SER-287 Phase 1b topline study results in patients with mild-to-moderate Ulcerative Colitis October 2, 2017
|
|
|
- Silas Jefferson
- 5 years ago
- Views:
Transcription
1 SER-287 Phase 1b topline study results in patients with mild-to-moderate Ulcerative Colitis October 2, 2017 Leading the Microbiome Revolution
2 Forward Looking Statements Some of the statements in this presentation constitute forward looking statements under the Private Securities Litigation Reform Act of Such statements are subject to factors, risks and uncertainties (such as those detailed in the Company s periodic filings with the SEC) that may cause actual results to differ materially from those expressed or implied by such forward looking statements. Any forward looking statements included herein represent our views as of today only. We may update these statements, but we disclaim any obligation to do so. 2
3 Seres Overview Opportunity Platform Pipeline Phase 3 stage company developing microbiome-based therapeutics, a highly promising new area of medicine Leader in microbiome drug development with differentiated capabilities Broad pipeline in infectious, inflammatory and immune, metabolic and liver diseases Team Experienced, accomplished leadership team Runway Strong cash and strategic position 3
4 Robust Microbiome Therapeutics Pipeline PRECLINICAL PHASE 1 PHASE 2 PHASE 3 SER-109 Recurrent C. difficile SER-262 SER-287 SER-301 SER-155 Primary C. difficile Ulcerative colitis Inflammatory Bowel Disease (IBD) Prevention of infection and GVHD following hematopoietic stem cell or solid organ transplant Positive top-line Phase 1b results Synthetically fermented Biologically sourced Infectious Inflammatory DISCOVERY EFFORTS ACADEMIC COLLABORATOR Immuno-oncology and hematopoietic stem cell transplant Inflammatory bowel diseases Primary sclerosing cholangitis, NASH and other liver diseases 4Obesity/metabolic syndrome Genetic metabolic diseases Based on interactions with the U.S. Food and Drug Administration, ECOSPOR III will be designated a Phase 3 trial and the company expects that this single pivotal study may support SER-109 registration and approval. Collaboration with Nestlé Health Science regarding C. difficile and IBD programs for markets only outside of North America 4
5 SER-287 Overview Biologically sourced consortium of bacterial spores Orally administered Strong intellectual property protection supported by multiple patent claims covering Ulcerative Colitis, as well as regulatory data protection Hypothesized to act by modulating the dysbiotic microbiome, reducing inflammation without immunosuppression effects 1,2 Rationale for development supported by: Preclinical results from microbiome modification in animal models of colitis Multiple randomized, placebo controlled clinical studies examining the the treatment effects of multiple fecal microbiota transplantation in patients with Ulcerative Colitis 1. Blander JM et al., Regulation of inflammation by microbiota interactions with the host, Nature Immunology, Lynch S and Pedersen O, The Human Intestinal Microbiome in Health and Disease, The New England Journal of Medicine, Costello et al. Systematic review with meta-analysis: faecal microbiota transplantation for the induction of remission for active ulcerative colitis, Alimentary Pharmacology & Therapeutics,
6 SER-287 Opportunity in Ulcerative Colitis Significant need for improved Ulcerative Colitis therapies Large population: ~700K ulcerative colitis (US figures) Many ulcerative colitis patients do not respond to current therapies, both during induction and maintenance, and there is need for new treatment modalities Several leading ulcerative colitis therapies are immunosuppressive, increasing the risk of opportunistic infections and certain cancers, and limiting widespread use SER-287 target profile: Oral Alternative mechanistic approach, potential as a mono or combination therapy Potential highly favorable safety profile 6
7 SER-287 Phase 1b Ulcerative Colitis Study Design Arm A: Placebo pre-treatment / Placebo once daily for 8 weeks (n=11) Intent to treat, observed case population for efficacy analysis = 10 subjects 58 mildmoderate UC patients failing standard-ofcare* Arm B: Vancomycin pretreatment / SER-287 once daily dosing for 8 weeks (n=15) Arm C: Placebo pre-treatment / SER-287 once weekly dosing for 8 weeks (n=15) Intent to treat, observed case population for efficacy analysis = 15 subjects Intent to treat, observed case population for efficacy analysis = 14 subjects Arm D: Vancomycin pre-treatment / SER-287 once weekly dosing for 8 weeks (n=17) Intent to treat, observed case population for efficacy analysis = 14 subjects Note: Study designed to enroll 55 patients, with 15 in SER-287 treatment arms and 10 in the placebo / placebo arm 7
8 SER-287 Phase 1b Study Endpoints Safety and tolerability Primary Objectives Change in composition of intestinal microbiome at 8 weeks * Secondary Objectives Clinical remission, endoscopic improvement and response through measure of the total modified Mayo Score Change in serum and fecal biomarkers * Complement of microbiome metabolic pathways from stool, urine and blood * Immunological and pathologic changes in mucosal biopsies * * Data expected in the coming months 8
9 Phase 1b Study Patient Demographics Statistic (Placebo/ Placebo) (N = 11) (Vancomycin / SER-287 daily) (N = 15) (Placebo/SER- 287 weekly) (N = 15) (Vancomycin / SER-287 weekly) (N = 17) Ser-287 (N = 47) Overall (N = 58) Characteristic Age (years) n Mean (SD) 45.8 (15.20) 47.8 (18.59) 46.5 (16.12) 47.9 (11.18) 47.4 (15.10) 47.1 (15.00) Sex Male n (%) 4 (36.4) 7 (46.7) 6 (40.0) 10 (58.8) 23 (48.9) 27 (46.6) Female n (%) 7 (63.6) 8 (53.3) 9 (60.0) 7 (41.2) 24 (51.1) 31 (53.4) Race White n (%) 8 (72.7) 12 (80.0) 12 (80.0) 15 (88.2) 39 (83.0) 47 (81.0) Asian n (%) 1 (9.1) (5.9) 1 (2.1) 2 (3.4) Black or African American n (%) 1 (9.1) 2 (13.3) 3 (20.0) 1 (5.9) 6 (12.8) 7 (12.1) Other - Indian n (%) 1 (9.1) 1 (6.7) (2.1) 2 (3.4) Montreal Classification Ulcerative Proctitis n (%) 0 4 (26.7) 0 2 (11.8) 6 (12.8) 6 (10.3) Left-sided UC (distal UC) n (%) 8 (72.7) 5 (33.3) 10 (66.7) 10 (58.8) 25 (53.2) 33 (56.9) Extensive UC (pancolitis) n (%) 3 (27.3) 6 (40.0) 5 (33.3) 5 (29.4) 16 (34.0) 19 (32.8) Severity of UC Mild n (%) 3 (27.3) 6 (40.0) 6 (40.0) 9 (52.9) 21 (44.7) 24 (41.4) Moderate n (%) 8 (72.7) 9 (60.0) 9 (60.0) 7 (41.2) 25 (53.2) 33 (56.9) Receiving Current UC Treatment Prior to or On Date of 1st Pretreatment Dose No n (%) 1 (9.1) 3 (20.0) 2 (13.3) 6 (35.3) 11 (23.4) 12 (20.7) 5-ASA n (%) 7 (63.6) 11 (73.3) 11 (73.3) 9 (52.9) 31 (66.0) 38 (65.5) Immunomodulator n (%) 2 (18.2) 1 (6.7) 4 (26.7) 2 (11.8) 7 (14.9) 9 (15.5) Steroid n (%) 3 (27.3) 3 (20.0) 2 (13.3) 0 5 (10.6) 8 (13.8) Other n (%) 1 (9.1) (5.9) 1 (2.1) 2 (3.4) Time since first UC diagnosis n (months) Mean (SD) (85.91) (143.77) (141.34) (105.41) (127.51) (120.12) 9
