Vesicular Demyelination in MS A Pattern of Humoral Immune Pathology Multiple Sclerosis: B-Cells Take Center Stage
|
|
|
- Raymond Patrick
- 6 years ago
- Views:
Transcription
1 Vesicular Demyelination in MS A Pattern of Humoral Immune Pathology Multiple Sclerosis: B-Cells Take Center Stage Stephen L. Hauser, M.D. CD2 B cells in an MS lesion CD2 B cells in an MS lesion 1
2 CD138 plasma cells in an MS lesion B Cells in the Pathogenesis of MS Possible Roles: Antigen uptake, processing and presentation to T cells (APC) Activation of dendritic cells, microglia Co-stimulation of T cells Recruitment of cells to inflammatory sites Demyelination, myelin opsonization by Ab +/- C Site of Epstein-Barr virus persistence Enhance remyelination* T cell anergy* Regulation by anti-idiotypic Abs* * Potentially beneficial B-Cell Development Short-lived PC (secrete IgM) Death Long-lived PC Anti-CD2 Monoclonal Antibody is a genetically engineered chimeric (mouse-human) monoclonal antibody that targets CD2-positive B lymphocytes Ag APC T cells Ag CD2 is present on B and pre-b lymphocytes but not on stem cells or plasma cells Naïve B cell Ag = antigen APC = antigen-presenting cells PC = plasma cell Activated B cell Germinal center B cell Memory B cell Affinity maturation of B-cell receptor Long duration of action FDA approval for B-cell lymphoma (1997) and RA (26) Maloney DG, Grillo-López AJ, Bodkin D, Ahmed R et al. Immunology of Infectious Diseases; 22:
3 Phase II RRMS Study Design 1 Treatment Cycle and 9 MRIs Over 1Year Screen (4 wks) MRI Week / Infusion Wk 4 MRI to evaluate safety Treatment period (48 wks) Primary Endpoint Total number of Gd-enhancing lesions at Wks 12, 16, 2, and 24 Secondary Endpoints Proportion of patients relapsing Wks -24 New Gd-enhancing lesions T2 lesion volume changes B/L-Wk 24 2:1 Randomization stratified by site, EDSS ( 2.5, >2.5), and prior therapy (none or stopped >6 months or 6 months) Hauser et al. NEJM, in press 28 Primary Endpoint Total New Gd Lesion Count at Weeks 12, 16, 2 and 24 (ITT population) Total new Gd lesion count >-1 >1-2 >2-3 >3 Mean (SD) Minimum, Maximum 19 (54.3%) 3 (8.6%) 5 (14.3%) 1 (2.9%) 7 (2.%) 4.5 (12.58).., 68.7 (n=65) 54 (83.1%) 8 (12.3%) 3 (4.6%).16 (.43).., 2. <.1 1 *The p-value is based on Van Elteren test stratified by EDSS (<2.5, >2.5), prior therapy (<6 months, >6 months), and baseline gad (, >1) Gadolinium-Enhancing Lesions in Each Treatment Group from Baseline to Week 48 Panel B shows the number of new gadolinium-enhancing lesions by week. Missing values were imputed by an average of the available data. Panel A shows the mean total number of gadolinium enhancing lesions by week. Hauser, et. al. NEJM: Awaiting Publication Proportion of Subjects with Relapses During 24, 36 and 48 Weeks Proportion 1 24 Weeks Relative Risk 2 9% CI for RR 3 Proportion 1 Relative Risk 2 9% CI for RR 3 Proportion 1 36 Weeks 48 Weeks Relative Risk 2 9% CI for RR 3 12 (34.3)% 14 (4.)% 14 (4.)% (n=69) 1 (14.5%) 2.31 (1.25,4.26) (n=69) 12 (17.4%) 2.24 (1.3,3.88) (n=69) 14 (2.3%) 1.9 (1.3,3.88) (n=14).238 (n=14).145 (n=14).37 *The p-value is based on CMH test stratified for EDSS and prior treatment 1 The patients who discontinued before analysis are considered relapse-free if they did not experience any relapse during their study period. 2 Relative Risk of having relapse comparing to placebo, 3 9% CI are based on logit-adjusted 3
4 Change in T2 Lesion Volume T2 Lesion Volume at Baseline, mm 3 Mean ± SD Mean ± SD 432 T2 Lesion Volume Change from Baseline to Week 24**, mm ± ± 1188 T2 Lesion Volume Change from Baseline to Week 36**, mm 3 Mean ± SD (N=35) 5723 ± ± (N=68) 6452 ± ± P- Value* *Based on ANCOVA (ranked data), adjusted for baseline T2 lesion volume and stratification factors of EDSS ( 2.5, >2.5) and prior therapy (none or >6 months, 6 months) **Last observation carried forward median peripheral CD19+ B cell count CD19 Counts weeks placebo Ritxuan Effect of on Ig Levels HACA (Human Anti-Chimeric Antibodies) Relationship to Primary Outcome Values Below LLN at Week 48 (% at 24wks) Week Week Week 24 Week 48 Rituxan HACA + /69 (%) /62 (%) 14/58 (24 %) IgG IgM IgA (3.1%) (8.8%) 9.8% (6.3%) 21.6% (13.4%) Outcome Gad Mean (SD) Proportion relapse-free at week 48 HACA+ (N=14).5 (.85) 12(85.7%) HACA- (N=55).53 (2.2) 43(78.2%) 9% CI (7%, 1%) (69%, 87%) There was no apparent relationship between the HACA data and these outcomes 4
5 AEs within 24 Hours of Infusion (Infusion-Associated AEs) Drug-Related* AEs 1% (N=35) (N=69) 1 Any Drug-Related AE 17 (48.6%) 53 (76.8%) % patients st Infusion N=35 N=69 N=35 N=67 2nd Infusion No glucocorticoids given prior to infusion Infusion AEs markedly decreased with 2nd infusion One serious AE (pyrexia) was reported with first rituximab infusion Chills Headache Nausea Pruritus Pyrexia Fatigue Throat Irritation Pharyngolaryngeal Pain *As assessed by investigator 7 (2%) 4 (11.4%) 2 (5.7%) 14 (2.3%) 13 (18.8%) 12 (17.4%) 11 (15.9%) 9 (13.%) 8 (11.6%) 8 (11.6%) 7 (1.1%) Safety Summary Most AEs were mild to moderate in severity (Grade 1-2); none were serious Infusion-associated AEs decreased with each infusion and were mild to moderate in severity Reported infections were mild to moderate and were comparable between the rituximab and placebo groups Nearly one-quarter of patients became HACA-positive at 48 weeks (higher than in RA) Conclusions In RRMS, treatment with rituximab (2 x 1 mg) led to significantly (P<.1) fewer inflammatory brain lesions over 6 months compared with the placebo group a 91% relative reduction The proportion of patients with relapses during 24 weeks was also significantly reduced in the rituximab-treated group compared with the placebo group (P=.238) a 58% relative reduction Patients receiving rituximab showed a significant (P=.77) reduction from baseline to Week 24 in T2 lesion volume compared with placebo group was generally safe and well tolerated These data provide proof of principle that B cells play a role in the pathophysiology of RRMS 5
