The development of a manageable medical
|
|
|
- Esther Holt
- 5 years ago
- Views:
Transcription
1 Developing a Rational Approach for the Use of Growth Hormone in npediatric Patients David Cook, MD; and Gary Owens, MD The development of a manageable medical policy that ensures appropriate use of recombinant human growth hormone (hgh) has been elusive. Sorting through the misconceptions, fads, cosmetic uses, and economics of this therapy is difficult even for the most sophisticated medical policy or pharmacy and therapeutics committees. An effective policy enables the quick identification of appropriate patients using minimal resources. A poorly structured policy can create a hostile environment in which extra resources are spent on many members in an effort to prevent inappropriate use by a few members. The following article applies the principles of ethics, expert scientific input, and an appreciation of the economic impact each approved treatment regimen will place on the healthcare system. The roundtable faculty functioned as a medical policy committee, and the sample policies are presented to stimulate discussions within health plans to assess current growth hormone (GH) policies and challenge the standard of care while respecting the need to manage hgh proactively. Medical Policy for Adults Defining the appropriate adult hgh patient population is controversial. Based on the US Food and Drug Administration (FDA) labeling, published studies, and expert opinions, the panel stratified the potential uses into 4 categories: Approved: FDA labeled indication Accepted: Use supported by numerous clinical trials published in peer-reviewed journals; widely accepted use within the endocrinology community Possible: Medical rationale for use (less published support) is that it improves outcomes Medical Rationale: Uses described in case reports or fad uses; no clinical trials available to support improvement in clinical outcomes The objective of the committee was to prioritize uses that have documented clinical benefit in reducing morbidity and/or mortality. By employing the above criteria, the cosmetic uses of hgh were placed in the lowest categories. Table 1 summarizes the current uses by category. The uses in the approved and accepted categories are routinely incorporated in many medical policies of managed care organizations (MCOs). These categories represent the core of an inclusive medical policy. However, some health plans have chosen to limit their definition of appropriate use to a subset of these clinical uses based on their own review of the literature and the recommendations of local endocrinologists. The variation in covered uses leads to confusion within the provider community and may result in additional time for the plan and endocrinologist to verify plan coverage and navigate the prior authorization process. In adult patients, accepted hgh therapy included treatment of growth hormone deficiency (GHD) resulting from pituitary disease, hypothalamic disease, irradiation or trauma to the pituitary, and reconfirmed childhood GHD. In addition, GH replacement therapy for, chronic renal failure,, acquired immune deficiency syndrome (AIDS) wasting, and short-bowel syndrome are supported by significant clinical data. On the other hand, a host of other conditions were S436 THE AMERICAN JOURNAL OF MANAGED CARE OCTOBER 2004
2 Developing a Rational Approach for the Use of Growth Hormone in npediatric Patients considered inappropriate therapy, including fibromyalgia, chronic fatigue syndrome, and obesity, among others. The treatment of idiopathic short stature and isolated GHD were considered gray areas areas in which treatment would be possible, but would need to be considered on a case-by-case basis. The diagnostic workup requirements to establish GHD, as defined by medical policy, are often a source of considerable confusion and frustration. Documentation of GHD can be convoluted based on the patient presentation and the sensitivity of the test specified in the policy. A previous article in this supplement discussed the benefits and challenges associated with each test. Often MCO medical policies specify the need for 2 tests or do not specify which tests are preferred. One health plan reported a pattern of some endocrinologists to require clonidine and levodopa for their policy. Because of low specificity of these tests, a number of patients with questionable criteria have been approved. The concept of using a stringent test (ie, a test that payers and providers can rely on) is emerging. The insulin tolerance test (ITT) and/or the arginine GHreleasing hormone (GHRH) tests currently fit this description. This case illustrates that only defining when to test and the number of tests in a medical policy is not sufficient; a list of which tests are acceptable for a diagnosis needs to be included. Follow-up monitoring requirements can also be confusing. Health plans waste resources because testing requirements are not necessary, inappropriate, or place the member at undo risk. Clearly, there is a need to develop evidence-based guidelines for the use of hgh in adults to ensure that the appropriate patients are tested for GHD and receive hgh therapy. The Figure displays an algorithm for the assessment and treatment of GHD in adults, based on the workshop discussion and the clinical evidence presented in this supplement. When this algorithm is coupled with the use categories listed in Table 1 and streamlined testing requirements, a degree of clarity emerges to provide an objective assessment of each potential GH patient. Limiting tests to only those with the greatest specificity and removing testing require- Table 1. hgh Uses in Adults Approved uses Accepted uses Possible uses medical rationale Long-term replacement therapy in adults with GHD of either childhood- or adultonset etiology. GHD either alone or with multiple hormone deficiencies (hypopituitarism) as a result of pituitary disease, hypothalamic disease, surgery, radiation therapy, or trauma Reconfirmed childhood GHD AIDS wasting or cachexia CRF up to time of renal transplantation CRF Panhypopituitarism AIDS wasting Idiopathic short stature Short-bowel syndrome Fibromyalgia Chronic fatigue Obesity Performance enhancement Short stature congenital conditions such as Down s, Fanconi s, or Bloom s syndrome Cystic fibrosis Antiaging End-stage CHF Osteoporosis Infertility Muscular dystrophy Enhanced sexual performance Regrowth of heart, liver, spleen, kidneys, and other organs that shrink with age Improved resistance to infection and cancer Faster wound healing Hair regrowth Sharper vision and hearing Mood elevation Crohn s disease hgh indicates recombinant human growth hormone; GHD, growth hormone deficiency; AIDS, acquired immune deficiency syndrome; CRF, chronic renal failure; CHF, congestive heart failure. VOL. 10, NO. 13, SUP. THE AMERICAN JOURNAL OF MANAGED CARE S437
3 Figure. Sample Adult hgh Coverage Policy Algorithm Overall Assessment Other Pituitary Deficiencies TSH, ACTH, LH/FSH, and ADH 3 Stim Test Needed Possible 1-2 Deficiencies IGF-1 Baseline MRI R/O Other Causes MRI Start GH Tumor GHD Likely Slim Test Tumor Perform ARG/L-dopa 5 Further documentation needed Possible denial Assess for Contraindication IGF-1 Baseline Approval Start GH Perform ITT or ARG/GHRH 5 Further documentation needed Possible denial hgh indicates recombinant human growth hormone; TSH, thyroid-stimulating hormone; ACTH, adrenocorticotropic hormone; LH/FSH, luteinizing hormone/follicle-stimulating hormone; ADH, antidiuretic hormone; GHD, growth hormone deficiency; IGF-1, insulin-like growth factor 1; MRI, magnetic resonance imaging; R/O, rule out; ITT, insulin tolerance test; ARG, adrenergic receptor variant allele; L-dopa, levodopa; GHRH, growth hormone releasing hormone. ments in those patients with the highest probability of disease quickly identifies appropriate patients. As with all algorithms, flexibility to manage specific situations needs to be included to accommodate patients with contraindications to specific tests or an atypical GHD presentation. Obtaining an insulinlike growth factor 1 (IGF-1) level at baseline is also recommended prior to starting therapy. Since IGF-1 levels are an effective method to monitor the response to hgh therapy, this baseline level provides a monitoring mechanism that is reliable and cost effective. Managing the Transitional Patient The transitional population is unique because persons of the same age may be classified as pediatric, adult, or transitional. The definition of a transitional patient includes those who received hgh as a child for a pediatric indication and those whose hgh therapy was stopped upon reaching epiphyses fusion. This definition excludes patients diagnosed with GHD later in life and recognizes these individuals as adults. Childhood-onset GHD because of anatomical damage to the pituitary, irradiation, or trauma, or the presence of panhypopituitarism is sufficient to consider continuing hgh therapy into adulthood (Table 2). However, the panel could not reach a consensus as to whether isolated GHD, longterm renal insufficiency, or the presence of or were compelling indications for the use of hgh in transitional patients. Other gray areas for the use of hgh in transitional patients include the treatment of shortbowel syndrome and AIDS wasting. The primary issue is determining which individuals with known GHD in childhood S438 THE AMERICAN JOURNAL OF MANAGED CARE OCTOBER 2004
