MATRIX VHD FORM. State the name of the patient ( Product Recipient ) for whom you are providing the information contained in this form.
|
|
|
- Mitchell Stewart
- 5 years ago
- Views:
Transcription
1 MATRIX VHD FORM A. Patient Information State the name of the patient ( Product Recipient ) for whom you are providing the information contained in this form. (First Name) (Middle Initial) (Last Name) (Date of Birth) (Date of Diagnosis with FDA Positive Condition as defined in Section D) B. Physician Information State your name, office address, telephone number, address, if any and medical specialty. (First Name) (Middle Initial) (Last Name) (Office Address) (City) Prov/Terr (Postal Code) (Area Code and Telephone Number) (Medical Specialty) 1
2 C. Echocardiogram Information List the date(s) when the Echocardiogram(s) upon which the Product Recipient s claim is based was/were performed and the Product Recipient s height and weight at the time if available. Date: Ht: Wt: (MM/DD/YYYY) Date: Ht: Wt: (MM/DD/YYYY) Date: Ht: Wt: (MM/DD/YYYY) NOTE: If the Product Recipient has had more than three Echocardiograms upon which the claim is based, please provide the information on a separate sheet of paper and attach it to this Form. D. Definitions In answering the questions on this form, please apply the following definitions: Arrhythmias or Chronic Atrial Fibrillation shall mean chronic atrial fibrillation/flutter that cannot be converted to normal sinus rhythm, or atrial fibrillation/flutter requiring ongoing medical therapy and the Product Recipient was under the age of 70 years at the date of diagnosis of the arrhythmia and the arrhythmia was not caused by hyperthyroidism, previously diagnosed coronary disease, alcohol abuse or systemic hypertension (3 readings of greater than 160/95 within 2 years prior to the diagnosis of the arrhythmia). BSA Indexing shall mean the calculation derived from dividing the patient s chamber dimension by the patient s body surface area using the Dubois method. FDA Positive Regurgitation 1 shall mean mitral regurgitation greater than 20% RJA/LAA or aortic regurgitation greater than 10% JH/LVOTH. Hemodynamically Significant MR shall mean mitral regurgitation >30% RJA/LAA, unless the echo recording is not available, in which case a written description of moderate or greater mitral regurgitation in the echocardiogram report will be sufficient. Hemodynamically Significant AR shall mean aortic regurgitation >45 JH/LVOTH, unless the echo recording is not available, in which case a written description of moderate or greater aortic regurgitation in the echocardiogram report will be sufficient. 2
3 Pulmonary Hypertension shall mean a mean systolic pulmonary artery pressure >25 mm Hg at rest or >30 mm Hg with exercise as measured by cardiac catheterization or with a peak systolic pulmonary artery pressure >45 mm Hg at rest as measured by Doppler Echocardiography, assuming a right atrial pressure of 5 mm Hg where the inferior vena cava is less than 2.0 cm and collapse is greater than 50%; assuming 10 mm Hg where the inferior vena cava is greater than 2.0 cm and collapse is less than or equal to 50%; and assuming 20 mm Hg where the inferior vena cava is greater than or equal to 2.0 cm and does not collapse; and where the inferior vena cava measurement is unavailable, a right atrial pressure of 5 mm Hg will be assumed. Products shall mean shall mean Ponderal, Ponderal Pacaps and/or Redux, or any one of them. Product Recipient shall mean the patient who ingested the Products. Severe renal failure shall mean as having creatinine clearance of less than 30 cc/min for greater than six months. Significant damage to the heart muscle shall mean either left ventricular ejection fraction <30% with aortic regurgitation or a left ventricular ejection fraction <35% with mitral regurgitation in Product Recipients who have not had surgery; or left ventricular ejection fraction <40% persisting for at least six months after valvular repair or replacement surgery in Product Recipients who have had such surgery. VHD shall mean valvular heart disease. E. Matrix VHD Claim Each of the following questions identifies a condition which may serve as the basis for a claim. To the best of your knowledge, does the Product Recipient have any of the following conditions? (Check each that applies): I. Matrix Level I Regurgitation with Complications 1. Hemodynamically Significant MR with evidence of left ventricular enlargement as documented by abnormal left ventricular end-diastolic dimension of >63 mm or >34 mm/m 2 by BSA Indexing and abnormal left atrial antero-posterior systolic dimension >40 mm or >22 mm/m 2 by BSA Indexing? 2. Hemodynamically Significant AR with evidence of left ventricular enlargement as documented by abnormal left ventricular end-diastolic dimension of >65mm or >34.5 mm/m 2 by BSA Indexing, or abnormal left ventricular end-systolic dimension of >45 mm or >24 mm/m 2 by BSA Indexing? 3
4 3. FDA Positive Regurgitation 2 with bacterial endocarditis in the valve upon which the claim is based? II. Matrix Level II Regurgitation with More Serious Complications 1. Hemodynamically Significant MR with evidence of left ventricular enlargement as documented by abnormal left ventricular end-diastolic dimension of >63 mm or >34 mm/m 2 by BSA Indexing and abnormal left atrial antero-posterior systolic dimension >40 mm or >22 mm/m 2 by BSA Indexing and one of the following complications: Arrhythmias? Pulmonary Hypertension secondary to regurgitation? Left Ventricular Ejection Fraction of <60% by echo? (d) Greater left ventricular enlargement defined as left ventricular end-diastolic dimension >67 mm or >36 mm/m 2 by BSA Indexing, or end-systolic dimension 45 mm or 24.5 mm/m 2 by BSA Indexing? 2. Mitral Regurgitation (defined as >30% RJA/LAA unless the echo recording is not available in which case a written description of greater than moderate mitral regurgitation in the echocardiogram report will be sufficient) with left ventricular enddiastolic dimension of >60 mm or >32.5 mm/m 2 by BSA Indexing and one of the following complications: A pulmonary artery systolic pressure of greater than 55 mm Hg and an abnormal left atrial antero-posterior systolic dimension >40 mm or >22 mm/m 2 by BSA Indexing? 4
5 Arrhythmias and an abnormal left atrial antero-posterior systolic dimension >48 mm or >26 mm/m 2 by BSA Indexing to be added to Matrix Level I? Left Ventricular Ejection Fraction of <50% by echocardiogram? 3. Hemodynamically significant AR with evidence of left ventricular enlargement as documented by abnormal left ventricular end-diastolic dimension of >65mm or >34.5 mm/m 2 by BSA Indexing, or evidence of abnormal left ventricular end-systolic dimension of >45 mm or >24 mm/m 2 by BSA Indexing and one of the following complications: Left Ventricular Ejection Fraction of <50% by echocardiogram? Pulmonary Hypertension secondary to regurgitation? Greater left ventricular enlargement defined as left ventricular end-diastolic dimension >70 mm or >37mm/m 2 by BSA Indexing, or end-systolic dimension >50 mm or >26.5 mm/m 2 by BSA Indexing? III. Matrix Level III Surgery or Surgical Candidate 1. Surgery to repair or replace the aortic and/or mitral valve(s) following the use of the Products? 2. Qualification for benefits at Matrix Level I or II but no surgery, and the Product Recipient has ACC/AHA Class I or greater indications for surgery to repair or replace the aortic 3 and/or mitral 4 valve(s) and a statement from the attending Certified Cardiac Surgeon or Certified Cardiologist regarding why the surgery is not being performed? 5
