distension of the stomach wall.
|
|
|
- Sheena Franklin
- 5 years ago
- Views:
Transcription
1 J. Phy8ii. (1967), 19, pp With 1 text-figure Printed in Great Britain ACCELERATED MOBILIZATION AND FORMATION OF HISTAMINE IN THE GASTRIC MUCOSA EVOKED BY VAGAL EXCITATION BY G. KAHLSON, ELSA ROSENGREN AND R. THUNBERG From the Institute of Physiology, University of Lund, Sweden (Received 28 June 1966) SUMMARY 1. The changes in the rate of histamine formation and in the histamine content of the parietal cell containing region of the gastric mucosa have been studied in rats under the influence of agents which evoke or abolish vagal excitation. 2. The hypoglycaemia producing agents, insulin and 2-deoxyglucose (2-DG), raised the mucosal histamine-forming capacity (HFC) in a way similar to that previously observed on re-feeding, gastrin injection, and distension of the stomach wall. 3. In cats, insulin injection elicited an elevation of mucosal HFC similar to the corresponding effect of insulin in rats. 4. Hoechst 998, which inhibits post-ganglionic cholinergic transmission, counteracted the elevation of mucosal HFC following vagal excitation, but did not inhibit changes produced by gastrin, thus indicating the absence of a cholinergic intermediary link between gastrin and changes in mucosal histamine. 5. It is emphasized that although re-feeding, vagus excitation, gastrin and distension all produce similar changes in mucosal histamine, the clarification of the precise role of histamine as a natural stimulant for the parietal cells may require a fresh kind of approach. INTRODUCTION Re-feeding of fasted rats, mice and frogs evokes a mobilization of histamine and a concurrent acceleration of the rate of histamine formation in the parietal cell containing region of the gastric mucosa. The changes in mucosal histamine pertaining to feeding can be reproduced by injecting a purified gastrin preparation. These changes in mucosal histamine are not merely a product of secretory activity since they do not occur when acid secretion is excited by injection of histamine. Nor do they occur in the 29-2
2 456 G. KAHLSON, ELSA ROSENGREN AND R. THUNBERG non-parietal cell containing region in the rat, the only species so far studied in this particular respect (Kahlson, Rosengren, Svahn & Thunberg, 1964). In the course of the above-mentioned work a feed-back relation between mucosal content of histamine and level of histidine decarboxylase was disclosed whereby lessening of end-product repression (histamine release) resulted in increased enzyme activity and, conversely, increasing the concentration of the end-product by injecting histamine depressed the level of enzyme activity. The nature of the enzyme histidine decarboxylase responsible for mucosal HFC has been studied in its essential features by Kahlson, Rosengren & Thunberg (1963). These authors found that the enzyme responsible for histamine formation in vivo in the species studied is strongly inhibited by a-methylhistidine, and weakly inhibited by ac-methyldopa, and they assessed quantitatively in vitro and in vivo the significance of pyridoxal phosphate as a co-factor. On re-feeding, three distinct, but interrelated, excitatory processes come into operation, vagal excitation, gastrin release, and distension of the stomach. Having investigated the changes in mucosal histamine evoked by gastrin and distension it appeared essential to study changes which might result from vagal excitation. Because of observations on acid secretion suggesting that histamine acts through nerves (Clark, Curnow, Murray, Stephens & Wyllie, 1964), and further, that gastrin stimulates secretion by acting on nerves to release acetylcholine (Bennett, 1965) the effect of a compound blocking cholinergic transmission was also studied. METHODS Animals. Female rats of the Sprague-Dawley strain, weighing g, were used throughout. Young cats of two different litters, weighing 55-8 g, and g, respectively, were employed. Food was withheld for 15 hr in all animals before injections. Drugs. In a first series of experiments in rats the two vagus nerves to the stomach were dissected below the diaphragm and stimulated electrically. Motor responses were obtained but acid secretion was scanty. This mode of vagal excitation was abandoned and replaced by injection of either insulin, 5 i.u./kg, or 2-DG, 1 mg/kg (Hirschowitz & Sachs, 1965). Post-ganglionic cholinergic influences were antagonized by the compound Hoechst 998 (piperidino-ethyl-diphenyl-acetamide),.1 mg/kg (for references see Emmelin & Henriksson, 1953). Purified gastrin 11 was obtained from Professor Gregory, Liverpool, and injected in a dose of 1,ug/kg. Controls received NaCl solution (-9 g/1 ml.), 1 ml./kg. All injections were given subcutaneously. The animals were killed by a blow on the head and bled by the carotid arteries 3j hr after injection of insulin or 2-DG, and 1 hr after gastrin injection; Hoechst 998 was given 4 hr before killing the animal. Tissue preparation. The gastric mucosa and submucosa of the parietal cell containing region were removed by scraping with a scalpel after the stomach had been opened along the minor curvature, washed in ice-cold NaCl solution and pinned flat. The tissue was minced with scissors and examined for histamine content and histidine decarboxylase activity.
3 VAGUS AND GASTRIC MUCOSAL HISTAMINE 457 Determination of histamine content was carried out by a modification of the origina. fluorometriemethod of Shore, Burkhalter& Cohn(1959). Briefly, themodification, as employed by Kahlson et al. (1963), involves isolation of histamine by ion exchange chromatography on Dowex 5 instead of extraction into butanol. A similar procedure has subsequently been used in fluorometric determination of histamine by Green & Erickson (1964). The advantage of the procedure is to improve specificity. It should be mentioned here that the fluorometric method, as employed in this laboratory, is also suitable for determination of free histamine in urine (R. Thunberg, to be published). In thirty-nine instances parallel determinations were made on the same samples with the fluorometric method and the conventional extraction procedure followed by biological assay on the guinea-pig's gut. In some instances additional determinations were carried out by a method involving reaction with dinitrofluorobenzene and thinlayer chromatography as devised by White (1966). Good agreement was found between the three methods as will be recorded in a separate report. Determination of HFC in vitro. The isotopic method originally elaborated by Schayer, and modified in this laboratory, was employed (Kahlson et al. 1963). This involved incubation of tissue samples with [14C]histidine and measurement of the [14C]histamine formed. In this method the concentration of [14C]histidine present in the incubation mixture is very low. Consequently the enzyme with a high affinity for this substrate is measured, whereas other enzymes with low affinity for histidine, i.e. DOPA-decarboxylase, and similar enzymes referred to as non-specific histidine decarboxylases, do not significantly contribute to the formation of ['4C]histamine. The enzyme measured should thus be designated simply as histidine decarboxylase. As already pointed out different amino acid decarboxylating enzymes can be differentiated by the use of inhibitors such as a-methylhistidine and a-methyldopa (Kahlson et al. 1963). RESULTS The changes in levels of mucosal HFC and histamine content were investigated in rats under the influence of vagal excitation produced by insulin or 2-DG. The cholinergic blocking compound Hoechst 998 was employed in order to antagonize changes thus produced. This compound was also used in experiments with the view to see whether a cholinergic mechanism is essential to the pertinent changes produced by gastrin. The results with these various influences and compounds are illustrated in Fig. 1 and detailed in Table 1. In the figure each column depicts the mean value and standard deviation of determinations of HFC and histamine content in five rats. Insulin. The HFC becomes elevated and the histamine content falls after injection of insulin. The change in HFC is largely reduced by pretreatment with Hoechst 998. The connexion between corresponding alterations in histamine content and HFC will presently be discussed in relation to observations on cats. 2-deoxyglucose (2-DG). This compound, alleged to excite gastric secretion via the vagus nerves (Hirschowitz & Sachs, 1965), likewise induced a substantial elevation of mucosal HFC and a fall in histamine content of low significance (P about -5). It was not investigated whether larger doses of
4 458 G. KAHLSON, ELSA ROSENGREN AND R. THUNBERG this compound would produce greater changes. The elevation of HFC evoked by 2-DG was abolished by Hoechst 998. Gastrin. The elevation of HFC and the fall in histamine content observed agree with previous results (Kahlson et al. 1964). These changes are not Control Insulin 2-DG Gastrin 4 3 Fig. 1. HFC (pg/g.3 hr) and histamine content (pug/g) of rat gastric mucosa, in controls and under the influence of insulin, 2-DO and gastrin. The open and the hatched colulmns stand for values obtained in animals injected with NaCl solution and Hoechst 998, respectively, before the injection of NaCl solution (control), insulin, 2-DOS or gastrin. Each column depicts the mean value and standard deviation of determinations in five rats. abolished by treatment with Hoechst 998. Rather, the increase in HFO is even greater. No attempts were made to explain this seemingly paradoxical result. Hoechst 998. This drug was employed in place of atropine because it was found that atropine by itself elevated mucosal HFC, a disturbing phenomenon possibly related to its character as a histamine releaser. In search for a substitute, Hoechst 998 was chosen, in doses sufficient to antagonize the elevation of HFC by insulin, because it did not by itself significantly alter the level of HFO, as shown in Table 1 and in the extreme left section of Fig. 1. Observations in cats. Eight young cats from two litters were examined for mucosal HFC: and histamine content. Each litter was divided into two groups, the one serving as controls injected with NaOl solution, the other injected with insulin. Results listed in Table 2 show that in cats insulin produces changes of HFC which are similar to those seen in rats. The prerequisites for obtaining maxrimum elevation of mucosal HFC under
5 VAGUS AND GASTRIC MUCOSAL HISTAMINE 459 o r o c+1. '4 to n- CP r~ o _ + o ~ o - X 9I +1 i o -4 n 14 bo, bo. CO to ;.I * ec i OWX O CN C= N C 4,4 CQ co c)es ta_ o csr C 1 'alo t - o_ct ex o~co too..c~ C CO - CO CO +._ 4 t- +1 NṾ1 COD -v.. -_ UCOtD OQ 1 V co 4Q +1.. CO +1; o~_~ C o Q _4 CO 1 _ 4 _'H4^4^ 1 E LlO COtI -4 U. 1 Oo OC I*
