Characteristics of evaluation panel used for Round 2 of WHO malaria RDT product testing at U.S. CDC, 2009 WHO-FIND malaria RDT evaluation programme
|
|
|
- Alexander Waters
- 5 years ago
- Views:
Transcription
1 (FIND) Special Programme for Research and Training in Tropical Diseases (TDR) Malaria ranch, Division of Parasitic Characteristics of evaluation panel used for Round 2 of WHO malaria RDT product testing at U.S. CDC, 2009 WHO-FIND malaria RDT evaluation programme Introduction The second round of product testing of malaria RDTs under the WHO-FIND malaria RDT evaluation programme was completed in November 2009 at the Malaria ranch, Division of Parasitic. The panel is a sub-set of the global specimen bank, maintained by WHO, FIND and U.S. CDC. This document summarizes the characterization reports of the evaluation panel. The report of the evaluation was released in April Sample collection and preparation Evaluation panel samples consist of culture-derived P. falciparum, 'wild-type' P. falciparum or P. vivax derived from febrile malaria patients in malaria-endemic settings, or parasite-negative samples from people with or without differential diagnoses of malaria or other known blood characteristics that may influence test results. Panel samples are stored at below -70 C, and used once after thawing. Samples were collected, prepared, and characterized at the following collection sites in partnership with the WHO and FIND. Country ISO Institute Address Code Australia AU Army Malaria Institute Enoggera, risbane Cambodia KH Institut Pasteur Cambodge Phnom Penh Central African CF Institut Pasteur angui angui Republic Colombia CO CIDEIM Cali Kenya KE Kenya Medical Research Institute Kisumu Madagascar MG Institut Pasteur de Madagascar Antananarivo Myanmar MM Department of Medical Research Yangon Nigeria NG University of Lagos Lagos Peru PE Instituto de Medicina Tropical Alexander von Lima Humboldt, Universidad Peruana Cayetano Heredia Philippines PH Research Institute for Tropical Medicine Muntinlupa City, Manila Senegal SN Université Cheikh Anta DIOP Dakar Tanzania TZ Ifakara Health Research and Development agamoyo Centre United Kingdom G Hospital for Tropical Disease London USA US Malaria ranch, Division of Parasitic Diseases Centers for Disease Control and Prevention Atlanta 1 WHO/TDR/FIND. Malaria Rapid DiagnosticTest Performance - results of WHO product testing of malaria RDTs: Round 2(2009). Geneva, WHO, Page 1 of 9
2 (FIND) Special Programme for Research and Training in Tropical Diseases (TDR) Malaria ranch, Division of Parasitic Map of sample collection, preparation and characterization institutions. Collection and testing Specimen characterization Global specimen bank HTD CDC DMR CIDEIM UCAD UL IP KEMRI IPC RITM IMT IHRDC IPM QIMR Source: WHO, All rights reserved Abbreviations: CDC Centers for Disease Control and Prevention (Atlanta, United States of America); CIDEIM Centro Internacional de Entrenamiento y Investigaciones Médicas (Cali, Colombia); DMR Experimental Medicine Research Division (Department of Medical Research, Yangon, Myanmar); HTD Hospital for Tropical Diseases (London, United Kingdom of Great ritain and Ireland); IHRDC Ifakara Health Research and Development Center (agamoyo, The United republic of Tanzania); IMT Instituto de Medicina Tropical (Universidad Peruana Cayetano Heredia, Lima, Peru); IP Institut Pasteur de angui (angui, Central African Republic); IPC Institut Pasteur du Cambodge (Phnom Penh, Cambodia); IPM Institut Pasteur de Madagascar (Antananarivo, Madagascar); KEMRI: Kenya Medical Research Institute (Kisumu, Kenya); QIMR Queensland Institute of Medical Research (risbane, Australia); RITM Research Institute of Tropical Medicine (Manila, The Philippines); UCAD: Université Cheikh Anta DIOP (Dakar, Senegal); UL University of Lagos (Lagos, Nigeria).. Details of specimen collection, preparation, characterization and use of panel samples are found in the Methods Manual for Laboratory Quality Control Testing or Malaria Rapid Diagnostic Tests (1), and the Methods Manual for Product Testing of Malaria Rapid Diagnostic Tests (2). Collection and characterization of parasite-positive samples is as follows: Parasites are derived from monoclonal culture lines, or volunteering patients with fever and evidence of malaria parasitaemia. Page 2 of 9
3 (FIND) Special Programme for Research and Training in Tropical Diseases (TDR) Malaria ranch, Division of Parasitic Parasite-positive samples have parasite density determined by two pre-qualified microscopists performing blinded readings, with a required concordance on parasite density within 20%. Dilution to standardized parasite densities is then based on a mean of these two results. All wild-type parasite samples are screened for HIV, hepatitis and hepatitis C viruses, and virus-positive (HIV, Hep, HepC) samples are excluded. Species is confirmed by PCR for P. falciparum, P. vivax, P. malariae, P. ovale and P. knowlesi, and multi-species infections are excluded. The concentration of the three target antigens, HRP2 (falciparum only); pldh; and aldolase, which is determined by quantitative ELISA. HRP2 primary sequence is determined. Collection and characterization of parasite-negative samples is as follows: Parasite-negative samples are derived from volunteers with specific blood types or diseases considered differential diagnoses of malaria, or from stored blood bank blood of other volunteers. Malaria is excluded by PCR for P. falciparum, P. vivax, P. malariae, P. ovale and P. knowlesi. Note on HRP2 sequence classification: The primary structure of HRP2 is classified according to a value calculated using the number of the common Type 2 and Type 7 amino acid repeat sequences in the protein, as identified by aker et al. (3): A ( 100 repeats), Type (50 to 99 repeats), and Type C ( 50 repeats). The primary structure is determined as it could theoretically affect RDT sensitivity, but its effect may be limited (5,6). Samples used in Round 2 evaluation panel RDTs in Round 2 were evaluated against three panels, specifically: i) P. falciparum culture lines (includes a subset, 'manufacturer's panel') at low (200 parasites/μl) and high parasite densities (2000 or 5000 parasites/μl), one sample (US01FNigeriaXII) was also used as the standard for heat stability testing. ii) Wild-type Plasmodium samples (P. falciparum, P. vivax) from naturally infected humans and parasite-negative samples at low (200 parasites/μl) and high densities (2000 or 5000 parasites/μl). iii) Parasite-negative panel ('clean' samples and disease-specific or blood factorspecific samples). A summary of outcomes of characterization of samples, including summary statistics for antigen concentrations at 200 parasites/μl, are given below. Page 3 of 9
4 (FIND) Special Programme for Research and Training in Tropical Diseases (TDR) Malaria ranch, Division of Parasitic Phase 1 (culture-derived) panel HRP2 primary structure classification of Phase 1 panel Culture line US01F7G8 US01FeninI US01ForneoI US01FECP US01FFC27/A3 US01FFCH4 US01FGhanaIII US01FGhanaV US01FGuineaI US01FH3 US01FKE4 US01FKI28 US01FMalawiI US01FNigeriaXII US01FPeru01064 US01FPH1 US01FPNG144 US01FSabahIII US01FSantaLucia US01FUgandaII HRP2 structure A A C C A Antigen concentration (ng/ml) of Phase 1 panel at 200 parasites/μl pldh HRP2 Aldolase Mean Median Standard deviation Maximum Minimum Results based on 20 samples (HRP2, pldh) and 12 samples (aldolase) Page 4 of 9
5 (FIND) Special Programme for Research and Training in Tropical Diseases (TDR) Malaria ranch, Division of Parasitic Phase 2 patient-derived P. falciparum parasite panel Geographical origin of Phase 2 P. falciparum -positive panel Region Country N South America Colombia 10 Peru 4 Central African Republic 13 Kenya 2 Africa Madagascar 6 Nigeria 23 Tanzania 18 Asia Cambodia 19 Myanmar 2 Philippines 3 Total 100 Antigen concentration (ng/ml) of Phase 2 (patient derived) P. falciparum -positive panel at 200 parasites/μl pldh HRP2 Aldolase Mean Median Standard deviation Maximum Minimum Concentrations of 1 or 2 antigens excluded from 11 samples where insufficient correctly-labeled sample were available for analysis Page 5 of 9
