PARA-TECT Cryptosporidium/Giardia Direct Fluorescent Assay Directions For Use For In Vitro Diagnostic Use
|
|
|
- Jonah Parrish
- 5 years ago
- Views:
Transcription
1 Medical Chemical Corp., Van Ness Avenue, Torrance, California (310) , Fax (310) Catalog # MCC-C/G - DFA, 75 Test PARA-TECT Cryptosporidium/Giardia Direct Fluorescent Assay Directions For Use For In Vitro Diagnostic Use Intended Use This direct fluorescent assay (DFA) is an in vitro immunoassay for the qualitative determination of Cryptosporidium and Giardia oocysts and cysts in feces. This assay may be used with stools preserved in 10% formalin, SAF or MCC s Universal Fixative. Summary and Explanation Giardia lamblia is the protozoan parasite responsible for the disease giardiasis. Symptoms of acute giardiasis include diarrhea, nausea, weight loss, malabsorption, abdominal cramps, flatulence and anemia. 1 The disease may manifest itself as an acute, chronic or as an asymptomatic infection. Giardiasis is the most prevalent parasitic disease in the United States and is responsible for an estimated 100 million mild infections and 1 million severe infections each year. 3 The mode of transmission of Giardia is through fecal-oral ingestion of cysts. Epidemics of giardiasis have been documented in day care centers and by drinking contaminated water. 1,2 Day care centers may be directly or indirectly responsible for 45% of diagnosed Giardia infections in the United States. 4 One study found 54% of the children at a day care center were infected. 1 Another important source of Giardia infection is among homosexual men. Prevalence rates of 5 to 19% for this population have been reported. 16 Cryptosporidium is a coccidian parasite that is recognized as an important enteric pathogen. The organism causes an acute, though selflimiting infection in immunocompetent individuals. Incubation periods of 1 to 12 days have been reported with most oocyst shedding ending by day 21. Symptoms range from mild to severe diarrhea with a variety of complications. 5,6,7,8,9,10 The infection in immunocompromised patients is much more severe and may often be life threatening. Passage of fluid, up to 12 liters per day, has been reported. 5,11,12,13,14,15 Multiple pathways of Cryptosporidium transmission have been implicated. These include animal to human, water contamination and person-to-person. The latter may include contact between members of the same household, day care centers, and homosexual men. 5,11,13,14,15 Principle of Procedure This assay utilizes the principle of direct immunofluorescence. The conjugate contains a mixture of FITC-labeled monoclonal antibodies directed against Cryptosporidium oocysts and Giardia cysts, which, if present, are affixed to the slide. The slide is then rinsed to remove unbound conjugate and examined under a fluorescent microscope looking for an apple-green color and the characteristic morphology of the Cryptosporidium oocysts and the Giardia cysts. Reagents Conjugate: One 4 ml vial of FITC-labeled anti-cryptosporidium and anti-giardia monoclonal antibodies Positive Control: One 1 ml vial of formalinized stool supernatant containing Cryptosporidium oocysts and Giardia cysts Negative Control: One 1 ml vial of formalinized stool supernatant Wash Buffer: One 25 ml bottle of concentrated (20X) buffer Counterstain: One 4 ml bottle of Eriochrome Black Mounting Media: One 3 ml vial Transfer loops: One package of 80 Slides: One package of 25, 3-well treated slides Slide Coverslips: One Package of 25 Instructions for Catalog # MCC-C/G-DFA, Page 1 of 5
2 Warnings/Precautions For In Vitro Diagnostic Use Do not use solutions if they precipitate or become cloudy. Exception: Wash concentrate may precipitate during refrigerated storage, but will dissolve upon warming. Do not add azides to the samples or any of the reagents. Controls and some reagents contain Thimerosal as a preservative. Treat all reagents and samples as potentially infectious materials. Use care to prevent aerosols and decontaminate any spills of samples. Persons who are color blind or visually impaired may not be able to read the test and should use another method (such as an EIA) to test the samples. Incubation Stage 2 should be performed in a light protected area. Take care not to scratch the surface of the slide test wells. Storage Conditions Store between 2-8º C. Squeeze bottle containing diluted wash buffer may be stored at room temperature. Specimen Collection and Preparation Collection of Stool (Feces) No modification of collection techniques used for standard microscopic O&P examinations is needed. Stool samples may be preserved in 10% formalin, SAF, MCC s Unifix or Total-Fix. Preserved samples may be kept at room temperature (15-25º C) or refrigerated. Wash Buffer Preparation Remove cap and add contents of one bottle of Wash Concentrate to a squeeze bottle containing 475 ml of DI water.. Swirl to mix. Preparation of Preserved Stools Mix contents thoroughly inside collection container. No further processing is required. Samples with low numbers of oocysts or cysts may benefit by concentration of the sample prior to testing. Use of the Micro-Sed, Sed- Connect and Para-Sed concentration kits (Medical Chemical Corporation, Torrance, CA) have been validated with this assay. Cryptosporidium/Giardia DFA Kit Procedure Materials Provided Materials Required But Not Provided Applicator Sticks Squeeze bottle for washing strips Graduated Cylinder Reagent grade (DI) water Humidity Chamber (place in light protected area) Fluorescent Microscope (excitation wavelength of , barrier filter of ) Proper Temperature All incubations are at room temperature (15 to 25º C) Test Procedure INITIAL SETUP 1. Bring the 20X Wash Solution to 500 ml with distilled water. MIX WELL 2. Fill a squeeze bottle with the resulting 1X wash buffer. 3. Thoroughly mix all reagents and specimens before use INCUBATION STAGE 1 4. Use a new transfer loop to transfer the Positive Control to a slide well. One can also use a transfer pipet to dispense one drop onto the slide. Carefully spread the material over the entire well taking care not to scratch the treated surface. 5. Use a new transfer loop to transfer the Negative Control to a slide well. One can also use a transfer pipet to dispense one drop onto the slide. Carefully spread the material over the entire well taking care not to scratch the treated surface. 6. Use a new transfer loop to transfer each patient s stool specimen to a slide well. Carefully spread the material over the entire well Instructions for Catalog # MCC-C/G-DFA, Page 2 of 5
