Pharmacy Medical Policy Bisphosphonates and Monoclonal Antibodies, Infusion/Injection
|
|
|
- Ernest Fields
- 6 years ago
- Views:
Transcription
1 Pharmacy Medical Policy Bisphosphonates and Monoclonal Antibodies, Infusion/Injection Table of Contents Policy: Commercial Policy History References Coding Information Information Pertaining to All Policies Forms Policy Number: 061 BCBSA Reference Number: None Related Policies None Policy Commercial Members: Managed Care (HMO and POS), PPO, and Indemnity Note: All requests for outpatient retail pharmacy for indications listed and not listed on the medical policy guidelines may be submitted to BCBSMA Clinical Pharmacy Operations by completing the Prior Authorization Form on the last page of this document. Physicians may also call BCBSMA Pharmacy Operations department at (800) to request a prior authorization/formulary exception verbally. Physicians may also submit requests for retail pharmacy exceptions via the web using Express PAth which can be found on the BCBSMA provider website or directly on the web at Patients must have pharmacy benefits under their subscriber certificates. Note: Reclast and Zometa are not covered under the retail pharmacy benefit and must be billed under the medical benefit. We cover Boniva (ibandronate) and Reclast (zoledronic acid) for a documented diagnosis of osteoporosis in women or men when one of the following criteria is met: Previous treatment failure with one or more oral bisphosphonates (alendronate, Fosamax, Actonel) resulting in intolerability to the oral product Inability to swallow Inability to remain in an upright position during post oral bisphosphonate administration. We cover Reclast (zoledronic acid) for a documented diagnosis of Paget s disease of the bone and for prevention of osteoporosis/osteopenia. We cover Prolia (denosumab) for a documented diagnosis of osteoporosis in postmenopausal women when one of the following criteria is met: Previous treatment failure with one or more oral bisphosphonates (alendronate, Fosamax, Actonel) resulting in intolerability to the oral product
2 Inability to swallow Inability to remain in an upright position during post oral bisphosphonate administration. We cover Prolia (denosumab) for the treatment of androgen deprivation-induced bone loss in men with prostate cancer. We cover Prolia (denosumab) for the treatment of aromatase inhibitor-induced bone loss in women with breast cancer. We cover Xgeva (denosumab) for the prevention of skeletal related events in bone metastases from solid tumors. We cover Zometa (zoledronic acid) for a documented diagnosis of any of the following: Hypercalcemia of malignancy Multiple myeloma Documented bone metastases from solid tumors. We do not cover the above drugs for other conditions not listed above. Other Information Blue Cross Blue Shield of Massachusetts (BCBSMA*) members (other than Medex ; Blue MedicareRx, Medicare Advantage plans that include prescription drug coverage) will be required to fill their prescriptions for the above medications at one of the providers in our retail specialty pharmacy network, as listed below: Retail Specialty Pharmacy Contact Information: AcariaHealth. Phone: Fax: Website: Accredo Health Group Phone: Fax: Website: Caremark, Inc. Phone: Fax: Website: Individual Consideration All our medical policies are written for the majority of people with a given condition. Each policy is based on medical science. For many of our medical policies, each individual s unique clinical circumstances may be considered in light of current scientific literature. Physicians may send relevant clinical information for individual patients for consideration to: Blue Cross Blue Shield of Massachusetts Pharmacy Operations Department One Enterprise Drive
3 Quincy, MA Tel: Fax: Managed Care Authorization Instructions Prior authorization is required for all out patient sites of service For retail pharmacy requests, physicians may call BCBSMA Pharmacy Operations department to request a review for prior authorization for patients. Pharmacy Operations: (800) For all outpatient sites of service, physicians may also fax or mail the attached form to the address above. The Formulary Exception/Prior Authorization form is included as part of this document for physicians to submit for patients. For all outpatient sites of service, physicians may also submit requests for retail pharmacy exceptions via the web using Express PAth which can be found on the BCBSMA provider website or directly on the web at PPO and Indemnity Authorization Instructions Prior authorization is required when these medications are processed under the retail pharmacy benefit and home infusion therapy benefit. Prior authorization is not required when these drugs are purchased by the physician and administered in the office in accordance with this medical policy. For retail pharmacy requests, physicians may call BCBSMA Pharmacy Operations department to request a review for prior authorization for patients. Pharmacy Operations: (800) Physicians may also fax or mail the attached form to the address above. The Formulary Exception/Prior Authorization form is included as part of this document for physicians to submit for patients. Physicians may also submit requests for retail pharmacy exceptions via the web using Express PAth which can be found on the BCBSMA provider website or directly on the web at CPT Codes / HCPCS Codes / ICD-9 Codes The following codes are included below for informational purposes. Inclusion or exclusion of a code does not constitute or imply member coverage or provider reimbursement. Please refer to the member s contract benefits in effect at the time of service to determine coverage or non-coverage as it applies to an individual member. A draft of future ICD-10 Coding related to this document, as it might look today, is included below for your reference. Providers should report all services using the most up-to-date industry-standard procedure, revenue, and diagnosis codes, including modifiers where applicable. CPT Codes There is no specific CPT code for this service. HCPCS Codes HCPCS codes: J0897 J1740 Diagnosis coding ICD-9-CM Code Description Injection, denosumab, 1 mg ((Prolia and Xgeva) Injection, ibandronate sodium, 1 mg. (Boniva) diagnosis codes: Code Description Secondary malignant neoplasm of bone and bone marrow Multiple myeloma, without mention of having achieved remission Multiple myeloma, in remission
