Texas Prior Authorization Program Clinical Edit Criteria
|
|
|
- Kellie Andrews
- 6 years ago
- Views:
Transcription
1 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Provigil (Modafinil) Drugs requiring prior authorization: the list of drugs requiring prior authorization for this clinical edit Prior authorization criteria logic: a description of how the prior authorization request will be evaluated against the clinical edit criteria rules Logic diagram: a visual depiction of the clinical edit criteria logic Supporting tables: a collection of information associated with the steps within the criteria (diagnosis codes, procedure codes, and therapy codes); provided when applicable References: clinical publications and sources relevant to this clinical edit te: Click the hyperlink to navigate directly to that section. Nuvigil (Armodafinil) Drugs requiring prior authorization: the list of drugs requiring prior authorization for this clinical edit Prior authorization criteria logic: a description of how the prior authorization request will be evaluated against the clinical edit criteria rules Logic diagram: a visual depiction of the clinical edit criteria logic Supporting tables: a collection of information associated with the steps within the criteria (diagnosis codes, procedure codes, and therapy codes); provided when applicable References: clinical publications and sources relevant to this clinical edit te: Click the hyperlink to navigate directly to that section. October 5, 2017 Copyright 2017 Health Information Designs, LLC 1
2 Revision tes Annual review by staff Added dose check to modafinil logic and diagram, pages 4-5 Added armodafinil to clinical edit Added criteria logic and diagram for armodafinil, pages Updated references, page 17 October 5, 2017 Copyright 2017 Health Information Designs, LLC 2
3 Provigil Drugs Requiring Prior Authorization Drugs Requiring Prior Authorization Label Name GCN MODAFINIL 100 MG TABLET MODAFINIL 200 MG TABLET PROVIGIL 100 MG TABLET PROVIGIL 200 MG TABLET October 5, 2017 Copyright 2017 Health Information Designs, LLC 3
4 Provigil Clinical Edit Criteria Logic 1. Is the client greater than or equal to () 16 years of age? [ ] (Go to #2) [ ] (Deny) 2. Does the client have a diagnosis of narcolepsy or shift work disorder in the last 730 days? [ ] (Go to #5) [ ] (Go to #3) 3. Does the client have a diagnosis of obstructive sleep apnea in the last 730 days? [ ] (Go to #4) [ ] (Deny) 4. Does the client have a procedure code for CPAP or BiPAP in the last 730 days? [ ] (Go to #5) [ ] (Deny) 5. Does the client have a diagnosis of severe hepatic impairment in the past 365 days? [ ] (Go to #6) [ ] (Go to #7) 6. Is the dose less than or equal to ( ) 100mg per day? [ ] (Approve 365 days) [ ] (Deny) 7. Is the dose less than or equal to ( ) 200mg per day? [ ] (Approve 365 days) [ ] (Deny) October 5, 2017 Copyright 2017 Health Information Designs, LLC 4
5 Provigil Clinical Edit Criteria Logic Diagram Step 1 Is the client 16 years of age? Deny Request Step 2 Step 5 Step 6 Does the client have a diagnosis of narcolepsy or shift work disorder in the last 730 days? Does the client have a diagnosis of severe hepatic impairment in the last 365 days? Is the dose 100mg per day? Approve Request (365 days) Step 3 Step 4 Step 7 Does the client have a diagnosis of obstructive sleep apnea in the last 730 days? Does the client have a procedure code for CPAP or BiPAP in the last 730 days? Is the dose 200mg per day? Deny Request Deny Request Deny Request Approve Request (365 days) October 5, 2017 Copyright 2017 Health Information Designs, LLC 5
6 Provigil Clinical Edit Criteria Supporting Tables ICD-9 Code Step 2 (diagnosis of narcolepsy or shift work disorder) Description Required diagnosis: 1 Look back timeframe: 730 days CIRCADIAN RHYTHM SLEEP DISORDER, OF NON ORGANIC CIRCADIAN RHYTHM SLEEP DISORDER, SHIFT WORK TYPE NARCOLEPSY,WITHOUT CATAPLEXY NARCOLEPSY,WITH CATAPLEXY NARCOLEPSY IN CONDITIONS CLASSIFIED ELSEWHERE, WITHOUT CATAPLEXY NARCOLEPSY IN CONDITIONS CLASSIFIED ELSEWHERE, WITH CATAPLEXY ICD-10 Code Description F518 G4726 G47411 G47419 G47421 G47429 OTHER SLEEP DISORDERS NOT DUE TO A SUBSTANCE OR KNOWN PHYSIOLOGICAL CONDITION CIRCADIAN RHYTHM SLEEP DISORDER, SHIFT WORK TYPE NARCOLEPSY WITH CATAPLEXY NARCOLEPSY WITHOUT CATAPLEXY NARCOLEPSY IN CONDITIONS CLASSIFIED ELSEWHERE WITH CATAPLEXY NARCOLEPSY IN CONDITIONS CLASSIFIED ELSEWHERE WITHOUT CATAPLEXY Step 3 (diagnosis of obstructive sleep apnea) Required diagnosis: 1 Look back timeframe: 730 days ICD-9 Code Description OBSTRUCTIVE SLEEP APNEA (ADULT) INSOMNIA WITH SLEEP APNEA UNSPECIFIED HYPERSOMNIA WITH SLEEP APNEA UNSPECIFIED UNSPECIFIED SLEEP APNEA ICD-10 Code Description G4730 SLEEP APNEA, UNSPECIFIED G4733 OBSTRUCTIVE SLEEP APNEA (ADULT) (PEDIATRIC) October 5, 2017 Copyright 2017 Health Information Designs, LLC 6
7 Step 4 (procedure code for CPAP or BiPAP) Required procedure: 1 Look back timeframe: 730 days CPT Code Description 9390 CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) CPAP INITIATION AND MANAGEMENT SLEEP STAGING WITH INITIATION OF CPAP E0601 CPAP DEVICE E0470 CPAP WITH BI-LEVEL FAILURE E0471 CPAP WITH BI-LEVEL FAILURE E0472 CPAP WITH BI-LEVEL FAILURE Step 5 (diagnosis of clinically-significant hepatic impairment) Required diagnosis: 1 Look back timeframe: 365 days ICD-9 Description 0700 HEPATITIS A WITH COMA 0701 HEPATITIS A W/O COMA HPT B ACTE COMA WO DLTA HPT B ACTE COMA W DLTA HPT B CHRN COMA WO DLTA HPT B CHRN COMA W DLTA HPT B ACTE WO CM WO DLTA HPT B ACTE WO CM W DLTA HPT B CHRN WO CM WO DLTA HPT B CHRN WO CM W DLTA ACUTE HEPATITIS C WITH HEPATIC COMA HPT DLT WO B W HPT COMA HPT E W HEPAT COMA CHRNC HPT C W HEPAT COMA OTH VRL HEPAT W HPT COMA ACUTE HEPATITIS C WITHOUT MENTION OF HEPATIC COMA HPT DLT WO B WO HPT COMA HPT E WO HEPAT COMA CHRNC HPT C WO HPAT COMA OTH VRL HPAT WO HPT COMA 0706 VIRAL HEPAT NOS W COMA UNSPECIFIED VIRAL HEPATITIS C WITHOUT HEPATIC COMA October 5, 2017 Copyright 2017 Health Information Designs, LLC 7