10 SER-287 Phase 1b Study Clinical Efficacy Endpoints Endpoint Intent to Treat Population, Observed Case: Treatment Group Clinical Remission Difference from placebo (SER-287 minus placebo) Placebo / Placebo (N = 10) (%) Vancomycin / SER-287 daily (N = 15) (%) Placebo / SER-287 weekly (N = 14) (%) Vancomycin / SER- 287 weekly (N = 14) (%) 1/10 (10.0) 6/15 (40.0) 2/14 (14.3) 3/14 (21.4) 30.0% 4.3% 11.4% Endoscopic Improvement 1/10 (10.0) 6/15 (40.0) 5/14 (35.7) 4/14 (28.6) Difference from placebo (SER-287 minus placebo) Clinical Response Difference from placebo (SER-287 minus placebo) 30.0% 25.7% 18.6% 6/10 (60.0) 9/15 (60.0) 6/14 (42.9) 4/14 (28.6) 0.0% -17.1% -31.4% 10
11 Treatment-Emergent Adverse Events Incidence by Treatment and System Organ Class System Organ Class Safety Population (Placebo / placebo) (N = 11) n (%) E (Vancomycin / SER-287 daily) (N = 15) n (%) E (Placebo / SER- 287 weekly) (N = 15) n (%) E (Vancomycin / SER-287 weekly) (N = 17) n (%) E SER-287 (N = 47) n (%) E Gastrointestinal disorders 5 (45.5) 7 2 (13.3) 4 7 (46.7) 19 8 (47.1) (36.2) 44 General disorders and administration site conditions 1 (9.1) 1 1 (6.7) (17.6) 3 4 (8.5) 4 Immune system disorders (5.9) 1 1 (2.1) 1 Infections and infestations 3 (27.3) 3 4 (26.7) 6 1 (6.7) 3 6 (35.3) 6 11 (23.4) 15 Injury, poisoning and procedural complications 2 (18.2) Investigations (5.9) 2 1 (2.1) 2 Metabolism and nutrition disorders 0 1 (6.7) (5.9) 1 2 (4.3) 2 Musculoskeletal and connective tissue disorders 0 2 (13.3) 2 3 (20.0) 4 1 (5.9) 2 6 (12.8) 8 Nervous system disorders 0 3 (20.0) (5.9) 1 4 (8.5) 5 Psychiatric disorders 1 (9.1) 1 1 (6.7) (2.1) 1 Reproductive system and breast disorders (5.9) 1 1 (2.1) 1 Respiratory, thoracic and mediastinal disorders Skin and subcutaneous tissue disorders 0 1 (6.7) 1 1 (6.7) 1 2 (11.8) 2 4 (8.5) (20.0) (5.9) 1 4 (8.5) 4 11
12 SER-287 Phase 1b Overall Safety: Most Common (>5%) Adverse Events by Treatment Group Safety Population Preferred Term Placebo / Placebo (N = 11) n (%) Vancomycin / SER-287 daily (N = 15) n (%) Placebo / SER-287 weekly (N = 15) n (%) Vancomycin / SER-287 weekly (N = 17) n (%) SER-287 (N = 47) n (%) Treatment-Emergent Adverse Events (All) 7 (63.6) 8 (53.3) 9 (60.0) 14 (82.4) 31 (66.0) Abdominal pain 1 (9.1) 0 3 (20.0) 4 (23.5) 7 (14.9) Nausea 0 1 (6.7) 3 (20.0) 1 (5.9) 5 (10.6) Back pain 0 1 (6.7) 3 (20.0) 1 (5.9) 5 (10.6) Diarrhoea 2 (18.2) 0 2 (13.3) 2 (11.8) 4 (8.5) Headache 0 3 (20.0) 0 1 (5.9) 4 (8.5) Constipation 0 2 (13.3) 1 (6.7) 0 3 (6.4) Flatulence (17.6) 3 (6.4) Upper Respiratory Tract Infection 0 2 (13.3) 0 1 (5.9) 3 (6.4) 12
13 Advancing SER-287 Clinical Development Phase 1b top line results summary: o SER-287 microbiome treatment resulted in a benefit in clinical remission rates, as well as an improvement in endoscopic scores o No clinically significant safety or tolerability findings were observed o Microbiome results are expected in the coming months Company expects to meet with with the FDA as soon as possible to determine the most accelerated path to advance SER-287 development in mild, moderate and severe forms of Ulcerative Colitis, and in maintenance after induction therapy Company also intends to assess future development in Crohn s disease, and pediatric forms of inflammatory bowel disease 13
Corporate Overview. August Leading the Microbiome Revolution
 Corporate Overview August 2018 Leading the Microbiome Revolution Forward looking statements Some of the statements in this presentation constitute forward looking statements under the Private Securities
Corporate Overview August 2018 Leading the Microbiome Revolution Forward looking statements Some of the statements in this presentation constitute forward looking statements under the Private Securities
REWRITING CANCER TREATMENT THROUGH EPIGENETIC MEDICINES
 REWRITING CANCER TREATMENT THROUGH EPIGENETIC MEDICINES May 18, 2017 Molecularly Defined Solid Tumor Program Update FORWARD-LOOKING STATEMENTS Any statements in this press release about future expectations,
REWRITING CANCER TREATMENT THROUGH EPIGENETIC MEDICINES May 18, 2017 Molecularly Defined Solid Tumor Program Update FORWARD-LOOKING STATEMENTS Any statements in this press release about future expectations,
ESTABLISH 2 Top Line Data Release
 The Future of Anti-Infectives ESTABLISH 2 Top Line Data Release March 25, 2013 1 Forward Looking Statements Statements contained in this data release regarding matters that are not historical facts are
The Future of Anti-Infectives ESTABLISH 2 Top Line Data Release March 25, 2013 1 Forward Looking Statements Statements contained in this data release regarding matters that are not historical facts are
Phase 2b/3 Topline Trial Results
 Phase 2b/3 Topline Trial Results RP-G28 For the Treatment of Lactose Intolerance March 2017 Forward - Looking Statement To the extent that statements contained in this presentation are not descriptions
Phase 2b/3 Topline Trial Results RP-G28 For the Treatment of Lactose Intolerance March 2017 Forward - Looking Statement To the extent that statements contained in this presentation are not descriptions
Phase 3c Topline Results. Page 1
 Phase 3c Topline Results Page 1 Important Information Any statements in this presentation about future expectations, plans and prospects for the Company including the development and regulatory status
Phase 3c Topline Results Page 1 Important Information Any statements in this presentation about future expectations, plans and prospects for the Company including the development and regulatory status
Jefferies Healthcare Conference. June 6, 2018
 Jefferies Healthcare Conference June 6, 2018 Forward-Looking Statements and Non-GAAP Financial Information Some statements in this presentation may be forward-looking statements for purposes of the Private
Jefferies Healthcare Conference June 6, 2018 Forward-Looking Statements and Non-GAAP Financial Information Some statements in this presentation may be forward-looking statements for purposes of the Private
Bobby W. Sandage, Jr., PhD President & Chief Executive Officer. Lazard Capital Markets 8 th Annual Healthcare Conference
 Bobby W. Sandage, Jr., PhD President & Chief Executive Officer Lazard Capital Markets 8 th Annual Healthcare Conference Statements in this presentation that are not descriptions of historical facts are
Bobby W. Sandage, Jr., PhD President & Chief Executive Officer Lazard Capital Markets 8 th Annual Healthcare Conference Statements in this presentation that are not descriptions of historical facts are
AVEO Oncology Announces Strategic Restructuring. AVEO to Host Conference Call Wednesday, June 5 at 8:30 a.m. ET