4 Multiple myeloma, in relapse Hypercalcemia Osteitis deformans without mention of bone tumor Osteoporosis, unspecified Senile osteoporosis Idiopathic osteoporosis Disuse osteoporosis Other osteoporosis Disorder of bone and cartilage, unspecified ICD-10 Diagnosis Codes ICD-10-CM Diagnosis codes: Code Description C79.51 Secondary malignant neoplasm of bone C79.52 Secondary malignant neoplasm of bone marrow C90.00 Multiple myeloma not having achieved remission C90.01 Multiple myeloma in remission C90.02 Multiple myeloma in relapse E83.52 Hypercalcemia M81.0 Age-related osteoporosis without current pathological fracture M81.6 Localized osteoporosis [Lequesne] M81.8 Other osteoporosis without current pathological fracture M85.9 Disorder of bone density and structure, unspecified M88.0 Osteitis deformans of skull M88.1 Osteitis deformans of vertebrae M Osteitis deformans of right shoulder M Osteitis deformans of left shoulder M Osteitis deformans of unspecified shoulder M Osteitis deformans of right upper arm M Osteitis deformans of left upper arm M Osteitis deformans of unspecified upper arm M Osteitis deformans of right forearm M Osteitis deformans of left forearm M Osteitis deformans of unspecified forearm M Osteitis deformans of right hand M Osteitis deformans of left hand M Osteitis deformans of unspecified hand M Osteitis deformans of right thigh M Osteitis deformans of left thigh M Osteitis deformans of unspecified thigh M Osteitis deformans of right lower leg M Osteitis deformans of left lower leg M Osteitis deformans of unspecified lower leg M Osteitis deformans of right ankle and foot M Osteitis deformans of left ankle and foot M Osteitis deformans of unspecified ankle and foot M88.88 Osteitis deformans of other bones M88.89 Osteitis deformans of multiple sites M88.9 Osteitis deformans of unspecified bone Policy History Date Action
5 7/2014 Updated Coding section with ICD10 procedure and diagnosis codes, effective 10/ /2014 Removal of Curascripts from Specialty Pharmacy section. 1/2014 Updated to remove deleted HCPCS code J3487 and J /2013 Updated 1/2013 to include new FDA approved indications for Prolia. 11/2011-4/2012 Medical policy ICD 10 remediation: Formatting, editing and coding updates. No changes to policy statements. 1/2012 Updated to include MED UM requirements for Prolia and Xgeva. 7/2011 Updated to include coverage criteria for new FDA approved medication Xgeva. 5/2011 Updated to include coverage criteria for FDA approved indication of osteopenia for Reclast. 1/1/2011 Updated to include coverage criteria for new FDA approved medication Prolia. 10/2009 Updated to reflect UM requirements. 1/1/2009 New policy, effective 1/1/2009, describing covered and non-covered indications. References 1. Boniva injection [package insert]. Nutley, NJ: Roche Laboratories; February Reclast injection [package insert[. East Hanover, NJ: Novartis; August Zometa injection [package insert]. East Hanover, NJ: Novartis; September Reid IR, Miller P, Lyles K, et al. Comparison of a single infusion of zoledronic acid with risedronate for Paget s disease. N Engl J Med (9): Management of osteoporosis in postmenopausal women: 2006 position statement of the North American Menopause Society. Menopause. 2006;13: National Osteoporosis Foundation. Physician s guide to prevention and treatment of osteoporosis. Pharmacologic Options. Washington, D.C.; Updated September Devogelaer JP, Goemaere S, Boonen S, et al. Evidence-based guidelines for the prevention and treatment of glucocorticoid-induced osteoporosis: a consensus document by the Belgian Bone Club. Osteoporosis Int. 2006;17: Whyte MP. Paget s disease of bone. N Engl J Med. 2006;355: Siris ES, Lyles KW, Singer FR, Meunier PJ. Medical management of Paget s disease of bone: indications for treatment and review of current therapies. J Bone Miner Res. 2006;21 Suppl 2:P94-P Black DM, Delmas PD, Eastell R, et al, for the HIZON Pivotal Fracture Trial. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med. 2007;356(18): Lyles KW, Colon-Emeric CS, Magaziner JS, et al, for the HIZON Recurrent Fracture Trial. Zoledronic acid and clinical fractures and mortality after hip surgery. N Engl J Med. 2007;357(18): McClung M, Recker R, Miller P, et al. Intravenous zoledronic acid 5 mg in the treatment of postmenopausal women with low bone density previously treated with alendronate. Bone. 2007;41: Hosking D, Lyles K, Brown JP, et al. Long-term control of bone turnover in Paget s disease with zoledronic acid and risedronate. J Bone Miner Res. 2007;22(1): Delmas PD, Adami S, Strugala C, et al. Intravenous ibandronate injections in postmenopausal women with osteoporosis. One-year results from the dosing intravenous administration study. Arthritis Rheum. 2006;54(6): Ringe JD, Dorst A, Faber H, et al. Intermittent intravenous ibandronate injections reduce vertebral fractures risk in corticosteroid-induced osteoporosis: results from a long-term comparative study. Osteoporosis Int. 2003;14(10): Lamy O, Sandini L, Pache I, et al. Intravenous ibandronate in men with osteoporosis: an open pilot study over 2 years. J Endocrinol Invest. 2003;26(8): Emkey RD, Ettinger M. Improving compliance and persistence with bisphosphonate therapy for osteoporosis. Am J Med. 2006;119(4A):18S-24S. 18. Cooper A, Drake J, Brankin E, on behalf of the persist investigators. Treatment persistence with oncemonthly ibandronate and patient support vs. once-weekly alendronate: results form the PERSIST study. Int J Clin Pract. 2006;60(8): Aredia injection [package insert]. East Hanover, NJ: Novartis Pharmaceuticals; Michaelson MD, Kaufman DS, Lee H, et al. Randomized controlled trial of annual zoledronic acid to prevent gonadotropin-releasing hormone agonist-induced bone loss in men with prostate cancer. J Clin Oncol. 2007;25(9):
6 21. Brufsky A, Harker WG, Beck JT, et al. Zoledronic acid inhibits adjuvant letrozole-induced bone loss in postmenopausal women with early breast cancer. J Clin Oncol. 2007;25(7): Gnant MF, Mlineritsch B, Luschin-Ebengreuth G, et al. Zoledronic acid prevents cancer treatment-induced bone loss in premenopausal women receiving adjuvant endocrine therapy for hormone-responsive breast cancer: a report from the Austrian Breast and Colorectal Cancer Study Group. J Clin Oncol. 2007;25(7): D Souza AB, Grigg AP, Szer J, Ebeling PR. Zoledronic acid prevents bone loss after allogeneic haemopoietic stem cell transplantation. Intern Med J. 2006;36(9): Ringe JD, Farahmand P. Advances in the management of corticosteroid-induced osteoporosis with bisphosphonates. Clin Rheumatol. 2007;26(4): Keating GM, Scott LJ. Zoledronic acid: a review of its use in the treatment of Paget s disease of bone. Drugs. 2007;67(5): Guay DR. Ibandronate, an experimental intravenous bisphosphonate for osteoporosis, bone metastases, and hypercalcemia of malignancy. Pharmacotherapy. 2006;26(5): Kyle RA, Yee GC, Somerfield MR, et al. American Society of Clinical Oncology 2007 clinical practice guidelines update on the role of bisphosphonates in multiple myeloma. J Clin Oncol. 2007;25(17): Prolia injection [package insert]. Thousand Oaks, CA: Amgen Manufacturing Limited; June Xgeva [package insert]. Thousand Oaks, CA: Amgen Manufacturing Limited; November 2010.