6 46 G. KAHLSON, ELSA ROSENGREN AND R. THUNBERG the influence of insulin were not explored. A cat of litter 1 (figures in brackets in Table 2) intended to serve as a control, had escaped from the cage during the period of fasting. This cat did not have access to food but the antral part of the stomach was found stuffed with wooden shavings TABLE 2. HFC (ng/g. 3 hr) and histamine content (,glg) of gastric mucosa in eight young cats injected with NaCl solution (controls) and insulin, respectively. Figures in brackets refer to a cat with distended stomach HFC Content Controls Insulin Controls Insulin Litter 1 (86-93 g) Litter 2 (55-8 g) 38, (8) 44, 7 82, 92 82, 86 32, (23) 26, 27 23, 18 31, 2 and contained a highly acid juice, this in contrast to the other controls in which the stomach was seemingly devoid of juice, and the surface of the mucosa covered with mucus of about neutral reaction. In this cat the mucosal HFC was 8 ng/g. 3 hr. This finding is in agreement with previous observations in rats on the effect of distension of the stomach (Kahlson et al. 1964). With regard to alterations in histamine content it should be borne in mind that in cats the gastric mucosal histamine content in the normal state varies within a wide range among individuals, 5-39,ug/g in sixteen kittens (Haeger, Kahlson & Westling, 1953), 8-4 /tg/g in fifteen adult cats (Haeger & Kahlson, 1952). The number of experiments in Table 2 is too small for any conclusion to be drawn with respect to changes in histamine content. Further, it should be appreciated that a minor fall in content, which functionally might be significant, may not be detectable by the methods employed. Notwithstanding this, an initial but undetectable fall in histamine content might induce an elevation of HFC resulting in rapid replenishment of histamine content. Consequently, an occasional absence of a detectable fall in histamine content in the presence of an elevated HFC should not be taken as evidence against the operation of a feed-back coupling between tissue content of histamine and HFC. It should be noted (see Discussion) that insulin injection, i.e. vagus stimulation, is followed by an elevation of mucosal HFC. DISCUSSION The gastric mucosa of all species studied in this laboratory, man, dog, cat, rat, guinea-pig, hamster, mouse and frog, is rich in histidine decarboxylase. In the rat, mouse and cat mucosal HFC is much higher than in other tissues, exceeded only by the high levels of histidine decarboxylase discovered in embryonic tissues (for references see Rosengren, 1963, 1966). Extirpation of the whole stomach in the female rat is followed by a fall in
7 VAGUS AND GASTRIC MUCOSAL HISTAMINE 461 the urinary excretion of free histamine to considerably less than half of normal (High, Shepherd & Woodcock, 1965); a similar observation was also made in this laboratory (E. Rosengren, unpublished). These findings imply that presumably more than half of the whole-body histamine formation takes place in the stomach wall. Even in the fasting state the glandular mucosa produces histamine at high rates, hence the amine is continuously discharged into the blood stream, and partly excreted. Yet, interdigestive acid secretion is reported to be absent in some species, for example dog and man, and scanty in the rat and cat (Lane, Ivy & Ivy, 1957). A logical conclusion would be that histamine set free from the mucosa is a poor, or ineffective, stimulant of acid secretion. The disappearance of histamine from the gastric mucosa is accelerated by feeding, vagal excitation and injection of gastrin. Smith (1954, 196) reported that a crude gastrin preparation released histamine from the stomach wall and also from other tissues. The mucosal histamine, in particular, has been found susceptible to release by these means. Schayer & Ivy (1957, 1958) discovered that [14C]histamine, laid down in the gastric mucosa of rats following injection of [14C]histidine, was released on feeding, and appeared in the urine. Similarly, vagal excitation and injection of purified gastrin induce release of mucosal histamine (Kim & Shore, 1963; Haverback, Tecimer, Dyce, Cohen, Stubrin & Santa Ana, 1964; Kahlson et al. 1964). The observation that the mucosal histamine content falls under the influence of factors producing excitation of hydrochloric acid secretion has been taken by some authors to support the view originally expressed by Babkin (1938) that histamine constitutes the final link in the chain of events which results in excitation of the parietal cells to secrete (for references see Gregory, 1962; Code, 1965; Haverback, Stubrin & Dyce, 1965; Shore, 1965). Other workers in this field may feel that the mere observation that histamine disappears from the mucosa, whilst secretion subsequent to a meal proceeds for several hours, would be difficult to reconcile with the view as postulated. A different picture presents itself in face of the demonstration that feeding, as well as the principal excitatory components operating subsequent to feeding, i.e. vagal excitation, gastrin and distension of the stomach wall, induce an accelerated rate of histamine formation which persists for several hours. The elevation of histidine decarboxylase is presumed to be brought about by an initial lowering of the mucosal content of preformed histamine, the levels of the enzyme and the end-product being interrelated by the feed-back coupling already referred to. The newly formed histamine moves freely through the stomach wall, as evidenced by its appearance in the urine (Kahlson et al. 1964). It appears difficult to visualize an intramural compartmental arrangement so set as
8 462 G. KAHLSON, ELSA ROSENGREN AND R. THUNBERG to prevent functional contact between the newly formed histamine and the parietal cells, including the blood vessels draining the mucosa. Histamine newly formed at high rates thus appears potentially designed to provide secretory stimulation and, concomitantly, for the vasodilation prerequisite to secretion. This view appears acceptable only on the assumption that during the interdigestive phase the histamine formed at considerable rates either escapes functional contact with the parietal cells, or that the intramural concentration of newly formed histamine is subthreshold for stimulation. Both these assumptions appear perilous and at present do not lend themselves to the test of experiment. In this connexion it should be emphasized that the mucosal histamine content, i.e. the amount of preformed loosely held histamine, lacks stimulatory potency, as evidenced from the fact that the mucosal histamine content is higher in the nonsecreting state and lower during the course of secretion. Having elaborated means to inhibit histamine formation in vivo, Kahlson et al. (1963, 1964) found in rats, in which the mucosal histamine content had been lowered to about 5 % of normal by inhibiting histamine formation, that injection of gastrin still produced the typical elevation of mucosal HFC and an acid secretory response of normal magnitude. These authors conclude that the excitatory mechanism, whatever its precise nature, operates at the terminal link with a wide safety margin. By contrast, Levine (1965) reported that in rats in which histamine formation had been inhibited by a hydrazino analogue of histidine resulting in about 5 % fall in stomach and urinary histamine, the acid secretory response to insulin or gastrin was blocked. It has previously been shown that feeding, injection of gastrin and distension of the stomach induce acceleration of histamine formation by elevation of mucosal histidine decarboxylase activity. The present work reveals a similar change in mucosal histamine under the influence of agents producing vagal excitation. As the ultimate outcome of the various studies it appears clear that the three individual excitatory influences operating on feeding have a common functional feature, namely the power to accelerate greatly formation and turn-over rate of histamine in the parietal cell containing region of the gastric mucosa. The three influences appear to have a common goal, the full elucidation of which, owing to difficulties inherent in the problem, may well be far ahead. This work was supported by Research Grant 5 R1 HD255-6 from the National Institute of Health, Bethesda, Md.
9 VAGUS AND GASTRIC MUCOSAL HISTAMINE 463 REFERENCES BABKIN, B. P. (1938). The abnormal functioning of the gastric secretory mechanism as a possible factor in the pathogenesis of peptic ulcer. Can. med. Ass. J. 38, BENNETT, A. (1965). Effect of gastrin on isolated smooth muscle preparations. Nature, Lond. 28, CTARK, C. G., CURNOW, V. J., MURRAY, J. G., STEPHENS, F.. & WYLLIE, J. H. (1964). Mode of action of histamine in causing gastric secretion in man. Gut 5, CODE, C. F. (1965). Histamine and gastric secretion: a later look, Fedn Proc. 24, EMMELIN, N. & HENRIKSSON, K. G. (1953). Depressor activity of saliva after section of the chorda tympani. Acta physiol. scand. 3, suppl. 111, GREEN, H. & ERICKSON, R. W. (1964). Effect of some drugs upon rat brain histamine content. Int. J. Neuropharmac. 3, GREGORY, R. A. (1962). Secretory Mechanisms of the Gastro-intestinal Tract, 1st edn., pp London: E. Arnold Ltd. HAEGER, K., KAHLSON, G. & WESTLING, H. (1953). Evidence of a regulatory mechanism controlling the levels of histamine and histaminase in the gastro-intestinal tract. Acta physiol. scand. 3, suppl. 111, HAEGER, K. & KAHLSON, G. (1952). Distribution of histamine and histaminase in the gastrointestinal mucosa of fed and starved cats. Acta physiol. scand. 25, HAVERBACK, B. J., STUBRIN, M. I. & DYCE, B. J. (1965). Relationship of histamine to gastrini and other secretagogues. Fedn Proc. 24, HAVERBACK, B. J., TECIMER, L. B., DYCE, B. J., COHEN, M., STUBRIN, M. 1. & SANTA ANA, A. D. (1964). The effect of gastrin on stomach histamine in the rat. Life Sci. Oxford 3, HIGH, D. P., SHEPHERD, D. M. & WOODCOCK, B. G. (1965). Urinary histamine excretion in the gastrectomised rat. Life Sci. Oxford 4, HIRSCHOWITZ, B. I. & SACHS, G. (1965). Vagal gastric secretory stimulation by 2-deoxy-Dglucose. Am. J. Physiol. 29, KAHLSON, G., ROSENGREN, E., SVAHN, D. & THUNBERG, R. (1964). Mobilization and formation of histamine in the gastric mucosa as related to acid secretion. J. Physiol. 174, KAHLSON, G., ROSENGREN, E. & THUNBERG, R. (1963). Observations on the inhibition of histamine formation. J. Physiol. 169, KIM, K. S. & SHORE, P. A. (1963). Mechanism of action of reserpine and insulin on gastric amines and gastric acid secretion, and the effect of monoamine oxidase inhibition. J. Pharmac. exp. Ther. 141, LANE, A., IvY, A. C. & Ivy, E. K. (1957). Response of the chronic gastric fistula rat to histamine. Am. J. Physiol. 19, LEVINE, R. J. (1965). Effect of histidine decarboxylase inhibition on gastric acid secretion in the rat. Fedn Proc. 24, ROSENGREN, E. (1963). Histamine metabolism in the pregnant mouse. J. Physiol. 169, ROSENGREN, E. (1966). Histamine metabolism in pregnancy. Acta Univ. Lund. II, no. 8, pp SCHAYER, R. W. & Ivy, A. C. (1957). Evidence that histamine is a gastric secretory hormone in the rat. Am. J. Physiol. 189, SCHAYER, R. W. & Ivy, A. C. (1958). Release of C14-histamine from stomach and intestine on feeding. Am. J. Physiol. 193, SHORE, P. A. (1965). Release of histamine from stomach by vagus-stimulating drugs: association with gastric acid secretion. Fedn Proc. 24, SHORE, P. A., BURKHALTER, A. & COHN, V. H. (1959). A method for the fluorometric assay of histamine in tissues. J. Pharmac. exp. Ther. 127, SMITH, A. N. (1954). Gastrin and histamine release. J. Physiol. 123, 71-72P. SMITH, A. N. (196). The mode of action of gastrin as examined in the experimental animal and its action via the release of histamine in the stomach wall. Gut 1, 83. WHITE, T. (1966). Histamine and methylhistamine in cat brain and other tissues. Br. J. Pharmac. Chemother. 26,
Gastrin derivatives investigated for secretory potency and for changes in gastric mucosal histamine formation
 Br. J. Pharmac. (1970), 38, 473-477. Gastrin derivatives investigated for secretory potency and for changes in gastric mucosal histamine formation ELSA ROSENGREN AND S. E. SVENSSON Institute of Physiology,
Br. J. Pharmac. (1970), 38, 473-477. Gastrin derivatives investigated for secretory potency and for changes in gastric mucosal histamine formation ELSA ROSENGREN AND S. E. SVENSSON Institute of Physiology,
THE ROLE OF THE ANTRUM AND THE VAGUS NERVE IN THE FORMATION OF GASTRIC MUCOSAL HISTAMINE
 J. Phy8iol. (1969), 205, pp. 275-288 275 With 2 text-figures Printed in Great Britain THE ROLE OF THE ANTRUM AND THE VAGUS NERVE IN THE FORMATION OF GASTRIC MUCOSAL HISTAMINE By ELSA ROSENGREN AND S. E.