 NEWS RELEASE FOR IMMEDIATE RELEASE AVEO Oncology Announces Strategic Restructuring AVEO to Host Conference Call Wednesday, June 5 at 8:30 a.m. ET CAMBRIDGE, Mass., June 4, 2013 AVEO Oncology (NASDAQ: AVEO)
NEWS RELEASE FOR IMMEDIATE RELEASE AVEO Oncology Announces Strategic Restructuring AVEO to Host Conference Call Wednesday, June 5 at 8:30 a.m. ET CAMBRIDGE, Mass., June 4, 2013 AVEO Oncology (NASDAQ: AVEO)
Theravance Announces Positive Results from Phase 1 and Phase 2 Clinical Studies with TD-1211 in Development for Opioid-Induced Constipation
 Theravance Announces Positive Results from Phase 1 and Phase 2 Clinical Studies with TD-1211 in Development for Opioid-Induced Constipation TD-1211 Achieves Primary and Secondary Endpoints SOUTH SAN FRANCISCO,
Theravance Announces Positive Results from Phase 1 and Phase 2 Clinical Studies with TD-1211 in Development for Opioid-Induced Constipation TD-1211 Achieves Primary and Secondary Endpoints SOUTH SAN FRANCISCO,
AVEO and Astellas Announce TAURUS Patient Preference Clinical Study Comparing Tivozanib with Sunitinib in First-Line Kidney Cancer
 FOR IMMEDIATE RELEASE AVEO and Astellas Announce TAURUS Patient Preference Clinical Study Comparing Tivozanib with Sunitinib in First-Line Kidney Cancer Study designed to build upon safety profile demonstrated
FOR IMMEDIATE RELEASE AVEO and Astellas Announce TAURUS Patient Preference Clinical Study Comparing Tivozanib with Sunitinib in First-Line Kidney Cancer Study designed to build upon safety profile demonstrated
Oragenics Shareholder Update
 January 4, 2017 Oragenics Shareholder Update Advances Drug Development Programs Focused on Conditions with Significant Unmet Medical Needs TAMPA, Fla.--(BUSINESS WIRE)-- Oragenics, Inc. (NYSE MKT:OGEN.BC),
January 4, 2017 Oragenics Shareholder Update Advances Drug Development Programs Focused on Conditions with Significant Unmet Medical Needs TAMPA, Fla.--(BUSINESS WIRE)-- Oragenics, Inc. (NYSE MKT:OGEN.BC),
ARDELYX REPORTS POSITIVE T3MPO-2 PHASE 3 TRIAL RESULTS IN IBS-C
 ARDELYX REPORTS POSITIVE T3MPO-2 PHASE 3 TRIAL RESULTS IN IBS-C OCTOBER 11, 2017 NASDAQ: ARDX FORWARD-LOOKING STATEMENTS To the extent that statements contained in this presentation are not descriptions
ARDELYX REPORTS POSITIVE T3MPO-2 PHASE 3 TRIAL RESULTS IN IBS-C OCTOBER 11, 2017 NASDAQ: ARDX FORWARD-LOOKING STATEMENTS To the extent that statements contained in this presentation are not descriptions
UBS Global Healthcare Conference May 19, 2014
 UBS Global Healthcare Conference May 19, 2014 Safe Harbor Statement This presentation may contain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section
UBS Global Healthcare Conference May 19, 2014 Safe Harbor Statement This presentation may contain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section
XARACOLL Phase 3 Results Webcast. MATRIX 1 and MATRIX 2 Clinical Trials May 25, 2016
 XARACOLL Phase 3 Results Webcast MATRIX 1 and MATRIX 2 Clinical Trials May 25, 2016 Forward Looking Statements This presentation contains forward-looking statements about our ongoing development of XARACOLL
XARACOLL Phase 3 Results Webcast MATRIX 1 and MATRIX 2 Clinical Trials May 25, 2016 Forward Looking Statements This presentation contains forward-looking statements about our ongoing development of XARACOLL
IRONWOOD AND FOREST ANNOUNCE POSITIVE LINACLOTIDE RESULTS FROM PHASE 3 TRIAL IN PATIENTS WITH IRRITABLE BOWEL SYNDROME WITH CONSTIPATION
 FOR IMMEDIATE RELEASE Ironwood Contact: Forest Contact: Susan Brady Frank J. Murdolo Corporate Communications Vice President, Investor Relations 617.621.8304 212.224.6714 sbrady@ironwoodpharma.com frank.murdolo@frx.com
FOR IMMEDIATE RELEASE Ironwood Contact: Forest Contact: Susan Brady Frank J. Murdolo Corporate Communications Vice President, Investor Relations 617.621.8304 212.224.6714 sbrady@ironwoodpharma.com frank.murdolo@frx.com
Press Release. RedHill Biopharma Announces Enrollment of Last Patient in the BEKINDA Phase II Study for IBS-D
 Press Release RedHill Biopharma Announces Enrollment of Last Patient in the BEKINDA Phase II Study for IBS-D Top-line results are expected in the third quarter of 2017 The randomized, double-blind, placebo-controlled
Press Release RedHill Biopharma Announces Enrollment of Last Patient in the BEKINDA Phase II Study for IBS-D Top-line results are expected in the third quarter of 2017 The randomized, double-blind, placebo-controlled
Building a Fully Integrated Biopharmaceutical Company. June 2014
 Building a Fully Integrated Biopharmaceutical Company June 2014 Forward-Looking Statements This presentation contains forward-looking statements within the meaning of The Private Securities Litigation
Building a Fully Integrated Biopharmaceutical Company June 2014 Forward-Looking Statements This presentation contains forward-looking statements within the meaning of The Private Securities Litigation
ZIOPHARM / Intrexon Graft-Versus-Host Disease Exclusive Channel Collaboration SEPTEMBER 28, 2015
 ZIOPHARM / Intrexon Graft-Versus-Host Disease Exclusive Channel Collaboration SEPTEMBER 28, 2015 1 Forward-looking Statements This presentation contains certain forward-looking information about ZIOPHARM
ZIOPHARM / Intrexon Graft-Versus-Host Disease Exclusive Channel Collaboration SEPTEMBER 28, 2015 1 Forward-looking Statements This presentation contains certain forward-looking information about ZIOPHARM
AVEO and Astellas Announce Positive Findings from TIVO-1 Superiority Study of Tivozanib in First-Line Advanced RCC
 FOR IMMEDIATE RELEASE AVEO and Astellas Announce Positive Findings from TIVO-1 Superiority Study of Tivozanib in First-Line Advanced RCC - Tivozanib is the First Agent to Demonstrate Greater than One Year
FOR IMMEDIATE RELEASE AVEO and Astellas Announce Positive Findings from TIVO-1 Superiority Study of Tivozanib in First-Line Advanced RCC - Tivozanib is the First Agent to Demonstrate Greater than One Year
Capricor Therapeutics
 Therapeutics Conference Call to Discuss the HOPE-2 Clinical Trial NASDAQ: CAPR November 29, 2017 Forward-Looking Statements Statements in this presentation regarding the efficacy, safety, and intended
Therapeutics Conference Call to Discuss the HOPE-2 Clinical Trial NASDAQ: CAPR November 29, 2017 Forward-Looking Statements Statements in this presentation regarding the efficacy, safety, and intended
Third Quarter 2018 Financial Results. November 1, 2018
 Third Quarter 2018 Financial Results November 1, 2018 Agios Conference Call Participants Prepared Remarks Introduction RENEE LECK, Associate Director, Investor Relations Business Highlights & 2018 Key
Third Quarter 2018 Financial Results November 1, 2018 Agios Conference Call Participants Prepared Remarks Introduction RENEE LECK, Associate Director, Investor Relations Business Highlights & 2018 Key
Studies on inflammatory bowel disease and functional gastrointestinal disorders in children and adults Hoekman, D.R.
