Original Policy Date
|
|
|
- Antony Norman
- 5 years ago
- Views:
Transcription
1 MP Transcatheter Closure of Patent Ductus Arteriosus Medical Policy Section Surgery Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed with literature search/12:2013 Return to Medical Policy Index Disclaimer Our medical policies are designed for informational purposes only and are not an authorization, or an explanation of benefits, or a contract. Receipt of benefits is subject to satisfaction of all terms and conditions of the coverage. Medical technology is constantly changing, and we reserve the right to review and update our policies periodically. Description The ductus arteriosus is the vascular remnant of the left sixth aortic arch, connecting the main pulmonary artery to the aorta. A patent ductus arteriosus (PDA) is the persistent opening of the channel beyond its expected time of closure during the first few days of life. Catheter-based techniques have been developed to close PDAs to eliminate the need for general anesthesia, a thoracotomy, and an extended hospital stay and convalescence associated with open surgical PDA closure. The ductus arteriosus is the vascular remnant of the left sixth aortic arch, connecting the main pulmonary artery to the aorta. A patent ductus arteriosus (PDA) is the persistent opening of the channel beyond its expected time of closure during the first few days of life. Symptoms are related to the size of the ductus; a large non-restrictive ductus with a left to right shunt can cause cardiac failure, while small restrictive PDAs are associated with an increased risk of infective endarteritis. Because of the twin threats of heart failure or endarteritis, it is recommended that all PDAs that persist after the age of 2 years be surgically closed with ligation or division of the PDA. Open surgical treatment of the PDA is a low-risk procedure, if performed electively. However, over the past several decades there has been interest in developing a catheter-based technique to close PDAs, thus eliminating the need for general anesthesia, a thoracotomy, and an extended hospital stay and convalescence. A number of devices have been developed for this purpose. The Gianturco coil, also referred to as the Cook embolization coil, is an arterial and venous occlusive device that was marketed prior to 1976, when the U.S. Food and Drug Administration (FDA) formally acquired regulatory authority over devices. (The Gianturco coil is entirely different than the Gianturco stent, which is used in coronary arteries.) Therefore, the Gianturco device has never undergone formal FDA approval but is available for clinical use. Transcatheter insertion of the coil is typically an outpatient procedure performed in the catheterization lab. General anesthesia may only be required in those very young patients who cannot reliably hold 42 Memorial Drive Suite 1 Pinehurst, N.C Phone (910) Fax (910)
2 still during the procedure. General anesthesia in a child younger than 1-year-old may require overnight hospitalization. Regulatory Status In 2003, the Amplatzer Duct Occluder received FDA approval, with the specific indication for non-surgical closure of patent ductus arteriosus. This device is a self-expandable device made from a Nitinol wire mesh and polyester fabric. As the occluder is implanted, it expands outward, and the wires push against the wall of the ductus. The polyester fabric induces thrombosis, which closes the communication. Policy Transcatheter closure of a patent ductus arteriosus using an FDA-approved device may be consideredmedically necessary. Transcatheter closure of a patent ductus arteriosus using other non-fda-approved devices is consideredinvestigational. Policy Guidelines According to the labeled indications of the Amplatzer Duct Occluder, the following are contraindications for the use of this device: Patients weighing less than 6 kg Patients less than 6 months of age Presence of thrombus at the intended site of implant, or documented evidence of venous thrombus in the vessels through which access to the defect is gained Active endocarditis or other infections producing bacteremia Patients whose vasculature, through which access to the defect is gained, is inadequate to accommodate the appropriate sheath size. Patients with pulmonary hypertension with pulmonary vascular resistance of >8 Woods units or Rp/Rs of >0.4. Rationale This policy was created in February 2003 and next updated in December 2010, followed by periodic updates with literature review. The most recent update with literature review covers the period between 2011 and August A number of different devices have been used to close patent ductus arteriosus (PDAs), including coils and occlusion devices. The evidence on the efficacy of percutaneous closure devices for PDA primarily consists of clinical series and a small number of non-randomized comparative studies. A representative sample of some of the larger studies is discussed below. Efficacy of closure devices for PDA 42 Memorial Drive Suite 1 Pinehurst, N.C Phone (910) Fax (910)
3 The Gianturco coil has been successfully adapted for use in closure of PDAs. Studies suggest a success rate of more than 90% in abolishing a clinically detectable shunt. (1, 2) Postoperative angiography and/or Doppler echocardiography frequently detect the persistence of a subclinical or trace shunt. The clinical significance of this finding is thought to be minimal. For example, Latson points out that using the Rashkind device, there have been no late cases of infection in more than 1,800 patient years of follow-up in postimplantation patients who have had no clinical evidence of a residual shunt. (3) One nonrandomized comparative trial compared percutaneous closure to open surgery. In this study from China, Chen et al. (4) compared 72 patients treated with percutaneous closure with 183 patients treated with open surgery. The choice of procedure was made at the discretion of the patient and/or treating physician. There were more procedure-related events in the open surgery group compared to the percutaneous group (13.7% vs.1.4%, p=0.004), and recovery time was longer for the open surgery group (8.7 days vs. 1.3 days, p<0.001). Freedom from persistent residual shunt was higher in the percutaneous group (98.6% vs. 91.3%, p=0.04). Other clinical outcomes such as pulmonary arterial hypertension and left ventricular size were similar between groups. Other evidence on the efficacy of closure devices consists of case series. A large case series of 1,291 attempted PDA coil occlusions was reported from the European Paediatric Cardiology Registry. (5) Immediate occlusion was demonstrated in 59% of patients, and this increased to 95% 1 year after the procedure. A suboptimal outcome occurred in 10% of patients, defined as failure to implant, coil embolization, residual leak, hemolysis, duct recanalization and flow impairment to adjacent structures. Increasing size of the PDA greater than 2 mm and PDAs that were tubular in shape were associated with an increased likelihood of unfavorable outcome. In 2003, the Amplatzer Duct Occluder device received FDA approval for closure of PDA. (6) The clinical data submitted to the FDA as part of the FDA approval process consisted of results from a multi-center non-randomized pivotal study that enrolled 441 patients. The primary efficacy measure was complete closure and was achieved in greater than 98.6% of patients at 12 months. A total of 1.3% of patients was reported to have serious or major adverse events, and 4.8% were reported to have a minor adverse event. The Multicenter USA Amplatzer patent ductus arteriosus device trial (7) reported periprocedural and 1-year outcomes in 484 patients from 25 U.S. centers. Of the 484 patients enrolled, the Amplatzer device implantation was not attempted in 45; due to the size of the PDA or the morphology of the PDA was more suited for treatment with a coil. Of the 439 patients in whom implantation was attempted, the device was successfully implanted in 435 patients (99%). Immediate postprocedure occlusion was reported in 76% of patients, which increased to 89% on postprocedure day 1 and to 99% at 1 year. At last evaluation, PDA occlusion was documented in 98% of patients. At 1-year follow-up 359/360 (99.7%) evaluable patients have no evidence of a left to right shunt on echocardiography. Complications were uncommon, with one periprocedural death and major events reported in 2.3% (10/439) of patients. Examples of major events included device embolization (n=2), partial obstruction of the pulmonary artery (n=2), and bleeding requiring transfusion (n=2). Minor events occurred in 7.1% (31/439) of patients. Other case series of both the Amplatzer device and the Gianturco coils report similar outcomes. (8-13) These series vary in terms of patient selection, types of device, and outcomes reported. However, the case series are consistent in reporting a high rate of procedural success, a high rate of successful closure of the PDA, and a low rate of serious complications. 42 Memorial Drive Suite 1 Pinehurst, N.C Phone (910) Fax (910)