4 Developing a Rational Approach for the Use of Growth Hormone in npediatric Patients should be tested for ongoing treatment. As in the adult population, if a patient exhibits at least 3 pituitary hormone deficiencies, then provocation testing would not be clinically necessary; however, magnetic resonance imaging (MRI) and IGF-1 data should be obtained to establish baseline information of these patients. If only 1 or 2 pituitary deficiencies are present, provocation testing is warranted, preferably an ITT or arginine- GHRH. Then, an MRI should be performed as part of the workup for anterior pituitary disease, and an IGF-1 level should be obtained for baseline purposes. Transitional patients already have a significant risk for osteopenia, an increase of visceral fat, and decreased muscle mass. Consequently, they exhibit many of the features of adult GHD. Indeed, the metabolic effects of GHD are thought to be similar in transitional and adult patients with GHD. Patients with and, approved indications for hgh until epiphyses fusion, should receive an evaluation of need before approval to continue for the transitional patients. Duration of therapy for transitional patients is not clear. Periodic testing using IGF-1 is suggested to determine the continued need for hgh therapy. Conclusion Developing an evidence-based hgh medical policy for nonpediatric patients is a complex process. In addition, medical and prescription policies are never static. There is a continual need to balance evolving scientific information and an ever-increasing requirement to manage increasing medical costs. Practice patterns vary across the country, and an assessment of the plan s provider network prescribing practices is a starting point. Understanding the patient with GH deficiency that is routinely rejected provides an initial focus for policy revision or provider education. Overly restrictive policies may actually increase the long-term cost to the health plan as the metabolic complications of GHD develop. Broad policies, on the other hand, may yield lower than expected clinical outcomes, resulting in an increased cost to the plan with no measurable improvement in clinical outcomes. Table 2. hgh in the Transitional Patient Approved uses Accepted uses Possible uses medical rationale Multiple pituitary deficiencies and low IGF Child-onset GHD Pituitary tumor Pituitary surgical damage Hypothalamic disease Irradiation Trauma Reconfirmed idiopathic childhood GHD Chronic renal insufficiency AIDS wasting Short-bowel syndrome Continued height deficit at puberty Short for gestational age hgh indicates recombinant human growth hormone; IGF, insulin-like growth factor; GHD, growth hormone deficiency; AIDS, acquired immune deficiency syndrome. Because of emerging medical evidence highlighting the deleterious impact of GHD over a lifetime, payers and providers are beginning to recognize the importance of hgh therapy not only in the well-established pediatric setting but also in the treatment of transitional patients and adults. Transitional patients and adults with GHD are at an increased risk for metabolic disturbances that can increase morbidity and mortality and, in turn, long-term health plan costs. As the evidence is examined, the long-term benefit of this therapy may be the resolution of metabolic complications and an improvement in the quality of life for members with GHD. It is paramount for providers and payers to agree on which transitional and adult patients are appropriate for coverage and treatment. A consensus is starting to emerge among payers and providers on which transitional and adult patients should be tested for GHD, which provocation tests are most VOL. 10, NO. 13, SUP. THE AMERICAN JOURNAL OF MANAGED CARE S439
5 appropriate, and who ultimately should receive hgh therapy. A secondary consideration should be given to the approval process used by health plans. Various treatment approval forms are used by MCOs and specialty pharmacies to document an appropriate diagnosis. Some of these tools could actually limit the presentation of the clinical situation or require unnecessary tests resulting in less-than-optimal MCO decisions. In addition, gaps exist in providers knowledge of health plan reimbursement policies for hgh therapy. To overcome these impediments to quality cost-effective care, an efficient means for clear and consistent communication between payers and providers is clearly needed. Ideally, evidencebased guidelines should drive the use of hgh in transitional patients and adults to achieve the goal of balancing ethical and compassionate care for these patients with the mounting need to restrain payer and member prescription costs. A policy based on FDA-approved and extensively documented uses may appear adequate but, in reality, may deprive therapy for individuals with an atypical presentation of GHD. The process must include a mechanism for a physician specialist to exercise professional judgment because the diagnostic process is not always clearly defined. An open dialogue between medical directors and endocrinologists forms the foundation for a successful policy that meets the needs of the GH population and respects the financial burden this treatment places on the MCO. The resulting policy will clearly define appropriate patients and minimize provider, payer, and member frustration. S440 THE AMERICAN JOURNAL OF MANAGED CARE OCTOBER 2004
CIGNA HealthCare Prior Authorization Form - Growth Hormone Medications -
 Pharmacy Services Phone: (800)244-6224 Fax: (800)390-9745 CIGNA HealthCare Prior Authorization Form - Growth Hormone Medications - Notice: Failure to complete this form in its entirety may result in delayed
Pharmacy Services Phone: (800)244-6224 Fax: (800)390-9745 CIGNA HealthCare Prior Authorization Form - Growth Hormone Medications - Notice: Failure to complete this form in its entirety may result in delayed
2. Has this plan authorized this medication in the past for this member (i.e., previous authorization is on file under this plan)?
 Pharmacy Prior Authorization AETA BETTER HEALTH KETUCK Growth Hormone (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and
Pharmacy Prior Authorization AETA BETTER HEALTH KETUCK Growth Hormone (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and
Humatrope*, Norditropin*, Genotropin, Nutropin, Nutropin AQ, Omnitrope, Saizen
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.11 Subject: Growth Hormone Adult Page: 1 of 6 Last Review Date: December 8, 2017 Growth Hormone Adult
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.11 Subject: Growth Hormone Adult Page: 1 of 6 Last Review Date: December 8, 2017 Growth Hormone Adult
Humatrope*, Norditropin*, Genotropin, Nutropin, Nutropin AQ, Omnitrope, Saizen
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.08.11 Subject: Growth Hormone Adult Page: 1 of 6 Last Review Date: September 15, 2016 Growth Hormone
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.08.11 Subject: Growth Hormone Adult Page: 1 of 6 Last Review Date: September 15, 2016 Growth Hormone
Growth Hormones DRUG.00009
 Market DC Growth Hormones DRUG.00009 Override(s) Prior Authorization Quantity Limit Approval Duration WPM PAB Center: Thirty (30) day exception for recently expired (within the past 45 days) growth hormone
Market DC Growth Hormones DRUG.00009 Override(s) Prior Authorization Quantity Limit Approval Duration WPM PAB Center: Thirty (30) day exception for recently expired (within the past 45 days) growth hormone
Humatrope*, Norditropin*, Genotropin, Nutropin, Nutropin AQ, Omnitrope, Saizen
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.08.11 Subject: Growth Hormone Adult Page: 1 of 6 Last Review Date: December 5, 2014 Growth Hormone Adult
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.08.11 Subject: Growth Hormone Adult Page: 1 of 6 Last Review Date: December 5, 2014 Growth Hormone Adult
General Approval Criteria for ALL Growth Hormone agents: (ALL criteria must be met)
 Growth Hormone Agents Prior Authorization Criteria for Louisiana Fee for Service and MCO Medicaid Recipients Page 1 of 7 Preferred Agents Somatropin Pen (Norditropin ) Somatropin Pen (Nutropin AQ ) Non-Preferred
Growth Hormone Agents Prior Authorization Criteria for Louisiana Fee for Service and MCO Medicaid Recipients Page 1 of 7 Preferred Agents Somatropin Pen (Norditropin ) Somatropin Pen (Nutropin AQ ) Non-Preferred
Humatrope*, Norditropin*, Genotropin, Nutropin, Nutropin AQ, Omnitrope, Saizen, Zomacton
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.11 Subject: Growth Hormone Adult Page: 1 of 6 Last Review Date: September 20, 2018 Growth Hormone
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.11 Subject: Growth Hormone Adult Page: 1 of 6 Last Review Date: September 20, 2018 Growth Hormone
Request for Prior Authorization Growth Hormone (Norditropin
 Request for Prior Authorization Growth Hormone (Norditropin, Nutropin/AQ ) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Growth Hormone require a
Request for Prior Authorization Growth Hormone (Norditropin, Nutropin/AQ ) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Growth Hormone require a
Genotropin, Norditropin, Nutropin, Nutropin AQ, Humatrope, Saizen,
 Blue Cross Blue Shield of Vermont and The Vermont Health Plan Prior Approval Guidelines Human Growth Hormone Somatropin (Genotropin, Norditropin, Nutropin, Nutropin AQ, Humatrope, Serostim, Saizen, Zomacton/TevTropin,
Blue Cross Blue Shield of Vermont and The Vermont Health Plan Prior Approval Guidelines Human Growth Hormone Somatropin (Genotropin, Norditropin, Nutropin, Nutropin AQ, Humatrope, Serostim, Saizen, Zomacton/TevTropin,
First Name. Specialty: Fax. First Name DOB: Duration:
 Prescriber Information Last ame: First ame DEA/PI: Specialty: Phone - - Fax - - Member Information Last ame: First ame Member ID umber DOB: - - Medication Information: Drug ame and Strength: Diagnosis:
Prescriber Information Last ame: First ame DEA/PI: Specialty: Phone - - Fax - - Member Information Last ame: First ame Member ID umber DOB: - - Medication Information: Drug ame and Strength: Diagnosis:
Circle Yes or No Y N. [If yes, skip to question 30.] 2. Is this request for a child? Y N. [If no, skip to question 20.]