3 taking care not to scratch the treated surface. 7. Allow the slides to completely air-dry. This process usually takes between minutes depending upon environmental conditions. INCUBATION STAGE 2 8. Add 50 μl (1 drop) of the Conjugate to all used slide wells 9. Add 50 μl (1 drop) of the Counterstain to all used slide wells 10. Mix reagents with applicator stick and spread over entire well taking care not to scratch the treated surface. 11. Incubate the slides in a light-protected humidity chamber for 30 minutes. RINSE STAGE 12. Using the squeeze bottle from step 2, gently rinse the slides to remove excess conjugate and counterstain. 13. Tap the edge of the slide against a paper towel to remove excess buffer. DO NOT ALLOW SLIDE TO DRY. 14. Add 1 drop of mounting medium to each well and apply coverslip. INTERPRETATION STAGE 15. Scan each well at 400X looking for the presence of the characteristic FITC brilliant apple-green color and morphology identified in the Quality Control Section. Results Cryptosporidium Positive: The presence of at least one oocyst with the characteristic FITC brilliant apple-green color and morphology identified in the Quality Control Section. Giardia Positive: The presence of at least one cyst with the characteristic FITC brilliant apple-green color and morphology identified in the Quality Control Section. Negative: The absence of the characteristic FITC brilliant apple-green color and morphology identified in the Quality Control Section. Limitation of Procedure Test results should be used as an aid in diagnosis and should not be interpreted as diagnostic by themselves. The presence of Cryptosporidium oocysts and Giardia cysts does not preclude concurrent infections by other pathogens. Expected Values Normal healthy individuals should be free of Cryptosporidium and Giardia and should test negative. A positive reaction indicates that the patient is shedding detectable amounts of oocysts or cysts. This shedding may continue in some patients even after clinical recovery. Shedding may also occur in asymptomatic cases. Specific Performance Characteristics Study #1 A total of 170 unconcentrated stools (145 human stools and 25 bovine stools) examined by O&P microscopy were tested against the IVD DFA kit. The following results were obtained. DFA DFA Sensitivity: 100% 95% CI = 92% to 100% Specificity: 100% 95% CI = 97% to 100% Instructions for Catalog # MCC-C/G-DFA, Page 3 of 5
4 DFA DFA Sensitivity: 100% 95% CI = 91% to 100% Specificity: 100% 95% CI = 97% to 100% Study #2 A total of 53 unconcentrated stools examined by O&P microscopy were tested against the IVD DFA kit. The following results were obtained. DFA DFA Sensitivity: 100%95% CI = 79% to 100% Specificity: 100%95% CI = 90% to 100% DFA DFA Sensitivity: 100% 95% CI = 72% to 100% Specificity: 100% 95% CI = 92% to 100% Study #3 A total of 74 formalin and SAF preserved stools were tested against the IVD DFA test. DFA DFA Sensitivity: 100% 95% CI = 87% to 100% Specificity: 100% 95% CI = 93% to 100% DFA DFA Sensitivity: 95% 95% CI = 74% to 100% Specificity: 100% 95% CI = 94% to 100% No cross-reactions were seen with the following organisms: Entamoeba hartmanni, Endolimax nana, Entamoeba histolytica/dispar, Entamoeba coli, Blastocystis hominis, Dientamoeba fragilis, Chilomastix mesnili, Strongyloides stercoralis, Ascaris lumbricoides, Enterobius vermicularis, Diphyllobothrium species, Hymenolepis nana, Enteromonas hominis, Trichuris trichiura, Iodamoeba buetschlii, Hookworm, Taenia eggs, White Blood Cells, Cyclospora cayetanensis. Summary of Reproducibility Data Nine samples (3 negative, 3 Giardia positive and 3 Cryptosporidium positive) were tested by 3 different persons. All 3 people correctly identified the negative and positive samples. Instructions for Catalog # MCC-C/G-DFA, Page 4 of 5
5 Quality Control The positive and negative controls must be included whenever patient testing performed. Positive Control: Must show the presence of both the Cryptosporidium oocyst and the Giardia cyst. Cryptosporidium oocysts are 2-6μm in size are round to slightly ovoid in shape. The oocyst wall will stain the characteristic FITC brilliant apple-green color. Giardia cysts are 8-12μm in size and are oval shaped. The cyst wall will stain the characteristic FITC brilliant apple-green color. Negative Control: Must not exhibit any of the characteristic FITC brilliant apple-green color associated with either the oocyst or cyst morphology Background Staining: The background material in the control wells and patient wells will counterstain a dull orange-red color. There also may be non-specific fluorescence that is easily distinguished from the brilliant apple-green color and morphology of the oocyst and cyst. References 1. Black, R.. A.C. Dykes, S.P. Sinclair, and J.G. Wells. Giardiasis in day-care centers: evidence of person- to-person transmission Pediatrics. 60(4): Craun, G.. Waterborne giardiasis in the United States. Lancet Intestinal protozoa: cinderellas of parasitology. ASM News (10): Peters, C.S., R. Sable, W.M. Janda, A.L. Chittom, and F.E. Kocka.. Prevalence of enteric parasites in homosexual patients attending an outpatient clinic. J. Clin. Microbiol (4): Chapman, P.A. Cryptosporidiosis: recent trends in epidemiology, diagnosis, and treatment. Serodiag. & Immunother. Infect. Dis : Jokipii, L., S. Pohjola, and A.M. Jokipii. Cryptosporidium: a frequent finding in patients with gastrointestinal symptoms. Lancet (8346): Shepherd, R.C., C.L. Reed, and G.P. Sinha. Shedding of oocysts of Cryptosporidium in immunocompetent patients. J. Clin. Pathol (10): Holten-Andersen, W., J. Gerstoft, S.A. Henriksen, and N.S. Pedersen. Prevalence of Cryptosporidium Among patients with acute enteric infection. J. Infect (3): Jokipii, L. and M. Jokipii. Timing of symptoms and oocyst excretion in human Cryptosporidiosis. N. Engl. J. Med (26): Hart, M.H., R. Kruger, S. Nielsen, and S. Kaufman. Acute self-limited colitis associated with Cryptosporidium in an immunocompetent patient. J. Pediatr. Gastroenterol. Nutr (3): Meyer, E.A. Waterborne Giardia and Cryptosporidium. Parasit. Today (7): Garcia, L.S., D. Bruckner, and T. Brewer. Cryptosporidosis in patients with AIDS. ACPR. May Egger, M., D. Mausezahl, P. Odermatt, H.P. Marti, and M. Tanner. Symptoms and transmission of intestinal cryptosporidiosis. Arch. Dis. Child (4): Nwanyanwu, O., J.N. Baird, and G.R. Reeve. Cryptosporidiosis in a day-care Center. Texas Med (6): Current, W.L., and L.S. Garcia. Cryptosporidiosis. Clin. Microbiol. Rev (3): Kappus, K. and D. Juranek. Giardia in the well. JAMA (12): Revision Date: Manufactured by: Medical Chemical Corp Van Ness Ave. Torrance, CA USA Telephone: , FAX: Instructions for Catalog # MCC-C/G-DFA, Page 5 of 5