 UvA-DARE (Digital Academic Repository) Studies on inflammatory bowel disease and functional gastrointestinal disorders in children and adults Hoekman, D.R. Link to publication Citation for published version
UvA-DARE (Digital Academic Repository) Studies on inflammatory bowel disease and functional gastrointestinal disorders in children and adults Hoekman, D.R. Link to publication Citation for published version
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 20 October 2010
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 20 October 2010 MEZAVANT LP 1200 mg, prolonged-release gastro-resistant tablets B/60 (CIP code: 378 689-2) Applicant
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 20 October 2010 MEZAVANT LP 1200 mg, prolonged-release gastro-resistant tablets B/60 (CIP code: 378 689-2) Applicant
-- Edasalonexent Substantially Slowed Duchenne Muscular Dystrophy Disease Progression through 36 Weeks --
 Catabasis Pharmaceuticals Reports Positive Results from Open-Label Extension of Phase 2 MoveDMD Trial Evaluating Edasalonexent in Duchenne Muscular Dystrophy and Plans to Initiate Phase 3 Clinical Trial
Catabasis Pharmaceuticals Reports Positive Results from Open-Label Extension of Phase 2 MoveDMD Trial Evaluating Edasalonexent in Duchenne Muscular Dystrophy and Plans to Initiate Phase 3 Clinical Trial
Medical Therapy for Pediatric IBD: Efficacy and Safety
 Medical Therapy for Pediatric IBD: Efficacy and Safety Betsy Maxwell, MD Assistant Professor of Clinical Pediatrics Division of Gastroenterology, Hepatology, and Nutrition Pediatric IBD: Defining Remission
Medical Therapy for Pediatric IBD: Efficacy and Safety Betsy Maxwell, MD Assistant Professor of Clinical Pediatrics Division of Gastroenterology, Hepatology, and Nutrition Pediatric IBD: Defining Remission
ASCEND Phase 2 Trial of AXS-05 in MDD Topline Results Conference Call
 NASDAQ: AXSM ASCEND Phase 2 Trial of in MDD Topline Results Conference Call January 7, 2019 Overview in MDD ASCEND Phase 2 Trial Topline Results Introduction Mark Jacobson, Senior Vice President, Operations
NASDAQ: AXSM ASCEND Phase 2 Trial of in MDD Topline Results Conference Call January 7, 2019 Overview in MDD ASCEND Phase 2 Trial Topline Results Introduction Mark Jacobson, Senior Vice President, Operations
IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics
 IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics Oncology Autoimmune Diseases Rodman & Renshaw 19 th Annual Global Investment Conference Michael R. Garone, Principal Executive Officer and CFO Forward-Looking
IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics Oncology Autoimmune Diseases Rodman & Renshaw 19 th Annual Global Investment Conference Michael R. Garone, Principal Executive Officer and CFO Forward-Looking
Moderately to severely active ulcerative colitis
 Adalimumab in the Treatment of Moderate-to-Severe Ulcerative Colitis: ULTRA 2 Trial Results Sandborn WJ, van Assche G, Reinisch W, et al. Adalimumab induces and maintains clinical remission in patients
Adalimumab in the Treatment of Moderate-to-Severe Ulcerative Colitis: ULTRA 2 Trial Results Sandborn WJ, van Assche G, Reinisch W, et al. Adalimumab induces and maintains clinical remission in patients
Efficacy and Safety of Treatment for Pediatric IBD
 Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Assistant Professor of Clinical Pediatrics Division of Gastroenterology,
Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Assistant Professor of Clinical Pediatrics Division of Gastroenterology,
-- Single Global Phase 3 Trial Expected to Begin in First Half of
 Catabasis Pharmaceuticals Reports Edasalonexent Preserved Muscle Function and Substantially Slowed Duchenne Muscular Dystrophy Disease Progression Through More Than One Year of Treatment -- Consistent
Catabasis Pharmaceuticals Reports Edasalonexent Preserved Muscle Function and Substantially Slowed Duchenne Muscular Dystrophy Disease Progression Through More Than One Year of Treatment -- Consistent
Utrophin Modulation: A Universal Treatment Approach to DMD. End Duchenne Tour April 2018
 Utrophin Modulation: A Universal Treatment Approach to DMD April 2018 Legal Disclaimer Statements in this presentation, other than statements of historical fact, constitute forward-looking statements within
Utrophin Modulation: A Universal Treatment Approach to DMD April 2018 Legal Disclaimer Statements in this presentation, other than statements of historical fact, constitute forward-looking statements within
PEDIATRIC INFLAMMATORY BOWEL DISEASE
 PEDIATRIC INFLAMMATORY BOWEL DISEASE Alexis Rodriguez, MD Pediatric Gastroenterology Advocate Children s Hospital Disclosers Abbott Nutrition - Speaker Inflammatory Bowel Disease Chronic inflammatory disease
PEDIATRIC INFLAMMATORY BOWEL DISEASE Alexis Rodriguez, MD Pediatric Gastroenterology Advocate Children s Hospital Disclosers Abbott Nutrition - Speaker Inflammatory Bowel Disease Chronic inflammatory disease
I.V. CR845 Adaptive Phase 2/3 Post Operative Pain Study Results
 I.V. CR845 Adaptive Phase 2/3 Post Operative Pain Study Results A Multicenter, Randomized, Double-Blind, Placebo-Controlled, Adaptive Design Study Evaluating the Analgesic Efficacy and Safety of I.V. CR845
I.V. CR845 Adaptive Phase 2/3 Post Operative Pain Study Results A Multicenter, Randomized, Double-Blind, Placebo-Controlled, Adaptive Design Study Evaluating the Analgesic Efficacy and Safety of I.V. CR845
MANIFEST Phase 2 Enhancement / Expansion
 MANIFEST Phase 2 Enhancement / Expansion Investor Conference Call Stellar Science, Breakthrough Medicine October 11, 2018 Forward-Looking Statements This presentation contains forward-looking statements
MANIFEST Phase 2 Enhancement / Expansion Investor Conference Call Stellar Science, Breakthrough Medicine October 11, 2018 Forward-Looking Statements This presentation contains forward-looking statements
Five Prime Therapeutics, Inc. Corporate Overview
 Five Prime Therapeutics, Inc. Corporate Overview June 2015 NASDAQ:FPRX Forward-Looking Statements Disclaimer This presentation contains forward-looking statements within the meaning of the Private Securities
Five Prime Therapeutics, Inc. Corporate Overview June 2015 NASDAQ:FPRX Forward-Looking Statements Disclaimer This presentation contains forward-looking statements within the meaning of the Private Securities
Jefferies Healthcare Conference. June 25, 2008
 Jefferies Healthcare Conference June 25, 2008 Safe Harbor Statement Except for the historical information set forth herein, the matters set forth in this presentation,including without limitation statements
Jefferies Healthcare Conference June 25, 2008 Safe Harbor Statement Except for the historical information set forth herein, the matters set forth in this presentation,including without limitation statements