8 Step 5 (diagnosis of clinically-significant hepatic impairment) Required diagnosis: 1 Look back timeframe: 365 days UNSPECIFIED VIRAL HEPATITIS C WITH HEPATIC COMA 0709 VIRAL HEPAT NOS W/O COMA 5710 ALCOHOLIC FATTY LIVER 5711 AC ALCOHOLIC HEPATITIS 5712 ALCOHOL CIRRHOSIS LIVER 5713 ALCOHOL LIVER DAMAGE NOS CHRONIC HEPATITIS NOS CHR PERSISTENT HEPATITIS AUTOIMMUNE HEPATITIS CHRONIC HEPATITIS NEC 5715 CIRRHOSIS OF LIVER NOS 5716 BILIARY CIRRHOSIS 5718 CHRONIC LIVER DIS NEC 5719 CHRONIC LIVER DIS NOS 5720 ABSCESS OF LIVER 5721 PORTAL PYEMIA 5722 HEPATIC ENCEPHALOPATHY 5723 PORTAL HYPERTENSION 5724 HEPATORENAL SYNDROME 5728 OTH SEQUELA, CHR LIV DIS 5730 CHR PASSIV CONGEST LIVER 5731 HEPATITIS IN VIRAL DIS 5732 HEPATITIS IN OTH INF DIS 5733 HEPATITIS NOS 5734 HEPATIC INFARCTION 5738 LIVER DISORDERS NEC 5739 LIVER DISORDER NOS ICD-10 Code B150 B159 B160 B161 B162 B169 B170 B1710 Description HEPATITIS A WITH HEPATIC COMA HEPATITIS A WITHOUT HEPATIC COMA ACUTE HEPATITIS B WITH DELTA-AGENT WITH HEPATIC COMA ACUTE HEPATITIS B WITH DELTA-AGENT WITHOUT HEPATIC COMA ACUTE HEPATITIS B WITHOUT DELTA-AGENT WITH HEPATIC COMA ACUTE HEPATITIS B WITHOUT DELTA-AGENT AND WITHOUT HEPATIC COMA ACUTE DELTA-(SUPER) INFECTION OF HEPATITIS B CARRIER ACUTE HEPATITIS C WITHOUT HEPATIC COMA October 5, 2017 Copyright 2017 Health Information Designs, LLC 8
9 B1711 B172 B178 B179 B180 B181 B182 B188 B189 B190 B1910 B1911 B1920 B1921 B199 B251 K700 K7010 K7011 K702 K7030 K7031 K7040 K7041 K709 K710 K7110 K7111 K712 K713 K714 K7150 K7151 K716 K717 Step 5 (diagnosis of clinically-significant hepatic impairment) Required diagnosis: 1 Look back timeframe: 365 days ACUTE HEPATITIS C WITH HEPATIC COMA ACUTE HEPATITIS E OTHER SPECIFIED ACUTE VIRAL HEPATITIS ACUTE VIRAL HEPATITIS, UNSPECIFIED CHRONIC VIRAL HEPATITIS B WITH DELTA-AGENT CHRONIC VIRAL HEPATITIS B WITHOUT DELTA-AGENT CHRONIC VIRAL HEPATITIS C OTHER CHRONIC VIRAL HEPATITIS CHRONIC VIRAL HEPATITIS, UNSPECIFIED UNSPECIFIED VIRAL HEPATITIS WITH HEPATIC COMA UNSPECIFIED VIRAL HEPATITIS B WITHOUT HEPATIC COMA UNSPECIFIED VIRAL HEPATITIS B WITH HEPATIC COMA UNSPECIFIED VIRAL HEPATITIS C WITHOUT HEPATIC COMA UNSPECIFIED VIRAL HEPATITIS C WITH HEPATIC COMA UNSPECIFIED VIRAL HEPATITIS WITHOUT HEPATIC COMA CYTOMEGALOVIRAL HEPATITIS ALCOHOLIC FATTY LIVER ALCOHOLIC HEPATITIS WITHOUT ASCITES ALCOHOLIC HEPATITIS WITH ASCITES ALCOHOLIC FIBROSIS AND SCLEROSIS OF LIVER ALCOHOLIC CIRRHOSIS OF LIVER WITHOUT ASCITES ALCOHOLIC CIRRHOSIS OF LIVER WITH ASCITES ALCOHOLIC HEPATIC FAILURE WITHOUT COMA ALCOHOLIC HEPATIC FAILURE WITH COMA ALCOHOLIC LIVER DISEASE, UNSPECIFIED TOXIC LIVER DISEASE WITH CHOLESTASIS TOXIC LIVER DISEASE WITH HEPATIC NECROSIS, WITHOUT COMA TOXIC LIVER DISEASE WITH HEPATIC NECROSIS, WITH COMA TOXIC LIVER DISEASE WITH ACUTE HEPATITIS TOXIC LIVER DISEASE WITH CHRONIC PERSISTENT HEPATITIS TOXIC LIVER DISEASE WITH CHRONIC LOBULAR HEPATITIS TOXIC LIVER DISEASE WITH CHRONIC ACTIVE HEPATITIS WITHOUT ASCITES TOXIC LIVER DISEASE WITH CHRONIC ACTIVE HEPATITIS WITH ASCITES TOXIC LIVER DISEASE WITH HEPATITIS, NOT ELSEWHERE CLASSIFIED TOXIC LIVER DISEASE WITH FIBROSIS AND CIRRHOSIS OF LIVER October 5, 2017 Copyright 2017 Health Information Designs, LLC 9
10 Step 5 (diagnosis of clinically-significant hepatic impairment) Required diagnosis: 1 Look back timeframe: 365 days K718 K719 K7201 K7210 K7211 K7290 K7291 K730 K731 K732 K738 K739 K740 K741 K742 K743 K744 K745 K7460 K7469 K750 K751 K752 K753 K7581 K7589 K759 K760 K761 K763 K764 K765 K766 K767 K7689 K769 TOXIC LIVER DISEASE WITH OTHER DISORDERS OF LIVER TOXIC LIVER DISEASE, UNSPECIFIED ACUTE AND SUBACUTE HEPATIC FAILURE WITH COMA CHRONIC HEPATIC FAILURE WITHOUT COMA CHRONIC HEPATIC FAILURE WITH COMA HEPATIC FAILURE, UNSPECIFIED WITHOUT COMA HEPATIC FAILURE, UNSPECIFIED WITH COMA CHRONIC PERSISTENT HEPATITIS, NOT ELSEWHERE CLASSIFIED CHRONIC LOBULAR HEPATITIS, NOT ELSEWHERE CLASSIFIED CHRONIC ACTIVE HEPATITIS, NOT ELSEWHERE CLASSIFIED OTHER CHRONIC HEPATITIS, NOT ELSEWHERE CLASSIFIED CHRONIC HEPATITIS, UNSPECIFIED HEPATIC FIBROSIS HEPATIC SCLEROSIS HEPATIC FIBROSIS WITH HEPATIC SCLEROSIS PRIMARY BILIARY CIRRHOSIS SECONDARY BILIARY CIRRHOSIS BILIARY CIRRHOSIS, UNSPECIFIED UNSPECIFIED CIRRHOSIS OF LIVER OTHER CIRRHOSIS OF LIVER ABSCESS OF LIVER PHLEBITIS OF PORTAL VEIN NONSPECIFIC REACTIVE HEPATITIS GRANULOMATOUS HEPATITIS, NOT ELSEWHERE CLASSIFIED NONALCOHOLIC STEATOHEPATITIS (NASH) OTHER SPECIFIED INFLAMMATORY LIVER DISEASES INFLAMMATORY LIVER DISEASE, UNSPECIFIED FATTY (CHANGE OF) LIVER, NOT ELSEWHERE CLASSIFIED CHRONIC PASSIVE CONGESTION OF LIVER INFARCTION OF LIVER PELIOSIS HEPATIS HEPATIC VENO-OCCLUSIVE DISEASE PORTAL HYPERTENSION HEPATORENAL SYNDROME OTHER SPECIFIED DISEASES OF LIVER LIVER DISEASE, UNSPECIFIED October 5, 2017 Copyright 2017 Health Information Designs, LLC 10
11 Step 5 (diagnosis of clinically-significant hepatic impairment) Required diagnosis: 1 Look back timeframe: 365 days K77 LIVER DISORDERS IN DISEASES CLASSIFIED ELSEWHERE October 5, 2017 Copyright 2017 Health Information Designs, LLC 11
12 Nuvigil Drugs Requiring Prior Authorization Drugs Requiring Prior Authorization Label Name GCN ARMODAFINIL 50MG TABLET ARMODAFINIL 150MG TABLET ARMODAFINIL 200MG TABLET ARMODAFINIL 250MG TABLET NUVIGIL 50MG TABLET NUVIGIL 150MG TABLET NUVIGIL 200MG TABLET NUVIGIL 250MG TABLET October 5, 2017 Copyright 2017 Health Information Designs, LLC 12
13 Nuvigil Clinical Edit Criteria Logic 1. Is the client greater than or equal to () 16 years of age? [ ] (Go to #2) [ ] (Deny) 2. Does the client have a diagnosis of shift work disorder in the last 730 days? [ ] (Go to #3) [ ] (Go to #4) 3. Is the dose less than or equal to ( ) 150mg per day? [ ] (Approve 365 days) [ ] (Deny) 4. Does the client have a diagnosis of narcolepsy in the last 730 days? [ ] (Go to #7) [ ] (Go to #5) 5. Does the client have a diagnosis of obstructive sleep apnea in the last 730 days? [ ] (Go to #6) [ ] (Deny) 6. Does the client have a procedure code for CPAP or BiPAP in the last 730 days? [ ] (Go to #7) [ ] (Deny) 7. Is the dose less than or equal to ( ) 250mg per day? [ ] (Approve 365 days) [ ] (Deny) October 5, 2017 Copyright 2017 Health Information Designs, LLC 13
