Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
|
|
|
- Adela Hill
- 5 years ago
- Views:
Transcription
1 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease Disease alternative names please provide any alternative names you wish listed Disease please provide a brief description of the disease characteristics Disease - mode of inheritance Gene name(s) OMIM number for gene(s) Gene alternative names please provide any alternative names you wish listed Gene description(s) (including number of amplicons). Mutational spectrum for which you test including details of known common mutations. Technical Method (s) Validation Process Note: please eplain how this test has been validated for use in your laboratory Are you providing this test already? If yes, how many reports have you produced? Please give the number of mutation positive/negative samples you have reported For how long have you been providing this service? Progressive familial intrahepatic cholestasis PFIC3; MDR3 deficiency; cholestasis, progressive familial intrahepatic, with elevated serum gammaglutamyltransferase Progressive familial intrahepatic cholestasis type 3 is an autosomal recessive liver disorder characterized by early onset of cholestasis that progresses to hepatic fibrosis, cirrhosis, and end-stage liver disease in childhood. Autosomal recessive ABCB ATP-binding cassette, subfamily B, member 11; P- glycoprotein 3; PGY3; multidrug resistance 3; MDR3 ABCB4-26 amplicons Nonsense, missense, frameshift, small indels, splice site mutations Direct sequencing Direct sequencing in high throughput format used in laboratory for many conditions. Analysis of sequencing by mutation surveyor validated on a panel of 30 patients compared to manual methods. Primers all SNP checked using Manchester website. New amplicons tested using at least 8 normal controls to check quality and identity of sequences produced. Yes If Yes: Number of reports issued: 21 Number of reports mutation positive: 5(all of these were variants of unknown clinical significance) Number of reports mutation negative: 16 Since Jan
2 Is there specialised local clinical/research epertise for this disease? Yes Please provide details Are you testing for other genes/diseases closely allied to this one? Please give details Your Current Activity If applicable - How many tests do you currently provide annually in your laboratory? Your Capacity if Gene Dossier approved How many tests will you be able to provide annually in your laboratory if this gene dossier is approved and recommended for NHS funding? Based on eperience how many tests will be required nationally (UK wide)? National Activity (England, Scotland, Wales & Northern Ireland) If your laboratory is unable to provide the full national need please could you provide information on how the national requirement may be met. For eample, are you aware of any other labs (UKGTN members or otherwise) offering this test to NHS patients on a local area basis only? This question has been included In order to gauge if there could be any issues in equity of access for NHS patients. It is appreciated that some laboratories may not be able to answer this question. If this is the case please write unknown. Dr Jane Hartley Consultant hepatologist, Birmingham Children s Hospital Childhood liver diseases PFIC type 1, PFIC type 2, Niemann Pick type C, ARC, NICCD (Citrin deficiency) Inde cases: 21 Family members where mutation is known: 2 (Based on 2010 only) Inde cases: Family members where mutation is known: 100 Annual audit of liver activity (with review of 3 previous years) estimates the number of children who may require genetic testing for PFIC is approimately 32 per year This is based on the eperience and figures from the Liver Unit at Birmingham Children s Hospital which is one of the main centres for treatment of liver disease in children. Testing is also carried out at Kings laboratory is not listed on UKGTN. Laboratory is a research lab but they do issue reports. It is not clear how many samples they can accept. 2
3 Epidemiology Estimated prevalence of disease in the general UK population Estimated gene frequency (Carrier frequency or allele frequency) Estimated penetrance Target Population Description of the population to which this test will apply (i.e. description of the population as defined by the minimum criteria listed in the testing criteria) Estimated prevalence of disease in the target population The true incidence of PFIC (including type 3) is not precisely known, but PFIC is considered a rare disease with an estimated incidence of 1/50,000 to 1/100,000 births. All types of PFIC eist worldwide. Both sees seem to be equally affected. Davit-Spraul et al Progressive familial intrahepatic cholestasis Orphanet Journal of Rare Diseases 2009, 4:1 Carrier frequency is unknown (Gene Clinics) Unknown but presumed high Infants with intrahepatic cholestasis, pruritis or diarrhoea. Older children or adults with episodes of uneplained cholestasis because ABCB4 is also associated with a milder disease called Benign Recurrent Intrahepatic Cholestasis type 3 (BRIC3). PFIC (all types) estimated to be 10-15% Davit-Spraul et al Progressive familial intrahepatic cholestasis Orphanet Journal of Rare Diseases 2009, 4:1 Intended Use (Please use the questions in Anne A to inform your answers) Please tick the relevant clinical purpose of testing Diagnosis Treatment Prognosis & Management Presymptomatic testing Risk Assessment for family members Risk Assessment prenatal testing YES NO 3
4 Test Characteristics Analytical sensitivity and specificity This should be based on your own laboratory data for the specific test being applied for or the analytical sensitivity and specificity of the method/technique to be used in the case of a test yet to be set up. Clinical sensitivity and specificity of test in target population The clinical sensitivity of a test is the probability of a positive test result when disease is known to be present; the clinical specificity is the probability of a negative test result when disease is known to be absent. The denominator in this case is the number with the disease (for sensitivity) or the number without disease (for specificity) Clinical validity (positive and negative predictive value in the target population) The clinical validity of a genetic test is a measure of how well the test predicts the presence or absence of the phenotype, clinical disease or predisposition. It is measured by its positive predictive value (the probability of getting the disease given a positive test) and negative predictive value (the probability of not getting the disease given a negative test). Testing pathway Please include your testing strategy if more than one gene will be tested and data on the epected proportions of positive results for each part of the process. Please illustrate this with a flow diagram. This can be added to the