IMMUNOMEDICS, INC. November Advanced Antibody-Based Therapeutics. Oncology Autoimmune Diseases
 IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics Oncology Autoimmune Diseases November 2017 Forward-Looking Statements This presentation, in addition to historical information, contains certain
IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics Oncology Autoimmune Diseases November 2017 Forward-Looking Statements This presentation, in addition to historical information, contains certain
Merrill Lynch's Global Pharmaceutical, Biotechnology, and Medical Device Conference. February 7, 2007
 Merrill Lynch's Global Pharmaceutical, Biotechnology, and Medical Device Conference February 7, 2007 Information related to forward-looking statements This presentation includes forward-looking statements
Merrill Lynch's Global Pharmaceutical, Biotechnology, and Medical Device Conference February 7, 2007 Information related to forward-looking statements This presentation includes forward-looking statements
ADAPTIMMUNE INVESTOR PRESENTATION. August 2016
 ADAPTIMMUNE INVESTOR PRESENTATION August 2016 DISCLAIMER This presentation contains forward-looking statements, as that term is defined under the Private Securities Litigation Reform Act of 1995 (PSLRA),
ADAPTIMMUNE INVESTOR PRESENTATION August 2016 DISCLAIMER This presentation contains forward-looking statements, as that term is defined under the Private Securities Litigation Reform Act of 1995 (PSLRA),
AVEO and Astellas Report Final Overall Survival Results from TIVO-1
 AVEO and Astellas Report Final Overall Survival Results from TIVO-1 - Median Overall Survival of 28.8 Months Reported for Tivozanib in Patients with Advanced Kidney Cancer - CAMBRIDGE, Mass. and TOKYO,
AVEO and Astellas Report Final Overall Survival Results from TIVO-1 - Median Overall Survival of 28.8 Months Reported for Tivozanib in Patients with Advanced Kidney Cancer - CAMBRIDGE, Mass. and TOKYO,
LJPC-401 Phase 1 Results and Development Update. September 7, 2016
 LJPC-401 Phase 1 Results and Development Update September 7, 2016 Forward-Looking Statements These slides contain "forward-looking" statements within the meaning of the Private Securities Litigation Reform
LJPC-401 Phase 1 Results and Development Update September 7, 2016 Forward-Looking Statements These slides contain "forward-looking" statements within the meaning of the Private Securities Litigation Reform
Synergy Pharmaceuticals TRULANCE (Plecanatide) Receives U.S. FDA Approval for the Treatment of Adults with Chronic Idiopathic Constipation
 January 19, 2017 Synergy Pharmaceuticals TRULANCE (Plecanatide) Receives U.S. FDA Approval for the Treatment of Adults with Chronic Idiopathic Constipation NEW YORK--(BUSINESS WIRE)-- Synergy Pharmaceuticals
January 19, 2017 Synergy Pharmaceuticals TRULANCE (Plecanatide) Receives U.S. FDA Approval for the Treatment of Adults with Chronic Idiopathic Constipation NEW YORK--(BUSINESS WIRE)-- Synergy Pharmaceuticals
Targeted Therapeutics for Inflammatory Disease
 Targeted Therapeutics for Inflammatory Disease Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary contain forward-looking statements that involve substantial
Targeted Therapeutics for Inflammatory Disease Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary contain forward-looking statements that involve substantial
Use of extrapolation in small clinical trials:
 Use of extrapolation in small clinical trials: Infliximab for pediatric ulcerative colitis Jessica J. Lee, MD, MMSc Medical Officer Division of Gastroenterology and Inborn Errors Products CDER/ FDA 1 Learning
Use of extrapolation in small clinical trials: Infliximab for pediatric ulcerative colitis Jessica J. Lee, MD, MMSc Medical Officer Division of Gastroenterology and Inborn Errors Products CDER/ FDA 1 Learning
TARGET A BETTER NOW FORWARD-LOOKING STATEMENTS NASDAQ: IMGN. Current as of January 2018
 NASDAQ: IMGN TARGET A BETTER NOW Current as of January 2018 FORWARD-LOOKING STATEMENTS This presentation includes forward looking statements based on management's current expectations. These statements
NASDAQ: IMGN TARGET A BETTER NOW Current as of January 2018 FORWARD-LOOKING STATEMENTS This presentation includes forward looking statements based on management's current expectations. These statements
PRO 140. First self-administered antibody therapy for HIV in late-stage clinical development. March
 PRO 140 First self-administered antibody therapy for HIV in late-stage clinical development March 2018 Forward-Looking Statements This presentation includes forward-looking statements and forward-looking
PRO 140 First self-administered antibody therapy for HIV in late-stage clinical development March 2018 Forward-Looking Statements This presentation includes forward-looking statements and forward-looking
(39% 20%), (36% 20%) (36% 17%) MAP US,
 Press Release RedHill Biopharma Announces Additional Data from Positive Phase III Study of RHB-104 in Crohn s Disease at United European Gastroenterology (UEG) Week 2018 The presentation highlighted enhanced
Press Release RedHill Biopharma Announces Additional Data from Positive Phase III Study of RHB-104 in Crohn s Disease at United European Gastroenterology (UEG) Week 2018 The presentation highlighted enhanced
Developing & Commercializing Targeted Small Molecule Drugs in Cancer
 Developing & Commercializing Targeted Small Molecule Drugs in Cancer SAFE HARBOR STATEMENT 2 Forward-looking statements made in the course of this presentation are made pursuant to the safe harbor provisions
Developing & Commercializing Targeted Small Molecule Drugs in Cancer SAFE HARBOR STATEMENT 2 Forward-looking statements made in the course of this presentation are made pursuant to the safe harbor provisions
Actinium Pharmaceuticals Highlights Analysis of Pivotal Iomab-B Phase 3 SIERRA Trial Presented in Oral Session at ASH Annual Meeting
 December 4, 2018 Actinium Pharmaceuticals Highlights Analysis of Pivotal Iomab-B Phase 3 SIERRA Trial Presented in Oral Session at ASH Annual Meeting - Key highlights include near universal engraftment
December 4, 2018 Actinium Pharmaceuticals Highlights Analysis of Pivotal Iomab-B Phase 3 SIERRA Trial Presented in Oral Session at ASH Annual Meeting - Key highlights include near universal engraftment
ALMIRALL AND IRONWOOD ANNOUNCE POSITIVE RESULTS FROM A PHASE 3 TRIAL WITH LINACLOTIDE IN PATIENTS WITH IRRITABLE BOWEL SYNDROME WITH CONSTIPATION
 FOR IMMEDIATE RELEASE Ironwood Contact: Almirall Contact: Susan Brady Ketchum Pleon Corporate Communications Amanda Sefton 617.621.8304 +44 (0) 207.611.3653 sbrady@ironwoodpharma.com amanda.sefton@ketchumpleon.com
FOR IMMEDIATE RELEASE Ironwood Contact: Almirall Contact: Susan Brady Ketchum Pleon Corporate Communications Amanda Sefton 617.621.8304 +44 (0) 207.611.3653 sbrady@ironwoodpharma.com amanda.sefton@ketchumpleon.com