4 Comparative efficacy of different devices and/or different techniques Wang et al. (14) compared outcomes among 214 patients undergoing percutaneous closure with coils and 134 patients undergoing closure by an occluder device. Patients were selected for either group by the size of the PDA, with coils utilized for small to moderate PDAs and the occluder device utilized for larger PDAs. The procedural success rate was high for both the coils (96.7%) and the occluder (98.5%), with no significant difference between groups. There were higher complication rates reported for the coil group. Distal embolization occurred in 8.9% (19/214) of patients in the coil group compared with 1.5% (2/136) patients in the occluder group (p<0.01). Pulmonary artery stenosis occurred in 4.2% (9/214) patients in the coil group compared with zero in the occluder group (p<0.05). In a non-randomized study, Chen et al. compared PDA occlusion using the transcatheter Amplatzer occluder in 98 patients to video-assisted thoracoscopic surgery in 196 patients. (15) No deaths or late recanalizations occurred in either group. However, residual shunt and left ventricular overload occurred in 4 (4.3%) patients in the transcatheter occluder group compared to zero patients in the thoracoscopic group. Acute complications related to the procedures occurred in 10.2% of the transcatheter occluder group compared to 1.5% of the video-assisted thoracoscopic surgery group (p<0.05). After follow-up of 3.1 to 8 years (mean, 5.4 ± 1.2 years) in the transcatheter occluder group and 3-8 years (mean, 5.6 ± 2.8 years) in the thoracoscopic surgery group, heart structures in both groups returned to normal. While fewer complications occurred in the thoracoscopic group, the non-randomized nature of the study limits interpretation of the results. Hongxin and colleagues reported on a parasternal, perpulmonary approach for PDA device closure in a cohort study of 79 patients. (16) Complete PDA occlusion occurred in 61 (78%) patients immediately after device placement while 7 (9%) patients required device redeployment. Complete PDA closure was found by echocardiogram in 76 (97%) patients during the follow-up period of 3 months to 2 years. While this minimally invasive approach is theoretically designed to avoid or reduce the disadvantages of other PDA occlusion procedures and devices, such as the trauma of thoracotomy and risk of transcatheter device embolization, these devices are not available in the United States, and this approach still requires general anesthesia. Ongoing Clinical Trials A search of online site ClinicalTrials.gov identified 2 ongoing non-randomized studies of PDA occluders. In a Phase I study, the safety and PDA closure outcomes of a new PDA occluder by Occlutech is being evaluated in 50 patients (NCT ). This trial is expected to be completed in December 2012 and is still recruiting patients. In the Amplatzer Duct Occluder II (ADOII) study, the safety and PDA closure outcomes of the Amplatzer Duct Occluder II will be evaluated in 192 patients (NCT ). This study is ongoing but is no longer recruiting patients and is expected to be completed in July Summary The ductus arteriosus is the vascular remnant of the left sixth aortic arch, connecting the main pulmonary artery to the aorta. A patent ductus arteriosus (PDA) is the persistent opening of the channel beyond its expected time of closure during the first few days of life. Catheter-based 42 Memorial Drive Suite 1 Pinehurst, N.C Phone (910) Fax (910)
5 techniques have been developed to close PDAs to eliminate the need for general anesthesia, a thoracotomy, and an extended hospital stay and convalescence associated with open surgical PDA closure. The use of percutaneous closure devices has become the procedure of choice for closure of patent ductus arteriosus in suitable patients. The evidence base for percutaneous closure of PDAs consists of a large number of case series that report high success rates with low rates of adverse events. A few non-randomized comparative trials compare outcomes of different devices and techniques, and one such study reports better outcomes with a thoracoscopic approach compared to a percutaneous approach. However, these non-randomized studies are not adequately rigorous to form conclusions because there is a high likelihood of selection bias, resulting in populations that are not comparable. Based on the evidence that percutaneous closure achieves high success rates and avoids the morbidity of open surgery, this technique may be considered medically necessary. Practice Guidelines and Position Statements In 2008 the American College of Cardiology/American Hospital Association (ACC/AHA) published guidelines on the management of adults with congenital heart disease. (17) Class I indications for closure of a PDA were listed as: Left atrial enlargement, left ventricular enlargement, pulmonary arterial hypertension, or left-to-right shunt (Level of evidence C) Prior endarteritis (Level of evidence C) Class IIa indications for closure of a PDA were: It is reasonable to close an asymptomatic small PDA by catheter device (Level of evidence C) PDA closure is reasonable for patients with pulmonary arterial hypertension with a net left-to-right shunt (Level of evidence C) Medicare National Coverage There is no national coverage determination. References: 1. Hijazi ZM, Geggel RL. Results of anterograde transcatheter closure of patent ductus arteriosus using single or multiple Gianturco coils. Am J Cardiol 1994; 74(9): Rothman A, Lucas VW, Sklansky MS et al. Percutaneous coil occlusion of patent ductus arteriosus. J Pediatr 1997; 130(3): Latson LA. Residual shunts after transcatheter closure of patent ductus arteriosus. A major concern or benign "techno-malady"? Circulation 1991; 84(6): Chen ZY, Wu LM, Luo YK et al. Comparison of long-term clinical outcome between transcatheter Amplatzer occlusion and surgical closure of isolated patent ductus arteriosus. Chin Med J (Engl) 2009; 122(10): Memorial Drive Suite 1 Pinehurst, N.C Phone (910) Fax (910)
6 5. Magee AG, Huggon IC, Seed PT et al. Transcatheter coil occlusion of the arterial duct; results of the European Registry. Eur Heart J 2001; 22(19): McCallum RW, Lin Z, Forster J et al. Gastric electrical stimulation improves outcomes of patients with gastroparesis for up to 10 years. Clin Gastroenterol Hepatol 2011; 9(4): e1. 7. Pass RH, Hijazi Z, Hsu DT et al. Multicenter USA Amplatzer patent ductus arteriosus occlusion device trial: initial and one-year results. J Am Coll Cardiol 2004; 44(3): Butera G, De Rosa G, Chessa M et al. Transcatheter closure of persistent ductus arteriosus with the Amplatzer duct occluder in very young symptomatic children. Heart 2004; 90(12): Ghasemi A, Pandya S, Reddy SV et al. Trans-catheter closure of patent ductus arteriosus-what is the best device? Catheter Cardiovasc Interv 2010; 76(5): Masura J, Tittel P, Gavora P et al. Long-term outcome of transcatheter patent ductus arteriosus closure using Amplatzer duct occluders. Am Heart J 2006; 151(3):755 e7-55 e Saliba Z, El-rassi I, Helou D et al. Development of catheter-based treatment of patent ductus arteriosus: a medium-sized centre experience. Arch Cardiovasc Dis 2009; 102(2): Wang JK, Wu MH, Hwang JJ et al. Transcatheter closure of moderate to large patent ductus arteriosus with the Amplatzer duct occluder. Catheter Cardiovasc Interv 2007; 69(4): Yang SW, Zhou YJ, Hu DY et al. Feasibility and safety of transcatheter intervention for complex patent ductus arteriosus. Angiology 2010; 61(4): Wang JK, Hwang JJ, Chiang FT et al. A strategic approach to transcatheter closure of patent ductus: Gianturco coils for small-to-moderate ductus and Amplatzer duct occluder for large ductus. Int J Cardiol 2006; 106(1): Chen H, Weng G, Chen Z et al. Comparison of long-term clinical outcomes and costs between video-assisted thoracoscopic surgery and transcatheter amplatzer occlusion of the patent ductus arteriosus. Pediatr Cardiol 2012; 33(2): Hongxin L, Wenbin G, Zhu M et al. New minimally invasive technique of perpulmonary device closure of patent ductus arteriosus through a parasternal approach. Ann Thorac Surg 2012; 93(3): Warnes CA, Williams RG, Bashore TM et al. ACC/AHA 2008 Guidelines for the Management of Adults with Congenital Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (writing committee to develop guidelines on the management of adults with congenital heart disease). Circulation 2008; 118(23):e Codes Number Description CPT Transcatheter occlusion or embolization (e.g., 42 Memorial Drive Suite 1 Pinehurst, N.C Phone (910) Fax (910)
7 for tumor destruction, to achieve hemostasis, to occlude a vascular malformation), percutaneous, any method, non-central nervous system, non head or neck. (Describes placement of coil.) ICD-9 Diagnosis Patent ductus arteriosus ICD-9 Procedure Other surgical occlusion of vessels; thoracic vessel HCPCS No code ICD-10-CM (effective 10/1/14) Q25.0 Patent ductus arteriosus ICD-10-PCS (effetive 10/1/14) Type of Service Place of Service Index 02LR3CT 02LR3DT 02LR3ZT 02LR4CT 02LR4DT 02LR4ZT 02VR3CT 02VR3DT 02VR3ZT 02VR4CT 02VR4DT 02VR4ZT Cardiology Inpatient Occlusion of Ductus Arteriosus with Extraluminal Device, Percutaneous Occlusion of Ductus Arteriosus with Intraluminal Device, Percutaneous Occlusion of Ductus Arteriosus, Percutaneous Occlusion of Ductus Arteriosus with Extraluminal Device, Percutaneous Endoscopic Occlusion of Ductus Arteriosus with Intraluminal Device, Percutaneous Endoscopic Occlusion of Ductus Arteriosus, Percutaneous Endoscopic Restriction of Ductus Arteriosus with Extraluminal Device, Percutaneous Restriction of Ductus Arteriosus with Intraluminal Device, Percutaneous Restriction of Ductus Arteriosus, Percutaneous Restriction of Ductus Arteriosus with Extraluminal Device, Percutaneous Endoscopic Restriction of Ductus Arteriosus with Intraluminal Device, Percutaneous Endoscopic Restriction of Ductus Arteriosus, Percutaneous Endoscopic 42 Memorial Drive Suite 1 Pinehurst, N.C Phone (910) Fax (910)
8 Amplatzer Duct Occluder, Patent Ductus Arteriosus Coils, Gianturco Cook Embolization Coils Gianturco Coil Patent Ductus Arteriosus, Catheter Closure Transcatheter Closure of Patent Ductus Arteriosus 42 Memorial Drive Suite 1 Pinehurst, N.C Phone (910) Fax (910)
Transcatheter closure of the patent ductus arteriosus using the new Amplatzer duct occluder: Initial clinical applications in children
 Transcatheter closure of the patent ductus arteriosus using the new Amplatzer duct occluder: Initial clinical applications in children Basil Thanopoulos, MD, PhD, a Nikolaos Eleftherakis, MD, a Konstantinos
Transcatheter closure of the patent ductus arteriosus using the new Amplatzer duct occluder: Initial clinical applications in children Basil Thanopoulos, MD, PhD, a Nikolaos Eleftherakis, MD, a Konstantinos
Paediatrica Indonesiana
 Paediatrica Indonesiana VOLUME 53 July NUMBER 4 Original Article Transcatheter vs. surgical closure of patent ductus arteriosus: outcomes and cost analysis Mulyadi M Djer, Mochammading, Mardjanis Said
Paediatrica Indonesiana VOLUME 53 July NUMBER 4 Original Article Transcatheter vs. surgical closure of patent ductus arteriosus: outcomes and cost analysis Mulyadi M Djer, Mochammading, Mardjanis Said
TRANSCATHETER CLOSURE OF PERSISTENT DUCTUS ARTERIOSUS USING THE AMPLATZER DUCT OCCLUDER IN INFANTS
 TRANSCATHETER CLOSURE OF PERSISTENT DUCTUS ARTERIOSUS USING THE AMPLATZER DUCT OCCLUDER IN INFANTS Al-Hakim FA 1, Harahsheh AS 2 & Hijazi IS 3 ABSTRACT Background: Despite technical improvements over the
TRANSCATHETER CLOSURE OF PERSISTENT DUCTUS ARTERIOSUS USING THE AMPLATZER DUCT OCCLUDER IN INFANTS Al-Hakim FA 1, Harahsheh AS 2 & Hijazi IS 3 ABSTRACT Background: Despite technical improvements over the
AMPLATZER Septal Occluder Structural Heart Therapy. Over 15 years of. Demonstrated. Clinical Experience. We ll show you our data. Ask to see theirs.
