University of Groningen
|
|
|
- Maurice Wright
- 5 years ago
- Views:
Transcription
1 University of Groningen Nasal high flow therapy and PtCO2 in stable COPD McKinstry, Steven; Pilcher, Janine; Bardsley, George; Berry, James; van de Hei, Susanne; Braithwaite, Irene; Fingleton, James; Weatherall, Mark; Beasley, Richard Published in: Respirology DOI: /resp IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the document version below. Document Version Publisher's PDF, also known as Version of record Publication date: 2018 Link to publication in University of Groningen/UMCG research database Citation for published version (APA): McKinstry, S., Pilcher, J., Bardsley, G., Berry, J., Van de Hei, S., Braithwaite, I.,... Beasley, R. (2018). Nasal high flow therapy and PtCO2 in stable COPD: A randomized controlled cross-over trial. Respirology, 23(4), DOI: /resp Copyright Other than for strictly personal use, it is not permitted to download or to forward/distribute the text or part of it without the consent of the author(s) and/or copyright holder(s), unless the work is under an open content license (like Creative Commons). Take-down policy If you believe that this document breaches copyright please contact us providing details, and we will remove access to the work immediately and investigate your claim. Downloaded from the University of Groningen/UMCG research database (Pure): For technical reasons the number of authors shown on this cover page is limited to 10 maximum. Download date:
2 Editor's Choice ORIGINAL ARTICLE Nasal high flow therapy and PtCO 2 in stable COPD: A randomized controlled cross-over trial STEVEN MCKINSTRY, 1,2,3 JANINE PILCHER, 1,2,3 GEORGE BARDSLEY, 1,3 JAMES BERRY, 1 SUSANNE VAN DE HEI, 4 IRENE BRAITHWAITE, 1,3 JAMES FINGLETON, 1,2,3 MARK WEATHERALL 3,5 AND RICHARD BEASLEY 1,2,3 1 Medical Research Institute of New Zealand, Wellington; 2 School of Biological Sciences, Victoria University of Wellington, Wellington; 3 Capital and Coast District Health Board, Wellington; 5 School of Medicine and Health Sciences, University of Otago Wellington, Wellington, New Zealand; 4 University of Groningen, Groningen, The Netherlands ABSTRACT Background and objective: Hypercapnia is associated with worse clinical outcomes in exacerbations of COPD. The present study aimed to determine the effects of nasal high flow (NHF) therapy on transcutaneous partial pressure of carbon dioxide (PtCO 2 ) in stable COPD patients. Methods: In a single-blind randomized controlled crossover trial, 48 participants with COPD were allocated in random order to all of four 20 min interventions: NHF at 15 L/min, 30 L/min and 45 L/min or breathing room air with each intervention followed by a washout period of 15 min. The primary outcome measure was PtCO 2 at 20 min, adjusted for baseline PtCO 2. Secondary outcomes included respiratory rate at 20 min, adjusted for baseline. Results: The mean (95% CI) change in PtCO 2 at 20 min was 0.6 mm Hg ( 1.1 to 0.0), P = 0.06; 1.3 mm Hg ( 1.9 to 0.8), ; and 2.4 mm Hg ( 2.9 to 1.8), ; for NHF at 15 L/min, 30 L/min and 45 L/min compared with room air, respectively. The mean (95% CI) change in respiratory rate at 20 min was 1.5 ( 2.7 to 0.3), P = 0.02; 4.1 ( 5.3 to 2.9), ; and 4.3 ( 5.5 to 3.1), ; breaths per minute compared with room air, respectively. Conclusion: NHF results in a small flow-dependent reduction in PtCO 2 and respiratory rate in patients with stable COPD. Clinical trial registration: ACTRN at anzctr. org.au Key words: arterial partial pressure, carbon dioxide, chronic obstructive respiratory disease, nasal high flow, randomized controlled trial. Abbreviations: FEV 1, forced expiratory volume in 1 s; FVC, forced vital capacity; NHF, nasal high flow; NIV, non-invasive ventilation; PaCO 2, partial pressure of arterial carbon dioxide; PtCO 2, transcutaneous partial pressure of carbon dioxide; RIP, Respiratory Inductance Plethysmography; StO 2, transcutaneous oxygen saturation. Correspondence: Steven McKinstry, Medical Research Institute of New Zealand, Private Bag 7902, Wellington 6242, New Zealand. steve.mckinstry@mrinz.ac.nz Received March ; revised July ; accepted September (Associate Editor: Maarten van den Berge; Senior Editor: Phan Nguyen). SUMMARY AT A GLANCE In patients with stable COPD, the administration of nasal high flow results in flow-dependent reductions in transcutaneous partial pressure of carbon dioxide and respiratory rate. INTRODUCTION In acute exacerbations of COPD, hypercapnia is associated with worse clinical outcomes including death. 1 Noninvasive ventilation (NIV) is recommended to provide respiratory support to patients with exacerbations of COPD who have hypercapnic respiratory failure despite optimal medical therapy. 2 Tolerability of NIV may be a barrier to effective use 3 and an alternative to NIV is a priority for the management of acute exacerbations of COPD. Nasal high flow (NHF) therapy may cause a modest reduction in the partial pressure of arterial carbon dioxide (PaCO 2 ) in both stable and acute COPD. 4 9 However, the interpretation of studies of NHF, and their applicability to clinical practice, remains variably limited by the confounding effect of concomitant oxygen therapy, absence of randomized controlled treatments and a lack of data on the dose response relationship across the range of flows used in clinical practice. The present study is a randomized controlled crossover trial of the effect of three different flow rates of NHF therapy compared with a control intervention of room air, in patients with stable COPD who do not need concomitant oxygen therapy. The main objective of the present study was to determine the flowresponse relationship of NHF therapy and PaCO 2 in stable COPD. The hypothesis was that NHF therapy would cause a flow-dependent reduction in PaCO 2 and respiratory rate in stable COPD. METHODS In this single-blind, randomized, controlled, four-way cross-over trial, 48 participants with a doctor s diagnosis of COPD, aged at least 40 years and with a tobacco 2017 Asian Pacific Society of Respirology Respirology (2018) 23, doi: /resp.13185
3 Nasal high flow therapy in stable COPD 379 smoking history of 10 pack years were recruited. Participants were excluded if their forced expiratory volume in 1 s (FEV 1 )/ forced vital capacity ratio was >0.7, or if they were on long-term oxygen therapy, had a current exacerbation of COPD requiring a short course of antibiotics or oral glucocorticoids or oxygen therapy, or hospitalization for an acute exacerbation of COPD within the last 6 weeks. People with nasal conditions potentially affecting the ability to use NHF were also excluded. Eligible participants attended a single study visit at the MRINZ Respiratory Physiology Laboratory at Wellington Regional Hospital. The present study was prospectively registered with ANZCTR (Trial ID: ACTRN ) and approved by the Health and Disability Ethics Committee of New Zealand (Ref: 15/NTA/4). Full written informed consent was completed before any studyspecific procedures. After demographic data was collected, spirometry was performed in accordance with American Thoracic Society/European Respiratory Society criteria 10 using a Jaeger Master screen body volume constant plethysmography unit with pneumotachograph and diffusion unit (Erich-Jaegar, Wurzburg, Germany). Measurements of transcutaneous partial pressure of carbon dioxide (PtCO 2 ), transcutaneous oxygen saturation (StO 2 ) and heart rate were made using the SenTec transcutaneous monitor (SenTec digital monitor with V-Sign Sensor VS-A/P/N, Therwil, Switzerland; further details in the Appendix S1 in Supplementary Information). The SenTec probe was kept on the patient for between 20 and 30 min before the subsequent study procedures to ensure a stable baseline measurement of PtCO 2. Minute ventilation was measured using Respiratory Inductance Plethysmography (RIP) bands (QDC-Pro device; CareFusion, Yorba Linda, California, USA). Further details are given in the Appendix S1 in Supplementary Information. Participants received all interventions for 20 min in a randomized order while seated. Each NHF flow setting was at a temperature of 37 C without oxygen: 15 L/ min, 30 L/min or 45 L/min; or the control setting of breathing room air only without the NHF attached, using the myairvo 2 device (PT101AZ; Fisher and Paykel Healthcare, Auckland, New Zealand). Further details are given in the Appendix S1 in Supplementary Information. Each of the four interventions was followed by a washout period breathing room air for at least 15 min, allowing the PtCO 2 to return to within 4 mm Hg of the baseline measurement for the particular intervention. The washout could be extended until this criterion was met. PtCO 2, StO 2, heart rate and respiratory rate were recorded at the start of each intervention and then every 5 min until the end of each washout period. The order of administration of the four treatments was randomized. The randomisation was computergenerated by the study statistician, who had no role in the recruitment, study visits or data collection. Treatment allocation and maintenance of blinding are described in the Appendix S1 in Supplementary Information. Respirology (2018) 23, Participant tolerability questionnaires were administered during the washout periods after each NHF intervention. Participants rated the ease of application, level of comfort, weight of the nasal interface, noisiness, amount of moisture in the nasal passages and likelihood of reusing the system on a continuous scale from most positive (0) to least positive (100). Outcomes The primary outcome was PtCO 2 at 20 min, adjusted for baseline PtCO 2. Secondary outcomes were: the proportion of participants who had a decrease in PtCO 2 4 mm Hg from baseline during the intervention; PtCO 2, respiratory rate, StO 2, heart rate and minute ventilation adjusted for