Cx601 ADMIRE-CD Top-Line Results Webcast. 24 August 2015
 Cx601 ADMIRE-CD Top-Line Results Webcast 24 August 2015 1 Cx601 ADMIRE-CD Top-Line Results Webcast Speakers Mr Eduardo Bravo, Chief Executive Officer Dr Julián Panés, Head of Gastroenterology Department,
Cx601 ADMIRE-CD Top-Line Results Webcast 24 August 2015 1 Cx601 ADMIRE-CD Top-Line Results Webcast Speakers Mr Eduardo Bravo, Chief Executive Officer Dr Julián Panés, Head of Gastroenterology Department,
Targeted Therapeutics for Inflammatory Disease Jefferies Healthcare Conference
 Targeted Therapeutics for Inflammatory Disease 2016 Jefferies Healthcare Conference Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary contain forward-looking
Targeted Therapeutics for Inflammatory Disease 2016 Jefferies Healthcare Conference Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary contain forward-looking
Inducing Tumor-Specific Ischemic Necrosis to Enhance the Efficacy of Checkpoint Inhibitors and Chemotherapy
 Inducing Tumor-Specific Ischemic Necrosis to Enhance the Efficacy of Checkpoint Inhibitors and Chemotherapy Company Overview, September 2018 Safe Harbor Statement This presentation contains forward-looking
Inducing Tumor-Specific Ischemic Necrosis to Enhance the Efficacy of Checkpoint Inhibitors and Chemotherapy Company Overview, September 2018 Safe Harbor Statement This presentation contains forward-looking
Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans. Presented by: Richard Veselý. An agency of the European Union
 Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans Presented by: Richard Veselý An agency of the European Union Adalimumab - Crohn s disease Indication: Treatment of severe,
Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans Presented by: Richard Veselý An agency of the European Union Adalimumab - Crohn s disease Indication: Treatment of severe,
Bank of America Merrill Lynch 2016 Health Care Conference
 Bank of America Merrill Lynch 2016 Health Care Conference Dr. Steven Stein Chief Medical Officer David Gryska Chief Financial Officer May 11, 2016 Forward Looking Statements Except for the historical information
Bank of America Merrill Lynch 2016 Health Care Conference Dr. Steven Stein Chief Medical Officer David Gryska Chief Financial Officer May 11, 2016 Forward Looking Statements Except for the historical information
WARNING: RISK OF SERIOUS DEHYDRATION IN PEDIATRIC PATIENTS
 June 5, 2018 Synergy Pharmaceuticals Highlights New Data at Digestive Disease Week (DDW) 2018 Linking Uroguanylin Deficiency to Chronic Idiopathic Constipation (CIC) and Irritable Bowel Syndrome with Constipation
June 5, 2018 Synergy Pharmaceuticals Highlights New Data at Digestive Disease Week (DDW) 2018 Linking Uroguanylin Deficiency to Chronic Idiopathic Constipation (CIC) and Irritable Bowel Syndrome with Constipation
Investor Call. May 19, Nasdaq: IMGN
 Investor Call May 19, 2017 Nasdaq: IMGN Forward-Looking Statements This presentation includes forward-looking statements based on management's current expectations. These statements include, but are not
Investor Call May 19, 2017 Nasdaq: IMGN Forward-Looking Statements This presentation includes forward-looking statements based on management's current expectations. These statements include, but are not
Conference Call for Investment Community. Nov 19, 2018
 Conference Call for Investment Community Nov 19, 2018 Agenda for Today s Call Topic Safe Harbor Statement Opening Remarks AR101 for Peanut Allergy Speaker Laura Hansen, PhD, VP, Investor Relations Jayson
Conference Call for Investment Community Nov 19, 2018 Agenda for Today s Call Topic Safe Harbor Statement Opening Remarks AR101 for Peanut Allergy Speaker Laura Hansen, PhD, VP, Investor Relations Jayson
Building a Premier Oncology Biotech
 Corporate Deck Building a Premier Oncology Biotech Dr. Helen Torley, President and CEO November 2018 Forward-Looking Statements All of the statements in this presentation that are not statements of historical
Corporate Deck Building a Premier Oncology Biotech Dr. Helen Torley, President and CEO November 2018 Forward-Looking Statements All of the statements in this presentation that are not statements of historical
About X-Linked Hypophosphatemia (XLH)
 Ultragenyx and Kyowa Kirin Announce Publication of Phase 2 Study Results Demonstrating that Crysvita (burosumab) Improved Outcomes in Children with X-linked Hypophosphatemia in the New England Journal
Ultragenyx and Kyowa Kirin Announce Publication of Phase 2 Study Results Demonstrating that Crysvita (burosumab) Improved Outcomes in Children with X-linked Hypophosphatemia in the New England Journal
Q4 Report Webcast February 7, 2019 Presenters: Renée Aguiar-Lucander, CEO Fredrik Johansson, CFO
 Q4 Report 2018 Webcast February 7, 2019 Presenters: Renée Aguiar-Lucander, CEO Fredrik Johansson, CFO Disclaimer Important information This presentation has been prepared by Calliditas Therapeutics AB
Q4 Report 2018 Webcast February 7, 2019 Presenters: Renée Aguiar-Lucander, CEO Fredrik Johansson, CFO Disclaimer Important information This presentation has been prepared by Calliditas Therapeutics AB
LION. Corporate Presentation June 2016 BIOTECHNOLOGIES. Leadership & Innovation in Oncology
 LION BIOTECHNOLOGIES Leadership & Innovation in Oncology Corporate Presentation June 2016 Forward-Looking Statements This presentation contains forward-looking statements within the meaning of the Private
LION BIOTECHNOLOGIES Leadership & Innovation in Oncology Corporate Presentation June 2016 Forward-Looking Statements This presentation contains forward-looking statements within the meaning of the Private
Corporate Medical Policy Fecal Microbiota Transplantation
 Corporate Medical Policy Fecal Microbiota Transplantation File Name: Origination: Last CAP Review: Next CAP Review: Last Review: Fecal_microbiota_transplantation 7/2014 11/2017 11/2018 11/2017 Description
Corporate Medical Policy Fecal Microbiota Transplantation File Name: Origination: Last CAP Review: Next CAP Review: Last Review: Fecal_microbiota_transplantation 7/2014 11/2017 11/2018 11/2017 Description
DISCUSSION GROUP #5 ISAPP 2014 THE MICROBIOME AND HEALTH: PROMISING ENDPOINTS FOR INTERVENTIONS. Friday, June 20, 2014
 DISCUSSION GROUP #5 ISAPP 2014 THE MICROBIOME AND HEALTH: PROMISING ENDPOINTS FOR INTERVENTIONS Friday, June 20, 2014 Chairs: George Fahey, Jens Walter 1 What do we want to achieve? Treatment of disease
DISCUSSION GROUP #5 ISAPP 2014 THE MICROBIOME AND HEALTH: PROMISING ENDPOINTS FOR INTERVENTIONS Friday, June 20, 2014 Chairs: George Fahey, Jens Walter 1 What do we want to achieve? Treatment of disease
ARQ 087 Overview. FGFR Inhibitor. March 2017
 ARQ 087 Overview FGFR Inhibitor March 2017 Safe Harbor This presentation and other statements by ArQule contain forward-looking statements within the meaning of the Private Securities Litigation Reform
