expressed per gland, decreased [Ohlin, 1962]; in the present study the reverse was observed in intact rats treated with hormones. It seems likely that the changes in enzyme activity are confined mainly to the cells of the granular tubules. When the enzyme activity is modified in one glandular structure only, the possibility of demonstrating changes 'is dependent on the enzyme activity in other glandular cells, especially when the activity is expressed per g. of total gland. When the activity of the respiratory enzymes was expressed per unit weight, the activity of succinic dehydrogenase decreased after hypophysectomy, but that of cytochrome oxidase and fumarase was unchanged [Ohlin, 1962]; on the other hand, the activity of all enzymes appeared unchanged after treatment with thyroxine or testosterone. The findings on enzyme activity per unit weight after hypophysectomy may indicate that the activity of succinic dehydrogenase decreased markedly in the cells of the granular tubules. It cannot 'be excluded, however, that the
9 454 Ohlin activity of cytochrome oxidase and fumarase also decreased in the tubuilar cells. The enzyme concentration did not seem to increase in the granular tubules after treatment with thyroxine or testosterone. The threshold doses of acetylcholine and methacholine seemed slightly decreased after treatment with thyroxine or testosterone. A tendency of the threshold dose of acetylcholine to increase was seen after hypophysectomy. The changes of the threshold doses of parasympathomimetic agents seem closely linked to structural modifications of the granular tubules. On the other hand, the threshold dose of adrenaline seemed to be independent of this structure since it was unaffected under the various experimental conditions. In this connection it should be recalled that sympathetic denervation of the submaxillary gland of rats affects only the acinar cells [Snell and Garrett, 1958] and that treatment with a catecholamine, isoprenaline, causes an enlargement of these cells [Selye et al., 1961]. Section of the parasympathetic nerve of the submaxillary gland of rats caused marked changes in glandular function [Ohlin, 1965]. In spite of this, after treatment with thyroxine or testosterone the weight of the gland and the maximal secretory response to pilocarpine, when expressed per gland, still increased. Treatment with thyroxine did not result in an increase of the maximal secretory response of the denervated glands when expressed per unit weight. ACKNOWLEDGEMENT This work was supported by a grant from the Faculty of Medicine in Lund. REFERENCES ARGONZ, J. J. and RRAL SALETA, J. M. DE (1961). 'Action de diverses hormones sur la glande sous-maxillaire du rat', C.R. Soc. Biol., Pari, 155, 172. BIXLER, D., MUHLER, J. C. and SHAFER, W. G. (1959). 'Growth hormone and thyroxine: effects on submaxillary gland of hypophysectomized rat', Proc. Soc. exp. Biol., N. Y. 1, GABE, M. (195). 'Action de la thyroxine sur la glande sous-maxillaire du rat hypophysectomise', C.R. Acad. Sci., Pari8, 23, GRAD, B. (1949). 'Joint actions of thyroxine and testosterone in thyroidectomizedcastrated albino rats', Anat. Rec. 13, 458. GRAD, B. and LEBLOND, C. P. (1949). 'The necessity of testis and thyroid hormones for the maintenance of the serous tubules of the submaxillary gland in the male rat', Endocrinology, 45, GRIESBACH, W. E., KENNEDY, T. H. and PURVES, H. D. (1949). 'The physiological activities of the stereoisomers of thyroxine', Endocrinology, 44, JABY, F. and LEESON, C. R. (1959). 'The post-natal development of the rat submaxillary gland', J. Anat., Lond. 93, JABSOHN, D. (196). 'Effects of thyroxine on growth of mammary glands, whole body, heart and liver in hypophysectomized rats treated with insulin, cortisone and ovarian steroids', Acta endocr., Copenhagen, 85,
10 Hormonal Influences on Rat's Submaxillary Gland 455 LACASSAGNE, A. (194 a). 'Dimorphisme sexuel de la glande sous-maxillaire chez la souris', C.R. Soc. Biol., Paris, 133, LACASSAGNE, A. (194 b). 'Reactions de la glande sous-maxillaire a l'hormone male, chez la souris et le rat', (C.R. Soc. Biol., Paris, 133, LEBLOND, C. P. and GRAD, B. (1948). 'Control of the serous acini of the rat submaxillary gland by the thyroid hormone', Anat. Rec. 1, 75. NORDENFELT, I., OHLIN, P. and STROMBLAD, B. C. R. (196). 'Effect of denervation on respiratory enzymes in salivary glands', J. Physiol. 155, OHLIN, P. (1962). 'The effect of hypophysectomy on respiratory enzymes in the submaxillary gland of the rat', Quart. J. exp. Physiol. 47, OHLIN, P. (1965). To be published. PADYKULA, H. (1952). 'The localization of succinic dehydrogenase in tissue sections of the rat', Amer. J. Anat. 91, SCHNEIDER, R. M. and PERSON, P. (196). 'The isolation of submaxillary gland acini and duct segments', Exp. Cell Res. 2, SCHNEYER, C. A. and SCHNEYER, L. H. (1961). 'Secretion by salivary glands deficient in acini', Amer. J. Physiol. 21, SELYE, H., VEILLEUX, R. and CANTIN, M. (1961). 'Excessive stimulation of salivary gland growth by isoproterenol', Science, 133, SHAFER, W. G. and MUHLER, J. C. (1953). 'Effect of gonadectomy and sex hormones on the structure of the rat salivary glands', J. dent. Res. 32, SHAFER, W. G., CLARK, P. G. and MUHLER, J. C. (1956). 'The inhibition of hypophysectomy-induced changes in the rat submaxillary glands', Endocrinology, 59, SHAFER, W. G. and MUHLER, J. C. (1956). 'The effect of desiccated thyroid, propylthiouracil, testosterone, and fluorine on the submaxillary glands of the rat', J. dent. Res. 35, SHAFER, W. G., CLARK, P. G., BIXLER, D. and MUHLER, J. C. (1958). 'Salivary gland function in rats. II. Effect of thyroid function on salivary flow and viscosity', Proc. Soc. exp. Biol., N.Y. 98, SNELL, R. S. and GARRETT, J. R. (1958). 'The effect of postganglionic sympathectomy on the structure of the submandibular and major sublingual salivary glands of the rat', Z. Zellforsch. 48, SREEBNY, L. M. (1953). 'Histologic and enzymatic differences in the submaxillary glands of normal and hypophysectomized male and female white rats', J. dent. Res. 32, 686. WELLS, H. and MUNSON, P. L. (1958). 'Effect of thyroxine on the weight of submaxillary salivary glands', J. dent. Res. 37, 55.
however, reduced after parasympathetic denervation [Nordenfelt et al., 1960]. opposite to those caused by parasympathetic denervation.