document as a separate sheet if necessary. Clinical utility of test in target population (Please refer to Appendi A) Please provide a description of the clinical care pathway. Sensitivity of sequencing is >99%. This test will not detect deletions or duplications in the ABCB4 gene; the fraction of PFIC3 patients this affects is unknown at present. Sensitivity 26% Specificity >99% ABCB4 sequencing in infants would only be carried out in those with cholestasis and high GGT. This clinical phenotype is indistinguishable from many other causes of neonatal cholestasis. Liver biopsy is also non-specific with portal inflammation and ductular proliferation. The only method of identifying PFIC3 accurately is through mutation identification and therefore in this population the positive and negative predictive value is 100%. In other populations such as those with intrahepatic cholestasis in pregnancy this is not so specific in that the clinical phenotype can arise without mutations in ABCB4. The positive and negative predictive value therefore in the adult population cannot be quantified. Neonatal cholestasis with high GGT and normal colour stool Normal ultrasound scan to eclude obstructive cause Alpha 1 antitrypsin deficiency, metabolic, infective, and endocrine causes ecluded by blood and urine sampling Liver biopsy: non-specific inflammation with ductular proliferation Refer for ABCB4 sequencing Cholestasis in infancy is the presenting feature of many liver disorders therefore making a diagnosis can be difficult. Often there is a prolonged period between presentation and making the diagnosis leading to multiple hospital admissions and invasive procedures such as liver biopsies. Confirming the diagnosis by identifying mutations in ABCB4 facilitates clinical management and enables accurate counselling of families as to the likely 4
5 How will the test add to the management of the patient or alter clinical outcome? What impact will this test have on the NHS i.e. by removing the need for alternative management and/or investigations for this clinical population? Please provide evidence from your own service. What are the consequences of not doing this genetic test. Commissioners have asked for specific information to support introduction of tests. Utility of test in the NHS In a couple of sentences eplain the utility of this test for the disease(s) Is there an alternative means of diagnosis or prediction that does not involve molecular diagnosis? If so (and in particular if there is a biochemical test) please state the added advantage of the molecular test Please describe any specific ethical, legal or social issues with this particular test? long-term prognosis. Children with PFIC3 require intensive support with high calorie medium chain triglyceride feeds often by nasogastric feeding and fat-soluble vitamin supplementation. The infants are often etremely pruritic leading to a poor quality of life for all the family and requiring multiple medications, which are often poorly effective. Most children with PFIC3 will require liver transplant in childhood as a life saving procedure. The etrahepatic manifestations (diarrhoea and pancreatitis) may only become manifest following transplantation. By providing a diagnosis intensive management to support the liver can be instigated prior to the development of complications. Invasive investigations will not be required and repeat investigations, which may be carried out when there is diagnostic uncertainty (such as liver biopsy, which is associated with morbidity and mortality), can be avoided. The clinical management of the child is optimised at an earlier stage and the essential counselling of the families for timely biliary diversion surgery or liver transplantation is facilitated. The test will reduce the number of admissions to hospital. It will reduce the number of children who develop complications which have developed due to not maimising the supportive management. It will facilitate timing of transplantation i.e. the diagnosis of PFIC1 means the child is likely to need transplantation and therefore the timing can be planned. Without a diagnosis there may be a delay. Diagnostic uncertainty leading to reduced input of intensive nutritional support, repeated hospitalisation for invasive procedures such as liver biopsy, inability to be able to prognosticate and therefore provide advice as to timing of biliary diversion or transplantation. The identification of mutations facilitates optimal medical management to support the liver and listing for liver transplant at the best possible time. There are no alternative methods of accurate diagnosing PFIC3 5
6 UKGTN Testing Criteria Name of Disease(s): CHOLESTASIS, PROGRESSIVE FAMILIAL INTRAHEPATIC, 3; PFIC3 (602347) Name of gene(s): ATP-binding cassette, sub-family B (MDR/TAP), member 4; ABCB4 (171060) Patient name: Patient postcode: Date of birth: NHS number: Name of referrer: Title/Position: Lab ID: Referrals will only be accepted from one of the following: Referrer Consultant Liver Specialist Consultant Hepatology Paediatrician Consultant Clinical Geneticist Tick if this refers to you. Minimum criteria required for testing to be appropriate as stated in the Gene Dossier: Criteria Cholestasis associated with raised gamma glutamyl transferase (GGT) At risk family members with known mutation Tick if this patient meets criteria If the sample does not fulfil the clinical criteria or you are not one of the specified types of referrer and you still feel that testing should be performed please contact the laboratory to discuss testing of the sample. 6
7 Neonatal cholestasis Low GGT High GGT Diarrhoea Pancreatitis Sensorineural deafness ATP8B1 ABCB11 If no mutations identified Contractures Renal dysfunction VPS33B If no mutations identified Low birth weight Poor weight gain Hepatomegaly Haemolytic anaemia Hypoglycaemia Hypoproteinaemia SLC25A13 Hepatosplenomegaly Neonatal hepatitis (can be severe) Older child: Supranuclear vertical gaze palsy Gelastic catapley Neurodegeneration VIPAR NPC1 ABCB4 If no mutations identified NPC2 7
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Epileptic encephalopathy, early infantile 4. OMIM number for disease 612164 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Epileptic encephalopathy, early infantile 4. OMIM number for disease 612164 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name HEMOCHROMATOSIS, TYPE 4; HFE4 OMIM number for disease #606069 Disease alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name HEMOCHROMATOSIS, TYPE 4; HFE4 OMIM number for disease #606069 Disease alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Leber congenital amaurosis OMIM number for disease 204000 Disease alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Leber congenital amaurosis OMIM number for disease 204000 Disease alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Genetic Causes of Hypothyroidism 1. Loss of function mutations in TSHR cause thyroid