7 Request for Outpatient Retail Pharmacy Prior Authorization Fax to: Clinical Pharmacy Program (800) Phone Authorization (800) or Web: We plan to respond to your request within two business days of our receipt. To ensure that we can confirm your request (required by NCQA), please be sure to include your fax number. We cannot process requests unless they contain all of the information requested below: Patient Information (REQUIRED) Name BCBSMA ID Number Is the patient a BCBSMA employee? If yes, please fax request to: (617) Date of Birth Patient s Diagnosis or ICD-9-CM code Physician Information (REQUIRED) Name Medical Specialty BCBSMA Provider number/npi number Telephone Number Fax Number Is this fax number secure for PHI receipt/transmission per HIPAA requirements? (circle one) Yes Contact Name (if different from physician) Please select one of the three following sections to complete, depending on the nature of your request for the above-named patient. Formulary Exception Request Name of non-covered drug you want to prescribe Reason for Individual Consideration Request (please check one): Treatment failure with the following covered drugs in class Documented adverse reaction to the following covered drugs Other clinical reason (please specify) Quality Care Dosing Override Request Drug name, strength and quantity requested: Clinical reason for override (please specify) Outpatient Retail Pharmacy Prior Authorization Request Drug name: Start/End date (must be one year or less): Associated Co-morbid diagnosis: MD Signature: Date: Yes No No
8 Servicing Provider Phone# Fax# Outpatient Medical Prior Authorization Form Please complete and fax to: (888) Please contact Pharmacy Operations with questions at (800) If the patient is a BCBSMA employee, please fax the form to: (617) Name: NPI Number: Requesting Provider Phone# Fax# Name: NPI Number: Contact Person: Contact Person: Patient Name: DOB / / Diagnosis: Patient BCBSMA ID# Is this fax number secure for PHI receipt/transmission per HIPAA requirements? (circle one) Yes No Drug/Therapy: Drug Name: Dates of Service: / / to / / Dose: Frequency: Additional Clinical (including previous treatment failure): Physician signature: Date: 1
Pharmacy Medical Policy IgE Receptor Binding Inhibitors
 Pharmacy Medical Policy IgE Receptor Binding Inhibitors Table of Contents Policy: Commercial Policy History Endnotes Policy: Medicare Information Pertaining to All Policies Forms Coding Information References
Pharmacy Medical Policy IgE Receptor Binding Inhibitors Table of Contents Policy: Commercial Policy History Endnotes Policy: Medicare Information Pertaining to All Policies Forms Coding Information References
Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management
 Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Policy: Medicare References
Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Policy: Medicare References
Pharmacy Medical Policy Natalizumab (Tysabri )
 Pharmacy Medical Policy Natalizumab (Tysabri ) Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Coding Information References Forms Policy History Policy Number: 062
Pharmacy Medical Policy Natalizumab (Tysabri ) Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Coding Information References Forms Policy History Policy Number: 062
FYI ONLY Generic Name. Generics available. zoledronic acid N/A
 Criteria Document: Reference #: PC/A011 Page 1 of 5 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services, Inc. (PAS) Non-ERISA PreferredOne Community
Criteria Document: Reference #: PC/A011 Page 1 of 5 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services, Inc. (PAS) Non-ERISA PreferredOne Community
AETNA BETTER HEALTH Prior Authorization guideline for Injectable Osteoporosis Agents
 AETNA BETTER HEALTH Prior Authorization guideline for Injectable Osteoporosis Agents Injectable Osteoporosis Agents Forteo (teriparatide); zoledronic acid Prolia (denosumab)] Authorization guidelines For
AETNA BETTER HEALTH Prior Authorization guideline for Injectable Osteoporosis Agents Injectable Osteoporosis Agents Forteo (teriparatide); zoledronic acid Prolia (denosumab)] Authorization guidelines For
Name of Policy: Zoledronic Acid (Reclast ) Injection
 Name of Policy: Zoledronic Acid (Reclast ) Injection Policy #: 355 Latest Review Date: May 2011 Category: Pharmacy Policy Grade: Active Policy but no longer scheduled for regular literature reviews and
Name of Policy: Zoledronic Acid (Reclast ) Injection Policy #: 355 Latest Review Date: May 2011 Category: Pharmacy Policy Grade: Active Policy but no longer scheduled for regular literature reviews and
Pharmacy Management Drug Policy
 SUBJECT: - Forteo (teriparatide), Prolia (denosumab), Tymlos (abaloparatide) POLICY NUMBER: Pharmacy-35 EFFECTIVE DATE: 9/07 LAST REVIEW DATE: 9/29/2017 If the member s subscriber contract excludes coverage
SUBJECT: - Forteo (teriparatide), Prolia (denosumab), Tymlos (abaloparatide) POLICY NUMBER: Pharmacy-35 EFFECTIVE DATE: 9/07 LAST REVIEW DATE: 9/29/2017 If the member s subscriber contract excludes coverage
[If no, skip to question 10.] Y N. 2. Does the member have a diagnosis of Paget s disease of bone? Y N. [If no, skip to question 4.
![[If no, skip to question 10.] Y N. 2. Does the member have a diagnosis of Paget s disease of bone? Y N. [If no, skip to question 4. [If no, skip to question 10.] Y N. 2. Does the member have a diagnosis of Paget s disease of bone? Y N. [If no, skip to question 4.](/thumbs/86/94943423.jpg) Pharmacy Prior Authorization AETA BETTER HEALTH EW JERSE (MEDICAID) Zoledronic Acid (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
Pharmacy Prior Authorization AETA BETTER HEALTH EW JERSE (MEDICAID) Zoledronic Acid (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
Pharmacy Management Drug Policy
 SUBJECT: - Forteo (teriparatide), Prolia (denosumab), Tymlos (abaloparatide), Boniva injection (Ibandronate) POLICY NUMBER: Pharmacy-35 EFFECTIVE DATE: 9/07 LAST REVIEW DATE: 10/15/2018 If the member s
SUBJECT: - Forteo (teriparatide), Prolia (denosumab), Tymlos (abaloparatide), Boniva injection (Ibandronate) POLICY NUMBER: Pharmacy-35 EFFECTIVE DATE: 9/07 LAST REVIEW DATE: 10/15/2018 If the member s
3. Has bone specific alkaline phosphatase level increased OR does the member have symptoms related to active Paget s?
 Pharmacy Prior Authorization AETA BETTER HEALTH VIRGIIA CCC PLUS and MEDALLIO/FAMIS 4.0 Zoledronic Acid (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Pharmacy Prior Authorization AETA BETTER HEALTH VIRGIIA CCC PLUS and MEDALLIO/FAMIS 4.0 Zoledronic Acid (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
HARVARD PILGRIM HEALTH CARE RECOMMENDED MEDICATION REQUEST GUIDELINES
 Generic Brand HICL GCN Exception/Other PROLIA, XGEVA 37012 If the caller wishes to initiate a request then a MRF must be completed. This drug requires a written request for prior authorization. All requests
Generic Brand HICL GCN Exception/Other PROLIA, XGEVA 37012 If the caller wishes to initiate a request then a MRF must be completed. This drug requires a written request for prior authorization. All requests
Osteoporosis Agents Drug Class Prior Authorization Protocol
 Osteoporosis Agents Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review of
Osteoporosis Agents Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review of
Prior Authorization Required: Yes as shown below
 PROLIA, XGEVA (denosumab) MB9409 Covered Service: Prior Authorization Required: Additional Information Medicare Policy: BadgerCare Plus Policy: Yes when meets criteria below Yes as shown below Must be
PROLIA, XGEVA (denosumab) MB9409 Covered Service: Prior Authorization Required: Additional Information Medicare Policy: BadgerCare Plus Policy: Yes when meets criteria below Yes as shown below Must be
Pharmacy Management Drug Policy
 Clinical criteria used to make utilization review decisions are based on credible scientific evidence published in peer reviewed medical literature generally recognized by the medical community. Guidelines
Clinical criteria used to make utilization review decisions are based on credible scientific evidence published in peer reviewed medical literature generally recognized by the medical community. Guidelines
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: denosumab_prolia_xgeva 3/2011 9/2017 9/2018 9/2017 Description of Procedure or Service Receptor activator
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: denosumab_prolia_xgeva 3/2011 9/2017 9/2018 9/2017 Description of Procedure or Service Receptor activator
PROLIA: Medical Coverage Policy Denosumab (Prolia and. Xgeva) EFFECTIVE DATE: POLICY LAST UPDATED:
 Medical Coverage Policy Denosumab (Prolia and Xgeva) EFFECTIVE DATE: 11 01 2016 POLICY LAST UPDATED: 12 19 2017 OVERVIEW Prolia (denosumab) is indicated for the treatment of postmenopausal women with osteoporosis
Medical Coverage Policy Denosumab (Prolia and Xgeva) EFFECTIVE DATE: 11 01 2016 POLICY LAST UPDATED: 12 19 2017 OVERVIEW Prolia (denosumab) is indicated for the treatment of postmenopausal women with osteoporosis
DENOSUMAB (PROLIA & XGEVA )
 DENOSUMAB (PROLIA & XGEVA ) UnitedHealthcare Oxford Clinical Policy Policy Number: PHARMACY 306.3 T2 Effective Date: July 2, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
DENOSUMAB (PROLIA & XGEVA ) UnitedHealthcare Oxford Clinical Policy Policy Number: PHARMACY 306.3 T2 Effective Date: July 2, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
Pharmacy Medical Policy Angiotensin II Receptor Antagonists
 Pharmacy Medical Policy Angiotensin II Receptor Antagonists Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Policy: Medicare References Forms Policy History Policy
Pharmacy Medical Policy Angiotensin II Receptor Antagonists Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Policy: Medicare References Forms Policy History Policy
Approval of a drug under this criteria document does not ensure full coverage of the drug.