J. Phy8iol. (1969), 205, pp. 275-288 275 With 2 text-figures Printed in Great Britain THE ROLE OF THE ANTRUM AND THE VAGUS NERVE IN THE FORMATION OF GASTRIC MUCOSAL HISTAMINE By ELSA ROSENGREN AND S. E.
EXPERIMENTS ON THE INHIBITION OF HISTAMINE FORMATION IN THE RAT
 Br. J. Pharmac. Chemother. (1967), 30, 274-282. EXPERIMENTS ON THE INHIBITION OF HISTAMINE FORMATION IN THE RAT BY MARIAN JOHNSTON* AND G. KAHLSON From the Institute of Physiology, University of Lund,
Br. J. Pharmac. Chemother. (1967), 30, 274-282. EXPERIMENTS ON THE INHIBITION OF HISTAMINE FORMATION IN THE RAT BY MARIAN JOHNSTON* AND G. KAHLSON From the Institute of Physiology, University of Lund,
EFFECT OF THYROID HORMONES ON HISTAMINE FORMATION IN THE RAT
 Brit. J. Pharmacol. (1961), 17, 479-487. EFFECT OF THYROID HORMONES ON HISTAMINE FORMATION IN THE RAT BY T. BJURO, H. WESTLING AND H. WETTERQVIST From the Department of Clinical Physiology, University
Brit. J. Pharmacol. (1961), 17, 479-487. EFFECT OF THYROID HORMONES ON HISTAMINE FORMATION IN THE RAT BY T. BJURO, H. WESTLING AND H. WETTERQVIST From the Department of Clinical Physiology, University
possibility that the "gastric hormone" may not as yet have been extracted investigation of any part of the stomach other than the pyloric mucosa.
 234 6I2.32.014.2I :547x78I.5 HISTAMINE IN CANINE GASTRIC TISSUES. BY GERTRUDE GAVIN, E. W. McHENRY AmD M. J. WILSON. (From the Department of Physiological Hygiene, School of Hygiene, University of Toronto.)
234 6I2.32.014.2I :547x78I.5 HISTAMINE IN CANINE GASTRIC TISSUES. BY GERTRUDE GAVIN, E. W. McHENRY AmD M. J. WILSON. (From the Department of Physiological Hygiene, School of Hygiene, University of Toronto.)
ALTERATIONS IN HISTIDINE DECARBOXYLASE ACTIVITY DURING ANAPHYLACTIC SHOCK IN THE RAT
 Br. J. Pharmac. Chemother. (1967), 30, 392-399. ALTERATIONS IN HISTIDINE DECARBOXYLASE ACTIVITY DURING ANAPHYLACTIC SHOCK IN THE RAT BY A. G. RADWAN AND G. B. WEST* From the Department of Pharmacology,
Br. J. Pharmac. Chemother. (1967), 30, 392-399. ALTERATIONS IN HISTIDINE DECARBOXYLASE ACTIVITY DURING ANAPHYLACTIC SHOCK IN THE RAT BY A. G. RADWAN AND G. B. WEST* From the Department of Pharmacology,
THE EFFECT OF SODIUM INTAKE ON THE URINARY HISTAMINE IN ADRENALECTOMIZED RATS
 Brit. J. Pharmacol. (1964), 22, 453-462. THE EFFECT OF SODIUM INTAKE ON THE URINARY HISTAMINE IN ADRENALECTOMIZED RATS BY T. BJURO AND H. WESTLING* From the Department of Clinical Physiology, University
Brit. J. Pharmacol. (1964), 22, 453-462. THE EFFECT OF SODIUM INTAKE ON THE URINARY HISTAMINE IN ADRENALECTOMIZED RATS BY T. BJURO AND H. WESTLING* From the Department of Clinical Physiology, University
LEAKAGE OF TRANSMITTERS IN SALIVARY GLANDS
 Brit. J. Pharmacol. (1964), 22, 119-125. LEAKAGE OF TRANSMITTERS IN SALIVARY GLANDS BY N. ASSARSON AND N. EMMELIN From the Institute of Physiology, University of Lund, Sweden (Received October 8, 1963)
Brit. J. Pharmacol. (1964), 22, 119-125. LEAKAGE OF TRANSMITTERS IN SALIVARY GLANDS BY N. ASSARSON AND N. EMMELIN From the Institute of Physiology, University of Lund, Sweden (Received October 8, 1963)
however, reduced after parasympathetic denervation [Nordenfelt et al., 1960]. opposite to those caused by parasympathetic denervation.
![however, reduced after parasympathetic denervation [Nordenfelt et al., 1960]. opposite to those caused by parasympathetic denervation. however, reduced after parasympathetic denervation [Nordenfelt et al., 1960]. opposite to those caused by parasympathetic denervation.](/thumbs/90/101412039.jpg) CHOLINE ACETYLASE IN SALIVARY GLANDS OF THE CAT AFTER SYMPATHETIC DENERVATION. By IVAR NORDENFELT. From the Institute of Physiology, University of Lund, Sweden. (Received for publication 20th April 1964)
CHOLINE ACETYLASE IN SALIVARY GLANDS OF THE CAT AFTER SYMPATHETIC DENERVATION. By IVAR NORDENFELT. From the Institute of Physiology, University of Lund, Sweden. (Received for publication 20th April 1964)
MECHANISM BY WHICH FAT IN THE UPPER SMALL INTESTINE INHIBITS GASTRIC ACID
 GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.3 Printea in U.S.A. MECHANISM BY WHICH FAT IN THE UPPER SMALL INTESTINE INHIBITS GASTRIC ACID H. T. DEBAS, M.D., B. S. BEDI, M.B.,
GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.3 Printea in U.S.A. MECHANISM BY WHICH FAT IN THE UPPER SMALL INTESTINE INHIBITS GASTRIC ACID H. T. DEBAS, M.D., B. S. BEDI, M.B.,
Action of drugs on denervated myoepithelial cells of salivary glands
 Br. J. Pharmac. (1973), 48, 73-79. Action of drugs on denervated myoepithelial cells of salivary glands N. EMMELIN AND A. THULIN Institute of Physiology, University of Lund, Sweden Summary 1. The pressure
Br. J. Pharmac. (1973), 48, 73-79. Action of drugs on denervated myoepithelial cells of salivary glands N. EMMELIN AND A. THULIN Institute of Physiology, University of Lund, Sweden Summary 1. The pressure
pouches and in Heidenhain pouches the responses to 2-deoxy-D-glucose and to food were established, as were also the responses to gastrin pentapeptide
 J. Physiol. (197), 27, pp. 329-35 329 With 9 text-ftgure8 Printed in Great Britain THE SECRETORY PATTERN OF THREE STOMACH PREPARATIONS IN THE RAT BY S. E. SVENSSON From the Institute of Physiology, University
J. Physiol. (197), 27, pp. 329-35 329 With 9 text-ftgure8 Printed in Great Britain THE SECRETORY PATTERN OF THREE STOMACH PREPARATIONS IN THE RAT BY S. E. SVENSSON From the Institute of Physiology, University
ROLE OF ADRENAL HORMONES IN MAINTAINING TISSUE STORES OF NORADRENALINE DURING INCREASED SYMPATHETIC ACTIVITY
 Br. J. Pharmac. Chemother. (1966), 27, 532-535. ROLE OF ADRENAL HORMONES IN MAINTAINING TISSUE STORES OF NORADRENALINE DURING INCREASED SYMPATHETIC ACTIVITY BY V. M. AVAKIAN* AND MARTHE VOGT From the Agricultural
Br. J. Pharmac. Chemother. (1966), 27, 532-535. ROLE OF ADRENAL HORMONES IN MAINTAINING TISSUE STORES OF NORADRENALINE DURING INCREASED SYMPATHETIC ACTIVITY BY V. M. AVAKIAN* AND MARTHE VOGT From the Agricultural
SUPERSENSITIVITY OF THE SUBMAXILLARY GLAND FOLLOWING EXCLUSION OF THE POSTGANGLIONIC PARASYMPATHETIC NEURONE
 Brit. J. Pharmacol. (1960), 15, 356. SUPERSENSITIVITY OF THE SUBMAXILLARY GLAND FOLLOWING EXCLUSION OF THE POSTGANGLIONIC PARASYMPATHETIC NEURONE BY N. EMMELIN From the Institute of Physiology, University
Brit. J. Pharmacol. (1960), 15, 356. SUPERSENSITIVITY OF THE SUBMAXILLARY GLAND FOLLOWING EXCLUSION OF THE POSTGANGLIONIC PARASYMPATHETIC NEURONE BY N. EMMELIN From the Institute of Physiology, University
PEPSIN SECRETION DURING DAMAGE BY ETHANOL AND SALICYLIC ACID
 GASTROENTEROLOGY Copyriht 1972 by The Williams & Wilkins Co. Vol. 62. No. 3 Printed in U.S. A. PEPSIN SECRETION DURING DAMAGE BY ETHANOL AND SALICYLIC ACID LEONARD R. JOHNSON, PH.D. Department of Physiology
GASTROENTEROLOGY Copyriht 1972 by The Williams & Wilkins Co. Vol. 62. No. 3 Printed in U.S. A. PEPSIN SECRETION DURING DAMAGE BY ETHANOL AND SALICYLIC ACID LEONARD R. JOHNSON, PH.D. Department of Physiology
University of Buea. Faculty of Health Sciences. Programme in Medicine
 Faculty of Health Sciences University of Buea Wednesday, 28 th January 2009 Time: 8 00-10 00 Programme in Medicine MED 303 (Gastrointestinal Physiology) EXAMS (2008-2009) Identify the letter of the choice
Faculty of Health Sciences University of Buea Wednesday, 28 th January 2009 Time: 8 00-10 00 Programme in Medicine MED 303 (Gastrointestinal Physiology) EXAMS (2008-2009) Identify the letter of the choice
THE EXTRACTABLE HISTAMINE CONTENT OF GASTRIC
 THE EXTRACTABLE HISTAMINE CONTENT OF GASTRIC MUCOSA AND LUNG BEFORE AND AFTER PARENTERAL HISTAMINE ADMINISTRATION* LOUIS E. YOUNGt Edkins in 19064 stated that aqueous extracts of the pyloric and cardiac
THE EXTRACTABLE HISTAMINE CONTENT OF GASTRIC MUCOSA AND LUNG BEFORE AND AFTER PARENTERAL HISTAMINE ADMINISTRATION* LOUIS E. YOUNGt Edkins in 19064 stated that aqueous extracts of the pyloric and cardiac
CATABOLISM IN MAN (RECEIVED MARCH 14, 1960) aminoguanidine and iproniazid than could be expected from the animal experiments of Schayer.