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Genetic Causes of Hypothyroidism 1. Loss of function mutations in TSHR cause thyroid
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Choroideremia OMIM number for disease 303100 Disease alternative names please
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Choroideremia OMIM number for disease 303100 Disease alternative names please
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Parkinson disease 8, automsomal dominant OMIM number for disease 607060 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Parkinson disease 8, automsomal dominant OMIM number for disease 607060 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 147920 Disease alternative names Please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 147920 Disease alternative names Please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Amyotrophic Lateral Sclerosis 10 (ALS10) and Amyotrophic Lateral Sclerosis 6 (ALS6)
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Amyotrophic Lateral Sclerosis 10 (ALS10) and Amyotrophic Lateral Sclerosis 6 (ALS6)
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 231300 Disease alternative names Please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 231300 Disease alternative names Please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: Cambridge RGC Approved: September 2012
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: Cambridge RGC Approved: September 2012
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names Osteogenesis Imperfecta
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names Osteogenesis Imperfecta
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Loeys-Dietz Syndrome OMIM number for disease 609192; 608967; 610380; 610168 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Loeys-Dietz Syndrome OMIM number for disease 609192; 608967; 610380; 610168 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISORDER/CONDITION POPULATION TRIAD Submitting laboratory: Exeter RGC Approved: Sept 2013 1. Disorder/condition
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISORDER/CONDITION POPULATION TRIAD Submitting laboratory: Exeter RGC Approved: Sept 2013 1. Disorder/condition
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) Glucocorticoid-remediable
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) Glucocorticoid-remediable
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: Birmingham RGC Approved: September 2012
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: Birmingham RGC Approved: September 2012
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Application to be an additional provider for existing test on the NHS Directory of Molecular Genetic Testing Additional Provider form
 Application to be an additional provider for existing test on the NHS Directory of Molecular Genetic Testing Additional Provider form Disease: Gene: Cystic Fibrosis (CF) (carrier testing in reproductive
Application to be an additional provider for existing test on the NHS Directory of Molecular Genetic Testing Additional Provider form Disease: Gene: Cystic Fibrosis (CF) (carrier testing in reproductive
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: London North East RGC GOSH Approved: September
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: London North East RGC GOSH Approved: September
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Polycystic Kidney Disease, Autosomal Dominant OMIM number for disease 173900 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Polycystic Kidney Disease, Autosomal Dominant OMIM number for disease 173900 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 312060 Disease alternative names please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 312060 Disease alternative names please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: Manchester RGC Approved: September 2013
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: Manchester RGC Approved: September 2013
Submitting Laboratory: London NE RGC GOSH
 Submitting laboratory: London NE RGC GOSH 1. Disorder/condition approved name (please provide UK spelling if different from US) and symbol as published on the OMIM database (alternative names will be listed
Submitting laboratory: London NE RGC GOSH 1. Disorder/condition approved name (please provide UK spelling if different from US) and symbol as published on the OMIM database (alternative names will be listed
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: London North East RGC GOSH Approved: September
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: London North East RGC GOSH Approved: September
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: Birmingham RGC 1. Disorder/condition approved name (please provide UK spelling
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: Birmingham RGC 1. Disorder/condition approved name (please provide UK spelling
PROGRESSIVE FAMILIAL INTRAHEPATIC CHOLESTASIS (PFIC)
 The Childhood Liver Disease Research Network strives to provide information and support to individuals and families affected by liver disease through its many research programs. PROGRESSIVE FAMILIAL INTRAHEPATIC
The Childhood Liver Disease Research Network strives to provide information and support to individuals and families affected by liver disease through its many research programs. PROGRESSIVE FAMILIAL INTRAHEPATIC
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISORDER/CONDITION POPULATION TRIAD Submitting laboratory: Cardiff SAS Porphyria Approved: Sept
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISORDER/CONDITION POPULATION TRIAD Submitting laboratory: Cardiff SAS Porphyria Approved: Sept
Dhanpat Jain Yale University School of Medicine, New Haven, CT
 Dhanpat Jain Yale University School of Medicine, New Haven, CT Case history 15 years old female presented with fatigue. Found to have features suggestive of cirrhosis with esophageal varices, splenomegaly
Dhanpat Jain Yale University School of Medicine, New Haven, CT Case history 15 years old female presented with fatigue. Found to have features suggestive of cirrhosis with esophageal varices, splenomegaly
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Submitting laboratory: Bristol RGC 1. Disorder/condition approved name and symbol as published on the OMIM database (alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Submitting laboratory: Bristol RGC 1. Disorder/condition approved name and symbol as published on the OMIM database (alternative
Metabolic Liver Disease
 Metabolic Liver Disease Peter Eichenseer, MD No relationships to disclose. Outline Overview Alpha-1 antitrypsin deficiency Wilson s disease Hereditary hemochromatosis Pathophysiology Clinical features