 Criteria Document: Reference #: PC/A011 Page 1 of 8 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services, Inc. (PAS) Non-ERISA PreferredOne Community
Criteria Document: Reference #: PC/A011 Page 1 of 8 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services, Inc. (PAS) Non-ERISA PreferredOne Community
This Coverage Policy applies to Individual Health Insurance Marketplace benefit plans only.
 This Coverage Policy applies to Individual Health Insurance Marketplace benefit plans only. INJECTABLE OSTEOPOSIS AGENTS SUBJECT Pharmacologic Agents: Bisphosphonates: Boniva IV (ibandronate) Reclast (zoledronic
This Coverage Policy applies to Individual Health Insurance Marketplace benefit plans only. INJECTABLE OSTEOPOSIS AGENTS SUBJECT Pharmacologic Agents: Bisphosphonates: Boniva IV (ibandronate) Reclast (zoledronic
Pharmacy Medical Policy Alglucerase (Ceredase and Cerezyme ) for Gaucher Disease
 Pharmacy Medical Policy Alglucerase (Ceredase and Cerezyme ) for Gaucher Disease Table of Contents Policy: Commercial Policy History Endnotes Policy: Medicare Information Pertaining to All Policies Forms
Pharmacy Medical Policy Alglucerase (Ceredase and Cerezyme ) for Gaucher Disease Table of Contents Policy: Commercial Policy History Endnotes Policy: Medicare Information Pertaining to All Policies Forms
Xgeva. Xgeva (denosumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.18 Subject: Xgeva Page: 1 of 5 Last Review Date: March 16, 2018 Xgeva Description Xgeva (denosumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.18 Subject: Xgeva Page: 1 of 5 Last Review Date: March 16, 2018 Xgeva Description Xgeva (denosumab)
Xgeva. Xgeva (denosumab) Description. Section: Prescription Drugs Effective Date: January 1, 2016
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.18 Subject: Xgeva Page: 1 of 5 Last Review Date: December 3, 2015 Xgeva Description Xgeva (denosumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.18 Subject: Xgeva Page: 1 of 5 Last Review Date: December 3, 2015 Xgeva Description Xgeva (denosumab)
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Prolia, Xgeva) Reference Number: CP.PHAR.58 Effective Date: 03.01.11 Last Review Date: 02.19 Line of Business: Commercial, HIM, Medicaid Coding Implications Revision Log See Important
Clinical Policy: (Prolia, Xgeva) Reference Number: CP.PHAR.58 Effective Date: 03.01.11 Last Review Date: 02.19 Line of Business: Commercial, HIM, Medicaid Coding Implications Revision Log See Important
Bisphosphonates for Injection* Effective Date: July 28, 2009
 Subject: Bisphosphonates for Injection* Effective Date: July 28, 2009 Department(s): Utilization Management Policy: Objective: Procedure: The use of injectable bisphosphonates is reimbursable for Plans
Subject: Bisphosphonates for Injection* Effective Date: July 28, 2009 Department(s): Utilization Management Policy: Objective: Procedure: The use of injectable bisphosphonates is reimbursable for Plans
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Prolia, Xgeva) Reference Number: CP.PHAR.58 Effective Date: 03.01.11 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder
Clinical Policy: (Prolia, Xgeva) Reference Number: CP.PHAR.58 Effective Date: 03.01.11 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder
Bisphosphonate Step Therapy Criteria
 ϯ ϯ ϯ A Division of Health Care Service Corporation, a Mutual Legal Reserve Company, an Independent Licensee of the Blue Cross and Blue Shield Association Bisphosphonate Step Therapy Criteria Program may
ϯ ϯ ϯ A Division of Health Care Service Corporation, a Mutual Legal Reserve Company, an Independent Licensee of the Blue Cross and Blue Shield Association Bisphosphonate Step Therapy Criteria Program may
Medical Policy. MP Specialty Drugs. Related Policies Guidelines for Prior Authorization of Pharmacologic Therapies
 Medical Policy Last Review: 04/30/2018 Effective Date: 07/01/2018 Section: Prescription Drug Related Policies 5.01.501 Guidelines for Prior Authorization of Pharmacologic Therapies DISCLAIMER Our medical
Medical Policy Last Review: 04/30/2018 Effective Date: 07/01/2018 Section: Prescription Drug Related Policies 5.01.501 Guidelines for Prior Authorization of Pharmacologic Therapies DISCLAIMER Our medical
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Reclast, Zometa) Reference Number: CP.PHAR.59 Effective Date: 03.11 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder
Clinical Policy: (Reclast, Zometa) Reference Number: CP.PHAR.59 Effective Date: 03.11 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder
4.7 Studies of Quality Holy Cross Hospital Bone Health Early Stage I ER/PR Positive Breast Cancer Patients December 13, 2017
 4.7 Studies of Quality Holy Cross Hospital 2017 Bone Health Early Stage I ER/PR Positive Breast Cancer Patients December 13, 2017 Bone Health in Stage I ER/PR Positive Breast Cancer Patients To review
4.7 Studies of Quality Holy Cross Hospital 2017 Bone Health Early Stage I ER/PR Positive Breast Cancer Patients December 13, 2017 Bone Health in Stage I ER/PR Positive Breast Cancer Patients To review
Name of Policy: Boniva (Ibandronate Sodium) Infusion
 Name of Policy: Boniva (Ibandronate Sodium) Infusion Policy #: 266 Latest Review Date: April 2010 Category: Pharmacology Policy Grade: Active Policy but no longer scheduled for regular literature reviews
Name of Policy: Boniva (Ibandronate Sodium) Infusion Policy #: 266 Latest Review Date: April 2010 Category: Pharmacology Policy Grade: Active Policy but no longer scheduled for regular literature reviews
denosumab (Prolia ) Policy # Original Effective Date: 07/21/2011 Current Effective Date: 04/19/2017
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Based on review of available data, the Company may consider the use of denosumab (Prolia) for the
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Horizon Scanning Technology Briefing. Zoledronic Acid (Aclasta) once yearly treatment for postmenopausal. National Horizon Scanning Centre
 Horizon Scanning Technology Briefing National Horizon Scanning Centre Zoledronic Acid (Aclasta) once yearly treatment for postmenopausal osteoporosis December 2006 This technology summary is based on information
Horizon Scanning Technology Briefing National Horizon Scanning Centre Zoledronic Acid (Aclasta) once yearly treatment for postmenopausal osteoporosis December 2006 This technology summary is based on information