6 (FIND) Special Programme for Research and Training in Tropical Diseases (TDR) Malaria ranch, Division of Parasitic HRP2 primary structure classification of Phase 2 P. falciparum-positive panel Sample HRP2 HRP2 HRP2 Sample HRP2 ID type 1 Sample ID type 1 Sample ID type 1 ID type 1 CF01F01 KH03F06 C NG01F17 TZ01F13 MC CF01F02 MC KH03F07 C NG01F20 MC TZ01F14 A CF01F04 KH03F08 NG01F22 TZ01F15 CF01F05 A KH03F09 NG01F23 TZ01F16 MC CF01F06 C KH04F01 C NG01F24 TZ01F17 MC CF01F07 KH04F02 C NG01F25 A TZ01F18 CF01F09 C KH04F03 C NG02F01 A TZ01F20 CF01F13 KH04F04 NG02F02 TZ01F23 CF01F15 KH04F05 C NG02F08 TZ01F24 CF01F18 MC KH04F06 NG02F13 A TZ01F25 CF01F19 NA KH05F01 A NG02F16 TZ01F26 MC CF01F22 NA KH05F06 NG02F17 TZ01F27 A CF01F23 NA KH05F09 NG02F19 TZ01F28 A CO01F02 KH05F10 MC NG02F22 A CO01F04 KH05F11 NG02F25 CO01F05 MG01F01 A NG02F26 CO01F08 MG01F02 NG02F33 CO01F09 MG01F14 PE01F04 CO01F11 MG01F19 PE02F01 CO01F12 MG01F20 PE02F04 CO01F13 MG01F22 PE02F29 CO01F16 MM04F02 NA PH7F04 A CO01F18 MM09F01 PH08F07 A KE01F05 NA NG01F01 PH08F09 C KE01F14 NA NG01F02 TZ01F01 MC KH03F01 MC NG01F05 TZ01F02 MC KH03F03 NG01F06 TZ01F07 C KH03F04 NG01F08 A TZ01F11 KH03F05 NG01F16 TZ01F12 1. MC: Probable multi-clone P. falciparum infection 2. NA: Not available Page 6 of 9
7 (FIND) Special Programme for Research and Training in Tropical Diseases (TDR) Malaria ranch, Division of Parasitic Phase 2 patient-derived P. vivax parasite panel Geographical origin of Phase 2 P. vivax-positive panel Region Country N South America Colombia 4 Peru 22 Africa Madagascar 1 Asia Cambodia 11 Philippines 2 Total 40 Antigen concentration (ng/ml) of Phase 2 P. vivaxpositive panel at 200 parasites/μl pldh Aldolase Mean Median Standard deviation Maximum Minimum Concentrations of 1 or 2 antigens excluded from 5 samples where insufficient correctly-labeled sample were available for analysis The distribution of antigen levels for Pf HRP2, Pf pldh and Pv pldh were compared between the Round 1 and Round 2 parasite panels to ensure consistency. No statistically significant differences in median antigen levels between the Round 1 and 2 panels were detected for any of the 3 antigens (P>0.2, Mann-Whitney test). Page 7 of 9
8 (FIND) Special Programme for Research and Training in Tropical Diseases (TDR) Malaria ranch, Division of Parasitic Phase 2 parasite-negative panel Geographical origin of 'clean' parasite-negative panel Country N Madagascar 1 Nigeria 3 Philippines 20 Senegal 8 USA 18 Total 50 Parasite-negative panel with known disease or blood abnormality characteristics Sample type N Dengue 4 RPR positive 9 ANA 13 Schistosomiasis 10 Rheumatoid Factor 4 Leishmaniasis 5 Chagas 2 Human anti-mouse antibody 3 Total 50 Page 8 of 9
9 (FIND) Special Programme for Research and Training in Tropical Diseases (TDR) Malaria ranch, Division of Parasitic References 1. WHO/FIND/TDR, Rapid Diagnostic Tests for Malaria: Methods Manual for Laboratory Quality Control Testing (Version 6) 2010, WHO - Regional Office for the Western Pacific, TDR, FIND WHO/FIND/CDC, Methods manual for product testing of malaria rapid diagnostic tests (version 3) aker, J., et al., Genetic diversity of Plasmodium falciparum histidine-rich protein 2 (PfHRP2) and its effect on the performance of PfHRP2-based rapid diagnostic tests. J Infect Dis, (5): p Lee, N., et al., Effect of sequence variation in Plasmodium falciparum histidinerich protein 2 on binding of specific monoclonal antibodies: Implications for rapid diagnostic tests for malaria. J Clin Microbiol, (8): p Lee, N., et al., Assessing the genetic diversity of the aldolase genes of Plasmodium falciparum and Plasmodium vivax and its potential effect on performance of aldolase-detecting rapid diagnostic tests. J Clin Microbiol, (12): p Talman, A.M., et al., Evaluation of the intra- and inter-specific genetic variability of Plasmodium lactate dehydrogenase. Malar J, : p Page 9 of 9
Characteristics of evaluation panel used for Round 4 of WHO Malaria RDT Product Testing at U.S. CDC,
 Programme for Research and Training in Tropical Diseases (TDR) Malaria ranch, Division of Parasitic Diseases Centers for Characteristics of evaluation panel used for Round 4 of WHO Malaria RDT Product
Programme for Research and Training in Tropical Diseases (TDR) Malaria ranch, Division of Parasitic Diseases Centers for Characteristics of evaluation panel used for Round 4 of WHO Malaria RDT Product
Initiative for Quality Assurance of Malaria Rapid Diagnostic Tests
 Initiative for Quality Assurance of Malaria Rapid Diagnostic Tests Outline of product testing and associated protocols World Health Organization (WHO) Regional Office for the Western Pacific UNICEF / UNDP
Initiative for Quality Assurance of Malaria Rapid Diagnostic Tests Outline of product testing and associated protocols World Health Organization (WHO) Regional Office for the Western Pacific UNICEF / UNDP
Lot Testing of malaria RDTs: achievements and lessons learned from a 7 year-long experience
 Lot Testing of malaria RDTs: achievements and lessons learned from a 7 year-long experience Incardona S., Luchavez J., Nhem S., Barnwell J., Champouillon N., Chiodini P., Luna C., Ménard D., Meth R., Rees-Channer
Lot Testing of malaria RDTs: achievements and lessons learned from a 7 year-long experience Incardona S., Luchavez J., Nhem S., Barnwell J., Champouillon N., Chiodini P., Luna C., Ménard D., Meth R., Rees-Channer
Malaria Rapid Diagnostic Test Performance. Results of WHO product testing of malaria RDTs: round 8 ( )
 Malaria Rapid Diagnostic Test Performance Results of WHO product testing of malaria RDTs: round 8 (2016 2018) Malaria Rapid Diagnostic Test Performance Results of WHO product testing of malaria RDTs:
Malaria Rapid Diagnostic Test Performance Results of WHO product testing of malaria RDTs: round 8 (2016 2018) Malaria Rapid Diagnostic Test Performance Results of WHO product testing of malaria RDTs:
Malaria Rapid Diagnostic Test Performance. Results of WHO product testing of malaria RDTs: round 7 ( )
 Malaria Rapid Diagnostic Test Performance Results of WHO product testing of malaria RDTs: round 7 (2015 2016) Malaria Rapid Diagnostic Test Performance Results of WHO product testing of malaria RDTs:
Malaria Rapid Diagnostic Test Performance Results of WHO product testing of malaria RDTs: round 7 (2015 2016) Malaria Rapid Diagnostic Test Performance Results of WHO product testing of malaria RDTs:
Molecular Profile and Survey of HRP2 and HRP3 Genetic Deletions in South America: An Update
 Molecular Profile and Survey of HRP2 and HRP3 Genetic Deletions in South America: An Update John W. Barnwell and Kumar Udhayakumar Malaria Research & Development Laboratory Unit Malaria Branch, DPDM, CGH
Molecular Profile and Survey of HRP2 and HRP3 Genetic Deletions in South America: An Update John W. Barnwell and Kumar Udhayakumar Malaria Research & Development Laboratory Unit Malaria Branch, DPDM, CGH
Malaria Rapid Diagnostic Tests: role and place in the diagnosis of malaria
 Malaria Rapid Diagnostic Tests: role and place in the diagnosis of malaria 1 Plasmodium falciparum Malaria : an overview 2 Plasmodium vivax 3 Plasmodium ovale Most serious Jan Jacobs Institute of Tropical
Malaria Rapid Diagnostic Tests: role and place in the diagnosis of malaria 1 Plasmodium falciparum Malaria : an overview 2 Plasmodium vivax 3 Plasmodium ovale Most serious Jan Jacobs Institute of Tropical
HUMASIS MALARIA ANTIGEN TEST HIGH SENSITIVE DIFFERENTIAL DIAGNOSIS OF MALARIA INFECTION
 HUMASIS MALARIA ANTIGEN TEST HIGH SENSITIVE DIFFERENTIAL DIAGNOSIS OF MALARIA INFECTION References 1) World Malaria Report 2010, WHO 2) Rapid diagnostic tests for malaria parasites, Clin. Microbiol. Rev.