Metabolic Liver Disease Peter Eichenseer, MD No relationships to disclose. Outline Overview Alpha-1 antitrypsin deficiency Wilson s disease Hereditary hemochromatosis Pathophysiology Clinical features
2. Liver blood tests and what they mean p2 Acute and chronic liver screen
 Hepatology referral pathways for GP 1 Scope For use within hepatology Contents 2. Liver blood tests and what they mean p2 Acute and chronic liver screen p2 Common reasons for hepatology referral 3. Raised
Hepatology referral pathways for GP 1 Scope For use within hepatology Contents 2. Liver blood tests and what they mean p2 Acute and chronic liver screen p2 Common reasons for hepatology referral 3. Raised
UKGTN Testing Criteria
 Test name: Neonatal Diabetes 22 Gene Panel UKGTN Testing Criteria Approved name and symbol of disorder/condition(s): See Appendix 1 Approved name and symbol of gene(s): See Appendix 1 number(s): number(s):
Test name: Neonatal Diabetes 22 Gene Panel UKGTN Testing Criteria Approved name and symbol of disorder/condition(s): See Appendix 1 Approved name and symbol of gene(s): See Appendix 1 number(s): number(s):
Approach to a case of Neonatal Cholestasis
 Approach to a case of Neonatal Cholestasis Ira Shah (Co-Incharge, Consultant Pediatrician, Pediatric Liver Clinic) Gunjan Narkhede (Resident in Pediatric Liver Clinic) Pediatric Hepatobiliary Clinic, B.J.Wadia
Approach to a case of Neonatal Cholestasis Ira Shah (Co-Incharge, Consultant Pediatrician, Pediatric Liver Clinic) Gunjan Narkhede (Resident in Pediatric Liver Clinic) Pediatric Hepatobiliary Clinic, B.J.Wadia
General Guidelines for Health professionals
 General Guidelines for Health professionals Page 1 Haemochromatosis Introduction Hereditary haemochromatosis (HH) now easily screened for as most symptomatic individuals are homozygous for the C282Y mutation
General Guidelines for Health professionals Page 1 Haemochromatosis Introduction Hereditary haemochromatosis (HH) now easily screened for as most symptomatic individuals are homozygous for the C282Y mutation
CURRENT GENETIC TESTING TOOLS IN NEONATAL MEDICINE. Dr. Bahar Naghavi
 2 CURRENT GENETIC TESTING TOOLS IN NEONATAL MEDICINE Dr. Bahar Naghavi Assistant professor of Basic Science Department, Shahid Beheshti University of Medical Sciences, Tehran,Iran 3 Introduction Over 4000
2 CURRENT GENETIC TESTING TOOLS IN NEONATAL MEDICINE Dr. Bahar Naghavi Assistant professor of Basic Science Department, Shahid Beheshti University of Medical Sciences, Tehran,Iran 3 Introduction Over 4000
Neonatal Cholestasis. What is Cholestasis? Congenital and Pediatric liver diseases 4/26/18
 Congenital and Pediatric liver diseases Nitika Gupta, M.D. Personal/Professional Financial Relationships with Industry in the past year External Industry Relationships * Equity, stock, or options in biomedical
Congenital and Pediatric liver diseases Nitika Gupta, M.D. Personal/Professional Financial Relationships with Industry in the past year External Industry Relationships * Equity, stock, or options in biomedical
Natural history of α-1-atd in children
 Natural history of α-1-atd in children Agnieszka Bakuła Dpt of Gastroenterology, Hepatology, Nutrition Disorders and Paediatrics The Children s Memorial Health Institute Warsaw, Poland Topics to be discussed
Natural history of α-1-atd in children Agnieszka Bakuła Dpt of Gastroenterology, Hepatology, Nutrition Disorders and Paediatrics The Children s Memorial Health Institute Warsaw, Poland Topics to be discussed
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISORDER/CONDITION POPULATION TRIAD Submitting laboratory: Bristol RGC Approved: Sept 2013 1. Disorder/condition
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISORDER/CONDITION POPULATION TRIAD Submitting laboratory: Bristol RGC Approved: Sept 2013 1. Disorder/condition
CYSTIC FIBROSIS. The condition:
 CYSTIC FIBROSIS Both antenatal and neonatal screening for CF have been considered. Antenatal screening aims to identify fetuses affected by CF so that parents can be offered an informed choice as to whether
CYSTIC FIBROSIS Both antenatal and neonatal screening for CF have been considered. Antenatal screening aims to identify fetuses affected by CF so that parents can be offered an informed choice as to whether
2. Liver blood tests and what they mean p2 Acute and chronic liver screen
 1 Scope For use within hepatology Contents 2. Liver blood tests and what they mean p2 Acute and chronic liver screen p2 Common reasons for referral 3. Raised ALT +/- GGT p3 4. Non alcoholic fatty liver
1 Scope For use within hepatology Contents 2. Liver blood tests and what they mean p2 Acute and chronic liver screen p2 Common reasons for referral 3. Raised ALT +/- GGT p3 4. Non alcoholic fatty liver
NGS Types of gene dossier applications UKGTN can evaluate
 NGS Types of gene dossier applications UKGTN can evaluate Jo Whittaker on behalf of the Genetic Test Evaluation Working Group & UKGTN Project team NGS used in a number of ways Replacement technology for
NGS Types of gene dossier applications UKGTN can evaluate Jo Whittaker on behalf of the Genetic Test Evaluation Working Group & UKGTN Project team NGS used in a number of ways Replacement technology for
THALASSEMIA AND COMPREHENSIVE CARE
 1 THALASSEMIA AND COMPREHENSIVE CARE Melanie Kirby MBBS, FRCP (C), Hospital for Sick Children, Toronto Associate Professor of Paediatrics, University of Toronto. Objectives 2 By the end of this presentation,
1 THALASSEMIA AND COMPREHENSIVE CARE Melanie Kirby MBBS, FRCP (C), Hospital for Sick Children, Toronto Associate Professor of Paediatrics, University of Toronto. Objectives 2 By the end of this presentation,
Biliary Atresia. Who is at risk for biliary atresia?
 Biliary Atresia Biliary atresia is a life-threatening condition in infants in which the bile ducts inside or outside the liver do not have normal openings. Bile ducts in the liver, also called hepatic
Biliary Atresia Biliary atresia is a life-threatening condition in infants in which the bile ducts inside or outside the liver do not have normal openings. Bile ducts in the liver, also called hepatic
Biliary Atresia. Karen F. Murray, MD Professor of Pediatrics Director, Hepatobiliary Program Seattle Children s
 Biliary Atresia Karen F. Murray, MD Professor of Pediatrics Director, Hepatobiliary Program Seattle Children s Biliary Atresia Incidence: 1/8,000-15,000 live births Girls > boys 1.5:1 The most common cause
Biliary Atresia Karen F. Murray, MD Professor of Pediatrics Director, Hepatobiliary Program Seattle Children s Biliary Atresia Incidence: 1/8,000-15,000 live births Girls > boys 1.5:1 The most common cause
Interpreting Liver Function Tests
 PSH Clinical Guidelines Statement 2017 Interpreting Liver Function Tests Dr. Asad A Chaudhry Consultant Hepatologist, Chaudhry Hospital, Gujranwala, Pakistan. Liver function tests (LFTs) generally refer
PSH Clinical Guidelines Statement 2017 Interpreting Liver Function Tests Dr. Asad A Chaudhry Consultant Hepatologist, Chaudhry Hospital, Gujranwala, Pakistan. Liver function tests (LFTs) generally refer