Parathyroid Hormone Analog for Osteoporosis Prior Authorization with Quantity Limit Criteria Program Summary
 Parathyroid Hormone Analog for Osteoporosis Prior Authorization with Quantity Limit Criteria Program Summary This prior authorization program applies to Commercial, NetResults A series, NetResults F series
Parathyroid Hormone Analog for Osteoporosis Prior Authorization with Quantity Limit Criteria Program Summary This prior authorization program applies to Commercial, NetResults A series, NetResults F series
Medication Prior Authorization Form
 Reclast (Zoledronic Acid) Policy Number: 1050 Policy History Approve Date: 12/11/2015 Revise Dates: 8/22/2016 Next Review: 12/11/2016 Review Dates: Preauthorization All Plans Benefit plans vary in coverage
Reclast (Zoledronic Acid) Policy Number: 1050 Policy History Approve Date: 12/11/2015 Revise Dates: 8/22/2016 Next Review: 12/11/2016 Review Dates: Preauthorization All Plans Benefit plans vary in coverage
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Actonel, Atelvia) Reference Number: CP.PMN.100 Effective Date: 03.01.18 Last Review Date: 02.19 Line of Business: Commercial, HIM, Medicaid Revision Log See Important Reminder at the
Clinical Policy: (Actonel, Atelvia) Reference Number: CP.PMN.100 Effective Date: 03.01.18 Last Review Date: 02.19 Line of Business: Commercial, HIM, Medicaid Revision Log See Important Reminder at the
Bisphosphonate treatment break
 Bulletin 110 December 2015 Bisphosphonate treatment break Bisphosphonates have been widely used in the treatment of osteoporosis with robust data demonstrating efficacy in fracture risk reduction over
Bulletin 110 December 2015 Bisphosphonate treatment break Bisphosphonates have been widely used in the treatment of osteoporosis with robust data demonstrating efficacy in fracture risk reduction over
Prolia /Xgeva (denosumab) Document Number: IC-0098
 / (denosumab) Document Number: IC-0098 Last Review Date: 5/30/2017 Date of Origin: 11/28/2011 Dates Reviewed: 12/2011, 03/2012, 06/2012, 09/2012, 12/2012, 03/2013, 06/2013, 03/2014, 06/2014, 09/2014, 12/2014,
/ (denosumab) Document Number: IC-0098 Last Review Date: 5/30/2017 Date of Origin: 11/28/2011 Dates Reviewed: 12/2011, 03/2012, 06/2012, 09/2012, 12/2012, 03/2013, 06/2013, 03/2014, 06/2014, 09/2014, 12/2014,
Clinical Policy: Denosumab (Prolia, Xgeva) Reference Number: CP.PHAR.58
 Clinical Policy: (Prolia, Xgeva) Reference Number: CP.PHAR.58 Effective Date: 03/11 Last Review Date: 08/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Prolia, Xgeva) Reference Number: CP.PHAR.58 Effective Date: 03/11 Last Review Date: 08/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important
Questions and Answers About Breast Cancer, Bone Metastases, & Treatment-Related Bone Loss. A Publication of The Bone and Cancer Foundation
 Questions and Answers About Breast Cancer, Bone Metastases, & Treatment-Related Bone Loss A Publication of The Bone and Cancer Foundation Contents This publication includes important information about
Questions and Answers About Breast Cancer, Bone Metastases, & Treatment-Related Bone Loss A Publication of The Bone and Cancer Foundation Contents This publication includes important information about
BONIVA (ibandronate sodium)
 BONIVA (ibandronate sodium) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices
BONIVA (ibandronate sodium) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices
Subject: Denosumab (Prolia ; Xgeva ) Injection
 09-J1000-25 Original Effective Date: 08/15/10 Reviewed: 03/14/18 Revised: 12/15/18 Next Review: 01/09/19 Subject: Denosumab (Prolia ; Xgeva ) Injection THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION,
09-J1000-25 Original Effective Date: 08/15/10 Reviewed: 03/14/18 Revised: 12/15/18 Next Review: 01/09/19 Subject: Denosumab (Prolia ; Xgeva ) Injection THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION,
Medication Policy Manual. Topic: Prolia, denosumab Date of Origin: August 11, 2010
 Independent licensees of the Blue Cross and Blue Shield Association Medication Policy Manual Policy No: dru223 Topic: Prolia, denosumab Date of Origin: August 11, 2010 Committee Approval Date: August 11,
Independent licensees of the Blue Cross and Blue Shield Association Medication Policy Manual Policy No: dru223 Topic: Prolia, denosumab Date of Origin: August 11, 2010 Committee Approval Date: August 11,
Contents Please refer to Medical Policy I-40 Pertuzumab (Perjeta) for additional information.
 May 2018 In This Issue Coverage Guidelines Revised for Azacitidine (Vidaza)... 3 Coverage Guidelines Revised for Fulvestrant (Faslodex)... 4 Place of Service Revised for Total Hip and Total Knee Arthroplasty...
May 2018 In This Issue Coverage Guidelines Revised for Azacitidine (Vidaza)... 3 Coverage Guidelines Revised for Fulvestrant (Faslodex)... 4 Place of Service Revised for Total Hip and Total Knee Arthroplasty...
Forteo (teriparatide) Prior Authorization Program Summary
 Forteo (teriparatide) Prior Authorization Program Summary FDA APPROVED INDICATIONS DOSAGE 1 FDA Indication 1 : Forteo (teriparatide) is indicated for: the treatment of postmenopausal women with osteoporosis
Forteo (teriparatide) Prior Authorization Program Summary FDA APPROVED INDICATIONS DOSAGE 1 FDA Indication 1 : Forteo (teriparatide) is indicated for: the treatment of postmenopausal women with osteoporosis
Collagen Crosslinks, Any Method
 190.19 - Collagen Crosslinks, Any Method Collagen crosslinks, part of the matrix of bone upon which bone mineral is deposited, are biochemical markers the excretion of which provides a quantitative measurement
190.19 - Collagen Crosslinks, Any Method Collagen crosslinks, part of the matrix of bone upon which bone mineral is deposited, are biochemical markers the excretion of which provides a quantitative measurement
Medication Policy Manual. Topic: Prolia, Xgeva, denosumab Date of Origin: August 11, 2010
 Medication Policy Manual Policy No: dru223 Topic: Prolia, Xgeva, denosumab Date of Origin: August 11, 2010 Committee Approval Date: June 20, 2014 Next Review Date: June 2015 Effective Date: July 1, 2014
Medication Policy Manual Policy No: dru223 Topic: Prolia, Xgeva, denosumab Date of Origin: August 11, 2010 Committee Approval Date: June 20, 2014 Next Review Date: June 2015 Effective Date: July 1, 2014
Bisphosphonates in the Management of. Myeloma Bone Disease