 AMPLATZER Septal Occluder Structural Heart Therapy Over 15 years of Demonstrated Clinical Experience We ll show you our data. Ask to see theirs. Leading the Standard of Care 1,2 The AMPLATZER Septal Occluder
AMPLATZER Septal Occluder Structural Heart Therapy Over 15 years of Demonstrated Clinical Experience We ll show you our data. Ask to see theirs. Leading the Standard of Care 1,2 The AMPLATZER Septal Occluder
COMPLEMENTARY TECHNIQUES OF PERCUTANEOUS CLOSURE OF DUCTUS ARTERIOSUS USING DETACHABLE COOK COILS AND AMPLATZER DEVICES
 214 East African Medical Journal July 2013 East African Medical Journal Vol. 90 No. 7 July 2013 COMPLEMENTARY TECHNIQUES OF PERCUTANEOUS CLOSURE OF DUCTUS ARTERIOSUS USING DETACHABLE COOK COILS AND AMPLATZER
214 East African Medical Journal July 2013 East African Medical Journal Vol. 90 No. 7 July 2013 COMPLEMENTARY TECHNIQUES OF PERCUTANEOUS CLOSURE OF DUCTUS ARTERIOSUS USING DETACHABLE COOK COILS AND AMPLATZER
Patient guide: pfm Nit-Occlud PDA coil occlusion system. Catheter occlusion of. Patent Ductus Arteriosus. with the
 Patient guide: Catheter occlusion of Patent Ductus Arteriosus with the pfm Nit-Occlud PDA coil occlusion system pfm Produkte für die Medizin - AG Wankelstr. 60 D - 50996 Cologne Phone: +49 (0) 2236 96
Patient guide: Catheter occlusion of Patent Ductus Arteriosus with the pfm Nit-Occlud PDA coil occlusion system pfm Produkte für die Medizin - AG Wankelstr. 60 D - 50996 Cologne Phone: +49 (0) 2236 96
CY2015 Hospital Outpatient: Endovascular Procedure APCs and Complexity Adjustments
 CY2015 Hospital Outpatient: Endovascular Procedure APCs Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) CMS finalized the implementation of 25 Comprehensive APC to further
CY2015 Hospital Outpatient: Endovascular Procedure APCs Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) CMS finalized the implementation of 25 Comprehensive APC to further
Type Size AP/PS RP/RS Qp/Qs. Ia Resistive <0.3 < Ib Resistive <0.3 < IIa Resistive <0.5 >2
 Transcatheter closure of VSD using Duct Occluder device Nguyen Lan Hieu, MD, PhD Hanoi Medical University Vietnam Heart Institute Anatomy of VSD 1. Perimembranous VSD: Aneurysm septal membranous(tv or
Transcatheter closure of VSD using Duct Occluder device Nguyen Lan Hieu, MD, PhD Hanoi Medical University Vietnam Heart Institute Anatomy of VSD 1. Perimembranous VSD: Aneurysm septal membranous(tv or
IMAGES. in PAEDIATRIC CARDIOLOGY. Abstract
 IMAGES in PAEDIATRIC CARDIOLOGY Images Paediatr Cardiol. 2008 Apr-Jun; 10(2): 11 17. PMCID: PMC3232589 Transcatheter closure of symptomatic aortopulmonary window in an infant F Pillekamp, 1 T Hannes, 1
IMAGES in PAEDIATRIC CARDIOLOGY Images Paediatr Cardiol. 2008 Apr-Jun; 10(2): 11 17. PMCID: PMC3232589 Transcatheter closure of symptomatic aortopulmonary window in an infant F Pillekamp, 1 T Hannes, 1
Jian Fang 1, Shaobo Xie 2, Lunchao Ma 2, Chao Yang 2. Original Article
 Original Article Anatomic and surgical factors affecting the switch from minimally invasive transthoracic occlusion to open surgery during ventricular septal defect repair Jian Fang 1, Shaobo Xie 2, Lunchao
Original Article Anatomic and surgical factors affecting the switch from minimally invasive transthoracic occlusion to open surgery during ventricular septal defect repair Jian Fang 1, Shaobo Xie 2, Lunchao
Ovarian and Internal Iliac Vein Embolization as a Treatment of Pelvic Congestion Syndrome. Original Policy Date
 MP 4.01.11 Ovarian and Internal Iliac Vein Embolization as a Treatment of Pelvic Congestion Syndrome Medical Policy Section OB/Gyn/Reproduction Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date
MP 4.01.11 Ovarian and Internal Iliac Vein Embolization as a Treatment of Pelvic Congestion Syndrome Medical Policy Section OB/Gyn/Reproduction Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date
Reopening After Successful Coil Occlusion for Patent Ductus Arteriosus
 444 JACC Vol. 31, No. 2 PEDIATRIC CARDIOLOGY Reopening After Successful Coil Occlusion for Patent Ductus Arteriosus CURT J. DANIELS, MD, STEVEN C. CASSIDY, MD, FACC, DOUGLAS W. TESKE, MD, FACC, JOHN J.
444 JACC Vol. 31, No. 2 PEDIATRIC CARDIOLOGY Reopening After Successful Coil Occlusion for Patent Ductus Arteriosus CURT J. DANIELS, MD, STEVEN C. CASSIDY, MD, FACC, DOUGLAS W. TESKE, MD, FACC, JOHN J.