6 3. The Product Recipient has not had surgery and would not qualify for benefits at Matrix Level I or II but has greater than moderate valvular regurgitation and demonstrates, where a record is available, that his or her left ventricular end-diastolic pressure is greater than 15 mm Hg following use of the Products and the Product Recipient has ACC/AHA Class I or greater indications for surgery to repair or replace the aortic 5 and/or mitral 6 valve(s) and a statement from the attending Certified Cardiac Surgeon or Certified Cardiologist regarding why the surgery is not being performed? 4. Qualification at Matrix Level I or II, and a stroke which results in a permanent condition which meets the criteria of the AHA Stroke Outcome Classification System, 7 Functional Level II, determined at least six months after the event, due to: Bacterial endocarditis in the valve upon which the claim is based? chronic atrial fibrillation with left atrial enlargement caused by VHD and not due to an unrelated heart condition? IV. Matrix Level IV Severe Complications of Regurgitation and/or Surgery 1. Qualification at Matrix Levels I, II or III, and a stroke which meets the criteria of the AHA Stroke Outcome Classification System, 8 Functional Level III, determined at least six months after the event, due to: bacterial endocarditis in the valve upon which the claim is based? chronic atrial fibrillation with left atrial enlargement? 2. Qualification at Matrix Levels I, II or III, and a peripheral embolus due to bacterial endocarditis in the valve upon which the claim is based, or as a consequence of atrial fibrillation with left atrial enlargement, which resulted in severe permanent impairment to the kidneys, abdominal organs, or extremities, where severe permanent impairment means: 6
7 for the kidneys, severe renal failure? for the abdominal organs, impairment requiring intra-abdominal surgery? for the extremities, impairment requiring amputation of a major limb? 3. Qualification for payment at Matrix Level III, and New York Heart Association Functional Class I or Class II symptoms as documented by the attending Certified Cardiac Surgeon or Certified Cardiologist, and significant damage to the heart muscle? 4. Surgery to repair or replace the aortic and/or mitral valve(s), and, within 30 days after surgery, or during the same hospital stay as the surgery, one or more of the following complications occurred either during surgery: Severe renal failure? Peripheral embolus following surgery resulting in severe permanent impairment of the kidneys, abdominal organs, or extremities? Paraplegia resulting from cervical spine injury during valvular heart surgery? 5. Surgery to repair or replace the aortic and/or mitral valve(s), and a stroke caused by that surgery where the stroke has produced a permanent condition which meets the criteria of the AHA Stroke Outcome Classification 9 Functional Levels II or III determined at least six months after the event? 7
8 6. Surgery to repair or replace the aortic and/or mitral valve(s) with: post-operative endocarditis, mediastinitis or sternal osteomyelitis within six months of surgery, any of which required reopening of the median sternotomy for treatment, a post-operative serious infection defined as HIV or Hepatitis other than Hepatitis A, as a result of blood transfusion associated with the heart valve surgery? 7. Surgery to repair or replace the aortic and/or mitral valve(s) and a second surgery through the sternum due to prosthetic valve malfunction, poor fit, or complications reasonably related to the initial surgery but not due to replacement of a valve at the end of its functional life as determined by the Treating Physician? 8. Death resulting from a condition caused by valvular heart disease or valvular repair/replacement surgery following use of the Products and a statement from the attending Certified Cardiac Surgeon or Certified Cardiologist regarding why the surgery is not being performed? V. Matrix Level V Incapacitating Complications of Regurgitation and/or Surgery 1. Qualification for Matrix Level I, II, III, or IV with one or more of the following: A severe stroke contracted after use of the Products which has resulted in a permanent condition which meets the criteria of AHA Stroke Outcome Classification 10 Functional Levels IV or V, determined at least six months after the event, which was caused by one of the following: (i) aortic and/or mitral valve surgery, (ii) bacterial endocarditis in the valve upon which the claim is based, 8
9 (iii) chronic atrial fibrillation with left atrial enlargement? Quadriplegia resulting from cervical spine injury during valvular heart surgery? Qualification for payment at Matrix Levels III or IV and New York Heart Association Functional Class III or Class IV symptoms as documented by the attending Certified Cardiac Surgeon or Certified Cardiologist and significant damage to the heart muscle 11? (d) Heart transplant as a consequence of valvular heart disease of a nature subject to benefits under the Settlement? (e) Irreversible pulmonary hypertension secondary to valvular heart disease defined as peak systolic pulmonary artery pressure > 50 mm Hg at rest by cardiac catheterization or > 60 mm Hg by Doppler echocardiogram, where cardiac catheterization is medically contraindicated, following repair or replacement surgery? (f) A persistent non-cognitive state 12 caused by a complication of valvular heart disease (e.g. cardiac arrest) or valvular repair/replacement surgery? 2. The Product Recipient otherwise qualifies for payment at Matrix Level II, III or IV and ventricular fibrillation or sustained ventricular tachycardia which results in hemodynamic compromise, in the absence of hemodynamically significant coronary artery disease as demonstrated by cardiac catheterization except where medically contraindicated? 9
10 F. Valve Pathology Evidence Does the Product Recipient have evidence of valve pathology produced on Verhoeff-Van Gieson stain or Movat pentachrome stain which demonstrates the valve pathology associated with ingestion of the Products as defined by Volmar 13? G. Additional Medical Factors To Be Considered To the best of your knowledge, does the Product Recipient have any of the following conditions? (Check each that applies): 1. In the case of an aortic valve claim, does the Product Recipient have one of the following conditions? Moderate or greater aortic sclerosis as defined by Otto 14 in a Product Recipients who is 60 years old as of the time they are first diagnosed as with an FDA Positive condition? Diseases of the aortic root in conjunction with aortic root dilatation of cm measured at the ascending aorta? 2. In the case of a mitral valve claim, does the Product Recipient have one of the following conditions? Moderate or greater mitral annular calcification as defined by Otto 15? Chronic, inadequately treated, severe systemic hypertension (greater than 160/100) of at least five years duration with left ventricular ejection fraction of 40% or less? 10
11 Evidence of greater than or equal to Grade III systolic murmur on auscultation that was not refuted by echocardiography and that was attributed by the Treating Physician to mitral regurgitation prior to use of the Products? 3. In the case of either an aortic valve claim or a mitral valve claim, does the Product Recipient have one of the following conditions? Bacterial endocarditis prior to use of the Products in the valve which is the basis of the claim? FDA Positive Regurgitation confirmed by echocardiogram prior to use of the Products for the valve that is the basis of the claim? Prior history of daily use of methysergide or ergotamines for a continuous period of longer than 120 days? (d) Prior history of daily use of pergolide or bromocriptine for a continuous period of at least two years? H. Exclusionary Conditions To the best of your knowledge, does the Product Recipient have any of the following conditions? (Check each that applies): 1. In the case of an aortic valve claim, does the Product Recipient have one of the following conditions? Aortic root disease in conjunction with aortic root dilatation >4.5 cm measured at the ascending aorta? 11
12 Aortic stenosis and aortic valve area <2.0 cm 2 by the Continuity Equation? Evidence of diastolic murmur on auscultation that was not refuted by echocardiography and that was attributed by the Treating Physician to aortic insufficiency or to aortic valve disease prior to use of the Products? (d) The following congenital aortic valve abnormalities: unicuspid, bicuspid or quadricuspid aortic valve, or ventricular septal defect associated with aortic regurgitation? (e) Aortic dissection involving the aortic root and/or aortic valve? 2. In the case of a mitral valve claim, does the Product Recipient have one of the following conditions? Mitral Valve Prolapse of the degree defined by Freed 16 as a condition where the echocardiogram recording includes the parasternal long axis view and that echocardiographic view shows displacement of one or both mitral leaflets >2mm above the atrial-ventricular border prolapsing into the left atrium during systole, and >5mm mitral leaflet thickening during diastole, and as determined by a Certified Cardiologist? M-Mode and 2-D echocardiographic evidence of rheumatic mitral valves (doming of the anterior leaflet and/or anterior motion of the posterior leaflet and/or commissural fusion), except where a Certified Pathologist has examined mitral valve tissue and provided a statement confirming that he/she has determined that there was no evidence of rheumatic valve disease? Evidence of mitral valve obstruction with mitral valve area of <2.0 cm 2 as measured by the Continuity Equation? 12