 Bisphosphonates in the Management of Myeloma Bone Disease James R. Berenson, MD Medical & Scientific Director Institute for Myeloma & Bone Cancer Research Los Angeles, CA Myeloma Bone Disease Myeloma cells
Bisphosphonates in the Management of Myeloma Bone Disease James R. Berenson, MD Medical & Scientific Director Institute for Myeloma & Bone Cancer Research Los Angeles, CA Myeloma Bone Disease Myeloma cells
Parathyroid Hormone Analogs
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.36 Subject: Parathyroid Hormone Analogs Page: 1 of 6 Last Review Date: September 15, 2017 Parathyroid
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.36 Subject: Parathyroid Hormone Analogs Page: 1 of 6 Last Review Date: September 15, 2017 Parathyroid
CASE 1 WHY IS IT IMPORTANT TO TREAT? FACTS CONCERNS
 4:30-5:15pm Ask the Expert: Osteoporosis SPEAKERS Silvina Levis, MD OSTEOPOROSIS - FACTS 1:3 older women and 1:5 older men will have a fragility fracture after age 50 After 3 years of treatment, depending
4:30-5:15pm Ask the Expert: Osteoporosis SPEAKERS Silvina Levis, MD OSTEOPOROSIS - FACTS 1:3 older women and 1:5 older men will have a fragility fracture after age 50 After 3 years of treatment, depending
CLINICAL MEDICAL POLICY
 CLINICAL MEDICAL POLICY Policy Name: Faslodex (fulvestrant) Policy Number: MP-044-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Original Effective
CLINICAL MEDICAL POLICY Policy Name: Faslodex (fulvestrant) Policy Number: MP-044-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Original Effective
BREAST CANCER AND BONE HEALTH
 BREAST CANCER AND BONE HEALTH Rowena Ridout, MD, FRCPC Toronto Western Hospital Osteoporosis Program University Health Network / Mount Sinai Hospital rowena.ridout@uhn.ca None to declare Conflicts of Interest
BREAST CANCER AND BONE HEALTH Rowena Ridout, MD, FRCPC Toronto Western Hospital Osteoporosis Program University Health Network / Mount Sinai Hospital rowena.ridout@uhn.ca None to declare Conflicts of Interest
Collagen Crosslinks, Any Method
 190.19 - Collagen Crosslinks, Any Method Collagen crosslinks, part of the matrix of bone upon which bone mineral is deposited, are biochemical markers the excretion of which provides a quantitative measurement
190.19 - Collagen Crosslinks, Any Method Collagen crosslinks, part of the matrix of bone upon which bone mineral is deposited, are biochemical markers the excretion of which provides a quantitative measurement
Vol. 19, Bulletin No. 108 August-September 2012 Also in the Bulletin: Denosumab 120mg for Bone Metastases
 ה מ ר א פ הביטאון לענייני תרופות ISRAEL DRUG BULLETIN 19 years of unbiased and independent drug information P H A R x M A Vol. 19, Bulletin No. 108 August-September 2012 Also in the Bulletin: Denosumab
ה מ ר א פ הביטאון לענייני תרופות ISRAEL DRUG BULLETIN 19 years of unbiased and independent drug information P H A R x M A Vol. 19, Bulletin No. 108 August-September 2012 Also in the Bulletin: Denosumab
Tymlos (abaloparatide)
 Tymlos (abaloparatide) Policy Number: 5.01.638 Last Review: 11/2018 Origination: 10/2017 Next Review: 11/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Tymlos
Tymlos (abaloparatide) Policy Number: 5.01.638 Last Review: 11/2018 Origination: 10/2017 Next Review: 11/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Tymlos
Osteoporosis: Are your bones at risk of fracturing? Rachel Wallwork, MD Internal medicine resident Massachusetts General Hospital
 Osteoporosis: Are your bones at risk of fracturing? Rachel Wallwork, MD Internal medicine resident Massachusetts General Hospital What is Osteoporosis? Osteoporosis causes bones to lose density, become
Osteoporosis: Are your bones at risk of fracturing? Rachel Wallwork, MD Internal medicine resident Massachusetts General Hospital What is Osteoporosis? Osteoporosis causes bones to lose density, become
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Reference Number: HIM.PA.51 Effective Date: 12/14 Last Review Date: 08/17 Line of Business: Health Insurance Marketplace Revision Log See Important Reminder at the end of this policy for
Clinical Policy: Reference Number: HIM.PA.51 Effective Date: 12/14 Last Review Date: 08/17 Line of Business: Health Insurance Marketplace Revision Log See Important Reminder at the end of this policy for
MEDICAL POLICY MEDICARE ADVANTAGE ONLY (See Product Variations)
 Original Issue Date (Created): 11/1/2010 Most Recent Review Date (Revised): 1/18/2018 Effective Date: 9/1/2018 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS
Original Issue Date (Created): 11/1/2010 Most Recent Review Date (Revised): 1/18/2018 Effective Date: 9/1/2018 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Forteo) Reference Number: CP.PHAR.188 Effective Date: 11.15.17 Last Review Date: 02.19 Line of Business: Commercial* (Exchange Plans), HIM, Medicaid Coding Implications Revision Log See
Clinical Policy: (Forteo) Reference Number: CP.PHAR.188 Effective Date: 11.15.17 Last Review Date: 02.19 Line of Business: Commercial* (Exchange Plans), HIM, Medicaid Coding Implications Revision Log See
What is Osteoporosis?
 What is Osteoporosis? 2000 NIH Definition A skeletal disorder characterized by compromised bone strength predisposing a person to an increased risk of fracture. Bone strength reflects the integration of
What is Osteoporosis? 2000 NIH Definition A skeletal disorder characterized by compromised bone strength predisposing a person to an increased risk of fracture. Bone strength reflects the integration of
John J. Wolf, DO Family Medicine
 John J. Wolf, DO Family Medicine Objectives: 1. Review incidence & Risk of Osteoporosis 2.Review indications for testing 3.Review current pharmacologic & Non pharmacologic Tx options 4.Understand & Utilize
John J. Wolf, DO Family Medicine Objectives: 1. Review incidence & Risk of Osteoporosis 2.Review indications for testing 3.Review current pharmacologic & Non pharmacologic Tx options 4.Understand & Utilize
OSTEOPOROSIS MEDICINES
 Bone Basics 2010. NOF. All rights reserved. National Osteoporosis Foundation 1150 17th Street, NW, Suite 850 Washington, DC 20036 (800) 223-9994 www.nof.org OSTEOPOROSIS MEDICINES Although there is no
Bone Basics 2010. NOF. All rights reserved. National Osteoporosis Foundation 1150 17th Street, NW, Suite 850 Washington, DC 20036 (800) 223-9994 www.nof.org OSTEOPOROSIS MEDICINES Although there is no
TYMLOS (abaloparatide)
 TYMLOS (abaloparatide) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