baseline for each 5-min time-point during the intervention and the subsequent 15 min washout period; the proportion of participants who withdrew from the intervention before it was completed; and results of the tolerability questionnaires. Statistical analysis The paired SDs of PtCO 2 in a previous study investigating oxygen administration to patients with stable COPD were between 1.8 and 4.4 mm Hg. 11 Based on the highest PtCO 2 SD of 4.4 mm Hg, and an alpha value of (to take into account the potential for six possible comparisons for the four-way cross-over study) a sample size of 48 had 90% power to detect a difference in PtCO 2 of 3.8 mm Hg. The comparison of each of the three NHF treatments compared to room air was by mixed linear model with fixed effects for the randomisation sequence, the baseline measurement of the particular variable, and the randomized treatment, time and their interaction; and a random effect for each participant, with an exponential time correlation structure for the repeated measurements. The comparison of paired proportions for those that had a decrease from baseline PtCO 2 of 4 mm Hg was by an exact McNemar s test and estimation of the CI for the differences in paired proportions, NHF intervention minus room air, by an asymptotic method. The comparison of device questionnaire scores was by a mixed linear model with fixed effects for the randomization sequence and treatment; and a random effect for the participant. In a post hoc analysis, the statistical test of whether the PtCO 2 response to treatment differed by whether the baseline PtCO 2 was >45 mm Hg or not was the interaction P value. The above-mentioned analysis was carried out using the SAS (SAS, Cary, NC, USA) version 9.4 package. RESULTS Participant characteristics Contact was made with 84 potentially eligible participants of whom 48 were randomized between May 2015 and February 2016 (Fig. 1). Three participants required an extended washout in at least one of the interventions past the planned 15 min washout for the PtCO 2 to return to within 4 mm 2017 Asian Pacific Society of Respirology
4 380 S McKinstry et al. Screening phone call and/or PIS sent to potentially eligible participants on MRINZ database (n = 84) Randomized (n = 48) to 4 interventions: NHF 15 L/min NHF 30 L/min NHF 45 L/min Room air Completed all 4 interventions (n = 48) Analysis completed (n = 48) RIP data not analysed Excluded (n = 36) - Not meeting inclusion criteria (n = 5) On LTOT (n = 1) Current exacerbation (n = 3) No COPD (n = 1) - No further contact (n = 14) - Declined (n = 12) - Unsuitable, other reasons (n = 5) Figure 1 Participant flow through the study and allocation of interventions. LTOT, long-term oxygen therapy; NHF, nasal high flow; PIS, participant information sheet; RIP, Respiratory Inductance Plethysmography. Hg of the time point zero reading, with the longest extension being 10 min. Participant characteristics are shown in Table 1. Twenty-nine of the participants were male and 6/48 (12.5%) were hypercapnic, with PtCO 2 > 45 mm Hg, at randomization. Twenty-four participants (50%) had severe or very severe COPD according to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) classification. 12 Transcutaneous partial pressure of carbon dioxide The mean PtCO 2 adjusted for baseline after 20 min compared to room air was lower for NHF with a flowdependent reduction in PtCO 2 (Table 2). The mean difference in PtCO 2 compared to room air was 2.4 mm Hg (95% CI: 2.9 to 1.8),, 1.3 mm Hg (95% CI: 1.9 to 0.8), and 0.6 mm Hg (95% CI: 1.1 to 0.0), P = 0.06 for NHF at 45 L/min, 30 L/min and 15 L/min, respectively. There was no significant interaction between treatment response and whether the baseline PtCO 2 was greater than 45 mm Hg or not, P = The proportion of participants with at least one measurement of PtCO 2 which decreased from baseline 4 mm Hg, up to and including the 20-min treatment period was 15/48 (31.2%), 8/48 (16.7%) and 2/48 (4.2%) for NHF at 45 L/min, 30 L/min and 15 L/min, respectively, and 1/48 (2.1%) for room air. The paired proportions difference for NHF minus room air was 29.7% (95% CI: ),, 14.6% (95% CI: ), P = and 2.1% (95% CI: 2.0 to 6.1), P = 0.99, for NHF at 45 L/min, 30 L/min and 15 L/min, respectively. Four participants (8.3%) had at least one measurement of PtCO 2 which decreased from baseline 8 mm Hg, up to and including the 20-min treatment period for NHF at 45 L/min. Respiratory rate There were significant reductions in respiratory rate between NHF compared to room air at 20 min with a flow rate dependent effect (Table 3). The maximum point estimate difference in respiratory rate was 5.0 breaths per minute (95% CI: 6.2 to 3.8),, at 5 min with NHF at 45 L/min compared to room air, representing a 28% reduction from the baseline respiratory rate. Oxygen saturation and heart rate The StO 2 was higher for NHF 45 L/min compared to room air at the 5, 15 and 20 min time points. The mean maximum difference in StO 2 was 0.8% (95% CI: ),, observed after 5 min for NHF 45 L/min compared to room air (Table S1 (Supplementary Information)). Heart rate remained largely constant throughout the interventions with no statistically significant differences between any of the NHF interventions and room air, with the exception of the 20-min time point for the 15 L/min where it was 2 beats per minute higher (95% CI: ), P = (Table S2 (Supplementary Information)). The full-set of mean data for each variable is shown in the Tables S3 S6 in Supplementary Information. Minute ventilation In 97/192 (51%) interventions, the RIP measurements were valid, and of the 144 planned comparisons between NHF and room air, in only 52 (36%) were both NHF and room air measurements valid. For this reason, the RIP data is not presented. Tolerability questionnaires Participant feedback was that NHF at 45 L/min was less comfortable and noisier, but moister than NHF at 15 L/min (Table 4). NHF at 30 L/min was generally more tolerable than 45 L/min. DISCUSSION The NHF device resulted in a small flow-dependent reduction in the PtCO 2 in participants with stable COPD. There was a marked flow-dependent reduction in respiratory rate with the use of NHF. These findings suggest a favourable physiological effect with NHF in stable COPD. There are a number of methodological issues relevant to the interpretation of the study findings. Our study was single-blinded in that although participants were blinded to the actual flow rate they received, they could feel the difference between low, medium and high flows. The interventions were applied for 20 min periods, which was sufficient time to observe an effect on PtCO 2 with the maximum change usually observed at the 5-min time point. There was a washout period 2017 Asian Pacific Society of Respirology Respirology (2018) 23,
5 Nasal high flow therapy in stable COPD 381 Table 1 Baseline participant characteristics Characteristic n = 48 for all Median (IQR) Min to max Age (years) 69.4 (8.6) 70 (62 74) BMI (kg/m 2 ) 27.6 (6.7) 25.7 ( ) FEV 1 (L) 1.55 (0.64) 1.44 ( ) FEV 1 /FVC (%) 47.2 (11.5) 47.5 ( ) FEV 1 % predicted 52.5 (19.6) 49.6 ( ) FVC (L) 3.24 (0.88) 3.10 ( ) Smoking pack years 46.1 (31.2) 41.5 ( ) MMRC 1.17 (0.31) 1 (0 2) 0 3 PtCO 2 (mm Hg) 37.8 (6.1) 36.7 ( ) StO 2 (%) 94.9 (2.4) 95.0 ( ) Respiratory rate (breaths per minute) 17.8 (5.5) 16.5 ( ) Heart rate (beats per minute 74.6 (14.1) 74.5 ( ) n/48 (%) Gender male 29 (60.4) Co-morbidities Asthma 4 (8.3) Bronchiectasis 1 (2.1) Heart failure 3 (6.3) Ethnicity European 32 (66.7) Maori 9 (18.8) Other 6 (12.5) Pacific 1 (2.1) Treatment Inhaled corticosteroid 34 (70.8) Long-acting beta-agonist 34 (70.8) Long-acting muscarinic antagonist 19 (40.0) Short-acting beta-agonist 34 (70.8) Short-acting muscarinic antagonist 15 (31.3) FEV 1, forced expiratory volume in 1 s; FVC, forced vital capacity; IQR, interquartile range; MMRC, Modified Medical Research Council; PtCO 2, transcutaneous carbon dioxide, StO 2, transcutaneous oxygen saturation. which allowed each of the four intervention periods to begin within a similar baseline PtCO 2. The external validity of the findings was limited in the respect that participants with stable COPD were recruited, rather than during a severe exacerbation, in which NHF is more likely to be administered. However, this design enhanced the internal validity, allowing a cross-over design to be utilized with a single study visit, which importantly enabled a stable baseline PtCO 2 to be achieved before each intervention. It also avoided the confounding effect of supplemental oxygen use, a potential limiting factor in previous studies of NHF therapy in exacerbations of COPD, in which lower inspired concentrations of oxygen with NHF may have contributed to the reductions in PaCO 2 observed. 4 6 There was a broad cross-section of severity of COPD, with one in eight having hypercapnia and one in two having an FEV 1 < 50% predicted. A post hoc analysis showed no evidence that the change in PtCO 2 in response to treatment varied by whether the patient was in chronic hypercapnic respiratory failure. The transcutaneous SenTec monitor has been validated and used as a surrogate measure of PaCO 2, allowing continuous monitoring and the avoidance of multiple arterial blood gas punctures The RIP Respirology (2018) 23, measures were not valid for most interventions and so it was not possible to directly measure the effect of NHF on minute ventilation or tidal volume. Our observations showed that NHF reduces PtCO 2 in a flow-dependent manner complements previous work. The small reduction in PtCO 2 of 2.4 mm Hg at 45 L/ min is similar to the 3.4 mm Hg reduction with NHF at 30 L/min for 20 min in COPD patients on long-term oxygen therapy, 4 the 3.1 mm Hg reduction with NHF at 20 L/min for 45 min in COPD patients requiring supplemental oxygen at 2 L/min in hospital, 6 and the reduction of 1.4 mm Hg observed in our previous study of patients hospitalized with exacerbations of COPD, where supplemental oxygen delivered with NHF was titrated to maintain patient StO 2 at hospital pre-study levels. 7 However, it is less than the 5.2 mm Hg and 7.3 mm Hg reduction in PtCO 2 observed with 2 h of NHF treatment, at 20 L/min and 30 L/min respectively, in the uncontrolled trial of hospitalized COPD patients 8 and the non-significant 4.0 mm Hg and 5.5 mm Hg reduction in PaCO 2 in COPD patients with chronic hypercapnic respiratory failure receiving NHF therapy for 30 min at 20 L/min and 30 L/min, respectively. 9 While the mean reduction in PtCO 2 of 2.4 mm Hg found in our study is of uncertain clinical 2017 Asian Pacific Society of Respirology