6 Phase I RRMS Study 2 Cycles of MRI: Study Drug: Week: -2 to Screen (4 weeks) Endpoint Primary Secondary Treatment period (72 wks) Study Outcome Measures Safety through Wk Frequency of relapse 2. MRI evaluation of activity (descriptive) Bar-Or et al. Ann Neurol, in press 28 Phase 1 RRMS Study Mean Total Gd Lesions at Each Timepoint Mean Gd-Enhanced Lesion Count Treatment 1 (Wk + 2) Retreatment (Wks ) Weeks (N=26) in Neuromyelitis Optica Pre- and Post-Treatment Attack Rate Annual Attack Rate Pre- p =.78 Time Of Follow-up = 12 months Post- Phase II/III PPMS Study Design 4 Cycles of and 5 MRIs over 126 Weeks Cree et al. Neurol 64:127, 25 6
7 Progressive Multifocal Leukoencephalopathy (PML) Cases of PML have been reported in patients with lymphoid malignancies and in patients with autoimmune diseases including SLE and vascultitis, during or up to one year after treatment with rituximab No cases of PML have been reported in MS patients exposed to rituximab either within or outside of the rituximab clinical development program The UCSF MS Group 7
Biologics and Beyond: Treatment of Multiple Sclerosis. Rita Jebrin, PharmD, BCPS
 Biologics and Beyond: Treatment of Multiple Sclerosis Rita Jebrin, PharmD, BCPS Disclosure Information Biologics and Beyond: Treatment of Multiple Sclerosis Rita Jebrin, PharmD, BCPS I have no financial
Biologics and Beyond: Treatment of Multiple Sclerosis Rita Jebrin, PharmD, BCPS Disclosure Information Biologics and Beyond: Treatment of Multiple Sclerosis Rita Jebrin, PharmD, BCPS I have no financial
Mitzi Joi Williams, MD Neurologist MS Center of Atlanta Atlanta, GA
 Mitzi Joi Williams, MD Neurologist MS Center of Atlanta Atlanta, GA Disclosures Consultant and Speaker Bureau member for Biogen-Idec, Pfizer, TEVA Neuroscience, Bayer, EMD Serrono, Questcor, Novartis,
Mitzi Joi Williams, MD Neurologist MS Center of Atlanta Atlanta, GA Disclosures Consultant and Speaker Bureau member for Biogen-Idec, Pfizer, TEVA Neuroscience, Bayer, EMD Serrono, Questcor, Novartis,
Ocrevus. (ocrelizumab) New Product Slideshow
 Ocrevus (ocrelizumab) New Product Slideshow Introduction Brand name: Ocrevus Generic name: Ocrelizumab Pharmacological class: CD20-directed cytolytic monoclonal antibody Strength and Formulation: 30mg/mL;
Ocrevus (ocrelizumab) New Product Slideshow Introduction Brand name: Ocrevus Generic name: Ocrelizumab Pharmacological class: CD20-directed cytolytic monoclonal antibody Strength and Formulation: 30mg/mL;
GENENTECH AND BIOGEN IDEC RECEIVE A COMPLETE RESPONSE FROM FDA FOR EARLIER USE OF RITUXAN FOR RHEUMATOID ARTHRITIS
 NEWS RELEASE Genentech Contacts: Media / Advocacy: Megan Pace (650) 467-7334 Investor: Susan Morris (650) 225-6523 Karl Mahler 011 41 61 68785 03 Biogen Idec Contacts: Media: Amy Reilly (617) 914-6524
NEWS RELEASE Genentech Contacts: Media / Advocacy: Megan Pace (650) 467-7334 Investor: Susan Morris (650) 225-6523 Karl Mahler 011 41 61 68785 03 Biogen Idec Contacts: Media: Amy Reilly (617) 914-6524
Clinician s view of Benefit-Risk
 Clinician s view of Benefit-Risk Gordon Francis, MD Novartis, Clinical Development Clinician s View of Benefit-Risk: a need for reliable metrics A tale of 3 drugs Natalizumab MS Crohn s Disease Fingolimod
Clinician s view of Benefit-Risk Gordon Francis, MD Novartis, Clinical Development Clinician s View of Benefit-Risk: a need for reliable metrics A tale of 3 drugs Natalizumab MS Crohn s Disease Fingolimod
Mellen Center Approach to Ocrelizumab (Ocrevus)
 Mellen Center Approach to Ocrelizumab (Ocrevus) Q: What is mechanism of action of ocrelizumab? A; Ocrelizumab is a novel humanized monoclonal anti-cd20 antibody constructed with recombinant DNA techniques
Mellen Center Approach to Ocrelizumab (Ocrevus) Q: What is mechanism of action of ocrelizumab? A; Ocrelizumab is a novel humanized monoclonal anti-cd20 antibody constructed with recombinant DNA techniques
OCREVUS (ocrelizumab) Injection, for Intravenous Use
 OCREVUS (ocrelizumab) Injection, for Intravenous Use Heidi Crayton, MD Medical Director Multiple Sclerosis Center of Greater Washington Vienna, VA OCR/071916/0181(1) Disclaimer This program is presented
OCREVUS (ocrelizumab) Injection, for Intravenous Use Heidi Crayton, MD Medical Director Multiple Sclerosis Center of Greater Washington Vienna, VA OCR/071916/0181(1) Disclaimer This program is presented
Rapid and Robust B Cell Depletion in Preliminary Results of a Phase 2 Study of Ublituximab, Novel Glycoengineered Anti-CD20 Mab, RMS Patients
 Rapid and Robust B Cell Depletion in Preliminary Results of a Phase 2 Study of Ublituximab, Novel Glycoengineered Anti-CD20 Mab, RMS Patients Amy Lovett-Racke, PhD May 26, 2017 2 Slide 1 2 Go to slide
Rapid and Robust B Cell Depletion in Preliminary Results of a Phase 2 Study of Ublituximab, Novel Glycoengineered Anti-CD20 Mab, RMS Patients Amy Lovett-Racke, PhD May 26, 2017 2 Slide 1 2 Go to slide
Ocrevus (ocrelizumab)
 Ocrevus (ocrelizumab) Policy Number: 5.01.629 Last Review: 04/2018 Origination: 05/2017 Next Review: 04/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Ocrevus
Ocrevus (ocrelizumab) Policy Number: 5.01.629 Last Review: 04/2018 Origination: 05/2017 Next Review: 04/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Ocrevus
How the Innate Immune System Profiles Pathogens
 How the Innate Immune System Profiles Pathogens Receptors on macrophages, neutrophils, dendritic cells for bacteria and viruses Broad specificity - Two main groups of bacteria: gram positive, gram-negative
How the Innate Immune System Profiles Pathogens Receptors on macrophages, neutrophils, dendritic cells for bacteria and viruses Broad specificity - Two main groups of bacteria: gram positive, gram-negative
Get on with life, we ll see you. in 6 months. Living your life your way with MS
 Get on with life, we ll see you in 6 months. Living your life your way with MS If you re one of the 4000 New Zealanders affected by MS, ask your doctor or nurse if OCREVUS is right for you. Contents What
Get on with life, we ll see you in 6 months. Living your life your way with MS If you re one of the 4000 New Zealanders affected by MS, ask your doctor or nurse if OCREVUS is right for you. Contents What