TYMLOS (abaloparatide) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Integrated care: guidance on fracture prevention in cancer-associated bone disease; treatment options
 Paris, November 1st 2016 Integrated care: guidance on fracture prevention in cancer-associated bone disease; treatment options René Rizzoli MD International Osteoporosis Foundation and Division of Bone
Paris, November 1st 2016 Integrated care: guidance on fracture prevention in cancer-associated bone disease; treatment options René Rizzoli MD International Osteoporosis Foundation and Division of Bone
Bisphosphonates. Making intelligent drug choices
 Making intelligent drug choices Bisphosphonates are a first choice for treating osteoporosis, according to Kedrin E. Van Steenwyk, DO, an obstetrician/gynecologist at Sycamore Women s Center, Miamisburg,
Making intelligent drug choices Bisphosphonates are a first choice for treating osteoporosis, according to Kedrin E. Van Steenwyk, DO, an obstetrician/gynecologist at Sycamore Women s Center, Miamisburg,
Hot Topics in Bone Disease in 2017: Building Better Bones Breaking News in Osteoporosis
 Hot Topics in Bone Disease in 2017: Building Better Bones Breaking News in Osteoporosis Aromatase Inhibitor-Induced Bone Loss in Early Breast Cancer Rachel Pessah-Pollack, M.D., F.A.C.E. Mount Sinai School
Hot Topics in Bone Disease in 2017: Building Better Bones Breaking News in Osteoporosis Aromatase Inhibitor-Induced Bone Loss in Early Breast Cancer Rachel Pessah-Pollack, M.D., F.A.C.E. Mount Sinai School
Bad to the bones: treatments for breast and prostate cancer
 12 th Annual Osteoporosis: New Insights in Research, Diagnosis, and Clinical Care 23 rd July 2015 Bad to the bones: treatments for breast and prostate cancer Richard Eastell, MD FRCP (Lond, Edin, Ireland)
12 th Annual Osteoporosis: New Insights in Research, Diagnosis, and Clinical Care 23 rd July 2015 Bad to the bones: treatments for breast and prostate cancer Richard Eastell, MD FRCP (Lond, Edin, Ireland)
Breast Cancer and Bone Loss. One in seven women will develop breast cancer during a lifetime
 Breast Cancer and Bone Loss One in seven women will develop breast cancer during a lifetime Causes of Bone Loss in Breast Cancer Patients Aromatase inhibitors Bil Oophorectomy Hypogonadism Steroids Chemotherapy
Breast Cancer and Bone Loss One in seven women will develop breast cancer during a lifetime Causes of Bone Loss in Breast Cancer Patients Aromatase inhibitors Bil Oophorectomy Hypogonadism Steroids Chemotherapy
IBRANCE (palbociclib) oral capsule
 IBRANCE (palbociclib) oral capsule Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
IBRANCE (palbociclib) oral capsule Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
Horizon Scanning Centre March Denosumab for glucocorticoidinduced SUMMARY NIHR HSC ID: 6329
 Horizon Scanning Centre March 2014 Denosumab for glucocorticoidinduced osteoporosis SUMMARY NIHR HSC ID: 6329 This briefing is based on information available at the time of research and a limited literature
Horizon Scanning Centre March 2014 Denosumab for glucocorticoidinduced osteoporosis SUMMARY NIHR HSC ID: 6329 This briefing is based on information available at the time of research and a limited literature
Bone Mass Measurement BONE MASS MEASUREMENT HS-042. Policy Number: HS-042. Original Effective Date: 8/25/2008
 Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Questions and Answers About Breast Cancer, Bone Metastases, & Treatment-Related Bone Loss. A Publication of The Bone and Cancer Foundation
 Questions and Answers About Breast Cancer, Bone Metastases, & Treatment-Related Bone Loss A Publication of The Bone and Cancer Foundation Contents This publication includes important information about
Questions and Answers About Breast Cancer, Bone Metastases, & Treatment-Related Bone Loss A Publication of The Bone and Cancer Foundation Contents This publication includes important information about
Disclosures. Diagnostic Challenges in Osteoporosis: Whom To Treat 9/25/2014
 Disclosures Diagnostic Challenges in Osteoporosis: Whom To Treat Ethel S. Siris, MD Columbia University Medical Center New York, NY Consultant on scientific issues for: AgNovos Amgen Eli Lilly Merck Novartis
Disclosures Diagnostic Challenges in Osteoporosis: Whom To Treat Ethel S. Siris, MD Columbia University Medical Center New York, NY Consultant on scientific issues for: AgNovos Amgen Eli Lilly Merck Novartis
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 21 July 2010
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 21 July 2010 ACTONEL 5 mg, film-coated tablet B/14 (CIP code: 354 362-3) ACTONEL 30 mg, film-coated tablet B/28 (CIP
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 21 July 2010 ACTONEL 5 mg, film-coated tablet B/14 (CIP code: 354 362-3) ACTONEL 30 mg, film-coated tablet B/28 (CIP
2. Does the patient have a diagnosis of Crohn s disease? Y N
 Pharmacy Prior Authorization MERC CARE PLA (MEDICAID) Stelara (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE PLA (MEDICAID) Stelara (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Jaw Osteonecrosis Tied To Infusion Exposure.(Clinical Rounds): An Article From: Family Practice News [HTML] [Digital] By Kerri Wachter READ ONLINE
![Jaw Osteonecrosis Tied To Infusion Exposure.(Clinical Rounds): An Article From: Family Practice News [HTML] [Digital] By Kerri Wachter READ ONLINE Jaw Osteonecrosis Tied To Infusion Exposure.(Clinical Rounds): An Article From: Family Practice News [HTML] [Digital] By Kerri Wachter READ ONLINE](/thumbs/81/84618564.jpg) Jaw Osteonecrosis Tied To Infusion Exposure.(Clinical Rounds): An Article From: Family Practice News [HTML] [Digital] By Kerri Wachter READ ONLINE If you are searching for the book Jaw osteonecrosis tied
Jaw Osteonecrosis Tied To Infusion Exposure.(Clinical Rounds): An Article From: Family Practice News [HTML] [Digital] By Kerri Wachter READ ONLINE If you are searching for the book Jaw osteonecrosis tied
OSTEOPOROSIS MANAGEMENT AND INVESTIGATION. David A. Hanley, MD, FRCPC
 OSTEOPOROSIS MANAGEMENT AND INVESTIGATION David A. Hanley, MD, FRCPC There is a huge care gap in the management of osteoporosis in this country. As yet unpublished findings from the Canadian Multicentre
OSTEOPOROSIS MANAGEMENT AND INVESTIGATION David A. Hanley, MD, FRCPC There is a huge care gap in the management of osteoporosis in this country. As yet unpublished findings from the Canadian Multicentre
Prolia 2 shots a year proven to help strengthen bones.