6 382 S McKinstry et al. Table 2 PtCO 2 values and mixed linear models for difference in PtCO 2 of NHF minus room air adjusted for baseline (time zero) Time point (min) Air PtCO 2 (mm Hg) PtCO2 (mm Hg) NHF 15 L/min NHF 30 L/min NHF 45 L/min baseline (mm Hg) Mean (95% CI) P value PtCO 2 (mm Hg) baseline (mm Hg) Mean (95% CI) P value PtCO 2 (mm Hg) baseline (mm Hg) Mean (95% CI) P value (5.5) 37.9 (5.5) 38.0 (5.6) 38.2 (5.1) (5.3) 37.4 (5.3) 0.95 ( 1.53 to 0.37) P = (5.3) 37.6 (5.4) 0.74 ( 1.31 to 0.16) P = (5.2) 37.9 (5.3) 0.50 ( 1.08 to 0.07 P = (5.0) 38.0 (5.3) 0.55 ( 1.12 to 0.03 P = (5.9) 1.43 ( 2.00 to 0.85) 36.9 (6.1) 1.51 ( 2.09 to 0.94) 37.1 (6.0) 1.44 ( 2.01 to 0.86) 37.3 (6.0) 1.32 ( 1.90 to 0.75) 36.3 (6.0) 2.21 ( 2.78 to 1.63) 36.0 (6.1) 2.47 ( 3.05 to 1.90) 36.4 (5.9) 2.15 ( 2.72 to 1.57) 36.3 (5.6) 2.37 ( 2.94 to 1.79) NHF, nasal high flow; PtCO 2 : transcutaneous partial pressure of carbon dioxide. Table 3 Mixed linear models for difference in respiratory rate of NHF minus room air adjusted for baseline (time zero) Time points (min) Air RR (bpm) RR (bpm) NHF 15 L/min NHF 30 L/min NHF 45 L/min baseline (%) Mean (95% CI) P value RR (bpm) baseline (%) Mean (95% CI) P value RR (bpm) baseline (%) Mean (95% CI) P value (4.7) 17.6 (4.9) 18.1 (5.7) 18.3 (4.8) (4.9) 15.4 (4.9) 2.45 ( 3.65 to 1.24) (4.6) 15.0 (5.0) 1.99 ( 3.19 to 0.78) P = (4.9) 15.9 (5.3) 1.15 ( 2.36 to 0.05 P = (4.8) 16.0 (5.7) 1.47 ( 2.67 to 0.26) P = (4.8) 4.59 ( 5.79 to 3.39) 12.6 (4.4) 4.42 ( 5.63 to 3.22) 13.6 (5.2) 3.53 ( 4.73 to 2.32) 13.4 (5.2) 4.13 ( 5.34 to 2.93) 12.9 (5.5) 4.98 ( 6.19 to 3.78) 12.9 (5.7) 4.13 ( 5.33 to 2.93) 12.9 (5.0) 4.23 ( 5.44 to 3.03) 13.3 (4.8) 4.25 ( 5.46 to 3.05) bpm: breaths per minute; NHF, nasal high flow; RR, respiratory rate 2017 Asian Pacific Society of Respirology Respirology (2018) 23,
7 Nasal high flow therapy in stable COPD 383 Table 4 Mixed linear models for difference in questionnaire responses between NHF interventions. Values are on a continuous scale from most positive (0) to least positive (100) Mean difference (95% CI) P value for individual comparison Question NHF 30 L/min minus NHF 15 L/min NHF 45 L/min minus NHF 15 L/min P overall Ease of application 3.7 (0.7 to 6.7) P = Overall comfort 11.0 (4.5 to 17.4) P = Moisture in nasal passages 0.02 ( 4.3 to 4.2) P = 0.99 Noisiness 11.6 (4.1 to 19.1) P = Likelihood of reusing NHF 3.0 ( 2.9 to 9.0) P = 0.31 Weight of nasal cannula 1.5 ( 3.5 to 6.4) P = ( 0.3 to 5.8) P = (13.8 to 26.7) < ( 11.9 to 3.4) < (20.9 to 35.9) < ( 3.4 to 8.5) 0.55 P = ( 1.8 to 8.1) 0.46 P = 0.21 NHF, nasal high flow. n = 47. significance, the reduction in PtCO 2 from baseline of 8 mm Hg in 4/48 participants on NHF at 45 L/min suggests this therapy may have clinically important effects on PtCO 2 in a proportion of patients with COPD. The reduction in respiratory rate with NHF we observed has been reported in healthy volunteers, 18 in COPD patients, 4 6,19 and in other clinical situations such as pulmonary fibrosis and post-cardiac surgical patients. 5,20 The magnitude of the reduction in respiratory rate was marked with a maximum 5 breaths per minute reduction after 5 min of NHF at 45 L/min, representing a >25% reduction in respiratory rate. It has recently been reported that in patients with COPD and chronic hypercapnic respiratory failure that a reduction in respiratory rate of this magnitude with NHF therapy is associated with reduced respiratory muscle load, with a reduction in transdiaphragmatic pressures and an increase in expiratory time. 9 The flow-dependent physiological effects on PtCO 2 and respiratory rate we observed with NHF in COPD patients was consistent with the observed flowdependent increase in airway pressure, end expiratory pressures, end expiratory lung volume and inspiratory pressures observed in post-cardiac surgery patients, 20,21 and airway pressures in healthy volunteers 18 and COPD patients. 8 The modest reduction in PtCO 2 indicates alveolar ventilation was increased despite the marked reduction in respiratory rate. The relative contributions of increases in alveolar volume and/or reductions in physiological dead space to the increase in alveolar ventilation were not assessed in this study. Other mechanisms which may play a role but were not assessed in the study include an increase in tidal volume, a small positive end-expiratory pressure effect, reduction in upper airway resistance and improved mucociliary clearance from humidification of the airways. 4,8,22 24 Previous studies have shown improved tolerability of NHF compared to both face masks and standard nasal Respirology (2018) 23, prongs and it was generally well tolerated in our study. Given NHF at 30 L/min reduced PtCO 2 and respiratory rate by a similar amount to NHF at 45 L/ min, but was more comfortable, this flow rate may be preferred in clinical practice. In conclusion, NHF results in a small reduction in PtCO 2 and a marked reduction in respiratory rate, in a flow-dependent manner with the higher the flow the greater the effect. Further studies need to be undertaken to compare NHF against the gold standard of NIV in acute exacerbations of COPD associated with respiratory failure. Acknowledgements The Medical Research Institute of New Zealand is supported by Health Research Council of New Zealand Independent Research Organization Funding. S.M, J.P. and I.B. are Health Research Council of New Zealand Clinical Training Fellows. Disclosure statement The study was funded by Fisher and Paykel Healthcare New Zealand. Study design, conduct, data analysis and manuscript write-up were performed by the Medical Research Institute of New Zealand, independently of the funder. REFERENCES 1 Steer J, Gibson GJ, Bourke SC. Predicting outcomes following hospitalization for acute exacerbations of COPD. QJM 2010; 103: Davidson AC, Banham S, Elliott M, Kennedy D, Gelder C, Glossop A, Church AC, Creagh-Brown B, Dodd JW, Felton T et al. BTS/ICS guideline for the ventilatory management of acute hypercapnic respiratory failure in adults. Thorax 2016; 71(Suppl 2): ii Baudouin S, Blumenthal S, Cooper B, Davidson C, Davison A, Elliot M, Kinnear W, Paton R, Sawicka E, Turner L. Non-invasive ventilation in acute respiratory failure. Thorax 2002; 57: Fraser JF, Spooner AJ, Dunster KR, Anstey CM, Corley A. Nasal high flow oxygen therapy in patients with COPD reduces 2017 Asian Pacific Society of Respirology
8 384 S McKinstry et al. respiratory rate and tissue carbon dioxide while increasing tidal and end-expiratory lung volumes: a randomised crossover trial. Thorax 2016; 71: Braunlich J, Beyer D, Mai D, Hammerschmidt S, Seyfarth HJ, Wirtz H. Effects of nasal high flow on ventilation in volunteers, COPD and idiopathic pulmonary fibrosis patients. Respiration 2013; 85: Nilius G, Franke KJ, Domanski U, Ruhle KH, Kirkness JP, Schneider H. Effects of nasal insufflation on arterial gas exchange and breathing pattern in patients with chronic obstructive pulmonary disease and hypercapnic respiratory failure. Adv. Exp. Med. Biol. 2013; 755: Pilcher J, Eastlake L, Richards M, Power S, Cripps T, Bibby S, Braithwaite I, Weatherall M, Beasley R. Physiological effects of titrated oxygen via nasal high flow cannuale in exacerbations of COPD: a randomised controlled cross-over trial. Respirology 2017; 22: Braunlich J, Kohler M, Wirtz H. Nasal highflow improves ventilation in patients with COPD. Int. J. COPD 2016; 11: Pisani L, Fasano L, Corcione N, Comellini V, Musti MA, Brandao M, Bottone D, Calderini E, Navalesi P, Nava S. Change in pulmonary mechanics and the effect on breathing pattern of high flow oxygen therapy in stable hypercapnic COPD. Thorax 2017; 72: Miller MR, Hankinson J, Brusasco V, Burgos F, Casaburi R, Coates A, Crapo R, Enright P, van der Grinten CP, Gustafsson P et al. Standardisation of spirometry. Eur. Respir. J. 2005; 26: Edwards L, Perrin K, Williams M, Weatherall M, Beasley R. Randomised controlled crossover trial of the effect on PtCO 2 of oxygendriven versus air-driven nebulisers in severe chronic obstructive pulmonary disease. Emerg. Med. J. 2012; 29: Vogelmeier C, Criner G, Martinez F, Anzueto A, Barnes P, Bourbeau J, Celli B, Chen R, Decramer M, Fabbri L et al. Global strategy for the diagnosis, management, and prevention of Chronic Obstructive Pulmonary Disease 2017 report. GOLD executive summary. Am J Resp. Crit. Care Med. 2017; 195: Domingo C, Canturri E, Lujan M, Moreno A, Espuelas H, Marin A. Transcutaneous measurement of partial pressure of carbon dioxide and oxygen saturation: validation of the SenTec monitor. Arch. Bronconeumol. 2006; 42: Rodriguez P, Lellouche F, Aboab J, Buisson CB, Brochard L. Transcutaneous arterial carbon dioxide pressure monitoring in critically ill adult patients. Intensive Care Med. 2006; 32: Roediger R, Beck-Schimmer B, Theusinger OM, Rusch D, Seifert B, Spahn DR, Schmid ER, Baulig W. The revised digital transcutaneous PCO 2 /SpO 2 ear sensor is a reliable noninvasive monitoring tool in patients after cardiac surgery. J. Cardiothorac. Vasc. Anesth. 