Crypto / Giardia Ag Combo (Fecal) ELISA
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Cryptosporidium Veterinary
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Catalog # OKDA00123 SUMMARY AND EXPLANATION
 Aviva Systems Biology 5754 Pacific Center Blvd., Suite 201, San Diego, CA 92121, USA Tel: 858-552-6979 Fax: 858-552-6975 www.avivasysbio.com Email: info@avivasysbio.com Giardia lamblia ELISA Kit Catalog
Aviva Systems Biology 5754 Pacific Center Blvd., Suite 201, San Diego, CA 92121, USA Tel: 858-552-6979 Fax: 858-552-6975 www.avivasysbio.com Email: info@avivasysbio.com Giardia lamblia ELISA Kit Catalog
Sure-Vue Signature Cryptosporidium/Giardia Test
 SAMPLE PROCEDURE This procedure is provided to Fisher Healthcare customers to assist with the development of laboratory procedures. This document was derived from, and was current with, the instructions
SAMPLE PROCEDURE This procedure is provided to Fisher Healthcare customers to assist with the development of laboratory procedures. This document was derived from, and was current with, the instructions
American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX
 Q3 2018 Parasitology American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX 77005 800-234-5315 281-436-5357 Specimen 1 Referees Extent 1 Extent 2 Total Few to 534 Giardia lamblia Many
Q3 2018 Parasitology American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX 77005 800-234-5315 281-436-5357 Specimen 1 Referees Extent 1 Extent 2 Total Few to 534 Giardia lamblia Many
News and Notes. Parasitology Comprehensive 2 October Sample Preparation and Quality Control. 12 K (All Parasites)
 NEW YORK STATE Parasitology Proficiency Testing Program News and Notes Recent reports in the literature have indicated a high rate of Cryptosporidium sp. false positive associated with Rapid Cartridge
NEW YORK STATE Parasitology Proficiency Testing Program News and Notes Recent reports in the literature have indicated a high rate of Cryptosporidium sp. false positive associated with Rapid Cartridge
Coccidians. Cryptosporidium Cystoisospora belli Cyclospora cayetanensis. by author
 Coccidians Cryptosporidium Cystoisospora belli Cyclospora cayetanensis Diagnostic techniques Cryptosporidium Cyclospora cayetanensis Cystoisospora belli sporulation In host weeks days seize 2-5 μm
Coccidians Cryptosporidium Cystoisospora belli Cyclospora cayetanensis Diagnostic techniques Cryptosporidium Cyclospora cayetanensis Cystoisospora belli sporulation In host weeks days seize 2-5 μm
News and Notes. Parasitology Comprehensive 5 November 2013
 NEW YORK STATE Parasitology Proficiency Testing Program News and Notes Beginning with the May 2013 event a separate set of 3 samples were supplied for laboratories performing antigen detection. For the
NEW YORK STATE Parasitology Proficiency Testing Program News and Notes Beginning with the May 2013 event a separate set of 3 samples were supplied for laboratories performing antigen detection. For the
Cryptosporidium spp. ELISA Kit
 Cryptosporidium spp. ELISA Kit Catalog Number KA2094 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Cryptosporidium spp. ELISA Kit Catalog Number KA2094 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
COMPREHENSIVE STOOL ANALYSIS
 COMPREHENSIVE STOOL ANALYSIS Intestinal parasites: Normal value = 0 (not marked) Reference range: 0 (negative) - 4 (heavy presence) Specimens fixed and transported in SAF and concentrated using CONSED
COMPREHENSIVE STOOL ANALYSIS Intestinal parasites: Normal value = 0 (not marked) Reference range: 0 (negative) - 4 (heavy presence) Specimens fixed and transported in SAF and concentrated using CONSED
Campylobacter Antigen ELISA Kit
 Campylobacter Antigen ELISA Kit Catalog Number KA3204 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Campylobacter Antigen ELISA Kit Catalog Number KA3204 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Giardia lamblia/ Cryptosporidium spp. ELISA Kit
 Giardia lamblia/ Cryptosporidium spp. ELISA Kit Catalog Number KA2095 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background...
Giardia lamblia/ Cryptosporidium spp. ELISA Kit Catalog Number KA2095 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background...
Origination Date: 25 Mar 2004 Total Pages: Effective Date: 18 Sep 2012 SOP Number LTC-SOP-17 v2.0. Network Name, Title Signature Date
 Title: Stool Processing and Cryptosporidium/Giardia Origination Date: 25 Mar 2004 Total Pages: Effective Date: 18 Sep 2012 SOP Number LTC-SOP-17 v2.0 Prepared By: Bill Kabat and Nancy Isham of the Supersedes
Title: Stool Processing and Cryptosporidium/Giardia Origination Date: 25 Mar 2004 Total Pages: Effective Date: 18 Sep 2012 SOP Number LTC-SOP-17 v2.0 Prepared By: Bill Kabat and Nancy Isham of the Supersedes
CoproELISA Giardia/Cryptosporidium
 CoproELISA Giardia/Cryptosporidium Enzyme-Linked Immunosorbent Assay (ELISA) for the detection of Giardia lamblia and Cryptosporidium spp. antigens in human feces Instruction Manual Test kit for 96 determinations
CoproELISA Giardia/Cryptosporidium Enzyme-Linked Immunosorbent Assay (ELISA) for the detection of Giardia lamblia and Cryptosporidium spp. antigens in human feces Instruction Manual Test kit for 96 determinations
E. Histolytica IgG (Amebiasis)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
NEW YORK STATE Parasitology Proficiency Testing Program. Parasitology (General) 02 February Sample Preparation and Quality Control
 NEW YORK STATE Parasitology Proficiency Testing Program Parasitology (General) 02 February 2010 The purpose of the New York State Proficiency Testing Program in the category of Parasitology (General) is
NEW YORK STATE Parasitology Proficiency Testing Program Parasitology (General) 02 February 2010 The purpose of the New York State Proficiency Testing Program in the category of Parasitology (General) is
Recent Diagnostic Methods for Intestinal Parasitic Infections