Anisha Keshavan. Roland Henry, PhD. Proportion of Patients (%) Rituximab 2000mg (n=66) Placebo (n=35)
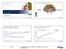 Two Faces of Multiple Sclerosis February 8, 217 5 T H A N N U A L R E C E N T A D V A N C E S I N N E U R O L O G Y B CELL THERAPY FOR MULTIPLE SCLEROSIS: A NEW DAY? Inflammation Relapsing MS Anisha Keshavan
Two Faces of Multiple Sclerosis February 8, 217 5 T H A N N U A L R E C E N T A D V A N C E S I N N E U R O L O G Y B CELL THERAPY FOR MULTIPLE SCLEROSIS: A NEW DAY? Inflammation Relapsing MS Anisha Keshavan
Arzerra. Arzerra (ofatumumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.03 Subject: Arzerra Page: 1 of 5 Last Review Date: June 22, 2017 Arzerra Description Arzerra (ofatumumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.03 Subject: Arzerra Page: 1 of 5 Last Review Date: June 22, 2017 Arzerra Description Arzerra (ofatumumab)
New Evidence reports on presentations given at ACR Improving Radiographic, Clinical, and Patient-Reported Outcomes with Rituximab
 New Evidence reports on presentations given at ACR 2009 Improving Radiographic, Clinical, and Patient-Reported Outcomes with Rituximab From ACR 2009: Rituximab Rituximab in combination with methotrexate
New Evidence reports on presentations given at ACR 2009 Improving Radiographic, Clinical, and Patient-Reported Outcomes with Rituximab From ACR 2009: Rituximab Rituximab in combination with methotrexate
Presented at the Annual Congress of ECTRIMS, October 11, 2018, Berlin, Germany
 Final Results of a Placebo Controlled, Phase 2 Multicenter Study of Ublituximab (UTX), a Novel Glycoengineered Anti-CD20 Monoclonal Antibody (mab), in Patients with Relapsing Forms of Multiple Sclerosis
Final Results of a Placebo Controlled, Phase 2 Multicenter Study of Ublituximab (UTX), a Novel Glycoengineered Anti-CD20 Monoclonal Antibody (mab), in Patients with Relapsing Forms of Multiple Sclerosis
Potential Rebalancing of the Immune System by Anti-CD52 Therapy
 Potential Rebalancing of the Immune System by Anti-CD52 Therapy Johanne Kaplan, PhD VP Neuroimmunology Research Genzyme March 26, 2013 RESTRICTED USE SEE TRAINING MEMO 2011 DO Genzyme NOT 1COPY Corporation
Potential Rebalancing of the Immune System by Anti-CD52 Therapy Johanne Kaplan, PhD VP Neuroimmunology Research Genzyme March 26, 2013 RESTRICTED USE SEE TRAINING MEMO 2011 DO Genzyme NOT 1COPY Corporation
OCREVUS TM (ocrelizumab) injection, for intravenous use Initial U.S. Approval: 2017
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OCREVUS safely and effectively. See full prescribing information for OCREVUS. OCREVUS TM (ocrelizumab)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OCREVUS safely and effectively. See full prescribing information for OCREVUS. OCREVUS TM (ocrelizumab)
Rituxan Hycela. Rituxan Hycela (rituximab and hyaluronidase human) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.96 Subject: Rituxan Hycela Page: 1 of 5 Last Review Date: September 15, 2017 Rituxan Hycela Description
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.96 Subject: Rituxan Hycela Page: 1 of 5 Last Review Date: September 15, 2017 Rituxan Hycela Description
Infusion Checklist for OCREVUS in relapsing or primary progressive forms of multiple sclerosis
 Infusion Checklist for OCREVUS in relapsing or primary progressive forms of multiple sclerosis 1 PREINFUSION COUNSELING Provide medication guide and review with your patient Inform patients about the signs
Infusion Checklist for OCREVUS in relapsing or primary progressive forms of multiple sclerosis 1 PREINFUSION COUNSELING Provide medication guide and review with your patient Inform patients about the signs
Local Natalizumab Treatment Protocol
 Local Natalizumab Treatment Protocol 1. New medicine name: Natalizumab 300mg concentrate for solution for infusion (Natalizumab ) 2. Licensed indication(s): Natalizumab is indicated for single disease
Local Natalizumab Treatment Protocol 1. New medicine name: Natalizumab 300mg concentrate for solution for infusion (Natalizumab ) 2. Licensed indication(s): Natalizumab is indicated for single disease
Immune checkpoint inhibitors in Hodgkin and non-hodgkin Lymphoma: How do they work? Where will we use them? Stephen M. Ansell, MD, PhD Mayo Clinic
 Immune checkpoint inhibitors in Hodgkin and non-hodgkin Lymphoma: How do they work? Where will we use them? Stephen M. Ansell, MD, PhD Mayo Clinic Conflicts of Interest Research Funding from Bristol Myers
Immune checkpoint inhibitors in Hodgkin and non-hodgkin Lymphoma: How do they work? Where will we use them? Stephen M. Ansell, MD, PhD Mayo Clinic Conflicts of Interest Research Funding from Bristol Myers
Corporate Medical Policy
 Corporate Medical Policy Rituximab for the Treatment of Rheumatoid Arthritis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: rituximab_for_the_treatment_of_rheumatoid_arthritis 4/2008
Corporate Medical Policy Rituximab for the Treatment of Rheumatoid Arthritis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: rituximab_for_the_treatment_of_rheumatoid_arthritis 4/2008
Gazyva. Gazyva (obinutuzumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.29 Subject: Gazyva Page: 1 of 7 Last Review Date: March 16, 2018 Gazyva Description Gazyva (obinutuzumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.29 Subject: Gazyva Page: 1 of 7 Last Review Date: March 16, 2018 Gazyva Description Gazyva (obinutuzumab)
Gazyva. Gazyva (obinutuzumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.29 Subject: Gazyva Page: 1 of 7 Last Review Date: September 15, 2016 Gazyva Description Gazyva (obinutuzumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.29 Subject: Gazyva Page: 1 of 7 Last Review Date: September 15, 2016 Gazyva Description Gazyva (obinutuzumab)
2.0 Synopsis. Adalimumab R&D/04/118. (For National Authority Use Only) Referring to Part of Dossier: Volume:
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
2.0 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
XP23829 PHASE 2 PSORIASIS TRIAL PRELIMINARY TOPLINE DATA PRESENTATION SEPTEMBER 15, 2015 COPYRIGHT 2015 XENOPORT, INC. ALL RIGHTS RESERVED.