![however, reduced after parasympathetic denervation [Nordenfelt et al., 1960]. opposite to those caused by parasympathetic denervation. however, reduced after parasympathetic denervation [Nordenfelt et al., 1960]. opposite to those caused by parasympathetic denervation.](/thumbs/90/101412039.jpg) CHOLINE ACETYLASE IN SALIVARY GLANDS OF THE CAT AFTER SYMPATHETIC DENERVATION. By IVAR NORDENFELT. From the Institute of Physiology, University of Lund, Sweden. (Received for publication 20th April 1964)
CHOLINE ACETYLASE IN SALIVARY GLANDS OF THE CAT AFTER SYMPATHETIC DENERVATION. By IVAR NORDENFELT. From the Institute of Physiology, University of Lund, Sweden. (Received for publication 20th April 1964)
Action of drugs on denervated myoepithelial cells of salivary glands
 Br. J. Pharmac. (1973), 48, 73-79. Action of drugs on denervated myoepithelial cells of salivary glands N. EMMELIN AND A. THULIN Institute of Physiology, University of Lund, Sweden Summary 1. The pressure
Br. J. Pharmac. (1973), 48, 73-79. Action of drugs on denervated myoepithelial cells of salivary glands N. EMMELIN AND A. THULIN Institute of Physiology, University of Lund, Sweden Summary 1. The pressure
LEAKAGE OF TRANSMITTERS IN SALIVARY GLANDS
 Brit. J. Pharmacol. (1964), 22, 119-125. LEAKAGE OF TRANSMITTERS IN SALIVARY GLANDS BY N. ASSARSON AND N. EMMELIN From the Institute of Physiology, University of Lund, Sweden (Received October 8, 1963)
Brit. J. Pharmacol. (1964), 22, 119-125. LEAKAGE OF TRANSMITTERS IN SALIVARY GLANDS BY N. ASSARSON AND N. EMMELIN From the Institute of Physiology, University of Lund, Sweden (Received October 8, 1963)
SUPERSENSITIVITY OF THE SUBMAXILLARY GLAND FOLLOWING EXCLUSION OF THE POSTGANGLIONIC PARASYMPATHETIC NEURONE
 Brit. J. Pharmacol. (1960), 15, 356. SUPERSENSITIVITY OF THE SUBMAXILLARY GLAND FOLLOWING EXCLUSION OF THE POSTGANGLIONIC PARASYMPATHETIC NEURONE BY N. EMMELIN From the Institute of Physiology, University
Brit. J. Pharmacol. (1960), 15, 356. SUPERSENSITIVITY OF THE SUBMAXILLARY GLAND FOLLOWING EXCLUSION OF THE POSTGANGLIONIC PARASYMPATHETIC NEURONE BY N. EMMELIN From the Institute of Physiology, University
Observations on the function of the gland after denervation have usually
 Quarterly Journal of Experimental Phy8iology (1974) 59,1-9 REINNERVATION OF THE DENERVATED PAROTID GLAND OF THE CAT. By J. EKSTROM and N. EMMELIN. From the Institute of Physiology, University of Lund,
Quarterly Journal of Experimental Phy8iology (1974) 59,1-9 REINNERVATION OF THE DENERVATED PAROTID GLAND OF THE CAT. By J. EKSTROM and N. EMMELIN. From the Institute of Physiology, University of Lund,
fumarase, which are all important in the respiratory activity ofmammalian
 J. Physiol. (1960), 152, pp. 99-107 99 With 6 text-flgures Printed in Great Britain EFFECT OF DENERVATION ON RESPIRATORY ENZYMES IN SALIVARY GLANDS BY I. NORDENFELT, P. OHLIN AND B. C. R. STROMBLAD From
J. Physiol. (1960), 152, pp. 99-107 99 With 6 text-flgures Printed in Great Britain EFFECT OF DENERVATION ON RESPIRATORY ENZYMES IN SALIVARY GLANDS BY I. NORDENFELT, P. OHLIN AND B. C. R. STROMBLAD From
REINNERVATION OF SUBMAXILLARY GLANDS AFTER PARTIAL. mainly in sympathetic ganglia, in which marked sprouting has been shown
 Q. Jl exp. Phy8iol. (1968) 53, 1-18 REINNERVATION OF SUBMAXILLARY GLANDS AFTER PARTIAL POSTGANGLIONIC DENERVATION. By N. EMMELIN and C. PEREC*. From the Institute of Physiology, University of Lund, Sweden.
Q. Jl exp. Phy8iol. (1968) 53, 1-18 REINNERVATION OF SUBMAXILLARY GLANDS AFTER PARTIAL POSTGANGLIONIC DENERVATION. By N. EMMELIN and C. PEREC*. From the Institute of Physiology, University of Lund, Sweden.
EFFECT OF DENERVATION AND OF COCAINE ON THE ACTION OF SYMPATHOMIMETIC AMINES
 Brit. J. Pharmacol. (1960), 15, 328. EFFECT OF DENERVATION AND OF COCAINE ON THE ACTION OF SYMPATHOMIMETIC AMINES BY B. C. R. STROMBLAD From the Institute of Physiology, Lund, Sweden (RECEIVED FEBRUARY
Brit. J. Pharmacol. (1960), 15, 328. EFFECT OF DENERVATION AND OF COCAINE ON THE ACTION OF SYMPATHOMIMETIC AMINES BY B. C. R. STROMBLAD From the Institute of Physiology, Lund, Sweden (RECEIVED FEBRUARY
auriculo-temporal nerve. The secretory response to various cholinesterase
 Quarterly Journal of Experimental Phy8iology (1974) 59, 11-17 THE SECRETORY INNERVATION OF THE PAROTID GLAND OF THE CAT: AN UNEXPECTED COMPONENT. By J. EKSTROM and N. EMMELIN. From the Institute of Physiology,
Quarterly Journal of Experimental Phy8iology (1974) 59, 11-17 THE SECRETORY INNERVATION OF THE PAROTID GLAND OF THE CAT: AN UNEXPECTED COMPONENT. By J. EKSTROM and N. EMMELIN. From the Institute of Physiology,
THE INFLUENCE OF ISOPRENALINE AND PROPRANOLOL ON THE SUBMAXILLARY GLAND OF THE RAT
 THE INFLUENCE OF ISOPRENALINE AND PROPRANOLOL ON THE SUBMAXILLARY GLAND OF THE RAT MICHIO FUKUDA Department of Surgery, Osaka University Dental School, Kita-ku, Osaka Received for publication December
THE INFLUENCE OF ISOPRENALINE AND PROPRANOLOL ON THE SUBMAXILLARY GLAND OF THE RAT MICHIO FUKUDA Department of Surgery, Osaka University Dental School, Kita-ku, Osaka Received for publication December
OBSERVATIONS ON THE ISOLATED VAS DEFERENS
 Brit. J. Pharmacol. (1963), 20, 299-306. OBSERVATIONS ON THE ISOLATED VAS DEFERENS BY P. OHLIN AND B. C. R. STROMBLAD* From the Institute of Physiology, University of Lund, Sweden (Received December 3,
Brit. J. Pharmacol. (1963), 20, 299-306. OBSERVATIONS ON THE ISOLATED VAS DEFERENS BY P. OHLIN AND B. C. R. STROMBLAD* From the Institute of Physiology, University of Lund, Sweden (Received December 3,
hypophysectomized rat. Marenzi & Gerschman [1934] studied six of the University and Royal Infirmary, Glasgow (Received 13 December 1937)
![hypophysectomized rat. Marenzi & Gerschman [1934] studied six of the University and Royal Infirmary, Glasgow (Received 13 December 1937) hypophysectomized rat. Marenzi & Gerschman [1934] studied six of the University and Royal Infirmary, Glasgow (Received 13 December 1937)](/thumbs/96/129025719.jpg) 124 J. Physiol. (I938) 92, I24-130 6i2.492.5:6I2.I26 THE EFFECT OF HYPOPHYSECTOMY ON THE BLOOD CALCIUM AND PHOSPHORUS OF THE RAT BY A. B. ANDERSON AND E. G. OASTLER From the Biochemical Laboratory, Department
124 J. Physiol. (I938) 92, I24-130 6i2.492.5:6I2.I26 THE EFFECT OF HYPOPHYSECTOMY ON THE BLOOD CALCIUM AND PHOSPHORUS OF THE RAT BY A. B. ANDERSON AND E. G. OASTLER From the Biochemical Laboratory, Department
6I :6I2.I83 BY ALISON S. DALE. concluded that the apparent vaso-constriction obtained by F r6 hli c h and
 6I2.313.87:6I2.I83 A REVERSED ACTION OF THE CHORDA TYMPANI ON THE VENOUS OUTFLOW FROM THE SUBMAXILLARY GLAND. BY ALISON S. DALE. (From the Physiological Laboratory, Cambridcgel.) INTRODUCTORY. FROiHLICH
6I2.313.87:6I2.I83 A REVERSED ACTION OF THE CHORDA TYMPANI ON THE VENOUS OUTFLOW FROM THE SUBMAXILLARY GLAND. BY ALISON S. DALE. (From the Physiological Laboratory, Cambridcgel.) INTRODUCTORY. FROiHLICH
different ratios of PMSG and HCG on the occurrence of follicular haemorrhage THE induction of ovulation with PMSG and HCG in the rat has been studied
 Q. Jl exp. Physiol. (1968) 53, 129-135 THE INDUCTION OF OVULATION IN IMMATURE RATS TREATED WITH PREGNANT MARE'S SERUM GONADOTROPHIN AND HUMAN CHORIONIC GONADOTROPHIN. By S. F. LUNN and E. T. BELL. From
Q. Jl exp. Physiol. (1968) 53, 129-135 THE INDUCTION OF OVULATION IN IMMATURE RATS TREATED WITH PREGNANT MARE'S SERUM GONADOTROPHIN AND HUMAN CHORIONIC GONADOTROPHIN. By S. F. LUNN and E. T. BELL. From
Citation Acta medica Nagasakiensia. 1961, 5(
 NAOSITE: Nagasaki University's Ac Title Responsiveness of the Denervated Ad Author(s) Yamashita, Kazukuni; Jinnai, Seiich Citation Acta medica Nagasakiensia. 1961, 5( Issue Date 1961-03-25 URL http://hdl.handle.net/10069/15441
NAOSITE: Nagasaki University's Ac Title Responsiveness of the Denervated Ad Author(s) Yamashita, Kazukuni; Jinnai, Seiich Citation Acta medica Nagasakiensia. 1961, 5( Issue Date 1961-03-25 URL http://hdl.handle.net/10069/15441
gland, the tongue and the sweat glands of the cat. The submaxillary
 306 547.435-292:6I2.8I7 THE LIBERATION OF ACETYLCHOLINE BY POTASSIUM. BY W. FELDBERG1 AND J. A. GUIMARAIS1,2. (From the National Institute for Medical Research, London, N.W. 3.) (Received November 22,
306 547.435-292:6I2.8I7 THE LIBERATION OF ACETYLCHOLINE BY POTASSIUM. BY W. FELDBERG1 AND J. A. GUIMARAIS1,2. (From the National Institute for Medical Research, London, N.W. 3.) (Received November 22,
it by the sympathetic nerve.