Originally Posted: November 15, 2014 BRUIT IN THE GROIN
 Originally Posted: November 15, 2014 BRUIT IN THE GROIN Resident(s): Donald ML Tse, MD Attending(s): KT Tan, MD Program/Dept(s): University Health Network/Mount Sinai Hospital, Toronto, ON, Canada CHIEF
Originally Posted: November 15, 2014 BRUIT IN THE GROIN Resident(s): Donald ML Tse, MD Attending(s): KT Tan, MD Program/Dept(s): University Health Network/Mount Sinai Hospital, Toronto, ON, Canada CHIEF
Original Policy Date
 MP 7.01.111 Transcatheter Pulmonary Valve Implantation Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to
MP 7.01.111 Transcatheter Pulmonary Valve Implantation Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to
IMAGES. in PAEDIATRIC CARDIOLOGY
 IMAGES in PAEDIATRIC CARDIOLOGY Images Paediatr Cardiol. 2005 Jan-Mar; 7(1): 12 17. PMCID: PMC3232568 Stent implantation for coarctation facilitated by the anterograde trans-septal approach N Sreeram and
IMAGES in PAEDIATRIC CARDIOLOGY Images Paediatr Cardiol. 2005 Jan-Mar; 7(1): 12 17. PMCID: PMC3232568 Stent implantation for coarctation facilitated by the anterograde trans-septal approach N Sreeram and
Initial Human Experience. Objectives: To evaluate the feasibility, safety, and efficacy of the Occlutech VR
 Catheterization and Cardiovascular Interventions 00:00 00 (2015) Original Studies The New Occlutech V R PDA Occluder: Initial Human Experience M. A. Elbashier Abdelbasit, 1 * MBBS, MD, Mazeni Alwi, 1 MBBS,
Catheterization and Cardiovascular Interventions 00:00 00 (2015) Original Studies The New Occlutech V R PDA Occluder: Initial Human Experience M. A. Elbashier Abdelbasit, 1 * MBBS, MD, Mazeni Alwi, 1 MBBS,
Percutaneous VSD closure
 Percutaneous VSD closure Gianfranco Butera San Donato Milanese - Italy Patients selection Pts having hemodynamically significant VSD Left ventricular enlargement (left ventricular overload),defined as
Percutaneous VSD closure Gianfranco Butera San Donato Milanese - Italy Patients selection Pts having hemodynamically significant VSD Left ventricular enlargement (left ventricular overload),defined as
Index. interventional.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Alagille syndrome, pulmonary artery stenosis in, 143 145, 148 149 Amplatz devices for atrial septal defect closure, 42 46 for coronary
Index Note: Page numbers of article titles are in boldface type. A Alagille syndrome, pulmonary artery stenosis in, 143 145, 148 149 Amplatz devices for atrial septal defect closure, 42 46 for coronary
Long-Term Results of Transcatheter Closure of Patent Ductus Arteriosus in Infants Using Amplatzer Duct Occluder
 Original Article Iran J Pediatr Aug 2013; Vol 23 (No 4), Pp: 411-416 Long-Term Results of Transcatheter Closure of Patent Ductus Arteriosus in Infants Using Amplatzer Duct Occluder Mostafa Behjati-Ardakani*
Original Article Iran J Pediatr Aug 2013; Vol 23 (No 4), Pp: 411-416 Long-Term Results of Transcatheter Closure of Patent Ductus Arteriosus in Infants Using Amplatzer Duct Occluder Mostafa Behjati-Ardakani*
VIRTUS: Trial Design and Primary Endpoint Results
 VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
CY2017 Hospital Outpatient: Vascular Procedure APCs and Complexity Adjustments
 CY2017 Hospital Outpatient: Vascular Procedure APCs and Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) In CY2015 and in an effort to help pay providers for quality, not
CY2017 Hospital Outpatient: Vascular Procedure APCs and Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) In CY2015 and in an effort to help pay providers for quality, not
Transcatheter device occlusion of patent ductus arteriosus
 CONGENITAL HEART DISEASE The New Occlutech Duct Occluder: Immediate Results, Procedural Challenges, and Short-Term Follow-Up Vikram Kudumula, MRCPCH; Demetris Taliotis, MRCPCH; Christopher Duke, FRCP ABSTRACT:
CONGENITAL HEART DISEASE The New Occlutech Duct Occluder: Immediate Results, Procedural Challenges, and Short-Term Follow-Up Vikram Kudumula, MRCPCH; Demetris Taliotis, MRCPCH; Christopher Duke, FRCP ABSTRACT:
Fabien Praz, Andreas Wahl, Sophie Beney, Stephan Windecker, Heinrich P. Mattle*, Bernhard Meier
 Procedural Outcome after Percutaneous Closure of Patent Foramen Ovale using the Amplatzer PFO Occluder Without Intra-Procedural Echocardiography in 1,000 Patients Fabien Praz, Andreas Wahl, Sophie Beney,
Procedural Outcome after Percutaneous Closure of Patent Foramen Ovale using the Amplatzer PFO Occluder Without Intra-Procedural Echocardiography in 1,000 Patients Fabien Praz, Andreas Wahl, Sophie Beney,
AMPLATZER Septal Occluder
 AMPLATZER Septal Occluder A Patient s Guide to the Non-Surgical Closure of the Atrial Septal Defect Using the AMPLATZER Septal Occluder System leadership through innovation TM AGA Medical Corporation 5050
AMPLATZER Septal Occluder A Patient s Guide to the Non-Surgical Closure of the Atrial Septal Defect Using the AMPLATZER Septal Occluder System leadership through innovation TM AGA Medical Corporation 5050
CONGENITAL HEART DEFECTS IN ADULTS
 CONGENITAL HEART DEFECTS IN ADULTS THE ROLE OF CATHETER INTERVENTIONS Mario Carminati CONGENITAL HEART DEFECTS IN ADULTS CHD in natural history CHD with post-surgical sequelae PULMONARY VALVE STENOSIS
CONGENITAL HEART DEFECTS IN ADULTS THE ROLE OF CATHETER INTERVENTIONS Mario Carminati CONGENITAL HEART DEFECTS IN ADULTS CHD in natural history CHD with post-surgical sequelae PULMONARY VALVE STENOSIS
CIPG Transcatheter Aortic Valve Replacement- When Is Less, More?
 CIPG 2013 Transcatheter Aortic Valve Replacement- When Is Less, More? James D. Rossen, M.D. Professor of Medicine and Neurosurgery Director, Cardiac Catheterization Laboratory and Interventional Cardiology
CIPG 2013 Transcatheter Aortic Valve Replacement- When Is Less, More? James D. Rossen, M.D. Professor of Medicine and Neurosurgery Director, Cardiac Catheterization Laboratory and Interventional Cardiology
PDA: Closure Using Coils and Devices: Indications, Technique & Outcome
 PDA: Closure Using Coils and Devices: Indications, Technique & Outcome Daniel Levi, MD, FSCAI Associate Professor of Pediatrics UCLA Biomedical Engineering Mattel Children s Hospital at UCLA, Division
PDA: Closure Using Coils and Devices: Indications, Technique & Outcome Daniel Levi, MD, FSCAI Associate Professor of Pediatrics UCLA Biomedical Engineering Mattel Children s Hospital at UCLA, Division
Index. cardiology.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A ACHD. See Adult congenital heart disease (ACHD) Adult congenital heart disease (ACHD), 503 512 across life span prevalence of, 504 506
Index Note: Page numbers of article titles are in boldface type. A ACHD. See Adult congenital heart disease (ACHD) Adult congenital heart disease (ACHD), 503 512 across life span prevalence of, 504 506
Indication, Timing, Assessment and Update on TAVI
 Indication, Timing, Assessment and Update on TAVI Swedish Heart and Vascular Institute Ming Zhang MD PhD Interventional Cardiology Structure Heart Disease Conflict of Interest None Starr- Edwards Mechanical
Indication, Timing, Assessment and Update on TAVI Swedish Heart and Vascular Institute Ming Zhang MD PhD Interventional Cardiology Structure Heart Disease Conflict of Interest None Starr- Edwards Mechanical
CPT Code Details
 CPT Code 93572 Details Code Descriptor Intravascular Doppler velocity and/or pressure derived coronary flow reserve measurement (coronary vessel or graft) during coronary angiography including pharmacologically
CPT Code 93572 Details Code Descriptor Intravascular Doppler velocity and/or pressure derived coronary flow reserve measurement (coronary vessel or graft) during coronary angiography including pharmacologically
For Personal Use. Copyright HMP 2013
 Case Report J INVASIVE CARDIOL 2013;25(2):E42-E44 Percutaneous Closure of Iatrogenic Ventricular Septal Defect Following Surgical Aortic Valve Replacement Using Two Different Approaches Takashi Matsumoto,