14 Nuvigil Clinical Edit Criteria Logic Diagram Step 1 Is the client 16 years of age? Deny Request Step 2 Step 3 Does the client have a diagnosis of shift work disorder in the last 730 days? Is the dose 150mg per day? Approve Request (365 days) Step 4 Step 7 Does the client have a diagnosis of narcolepsy in the last 730 days? Is the dose 250mg per day? Approve Request (365 days) Step 5 Step 6 Does the client have a diagnosis of obstructive sleep apnea in the last 730 days? Does the client have a procedure code for CPAP or BiPAP in the last 730 days? Deny Request Deny Request Deny Request October 5, 2017 Copyright 2017 Health Information Designs, LLC 14
15 Nuvigil Clinical Edit Criteria Supporting Tables ICD-9 Code Step 2 (diagnosis of shift work disorder) Description Required diagnosis: 1 Look back timeframe: 730 days CIRCADIAN RHYTHM SLEEP DISORDER, OF NON ORGANIC CIRCADIAN RHYTHM SLEEP DISORDER, SHIFT WORK TYPE ICD-10 Code F518 G4726 Description OTHER SLEEP DISORDERS NOT DUE TO A SUBSTANCE OR KNOWN PHYSIOLOGICAL CONDITION CIRCADIAN RHYTHM SLEEP DISORDER, SHIFT WORK TYPE ICD-9 Code Description Step 4 (diagnosis of narcolepsy) Required diagnosis: 1 Look back timeframe: 730 days NARCOLEPSY,WITHOUT CATAPLEXY NARCOLEPSY,WITH CATAPLEXY NARCOLEPSY IN CONDITIONS CLASSIFIED ELSEWHERE, WITHOUT CATAPLEXY NARCOLEPSY IN CONDITIONS CLASSIFIED ELSEWHERE, WITH CATAPLEXY ICD-10 Code Description G47411 G47419 G47421 G47429 NARCOLEPSY WITH CATAPLEXY NARCOLEPSY WITHOUT CATAPLEXY NARCOLEPSY IN CONDITIONS CLASSIFIED ELSEWHERE WITH CATAPLEXY NARCOLEPSY IN CONDITIONS CLASSIFIED ELSEWHERE WITHOUT CATAPLEXY October 5, 2017 Copyright 2017 Health Information Designs, LLC 15
16 Step 5 (diagnosis of obstructive sleep apnea) Required quantity: 1 Look back timeframe: 730 days For the list of diagnosis codes that pertain to this step, see the Obstructive Sleep Apnea table in the previous Supporting Tables section. te: Click the hyperlink to navigate directly to the table. Step 6 (procedure code for CPAP or BiPAP) Required quantity: 1 Look back timeframe: 730 days For the list of procedure codes that pertain to this step, see the CPAP/BiPAP table in the previous Supporting Tables section. te: Click the hyperlink to navigate directly to the table. October 5, 2017 Copyright 2017 Health Information Designs, LLC 16
17 Clinical Edit Criteria References 1. Ballon JS, Feifel D. A systematic review of modafinil: Potential clinical uses and mechanisms of action. J Clin Psychiatry 2006;67: Psychopharmacologic Drugs Advisory Committee, United States Food and Drug Administration. March 23, Meeting minutes available at: Accessed on August 11, American Medical Association data files ICD-9-CM Diagnosis Codes. Available at 4. American Medical Association data files ICD-10-CM Diagnosis Codes. Available at 5. Clinical Pharmacology [online database]. Tampa, FL: Elsevier/Gold Standard, Inc.; Available at Accessed on June 9, Micromedex [online database]. Available at Accessed on June 9, Provigil Prescribing Information. rth Wales, PA. Teva Pharmaceuticals. January Nuvigil Prescribing Information. rth Wales, PA. Teva Pharmaceuticals. February Krahn LE, Hershner S, Loeding, LD, et al. Quality Measures for the Care of Patients with Narcolepsy. J Clin Sleep Med 2015;11(3): Epstein LJ, Kristo D, Strollo PJ, et al. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med 2009;5(3): Sack RL, Auckley D, Auger RR, et al. Circadian rhythm sleep disorders: Part I, basic principles, shift work and jet lag disorders. SLEEP 2007;30(11): October 5, 2017 Copyright 2017 Health Information Designs, LLC 17
18 Publication History The Publication History records the publication iterations and revisions to this document. tes for the most current revision are also provided in the Revision tes on the first page of this document. Publication Date tes 01/31/2011 Initial publication and posting to website 04/13/2012 Added a new section to specify the drugs requiring prior authorization In the Clinical Edit Supporting Tables section, revised tables to specify the diagnosis codes pertinent to steps 2, 3 and 4 of the logic diagram In the Clinical Edit Supporting Tables section, revised table to specify the procedure codes pertinent to step 5 of the logic diagram 10/26/2012 Added Modafinil 100mg and 200mg tablets to table of drugs requiring prior authorization 04/03/2015 Updated to include ICD-10s 10/05/2017 Annual review by staff Added dose check for modafinil to logic and diagram, pages 4-5 Added armodafinil to clinical edit Added criteria logic and diagram for armodafinil, pages Updated references, page 17 October 5, 2017 Copyright 2017 Health Information Designs, LLC 18
Texas Prior Authorization Program Clinical Edit Criteria
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Sodium-Glucose Cotransporter 2 (SGLT2) Inhibitor Combination Agents
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Sodium-Glucose Cotransporter 2 (SGLT2) Inhibitor Combination Agents This criteria was recommended for review by an MCO to ensure appropriate
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Sodium-Glucose Cotransporter 2 (SGLT2) Inhibitor Combination Agents This criteria was recommended for review by an MCO to ensure appropriate
Supplementary Table 1 ICD -8 and ICD- 9 codes for liver disease and the three definitions of liver cirrhosis (LC)
 Supplementary Table 1 ICD -8 and ICD- 9 codes for liver disease and the three definitions of liver cirrhosis () ICD version Code Description Liver disease 1 2 3 ICD-8 5700 Acute/subacute necrosis of liver
Supplementary Table 1 ICD -8 and ICD- 9 codes for liver disease and the three definitions of liver cirrhosis () ICD version Code Description Liver disease 1 2 3 ICD-8 5700 Acute/subacute necrosis of liver
Texas Prior Authorization Program Clinical Criteria
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Criteria Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Criteria Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Sitagliptin (Januvia)
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document 25mg Drugs requiring prior authorization: the list of drugs requiring prior
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document 25mg Drugs requiring prior authorization: the list of drugs requiring prior
Texas Prior Authorization Program Clinical Edit Criteria
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Xifaxan 200mg Drugs requiring prior authorization: the list of drugs requiring