 OBSERVATIONS ON AUGMENTED SALIVARY SECRETION. BY G. V. ANREP. * (From the Institute of Physiology, University College, London.) IN 1889 Langley described a peculiar effect of stimulation of the cerebral
OBSERVATIONS ON AUGMENTED SALIVARY SECRETION. BY G. V. ANREP. * (From the Institute of Physiology, University College, London.) IN 1889 Langley described a peculiar effect of stimulation of the cerebral
mice, rats and rabbits produces a temporary fall in the absolute number of
 328 J. Physiol. (I950) III, 328-334 6I2.492:6I2.II2 PITUITARY GLAND AND BLOOD LYMPHOCYTES BY H. F. COLFER, J. DE GROOT AND G. W. HARRIS From the Physiological Laboratory, University of Cambridge (Received
328 J. Physiol. (I950) III, 328-334 6I2.492:6I2.II2 PITUITARY GLAND AND BLOOD LYMPHOCYTES BY H. F. COLFER, J. DE GROOT AND G. W. HARRIS From the Physiological Laboratory, University of Cambridge (Received
J. Physiol. (I959) I45, 595-6IO
 595 J. Physiol. (I959) I45, 595-6IO CONCENTRATION OF SALIVARY IODIDE: A COMPARATIVE STUDY BY B. COHEN* AND N. B. MYANT From the Department of Dental Science, Royal College of Surgeons of England, London,
595 J. Physiol. (I959) I45, 595-6IO CONCENTRATION OF SALIVARY IODIDE: A COMPARATIVE STUDY BY B. COHEN* AND N. B. MYANT From the Department of Dental Science, Royal College of Surgeons of England, London,
might be due to a direct action on the thyroid, like that of the thiouracil
 288 J. Physiol. (1953) I20, 288-297 COMPARISON OF THE EFFECTS OF THIOURACIL, THY- ROXINE AND CORTISONE ON THE THYROID FUNCTION OF RABBITS BY N. B. MYANT* From the Department of Clinical Research, University
288 J. Physiol. (1953) I20, 288-297 COMPARISON OF THE EFFECTS OF THIOURACIL, THY- ROXINE AND CORTISONE ON THE THYROID FUNCTION OF RABBITS BY N. B. MYANT* From the Department of Clinical Research, University
(Received April 6, 1936)
 275 6I2.26 RESPIRATION AND FUNCTIONAL ACTIVITY BY W. DEUTSCH AND H. S. RAPER (From the Department of Physiology, University of Manchester) (Received April 6, 1936) IT has long been established that in
275 6I2.26 RESPIRATION AND FUNCTIONAL ACTIVITY BY W. DEUTSCH AND H. S. RAPER (From the Department of Physiology, University of Manchester) (Received April 6, 1936) IT has long been established that in
(17fl-hydroxyandrost-4-en-3-one) to produce this syndrome experimentally
 J. Physiol. (1964), 176, pp. 91-14 91 Printed in Great Britain THE EFFECT OF TESTOSTERONE DURING THE NEONATAL PERIOD ON THE THYROID GLAND OF MALE AND FEMALE RATS BY K. BROWVN-GRANT* From the Department
J. Physiol. (1964), 176, pp. 91-14 91 Printed in Great Britain THE EFFECT OF TESTOSTERONE DURING THE NEONATAL PERIOD ON THE THYROID GLAND OF MALE AND FEMALE RATS BY K. BROWVN-GRANT* From the Department
Columbia University, New York.)
 15 6I2.62I:6I2.4 THE EFFECT OF OVARIAN HORMONE ON THE PITUITARY, THYROID, AND ADRENAL GLANDS OF SPAYED FEMALE RATS. BY DOROTHY H. ANDERSEN. (From the Department of Pathology, College of Physicians and
15 6I2.62I:6I2.4 THE EFFECT OF OVARIAN HORMONE ON THE PITUITARY, THYROID, AND ADRENAL GLANDS OF SPAYED FEMALE RATS. BY DOROTHY H. ANDERSEN. (From the Department of Pathology, College of Physicians and
Stromblad, 1958]. Further, nerves stained for acetylcholinesterase were looked
![Stromblad, 1958]. Further, nerves stained for acetylcholinesterase were looked Stromblad, 1958]. Further, nerves stained for acetylcholinesterase were looked](/thumbs/89/99071497.jpg) Quarterly Journal of Experimntal Phy8iology (1976) 61, 127-132 UNCHARTED CHOLINERGIC NERVES IN THE RABBIT PAROTID GLAND. By P. ALM and J. EKSTROM. From the Institute of Anatomy and Histology, and the Institute
Quarterly Journal of Experimntal Phy8iology (1976) 61, 127-132 UNCHARTED CHOLINERGIC NERVES IN THE RABBIT PAROTID GLAND. By P. ALM and J. EKSTROM. From the Institute of Anatomy and Histology, and the Institute
612.6I7.5:612.6I6.I. different, but most of them appear to be satisfactory from a qualitative
 442 612.6I7.5:612.6I6.I SIZE CHANGES IN THE SEMINAL VESICLES OF THE MOUSE DURING DEVELOPMENT AND AFTER CASTRATION. BY RUTH DEANESLY AND A. S. PARKES'. (From the National Institute for Medical Research,
442 612.6I7.5:612.6I6.I SIZE CHANGES IN THE SEMINAL VESICLES OF THE MOUSE DURING DEVELOPMENT AND AFTER CASTRATION. BY RUTH DEANESLY AND A. S. PARKES'. (From the National Institute for Medical Research,
Effect of Muscular Exercise on Adrenaline and Noradrenaline Secretion of the Adrenal Gland in the Dog
 Tohoku J. exp. Med., 1966, 88, 361-366 Effect of Muscular Exercise on Adrenaline and Noradrenaline Secretion of the Adrenal Gland in the Dog Sennosuke Ohukuzi Deparment of Physiology (Prof. T. Suzuki),
Tohoku J. exp. Med., 1966, 88, 361-366 Effect of Muscular Exercise on Adrenaline and Noradrenaline Secretion of the Adrenal Gland in the Dog Sennosuke Ohukuzi Deparment of Physiology (Prof. T. Suzuki),
THE DIABETOGENIC HORMONE OF THE PITUITARY GLAND
 VOL. XIII, i JANUARY, 96 THE DIABETOGENIC HORMONE OF THE PITUITARY GLAND BY DAVID SLOME. (From the Department of Social Biology, the University of London, and the Buckston Browne Research Farm, Royal College
VOL. XIII, i JANUARY, 96 THE DIABETOGENIC HORMONE OF THE PITUITARY GLAND BY DAVID SLOME. (From the Department of Social Biology, the University of London, and the Buckston Browne Research Farm, Royal College
THYROID ACTIVITY AND AMINE OXIDASE IN THE LIVER
 Brit. J. Pharmacol. (1952), 7, 93. THYROID ACTIVITY AND AMINE OXIDASE IN THE LIVER BY A. SPINKS AND J. H. BURN From the Department of Pharmacology, Oxford University (Received November 13, 1951) So long
Brit. J. Pharmacol. (1952), 7, 93. THYROID ACTIVITY AND AMINE OXIDASE IN THE LIVER BY A. SPINKS AND J. H. BURN From the Department of Pharmacology, Oxford University (Received November 13, 1951) So long
THE ACTION OF NICOTINE ON THE CILIARY GANGLION
 Brit. J. Pharmnacol. (1952), 7, 665. THE ACTION OF NICOTINE ON THE CILIARY GANGLION BY BRENDA M. SCHOFIELD From the Department of Pharmacology, University of Oxford (Received June 7, 1952) The existing
Brit. J. Pharmnacol. (1952), 7, 665. THE ACTION OF NICOTINE ON THE CILIARY GANGLION BY BRENDA M. SCHOFIELD From the Department of Pharmacology, University of Oxford (Received June 7, 1952) The existing
Lec.2 Histology Glandular Epithelium A gland 1. Endocrine Glands 2. Exocrine Glands Endocrine Glands Exocrine Glands
 Lec.2 Histology Glandular Epithelium A gland is one or more cells that produce and secrete a specific product. The product is always a water-based fluid (aqueous) and usually contains proteins (the product
Lec.2 Histology Glandular Epithelium A gland is one or more cells that produce and secrete a specific product. The product is always a water-based fluid (aqueous) and usually contains proteins (the product
extensively investigated (see review by Maqsood, 1952), but the possible
 390 J. Physiol. (I 955) I 27, 390-399 THE EFFECTS OF SOME GONADAL HORMONES ON THYROID ACTIVITY IN THE RABBIT BY K. BROWN-GRANT From the Department of Neuroendocrinology, Maudsley Hospital, London, S.E.5
390 J. Physiol. (I 955) I 27, 390-399 THE EFFECTS OF SOME GONADAL HORMONES ON THYROID ACTIVITY IN THE RABBIT BY K. BROWN-GRANT From the Department of Neuroendocrinology, Maudsley Hospital, London, S.E.5
(d) Loss of the anti-diuretic and pressor substance from the posterior. (c) Complete hypophysectomy is followed by a transitory polyuria
 202 J. Physiol. (I937) 9I, 202-2II 6I2.492:6I2.463 THE PITUITARY GLAND AND THE CONTROL OF URINARY SECRETION BY E. C. DODDS, R. L. NOBLE AND P. C. WILLIAMS From the Courtauld Institute of Biochemistry,
202 J. Physiol. (I937) 9I, 202-2II 6I2.492:6I2.463 THE PITUITARY GLAND AND THE CONTROL OF URINARY SECRETION BY E. C. DODDS, R. L. NOBLE AND P. C. WILLIAMS From the Courtauld Institute of Biochemistry,
THE EFFECT OF UNILATERAL CASTRATION ON THE REMAINING TESTIS OF THE MOUSE
 402 THE EFFECT OF UNILATERAL CASTRATION ON THE REMAINING TESTIS OF THE MOUSE BY I. W. ROWLANDS. (From the Department of Zoology, University College of North Wales, Bangor.) (Received 14th April, 1934.)