2011; 25: Storre JH, Magnet FS, Dreher M, Windisch W. Transcutaneous monitoring as a replacement for arterial PCO(2) monitoring during nocturnal non-invasive ventilation. Respir. Med. 2011; 105: Fingleton J, McKinstry S, Pilcher J, Weatherall M, Beasley R, Bardsley G. Accuracy of transcutaneous carbon dioxide measurement for change over time. In TSANZ oral presentations. Respirology 2017; 22: Ritchie JE, Williams AB, Gerard C, Hockey H. Evaluation of a humidified nasal high-flow oxygen system, using oxygraphy, capnography and measurement of upper airway pressures. Anaesth. Intensive Care 2011; 39: Chatila W, Nugent T, Vance G, Gaughan J, Criner GJ. The effects of high-flow vs low-flow oxygen on exercise in advanced obstructive airways disease. Chest 2004; 126: Corley A, Caruana LR, Barnett AG, Tronstad O, Fraser JF. Oxygen delivery through high-flow nasal cannulae increase end-expiratory lung volume and reduce respiratory rate in post-cardiac surgical patients. Br. J. Anaesth. 2011; 107: Parke RL, McGuinness SP. Pressures delivered by nasal high flow oxygen during all phases of the respiratory cycle. Respir. Care 2013; 58: Lee JH, Rehder KJ, Williford L, Cheifetz IM, Turner DA. Use of high flow nasal cannula in critically ill infants, children, and adults: a critical review of the literature. Intensive Care Med. 2013; 39: Nishimura M. High-flow nasal cannula oxygen therapy in adults: physiological benefits, indication, clinical benefits, and adverse effects. Respir. Care 2016; 61: Dysart K, Miller TL, Wolfson MR, Shaffer TH. Research in high flow therapy: mechanisms of action. Respir. Med. 2009; 103: Cuquemelle E, Pham T, Papon JF, Louis B, Danin PE, Brochard L. Heated and humidified high-flow oxygen therapy reduces discomfort during hypoxemic respiratory failure. Respir. Care 2012; 57: Maggiore SM, Idone FA, Vaschetto R, Festa R, Cataldo A, Antonicelli F, Montini L, De Gaetano A, Navalesi P, Antonelli M. Nasal high-flow versus Venturi mask oxygen therapy after extubation. Effects on oxygenation, comfort, and clinical outcome. Am. J. Respir. Crit. Care Med. 2014; 190: Roca O, Riera J, Torres F, Masclans JR. High-flow oxygen therapy in acute respiratory failure. Respir. Care 2010; 55: Supplementary Information Additional supplementary information can be accessed via the html version of this article at the publisher s website. Appendix S1 Methods. Table S1 Mixed linear models for Oxygen saturation difference in NHF minus room air adjusted for baseline (time zero). Table S2 Mixed linear models for Heart rate difference in NHF minus room air adjusted for baseline (time zero). Table S3 Data description for transcutaneous carbon dioxide (PtCO 2 ) by intervention and time. Table S4 Data description for respiratory rate by intervention and time. Table S5 Data description for heart rate by intervention and time. Table S6 Data description for transcutaneous oxygen saturation (StO 2 ) by intervention and time Asian Pacific Society of Respirology Respirology (2018) 23,
Oxygen therapy increases the arterial partial pressure of
 High flow or titrated oxygen for obese medical inpatients: a randomised crossover trial Janine Pilcher 1, Michael Richards 1, Leonie Eastlake 1, Steven J McKinstry 1, George Bardsley 1, Sarah Jefferies
High flow or titrated oxygen for obese medical inpatients: a randomised crossover trial Janine Pilcher 1, Michael Richards 1, Leonie Eastlake 1, Steven J McKinstry 1, George Bardsley 1, Sarah Jefferies
PRESSURES DELIVERED BY NASAL HIGH FLOW THERAPY DURING
 PRESSURES DELIVERED BY NASAL HIGH FLOW THERAPY DURING ALL PHASES OF THE RESPIRATORY CYCLE Rachael L. Parke, RN, MHSc (Hons) Cardiothoracic and Vascular Intensive Care Unit Auckland City Hospital Private
PRESSURES DELIVERED BY NASAL HIGH FLOW THERAPY DURING ALL PHASES OF THE RESPIRATORY CYCLE Rachael L. Parke, RN, MHSc (Hons) Cardiothoracic and Vascular Intensive Care Unit Auckland City Hospital Private
High Flow Humidification Therapy, Updates.
 High Flow Humidification Therapy, Updates. Bernardo Selim, M.D. I have no relevant financial relationships to disclose. Assistant Professor, Pulmonary, Critical Care and Sleep Medicine, Mayo Clinic What
High Flow Humidification Therapy, Updates. Bernardo Selim, M.D. I have no relevant financial relationships to disclose. Assistant Professor, Pulmonary, Critical Care and Sleep Medicine, Mayo Clinic What
Effectiveness of nasal highflow in hypercapnic COPD patients is flow and leakage dependent
 Bräunlich et al. BMC Pulmonary Medicine (2018) 18:14 DOI 10.1186/s12890-018-0576-x RESEARCH ARTICLE Open Access Effectiveness of nasal highflow in hypercapnic COPD patients is flow and leakage dependent
Bräunlich et al. BMC Pulmonary Medicine (2018) 18:14 DOI 10.1186/s12890-018-0576-x RESEARCH ARTICLE Open Access Effectiveness of nasal highflow in hypercapnic COPD patients is flow and leakage dependent
Nasal High Flow Humidification with or without Oxygen for COPD Management. Shereen Bailey, RCP, RRT, NPS
 Nasal High Flow Humidification with or without Oxygen for COPD Management Shereen Bailey, RCP, RRT, NPS Objectives How it works COPD Management today The role of NHFC Evidence Research/Case Studies Types
Nasal High Flow Humidification with or without Oxygen for COPD Management Shereen Bailey, RCP, RRT, NPS Objectives How it works COPD Management today The role of NHFC Evidence Research/Case Studies Types
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. This online publication has been corrected. The corrected
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. This online publication has been corrected. The corrected
Effect of Very-High-Flow Nasal Therapy on Airway Pressure and End-Expiratory Lung Impedance in Healthy Volunteers
 Effect of Very-High-Flow Nasal Therapy on Airway Pressure and End-Expiratory Lung Impedance in Healthy Volunteers Rachael L Parke RN PhD, Andreas Bloch MD, and Shay P McGuinness MB ChB BACKGROUND: Previous
Effect of Very-High-Flow Nasal Therapy on Airway Pressure and End-Expiratory Lung Impedance in Healthy Volunteers Rachael L Parke RN PhD, Andreas Bloch MD, and Shay P McGuinness MB ChB BACKGROUND: Previous
pat hways Medtech innovation briefing Published: 1 October 2018 nice.org.uk/guidance/mib161
 pat hways myairvo2 for the treatment of chronic obstructive pulmonary disease Medtech innovation briefing Published: 1 October 2018 nice.org.uk/guidance/mib161 Summary The technology described in this
pat hways myairvo2 for the treatment of chronic obstructive pulmonary disease Medtech innovation briefing Published: 1 October 2018 nice.org.uk/guidance/mib161 Summary The technology described in this
High Flow Oxygen Therapy in Acute Respiratory Failure. Laurent Brochard Toronto
 High Flow Oxygen Therapy in Acute Respiratory Failure Laurent Brochard Toronto Conflicts of interest Our clinical research laboratory has received research grants for clinical research projects from the
High Flow Oxygen Therapy in Acute Respiratory Failure Laurent Brochard Toronto Conflicts of interest Our clinical research laboratory has received research grants for clinical research projects from the
Adult Nasal High Flow: Clinical Paper Summaries
 Adult Nasal High Flow: Clinical Paper Summaries Table of Contents KEY REFERENCES THERAPY OVERVIEW Research in high flow therapy (Dysart).. S1 SPECIFIC PATIENT POPULATION High-flow nasal cannulae in post-cardiac
Adult Nasal High Flow: Clinical Paper Summaries Table of Contents KEY REFERENCES THERAPY OVERVIEW Research in high flow therapy (Dysart).. S1 SPECIFIC PATIENT POPULATION High-flow nasal cannulae in post-cardiac
pounds in the UK[2]. Most acute exacerbations are secondary to infection[3] but many are
![pounds in the UK[2]. Most acute exacerbations are secondary to infection[3] but many are pounds in the UK[2]. Most acute exacerbations are secondary to infection[3] but many are](/thumbs/92/110813590.jpg) ONLINE REPOSITORY MATERIAL INTRODUCTION Cost estimates associated with the disease are over 50 billion dollars in the USA[1] and over a billion pounds in the UK[2]. Most acute exacerbations are secondary
ONLINE REPOSITORY MATERIAL INTRODUCTION Cost estimates associated with the disease are over 50 billion dollars in the USA[1] and over a billion pounds in the UK[2]. Most acute exacerbations are secondary
Bi-Level Therapy: Boosting Comfort & Compliance in Apnea Patients
 Bi-Level Therapy: Boosting Comfort & Compliance in Apnea Patients Objectives Describe nocturnal ventilation characteristics that may indicate underlying conditions and benefits of bilevel therapy for specific
Bi-Level Therapy: Boosting Comfort & Compliance in Apnea Patients Objectives Describe nocturnal ventilation characteristics that may indicate underlying conditions and benefits of bilevel therapy for specific
What you need to know about: High flow nasal oxygen therapy
 What you need to know about: High flow nasal oxygen therapy Main introduction Adequate oxygenation is essential in many disorders, and this article will discuss the physiology, practicalities and indications
What you need to know about: High flow nasal oxygen therapy Main introduction Adequate oxygenation is essential in many disorders, and this article will discuss the physiology, practicalities and indications
Use of High-Flow Nasal Cannula for Acute Dyspnea and Hypoxemia in the Emergency Department
 Use of High-Flow Nasal Cannula for Acute Dyspnea and Hypoxemia in the Emergency Department Nuttapol Rittayamai MD, Jamsak Tscheikuna MD, Nattakarn Praphruetkit MD, and Sunthorn Kijpinyochai MD BACKGROUND:
Use of High-Flow Nasal Cannula for Acute Dyspnea and Hypoxemia in the Emergency Department Nuttapol Rittayamai MD, Jamsak Tscheikuna MD, Nattakarn Praphruetkit MD, and Sunthorn Kijpinyochai MD BACKGROUND:
Effectiveness of high-flow nasal cannula oxygen therapy for acute respiratory failure with hypercapnia
 Original Article Effectiveness of high-flow nasal cannula oxygen therapy for acute respiratory failure with hypercapnia Eun Sun Kim, Hongyeul Lee, Se Joong Kim, Jisoo Park, Yeon Joo Lee, Jong Sun Park,