13 (d) The following congenital mitral valve abnormalities: parachute valve, or cleft of the mitral valve? (e) Chordae rupture with flail leaflet? (f) Acute myocardial infarction associated with papillary muscle dysfunction or rupture? (g) Dilated cardiomyopathy diagnosed prior to use of the Products? (h) Chronic, inadequately treated, severe systemic hypertension (greater than 160/110) of at least ten years duration with LVEF of 40% or less? 3. In the case of either an aortic valve claim or a mitral valve claim, does the Product Recipient have one of the following conditions? Heart valve surgery on the valve that is the basis of the claims prior to use of the Products? A diagnosis of Systemic Lupus Erythematosis or a diagnosis of Rheumatoid Arthritis as defined by Harrison 17 or a diagnosis of Ehler s Danlos syndrome, and valvular abnormalities of a type associated with those conditions as defined by Otto 18? End-stage renal disease diagnosed prior to use of the Products? 13
14 (d) No evidence of the valve pathology alleged to be associated with ingestion of the Products as defined by Volmar 19 where such pathology is available on Verhoeff- Van Gieson stain or Movat pentachrome stain? (e) Carcinoid tumor of a type associated with aortic and/or mitral valve lesions or carcinoid syndrome? (f) Mucopolysaccharidoses? (g) I. Declaration Gross or microscopic pathology of explanted valves diagnostic of VHD attributable to another cause listed in sections G or H? I declare under penalty of perjury that it is my opinion, to a reasonable degree of medical certainty, and to the best of my knowledge, that: (d) (e) the Product Recipient has been diagnosed with the medical condition(s) specified in Section E above which serves as the basis of the claim; the specific medical condition which serves as the basis of the claim was not present in the Product Recipient prior to his or her first use of the Products; the Product Recipient does or does not have the conditions listed in Sections G and H, as indicated above; if one of the conditions listed in Sections G or H is present, there is nevertheless a possibility that the Product Recipient s condition was caused by ingestion of the Products; the echocardiogram(s) that serve as the basis of this claim meet the criteria set forth in the Appendix attached to this form. (Date: MM/DD/YYYY) (Signature of Treating Physician) This form is an official Court document sanctioned by the Court and submitting it to the Ponderal/Redux Settlement Administrator is equivalent to filing it with a Court. 14
15 1 See Centers for Disease Control and Prevention, U.S. Dep t of Health and Human Services, Cardiac Valvulopathy Associated with Exposure to Fenfluramine or Dexfenfluramine: US Department of Health and Human Services Interim Public Health Recommendations, 46 Morbidity & Mortality Weekly Rep. 1061, (1997). 2 See supra, note 1 3 Robert O. Bonow, et al., Guidelines for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on Management of Patients With Valvular Heart Disease), 32 J. Am. C. Cardiology 1486 (1998) at See supra, note 3 at See supra, note 2. 6 See supra, note 3. 7 M. Kelley-Hayes, et al., The American Heart Association Stroke Outcome Classification, 29 Stroke , (1998). (Note: approved by the American Heart Association Science Advisory and Coordinating committee). 8 See supra, note 6. 9 See supra, note See supra, note See supra, note (16). 12 Adelman, G. ed., Encyclopedia of Neuroscience at 268 (1987). 13 Volmar et al., Aortic and Mitral Fenfluramine-Phentermine Valvulopathy in 64 Patients Treated with Anorectic Agents, 125 Arch. Pathol. Lab. Med (Dec. 2001). 14 Otto, C.M. et al., Association of Aortic Valve Sclerosis with Cardiovascular Mortality and Morbidity in the Elderly, 341 New Eng. J. Med., (1999). 15 Otto, C.M., The Practice of Clinical Echocardiography 808 (2d Ed. 2002). 16 Lisa A. Freed, et al., Prevalence and Clinical Outcomes of Mitral Valve Prolapse, 341 New Eng J. Med.1, 2 (1999). 17 Harrison s Principles of Internal Medicine 1878, 1885 (14 th ed. 1998). 18 See supra, note (26 at ). 19 See supra, note
IN THE UNITED STATES DISTRICT COURT FOR THE EASTERN DISTRICT OF PENNSYLVANIA
 IN THE UNITED STATES DISTRICT COURT FOR THE EASTERN DISTRICT OF PENNSYLVANIA IN RE: DIET DRUGS (PHENTERMINE / : MDL DOCKET NO. FENFLURAMINE/DEXFENFLURAMINE) : 2 :15 MD1203 PRODUCTS LIABILITY LITIGATION
IN THE UNITED STATES DISTRICT COURT FOR THE EASTERN DISTRICT OF PENNSYLVANIA IN RE: DIET DRUGS (PHENTERMINE / : MDL DOCKET NO. FENFLURAMINE/DEXFENFLURAMINE) : 2 :15 MD1203 PRODUCTS LIABILITY LITIGATION
Degenerative Mitral Regurgitation: Etiology and Natural History of Disease and Triggers for Intervention
 Degenerative Mitral Regurgitation: Etiology and Natural History of Disease and Triggers for Intervention John N. Hamaty D.O. FACC, FACOI November 17 th 2017 I have no financial disclosures Primary Mitral
Degenerative Mitral Regurgitation: Etiology and Natural History of Disease and Triggers for Intervention John N. Hamaty D.O. FACC, FACOI November 17 th 2017 I have no financial disclosures Primary Mitral
Adult Echocardiography Examination Content Outline
 Adult Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 2 3 4 5 Anatomy and Physiology Pathology Clinical Care and Safety Measurement Techniques, Maneuvers,
Adult Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 2 3 4 5 Anatomy and Physiology Pathology Clinical Care and Safety Measurement Techniques, Maneuvers,
ICE: Echo Core Lab-CRF
 APPENDIX 1 ICE: Echo Core Lab-CRF Study #: - Pt Initials: 1. Date of study: / / D D M M M Y Y Y Y 2. Type of Study: TTE TEE 3. Quality of Study: Poor Moderate Excellent Ejection Fraction 4. Ejection Fraction
APPENDIX 1 ICE: Echo Core Lab-CRF Study #: - Pt Initials: 1. Date of study: / / D D M M M Y Y Y Y 2. Type of Study: TTE TEE 3. Quality of Study: Poor Moderate Excellent Ejection Fraction 4. Ejection Fraction
Common Codes for ICD-10
 Common Codes for ICD-10 Specialty: Cardiology *Always utilize more specific codes first. ABNORMALITIES OF HEART RHYTHM ICD-9-CM Codes: 427.81, 427.89, 785.0, 785.1, 785.3 R00.0 Tachycardia, unspecified
Common Codes for ICD-10 Specialty: Cardiology *Always utilize more specific codes first. ABNORMALITIES OF HEART RHYTHM ICD-9-CM Codes: 427.81, 427.89, 785.0, 785.1, 785.3 R00.0 Tachycardia, unspecified
8/31/2016. Mitraclip in Matthew Johnson, MD
 Mitraclip in 2016 Matthew Johnson, MD 1 Abnormal Valve Function Valve Stenosis Obstruction to valve flow during that phase of the cardiac cycle when the valve is normally open. Hemodynamic hallmark - pressure
Mitraclip in 2016 Matthew Johnson, MD 1 Abnormal Valve Function Valve Stenosis Obstruction to valve flow during that phase of the cardiac cycle when the valve is normally open. Hemodynamic hallmark - pressure
Certificate in Clinician Performed Ultrasound (CCPU) Syllabus. Rapid Cardiac Echo (RCE)
 Certificate in Clinician Performed Ultrasound (CCPU) Syllabus Rapid Cardiac Echo (RCE) Purpose: Rapid Cardiac Echocardiography (RCE) This unit is designed to cover the theoretical and practical curriculum
Certificate in Clinician Performed Ultrasound (CCPU) Syllabus Rapid Cardiac Echo (RCE) Purpose: Rapid Cardiac Echocardiography (RCE) This unit is designed to cover the theoretical and practical curriculum
Echocardiographic Cardiovascular Risk Stratification: Beyond Ejection Fraction
 Echocardiographic Cardiovascular Risk Stratification: Beyond Ejection Fraction October 4, 2014 James S. Lee, M.D., F.A.C.C. Associates in Cardiology, P.A. Silver Spring, M.D. Disclosures Financial none
Echocardiographic Cardiovascular Risk Stratification: Beyond Ejection Fraction October 4, 2014 James S. Lee, M.D., F.A.C.C. Associates in Cardiology, P.A. Silver Spring, M.D. Disclosures Financial none
MITRAL STENOSIS. Joanne Cusack
 MITRAL STENOSIS Joanne Cusack BSE Breakdown Recognition of rheumatic mitral stenosis Qualitative description of valve and sub-valve calcification and fibrosis Measurement of orifice area by planimetry