402 THE EFFECT OF UNILATERAL CASTRATION ON THE REMAINING TESTIS OF THE MOUSE BY I. W. ROWLANDS. (From the Department of Zoology, University College of North Wales, Bangor.) (Received 14th April, 1934.)
Effect of Gonadotropic Hormones on Hypophysectomized (Anterior Lobe) Male Rana Pipiens
 The Ohio State University Knowledge Bank kb.osu.edu Ohio Journal of Science (Ohio Academy of Science) Ohio Journal of Science: Volume 54, Issue 3 (May, 1954) 19545 Effect of Gonadotropic Hormones on Hypophysectomized
The Ohio State University Knowledge Bank kb.osu.edu Ohio Journal of Science (Ohio Academy of Science) Ohio Journal of Science: Volume 54, Issue 3 (May, 1954) 19545 Effect of Gonadotropic Hormones on Hypophysectomized
EFFECTS OF EXOGENOUSLY ADDED SHORT-CHAIN FATTY ACIDS ON PANCREATIC EXOCRINE SECRETION IN DOMESTIC RABBIT
 EFFECTS OF EXOGENOUSLY ADDED SHORT-CHAIN FATTY ACIDS ON PANCREATIC EXOCRINE SECRETION IN DOMESTIC RABBIT DOJANA N 1., POP A 2., PAPUC C 3. 1 Department of Animal Physiology, Faculty of Veterinary Medicine,
EFFECTS OF EXOGENOUSLY ADDED SHORT-CHAIN FATTY ACIDS ON PANCREATIC EXOCRINE SECRETION IN DOMESTIC RABBIT DOJANA N 1., POP A 2., PAPUC C 3. 1 Department of Animal Physiology, Faculty of Veterinary Medicine,
CHEMICALLY INDUCED BREAST TUMOURS AND THE MAMMARY TUM,OUR AGENT.
 52 CHEMICALLY INDUCED BREAST TUMOURS AND THE MAMMARY TUM,OUR AGENT. L. DMOCHOWSKI AND J. W. ORR. From the Department of Experimental Pathology and Cancer Research, Medical School, University of Leeds,
52 CHEMICALLY INDUCED BREAST TUMOURS AND THE MAMMARY TUM,OUR AGENT. L. DMOCHOWSKI AND J. W. ORR. From the Department of Experimental Pathology and Cancer Research, Medical School, University of Leeds,
Hormone-induced granular convoluted tubule-like cells in mouse parotid gland
 290 MINI-REVIEW Hormone-induced granular convoluted tubule-like cells in mouse parotid gland Shingo Kurabuchi 1, Takanori Matsuoka 1, and Kazuo Hosoi 2 1 Department of Histology, The Nippon Dental University
290 MINI-REVIEW Hormone-induced granular convoluted tubule-like cells in mouse parotid gland Shingo Kurabuchi 1, Takanori Matsuoka 1, and Kazuo Hosoi 2 1 Department of Histology, The Nippon Dental University
published by Hektoen and SchuIhof (1), and since the appearance
 ON THE INNERVATION AND SECRETORY PATH OF THE THYROID GLAND. BY C. S. HICKS (Beit Memorial Fellow). (From the Biochemistry Laboratories, Cambridge, and Physiology Department, University of Adelaide.) IN
ON THE INNERVATION AND SECRETORY PATH OF THE THYROID GLAND. BY C. S. HICKS (Beit Memorial Fellow). (From the Biochemistry Laboratories, Cambridge, and Physiology Department, University of Adelaide.) IN
THE INHIBITORY EFFECT OF STILBOESTROL ON GASTRIC SECRETION IN CATS
 Brit. J. Pharmacol. (1950), 5, 3S9. THE INHIBITORY EFFECT OF STILBOESTROL ON GASTRIC SECRETION IN CATS BY K. N. OJHA* AND D. R. WOOD From the Department of Pharmacology and Therapeutics, University of
Brit. J. Pharmacol. (1950), 5, 3S9. THE INHIBITORY EFFECT OF STILBOESTROL ON GASTRIC SECRETION IN CATS BY K. N. OJHA* AND D. R. WOOD From the Department of Pharmacology and Therapeutics, University of
(Received 14 February 1951)
 510 J. Physiol. (I95I) II4, 5I0-54 PHYSIOLOGICAL SIGNIFICANCE OF THE SWEAT RESPONSE TO ADRENALINE IN MAN BY T. M. CHALMERS jam C. A. KEELE From the Department of Pharmacology, Middlesex Hospital Medical
510 J. Physiol. (I95I) II4, 5I0-54 PHYSIOLOGICAL SIGNIFICANCE OF THE SWEAT RESPONSE TO ADRENALINE IN MAN BY T. M. CHALMERS jam C. A. KEELE From the Department of Pharmacology, Middlesex Hospital Medical
Since, for many months after section of the right vagus in the neck,
 THE INFLUENCE OF THE VAGUS ON THE ISLETS OF LANGERHANS. Part II. The effect of cutting the vagus upon sugar tolerance. BY G. A. CLARK. (From the Physiological Laboratory, Sheffield University.) IN Part
THE INFLUENCE OF THE VAGUS ON THE ISLETS OF LANGERHANS. Part II. The effect of cutting the vagus upon sugar tolerance. BY G. A. CLARK. (From the Physiological Laboratory, Sheffield University.) IN Part
KALLIKREIN AND VASODILATION IN THE SUBMAXILLARY GLAND
 GASTROENTEROLOGY Copyright 1967 by The Williams & Wilk' ns Co. Vol. 52, No.2, Part 2 Printed in U.S.A. KALLIKREIN AND VASODILATION IN THE SUBMAXILLARY GLAND MELVILLE SCHACHTER, M.D., AND SUSANNE BEILENSON,
GASTROENTEROLOGY Copyright 1967 by The Williams & Wilk' ns Co. Vol. 52, No.2, Part 2 Printed in U.S.A. KALLIKREIN AND VASODILATION IN THE SUBMAXILLARY GLAND MELVILLE SCHACHTER, M.D., AND SUSANNE BEILENSON,
Sprague-Dawley rats under chloralose anaesthesia. Adrenergic responses were
 Journal of Physiology (1988), 403, pp. 105-116 105 With 4 text-figures Printed in Great Britain THE EFFECTS OF ATROPINE AND CHRONIC SYMPATHECTOMY ON MAXIMAL PARASYMPATHETIC STIMULATION OF PAROTID SALIVA
Journal of Physiology (1988), 403, pp. 105-116 105 With 4 text-figures Printed in Great Britain THE EFFECTS OF ATROPINE AND CHRONIC SYMPATHECTOMY ON MAXIMAL PARASYMPATHETIC STIMULATION OF PAROTID SALIVA
Interrelationship between Angiotensin Catecholamines. Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D.