Original Article Effectiveness of high-flow nasal cannula oxygen therapy for acute respiratory failure with hypercapnia Eun Sun Kim, Hongyeul Lee, Se Joong Kim, Jisoo Park, Yeon Joo Lee, Jong Sun Park,
High-Flow Nasal Cannula in a Mixed Adult ICU
 High-Flow Nasal Cannula in a Mixed Adult ICU Kristina A Gaunt MD, Sarah K Spilman MA, Meghan E Halub MD, Julie A Jackson RRT-ACCS, Keith D Lamb RRT-ACCS, and Sheryl M Sahr MD MSc BACKGROUND: Humidified,
High-Flow Nasal Cannula in a Mixed Adult ICU Kristina A Gaunt MD, Sarah K Spilman MA, Meghan E Halub MD, Julie A Jackson RRT-ACCS, Keith D Lamb RRT-ACCS, and Sheryl M Sahr MD MSc BACKGROUND: Humidified,
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
Haut debit nasal ou BiPAP? Laurent Brochard Toronto
 Haut debit nasal ou BiPAP? Laurent Brochard Toronto Conflicts of interest Our clinical research laboratory has received research grants for clinical trials from the following companies: General Electric
Haut debit nasal ou BiPAP? Laurent Brochard Toronto Conflicts of interest Our clinical research laboratory has received research grants for clinical trials from the following companies: General Electric
Rachael L Parke MHSc, Shay P McGuinness, and Michelle L Eccleston RN
 Original Research A Preliminary Randomized Controlled Trial to Assess Effectiveness of Nasal High-Flow Oxygen in Intensive Care Patients Rachael L Parke MHSc, Shay P McGuinness, and Michelle L Eccleston
Original Research A Preliminary Randomized Controlled Trial to Assess Effectiveness of Nasal High-Flow Oxygen in Intensive Care Patients Rachael L Parke MHSc, Shay P McGuinness, and Michelle L Eccleston
Emergency Medicine High Velocity Nasal Insufflation (Hi-VNI) VAPOTHERM POCKET GUIDE
 Emergency Medicine High Velocity Nasal Insufflation (Hi-VNI) VAPOTHERM POCKET GUIDE Indications for Vapotherm High Velocity Nasal Insufflation (Hi-VNI ) administration, the patient should be: Spontaneously
Emergency Medicine High Velocity Nasal Insufflation (Hi-VNI) VAPOTHERM POCKET GUIDE Indications for Vapotherm High Velocity Nasal Insufflation (Hi-VNI ) administration, the patient should be: Spontaneously
High-Flow Nasal Cannula in a Mixed Adult ICU
 High-Flow Nasal Cannula in a Mixed Adult ICU Kristina A Gaunt MD, Sarah K Spilman MA, Meghan E Halub MD, Julie A Jackson RRT-ACCS, Keith D Lamb RRT-ACCS, and Sheryl M Sahr MD MSc BACKGROUND: Humidified,
High-Flow Nasal Cannula in a Mixed Adult ICU Kristina A Gaunt MD, Sarah K Spilman MA, Meghan E Halub MD, Julie A Jackson RRT-ACCS, Keith D Lamb RRT-ACCS, and Sheryl M Sahr MD MSc BACKGROUND: Humidified,
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
Evaluation of Effect of Breathe Ventilation System on Work of Breathing in COPD patients. Matthew Cohn, M.D.
 Evaluation of Effect of Breathe Ventilation System on Work of Breathing in COPD patients Matthew Cohn, M.D. 1 11/4/2013 Disclosure Slide- Matthew Cohn, M.D. Personal financial relationships with commercial
Evaluation of Effect of Breathe Ventilation System on Work of Breathing in COPD patients Matthew Cohn, M.D. 1 11/4/2013 Disclosure Slide- Matthew Cohn, M.D. Personal financial relationships with commercial
Decramer 2014 a &b [21]
![Decramer 2014 a &b [21] Decramer 2014 a &b [21]](/thumbs/90/101390504.jpg) Buhl 2015 [19] Celli 2014 [20] Decramer 2014 a &b [21] D Urzo 2014 [22] Maleki-Yazdi 2014 [23] Inclusion criteria: Diagnosis of chronic obstructive pulmonary disease; 40 years of age or older; Relatively
Buhl 2015 [19] Celli 2014 [20] Decramer 2014 a &b [21] D Urzo 2014 [22] Maleki-Yazdi 2014 [23] Inclusion criteria: Diagnosis of chronic obstructive pulmonary disease; 40 years of age or older; Relatively
Improving Care & Outcomes
 Improving Care & Outcomes Macquarie Technology Day, 20 October 2011 1 Improving Care & Outcomes The Care Continuum - Matthew Payton F&P Optiflow - Matthew Payton F&P Info Technologies - Lewis Gradon ICON
Improving Care & Outcomes Macquarie Technology Day, 20 October 2011 1 Improving Care & Outcomes The Care Continuum - Matthew Payton F&P Optiflow - Matthew Payton F&P Info Technologies - Lewis Gradon ICON
Títle: Efficacy of high-flow oxygen and active humidification in a patient with acute respiratory failure of neuromuscular origin.
 Títle: Efficacy of high-flow oxygen and active humidification in a patient with acute respiratory failure of neuromuscular origin. Authors: Díaz- Lobato S, MD, PhD; Folgado MA*, MD; Chapa A*, MD; Mayoralas
Títle: Efficacy of high-flow oxygen and active humidification in a patient with acute respiratory failure of neuromuscular origin. Authors: Díaz- Lobato S, MD, PhD; Folgado MA*, MD; Chapa A*, MD; Mayoralas
Ron Hosp, MS-HSA, RRT Regional Respiratory Specialist. This program has been approved for 1 hour of continuing education credit.
 Ron Hosp, MS-HSA, RRT Regional Respiratory Specialist This program has been approved for 1 hour of continuing education credit. Course Objectives Identify at least four goals of home NIV Identify candidates
Ron Hosp, MS-HSA, RRT Regional Respiratory Specialist This program has been approved for 1 hour of continuing education credit. Course Objectives Identify at least four goals of home NIV Identify candidates
SenTec Digital Monitoring System Transcutaneous PCO2 monitoring of patients with chronic respiratory failure PCO2 SpO2 PR
 Digital Transcutaneous Blood Gas Monitoring SenTec Digital Monitoring System Transcutaneous PCO2 monitoring of patients with chronic respiratory failure PCO2 SpO2 PR Continuous Noninvasive Accurate Safe
Digital Transcutaneous Blood Gas Monitoring SenTec Digital Monitoring System Transcutaneous PCO2 monitoring of patients with chronic respiratory failure PCO2 SpO2 PR Continuous Noninvasive Accurate Safe
Average volume-assured pressure support
 Focused review Average volume-assured pressure support Abdurahim Aloud MD Abstract Average volume-assured pressure support (AVAPS) is a relatively new mode of noninvasive positive pressure ventilation
Focused review Average volume-assured pressure support Abdurahim Aloud MD Abstract Average volume-assured pressure support (AVAPS) is a relatively new mode of noninvasive positive pressure ventilation
SGRQ Questionnaire assessing respiratory disease-specific quality of life. Questionnaire assessing general quality of life
 SUPPLEMENTARY MATERIAL e-table 1: Outcomes studied in present analysis. Outcome Abbreviation Definition Nature of data, direction indicating adverse effect (continuous only) Clinical outcomes- subjective
SUPPLEMENTARY MATERIAL e-table 1: Outcomes studied in present analysis. Outcome Abbreviation Definition Nature of data, direction indicating adverse effect (continuous only) Clinical outcomes- subjective
Supplementary Online Content
 Supplementary Online Content Regan EA, Lynch DA, Curran-Everett D, et al; Genetic Epidemiology of COPD (COPDGene) Investigators. Clinical and radiologic disease in smokers with normal spirometry. Published
Supplementary Online Content Regan EA, Lynch DA, Curran-Everett D, et al; Genetic Epidemiology of COPD (COPDGene) Investigators. Clinical and radiologic disease in smokers with normal spirometry. Published
Patients with COPD are frequently limited in their. The Effects of High-Flow vs Low-Flow Oxygen on Exercise in Advanced Obstructive Airways Disease*
 The Effects of High-Flow vs Low-Flow Oxygen on Exercise in Advanced Obstructive Airways Disease* Wissam Chatila, MD; Tom Nugent, MD; Gwendolyn Vance, RN; John Gaughan, PhD; and Gerard J. Criner, MD Study
The Effects of High-Flow vs Low-Flow Oxygen on Exercise in Advanced Obstructive Airways Disease* Wissam Chatila, MD; Tom Nugent, MD; Gwendolyn Vance, RN; John Gaughan, PhD; and Gerard J. Criner, MD Study
Clinical and radiographic predictors of GOLD-Unclassified smokers in COPDGene
 Clinical and radiographic predictors of GOLD-Unclassified smokers in COPDGene Emily S. Wan, John E. Hokanson, James R. Murphy, Elizabeth A. Regan, Barry J. Make, David A. Lynch, James D. Crapo, Edwin K.
Clinical and radiographic predictors of GOLD-Unclassified smokers in COPDGene Emily S. Wan, John E. Hokanson, James R. Murphy, Elizabeth A. Regan, Barry J. Make, David A. Lynch, James D. Crapo, Edwin K.
NIV in Acute Respiratory Failure: Where we fail? Dr Shrikanth Srinivasan MD,DNB,FNB,EDIC Consultant, Critical Care Medicine Medanta, The Medicity
 NIV in Acute Respiratory Failure: Where we fail? Dr Shrikanth Srinivasan MD,DNB,FNB,EDIC Consultant, Critical Care Medicine Medanta, The Medicity Use of NIV 1998-2010 50 45 40 35 30 25 20 15 10 5 0 1998
NIV in Acute Respiratory Failure: Where we fail? Dr Shrikanth Srinivasan MD,DNB,FNB,EDIC Consultant, Critical Care Medicine Medanta, The Medicity Use of NIV 1998-2010 50 45 40 35 30 25 20 15 10 5 0 1998
This is a cross-sectional analysis of the National Health and Nutrition Examination
 SUPPLEMENTAL METHODS Study Design and Setting This is a cross-sectional analysis of the National Health and Nutrition Examination Survey (NHANES) data 2007-2008, 2009-2010, and 2011-2012. The NHANES is
SUPPLEMENTAL METHODS Study Design and Setting This is a cross-sectional analysis of the National Health and Nutrition Examination Survey (NHANES) data 2007-2008, 2009-2010, and 2011-2012. The NHANES is
A study of non-invasive ventilation in acute respiratory failure
 Original Research Article A study of non-invasive ventilation in acute respiratory failure Nilima Manohar Mane 1, Jayant L. Pednekar 2, Sangeeta Pednekar 3* 1 Consultant Physician and Diabetologist, Apollo
Original Research Article A study of non-invasive ventilation in acute respiratory failure Nilima Manohar Mane 1, Jayant L. Pednekar 2, Sangeeta Pednekar 3* 1 Consultant Physician and Diabetologist, Apollo
The Use of Active Cycle of Breathing Technique (ACBT) In Pulmonary Physiotherapy: A Critical Review of the Literature Lauro G. Villegas Jr.