![Circle Yes or No Y N. [If yes, skip to question 30.] 2. Is this request for a child? Y N. [If no, skip to question 20.] Circle Yes or No Y N. [If yes, skip to question 30.] 2. Is this request for a child? Y N. [If no, skip to question 20.]](/thumbs/89/99261437.jpg) 05/20/2015 Prior Authorization MERC CARE PLA (MEDICAID) Growth Hormone (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and
05/20/2015 Prior Authorization MERC CARE PLA (MEDICAID) Growth Hormone (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and
Clinical Guideline POSITION STATEMENT ON THE INVESTIGATION AND TREATMENT OF GROWTH HORMONE DEFICIENCY IN TRANSITION
 Clinical Guideline POSITION STATEMENT ON THE INVESTIGATION AND TREATMENT OF GROWTH HORMONE DEFICIENCY IN TRANSITION Date of First Issue 01/04/2015 Approved 28/01/2016 Current Issue Date 28/01/2016 Review
Clinical Guideline POSITION STATEMENT ON THE INVESTIGATION AND TREATMENT OF GROWTH HORMONE DEFICIENCY IN TRANSITION Date of First Issue 01/04/2015 Approved 28/01/2016 Current Issue Date 28/01/2016 Review
GROWTH HORMONE DEFICIENCY AND OTHER INDICATIONS FOR GROWTH HORMONE THERAPY CHILD AND ADOLESCENT
 1. Medical Condition TUEC Guidelines GROWTH HORMONE DEFICIENCY AND OTHER INDICATIONS FOR GROWTH HORMONE THERAPY CHILD AND ADOLESCENT Growth Hormone Deficiency and other indications for growth hormone therapy
1. Medical Condition TUEC Guidelines GROWTH HORMONE DEFICIENCY AND OTHER INDICATIONS FOR GROWTH HORMONE THERAPY CHILD AND ADOLESCENT Growth Hormone Deficiency and other indications for growth hormone therapy
GROWTH HORMONE DEFICIENCY AND OTHER INDICATIONS FOR GROWTH HORMONE THERAPY ADULT
 1. Medical Condition GROWTH HORMONE DEFICIENCY AND OTHER INDICATIONS FOR GROWTH HORMONE THERAPY ADULT Growth Hormone Deficiency and other indications for growth hormone therapy (adult). 2. Diagnosis A.
1. Medical Condition GROWTH HORMONE DEFICIENCY AND OTHER INDICATIONS FOR GROWTH HORMONE THERAPY ADULT Growth Hormone Deficiency and other indications for growth hormone therapy (adult). 2. Diagnosis A.
HUMAN GROWTH HORMONE GENOTROPIN
 Drug Prior Authorization Guideline HUMAN GROWTH HORMONE GENOTROPIN (somatropin) PA9728 Covered Service: Yes when meets criteria below Prior Authorization Required: Yes Additional Information: Medicare
Drug Prior Authorization Guideline HUMAN GROWTH HORMONE GENOTROPIN (somatropin) PA9728 Covered Service: Yes when meets criteria below Prior Authorization Required: Yes Additional Information: Medicare
ATHLETES & PRESCRIBING PHYSICIANS PLEASE READ
 ATHLETES & PRESCRIBING PHYSICIANS PLEASE READ USADA can grant a Therapeutic Use Exemption (TUE) in compliance with the World Anti-Doping Agency International Standard for TUEs. The TUE application process
ATHLETES & PRESCRIBING PHYSICIANS PLEASE READ USADA can grant a Therapeutic Use Exemption (TUE) in compliance with the World Anti-Doping Agency International Standard for TUEs. The TUE application process
2. Is the request for Humatrope? Y N [If no, skip to question 6.]
![2. Is the request for Humatrope? Y N [If no, skip to question 6.] 2. Is the request for Humatrope? Y N [If no, skip to question 6.]](/thumbs/90/101419747.jpg) Pharmacy Prior Authorization AETA BETTER HEALTH FLORIDA Growth Hormone Agents This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date.
Pharmacy Prior Authorization AETA BETTER HEALTH FLORIDA Growth Hormone Agents This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date.