HUMASIS MALARIA ANTIGEN TEST HIGH SENSITIVE DIFFERENTIAL DIAGNOSIS OF MALARIA INFECTION References 1) World Malaria Report 2010, WHO 2) Rapid diagnostic tests for malaria parasites, Clin. Microbiol. Rev.
False positive results for malaria rapid diagnostic tests. in patients with rheumatoid factor. Seung Gyu Yun c, Chae Seung Lim a *
 JCM Accepts, published online ahead of print on 23 July 2014 J. Clin. Microbiol. doi:10.1128/jcm.01797-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 2 False positive results
JCM Accepts, published online ahead of print on 23 July 2014 J. Clin. Microbiol. doi:10.1128/jcm.01797-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 2 False positive results
Malaria Rapid Diagnostic Tests
 Clinical Infectious Diseases Advance Access published May 1, 2012 INVITED ARTICLE MEDICAL MICROBIOLOGY L. Barth Reller and Melvin P. Weinstein, Section Editors Malaria Rapid Diagnostic Tests Michael L.
Clinical Infectious Diseases Advance Access published May 1, 2012 INVITED ARTICLE MEDICAL MICROBIOLOGY L. Barth Reller and Melvin P. Weinstein, Section Editors Malaria Rapid Diagnostic Tests Michael L.
Proposed WHO Technical Consultation on external competence assessment for malaria microscopy
 Proposed WHO Technical Consultation on external competence assessment for malaria microscopy Malaria Policy Advisory Committee Meeting 17-19 October 2018, Executive Board meeting room World Health Organization,
Proposed WHO Technical Consultation on external competence assessment for malaria microscopy Malaria Policy Advisory Committee Meeting 17-19 October 2018, Executive Board meeting room World Health Organization,
Performance of Rapid Diagnostic Tests for Plasmodium ovale Malaria in Japanese Travellers
 Tropical Medicine and Health Vol. 42 No.4, 2014, 149 153 doi: 10.2149/tmh.2014-07 Copyright 2014 by The Japanese Society of Tropical Medicine 149 Original Paper Performance of Rapid Diagnostic Tests for
Tropical Medicine and Health Vol. 42 No.4, 2014, 149 153 doi: 10.2149/tmh.2014-07 Copyright 2014 by The Japanese Society of Tropical Medicine 149 Original Paper Performance of Rapid Diagnostic Tests for
T he diagnosis of malaria in clinical laboratories in the UK
 862 ORIGINAL ARTICLE Use of rapid diagnostic tests for diagnosis of malaria in the UK D Chilton, A N J Malik, M Armstrong, M Kettelhut, J Parker-Williams, P L Chiodini... See end of article for authors
862 ORIGINAL ARTICLE Use of rapid diagnostic tests for diagnosis of malaria in the UK D Chilton, A N J Malik, M Armstrong, M Kettelhut, J Parker-Williams, P L Chiodini... See end of article for authors
Malaria Rapid Diagnostic Test Performance. Summary results of WHO product testing of malaria RDTs: round 1-7 ( )
 Malaria Rapid Diagnostic Test Performance Summary results of WHO product testing of malaria RDTs: round 1-7 (2008 2016) Malaria Rapid Diagnostic Test Performance Summary results of WHO product testing
Malaria Rapid Diagnostic Test Performance Summary results of WHO product testing of malaria RDTs: round 1-7 (2008 2016) Malaria Rapid Diagnostic Test Performance Summary results of WHO product testing
Annex 2 A. Regional profile: West Africa
 Annex 2 A. Regional profile: West Africa 355 million people at risk for malaria in 215 297 million at high risk A. Parasite prevalence, 215 Funding for malaria increased from US$ 233 million to US$ 262
Annex 2 A. Regional profile: West Africa 355 million people at risk for malaria in 215 297 million at high risk A. Parasite prevalence, 215 Funding for malaria increased from US$ 233 million to US$ 262
Sysmex Educational Enhancement and Development No
 SEED Malaria Sysmex Educational Enhancement and Development No 1 2017 Malaria diagnostics in the era of improved malaria control The purpose of this newsletter is to provide an overview of the role and
SEED Malaria Sysmex Educational Enhancement and Development No 1 2017 Malaria diagnostics in the era of improved malaria control The purpose of this newsletter is to provide an overview of the role and
Downloaded from:
 Gatton, ML; Ciketic, S; Barnwell, JW; Cheng, Q; Chiodini, PL; Incardona, S; Bell, D; Cunningham, J; Gonzlez, IJ (2018) An assessment of false positive rates for malaria rapid diagnostic tests caused by
Gatton, ML; Ciketic, S; Barnwell, JW; Cheng, Q; Chiodini, PL; Incardona, S; Bell, D; Cunningham, J; Gonzlez, IJ (2018) An assessment of false positive rates for malaria rapid diagnostic tests caused by
WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT
 WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT Product: One Step test for Malaria Pf/Pv Ag MERISCREEN Malaria Pf/Pv Ag WHO reference number: PQDx 0294-074-00 One Step test for Malaria Pf/Pv
WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT Product: One Step test for Malaria Pf/Pv Ag MERISCREEN Malaria Pf/Pv Ag WHO reference number: PQDx 0294-074-00 One Step test for Malaria Pf/Pv
DOWNLOAD OR READ : TROPICAL INFECTIOUS DISEASES SECOND EDITION PDF EBOOK EPUB MOBI
 DOWNLOAD OR READ : TROPICAL INFECTIOUS DISEASES SECOND EDITION PDF EBOOK EPUB MOBI Page 1 Page 2 tropical infectious diseases second edition tropical infectious diseases second pdf tropical infectious
DOWNLOAD OR READ : TROPICAL INFECTIOUS DISEASES SECOND EDITION PDF EBOOK EPUB MOBI Page 1 Page 2 tropical infectious diseases second edition tropical infectious diseases second pdf tropical infectious
Evaluation of a rapid diagnostic test specific for Plasmodium vivax
 Tropical Medicine and International Health doi:10.1111/j.1365-3156.2008.02163.x volume 13 no 12 pp 1495 1500 december 2008 Evaluation of a rapid diagnostic test specific for Plasmodium vivax Sun Hyung
Tropical Medicine and International Health doi:10.1111/j.1365-3156.2008.02163.x volume 13 no 12 pp 1495 1500 december 2008 Evaluation of a rapid diagnostic test specific for Plasmodium vivax Sun Hyung
For research on diseases of poverty. UNICEF UNDP World Bank WHO TDR: HEALTH RESEARCH THAT MAKES AN IMPACT
 For research on diseases of poverty UNICEF UNDP World Bank WHO TDR: HEALTH RESEARCH THAT MAKES AN IMPACT TDR page 1 page 2 TDR QUICK FACTS ABOUT TDR TDR supports infectious disease research and strengthens
For research on diseases of poverty UNICEF UNDP World Bank WHO TDR: HEALTH RESEARCH THAT MAKES AN IMPACT TDR page 1 page 2 TDR QUICK FACTS ABOUT TDR TDR supports infectious disease research and strengthens
Kato, N. et al, Nature, 2016, Accelerated Article Preview. Celeste Alverez Current Literature 9/10/ /10/2016 Celeste Wipf Group 1
 Kato, N. et al, Nature, 2016, Accelerated Article Preview Celeste Alverez Current Literature 9/10/2016 9/10/2016 Celeste Alverez @ Wipf Group 1 Malaria Caused by Plasmodium parasites Carried by 30-40 species
Kato, N. et al, Nature, 2016, Accelerated Article Preview Celeste Alverez Current Literature 9/10/2016 9/10/2016 Celeste Alverez @ Wipf Group 1 Malaria Caused by Plasmodium parasites Carried by 30-40 species
The Three Millennium Development Goal Fund A guide for the selection of Malaria Rapid Diagnostic Test kits purchased with 3MDG grants
 The Three Millennium Development Goal Fund A guide for the selection of Malaria Rapid Diagnostic Test kits purchased with 3MDG grants Version 4.0 February 2014 1. Contents A GUIDE FOR THE SELECTION OF