MITRAL STENOSIS Joanne Cusack BSE Breakdown Recognition of rheumatic mitral stenosis Qualitative description of valve and sub-valve calcification and fibrosis Measurement of orifice area by planimetry
Chapter 24: Diagnostic workup and evaluation: eligibility, risk assessment, FDA guidelines Ashwin Nathan, MD, Saif Anwaruddin, MD, FACC Penn Medicine
 Chapter 24: Diagnostic workup and evaluation: eligibility, risk assessment, FDA guidelines Ashwin Nathan, MD, Saif Anwaruddin, MD, FACC Penn Medicine Mitral regurgitation, regurgitant flow between the
Chapter 24: Diagnostic workup and evaluation: eligibility, risk assessment, FDA guidelines Ashwin Nathan, MD, Saif Anwaruddin, MD, FACC Penn Medicine Mitral regurgitation, regurgitant flow between the
PROSTHETIC VALVE BOARD REVIEW
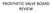 PROSTHETIC VALVE BOARD REVIEW The correct answer D This two chamber view shows a porcine mitral prosthesis with the typical appearance of the struts although the leaflets are not well seen. The valve
PROSTHETIC VALVE BOARD REVIEW The correct answer D This two chamber view shows a porcine mitral prosthesis with the typical appearance of the struts although the leaflets are not well seen. The valve
ECHOCARDIOGRAPHY DATA REPORT FORM
 Patient ID Patient Study ID AVM - - Date of form completion / / 20 Initials of person completing the form mm dd yyyy Study period Preoperative Postoperative Operative 6-month f/u 1-year f/u 2-year f/u
Patient ID Patient Study ID AVM - - Date of form completion / / 20 Initials of person completing the form mm dd yyyy Study period Preoperative Postoperative Operative 6-month f/u 1-year f/u 2-year f/u
DISCLOSURE. Echocardiography in Systemic Diseases: Questions. Relevant Financial Relationship(s) None. Off Label Usage None 5/7/2018
 Echocardiography in Systemic Diseases: Questions Sunil Mankad, MD, FACC, FCCP, FASE Associate Professor of Medicine Mayo Clinic College of Medicine Director, Transesophageal Echocardiography Associate
Echocardiography in Systemic Diseases: Questions Sunil Mankad, MD, FACC, FCCP, FASE Associate Professor of Medicine Mayo Clinic College of Medicine Director, Transesophageal Echocardiography Associate
Echocardiography as a diagnostic and management tool in medical emergencies
 Echocardiography as a diagnostic and management tool in medical emergencies Frank van der Heusen MD Department of Anesthesia and perioperative Care UCSF Medical Center Objective of this presentation Indications
Echocardiography as a diagnostic and management tool in medical emergencies Frank van der Heusen MD Department of Anesthesia and perioperative Care UCSF Medical Center Objective of this presentation Indications
Congenital Heart Disease Cases
 Congenital Heart Disease Cases Sabrina Phillips, MD FACC FASE Mayo Clinic Congenital Heart Disease Center 2013 MFMER slide-1 No Disclosures 2013 MFMER slide-2 1 CASE 1 2013 MFMER slide-3 63 year old Woman
Congenital Heart Disease Cases Sabrina Phillips, MD FACC FASE Mayo Clinic Congenital Heart Disease Center 2013 MFMER slide-1 No Disclosures 2013 MFMER slide-2 1 CASE 1 2013 MFMER slide-3 63 year old Woman
Mitral Valve Disease, When to Intervene
 Mitral Valve Disease, When to Intervene Swedish Heart and Vascular Institute Ming Zhang MD PhD Interventional Cardiology Structure Heart Disease Conflict of Interest None Current ACC/AHA guideline Stages
Mitral Valve Disease, When to Intervene Swedish Heart and Vascular Institute Ming Zhang MD PhD Interventional Cardiology Structure Heart Disease Conflict of Interest None Current ACC/AHA guideline Stages
Valvular Heart Disease in Clinical Practice
 Valvular Heart Disease in Clinical Practice Michael Y. Henein Editor Valvular Heart Disease in Clinical Practice 123 Editor Michael Y. Henein Consultant Cardiologist Umea Heart Centre Umea University
Valvular Heart Disease in Clinical Practice Michael Y. Henein Editor Valvular Heart Disease in Clinical Practice 123 Editor Michael Y. Henein Consultant Cardiologist Umea Heart Centre Umea University
Index. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Acute coronary syndrome(s), anticoagulant therapy in, 706, 707 antiplatelet therapy in, 702 ß-blockers in, 703 cardiac biomarkers in,
Index Note: Page numbers of article titles are in boldface type. A Acute coronary syndrome(s), anticoagulant therapy in, 706, 707 antiplatelet therapy in, 702 ß-blockers in, 703 cardiac biomarkers in,
pulmonary valve on, 107 pulmonary valve vegetations on, 113
 INDEX Adriamycin-induced cardiomyopathy, 176 Amyloidosis, 160-161 echocardiographic abnormalities in, 160 intra-mural tumors similar to, 294 myocardial involvement in, 160-161 two-dimensional echocardiography
INDEX Adriamycin-induced cardiomyopathy, 176 Amyloidosis, 160-161 echocardiographic abnormalities in, 160 intra-mural tumors similar to, 294 myocardial involvement in, 160-161 two-dimensional echocardiography
Aortic Valve Practice Guidelines: What Has Changed and What You Need to Know
 Aortic Valve Practice Guidelines: What Has Changed and What You Need to Know James F. Burke, MD Program Director Cardiovascular Disease Fellowship Lankenau Medical Center Disclosure Dr. Burke has no conflicts
Aortic Valve Practice Guidelines: What Has Changed and What You Need to Know James F. Burke, MD Program Director Cardiovascular Disease Fellowship Lankenau Medical Center Disclosure Dr. Burke has no conflicts
HISTORY. Question: How do you interpret the patient s history? CHIEF COMPLAINT: Dyspnea of two days duration. PRESENT ILLNESS: 45-year-old man.
 HISTORY 45-year-old man. CHIEF COMPLAINT: Dyspnea of two days duration. PRESENT ILLNESS: His dyspnea began suddenly and has been associated with orthopnea, but no chest pain. For two months he has felt
HISTORY 45-year-old man. CHIEF COMPLAINT: Dyspnea of two days duration. PRESENT ILLNESS: His dyspnea began suddenly and has been associated with orthopnea, but no chest pain. For two months he has felt
PART II ECHOCARDIOGRAPHY LABORATORY OPERATIONS ADULT TRANSTHORACIC ECHOCARDIOGRAPHY TESTING
 PART II ECHOCARDIOGRAPHY LABORATORY OPERATIONS ADULT TRANSTHORACIC ECHOCARDIOGRAPHY TESTING STANDARD - Primary Instrumentation 1.1 Cardiac Ultrasound Systems SECTION 1 Instrumentation Ultrasound instruments
PART II ECHOCARDIOGRAPHY LABORATORY OPERATIONS ADULT TRANSTHORACIC ECHOCARDIOGRAPHY TESTING STANDARD - Primary Instrumentation 1.1 Cardiac Ultrasound Systems SECTION 1 Instrumentation Ultrasound instruments
Clinicians and Facilities: RESOURCES WHEN CARING FOR WOMEN WITH ADULT CONGENITAL HEART DISEASE OR OTHER FORMS OF CARDIOVASCULAR DISEASE!!
 Clinicians and Facilities: RESOURCES WHEN CARING FOR WOMEN WITH ADULT CONGENITAL HEART DISEASE OR OTHER FORMS OF CARDIOVASCULAR DISEASE!! Abha'Khandelwal,'MD,'MS' 'Stanford'University'School'of'Medicine'
Clinicians and Facilities: RESOURCES WHEN CARING FOR WOMEN WITH ADULT CONGENITAL HEART DISEASE OR OTHER FORMS OF CARDIOVASCULAR DISEASE!! Abha'Khandelwal,'MD,'MS' 'Stanford'University'School'of'Medicine'
Detailed Order Request Checklists for Cardiology
 Next Generation Solutions Detailed Order Request Checklists for Cardiology 8600 West Bryn Mawr Avenue South Tower Suite 800 Chicago, IL 60631 www.aimspecialtyhealth.com Appropriate.Safe.Affordable 2018
Next Generation Solutions Detailed Order Request Checklists for Cardiology 8600 West Bryn Mawr Avenue South Tower Suite 800 Chicago, IL 60631 www.aimspecialtyhealth.com Appropriate.Safe.Affordable 2018
Case # 1. Page: 8. DUKE: Adams
 Case # 1 Page: 8 1. The cardiac output in this patient is reduced because of: O a) tamponade physiology O b) restrictive physiology O c) coronary artery disease O d) left bundle branch block Page: 8 1.
Case # 1 Page: 8 1. The cardiac output in this patient is reduced because of: O a) tamponade physiology O b) restrictive physiology O c) coronary artery disease O d) left bundle branch block Page: 8 1.
Ethiology of the disease - What may cause it?: One of the following numerous causes can cause aortic regurgitation:
 AORTIC REGURGITATION Aortic regurgitation is one of the aortic valve diseases, the aorta being the artery that brings oxygenated blood to the systemic circulation. The other one is the aortic valve stenosis.
AORTIC REGURGITATION Aortic regurgitation is one of the aortic valve diseases, the aorta being the artery that brings oxygenated blood to the systemic circulation. The other one is the aortic valve stenosis.