 Brit. J. Pharmacol. (1960), t5, 351. THE EFFECT OF ENZYME INHIBITORS ON HISTAMINE CATABOLISM IN MAN BY S. E. LINDELL,* KAREN NILSSON, B.-E. ROOS, AND H. WESTLING From the Institute of Physiology, University
Brit. J. Pharmacol. (1960), t5, 351. THE EFFECT OF ENZYME INHIBITORS ON HISTAMINE CATABOLISM IN MAN BY S. E. LINDELL,* KAREN NILSSON, B.-E. ROOS, AND H. WESTLING From the Institute of Physiology, University
STUDIES ON THE EXPERIMENTAL GASTROINTESTINAL ULCERS PRODUCED BY RESERPINE AND STRESS II. ULCEROGENIC ACTIVITIES OF RESERPINE AND ITS ANALOGUES
 STUDIES ON THE EXPERIMENTAL GASTROINTESTINAL ULCERS PRODUCED BY RESERPINE AND STRESS II. ULCEROGENIC ACTIVITIES OF RESERPINE AND ITS ANALOGUES MASAMI DOTEUCHI Division of Neuropharmacology, Shionogi Research
STUDIES ON THE EXPERIMENTAL GASTROINTESTINAL ULCERS PRODUCED BY RESERPINE AND STRESS II. ULCEROGENIC ACTIVITIES OF RESERPINE AND ITS ANALOGUES MASAMI DOTEUCHI Division of Neuropharmacology, Shionogi Research
INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT
 Brit. J. Phawmacol. (1951), 6, 289. INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT BY From the Pharmacological Laboratory, University of St. Andrews, Medical School, Dundee (Received February 2, 1951)
Brit. J. Phawmacol. (1951), 6, 289. INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT BY From the Pharmacological Laboratory, University of St. Andrews, Medical School, Dundee (Received February 2, 1951)
THE ROLE OF GASTRINE AND ACETYLCHOLINE IN THE GASTRIC ACID SECRETION MECHANISM
 ANALELE UNIVERSITATII DIN ORADEA, Fascicula Ecotoxicologie, Zootehnie si Tehnologii de Industrie Alimentara THE ROLE OF GASTRINE AND ACETYLCHOLINE IN THE GASTRIC ACID SECRETION MECHANISM Gavril Paul, Ioana
ANALELE UNIVERSITATII DIN ORADEA, Fascicula Ecotoxicologie, Zootehnie si Tehnologii de Industrie Alimentara THE ROLE OF GASTRINE AND ACETYLCHOLINE IN THE GASTRIC ACID SECRETION MECHANISM Gavril Paul, Ioana
THE EFFECT OF ESERINE ON THE RESPONSE OF THE VAS DEFERENS TO HYPOGASTRIC NERVE STIMULATION
 Brit. J. Pharmacol. (1963), 20, 74-82. THE EFFECT OF ESERINE ON THE RESPONSE OF THE VAS DEFERENS TO HYPOGASTRIC NERVE STIMULATION BY J. H. BURN AND D. F. WEETMAN From the Biological Research Laboratories,
Brit. J. Pharmacol. (1963), 20, 74-82. THE EFFECT OF ESERINE ON THE RESPONSE OF THE VAS DEFERENS TO HYPOGASTRIC NERVE STIMULATION BY J. H. BURN AND D. F. WEETMAN From the Biological Research Laboratories,
Histologically the pyloric glands and the glands of Brunner in the. Ivy(7) and his co-workers seem to believe, at the present time, in the
 6I2.323[6I2.33 6I2.OI5.2I THE SMALL INTESTINE AND GASTRIC SECRETION. (With special reference to Brunner's Glands.) BY MARGARET M. MURRAY. (From the Department of Physiology, Bedford College.) EVIDENCE
6I2.323[6I2.33 6I2.OI5.2I THE SMALL INTESTINE AND GASTRIC SECRETION. (With special reference to Brunner's Glands.) BY MARGARET M. MURRAY. (From the Department of Physiology, Bedford College.) EVIDENCE
RECEPTORS MEDIATING SOME ACTIONS OF
 Br. J. Phannac. Chemother. (1966), 27, 427-439. RECEPTORS MEDIATING SOME ACTIONS OF HISTAMINE BY A. S. F. ASH AND H. 0. SCHILD From the Department of Pharmacology, University College London (Received April
Br. J. Phannac. Chemother. (1966), 27, 427-439. RECEPTORS MEDIATING SOME ACTIONS OF HISTAMINE BY A. S. F. ASH AND H. 0. SCHILD From the Department of Pharmacology, University College London (Received April
EFFECT OF DENERVATION AND OF COCAINE ON THE ACTION OF SYMPATHOMIMETIC AMINES
 Brit. J. Pharmacol. (1960), 15, 328. EFFECT OF DENERVATION AND OF COCAINE ON THE ACTION OF SYMPATHOMIMETIC AMINES BY B. C. R. STROMBLAD From the Institute of Physiology, Lund, Sweden (RECEIVED FEBRUARY
Brit. J. Pharmacol. (1960), 15, 328. EFFECT OF DENERVATION AND OF COCAINE ON THE ACTION OF SYMPATHOMIMETIC AMINES BY B. C. R. STROMBLAD From the Institute of Physiology, Lund, Sweden (RECEIVED FEBRUARY
INTRODUCTION TO GASTROINTESTINAL FUNCTIONS
 1 INTRODUCTION TO GASTROINTESTINAL FUNCTIONS 2 Learning outcomes List two main components that make up the digestive system Describe the 6 essential functions of the GIT List factors (neurological, hormonal
1 INTRODUCTION TO GASTROINTESTINAL FUNCTIONS 2 Learning outcomes List two main components that make up the digestive system Describe the 6 essential functions of the GIT List factors (neurological, hormonal
(Axelsson & Thesleff, 1959; Miledi, 1960). Recently, it has become
 J. Physiol. (1973), 230, pp. 613-618 613 With 1 text-figure Printed in Great Britain INFLUENCE OF CHRONIC NEOSTIGMINE TREATMENT ON THE NUMBER OF ACETYLCHOLINE RECEPTORS AND THE RELEASE OF ACETYLCHOLINE
J. Physiol. (1973), 230, pp. 613-618 613 With 1 text-figure Printed in Great Britain INFLUENCE OF CHRONIC NEOSTIGMINE TREATMENT ON THE NUMBER OF ACETYLCHOLINE RECEPTORS AND THE RELEASE OF ACETYLCHOLINE
Physiological processes in the GI tract:
 Gastrointestinal physiology for medical students General principal of gastrointestinal function Motility, nervous control and blood circulation Physiological processes in the GI tract: Motility Secretion
Gastrointestinal physiology for medical students General principal of gastrointestinal function Motility, nervous control and blood circulation Physiological processes in the GI tract: Motility Secretion
to food and histamine
 Gut, 97,, 53-57 Maximal acid response of Pavlov pouches to food and histamine A. MARVIN BROOKS AND MORTON I. GROSSMAN From the Veterans Administration Center and UCLA School of Medicine, Departments of
Gut, 97,, 53-57 Maximal acid response of Pavlov pouches to food and histamine A. MARVIN BROOKS AND MORTON I. GROSSMAN From the Veterans Administration Center and UCLA School of Medicine, Departments of
ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE
![ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE](/thumbs/74/71109260.jpg) Brit. J. Pharmacol. (1962), 18, 161-166. ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE BY G. HERTTING,* J. AXELROD AND R. W. PATRICK From the Laboratory of Clinical
Brit. J. Pharmacol. (1962), 18, 161-166. ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE BY G. HERTTING,* J. AXELROD AND R. W. PATRICK From the Laboratory of Clinical
ISOLATED AND INNERVATED ATRIA AND VESSELS
 Brit. J. Pharmacol. (1960), 15, 117. THE ACTION OF SYMPATHETIC BLOCKING AGENTS ON ISOLATED AND INNERVATED ATRIA AND VESSELS BY S. HUKOVIC* From the Department of Pharmacology, University of Oxford (RECEIVED
Brit. J. Pharmacol. (1960), 15, 117. THE ACTION OF SYMPATHETIC BLOCKING AGENTS ON ISOLATED AND INNERVATED ATRIA AND VESSELS BY S. HUKOVIC* From the Department of Pharmacology, University of Oxford (RECEIVED