 The Use of Active Cycle of Breathing Technique (ACBT) In Pulmonary Physiotherapy: A Critical Review of the Literature Lauro G. Villegas Jr., PTRP Keywords: Active Cycle of Breathing Technique (ACBT), Pulmonary
The Use of Active Cycle of Breathing Technique (ACBT) In Pulmonary Physiotherapy: A Critical Review of the Literature Lauro G. Villegas Jr., PTRP Keywords: Active Cycle of Breathing Technique (ACBT), Pulmonary
Randomised controlled trial of high concentration versus titrated oxygen therapy in severe exacerbations of asthma
 See Editorial, p 931 < Additional tables are published online only. To view these files please visit the journal online (http://thorax.bmj. com). 1 Medical Research Institute of New Zealand, Wellington,
See Editorial, p 931 < Additional tables are published online only. To view these files please visit the journal online (http://thorax.bmj. com). 1 Medical Research Institute of New Zealand, Wellington,
Georg Nilius, Karl-Josef Franke, Ulrike Domanski, Karl-Heinz Rühle, Jason P. Kirkness, and Hartmut Schneider
 Chapter 4 Effects of Nasal Insuf fl ation on Arterial Gas Exchange and Breathing Pattern in Patients with Chronic Obstructive Pulmonary Disease and Hypercapnic Respiratory Failure Georg Nilius, Karl-Josef
Chapter 4 Effects of Nasal Insuf fl ation on Arterial Gas Exchange and Breathing Pattern in Patients with Chronic Obstructive Pulmonary Disease and Hypercapnic Respiratory Failure Georg Nilius, Karl-Josef
Supplementary Online Content
 Supplementary Online Content Murphy PB, Rehal S, Arbane G, et al. Effect of home noninvasive ventilation with oxygen therapy vs oxygen therapy alone on hospital readmission or death after an acute COPD
Supplementary Online Content Murphy PB, Rehal S, Arbane G, et al. Effect of home noninvasive ventilation with oxygen therapy vs oxygen therapy alone on hospital readmission or death after an acute COPD
2/4/2019. GOLD Objectives. GOLD 2019 Report: Chapters
 GOLD Objectives To provide a non biased review of the current evidence for the assessment, diagnosis and treatment of patients with COPD. To highlight short term and long term treatment objectives organized
GOLD Objectives To provide a non biased review of the current evidence for the assessment, diagnosis and treatment of patients with COPD. To highlight short term and long term treatment objectives organized
BiPAPS/TVAPSCPAPASV???? Lori Davis, B.Sc., R.C.P.T.(P), RPSGT
 BiPAPS/TVAPSCPAPASV???? Lori Davis, B.Sc., R.C.P.T.(P), RPSGT Modes Continuous Positive Airway Pressure (CPAP): One set pressure which is the same on inspiration and expiration Auto-PAP (APAP) - Provides
BiPAPS/TVAPSCPAPASV???? Lori Davis, B.Sc., R.C.P.T.(P), RPSGT Modes Continuous Positive Airway Pressure (CPAP): One set pressure which is the same on inspiration and expiration Auto-PAP (APAP) - Provides
Intensive Care Medicine Experimental. Yu Onodera * , Ryo Akimoto, Hiroto Suzuki, Masayuki Okada, Masaki Nakane and Kaneyuki Kawamae
 Onodera et al. Intensive Care Medicine Experimental (2018) 6:7 https://doi.org/10.1186/s40635-018-0172-7 Intensive Care Medicine Experimental RESEARCH Open Access A high-flow nasal cannula system with
Onodera et al. Intensive Care Medicine Experimental (2018) 6:7 https://doi.org/10.1186/s40635-018-0172-7 Intensive Care Medicine Experimental RESEARCH Open Access A high-flow nasal cannula system with
POLICY. Number: Title: APPLICATION OF NON INVASIVE VENTILATION FOR ACUTE RESPIRATORY FAILURE. Authorization
 POLICY Number: 7311-60-024 Title: APPLICATION OF NON INVASIVE VENTILATION FOR ACUTE RESPIRATORY FAILURE Authorization [ ] President and CEO [ x ] Vice President, Finance and Corporate Services Source:
POLICY Number: 7311-60-024 Title: APPLICATION OF NON INVASIVE VENTILATION FOR ACUTE RESPIRATORY FAILURE Authorization [ ] President and CEO [ x ] Vice President, Finance and Corporate Services Source:
Keeping Patients Off the Vent: Bilevel, HFNC, Neither?
 Keeping Patients Off the Vent: Bilevel, HFNC, Neither? Robert Kempainen, MD Pulmonary and Critical Care Medicine Hennepin County Medical Center University of Minnesota School of Medicine Objectives Summarize
Keeping Patients Off the Vent: Bilevel, HFNC, Neither? Robert Kempainen, MD Pulmonary and Critical Care Medicine Hennepin County Medical Center University of Minnesota School of Medicine Objectives Summarize
Web Appendix 1: Literature search strategy. BTS Acute Hypercapnic Respiratory Failure (AHRF) write-up. Sources to be searched for the guidelines;
 Web Appendix 1: Literature search strategy BTS Acute Hypercapnic Respiratory Failure (AHRF) write-up Sources to be searched for the guidelines; Cochrane Database of Systematic Reviews (CDSR) Database of
Web Appendix 1: Literature search strategy BTS Acute Hypercapnic Respiratory Failure (AHRF) write-up Sources to be searched for the guidelines; Cochrane Database of Systematic Reviews (CDSR) Database of
Comparison of two humidification systems for long-term noninvasive mechanical ventilation
 Eur Respir J 2008; 32: 460 464 DOI: 10.1183/09031936.00000208 CopyrightßERS Journals Ltd 2008 Comparison of two humidification systems for long-term noninvasive mechanical ventilation S. Nava*, S. Cirio*,
Eur Respir J 2008; 32: 460 464 DOI: 10.1183/09031936.00000208 CopyrightßERS Journals Ltd 2008 Comparison of two humidification systems for long-term noninvasive mechanical ventilation S. Nava*, S. Cirio*,
Concerns and Controversial Issues in NPPV. Concerns and Controversial Issues in Noninvasive Positive Pressure Ventilation
 : Common Therapy in Daily Practice Concerns and Controversial Issues in Noninvasive Positive Pressure Ventilation Rongchang Chen Guangzhou Institute of Respiratory Disease as the first choice of mechanical
: Common Therapy in Daily Practice Concerns and Controversial Issues in Noninvasive Positive Pressure Ventilation Rongchang Chen Guangzhou Institute of Respiratory Disease as the first choice of mechanical
Chapter. Diffusion capacity and BMPR2 mutations in pulmonary arterial hypertension
 Chapter 7 Diffusion capacity and BMPR2 mutations in pulmonary arterial hypertension P. Trip B. Girerd H.J. Bogaard F.S. de Man A. Boonstra G. Garcia M. Humbert D. Montani A. Vonk Noordegraaf Eur Respir
Chapter 7 Diffusion capacity and BMPR2 mutations in pulmonary arterial hypertension P. Trip B. Girerd H.J. Bogaard F.S. de Man A. Boonstra G. Garcia M. Humbert D. Montani A. Vonk Noordegraaf Eur Respir
BTS Guideline for Home Oxygen use in adults Appendix 9 (online only) Key Questions - PICO 10 December 2012
 BTS Guideline for Home Oxygen use in adults Appendix 9 (online only) Key Questions - PICO 10 December 2012 Evidence base for Home Oxygen therapy in COPD, non-copd respiratory disease and nonrespiratory
BTS Guideline for Home Oxygen use in adults Appendix 9 (online only) Key Questions - PICO 10 December 2012 Evidence base for Home Oxygen therapy in COPD, non-copd respiratory disease and nonrespiratory
Chronic Obstructive Pulmonary Disease Guidelines and updates
 Chronic Obstructive Pulmonary Disease Guidelines and updates October 20, 2018 Saratoga Springs, NY COPD (Chronic obstructive pulmonary disease) is a major cause of mortality and morbidity in the United
Chronic Obstructive Pulmonary Disease Guidelines and updates October 20, 2018 Saratoga Springs, NY COPD (Chronic obstructive pulmonary disease) is a major cause of mortality and morbidity in the United
AECOPD: Management and Prevention
 Neil MacIntyre MD Duke University Medical Center Durham NC Professor P.J. Barnes, MD, National Heart and Lung Institute, London UK Professor Peter J. Barnes, MD National Heart and Lung Institute, London
Neil MacIntyre MD Duke University Medical Center Durham NC Professor P.J. Barnes, MD, National Heart and Lung Institute, London UK Professor Peter J. Barnes, MD National Heart and Lung Institute, London
IICU Staff Meeting Minutes May 15 and 16, 2013 IICU Conference Room
 IICU Staff Meeting Minutes May 15 and 16, 2013 IICU Conference Room 1) Decreasing Telemetry Alarms Janice Marlett, BSN, RN, Nursing Staff Educator To decrease tele alarms: Properly prep the skin Shave
IICU Staff Meeting Minutes May 15 and 16, 2013 IICU Conference Room 1) Decreasing Telemetry Alarms Janice Marlett, BSN, RN, Nursing Staff Educator To decrease tele alarms: Properly prep the skin Shave
Respiratory Medicine. Some pet peeves and other random topics. Kyle Perrin
 Respiratory Medicine Some pet peeves and other random topics Kyle Perrin Overview 1. Acute asthma Severity assessment and management 2. Acute COPD NIV and other management 3. Respiratory problems in the
Respiratory Medicine Some pet peeves and other random topics Kyle Perrin Overview 1. Acute asthma Severity assessment and management 2. Acute COPD NIV and other management 3. Respiratory problems in the
Non-Invasive PCO 2 Monitoring in Infants Hospitalized with Viral Bronchiolitis
 Non-Invasive PCO 2 Monitoring in Infants Hospitalized with Viral Bronchiolitis Gal S, Riskin A, Chistyakov I, Shifman N, Srugo I, and Kugelman A Pediatric Department and Pediatric Pulmonary Unit Bnai Zion
Non-Invasive PCO 2 Monitoring in Infants Hospitalized with Viral Bronchiolitis Gal S, Riskin A, Chistyakov I, Shifman N, Srugo I, and Kugelman A Pediatric Department and Pediatric Pulmonary Unit Bnai Zion
Efficacy of High-Flow Nasal Cannula Therapy in Acute Hypoxemic Respiratory Failure: Decreased Use of Mechanical Ventilation
 Efficacy of High-Flow Nasal Cannula Therapy in Acute Hypoxemic Respiratory Failure: Decreased Use of Mechanical Kazuma Nagata MD, Takeshi Morimoto MD PhD MPH, Daichi Fujimoto MD, Takehiro Otoshi MD, Atsushi
Efficacy of High-Flow Nasal Cannula Therapy in Acute Hypoxemic Respiratory Failure: Decreased Use of Mechanical Kazuma Nagata MD, Takeshi Morimoto MD PhD MPH, Daichi Fujimoto MD, Takehiro Otoshi MD, Atsushi
Respiratory Pathophysiology Cases Linda Costanzo Ph.D.