Regurgitant Lesions. Bicol Hospital, Legazpi City, Philippines July Gregg S. Pressman MD, FACC, FASE Einstein Medical Center Philadelphia, USA
 Regurgitant Lesions Bicol Hospital, Legazpi City, Philippines July 2016 Gregg S. Pressman MD, FACC, FASE Einstein Medical Center Philadelphia, USA Aortic Insufficiency Valve anatomy and function LVOT and
Regurgitant Lesions Bicol Hospital, Legazpi City, Philippines July 2016 Gregg S. Pressman MD, FACC, FASE Einstein Medical Center Philadelphia, USA Aortic Insufficiency Valve anatomy and function LVOT and
Mitral Valve Disease. Prof. Sirchak Yelizaveta Stepanovna
 Mitral Valve Disease Prof. Sirchak Yelizaveta Stepanovna Fall 2008 Mitral Valve Stenosis Lecture Outline Mitral Stenosis Mitral Regurgitation Etiology Pathophysiology Clinical features Diagnostic testing
Mitral Valve Disease Prof. Sirchak Yelizaveta Stepanovna Fall 2008 Mitral Valve Stenosis Lecture Outline Mitral Stenosis Mitral Regurgitation Etiology Pathophysiology Clinical features Diagnostic testing
Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease
 Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease TIRONE E. DAVID, MD ; SEMIN THORAC CARDIOVASC SURG 19:116-120c 2007 ELSEVIER INC. PRESENTED BY INTERN 許士盟 Mitral valve
Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease TIRONE E. DAVID, MD ; SEMIN THORAC CARDIOVASC SURG 19:116-120c 2007 ELSEVIER INC. PRESENTED BY INTERN 許士盟 Mitral valve
SONOGRAPHER & NURSE LED VALVE CLINICS
 SONOGRAPHER & NURSE LED VALVE CLINICS Frequency of visits and alerts AORTIC STENOSIS V max > 4.0 m/s or EOA < 1.0 cm 2 V max 3.5 4.0 m/s + Ca+ V max 3.0 4.0 m/s or EOA 1.0-1.5 cm 2 V max 2.5 3.0 m/s every
SONOGRAPHER & NURSE LED VALVE CLINICS Frequency of visits and alerts AORTIC STENOSIS V max > 4.0 m/s or EOA < 1.0 cm 2 V max 3.5 4.0 m/s + Ca+ V max 3.0 4.0 m/s or EOA 1.0-1.5 cm 2 V max 2.5 3.0 m/s every
Rotation: Echocardiography: Transthoracic Echocardiography (TTE)
 Rotation: Echocardiography: Transthoracic Echocardiography (TTE) Rotation Format and Responsibilities: Fellows rotate in the echocardiography laboratory in each clinical year. Rotations during the first
Rotation: Echocardiography: Transthoracic Echocardiography (TTE) Rotation Format and Responsibilities: Fellows rotate in the echocardiography laboratory in each clinical year. Rotations during the first
Pediatric Echocardiography Examination Content Outline
 Pediatric Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 Anatomy and Physiology Normal Anatomy and Physiology 10% 2 Abnormal Pathology and Pathophysiology
Pediatric Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 Anatomy and Physiology Normal Anatomy and Physiology 10% 2 Abnormal Pathology and Pathophysiology
MITRAL VALVE DISEASE- ASSESSMENT AND MANAGEMENT. Irene Frantzis P year, SGUL Sheba Medical Center
 MITRAL VALVE DISEASE- ASSESSMENT AND MANAGEMENT Irene Frantzis P year, SGUL Sheba Medical Center MITRAL VALVE DISEASE Mitral Valve Regurgitation Mitral Valve Stenosis Mitral Valve Prolapse MITRAL REGURGITATION
MITRAL VALVE DISEASE- ASSESSMENT AND MANAGEMENT Irene Frantzis P year, SGUL Sheba Medical Center MITRAL VALVE DISEASE Mitral Valve Regurgitation Mitral Valve Stenosis Mitral Valve Prolapse MITRAL REGURGITATION
TSDA Boot Camp September 13-16, Introduction to Aortic Valve Surgery. George L. Hicks, Jr., MD
 TSDA Boot Camp September 13-16, 2018 Introduction to Aortic Valve Surgery George L. Hicks, Jr., MD Aortic Valve Pathology and Treatment Valvular Aortic Stenosis in Adults Average Course (Post mortem data)
TSDA Boot Camp September 13-16, 2018 Introduction to Aortic Valve Surgery George L. Hicks, Jr., MD Aortic Valve Pathology and Treatment Valvular Aortic Stenosis in Adults Average Course (Post mortem data)
Data Collected: June 17, Reported: June 30, Survey Dates 05/24/ /07/2010
 Job Task Analysis for ARDMS Pediatric Echocardiography Data Collected: June 17, 2010 Reported: Analysis Summary For: Pediatric Echocardiography Exam Survey Dates 05/24/2010-06/07/2010 Invited Respondents
Job Task Analysis for ARDMS Pediatric Echocardiography Data Collected: June 17, 2010 Reported: Analysis Summary For: Pediatric Echocardiography Exam Survey Dates 05/24/2010-06/07/2010 Invited Respondents
Aortic Stenosis: Spectrum of Disease, Low Flow/Low Gradient and Variants
 Aortic Stenosis: Spectrum of Disease, Low Flow/Low Gradient and Variants Martin G. Keane, MD, FASE Professor of Medicine Lewis Katz School of Medicine at Temple University Basic root structure Parasternal
Aortic Stenosis: Spectrum of Disease, Low Flow/Low Gradient and Variants Martin G. Keane, MD, FASE Professor of Medicine Lewis Katz School of Medicine at Temple University Basic root structure Parasternal
HISTORY. Question: What category of heart disease is suggested by this history? CHIEF COMPLAINT: Heart murmur present since early infancy.
 HISTORY 18-year-old man. CHIEF COMPLAINT: Heart murmur present since early infancy. PRESENT ILLNESS: Although normal at birth, a heart murmur was heard at the six week check-up and has persisted since
HISTORY 18-year-old man. CHIEF COMPLAINT: Heart murmur present since early infancy. PRESENT ILLNESS: Although normal at birth, a heart murmur was heard at the six week check-up and has persisted since
Tricuspid and Pulmonic Valve Disease
 Chapter 31 Tricuspid and Pulmonic Valve Disease David A. Tate Acquired disease of the right-sided cardiac valves is much less common than disease of the leftsided counterparts, possibly because of the
Chapter 31 Tricuspid and Pulmonic Valve Disease David A. Tate Acquired disease of the right-sided cardiac valves is much less common than disease of the leftsided counterparts, possibly because of the
Index of subjects. effect on ventricular tachycardia 30 treatment with 101, 116 boosterpump 80 Brockenbrough phenomenon 55, 125
 145 Index of subjects A accessory pathways 3 amiodarone 4, 5, 6, 23, 30, 97, 102 angina pectoris 4, 24, 1l0, 137, 139, 140 angulation, of cavity 73, 74 aorta aortic flow velocity 2 aortic insufficiency
145 Index of subjects A accessory pathways 3 amiodarone 4, 5, 6, 23, 30, 97, 102 angina pectoris 4, 24, 1l0, 137, 139, 140 angulation, of cavity 73, 74 aorta aortic flow velocity 2 aortic insufficiency
Percutaneous Mitral Valve Repair: What Can We Treat and What Should We Treat
 Percutaneous Mitral Valve Repair: What Can We Treat and What Should We Treat Innovative Procedures, Devices & State of the Art Care for Arrhythmias, Heart Failure & Structural Heart Disease October 8-10,
Percutaneous Mitral Valve Repair: What Can We Treat and What Should We Treat Innovative Procedures, Devices & State of the Art Care for Arrhythmias, Heart Failure & Structural Heart Disease October 8-10,
MR echo case. N.Koutsogiannis Department of Cardiology University Hospital Of Patras
 MR echo case N.Koutsogiannis Department of Cardiology University Hospital Of Patras Case A 35 years old male came to the echo lab for a third opinion for his valvulopathy. He reports a long standing MR
MR echo case N.Koutsogiannis Department of Cardiology University Hospital Of Patras Case A 35 years old male came to the echo lab for a third opinion for his valvulopathy. He reports a long standing MR
Long-term results (22 years) of the Ross Operation a single institutional experience
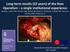 Long-term results (22 years) of the Ross Operation a single institutional experience Authors: Costa FDA, Schnorr GM, Veloso M,Calixto A, Colatusso D, Balbi EM, Torres R, Ferreira ADA, Colatusso C Department
Long-term results (22 years) of the Ross Operation a single institutional experience Authors: Costa FDA, Schnorr GM, Veloso M,Calixto A, Colatusso D, Balbi EM, Torres R, Ferreira ADA, Colatusso C Department
Heart Valve disease: MR. AS tough patient When to echo, When to refer, What s new
 Heart Valve disease: MR. AS tough patient When to echo, When to refer, What s new B. Sonnenberg UAH Cardiology CME Day 5 May 2015 Disclosures Speaker s or Advisory Boards: none Research grants: none (co-investigator
Heart Valve disease: MR. AS tough patient When to echo, When to refer, What s new B. Sonnenberg UAH Cardiology CME Day 5 May 2015 Disclosures Speaker s or Advisory Boards: none Research grants: none (co-investigator
In contrast to aortic stenosis, which essentially has 3
 Valvular Heart Disease Causes of Pure Aortic Regurgitation in Patients Having Isolated Aortic Valve Replacement at a Single US Tertiary Hospital (1993 to 2005) William Clifford Roberts, MD; Jong Mi Ko,
Valvular Heart Disease Causes of Pure Aortic Regurgitation in Patients Having Isolated Aortic Valve Replacement at a Single US Tertiary Hospital (1993 to 2005) William Clifford Roberts, MD; Jong Mi Ko,
HOW IMPORTANT ARE THESE ECHO MEASUREMENTS ANYWAY?