ATHLETES & PRESCRIBING PHYSICIANS PLEASE READ
 ATHLETES & PRESCRIBING PHYSICIANS PLEASE READ USADA can grant a Therapeutic Use Exemption (TUE) in compliance with the World Anti- Doping Agency International Standard for TUEs. The TUE application process
ATHLETES & PRESCRIBING PHYSICIANS PLEASE READ USADA can grant a Therapeutic Use Exemption (TUE) in compliance with the World Anti- Doping Agency International Standard for TUEs. The TUE application process
Aetna Better Health of Virginia
 Genotropin Nutropin Serostim Zomacton Humatrope Omnitrope Zorbtive somatropin Norditropin Saizen General Criteria for Approval: Omnitrope vial formulation is the preferred Growth Hormone product; consideration
Genotropin Nutropin Serostim Zomacton Humatrope Omnitrope Zorbtive somatropin Norditropin Saizen General Criteria for Approval: Omnitrope vial formulation is the preferred Growth Hormone product; consideration
Growth Hormone!gents. WA.PHAR.50 Growth Hormone Agents
 Growth Hormone!gents WA.PHAR.50 Growth Hormone Agents Background: Human growth hormone, also known as somatotropin, is produced in the anterior lobe of the pituitary gland. This hormone plays an important
Growth Hormone!gents WA.PHAR.50 Growth Hormone Agents Background: Human growth hormone, also known as somatotropin, is produced in the anterior lobe of the pituitary gland. This hormone plays an important
HARVARD PILGRIM HEALTH CARE RECOMMENDED MEDICATION REQUEST GUIDELINES
 Generic Brand HICL GCN Exception/Other SOMATROPIN HUMATROPE GENOTROPIN NORDITROPIN NORDITROPIN FLEXPRO NORDITROPIN NORDIFLEX NUTROPIN NUTROPIN AQ OMNITROPE SAIZEN ZOMACTON 02824 BRAND ZORBTIVE BRAND SEROSTIM
Generic Brand HICL GCN Exception/Other SOMATROPIN HUMATROPE GENOTROPIN NORDITROPIN NORDITROPIN FLEXPRO NORDITROPIN NORDIFLEX NUTROPIN NUTROPIN AQ OMNITROPE SAIZEN ZOMACTON 02824 BRAND ZORBTIVE BRAND SEROSTIM
PHARMACY POLICY STATEMENT Indiana Medicaid
 DRUG NAME BILLING CODE BENEFIT TYPE SITE OF SERVICE ALLOWED COVERAGE REQUIREMENTS LIST OF DIAGNOSES CONSIDERED NOT MEDICALLY NECESSARY PHARMACY POLICY STATEMENT Indiana Medicaid Zomacton (somatropin) Must
DRUG NAME BILLING CODE BENEFIT TYPE SITE OF SERVICE ALLOWED COVERAGE REQUIREMENTS LIST OF DIAGNOSES CONSIDERED NOT MEDICALLY NECESSARY PHARMACY POLICY STATEMENT Indiana Medicaid Zomacton (somatropin) Must
PHARMACY POLICY STATEMENT Indiana Medicaid
 DRUG NAME BILLING CODE BENEFIT TYPE SITE OF SERVICE ALLOWED COVERAGE REQUIREMENTS LIST OF DIAGNOSES CONSIDERED NOT MEDICALLY NECESSARY PHARMACY POLICY STATEMENT Indiana Medicaid Norditropin (somatropin)
DRUG NAME BILLING CODE BENEFIT TYPE SITE OF SERVICE ALLOWED COVERAGE REQUIREMENTS LIST OF DIAGNOSES CONSIDERED NOT MEDICALLY NECESSARY PHARMACY POLICY STATEMENT Indiana Medicaid Norditropin (somatropin)
AETNA BETTER HEALTH Non-Formulary Prior Authorization guideline for Growth Hormone and related agents
 Aetna Better Health 2000 Market Street, Suite 850 Philadelphia, PA 19103 AETNA BETTER HEALTH Non-Formulary Prior Authorization guideline for Growth Hormone and related agents Revised April 2014 Growth
Aetna Better Health 2000 Market Street, Suite 850 Philadelphia, PA 19103 AETNA BETTER HEALTH Non-Formulary Prior Authorization guideline for Growth Hormone and related agents Revised April 2014 Growth
Growth Hormone Therapy Guidelines: Clinical and Managed Care Perspectives
 At a Glance Review Article Practical Implications e135 Author Information e144 Full text and PDF Growth Hormone Therapy Guidelines: Clinical and Managed Care Perspectives Susan R. Rose, MD; David M. Cook,
At a Glance Review Article Practical Implications e135 Author Information e144 Full text and PDF Growth Hormone Therapy Guidelines: Clinical and Managed Care Perspectives Susan R. Rose, MD; David M. Cook,
Original Effective Date: 7/5/2007
 Subject: Recombinant Human Growth Hormone (somatropin)_adult Growth Hormone Deficiency GHD HIV/AIDS-associated wasting and cachexia Short Bowel Syndrome (SBS) Policy Number: MCP-004-D Review Dates: 4/28/2010,
Subject: Recombinant Human Growth Hormone (somatropin)_adult Growth Hormone Deficiency GHD HIV/AIDS-associated wasting and cachexia Short Bowel Syndrome (SBS) Policy Number: MCP-004-D Review Dates: 4/28/2010,
Original Effective Date: 7/5/2007
 Subject: Recombinant Human Growth Hormone: PEDIATRIC_GENETIC DISEASES with Primary Effects on Growth Turner syndrome Noonan syndrome Prader-Willi syndrome SHOX mutations DISCLAIMER Original Effective Date:
Subject: Recombinant Human Growth Hormone: PEDIATRIC_GENETIC DISEASES with Primary Effects on Growth Turner syndrome Noonan syndrome Prader-Willi syndrome SHOX mutations DISCLAIMER Original Effective Date:
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 Program Number 2018 P 1039-8 Program UnitedHealthcare Pharmacy Clinical Pharmacy Programs Prior Authorization/Notification Human Growth Hormone, Growth Stimulating Products Medication Human Growth Hormone:
Program Number 2018 P 1039-8 Program UnitedHealthcare Pharmacy Clinical Pharmacy Programs Prior Authorization/Notification Human Growth Hormone, Growth Stimulating Products Medication Human Growth Hormone:
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 2016-8 Program Prior Authorization/Medical Necessity Medication Human Growth Hormone: Somatropin (Genotropin *, Humatrope *, Norditropin
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 2016-8 Program Prior Authorization/Medical Necessity Medication Human Growth Hormone: Somatropin (Genotropin *, Humatrope *, Norditropin
Growth hormone therapy for short stature in adolescents the experience in the University Medical Unit, National Hospital of Sri Lanka
 Growth hormone therapy for short stature in adolescents Growth hormone therapy for short stature in adolescents the experience in the University Medical Unit, National Hospital of Sri Lanka K K K Gamage,
Growth hormone therapy for short stature in adolescents Growth hormone therapy for short stature in adolescents the experience in the University Medical Unit, National Hospital of Sri Lanka K K K Gamage,
Clinical Standards for GH Treatment in Childhood & Adolescence.
