Journal of the COPD Foundation The COPD Pipeline XXXVI Nicholas Gross, MD, PhD 1
|
|
|
- Marshall Reynolds
- 6 years ago
- Views:
Transcription
1 320 COPD Pipeline Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation The COPD Pipeline XXXVI Nicholas Gross, MD, PhD 1 Abbreviations: chronic obstructive pulmonary disease, COPD; Food and Drug Administration, FDA; long-acting muscarinic antagonist, LAMA; forced expiratory volume in 1 second, FEV1; phosphodiesterase, PDE; epithelial sodium channel, ENaC; cystic fibrosis transmembrane conductance regulator, CFTR; acute respiratory distress syndrome, ARDS; acute exacerbations of COPD, AECOPD Citation: Gross N. The COPD pipeline XXXVI. Chronic Obstr Pulm Dis. 2017;4(4): doi: jcopdf University Medical Research, Saint Francis Hospital and Medical Center, Hartford, Connecticut Address correspondence to: Nicholas Gross, MD, PhD grossnicholas1@gmail.com Contract Pharma Contract Pharma is a publication that reviews, among other things, the pharmaceutical industry annually. Those of us who work in chronic obstructive pulmonary disease (COPD) take an interest in the source of the drugs we use and Contract Pharma brings that information to us. Each year they report the status of the top 25 pharmaceutical companies. 1 For each pharma, a report lists the newly approved drugs, those pending approval, drugs in phase 2, those in early research, drugs coming off patent, and some other less interesting data. There is usually a tussle between Pfizer and Novartis for the top. For the 2017 report (which is based on 2016 sales) Pfizer, Inc., is number 1. As I sift through the top 25 pharma companies, I look for drugs that might be the ones we will one day be prescribing for our patients with COPD. Pfizer has no new COPD drugs anywhere in the process of development unless one includes Prevnar 13 which was just approved. Next is Novartis which has Fevipiprant, an interesting CRTh2 antagonist in phase 1. But it is for an asthma indication. Merck, at number 3, has MK-1029, also a CRTh2 antagonist, but also in trials as a potential asthma therapy. Merck has no agents in development for COPD. Nor, going down the list, has Roche. GlaxoSmithKline has Relvar Elipta (recently approved), a triple for COPD that is pending the Food and Drug Administration s (FDA s) review, and mepolizumab for COPD. Next on the list are Sanofi, Johnson & Johnson, Gilead, and ABBVie with no COPD drugs. Then AstraZeneca with Bevespi/Aerosphere and benralizumab both in phase 2 for a COPD indication. Teva has several drugs for asthma but none for COPD. Next on the list are Lilly, Bristol-Myers Squibb, and Bayer. Finally, Boehringer Ingelheim has worked with Hamni Pharmaceuticals on HCP1202 for COPD. It is in a Phase 2 trial. Then, moving down the list, comes a desert with no potential COPD drugs, - Novo Nordisk, Merck KGAA, Takeda, Allergan, Biogen, Shire, and Celgene. Mylan brings the list to a close with its revefenacin, a nebulized long-acting muscarinic antagonist (LAMA) in phase 3. The point here is that the third largest cause of death in most of the world needs more novel therapies. Other than the me-too s, we have just 2 -lizumabs in the works for COPD. Cancer, heart, and diseases of ageing are all well represented in late stage development, which is appropriate. But COPD should be up there with them. Autologous bronchial basal cells transplantation is Autologous Bronchial Basal Cells Transplantation a possible treatment for chronic respiratory diseases including COPD and bronchiectasis. Regend Therapeutics has the novel and interesting idea that autologous bronchial cells can be obtained by bronchoscopy, expanded in vitro, and reinjected directly into lesions via bronchoscopy. Progress of the treatments will be followed for 6 months (but it is not clear how the donated cells will be identified). Two phase 1/2 trials will be conducted, NCT and NCT , 3 and the studies will be singlecentered, non-randomized, and conducted in 30 and 20 stable COPD patients, respectively. Primary outcomes will be forced expiratory volume in 1 second (FEV1) and diffusing capacity of the lungs for carbon monoxide.
2 321 COPD Pipeline GSK is a phosphoinositide-3 kinase delta GSK inhibitor that is being investigated for an antiinflammatory action in COPD. A recently completed phase 1 study enrolled 12 healthy participants who received the agent by dry powder inhalation. The primary outcomes, apart from the usual pharmacokinetic and safety outcomes, were a range of lung function measurements over the following 6 days (results pending) (NCT ). 4 RPL554 RPL554 is a dual phosphodiesterase (PDE) 3/4 inhibitor nebulized for acute exacerbations of COPD. The molecule displays simultaneously bronchodilator and anti-inflammatory properties. In a recently completed trial, 40 individuals with COPD were entered in a phase 2 trial with 3 arms, namely 2 doses of RPL554 plus tiotropium and a control group that received only tiotropium. The primary outcome was peak FEV1 on day 3 of treatment (NCT ) (results pending). 5 Colistimethate Sodium Colistimethate sodium, also known as polymixin E, is a well-known antibiotic that is effective against most gram-negative bacilli. It fell out of use due to renal toxicity but remains a last resort against Pseudomonas aeruginosa, Klebsiella pneumoniae, and Acinetobacter. In a placebo-controlled trial including 264 participants, the primary outcome will be time to first exacerbation. 6 A Triple for COPD? A trial of a fixed combination of fluticasone furoate/ umeclidinium/vilanterol has been announced in clinicaltrials.gov (NCT ). 7 Previous developments of fixed combinations of 2 components have faced the FDA combination rule that the optimal dose of each component must be determined in the presence of the other component. The rationale has been that the optimal dose of a drug cannot be assumed to be the same in the presence of another drug as it was in isolation. This rule often required trials of 8 or more arms to find acceptable doses of each component and often several such multi-armed trials were needed. The costs of these trials often went into 7 figures. The mathematics of a triple required a logarithmic increase in the number of arms each trial would require--a daunting task. Which is why molecules with 2 actions, e.g., an antimuscarinic moiety and an adrenergic moiety, are being developed as single agents by some pharmaceutical companies. (That tactic has brought its own problems including the finding that it has been difficult to fashion a hybrid molecule in which the 2 moieties have significant and roughly equal potencies). The NCT trial seems to suggest either that the large number of arms and trials will be undertaken or that the FDA rule has been relaxed. The present study, by GlaxoSmithKline, will put all 3 components, beta-agonist, anticholinergic, and corticosteroid, on the same molecule. It is referred to as a single inhaler triple. Enrollment will be a single participant. Yes, 1 patient. The dual primary outcomes will be evaluation of the key relevant attributes of COPD treatment (time frame: up to 368 hours) and evaluation of the preferences, priorities and treatment goals of subjects with COPD for inhaled treatments. 7 Acumapimod Acumapimod (BCT-197) is an oral p38 MAP kinase inhibitor that is being developed as a therapy for acute exacerbations of COPD. Previous studies undertaken by Novartis, showed the drug has the capacity to reduce the inflammatory marker TNFα and to increase FEV1. Previous studies showed the drug to be well tolerated in the target population. The aim is to shorten the duration of acute exacerbations of COPD, something that would be novel. A placebo-controlled randomized study will enroll 255 individuals with COPD who will receive either the drug or placebo to standard of care for the treatment of an acute exacerbation in a phase 2 trial (NCT ). 8 Primary outcomes are safety and pharmacokinetics. Emera 003COPD Emera 003COPD is an antioxidant and metal chelator that is being developed by Emeramed for patients with COPD with bronchitis (NCT ). 9 The double-blind placebo controlled trial will be in phase 2. The only previous trial of this agents was as a treatment for mercury intoxication (NCT ). 10 I cannot find any other information about this agent.