Alpha-1 Antitrypsin Deficiency: Liver Disease
 Alpha-1 Antitrypsin Deficiency: Liver Disease Who is at risk to develop Alpha-1 liver disease? Alpha-1 liver disease may affect children and adults who have abnormal Alpha-1 antitrypsin genes. Keys to
Alpha-1 Antitrypsin Deficiency: Liver Disease Who is at risk to develop Alpha-1 liver disease? Alpha-1 liver disease may affect children and adults who have abnormal Alpha-1 antitrypsin genes. Keys to
Presentation of Progressive Familial Intrahepatic Cholestasis Type 3 Mimicking Wilson Disease: Molecular Genetic Diagnosis and Response to Treatment
 pissn: 2234-8646 eissn: 2234-8840 http://dx.doi.org/10.5223/pghn.2015.18.3.202 Pediatr Gastroenterol Hepatol Nutr 2015 September 18(3):202-208 Case Report PGHN Presentation of Progressive Familial Intrahepatic
pissn: 2234-8646 eissn: 2234-8840 http://dx.doi.org/10.5223/pghn.2015.18.3.202 Pediatr Gastroenterol Hepatol Nutr 2015 September 18(3):202-208 Case Report PGHN Presentation of Progressive Familial Intrahepatic
Hereditary Haemochromatosis For GPs
 Hereditary Haemochromatosis For GPs What is Hereditary Haemochromatosis? Hereditary Haemochromatosis () is a common autosomal recessive disease resulting in excessive absorption of dietary iron from the
Hereditary Haemochromatosis For GPs What is Hereditary Haemochromatosis? Hereditary Haemochromatosis () is a common autosomal recessive disease resulting in excessive absorption of dietary iron from the
Lab Activity Report: Mendelian Genetics - Genetic Disorders
 Name Date Period Lab Activity Report: Mendelian Genetics - Genetic Disorders Background: Sometimes genetic disorders are caused by mutations to normal genes. When the mutation has been in the population
Name Date Period Lab Activity Report: Mendelian Genetics - Genetic Disorders Background: Sometimes genetic disorders are caused by mutations to normal genes. When the mutation has been in the population
Fatty Acid Oxidation Disorders
 Genetic Fact Sheets for Parents Fatty Acid Oxidation Disorders Screening, Technology, and Research in Genetics is a multi-state project to improve information about the financial, ethical, legal, and social
Genetic Fact Sheets for Parents Fatty Acid Oxidation Disorders Screening, Technology, and Research in Genetics is a multi-state project to improve information about the financial, ethical, legal, and social
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: Leeds RGC 1. Disorder/condition approved name (please provide UK spelling if different
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: Leeds RGC 1. Disorder/condition approved name (please provide UK spelling if different
EAST LONDON INTEGRATED CARE
 CITY & HACKNEY ELIC EAST LONDON INTEGRATED CARE MANAGEMENT OF CHRONIC HEPATITIS B IN PRIMARY CARE Chronic Hepatitis B virus (HBV) is an important public health problem globally and a leading cause of liver
CITY & HACKNEY ELIC EAST LONDON INTEGRATED CARE MANAGEMENT OF CHRONIC HEPATITIS B IN PRIMARY CARE Chronic Hepatitis B virus (HBV) is an important public health problem globally and a leading cause of liver
CITY & HACKNEY ELIC EAST LONDON INTEGRATED CARE MANAGEMENT OF CHRONIC HEPATITIS B IN PRIMARY CARE
 CITY & HACKNEY ELIC EAST LONDON INTEGRATED CARE MANAGEMENT OF CHRONIC HEPATITIS B IN PRIMARY CARE Chronic Hepatitis B virus (HBV) is an important public health problem globally and a leading cause of liver
CITY & HACKNEY ELIC EAST LONDON INTEGRATED CARE MANAGEMENT OF CHRONIC HEPATITIS B IN PRIMARY CARE Chronic Hepatitis B virus (HBV) is an important public health problem globally and a leading cause of liver
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: Sheffield RGC 1. Disorder/condition approved name (please provide UK spelling
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: Sheffield RGC 1. Disorder/condition approved name (please provide UK spelling
Progressive Familial Intrahepatic Cholestasis (PFIC)
 fighting childhood liver disease Progressive Familial Intrahepatic Cholestasis (PFIC) a guide Medical Information Series Welcome This leaflet has been written for: Parents/carers of a child with PFIC Young
fighting childhood liver disease Progressive Familial Intrahepatic Cholestasis (PFIC) a guide Medical Information Series Welcome This leaflet has been written for: Parents/carers of a child with PFIC Young
YES NO UNKNOWN PENETRANCE ACTIONABILITY SIGNIFICANCE/BURDEN OF DISEASE NEXT STEPS. YES (Proceed to Stage II) YES ( 1 of above)
 Stage I: Rule-Out Dashboard GENE/GENE PANEL: ATP7B DISORDER: Wilson Disease HGNC ID: 870 OMIM ID: 277900 ACTIONABILITY 1. Is there a qualifying resource, such as a practice guideline or systematic review,
Stage I: Rule-Out Dashboard GENE/GENE PANEL: ATP7B DISORDER: Wilson Disease HGNC ID: 870 OMIM ID: 277900 ACTIONABILITY 1. Is there a qualifying resource, such as a practice guideline or systematic review,
Form 3. Template for a full review process for a condition being considered for addition to the newborn/child screening panel
 Form 3. Template for a full review process for a condition being considered for addition to the newborn/child screening panel **Note: please specify the basis for each answer and rely on published evidence
Form 3. Template for a full review process for a condition being considered for addition to the newborn/child screening panel **Note: please specify the basis for each answer and rely on published evidence
Cryptogenic Cirrhosis: An Approach To The Diagnosis In The Era Of Molecular and Genomic Medicine
 Cryptogenic Cirrhosis: An Approach To The Diagnosis In The Era Of Molecular and Genomic Medicine Introduction and historical perspective: Cryptogenic cirrhosis(cc) is defined as cases of cirrhosis where
Cryptogenic Cirrhosis: An Approach To The Diagnosis In The Era Of Molecular and Genomic Medicine Introduction and historical perspective: Cryptogenic cirrhosis(cc) is defined as cases of cirrhosis where
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Submitting laboratory: Bristol RGC 1. Disorder/condition approved name and symbol as published on the OMIM database (alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Submitting laboratory: Bristol RGC 1. Disorder/condition approved name and symbol as published on the OMIM database (alternative
Management of Hepatitis B - Information for primary care providers
 Management of Hepatitis B - Information for primary care providers July 2018 Chronic hepatitis B (CHB) is often a lifelong condition. Not everyone infected needs anti-viral therapy. This document outlines
Management of Hepatitis B - Information for primary care providers July 2018 Chronic hepatitis B (CHB) is often a lifelong condition. Not everyone infected needs anti-viral therapy. This document outlines
Precision Medicine and Genetic Counseling : Is Yes always the correct answer?