 XP23829 PHASE 2 PSORIASIS TRIAL PRELIMINARY TOPLINE DATA PRESENTATION SEPTEMBER 15, 2015 COPYRIGHT 2015 XENOPORT, INC. ALL RIGHTS RESERVED. SAFE HARBOR DISCLAIMER These slides and the accompanying oral
XP23829 PHASE 2 PSORIASIS TRIAL PRELIMINARY TOPLINE DATA PRESENTATION SEPTEMBER 15, 2015 COPYRIGHT 2015 XENOPORT, INC. ALL RIGHTS RESERVED. SAFE HARBOR DISCLAIMER These slides and the accompanying oral
Treating MS patients earlier in the disease progression may affect long-term outcomes 1-4
 Treating MS patients earlier in the disease progression may affect long-term outcomes 1-4 TIME DISEASE ONSET EARLY TREATMENT NATURAL COURSE OF MS LATER TREATMENT DISABILITY INCREASE The disease activity
Treating MS patients earlier in the disease progression may affect long-term outcomes 1-4 TIME DISEASE ONSET EARLY TREATMENT NATURAL COURSE OF MS LATER TREATMENT DISABILITY INCREASE The disease activity
Hot Topics Multiple Sclerosis. Natalie Parks, MD, FRCPC Assistant Professor, Dalhousie University June 27, 2018
 Hot Topics Multiple Sclerosis Natalie Parks, MD, FRCPC Assistant Professor, Dalhousie University June 27, 2018 Disclosures Natalie Parks has received compensation from Biogen, EMD Serono, Roche, and Sanofi
Hot Topics Multiple Sclerosis Natalie Parks, MD, FRCPC Assistant Professor, Dalhousie University June 27, 2018 Disclosures Natalie Parks has received compensation from Biogen, EMD Serono, Roche, and Sanofi
Update in Multiple Sclerosis
 Update in Multiple Sclerosis Kyle Smoot, MD Providence MS Center Portland, OR 1 Disclosures Research funds from Biogen. Consulting fees from Acorda, Biogen, EMDSerono, Genzyme, Novartis, and Teva. 2 Outline
Update in Multiple Sclerosis Kyle Smoot, MD Providence MS Center Portland, OR 1 Disclosures Research funds from Biogen. Consulting fees from Acorda, Biogen, EMDSerono, Genzyme, Novartis, and Teva. 2 Outline
Soliris in NMOSD Phase 3 PREVENT Study Topline Results September 24, 2018
 Soliris in NMOSD Phase 3 PREVENT Study Topline Results September 24, 2018 Forward-Looking Statements This presentation contains forward-looking statements within the meaning of the Private Securities Litigation
Soliris in NMOSD Phase 3 PREVENT Study Topline Results September 24, 2018 Forward-Looking Statements This presentation contains forward-looking statements within the meaning of the Private Securities Litigation
Understanding Autoimmune Diseases: Evolving Issues. Alvina D. Chu, M.D. April 23, 2009
 Understanding Autoimmune Diseases: Evolving Issues Alvina D. Chu, M.D. April 23, 2009 Objectives Define the key pathogenic characteristics of: Type I diabetes mellitus Multiple sclerosis Rheumatoid arthritis
Understanding Autoimmune Diseases: Evolving Issues Alvina D. Chu, M.D. April 23, 2009 Objectives Define the key pathogenic characteristics of: Type I diabetes mellitus Multiple sclerosis Rheumatoid arthritis
Positive Phase III results for Roche s investigational medicine OCREVUS (ocrelizumab) published in New England Journal of Medicine
 Media Release Basel 21 December 2016 Positive Phase III results for Roche s investigational medicine OCREVUS (ocrelizumab) published in New England Journal of Medicine OCREVUS is the first and only investigational
Media Release Basel 21 December 2016 Positive Phase III results for Roche s investigational medicine OCREVUS (ocrelizumab) published in New England Journal of Medicine OCREVUS is the first and only investigational
Final Labeling Text
 1.14.2.3 Final Labeling Text HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Rituxan safely and effectively. See full prescribing information for
1.14.2.3 Final Labeling Text HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Rituxan safely and effectively. See full prescribing information for
B cell activation and antibody production. Abul K. Abbas UCSF
 1 B cell activation and antibody production Abul K. Abbas UCSF 2 Lecture outline B cell activation; the role of helper T cells in antibody production Therapeutic targeting of B cells 3 Principles of humoral
1 B cell activation and antibody production Abul K. Abbas UCSF 2 Lecture outline B cell activation; the role of helper T cells in antibody production Therapeutic targeting of B cells 3 Principles of humoral
Idelalisib in the Treatment of Chronic Lymphocytic Leukemia
 Idelalisib in the Treatment of Chronic Lymphocytic Leukemia Jacqueline C. Barrientos, MD Assistant Professor of Medicine Hofstra North Shore LIJ School of Medicine North Shore LIJ Cancer Institute CLL
Idelalisib in the Treatment of Chronic Lymphocytic Leukemia Jacqueline C. Barrientos, MD Assistant Professor of Medicine Hofstra North Shore LIJ School of Medicine North Shore LIJ Cancer Institute CLL
SUSPECTED MECHANISMS INVOLVED IN MS AND PUTATIVE INTERACTIONS WITH HEPATITIS B VACCINE IN MS
 SUSPECTED MECHANISMS INVOLVED IN MS AND PUTATIVE INTERACTIONS WITH HEPATITIS B VACCINE IN MS Emmanuelle Waubant, M.D. Olaf Stüve, M.D., PhD UCSF MS Center, San Francisco March 11, 2002 EPIDEMIOLOGY OF
SUSPECTED MECHANISMS INVOLVED IN MS AND PUTATIVE INTERACTIONS WITH HEPATITIS B VACCINE IN MS Emmanuelle Waubant, M.D. Olaf Stüve, M.D., PhD UCSF MS Center, San Francisco March 11, 2002 EPIDEMIOLOGY OF
They determine if there will be an immune response. Determine functions associated with immune response, but not specific to Ag.