factors. directly. There are many, however, who regard the hydrochloric acid degree of stimulation of the stomach cells [Roseman, 1927; Katsch &
 308 J. Physiol. (I940) 97, 308-3I5 6I2.323.3 ON THE PRIMARY ACIDITY OF THE GASTRIC JUICE BY TORSTEN TEORELL From the Department of Medical Chemistry, University of Uppsala, Sweden (Received 10 July 1939)
308 J. Physiol. (I940) 97, 308-3I5 6I2.323.3 ON THE PRIMARY ACIDITY OF THE GASTRIC JUICE BY TORSTEN TEORELL From the Department of Medical Chemistry, University of Uppsala, Sweden (Received 10 July 1939)
OBSERVATIONS ON THE ISOLATED VAS DEFERENS
 Brit. J. Pharmacol. (1963), 20, 299-306. OBSERVATIONS ON THE ISOLATED VAS DEFERENS BY P. OHLIN AND B. C. R. STROMBLAD* From the Institute of Physiology, University of Lund, Sweden (Received December 3,
Brit. J. Pharmacol. (1963), 20, 299-306. OBSERVATIONS ON THE ISOLATED VAS DEFERENS BY P. OHLIN AND B. C. R. STROMBLAD* From the Institute of Physiology, University of Lund, Sweden (Received December 3,
THE ACTION OF PHYSOSTIGMINE AND THE DISTRIBUTION OF CHOLINESTERASES IN THE CHICKEN OESOPHAGUS
 Br. J. Phannac. Chemother. (1968), 33, 531-536. THE ACTION OF PHYSOSTIGMINE AND THE DISTRIBUTION OF CHOLINESTERASES IN THE CHICKEN OESOPHAGUS BY A. L. BARTLET AND T. HASSAN From the Department of Veterinary
Br. J. Phannac. Chemother. (1968), 33, 531-536. THE ACTION OF PHYSOSTIGMINE AND THE DISTRIBUTION OF CHOLINESTERASES IN THE CHICKEN OESOPHAGUS BY A. L. BARTLET AND T. HASSAN From the Department of Veterinary
neoplastic mast cells (Giarman, Potter & Day, 1960). According to Toh
 J. Phy8iol. (1963), 165, pp. 83-88 83 Printed in Great Britain RELEASE OF HISTAMINE FROM SPLEEN BY KIDNEY EXTRACT, RESERPINE AND COMPOUND 48/80 BY ANNIE B. ELLIOTT From the Department of Physiology, University
J. Phy8iol. (1963), 165, pp. 83-88 83 Printed in Great Britain RELEASE OF HISTAMINE FROM SPLEEN BY KIDNEY EXTRACT, RESERPINE AND COMPOUND 48/80 BY ANNIE B. ELLIOTT From the Department of Physiology, University
gland, the tongue and the sweat glands of the cat. The submaxillary
 306 547.435-292:6I2.8I7 THE LIBERATION OF ACETYLCHOLINE BY POTASSIUM. BY W. FELDBERG1 AND J. A. GUIMARAIS1,2. (From the National Institute for Medical Research, London, N.W. 3.) (Received November 22,
306 547.435-292:6I2.8I7 THE LIBERATION OF ACETYLCHOLINE BY POTASSIUM. BY W. FELDBERG1 AND J. A. GUIMARAIS1,2. (From the National Institute for Medical Research, London, N.W. 3.) (Received November 22,
Cytochemical Quantification of Physiologic Regulation of Oxyntic Cell Carbonic Anhydrase a
 Cytochemical Quantification of Physiologic Regulation of Oxyntic Cell Carbonic Anhydrase a A. I. VINIKb and A. A. HELDSINGER Department of Internal Medicine University of Michigan Medical School Ann Arbor,
Cytochemical Quantification of Physiologic Regulation of Oxyntic Cell Carbonic Anhydrase a A. I. VINIKb and A. A. HELDSINGER Department of Internal Medicine University of Michigan Medical School Ann Arbor,
marked secretion ofcatecholamines and a subsequent inhibition ofsecretion although the basal secretion shows an initial rise.
 J. Physiol. (1969), 2, pp. 797-85 797 With 7 text-ftgurem Printed in Great Britain SODIUM IONS AND THE SECRETION OF CATECHOLAMINES By P. BANKS, ROSEMARY BIGGINS, R. BISHOP, B. CHRISTIAN AND N. CURRIE From
J. Physiol. (1969), 2, pp. 797-85 797 With 7 text-ftgurem Printed in Great Britain SODIUM IONS AND THE SECRETION OF CATECHOLAMINES By P. BANKS, ROSEMARY BIGGINS, R. BISHOP, B. CHRISTIAN AND N. CURRIE From
(b) Stomach s function 1. Dilution of food materials 2. Acidification of food (absorption of dietary Fe in small intestine) 3. Partial chemical digest
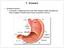 (1) General features a) Stomach is widened portion of gut-tube: between tubular and spherical; Note arranged of smooth muscle tissue in muscularis externa. 1 (b) Stomach s function 1. Dilution of food
(1) General features a) Stomach is widened portion of gut-tube: between tubular and spherical; Note arranged of smooth muscle tissue in muscularis externa. 1 (b) Stomach s function 1. Dilution of food
A comparison of the sensitivities of innervated and denervated rat vasa deferentia to agonist drugs
 Br. J. Pharmac. (1970), 39, 748-754. A comparison of the sensitivities of innervated and denervated rat vasa deferentia to agonist drugs A. T. BIRMINGHAM*, G. PATRSON AND J. W6JCICKIt Department of Pharmacology,
Br. J. Pharmac. (1970), 39, 748-754. A comparison of the sensitivities of innervated and denervated rat vasa deferentia to agonist drugs A. T. BIRMINGHAM*, G. PATRSON AND J. W6JCICKIt Department of Pharmacology,
McSwiney and Wadge [1930] described the effects on the stomach of
![McSwiney and Wadge [1930] described the effects on the stomach of McSwiney and Wadge [1930] described the effects on the stomach of](/thumbs/83/87976616.jpg) 6I2.328:6I2.898 THE SYMPATHETIC INNERVATION OF THE STOMACH. II. The effect of stimulation of the peri-arterial nerves on the stomach and small intestine. BY B. A. McSWINEY AND J. M. ROBSON. (Department
6I2.328:6I2.898 THE SYMPATHETIC INNERVATION OF THE STOMACH. II. The effect of stimulation of the peri-arterial nerves on the stomach and small intestine. BY B. A. McSWINEY AND J. M. ROBSON. (Department
RELEASE OF HISTAMINE INTO GASTRIC VENOUS BLOOD FOLLOWING INJURY BY ACETIC OR SALICYLIC ACID
 GASTROENTEROLOGY Copyright 1967 by The Williams & Wilkins Co. Vol. 52, No.3 Printed in U.S.A. RELEASE OF HISTAMINE INTO GASTRIC VENOUS BLOOD FOLLOWING INJURY BY ACETIC OR SALICYLIC ACID LEONARD R. JOHNSON
GASTROENTEROLOGY Copyright 1967 by The Williams & Wilkins Co. Vol. 52, No.3 Printed in U.S.A. RELEASE OF HISTAMINE INTO GASTRIC VENOUS BLOOD FOLLOWING INJURY BY ACETIC OR SALICYLIC ACID LEONARD R. JOHNSON
5. Which component of the duodenal contents entering the stomach causes the most severe changes to gastric mucosa:
 Gastro-intestinal disorders 1. Which are the most common causes of chronic gastritis? 1. Toxic substances 2. Chronic stress 3. Alimentary factors 4. Endogenous noxious stimuli 5. Genetic factors 2. Chronic
Gastro-intestinal disorders 1. Which are the most common causes of chronic gastritis? 1. Toxic substances 2. Chronic stress 3. Alimentary factors 4. Endogenous noxious stimuli 5. Genetic factors 2. Chronic
ACETYLCHOLINE, HISTAMINE AND GASTRIN
 Br. J. Pharmac. (1977), 61, 279-284 THE EFFECT OF ATROPINE ON ACID SECRETION STIMULATED BY ACETYLCHOLINE, HISTAMINE AND GASTRIN IN THE ISOLATED WHOLE STOMACH OF THE RAT K.T. BUNCE, GILLIAN F. MARSH & M.E.
Br. J. Pharmac. (1977), 61, 279-284 THE EFFECT OF ATROPINE ON ACID SECRETION STIMULATED BY ACETYLCHOLINE, HISTAMINE AND GASTRIN IN THE ISOLATED WHOLE STOMACH OF THE RAT K.T. BUNCE, GILLIAN F. MARSH & M.E.