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Xifaxan 200mg Drugs requiring prior authorization: the list of drugs requiring
Texas Prior Authorization Program Clinical Edit Criteria
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Amitiza (Lubiprostone)
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Dipeptidyl Peptidase-4 (DPP-4) Inhibitors
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Criteria Information Included in this Document DPP-4 Inhibitor Criteria A: Alogliptin 6.25mg, Januvia 25mg, Nesina 6.25mg, Onglyza
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Criteria Information Included in this Document DPP-4 Inhibitor Criteria A: Alogliptin 6.25mg, Januvia 25mg, Nesina 6.25mg, Onglyza
Texas Prior Authorization Program Clinical Criteria
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Information Included in this Document Xifaxan 200mg Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Information Included in this Document Xifaxan 200mg Drugs requiring prior authorization: the list of drugs requiring prior authorization
Prior Authorization Neurontin (gabapentin) 2016
 Drugs Requiring Prior Authorization Label Name GCN GABAPENTIN 100 MG CAPSULE 00780 GABAPENTIN 300 MG CAPSULE 00781 GABAPENTIN 400 MG CAPSULE 00782 GABAPENTIN 250 MG/5 ML SOLN 13235 GABAPENTIN 600 MG TABLET
Drugs Requiring Prior Authorization Label Name GCN GABAPENTIN 100 MG CAPSULE 00780 GABAPENTIN 300 MG CAPSULE 00781 GABAPENTIN 400 MG CAPSULE 00782 GABAPENTIN 250 MG/5 ML SOLN 13235 GABAPENTIN 600 MG TABLET
Diclofenac 3% Gel, Diclofenac 1.5% and 2% Topical Solution
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class, Diclofenac 1.5% and 2% Topical Solution This criteria was recommended for review by an MCO to ensure appropriate and safe utilization
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class, Diclofenac 1.5% and 2% Topical Solution This criteria was recommended for review by an MCO to ensure appropriate and safe utilization
Texas Prior Authorization Program Clinical Criteria
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Criteria Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Criteria Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Fentanyl Agents Clinical Edit Criteria
 Fentanyl Agents Clinical Edit Criteria Drug/Drug Class: Fentanyl Agents Superior HealthPlan follows the guidance of the Texas Vendor Drug Program (VDP) for all clinical edit criteria. Superior has adjusted
Fentanyl Agents Clinical Edit Criteria Drug/Drug Class: Fentanyl Agents Superior HealthPlan follows the guidance of the Texas Vendor Drug Program (VDP) for all clinical edit criteria. Superior has adjusted
Adverse Drug Event (ADE) Anti-Coagulant Usage - OPTION #2 Measure Definition Sheet
 Adverse Drug Event (ADE) Anti-Coagulant Usage - OPTION #2 Measure Definition Sheet Data Definition Percent of International Normalized Ratio (INR) Greater Than 5 Numerator: Total number of INR >5 readings.
Adverse Drug Event (ADE) Anti-Coagulant Usage - OPTION #2 Measure Definition Sheet Data Definition Percent of International Normalized Ratio (INR) Greater Than 5 Numerator: Total number of INR >5 readings.
Methylnaltrexone Bromide (Relistor)
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Prior Authorization Flexeril/Amrix (cyclobenzaprine) 2017
 Drugs Requiring Prior Authorization Label Name GCN AMRIX ER 15 MG CAPSULE 97959 AMRIX ER 30 MG CAPSULE 97960 CYCLOBENZAPRINE 10 MG TABLET 18020 CYCLOBENZAPRINE 5 MG TABLET 12805 CYCLOBENZAPRINE 7.5 MG
Drugs Requiring Prior Authorization Label Name GCN AMRIX ER 15 MG CAPSULE 97959 AMRIX ER 30 MG CAPSULE 97960 CYCLOBENZAPRINE 10 MG TABLET 18020 CYCLOBENZAPRINE 5 MG TABLET 12805 CYCLOBENZAPRINE 7.5 MG
Lidoderm (Lidocaine) Patch
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Aldara 5% Cream Drugs requiring prior authorization: the list of drugs requiring
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Aldara 5% Cream Drugs requiring prior authorization: the list of drugs requiring
Texas Prior Authorization Program Clinical Edit Criteria
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Thiazolidinediones Clinical Edit Information Included in this Document Thiazolidinediones Drugs requiring prior authorization: the
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Thiazolidinediones Clinical Edit Information Included in this Document Thiazolidinediones Drugs requiring prior authorization: the
Hypoglycemics, Lantus Insulin
 Texas Prior Authorization Program PDL Edit Criteria Drug/Drug Class Hypoglycemics, Lantus Insulin Information Included in this Document Hypoglycemics, Lantus Insulin Drugs requiring prior authorization:
Texas Prior Authorization Program PDL Edit Criteria Drug/Drug Class Hypoglycemics, Lantus Insulin Information Included in this Document Hypoglycemics, Lantus Insulin Drugs requiring prior authorization:
Texas Prior Authorization Program Clinical Criteria
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Aldurazyme Adagen Carbaglu Ceprotin Elaprase Fabrazyme Lumizyme Naglazyme Orfandin Ravicti Vimizim Note: Click the hyperlink to navigate
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Aldurazyme Adagen Carbaglu Ceprotin Elaprase Fabrazyme Lumizyme Naglazyme Orfandin Ravicti Vimizim Note: Click the hyperlink to navigate
Texas Prior Authorization Program Clinical Edit Criteria
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document - Oral Drugs requiring prior authorization: the list of drugs requiring prior
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document - Oral Drugs requiring prior authorization: the list of drugs requiring prior
Hepatitis Panel/Acute Hepatitis Panel
 190.33 - Hepatitis Panel/Acute Hepatitis Panel This panel consists of the following tests: Hepatitis A antibody (HAAb), IgM antibody; Hepatitis B core antibody (HBcAb), IgM antibody; Hepatitis B surface
190.33 - Hepatitis Panel/Acute Hepatitis Panel This panel consists of the following tests: Hepatitis A antibody (HAAb), IgM antibody; Hepatitis B core antibody (HBcAb), IgM antibody; Hepatitis B surface
Texas Prior Authorization Program Clinical Criteria. This criteria was recommended for review by an MCO to ensure appropriate and safe utilization.