The Three Millennium Development Goal Fund A guide for the selection of Malaria Rapid Diagnostic Test kits purchased with 3MDG grants Version 4.0 February 2014 1. Contents A GUIDE FOR THE SELECTION OF
Int.J.Curr.Microbiol.App.Sci (2015) 4(3):
 ISSN: 2319-7706 Volume 4 Number 3 (2015) pp. 98-107 http://www.ijcmas.com Original Research Article Role of a Parasite Lactate Dehydrogenase-Based Immunochromatographic Antigen Detection Assay for the
ISSN: 2319-7706 Volume 4 Number 3 (2015) pp. 98-107 http://www.ijcmas.com Original Research Article Role of a Parasite Lactate Dehydrogenase-Based Immunochromatographic Antigen Detection Assay for the
Malaria Rapid Diagnostic Test Performance. Summary results of WHO product testing of malaria RDTs: round 1-8 ( )
 Malaria Rapid Diagnostic Test Performance Summary results of WHO product testing of malaria RDTs: round 1-8 (2008 2018) Malaria Rapid Diagnostic Test Performance Summary results of WHO product testing
Malaria Rapid Diagnostic Test Performance Summary results of WHO product testing of malaria RDTs: round 1-8 (2008 2018) Malaria Rapid Diagnostic Test Performance Summary results of WHO product testing
Malaria Journal. Open Access RESEARCH
 https://doi.org/10.1186/s12936-017-2151-y Malaria Journal RESEARCH Unclear association between levels of Plasmodium falciparum lactate dehydrogenase (PfLDH) in saliva of malaria patients and blood parasitaemia:
https://doi.org/10.1186/s12936-017-2151-y Malaria Journal RESEARCH Unclear association between levels of Plasmodium falciparum lactate dehydrogenase (PfLDH) in saliva of malaria patients and blood parasitaemia:
PARASITOLOGY CASE HISTORY #14 (BLOOD PARASITES) (Lynne S. Garcia)
 PARASITOLOGY CASE HISTORY #14 (BLOOD PARASITES) (Lynne S. Garcia) A 37-year-old woman, who had traveled to New Guinea for several weeks, presented to the medical clinic with fever, chills, and rigors within
PARASITOLOGY CASE HISTORY #14 (BLOOD PARASITES) (Lynne S. Garcia) A 37-year-old woman, who had traveled to New Guinea for several weeks, presented to the medical clinic with fever, chills, and rigors within
Rapid diagnostic tests failing to detect Plasmodium falciparum infections in Eritrea: an investigation of reported false negative RDT results
 DOI 10.1186/s12936-017-1752-9 Malaria Journal RESEARCH Open Access Rapid diagnostic tests failing to detect Plasmodium falciparum infections in Eritrea: an investigation of reported false negative RDT
DOI 10.1186/s12936-017-1752-9 Malaria Journal RESEARCH Open Access Rapid diagnostic tests failing to detect Plasmodium falciparum infections in Eritrea: an investigation of reported false negative RDT
Target Product Profile: Point-of-Care Malaria Plasmodium falciparum Highly Sensitive Rapid Diagnostic Test
 1 Target Product Profile: Point-of-Care Malaria Plasmodium falciparum Highly Sensitive Rapid Diagnostic Test For rapid detection of low-density, malaria infections Updated November 2015 2 Context Defining
1 Target Product Profile: Point-of-Care Malaria Plasmodium falciparum Highly Sensitive Rapid Diagnostic Test For rapid detection of low-density, malaria infections Updated November 2015 2 Context Defining
Malaria Pf/pan antigen Rapid Test
 Malaria Pf/pan antigen Rapid Test Cat. No.:IVDTS003 Pkg.Size:10T/50T Intended use The Malaria Pf/pan antigen Rapid Test is a self-performing, qualitative, sandwich immunoassay, utilizing whole blood for
Malaria Pf/pan antigen Rapid Test Cat. No.:IVDTS003 Pkg.Size:10T/50T Intended use The Malaria Pf/pan antigen Rapid Test is a self-performing, qualitative, sandwich immunoassay, utilizing whole blood for
Towards an Atlas of Human Helminth Infection in sub-saharan Africa: The Use of Geographical Information Systems (GIS)
 Towards an Atlas of Human Helminth Infection in sub-saharan Africa: The Use of Geographical Information Systems (GIS) S. Brooker, M. Rowlands, L. Haller, L. Savioli and D.A.P. Bundy * *Ndir, O. et al.
Towards an Atlas of Human Helminth Infection in sub-saharan Africa: The Use of Geographical Information Systems (GIS) S. Brooker, M. Rowlands, L. Haller, L. Savioli and D.A.P. Bundy * *Ndir, O. et al.
ESCMID Online Lecture Library. by author
 Laboratory diagnosis of Malaria handout 1 Lisette van Lieshout & Eric Brienen LvanLieshout@Lumc.nl healthy blood Microscopy thick blood smear haemolysis RBC + staining healthy blood Microscopy thin blood
Laboratory diagnosis of Malaria handout 1 Lisette van Lieshout & Eric Brienen LvanLieshout@Lumc.nl healthy blood Microscopy thick blood smear haemolysis RBC + staining healthy blood Microscopy thin blood
Summary World Malaria Report 2010
 Summary The summarizes information received from 106 malaria-endemic countries and other partners and updates the analyses presented in the 2009 Report. It highlights continued progress made towards meeting
Summary The summarizes information received from 106 malaria-endemic countries and other partners and updates the analyses presented in the 2009 Report. It highlights continued progress made towards meeting
The global context for financing delivery innovations in fever case management and malaria treatment. Ramanan Laxminarayan
 The global context for financing delivery innovations in fever case management and malaria treatment Ramanan Laxminarayan Changed environment 1. Greater challenges in continued funding for global health
The global context for financing delivery innovations in fever case management and malaria treatment Ramanan Laxminarayan Changed environment 1. Greater challenges in continued funding for global health
Malaria. Edwin J. Asturias, MD
 Malaria Edwin J. Asturias, MD Associate Professor of Pediatrics and Epidemiology Director for Latin America Center for Global Health, Colorado School of Public Health Global Health and Disasters Course
Malaria Edwin J. Asturias, MD Associate Professor of Pediatrics and Epidemiology Director for Latin America Center for Global Health, Colorado School of Public Health Global Health and Disasters Course
Status of RTS,S/AS01 malaria vaccine candidate
 Status of RTS,S/AS01 malaria vaccine candidate Multi-center RTS,S malaria vaccine efficacy trial Phase 3, randomized, controlled, double-blind trial conducted in 11 centers in 7 African countries 15,460
Status of RTS,S/AS01 malaria vaccine candidate Multi-center RTS,S malaria vaccine efficacy trial Phase 3, randomized, controlled, double-blind trial conducted in 11 centers in 7 African countries 15,460
Malaria in pregnancy programmes: challenges and priorities in antimalarial drug development for African pregnant women
 7 th MIM Pan African Malaria Conference 15-20 April 2018 Dakar, Senegal The power of sharing science EDCTP session on Malaria in pregnancy programmes: challenges and priorities in antimalarial drug development
7 th MIM Pan African Malaria Conference 15-20 April 2018 Dakar, Senegal The power of sharing science EDCTP session on Malaria in pregnancy programmes: challenges and priorities in antimalarial drug development
Pandemic H1N Dr. Maria Neira Global Influenza Programme WHO, Geneva
 Pandemic H1N1 2010 Dr. Maria Neira Global Influenza Programme WHO, Geneva WHO Role during pandemic (H1N1) 2009 Under the International Health Regulations (2005) Detect event (notification by Member States