LOCALIZATION OF MONOAMINE OXIDASE IN MAMMALIAN
 Brit. Heart J., 1965, 27, 116. LOCALIZATION OF MONOAMINE OXIDASE IN MAMMALIAN AND REPTILIAN HEART BY E. MULLER* AND A. G. E. PEARSEt From the Department ofpathology, Postgraduate Medical School, London
Brit. Heart J., 1965, 27, 116. LOCALIZATION OF MONOAMINE OXIDASE IN MAMMALIAN AND REPTILIAN HEART BY E. MULLER* AND A. G. E. PEARSEt From the Department ofpathology, Postgraduate Medical School, London
(Received April 6, 1936)
 275 6I2.26 RESPIRATION AND FUNCTIONAL ACTIVITY BY W. DEUTSCH AND H. S. RAPER (From the Department of Physiology, University of Manchester) (Received April 6, 1936) IT has long been established that in
275 6I2.26 RESPIRATION AND FUNCTIONAL ACTIVITY BY W. DEUTSCH AND H. S. RAPER (From the Department of Physiology, University of Manchester) (Received April 6, 1936) IT has long been established that in
blood-vessels of the isolated perfused lungs of the rat. Both Hirakawa
 547.435-292: 547.781.5: 577.174.5: 612.215 THE ACTION OF ADRENALINE, ACETYLCHOLINE, AND HIS- TAMINE ON THE LUNGS OF THE RAT. By P. FoGGIE. From the Physiology Department, University of Edinburgh. (Received
547.435-292: 547.781.5: 577.174.5: 612.215 THE ACTION OF ADRENALINE, ACETYLCHOLINE, AND HIS- TAMINE ON THE LUNGS OF THE RAT. By P. FoGGIE. From the Physiology Department, University of Edinburgh. (Received
'the perfusion of the cat's lung a cannula was tied into the left auricle and :547.78I.5
 280 576.809.73:547.78I.5 LIBERATION OF HISTAMINE FROM THE PERFUSED LUNG BY STAPHYLOCOCCAL TOXIN BY W. FELDBERG AND E. V. KEOGH1 From The Walter and Eliza Hall Institute, Melbourne (Received 5 March 1937)
280 576.809.73:547.78I.5 LIBERATION OF HISTAMINE FROM THE PERFUSED LUNG BY STAPHYLOCOCCAL TOXIN BY W. FELDBERG AND E. V. KEOGH1 From The Walter and Eliza Hall Institute, Melbourne (Received 5 March 1937)
INTRODUCTION. IN a previous paper(l) we have been able to show that adrenaline may
 REVERSAL OF THE ACTION OF ADRENALINE. BY B. A. McSWINEY AND G. L. BROWN. (From the Department of Physiology, University of Manchester.) INTRODUCTION. IN a previous paper(l) we have been able to show that
REVERSAL OF THE ACTION OF ADRENALINE. BY B. A. McSWINEY AND G. L. BROWN. (From the Department of Physiology, University of Manchester.) INTRODUCTION. IN a previous paper(l) we have been able to show that
MAST-CELLS are present in the digestive tract of all classes of vertebrates
 The Distribution of Mast-Cells in the Digestive Tract of Laboratory Animals: Its Bearings on the Problem of the Location of Histamine in Tissues By I. MOTA, A. G. FERRI, AND S. YONEDA 251 (From the Laboratory
The Distribution of Mast-Cells in the Digestive Tract of Laboratory Animals: Its Bearings on the Problem of the Location of Histamine in Tissues By I. MOTA, A. G. FERRI, AND S. YONEDA 251 (From the Laboratory
INHIBITION OF AUDITORY NERVE ACTION POTENTIALS BY ACETYLCHOLINE AND PHYSOSTIGMINE
 Br. J. Pharmac. Chemother. (1966), 28, 207-211. INHIBITION OF AUDITORY NERVE ACTION POTENTIALS BY ACETYLCHOLINE AND PHYSOSTIGMINE BY J. AMARO, P. S. GUTH AND L. WANDERLINDER From the Department of Pharmacology,
Br. J. Pharmac. Chemother. (1966), 28, 207-211. INHIBITION OF AUDITORY NERVE ACTION POTENTIALS BY ACETYLCHOLINE AND PHYSOSTIGMINE BY J. AMARO, P. S. GUTH AND L. WANDERLINDER From the Department of Pharmacology,
INTESTINAL ABSORPTION OF AMINO ACIDS I. THE EFFECT OF VITAMIN B6 ON THE ABSORPTION OF L-AMINO ACIDS THROUGH THE INTESTINE
 The Journal of Biochemistry, Vol. 47, No. 1, 1960 INTESTINAL ABSORPTION OF AMINO ACIDS I. THE EFFECT OF VITAMIN B6 ON THE ABSORPTION OF L-AMINO ACIDS THROUGH THE INTESTINE By HITOSHI AKEDO, TADASHI SUGAWA,
The Journal of Biochemistry, Vol. 47, No. 1, 1960 INTESTINAL ABSORPTION OF AMINO ACIDS I. THE EFFECT OF VITAMIN B6 ON THE ABSORPTION OF L-AMINO ACIDS THROUGH THE INTESTINE By HITOSHI AKEDO, TADASHI SUGAWA,
hexamethonium, the effects of acetylcholine were on muscarinic receptors, probably
 J. Phyaiol. (1978), 279, pp. 39-32 39 With 8 text-figure8 Printed in Great Britain MODULATION OF CANINE ANTRAL CIRCULAR SMOOTH MUSCLE BY ACETYLCHOLINE, NORADRENALINE AND PENTAGASTRIN BY T. Y. EL-SHARKAWY
J. Phyaiol. (1978), 279, pp. 39-32 39 With 8 text-figure8 Printed in Great Britain MODULATION OF CANINE ANTRAL CIRCULAR SMOOTH MUSCLE BY ACETYLCHOLINE, NORADRENALINE AND PENTAGASTRIN BY T. Y. EL-SHARKAWY
University of Essen, Essen, F.R.G.
 J. Phy8iol. (1985), 369, pp. 355-364 355 With 4 text-figure8 Printed in Great Britain GASTRIN RESPONSE TO A MEAL BEFORE AND AFTER CUTTING THE EXTRINSIC NERVES OF THE STOMACH IN THE DOG By V. E. EYSSELEIN,
J. Phy8iol. (1985), 369, pp. 355-364 355 With 4 text-figure8 Printed in Great Britain GASTRIN RESPONSE TO A MEAL BEFORE AND AFTER CUTTING THE EXTRINSIC NERVES OF THE STOMACH IN THE DOG By V. E. EYSSELEIN,
The absorption of water from the whole stomach. or one of its parts has not been demonstrated. Many years ago Pavlov showed that water was a
 GASTRIC SECRETION. III. THE ABSORPTION OF HEAVY WATER FROM POUCHES OF THE BODY AND ANTRUM OF THE STOMACH OF THE DOG By OLIVER COPE, HESTER BLATT, AND MARGARET R. BALL (From the Surgical Research Laboratories
GASTRIC SECRETION. III. THE ABSORPTION OF HEAVY WATER FROM POUCHES OF THE BODY AND ANTRUM OF THE STOMACH OF THE DOG By OLIVER COPE, HESTER BLATT, AND MARGARET R. BALL (From the Surgical Research Laboratories
Autonomic Nervous System
 Autonomic Nervous System Keri Muma Bio 6 Organization of the Nervous System Efferent Division Somatic Nervous System Voluntary control Effector = skeletal muscles Muscles must be excited by a motor neuron
Autonomic Nervous System Keri Muma Bio 6 Organization of the Nervous System Efferent Division Somatic Nervous System Voluntary control Effector = skeletal muscles Muscles must be excited by a motor neuron
abnormally high compared to those encountered when animals are fed by University of Iowa, Iowa City, Iowa, U.S.A.
 J. Phy8iol. (1965), 181, pp. 59-67 59 With 5 text-ftgure8 Printed in Great Britain THE ANALYSIS OF GLUCOSE MEASUREMENTS BY COMPUTER SIMULATION* BY R. G. JANES "D J. 0. OSBURN From the Departments of Anatomy
J. Phy8iol. (1965), 181, pp. 59-67 59 With 5 text-ftgure8 Printed in Great Britain THE ANALYSIS OF GLUCOSE MEASUREMENTS BY COMPUTER SIMULATION* BY R. G. JANES "D J. 0. OSBURN From the Departments of Anatomy
Circulating Levels of Histamine and Histaminase under the Influence of An Indigenous Drug Abana in Chemically induced Ischaemic Heart Disease
 [Current Medical Practice (1988): (5), 115] Circulating Levels of Histamine and Histaminase under the Influence of An Indigenous Drug Abana in Chemically induced Ischaemic Heart Disease Lokesh Upadhyaya,
[Current Medical Practice (1988): (5), 115] Circulating Levels of Histamine and Histaminase under the Influence of An Indigenous Drug Abana in Chemically induced Ischaemic Heart Disease Lokesh Upadhyaya,
The actions of bombesin on gastric secretion of the dog and the rat
 Br. J. Pharmac. (1973), 49, 437-444. The actions of bombesin on gastric secretion of the dog and the rat G. BERTACCINI, V. ERSPAMER AND M. IMPICCIATORE Institutes of Pharmacology of the Universities of
Br. J. Pharmac. (1973), 49, 437-444. The actions of bombesin on gastric secretion of the dog and the rat G. BERTACCINI, V. ERSPAMER AND M. IMPICCIATORE Institutes of Pharmacology of the Universities of
Chapter 20 The Digestive System Exam Study Questions
 Chapter 20 The Digestive System Exam Study Questions 20.1 Overview of GI Processes 1. Describe the functions of digestive system. 2. List and define the four GI Processes: 20.2 Functional Anatomy of the
Chapter 20 The Digestive System Exam Study Questions 20.1 Overview of GI Processes 1. Describe the functions of digestive system. 2. List and define the four GI Processes: 20.2 Functional Anatomy of the
Digestive System Module 4: The Stomach *
 OpenStax-CNX module: m49286 1 Digestive System Module 4: The * Donna Browne Based on The by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0
OpenStax-CNX module: m49286 1 Digestive System Module 4: The * Donna Browne Based on The by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0
THE INHIBITORY EFFECT OF STILBOESTROL ON GASTRIC SECRETION IN CATS
 Brit. J. Pharmacol. (1950), 5, 3S9. THE INHIBITORY EFFECT OF STILBOESTROL ON GASTRIC SECRETION IN CATS BY K. N. OJHA* AND D. R. WOOD From the Department of Pharmacology and Therapeutics, University of
Brit. J. Pharmacol. (1950), 5, 3S9. THE INHIBITORY EFFECT OF STILBOESTROL ON GASTRIC SECRETION IN CATS BY K. N. OJHA* AND D. R. WOOD From the Department of Pharmacology and Therapeutics, University of
REVERSAL OF NICOTINE ACTION ON THE INTESTINE BY ATROPINE
 Brit. J. Pharmacol. (1951), 6, 31 1. REVERSAL OF NICOTINE ACTION ON THE INTESTINE BY ATROPINE BY N. AMBACHE AND J. EDWARDS From the Medical Research Council, Ophthalmological Research Unit, Institute Ophthalmology,
Brit. J. Pharmacol. (1951), 6, 31 1. REVERSAL OF NICOTINE ACTION ON THE INTESTINE BY ATROPINE BY N. AMBACHE AND J. EDWARDS From the Medical Research Council, Ophthalmological Research Unit, Institute Ophthalmology,
(From the Physiological Laboratory, University College.)