 Clinical Standards for GH Treatment in Childhood & Adolescence. The Clinical Standards for GH treatment have been produced by the Clinical Committee of the BSPED. They are evidence based where possible
Clinical Standards for GH Treatment in Childhood & Adolescence. The Clinical Standards for GH treatment have been produced by the Clinical Committee of the BSPED. They are evidence based where possible
Growth Hormone Therapy
 Growth Hormone Therapy Policy Number: Original Effective Date: MM.04.011 05/21/1999 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 05/23/2014 Section: Prescription Drugs Place(s)
Growth Hormone Therapy Policy Number: Original Effective Date: MM.04.011 05/21/1999 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 05/23/2014 Section: Prescription Drugs Place(s)
Prior Authorization Criteria Form This form applies to Paramount Commercial Members Only. Non-Preferred Growth Hormone Products
 Prior Authorization Criteria Form This form applies to Paramount Commercial Members Only Criteria: P0078 Approved: 3/2017 Reviewed: Non-Preferred Growth Hormone Products Complete/review information, sign
Prior Authorization Criteria Form This form applies to Paramount Commercial Members Only Criteria: P0078 Approved: 3/2017 Reviewed: Non-Preferred Growth Hormone Products Complete/review information, sign
Growth promoting treatment: When discretion is the better part of value
 Growth promoting treatment: When discretion is the better part of value David B. Allen, MD Professor of Pediatrics University of Wisconsin School of Medicine and Public Health Head of Division of Diabetes
Growth promoting treatment: When discretion is the better part of value David B. Allen, MD Professor of Pediatrics University of Wisconsin School of Medicine and Public Health Head of Division of Diabetes
Pharmacy Management Drug Policy
 SUBJECT: Policy POLICY NUMBER: Pharmacy-18 EFFECTIVE DATE: 08/03 LAST REVIEW DATE: 9/24/2018 If the member s subscriber contract excludes coverage for a specific service or prescription drug, it is not
SUBJECT: Policy POLICY NUMBER: Pharmacy-18 EFFECTIVE DATE: 08/03 LAST REVIEW DATE: 9/24/2018 If the member s subscriber contract excludes coverage for a specific service or prescription drug, it is not
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Human Growth Hormone Page 1 of 44 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Human Growth Hormone Pre-Determination of Services IS REQUIRED by the Member s
Human Growth Hormone Page 1 of 44 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Human Growth Hormone Pre-Determination of Services IS REQUIRED by the Member s
Pharmacy Prior Authorization Growth Hormone- Clinical Guidelines
 Genotropin, Humatrope, Norditropin, Nutropin, Omnitrope, Saizen, Serostim, somatropin, Zorbtive, Zomacton I. Growth Hormone Deficiency in Children and Adolescents: Note: Provider must submit chart notes
Genotropin, Humatrope, Norditropin, Nutropin, Omnitrope, Saizen, Serostim, somatropin, Zorbtive, Zomacton I. Growth Hormone Deficiency in Children and Adolescents: Note: Provider must submit chart notes
TRANSITIONING FROM A PEDIATRIC TO AN ADULT ENDOCRINOLOGIST CARLOS A. LEYVA JORDÁN, M.D. PEDIATRIC ENDOCRINOLOGIST
 TRANSITIONING FROM A PEDIATRIC TO AN ADULT ENDOCRINOLOGIST CARLOS A. LEYVA JORDÁN, M.D. PEDIATRIC ENDOCRINOLOGIST DISCLOSURE No potential conflict of interest OBJECTIVES Review timing considerations for
TRANSITIONING FROM A PEDIATRIC TO AN ADULT ENDOCRINOLOGIST CARLOS A. LEYVA JORDÁN, M.D. PEDIATRIC ENDOCRINOLOGIST DISCLOSURE No potential conflict of interest OBJECTIVES Review timing considerations for
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Human Growth Hormone Page 1 of 57 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Human Growth Hormone Pre-Determination of Services IS REQUIRED by the Member s
Human Growth Hormone Page 1 of 57 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Human Growth Hormone Pre-Determination of Services IS REQUIRED by the Member s
Growth Hormone: Review of the Evidence
 Drug Use Research & Management Program DHS Division of Medical Assistance Programs, 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Growth Hormone: Review of the Evidence
Drug Use Research & Management Program DHS Division of Medical Assistance Programs, 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Growth Hormone: Review of the Evidence
Clinical and Reimbursement Issues in Growth Hormone Use in Adults
 Clinical and Reimbursement Issues in Growth Hormone Use in Adults Based on presentations by Beverly M.K. Biller, MD; Mary Lee Vance, MD; David L. Kleinberg, MD; David M. Cook, MD; and Terry Gordon, MD,
Clinical and Reimbursement Issues in Growth Hormone Use in Adults Based on presentations by Beverly M.K. Biller, MD; Mary Lee Vance, MD; David L. Kleinberg, MD; David M. Cook, MD; and Terry Gordon, MD,
Policy: Growth Hormones Reference Number: TCHP.PHAR.184 Effective Date: Last Review Date:
 Policy: Growth Hormones Reference Number: TCHP.PHAR.184 Effective Date: 07.01.2018 Last Review Date: 04.13.2018 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy
Policy: Growth Hormones Reference Number: TCHP.PHAR.184 Effective Date: 07.01.2018 Last Review Date: 04.13.2018 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy
10.2 Summary of the Votes and Considerations for Policy
 CEPAC Voting and Policy Implications Summary Supplemental Screening for Women with Dense Breast Tissue December 13, 2013 The last CEPAC meeting addressed the comparative clinical effectiveness and value
CEPAC Voting and Policy Implications Summary Supplemental Screening for Women with Dense Breast Tissue December 13, 2013 The last CEPAC meeting addressed the comparative clinical effectiveness and value
Endocrine part two. Presented by Dr. Mohammad Saadeh The requirements for the Clinical Chemistry Philadelphia University Faculty of pharmacy
 Endocrine part two Presented by Dr. Mohammad Saadeh The requirements for the Clinical Chemistry Philadelphia University Faculty of pharmacy Cushing's disease: increased secretion of adrenocorticotropic
Endocrine part two Presented by Dr. Mohammad Saadeh The requirements for the Clinical Chemistry Philadelphia University Faculty of pharmacy Cushing's disease: increased secretion of adrenocorticotropic
Medical and Rehabilitation Innovations Neuroendocrine Screening and Hormone Replacement Therapy in Trauma Related Acquired Brain Injury
 Medical and Rehabilitation Innovations Neuroendocrine Screening and Hormone Replacement Therapy in Trauma Related Acquired Brain Injury BACKGROUND Trauma related acquired brain injury (ABI) is known to
Medical and Rehabilitation Innovations Neuroendocrine Screening and Hormone Replacement Therapy in Trauma Related Acquired Brain Injury BACKGROUND Trauma related acquired brain injury (ABI) is known to
This Coverage Policy applies to Individual Health Insurance Marketplace benefit plans only.
 This Coverage Policy applies to Individual Health Insurance Marketplace benefit plans only. GROWTH HORMONE THERAPY POLICY Omnitrope is the EXCLUSIVE growth hormone covered if patient meets criteria. COVERAGE
This Coverage Policy applies to Individual Health Insurance Marketplace benefit plans only. GROWTH HORMONE THERAPY POLICY Omnitrope is the EXCLUSIVE growth hormone covered if patient meets criteria. COVERAGE
UW MEDICINE PATIENT EDUCATION. Acromegaly Symptoms and treatments. What is acromegaly? DRAFT. What are the symptoms? How is it diagnosed?