Case Report J INVASIVE CARDIOL 2013;25(2):E42-E44 Percutaneous Closure of Iatrogenic Ventricular Septal Defect Following Surgical Aortic Valve Replacement Using Two Different Approaches Takashi Matsumoto,
Predictors of unfavorable outcome after atrial septal defect closure in adults
 after atrial septal defect closure in adults H. M. Gabriel 1, M. Humenberger 1, R. Rosenhek 1, GP. Diller 2, G. Kaleschke 2, TH. Binder 1, P. Probst 1, G. Maurer 1, H. Baumgartner 2 (1) Medical University
after atrial septal defect closure in adults H. M. Gabriel 1, M. Humenberger 1, R. Rosenhek 1, GP. Diller 2, G. Kaleschke 2, TH. Binder 1, P. Probst 1, G. Maurer 1, H. Baumgartner 2 (1) Medical University
Transcatheter Closure of Patent Ductus Arteriosus Using ADO Device: Retrospective Study of 149 Patients
 Original Article Transcatheter Closure of Patent Ductus Arteriosus Using ADO Device: Retrospective Study of 149 Patients ABSTRACT Sadiq M. Al-Hamash, Hussein Abdul Wahab 1, Zayir H. Khalid 1, Isam V. Nasser
Original Article Transcatheter Closure of Patent Ductus Arteriosus Using ADO Device: Retrospective Study of 149 Patients ABSTRACT Sadiq M. Al-Hamash, Hussein Abdul Wahab 1, Zayir H. Khalid 1, Isam V. Nasser
Perimembranous ventricular septal defects (pmvsds) are
 Transcatheter Closure of Congenital Perimembranous Ventricular Septal Defect in Children Using Symmetric Occluders: An 8-Year Multiinstitutional Experience Lei Wang, MD, PhD,* Sancheng Cao, MD, Jun Li,
Transcatheter Closure of Congenital Perimembranous Ventricular Septal Defect in Children Using Symmetric Occluders: An 8-Year Multiinstitutional Experience Lei Wang, MD, PhD,* Sancheng Cao, MD, Jun Li,
Original Policy Date
 MP 7.01.39 Transurethral Microwave Thermotherapy Medical Policy Section Surgery Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed with literature search/12:2013 Return to Medical
MP 7.01.39 Transurethral Microwave Thermotherapy Medical Policy Section Surgery Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed with literature search/12:2013 Return to Medical
Peripheral and Cardiology Coder 2018
 Peripheral and Cardiology Coder 2018 Cardiovascular Services and Procedures Prepared and Published By: MedLearn Publishing A Division of MedLearn Media, Inc. 445 Minnesota Street, Suite 514 St. Paul, MN
Peripheral and Cardiology Coder 2018 Cardiovascular Services and Procedures Prepared and Published By: MedLearn Publishing A Division of MedLearn Media, Inc. 445 Minnesota Street, Suite 514 St. Paul, MN
Original Policy Date 12:2013
 MP 6.01.30 Magnetic Resonance Cholangiopancreatography Medical Policy Section Radiology Is12:2013sue 3:2005 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index Disclaimer
MP 6.01.30 Magnetic Resonance Cholangiopancreatography Medical Policy Section Radiology Is12:2013sue 3:2005 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index Disclaimer
Devices to Protect Against Stroke in Atrial Fibrillation
 Devices to Protect Against Stroke in Atrial Fibrillation Jonathan C. Hsu, MD, MAS Associate Clinical Professor Division of Cardiology, Section of Cardiac Electrophysiology June 2, 2018 Disclosures Honoraria
Devices to Protect Against Stroke in Atrial Fibrillation Jonathan C. Hsu, MD, MAS Associate Clinical Professor Division of Cardiology, Section of Cardiac Electrophysiology June 2, 2018 Disclosures Honoraria
Diagnostic & Therapeutic Cardiac Catheterization Coder 2017
 Diagnostic & Therapeutic Cardiac Catheterization Coder 2017 Including peripheral and cardiovascular services and procedures Prepared and Published By: MedLearn Publishing A Division of Panacea Healthcare
Diagnostic & Therapeutic Cardiac Catheterization Coder 2017 Including peripheral and cardiovascular services and procedures Prepared and Published By: MedLearn Publishing A Division of Panacea Healthcare
Outcomes Associated with the Off-label Use of Medical Devices in Congenital Heart Disease at a Single Institute
 Original Article Print ISSN 1738-5520 On-line ISSN 1738-5555 Korean Circulation Journal Outcomes Associated with the Off-label Use of Medical Devices in Congenital Heart Disease at a Single Institute Young
Original Article Print ISSN 1738-5520 On-line ISSN 1738-5555 Korean Circulation Journal Outcomes Associated with the Off-label Use of Medical Devices in Congenital Heart Disease at a Single Institute Young
INTEGRATING ECHOCARDIOGRAPHY WITH CATHETER INTERVENTIONS FOR CONGENITAL HEART DISEASE. Krishna Kumar SevenHills Hospital, Mumbai, India
 INTEGRATING ECHOCARDIOGRAPHY WITH CATHETER INTERVENTIONS FOR CONGENITAL HEART DISEASE Krishna Kumar SevenHills Hospital, Mumbai, India Why talk about it? What is the big deal? Are we not stating the obvious?
INTEGRATING ECHOCARDIOGRAPHY WITH CATHETER INTERVENTIONS FOR CONGENITAL HEART DISEASE Krishna Kumar SevenHills Hospital, Mumbai, India Why talk about it? What is the big deal? Are we not stating the obvious?
PDA Device Closure with and without Arterial Access
 Research Article imedpub Journals www.imedpub.com Interventional Cardiology Journal DOI: 10.21767/2471-8157.100061 Abstract PDA Device Closure with and without Arterial Access Background: PDA device closure
Research Article imedpub Journals www.imedpub.com Interventional Cardiology Journal DOI: 10.21767/2471-8157.100061 Abstract PDA Device Closure with and without Arterial Access Background: PDA device closure
Evaluation of Gianturco Coils for Closure of Large (> 3.5 mm) Patent Ductus Arteriosus
 1856 JACC Vol. 30, No. 7 Evaluation of Gianturco Coils for Closure of Large (> 3.5 mm) Patent Ductus Arteriosus CARL Y. OWADA, MD, DAVID F. TEITEL, MD, PHILLIP MOORE, MD San Francisco, California Objectives.
1856 JACC Vol. 30, No. 7 Evaluation of Gianturco Coils for Closure of Large (> 3.5 mm) Patent Ductus Arteriosus CARL Y. OWADA, MD, DAVID F. TEITEL, MD, PHILLIP MOORE, MD San Francisco, California Objectives.
Case Report Hemostasis of Left Atrial Appendage Bleed With Lariat Device
 273 Case Report Hemostasis of Left Atrial Appendage Bleed With Lariat Device Amena Hussain MD, Muhamed Saric MD, Scott Bernstein MD, Douglas Holmes MD, Larry Chinitz MD NYU Langone Medical Center, United
273 Case Report Hemostasis of Left Atrial Appendage Bleed With Lariat Device Amena Hussain MD, Muhamed Saric MD, Scott Bernstein MD, Douglas Holmes MD, Larry Chinitz MD NYU Langone Medical Center, United
Indications of Coronary Angiography Dr. Shaheer K. George, M.D Faculty of Medicine, Mansoura University 2014
 Indications of Coronary Angiography Dr. Shaheer K. George, M.D Faculty of Medicine, Mansoura University 2014 Indications for cardiac catheterization Before a decision to perform an invasive procedure such
Indications of Coronary Angiography Dr. Shaheer K. George, M.D Faculty of Medicine, Mansoura University 2014 Indications for cardiac catheterization Before a decision to perform an invasive procedure such
UNMH Pediatric Cardiology Clinical Privileges. Name: Effective Dates: From To
 All new applicants must meet the following requirements as approved by the UNMH Board of Trustees, effective August 18, 2017: Initial Privileges (initial appointment) Renewal of Privileges (reappointment)
All new applicants must meet the following requirements as approved by the UNMH Board of Trustees, effective August 18, 2017: Initial Privileges (initial appointment) Renewal of Privileges (reappointment)
Hybrid Muscular VSD Closure in Small Weight Children
 Hybrid Muscular VSD Closure in Small Weight Children Shakeel A Qureshi, on behalf of: John P. Cheatham, MD George H. Dunlap Endowed Chair in Interventional Cardiology Director Cardiac Catheterization &
Hybrid Muscular VSD Closure in Small Weight Children Shakeel A Qureshi, on behalf of: John P. Cheatham, MD George H. Dunlap Endowed Chair in Interventional Cardiology Director Cardiac Catheterization &
1. Definition/Overview
 VCAwestlaspecialty.com VCA West Los Angeles recently performed closure of a patent ductus arteriosus via percutaneous transarterial embolization with an Amplatz canine ductal occluder on a female, four-month-old