 Appendices A They determine if there will be an immune response. Antigen receptor genes in T cells (TCR) and B cell (Ig) Determine functions associated with immune response, but not specific to Ag. MHC
Appendices A They determine if there will be an immune response. Antigen receptor genes in T cells (TCR) and B cell (Ig) Determine functions associated with immune response, but not specific to Ag. MHC
Demyelinating Diseases: Multiple Sclerosis January 10, 2018 Dr. Ostrow
 Demyelinating Diseases: Multiple Sclerosis January 10, 2018 Dr. Ostrow Reading: Robbins & Cotran, 9 th edition, pp 1283-1286 Robbins Basic Pathology, 9 th edition, 832-835 Overview: Grossly, myelin is
Demyelinating Diseases: Multiple Sclerosis January 10, 2018 Dr. Ostrow Reading: Robbins & Cotran, 9 th edition, pp 1283-1286 Robbins Basic Pathology, 9 th edition, 832-835 Overview: Grossly, myelin is
Progress in MS: Current and Emerging Therapies. Presented by: Dr. Kathryn Giles, MD MSc FRCPC Cambridge, Ontario, Canada
 Progress in MS: Current and Emerging Therapies Presented by: Dr. Kathryn Giles, MD MSc FRCPC Cambridge, Ontario, Canada Today s Discussion Natural History and Classification of MS Treating MS Management
Progress in MS: Current and Emerging Therapies Presented by: Dr. Kathryn Giles, MD MSc FRCPC Cambridge, Ontario, Canada Today s Discussion Natural History and Classification of MS Treating MS Management
European Commission Grants Approval for Mavenclad (Cladribine Tablets)
 Your Contact Friederike Segeberg +49 6151 72-6328 Investor Relations +49 6151 72-3321 August 25, 2017 European Commission Grants Approval for Mavenclad (Cladribine Tablets) First oral short-course treatment
Your Contact Friederike Segeberg +49 6151 72-6328 Investor Relations +49 6151 72-3321 August 25, 2017 European Commission Grants Approval for Mavenclad (Cladribine Tablets) First oral short-course treatment
Rituximab for the treatment of Immune (Idiopathic) Thrombocytopenic Purpura (ITP)
 Rituximab for the treatment of Immune (Idiopathic) Thrombocytopenic Purpura (ITP) Lead author: Stephen Erhorn Regional Drug & Therapeutics Centre (Newcastle) February 2015 2015 Summary Rituximab (MabThera,
Rituximab for the treatment of Immune (Idiopathic) Thrombocytopenic Purpura (ITP) Lead author: Stephen Erhorn Regional Drug & Therapeutics Centre (Newcastle) February 2015 2015 Summary Rituximab (MabThera,
BR is an established treatment regimen for CLL in the front-line and R/R settings
 Idelalisib plus bendamustine and rituximab (BR) is superior to BR alone in patients with relapsed/refractory CLL: Results of a phase III randomized double-blind placebo-controlled study Andrew D. Zelenetz,
Idelalisib plus bendamustine and rituximab (BR) is superior to BR alone in patients with relapsed/refractory CLL: Results of a phase III randomized double-blind placebo-controlled study Andrew D. Zelenetz,
TarGeting B-Cell Diseases
 TarGeting B-Cell Diseases Forward Looking Safe Harbor Statement This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These statements
TarGeting B-Cell Diseases Forward Looking Safe Harbor Statement This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These statements
Autologous Hematopoietic Stem Cell Transplantation for the Treatment of Neuromyelitis Optica in Singapore
 Case Reports 26 Autologous Hematopoietic Stem Cell Transplantation for the Treatment of Neuromyelitis Optica in Singapore Koh Yeow Hoay, Pavanni Ratnagopal Abstract Introduction: Neuromyelitis optica (NMO)
Case Reports 26 Autologous Hematopoietic Stem Cell Transplantation for the Treatment of Neuromyelitis Optica in Singapore Koh Yeow Hoay, Pavanni Ratnagopal Abstract Introduction: Neuromyelitis optica (NMO)
PATIENT INFORMATION: Patient Surname First Name Middle Initial Sex Date of Birth Alberta Personal Health Number M / F Year Month Day
 Applicant must be covered on an Alberta Government sponsored drug program. Page 1 of 6 PATIENT INFORMATION: Patient Surname First Name Middle Initial Sex Date of Birth Alberta Personal Health Number M
Applicant must be covered on an Alberta Government sponsored drug program. Page 1 of 6 PATIENT INFORMATION: Patient Surname First Name Middle Initial Sex Date of Birth Alberta Personal Health Number M
Edward Fox, MD, PhD Clinical Associate Professor, University of Texas Dell Medical School
 Phase 2 Multicenter Study Results of Ublituximab, a Novel Glycoengineered AntiCD20 Monoclonal Antibody (mab), in Patients with Relapsing Multiple Sclerosis (RMS) Edward Fox, MD, PhD Clinical Associate
Phase 2 Multicenter Study Results of Ublituximab, a Novel Glycoengineered AntiCD20 Monoclonal Antibody (mab), in Patients with Relapsing Multiple Sclerosis (RMS) Edward Fox, MD, PhD Clinical Associate
Autoimmune Diseases. Betsy Kirchner CNP The Cleveland Clinic
 Autoimmune Diseases Betsy Kirchner CNP The Cleveland Clinic Disclosures (financial) No relevant disclosures Learning Objectives Explain the pathophysiology of autoimmune disease Discuss safe administration
Autoimmune Diseases Betsy Kirchner CNP The Cleveland Clinic Disclosures (financial) No relevant disclosures Learning Objectives Explain the pathophysiology of autoimmune disease Discuss safe administration
Targeting B cells in relapsing remitting multiple sclerosis: from pathophysiology to optimal clinical management
 666741TAN0010.1177/1756285616666741Therapeutic Advances in Neurological DisordersS Bittner, T Ruck research-article2016 Therapeutic Advances in Neurological Disorders Review Targeting B cells in relapsing
666741TAN0010.1177/1756285616666741Therapeutic Advances in Neurological DisordersS Bittner, T Ruck research-article2016 Therapeutic Advances in Neurological Disorders Review Targeting B cells in relapsing
1996 vs 2013 MS Phenotype Descriptions of Progressive Disease
 Learning Objectives Upon completion, participants should be able to: Describe methods of distinguishing among RRMS, SPMS, and PPMS Incorporate available evidence about emerging and recently approved novel
Learning Objectives Upon completion, participants should be able to: Describe methods of distinguishing among RRMS, SPMS, and PPMS Incorporate available evidence about emerging and recently approved novel
Biologic Therapy for Inflammatory. Is Top-Down Too Top-Heavy? S. Devi Rampertab, MD, FACG, AGAF Associate Professor of Medicine University of Florida
 Biologic Therapy for Inflammatory Bowel Disease: Is Top-Down Too Top-Heavy? S. Devi Rampertab, MD, FACG, AGAF Associate Professor of Medicine University of Florida Learning Objectives Evaluate evidence
Biologic Therapy for Inflammatory Bowel Disease: Is Top-Down Too Top-Heavy? S. Devi Rampertab, MD, FACG, AGAF Associate Professor of Medicine University of Florida Learning Objectives Evaluate evidence
Defensive mechanisms include :
 Acquired Immunity Defensive mechanisms include : 1) Innate immunity (Natural or Non specific) 2) Acquired immunity (Adaptive or Specific) Cell-mediated immunity Humoral immunity Two mechanisms 1) Humoral
Acquired Immunity Defensive mechanisms include : 1) Innate immunity (Natural or Non specific) 2) Acquired immunity (Adaptive or Specific) Cell-mediated immunity Humoral immunity Two mechanisms 1) Humoral