 HOW IMPORTANT ARE THESE ECHO MEASUREMENTS ANYWAY? John D. Carroll, MD Professor, Director of Interventional Cardiology and Co-Medical Director of the Cardiac and Vascular Center, University of Colorado
HOW IMPORTANT ARE THESE ECHO MEASUREMENTS ANYWAY? John D. Carroll, MD Professor, Director of Interventional Cardiology and Co-Medical Director of the Cardiac and Vascular Center, University of Colorado
The production of murmurs is due to 3 main factors:
 Heart murmurs The production of murmurs is due to 3 main factors: high blood flow rate through normal or abnormal orifices forward flow through a narrowed or irregular orifice into a dilated vessel or
Heart murmurs The production of murmurs is due to 3 main factors: high blood flow rate through normal or abnormal orifices forward flow through a narrowed or irregular orifice into a dilated vessel or
Catheter-based mitral valve repair MitraClip System
 Percutaneous Mitral Valve Repair: Results of the EVEREST II Trial William A. Gray MD Director of Endovascular Services Associate Professor of Clinical Medicine Columbia University Medical Center The Cardiovascular
Percutaneous Mitral Valve Repair: Results of the EVEREST II Trial William A. Gray MD Director of Endovascular Services Associate Professor of Clinical Medicine Columbia University Medical Center The Cardiovascular
Cases in Adult Congenital Heart Disease
 Cases in Adult Congenital Heart Disease Sabrina Phillips, MD FACC FASE Associate Professor of Medicine The University of Oklahoma Health Sciences Center No Disclosures I Have Palpitations 18 Year old Man
Cases in Adult Congenital Heart Disease Sabrina Phillips, MD FACC FASE Associate Professor of Medicine The University of Oklahoma Health Sciences Center No Disclosures I Have Palpitations 18 Year old Man
Valvular Heart Disease
 Valvular Heart Disease Roman M. Sniecinski, MD, FASE Associate Professor of Anesthesiology Emory University School of Medicine Learning Objectives Review the major pathophysiology of the most common heart
Valvular Heart Disease Roman M. Sniecinski, MD, FASE Associate Professor of Anesthesiology Emory University School of Medicine Learning Objectives Review the major pathophysiology of the most common heart
Stress Testing in Valvular Disease
 2017 ASE Florida Orlando, FL October 10, 2017 2:40 2:50 PM 10 min Grand Harbor Ballroom South Stress Testing in Valvular Disease Muhamed Sarić MD, PhD, MPA Director of Noninvasive Cardiology Echo Lab Associate
2017 ASE Florida Orlando, FL October 10, 2017 2:40 2:50 PM 10 min Grand Harbor Ballroom South Stress Testing in Valvular Disease Muhamed Sarić MD, PhD, MPA Director of Noninvasive Cardiology Echo Lab Associate
Mitral Valve Disease. Chapter 29
 Chapter 29 Mitral Valve Disease Thomas R. Griggs Mitral valve leaflets consist of thin, pliable, fibrous material. The two leaflets anterior and posterior open by unfolding against the ventricular wall
Chapter 29 Mitral Valve Disease Thomas R. Griggs Mitral valve leaflets consist of thin, pliable, fibrous material. The two leaflets anterior and posterior open by unfolding against the ventricular wall
Congenital. Unicuspid Bicuspid Quadricuspid
 David Letterman s Top 10 Aortic Stenosis The victim can be anyone: Echo is the question and the answer!!!! Hilton Head Island Echocardiography Conference 2012 Timothy E. Paterick, MD, JD, MBA Christopher
David Letterman s Top 10 Aortic Stenosis The victim can be anyone: Echo is the question and the answer!!!! Hilton Head Island Echocardiography Conference 2012 Timothy E. Paterick, MD, JD, MBA Christopher
Aortic Regurgitation & Aorta Evaluation
 VALVULAR HEART DISEASE Regurgitation Valvular Lessions 2017 Aortic Regurgitation & Aorta Evaluation Jorge Eduardo Cossío-Aranda MD, FACC Chairman of Outpatient Care Department Instituto Nacional de Cardiología
VALVULAR HEART DISEASE Regurgitation Valvular Lessions 2017 Aortic Regurgitation & Aorta Evaluation Jorge Eduardo Cossío-Aranda MD, FACC Chairman of Outpatient Care Department Instituto Nacional de Cardiología
The Cardiovascular System Part I: Heart Outline of class lecture After studying part I of this chapter you should be able to:
 The Cardiovascular System Part I: Heart Outline of class lecture After studying part I of this chapter you should be able to: 1. Describe the functions of the heart 2. Describe the location of the heart,
The Cardiovascular System Part I: Heart Outline of class lecture After studying part I of this chapter you should be able to: 1. Describe the functions of the heart 2. Describe the location of the heart,
Cardiology/Cardiothoracic
 Cardiology/Cardiothoracic ICD-9-CM to ICD-10-CM Code Mapper 800-334-5724 www.contexomedia.com 2013 ICD-9-CM 272.0 Pure hypercholesterolemia 272.2 Mixed hyperlipidemia 272.4 Other and hyperlipidemia 278.00
Cardiology/Cardiothoracic ICD-9-CM to ICD-10-CM Code Mapper 800-334-5724 www.contexomedia.com 2013 ICD-9-CM 272.0 Pure hypercholesterolemia 272.2 Mixed hyperlipidemia 272.4 Other and hyperlipidemia 278.00
A Health Care Professional s Guide Aortic Stenosis in Seniors
 A Health Care Professional s Guide Aortic Stenosis in Seniors With highlights from the 2014 ACC/AHA practice guidelines for valve disease Aortic stenosis (AS) is primarily caused by calcification of the
A Health Care Professional s Guide Aortic Stenosis in Seniors With highlights from the 2014 ACC/AHA practice guidelines for valve disease Aortic stenosis (AS) is primarily caused by calcification of the
ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT
 ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT Karen Stout, MD, FACC Divisions of Cardiology University of Washington Medical Center Seattle Children s Hospital NO DISCLOSURES
ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT Karen Stout, MD, FACC Divisions of Cardiology University of Washington Medical Center Seattle Children s Hospital NO DISCLOSURES
Valvular Heart Disease
 Valvular Heart Disease MITRAL STENOSIS Pathophysiology rheumatic fever. calcific degeneration, malignant carcinoid disease, congenital mitral stenosis. SLE. The increased pressure gradient across the mitral
Valvular Heart Disease MITRAL STENOSIS Pathophysiology rheumatic fever. calcific degeneration, malignant carcinoid disease, congenital mitral stenosis. SLE. The increased pressure gradient across the mitral
Hemodynamic Assessment. Assessment of Systolic Function Doppler Hemodynamics
 Hemodynamic Assessment Matt M. Umland, RDCS, FASE Aurora Medical Group Milwaukee, WI Assessment of Systolic Function Doppler Hemodynamics Stroke Volume Cardiac Output Cardiac Index Tei Index/Index of myocardial
Hemodynamic Assessment Matt M. Umland, RDCS, FASE Aurora Medical Group Milwaukee, WI Assessment of Systolic Function Doppler Hemodynamics Stroke Volume Cardiac Output Cardiac Index Tei Index/Index of myocardial
By the end of this session, the student should be able to:
 Valvular Heart disease HVD By Dr. Ashraf Abdelfatah Deyab VHD- Objectives By the end of this session, the student should be able to: Define and classify valvular heart disease. Enlist the causes of acquired
Valvular Heart disease HVD By Dr. Ashraf Abdelfatah Deyab VHD- Objectives By the end of this session, the student should be able to: Define and classify valvular heart disease. Enlist the causes of acquired
HISTORY. Question: What category of heart disease is suggested by the fact that a murmur was heard at birth?
 HISTORY 23-year-old man. CHIEF COMPLAINT: Decreasing exercise tolerance of several years duration. PRESENT ILLNESS: The patient is the product of an uncomplicated term pregnancy. A heart murmur was discovered
HISTORY 23-year-old man. CHIEF COMPLAINT: Decreasing exercise tolerance of several years duration. PRESENT ILLNESS: The patient is the product of an uncomplicated term pregnancy. A heart murmur was discovered
Which one of the following echocardiographic profiles most strongly indicates the need for aortic valve replacement? FS [
 Question 46 A 45-year-old asymptomatic man returns for follow-up. He was diagnosed 10 years ago with aortic regurgitation due to a congentia lbicuspid aortic valve, He has never had endocarditis Which
Question 46 A 45-year-old asymptomatic man returns for follow-up. He was diagnosed 10 years ago with aortic regurgitation due to a congentia lbicuspid aortic valve, He has never had endocarditis Which
(Ann Thorac Surg 2008;85:845 53)
 I Made Adi Parmana The utility of intraoperative TEE has become increasingly more evident as anesthesiologists, cardiologists, and surgeons continue to appreciate its potential application as an invaluable
I Made Adi Parmana The utility of intraoperative TEE has become increasingly more evident as anesthesiologists, cardiologists, and surgeons continue to appreciate its potential application as an invaluable
Congenital heart disease. By Dr Saima Ali Professor of pediatrics
 Congenital heart disease By Dr Saima Ali Professor of pediatrics What is the most striking clinical finding in this child? Learning objectives By the end of this lecture, final year student should be able
Congenital heart disease By Dr Saima Ali Professor of pediatrics What is the most striking clinical finding in this child? Learning objectives By the end of this lecture, final year student should be able
Cardiac MRI in ACHD What We. ACHD Patients
 Cardiac MRI in ACHD What We Have Learned to Apply to ACHD Patients Faris Al Mousily, MBChB, FAAC, FACC Consultant, Pediatric Cardiology, KFSH&RC/Jeddah Adjunct Faculty, Division of Pediatric Cardiology
Cardiac MRI in ACHD What We Have Learned to Apply to ACHD Patients Faris Al Mousily, MBChB, FAAC, FACC Consultant, Pediatric Cardiology, KFSH&RC/Jeddah Adjunct Faculty, Division of Pediatric Cardiology
7. Echocardiography Appropriate Use Criteria (by Indication)
 Criteria for Echocardiography 1133 7. Echocardiography Criteria (by ) Table 1. TTE for General Evaluation of Cardiac Structure and Function Suspected Cardiac Etiology General With TTE 1. Symptoms or conditions
Criteria for Echocardiography 1133 7. Echocardiography Criteria (by ) Table 1. TTE for General Evaluation of Cardiac Structure and Function Suspected Cardiac Etiology General With TTE 1. Symptoms or conditions
Presenter: Steven Brust, HCS-D, HCS-H Product Manager, Home Health Coding Center
 Presenter: Steven Brust, HCS-D, HCS-H Product Manager, Home Health Coding Center Pinpoint & properly assign the appropriate heart failure codes Left- vs. Right-sided Left ventricular failure (LVF) may
Presenter: Steven Brust, HCS-D, HCS-H Product Manager, Home Health Coding Center Pinpoint & properly assign the appropriate heart failure codes Left- vs. Right-sided Left ventricular failure (LVF) may
Managing Hypertrophic Cardiomyopathy with Imaging. Gisela C. Mueller University of Michigan Department of Radiology
 Managing Hypertrophic Cardiomyopathy with Imaging Gisela C. Mueller University of Michigan Department of Radiology Disclosures Gadolinium contrast material for cardiac MRI Acronyms Afib CAD Atrial fibrillation
Managing Hypertrophic Cardiomyopathy with Imaging Gisela C. Mueller University of Michigan Department of Radiology Disclosures Gadolinium contrast material for cardiac MRI Acronyms Afib CAD Atrial fibrillation
What are the best diagnostic tools to quantify aortic regurgitation?