 UW MEDICINE PATIENT EDUCATION Acromegaly Symptoms and treatments This handout explains a health condition called acromegaly. It describes tests that are used to diagnose the condition and gives basic instructions
UW MEDICINE PATIENT EDUCATION Acromegaly Symptoms and treatments This handout explains a health condition called acromegaly. It describes tests that are used to diagnose the condition and gives basic instructions
AETNA BETTER HEALTH Prior Authorization guideline for Growth Hormone Agents
 AETNA BETTER HEALTH Prior Authorization guideline for Growth Hormone Agents Growth Hormone and related agents Formulary: Omnitrope vials Non-Formulary: Genotropin, Humatrope, Saizen, Serostim, Tev-Tropin,
AETNA BETTER HEALTH Prior Authorization guideline for Growth Hormone Agents Growth Hormone and related agents Formulary: Omnitrope vials Non-Formulary: Genotropin, Humatrope, Saizen, Serostim, Tev-Tropin,
Clinical Policy: Somatropin (Recombinant Human Growth Hormone) Reference Number: CP.PHAR.55 Effective Date: 03/11 Last Review Date: 06/17
 Clinical Policy: (Recombinant Human Growth Hormone) Reference Number: CP.PHAR.55 Effective Date: 03/11 Last Review Date: 06/17 Line of Business: Medicaid Revision Log See Important Reminder at the end
Clinical Policy: (Recombinant Human Growth Hormone) Reference Number: CP.PHAR.55 Effective Date: 03/11 Last Review Date: 06/17 Line of Business: Medicaid Revision Log See Important Reminder at the end
Growth and DMD Endocrine aspects of care
 Growth and DMD Endocrine aspects of care Meilan Rutter, MB,BCh, FRACP Division of Endocrinology Cincinnati Children s Hospital Medical Center July 2007 Where are we now? Inactive Reactive Proactive CCHMC
Growth and DMD Endocrine aspects of care Meilan Rutter, MB,BCh, FRACP Division of Endocrinology Cincinnati Children s Hospital Medical Center July 2007 Where are we now? Inactive Reactive Proactive CCHMC
Hypothalamus & Pituitary Gland
 Hypothalamus & Pituitary Gland Hypothalamus and Pituitary Gland The hypothalamus and pituitary gland form a unit that exerts control over the function of several endocrine glands (thyroid, adrenals, and
Hypothalamus & Pituitary Gland Hypothalamus and Pituitary Gland The hypothalamus and pituitary gland form a unit that exerts control over the function of several endocrine glands (thyroid, adrenals, and
GROWTH HORMONE THERAPY
 GROWTH HORMONE THERAPY Line(s) of Business: HMO; PPO; QUEST Integration Original Effective Date: 05/21/1999 Current Effective Date: 10/01/2015 POLICY A. INDICATIONS The indications below including FDA-approved
GROWTH HORMONE THERAPY Line(s) of Business: HMO; PPO; QUEST Integration Original Effective Date: 05/21/1999 Current Effective Date: 10/01/2015 POLICY A. INDICATIONS The indications below including FDA-approved
TEXAS MEDICAID Clinical Edit Prior Authorization Growth Hormones: GENOTROPIN & NORDITROPIN Texas Children s Health Plan Only
 TEXAS MEDICAID Clinical Edit Prior Authorization Growth Hormones: GENOTROPIN & NORDITROPIN Texas Children s Health Plan Only STEP 1: CLEARLY PRINT AND COMPLETE TO EXPEDITE PROCESSING Date: Prescriber First
TEXAS MEDICAID Clinical Edit Prior Authorization Growth Hormones: GENOTROPIN & NORDITROPIN Texas Children s Health Plan Only STEP 1: CLEARLY PRINT AND COMPLETE TO EXPEDITE PROCESSING Date: Prescriber First
PENS 2017 Minneapolis, MN April 27, Disclosure. Objectives: Growth Hormone Guidelines Roundtable
 Growth Hormone Guidelines Roundtable PENS 2017 Minneapolis, MN April 27, 2017 Panelists: Mary S. Burr, DNP, CPNP-PC Catherine P. Metzinger, AAS, RN, CDE Bradley S. Miller, MD, PhD Disclosure Dr. Miller
Growth Hormone Guidelines Roundtable PENS 2017 Minneapolis, MN April 27, 2017 Panelists: Mary S. Burr, DNP, CPNP-PC Catherine P. Metzinger, AAS, RN, CDE Bradley S. Miller, MD, PhD Disclosure Dr. Miller
PITUITARY: JUST THE BASICS PART 2 THE PATIENT
 PITUITARY: JUST THE BASICS PART 2 THE PATIENT DISCLOSURE Relevant relationships with commercial entities none Potential for conflicts of interest within this presentation none Steps taken to review and
PITUITARY: JUST THE BASICS PART 2 THE PATIENT DISCLOSURE Relevant relationships with commercial entities none Potential for conflicts of interest within this presentation none Steps taken to review and
Humatrope*, Norditropin*, Genotropin, Nutropin, Nutropin AQ, Omnitrope, Saizen, Zomacton (aka. Tev-Tropin)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.12 Subject: Growth Hormone Pediatric Page: 1 of 6 Last Review Date: September 15, 2016 Growth Hormone
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.12 Subject: Growth Hormone Pediatric Page: 1 of 6 Last Review Date: September 15, 2016 Growth Hormone
Diagnosing Growth Disorders. PE Clayton School of Medical Sciences, Faculty of Biology, Medicine & Health
 Diagnosing Growth Disorders PE Clayton School of Medical Sciences, Faculty of Biology, Medicine & Health Content Normal pattern of growth and its variation Using growth charts Interpreting auxological
Diagnosing Growth Disorders PE Clayton School of Medical Sciences, Faculty of Biology, Medicine & Health Content Normal pattern of growth and its variation Using growth charts Interpreting auxological
GROWTH HORMONE THERAPY
 GROWTH HORMONE THERAPY Line(s) of Business: HMO; PPO; QUEST Integration Original Effective Date: 05/21/1999 Current Effective Date: 03/01/201804/01/2019 POLICY A. INDICATIONS The indications below including
GROWTH HORMONE THERAPY Line(s) of Business: HMO; PPO; QUEST Integration Original Effective Date: 05/21/1999 Current Effective Date: 03/01/201804/01/2019 POLICY A. INDICATIONS The indications below including
GROWTH HORMONE THERAPY
 GROWTH HORMONE THERAPY Line(s) of Business: HMO; PPO; QUEST Integration Original Effective Date: 05/21/1999 Current Effective Date: 12/30/201601/01/2018TBD03/01/2018 POLICY A. INDICATIONS The indications
GROWTH HORMONE THERAPY Line(s) of Business: HMO; PPO; QUEST Integration Original Effective Date: 05/21/1999 Current Effective Date: 12/30/201601/01/2018TBD03/01/2018 POLICY A. INDICATIONS The indications
TEXAS MEDICAID Clinical Edit Prior Authorization Growth Hormones: HUMATROPE, NUTROPIN AQ, OMNITROPE, SAIZEN
 TEXAS MEDICAID Clinical Edit Prior Authorization Growth Hormones: HUMATROPE, NUTROPIN AQ, OMNITROPE, SAIZEN STEP 1: CLEARLY PRINT AND COMPLETE TO EXPEDITE PROCESSING Date: Prescriber First & Last Name:
TEXAS MEDICAID Clinical Edit Prior Authorization Growth Hormones: HUMATROPE, NUTROPIN AQ, OMNITROPE, SAIZEN STEP 1: CLEARLY PRINT AND COMPLETE TO EXPEDITE PROCESSING Date: Prescriber First & Last Name:
Craniopharyngioma. Michael Gottschalk, MD,PhD University of California San Diego Rady Children s Hospital
 Craniopharyngioma Michael Gottschalk, MD,PhD University of California San Diego Rady Children s Hospital Objectives Incidence Clinical Presentation Treatment Options Perioperative concerns Long-term endocrine
Craniopharyngioma Michael Gottschalk, MD,PhD University of California San Diego Rady Children s Hospital Objectives Incidence Clinical Presentation Treatment Options Perioperative concerns Long-term endocrine
Endocrine Pharmacology
 Endocrine Pharmacology 17-2-2013 DRUGS AFFECTING THE ENDOCRINE SYSTEM The endocrine system is the system of glands, each of which secretes a type of hormone directly into the bloodstream to regulate the
Endocrine Pharmacology 17-2-2013 DRUGS AFFECTING THE ENDOCRINE SYSTEM The endocrine system is the system of glands, each of which secretes a type of hormone directly into the bloodstream to regulate the
Endocrine system overview
 Endocrine system overview Nature of the hormonal system -Major integrator of body function Classification of hormones Endocrine vs paracrine Nature of hormone-receptor systems Role of the hypothalamuspituitary