 Recent Diagnostic Methods for Intestinal Parasitic Infections By Dr. Doaa Abdel Badie Salem Lecturer of Medical Parasitology, Mansoura Faculty of Medicine Agenda Intestinal parasites. Traditional Diagnostic
Recent Diagnostic Methods for Intestinal Parasitic Infections By Dr. Doaa Abdel Badie Salem Lecturer of Medical Parasitology, Mansoura Faculty of Medicine Agenda Intestinal parasites. Traditional Diagnostic
Giardia lamblia ELISA Kit
 Giardia lamblia ELISA Kit Catalog Number KA2093 96 assay Version: 01 Intend for research use only www.abnova.com Introduction and Background A. Intended Use Giardia lamblia ELISA Kit is an Enzyme-Linked
Giardia lamblia ELISA Kit Catalog Number KA2093 96 assay Version: 01 Intend for research use only www.abnova.com Introduction and Background A. Intended Use Giardia lamblia ELISA Kit is an Enzyme-Linked
E. Histolytica IgG ELISA Kit
 E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
ELISA TEST FOR DETECTION OF BLASTOCYSTIS SPP. IN HUMAN FAECES: COMPARISON OF THREE METHODS
 ELISA TEST FOR DETECTION OF BLASTOCYSTIS SPP. IN HUMAN FAECES: COMPARISON OF THREE METHODS István Kucsera, Mónika Molnár, Eszter Gályász, József Danka, Erika Orosz National Center for Epidemiology, Department
ELISA TEST FOR DETECTION OF BLASTOCYSTIS SPP. IN HUMAN FAECES: COMPARISON OF THREE METHODS István Kucsera, Mónika Molnár, Eszter Gályász, József Danka, Erika Orosz National Center for Epidemiology, Department
Overview IMPORTANCE CLASSIFICATION SPECIMEN COLLECTION PROTOZOA WORMS BLOOD PARASITES ARTHROPODS DELUSIONAL PARASITOSIS QUIZZES GROSS
 IMPORTANCE Overview CLASSIFICATION SPECIMEN COLLECTION PROTOZOA WORMS BLOOD PARASITES ARTHROPODS DELUSIONAL PARASITOSIS QUIZZES GROSS - Of 6.6 billion people on Earth, 2.7 billion live on
IMPORTANCE Overview CLASSIFICATION SPECIMEN COLLECTION PROTOZOA WORMS BLOOD PARASITES ARTHROPODS DELUSIONAL PARASITOSIS QUIZZES GROSS - Of 6.6 billion people on Earth, 2.7 billion live on
A New Multiplex Real-time PCR Assay For Detection Of Intestinal Parasites
 A New Multiplex Real-time PCR Assay For Detection Of Intestinal Parasites Dr. Andreas Simons Worldwide provider of diagnostic assay solutions Offers a variety of test kit methodologies R-Biopharm Headquarters
A New Multiplex Real-time PCR Assay For Detection Of Intestinal Parasites Dr. Andreas Simons Worldwide provider of diagnostic assay solutions Offers a variety of test kit methodologies R-Biopharm Headquarters
PARATEST STABILITY STUDY, SYSTEM USED FOR DIAGNOSIS OF INTESTINAL PARASITES
 PARATEST STABILITY STUDY, SYSTEM USED FOR DIAGNOSIS OF INTESTINAL PARASITES Rev.00 January 10, 2011 1. Introduction Intestinal parasitic diseases represent a serious public health issue in underdeveloped
PARATEST STABILITY STUDY, SYSTEM USED FOR DIAGNOSIS OF INTESTINAL PARASITES Rev.00 January 10, 2011 1. Introduction Intestinal parasitic diseases represent a serious public health issue in underdeveloped
Hompes Method. Practitioner Training Level II. Lesson Five (b) Bad Bugs - Parasites
 Hompes Method Practitioner Training Level II Lesson Five (b) Bad Bugs - Parasites Health for the People Ltd not for reuse without expressed permission Hompes Method is a trading name of Health For The
Hompes Method Practitioner Training Level II Lesson Five (b) Bad Bugs - Parasites Health for the People Ltd not for reuse without expressed permission Hompes Method is a trading name of Health For The
News and Notes. Parasitology Comprehensive 5 February Sample Preparation and Quality Control. 13 A Helminthes Only
 NEW YORK STATE Parasitology Proficiency Testing Program News and Notes Recent reports in the literature have indicated a high rate of Cryptosporidium sp. false positive associated with Rapid Cartridge
NEW YORK STATE Parasitology Proficiency Testing Program News and Notes Recent reports in the literature have indicated a high rate of Cryptosporidium sp. false positive associated with Rapid Cartridge
TYPES OF ORGANISM RELATIONSHIPS
 TYPES OF ORGANISM RELATIONSHIPS Normal Flora. Normal flora consists of microorganisms that are normally and consistently found in or on the body in the absence of disease. Symbiosis. This is the close
TYPES OF ORGANISM RELATIONSHIPS Normal Flora. Normal flora consists of microorganisms that are normally and consistently found in or on the body in the absence of disease. Symbiosis. This is the close
Giardia lamblia (flagellates)
 Giardia lamblia (flagellates) Dr. Hala Al Daghistani Giardia lamblia (Giardia duodenalis or Giardia intestinalis) is the causative agent of giardiasis and is the only common pathogenic protozoan found
Giardia lamblia (flagellates) Dr. Hala Al Daghistani Giardia lamblia (Giardia duodenalis or Giardia intestinalis) is the causative agent of giardiasis and is the only common pathogenic protozoan found
CLIENT NOTICE UPDATE - August 24, *** NEW COLLECTION CONTAINTER for PARASITE BY PCR ***
 CLIENT NOTICE UPDATE - August 24, 2017 LifeLabs Introduces PCR Testing For Detection Of Parasites In Stool Samples On August 8 th, 2017, LifeLabs Medical Laboratories has started detection of parasites
CLIENT NOTICE UPDATE - August 24, 2017 LifeLabs Introduces PCR Testing For Detection Of Parasites In Stool Samples On August 8 th, 2017, LifeLabs Medical Laboratories has started detection of parasites
Parasites List of Pinworm (Enterobius vermicularis) Giardia ( Giardia lamblia Coccidia ( Cryptosporidium
 Parasites List of Pinworm (Enterobius vermicularis) Pinworm, also known as seatworm or threadworm, is one of the most prevalent intestinal parasites in the United States, with approximately 40 million
Parasites List of Pinworm (Enterobius vermicularis) Pinworm, also known as seatworm or threadworm, is one of the most prevalent intestinal parasites in the United States, with approximately 40 million
Study of Intestinal Protozoan Parasites in Rural Inhabitants of Mazandaran Province, Northern Iran
 Original Article Study of Intestinal Protozoan Parasites in Rural Inhabitants of Mazandaran Province, Northern Iran EB Kia 1, M Hosseini 2, MR Nilforoushan 1, AR Meamar 3, *M Rezaeian 1 1 Dept. of Medical
Original Article Study of Intestinal Protozoan Parasites in Rural Inhabitants of Mazandaran Province, Northern Iran EB Kia 1, M Hosseini 2, MR Nilforoushan 1, AR Meamar 3, *M Rezaeian 1 1 Dept. of Medical
Comparison of Lateral Flow Immunoassays to Current Stool Evaluation Methods for the Detection of Giardia and Cryptosporidium. Laurianne T.