ARQ 087 Overview FGFR Inhibitor March 2017 Safe Harbor This presentation and other statements by ArQule contain forward-looking statements within the meaning of the Private Securities Litigation Reform
New treatment options in IBD: today and the future. Silvio Danese Istituto Clinico Humanitas, Milan, Italy
 New treatment options in IBD: today and the future Silvio Danese Istituto Clinico Humanitas, Milan, Italy Date of preparation: October 2014 GLO/EYV/2014-00010h Overview of the late-stage IBD drug pipeline*
New treatment options in IBD: today and the future Silvio Danese Istituto Clinico Humanitas, Milan, Italy Date of preparation: October 2014 GLO/EYV/2014-00010h Overview of the late-stage IBD drug pipeline*
Syndax Announces Updated Results from Phase 2 ENCORE 601 Trial of Entinostat in Combination with KEYTRUDA (pembrolizumab)
 Syndax Announces Updated Results from Phase 2 ENCORE 601 Trial of Entinostat in Combination with KEYTRUDA (pembrolizumab) Ongoing ENCORE 601 biomarker analyses suggest enhanced clinical benefit in subpopulation
Syndax Announces Updated Results from Phase 2 ENCORE 601 Trial of Entinostat in Combination with KEYTRUDA (pembrolizumab) Ongoing ENCORE 601 biomarker analyses suggest enhanced clinical benefit in subpopulation
Dynavax Corporate Presentation
 Dynavax Corporate Presentation Forward-Looking Statements This presentation contains forward-looking statements, including statements regarding our HEPLISAV-B TM regulatory submissions, product profile,
Dynavax Corporate Presentation Forward-Looking Statements This presentation contains forward-looking statements, including statements regarding our HEPLISAV-B TM regulatory submissions, product profile,
Tonix Pharmaceuticals Reports Top Line Results From Phase 2b BESTFIT Trial of TNX-102 SL in Patients With Fibromyalgia
 September 29, 2014 Tonix Pharmaceuticals Reports Top Line Results From Phase 2b BESTFIT Trial of TNX-102 SL in Patients With Fibromyalgia Conference Call Today at 8:30 a.m. ET NEW YORK, Sept. 29, 2014
September 29, 2014 Tonix Pharmaceuticals Reports Top Line Results From Phase 2b BESTFIT Trial of TNX-102 SL in Patients With Fibromyalgia Conference Call Today at 8:30 a.m. ET NEW YORK, Sept. 29, 2014
Inarigivir ACHIEVE Trial Results and HBV Clinical Program Update. August 2, 2018
 Inarigivir ACHIEVE Trial Results and HBV Clinical Program Update August 2, 2018 FORWARD LOOKING STATEMENT This presentation includes forward-looking statements within the meaning of the Private Securities
Inarigivir ACHIEVE Trial Results and HBV Clinical Program Update August 2, 2018 FORWARD LOOKING STATEMENT This presentation includes forward-looking statements within the meaning of the Private Securities
Broad and clinically important benefits beyond the initial registrational endpoints are now reported.
 Biohaven Announces Robust Clinical Data with Single Dose Rimegepant That Defines Acute and Durable Benefits to Patients: The First Oral CGRP Receptor Antagonist to Deliver Positive Data on Pain Freedom
Biohaven Announces Robust Clinical Data with Single Dose Rimegepant That Defines Acute and Durable Benefits to Patients: The First Oral CGRP Receptor Antagonist to Deliver Positive Data on Pain Freedom
Efficacy and Safety of Treatment for Pediatric IBD
 Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Associate Professor of Clinical Pediatrics Division of Gastroenterology,
Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Associate Professor of Clinical Pediatrics Division of Gastroenterology,
Corporate Presentation May Transforming Immuno-Oncology Using Next-Generation Immune Cell Engagers
 Corporate Presentation May 2016 Transforming Immuno-Oncology Using Next-Generation Immune Cell Engagers Forward-looking statements / safe harbor This presentation and the accompanying oral commentary contain
Corporate Presentation May 2016 Transforming Immuno-Oncology Using Next-Generation Immune Cell Engagers Forward-looking statements / safe harbor This presentation and the accompanying oral commentary contain
HTX-011 Postoperative Pain Program Topline Results From Phase 3. March 19, 2018
 HTX-011 Postoperative Pain Program Topline Results From Phase 3 March 19, 2018 Forward-Looking Statements This presentation contains "forward-looking statements" as defined by the Private Securities Litigation
HTX-011 Postoperative Pain Program Topline Results From Phase 3 March 19, 2018 Forward-Looking Statements This presentation contains "forward-looking statements" as defined by the Private Securities Litigation
Credit Suisse 27 th Annual Healthcare Conference
 CHANGING THE COURSE OF HUMAN HEALTH THROUGH BOLD PURSUITS IN SCIENCE Credit Suisse 27 th Annual Healthcare Conference November 14, 2018 Forward-Looking Statements and Adjusted Financial Information This
CHANGING THE COURSE OF HUMAN HEALTH THROUGH BOLD PURSUITS IN SCIENCE Credit Suisse 27 th Annual Healthcare Conference November 14, 2018 Forward-Looking Statements and Adjusted Financial Information This
AVX-470, an Orally Delivered Anti-TNF Antibody for Treatment of Acute Ulcerative Colitis: Results of a First-in- Human Trial
 AVX-470, an Orally Delivered Anti-TNF Antibody for Treatment of Acute Ulcerative Colitis: Results of a First-in- Human Trial M Scott Harris 1, Deborah Hartman 1, Sharon Spence 1, Sally Kennedy 1, Theodore
AVX-470, an Orally Delivered Anti-TNF Antibody for Treatment of Acute Ulcerative Colitis: Results of a First-in- Human Trial M Scott Harris 1, Deborah Hartman 1, Sharon Spence 1, Sally Kennedy 1, Theodore
VeriStrat Poor Patients Show Encouraging Overall Survival and Progression Free Survival Signal; Confirmatory Phase 2 Study Planned by Year-End
 AVEO and Biodesix Announce Exploratory Analysis of VeriStrat-Selected Patients with Non-Small Cell Lung Cancer in Phase 2 Study of Ficlatuzumab Presented at ESMO 2014 Congress VeriStrat Poor Patients Show
AVEO and Biodesix Announce Exploratory Analysis of VeriStrat-Selected Patients with Non-Small Cell Lung Cancer in Phase 2 Study of Ficlatuzumab Presented at ESMO 2014 Congress VeriStrat Poor Patients Show
Liquid Biopsies. Next Generation Cancer Molecular Diagnostics
 Liquid Biopsies Next Generation Cancer Molecular Diagnostics Forward Looking Statements 2 Statements pertaining to future financial and/or operating results, future research, diagnostic tests and technology
Liquid Biopsies Next Generation Cancer Molecular Diagnostics Forward Looking Statements 2 Statements pertaining to future financial and/or operating results, future research, diagnostic tests and technology
New Ideas. Better Medicines. Third Quarter Financial Results Conference Call
 New Ideas. Better Medicines. Third Quarter 2018 Financial Results Conference Call Forward-Looking Statements 2 This presentation contains forward-looking statements that involve substantial risks and uncertainties.