Endocrine system overview Nature of the hormonal system -Major integrator of body function Classification of hormones Endocrine vs paracrine Nature of hormone-receptor systems Role of the hypothalamuspituitary
PedsCases Podcast Scripts
 PedsCases Podcast Scripts This is a text version of a podcast from Pedscases.com on the Approach to Pediatric Anemia and Pallor. These podcasts are designed to give medical students an overview of key
PedsCases Podcast Scripts This is a text version of a podcast from Pedscases.com on the Approach to Pediatric Anemia and Pallor. These podcasts are designed to give medical students an overview of key
Hypothalamic Pituitary and Growth Disorders in Survivors of Childhood Cancer: An Endocrine Society* Clinical Practice Guideline
 CLINICAL PRACTICE GUIDELINE Hypothalamic Pituitary and Growth Disorders in Survivors of Childhood Cancer: An Endocrine Society* Clinical Practice Guideline Charles A. Sklar, 1 Zoltan Antal, 1,2 Wassim
CLINICAL PRACTICE GUIDELINE Hypothalamic Pituitary and Growth Disorders in Survivors of Childhood Cancer: An Endocrine Society* Clinical Practice Guideline Charles A. Sklar, 1 Zoltan Antal, 1,2 Wassim
Growth hormone therapy in a girl with Turner syndrome showing a large increase over the initially predicted ht of 4 5
 Disorders of Growth and Puberty: How to Recognize the Normal Variants vs Patients Who Need to be Evaluated Paul Kaplowitz, M.D Pediatric Endocrinology. VCU School of Medicine Interpretation of Growth Charts
Disorders of Growth and Puberty: How to Recognize the Normal Variants vs Patients Who Need to be Evaluated Paul Kaplowitz, M.D Pediatric Endocrinology. VCU School of Medicine Interpretation of Growth Charts
Prior Authorization Criteria Update: Androgens, Topical and Parenteral
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
MEDICAL POLICY I. POLICY HUMAN GROWTH HORMONE MP POLICY TITLE POLICY NUMBER
 g Original Issue Date (Created): July 1, 2002 Most Recent Review Date (Revised): March 25, 2014 Effective Date: June 1, 2014 I. POLICY Recombinant human growth hormone (GH) therapy may be considered medically
g Original Issue Date (Created): July 1, 2002 Most Recent Review Date (Revised): March 25, 2014 Effective Date: June 1, 2014 I. POLICY Recombinant human growth hormone (GH) therapy may be considered medically
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject: Somatropin Table of Contents Coverage Policy...1 General Background...9 Coding/Billing Information... 12 References... 12 Effective Date.. 01/15/2018 Next
Cigna Drug and Biologic Coverage Policy Subject: Somatropin Table of Contents Coverage Policy...1 General Background...9 Coding/Billing Information... 12 References... 12 Effective Date.. 01/15/2018 Next
An Overview of Growth Hormone Deficiency in Adults
 An Overview of Growth Hormone Deficiency in Adults Table of contents page What this booklet is about........................3 What is adult growth hormone deficiency (GHD)?......3 What is the pituitary
An Overview of Growth Hormone Deficiency in Adults Table of contents page What this booklet is about........................3 What is adult growth hormone deficiency (GHD)?......3 What is the pituitary
Chapter 13 worksheet
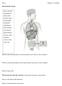 Name: Chapter 13 worksheet The Endocrine System Please label the: hypothalamus pineal gland pituitary gland thyroid gland parathyroid gland thymus heart stomach liver adrenal glands kidneys pancreas small
Name: Chapter 13 worksheet The Endocrine System Please label the: hypothalamus pineal gland pituitary gland thyroid gland parathyroid gland thymus heart stomach liver adrenal glands kidneys pancreas small
GUIDELINES ON. Introduction. G.R. Dohle, S. Arver, C. Bettocchi, S. Kliesch, M. Punab, W. de Ronde
 GUIDELINES ON Male Hypogonadism G.R. Dohle, S. Arver,. Bettocchi, S. Kliesch, M. Punab, W. de Ronde Introduction Male hypogonadism is a clinical syndrome caused by androgen deficiency. It may adversely
GUIDELINES ON Male Hypogonadism G.R. Dohle, S. Arver,. Bettocchi, S. Kliesch, M. Punab, W. de Ronde Introduction Male hypogonadism is a clinical syndrome caused by androgen deficiency. It may adversely
National Screening Committee. Child Health Sub-Group Report Growth Disorders
 National Screening Committee Child Health Sub-Group Report Growth Disorders September 2004 Growth Disorders The condition Growth disorders. There are many conditions that cause abnormally slow or fast
National Screening Committee Child Health Sub-Group Report Growth Disorders September 2004 Growth Disorders The condition Growth disorders. There are many conditions that cause abnormally slow or fast
What we will cover. Evaluation of the Child with Suspected Pituitary Disease. ituitary
 Evaluation of the Child with Suspected Pituitary Disease Craig Alter, MD University of Pennsylvania Children s Hospital of Philadelphia What we will cover * What laboratory tests to order * MRI: common
Evaluation of the Child with Suspected Pituitary Disease Craig Alter, MD University of Pennsylvania Children s Hospital of Philadelphia What we will cover * What laboratory tests to order * MRI: common
David M. Cook, MD, FACE; Kevin C.J. Yuen, MD; Beverly M.K. Biller, MD; Stephen F. Kemp, MD, PhD, FACE; Mary Lee Vance, MD
 AACE Guidelines AMERICAN ASSOCIATION OF CLINICAL ENDOCRINOLOGISTS MEDICAL GUIDELINES FOR CLINICAL PRACTICE FOR GROWTH HORMONE USE IN GROWTH HORMONE-DEFICIENT ADULTS AND TRANSITION PATIENTS 2009 UPDATE
AACE Guidelines AMERICAN ASSOCIATION OF CLINICAL ENDOCRINOLOGISTS MEDICAL GUIDELINES FOR CLINICAL PRACTICE FOR GROWTH HORMONE USE IN GROWTH HORMONE-DEFICIENT ADULTS AND TRANSITION PATIENTS 2009 UPDATE
Growth Hormone Deficiency and Related Diagnoses
 Care1st Health Plan Arizona, Inc. Easy Choice Health Plan Harmony Health Plan of Illinois Missouri Care Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona OneCare (Care1st Health
Care1st Health Plan Arizona, Inc. Easy Choice Health Plan Harmony Health Plan of Illinois Missouri Care Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona OneCare (Care1st Health
Pituitary for the General Practitioner. Marilyn Lee Consultant physician and endocrinologist
 Pituitary for the General Practitioner Marilyn Lee Consultant physician and endocrinologist Pituitary tumours Anterior/posterior pituitary Extension of adenoma upwards/downwards/sideways Producing too
Pituitary for the General Practitioner Marilyn Lee Consultant physician and endocrinologist Pituitary tumours Anterior/posterior pituitary Extension of adenoma upwards/downwards/sideways Producing too
9.2 Hormonal Regulation of Growth
 9.2 Hormonal Regulation of Growth Hormonal Regulation of Growth Pituitary gland regulates growth and development Thyroid gland regulates metabolic rate (exception: some hormones for growth and development)
9.2 Hormonal Regulation of Growth Hormonal Regulation of Growth Pituitary gland regulates growth and development Thyroid gland regulates metabolic rate (exception: some hormones for growth and development)
Hypothalamic & Pituitary Hormones
 1 Hypothalamic & Pituitary Hormones Pharmacologic Applications: Drugs that mimic or block the effects of hypothalamic or pituitary hormones have the following applications: 1. Replacement therapy for hormone
1 Hypothalamic & Pituitary Hormones Pharmacologic Applications: Drugs that mimic or block the effects of hypothalamic or pituitary hormones have the following applications: 1. Replacement therapy for hormone
The University of Arizona Pediatric Residency Program. Primary Goals for Rotation. Endocrinology
 The University of Arizona Pediatric Residency Program Primary Goals for Rotation Endocrinology 1. GOAL: Understand the role of the pediatrician in preventing endocrine dysfunction, and in counseling and
The University of Arizona Pediatric Residency Program Primary Goals for Rotation Endocrinology 1. GOAL: Understand the role of the pediatrician in preventing endocrine dysfunction, and in counseling and