Pandemic H1N1 2010 Dr. Maria Neira Global Influenza Programme WHO, Geneva WHO Role during pandemic (H1N1) 2009 Under the International Health Regulations (2005) Detect event (notification by Member States
O c t o b e r 1 0,
 STUDENT VOICES FROM THE FIELD: WORLD HEALTH ORGANIZATION GENEVA, SWITZERLAND O c t o b e r 1 0, 2 0 1 6 M o l l y P e z z u l o University at Albany School of Public Health MPH Epidemiology Candidate 17
STUDENT VOICES FROM THE FIELD: WORLD HEALTH ORGANIZATION GENEVA, SWITZERLAND O c t o b e r 1 0, 2 0 1 6 M o l l y P e z z u l o University at Albany School of Public Health MPH Epidemiology Candidate 17
Geographic Opportunistic Infections
 Epidemiology of HIV Associated Opportunistic Infections in the United States Siriluck Anunnatsiri, MD Division of Infectious Diseases and Tropical Medicine Department of Medicine, Khon Kaen University
Epidemiology of HIV Associated Opportunistic Infections in the United States Siriluck Anunnatsiri, MD Division of Infectious Diseases and Tropical Medicine Department of Medicine, Khon Kaen University
International Journal of Pharma and Bio Sciences
 Research Article Medical Microbiology International Journal of Pharma and Bio Sciences ISSN 0975-6299 COMPARISON OF RAPID IMMUNOCHROMATOGRAPHIC ASSAYS (ICT MALARIA P.F. /P.V. TEST AND OPTIMAL TEST) WITH
Research Article Medical Microbiology International Journal of Pharma and Bio Sciences ISSN 0975-6299 COMPARISON OF RAPID IMMUNOCHROMATOGRAPHIC ASSAYS (ICT MALARIA P.F. /P.V. TEST AND OPTIMAL TEST) WITH
Post Travel Fever. Dr. Eyal Leshem. Center for Geographic Medicine Sheba Medical Center Tel Hashomer, Israel
 Post Travel Fever Dr. Eyal Leshem Center for Geographic Medicine Sheba Medical Center Tel Hashomer, Israel Introduction Fever in returned traveler: Trivial vs. life threatening infections Tropical and
Post Travel Fever Dr. Eyal Leshem Center for Geographic Medicine Sheba Medical Center Tel Hashomer, Israel Introduction Fever in returned traveler: Trivial vs. life threatening infections Tropical and
Malaria (Pan-LDH) W/B
 CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
Fighting Harder and Smarter Against Malaria. Dr.Bernard Nahlen Deputy US Global Malaria Coordinator University of Georgia, February 23, 2010
 Fighting Harder and Smarter Against Malaria Dr.Bernard Nahlen Deputy US Global Malaria Coordinator University of Georgia, February 23, 2010 Outline Burden of malaria Global support for rolling back malaria
Fighting Harder and Smarter Against Malaria Dr.Bernard Nahlen Deputy US Global Malaria Coordinator University of Georgia, February 23, 2010 Outline Burden of malaria Global support for rolling back malaria
1 introduction strengthening and ac- celerating future vaccine innovation and development for diseases of poverty.
 European Vaccine Initiative 1 Introduction Infectious diseases are key contributors to the devastating poverty in much of the world today. Every year diseases of poverty kill almost 9 million people, many
European Vaccine Initiative 1 Introduction Infectious diseases are key contributors to the devastating poverty in much of the world today. Every year diseases of poverty kill almost 9 million people, many
Challenges and Opportunities for Responding to HIV/AIDS in LDCs. Mazuwa Banda Department of HIV/AIDS World Health Organization
 Challenges and Opportunities for Responding to HIV/AIDS in LDCs Mazuwa Banda Department of HIV/AIDS World Health Organization Outline of Presentation Global HIV/AIDS epidemic HIV/AIDS and LDCs Challenges
Challenges and Opportunities for Responding to HIV/AIDS in LDCs Mazuwa Banda Department of HIV/AIDS World Health Organization Outline of Presentation Global HIV/AIDS epidemic HIV/AIDS and LDCs Challenges
Factsheet about Chikungunya
 Factsheet about Chikungunya Chikungunya fever is a viral disease transmitted to humans by infected mosquitoes that is characterized by fever, headache, rash, and severe joint and muscle pain. The name
Factsheet about Chikungunya Chikungunya fever is a viral disease transmitted to humans by infected mosquitoes that is characterized by fever, headache, rash, and severe joint and muscle pain. The name
PARASITOLOGY CASE HISTORY #11 (BLOOD PARASITES) (Lynne S. Garcia)
 PARASITOLOGY CASE HISTORY #11 (BLOOD PARASITES) (Lynne S. Garcia) A 39-year old male traveler developed fever and thrombocytopenia after returning from a trip to the Philippines. The parasitemia was 10,000
PARASITOLOGY CASE HISTORY #11 (BLOOD PARASITES) (Lynne S. Garcia) A 39-year old male traveler developed fever and thrombocytopenia after returning from a trip to the Philippines. The parasitemia was 10,000
REPORT OF THE TECHNICAL CONSULTATION BACKGROUND
 Defining and validating a measure of parasite resistance to sulfadoxinepyrimethamine (SP) that would be indicative of the protective efficacy of SP for intermittent preventive treatment in infancy (SP-IPTi)
Defining and validating a measure of parasite resistance to sulfadoxinepyrimethamine (SP) that would be indicative of the protective efficacy of SP for intermittent preventive treatment in infancy (SP-IPTi)
Malaria Rapid Diagnostic Test: A Study of Accuracy among Adults in North Central Nigeria
 Journal of Medicine in the Tropics (2011) 14:1: 7-11 Journal of Medicine in the Tropics Original Article Malaria Rapid Diagnostic Test: A Study of Accuracy among Adults in North Central Nigeria 1 1 2 2
Journal of Medicine in the Tropics (2011) 14:1: 7-11 Journal of Medicine in the Tropics Original Article Malaria Rapid Diagnostic Test: A Study of Accuracy among Adults in North Central Nigeria 1 1 2 2
Malaria Combo Test Kit
 CLIAwaived,Inc. Combo Test Kit A rapid test for the qualitative detection of Plasmodium falciparum and/or Plasmodium vivax, Plasmodium orale and Plasmodium malariae antigen in whole blood. For in-vitro
CLIAwaived,Inc. Combo Test Kit A rapid test for the qualitative detection of Plasmodium falciparum and/or Plasmodium vivax, Plasmodium orale and Plasmodium malariae antigen in whole blood. For in-vitro
Good practices for selecting and procuring
 WHO Global Malaria Programme Good practices for selecting and procuring rapid diagnostic tests for malaria Good practices for selecting and procuring rapid diagnostic tests for malaria Good practices for
WHO Global Malaria Programme Good practices for selecting and procuring rapid diagnostic tests for malaria Good practices for selecting and procuring rapid diagnostic tests for malaria Good practices for
Rapid diagnostic tests for diagnosing uncomplicated non-falciparum or Plasmodium vivax malaria in endemic countries (Review)
 Rapid diagnostic tests for diagnosing uncomplicated nonfalciparum or Plasmodium vivax malaria in endemic countries (Review) Abba K, Kirkham AJ, Olliaro PL, Deeks JJ, Donegan S, Garner P, Takwoingi Y This
Rapid diagnostic tests for diagnosing uncomplicated nonfalciparum or Plasmodium vivax malaria in endemic countries (Review) Abba K, Kirkham AJ, Olliaro PL, Deeks JJ, Donegan S, Garner P, Takwoingi Y This
or Plasmodium vivax malaria in endemic countries (Review)
 Cochrane Database of Systematic Reviews Rapid diagnostic tests for diagnosing uncomplicated nonfalciparum or Plasmodium vivax malaria in endemic countries (Review) AbbaK,KirkhamAJ,OlliaroPL,DeeksJJ,DoneganS,GarnerP,TakwoingiY