 Interrelationship between Angiotensin and Catecholamines Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D. SUMMARY Urinary catecholamines were measured with an attempt
Interrelationship between Angiotensin and Catecholamines Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D. SUMMARY Urinary catecholamines were measured with an attempt
CAROTID SINUS REFLEX AND CONTRACTION
 Brit. J. Pharmacol. (1950), 5, 505. CAROTID SINUS REFLEX AND CONTRACTION OF THE SPLEEN BY ROBERT L. DRIVER AND MARTHE VOGT From the Department of Pharmacology, University of Edinburgh (Received July 12,
Brit. J. Pharmacol. (1950), 5, 505. CAROTID SINUS REFLEX AND CONTRACTION OF THE SPLEEN BY ROBERT L. DRIVER AND MARTHE VOGT From the Department of Pharmacology, University of Edinburgh (Received July 12,
Received for publication February' 7, 1973
 T he J ournal of H istochemistry and Cytochemistry Copyright 1973 by The Histochemical Society, Inc. Vol. 21. No. 7. pp. 661-673, 1973 Printed in U.S.A. A QUALITATIVE AND QUANTITATIVE HISTOCHEMICAL INVESTIGATION
T he J ournal of H istochemistry and Cytochemistry Copyright 1973 by The Histochemical Society, Inc. Vol. 21. No. 7. pp. 661-673, 1973 Printed in U.S.A. A QUALITATIVE AND QUANTITATIVE HISTOCHEMICAL INVESTIGATION
supplied with a parasympathetic motor innervation which can cause them only secretory and vasomotor fibres, but also motor fibres supplying the
 J. Phy8iol. (1968), 196, pp. 381-396 381 With 1 plate and 10 text-figures Printed in Great Britain NEURAL CONTROL OF SALIVARY MYOEPITHELIAL CELLS BY N. EMMELIN, J. R. GARRETT AND P. OHLIN From the Institute
J. Phy8iol. (1968), 196, pp. 381-396 381 With 1 plate and 10 text-figures Printed in Great Britain NEURAL CONTROL OF SALIVARY MYOEPITHELIAL CELLS BY N. EMMELIN, J. R. GARRETT AND P. OHLIN From the Institute
THE ACTION OF PROMETHAZINE (PHENERGAN) DUE TO HISTAMINE IN PROTECTING MICE AGAINST DEATH
 Brit. J. Pharmacol. (1950), 5, 510. THE ACTION OF PROMETHAZINE (PHENERGAN) IN PROTECTING MICE AGAINST DEATH DUE TO HISTAMINE BY B. N. HALPERN * AND D. R. WOODt From the Clinique Medicale Propedeutique
Brit. J. Pharmacol. (1950), 5, 510. THE ACTION OF PROMETHAZINE (PHENERGAN) IN PROTECTING MICE AGAINST DEATH DUE TO HISTAMINE BY B. N. HALPERN * AND D. R. WOODt From the Clinique Medicale Propedeutique
THE EFFECT OF TESTICULAR EXTRACTS ON THE BLOOD CALCIUM
 55 THE EFFECT OF TESTICULAR EXTRACTS ON THE BLOOD CALCIUM BY L. MIRVISH AND L. P. BOSMAN. (From the Department of Biochemistry, University of Cape Town.) {Received 12th February 1929.) IT has long been
55 THE EFFECT OF TESTICULAR EXTRACTS ON THE BLOOD CALCIUM BY L. MIRVISH AND L. P. BOSMAN. (From the Department of Biochemistry, University of Cape Town.) {Received 12th February 1929.) IT has long been
clearing activity is produced and destroyed in the rat. Both the
 THE SITES AT WHICH PLASMA CLEARING ACTIVITY IS PRODUCED AND DESTROYED IN THE RAT. By G. H. JEFFRIES. From the Sir William Dunn School of Pathology, Oxford. (Received for publication 25th June 1954.) CLEARING
THE SITES AT WHICH PLASMA CLEARING ACTIVITY IS PRODUCED AND DESTROYED IN THE RAT. By G. H. JEFFRIES. From the Sir William Dunn School of Pathology, Oxford. (Received for publication 25th June 1954.) CLEARING
THE RESTORATION OF LOST ORGAN TISSUE* THE RATE AND DEGREE OF RESTORATION BY T. ADDIS, M.D., AND W. LEW
 Published Online: 1 March, 1940 Supp Info: http://doi.org/10.1084/jem.71.3.325 Downloaded from jem.rupress.org on November 20, 2018 THE RESTORATION OF LOST ORGAN TISSUE* THE RATE AND DEGREE OF RESTORATION
Published Online: 1 March, 1940 Supp Info: http://doi.org/10.1084/jem.71.3.325 Downloaded from jem.rupress.org on November 20, 2018 THE RESTORATION OF LOST ORGAN TISSUE* THE RATE AND DEGREE OF RESTORATION
THE EFFECT OF EXTRACTS OF SUPRARENAL CORTEX ON THE BLOOD CALCIUM
 35 THE EFFECT OF EXTRACTS OF SUPRARENAL CORTEX ON THE BLOOD CALCIUM BY L. MIRVISH AND L. P. BOSMAN. (From the Department of Biochemistry, University of Cape Town.) (Received 12th February 1929.) INTRODUCTION.
35 THE EFFECT OF EXTRACTS OF SUPRARENAL CORTEX ON THE BLOOD CALCIUM BY L. MIRVISH AND L. P. BOSMAN. (From the Department of Biochemistry, University of Cape Town.) (Received 12th February 1929.) INTRODUCTION.
Franklin, 1933; Waterman, 1933]; indeed, the only negative findings, [Waterman, 1933]. Inasmuch, then, as Donegan was misled with
![Franklin, 1933; Waterman, 1933]; indeed, the only negative findings, [Waterman, 1933]. Inasmuch, then, as Donegan was misled with Franklin, 1933; Waterman, 1933]; indeed, the only negative findings, [Waterman, 1933]. Inasmuch, then, as Donegan was misled with](/thumbs/91/106294335.jpg) 381 6I2.I34:6I2.893 THE CONSTRICTOR RESPONSE OF THE INFERIOR VENA CAVA TO STIMULATION OF THE SPLANCHNIC NERVE BY K. J. FRANKLIN AND A. D. McLACHLIN (From the University Department of Pharmacology, Oxford)
381 6I2.I34:6I2.893 THE CONSTRICTOR RESPONSE OF THE INFERIOR VENA CAVA TO STIMULATION OF THE SPLANCHNIC NERVE BY K. J. FRANKLIN AND A. D. McLACHLIN (From the University Department of Pharmacology, Oxford)
Effect of Orchiectomy on Pituitary Secretion of ACTH MARY D. COYNE AND JULIAN I. KITAY
 Excerpted from: Journal Title: Endocrinology. Volume: 89 Issue: 4 October 1971 Pages: 1024-8 Effect of Orchiectomy on Pituitary Secretion of ACTH MARY D. COYNE AND JULIAN I. KITAY Department of Physiology,
Excerpted from: Journal Title: Endocrinology. Volume: 89 Issue: 4 October 1971 Pages: 1024-8 Effect of Orchiectomy on Pituitary Secretion of ACTH MARY D. COYNE AND JULIAN I. KITAY Department of Physiology,
INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT
 Brit. J. Phawmacol. (1951), 6, 289. INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT BY From the Pharmacological Laboratory, University of St. Andrews, Medical School, Dundee (Received February 2, 1951)
Brit. J. Phawmacol. (1951), 6, 289. INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT BY From the Pharmacological Laboratory, University of St. Andrews, Medical School, Dundee (Received February 2, 1951)
ROLE OF ADRENAL HORMONES IN MAINTAINING TISSUE STORES OF NORADRENALINE DURING INCREASED SYMPATHETIC ACTIVITY
 Br. J. Pharmac. Chemother. (1966), 27, 532-535. ROLE OF ADRENAL HORMONES IN MAINTAINING TISSUE STORES OF NORADRENALINE DURING INCREASED SYMPATHETIC ACTIVITY BY V. M. AVAKIAN* AND MARTHE VOGT From the Agricultural
Br. J. Pharmac. Chemother. (1966), 27, 532-535. ROLE OF ADRENAL HORMONES IN MAINTAINING TISSUE STORES OF NORADRENALINE DURING INCREASED SYMPATHETIC ACTIVITY BY V. M. AVAKIAN* AND MARTHE VOGT From the Agricultural
androgen on the seminal vesicles it had neither a blocking effect on the penile
 MORPHOLOGICAL AND BEHAVIOURAL EFFECTS OF AN 'ANTIANDROGEN' IN MALE RATS F. A. BEACH and W. H. WESTBROOK Department of Psychology, University of California, Berkeley, California 94720, U.S.A. (Received
MORPHOLOGICAL AND BEHAVIOURAL EFFECTS OF AN 'ANTIANDROGEN' IN MALE RATS F. A. BEACH and W. H. WESTBROOK Department of Psychology, University of California, Berkeley, California 94720, U.S.A. (Received
-The process by which organisms maintain, control, and coordinate their internal environment with a constantly changing external environment
 -The process by which organisms maintain, control, and coordinate their internal environment with a constantly changing external environment -It is all of the activities that help to maintain an organism
-The process by which organisms maintain, control, and coordinate their internal environment with a constantly changing external environment -It is all of the activities that help to maintain an organism
by an increase in the ovarian content of acid mucopolysaccharides with a sub sequent tendency to develop polycystic ovaries (Thorsee 1961).