3 322 COPD Pipeline AZD8871 AZD8871 is another bifunctional molecule with both anti-muscarinic and beta-adrenergic functions being developed for both asthma and COPD. It is in phase 1 and if successful will probably be married up with a corticosteroid to form, effectively, a triple inhalation for airways disorders that seems to have become the holy grail for inflammatory airways disorders. In addition to the present study (NCT ), 11 3 other trials are registered in clinicaltrials.gov. SPX-101 The epithelial sodium channel (ENaC) is responsible for reduced airway hydration causing mucus dehydration and decreased mucociliary clearance. 12 It is therefore a target for the management of cystic fibrosis. SPX-101 inhibits ENaC. It is an inhalation in a recently completed phase 1 trial (NCT ) 13 for 5 individuals with cystic fibrosis independent of their underlying cystic fibrosis transmembrane conductance regulator (CFTR) gene mutation. If successful, one wonders whether it might be of interest in COPD. IC14 IC14 is an anti-cd14 antibody that blocks TNFα production. 14 (It was first published in the early 2000 s as an agent that might address the problem of inflammation in severe sepsis. After a flurry of about 6 or 7 publications, it seems that trials on its potential to address sepsis were disappointing. A further analysis of its potential is about to be renewed [NCT ]). 15 In a phase 2 study, 160 individuals with acute respiratory distress syndrome (ARDS) will be randomized to receive either the IC14 or placebo by intravenous injection in daily doses for 4 days. The primary outcome will be safety, secondary outcomes will be changes in ARDS markers, e.g., IL-8, stnfr1, and IL-6. ZL-2102 ZL-2102 is a novel agent from China. The drug is in phase 1 in a double-blind, placebo-controlled, randomized trial. None of the other trials of the same agent, nor its sponsor, Zai Lab Pty Ltd, provide any information about the action of ZL There are no citations of ZL-2102 in Pub Med. However, the drug s sponsor claims that it aims to transform patients lives rather than simply relieve symptoms, (more or less exactly what we need). Asthma and COPD are stated targets in a 14-day, 3-stage trial involving 104 normal healthy participants (NCT ). 16 Viral and Bacterial Pathogens in the Respiratory Tract A new study will examine the occurrence of potential bacterial and viral pathogens in both stable and acute exacerbations of COPD in several Asian Pacific regions (NCT ). 17 It has been suggested that the infectious etiology of acute exacerbations of COPD (AECOPD) varies according to geographical region: the primary purpose of this study (which will be conducted in several countries in Asia Pacific) is to evaluate the occurrence of bacterial and viral pathogens in the sputum of stable COPD patients and at the time of AECOPD. Given the increasing and projected burden of COPD in the Asian Pacific region, this study will also evaluate the frequency, severity and duration of AECOPD, as well as the impact of AECOPD on healthrelated quality of life, health care utilization and lung function, as stated on ClinicalTrials.gov. 17 Yet Another LAMA/LABA Just when you thought we had enough doubles, there is another. However, with at least 3 LAMAs and 5 or 6 longacting beta2-agonists and several delivery pathways, and durations of actions, etc, one may compute 100 possibilities for essentially the same treatment. They are or will all be safe and effective. You have seen them all advertised and detailed to you and have yourself detailed them to your colleagues and this column is not about things you already know very well. Chaperones for Cystic Fibrosis It is estimated that about 30% of cases of cystic fibrosis are due to protein misfolding due in turn to genetic abnormalities. The misfolded nascent proteins are, for one reason or another, unable to do their fluid and electrolyte duties at the cell surface. Chaperone proteins are proteins that promote the correct folding and assembly of other proteins. It is postulated that molecules can be synthesized that correct misfolded proteins in a chaperone manner. The pharmaceutical
4 323 COPD Pipeline company Vertex has obtained FDA approval for 2 drugs that can perform that function in cystic fibrosis: ivacaftor (Kalydeco) and a fixed combination of lumacaftor and ivacaftor (Orkambi). The same company has at least 4 other studies in phase 2 of fixed combinations of, essentially, triple combinations of either VX-659, or VX-440, or VX-445, or VX-152 in combination with a tezacaftor/ivacaftor combination. 18 The concept is that each agent (chaperone) has its own effect and improves the overall outcome. Acknowledgement Special thanks to Johan Karlberg, MD, PhD, of Clinical Trial Magnifier newsletter, for much of the information in this report.
5 324 COPD Pipeline References Wright T, Brooks K. Top 25 pharma and biopharma report. Contract Pharma July. heaps/view/3659/1 Published July 27, Accessed October National Institutes of Health. Autologous bronchial basal cells transplantation for treatment of CRD including COPD, BE and PF. Clinicaltrials.gov website. show/nct Published May Accessed October National Institutes of Health. Autologous bronchial basal cell transplantation for treatment of COPD. ClinicalTrials.gov website. Published June Accessed National Institutes of Health. A study to evaluate the safety, tolerability and pharmacokinetics of single and repeat doses of the dry powder formulation of GSK in healthy subjects. ClinicalTrials.gov website. NCT Published January 8, Updated July 13, Accessed 5. National Institutes of Health. The effects of RPL554 in addition to tiotropium in COPD patients.clinicaltrials.gov website clinicaltrials.gov/ct2/show/nct Published January 23, Updated September 12, Accessed Vardakas KZ, et al. Inhaled colistin monotherapy for respiratory tract infections in adults without cystic fibrosis: A systematic review and meta-analysis. J Antimicrob Agents. In press. doi: National Institutes of Health. FF/UMEC/VI Inhaler: Qualitative analysis and subject preference survey. ClinicalTrials.gov website. Published March Updated September Accessed National Institutes of Health. Efficacy and safety of BCT197 in subjects with acute respiratory exacerbations of chronic obstructive pulmonary disease. ClinicalTrials.gov clinicaltrials.gov/ct2/show/nct Updated June Accessed October ber 11. National Institutes of Health. Two-part safety, tolerability, pharmacodynamic and -kinetic study of inhaled azd8871 in asthmatic and COPD subjects. ClinicalTrials.gov clinicaltrials.gov/ct2/show/nct Published October Updated September Accessed Scott DW, Walker MP, Sesma J, et al. SPX-101 is a novel ENaCtargeted therapeutic for cystic fibrosis that restores mucus transport. Am J Respir Crit Care Med. 2017;196(6): doi: doi.org/ /rccm oc. National Institutes of Health. A safety study of SPX-101 inhalation solution in subjects with cystic fibrosis. ClinicalTrials. gov website. Published February 17, Updated September Accessed Genth-Zotz S, Van Haehling S, Bolger AP, et al. The anti-cd14 antibody IC14 suppresses ex vivo endotoxin stimulated tumor necrosis factor-alpha in patients with chronic heart failure. Eur J Heart Fail. 2006;8(4): National Institutes of Health. A phase 2 study of IC14 in acute respiratory distress syndrome. ClinicalTrials.gov website. Published January Accessed 16. National Institutes of Health. Study of the tolerability and pharmacokinetic of ZL-2102 with an investigation of food effect in healthy male subjects. ClinicalTrials.gov website. clinicaltrials.gov/ct2/show/nct Published March Updated June Accessed 17. National Institutes of Health. Occurrence of potential bacterial and viral pathogens in stable chronic obstructive pulmonary disease and during acute exacerbations of the disease, in Asia Pacific. ClinicalTrials.gov website. show/nct Published May Updated September Accessed October Vertex Pharmaceuticals. Research and pipeline: Cystic fibrosis. Vertex website. stigational-medicines-pipeline Published March Accessed 9. National Institutes of Health. Clinical study on COPD (Emera003COPD). ClinicalTrials.gov gov/ct2/show/nct Published April Accessed 10. National Institutes of Health. Pilot proof of concept study of NBMI treatment of mercury intoxication (NBMI). ClinialTrials. gov website. Published July 1, Updated April 7, Accessed October