 Comparison of Lateral Flow Immunoassays to Current Stool Evaluation Methods for the Detection of Giardia and Cryptosporidium Laurianne T. Mullinax A thesis submitted in partial fulfillment of the requirements
Comparison of Lateral Flow Immunoassays to Current Stool Evaluation Methods for the Detection of Giardia and Cryptosporidium Laurianne T. Mullinax A thesis submitted in partial fulfillment of the requirements
Parasite Organism Chart Parasite Description Habitat/Sources of Isolation Blastocystis hominis
 Blastocystis hominis B. hominis has recently been reclassified as a protozoan, of which there are thought to be four separate serologic groups. 1 This organism is transmitted via the fecal-oral route or
Blastocystis hominis B. hominis has recently been reclassified as a protozoan, of which there are thought to be four separate serologic groups. 1 This organism is transmitted via the fecal-oral route or
Amebiasis Ab E. histolytica IgG
 Code: HP003 96 Test distribuito da: Amebiasis Ab E. histolytica IgG Amebiasis E. histolytica lgg test is an enzyme immunoassay for the detection of specific lgg antibodies against Anti - Entamoeba histolytica
Code: HP003 96 Test distribuito da: Amebiasis Ab E. histolytica IgG Amebiasis E. histolytica lgg test is an enzyme immunoassay for the detection of specific lgg antibodies against Anti - Entamoeba histolytica
Taenia solium IgG ELISA Kit
 Taenia solium IgG ELISA Kit Catalog Number KA3191 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Taenia solium IgG ELISA Kit Catalog Number KA3191 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
RIDA QUICK Cryptosporidium/Giardia/Entamoeba Combi
 RIDA QUICK Cryptosporidium/Giardia/Entamoeba Combi Article no: N1723 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1.
RIDA QUICK Cryptosporidium/Giardia/Entamoeba Combi Article no: N1723 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1.
The Changing Landscape of Stool Parasite Diagnosis and Surveillance
 The Changing Landscape of Stool Parasite Diagnosis and Surveillance Ann Valley- Advanced Microbiologist Tim Monson- Microbiologist Supervisor Communicable Disease Division Wisconsin State Laboratory of
The Changing Landscape of Stool Parasite Diagnosis and Surveillance Ann Valley- Advanced Microbiologist Tim Monson- Microbiologist Supervisor Communicable Disease Division Wisconsin State Laboratory of
The Changing Landscape of Stool Parasite Diagnosis and Surveillance
 The Changing Landscape of Stool Parasite Diagnosis and Surveillance Ann Valley- Advanced Microbiologist Tim Monson- Microbiologist Supervisor Communicable Disease Division Wisconsin State Laboratory of
The Changing Landscape of Stool Parasite Diagnosis and Surveillance Ann Valley- Advanced Microbiologist Tim Monson- Microbiologist Supervisor Communicable Disease Division Wisconsin State Laboratory of
Please use only the valid version of the package insert provided with the kit.
 Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Trichinella using an Enzyme Linked Immunoabsorbant Assay
Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Trichinella using an Enzyme Linked Immunoabsorbant Assay
Rapid-VIDITEST. Rota-Adeno Blister. One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual
 Rapid-VIDITEST Rota-Adeno Blister One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual INTENDED USE: The Rapid-VIDITEST Rota-Adeno Blister test is a rapid chromatographic immunoassay
Rapid-VIDITEST Rota-Adeno Blister One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual INTENDED USE: The Rapid-VIDITEST Rota-Adeno Blister test is a rapid chromatographic immunoassay
Interpretation At-a-Glance Bacteriology. Parasitology
 Patient: EMILY TET DOB: December 31, 1980 ex: F MN: 0001558266 Order Number: M9301014 Completed: January 31, 2019 eceived: January 30, 2019 Collected: January 30, 2019 Test Office Test POD Test MD, DO,
Patient: EMILY TET DOB: December 31, 1980 ex: F MN: 0001558266 Order Number: M9301014 Completed: January 31, 2019 eceived: January 30, 2019 Collected: January 30, 2019 Test Office Test POD Test MD, DO,
Non_ pathogenic Amoeba of humans:
 The parasite protozoa Phylum: Sarcomastigophora Sarcodina االميبات Amoebae Non_ pathogenic Amoeba of humans: 1. Entamoeba hartmanii, contain trophozoite and cyst 2. Entamoeba coli, cyst and trophozoite
The parasite protozoa Phylum: Sarcomastigophora Sarcodina االميبات Amoebae Non_ pathogenic Amoeba of humans: 1. Entamoeba hartmanii, contain trophozoite and cyst 2. Entamoeba coli, cyst and trophozoite
Rapid-VIDITEST Enterovirus
 Rapid-VIDITEST Enterovirus A rapid one step Enterovirus Card test for the qualitative detection of Enterovirus antigens in human feces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
Rapid-VIDITEST Enterovirus A rapid one step Enterovirus Card test for the qualitative detection of Enterovirus antigens in human feces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
IVD. DRG Toxocara canis ELISA (EIA-3518) Revised 12 Feb rm (Vers. 6.1)
 Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Toxocara using an Enzyme-Linked Immunosorbent Assay
Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Toxocara using an Enzyme-Linked Immunosorbent Assay
Toxoplasma gondii IgM ELISA Kit
 Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Rotavirus Test Kit. Instructions For Use. Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA
 Rotavirus Test Kit Instructions For Use Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA * Please read the instructions carefully before use INTENDED USE Velotest Rotavirus Test is
Rotavirus Test Kit Instructions For Use Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA * Please read the instructions carefully before use INTENDED USE Velotest Rotavirus Test is
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia)
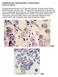 PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
Trichinella Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
NEW YORK STATE Parasitology Proficiency Testing Program. Parasitology (General) 01 February Sample Preparation and Quality Control
 NEW YORK STATE Parasitology Proficiency Testing Program Parasitology (General) 01 February 2011 The purpose of the New York State Proficiency Testing Program in the category of Parasitology (General) is
NEW YORK STATE Parasitology Proficiency Testing Program Parasitology (General) 01 February 2011 The purpose of the New York State Proficiency Testing Program in the category of Parasitology (General) is
Giardiasis. Table of Contents
 Table of Contents Case Definition... Error! Bookmark not defined. Reporting Requirements... 2 Etiology... Error! Bookmark not defined. Clinical Presentation... Error! Bookmark not defined. Diagnosis...