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class This criteria was recommended for review by an MCO to ensure appropriate and safe utilization. Clinical Information Included in this
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class This criteria was recommended for review by an MCO to ensure appropriate and safe utilization. Clinical Information Included in this
Agents for the Treatment of Hepatitis C
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria. H.P. Acthar
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Flexeril/Amrix (Cyclobenzaprine) Clinical Edit Criteria
 Flexeril/Amrix (Cyclobenzaprine) Clinical Edit Criteria Drug/Drug Class: Flexeril/Amrix (Cyclobenzaprine) Superior HealthPlan follows the guidance of the Texas Vendor Drug Program (VDP) for all clinical
Flexeril/Amrix (Cyclobenzaprine) Clinical Edit Criteria Drug/Drug Class: Flexeril/Amrix (Cyclobenzaprine) Superior HealthPlan follows the guidance of the Texas Vendor Drug Program (VDP) for all clinical
Victoza (Liraglutide) Solution for Injection
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Hepatitis Panel/Acute Hepatitis Panel
 190.33 - Hepatitis Panel/Acute Hepatitis Panel This panel consists of the following tests: Hepatitis A antibody (HAAb), IgM antibody; Hepatitis B core antibody (HBcAb), IgM antibody; Hepatitis B surface
190.33 - Hepatitis Panel/Acute Hepatitis Panel This panel consists of the following tests: Hepatitis A antibody (HAAb), IgM antibody; Hepatitis B core antibody (HBcAb), IgM antibody; Hepatitis B surface
Clinical Policy: Modafinil (Provigil) Reference Number: CP.PMN.39. Line of Business: Medicaid
 Clinical Policy: (Provigil) Reference Number: CP.PMN.39 Effective Date: 05/08 Last Review Date: 05/17 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of this
Clinical Policy: (Provigil) Reference Number: CP.PMN.39 Effective Date: 05/08 Last Review Date: 05/17 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of this
Texas Prior Authorization Program Clinical Criteria. Allergen Extracts
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Information Included in this Document Oralair (Mixed Grass Pollens Allergen Extract)) Drugs requiring prior authorization: the
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Information Included in this Document Oralair (Mixed Grass Pollens Allergen Extract)) Drugs requiring prior authorization: the
Injectable Agents for the Treatment of Pulmonary Arterial Hypertension (PAH)
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Injectable Agents for the Treatment of Pulmonary Arterial Hypertension (PAH) Clinical Edit Information Included in this Document
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Injectable Agents for the Treatment of Pulmonary Arterial Hypertension (PAH) Clinical Edit Information Included in this Document
Flexeril/Amrix (Cyclobenzaprine)
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Provigil Nuvigil. Provigil (modafinil) / Nuvigil (armodafinil) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.60.14 Subject: Provigil Nuvigil Page: 1 of 6 Last Review Date: December 8. 2017 Provigil Nuvigil Description
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.60.14 Subject: Provigil Nuvigil Page: 1 of 6 Last Review Date: December 8. 2017 Provigil Nuvigil Description
Provigil / Nuvigil. Provigil (modafinil) / Nuvigil (armodafinil) Description. Section: Prescription Drugs Effective Date: July 1, 2014
 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Section: Prescription Drugs Effective Date: July 1, 2014 Subject: Provigil / Nuvigil Page: 1 of 7 Last Review Date: June 12, 2014
1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Section: Prescription Drugs Effective Date: July 1, 2014 Subject: Provigil / Nuvigil Page: 1 of 7 Last Review Date: June 12, 2014
Byetta (Exenatide Injection)
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Agents for Cystic Fibrosis
 Texas Prior Authorization Program Clinical Edit Criteria Clinical Edit Information Included in this Document Kalydeco (Ivacaftor) Drugs requiring prior authorization: the list of drugs requiring prior
Texas Prior Authorization Program Clinical Edit Criteria Clinical Edit Information Included in this Document Kalydeco (Ivacaftor) Drugs requiring prior authorization: the list of drugs requiring prior
Xyrem (Sodium Oxybate)
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Criteria Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Criteria Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Drugs requiring prior authorization: the list of drugs requiring prior authorization for this clinical criteria
 Drug/Drug Class Antipsychotics Clinical Criteria Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization for this clinical criteria Prior
Drug/Drug Class Antipsychotics Clinical Criteria Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization for this clinical criteria Prior
Sample page. Laboratory. Cross Coder. Essential links from CPT codes to ICD-10-CM and HCPCS
 Cross Coder 2018 Laboratory Essential links from CPT codes to ICD-10-CM and HCPCS POWER UP YOUR CODING with Optum360, your trusted coding partner for 32 years. Visit optum360coding.com. Contents Introduction...i
Cross Coder 2018 Laboratory Essential links from CPT codes to ICD-10-CM and HCPCS POWER UP YOUR CODING with Optum360, your trusted coding partner for 32 years. Visit optum360coding.com. Contents Introduction...i
Glucagon-Like Peptide (GLP-1) Receptor Agonists Clinical Edit Criteria
 Glucagon-Like Peptide (GLP-1) Receptor Agonists Clinical Edit Criteria Drug/Drug Class: Glucagon-Like Peptide-1 (GLP-1) Receptor Agonists Superior HealthPlan follows the guidance of the Texas Vendor Drug
Glucagon-Like Peptide (GLP-1) Receptor Agonists Clinical Edit Criteria Drug/Drug Class: Glucagon-Like Peptide-1 (GLP-1) Receptor Agonists Superior HealthPlan follows the guidance of the Texas Vendor Drug
Sleep 101. Kathleen Feeney RPSGT, RST, CSE Business Development Specialist
 Sleep 101 Kathleen Feeney RPSGT, RST, CSE Business Development Specialist 2016 Why is Sleep Important More than one-third of the population has trouble sleeping (Gallup) Obstructive Sleep Apnea Untreated
Sleep 101 Kathleen Feeney RPSGT, RST, CSE Business Development Specialist 2016 Why is Sleep Important More than one-third of the population has trouble sleeping (Gallup) Obstructive Sleep Apnea Untreated
Vesicular Monoamine Transporter 2 (VMAT2) Inhibitors
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Vesicular Monoamine Transporter 2 (VMAT2) Inhibitors Clinical Edit Information Included in this Document Austedo (Deutetrabenazine) /
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Vesicular Monoamine Transporter 2 (VMAT2) Inhibitors Clinical Edit Information Included in this Document Austedo (Deutetrabenazine) /
INDIANA HEALTH COVERAGE PROGRAMS
 INDIANA HEALTH COVERAGE PROGRAMS PROVIDER CODE TABLES Note: Due to possible changes in Indiana Health Coverage Programs (IHCP) policy or national coding updates, inclusion of a code on the code tables
INDIANA HEALTH COVERAGE PROGRAMS PROVIDER CODE TABLES Note: Due to possible changes in Indiana Health Coverage Programs (IHCP) policy or national coding updates, inclusion of a code on the code tables
Icd 10 code for ischemic hepatitis
 Feb 8, 2016. Coding of Shock Liver". ICD-10 has no coding option of shock liver or ischemic hepatitis. Due to the necrosis that occurs from lack of blood supply, this is where we would start, resulting
Feb 8, 2016. Coding of Shock Liver". ICD-10 has no coding option of shock liver or ischemic hepatitis. Due to the necrosis that occurs from lack of blood supply, this is where we would start, resulting
Index SLEEP MEDICINE CLINICS. Note: Page numbers of article titles are in boldface type. Cerebrospinal fluid analysis, for Kleine-Levin syndrome,
 165 SLEEP MEDICINE CLINICS Index Sleep Med Clin 1 (2006) 165 170 Note: Page numbers of article titles are in boldface type. A Academic performance, effects of sleepiness in children on, 112 Accidents,
165 SLEEP MEDICINE CLINICS Index Sleep Med Clin 1 (2006) 165 170 Note: Page numbers of article titles are in boldface type. A Academic performance, effects of sleepiness in children on, 112 Accidents,
Latest Press Release. polar lights paradisebirds
 corp@stantec.com Latest Press Release polar lights paradisebirds S I50 Heart failure. I50.1 Left ventricular failure, unspecified; I50.2 Systolic (congestive) heart failure. I50.20 Unspecified systolic
corp@stantec.com Latest Press Release polar lights paradisebirds S I50 Heart failure. I50.1 Left ventricular failure, unspecified; I50.2 Systolic (congestive) heart failure. I50.20 Unspecified systolic
Pharmacy Benefit Determination Policy
 Policy Subject: CNS Stimulant Medications Policy Number: SHS PBD06 Category: CNS Drugs Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS ASO PPO Individual
Policy Subject: CNS Stimulant Medications Policy Number: SHS PBD06 Category: CNS Drugs Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS ASO PPO Individual
Coding for Sleep Disorders Jennifer Rose V. Molano, MD
 Practice Coding for Sleep Disorders Jennifer Rose V. Molano, MD Accurate coding is an important function of neurologic practice. This section of is part of an ongoing series that presents helpful coding