Journal of the COPD Foundation. The COPD Pipeline XXXIII Nicholas Gross, MD, PhD 1. Chronic Obstructive Pulmonary Diseases:
 65 COPD Pipeline Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation The COPD Pipeline XXXIII Nicholas Gross, MD, PhD 1 Abbreviations: chronic obstructive pulmonary disease, COPD; phosphoinositide
65 COPD Pipeline Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation The COPD Pipeline XXXIII Nicholas Gross, MD, PhD 1 Abbreviations: chronic obstructive pulmonary disease, COPD; phosphoinositide
Journal of the COPD Foundation The COPD Pipeline XXXVII Nicholas Gross, MD, PhD 1
 66 COPD Pipeline Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation The COPD Pipeline XXXVII Nicholas Gross, MD, PhD 1 Abbreviations: chronic obstructive pulmonary disease, COPD; neutrophil
66 COPD Pipeline Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation The COPD Pipeline XXXVII Nicholas Gross, MD, PhD 1 Abbreviations: chronic obstructive pulmonary disease, COPD; neutrophil
Breathtaking science. Developing respiratory drugs to improve health and quality of life. H.C. Wainwright Global Life Sciences Conference April 2018
 Breathtaking science Developing respiratory drugs to improve health and quality of life H.C. Wainwright Global Life Sciences Conference April 2018 www.veronapharma.com Forward-Looking Statements This presentation
Breathtaking science Developing respiratory drugs to improve health and quality of life H.C. Wainwright Global Life Sciences Conference April 2018 www.veronapharma.com Forward-Looking Statements This presentation
Breathtaking science. Developing respiratory drugs to improve health and quality of life
 Breathtaking science Developing respiratory drugs to improve health and quality of life Wedbush PacGrow Healthcare Conference New York City, August 2018 NASDAQ VRNA www.veronapharma.com Forward-Looking
Breathtaking science Developing respiratory drugs to improve health and quality of life Wedbush PacGrow Healthcare Conference New York City, August 2018 NASDAQ VRNA www.veronapharma.com Forward-Looking
Cystic Fibrosis: Progress in Treatment Management. Patrick A. Flume, M.D. Medical University of South Carolina
 Cystic Fibrosis: Progress in Treatment Management Patrick A. Flume, M.D. Medical University of South Carolina Disclosures Grant support Mpex Pharmaceuticals, Inc Gilead Sciences, Inc Bayer Healthcare AG
Cystic Fibrosis: Progress in Treatment Management Patrick A. Flume, M.D. Medical University of South Carolina Disclosures Grant support Mpex Pharmaceuticals, Inc Gilead Sciences, Inc Bayer Healthcare AG
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Calverley P M A, Anzueto A R, Carter K, et
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Calverley P M A, Anzueto A R, Carter K, et
Surveillance report Published: 6 April 2016 nice.org.uk. NICE All rights reserved.
 Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Cystic Fibrosis Transmembrane Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) Prime Therapeutics
Cystic Fibrosis Transmembrane Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) Prime Therapeutics
National Horizon Scanning Centre. Mannitol dry powder for inhalation (Bronchitol) for cystic fibrosis. April 2008
 Mannitol dry powder for inhalation (Bronchitol) for cystic fibrosis April 2008 This technology summary is based on information available at the time of research and a limited literature search. It is not
Mannitol dry powder for inhalation (Bronchitol) for cystic fibrosis April 2008 This technology summary is based on information available at the time of research and a limited literature search. It is not
Breathtaking science. Developing respiratory drugs to improve health and quality of life
 Breathtaking science Developing respiratory drugs to improve health and quality of life June 2017 Forward-Looking Statements This presentation contains forward looking statements that are based on the
Breathtaking science Developing respiratory drugs to improve health and quality of life June 2017 Forward-Looking Statements This presentation contains forward looking statements that are based on the
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Cystic Fibrosis Transmembrane Page 1 of 11 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) Prime Therapeutics
Cystic Fibrosis Transmembrane Page 1 of 11 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) Prime Therapeutics
The Journal Club: COPD Exacerbations
 252 The Journal Club: COPD Exacerbations Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Journal Club The Journal Club: COPD Exacerbations Ron Balkissoon, MD, MSc, DIH, FRCPC 1 Abbreviations:
252 The Journal Club: COPD Exacerbations Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Journal Club The Journal Club: COPD Exacerbations Ron Balkissoon, MD, MSc, DIH, FRCPC 1 Abbreviations:
Transformational Treatments. PRESTON W. CAMPBELL, III, M.D. Executive Vice President for Medical Affairs
 Transformational Treatments PRESTON W. CAMPBELL, III, M.D. Executive Vice President for Medical Affairs Symptom-based CF Therapies 45 Median Predicted Survival Age of US Patients with Cystic Fibrosis 41
Transformational Treatments PRESTON W. CAMPBELL, III, M.D. Executive Vice President for Medical Affairs Symptom-based CF Therapies 45 Median Predicted Survival Age of US Patients with Cystic Fibrosis 41
#POMAD8 #ChoosePOMA #POMAD8 #ChoosePOMA #POMAD8 #ChoosePOMA
 Where There s Smoke There s Obstruction Stephen G. Basheda, D.O., F.C.C.P. 1 Disclosures Dr. Basheda is an independent contractor for AstraZeneca Pharmaceuticals, LP and GlaxoSmithKline and on the speaker
Where There s Smoke There s Obstruction Stephen G. Basheda, D.O., F.C.C.P. 1 Disclosures Dr. Basheda is an independent contractor for AstraZeneca Pharmaceuticals, LP and GlaxoSmithKline and on the speaker
Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984]
![Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984] Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984]](/thumbs/85/91889043.jpg) Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984] 1 st Appraisal Committee meeting Background & Clinical Effectiveness John McMurray 11 th January 2016 For
Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984] 1 st Appraisal Committee meeting Background & Clinical Effectiveness John McMurray 11 th January 2016 For
New and Novel Medications for Respiratory Care
 New and Novel Medications for Respiratory Care JASON MOORE, PHARM.D. BCCCP CLINICAL STAFF PHARMACIST STORMONT-VAIL HEALTH Objectives Quick overview of the newest FDA-approved repiratory-related medications
New and Novel Medications for Respiratory Care JASON MOORE, PHARM.D. BCCCP CLINICAL STAFF PHARMACIST STORMONT-VAIL HEALTH Objectives Quick overview of the newest FDA-approved repiratory-related medications
Verona Pharma Announces Positive Top-Line Data from Phase 2a Clinical Trial in COPD with RPL554 Dosed in Addition to Tiotropium (Spiriva )
 Press release 7 September 2017 Verona Pharma Announces Positive Top-Line Data from Phase 2a Clinical Trial in COPD with RPL554 Dosed in Addition to Tiotropium (Spiriva ) Achieved significant and clinically
Press release 7 September 2017 Verona Pharma Announces Positive Top-Line Data from Phase 2a Clinical Trial in COPD with RPL554 Dosed in Addition to Tiotropium (Spiriva ) Achieved significant and clinically
Choosing an inhaler for COPD made simple. Dr Simon Hart Castle Hill Hospital
 Choosing an inhaler for COPD made simple Dr Simon Hart Castle Hill Hospital 1 Declaration of interests I have received speaker fees, sponsorship to attend conferences, and funding for research from companies
Choosing an inhaler for COPD made simple Dr Simon Hart Castle Hill Hospital 1 Declaration of interests I have received speaker fees, sponsorship to attend conferences, and funding for research from companies
AND COPD ADVANCES IN COMBINATION THERAPY FOR ASTHMA. Jan Lotvall. Editor. Krefting Research Centre, University of Gothenburg,