Table of Contents Case Definition... Error! Bookmark not defined. Reporting Requirements... 2 Etiology... Error! Bookmark not defined. Clinical Presentation... Error! Bookmark not defined. Diagnosis...
Human HIV (1+2) antigen&antibody ELISA Kit
 Human HIV (1+2) antigen&antibody ELISA Kit Catalog Number. CSB-E18042h For the qualitative determination of human HIV (1+2) antibody and P24 antigen concentrations in serum, plasma. This package insert
Human HIV (1+2) antigen&antibody ELISA Kit Catalog Number. CSB-E18042h For the qualitative determination of human HIV (1+2) antibody and P24 antigen concentrations in serum, plasma. This package insert
HBeAg and HBeAg Ab ELISA Kit
 HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 17 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3
HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 17 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3
Practice Guidelines for Ordering Stool Ova and Parasite Testing in a Pediatric Population
 CLINICAL MICROBIOLOGY AND INFECTIOUS DISEASE Original Article Practice Guidelines for Ordering Stool Ova and Parasite Testing in a Pediatric Population AMIN KABANI, MD, FRCPC, GISELE CADRAIN, RT, CYNTHIA
CLINICAL MICROBIOLOGY AND INFECTIOUS DISEASE Original Article Practice Guidelines for Ordering Stool Ova and Parasite Testing in a Pediatric Population AMIN KABANI, MD, FRCPC, GISELE CADRAIN, RT, CYNTHIA
Comparison of Three Collection-Preservation Methods for
 JOURNAL OF CLINICAL MICROBIOLOGY, Dec. 1981, p. 656-660 0095-1 137/81/120656-05$02.00/0 Vol. 14, No. 6 Comparison of Three Collection-Preservation Methods for Detection of Intestinal Parasites DONALD L.
JOURNAL OF CLINICAL MICROBIOLOGY, Dec. 1981, p. 656-660 0095-1 137/81/120656-05$02.00/0 Vol. 14, No. 6 Comparison of Three Collection-Preservation Methods for Detection of Intestinal Parasites DONALD L.
Apolipoprotein A-1 ELISA
 Apolipoprotein A-1 ELISA For the quantitative determination of apolipoprotein A1 in serum and plasma. For Research Use Only. Not For Use In Diagnostic Procedures. Please read carefully due to Critical
Apolipoprotein A-1 ELISA For the quantitative determination of apolipoprotein A1 in serum and plasma. For Research Use Only. Not For Use In Diagnostic Procedures. Please read carefully due to Critical
RIDA QUICK Entamoeba. Article no: N 1703
 RIDA QUICK Entamoeba Article no: N 1703 R-Biopharm AG, Landwehrstr. 54, D-64293 Darmstadt, Germany Tel: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic use.
RIDA QUICK Entamoeba Article no: N 1703 R-Biopharm AG, Landwehrstr. 54, D-64293 Darmstadt, Germany Tel: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic use.
Toxoplasma gondii IgM ELISA Kit
 Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Hepatitis A virus IgM ELISA Kit
 Hepatitis A virus IgM ELISA Kit Catalog Number KA0285 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Hepatitis A virus IgM ELISA Kit Catalog Number KA0285 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Rapid-VIDITEST Swine Flu
 Rapid-VIDITEST Swine Flu One Step Influenza type A Antigen Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
Rapid-VIDITEST Swine Flu One Step Influenza type A Antigen Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
Serum Amyloid A ELISA
 Serum Amyloid A ELISA For the quantitative determination of serum amyloid A (SAA) in serum plasma For Research use Only. Not for Use in Diagnostic Procedures Please see Appendix A for Reference Serum information
Serum Amyloid A ELISA For the quantitative determination of serum amyloid A (SAA) in serum plasma For Research use Only. Not for Use in Diagnostic Procedures Please see Appendix A for Reference Serum information
Toxoplasma gondii IgM (Toxo IgM)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Washington State Department of Health (DOH) Cryptosporidium in Drinking Water Position Paper. Purpose
 Washington State Department of Health (DOH) Cryptosporidium in Drinking Water Position Paper Purpose Cryptosporidium is a micro-organism which has gained increased public health significance as a result
Washington State Department of Health (DOH) Cryptosporidium in Drinking Water Position Paper Purpose Cryptosporidium is a micro-organism which has gained increased public health significance as a result
The Prevalence of Intestinal Parasitic Infections in a Tertiary Care Hospital in Southern India - A Retrospective Study
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 5 Number 10 (2016) pp. 718-723 Journal homepage: http://www.ijcmas.com Original Research Article http://dx.doi.org/10.20546/ijcmas.2016.510.078
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 5 Number 10 (2016) pp. 718-723 Journal homepage: http://www.ijcmas.com Original Research Article http://dx.doi.org/10.20546/ijcmas.2016.510.078
Human Influenza A (Swine Flu) Rapid test
 Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Toxocara. Cat # Toxocara ELISA. Enzyme Linked Immunosorbent Assay. ELISA-Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Rapid-VIDITEST. Helicobacter pylori. One step Helicobacter pylori Blister test. Instruction manual
 Rapid-VIDITEST Helicobacter pylori One step Helicobacter pylori Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565,
Rapid-VIDITEST Helicobacter pylori One step Helicobacter pylori Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565,
RIDA QUICK Entamoeba. Article no: N1702
 RIDA QUICK Entamoeba Article no: N1702 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic
RIDA QUICK Entamoeba Article no: N1702 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic
HIV-1 p24 ELISA Kit. Catalog Number KA assays Version: 06. Intended for research use only.