 Connective Tissue Laboratory (Prof. G. Asboe-Hansen, M. D.), University Institute of Medical Anatomy, Copenhagen INHIBITION OF OVULATION AND CHANGES IN OVARIAN MUCOPOLYSACCHARIDES INDUCED BY THYROIDECTOMY
Connective Tissue Laboratory (Prof. G. Asboe-Hansen, M. D.), University Institute of Medical Anatomy, Copenhagen INHIBITION OF OVULATION AND CHANGES IN OVARIAN MUCOPOLYSACCHARIDES INDUCED BY THYROIDECTOMY
THE ACTION OF GUANETHIDINE WITH PARTICULAR REFERENCE TO THE SYMPATHETIC NERVOUS SYSTEM
 Brit. J. Pharinacol. (1963), 20, 171-177. THE ACTION OF GUANETHIDINE WITH PARTICULAR REFERENCE TO THE SYMPATHETIC NERVOUS SYSTEM BY G. F. ABERCROMBIE AND B. N. DAVIES From the Department of Physiology,
Brit. J. Pharinacol. (1963), 20, 171-177. THE ACTION OF GUANETHIDINE WITH PARTICULAR REFERENCE TO THE SYMPATHETIC NERVOUS SYSTEM BY G. F. ABERCROMBIE AND B. N. DAVIES From the Department of Physiology,
INDUCTION OF OVULATION IN URETHANE-TREATED RATS
 5 INDUCTION OF OVULATION IN URETHANE-TREATED RATS Ronald D. Johnson* and Barbara Shirley Faculty of Natural Sciences, University of Tulsa, Tulsa, Oklahoma 74104 Subcutaneous injection of urethane (1 g/kg
5 INDUCTION OF OVULATION IN URETHANE-TREATED RATS Ronald D. Johnson* and Barbara Shirley Faculty of Natural Sciences, University of Tulsa, Tulsa, Oklahoma 74104 Subcutaneous injection of urethane (1 g/kg
found to increase with increasing flow rate [Anderson, 1949; Thaysen, Thorn
 Quart. J. exp. Phy8iol. (1966) 51, 202-206 CARBONIC ANHYDRASE ACTIVITY IN SALIVA IN MAN. By I. SZABO,* D. K. MASON and R. McG. HARDEN. From the Dental Hospital and School, and University Department of
Quart. J. exp. Phy8iol. (1966) 51, 202-206 CARBONIC ANHYDRASE ACTIVITY IN SALIVA IN MAN. By I. SZABO,* D. K. MASON and R. McG. HARDEN. From the Dental Hospital and School, and University Department of
suffering from hyperthyroidism before and after treatment have shown variable J. Physiol. (1958) I42,
 447 J. Physiol. (1958) I42, 447-452 BONE CALCIUM AND SODIUM CONTENT AND THE EXCHANGE OF RADIOSODIUM IN BONES FROM RATS TREATED WITH THYROXINE AND PARATHORMONE BY D. S. MUNRO, R. S. SATOSKAR AND G. M. WILSON
447 J. Physiol. (1958) I42, 447-452 BONE CALCIUM AND SODIUM CONTENT AND THE EXCHANGE OF RADIOSODIUM IN BONES FROM RATS TREATED WITH THYROXINE AND PARATHORMONE BY D. S. MUNRO, R. S. SATOSKAR AND G. M. WILSON
HYPOTHALAMIC ELECTRICAL ACTIVITIES PRODUCED BY FACTORS CAUSING DISCHARGE OF PITUITARY HORMONES
 HYPOTHALAMIC ELECTRICAL ACTIVITIES PRODUCED BY FACTORS CAUSING DISCHARGE OF PITUITARY HORMONES TERUO NAKAYAMA* Institute of Physiology, School of Medicine, University of Nagoya It is known that electrical
HYPOTHALAMIC ELECTRICAL ACTIVITIES PRODUCED BY FACTORS CAUSING DISCHARGE OF PITUITARY HORMONES TERUO NAKAYAMA* Institute of Physiology, School of Medicine, University of Nagoya It is known that electrical
possibility of a secretion of adrenaline from the suprarenal glands resulting
 355 J Physiol. (I942) IOI, 355-36I 6i2.014.465:577 I74.5 THE EFFECT OF ANAESTHESIA ON THE ADRENALINE CONTENT OF THE SUPRARENAL GLANDS BY P. C. ELMES AND A. A. JEFFERSON From the Department of Pharmacology,
355 J Physiol. (I942) IOI, 355-36I 6i2.014.465:577 I74.5 THE EFFECT OF ANAESTHESIA ON THE ADRENALINE CONTENT OF THE SUPRARENAL GLANDS BY P. C. ELMES AND A. A. JEFFERSON From the Department of Pharmacology,
THE ACTION OF HANSON'S THYMUS EXTRACT ON THE MOUSE
 140 THE ACTION OF HANSON'S THYMUS EXTRACT ON THE MOUSE BY M. LAFON (From the Department of Genetics, University College, London) (Received July 26, 19) IN a recent paper Hanson (19) described important
140 THE ACTION OF HANSON'S THYMUS EXTRACT ON THE MOUSE BY M. LAFON (From the Department of Genetics, University College, London) (Received July 26, 19) IN a recent paper Hanson (19) described important
Studies on Induced Ovulation in the Intact Immature Hamster. Charles W. Bodemer, Ph.D., Ruth E. Rumery, Ph.D., and Richard J. Blandau, Ph.D., M.D.
 Studies on Induced Ovulation in the Intact Immature Hamster Charles W. Bodemer, Ph.D., Ruth E. Rumery, Ph.D., and Richard J. Blandau, Ph.D., M.D. IT IS WELL KNOWN that gonadotropins are incapable of inducing
Studies on Induced Ovulation in the Intact Immature Hamster Charles W. Bodemer, Ph.D., Ruth E. Rumery, Ph.D., and Richard J. Blandau, Ph.D., M.D. IT IS WELL KNOWN that gonadotropins are incapable of inducing
norepinephrinee." 2 PNMT activity is stimulated by certain adrenocortical markedly,3' 4 but can be restored to normal by the administration of
 IMPAIRED SECRETION OF EPINEPHRINE IN RESPONSE TO INSULIN AMONG HYPOPHYSECTOMIZED DOGS* BY RICHARD J. WURTMAN, ALFRED CASPER, LARISSA A. POHORECKY, AND FREDERIC C. BARTTER DEPARTMENT OF NUTRITION AND FOOD
IMPAIRED SECRETION OF EPINEPHRINE IN RESPONSE TO INSULIN AMONG HYPOPHYSECTOMIZED DOGS* BY RICHARD J. WURTMAN, ALFRED CASPER, LARISSA A. POHORECKY, AND FREDERIC C. BARTTER DEPARTMENT OF NUTRITION AND FOOD
(From the Physiotogicat Laboratory, Cambridge.)
 THE OXYGEN EXCHANGE OF THE SUPRARENAL GLAND. BY K. 0. NEUMAN. (From the Physiotogicat Laboratory, Cambridge.) THIS paper deals with the question of the amount of oxygen taken in by a unit weight of the
THE OXYGEN EXCHANGE OF THE SUPRARENAL GLAND. BY K. 0. NEUMAN. (From the Physiotogicat Laboratory, Cambridge.) THIS paper deals with the question of the amount of oxygen taken in by a unit weight of the
(From the Physiological Laboratory, Cambridge.)
 THE INNERVATION OF THE PYLORIC SPHINCTER OF THE RAT. BY M. NAKANISHI. (From the Physiological Laboratory, Cambridge.) WHILST numerous observations have been made on the behaviour of the pyloric region
THE INNERVATION OF THE PYLORIC SPHINCTER OF THE RAT. BY M. NAKANISHI. (From the Physiological Laboratory, Cambridge.) WHILST numerous observations have been made on the behaviour of the pyloric region
1. What is the embryologic origin of the major salivary glands, and when do they develop? 2. What is the embryologic origin of minor salivary glands?