 What are the best diagnostic tools to quantify aortic regurgitation? Agnès Pasquet, MD, PhD Pôle de Recherche Cardiovasculaire Institut de Recherche Expérimentale et Clinique Université catholique de Louvain
What are the best diagnostic tools to quantify aortic regurgitation? Agnès Pasquet, MD, PhD Pôle de Recherche Cardiovasculaire Institut de Recherche Expérimentale et Clinique Université catholique de Louvain
Imaging Assessment of Aortic Stenosis/Aortic Regurgitation
 Imaging Assessment of Aortic Stenosis/Aortic Regurgitation Craig E Fleishman, MD FACC FASE The Heart Center at Arnold Palmer Hospital for Children, Orlando SCAI Fall Fellows Course 2014 Las Vegas Disclosure
Imaging Assessment of Aortic Stenosis/Aortic Regurgitation Craig E Fleishman, MD FACC FASE The Heart Center at Arnold Palmer Hospital for Children, Orlando SCAI Fall Fellows Course 2014 Las Vegas Disclosure
Aortic regurgitation. Physiopathology. Dr Pilar Tornos Hospital Vall d Hebron. Barcelona Eurovalve 2014
 Aortic regurgitation. Physiopathology Dr Pilar Tornos Hospital Vall d Hebron. Barcelona Eurovalve 2014 Faculty disclosure Pilar Tornos I have no financial relationships to disclose. Acute AR Etiology:
Aortic regurgitation. Physiopathology Dr Pilar Tornos Hospital Vall d Hebron. Barcelona Eurovalve 2014 Faculty disclosure Pilar Tornos I have no financial relationships to disclose. Acute AR Etiology:
Physical Exam Part II
 Physical Exam Part II University of Michigan Cardiovascular Center Kim A. Eagle, MD Albion Walter Hewlett Professor Director Physical Exam: Part II Heart Sounds Heart Murmurs HEART SOUNDS S1 MITRAL + TRICUSPID
Physical Exam Part II University of Michigan Cardiovascular Center Kim A. Eagle, MD Albion Walter Hewlett Professor Director Physical Exam: Part II Heart Sounds Heart Murmurs HEART SOUNDS S1 MITRAL + TRICUSPID
A new era in cardiac valve surgery has begun...
 THE CENTER FOR VALVE SURGERY A new era in cardiac valve surgery has begun... Good Help to Those in Need Rawn Salenger, MD, FACS, Director, The Center for Valve Surgery Edward F. Lundy, MD, PhD, Chief of
THE CENTER FOR VALVE SURGERY A new era in cardiac valve surgery has begun... Good Help to Those in Need Rawn Salenger, MD, FACS, Director, The Center for Valve Surgery Edward F. Lundy, MD, PhD, Chief of
The Doppler Examination. Katie Twomley, MD Wake Forest Baptist Health - Lexington
 The Doppler Examination Katie Twomley, MD Wake Forest Baptist Health - Lexington OUTLINE Principles/Physics Use in valvular assessment Aortic stenosis (continuity equation) Aortic regurgitation (pressure
The Doppler Examination Katie Twomley, MD Wake Forest Baptist Health - Lexington OUTLINE Principles/Physics Use in valvular assessment Aortic stenosis (continuity equation) Aortic regurgitation (pressure
A DAYS CARDIOVASCULAR UNIT GUIDE DUE WEDNESDAY 4/12
 A DAYS CARDIOVASCULAR UNIT GUIDE DUE WEDNESDAY 4/12 MONDAY TUESDAY WEDNESDAY THURSDAY FRIDAY 3/20 - B 3/21 - A 3/22 - B 3/23 - A 3/24 - B 3/27 - A Dissection Ethics Debate 3/28 - B 3/29 - A Intro to Cardiovascular
A DAYS CARDIOVASCULAR UNIT GUIDE DUE WEDNESDAY 4/12 MONDAY TUESDAY WEDNESDAY THURSDAY FRIDAY 3/20 - B 3/21 - A 3/22 - B 3/23 - A 3/24 - B 3/27 - A Dissection Ethics Debate 3/28 - B 3/29 - A Intro to Cardiovascular
SUPPLEMENTAL MATERIAL
 SUPPLEMENTAL MATERIAL Table S1: Number and percentage of patients by age category Distribution of age Age
SUPPLEMENTAL MATERIAL Table S1: Number and percentage of patients by age category Distribution of age Age
Appendix II: ECHOCARDIOGRAPHY ANALYSIS
 Appendix II: ECHOCARDIOGRAPHY ANALYSIS Two-Dimensional (2D) imaging was performed using the Vivid 7 Advantage cardiovascular ultrasound system (GE Medical Systems, Milwaukee) with a frame rate of 400 frames
Appendix II: ECHOCARDIOGRAPHY ANALYSIS Two-Dimensional (2D) imaging was performed using the Vivid 7 Advantage cardiovascular ultrasound system (GE Medical Systems, Milwaukee) with a frame rate of 400 frames
Indications of Coronary Angiography Dr. Shaheer K. George, M.D Faculty of Medicine, Mansoura University 2014
 Indications of Coronary Angiography Dr. Shaheer K. George, M.D Faculty of Medicine, Mansoura University 2014 Indications for cardiac catheterization Before a decision to perform an invasive procedure such
Indications of Coronary Angiography Dr. Shaheer K. George, M.D Faculty of Medicine, Mansoura University 2014 Indications for cardiac catheterization Before a decision to perform an invasive procedure such
MITRAL REGURGITATION ECHO PARAMETERS TOOL
 Comprehensive assessment of qualitative and quantitative parameters, along with the use of standardized nomenclature when reporting echocardiographic findings, helps to better define a patient s MR and
Comprehensive assessment of qualitative and quantitative parameters, along with the use of standardized nomenclature when reporting echocardiographic findings, helps to better define a patient s MR and
Valvular Heart Disease: Recognition and Management in the Outpatient Setting
 Valvular Heart Disease: Recognition and Management in the Outpatient Setting Ian S. Harris UCSF Division of Cardiology Case 1: 80 year old man with a history of hypertension, complaining of exertional
Valvular Heart Disease: Recognition and Management in the Outpatient Setting Ian S. Harris UCSF Division of Cardiology Case 1: 80 year old man with a history of hypertension, complaining of exertional
G. AORTIC STENOSIS (AS)
 G. AORTIC STENOSIS (AS) DEFINITION THE FACTS Aortic stenosis (AS) is a narrowing/thickening/obstruction of the aortic valve (AOV) that impedes systolic flow traveling from the left ventricle, through the
G. AORTIC STENOSIS (AS) DEFINITION THE FACTS Aortic stenosis (AS) is a narrowing/thickening/obstruction of the aortic valve (AOV) that impedes systolic flow traveling from the left ventricle, through the
ECHOCARDIOGRAPHY. Patient Care. Goals and Objectives PF EF MF LF Aspirational
 Patient Care Be able to: Perform and interpret basic TTE and X cardiac Doppler examinations Perform and interpret a comprehensive X TTE and cardiac Doppler examination Perform and interpret a comprehensive
Patient Care Be able to: Perform and interpret basic TTE and X cardiac Doppler examinations Perform and interpret a comprehensive X TTE and cardiac Doppler examination Perform and interpret a comprehensive
Percutaneous Mitral Valve Repair
 Percutaneous Mitral Valve Repair MitraClip: Procedure, Data, Patient Selection Chad Rammohan, MD FACC Director, Cardiac Cath Lab El Camino Hospital Mountain View, California Mitral Regurgitation MitraClip
Percutaneous Mitral Valve Repair MitraClip: Procedure, Data, Patient Selection Chad Rammohan, MD FACC Director, Cardiac Cath Lab El Camino Hospital Mountain View, California Mitral Regurgitation MitraClip
C1: Medical Standards for Safety Critical Workers with Cardiovascular Disorders
 C1: Medical Standards for Safety Critical Workers with Cardiovascular Disorders GENERAL ISSUES REGARDING MEDICAL FITNESS-FOR-DUTY 1. These medical standards apply to Union Pacific Railroad (UPRR) employees
C1: Medical Standards for Safety Critical Workers with Cardiovascular Disorders GENERAL ISSUES REGARDING MEDICAL FITNESS-FOR-DUTY 1. These medical standards apply to Union Pacific Railroad (UPRR) employees
Bogdan A. Popescu. University of Medicine and Pharmacy Bucharest, Romania. EAE Course, Bucharest, April 2010
 Bogdan A. Popescu University of Medicine and Pharmacy Bucharest, Romania EAE Course, Bucharest, April 2010 This is how it started Mitral stenosis at a glance 2D echo narrow diastolic opening of MV leaflets
Bogdan A. Popescu University of Medicine and Pharmacy Bucharest, Romania EAE Course, Bucharest, April 2010 This is how it started Mitral stenosis at a glance 2D echo narrow diastolic opening of MV leaflets
LV FUNCTION ASSESSMENT: WHAT IS BEYOND EJECTION FRACTION
 LV FUNCTION ASSESSMENT: WHAT IS BEYOND EJECTION FRACTION Jamilah S AlRahimi Assistant Professor, KSU-HS Consultant Noninvasive Cardiology KFCC, MNGHA-WR Introduction LV function assessment in Heart Failure:
LV FUNCTION ASSESSMENT: WHAT IS BEYOND EJECTION FRACTION Jamilah S AlRahimi Assistant Professor, KSU-HS Consultant Noninvasive Cardiology KFCC, MNGHA-WR Introduction LV function assessment in Heart Failure:
Ch.15 Cardiovascular System Pgs {15-12} {15-13}
 Ch.15 Cardiovascular System Pgs {15-12} {15-13} E. Skeleton of the Heart 1. The skeleton of the heart is composed of rings of dense connective tissue and other masses of connective tissue in the interventricular
Ch.15 Cardiovascular System Pgs {15-12} {15-13} E. Skeleton of the Heart 1. The skeleton of the heart is composed of rings of dense connective tissue and other masses of connective tissue in the interventricular
When should we intervene surgically in pediatric patient with MR?