 Respiratory Pathophysiology Cases Linda Costanzo Ph.D. I. Case of Pulmonary Fibrosis Susan was diagnosed 3 years ago with diffuse interstitial pulmonary fibrosis. She tries to continue normal activities,
Respiratory Pathophysiology Cases Linda Costanzo Ph.D. I. Case of Pulmonary Fibrosis Susan was diagnosed 3 years ago with diffuse interstitial pulmonary fibrosis. She tries to continue normal activities,
Identification and Treatment of the Patient with Sleep Related Hypoventilation
 Identification and Treatment of the Patient with Sleep Related Hypoventilation Hillary Loomis-King, MD Pulmonary and Critical Care of NW MI Munson Sleep Disorders Center X Conflict of Interest Disclosures
Identification and Treatment of the Patient with Sleep Related Hypoventilation Hillary Loomis-King, MD Pulmonary and Critical Care of NW MI Munson Sleep Disorders Center X Conflict of Interest Disclosures
Disclosure. Learning Objectives. Bernadette Zelaya, RRT. Area Clinical Manager
 High Velocity Nasal Insufflation An Important Therapeutic Approach for Use in the Emergency Department Presented by Vapotherm Accredited for 1 CEU by the American Association for Respiratory Care Provider
High Velocity Nasal Insufflation An Important Therapeutic Approach for Use in the Emergency Department Presented by Vapotherm Accredited for 1 CEU by the American Association for Respiratory Care Provider
Over the last several years various national and
 Recommendations for the Management of COPD* Gary T. Ferguson, MD, FCCP Three sets of guidelines for the management of COPD that are widely recognized (from the European Respiratory Society [ERS], American
Recommendations for the Management of COPD* Gary T. Ferguson, MD, FCCP Three sets of guidelines for the management of COPD that are widely recognized (from the European Respiratory Society [ERS], American
Citation for published version (APA): Westerman, E. M. (2009). Studies on antibiotic aerosols for inhalation in cystic fibrosis s.n.
 University of Groningen Studies on antibiotic aerosols for inhalation in cystic fibrosis Westerman, Elisabeth Mechteld IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF)
University of Groningen Studies on antibiotic aerosols for inhalation in cystic fibrosis Westerman, Elisabeth Mechteld IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF)
Chronic obstructive pulmonary disease
 0 Chronic obstructive pulmonary disease Implementing NICE guidance June 2010 NICE clinical guideline 101 What this presentation covers Background Scope Key priorities for implementation Discussion Find
0 Chronic obstructive pulmonary disease Implementing NICE guidance June 2010 NICE clinical guideline 101 What this presentation covers Background Scope Key priorities for implementation Discussion Find
High-flow nasal cannula use in a paediatric intensive care unit over 3 years
 High-flow nasal cannula use in a paediatric intensive care unit over 3 years Tracey I Wraight and Subodh S Ganu Respiratory illness and/or distress is the commonest reason for non-elective paediatric intensive
High-flow nasal cannula use in a paediatric intensive care unit over 3 years Tracey I Wraight and Subodh S Ganu Respiratory illness and/or distress is the commonest reason for non-elective paediatric intensive
Chieko Mitaka 1*, Masahiko Odoh 1, Daizoh Satoh 1, Tadasuke Hashiguchi 2 and Eiichi Inada 1
 Mitaka et al. Journal of Medical Case Reports (2018) 12:292 https://doi.org/10.1186/s13256-018-1832-7 CASE REPORT Open Access High-flow oxygen via tracheostomy facilitates weaning from prolonged mechanical
Mitaka et al. Journal of Medical Case Reports (2018) 12:292 https://doi.org/10.1186/s13256-018-1832-7 CASE REPORT Open Access High-flow oxygen via tracheostomy facilitates weaning from prolonged mechanical
Study No.: Title: Rationale: Phase: Study Period Study Design: Centres: Indication: Treatment: Objectives : Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Surveillance report Published: 6 April 2016 nice.org.uk. NICE All rights reserved.
 Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Keywords: Non-invasive mechanical ventilation, Respiratory Failure, Respiratory muscles, Hypercapnia, Breathing pattern.
 Monaldi Arch Chest Dis 2004; 61: 2, 81-85 ORIGINAL ARTICLE Inspiratory muscle workload due to dynamic intrinsic PEEP in stable COPD patients: effects of two different settings of non-invasive pressure-support
Monaldi Arch Chest Dis 2004; 61: 2, 81-85 ORIGINAL ARTICLE Inspiratory muscle workload due to dynamic intrinsic PEEP in stable COPD patients: effects of two different settings of non-invasive pressure-support
Oxygen: Is there a problem? Tom Heaps Acute Physician
 Oxygen: Is there a problem? Tom Heaps Acute Physician Case 1 79-year-old female, diabetic, morbidly obese Admitted with LVF Overnight Reduced GCS?cause 15l NRB in situ ABG showed ph 6.9, pco 2 15.9kPa
Oxygen: Is there a problem? Tom Heaps Acute Physician Case 1 79-year-old female, diabetic, morbidly obese Admitted with LVF Overnight Reduced GCS?cause 15l NRB in situ ABG showed ph 6.9, pco 2 15.9kPa
GE Healthcare. Non Invasive Ventilation (NIV) For the Engström Ventilator. Relief, Relax, Recovery
 GE Healthcare Non Invasive Ventilation (NIV) For the Engström Ventilator Relief, Relax, Recovery COPD is currently the fourth leading cause of death in the world, and further increases in the prevalence
GE Healthcare Non Invasive Ventilation (NIV) For the Engström Ventilator Relief, Relax, Recovery COPD is currently the fourth leading cause of death in the world, and further increases in the prevalence
Chronic Obstructive Pulmonary Disease (COPD) Measures Document
 Chronic Obstructive Pulmonary Disease (COPD) Measures Document COPD Version: 3 - covering patients discharged between 01/10/2017 and present. Programme Lead: Jo Higgins Clinical Lead: Dr Paul Albert Number
Chronic Obstructive Pulmonary Disease (COPD) Measures Document COPD Version: 3 - covering patients discharged between 01/10/2017 and present. Programme Lead: Jo Higgins Clinical Lead: Dr Paul Albert Number
Chronic Obstructive Pulmonary Diseases:
 313 High Intensity Non-Invasive Ventilation for Hypercapnic COPD Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Original Research High Intensity Non-Invasive Positive Pressure Ventilation
313 High Intensity Non-Invasive Ventilation for Hypercapnic COPD Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Original Research High Intensity Non-Invasive Positive Pressure Ventilation
Dr Conroy Wong. Professor Richard Beasley. Dr Sarah Mooney. Professor Innes Asher
 Professor Richard Beasley University of Otago Director Medical Research Institute of New Zealand Wellington Dr Sarah Mooney Physiotherapy Advanced Clinician Counties Manukau Health NZ Respiratory and Sleep
Professor Richard Beasley University of Otago Director Medical Research Institute of New Zealand Wellington Dr Sarah Mooney Physiotherapy Advanced Clinician Counties Manukau Health NZ Respiratory and Sleep
CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) DEFINITION
 CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) DEFINITION Method of maintaining low pressure distension of lungs during inspiration and expiration when infant breathing spontaneously Benefits Improves oxygenation
CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) DEFINITION Method of maintaining low pressure distension of lungs during inspiration and expiration when infant breathing spontaneously Benefits Improves oxygenation
Policy Specific Section: October 1, 2010 January 21, 2013
 Medical Policy Bi-level Positive Airway Pressure (BPAP/NPPV) Type: Medical Necessity/Not Medical Necessity Policy Specific Section: Durable Medical Equipment Original Policy Date: Effective Date: October
Medical Policy Bi-level Positive Airway Pressure (BPAP/NPPV) Type: Medical Necessity/Not Medical Necessity Policy Specific Section: Durable Medical Equipment Original Policy Date: Effective Date: October
Reasons Providers Use Bilevel
 Reasons Providers Use Bilevel More comfort, improve therapy compliance Noncompliant OSA (NCOSA) 1 Scripts from lab referrals Central/Complex Sleep Apnea 2 For ventilations needs Restrictive Thoracic Disorders/Neuromuscular
Reasons Providers Use Bilevel More comfort, improve therapy compliance Noncompliant OSA (NCOSA) 1 Scripts from lab referrals Central/Complex Sleep Apnea 2 For ventilations needs Restrictive Thoracic Disorders/Neuromuscular
Page 1. Nasal High Flow Reduces Dead Space. Research, Max-Lebsche-Platz 31, Munich, Germany
 Articles in PresS. J Appl Physiol (November 17, 2016). doi:10.1152/japplphysiol.00584.2016 Page 1 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 Nasal High Flow
Articles in PresS. J Appl Physiol (November 17, 2016). doi:10.1152/japplphysiol.00584.2016 Page 1 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 Nasal High Flow
Systems differ in their ability to deliver optimal humidification
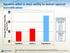 Average Absolute Humidity (mg H 2 O/L) Systems differ in their ability to deliver optimal humidification 45 Flows Tested 40 35 30 Optiflow Airvo 2 Vapotherm Vapotherm 5 L/min 10L/min 20L/min 30L/min 40L/min
Average Absolute Humidity (mg H 2 O/L) Systems differ in their ability to deliver optimal humidification 45 Flows Tested 40 35 30 Optiflow Airvo 2 Vapotherm Vapotherm 5 L/min 10L/min 20L/min 30L/min 40L/min
TORCH: Salmeterol and Fluticasone Propionate and Survival in COPD
 TORCH: and Propionate and Survival in COPD April 19, 2007 Justin Lee Pharmacy Resident University Health Network Outline Overview of COPD Pathophysiology Pharmacological Treatment Overview of the TORCH