Cost-effectiveness of cladribine (Mavenclad ) for the
 Cost-effectiveness of cladribine (Mavenclad ) for the treatment of adult patients with highly active relapsing multiple sclerosis (MS) as defined by clinical or imaging features The NCPE has issued a recommendation
Cost-effectiveness of cladribine (Mavenclad ) for the treatment of adult patients with highly active relapsing multiple sclerosis (MS) as defined by clinical or imaging features The NCPE has issued a recommendation
Neuroimmunology. Innervation of lymphoid organs. Neurotransmitters. Neuroendocrine hormones. Cytokines. Autoimmunity
 Neuroimmunology Innervation of lymphoid organs Neurotransmitters Neuroendocrine hormones Cytokines Autoimmunity CNS has two ways of contacting and regulating structures in the periphery Autonomic
Neuroimmunology Innervation of lymphoid organs Neurotransmitters Neuroendocrine hormones Cytokines Autoimmunity CNS has two ways of contacting and regulating structures in the periphery Autonomic
NEWS RELEASE Genentech Contacts: Media: Joe St. Martin (650) Investor: Karl Mahler Thomas Kudsk Larsen (973)
 NEWS RELEASE Genentech Contacts: Media: Joe St. Martin (650) 467-6800 Investor: Karl Mahler 011 41 61 687 8503 Thomas Kudsk Larsen (973) 235-3655 Biogen Idec Contacts: Media: Christina Chan (781) 464-3260
NEWS RELEASE Genentech Contacts: Media: Joe St. Martin (650) 467-6800 Investor: Karl Mahler 011 41 61 687 8503 Thomas Kudsk Larsen (973) 235-3655 Biogen Idec Contacts: Media: Christina Chan (781) 464-3260
Mucosal Immune System
 Exam Format 100 points - 60 pts mandatory; 40 points where 4, 10 point questions will be chosen Some open-ended questions, some short answer. Kuby question Cytokines Terminology How do cytokines achieve
Exam Format 100 points - 60 pts mandatory; 40 points where 4, 10 point questions will be chosen Some open-ended questions, some short answer. Kuby question Cytokines Terminology How do cytokines achieve
Chronic Lymphocytic Leukemia Update. Learning Objectives
 Chronic Lymphocytic Leukemia Update Ashley Morris Engemann, PharmD, BCOP, CPP Clinical Associate Adult Stem Cell Transplant Program Duke University Medical Center August 8, 2015 Learning Objectives Recommend
Chronic Lymphocytic Leukemia Update Ashley Morris Engemann, PharmD, BCOP, CPP Clinical Associate Adult Stem Cell Transplant Program Duke University Medical Center August 8, 2015 Learning Objectives Recommend
Post-natalizumab clinical and radiological findings in a cohort of multiple sclerosis patients: 12-month follow-up
 DOI 10.1007/s10072-013-1527-1 ORIGINAL ARTICLE Post-natalizumab clinical and radiological findings in a cohort of multiple sclerosis patients: 12-month follow-up Marta Melis Eleonora Cocco Jessica Frau
DOI 10.1007/s10072-013-1527-1 ORIGINAL ARTICLE Post-natalizumab clinical and radiological findings in a cohort of multiple sclerosis patients: 12-month follow-up Marta Melis Eleonora Cocco Jessica Frau
Inspiration for this talk. Introduction to Rituximab. Introduction to Rituximab (RTX) Introduction to Rituximab. Introduction to Rituximab
 It was the best of times, it was the worst of times The role of Rituximab in the treatment of Autoimmune Disease Inspiration for this talk Introduction to Rituximab (RTX) Chimeric anti-cd20 mab Approved
It was the best of times, it was the worst of times The role of Rituximab in the treatment of Autoimmune Disease Inspiration for this talk Introduction to Rituximab (RTX) Chimeric anti-cd20 mab Approved
Progressive Multifocal Leukoencephalopathy (PML)
 Progressive Multifocal Leukoencephalopathy (PML) By Jamie Meier Introduction: Progressive multifocal leukoencephalopathy (PML) occurs when change in an individual s immune status triggers reactivation
Progressive Multifocal Leukoencephalopathy (PML) By Jamie Meier Introduction: Progressive multifocal leukoencephalopathy (PML) occurs when change in an individual s immune status triggers reactivation
Advances in the Management of Multiple Sclerosis: A closer look at novel therapies. Disclosures
 Advances in the Management of Multiple Sclerosis: A closer look at novel therapies Lily Jung Henson, MD, MMM, FAAN Chief of Neurology Piedmont Healthcare, Atlanta, GA National Association of Managed Care
Advances in the Management of Multiple Sclerosis: A closer look at novel therapies Lily Jung Henson, MD, MMM, FAAN Chief of Neurology Piedmont Healthcare, Atlanta, GA National Association of Managed Care
CLINICAL MEDICAL POLICY
 CLINICAL MEDICAL POLICY Policy Name: Rituxan (rituximab) Policy Number: MP-031-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Issue Date: 11/01/2017
CLINICAL MEDICAL POLICY Policy Name: Rituxan (rituximab) Policy Number: MP-031-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Issue Date: 11/01/2017
BESPONSA (inotuzumab ozogamicin)
 BESPONSA (inotuzumab ozogamicin) Fact Sheet BESPONSA (inotuzumab ozogamicin) is an antibody-drug conjugate (ADC) composed of a monoclonal antibody (mab) targeting CD22, a cell surface antigen expressed
BESPONSA (inotuzumab ozogamicin) Fact Sheet BESPONSA (inotuzumab ozogamicin) is an antibody-drug conjugate (ADC) composed of a monoclonal antibody (mab) targeting CD22, a cell surface antigen expressed
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION
 PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION Pr OCREVUS TM Ocrelizumab for injection Concentrate for intravenous infusion 300 mg/10 ml (30 mg/ml) Selective Immunomodulator Treatment with
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION Pr OCREVUS TM Ocrelizumab for injection Concentrate for intravenous infusion 300 mg/10 ml (30 mg/ml) Selective Immunomodulator Treatment with
Research Article Safety and Efficacy of Rituximab in Multiple Sclerosis: A Retrospective Observational Study
 Journal of Immunology Research, Article ID 9084759, 9 pages https://doi.org/10.1155/2018/9084759 Research Article Safety and Efficacy of Rituximab in Multiple Sclerosis: A Retrospective Observational Study
Journal of Immunology Research, Article ID 9084759, 9 pages https://doi.org/10.1155/2018/9084759 Research Article Safety and Efficacy of Rituximab in Multiple Sclerosis: A Retrospective Observational Study
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: belimumab_benlysta 6/2011 2/2018 2/2019 3/2018 Description of Procedure or Service Belimumab (Benlysta) is
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: belimumab_benlysta 6/2011 2/2018 2/2019 3/2018 Description of Procedure or Service Belimumab (Benlysta) is
DOWNLOAD OR READ : THE PROGRESSIVE VIRUS PDF EBOOK EPUB MOBI
 DOWNLOAD OR READ : THE PROGRESSIVE VIRUS PDF EBOOK EPUB MOBI Page 1 Page 2 the progressive virus the progressive virus pdf the progressive virus Progressive Multifocal Leukoencephalopathy (PML) is an opportunistic
DOWNLOAD OR READ : THE PROGRESSIVE VIRUS PDF EBOOK EPUB MOBI Page 1 Page 2 the progressive virus the progressive virus pdf the progressive virus Progressive Multifocal Leukoencephalopathy (PML) is an opportunistic
Effector T Cells and
 1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
chapter 17: specific/adaptable defenses of the host: the immune response