 ON THE UNIFORMITY OF THE PANCREATIC ME- CHANISM IN VERTEBRATA. BY W. M. BAYLISS AND E. H. STARLING. (Nine Figures in Text.) (From the Physiological Laboratory, University College.) In a previous paper'
ON THE UNIFORMITY OF THE PANCREATIC ME- CHANISM IN VERTEBRATA. BY W. M. BAYLISS AND E. H. STARLING. (Nine Figures in Text.) (From the Physiological Laboratory, University College.) In a previous paper'
Using a technique by which it is possible to study gastro-intestinal absorption
 531 J. Physiol. (I956) I34, 53I-537 THE ABSORPTION OF GLUCOSE BY THE INTACT RAT BY P. C. REYNELL AND G. H. SPRAY From the Nuffield Department of Clinical Medicine, University of Oxford (Received 30 May
531 J. Physiol. (I956) I34, 53I-537 THE ABSORPTION OF GLUCOSE BY THE INTACT RAT BY P. C. REYNELL AND G. H. SPRAY From the Nuffield Department of Clinical Medicine, University of Oxford (Received 30 May
THE EFFECTS OF ION CHANGES ON THE CONTRACTION OF THE RAT UTERUS STIMULATED BY OXYTOCIN
 Brit. J. Pharmacol. (1961), 16, 45-49. THE EFFECTS OF ION CHANGES ON THE CONTRACTION OF THE RAT UTERUS STIMULATED BY OXYTOCIN BY P. J. BENTLEY AND ELEANOR McEWEN From the Department of Physiology, The
Brit. J. Pharmacol. (1961), 16, 45-49. THE EFFECTS OF ION CHANGES ON THE CONTRACTION OF THE RAT UTERUS STIMULATED BY OXYTOCIN BY P. J. BENTLEY AND ELEANOR McEWEN From the Department of Physiology, The
Development of the Rodent Gastrointestinal Tract:
 White Paper Development of the Rodent Gastrointestinal Tract: The regulation of the development of the GI tract is unique and complex. With respect to the GI tract there are several considerations that
White Paper Development of the Rodent Gastrointestinal Tract: The regulation of the development of the GI tract is unique and complex. With respect to the GI tract there are several considerations that
3, 4), although its concentration in mixed gastric
 THE VALUE OF THE ACID TEST MEAL: A STUDY OF NORMAL PERSONS AND OF PERSONS WITH DUODENAL ULCER By C. STUART WELCH AND MANDRED W. COMFORT (From The Mayo Foundation and the Division of Medicine, The Mayo
THE VALUE OF THE ACID TEST MEAL: A STUDY OF NORMAL PERSONS AND OF PERSONS WITH DUODENAL ULCER By C. STUART WELCH AND MANDRED W. COMFORT (From The Mayo Foundation and the Division of Medicine, The Mayo
A. Incorrect! Histamine is a secretagogue for stomach acid, but this is not the only correct answer.
 Pharmacology - Problem Drill 21: Drugs Used To Treat GI Disorders No. 1 of 10 1. Endogenous secretagogues for stomach acid include: #01 (A) Histamine (B) Gastrin (C) PGE1 (D) A and B (E) A, B and C Histamine
Pharmacology - Problem Drill 21: Drugs Used To Treat GI Disorders No. 1 of 10 1. Endogenous secretagogues for stomach acid include: #01 (A) Histamine (B) Gastrin (C) PGE1 (D) A and B (E) A, B and C Histamine
QUARTERLY JOURNAL OF EXPERIMENTAL PHYSIOLOGY
 612.337:612.013.88 "e ra4 78D QUARTERLY JOURNAL OF EXPERIMENTAL PHYSIOLOGY AND COGNATE MEDICAL SCIENCES FACTORS INFLUENCING THE MOTILITY OF THE PERFUSED HORSE INTESTINE. By FRANK ALEXANDER. From the Department
612.337:612.013.88 "e ra4 78D QUARTERLY JOURNAL OF EXPERIMENTAL PHYSIOLOGY AND COGNATE MEDICAL SCIENCES FACTORS INFLUENCING THE MOTILITY OF THE PERFUSED HORSE INTESTINE. By FRANK ALEXANDER. From the Department
Drug Therapy of Parkinsonism. Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia
 Drug Therapy of Parkinsonism Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Parkinsonism is a progressive neurological disorder of muscle movement, usually
Drug Therapy of Parkinsonism Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Parkinsonism is a progressive neurological disorder of muscle movement, usually
GI Secretion 1: Salivary and Gastric Secretion Jack Grider, Ph.D.
 GI Secretion 1: Salivary and Gastric Secretion Jack Grider, Ph.D. OBJECTIVES: 1. List the volumes of secretion by various regions. 2. Predict the components of salivary secretion at different flow rates.
GI Secretion 1: Salivary and Gastric Secretion Jack Grider, Ph.D. OBJECTIVES: 1. List the volumes of secretion by various regions. 2. Predict the components of salivary secretion at different flow rates.
STUDIES ON THE MODE OF ACTION OF THE DIAPAUSE HORMONE IN THE SILKWORM, BOMBYX MORI L.
 J. Exp. Biol. (4), 4, 55-55 Printed in Great Britain STUDIES ON THE MODE OF ACTION OF THE DIAPAUSE HORMONE IN THE SILKWORM, BOMBYX MORI L. II. CONTENT OF DIAPAUSE HORMONE IN THE SUBOESOPHAGEAL GANGLION
J. Exp. Biol. (4), 4, 55-55 Printed in Great Britain STUDIES ON THE MODE OF ACTION OF THE DIAPAUSE HORMONE IN THE SILKWORM, BOMBYX MORI L. II. CONTENT OF DIAPAUSE HORMONE IN THE SUBOESOPHAGEAL GANGLION
Nervous and chemical regulation heart
 Nervous and chemical regulation heart Myogenic heart when separated out from the body keep on beating on their own but when in the body the rate of heart beat is modified through various stimuli such as
Nervous and chemical regulation heart Myogenic heart when separated out from the body keep on beating on their own but when in the body the rate of heart beat is modified through various stimuli such as
Adrenergic fibres in the human intestine
 Gut, 1968, 9, 678-682 Adrenergic fibres in the human intestine L. CAPURSO,1 C. A. FRIEDMANN, AND A. G. PARKS From the Research Department, St Mark's Hospital, London, and the London Hospital, Whitechapel,
Gut, 1968, 9, 678-682 Adrenergic fibres in the human intestine L. CAPURSO,1 C. A. FRIEDMANN, AND A. G. PARKS From the Research Department, St Mark's Hospital, London, and the London Hospital, Whitechapel,
Composed by Natalia Leonidovna Svintsitskaya, Associate professor of the Chair of Human Anatomy, Candidate of Medicine
 Theoretical background to the study of the autonomic nervous system. Sympathetic and parasympathetic divisions of the autonomic nervous system. Features of the structure, function Composed by Natalia Leonidovna
Theoretical background to the study of the autonomic nervous system. Sympathetic and parasympathetic divisions of the autonomic nervous system. Features of the structure, function Composed by Natalia Leonidovna
The Autonomic Nervous
 Autonomic Nervous System The Autonomic Nervous Assess Prof. Fawzia Al-Rouq System Department of Physiology College of Medicine King Saud University LECTUR (1) Functional Anatomy & Physiology of Autonomic
Autonomic Nervous System The Autonomic Nervous Assess Prof. Fawzia Al-Rouq System Department of Physiology College of Medicine King Saud University LECTUR (1) Functional Anatomy & Physiology of Autonomic
EFFECT OF SEX HORMONES ON THE EXCRETION OF FREE HISTAMINE BY MALE AND FEMALE RATS
 Brit. J. Pharmacol. (1961), 16, 50-58. EFFECT OF SEX HORMONES ON THE EXCRETION OF FREE HISTAMINE BY MALE AND FEMALE RATS BY P. B. MARSHALL From the Department of Pharmacology and Therapeutics, University
Brit. J. Pharmacol. (1961), 16, 50-58. EFFECT OF SEX HORMONES ON THE EXCRETION OF FREE HISTAMINE BY MALE AND FEMALE RATS BY P. B. MARSHALL From the Department of Pharmacology and Therapeutics, University
(Received 8 December 1966)
 J. Physiol. (1967), 189, pp. 545-550 545 With 2 text-figure8 Printed in Great Britain FUSIMOTOR STIMULATION AND THE DYNAMIC SENSITIVITY OF THE SECONDARY ENDING OF THE MUSCLE SPINDLE BY M. C. BROWN, I.
J. Physiol. (1967), 189, pp. 545-550 545 With 2 text-figure8 Printed in Great Britain FUSIMOTOR STIMULATION AND THE DYNAMIC SENSITIVITY OF THE SECONDARY ENDING OF THE MUSCLE SPINDLE BY M. C. BROWN, I.
show-n to give off a branch, and sometimes two or three branches, to
 THE EFFECT OF STIMULATION OF THE VAGI ON THE PYLORIC REGION OF THE STOMACH. BY E. D. McCREA1 AND B. A. McSWINEY. (From the Department of Physiology, University of Manchester.) THE course taken by the vagus
THE EFFECT OF STIMULATION OF THE VAGI ON THE PYLORIC REGION OF THE STOMACH. BY E. D. McCREA1 AND B. A. McSWINEY. (From the Department of Physiology, University of Manchester.) THE course taken by the vagus
Since peripheral vasodilatation is one of the consequences of the administration
 J. Phy8iol. (1961), 155, pp. 161-174 161 With 7 text-figure8 Printed in Great Britain THE ACTION OF POSTERIOR PITUITARY HORMONES AND OESTROGENS ON THE VASCULAR SYSTEM OF THE RAT BY SYBIL LLOYD AND MARY
J. Phy8iol. (1961), 155, pp. 161-174 161 With 7 text-figure8 Printed in Great Britain THE ACTION OF POSTERIOR PITUITARY HORMONES AND OESTROGENS ON THE VASCULAR SYSTEM OF THE RAT BY SYBIL LLOYD AND MARY
Vagal Regulation of Acid Secretion and Gastrin Release
 YALE JOURNAL OF BIOLOGY AND MEDICINE 67 (1994), pp. 145-151. Copyright 1995. All rights reserved. Vagal Regulation of Acid Secretion and Gastrin Release Haile T. Debasa and Sam H. Carvajal Department ofsurgery
YALE JOURNAL OF BIOLOGY AND MEDICINE 67 (1994), pp. 145-151. Copyright 1995. All rights reserved. Vagal Regulation of Acid Secretion and Gastrin Release Haile T. Debasa and Sam H. Carvajal Department ofsurgery
HISTAMINE. Mr. D.Raju, M.pharm, Lecturer
 HISTAMINE Mr. D.Raju, M.pharm, Lecturer INTRODUCTION: Histamine is a biogenic amine, with a imidazoline ring and act as a neurotransmitter. It is involved in local immune responses as well as regulating
HISTAMINE Mr. D.Raju, M.pharm, Lecturer INTRODUCTION: Histamine is a biogenic amine, with a imidazoline ring and act as a neurotransmitter. It is involved in local immune responses as well as regulating
EFFECT OF HISTAMINE, BRADYKININ AND MORPHINE ON ADRENALINE RELEASE FROM RAT ADRENAL GLAND
 EFFECT OF HISTAMINE, BRADYKININ AND MORPHINE ON ADRENALINE RELEASE FROM RAT ADRENAL GLAND Toshio YOSHIZAKI Shionogi Research Laboratory, Shionogi & Co., Ltd., Fukushima-ku, Osaka, 553 Japan Accepted March
EFFECT OF HISTAMINE, BRADYKININ AND MORPHINE ON ADRENALINE RELEASE FROM RAT ADRENAL GLAND Toshio YOSHIZAKI Shionogi Research Laboratory, Shionogi & Co., Ltd., Fukushima-ku, Osaka, 553 Japan Accepted March
Why would fatty foods aggravate the patient s RUQ pain? What effect does cholecystokinin (CCK) have on gastric emptying?