Cochrane Database of Systematic Reviews Rapid diagnostic tests for diagnosing uncomplicated nonfalciparum or Plasmodium vivax malaria in endemic countries (Review) AbbaK,KirkhamAJ,OlliaroPL,DeeksJJ,DoneganS,GarnerP,TakwoingiY
Roadmaps as a Vehicle for Addressing Large- Scale Public Health Challenges: Lessons from the Malaria Vaccine Technology Roadmap
 Background to development of vaccine roadmaps, to include priority areas for research, product development, capacities, and policy and commercialization Roadmaps as a Vehicle for Addressing Large- Scale
Background to development of vaccine roadmaps, to include priority areas for research, product development, capacities, and policy and commercialization Roadmaps as a Vehicle for Addressing Large- Scale
GLOBAL TB IMPACT MEASUREMENT
 GLOBAL TB IMPACT MEASUREMENT What is it? Why is it important? How can it be done? What will it cost? 1. What is global TB impact measurement and why is it important? Global TB Impact Measurement is the
GLOBAL TB IMPACT MEASUREMENT What is it? Why is it important? How can it be done? What will it cost? 1. What is global TB impact measurement and why is it important? Global TB Impact Measurement is the
Field evaluation of HRP2 and pan pldh-based immunochromatographic assay in therapeutic monitoring of uncomplicated falciparum malaria in Myanmar
 Nyunt et al. Malaria Journal 2013, 12:123 RESEARCH Open Access Field evaluation of HRP2 and pan pldh-based immunochromatographic assay in therapeutic monitoring of uncomplicated falciparum malaria in Myanmar
Nyunt et al. Malaria Journal 2013, 12:123 RESEARCH Open Access Field evaluation of HRP2 and pan pldh-based immunochromatographic assay in therapeutic monitoring of uncomplicated falciparum malaria in Myanmar
Influenza Update N 159
 Influenza Update N 159 10 May 2012 Summary The seasonal peak for influenza has passed in most countries in the temperate regions of the northern hemisphere. Different viruses have predominated in different
Influenza Update N 159 10 May 2012 Summary The seasonal peak for influenza has passed in most countries in the temperate regions of the northern hemisphere. Different viruses have predominated in different
- LABELING. I. Labeling of the RDT Box: Product code (or CDC identification):
 Product code (or CDC identification): Technician name: Manufacturer: Technician ID: NOTE: Questions in yellow are aimed to be addressed by/after manufacturers - LABELING I. Labeling of the RDT Box: Reply
Product code (or CDC identification): Technician name: Manufacturer: Technician ID: NOTE: Questions in yellow are aimed to be addressed by/after manufacturers - LABELING I. Labeling of the RDT Box: Reply
Blood Smears Only 6 October Sample Preparation and Quality Control 15B-K
 NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 6 October 5 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only is
NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 6 October 5 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only is
Xiaodong et al. Xiaodong et al. Malaria Journal 2013, 12:6
 Diagnostic performance of CareStart malaria HRP2/pLDH (Pf/pan) combo test versus standard microscopy on falciparum and vivax malaria between China-Myanmar endemic borders Xiaodong et al. Xiaodong et al.
Diagnostic performance of CareStart malaria HRP2/pLDH (Pf/pan) combo test versus standard microscopy on falciparum and vivax malaria between China-Myanmar endemic borders Xiaodong et al. Xiaodong et al.
MP Biomedicals Asia Pacific Pte. Ltd. (formerly Genelabs Diagnostics Pte. Ltd.)
 Revision: 12 May 2005 1 WESTERN BLOT / IMMUNOBLOT HIV-1 BLOT Version 1.3* 11010-018 HIV-1 viral lysate Western Blot assay for the 11010-036 detection of antibodies to HIV-1 with serum 11010-108 108 strips
Revision: 12 May 2005 1 WESTERN BLOT / IMMUNOBLOT HIV-1 BLOT Version 1.3* 11010-018 HIV-1 viral lysate Western Blot assay for the 11010-036 detection of antibodies to HIV-1 with serum 11010-108 108 strips
Swaziland National Malaria Elimination Policy
 DRAFT 21 April 2010 Swaziland National Malaria Elimination Policy Version 1.0 May 2010 Kingdom of Swaziland Ministry of Health National Malaria Control Programme TABLE OF CONTENTS Acronyms... 3 Foreword...
DRAFT 21 April 2010 Swaziland National Malaria Elimination Policy Version 1.0 May 2010 Kingdom of Swaziland Ministry of Health National Malaria Control Programme TABLE OF CONTENTS Acronyms... 3 Foreword...
A rapid test for the qualitative detection of Malaria pf and pv antigen in human blood sample
 A rapid test for the qualitative detection of Malaria pf and pv antigen in human blood sample For in vitro use only Intended Use For the rapid qualitative determination of Malaria P. falciparum specific
A rapid test for the qualitative detection of Malaria pf and pv antigen in human blood sample For in vitro use only Intended Use For the rapid qualitative determination of Malaria P. falciparum specific
Antimalarial drug resistance
 Antimalarial drug resistance Md Mushfiqur Rahman*, Leonard Ortega**, R M Rastogi* and Krongthong Thimasarn* Abstract Antimalarial drug resistance is of great concern in the WHO South-East Asia (SEA) Region.
Antimalarial drug resistance Md Mushfiqur Rahman*, Leonard Ortega**, R M Rastogi* and Krongthong Thimasarn* Abstract Antimalarial drug resistance is of great concern in the WHO South-East Asia (SEA) Region.
Is there Artemisinin Resistance in Western Cambodia?
 Is there Artemisinin Resistance in Western Cambodia? Preliminary results, February 2008 Arjen Dondorp on behalf of the Task Force on Antimalarial Drug Resistance in Cambodia S769N PfATPase6 mutation Artemether
Is there Artemisinin Resistance in Western Cambodia? Preliminary results, February 2008 Arjen Dondorp on behalf of the Task Force on Antimalarial Drug Resistance in Cambodia S769N PfATPase6 mutation Artemether
TECHNICAL REPORT SECOND SLIDE PANEL EXTERNAL QUALITY ASSURANCE PROGRAM FOR MALARIA MICROSCOPY DIAGNOSIS
 TECHNICAL REPORT SECOND SLIDE PANEL 2012-2013 EXTERNAL QUALITY ASSURANCE PROGRAM FOR MALARIA MICROSCOPY DIAGNOSIS REGIONAL MALARIA PROGRAM NEGLECTED, TROPICAL AND VECTOR-BORNE DISEASES COMMUNICABLE DISEASES
TECHNICAL REPORT SECOND SLIDE PANEL 2012-2013 EXTERNAL QUALITY ASSURANCE PROGRAM FOR MALARIA MICROSCOPY DIAGNOSIS REGIONAL MALARIA PROGRAM NEGLECTED, TROPICAL AND VECTOR-BORNE DISEASES COMMUNICABLE DISEASES
Anopheles freeborni. Courtesy
 Anopheles freeborni Courtesy Plasmodia seen with the microscope M. Lontie, MCH, Leuven, 2012 Diagnosis of malaria Thin film (better for species identification). Thick film (more sensitive). QBC (quantitative
Anopheles freeborni Courtesy Plasmodia seen with the microscope M. Lontie, MCH, Leuven, 2012 Diagnosis of malaria Thin film (better for species identification). Thick film (more sensitive). QBC (quantitative
Original Article. Abstract. Introduction
 Original Article Role of ICT Malaria Immunochromatographic Test for Rapid diagnosis of Malaria Mahadev S. Harani, M.Asim Beg 2, Lubna Khaleeq 3, Salman N. Adil 4, Ghulam Nabi Kakepoto 5, Mohammad Khurshid