 chapter 17: specific/adaptable defenses of the host: the immune response defense against infection & illness body defenses innate/ non-specific adaptable/ specific epithelium, fever, inflammation, complement,
chapter 17: specific/adaptable defenses of the host: the immune response defense against infection & illness body defenses innate/ non-specific adaptable/ specific epithelium, fever, inflammation, complement,
Introduction to Immune System
 Introduction to Immune System Learning outcome You will be able to understand, at a fundamental level, the STRUCTURES and FUNCTIONS of cell surface and soluble molecules involved in recognition of foreign
Introduction to Immune System Learning outcome You will be able to understand, at a fundamental level, the STRUCTURES and FUNCTIONS of cell surface and soluble molecules involved in recognition of foreign
Current Enrolling Clinical Trials
 ASSESS RRMS patients with active disease who are still able to walk. Mariko Kita MD Description of Study/Trial: A 12-month, randomized, rater- and dose-blinded study to compare the efficacy and safety
ASSESS RRMS patients with active disease who are still able to walk. Mariko Kita MD Description of Study/Trial: A 12-month, randomized, rater- and dose-blinded study to compare the efficacy and safety
Multiple Sclerosis Update
 Multiple Sclerosis Update Amanda Stahnke, PharmD, BCACP University of Missouri-Kansas City School of Pharmacy Kansas City Veterans Affairs Honor Annex Kelsey Morris, PharmD University of Kansas Health-System
Multiple Sclerosis Update Amanda Stahnke, PharmD, BCACP University of Missouri-Kansas City School of Pharmacy Kansas City Veterans Affairs Honor Annex Kelsey Morris, PharmD University of Kansas Health-System
RiTUXimab 375 mg/m 2 Therapy-7 day
 RiTUXimab 375 mg/m 2 Therapy-7 day This regimen supercedes NCCP Regimen 00208 rituximab 375mg/m2 therapy-follicular lymphoma as of February 2019 INDICATIONS FOR USE: Regimen *Reimbursement INDICATION ICD10
RiTUXimab 375 mg/m 2 Therapy-7 day This regimen supercedes NCCP Regimen 00208 rituximab 375mg/m2 therapy-follicular lymphoma as of February 2019 INDICATIONS FOR USE: Regimen *Reimbursement INDICATION ICD10
Current & Emerging Treatment & Novel Strategies in the Management of MS
 Current & Emerging Treatment & Novel Strategies in the Management of MS Dr Shiv Saidha, MD, MRCPI Assistant Professor of Neurology, Johns Hopkins University School of Medicine Disclosures Dr. Saidha receives
Current & Emerging Treatment & Novel Strategies in the Management of MS Dr Shiv Saidha, MD, MRCPI Assistant Professor of Neurology, Johns Hopkins University School of Medicine Disclosures Dr. Saidha receives
Is it CVID? Not Necessarily HAIG TCHEUREKDJIAN, MD
 Is it CVID? Not Necessarily HAIG TCHEUREKDJIAN, MD Current Paradigm of Pathogenesis Genetic defect(s) Molecular defect(s) Cellular defect(s) Clinical disease Current Paradigm of Pathogenesis Genetic defect(s)
Is it CVID? Not Necessarily HAIG TCHEUREKDJIAN, MD Current Paradigm of Pathogenesis Genetic defect(s) Molecular defect(s) Cellular defect(s) Clinical disease Current Paradigm of Pathogenesis Genetic defect(s)
USTEKINUMAB and BRIAKINUMAB
 USTEKINUMAB and BRIAKINUMAB Andrew Blauvelt, M.D. Professor, Dept. of Dermatology and Dept. of Molecular Microbiology & Immunology Oregon Health & Science University Chief, Dermatology Service Veteran
USTEKINUMAB and BRIAKINUMAB Andrew Blauvelt, M.D. Professor, Dept. of Dermatology and Dept. of Molecular Microbiology & Immunology Oregon Health & Science University Chief, Dermatology Service Veteran
Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas
 Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas Chapter 04: Antigen Recognition in the Adaptive Immune System Test Bank MULTIPLE CHOICE 1. Most T lymphocytes
Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas Chapter 04: Antigen Recognition in the Adaptive Immune System Test Bank MULTIPLE CHOICE 1. Most T lymphocytes
Synopsis (C1034T02) CNTO 95 Module 5.3 Clinical Study Report C1034T02
 Module 5.3 Protocol: EudraCT No.: 2004-002130-18 Title of the study: A Phase 1/2, Multi-Center, Blinded, Randomized, Controlled Study of the Safety and Efficacy of the Human Monoclonal Antibody to Human
Module 5.3 Protocol: EudraCT No.: 2004-002130-18 Title of the study: A Phase 1/2, Multi-Center, Blinded, Randomized, Controlled Study of the Safety and Efficacy of the Human Monoclonal Antibody to Human
Th1/Th17 Cytokine Dysregulation during Different Stages of Multiple Sclerosis
 Th1/Th17 Cytokine Dysregulation during Different Stages of Multiple Sclerosis Benjamin M. Segal, M.D. Holtom-Garrett Professor of Neurology Director, Multiple Sclerosis Program University of Michigan Disclosures
Th1/Th17 Cytokine Dysregulation during Different Stages of Multiple Sclerosis Benjamin M. Segal, M.D. Holtom-Garrett Professor of Neurology Director, Multiple Sclerosis Program University of Michigan Disclosures
Class Update with New Drug Evaluation: Disease-Modifying Drugs for Multiple Sclerosis
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Mon, Wed, Fri 11:00 AM-12:00 PM. Owen, Judy, Jenni Punt, and Sharon Stranford Kuby-Immunology, 7th. Edition. W.H. Freeman and Co., New York.
 Course Title: Course Number: Immunology Biol-341/541 Semester: Fall 2013 Location: HS 268 Time: Instructor: 8:00-9:30 AM Tue/Thur Dr. Colleen M. McDermott Office: Nursing Ed 101 (424-1217) E-mail*: mcdermot@uwosh.edu
Course Title: Course Number: Immunology Biol-341/541 Semester: Fall 2013 Location: HS 268 Time: Instructor: 8:00-9:30 AM Tue/Thur Dr. Colleen M. McDermott Office: Nursing Ed 101 (424-1217) E-mail*: mcdermot@uwosh.edu
Mechanisms of Autontibodies
 Mechanisms of Autontibodies Production in Rheumatic Diseases Eisa Salehi PhD Tehran University of Medical Sciences Immunology Department Introduction Rheumatic diseases: Cause inflammation, swelling, and
Mechanisms of Autontibodies Production in Rheumatic Diseases Eisa Salehi PhD Tehran University of Medical Sciences Immunology Department Introduction Rheumatic diseases: Cause inflammation, swelling, and
Investor Update. Downloads. Services PDF. Basel, 23 June 2017
 Investor Update Basel, 23 June 2017 FDA approves Rituxan Hycela (rituximab and hyaluronidase human) for subcutaneous injection in certain blood cancers Treatment can be administered in five to seven minutes,
Investor Update Basel, 23 June 2017 FDA approves Rituxan Hycela (rituximab and hyaluronidase human) for subcutaneous injection in certain blood cancers Treatment can be administered in five to seven minutes,
Biogen Idec Oncology Pipeline. Greg Reyes, MD, PhD SVP, Oncology Research & Development
 Biogen Idec Oncology Pipeline Greg Reyes, MD, PhD SVP, Oncology Research & Development March 25, 2009 Biogen Idec Strategy in Lymphoma / Leukemia CLL RITUXAN NHL FC-RITUXAN GA101 RITUXAN-CVP RITUXAN-CHOP
Biogen Idec Oncology Pipeline Greg Reyes, MD, PhD SVP, Oncology Research & Development March 25, 2009 Biogen Idec Strategy in Lymphoma / Leukemia CLL RITUXAN NHL FC-RITUXAN GA101 RITUXAN-CVP RITUXAN-CHOP
PEDIATRIC MULTIPLE SCLEROSIS CLINICAL AND TRANSCRIPTIONAL ASPECTS M U L T I P L E S C L E R O S I S C E N T E R