 HIV-1 p24 ELISA Kit Catalog Number KA0452 96 assays Version: 06 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Principle of the Assay... 3 General Information... 4 Materials
HIV-1 p24 ELISA Kit Catalog Number KA0452 96 assays Version: 06 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Principle of the Assay... 3 General Information... 4 Materials
Aac Reagent Set ELISA for the detection of Acidovorax avenae subsp. citrulli Catalog number: SRA 14800
 List of contents Lot number Aac Reagent Set Item 96 wells 500 wells 1000 wells 5000 wells Capture antibody 0.150 ml 0.275 ml 0.525 ml 2.525 ml Alkaline phosphatase enzyme conjugate 0.150 ml 0.275 ml 0.525
List of contents Lot number Aac Reagent Set Item 96 wells 500 wells 1000 wells 5000 wells Capture antibody 0.150 ml 0.275 ml 0.525 ml 2.525 ml Alkaline phosphatase enzyme conjugate 0.150 ml 0.275 ml 0.525
Rapid-VIDITEST Calprotectin
 Rapid-VIDITEST Calprotectin One Step Calprotectin Card Test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz INTENDED
Rapid-VIDITEST Calprotectin One Step Calprotectin Card Test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz INTENDED
Rapid-VIDITEST FOB Blister
 Rapid-VIDITEST FOB Blister One Step Fecal Occult Blood Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420 261 090 565,
Rapid-VIDITEST FOB Blister One Step Fecal Occult Blood Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420 261 090 565,
Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister
 Li StarFish S.r.l. Via Cavour, 35-20063 Cernusco S/N (MI), Italy Tel. +39-02-92150794 - Fax. +39-02-92157285 info@listarfish.it -www.listarfish.it Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister One
Li StarFish S.r.l. Via Cavour, 35-20063 Cernusco S/N (MI), Italy Tel. +39-02-92150794 - Fax. +39-02-92157285 info@listarfish.it -www.listarfish.it Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister One
Outline EP1201. NEHA 2012 AEC June 2012
 Food and Water Borne Enteric Protozoa: Environmental Health Perspectives Stephanie M. Fletcher, PhD (c) NEHA AEC & Exhibition San Diego June 30, 2012 Outline Introduction Overview of the Epidemiology of
Food and Water Borne Enteric Protozoa: Environmental Health Perspectives Stephanie M. Fletcher, PhD (c) NEHA AEC & Exhibition San Diego June 30, 2012 Outline Introduction Overview of the Epidemiology of
PARASITE CONCENTRATOR FOR THE LABORATORY DIAGNOSIS OF INTESTINAL PARASITISM
 EVALUATION OF MINI PARASEP SF FAECAL PARASITE CONCENTRATOR FOR THE LABORATORY DIAGNOSIS OF INTESTINAL PARASITISM 1 IKEH, E.I. AND 2 ELUJOLA, M. 1 Department of Medical Microbiology/Medical Laboratory Science
EVALUATION OF MINI PARASEP SF FAECAL PARASITE CONCENTRATOR FOR THE LABORATORY DIAGNOSIS OF INTESTINAL PARASITISM 1 IKEH, E.I. AND 2 ELUJOLA, M. 1 Department of Medical Microbiology/Medical Laboratory Science
Indian Journal of Basic and Applied Medical Research; September 2014: Vol.-3, Issue- 4, P
 Original article: Comparison of stool concentration methods for detection of prevalence of enteroparasitic infection in rural tertiary care teaching hospital of Maharashtra 1 Dr. Jaishree Puri, 2 Dr. S.
Original article: Comparison of stool concentration methods for detection of prevalence of enteroparasitic infection in rural tertiary care teaching hospital of Maharashtra 1 Dr. Jaishree Puri, 2 Dr. S.
HAV IgG/IgM Rapid Test
 HAV IgG/IgM Rapid Test Cat. No.:DTS649 Pkg.Size: Intended use CD HAV IgG/IgM Rapid Test is a solid phase immunochromatographic assay for the rapid, qualitative and differential detection of IgG and IgM
HAV IgG/IgM Rapid Test Cat. No.:DTS649 Pkg.Size: Intended use CD HAV IgG/IgM Rapid Test is a solid phase immunochromatographic assay for the rapid, qualitative and differential detection of IgG and IgM
HIV and Parasitic Infection and the Effect of Treatment among Adult Outpatients in Malawi
 BRIEF REPORT HIV and Parasitic Infection and the Effect of Treatment among Adult Outpatients in Malawi Mina C. Hosseinipour, 1,2 Sonia Napravnik, 2 George Joaki, 1 Syze Gama, 1 Nyanyiwe Mbeye, 1 Blessings
BRIEF REPORT HIV and Parasitic Infection and the Effect of Treatment among Adult Outpatients in Malawi Mina C. Hosseinipour, 1,2 Sonia Napravnik, 2 George Joaki, 1 Syze Gama, 1 Nyanyiwe Mbeye, 1 Blessings
Egg Protein Residue ELISA Kit
 Egg Protein Residue ELISA Kit Catalog Number KA3208 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Egg Protein Residue ELISA Kit Catalog Number KA3208 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
1. Parasitology Protozoa 4
 Contents 1. Parasitology 1 Host Parasite Relationship 2 Mode of Transmission 2 2. Protozoa 4 Classification 4 Reproduction 5 Immunity 5 Pathogenesis 6 Laboratory Diagnosis 6 Treatment 8 Entamoeba 8 Classification
Contents 1. Parasitology 1 Host Parasite Relationship 2 Mode of Transmission 2 2. Protozoa 4 Classification 4 Reproduction 5 Immunity 5 Pathogenesis 6 Laboratory Diagnosis 6 Treatment 8 Entamoeba 8 Classification
Rapid-VIDITEST. Rota-Adeno. One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual
 Rapid-VIDITEST Rota-Adeno One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420
Rapid-VIDITEST Rota-Adeno One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420
Physician Use of Parasite Tests in the United States from 1997 to 2006 and in a Utah Cryptosporidium Outbreak in 2007
 JOURNAL OF CLINICAL MICROBIOLOGY, Feb. 2011, p. 591 596 Vol. 49, No. 2 0095-1137/11/$12.00 doi:10.1128/jcm.01806-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. Physician Use
JOURNAL OF CLINICAL MICROBIOLOGY, Feb. 2011, p. 591 596 Vol. 49, No. 2 0095-1137/11/$12.00 doi:10.1128/jcm.01806-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. Physician Use
Bacillary Dysentery (Shigellosis)
 Bacillary Dysentery (Shigellosis) An acute bacterial disease involving the large and distal small intestine, caused by the bacteria of the genus shigella. Infectious agent Shigella is comprised of four
Bacillary Dysentery (Shigellosis) An acute bacterial disease involving the large and distal small intestine, caused by the bacteria of the genus shigella. Infectious agent Shigella is comprised of four
Prevalence of Intestinal Parasites in HIV Seropositive Patients with and without Diarrhoea and its Correlation with CD4 Counts
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 5 Number 10 (2016) pp. 527-532 Journal homepage: http://www.ijcmas.com Original Research Article http://dx.doi.org/10.20546/ijcmas.2016.510.058
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 5 Number 10 (2016) pp. 527-532 Journal homepage: http://www.ijcmas.com Original Research Article http://dx.doi.org/10.20546/ijcmas.2016.510.058
Hosta virus X ELISA KIT
 Hosta virus X ELISA KIT Cat. No.:DEIA9256 Pkg.Size:96T Intended use The Hosta virus X ELISA KIT is used to detect HVX in both leaves and roots of hosta plants. Reagents And Materials Provided 1. Capture
Hosta virus X ELISA KIT Cat. No.:DEIA9256 Pkg.Size:96T Intended use The Hosta virus X ELISA KIT is used to detect HVX in both leaves and roots of hosta plants. Reagents And Materials Provided 1. Capture
Classification - Protozoa. Parasitology Intestinal Amoeba. Stools for O&P Examination. Stool Specimen. What to look for.