TORCH: and Propionate and Survival in COPD April 19, 2007 Justin Lee Pharmacy Resident University Health Network Outline Overview of COPD Pathophysiology Pharmacological Treatment Overview of the TORCH
Increased difference between slow and forced vital capacity is associated with reduced exercise tolerance in COPD patients
 Yuan et al. BMC Pulmonary Medicine 2014, 14:16 RESEARCH ARTICLE Open Access Increased difference between slow and forced vital capacity is associated with reduced exercise tolerance in COPD patients Wei
Yuan et al. BMC Pulmonary Medicine 2014, 14:16 RESEARCH ARTICLE Open Access Increased difference between slow and forced vital capacity is associated with reduced exercise tolerance in COPD patients Wei
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Calverley P M A, Anzueto A R, Carter K, et
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Calverley P M A, Anzueto A R, Carter K, et
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 DRAFT NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Chronic obstructive pulmonary disease: the management of adults with chronic obstructive pulmonary disease in primary
DRAFT NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Chronic obstructive pulmonary disease: the management of adults with chronic obstructive pulmonary disease in primary
PFT Interpretation and Reference Values
 PFT Interpretation and Reference Values September 21, 2018 Eric Wong Objectives Understand the components of PFT Interpretation of PFT Clinical Patterns How to choose Reference Values 3 Components Spirometry
PFT Interpretation and Reference Values September 21, 2018 Eric Wong Objectives Understand the components of PFT Interpretation of PFT Clinical Patterns How to choose Reference Values 3 Components Spirometry
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Chronic obstructive pulmonary disease: the management of adults with chronic obstructive pulmonary disease in primary and secondary
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Chronic obstructive pulmonary disease: the management of adults with chronic obstructive pulmonary disease in primary and secondary
Relevant Papers: eight relevant articles were found, but four were reviewed because they were most directly related to the topic
 Topic: Prehospital use of bromide paired with salbutamol as treatment for shortness of breath. Author: Lisa Henderson Clinical Scenario: Two primary care paramedics respond code 4 for a 55 year old male
Topic: Prehospital use of bromide paired with salbutamol as treatment for shortness of breath. Author: Lisa Henderson Clinical Scenario: Two primary care paramedics respond code 4 for a 55 year old male
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Oxygen & High flow nasal Oxygen therapy. Learning points. Why? 18/07/
 Oxygen & High flow nasal Oxygen therapy 13.07.2017 Learning points Update on BTS guidance May 2017 Help you understand the mechanism of action of high flow nasal oxygen therapy Help you think about the
Oxygen & High flow nasal Oxygen therapy 13.07.2017 Learning points Update on BTS guidance May 2017 Help you understand the mechanism of action of high flow nasal oxygen therapy Help you think about the
Capnography 101. James A Temple BA, NRP, CCP
 Capnography 101 James A Temple BA, NRP, CCP Expected Outcomes 1. Gain a working knowledge of the physiology and science behind End-Tidal CO2. 2.Relate End-Tidal CO2 to ventilation, perfusion, and metabolism.
Capnography 101 James A Temple BA, NRP, CCP Expected Outcomes 1. Gain a working knowledge of the physiology and science behind End-Tidal CO2. 2.Relate End-Tidal CO2 to ventilation, perfusion, and metabolism.
The Role of Facemask Spirometry in Motor Neurone Disease (MND) Respiratory Sleep and Support Centre, Papworth Hospital NHS Trust, Papworth
 The Role of Facemask Spirometry in Motor Neurone Disease (MND) Banerjee SK 1, Davies MG 1, Sharples LD 2, Smith IE 1 1 Respiratory Sleep and Support Centre, Papworth Hospital NHS Trust, Papworth Everard,
The Role of Facemask Spirometry in Motor Neurone Disease (MND) Banerjee SK 1, Davies MG 1, Sharples LD 2, Smith IE 1 1 Respiratory Sleep and Support Centre, Papworth Hospital NHS Trust, Papworth Everard,
High-flow nasal oxygen therapy and noninvasive ventilation in the management of acute hypoxemic respiratory failure
 Review Article Page 1 of 8 High-flow nasal oxygen therapy and noninvasive ventilation in the management of acute hypoxemic respiratory failure Jean-Pierre Frat 1,2,3, Rémi Coudroy 1,2,3, Nicolas Marjanovic
Review Article Page 1 of 8 High-flow nasal oxygen therapy and noninvasive ventilation in the management of acute hypoxemic respiratory failure Jean-Pierre Frat 1,2,3, Rémi Coudroy 1,2,3, Nicolas Marjanovic
This is the publisher s version. This version is defined in the NISO recommended practice RP
 Journal Article Version This is the publisher s version. This version is defined in the NISO recommended practice RP-8-2008 http://www.niso.org/publications/rp/ Suggested Reference Chong, J., Karner, C.,
Journal Article Version This is the publisher s version. This version is defined in the NISO recommended practice RP-8-2008 http://www.niso.org/publications/rp/ Suggested Reference Chong, J., Karner, C.,
COPD is a syndrome of chronic limitation in expiratory airflow encompassing emphysema or chronic bronchitis.
 1 Definition of COPD: COPD is a syndrome of chronic limitation in expiratory airflow encompassing emphysema or chronic bronchitis. Airflow obstruction may be accompanied by airway hyper-responsiveness
1 Definition of COPD: COPD is a syndrome of chronic limitation in expiratory airflow encompassing emphysema or chronic bronchitis. Airflow obstruction may be accompanied by airway hyper-responsiveness
What is the next best step?
 Noninvasive Ventilation William Janssen, M.D. Assistant Professor of Medicine National Jewish Health University of Colorado Denver Health Sciences Center What is the next best step? 65 year old female
Noninvasive Ventilation William Janssen, M.D. Assistant Professor of Medicine National Jewish Health University of Colorado Denver Health Sciences Center What is the next best step? 65 year old female
COPD Challenge CASE PRESENTATION
 Chronic obstructive pulmonary disease (COPD) exacerbations may make up more than 10% of acute medical admissions [1], and they are increasingly recognised as a cause of significant morbidity and mortality
Chronic obstructive pulmonary disease (COPD) exacerbations may make up more than 10% of acute medical admissions [1], and they are increasingly recognised as a cause of significant morbidity and mortality
Long-term non-invasive ventilation in cystic fibrosis Experience over two decades
 Journal of Cystic Fibrosis 11 (2012) 187 192 www.elsevier.com/locate/jcf Original Article Long-term non-invasive ventilation in cystic fibrosis Experience over two decades William G. Flight a, b,, Jonathan
Journal of Cystic Fibrosis 11 (2012) 187 192 www.elsevier.com/locate/jcf Original Article Long-term non-invasive ventilation in cystic fibrosis Experience over two decades William G. Flight a, b,, Jonathan
Chronic Obstructive Pulmonary Disease (COPD).
 Chronic Obstructive Pulmonary Disease (COPD). Linde: Living healthcare 02 03 Chronic Obstructive Pulmonary Disease (COPD). A pocket guide for healthcare professionals. COPD the facts Moderate to severe
Chronic Obstructive Pulmonary Disease (COPD). Linde: Living healthcare 02 03 Chronic Obstructive Pulmonary Disease (COPD). A pocket guide for healthcare professionals. COPD the facts Moderate to severe
Patient characteristics Intervention Comparison Length of followup
 ORAL MUCOLYTICS Ref ID: 2511 Bachh AA, Shah NN, Bhargava R et al. Effect oral N- in COPD - A randomised controlled trial. JK Practitioner. 2007; 14(1):12-16. Ref ID: 2511 RCT Single blind; unclear allocation
ORAL MUCOLYTICS Ref ID: 2511 Bachh AA, Shah NN, Bhargava R et al. Effect oral N- in COPD - A randomised controlled trial. JK Practitioner. 2007; 14(1):12-16. Ref ID: 2511 RCT Single blind; unclear allocation
Learning Objectives. 1. Indications versus contra-indications 2. CPAP versus NiVS 3. Clinical evidence
 Learning Objectives 1. Indications versus contra-indications 2. CPAP versus NiVS 3. Clinical evidence Pre-hospital Non-invasive vventilatory support Marc Gillis, MD Imelda Bonheiden Our goal out there
Learning Objectives 1. Indications versus contra-indications 2. CPAP versus NiVS 3. Clinical evidence Pre-hospital Non-invasive vventilatory support Marc Gillis, MD Imelda Bonheiden Our goal out there
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A.
 aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
Josh Stanton and Michael Epton Respiratory Physiology Laboratory, Canterbury Respiratory Research Group Christchurch Hospital
 Josh Stanton and Michael Epton Respiratory Physiology Laboratory, Canterbury Respiratory Research Group Christchurch Hospital Setting Scene Advancements in neonatal care over past 30 years has resulted
Josh Stanton and Michael Epton Respiratory Physiology Laboratory, Canterbury Respiratory Research Group Christchurch Hospital Setting Scene Advancements in neonatal care over past 30 years has resulted
THE CHALLENGES OF COPD MANAGEMENT IN PRIMARY CARE An Expert Roundtable
 THE CHALLENGES OF COPD MANAGEMENT IN PRIMARY CARE An Expert Roundtable This activity is supported by an educational grant from Sunovion Pharmaceuticals Inc. COPD in the United States Third leading cause
THE CHALLENGES OF COPD MANAGEMENT IN PRIMARY CARE An Expert Roundtable This activity is supported by an educational grant from Sunovion Pharmaceuticals Inc. COPD in the United States Third leading cause