Practice Coding for Sleep Disorders Jennifer Rose V. Molano, MD Accurate coding is an important function of neurologic practice. This section of is part of an ongoing series that presents helpful coding
Drug Prior Authorization Form Alertec (modafinil)
 This document contains both information and form fields. To read information, use the Down Arrow from a form field. Drug Prior Authorization Form The purpose of this form is to obtain information required
This document contains both information and form fields. To read information, use the Down Arrow from a form field. Drug Prior Authorization Form The purpose of this form is to obtain information required
Fatty liver cirrhosis icd 10 code
 Fatty liver cirrhosis icd 10 code 08/14/2017 Dcuo generators no survey 08/15/2017 Xfinity tg1682g default password 08/17/2017 -Hydraulic filter size chart -Btd 5 hacked unlimited money unblocked 08/18/2017
Fatty liver cirrhosis icd 10 code 08/14/2017 Dcuo generators no survey 08/15/2017 Xfinity tg1682g default password 08/17/2017 -Hydraulic filter size chart -Btd 5 hacked unlimited money unblocked 08/18/2017
Available Strengths Limits. 200 mg tablets PA. 50 mg, 150 mg, 200 mg, 250 mg tablets. 500 mg/ml solution PA
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY Sleeping Disorders P&T DATE: 12/14/2016 THERAPEUTIC CLASS Psychiatric Disorders REVIEW HISTORY: 2/16, 5/15, 2/12 LOB AFFECTED
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY Sleeping Disorders P&T DATE: 12/14/2016 THERAPEUTIC CLASS Psychiatric Disorders REVIEW HISTORY: 2/16, 5/15, 2/12 LOB AFFECTED
Drug Regimen Optimization
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Criteria Information Included in this Document Excluding Valsartan / Ramipril Prior authorization criteria logic: a description
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Clinical Criteria Information Included in this Document Excluding Valsartan / Ramipril Prior authorization criteria logic: a description
Alpha-fetoprotein
 Other Names/Abbreviations AFP 190.25 - Alpha-fetoprotein Alpha-fetoprotein (AFP) is a polysaccharide found in some carcinomas. It is effective as a biochemical marker for monitoring the response of certain
Other Names/Abbreviations AFP 190.25 - Alpha-fetoprotein Alpha-fetoprotein (AFP) is a polysaccharide found in some carcinomas. It is effective as a biochemical marker for monitoring the response of certain
Drug Regimen Optimization
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Excluding Valsartan / Ramipril Prior authorization criteria logic: a description
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Excluding Valsartan / Ramipril Prior authorization criteria logic: a description
CHALLENGES OF RECOGNIZING AND TREATING EXCESSIVE SLEEPINESS
 CHALLENGES OF RECOGNIZING AND TREATING EXCESSIVE SLEEPINESS Russell Rosenberg, Ph.D. Neurotrials Research and Atlanta School of Sleep Medicine DISCLOSURES Research grant funds from: Merck Jazz Pharmaceuticals
CHALLENGES OF RECOGNIZING AND TREATING EXCESSIVE SLEEPINESS Russell Rosenberg, Ph.D. Neurotrials Research and Atlanta School of Sleep Medicine DISCLOSURES Research grant funds from: Merck Jazz Pharmaceuticals
Sildenafil / Tadalafil
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Sildenafil / Tadalafil Clinical Criteria Information Included in this Document Drugs requiring prior authorization: the list of drugs
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Sildenafil / Tadalafil Clinical Criteria Information Included in this Document Drugs requiring prior authorization: the list of drugs
Icd 10 code for end stage liver disease. Pinni Meedha Mojutho Ammanu Dengina Koduku Part 1 Kama Kathalu
 Search for: Search Search Icd 10 code for end stage liver disease Pinni Meedha Mojutho Ammanu Dengina Koduku Part 1 Kama Kathalu Medical uses Malignancy. Radical cystectomy is the recommended treatment
Search for: Search Search Icd 10 code for end stage liver disease Pinni Meedha Mojutho Ammanu Dengina Koduku Part 1 Kama Kathalu Medical uses Malignancy. Radical cystectomy is the recommended treatment
Guide for Prescribers
 Pr ALERTEC * Modafinil Tablets 100 mg Central Nervous System Stimulant Guide for Prescribers ALERTEC is manufactured by: Teva Canada Limited Toronto, Ontario M1B 2K9 ALERTEC is distributed by: Teva Canada
Pr ALERTEC * Modafinil Tablets 100 mg Central Nervous System Stimulant Guide for Prescribers ALERTEC is manufactured by: Teva Canada Limited Toronto, Ontario M1B 2K9 ALERTEC is distributed by: Teva Canada
Observational Medical Outcomes Partnership
 Implications of Health Outcomes of Interest Definitions: Acute Liver Injury Case Study Judy Racoosin, Patrick Ryan on behalf of OMOP Research Team Observational Medical Outcomes Partnership Established
Implications of Health Outcomes of Interest Definitions: Acute Liver Injury Case Study Judy Racoosin, Patrick Ryan on behalf of OMOP Research Team Observational Medical Outcomes Partnership Established
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 2021-10 Program Prior Authorization/Medical Necessity Medication Xyrem (sodium oxybate) P&T Approval Date 4/2014, 8/2014, 4/2015,
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 2021-10 Program Prior Authorization/Medical Necessity Medication Xyrem (sodium oxybate) P&T Approval Date 4/2014, 8/2014, 4/2015,
Sleep Medicine. Maintenance of Certification Examination Blueprint. Purpose of the exam
 Sleep Medicine Maintenance of Certification Examination Blueprint Purpose of the exam The exam is designed to evaluate the knowledge, diagnostic reasoning, and clinical judgment skills expected of the
Sleep Medicine Maintenance of Certification Examination Blueprint Purpose of the exam The exam is designed to evaluate the knowledge, diagnostic reasoning, and clinical judgment skills expected of the
Texas Prior Authorization Program Clinical Criteria
 Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Ketorolac Clinical Criteria Information Included in this Document Ketorolac Oral Drugs requiring prior authorization: the list of drugs
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Ketorolac Clinical Criteria Information Included in this Document Ketorolac Oral Drugs requiring prior authorization: the list of drugs
Alpha-fetoprotein
 Other Names/Abbreviations AFP 190.25 - Alpha-fetoprotein Alpha-fetoprotein (AFP) is a polysaccharide found in some carcinomas. It is effective as a biochemical marker for monitoring the response of certain
Other Names/Abbreviations AFP 190.25 - Alpha-fetoprotein Alpha-fetoprotein (AFP) is a polysaccharide found in some carcinomas. It is effective as a biochemical marker for monitoring the response of certain
What Is Cirrhosis? CIRRHOSIS. Cirrhosis occurs when the liver is. by chronic conditions and diseases. permanently scarred or injured
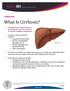 What Is Cirrhosis? Cirrhosis occurs when the liver is permanently scarred or injured by chronic conditions and diseases. Common causes of cirrhosis include: Long-term alcohol abuse. Chronic viral hepatitis
What Is Cirrhosis? Cirrhosis occurs when the liver is permanently scarred or injured by chronic conditions and diseases. Common causes of cirrhosis include: Long-term alcohol abuse. Chronic viral hepatitis
Alpha-fetoprotein
 Other Names/Abbreviations AFP 190.25 - Alpha-fetoprotein Alpha-fetoprotein (AFP) is a polysaccharide found in some carcinomas. It is effective as a biochemical marker for monitoring the response of certain
Other Names/Abbreviations AFP 190.25 - Alpha-fetoprotein Alpha-fetoprotein (AFP) is a polysaccharide found in some carcinomas. It is effective as a biochemical marker for monitoring the response of certain
Managing Sleep Disorders in Primary Care. Objectives. Disclosures. Nancy Nadolski, FNP, MSN, M.Ed Insomnia Medicine of Idaho Boise Counseling Center
 Managing Sleep Disorders in Primary Care Nancy Nadolski, FNP, MSN, M.Ed Insomnia Medicine of Idaho Boise Counseling Center www.sleeplessinidaho.com 1 Objectives 1) Identify stages and behaviors for normal
Managing Sleep Disorders in Primary Care Nancy Nadolski, FNP, MSN, M.Ed Insomnia Medicine of Idaho Boise Counseling Center www.sleeplessinidaho.com 1 Objectives 1) Identify stages and behaviors for normal
Pegylated Interferons and Ribavirins
 Pegylated Interferons and Ribavirins Goal(s): Cover drugs only for those clients where there is evidence of effectiveness and safety Length of Authorization: 16 weeks plus 12-36 additional weeks or 12
Pegylated Interferons and Ribavirins Goal(s): Cover drugs only for those clients where there is evidence of effectiveness and safety Length of Authorization: 16 weeks plus 12-36 additional weeks or 12
Cystic Fibrosis Agents
 Texas Prior Authorization Program Clinical Criteria Clinical Information Included in this Document Kalydeco (Ivacaftor) Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Criteria Clinical Information Included in this Document Kalydeco (Ivacaftor) Drugs requiring prior authorization: the list of drugs requiring prior authorization
HEALTHY LIFESTYLE, HEALTHY SLEEP. There are many different sleep disorders, and almost all of them can be improved with lifestyle changes.