 Classification - Protozoa Parasitology Intestinal Amoeba Phylum Phylum Sarcomastigophora Sarcomastigophora Subkingdom Subkingdom Protozoa Protozoa Phylum Phylum Ciliophora Ciliophora Phylum Phylum Apicomplexa
Classification - Protozoa Parasitology Intestinal Amoeba Phylum Phylum Sarcomastigophora Sarcomastigophora Subkingdom Subkingdom Protozoa Protozoa Phylum Phylum Ciliophora Ciliophora Phylum Phylum Apicomplexa
Canine Thyroid Stimulating Hormone, TSH ELISA Kit
 Canine Thyroid Stimulating Hormone, TSH ELISA Kit Catalog No: E0463c 96 Tests Operating instruction www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH
Canine Thyroid Stimulating Hormone, TSH ELISA Kit Catalog No: E0463c 96 Tests Operating instruction www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH
Prevalence of Intestinal Parasitic Infections in HIV-Positive Patients
 ISSN: 2319-7706 Volume 4 Number 5 (2015) pp. 269-273 http://www.ijcmas.com Original Research Article Prevalence of Intestinal Parasitic Infections in HIV-Positive Patients Vasundhara*, Haris M.Khan, Harekrishna
ISSN: 2319-7706 Volume 4 Number 5 (2015) pp. 269-273 http://www.ijcmas.com Original Research Article Prevalence of Intestinal Parasitic Infections in HIV-Positive Patients Vasundhara*, Haris M.Khan, Harekrishna
SIV p27 Antigen ELISA Catalog Number:
 INTENDED USE The RETRO-TEK SIV p27 Antigen ELISA is for research use only and is not intended for in vitro diagnostic use. The RETRO-TEK SIV p27 Antigen ELISA is an enzyme linked immunoassay used to detect
INTENDED USE The RETRO-TEK SIV p27 Antigen ELISA is for research use only and is not intended for in vitro diagnostic use. The RETRO-TEK SIV p27 Antigen ELISA is an enzyme linked immunoassay used to detect
Mycoplasma pneumoniae IgG ELISA Kit
 Mycoplasma pneumoniae IgG ELISA Kit Catalog Number KA2260 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Mycoplasma pneumoniae IgG ELISA Kit Catalog Number KA2260 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Dr. Jabar Etaby Lecture GIARDIASIS(lambliasis) Etiology: Giardia lamblia (flagellate)
 Dr. Jabar Etaby Lecture Two GIARDIASIS(lambliasis) Etiology: Giardia lamblia (flagellate) Epidemiology: It has worldwide distribution and is not uncommon in South Carolina. It is the most frequent protozoan
Dr. Jabar Etaby Lecture Two GIARDIASIS(lambliasis) Etiology: Giardia lamblia (flagellate) Epidemiology: It has worldwide distribution and is not uncommon in South Carolina. It is the most frequent protozoan
MALARIA P. FALCIPARUM / P. VIVAX
 MALARIA P. FALCIPARUM / P. VIVAX 1. EXPLANATION OF THE TEST: Malaria is a serious, sometimes fatal, parasitic disease characterized by fever, chills, and anemia and is caused by a parasite that is transmitted
MALARIA P. FALCIPARUM / P. VIVAX 1. EXPLANATION OF THE TEST: Malaria is a serious, sometimes fatal, parasitic disease characterized by fever, chills, and anemia and is caused by a parasite that is transmitted
IgG Antibodies To Toxoplasma Gondii ELISA Kit Protocol
 IgG Antibodies To Toxoplasma Gondii ELISA Kit Protocol (Cat. No.:EK-310-85) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED
IgG Antibodies To Toxoplasma Gondii ELISA Kit Protocol (Cat. No.:EK-310-85) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED
Chorionic Gonadotropin Human ELISA Kit
 ab108637 Chorionic Gonadotropin Human ELISA Kit Instructions for Use For the qualitative measurement of Human Chorionic Gonadotropin concentrations in serum and urine. This product is for research use
ab108637 Chorionic Gonadotropin Human ELISA Kit Instructions for Use For the qualitative measurement of Human Chorionic Gonadotropin concentrations in serum and urine. This product is for research use
DRG International, Inc., USA Fax: (908)
 Please use only the valid version of the package insert provided with the kit. 1 INTENDED USE The human Annexin V ELISA is an enzyme-linked immunosorbent assay for determination of human Annexin V. The
Please use only the valid version of the package insert provided with the kit. 1 INTENDED USE The human Annexin V ELISA is an enzyme-linked immunosorbent assay for determination of human Annexin V. The
HIV-1 p24 ELISA Kit. Cat.No: DEIA10155 Lot. No. (See product label) Size. Storage. Principle Of The Test. Reagents And Materials Provided
 HIV-1 p24 ELISA Kit Cat.No: DEIA10155 Lot. No. (See product label) Size 96T Storage All reagents should be stored at 2-8 C, and should not be used beyond the expiration date on the label. Once opened,
HIV-1 p24 ELISA Kit Cat.No: DEIA10155 Lot. No. (See product label) Size 96T Storage All reagents should be stored at 2-8 C, and should not be used beyond the expiration date on the label. Once opened,
Malaria Pf/pan antigen Rapid Test
 Malaria Pf/pan antigen Rapid Test Cat. No.:IVDTS003 Pkg.Size:10T/50T Intended use The Malaria Pf/pan antigen Rapid Test is a self-performing, qualitative, sandwich immunoassay, utilizing whole blood for
Malaria Pf/pan antigen Rapid Test Cat. No.:IVDTS003 Pkg.Size:10T/50T Intended use The Malaria Pf/pan antigen Rapid Test is a self-performing, qualitative, sandwich immunoassay, utilizing whole blood for
CHEMILUMINESCENCE ENZYME IMMUNOASSAY (CLIA) Toxoplasma IgG. Cat # (20-25 C Room temp.) Volume
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
INSTRUCTION MANUAL FOR HBsAg SUBTYPE EIA
 INSTRUCTION MANUAL FOR HBsAg SUBTYPE EIA EIA based hepatitis B surface antigen subtyping kit with monoclonal antibodies I. Kit configuration 1. Microplate coated with anti-hbs a (96 wells)... 1 plate 2.
INSTRUCTION MANUAL FOR HBsAg SUBTYPE EIA EIA based hepatitis B surface antigen subtyping kit with monoclonal antibodies I. Kit configuration 1. Microplate coated with anti-hbs a (96 wells)... 1 plate 2.