VCAwestlaspecialty.com VCA West Los Angeles recently performed closure of a patent ductus arteriosus via percutaneous transarterial embolization with an Amplatz canine ductal occluder on a female, four-month-old
Index. cardiology.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Acute ischemic stroke TOAST classification of, 270 Acute myocardial infarction (AMI) cardioembolic stroke following, 207 208 noncardioembolic
Index Note: Page numbers of article titles are in boldface type. A Acute ischemic stroke TOAST classification of, 270 Acute myocardial infarction (AMI) cardioembolic stroke following, 207 208 noncardioembolic
Atrial Septal Defect Closure. Stephen Brecker Director, Cardiac Catheterisation Labs
 Stephen Brecker Director, Cardiac Catheterisation Labs ADVANCED ANGIOPLASTY Incorporating The Left Main 5 Plus Course Conflicts of Interest The following companies have supported educational courses held
Stephen Brecker Director, Cardiac Catheterisation Labs ADVANCED ANGIOPLASTY Incorporating The Left Main 5 Plus Course Conflicts of Interest The following companies have supported educational courses held
National Imaging Associates, Inc. Clinical guidelines CARDIAC CATHETERIZATION -LEFT HEART CATHETERIZATION. Original Date: October 2015 Page 1 of 5
 National Imaging Associates, Inc. Clinical guidelines CARDIAC CATHETERIZATION -LEFT HEART CATHETERIZATION CPT Codes: 93451, 93452, 93453, 93454, 93455, 93456, 93457, 93458, 93459, 93460, 93461 LCD ID Number:
National Imaging Associates, Inc. Clinical guidelines CARDIAC CATHETERIZATION -LEFT HEART CATHETERIZATION CPT Codes: 93451, 93452, 93453, 93454, 93455, 93456, 93457, 93458, 93459, 93460, 93461 LCD ID Number:
Pathophysiology: Left To Right Shunts
 Pathophysiology: Left To Right Shunts Daphne T. Hsu, MD dh17@columbia.edu Learning Objectives Learn the relationships between pressure, blood flow, and resistance Review the transition from fetal to mature
Pathophysiology: Left To Right Shunts Daphne T. Hsu, MD dh17@columbia.edu Learning Objectives Learn the relationships between pressure, blood flow, and resistance Review the transition from fetal to mature
Case submission for CSI Asia-Pacific Case 2
 Case submission for CSI Asia-Pacific 2018- Case 2 Title Page Case category: Coarctation and ducts, valves Title: Simultaneous balloon aortic valvuloplasty with transcatheter closure of large hypertensive
Case submission for CSI Asia-Pacific 2018- Case 2 Title Page Case category: Coarctation and ducts, valves Title: Simultaneous balloon aortic valvuloplasty with transcatheter closure of large hypertensive
LAA Occluders: The Right Device for the Right Patient ACC/SHA MEETING OCTOBER 31 ST 2015 JEDDAH, KSA OMER A. M. ELAMIN, MD, FACC
 LAA Occluders: The Right Device for the Right Patient ACC/SHA MEETING OCTOBER 31 ST 2015 JEDDAH, KSA OMER A. M. ELAMIN, MD, FACC INTERVENTIONAL CARDIOLOGIST DIRECTOR, ADULT CARDIOLOGY FELLOWSHIP TRAINING
LAA Occluders: The Right Device for the Right Patient ACC/SHA MEETING OCTOBER 31 ST 2015 JEDDAH, KSA OMER A. M. ELAMIN, MD, FACC INTERVENTIONAL CARDIOLOGIST DIRECTOR, ADULT CARDIOLOGY FELLOWSHIP TRAINING
My personal experience with INCRAFT in standard and challenging cases
 My personal experience with INCRAFT in standard and challenging cases G Pratesi, MD Vascular Surgery University of Rome Tor Vergata giovanni.pratesi@uniroma2.it Disclosure Speaker name: Giovanni Pratesi,
My personal experience with INCRAFT in standard and challenging cases G Pratesi, MD Vascular Surgery University of Rome Tor Vergata giovanni.pratesi@uniroma2.it Disclosure Speaker name: Giovanni Pratesi,
Corporate Medical Policy
 Corporate Medical Policy Congenital Heart Defect, Repair Devices File Name: Origination: Last CAP Review: Next CAP Review: Last Review: congenital_heart_defect_repair_devices 10/2000 6/2017 6/2018 6/2017
Corporate Medical Policy Congenital Heart Defect, Repair Devices File Name: Origination: Last CAP Review: Next CAP Review: Last Review: congenital_heart_defect_repair_devices 10/2000 6/2017 6/2018 6/2017
Percutaneous atrial septal defect closure with the Occlutech Figulla Flex ASD Occluder.
 Percutaneous atrial septal defect closure with the Occlutech Figulla Flex ASD Occluder. First case with a novel delivery system. Werner Budts, Md, PhD, FESC Congenital and Structural Cardiology University
Percutaneous atrial septal defect closure with the Occlutech Figulla Flex ASD Occluder. First case with a novel delivery system. Werner Budts, Md, PhD, FESC Congenital and Structural Cardiology University
Transcatheter closure of right coronary artery fistula to the right ventricle
 Case Report Transcatheter closure of right coronary artery fistula to the right ventricle Abstract Coronary artery fistula (CAF) is an uncommon anomaly usually congenital but can be acquired. Although,
Case Report Transcatheter closure of right coronary artery fistula to the right ventricle Abstract Coronary artery fistula (CAF) is an uncommon anomaly usually congenital but can be acquired. Although,
Reliability of Echocardiography Measurement of Patent Ductus Arteriosus Minimum Diameter: A Meta-analysis
 International Journal of Cardiovascular and Cerebrovascular Disease 4(): 15-19, 016 DOI: 10.13189/ijccd.016.04001 http://www.hrpub.org Reliability of Echocardiography Measurement of Patent Ductus Arteriosus
International Journal of Cardiovascular and Cerebrovascular Disease 4(): 15-19, 016 DOI: 10.13189/ijccd.016.04001 http://www.hrpub.org Reliability of Echocardiography Measurement of Patent Ductus Arteriosus
COST AND EFFICACY OF SURGICAL LIGATION VERSUS TRANSCATHETER COIL OCCLUSION OF PATENT DUCTUS ARTERIOSUS
 COST AND EFFICACY OF SURGICAL LIGATION VERSUS TRANSCATHETER COIL OCCLUSION OF PATENT DUCTUS ARTERIOSUS John A. Hawkins, MD L. LuAnn Minich, MD Lloyd Y. Tani, MD Jane E. Sturtevant, BSN Garth S. Orsmond,
COST AND EFFICACY OF SURGICAL LIGATION VERSUS TRANSCATHETER COIL OCCLUSION OF PATENT DUCTUS ARTERIOSUS John A. Hawkins, MD L. LuAnn Minich, MD Lloyd Y. Tani, MD Jane E. Sturtevant, BSN Garth S. Orsmond,
Perimembranous VSD: When Do We Ask For A Surgical Closure? LI Xin. Department of Cardiothoracic Surgery Queen Mary Hospital Hong Kong
 Perimembranous VSD: When Do We Ask For A Surgical Closure? LI Xin Department of Cardiothoracic Surgery Queen Mary Hospital Hong Kong Classification (by Kirklin) I. Subarterial (10%) Outlet, conal, supracristal,
Perimembranous VSD: When Do We Ask For A Surgical Closure? LI Xin Department of Cardiothoracic Surgery Queen Mary Hospital Hong Kong Classification (by Kirklin) I. Subarterial (10%) Outlet, conal, supracristal,
PERCUTANEOUS TRANSCATHETER CLOSURE OF PARAVALVULAR LEAK
 Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs are dependent upon
Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs are dependent upon
Coronary Arteriovenous Malformation presenting as Acute Myocardial Infarction. Choon Ta NG, Aaron WONG, Foong-Koon CHEAH, Chi Keong CHING
 Coronary Arteriovenous Malformation presenting as Acute Myocardial Infarction Choon Ta NG, Aaron WONG, Foong-Koon CHEAH, Chi Keong CHING The patient 49 year old Male presented with Chest tightness x 1
Coronary Arteriovenous Malformation presenting as Acute Myocardial Infarction Choon Ta NG, Aaron WONG, Foong-Koon CHEAH, Chi Keong CHING The patient 49 year old Male presented with Chest tightness x 1
Premier Health Plan considers Intravascular Ultrasound (IVUS) for Coronary Vessels medically necessary for the following indications:
 Premier Health Plan POLICY AND PROCEDURE MANUAL MP.091.PH - Intravascular Ultrasound for Coronary Vessels This policy applies to the following lines of business: Premier Commercial Premier Employee Premier
Premier Health Plan POLICY AND PROCEDURE MANUAL MP.091.PH - Intravascular Ultrasound for Coronary Vessels This policy applies to the following lines of business: Premier Commercial Premier Employee Premier
Follow-up after VSD closure- what to look for?