 HEALTHY LIFESTYLE, HEALTHY SLEEP There are many different sleep disorders, and almost all of them can be improved with lifestyle changes. HEALTHY LIFESTYLE, HEALTHY SLEEP There are many different sleep
HEALTHY LIFESTYLE, HEALTHY SLEEP There are many different sleep disorders, and almost all of them can be improved with lifestyle changes. HEALTHY LIFESTYLE, HEALTHY SLEEP There are many different sleep
Checklist for Completion of Training Requirements in Sleep Medicine Pathway 2
 Checklist for Completion of Training Requirements in Sleep Medicine Pathway 2 This checklist follows the mandatory components of training in sleep medicine for physicians who have followed Pathway 2. Please
Checklist for Completion of Training Requirements in Sleep Medicine Pathway 2 This checklist follows the mandatory components of training in sleep medicine for physicians who have followed Pathway 2. Please
Prior Authorization Opioid Overutilization 2017
 Drugs Requiring Prior Authorization Label Name ACETAMINOPHEN/CAFFEINE/DIHYDROCODEINE CAPSULE ACETAMINOPHEN/CODEINE SOLUTION ACETAMINOPHEN/CODEINE TABLET ASCOMP/CODEINE CAPSULE BUTALBITAL/CAFFEINE/ACETAMINOPHEN/CODEINE
Drugs Requiring Prior Authorization Label Name ACETAMINOPHEN/CAFFEINE/DIHYDROCODEINE CAPSULE ACETAMINOPHEN/CODEINE SOLUTION ACETAMINOPHEN/CODEINE TABLET ASCOMP/CODEINE CAPSULE BUTALBITAL/CAFFEINE/ACETAMINOPHEN/CODEINE
Rpsgt, RST, CSE. Subtitle Here. Clinical Coordinator Crittenton Hospital Sleep Center Decoding Prior Authorization in Your Practice
 Lorraine Title Here Brierley Rpsgt, RST, CSE Subtitle Here Clinical Coordinator Crittenton Hospital Sleep Center Decoding Prior Authorization in Your Practice x YOU CAN MAKE A DIFFERENCE At the conclusion
Lorraine Title Here Brierley Rpsgt, RST, CSE Subtitle Here Clinical Coordinator Crittenton Hospital Sleep Center Decoding Prior Authorization in Your Practice x YOU CAN MAKE A DIFFERENCE At the conclusion
Icd 10 for cirrhosis with ascites
 Icd 10 for cirrhosis with ascites ICD-10 -CM Index; ICD - 10 -CM External Causes Index;. 2018 ICD-10 -CM Index. Alcoholic cirrhosis of liver with ascites. 1-10-2017 2018 ICD-10 -CM Diagnosis Code R18..
Icd 10 for cirrhosis with ascites ICD-10 -CM Index; ICD - 10 -CM External Causes Index;. 2018 ICD-10 -CM Index. Alcoholic cirrhosis of liver with ascites. 1-10-2017 2018 ICD-10 -CM Diagnosis Code R18..
Index. sleep.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Actigraphy, 475, 485, 496 Adolescents, sleep disorders in, 576 578 Adults, sleep disorders in, 578 580 Advanced sleep phase disorder, 482 Age,
Note: Page numbers of article titles are in boldface type. A Actigraphy, 475, 485, 496 Adolescents, sleep disorders in, 576 578 Adults, sleep disorders in, 578 580 Advanced sleep phase disorder, 482 Age,
Cystic Fibrosis Agents
 Texas Prior Authorization Program Clinical Criteria Clinical Information Included in this Document Kalydeco (Ivacaftor) Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Criteria Clinical Information Included in this Document Kalydeco (Ivacaftor) Drugs requiring prior authorization: the list of drugs requiring prior authorization
Cough/Cold Medications
 Texas Prior Authorization Program Clinical Edit Criteria Cough/Cold Medications NOTE: Claims for cough and cold products containing acetaminophen, ibuprofen, or hydrocodone are not covered by Texas Medicaid
Texas Prior Authorization Program Clinical Edit Criteria Cough/Cold Medications NOTE: Claims for cough and cold products containing acetaminophen, ibuprofen, or hydrocodone are not covered by Texas Medicaid
Texas Prior Authorization Program Clinical Edit Criteria
 Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
Texas Prior Authorization Program Clinical Edit Criteria Drug/Drug Class Clinical Edit Information Included in this Document Drugs requiring prior authorization: the list of drugs requiring prior authorization
CHAPTER 1. Alcoholic Liver Disease
 CHAPTER 1 Alcoholic Liver Disease Major Lesions of Alcoholic Liver Disease Alcoholic fatty liver - >90% of binge and chronic drinkers Alcoholic hepatitis precursor of cirrhosis Alcoholic cirrhosis end
CHAPTER 1 Alcoholic Liver Disease Major Lesions of Alcoholic Liver Disease Alcoholic fatty liver - >90% of binge and chronic drinkers Alcoholic hepatitis precursor of cirrhosis Alcoholic cirrhosis end
TOPIC: Continuing Coverage of CPAP Machines and Supplies for the Treatment of Obstructive Sleep Apnea
 These documents are not used to determine benefits or reimbursement. Please reference the appropriate certificate or contract for benefit information. BLUE CROSS BLUE SHIELD of MI MEDICAL POLICY Enterprise:
These documents are not used to determine benefits or reimbursement. Please reference the appropriate certificate or contract for benefit information. BLUE CROSS BLUE SHIELD of MI MEDICAL POLICY Enterprise:
PITFALLS IN THE DIAGNOSIS OF MEDICAL LIVER DISEASE WITH TWO CONCURRENT ETIOLOGIES I HAVE NOTHING TO DISCLOSE CURRENT ISSUES IN ANATOMIC PATHOLOGY 2017
 CURRENT ISSUES IN ANATOMIC PATHOLOGY 2017 I HAVE NOTHING TO DISCLOSE Linda Ferrell PITFALLS IN THE DIAGNOSIS OF MEDICAL LIVER DISEASE WITH TWO CONCURRENT ETIOLOGIES Linda Ferrell, MD, UCSF THE PROBLEM
CURRENT ISSUES IN ANATOMIC PATHOLOGY 2017 I HAVE NOTHING TO DISCLOSE Linda Ferrell PITFALLS IN THE DIAGNOSIS OF MEDICAL LIVER DISEASE WITH TWO CONCURRENT ETIOLOGIES Linda Ferrell, MD, UCSF THE PROBLEM
End stage liver disease icd-10 code
 End stage liver disease icd-10 code Sebastiani and Alberti (2012) chronic hepatitis C represents a major cause of progressive liver disease that can eventually evolve into cirrhosis and its end - stage.