 For women with postmenopausal osteoporosis at high risk for fracture: there s Prolia. Prolia 2 shots a year proven to help strengthen bones. Blythe Danner, Award winning actress taking Prolia. Prolia is
For women with postmenopausal osteoporosis at high risk for fracture: there s Prolia. Prolia 2 shots a year proven to help strengthen bones. Blythe Danner, Award winning actress taking Prolia. Prolia is
2. Is the patient responding to medication? Y N
 Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS ADD-ADHD Stimulants (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS ADD-ADHD Stimulants (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
New Developments in Oncology Bone Health. Learning Objectives. Disclosures 10/1/2014
 New Developments in Oncology Bone Health Kamakshi V. Rao, Pharm.D., BCPOP, CPP, FASHP Clinical Manager, Pharmacy Residency Programs Oncology and Bone Marrow Transplant Clinical Pharmacist University of
New Developments in Oncology Bone Health Kamakshi V. Rao, Pharm.D., BCPOP, CPP, FASHP Clinical Manager, Pharmacy Residency Programs Oncology and Bone Marrow Transplant Clinical Pharmacist University of
CLINICAL MEDICAL POLICY
 Policy Name: Policy Number: Approved By: Provider Notice Date: CLINICAL MEDICAL POLICY Portrazza (Necitumumab) MP-021-MD-WV Medical Management Original Effective Date: 06/02/2016 Annual Approval Date:
Policy Name: Policy Number: Approved By: Provider Notice Date: CLINICAL MEDICAL POLICY Portrazza (Necitumumab) MP-021-MD-WV Medical Management Original Effective Date: 06/02/2016 Annual Approval Date:
Breast Cancer and Bone Health. Robert Coleman, Cancer Research Centre, Weston Park Hospital, Sheffield
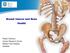 Breast Cancer and Bone Health Robert Coleman, Cancer Research Centre, Weston Park Hospital, Sheffield Breast Cancer and Bone Health Normal Bone Health Impact of Cancer Therapies on Bone Health Therapeutic
Breast Cancer and Bone Health Robert Coleman, Cancer Research Centre, Weston Park Hospital, Sheffield Breast Cancer and Bone Health Normal Bone Health Impact of Cancer Therapies on Bone Health Therapeutic
RETIRED. Maximum Drug Dose Policy
 RETIRED DRUG POLICY Policy Number 2017D0034A Maximum Drug Dose Policy Annual Approval Date 3/1/2017 Approved By UnitedHealthcare National Pharmacy & Therapeutics Committee United Healthcare Community Plan
RETIRED DRUG POLICY Policy Number 2017D0034A Maximum Drug Dose Policy Annual Approval Date 3/1/2017 Approved By UnitedHealthcare National Pharmacy & Therapeutics Committee United Healthcare Community Plan
Scottish Medicines Consortium
 Scottish Medicines Consortium zoledronic acid 5mg/100ml solution for infusion (Aclasta) No. (317/06) Novartis 8 September 2006 The Scottish Medicines Consortium (SMC) has completed its assessment of the
Scottish Medicines Consortium zoledronic acid 5mg/100ml solution for infusion (Aclasta) No. (317/06) Novartis 8 September 2006 The Scottish Medicines Consortium (SMC) has completed its assessment of the
Prolia 2 shots a year proven to help strengthen bones. next shot appointment
 Ask your doctor if Prolia (denosumab) is right for you and visit us at www.prolia.com next shot appointment For women with postmenopausal osteoporosis at high risk for fracture: there s Prolia. Prolia
Ask your doctor if Prolia (denosumab) is right for you and visit us at www.prolia.com next shot appointment For women with postmenopausal osteoporosis at high risk for fracture: there s Prolia. Prolia
NAMS Practice Pearl. Use of Drug Holidays in Women Taking Bisphosphonates. Released April 1, 2013
 NAMS Practice Pearl Use of Drug Holidays in Women Taking Bisphosphonates Released April 1, 2013 Dima L. Diab, MD 1, and Nelson B. Watts, MD 2 ( 1 Cincinnati VA Medical Center, Cincinnati, OH, 2 Mercy Health
NAMS Practice Pearl Use of Drug Holidays in Women Taking Bisphosphonates Released April 1, 2013 Dima L. Diab, MD 1, and Nelson B. Watts, MD 2 ( 1 Cincinnati VA Medical Center, Cincinnati, OH, 2 Mercy Health
To prevent bone loss and fractures in postmenopausal
 Online Exclusive Postmenopausal osteoporosis: Another approach to management The effectiveness of oral bisphosphonates is compromised by poor compliance. IV bisphosphonates provide another option. Practice
Online Exclusive Postmenopausal osteoporosis: Another approach to management The effectiveness of oral bisphosphonates is compromised by poor compliance. IV bisphosphonates provide another option. Practice
Medical Review. The following slides were medically reviewed by Dr. Nancy Dawson in June 2018.
 Bone Health Medical Review The following slides were medically reviewed by Dr. Nancy Dawson in June 2018. Presentation Overview 1. What is bone health? 2. How can cancer and cancer treatments affect your
Bone Health Medical Review The following slides were medically reviewed by Dr. Nancy Dawson in June 2018. Presentation Overview 1. What is bone health? 2. How can cancer and cancer treatments affect your
OSTEOPOROSIS IN MEN. Nelson B. Watts, MD OSTEOPOROSIS AND BONE HEALTH SERVICES CINCINNATI, OHIO
 OSTEOPOROSIS IN MEN Nelson B. Watts, MD OSTEOPOROSIS AND BONE HEALTH SERVICES CINCINNATI, OHIO DISCLOSURES Speakers Bureau: Amgen, Radius Consultant: Abbvie, Amgen, Janssen, Radius, Sanofi Watts NB et
OSTEOPOROSIS IN MEN Nelson B. Watts, MD OSTEOPOROSIS AND BONE HEALTH SERVICES CINCINNATI, OHIO DISCLOSURES Speakers Bureau: Amgen, Radius Consultant: Abbvie, Amgen, Janssen, Radius, Sanofi Watts NB et
Osteoporosis. Definition
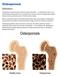 Osteoporosis Definition Osteoporosis causes bones to become weak and brittle so brittle that a fall or even mild stresses like bending over or coughing can cause a fracture. Osteoporosis-related fractures
Osteoporosis Definition Osteoporosis causes bones to become weak and brittle so brittle that a fall or even mild stresses like bending over or coughing can cause a fracture. Osteoporosis-related fractures
Osteoporosis. Overview
 v2 Osteoporosis Overview Osteoporosis is defined as compromised bone strength that increases risk of fracture (NIH Consensus Conference, 2000). Bone strength is characterized by bone mineral density (BMD)
v2 Osteoporosis Overview Osteoporosis is defined as compromised bone strength that increases risk of fracture (NIH Consensus Conference, 2000). Bone strength is characterized by bone mineral density (BMD)
ORILISSA (elagolix) oral tablet
 ORILISSA (elagolix) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
ORILISSA (elagolix) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
ERLEADA (apalutamide) oral tablet
 ERLEADA (apalutamide) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
ERLEADA (apalutamide) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
The Osteoporosis Center at St. Luke s Hospital
 The Osteoporosis Center at St. Luke s Hospital Desloge Outpatient Center (on the west side of 141) 121 St. Luke s Center Drive, Suite 504 Chesterfield, MO 63017 Phone 314 205-6633 Fax 314 590-5909 NEW
The Osteoporosis Center at St. Luke s Hospital Desloge Outpatient Center (on the west side of 141) 121 St. Luke s Center Drive, Suite 504 Chesterfield, MO 63017 Phone 314 205-6633 Fax 314 590-5909 NEW
1. UK List Price of Zoledronic acid (Zoledronate) 5 mg (Aclasta )
 Novartis Pharmaceuticals UK Ltd Frimley Business Park Frimley Camberley Surrey GU16 7SR Dr C M Longson Director, Centre for Health Technology Evaluation National Institute for Health and Clinical Excellence
Novartis Pharmaceuticals UK Ltd Frimley Business Park Frimley Camberley Surrey GU16 7SR Dr C M Longson Director, Centre for Health Technology Evaluation National Institute for Health and Clinical Excellence
Effective Health Care
 Number 12 Effective Health Care Comparative Effectiveness of Treatments To Prevent Fractures in Men and Women With Low Bone Density or Osteoporosis Executive Summary Background Osteoporosis is a systemic
Number 12 Effective Health Care Comparative Effectiveness of Treatments To Prevent Fractures in Men and Women With Low Bone Density or Osteoporosis Executive Summary Background Osteoporosis is a systemic
1
 www.osteoporosis.ca 1 2 Overview of the Presentation Osteoporosis: An Overview Bone Basics Diagnosis of Osteoporosis Drug Therapies Risk Reduction Living with Osteoporosis 3 What is Osteoporosis? Osteoporosis:
www.osteoporosis.ca 1 2 Overview of the Presentation Osteoporosis: An Overview Bone Basics Diagnosis of Osteoporosis Drug Therapies Risk Reduction Living with Osteoporosis 3 What is Osteoporosis? Osteoporosis:
PICATO (ingenol mebutate) gel
 PICATO (ingenol mebutate) gel Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
PICATO (ingenol mebutate) gel Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