 The 3rd Congress of Congenital heart disease Ventricular Septal Defect from A-Z January 9-11. 2013, Ho Chi Minh City, Vietnam Follow-up after VSD closure- what to look for? Nina Wunderlich University Hospital
The 3rd Congress of Congenital heart disease Ventricular Septal Defect from A-Z January 9-11. 2013, Ho Chi Minh City, Vietnam Follow-up after VSD closure- what to look for? Nina Wunderlich University Hospital
INTERVENTIONAL PROCEDURES PROGRAMME
 NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of balloon angioplasty for systemic to pulmonary arterial shunts Introduction This overview
NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of balloon angioplasty for systemic to pulmonary arterial shunts Introduction This overview
Pathophysiology: Left To Right Shunts
 Pathophysiology: Left To Right Shunts Daphne T. Hsu, MD dh17@columbia.edu Learning Objectives Learn the relationships between pressure, blood flow, and resistance Review the transition from fetal to mature
Pathophysiology: Left To Right Shunts Daphne T. Hsu, MD dh17@columbia.edu Learning Objectives Learn the relationships between pressure, blood flow, and resistance Review the transition from fetal to mature
Original Policy Date
 MP 7.01.66 Auditory Brainstem Implant Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to Medical Policy
MP 7.01.66 Auditory Brainstem Implant Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to Medical Policy
Atrial septal defects (ASDs) are one of the most
 Transcatheter Closure of Atrial Septal Defects An update on ASD occlusion devices. BY VICTOR-XAVIER TADROS, MD, MSc, AND ANITA W. ASGAR, MD, FRCPC, FACC Atrial septal defects (ASDs) are one of the most
Transcatheter Closure of Atrial Septal Defects An update on ASD occlusion devices. BY VICTOR-XAVIER TADROS, MD, MSc, AND ANITA W. ASGAR, MD, FRCPC, FACC Atrial septal defects (ASDs) are one of the most
Therapeutic Cardiac Catheterizations for Children with Congenital Heart Disease
 Therapeutic Cardiac Catheterizations for Children with Congenital Heart Disease Introduction A therapeutic cardiac catheterization is a procedure performed to treat your child s heart defect. A doctor
Therapeutic Cardiac Catheterizations for Children with Congenital Heart Disease Introduction A therapeutic cardiac catheterization is a procedure performed to treat your child s heart defect. A doctor
ABBOTT CODING GUIDE STRUCTURAL HEART AND VALVES CONGENITAL DEFECTS SURGICAL HEART VALVES AMPLATZER PFO OCCLUDER. Effective January 1, 2019
 ABBOTT CODING GUIDE STRUCTURAL HEART AND VALVES Effective January 1, 2019 STRUCTURAL HEART AND VALVES Effective Janaury 1, 2019 Introduction The Structural Heart and Valves Coding Guide is intended to
ABBOTT CODING GUIDE STRUCTURAL HEART AND VALVES Effective January 1, 2019 STRUCTURAL HEART AND VALVES Effective Janaury 1, 2019 Introduction The Structural Heart and Valves Coding Guide is intended to
Description. Page: 1 of 23. Closure Devices for Patent Foramen Ovale and Atrial Septal Defects. Last Review Status/Date: December 2014
 Last Review Status/Date: December 2014 Page: 1 of 23 Description Background Patent Foramen Ovale The foramen ovale, a component of fetal cardiovascular circulation, consists of a communication between
Last Review Status/Date: December 2014 Page: 1 of 23 Description Background Patent Foramen Ovale The foramen ovale, a component of fetal cardiovascular circulation, consists of a communication between
Debate in Management of native COA; Balloon Versus Surgery
 Debate in Management of native COA; Balloon Versus Surgery Dr. Amira Esmat, El Tantawy, MD Professor of Pediatrics Consultant Pediatric Cardiac Interventionist Faculty of Medicine Cairo University 23/2/2017
Debate in Management of native COA; Balloon Versus Surgery Dr. Amira Esmat, El Tantawy, MD Professor of Pediatrics Consultant Pediatric Cardiac Interventionist Faculty of Medicine Cairo University 23/2/2017
ΔΙΑΧΕΙΡΙΣΗ ΑΣΘΕΝΩΝ ΜΕ ΜΕΣΟΚΟΛΠΙΚΗ ΕΠΙΚΟΙΝΩΝΙΑ ΖΑΧΑΡΑΚΗ ΑΓΓΕΛΙΚΗ ΚΑΡΔΙΟΛΟΓΟΣ ΗΡΑΚΛΕΙΟ - ΚΡΗΤΗ
 ΔΙΑΧΕΙΡΙΣΗ ΑΣΘΕΝΩΝ ΜΕ ΜΕΣΟΚΟΛΠΙΚΗ ΕΠΙΚΟΙΝΩΝΙΑ ΖΑΧΑΡΑΚΗ ΑΓΓΕΛΙΚΗ ΚΑΡΔΙΟΛΟΓΟΣ ΗΡΑΚΛΕΙΟ - ΚΡΗΤΗ European Accreditation in TTE, TEE and CHD Echocardiography NOTHING TO DECLARE ATRIAL SEPTAL DEFECT TYPES SECUNDUM
ΔΙΑΧΕΙΡΙΣΗ ΑΣΘΕΝΩΝ ΜΕ ΜΕΣΟΚΟΛΠΙΚΗ ΕΠΙΚΟΙΝΩΝΙΑ ΖΑΧΑΡΑΚΗ ΑΓΓΕΛΙΚΗ ΚΑΡΔΙΟΛΟΓΟΣ ΗΡΑΚΛΕΙΟ - ΚΡΗΤΗ European Accreditation in TTE, TEE and CHD Echocardiography NOTHING TO DECLARE ATRIAL SEPTAL DEFECT TYPES SECUNDUM
Adult Echocardiography Examination Content Outline
 Adult Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 2 3 4 5 Anatomy and Physiology Pathology Clinical Care and Safety Measurement Techniques, Maneuvers,
Adult Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 2 3 4 5 Anatomy and Physiology Pathology Clinical Care and Safety Measurement Techniques, Maneuvers,
(2013 ) ACHD ACHD
 (0 ). ) ) ) ) ) ) (ACHD) / ( ) ACHD ACHD 0 6 (9 ) 9 8 8 6 0 6 / ACHD ACHD Adult congenital heart disease, Pediatric cardiologists, Adult cardiologists, Emergency admission, Transfer of the patients (congenital
(0 ). ) ) ) ) ) ) (ACHD) / ( ) ACHD ACHD 0 6 (9 ) 9 8 8 6 0 6 / ACHD ACHD Adult congenital heart disease, Pediatric cardiologists, Adult cardiologists, Emergency admission, Transfer of the patients (congenital
Intracardiac echocardiography: an ideal guiding tool for device closure of interatrial communications
 Eur J Echocardiography (2005) 6, 92e96 Intracardiac echocardiography: an ideal guiding tool for device closure of interatrial communications Thomas Bartel a, *, Thomas Konorza a, Ulrich Neudorf b, Tiko
Eur J Echocardiography (2005) 6, 92e96 Intracardiac echocardiography: an ideal guiding tool for device closure of interatrial communications Thomas Bartel a, *, Thomas Konorza a, Ulrich Neudorf b, Tiko
Description. Section: Surgery Effective Date: April 15, Subsection: Surgery Original Policy Date: December 6, 2012 Subject:
 Last Review Status/Date: March 2015 Page: 1 of 6 Description Wireless sensors implanted in an aortic aneurysm sac after endovascular repair are being investigated to measure post procedural pressure. It
Last Review Status/Date: March 2015 Page: 1 of 6 Description Wireless sensors implanted in an aortic aneurysm sac after endovascular repair are being investigated to measure post procedural pressure. It
Congenital heart disease. By Dr Saima Ali Professor of pediatrics
 Congenital heart disease By Dr Saima Ali Professor of pediatrics What is the most striking clinical finding in this child? Learning objectives By the end of this lecture, final year student should be able
Congenital heart disease By Dr Saima Ali Professor of pediatrics What is the most striking clinical finding in this child? Learning objectives By the end of this lecture, final year student should be able
Policy Specific Section: March 30, 2012 March 7, 2013
 Medical Policy Transcatheter Aortic Valve Replacement for Aortic Stenosis Type: Medical Necessity and Investigational / Experimental Policy Specific Section: Surgery Original Policy Date: Effective Date:
Medical Policy Transcatheter Aortic Valve Replacement for Aortic Stenosis Type: Medical Necessity and Investigational / Experimental Policy Specific Section: Surgery Original Policy Date: Effective Date:
Original Policy Date
 MP 2.01.26 Laser-Assisted Myringotomy and Tympanostomy Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed with literature search/12:2013 Return
MP 2.01.26 Laser-Assisted Myringotomy and Tympanostomy Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed with literature search/12:2013 Return
The Adolescent and Adult Congenital Heart Disease Program
 The Adolescent and Adult Congenital Heart Disease Program The Heart Center at Nationwide Children s Hospital & The Ohio State University D- Transposition of the Great Vessels D- transposition of the great
The Adolescent and Adult Congenital Heart Disease Program The Heart Center at Nationwide Children s Hospital & The Ohio State University D- Transposition of the Great Vessels D- transposition of the great
Centers for Medicare and Medicaid Services. National Coverage of Transcatheter Valve Technologies December 2015
 Centers for Medicare and Medicaid Services National Coverage of Transcatheter Valve Technologies December 2015 National Coverage Determination (NCD) for Transcatheter Aortic Valve Replacement (TAVR) (20.32)
Centers for Medicare and Medicaid Services National Coverage of Transcatheter Valve Technologies December 2015 National Coverage Determination (NCD) for Transcatheter Aortic Valve Replacement (TAVR) (20.32)
Patent Ductus Arteriosus (PDA)
 Patent Ductus Arteriosus (PDA) How does the heart work? The heart is the organ responsible for pumping blood to and from all tissues of the body. The heart is divided into right and left sides. The job
Patent Ductus Arteriosus (PDA) How does the heart work? The heart is the organ responsible for pumping blood to and from all tissues of the body. The heart is divided into right and left sides. The job
Implantation of Cardioverter Defibrillator After Percutaneous Closure of Atrial Septal Defect
 The Ochsner Journal 10:27 31, 2010 f Academic Division of Ochsner Clinic Foundation Implantation of Cardioverter Defibrillator After Percutaneous Closure of Atrial Septal Defect Anas Bitar, MD, Maria Malaya
The Ochsner Journal 10:27 31, 2010 f Academic Division of Ochsner Clinic Foundation Implantation of Cardioverter Defibrillator After Percutaneous Closure of Atrial Septal Defect Anas Bitar, MD, Maria Malaya
Nit-Occlud. Coil System for PDA Closure PATIENT EDUCATIONAL BROCHURE
 Nit-Occlud PDA Coil System for PDA Closure PATIENT EDUCATIONAL BROCHURE A Patient s Educational Brochure to Non-Surgical Closure of the Patent Ductus Arteriosus (PDA) Using the Nit-Occlud PDA Inside This
Nit-Occlud PDA Coil System for PDA Closure PATIENT EDUCATIONAL BROCHURE A Patient s Educational Brochure to Non-Surgical Closure of the Patent Ductus Arteriosus (PDA) Using the Nit-Occlud PDA Inside This
2) VSD & PDA - Dr. Aso
 2) VSD & PDA - Dr. Aso Ventricular Septal Defect (VSD) Most common cardiac malformation 25-30 % Types of VSD: According to position perimembranous, inlet, muscular. According to size small, medium, large.