 ADVANCES IN COMBINATION THERAPY FOR ASTHMA AND COPD Editor Jan Lotvall Krefting Research Centre, University of Gothenburg, Sweden A Wiley Sons, Ltd., Publication Contents Contributors xi Preface 1 Similarities
ADVANCES IN COMBINATION THERAPY FOR ASTHMA AND COPD Editor Jan Lotvall Krefting Research Centre, University of Gothenburg, Sweden A Wiley Sons, Ltd., Publication Contents Contributors xi Preface 1 Similarities
Theravance Biopharma, Inc. (NASDAQ: TBPH)
 Theravance Biopharma, Inc. (NASDAQ: TBPH) FDA Approval of YUPELRI TM (revefenacin) Inhalation Solution November 9, 2018 THERAVANCE and the Cross/Star logo are registered trademarks of the Theravance Biopharma
Theravance Biopharma, Inc. (NASDAQ: TBPH) FDA Approval of YUPELRI TM (revefenacin) Inhalation Solution November 9, 2018 THERAVANCE and the Cross/Star logo are registered trademarks of the Theravance Biopharma
Respiratory Subcommittee of PTAC meeting held 5 February (minutes for web publishing)
 Respiratory Subcommittee of PTAC meeting held 5 February 2010 (minutes for web publishing) Respiratory Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Respiratory Subcommittee of PTAC meeting held 5 February 2010 (minutes for web publishing) Respiratory Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Chronic Obstructive Pulmonary Disease (COPD) KAREN ALLEN MD PULMONARY & CRITICAL CARE MEDICINE VA HOSPITAL OKC / OUHSC
 Chronic Obstructive Pulmonary Disease (COPD) KAREN ALLEN MD PULMONARY & CRITICAL CARE MEDICINE VA HOSPITAL OKC / OUHSC I have no financial disclosures Definition COPD is a preventable and treatable disease
Chronic Obstructive Pulmonary Disease (COPD) KAREN ALLEN MD PULMONARY & CRITICAL CARE MEDICINE VA HOSPITAL OKC / OUHSC I have no financial disclosures Definition COPD is a preventable and treatable disease
COPD and Asthma Devices Market by Inhalers Type (Drug powder, Metered Dose, Soft Mist), Nebulizers (Compressor, Ultrasonic, Mesh) - Global
 COPD and Asthma Devices Market by Inhalers Type (Drug powder, Metered Dose, Soft Mist), Nebulizers (Compressor, Ultrasonic, Mesh) - Global Opportunity Analysis and Industry Forecasts, 2015-2022 COPD and
COPD and Asthma Devices Market by Inhalers Type (Drug powder, Metered Dose, Soft Mist), Nebulizers (Compressor, Ultrasonic, Mesh) - Global Opportunity Analysis and Industry Forecasts, 2015-2022 COPD and
Journal of the COPD Foundation. Impact of the IMPACT Trial Ron Balkissoon, MD, MSc, DIH, FRCPC 1. Chronic Obstructive Pulmonary Diseases: Journal Club
 221 Journal Club: The IMPACT Trial Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Journal Club Impact of the IMPACT Trial Ron Balkissoon, MD, MSc, DIH, FRCPC 1 Abbreviations: Global
221 Journal Club: The IMPACT Trial Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Journal Club Impact of the IMPACT Trial Ron Balkissoon, MD, MSc, DIH, FRCPC 1 Abbreviations: Global
Treatment. Assessing the outcome of interventions Traditionally, the effects of interventions have been assessed by measuring changes in the FEV 1
 58 COPD 59 The treatment of COPD includes drug therapy, surgery, exercise and counselling/psychological support. When managing COPD patients, it is particularly important to evaluate the social and family
58 COPD 59 The treatment of COPD includes drug therapy, surgery, exercise and counselling/psychological support. When managing COPD patients, it is particularly important to evaluate the social and family
COPD: Treatment Update Property of Presenter. Not for Reproduction. Barry Make, MD Professor of Medicine National Jewish Health
 COPD: Treatment Update Barry Make, MD Professor of Medicine National Jewish Health Disclosures Advisory board, consultant, multi-center trial, research funding, Data Safety Monitoring Board (DSMB), or
COPD: Treatment Update Barry Make, MD Professor of Medicine National Jewish Health Disclosures Advisory board, consultant, multi-center trial, research funding, Data Safety Monitoring Board (DSMB), or
2017 GOLD Report. Is it worth its weight in GOLD??? CSHP-NB Fall Education Day September 30, 2017
 2017 GOLD Report Is it worth its weight in GOLD??? CSHP-NB Fall Education Day September 30, 2017 Lauren Munro; BSc(Pharm) Amanda Burns; BSc(Pharm) Pharmacy Residents The Moncton Hospital Objectives Explain
2017 GOLD Report Is it worth its weight in GOLD??? CSHP-NB Fall Education Day September 30, 2017 Lauren Munro; BSc(Pharm) Amanda Burns; BSc(Pharm) Pharmacy Residents The Moncton Hospital Objectives Explain
Advancing COPD treatment strategies with evidencebased. 17:15 19:15 Monday 11 September 2017 ERS 2017, Milan, Italy
 Advancing COPD treatment strategies with evidencebased approaches 17:15 19:15 Monday 11 September 2017 ERS 2017, Milan, Italy Increasing understanding of COPD and the effect on guideline evolution. GOLD
Advancing COPD treatment strategies with evidencebased approaches 17:15 19:15 Monday 11 September 2017 ERS 2017, Milan, Italy Increasing understanding of COPD and the effect on guideline evolution. GOLD
Goals and Learning Objectives
 Small Airways (SAW) Symposium: Asthma Treatment Issues. New Bronchodilator for Asthma: A Patient Centric Approach for Treating Asthma Stephen P. Peters, MD, PhD, FAAAAI, FACP, FCCP, FCPP Thomas H. Davis
Small Airways (SAW) Symposium: Asthma Treatment Issues. New Bronchodilator for Asthma: A Patient Centric Approach for Treating Asthma Stephen P. Peters, MD, PhD, FAAAAI, FACP, FCCP, FCPP Thomas H. Davis
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD)
 Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) Zahava Picado, PharmD PGY1 Pharmacy Practice Resident Central Texas Veterans Healthcare System Temple, TX October
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) Zahava Picado, PharmD PGY1 Pharmacy Practice Resident Central Texas Veterans Healthcare System Temple, TX October
umeclidinium, 55 micrograms, powder for inhalation (Incruse ) SMC No. (1004/14) GlaxoSmithKline
 umeclidinium, 55 micrograms, powder for inhalation (Incruse ) SMC No. (1004/14) GlaxoSmithKline 07 November 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
umeclidinium, 55 micrograms, powder for inhalation (Incruse ) SMC No. (1004/14) GlaxoSmithKline 07 November 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
PATHOPHYSIOLOGICAL PROCESS TEMPLATE
 1 PATHOPHYSIOLOGICAL PROCESS TEMPLATE DISEASE: Chronic obstructive pulmonary disease (COPD) DEFINITION: COPD can be defined as a disease in which there is a significant damage to the lungs thus reducing
1 PATHOPHYSIOLOGICAL PROCESS TEMPLATE DISEASE: Chronic obstructive pulmonary disease (COPD) DEFINITION: COPD can be defined as a disease in which there is a significant damage to the lungs thus reducing
GINA. At-A-Glance Asthma Management Reference. for adults, adolescents and children 6 11 years. Updated 2017
 GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
Dr Stephen Child. General Physician Auckland. 14:20-14:40 Secondary Care Perspective
 Dr Stephen Child General Physician Auckland 14:20-14:40 Secondary Care Perspective Wheeze Witchery Stephen Child MD, FRACP, FRCPC General Physician Respiratory Interest Director of Clinical Training Auckland
Dr Stephen Child General Physician Auckland 14:20-14:40 Secondary Care Perspective Wheeze Witchery Stephen Child MD, FRACP, FRCPC General Physician Respiratory Interest Director of Clinical Training Auckland
roflumilast 500 microgram tablets (Daxas ) SMC No. (635/10) Nycomed Ltd
 roflumilast 500 microgram tablets (Daxas ) SMC No. (635/10) Nycomed Ltd 06 August 2010 (Issued 10 September 2010) The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
roflumilast 500 microgram tablets (Daxas ) SMC No. (635/10) Nycomed Ltd 06 August 2010 (Issued 10 September 2010) The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
FDCs for CO PD: From Famine to a Feast of Therapeutic Choices
 FDCs for CO PD: From Famine to a Feast of Therapeutic Choices LAURA RUNKEL, PhD Associate Director, CNS, AutoImmune/Inflammation, Ophthalmology Historically, there have been few therapies developed for
FDCs for CO PD: From Famine to a Feast of Therapeutic Choices LAURA RUNKEL, PhD Associate Director, CNS, AutoImmune/Inflammation, Ophthalmology Historically, there have been few therapies developed for
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A.
 aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
Optimum treatment for chronic obstructive pulmonary disease exacerbation prevention
 Commentary Page 1 of 5 Optimum treatment for chronic obstructive pulmonary disease exacerbation prevention Pradeep Karur, Dave Singh Centre for Respiratory Medicine and Allergy, Medicines Evaluation Unit,
Commentary Page 1 of 5 Optimum treatment for chronic obstructive pulmonary disease exacerbation prevention Pradeep Karur, Dave Singh Centre for Respiratory Medicine and Allergy, Medicines Evaluation Unit,
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
COPD Research at the University of Maryland School of Maryland
 COPD Research at the University of Maryland School of Maryland COPD Clinical Research Center A member of the National Heart Lung & Blood Institute National Institutes of Health Director: Steven M. Scharf,
COPD Research at the University of Maryland School of Maryland COPD Clinical Research Center A member of the National Heart Lung & Blood Institute National Institutes of Health Director: Steven M. Scharf,
QUANTITY LIMIT CRITERIA. BROVANA (arformoterol tartrate) SEREVENT DISKUS (salmeterol) STRIVERDI RESPIMAT (olodaterol)
 Carelirst. +.V Family of health care plans DRUG CLASS COMBINATIONS QUANTITY LIMIT CRITERIA LONG ACTING BETA2-ADRENERGIC AGONIST, ORAL INHALATION BRAND NAME (generic) LONG-ACTING BETA2-ADRENERGIC AGONISTS:
Carelirst. +.V Family of health care plans DRUG CLASS COMBINATIONS QUANTITY LIMIT CRITERIA LONG ACTING BETA2-ADRENERGIC AGONIST, ORAL INHALATION BRAND NAME (generic) LONG-ACTING BETA2-ADRENERGIC AGONISTS:
Decramer 2014 a &b [21]
![Decramer 2014 a &b [21] Decramer 2014 a &b [21]](/thumbs/90/101390504.jpg) Buhl 2015 [19] Celli 2014 [20] Decramer 2014 a &b [21] D Urzo 2014 [22] Maleki-Yazdi 2014 [23] Inclusion criteria: Diagnosis of chronic obstructive pulmonary disease; 40 years of age or older; Relatively
Buhl 2015 [19] Celli 2014 [20] Decramer 2014 a &b [21] D Urzo 2014 [22] Maleki-Yazdi 2014 [23] Inclusion criteria: Diagnosis of chronic obstructive pulmonary disease; 40 years of age or older; Relatively
your breathing problems worsen quickly. you use your rescue inhaler, but it does not relieve your breathing problems.
 MEDICATION GUIDE ADVAIR DISKUS [ad vair disk us] (fluticasone propionate and salmeterol inhalation powder) for oral inhalation What is the most important information I should know about ADVAIR DISKUS?
MEDICATION GUIDE ADVAIR DISKUS [ad vair disk us] (fluticasone propionate and salmeterol inhalation powder) for oral inhalation What is the most important information I should know about ADVAIR DISKUS?
Clinical Policy: Roflumilast (Daliresp) Reference Number: CP.PMN.46. Line of Business: Medicaid
 Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11/11 Last Review Date: 08/17 Line of Business: Medicaid See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11/11 Last Review Date: 08/17 Line of Business: Medicaid See Important Reminder at the end of this policy for important regulatory
THE CORPORATE REPUTATION OF PHARMA 2015 THE PERSPECTIVE OF 139 PATIENT GROUPS with an interest in DIABETES
 THE CORPORATE REPUTATION OF PHARMA 2015 THE PERSPECTIVE OF 139 PATIENT GROUPS with an interest in DIABETES PUBLISHED AUGUST 2016 There is growing distrust of pharma in light of several recent and dramatic
THE CORPORATE REPUTATION OF PHARMA 2015 THE PERSPECTIVE OF 139 PATIENT GROUPS with an interest in DIABETES PUBLISHED AUGUST 2016 There is growing distrust of pharma in light of several recent and dramatic
Journal of the COPD Foundation
 132 Predictors of Change in SGRQ Score Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Original Research Baseline Severity as Predictor of Change in St George s Respiratory Questionnaire
132 Predictors of Change in SGRQ Score Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Original Research Baseline Severity as Predictor of Change in St George s Respiratory Questionnaire
CHAPTER ONE: EXECUTIVE SUMMARY
 CHAPTER ONE: EXECUTIVE SUMMARY Introduction Scope and Methodology Total Size and Growth of the Market Issues and Trends Affecting Market Leading Competitors CHAPTER TWO: INTRODUCTION AND INDUSTRY TRENDS
CHAPTER ONE: EXECUTIVE SUMMARY Introduction Scope and Methodology Total Size and Growth of the Market Issues and Trends Affecting Market Leading Competitors CHAPTER TWO: INTRODUCTION AND INDUSTRY TRENDS
umeclidinium/vilanterol, 55/22 micrograms, inhalation powder (Anoro ) SMC No. (978/14) GlaxoSmithKline
 umeclidinium/vilanterol, 55/22 micrograms, inhalation powder (Anoro ) SMC No. (978/14) GlaxoSmithKline 04 July 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
umeclidinium/vilanterol, 55/22 micrograms, inhalation powder (Anoro ) SMC No. (978/14) GlaxoSmithKline 04 July 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
Chronic obstructive pulmonary disease
 0 Chronic obstructive pulmonary disease Implementing NICE guidance June 2010 NICE clinical guideline 101 What this presentation covers Background Scope Key priorities for implementation Discussion Find
0 Chronic obstructive pulmonary disease Implementing NICE guidance June 2010 NICE clinical guideline 101 What this presentation covers Background Scope Key priorities for implementation Discussion Find
Outcomes: Initially, our primary definitions of pneumonia was severe pneumonia, where the subject was hospitalized
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Re-Submission. roflumilast, 500 microgram, film-coated tablet (Daxas ) SMC No 635/10 AstraZeneca UK Ltd. Published 11 September
 Re-Submission roflumilast, 500 microgram, film-coated tablet (Daxas ) SMC No 635/10 AstraZeneca UK Ltd 4 August 2017 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
Re-Submission roflumilast, 500 microgram, film-coated tablet (Daxas ) SMC No 635/10 AstraZeneca UK Ltd 4 August 2017 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
The FDA Critical Path Initiative
 The FDA Critical Path Initiative Clinical Considerations for Demonstration of Dose-response for Inhaled Corticosteroids - Exhaled Nitric Oxide Model Badrul A. Chowdhury, MD, PhD Director Division of Pulmonary
The FDA Critical Path Initiative Clinical Considerations for Demonstration of Dose-response for Inhaled Corticosteroids - Exhaled Nitric Oxide Model Badrul A. Chowdhury, MD, PhD Director Division of Pulmonary
Inhalational antibacterial regimens in non-cystic fibrosis patients. Jeff Alder Bayer HealthCare
 Inhalational antibacterial regimens in non-cystic fibrosis patients Jeff Alder Bayer HealthCare Alder - Inhaled therapy for non-cf - EMA 25-26 Oct 2012 1 Inhalational antibacterials: two approaches 1.