Original Article Role of ICT Malaria Immunochromatographic Test for Rapid diagnosis of Malaria Mahadev S. Harani, M.Asim Beg 2, Lubna Khaleeq 3, Salman N. Adil 4, Ghulam Nabi Kakepoto 5, Mohammad Khurshid
BRIEFING ON RTS,S/AS01 MALARIA VACCINE FOR THE SEPTEMBER 2012 MEETING OF MPAC
 BRIEFING ON RTS,S/AS01 MALARIA VACCINE FOR THE SEPTEMBER 2012 MEETING OF MPAC Introduction Date: 12 August 2012. Author: WHO Secretariat with input from JTEG Chair The most advanced vaccine candidate against
BRIEFING ON RTS,S/AS01 MALARIA VACCINE FOR THE SEPTEMBER 2012 MEETING OF MPAC Introduction Date: 12 August 2012. Author: WHO Secretariat with input from JTEG Chair The most advanced vaccine candidate against
Malaria Updates. Fe Esperanza Espino Department of Parasitology Research Institute for Tropical Medicine
 Malaria Updates Fe Esperanza Espino Department of Parasitology Research Institute for Tropical Medicine Outline General epidemiology of malaria in the Philippines P falciparum Updates in treatment Recognizing
Malaria Updates Fe Esperanza Espino Department of Parasitology Research Institute for Tropical Medicine Outline General epidemiology of malaria in the Philippines P falciparum Updates in treatment Recognizing
4/16/2013 FARIDA OESMAN DEPARTMENT OF CLINICAL PATHOLOGY FACULTY OF MEDICINE, UNIVERSITY OF INDONESIA TYPHOID FEVER DENGUE FEVER MALARIA LEPTOSPIROSIS
 FARIDA OESMAN DEPARTMENT OF CLINICAL PATHOLOGY FACULTY OF MEDICINE, UNIVERSITY OF INDONESIA TYPHOID FEVER DENGUE FEVER MALARIA LEPTOSPIROSIS NOT SPECIFIC DEFINITIVE HOST RESPONSE HEMATOLOGY ACUTE PHASE
FARIDA OESMAN DEPARTMENT OF CLINICAL PATHOLOGY FACULTY OF MEDICINE, UNIVERSITY OF INDONESIA TYPHOID FEVER DENGUE FEVER MALARIA LEPTOSPIROSIS NOT SPECIFIC DEFINITIVE HOST RESPONSE HEMATOLOGY ACUTE PHASE
Malaria Situation and Plan for Elimination in China
 Malaria Situation and Plan for Elimination in China Dr. Guan Yayi National Institute of Parasitic Diseases, China CDC Current Situation Contents Vivax malaria in China The Support from Global Fund Malaria
Malaria Situation and Plan for Elimination in China Dr. Guan Yayi National Institute of Parasitic Diseases, China CDC Current Situation Contents Vivax malaria in China The Support from Global Fund Malaria
FARIDA OESMAN DEPARTMENT OF CLINICAL PATHOLOGY FACULTY OF MEDICINE, UNIVERSITY OF INDONESIA
 FARIDA OESMAN DEPARTMENT OF CLINICAL PATHOLOGY FACULTY OF MEDICINE, UNIVERSITY OF INDONESIA TYPHOID FEVER DENGUE FEVER MALARIA LEPTOSPIROSIS NOT SPECIFIC DEFINITIVE HOST RESPONSE HEMATOLOGY ACUTE PHASE
FARIDA OESMAN DEPARTMENT OF CLINICAL PATHOLOGY FACULTY OF MEDICINE, UNIVERSITY OF INDONESIA TYPHOID FEVER DENGUE FEVER MALARIA LEPTOSPIROSIS NOT SPECIFIC DEFINITIVE HOST RESPONSE HEMATOLOGY ACUTE PHASE
Field Performance of Wondfo and SD Bioline Malaria Pf/Pan Rapid Diagnostic Tests for Malaria Diagnosis in Koraput District, Odisha State, India
 American Journal of Epidemiology and Infectious Disease, 2015, Vol. 3, No. 2, 21-27 Available online at http://pubs.sciepub.com/ajeid/3/2/1 Science and Education Publishing DOI:10.12691/ajeid-3-2-1 Field
American Journal of Epidemiology and Infectious Disease, 2015, Vol. 3, No. 2, 21-27 Available online at http://pubs.sciepub.com/ajeid/3/2/1 Science and Education Publishing DOI:10.12691/ajeid-3-2-1 Field
INTRODUCTION MATERIALS AND METHODS
 Am. J. Trop. Med. Hyg., 76(3), 2007, pp. 481 485 Copyright 2007 by The American Society of Tropical Medicine and Hygiene WHICH MALARIA RAPID TEST FOR MADAGASCAR? FIELD AND LABORATORY EVALUATION OF THREE
Am. J. Trop. Med. Hyg., 76(3), 2007, pp. 481 485 Copyright 2007 by The American Society of Tropical Medicine and Hygiene WHICH MALARIA RAPID TEST FOR MADAGASCAR? FIELD AND LABORATORY EVALUATION OF THREE
EPIDEMIOLOGICAL SURVEILLANCE REPORT Malaria in Greece, 2012
 HELLENIC CENTRE FOR DISEASE CONTROL AND PREVENTION MINISTRY OF HEALTH Page 1 EPIDEMIOLOGICAL SURVEILLANCE REPORT Malaria in Greece, 2012 Introduction Malaria is a parasitic infection, transmitted through
HELLENIC CENTRE FOR DISEASE CONTROL AND PREVENTION MINISTRY OF HEALTH Page 1 EPIDEMIOLOGICAL SURVEILLANCE REPORT Malaria in Greece, 2012 Introduction Malaria is a parasitic infection, transmitted through
Malaria (Pf/Vivax) W/B
 CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
 Malaria parasites Malaria parasites are micro-organisms that belong to the genus Plasmodium. There are more than 100 species of Plasmodium, which can infect many animal species such as reptiles, birds,
Malaria parasites Malaria parasites are micro-organisms that belong to the genus Plasmodium. There are more than 100 species of Plasmodium, which can infect many animal species such as reptiles, birds,
IX. IMPROVING MATERNAL HEALTH: THE NEED TO FOCUS ON REACHING THE POOR. Eduard Bos The World Bank
 IX. IMPROVING MATERNAL HEALTH: THE NEED TO FOCUS ON REACHING THE POOR Eduard Bos The World Bank A. INTRODUCTION This paper discusses the relevance of the ICPD Programme of Action for the attainment of
IX. IMPROVING MATERNAL HEALTH: THE NEED TO FOCUS ON REACHING THE POOR Eduard Bos The World Bank A. INTRODUCTION This paper discusses the relevance of the ICPD Programme of Action for the attainment of
What happened earlier. APMEN-PATH CONSULTATION Manila, 10 / 11 February 2015
 What happened earlier APMEN-PATH CONSULTATION Manila, 10 / 11 February 2015 Background G6PD is essential enzyme of PPP G6PD gene is located on the X- chromosome: Homozygous, hemizygous individuals Heterozygous
What happened earlier APMEN-PATH CONSULTATION Manila, 10 / 11 February 2015 Background G6PD is essential enzyme of PPP G6PD gene is located on the X- chromosome: Homozygous, hemizygous individuals Heterozygous
Severe Plasmodium knowlesi infection with multiorgan involvement in north east peninsular Malaysia
 Tropical Biomedicine 31(1): 31 35 (2014) Severe Plasmodium knowlesi infection with multiorgan involvement in north east peninsular Malaysia Azidah, A.K. 1, Mohd Faizal, M.A. 1, Lili, H.Y. 1* and Zeehaida,
Tropical Biomedicine 31(1): 31 35 (2014) Severe Plasmodium knowlesi infection with multiorgan involvement in north east peninsular Malaysia Azidah, A.K. 1, Mohd Faizal, M.A. 1, Lili, H.Y. 1* and Zeehaida,
No relevant conflicts of interest to disclose WISCONSIN STATE LABORATORY OF HYGIENE - UNIVERSITY OF WISCONSIN
 No relevant conflicts of interest to disclose Disease background Diagnostics Contents Collecting blood Preparing smears Staining Interpreting slides Molecular and Antigen testing New and emerging causes
No relevant conflicts of interest to disclose Disease background Diagnostics Contents Collecting blood Preparing smears Staining Interpreting slides Molecular and Antigen testing New and emerging causes
GREATER MEKONG SUB-REGION ELIMINATION OF MALARIA (GEMS) PRIVATE SECTOR MALARIA SURVEILLANCE
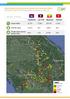 GREATER MEKONG SUB-REGION ELIMINATION OF MALARIA (GEMS) PRIVATE SECTOR MALARIA SURVEILLANCE JANUARY JUNE 218 Cambodia Lao PDR Myanmar Vietnam Cases tested 51,71 17,162 291,317 4,334 Positive cases 6,43
GREATER MEKONG SUB-REGION ELIMINATION OF MALARIA (GEMS) PRIVATE SECTOR MALARIA SURVEILLANCE JANUARY JUNE 218 Cambodia Lao PDR Myanmar Vietnam Cases tested 51,71 17,162 291,317 4,334 Positive cases 6,43
宮村参考人提出資料 資料 2-2