 Salivary Gland Chapters 37, 38, 39, 40 Shapiro 1. What is the embryologic origin of the major salivary glands, and when do they develop? Outpouchings of oral ectoderm in the 6th 8 th week 2. What is the
Salivary Gland Chapters 37, 38, 39, 40 Shapiro 1. What is the embryologic origin of the major salivary glands, and when do they develop? Outpouchings of oral ectoderm in the 6th 8 th week 2. What is the
Hospital, London, W. 12
 J. Physiol. (1967), 19, pp. 221-228 221 With 3 text-figure8 Printed in Great Britain THE TIME COURSE OF THE EFFECT OF THYROID HORMONES UPON BSL OXYGEN CONSUMPTION ND PLSM CONCENTRTION OF FREE FTTY CID
J. Physiol. (1967), 19, pp. 221-228 221 With 3 text-figure8 Printed in Great Britain THE TIME COURSE OF THE EFFECT OF THYROID HORMONES UPON BSL OXYGEN CONSUMPTION ND PLSM CONCENTRTION OF FREE FTTY CID
Sheet #6. Dr. Heba Kalbouneh. Dr. Heba Kalbouneh. Dr. Heba Kalbouneh
 Sheet #6 Dr. Heba Kalbouneh Dr. Heba Kalbouneh Dr. Heba Kalbouneh Ducts - In large glands, as you go away from the secretory unit, the duct becomes larger and the lining epithelium becomes thicker (from
Sheet #6 Dr. Heba Kalbouneh Dr. Heba Kalbouneh Dr. Heba Kalbouneh Ducts - In large glands, as you go away from the secretory unit, the duct becomes larger and the lining epithelium becomes thicker (from
blood-vessels of the isolated perfused lungs of the rat. Both Hirakawa
 547.435-292: 547.781.5: 577.174.5: 612.215 THE ACTION OF ADRENALINE, ACETYLCHOLINE, AND HIS- TAMINE ON THE LUNGS OF THE RAT. By P. FoGGIE. From the Physiology Department, University of Edinburgh. (Received
547.435-292: 547.781.5: 577.174.5: 612.215 THE ACTION OF ADRENALINE, ACETYLCHOLINE, AND HIS- TAMINE ON THE LUNGS OF THE RAT. By P. FoGGIE. From the Physiology Department, University of Edinburgh. (Received
SEASONAL CHANGES IN HISTOLOGYOF THE THYROID GLAND CALOTIS VERSICOLOR
 SEASONAL CHANGES IN HISTOLOGYOF THE THYROID GLAND CALOTIS VERSICOLOR M. D. Kulkarni And A. H. Shinde Department of Zoology Yashwantrao Chavan Arts & Science College, Mangrulpir Dist. Washim. (Received
SEASONAL CHANGES IN HISTOLOGYOF THE THYROID GLAND CALOTIS VERSICOLOR M. D. Kulkarni And A. H. Shinde Department of Zoology Yashwantrao Chavan Arts & Science College, Mangrulpir Dist. Washim. (Received
by the action of insulin. Loss in adrenocortical lipids is considered an index of
 222 J. Physiol. (I95I) I14, 222-233 THE ROLE OF HYPOGLYCAEMIA AND OF ADRENALINE IN THE RESPONSE OF THE ADRENAL CORTEX TO INSULIN BY MARTHE VOGT From the Departnent of Pharmacology, University of Edinburgh
222 J. Physiol. (I95I) I14, 222-233 THE ROLE OF HYPOGLYCAEMIA AND OF ADRENALINE IN THE RESPONSE OF THE ADRENAL CORTEX TO INSULIN BY MARTHE VOGT From the Departnent of Pharmacology, University of Edinburgh
PARTIAL CHARACTERIZATION OF MONOAMINE OXIDASE IN THE SALIVARY GLAND OF RATS. Kazuko NEMOTO, Seiichi HASHIMOTO, Takeyuki IKENO. and Hiroshi KUZUYA
 PARTIAL CHARACTERIZATION OF MONOAMINE OXIDASE IN THE SALIVARY GLAND OF RATS Kazuko NEMOTO, Seiichi HASHIMOTO, Takeyuki IKENO and Hiroshi KUZUYA Department of Biochemistry, Tohoku Dental University, Tomita-machi,
PARTIAL CHARACTERIZATION OF MONOAMINE OXIDASE IN THE SALIVARY GLAND OF RATS Kazuko NEMOTO, Seiichi HASHIMOTO, Takeyuki IKENO and Hiroshi KUZUYA Department of Biochemistry, Tohoku Dental University, Tomita-machi,
DEPLETION AND REPLACEMENT OF THE ADRENALINE AND NORADRENALINE CONTENTS OF THE RAT ADRENAL GLAND, FOLLOWING TREATMENT WITH RESERPINE
 Brit. J. Pharmacol. (1962), 18, 138-149. DEPLETION AND REPLACEMENT OF THE ADRENALINE AND NORADRENALINE CONTENTS OF THE RAT ADRENAL GLAND, FOLLOWING TREATMENT WITH RESERPINE BY B. A. CALLINGHAM AND MONICA
Brit. J. Pharmacol. (1962), 18, 138-149. DEPLETION AND REPLACEMENT OF THE ADRENALINE AND NORADRENALINE CONTENTS OF THE RAT ADRENAL GLAND, FOLLOWING TREATMENT WITH RESERPINE BY B. A. CALLINGHAM AND MONICA
fully established, although it seems reasonable to assume it is pseudocholinesterase
 DISTRIBUTION OF CHOLINESTERASES IN THE MAMMALIAN PANCREAS. By CATHERINE HEBB and K. J. HILL. From the Agricultural Research Council, Institute of Animal Physiology, Babraham, Cambridge. (Received for publication
DISTRIBUTION OF CHOLINESTERASES IN THE MAMMALIAN PANCREAS. By CATHERINE HEBB and K. J. HILL. From the Agricultural Research Council, Institute of Animal Physiology, Babraham, Cambridge. (Received for publication
Endocrine Glands System. Agha Zohaib Khan
 Endocrine Glands System Agha Zohaib Khan Introduction Endocrine means secreting internally. Indeed, the endocrine system is made up of glands whose secretions enter the blood stream. Hence these glands
Endocrine Glands System Agha Zohaib Khan Introduction Endocrine means secreting internally. Indeed, the endocrine system is made up of glands whose secretions enter the blood stream. Hence these glands
2. Epithelial Tissues Dr. Manal Othman
 Biology-232 GENERAL HISTOLOGY 2. Epithelial Tissues Dr. Manal Othman Anatomy Department CMMS, AGU HISTOLOGY: w Study of the structure and function of tissues and organs at the microscopic levels. w Tissues
Biology-232 GENERAL HISTOLOGY 2. Epithelial Tissues Dr. Manal Othman Anatomy Department CMMS, AGU HISTOLOGY: w Study of the structure and function of tissues and organs at the microscopic levels. w Tissues
Nevertheless, the specific concentrations of amylase and peroxidase (mg/mg protein)
 J. Phy8iol. (1984), 352, pp. 163-171 163 With 1 plate Printed in Great Britain INFLUENCE OF CIRCULATING CATECHOLAMINES ON PROTEIN SECRETION INTO RAT PAROTID SALIVA DURING PARASYMPATHETIC STIMULATION BY
J. Phy8iol. (1984), 352, pp. 163-171 163 With 1 plate Printed in Great Britain INFLUENCE OF CIRCULATING CATECHOLAMINES ON PROTEIN SECRETION INTO RAT PAROTID SALIVA DURING PARASYMPATHETIC STIMULATION BY
SOME OBSERVATIONS UPON SODIUM ALGINATE. By 0. M. SOLANDT. From the Physiological Laboratory, Cambridge.
 582.6 SOME OBSERVATIONS UPON SODIUM ALGINATE. By 0. M. SOLANDT. From the Physiological Laboratory, Cambridge. (Received for publication 13th December 1940.) ALGINIC acid was discovered by Stanford in 1883
582.6 SOME OBSERVATIONS UPON SODIUM ALGINATE. By 0. M. SOLANDT. From the Physiological Laboratory, Cambridge. (Received for publication 13th December 1940.) ALGINIC acid was discovered by Stanford in 1883
induced by sham feeding is accompanied by an increased excretion of University College, London.)