Inhalational antibacterial regimens in non-cystic fibrosis patients Jeff Alder Bayer HealthCare Alder - Inhaled therapy for non-cf - EMA 25-26 Oct 2012 1 Inhalational antibacterials: two approaches 1.
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Co-Primary Outcomes/Efficacy Variables:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Turning Science into Real Life Roflumilast in Clinical Practice. Roland Buhl Pulmonary Department Mainz University Hospital
 Turning Science into Real Life Roflumilast in Clinical Practice Roland Buhl Pulmonary Department Mainz University Hospital Therapy at each stage of COPD I: Mild II: Moderate III: Severe IV: Very severe
Turning Science into Real Life Roflumilast in Clinical Practice Roland Buhl Pulmonary Department Mainz University Hospital Therapy at each stage of COPD I: Mild II: Moderate III: Severe IV: Very severe
NHS Dumfries & Galloway Triple therapy in COPD patients over 16 years
 Title of Project: NHS Dumfries & Galloway Triple therapy in COPD patients over 16 years 1 Reason for the review Respiratory prescribing is long term and can be costly. Appropriate choice and use of inhaled
Title of Project: NHS Dumfries & Galloway Triple therapy in COPD patients over 16 years 1 Reason for the review Respiratory prescribing is long term and can be costly. Appropriate choice and use of inhaled
Medicines that make a difference
 Medicines that make a difference 11 th Annual Needham Healthcare Conference Mathai Mammen, M.D., Ph.D. Senior Vice President, Research and Early Clinical Development April 4, 2012 Theravance, Theravance
Medicines that make a difference 11 th Annual Needham Healthcare Conference Mathai Mammen, M.D., Ph.D. Senior Vice President, Research and Early Clinical Development April 4, 2012 Theravance, Theravance
Cystic Fibrosis the future
 Cystic Fibrosis the future Pathophysiologic cascade Abnormal Gene Abnormal CFTR Therapy Gene replacement Protein replacement Gene read through therapy Abnormal sodium chloride & water movement through
Cystic Fibrosis the future Pathophysiologic cascade Abnormal Gene Abnormal CFTR Therapy Gene replacement Protein replacement Gene read through therapy Abnormal sodium chloride & water movement through
Journal Club: Phosphodiesterase-4 Inhibitors Ron Balkissoon, MD, MSc, DIH, FRCPC 1
 693 Journal Club: Phosphodiesterase-4 Inhibitors Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Journal Club: Phosphodiesterase-4 Inhibitors Ron Balkissoon, MD, MSc, DIH, FRCPC
693 Journal Club: Phosphodiesterase-4 Inhibitors Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Journal Club: Phosphodiesterase-4 Inhibitors Ron Balkissoon, MD, MSc, DIH, FRCPC
Three better than 1 or 2?
 Three better than 1 or 2? DISCLOSURE Pam McLean-Veysey, Team Leader Drug Evaluation Unit DEU funded by the Drug Evaluation Alliance of NS. (DEANS). DEU prepares Drug Evaluation Reports for the Atlantic
Three better than 1 or 2? DISCLOSURE Pam McLean-Veysey, Team Leader Drug Evaluation Unit DEU funded by the Drug Evaluation Alliance of NS. (DEANS). DEU prepares Drug Evaluation Reports for the Atlantic
Methacholine versus Mannitol Challenge in the Evaluation of Asthma Clinical applications of methacholine and mannitol challenges
 Methacholine versus Mannitol Challenge in the Evaluation of Asthma Clinical applications of methacholine and mannitol challenges AAAAI San Antonio Tx February 2013 Catherine Lemière MD, MSc Hôpital du
Methacholine versus Mannitol Challenge in the Evaluation of Asthma Clinical applications of methacholine and mannitol challenges AAAAI San Antonio Tx February 2013 Catherine Lemière MD, MSc Hôpital du
COPD EXACERBATIONS AND HOSPITAL ADMISSIONS HOW CAN WE PREVENT THEM? Wisia Wedzicha National Heart and Lung Institute, Imperial College London, UK
 COPD EXACERBATIONS AND HOSPITAL ADMISSIONS HOW CAN WE PREVENT THEM? Wisia Wedzicha National Heart and Lung Institute, Imperial College London, UK Presenter Disclosures Wisia Wedzicha All disclosures prior
COPD EXACERBATIONS AND HOSPITAL ADMISSIONS HOW CAN WE PREVENT THEM? Wisia Wedzicha National Heart and Lung Institute, Imperial College London, UK Presenter Disclosures Wisia Wedzicha All disclosures prior
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Symdeko. Symdeko (tezacaftor and ivacaftor) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.45.10 Subject: Symdeko Page: 1 of 5 Last Review Date: June 22, 2018 Symdeko Description Symdeko (tezacaftor
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.45.10 Subject: Symdeko Page: 1 of 5 Last Review Date: June 22, 2018 Symdeko Description Symdeko (tezacaftor
Revefenacin (TD-4208) Phase 3 Efficacy Results
 Revefenacin (TD-4208) Phase 3 Efficacy Results Once-daily, Nebulized Long-Acting Muscarinic Antagonist (LAMA) October 20, 2016 THERAVANCE, the Cross/Star logo and MEDICINES THAT MAKE A DIFFERENCE are registered
Revefenacin (TD-4208) Phase 3 Efficacy Results Once-daily, Nebulized Long-Acting Muscarinic Antagonist (LAMA) October 20, 2016 THERAVANCE, the Cross/Star logo and MEDICINES THAT MAKE A DIFFERENCE are registered
14 41 minutes Vd 218 t ½ 22 to 70 Clearance hours Protein Binding 71%, active metabolite 42% Bioavailability 3%
 Brand Name: Yupelri Generic Name: revefenacin Manufacturer: Mylan Drug Class: Anticholinergic Uses: Labeled Uses: inhalation solution for maintenance treatment of COPD 1 Unlabeled Uses: no off-label indications
Brand Name: Yupelri Generic Name: revefenacin Manufacturer: Mylan Drug Class: Anticholinergic Uses: Labeled Uses: inhalation solution for maintenance treatment of COPD 1 Unlabeled Uses: no off-label indications
Respiratory Research Newsletter
 Date Published: September 2012 Respiratory Research Newsletter Contents Research at the Belfast Trust 1 NICRN Respiratory Health 2 The Respiratory Research Team 3 Our Partners 4 Getting Involved in Upcoming
Date Published: September 2012 Respiratory Research Newsletter Contents Research at the Belfast Trust 1 NICRN Respiratory Health 2 The Respiratory Research Team 3 Our Partners 4 Getting Involved in Upcoming
Bronchiectasis. What is bronchiectasis? What causes bronchiectasis?