Corporate Medical Policy Testosterone Pellet Implantation for Androgen Deficiency
 Corporate Medical Policy Testosterone Pellet Implantation for Androgen Deficiency File Name: Origination: Last CAP Review: Next CAP Review: Last Review: testosterone_pellet_implantation_for_androgen_deficiency
Corporate Medical Policy Testosterone Pellet Implantation for Androgen Deficiency File Name: Origination: Last CAP Review: Next CAP Review: Last Review: testosterone_pellet_implantation_for_androgen_deficiency
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Mecasermin (Increlex) Reference Number: CP.PHAR.150 Effective Date: 10.01.18 Last Review Date: 07.13.18 Line of Business: Oregon Health Plan Coding Implications Revision Log See Important
Clinical Policy: Mecasermin (Increlex) Reference Number: CP.PHAR.150 Effective Date: 10.01.18 Last Review Date: 07.13.18 Line of Business: Oregon Health Plan Coding Implications Revision Log See Important
Month/Year of Review: September 2013 Date of Last Review: September 2012
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Antidiuretic Hormone
 1 Antidiuretic Hormone 2 Physiology of the Posterior Pituitary The posterior pituitary gland secretes two hormones which are: oxytocin, increase uterine contractions during parturition Contraction of mammary
1 Antidiuretic Hormone 2 Physiology of the Posterior Pituitary The posterior pituitary gland secretes two hormones which are: oxytocin, increase uterine contractions during parturition Contraction of mammary
Provide preventive counseling to parents and patients with specific endocrine conditions about:
 Endocrinology Description: The resident will become familiar with the diagnosis, management, and treatment of endocrine problems. The resident will evaluate patients with a multitude of endocrine problems,
Endocrinology Description: The resident will become familiar with the diagnosis, management, and treatment of endocrine problems. The resident will evaluate patients with a multitude of endocrine problems,
Pathology of pituitary gland. By: Shifaa Qa qa
 Pathology of pituitary gland By: Shifaa Qa qa Sella turcica Adenohypophysis (80%): - epithelial cells - acidophil, basophil, chromophobe - Somatotrophs, Mammosomatotrophs, Corticotrophs, Thyrotrophs, Gonadotrophs
Pathology of pituitary gland By: Shifaa Qa qa Sella turcica Adenohypophysis (80%): - epithelial cells - acidophil, basophil, chromophobe - Somatotrophs, Mammosomatotrophs, Corticotrophs, Thyrotrophs, Gonadotrophs
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Mecasermin Table of Contents Coverage Policy... 1 General Background... 3 Coding/Billing Information... 5 References... 5 Effective Date... 5/15/2017 Next
Cigna Drug and Biologic Coverage Policy Subject Mecasermin Table of Contents Coverage Policy... 1 General Background... 3 Coding/Billing Information... 5 References... 5 Effective Date... 5/15/2017 Next
Effective September 30, 2011 Please refer to the Pharmacy policy for the new coverage criteria
 Effective September 30, 2011 Please refer to the Pharmacy policy for the new coverage criteria Name of Policy: Growth Hormone and Insulin Like Growth Factor-1 (IGF-1) Analogues Policy #: 067 Latest Review
Effective September 30, 2011 Please refer to the Pharmacy policy for the new coverage criteria Name of Policy: Growth Hormone and Insulin Like Growth Factor-1 (IGF-1) Analogues Policy #: 067 Latest Review
Children s Hospital & Research Center Oakland. Endocrinology
 Children s Hospital & Research Center Oakland Endocrinology 5.22 GOAL: Prevention, Counseling and Screening (Endocrine). Understand the role of the pediatrician in preventing endocrine PC, MK, dysfunction,
Children s Hospital & Research Center Oakland Endocrinology 5.22 GOAL: Prevention, Counseling and Screening (Endocrine). Understand the role of the pediatrician in preventing endocrine PC, MK, dysfunction,
DISCLAIMER SUMMARY OF EVIDENCE/POSITION
 Subject: Recombinant Human Growth Hormone Original Effective Date: 7/5/2007 (somatropin)_ PEDIATRIC Growth Hormone Deficiency Growth Failure in Children and Adolescents with Classic GHD [under 18 years]
Subject: Recombinant Human Growth Hormone Original Effective Date: 7/5/2007 (somatropin)_ PEDIATRIC Growth Hormone Deficiency Growth Failure in Children and Adolescents with Classic GHD [under 18 years]
CATEGORY Endocrine System Review. Provide labels for the following diagram CHAPTER 13 BLM
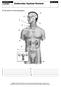 CHAPTER 13 BLM 13.1.1 CATEGORY Endocrine System Review Provide labels for the following diagram. 1. 6. 2. 7. 3. 8. 4. 9. 5. 10. CHAPTER 13 BLM 13.1.2 OVERHEAD Glands and Their Secretions Endocrine gland
CHAPTER 13 BLM 13.1.1 CATEGORY Endocrine System Review Provide labels for the following diagram. 1. 6. 2. 7. 3. 8. 4. 9. 5. 10. CHAPTER 13 BLM 13.1.2 OVERHEAD Glands and Their Secretions Endocrine gland
Pfizer Independent Grants for Learning & Change Track 2- Annual Meetings: Submission Deadlines, Areas of Interest & Educational Goals
 In order to submit a request for general meeting support through Track 2- Annual Meetings the answer to the following questions must be Yes : Does the activity align with Pfizer s areas of interest? (listed
In order to submit a request for general meeting support through Track 2- Annual Meetings the answer to the following questions must be Yes : Does the activity align with Pfizer s areas of interest? (listed
PHARMACY COVERAGE GUIDELINES ORIGINAL EFFECTIVE DATE: 4/1/19 SECTION: DRUGS LAST REVIEW DATE: 2/21/19 LAST CRITERIA REVISION DATE: ARCHIVE DATE:
 GROWTH HORMONE THERAPY: Genotropin (somatropin) subcutaneous injection Humatrope (somatropin) subcutaneous injection Norditropin (somatropin) subcutaneous injection Nutropin AQ (somatropin) subcutaneous
GROWTH HORMONE THERAPY: Genotropin (somatropin) subcutaneous injection Humatrope (somatropin) subcutaneous injection Norditropin (somatropin) subcutaneous injection Nutropin AQ (somatropin) subcutaneous
Growth IGF Analyte Information
 Growth IGF-1 Analyte Information - 1 - IGF-1 Introduction Insulin-like growth factor 1 (IGF-1, IGF-I) is a single chain polypeptide containing 70 amino acids and three disulfide bridges. It is structurally
Growth IGF-1 Analyte Information - 1 - IGF-1 Introduction Insulin-like growth factor 1 (IGF-1, IGF-I) is a single chain polypeptide containing 70 amino acids and three disulfide bridges. It is structurally
Growth and Puberty: A clinical approach. Dr Esko Wiltshire
 Growth and Puberty: A clinical approach Dr Esko Wiltshire NOTHING TO DISCLOSE Why is this character short? Food Psychosocial factors Major Systems (+drugs) Genetic potential Perinatal Classical Hormones
Growth and Puberty: A clinical approach Dr Esko Wiltshire NOTHING TO DISCLOSE Why is this character short? Food Psychosocial factors Major Systems (+drugs) Genetic potential Perinatal Classical Hormones
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: somatostatin_analogs 7/2016 7/2017 7/2018 7/2017 Description of Procedure or Service Somatostatin, a hypothalamic
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: somatostatin_analogs 7/2016 7/2017 7/2018 7/2017 Description of Procedure or Service Somatostatin, a hypothalamic
Pfizer Independent Grants for Learning & Change Track 2- Annual Meetings: Submission Deadlines, Areas of Interest & Educational Goals
 In order to submit a request for general meeting support through Track 2- Annual Meetings the answer to the following questions must be Yes : Does the activity align with Pfizer s areas of interest? (listed
In order to submit a request for general meeting support through Track 2- Annual Meetings the answer to the following questions must be Yes : Does the activity align with Pfizer s areas of interest? (listed