 宮村参考人提出資料 資料 - Highlights of New Wild Poliovirus and cvdpv Positives Reported Globally this Week WPV cvdpv AFP other AFP other Afghanistan 5 Pakistan 3 Nigeria Location: state/province/governorate Kunar
宮村参考人提出資料 資料 - Highlights of New Wild Poliovirus and cvdpv Positives Reported Globally this Week WPV cvdpv AFP other AFP other Afghanistan 5 Pakistan 3 Nigeria Location: state/province/governorate Kunar
APPLICATION FOR REVISION AND INCLUSION OF MALARIA MEDICINES IN WHO MODEL LIST OF ESSENTIAL MEDICINES
 APPLICATION FOR REVISION AND INCLUSION OF MALARIA MEDICINES IN WHO MODEL LIST OF ESSENTIAL MEDICINES The objective of this application is to assure compatibility between the WHO Model list of essential
APPLICATION FOR REVISION AND INCLUSION OF MALARIA MEDICINES IN WHO MODEL LIST OF ESSENTIAL MEDICINES The objective of this application is to assure compatibility between the WHO Model list of essential
Repellent Soap. The Jojoo Mosquito. Africa s innovative solution to Malaria prevention. Sapphire Trading Company Ltd
 The Jojoo Mosquito Repellent Soap Africa s innovative solution to Malaria prevention Sapphire Trading Company Ltd P.O.Box: 45938-00100 Nairobi, Kenya. Tel: +254 735 397 267 +254 733 540 868 +254 700 550
The Jojoo Mosquito Repellent Soap Africa s innovative solution to Malaria prevention Sapphire Trading Company Ltd P.O.Box: 45938-00100 Nairobi, Kenya. Tel: +254 735 397 267 +254 733 540 868 +254 700 550
WORLD MALARIA REPORT SUMMARY
 2016 WORLD MALARIA REPORT SUMMARY WHO/HTM/GMP/2017.4 World Health Organization 2017 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO
2016 WORLD MALARIA REPORT SUMMARY WHO/HTM/GMP/2017.4 World Health Organization 2017 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO
Evaluation of the ICT Malaria P.f/P.v and the OptiMal Rapid Diagnostic Tests for Malaria in Febrile Returned Travellers
 JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 2002, p. 4166 4171 Vol. 40, No. 11 0095-1137/02/$04.00 0 DOI: 10.1128/JCM.40.11.4166 4171.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved.
JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 2002, p. 4166 4171 Vol. 40, No. 11 0095-1137/02/$04.00 0 DOI: 10.1128/JCM.40.11.4166 4171.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved.
Global Alert & Response (GAR) Leptospirosis. Global Alert & Response (GAR)
 Leptospirosis Leptospirosis, a zoonotic and environmental disease a zoonotic and environmental disease Bacteria hosted in animals' kidneys for months/years Environment contaminated by urine (weeks/months)
Leptospirosis Leptospirosis, a zoonotic and environmental disease a zoonotic and environmental disease Bacteria hosted in animals' kidneys for months/years Environment contaminated by urine (weeks/months)
ASIA PACIFIC MALARIA GUIDE A QUICK REFERENCE FOR JOURNALISTS AND OTHERS INTERESTED IN MALARIA AND ITS ELIMINATION IN THE ASIA PACIFIC
 ASIA PACIFIC MALARIA GUIDE A QUICK REFERENCE FOR JOURNALISTS AND OTHERS INTERESTED IN MALARIA AND ITS ELIMINATION IN THE ASIA PACIFIC ACRONYMS AND ABBREVIATIONS ACT APLMA APMEN bn CDC DDT DOT FDA GHG IPT
ASIA PACIFIC MALARIA GUIDE A QUICK REFERENCE FOR JOURNALISTS AND OTHERS INTERESTED IN MALARIA AND ITS ELIMINATION IN THE ASIA PACIFIC ACRONYMS AND ABBREVIATIONS ACT APLMA APMEN bn CDC DDT DOT FDA GHG IPT
PROGRESS REPORT ON DECADE OF TRADITIONAL MEDICINE IN THE AFRICAN REGION. Progress Report. CONTENTS Paragraphs BACKGROUND PROGRESS MADE...
 5 July 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 17.2 PROGRESS REPORT ON DECADE OF TRADITIONAL
5 July 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 17.2 PROGRESS REPORT ON DECADE OF TRADITIONAL
Lessons learned from the IeDEA West Africa Collaboration
 Lessons learned from the IeDEA West Africa Collaboration François DABIS with the contribution of Didier Koumavi EKOUEVI INSERM U-897, Bordeaux, France, Programme PACCI, ANRS site, Abidjan, Côte d Ivoire
Lessons learned from the IeDEA West Africa Collaboration François DABIS with the contribution of Didier Koumavi EKOUEVI INSERM U-897, Bordeaux, France, Programme PACCI, ANRS site, Abidjan, Côte d Ivoire
PROGRESS REPORT ON CHILD SURVIVAL: A STRATEGY FOR THE AFRICAN REGION. Information Document CONTENTS
 29 June 2009 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, 31 August 4 September 2009 Provisional agenda item 9.2 PROGRESS REPORT ON CHILD SURVIVAL: A
29 June 2009 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, 31 August 4 September 2009 Provisional agenda item 9.2 PROGRESS REPORT ON CHILD SURVIVAL: A
Malaria screening of donors and donations - issues and outcomes
 Malaria screening of donors and donations - issues and outcomes lan Kitchen National Transfusion Microbiology NHS Blood & Transplant London IPF/PEI, Rome, May 2014 Blood and Transplant Key elements Nature
Malaria screening of donors and donations - issues and outcomes lan Kitchen National Transfusion Microbiology NHS Blood & Transplant London IPF/PEI, Rome, May 2014 Blood and Transplant Key elements Nature
Pre- Travel Case Studies (*with Key Pads)
 Pre- Travel Case Studies (*with Key Pads) Judi Piasecki, RN, BN Dana Male, RN, BN Pam White RN, BN Certificate in Travel Health A. Which year was the first Manitoba Travel Health Conference? 1. 1999 2.
Pre- Travel Case Studies (*with Key Pads) Judi Piasecki, RN, BN Dana Male, RN, BN Pam White RN, BN Certificate in Travel Health A. Which year was the first Manitoba Travel Health Conference? 1. 1999 2.
Financing malaria control
 Chapter 6. Financing malaria control The three major sources of funds for malaria control pro - grammes are national government spending, external assistance from donors and household or private out-of-pocket
Chapter 6. Financing malaria control The three major sources of funds for malaria control pro - grammes are national government spending, external assistance from donors and household or private out-of-pocket
Use of the Rapid BinaxNOW Malaria Test in a 24-Hour Laboratory Associated with
 JCM Accepts, published online ahead of print on 13 February 2013 J. Clin. Microbiol. doi:10.1128/jcm.00293-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 3 Use of the Rapid
JCM Accepts, published online ahead of print on 13 February 2013 J. Clin. Microbiol. doi:10.1128/jcm.00293-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 3 Use of the Rapid
Access to reproductive health care global significance and conceptual challenges
 08_XXX_MM1 Access to reproductive health care global significance and conceptual challenges Dr Lale Say World Health Organization Department of Reproductive Health and Research From Research to Practice:
08_XXX_MM1 Access to reproductive health care global significance and conceptual challenges Dr Lale Say World Health Organization Department of Reproductive Health and Research From Research to Practice:
Emerging TTIs How Singapore secure its blood supply
 Emerging TTIs How Singapore secure its blood supply Ms Sally Lam Acting Division Director I Blood Supply Management I Blood Services Group I Health Sciences Outline Risk Mitigation Strategies to secure
Emerging TTIs How Singapore secure its blood supply Ms Sally Lam Acting Division Director I Blood Supply Management I Blood Services Group I Health Sciences Outline Risk Mitigation Strategies to secure