 Precision Medicine and Genetic Counseling : Is Yes always the correct answer? Beverly M. Yashar, MS, PhD, CGC Director, Graduate Program in Genetic Counseling Professor, Department of Human Genetics. (yashar@umich.edu)
Precision Medicine and Genetic Counseling : Is Yes always the correct answer? Beverly M. Yashar, MS, PhD, CGC Director, Graduate Program in Genetic Counseling Professor, Department of Human Genetics. (yashar@umich.edu)
Survey of networks and arrangements for HIV clinical care
 1 of 6 20/08/2007 16:40 Survey of networks and arrangements for HIV clinical care Please ensure that you have read the [add link] invitation letter [/add link] before completing this survey. When you have
1 of 6 20/08/2007 16:40 Survey of networks and arrangements for HIV clinical care Please ensure that you have read the [add link] invitation letter [/add link] before completing this survey. When you have
Amino Acid Disorders. What is ASAL deficiency? Genetic Fact Sheets for Parents
 Genetic Fact Sheets for Parents Amino Acid Disorders Screening, Technology, and Research in Genetics is a multi-state project to improve information about the financial, ethical, legal, and social issues
Genetic Fact Sheets for Parents Amino Acid Disorders Screening, Technology, and Research in Genetics is a multi-state project to improve information about the financial, ethical, legal, and social issues
BILIARY ATRESIA. What is biliary atresia?
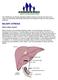 The Childhood Liver Disease Research Network strives to provide information and support to individuals and families affected by liver disease through its many research programs. BILIARY ATRESIA What is
The Childhood Liver Disease Research Network strives to provide information and support to individuals and families affected by liver disease through its many research programs. BILIARY ATRESIA What is
SETPEG GENETIC TESTING GUIDELINES Version 1.0, 5 th October 2017
 SETPEG GENETIC TESTING GUIDELINES Version 1.0, 5 th October 2017 1. The Epilepsy Genetic Diagnostic & Counselling Service at King s Health Partners Professor Deb Pal PhD MRCP (Consultant) deb.pal@nhs.net
SETPEG GENETIC TESTING GUIDELINES Version 1.0, 5 th October 2017 1. The Epilepsy Genetic Diagnostic & Counselling Service at King s Health Partners Professor Deb Pal PhD MRCP (Consultant) deb.pal@nhs.net
National Horizon Scanning Centre. Mannitol dry powder for inhalation (Bronchitol) for cystic fibrosis. April 2008
 Mannitol dry powder for inhalation (Bronchitol) for cystic fibrosis April 2008 This technology summary is based on information available at the time of research and a limited literature search. It is not
Mannitol dry powder for inhalation (Bronchitol) for cystic fibrosis April 2008 This technology summary is based on information available at the time of research and a limited literature search. It is not
How many disease-causing variants in a normal person? Matthew Hurles
 How many disease-causing variants in a normal person? Matthew Hurles Summary What is in a genome? What is normal? Depends on age What is a disease-causing variant? Different classes of variation Final
How many disease-causing variants in a normal person? Matthew Hurles Summary What is in a genome? What is normal? Depends on age What is a disease-causing variant? Different classes of variation Final
Amino Acid Disorders. Disorder name: Tyrosinemia, type 1. What is tyrosinemia 1? Genetic Fact Sheets for Parents
 Genetic Fact Sheets for Parents Amino Acid Disorders Screening, Technology, and Research in Genetics is a multi-state project to improve information about the financial, ethical, legal, and social issues
Genetic Fact Sheets for Parents Amino Acid Disorders Screening, Technology, and Research in Genetics is a multi-state project to improve information about the financial, ethical, legal, and social issues
Noncalculous Biliary Disease Dean Abramson, M.D. Gastroenterologists, P.C. Cedar Rapids. Cholestasis
 Noncalculous Biliary Disease Dean Abramson, M.D. Gastroenterologists, P.C. Cedar Rapids Cholestasis Biochemical hallmark Impaired bile flow from liver to small intestine Alkaline phosphatase is primary
Noncalculous Biliary Disease Dean Abramson, M.D. Gastroenterologists, P.C. Cedar Rapids Cholestasis Biochemical hallmark Impaired bile flow from liver to small intestine Alkaline phosphatase is primary
The University of Arizona Pediatric Residency Program. Primary Goals for Rotation. Genetics
 The University of Arizona Pediatric Residency Program Primary Goals for Rotation Genetics 1. GOAL: Understand the role of the pediatrician in preventing genetic disease, and in counseling and screening
The University of Arizona Pediatric Residency Program Primary Goals for Rotation Genetics 1. GOAL: Understand the role of the pediatrician in preventing genetic disease, and in counseling and screening
MEDICAL GENOMICS LABORATORY. Next-Gen Sequencing and Deletion/Duplication Analysis of NF1 Only (NF1-NG)
 Next-Gen Sequencing and Deletion/Duplication Analysis of NF1 Only (NF1-NG) Ordering Information Acceptable specimen types: Fresh blood sample (3-6 ml EDTA; no time limitations associated with receipt)
Next-Gen Sequencing and Deletion/Duplication Analysis of NF1 Only (NF1-NG) Ordering Information Acceptable specimen types: Fresh blood sample (3-6 ml EDTA; no time limitations associated with receipt)
THIAMINE TRANSPORTER TYPE 2 DEFICIENCY
 THIAMINE TRANSPORTER TYPE 2 DEFICIENCY WHAT IS THE THIAMINE TRANSPORTER TYPE 2 DEFICIENCY (hthtr2)? The thiamine transporter type 2 deficiency (hthtr2) is a inborn error of thiamine metabolism caused by
THIAMINE TRANSPORTER TYPE 2 DEFICIENCY WHAT IS THE THIAMINE TRANSPORTER TYPE 2 DEFICIENCY (hthtr2)? The thiamine transporter type 2 deficiency (hthtr2) is a inborn error of thiamine metabolism caused by
Autoimmune lymphoproliferative syndrome (ALPS)
 ALPS Autoimmune lymphoproliferative syndrome (ALPS) Information for families hello@piduk.org 0800 987 8986 www.piduk.org About this leaflet This leaflet is designed to help answer the questions families
ALPS Autoimmune lymphoproliferative syndrome (ALPS) Information for families hello@piduk.org 0800 987 8986 www.piduk.org About this leaflet This leaflet is designed to help answer the questions families
Fatty Acid Oxidation Disorders
 Genetic Fact Sheets for Parents Fatty Acid Oxidation Disorders Screening, Technology, and Research in Genetics is a multi-state project to improve information about the financial, ethical, legal, and social
Genetic Fact Sheets for Parents Fatty Acid Oxidation Disorders Screening, Technology, and Research in Genetics is a multi-state project to improve information about the financial, ethical, legal, and social
Guidance on format of the risk management plan (RMP) in the EU part II: Module SIV - Populations not studied in clinical trials
 25 July 2013 EMA/465927/2013 Rev.1 Patient Health Protection Guidance on format of the risk management plan (RMP) in the EU part II: Module SIV - Populations not studied in clinical trials Active substance
25 July 2013 EMA/465927/2013 Rev.1 Patient Health Protection Guidance on format of the risk management plan (RMP) in the EU part II: Module SIV - Populations not studied in clinical trials Active substance
Factor VII deficiency
 Factor VII deficiency Information for families Great Ormond Street Hospital for Children NHS Foundation Trust 1 Factor VII deficiency (also known as Alexander s disease) is a type of clotting disorder.