 PEDIATRIC MULTIPLE SCLEROSIS CLINICAL AND TRANSCRIPTIONAL ASPECTS M U L T I P L E S C L E R O S I S C E N T E R Gurevich M. PEDIATRIC MS Pediatric MS is defined as onset MS under the age of 18 years Pediatric
PEDIATRIC MULTIPLE SCLEROSIS CLINICAL AND TRANSCRIPTIONAL ASPECTS M U L T I P L E S C L E R O S I S C E N T E R Gurevich M. PEDIATRIC MS Pediatric MS is defined as onset MS under the age of 18 years Pediatric
Primary Immunodeficiency
 Primary Immunodeficiency DiGeorge Syndrome Severe Combined Immunodeficiency SCID X-Linked Agammaglobulinemia Common variable immunodeficiency (CVID) IgA deficiency Hyper- IgM Syndrome Wiskott-Aldrich syndrome
Primary Immunodeficiency DiGeorge Syndrome Severe Combined Immunodeficiency SCID X-Linked Agammaglobulinemia Common variable immunodeficiency (CVID) IgA deficiency Hyper- IgM Syndrome Wiskott-Aldrich syndrome
Roche data & results at EULAR 2006 Conference call Amsterdam, The Netherlands and Basel, Switzerland Friday, June 23, 2006
 We Innovate Healthcare 1 Roche data & results at EULAR 26 Conference call Amsterdam, The Netherlands and Basel, Switzerland Friday, June 23, 26 2 1 Forward-looking statements This presentation contains
We Innovate Healthcare 1 Roche data & results at EULAR 26 Conference call Amsterdam, The Netherlands and Basel, Switzerland Friday, June 23, 26 2 1 Forward-looking statements This presentation contains
News Release. March 29, 2019
 Your Contact Alice McGrail +1 781 738 8791 Investor Relations +49 6151 72 3321 March 29, 2019 FDA Approves MAVENCLAD (Cladribine) Tablets as First and Only Short-Course Oral Treatment for Relapsing-Remitting
Your Contact Alice McGrail +1 781 738 8791 Investor Relations +49 6151 72 3321 March 29, 2019 FDA Approves MAVENCLAD (Cladribine) Tablets as First and Only Short-Course Oral Treatment for Relapsing-Remitting
The Medical Letter. on Drugs and Therapeutics
 The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
Investor Presentation
 Investor Presentation September 12, 2018 Nasdaq: ATRA Forward-Looking Statements This presentation and the accompanying oral presentation contain forward-looking statements made pursuant to the safe harbor
Investor Presentation September 12, 2018 Nasdaq: ATRA Forward-Looking Statements This presentation and the accompanying oral presentation contain forward-looking statements made pursuant to the safe harbor
MCAT Biology - Problem Drill 16: The Lymphatic and Immune Systems
 MCAT Biology - Problem Drill 16: The Lymphatic and Immune Systems Question No. 1 of 10 1. Which of the following statements about pathogens is true? Question #01 (A) Both viruses and bacteria need to infect
MCAT Biology - Problem Drill 16: The Lymphatic and Immune Systems Question No. 1 of 10 1. Which of the following statements about pathogens is true? Question #01 (A) Both viruses and bacteria need to infect
Choices. Disease modifying treatments. Read me
 Choices Disease modifying treatments Read me Disease modifying treatments Disease modifying therapies (DMTs) are medications which modify the course of multiple sclerosis (MS) and are designed to reduce
Choices Disease modifying treatments Read me Disease modifying treatments Disease modifying therapies (DMTs) are medications which modify the course of multiple sclerosis (MS) and are designed to reduce
Autoimmunity and Primary Immune Deficiency
 Autoimmunity and Primary Immune Deficiency Mark Ballow, MD Division of Allergy & Immunology USF Morsani School of Medicine Johns Hopkins All Children s Hospital St Petersburg, FL The Immune System What
Autoimmunity and Primary Immune Deficiency Mark Ballow, MD Division of Allergy & Immunology USF Morsani School of Medicine Johns Hopkins All Children s Hospital St Petersburg, FL The Immune System What
Waldenstrom s Macroglobulinemia
 Waldenstrom s Macroglobulinemia : Monoclonal Antibodies Introduction Waldenstrom s macroglobulinemia (WM) is a lymphoma, or cancer of the lymphatic system. It occurs in a type of white blood cell called
Waldenstrom s Macroglobulinemia : Monoclonal Antibodies Introduction Waldenstrom s macroglobulinemia (WM) is a lymphoma, or cancer of the lymphatic system. It occurs in a type of white blood cell called
B-Cell Depletion with Rituximab in Relapsing Remitting Multiple Sclerosis
 T h e n e w e ng l a nd j o u r na l o f m e dic i n e original article B-Cell Depletion with in Relapsing Remitting Multiple Sclerosis Stephen L. Hauser, M.D., Emmanuelle Waubant, M.D., Ph.D., Douglas
T h e n e w e ng l a nd j o u r na l o f m e dic i n e original article B-Cell Depletion with in Relapsing Remitting Multiple Sclerosis Stephen L. Hauser, M.D., Emmanuelle Waubant, M.D., Ph.D., Douglas
Program Highlights. A multidisciplinary AAN working group identified areas for improvement in the diagnosis and management of patients with MS
 CasePerspectives: Illuminating Dark Pathways in Complex MS Cases Program Highlights Stephen Krieger, MD Associate Professor of Neurology Corinne Goldsmith Dickinson Center for MS Director, Neurology Residency
CasePerspectives: Illuminating Dark Pathways in Complex MS Cases Program Highlights Stephen Krieger, MD Associate Professor of Neurology Corinne Goldsmith Dickinson Center for MS Director, Neurology Residency
Efficacy and Safety of Belimumab in the treatment of Systemic Lupus Erythematosus: a Prospective Multicenter Study.
 1. Title Efficacy and Safety of Belimumab in the treatment of Systemic Lupus Erythematosus: a Prospective Multicenter Study. 2. Background Systemic Lupus Erythematosus (SLE) is a chronic, autoimmune and
1. Title Efficacy and Safety of Belimumab in the treatment of Systemic Lupus Erythematosus: a Prospective Multicenter Study. 2. Background Systemic Lupus Erythematosus (SLE) is a chronic, autoimmune and
Welcome to today s webinar: Learn about MS
 Welcome to today s webinar: Learn about MS Your Presenter is Dr Todd Hardy Your Facilitator is Belinda Saunders Housekeeping Thanks for joining us for this webinar welcome! You will be able to: hear the
Welcome to today s webinar: Learn about MS Your Presenter is Dr Todd Hardy Your Facilitator is Belinda Saunders Housekeeping Thanks for joining us for this webinar welcome! You will be able to: hear the
Multiple Sclerosis and Neuroinflammation: Considering Gender differences to design therapeutic agents HALINA OFFNER
 Multiple Sclerosis and Neuroinflammation: Considering Gender differences to design therapeutic agents HALINA OFFNER Sex differences in autoimmune disease Ratio Target Sex distribution of major autoimmune
Multiple Sclerosis and Neuroinflammation: Considering Gender differences to design therapeutic agents HALINA OFFNER Sex differences in autoimmune disease Ratio Target Sex distribution of major autoimmune
PRIMARY DISEASES OF MYELIN. By: Shifaa Al Qa qa
 PRIMARY DISEASES OF MYELIN By: Shifaa Al Qa qa Most diseases of myelin are primarily white matter disorders??? Myelinated axons most diseases of CNS myelin do not involve the peripheral nerves to any significant
PRIMARY DISEASES OF MYELIN By: Shifaa Al Qa qa Most diseases of myelin are primarily white matter disorders??? Myelinated axons most diseases of CNS myelin do not involve the peripheral nerves to any significant