2) VSD & PDA - Dr. Aso Ventricular Septal Defect (VSD) Most common cardiac malformation 25-30 % Types of VSD: According to position perimembranous, inlet, muscular. According to size small, medium, large.
Nit-Occlud. Coil System for PDA Closure IMPLANTATION POCKET GUIDE. Rx only CV / B. Braun Interventional Systems Inc.
 Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
Clinicians and Facilities: RESOURCES WHEN CARING FOR WOMEN WITH ADULT CONGENITAL HEART DISEASE OR OTHER FORMS OF CARDIOVASCULAR DISEASE!!
 Clinicians and Facilities: RESOURCES WHEN CARING FOR WOMEN WITH ADULT CONGENITAL HEART DISEASE OR OTHER FORMS OF CARDIOVASCULAR DISEASE!! Abha'Khandelwal,'MD,'MS' 'Stanford'University'School'of'Medicine'
Clinicians and Facilities: RESOURCES WHEN CARING FOR WOMEN WITH ADULT CONGENITAL HEART DISEASE OR OTHER FORMS OF CARDIOVASCULAR DISEASE!! Abha'Khandelwal,'MD,'MS' 'Stanford'University'School'of'Medicine'
Original Policy Date
 MP 8.03.02 Physical Therapy Medical Policy Section Therapy Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed by consensus/12/2013 Return to Medical Policy Index Disclaimer Our
MP 8.03.02 Physical Therapy Medical Policy Section Therapy Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed by consensus/12/2013 Return to Medical Policy Index Disclaimer Our
Transcatheter Pulmonary Valve Implantation
 Transcatheter Pulmonary Valve Implantation Policy Number: Original Effective Date: MM.06.022 09/01/2013 Lines of Business: Current Effective Date: HMO; PPO; QUEST Integration 01/23/2015 Section: Surgery
Transcatheter Pulmonary Valve Implantation Policy Number: Original Effective Date: MM.06.022 09/01/2013 Lines of Business: Current Effective Date: HMO; PPO; QUEST Integration 01/23/2015 Section: Surgery
Transcatheter Occlusion of Patent Ductus Arteriosus With a New Detachable Coil System (DuctOcclud)
 Jpn Circ J 1998; 62: 489 493 Transcatheter Occlusion of Patent Ductus Arteriosus With a New Detachable Coil System (DuctOcclud) a Multicenter Clinical Trial Shinichi Oho, MD; Akira Ishizawa, MD; Kazuyuki
Jpn Circ J 1998; 62: 489 493 Transcatheter Occlusion of Patent Ductus Arteriosus With a New Detachable Coil System (DuctOcclud) a Multicenter Clinical Trial Shinichi Oho, MD; Akira Ishizawa, MD; Kazuyuki
Uptofate Study Summary
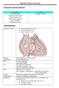 CONGENITAL HEART DISEASE Uptofate Study Summary Acyanotic Atrial septal defect Ventricular septal defect Patent foramen ovale Patent ductus arteriosus Aortic coartation Pulmonary stenosis Cyanotic Tetralogy
CONGENITAL HEART DISEASE Uptofate Study Summary Acyanotic Atrial septal defect Ventricular septal defect Patent foramen ovale Patent ductus arteriosus Aortic coartation Pulmonary stenosis Cyanotic Tetralogy
In the current era, transcatheter occlusion of the patent
 Safety of Percutaneous Patent Ductus Arteriosus Closure: An Unselected Multicenter Population Experience Howaida G. El-Said, MD, PhD; Andras Bratincsak, MD, PhD; Susan R. Foerster, MD; Joshua J. Murphy,
Safety of Percutaneous Patent Ductus Arteriosus Closure: An Unselected Multicenter Population Experience Howaida G. El-Said, MD, PhD; Andras Bratincsak, MD, PhD; Susan R. Foerster, MD; Joshua J. Murphy,
Stroke and ASA / FO REBUTTAL
 REBUTTAL Definition of an ischemic stroke Definition of a PFO Evidence for a causal role of PFO in stroke Evidence against a role of PFO in stroke Stroke recurrencies in stroke (PFO) patients Medical treatment
REBUTTAL Definition of an ischemic stroke Definition of a PFO Evidence for a causal role of PFO in stroke Evidence against a role of PFO in stroke Stroke recurrencies in stroke (PFO) patients Medical treatment
Mitral Valve Disease, When to Intervene
 Mitral Valve Disease, When to Intervene Swedish Heart and Vascular Institute Ming Zhang MD PhD Interventional Cardiology Structure Heart Disease Conflict of Interest None Current ACC/AHA guideline Stages
Mitral Valve Disease, When to Intervene Swedish Heart and Vascular Institute Ming Zhang MD PhD Interventional Cardiology Structure Heart Disease Conflict of Interest None Current ACC/AHA guideline Stages
CPT Code Details
 CPT Code 93572 Details Code Descriptor Intravascular Doppler velocity and/or pressure derived flow reserve measurement ( vessel or graft) during angiography pharmacologically induced stress; each additional
CPT Code 93572 Details Code Descriptor Intravascular Doppler velocity and/or pressure derived flow reserve measurement ( vessel or graft) during angiography pharmacologically induced stress; each additional
CARDIAC INTERVENTIONS IN PEDIATRIC CARDIOLOGY: THE FUTURE
 CARDIAC INTERVENTIONS IN PEDIATRIC CARDIOLOGY: THE FUTURE Vikas Kohli From the: Senior Consultant Pediatric Cardiology, Apollo Centre of Advance Pediatrics, Indraprastha Apollo Hospitals, New Delhi-110044.
CARDIAC INTERVENTIONS IN PEDIATRIC CARDIOLOGY: THE FUTURE Vikas Kohli From the: Senior Consultant Pediatric Cardiology, Apollo Centre of Advance Pediatrics, Indraprastha Apollo Hospitals, New Delhi-110044.
Tetralogy of Fallot Latest data in risk stratification and replacement of pulmonic valve
 Tetralogy of Fallot Latest data in risk stratification and replacement of pulmonic valve Alexandra A Frogoudaki Adult Congenital Heart Clinic Second Cardiology Department ATTIKON University Hospital No
Tetralogy of Fallot Latest data in risk stratification and replacement of pulmonic valve Alexandra A Frogoudaki Adult Congenital Heart Clinic Second Cardiology Department ATTIKON University Hospital No