Factor VII deficiency Information for families Great Ormond Street Hospital for Children NHS Foundation Trust 1 Factor VII deficiency (also known as Alexander s disease) is a type of clotting disorder.
TEST INFORMATION Test: CarrierMap GEN (Genotyping) Panel: CarrierMap Expanded Diseases Tested: 311 Genes Tested: 299 Mutations Tested: 2647
 Ordering Practice Jane Smith John Smith Practice Code: 675 Miller MD 374 Broadway New York, NY 10000 Physician: Dr. Frank Miller Report Generated: 2016-02-03 DOB: 1973-02-19 Gender: Female Ethnicity: European
Ordering Practice Jane Smith John Smith Practice Code: 675 Miller MD 374 Broadway New York, NY 10000 Physician: Dr. Frank Miller Report Generated: 2016-02-03 DOB: 1973-02-19 Gender: Female Ethnicity: European
Factor X deficiency. Information for families. Great Ormond Street Hospital for Children NHS Foundation Trust
 Factor X deficiency Information for families Great Ormond Street Hospital for Children NHS Foundation Trust 1 Factor X (previously known as the Stuart-Prower factor) deficiency is a type of clotting disorder.
Factor X deficiency Information for families Great Ormond Street Hospital for Children NHS Foundation Trust 1 Factor X (previously known as the Stuart-Prower factor) deficiency is a type of clotting disorder.
Specialised Services Commissioning Policy: CP34 Circumcision for children
 Specialised Services Commissioning Policy: CP34 Circumcision for children March 2019 Version 3.0 Document information Document purpose Document name Author Policy Circumcision for Children Welsh Health
Specialised Services Commissioning Policy: CP34 Circumcision for children March 2019 Version 3.0 Document information Document purpose Document name Author Policy Circumcision for Children Welsh Health
MEDICAL GENOMICS LABORATORY. Non-NF1 RASopathy panel by Next-Gen Sequencing and Deletion/Duplication Analysis of SPRED1 (NNP-NG)
 Non-NF1 RASopathy panel by Next-Gen Sequencing and Deletion/Duplication Analysis of SPRED1 (NNP-NG) Ordering Information Acceptable specimen types: Blood (3-6ml EDTA; no time limitations associated with
Non-NF1 RASopathy panel by Next-Gen Sequencing and Deletion/Duplication Analysis of SPRED1 (NNP-NG) Ordering Information Acceptable specimen types: Blood (3-6ml EDTA; no time limitations associated with
EVALUATION OF ABNORMAL LIVER TESTS
 EVALUATION OF ABNORMAL LIVER TESTS MIA MANABAT DO PGY6 MOA 119 TH ANNUAL SPRING SCIENTIFIC CONVENTION MAY 19, 2018 EVALUATION OF ABNORMAL LIVER TESTS Review of liver enzymes vs liver function tests Clinical
EVALUATION OF ABNORMAL LIVER TESTS MIA MANABAT DO PGY6 MOA 119 TH ANNUAL SPRING SCIENTIFIC CONVENTION MAY 19, 2018 EVALUATION OF ABNORMAL LIVER TESTS Review of liver enzymes vs liver function tests Clinical
The importance of pharmacogenetics in the treatment of epilepsy
 The importance of pharmacogenetics in the treatment of epilepsy Öner Süzer and Esat Eşkazan İstanbul University, Cerrahpaşa Faculty of Medicine, Department of Pharmacology and Clinical Pharmacology Introduction
The importance of pharmacogenetics in the treatment of epilepsy Öner Süzer and Esat Eşkazan İstanbul University, Cerrahpaşa Faculty of Medicine, Department of Pharmacology and Clinical Pharmacology Introduction
A review of Cystic Fibrosis
 A review of Cystic Fibrosis Jennifer Landry md F.R.C.P.(C) Pulmonary & Critical Care Medicine McGill University Health Center Cystic Fibrosis One of the most common lethal inherited AR disorders in the
A review of Cystic Fibrosis Jennifer Landry md F.R.C.P.(C) Pulmonary & Critical Care Medicine McGill University Health Center Cystic Fibrosis One of the most common lethal inherited AR disorders in the
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice SCOPE Clinical guideline title: Hepatitis B (chronic): diagnosis and management of chronic hepatitis B in children, young
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice SCOPE Clinical guideline title: Hepatitis B (chronic): diagnosis and management of chronic hepatitis B in children, young
Thalassaemia and Abnormal Haemoglobins in Pregnancy
 1. Purpose Thalassaemias and abnormal haemoglobins are detected in approximately 4% of patients of reproductive age attending the Women's. In almost half of these cases, the abnormality is not evident
1. Purpose Thalassaemias and abnormal haemoglobins are detected in approximately 4% of patients of reproductive age attending the Women's. In almost half of these cases, the abnormality is not evident
National Metabolic Biochemistry Network Best Practice Guidelines Neonatal Jaundice in Inherited Metabolic Disorders
 Introduction National Metabolic Biochemistry Network Best Practice Guidelines Neonatal Jaundice in Inherited Metabolic Disorders Jaundice is the most common clinical sign in the neonate, arising from either
Introduction National Metabolic Biochemistry Network Best Practice Guidelines Neonatal Jaundice in Inherited Metabolic Disorders Jaundice is the most common clinical sign in the neonate, arising from either
Cystic Biliary Atresia: Why Is It Important to Distinguish this from Congenital Choledochal Cyst?