 CASE 28 A 43-year-old woman presents to the emergency department with the acute onset of abdominal pain. Her pain is located to the right upper quadrant (RUQ) and radiates to the right shoulder. She reports
CASE 28 A 43-year-old woman presents to the emergency department with the acute onset of abdominal pain. Her pain is located to the right upper quadrant (RUQ) and radiates to the right shoulder. She reports
Department of Physiology, Okayama University Medical School
 The Japanese Journal of Physiology 15, pp.243-252, 1965 Department of Physiology, Okayama University Medical School BAYLISS and STARLING 1) and others 6, 7, 9, 12, 14, 15) have reported that the stimulation
The Japanese Journal of Physiology 15, pp.243-252, 1965 Department of Physiology, Okayama University Medical School BAYLISS and STARLING 1) and others 6, 7, 9, 12, 14, 15) have reported that the stimulation
THE DEPENDENCE OF EXOCRINE PANCREATIC SECRETION ON INSULIN IN SHEEP
 Quarterly Journal of Experimental Physiology (1984) 69, 35-39 3 5 Printed in Great Britain THE DEPENDENCE OF EXOCRINE PANCREATIC SECRETION ON INSULIN IN SHEEP STEFAN PIERZYNOWSKI AND W. BAREJ The Institute
Quarterly Journal of Experimental Physiology (1984) 69, 35-39 3 5 Printed in Great Britain THE DEPENDENCE OF EXOCRINE PANCREATIC SECRETION ON INSULIN IN SHEEP STEFAN PIERZYNOWSKI AND W. BAREJ The Institute
RESPONSES OF THE ISOLATED SYMPATHETIC NERVE-
 Brit. J. Pharmacol. (1961), 16, 188-194. RESPONSES OF THE ISOLATED SYMPATHETIC NERVE- DUCTUS DEFERENS PREPARATION OF THE GUINEA-PIG BY S. HUKOVIC From the Department of Pharmacology, Medical Faculty, University
Brit. J. Pharmacol. (1961), 16, 188-194. RESPONSES OF THE ISOLATED SYMPATHETIC NERVE- DUCTUS DEFERENS PREPARATION OF THE GUINEA-PIG BY S. HUKOVIC From the Department of Pharmacology, Medical Faculty, University
Autonomic Nervous System Dr. Ali Ebneshahidi
 Autonomic Nervous System Dr. Ali Ebneshahidi Nervous System Divisions of the nervous system The human nervous system consists of the central nervous System (CNS) and the Peripheral Nervous System (PNS).
Autonomic Nervous System Dr. Ali Ebneshahidi Nervous System Divisions of the nervous system The human nervous system consists of the central nervous System (CNS) and the Peripheral Nervous System (PNS).
by dialysis could be restored to their full activity by the addition of Laboratory and the Molteno Institute, Cambridge -(Received 22 August 1945)
 411 J. Physiol. (I946) I04, 4II-425 547.435-292:6I2.0I5.36:6I2.8 PROPERTIES AND DISTRIBUTION OF THE ENZYME SYSTEM WHICH SYNTHESIZES ACETYLCHOLINE IN NERVOUS TISSUE BY W. FELDBERG AND T. MANN, From the
411 J. Physiol. (I946) I04, 4II-425 547.435-292:6I2.0I5.36:6I2.8 PROPERTIES AND DISTRIBUTION OF THE ENZYME SYSTEM WHICH SYNTHESIZES ACETYLCHOLINE IN NERVOUS TISSUE BY W. FELDBERG AND T. MANN, From the
INHIBITION OF ANAPHYLACTIC SHOCK IN THE RAT BY ANTIHISTAMINES AND ASCORBIC ACID
 Br. J. Phannac. Chemother. (1966), 27, 249-255. INHIBITION OF ANAPHYLACTIC SHOCK IN THE RAT BY ANTIHISTAMINES AND ASCORBIC ACID BY W. DAWSON, M. S. STARR AND G. B. WEST From the Department of Pharmacology,
Br. J. Phannac. Chemother. (1966), 27, 249-255. INHIBITION OF ANAPHYLACTIC SHOCK IN THE RAT BY ANTIHISTAMINES AND ASCORBIC ACID BY W. DAWSON, M. S. STARR AND G. B. WEST From the Department of Pharmacology,
fumarase, which are all important in the respiratory activity ofmammalian
 J. Physiol. (1960), 152, pp. 99-107 99 With 6 text-flgures Printed in Great Britain EFFECT OF DENERVATION ON RESPIRATORY ENZYMES IN SALIVARY GLANDS BY I. NORDENFELT, P. OHLIN AND B. C. R. STROMBLAD From
J. Physiol. (1960), 152, pp. 99-107 99 With 6 text-flgures Printed in Great Britain EFFECT OF DENERVATION ON RESPIRATORY ENZYMES IN SALIVARY GLANDS BY I. NORDENFELT, P. OHLIN AND B. C. R. STROMBLAD From
STUDIES ON THE HISTIDINE-HISTAMINE RELATIONSHIP IN VIVO
 Br. J. Pharmac. Chemother. (1968), 32, 567-574. STUDIES ON THE HISTIDINE-HISTAMINE RELATIONSHIP IN VIVO BY MARGARET A. REILLY AND R. W. SCHAYER* From the Research Center, Rockland State Hospital, Orangeburg,
Br. J. Pharmac. Chemother. (1968), 32, 567-574. STUDIES ON THE HISTIDINE-HISTAMINE RELATIONSHIP IN VIVO BY MARGARET A. REILLY AND R. W. SCHAYER* From the Research Center, Rockland State Hospital, Orangeburg,
Our gut reactions to food or, gut reactions - to food
 Key concepts in Digestion. Our gut reactions to food or, gut reactions to food Prof. Barry Campbell Cellular & Molecular Physiology email: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl Swallowing
Key concepts in Digestion. Our gut reactions to food or, gut reactions to food Prof. Barry Campbell Cellular & Molecular Physiology email: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl Swallowing
OPERATIVE TREATMENT OF ULCER DISEASE
 Página 1 de 8 Copyright 2001 Lippincott Williams & Wilkins Greenfield, Lazar J., Mulholland, Michael W., Oldham, Keith T., Zelenock, Gerald B., Lillemoe, Keith D. Surgery: Scientific Principles & Practice,
Página 1 de 8 Copyright 2001 Lippincott Williams & Wilkins Greenfield, Lazar J., Mulholland, Michael W., Oldham, Keith T., Zelenock, Gerald B., Lillemoe, Keith D. Surgery: Scientific Principles & Practice,
Interrelationship between Angiotensin Catecholamines. Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D.
 Interrelationship between Angiotensin and Catecholamines Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D. SUMMARY Urinary catecholamines were measured with an attempt
Interrelationship between Angiotensin and Catecholamines Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D. SUMMARY Urinary catecholamines were measured with an attempt
EVIDENCE THAT TRANQUILIZING ACTION OF RESERPINE IS ASSOCIATED WITH CHANGE IN BRAIN SEROTONIN AND NOT IN BRAIN NOREPINEPHRINE
 835 EVIDENCE THAT TRANQUILIZING ACTION OF RESERPINE IS ASSOCIATED WITH CHANGE IN BRAIN SEROTONIN AND NOT IN BRAIN NOREPINEPHRINE EFFECTS OF RESERPINE, RAUNESCI NE, BENZOQUI NOLIZI NES, DIMETHYL- AMINOBENZOYL
835 EVIDENCE THAT TRANQUILIZING ACTION OF RESERPINE IS ASSOCIATED WITH CHANGE IN BRAIN SEROTONIN AND NOT IN BRAIN NOREPINEPHRINE EFFECTS OF RESERPINE, RAUNESCI NE, BENZOQUI NOLIZI NES, DIMETHYL- AMINOBENZOYL
INVESTIGATION OF ADRENALINE REVERSAL IN THE RAT UTERUS BY THE INDUCTION OF RESISTANCE TO ISOPRENALINE
 Br. J. Pharmac. Chemother. (1967), 29, 291-301. INVESTIGATION OF ADRENALINE REVERSAL IN THE RAT UTERUS BY THE INDUCTION OF RESISTANCE TO ISOPRENALINE BY From the Department of Pharmacology, Guy's Hospital
Br. J. Pharmac. Chemother. (1967), 29, 291-301. INVESTIGATION OF ADRENALINE REVERSAL IN THE RAT UTERUS BY THE INDUCTION OF RESISTANCE TO ISOPRENALINE BY From the Department of Pharmacology, Guy's Hospital
(Received 5 November 1963) rabbit were 65 and 80 mm Hg, respectively. The mean arterial blood
 J. Phy8iol. (1964), 174, pp. 136-171 163 With 5 text-figure8 Printed in Great Britain AORTIC BARORCPTOR THRSHOLD AND SNSITIVITY IN RABBITS AT DIFFRNT AGS BY C. M. BLOOR* From the Nuffield Institute for
J. Phy8iol. (1964), 174, pp. 136-171 163 With 5 text-figure8 Printed in Great Britain AORTIC BARORCPTOR THRSHOLD AND SNSITIVITY IN RABBITS AT DIFFRNT AGS BY C. M. BLOOR* From the Nuffield Institute for
auriculo-temporal nerve. The secretory response to various cholinesterase
 Quarterly Journal of Experimental Phy8iology (1974) 59, 11-17 THE SECRETORY INNERVATION OF THE PAROTID GLAND OF THE CAT: AN UNEXPECTED COMPONENT. By J. EKSTROM and N. EMMELIN. From the Institute of Physiology,
Quarterly Journal of Experimental Phy8iology (1974) 59, 11-17 THE SECRETORY INNERVATION OF THE PAROTID GLAND OF THE CAT: AN UNEXPECTED COMPONENT. By J. EKSTROM and N. EMMELIN. From the Institute of Physiology,