 THE METABOLISM OF THE SALIVARY GLANDS. II. The blood sugar metabolism of the Submaxillary Gland. BY G. V. ANREP AND R. K. CANNAN (Beit Memorial Research Fellow). (From the Institute of Physiology, University
THE METABOLISM OF THE SALIVARY GLANDS. II. The blood sugar metabolism of the Submaxillary Gland. BY G. V. ANREP AND R. K. CANNAN (Beit Memorial Research Fellow). (From the Institute of Physiology, University
NATURAL CAUSES OF VARIATIONS IN THE WEIGHT
 398 NATURAL CAUSES OF VARIATIONS IN THE WEIGHT OF SARCOMA I8 J. P. AUSTIN* AND E. M. GLASERt From the Riker Laboratories, Welwyn Garden City, Hertfordshire Received for publication November 11, 1969 SUMMARY.-The
398 NATURAL CAUSES OF VARIATIONS IN THE WEIGHT OF SARCOMA I8 J. P. AUSTIN* AND E. M. GLASERt From the Riker Laboratories, Welwyn Garden City, Hertfordshire Received for publication November 11, 1969 SUMMARY.-The
Anatomy PHL 212. Dr. Dina A. A. Hassan. -
 Anatomy PHL 212 Dr. Dina A. A. Hassan Associate Professor College of Pharmacy (Female Section) Sattam Bin Abdulaziz University Al kharj / Kingdom of Saudi Arabia Email :- da.hassan@psau.edu.sa 1 Anatomy
Anatomy PHL 212 Dr. Dina A. A. Hassan Associate Professor College of Pharmacy (Female Section) Sattam Bin Abdulaziz University Al kharj / Kingdom of Saudi Arabia Email :- da.hassan@psau.edu.sa 1 Anatomy
6. The diagram below represents an interaction between parts of an organism.
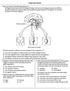 Endocrine Review 1. Base your answer to the following question on the diagram below and on your knowledge of biology. Each arrow in the diagram represents a different hormone released by the pituitary
Endocrine Review 1. Base your answer to the following question on the diagram below and on your knowledge of biology. Each arrow in the diagram represents a different hormone released by the pituitary
..'corisoe.rednis EFFECTS OF PREDNISONE, PREDNISOLONE, AND HYDROCORTISONE A COMPARISON OF THE PITUITARY INHIBITORY
 Brit. J. Pharmacol. (1958), 13, 98. A COMPARISON OF THE PITUITARY INHIBITORY EFFECTS OF PREDNISONE, PREDNISOLONE, AND HYDROCORTISONE BY J. R. HODGES AND JOAN VERNIKOS From the Department of Pharmacology,
Brit. J. Pharmacol. (1958), 13, 98. A COMPARISON OF THE PITUITARY INHIBITORY EFFECTS OF PREDNISONE, PREDNISOLONE, AND HYDROCORTISONE BY J. R. HODGES AND JOAN VERNIKOS From the Department of Pharmacology,
The Endocrine System Pearson Education, Inc.
 19 The Endocrine System Introduction The nervous system and the endocrine system work together to monitor the body s activities The nervous system: produces short-term, very specific responses The endocrine
19 The Endocrine System Introduction The nervous system and the endocrine system work together to monitor the body s activities The nervous system: produces short-term, very specific responses The endocrine
Chapter 20 Endocrine System
 Chapter 20 Endocrine System The endocrine system consists of glands and tissues that secrete Hormones are chemicals that affect other glands or tissues, many times far away from the site of hormone production
Chapter 20 Endocrine System The endocrine system consists of glands and tissues that secrete Hormones are chemicals that affect other glands or tissues, many times far away from the site of hormone production
Gastrin derivatives investigated for secretory potency and for changes in gastric mucosal histamine formation
 Br. J. Pharmac. (1970), 38, 473-477. Gastrin derivatives investigated for secretory potency and for changes in gastric mucosal histamine formation ELSA ROSENGREN AND S. E. SVENSSON Institute of Physiology,
Br. J. Pharmac. (1970), 38, 473-477. Gastrin derivatives investigated for secretory potency and for changes in gastric mucosal histamine formation ELSA ROSENGREN AND S. E. SVENSSON Institute of Physiology,
accompanied by a more rapid flow of secretion. But after administration
 612.34: 612.352.1 THE EFFECT OF INJECTED INSULIN ON THE STORAGE OF GLYCOGEN IN THE PANCREAS AND LIVER. By CATHERINE 0. HEBB. From the Department of Physiology, McGill University, Montreal, Canada. (Received
612.34: 612.352.1 THE EFFECT OF INJECTED INSULIN ON THE STORAGE OF GLYCOGEN IN THE PANCREAS AND LIVER. By CATHERINE 0. HEBB. From the Department of Physiology, McGill University, Montreal, Canada. (Received
CYCLIC MOUSE. and NEENA B. SCHWARTZ INTRODUCTION
 TIMING OF LH RELEASE AND OVULATION IN THE CYCLIC MOUSE AUDREY S. BINGEL and NEENA B. SCHWARTZ Department of Physiology, University of Illinois College of Medicine, Chicago, Illinois, U.S.A. (Received 1st
TIMING OF LH RELEASE AND OVULATION IN THE CYCLIC MOUSE AUDREY S. BINGEL and NEENA B. SCHWARTZ Department of Physiology, University of Illinois College of Medicine, Chicago, Illinois, U.S.A. (Received 1st
Biology of the Salivary Glands 513 (KEY) MID-TERM Examination June 1, 1998
 Biology of the Salivary Glands 513 (KEY) MID-TERM Examination June 1, 1998 1. In the two stage hypothesis of saliva production, the concentration of sodium ions a. in primary saliva is the same regardless
Biology of the Salivary Glands 513 (KEY) MID-TERM Examination June 1, 1998 1. In the two stage hypothesis of saliva production, the concentration of sodium ions a. in primary saliva is the same regardless
The Endocrine System. Lab Exercise 31. Objectives. Introduction
 Lab Exercise The Endocrine System Objectives - Become familiar with the major endocrine glands and their location. - Learn some of the hormones produced by each gland. - Become familiar with the anatomy
Lab Exercise The Endocrine System Objectives - Become familiar with the major endocrine glands and their location. - Learn some of the hormones produced by each gland. - Become familiar with the anatomy
ON GASTRIC SECRETION IN DOGS
 Gut, 960,, 345. THE EFFECT OF AN ADRENAL INHIBITOR (SU 4885) ON GASTRIC SECRETION IN DOGS BY J. W. McINTOSH, N. ANDERSON, H. L. DUTHIE, and A. P. M. FORREST From the University Department of Surgery, Western
Gut, 960,, 345. THE EFFECT OF AN ADRENAL INHIBITOR (SU 4885) ON GASTRIC SECRETION IN DOGS BY J. W. McINTOSH, N. ANDERSON, H. L. DUTHIE, and A. P. M. FORREST From the University Department of Surgery, Western
6I I:6I hypophysectomy. This diminution of diabetes is shown particularly as. hypophysectomized or totally decerebrated [Houssay and
 6I2.466.6I:6I2.492.5 KETOSIS IN THE PANCREATIC AND PHLORRHIZIN DIABETES OF HYPOPHYSECTOMIZED DOGS. BY CIRO T. RIETTI. (Institute of Physiology, Faculty of Medicine, Buenos Ayres.) IN the hypophysectomized
6I2.466.6I:6I2.492.5 KETOSIS IN THE PANCREATIC AND PHLORRHIZIN DIABETES OF HYPOPHYSECTOMIZED DOGS. BY CIRO T. RIETTI. (Institute of Physiology, Faculty of Medicine, Buenos Ayres.) IN the hypophysectomized
RUBENS MOREIRA ARCIERI and CELSO MARTINELLI Department of Oral Pathology, Aragatuba School of Dentistry, Araca.tuba, Brasil
 Tohoku J. exp. Med., 1977, 121, 105-110 Influence of Salivary Glands Extirpation on Procreation in Rats RUBENS MOREIRA ARCIERI and CELSO MARTINELLI Department of Oral Pathology, Aragatuba School of Dentistry,
Tohoku J. exp. Med., 1977, 121, 105-110 Influence of Salivary Glands Extirpation on Procreation in Rats RUBENS MOREIRA ARCIERI and CELSO MARTINELLI Department of Oral Pathology, Aragatuba School of Dentistry,
Histologically the pyloric glands and the glands of Brunner in the. Ivy(7) and his co-workers seem to believe, at the present time, in the
 6I2.323[6I2.33 6I2.OI5.2I THE SMALL INTESTINE AND GASTRIC SECRETION. (With special reference to Brunner's Glands.) BY MARGARET M. MURRAY. (From the Department of Physiology, Bedford College.) EVIDENCE
6I2.323[6I2.33 6I2.OI5.2I THE SMALL INTESTINE AND GASTRIC SECRETION. (With special reference to Brunner's Glands.) BY MARGARET M. MURRAY. (From the Department of Physiology, Bedford College.) EVIDENCE
THE ACTION OF ANTISYMPATHOMIMETIC DRUGS ON THE URINARY EXCRETION OF ADRENALINE AND NORADRENALINE
 Brit. J. Pharmacol. (1959), 14, 380. THE ACTION OF ANTISYMPATHOMIMETIC DRUGS ON THE URINARY EXCRETION OF ADRENALINE AND NORADRENALINE BY B. G. BENFEY, G. LEDOUX, AND M. SEGAL From the Department ofpharmacology,
Brit. J. Pharmacol. (1959), 14, 380. THE ACTION OF ANTISYMPATHOMIMETIC DRUGS ON THE URINARY EXCRETION OF ADRENALINE AND NORADRENALINE BY B. G. BENFEY, G. LEDOUX, AND M. SEGAL From the Department ofpharmacology,