 This factsheet explains what bronchiectasis is, what causes it, and how it is diagnosed and managed. More detailed information is available on the Bronchiectasis Patient Priorities website: www.europeanlunginfo.org/bronchiectasis
This factsheet explains what bronchiectasis is, what causes it, and how it is diagnosed and managed. More detailed information is available on the Bronchiectasis Patient Priorities website: www.europeanlunginfo.org/bronchiectasis
glycopyrronium 44 micrograms hard capsules of inhalation powder (Seebri Breezhaler ) SMC No. (829/12) Novartis Pharmaceuticals Ltd.
 glycopyrronium 44 micrograms hard capsules of inhalation powder (Seebri Breezhaler ) SMC No. (829/12) Novartis Pharmaceuticals Ltd. 07 December 2012 The Scottish Medicines Consortium (SMC) has completed
glycopyrronium 44 micrograms hard capsules of inhalation powder (Seebri Breezhaler ) SMC No. (829/12) Novartis Pharmaceuticals Ltd. 07 December 2012 The Scottish Medicines Consortium (SMC) has completed
Kalydeco. Kalydeco (ivacaftor) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.45.03 Subject: Kalydeco Page: 1 of 6 Last Review Date: November 30, 2018 Kalydeco Description Kalydeco
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.45.03 Subject: Kalydeco Page: 1 of 6 Last Review Date: November 30, 2018 Kalydeco Description Kalydeco
Update on Pulmonary Diseases. Jeffrey Lessar, MD
 Update on Pulmonary Diseases Jeffrey Lessar, MD 1 No disclosures to make No conflicts 2 Goals Update on key changes in Pulmonary Therapy 3 Spirometry Spirometry FEV1- forced expiratory Volume in 1 Sec
Update on Pulmonary Diseases Jeffrey Lessar, MD 1 No disclosures to make No conflicts 2 Goals Update on key changes in Pulmonary Therapy 3 Spirometry Spirometry FEV1- forced expiratory Volume in 1 Sec
12/18/2017. Disclosures. Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Changing Epidemiology: Quick Facts 9/28/2018. During the year New Treatment Options for COPD: Phenotypes, Endotypes or Treatable Traits?
 New Treatment Options for COPD: Phenotypes, Endotypes or Treatable Traits? Conflict of Interest Statement 2018 Speakers Bureau Astra Zeneca Boehringer Ingelheim Genentech Sunovion Ron Balkissoon MD DIH
New Treatment Options for COPD: Phenotypes, Endotypes or Treatable Traits? Conflict of Interest Statement 2018 Speakers Bureau Astra Zeneca Boehringer Ingelheim Genentech Sunovion Ron Balkissoon MD DIH
Forward Looking Statements
 Therapeutic products for respiratory diseases September 2008 Forward Looking Statements This presentation may contain forward-looking statements that are based on management s current expectations and
Therapeutic products for respiratory diseases September 2008 Forward Looking Statements This presentation may contain forward-looking statements that are based on management s current expectations and
PA Update: Oral Cystic Fibrosis Modulators
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Enabling CF Therapeutic Development
 Enabling CF Therapeutic Development PRESTON W. CAMPBELL, III, M.D. Executive Vice President for Medical Affairs No Disclosures Cystic Fibrosis In 1955 In 1955 most children with CF did not live long enough
Enabling CF Therapeutic Development PRESTON W. CAMPBELL, III, M.D. Executive Vice President for Medical Affairs No Disclosures Cystic Fibrosis In 1955 In 1955 most children with CF did not live long enough
Advances in the management of chronic obstructive lung diseases (COPD) David CL Lam Department of Medicine University of Hong Kong October, 2015
 Advances in the management of chronic obstructive lung diseases (COPD) David CL Lam Department of Medicine University of Hong Kong October, 2015 Chronic obstructive pulmonary disease (COPD) COPD in Hong
Advances in the management of chronic obstructive lung diseases (COPD) David CL Lam Department of Medicine University of Hong Kong October, 2015 Chronic obstructive pulmonary disease (COPD) COPD in Hong
CHRONIC OBSTRUCTIVE PULMONARY DISEASE
 CHRONIC OBSTRUCTIVE PULMONARY DISEASE INCIDENCE UP TO 380,000 PEOPLE IN IRELAND HSE FIGURES 110,000 DIAGNOSED AND 200,000 UNDIAGNOSED. AFFECTS MORE MEN THAN WOMEN BUT RATES ARE RISING 1500 DEATHS PER YEAR
CHRONIC OBSTRUCTIVE PULMONARY DISEASE INCIDENCE UP TO 380,000 PEOPLE IN IRELAND HSE FIGURES 110,000 DIAGNOSED AND 200,000 UNDIAGNOSED. AFFECTS MORE MEN THAN WOMEN BUT RATES ARE RISING 1500 DEATHS PER YEAR
To describe the impact of COPD exacerbations and the importance of the frequent exacerbator phenotype.
 Educational aims To describe the impact of COPD exacerbations and the importance of the frequent exacerbator phenotype. To describe the spectrum of pharmacological and non-pharmacological interventions
Educational aims To describe the impact of COPD exacerbations and the importance of the frequent exacerbator phenotype. To describe the spectrum of pharmacological and non-pharmacological interventions
COPD Treatable. Preventable.
 My COPD Action Plan Patient s Copy (Patient s Name) Date Canadian Respiratory COPD Treatable. Preventable. This is to tell me how I will take care of myself when I have a COPD flare-up. My goals are My
My COPD Action Plan Patient s Copy (Patient s Name) Date Canadian Respiratory COPD Treatable. Preventable. This is to tell me how I will take care of myself when I have a COPD flare-up. My goals are My
Roflumilast (Daxas) for chronic obstructive pulmonary disease
 Roflumilast (Daxas) for chronic obstructive pulmonary disease August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended
Roflumilast (Daxas) for chronic obstructive pulmonary disease August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended
Presented by UIC College of Nursing
 Presented by UIC College of Nursing Describe COPD. Identify red flags for a COPD exacerbation. Identify COPD triggers or risk factors. Differentiate between long-acting inhalers and emergency use inhalers.
Presented by UIC College of Nursing Describe COPD. Identify red flags for a COPD exacerbation. Identify COPD triggers or risk factors. Differentiate between long-acting inhalers and emergency use inhalers.
Chronic Obstructive Pulmonary Disease 1/18/2018
 Presented by UIC College of Nursing Describe COPD. Identify red flags for a COPD exacerbation. Identify COPD triggers or risk factors. Differentiate between long acting inhalers and emergency use inhalers.
Presented by UIC College of Nursing Describe COPD. Identify red flags for a COPD exacerbation. Identify COPD triggers or risk factors. Differentiate between long acting inhalers and emergency use inhalers.