End stage liver disease icd-10 code Sebastiani and Alberti (2012) chronic hepatitis C represents a major cause of progressive liver disease that can eventually evolve into cirrhosis and its end - stage.
 SLEEP DISORDERS IN WOMEN FROM MENARCHE THROUGH PREGNANCY TO MENOPAUSE A GUIDE FOR PRACTICAL MANAGEMENT CURRENT CLINICAL NEUROLOGY ALIENIST AND NEUROLOGIST A QUARTERLY JOURNAL OF SCIENTIFIC CLINICAL AND
SLEEP DISORDERS IN WOMEN FROM MENARCHE THROUGH PREGNANCY TO MENOPAUSE A GUIDE FOR PRACTICAL MANAGEMENT CURRENT CLINICAL NEUROLOGY ALIENIST AND NEUROLOGIST A QUARTERLY JOURNAL OF SCIENTIFIC CLINICAL AND
Gastrointestinal System: Accessory Organ Disorders
 Gastrointestinal System: Accessory Organ Disorders Mary DeLetter, PhD, RN Associate Professor Dept. of Baccalaureate and Graduate Nursing Eastern Kentucky University Disorders of Accessory Organs Portal
Gastrointestinal System: Accessory Organ Disorders Mary DeLetter, PhD, RN Associate Professor Dept. of Baccalaureate and Graduate Nursing Eastern Kentucky University Disorders of Accessory Organs Portal
ICD-10. An Introduction
 ICD-10 An Introduction ICD-10 Worldwide Other countries have been using ICD-10 for many years. Coding professionals there were better prepared with: Human Anatomy and Physiology Understanding of disease
ICD-10 An Introduction ICD-10 Worldwide Other countries have been using ICD-10 for many years. Coding professionals there were better prepared with: Human Anatomy and Physiology Understanding of disease
Fabrice Czarnecki, M.D., M.A., M.P.H., FACOEM I have no disclosures to make.
 Maryland College of Occupational and Environmental Medicine April 2014 Fabrice Czarnecki, M.D., M.A., M.P.H., FACOEM fczarnecki@gablesgroup.com I have no disclosures to make. Guidelines 2006: Joint Task
Maryland College of Occupational and Environmental Medicine April 2014 Fabrice Czarnecki, M.D., M.A., M.P.H., FACOEM fczarnecki@gablesgroup.com I have no disclosures to make. Guidelines 2006: Joint Task
LIVER SPECIALTY CONFERENCE USCAP Maha Guindi, M.D. Clinical Professor of Pathology Cedars-Sinai Medical Center Los Angeles, CA
 LIVER SPECIALTY CONFERENCE USCAP 2016 Maha Guindi, M.D. Clinical Professor of Pathology Cedars-Sinai Medical Center Los Angeles, CA Nothing to disclose Case History 47-year-old male, long standing ileal
LIVER SPECIALTY CONFERENCE USCAP 2016 Maha Guindi, M.D. Clinical Professor of Pathology Cedars-Sinai Medical Center Los Angeles, CA Nothing to disclose Case History 47-year-old male, long standing ileal
Pharmacy Medical Policy CNS Stimulants and Psychotherapeutic Agents
 Pharmacy Medical Policy CNS Stimulants and Psychotherapeutic Agents Table of Contents Policy: Commercial Policy History Forms Policy: Medicare Information Pertaining to All Policies Coding Information
Pharmacy Medical Policy CNS Stimulants and Psychotherapeutic Agents Table of Contents Policy: Commercial Policy History Forms Policy: Medicare Information Pertaining to All Policies Coding Information
Nash with cirrhosis icd 10
 Free, official coding info for 2018 ICD-10-CM K75.81 - includes detailed crosswalks, DRG grouping and more. Free, official coding info for 2018 ICD-10-CM K74.69 - includes detailed crosswalks, DRG grouping
Free, official coding info for 2018 ICD-10-CM K75.81 - includes detailed crosswalks, DRG grouping and more. Free, official coding info for 2018 ICD-10-CM K74.69 - includes detailed crosswalks, DRG grouping
Understanding your FibroScan Results
 PATIENT & CAREGIVER EDUCATION Understanding your FibroScan Results This information will help you understand your FibroScan results. About FibroScan FibroScan is a specialized ultrasound machine for your
PATIENT & CAREGIVER EDUCATION Understanding your FibroScan Results This information will help you understand your FibroScan results. About FibroScan FibroScan is a specialized ultrasound machine for your
Coding restrictive lung disease icd 10
 Progressive muscular atrophy; amyotrophic lateral sclerosis; Restrictive lung mechanics due to als. 1-10-2017 Free, official coding info for 2018 ICD - 10 -CM G12.21 - includes detailed rules, notes, synonyms,
Progressive muscular atrophy; amyotrophic lateral sclerosis; Restrictive lung mechanics due to als. 1-10-2017 Free, official coding info for 2018 ICD - 10 -CM G12.21 - includes detailed rules, notes, synonyms,
Diagnosis and treatment of sleep disorders
 Diagnosis and treatment of sleep disorders Normal human sleep Sleep cycle occurs about every 90 minutes, approximately 4-6 cycles occur per major sleep episode NREM (70-80%) slow wave sleep heart rate,
Diagnosis and treatment of sleep disorders Normal human sleep Sleep cycle occurs about every 90 minutes, approximately 4-6 cycles occur per major sleep episode NREM (70-80%) slow wave sleep heart rate,
UPDATES IN SLEEP APNEA:
 UPDATES IN SLEEP APNEA: CPAP,CPAP COMPLIANCE, & ALTERNATIVES MICHELLE ZETOONY, DO, FCCP, FACOI BOARD CERTIFIED PULMONARY, CRITICAL CARE, SLEEP & INTERNAL MEDICINE CLEARWATER, FL DISCLOSURE I have no conflicts
UPDATES IN SLEEP APNEA: CPAP,CPAP COMPLIANCE, & ALTERNATIVES MICHELLE ZETOONY, DO, FCCP, FACOI BOARD CERTIFIED PULMONARY, CRITICAL CARE, SLEEP & INTERNAL MEDICINE CLEARWATER, FL DISCLOSURE I have no conflicts
Transplant Hepatology
 Transplant Hepatology Certification Examination Blueprint Purpose of the exam The exam is designed to evaluate the knowledge, diagnostic reasoning, and clinical judgment skills expected of the certified
Transplant Hepatology Certification Examination Blueprint Purpose of the exam The exam is designed to evaluate the knowledge, diagnostic reasoning, and clinical judgment skills expected of the certified
Gastroenterology. Certification Examination Blueprint. Purpose of the exam
 Gastroenterology Certification Examination Blueprint Purpose of the exam The exam is designed to evaluate the knowledge, diagnostic reasoning, and clinical judgment skills expected of the certified gastroenterologist
Gastroenterology Certification Examination Blueprint Purpose of the exam The exam is designed to evaluate the knowledge, diagnostic reasoning, and clinical judgment skills expected of the certified gastroenterologist
Sleep Medicine Rotation Resident Goals and Objectives
 Sleep Medicine Rotation Resident Goals and Objectives Contact: Location: Parking: Arrival Time: Robyn Beard 747-7482 Sleep Center Center 40, Suite 600, 1600 S. Brentwood Blvd Behind the Center 40 building;
Sleep Medicine Rotation Resident Goals and Objectives Contact: Location: Parking: Arrival Time: Robyn Beard 747-7482 Sleep Center Center 40, Suite 600, 1600 S. Brentwood Blvd Behind the Center 40 building;