 When should we intervene surgically in pediatric patient with MR? DR.SAUD A. BAHAIDARAH CONSULTANT, PEDIATRIC CARDIOLOGY ASSISTANT PROFESSOR OF PEDIATRICS HEAD OF CARDIOLOGY AND CARDIAC SURGERY UNIT KAUH
When should we intervene surgically in pediatric patient with MR? DR.SAUD A. BAHAIDARAH CONSULTANT, PEDIATRIC CARDIOLOGY ASSISTANT PROFESSOR OF PEDIATRICS HEAD OF CARDIOLOGY AND CARDIAC SURGERY UNIT KAUH
The production of murmurs is due to 3 main factors:
 Heart murmurs The production of murmurs is due to 3 main factors: high blood flow rate through normal or abnormal orifices forward flow through a narrowed or irregular orifice into a dilated vessel or
Heart murmurs The production of murmurs is due to 3 main factors: high blood flow rate through normal or abnormal orifices forward flow through a narrowed or irregular orifice into a dilated vessel or
Uptofate Study Summary
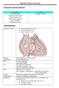 CONGENITAL HEART DISEASE Uptofate Study Summary Acyanotic Atrial septal defect Ventricular septal defect Patent foramen ovale Patent ductus arteriosus Aortic coartation Pulmonary stenosis Cyanotic Tetralogy
CONGENITAL HEART DISEASE Uptofate Study Summary Acyanotic Atrial septal defect Ventricular septal defect Patent foramen ovale Patent ductus arteriosus Aortic coartation Pulmonary stenosis Cyanotic Tetralogy
Case 47 Clinical Presentation
 93 Case 47 C Clinical Presentation 45-year-old man presents with chest pain and new onset of a murmur. Echocardiography shows severe aortic insufficiency. 94 RadCases Cardiac Imaging Imaging Findings C
93 Case 47 C Clinical Presentation 45-year-old man presents with chest pain and new onset of a murmur. Echocardiography shows severe aortic insufficiency. 94 RadCases Cardiac Imaging Imaging Findings C
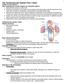
What Is Valvular Heart Disease? Heart valve disease occurs when your heart's valves do not work the way they should.
 What Is Valvular Heart Disease? Heart valve disease occurs when your heart's valves do not work the way they should. How Do Heart Valves Work? MAINTAIN ONE-WAY BLOOD FLOW THROUGH YOUR HEART The four heart
What Is Valvular Heart Disease? Heart valve disease occurs when your heart's valves do not work the way they should. How Do Heart Valves Work? MAINTAIN ONE-WAY BLOOD FLOW THROUGH YOUR HEART The four heart
Skin supplied by T1-4 (medial upper arm and neck) T5-9- epigastrium Visceral afferents from skin and heart are the same dorsal root ganglio
 Cardio 2 ECG... 3 Cardiac Remodelling... 11 Valvular Diseases... 13 Hypertension... 18 Aortic Coarctation... 24 Erythropoiesis... 27 Haemostasis... 30 Anaemia... 36 Atherosclerosis... 44 Angina... 48 Myocardial
Cardio 2 ECG... 3 Cardiac Remodelling... 11 Valvular Diseases... 13 Hypertension... 18 Aortic Coarctation... 24 Erythropoiesis... 27 Haemostasis... 30 Anaemia... 36 Atherosclerosis... 44 Angina... 48 Myocardial
Tricuspid and Pulmonary Valve Disease
 Tricuspid and Pulmonary Valve Disease Lawrence Rudski MD FRCPC FACC FASE Professor of Medicine Director, Division of Cardiology Jewish General Hospital McGill University Right Sided Failure Edema Gut congestion
Tricuspid and Pulmonary Valve Disease Lawrence Rudski MD FRCPC FACC FASE Professor of Medicine Director, Division of Cardiology Jewish General Hospital McGill University Right Sided Failure Edema Gut congestion
Adel Hasanin Ahmed 1
 Adel Hasanin Ahmed 1 PERICARDIAL DISEASE The pericardial effusion ends anteriorly to the descending aorta and is best visualised in the PLAX. PSAX is actually very useful sometimes for looking at posterior
Adel Hasanin Ahmed 1 PERICARDIAL DISEASE The pericardial effusion ends anteriorly to the descending aorta and is best visualised in the PLAX. PSAX is actually very useful sometimes for looking at posterior
For more information about how to cite these materials visit
 Author: Michael Shea, M.D., 2008 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Share Alike 3.0 License: http://creativecommons.org/licenses/by-sa/3.0/
Author: Michael Shea, M.D., 2008 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Share Alike 3.0 License: http://creativecommons.org/licenses/by-sa/3.0/
ASE 2011 Appropriate Use Criteria for Echocardiography
 ASE 2011 Appropriate Use Criteria for Echocardiography Table 1. TTE for General Evaluation of Cardiac Structure and Function 1 2 Suspected Cardiac Etiology General With TTE Symptoms or conditions potentially
ASE 2011 Appropriate Use Criteria for Echocardiography Table 1. TTE for General Evaluation of Cardiac Structure and Function 1 2 Suspected Cardiac Etiology General With TTE Symptoms or conditions potentially
Congestive Heart Failure or Heart Failure
 Congestive Heart Failure or Heart Failure Dr Hitesh Patel Ascot Cardiology Group Heart Failure Workshop April, 2014 Question One What is the difference between congestive heart failure and heart failure?
Congestive Heart Failure or Heart Failure Dr Hitesh Patel Ascot Cardiology Group Heart Failure Workshop April, 2014 Question One What is the difference between congestive heart failure and heart failure?
Pregnancy, Heart Disease and Imaging. Hemodynamics. Decreased systemic vascular resistance. Physiology anemia
 Pregnancy, Heart Disease and Imaging Sangeeta Shah, MD, FASE, FACC Associate Professor, Ochsner Clinical School of Medicine Advanced CV Imaging and Adult Congenital Heart Disease New Orleans, LA Hemodynamics
Pregnancy, Heart Disease and Imaging Sangeeta Shah, MD, FASE, FACC Associate Professor, Ochsner Clinical School of Medicine Advanced CV Imaging and Adult Congenital Heart Disease New Orleans, LA Hemodynamics
Mitral Valve Disorders
 Mitral Valve Disorders Echocardiography Findings and Assessment NEHOUA October 2013 Leominster, MA Adela de Loizaga, M.D. Proprietary Notice The material contained in this presentation has been prepared
Mitral Valve Disorders Echocardiography Findings and Assessment NEHOUA October 2013 Leominster, MA Adela de Loizaga, M.D. Proprietary Notice The material contained in this presentation has been prepared