 Bahrain Medical Bulletin, Vol. 36, No. 2, June 2014 Cystic Biliary Atresia: Why Is It Important to Distinguish this from Congenital Choledochal Cyst? Hussein Ahmed Mohammed Hamdy, MRCSEd, FEBPS* Hind Mustafa
Bahrain Medical Bulletin, Vol. 36, No. 2, June 2014 Cystic Biliary Atresia: Why Is It Important to Distinguish this from Congenital Choledochal Cyst? Hussein Ahmed Mohammed Hamdy, MRCSEd, FEBPS* Hind Mustafa
Corporate Medical Policy
 Corporate Medical Policy Invasive Prenatal (Fetal) Diagnostic Testing File Name: Origination: Last CAP Review: Next CAP Review: Last Review: invasive_prenatal_(fetal)_diagnostic_testing 12/2014 3/2018
Corporate Medical Policy Invasive Prenatal (Fetal) Diagnostic Testing File Name: Origination: Last CAP Review: Next CAP Review: Last Review: invasive_prenatal_(fetal)_diagnostic_testing 12/2014 3/2018
Corporate Medical Policy
 Corporate Medical Policy Genetic Testing for Alpha Thalassemia File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_alpha_thalassemia 9/2013 7/2017 7/2018 7/2017 Description
Corporate Medical Policy Genetic Testing for Alpha Thalassemia File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_alpha_thalassemia 9/2013 7/2017 7/2018 7/2017 Description
Information for health professionals
 Introduction of a new screening test for newborn babies in Wales Newborn bloodspot screening for Medium chain acyl-coa dehydrogenase deficiency (MCADD) Newborn bloodspot screening for MCADD is being introduced
Introduction of a new screening test for newborn babies in Wales Newborn bloodspot screening for Medium chain acyl-coa dehydrogenase deficiency (MCADD) Newborn bloodspot screening for MCADD is being introduced
Engagement Report for Clinical Commissioning Policies
 Engagement Report for Clinical Commissioning Policies Unique Reference Number Policy Title Lead Commissioner Clinical Reference Group 1670 Total pancreatectomy with islet auto transplant for chronic pancreatitis
Engagement Report for Clinical Commissioning Policies Unique Reference Number Policy Title Lead Commissioner Clinical Reference Group 1670 Total pancreatectomy with islet auto transplant for chronic pancreatitis
Genetic Counselling in relation to genetic testing
 Genetic Counselling in relation to genetic testing Dr Julie Vogt Consultant Geneticist West Midlands Regional Genetics Service September 2016 Disclosures for Research Support/P.I. Employee Consultant Major
Genetic Counselling in relation to genetic testing Dr Julie Vogt Consultant Geneticist West Midlands Regional Genetics Service September 2016 Disclosures for Research Support/P.I. Employee Consultant Major
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 Appendix B: Scope NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE Post publication note: The title of this guideline changed during development. This scope was published before the guideline
Appendix B: Scope NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE Post publication note: The title of this guideline changed during development. This scope was published before the guideline
Cystic Fibrosis and Liver Disease a guide
 fighting childhood liver disease Cystic Fibrosis and Liver Disease a guide Medical Information Series Cystic Fibrosis and Liver Disease a guide 1 Welcome This leaflet has been written for: Parents/carers
fighting childhood liver disease Cystic Fibrosis and Liver Disease a guide Medical Information Series Cystic Fibrosis and Liver Disease a guide 1 Welcome This leaflet has been written for: Parents/carers
Thalassaemia. What is thalassaemia? What causes thalassaemia? What are the different types of thalassaemia?
 Thalassaemia Thalassaemia is an inherited condition affecting the blood. There are different types, which vary from a mild condition with no symptoms, to a serious or lifethreatening condition. For the
Thalassaemia Thalassaemia is an inherited condition affecting the blood. There are different types, which vary from a mild condition with no symptoms, to a serious or lifethreatening condition. For the
Dr Banu Kaya Consultant Haematologist Barts Health NHS Trust Royal London Hospital, London, UK SICKLE CELL AND THALASSAEMIA OVERVIEW
 Dr Banu Kaya Consultant Haematologist Barts Health NHS Trust Royal London Hospital, London, UK SICKLE CELL AND THALASSAEMIA OVERVIEW Objectives Gain awareness of haemoglobinopathy inheritance, pathophysiology
Dr Banu Kaya Consultant Haematologist Barts Health NHS Trust Royal London Hospital, London, UK SICKLE CELL AND THALASSAEMIA OVERVIEW Objectives Gain awareness of haemoglobinopathy inheritance, pathophysiology
Horizon Scanning Technology Briefing. Prostate cancer gene 3 (Progensa PCA3) assay in the diagnosis of prostate cancer
 Horizon Scanning Technology Briefing National Horizon Scanning Centre Prostate cancer gene 3 (Progensa PCA3) assay in the diagnosis of prostate cancer December 2006 This technology summary is based on
Horizon Scanning Technology Briefing National Horizon Scanning Centre Prostate cancer gene 3 (Progensa PCA3) assay in the diagnosis of prostate cancer December 2006 This technology summary is based on