DR REBECCA THOMAS CONSULTANT RESPIRATORY PHYSICIAN YORK DISTRICT HOSPITAL
 DR REBECCA THOMAS CONSULTANT RESPIRATORY PHYSICIAN YORK DISTRICT HOSPITAL Definition Guidelines contact complicated definitions Central to this is Presence of symptoms Variable airflow obstruction Diagnosis
DR REBECCA THOMAS CONSULTANT RESPIRATORY PHYSICIAN YORK DISTRICT HOSPITAL Definition Guidelines contact complicated definitions Central to this is Presence of symptoms Variable airflow obstruction Diagnosis
Incruse Ellipta 55mcg (umeclidinium bromide) for the relief of symptoms in adult patients with Chronic Obstructive Pulmonary Disease (COPD):
 Incruse Ellipta 55mcg (umeclidinium bromide) for the relief of symptoms in adult patients with Chronic Obstructive Pulmonary Disease (COPD): An introduction for Clinical Commisioning Groups and Health
Incruse Ellipta 55mcg (umeclidinium bromide) for the relief of symptoms in adult patients with Chronic Obstructive Pulmonary Disease (COPD): An introduction for Clinical Commisioning Groups and Health
Therapeutic products for respiratory and autoimmune diseases. Annual General Meeting
 Therapeutic products for respiratory and autoimmune diseases Annual General Meeting November 2007 Summary Objective Lead products Discovery Listings Location Facility Employees Cash (30/6/07) Shares outstanding
Therapeutic products for respiratory and autoimmune diseases Annual General Meeting November 2007 Summary Objective Lead products Discovery Listings Location Facility Employees Cash (30/6/07) Shares outstanding
RESPIRATORY CARE IN GENERAL PRACTICE
 RESPIRATORY CARE IN GENERAL PRACTICE Definitions of Asthma and COPD Asthma is due to inflammation of the air passages in the lungs and affects the sensitivity of the nerve endings in the airways so they
RESPIRATORY CARE IN GENERAL PRACTICE Definitions of Asthma and COPD Asthma is due to inflammation of the air passages in the lungs and affects the sensitivity of the nerve endings in the airways so they
Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year,
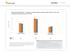 ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
Summary A pharmaceutical company which develops therapeutic products for
 Innovative products for respiratory diseases Annual General Meeting October 2012 Pharmaxis - company overview Summary A pharmaceutical company which develops therapeutic products for human chronic respiratory
Innovative products for respiratory diseases Annual General Meeting October 2012 Pharmaxis - company overview Summary A pharmaceutical company which develops therapeutic products for human chronic respiratory
COPD Device Workshop. Summary. Role of inhaler device in COPD. Why use inhaler device in COPD?
 Part 1 Role of inhaler device in COPD COPD Device Workshop Dr Philip Lee Respiratory and Sleep Physician St George Hospital, Sydney Part 2 Part 3 Part 4 Incorrect inhaler technique-adverse clinical outcomes
Part 1 Role of inhaler device in COPD COPD Device Workshop Dr Philip Lee Respiratory and Sleep Physician St George Hospital, Sydney Part 2 Part 3 Part 4 Incorrect inhaler technique-adverse clinical outcomes
Clinical Policy: Roflumilast (Daliresp) Reference Number: CP.PMN.46 Effective Date: Last Review Date: 08.18
 Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11.01.11 Last Review Date: 08.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11.01.11 Last Review Date: 08.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Verona Pharma (VRP.L)
 18 th May 2015 Pharma & Biotech Verona Pharma (VRP.L) Initiation Clinical data have increased probability of success Source: Fidessa Market data Price (p) 3.5 12m High (p) 3.9 12 Low (p) 1.0 Shares (m)
18 th May 2015 Pharma & Biotech Verona Pharma (VRP.L) Initiation Clinical data have increased probability of success Source: Fidessa Market data Price (p) 3.5 12m High (p) 3.9 12 Low (p) 1.0 Shares (m)
What s new in COPD? Apichart Khanichap MD. Department of Medicine, Faculty of Medicine, Thammasat university
 What s new in COPD? Apichart Khanichap MD. Department of Medicine, Faculty of Medicine, Thammasat university Management stable COPD Relieve symptoms Improve exercise tolerance Improve health status Prevent
What s new in COPD? Apichart Khanichap MD. Department of Medicine, Faculty of Medicine, Thammasat university Management stable COPD Relieve symptoms Improve exercise tolerance Improve health status Prevent
Shaping a Dynamic Future in Respiratory Practice. #DFResp
 Shaping a Dynamic Future in Respiratory Practice #DFResp www.dynamicfuture.co.uk Inhaled Therapy in COPD: Past, Present and Future Richard Russell Chest Physician West Hampshire Integrated Respiratory
Shaping a Dynamic Future in Respiratory Practice #DFResp www.dynamicfuture.co.uk Inhaled Therapy in COPD: Past, Present and Future Richard Russell Chest Physician West Hampshire Integrated Respiratory
without the permission of the author Not to be copied and distributed to others
 Emperor s Castle interior-prato What is the Role of Inhaled Polymyxins for Treatment of Respiratory Tract Infections? Helen Giamarellou CONCLUSIONS: Patients with Pseudomonas and Acinetobacter VAP may
Emperor s Castle interior-prato What is the Role of Inhaled Polymyxins for Treatment of Respiratory Tract Infections? Helen Giamarellou CONCLUSIONS: Patients with Pseudomonas and Acinetobacter VAP may
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Chronic obstructive pulmonary disease: the management of adults with chronic obstructive pulmonary disease in primary and secondary
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Chronic obstructive pulmonary disease: the management of adults with chronic obstructive pulmonary disease in primary and secondary
Chronic Obstructive Pulmonary Disease (COPD) Treatment Guidelines
 Chronic Obstructive Pulmonary Disease (COPD) Treatment Guidelines Where appropriate the following should be offered before commencing inhaled treatment: Offer treatment and support to stop smoking. Smoking
Chronic Obstructive Pulmonary Disease (COPD) Treatment Guidelines Where appropriate the following should be offered before commencing inhaled treatment: Offer treatment and support to stop smoking. Smoking
Dr Conroy Wong. Professor Richard Beasley. Dr Sarah Mooney. Professor Innes Asher
 Professor Richard Beasley University of Otago Director Medical Research Institute of New Zealand Wellington Dr Sarah Mooney Physiotherapy Advanced Clinician Counties Manukau Health NZ Respiratory and Sleep
Professor Richard Beasley University of Otago Director Medical Research Institute of New Zealand Wellington Dr Sarah Mooney Physiotherapy Advanced Clinician Counties Manukau Health NZ Respiratory and Sleep
CCLI. Bronchiectasis Treatment Antibiotics. Charles Haworth. Physician / Patient Conference, Georgetown University, May 2017
 Physician / Patient Conference, Georgetown University, May 2017 Bronchiectasis Treatment Antibiotics Charles Haworth CCLI Cambridge Centre for Lung Infection Disclosures Educational talks and / or consultancy
Physician / Patient Conference, Georgetown University, May 2017 Bronchiectasis Treatment Antibiotics Charles Haworth CCLI Cambridge Centre for Lung Infection Disclosures Educational talks and / or consultancy
CYSTIC FIBROSIS OBJECTIVES NO CONFLICT OF INTEREST TO DISCLOSE
 CYSTIC FIBROSIS Madhu Pendurthi MD MPH Staff Physician, Mercy Hospital Springfield, MO NO CONFLICT OF INTEREST TO DISCLOSE OBJECTIVES Epidemiology of Cystic Fibrosis (CF) Genetic basis and pathophysiology
CYSTIC FIBROSIS Madhu Pendurthi MD MPH Staff Physician, Mercy Hospital Springfield, MO NO CONFLICT OF INTEREST TO DISCLOSE OBJECTIVES Epidemiology of Cystic Fibrosis (CF) Genetic basis and pathophysiology