New Ideas. Better Medicines. Third Quarter 2018 Financial Results Conference Call Forward-Looking Statements 2 This presentation contains forward-looking statements that involve substantial risks and uncertainties.
More cancer patients are being treated with immunotherapy, but
 Bristol-Myers Squibb and Five Prime Present Phase 1a/1b Data Evaluating Cabiralizumab (anti-csf-1 receptor antibody) with Opdivo (nivolumab) in Patients with Advanced Solid Tumors PRINCETON, N.J. & SOUTH
Bristol-Myers Squibb and Five Prime Present Phase 1a/1b Data Evaluating Cabiralizumab (anti-csf-1 receptor antibody) with Opdivo (nivolumab) in Patients with Advanced Solid Tumors PRINCETON, N.J. & SOUTH
Building a Premier Oncology Biotech
 Wells Fargo Securities Healthcare Conference Building a Premier Oncology Biotech Dr. Helen Torley, President and CEO September 2018 Forward-Looking Statements All of the statements in this presentation
Wells Fargo Securities Healthcare Conference Building a Premier Oncology Biotech Dr. Helen Torley, President and CEO September 2018 Forward-Looking Statements All of the statements in this presentation
Determined to realize a future in which people with cancer live longer and better than ever before
 Determined to realize a future in which people with cancer live longer and better than ever before 4Q 2016 EARNINGS PRESENTATION MARCH 2017 1 Forward-looking statements disclosure This presentation contains
Determined to realize a future in which people with cancer live longer and better than ever before 4Q 2016 EARNINGS PRESENTATION MARCH 2017 1 Forward-looking statements disclosure This presentation contains
First self-administered antibody therapy for HIV in late-stage clinical trials. CytoDyn Annual Meeting of Stockholders August 24, 2017
 First self-administered antibody therapy for HIV in late-stage clinical trials CytoDyn Annual Meeting of Stockholders August 24, 2017 (OTCQB: CYDY) www.cytodyn.com Forward-Looking Statements This presentation
First self-administered antibody therapy for HIV in late-stage clinical trials CytoDyn Annual Meeting of Stockholders August 24, 2017 (OTCQB: CYDY) www.cytodyn.com Forward-Looking Statements This presentation
Clinical Study Clinical Study of the Relation between Mucosal Healing and Long-Term Outcomes in Ulcerative Colitis
 Hindawi Publishing Corporation Gastroenterology Research and Practice Volume 2013, Article ID 192794, 6 pages http://dx.doi.org/10.1155/2013/192794 Clinical Study Clinical Study of the Relation between
Hindawi Publishing Corporation Gastroenterology Research and Practice Volume 2013, Article ID 192794, 6 pages http://dx.doi.org/10.1155/2013/192794 Clinical Study Clinical Study of the Relation between
Bobby W. Sandage, Jr., PhD President & Chief Executive Officer. January 2012
 Bobby W. Sandage, Jr., PhD President & Chief Executive Officer January 2012 Statements in this presentation that are not descriptions of historical facts are forward-looking statements within the meaning
Bobby W. Sandage, Jr., PhD President & Chief Executive Officer January 2012 Statements in this presentation that are not descriptions of historical facts are forward-looking statements within the meaning
AXIM Biotechnologies Reports Year End 2017 Results
 March 19, 2018 AXIM Biotechnologies Reports Year End 2017 Results NEW YORK, March 19, 2018 (GLOBE NEWSWIRE) -- AXIM Biotechnologies, Inc. (AXIM Biotech) (OTC:AXIM), a world leader in cannabinoid research
March 19, 2018 AXIM Biotechnologies Reports Year End 2017 Results NEW YORK, March 19, 2018 (GLOBE NEWSWIRE) -- AXIM Biotechnologies, Inc. (AXIM Biotech) (OTC:AXIM), a world leader in cannabinoid research
Myeloid Differentiation Observed, Including Induction of CD38 in 85% of Evaluable Patients
 December 10, 2017 Syros Announces Initial Clinical Data from Ongoing Phase 2 Trial of SY-1425 Showing Biological and Clinical Activity as Single Agent in Genomically Defined AML and MDS Patients Clinical
December 10, 2017 Syros Announces Initial Clinical Data from Ongoing Phase 2 Trial of SY-1425 Showing Biological and Clinical Activity as Single Agent in Genomically Defined AML and MDS Patients Clinical
Edasalonexent (CAT-1004) Program
 Edasalonexent (CAT-1004) Program Oral small molecule designed to inhibit NF-κB for the treatment of Duchenne muscular dystrophy Joanne M. Donovan, MD, PhD Chief Medical Officer, Catabasis Pharmaceuticals
Edasalonexent (CAT-1004) Program Oral small molecule designed to inhibit NF-κB for the treatment of Duchenne muscular dystrophy Joanne M. Donovan, MD, PhD Chief Medical Officer, Catabasis Pharmaceuticals
Aurinia Pharmaceuticals Announces Voclosporin Meets Primary Endpoint in Phase IIB AURA-LV Study in Lupus Nephritis
 August 15, 2016 Aurinia Pharmaceuticals Announces Voclosporin Meets Primary Endpoint in Phase IIB AURA-LV Study in Lupus Nephritis Conference call and webcast at 8am ET First therapeutic agent to meet
August 15, 2016 Aurinia Pharmaceuticals Announces Voclosporin Meets Primary Endpoint in Phase IIB AURA-LV Study in Lupus Nephritis Conference call and webcast at 8am ET First therapeutic agent to meet
IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics. Jefferies 2014 Global Healthcare Conference Cynthia L. Sullivan, President and CEO
 IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics Oncology Autoimmune Diseases Jefferies 2014 Global Healthcare Conference Cynthia L. Sullivan, President and CEO Forward-Looking Statements This presentation,
IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics Oncology Autoimmune Diseases Jefferies 2014 Global Healthcare Conference Cynthia L. Sullivan, President and CEO Forward-Looking Statements This presentation,
Leerink Immuno-Oncology Roundtable Conference
 Leerink Immuno-Oncology Roundtable Conference September 28, 2017 NASDAQ:FPRX Forward-Looking Statements Disclaimer This presentation contains forward-looking statements within the meaning of the Private
Leerink Immuno-Oncology Roundtable Conference September 28, 2017 NASDAQ:FPRX Forward-Looking Statements Disclaimer This presentation contains forward-looking statements within the meaning of the Private
Combining HS-110 and anti-pd-1 in NSCLC. September 1, 2015
 Combining HS-110 and anti-pd-1 in NSCLC September 1, 2015 Forward Looking Statements This presentation includes statements that are, or may be deemed, forward-looking statements. In some cases, these forward-looking
Combining HS-110 and anti-pd-1 in NSCLC September 1, 2015 Forward Looking Statements This presentation includes statements that are, or may be deemed, forward-looking statements. In some cases, these forward-looking
Managing out-licensing collaborations: a big pharma perspective
 Managing out-licensing collaborations: a big pharma perspective Arne Wörn, PhD, Head Alliance Management IID European PLG Conference, Copenhagen, Sept. 5-6 These materials contain certain forward-looking
Managing out-licensing collaborations: a big pharma perspective Arne Wörn, PhD, Head Alliance Management IID European PLG Conference, Copenhagen, Sept. 5-6 These materials contain certain forward-looking
XP23829 PHASE 2 PSORIASIS TRIAL PRELIMINARY TOPLINE DATA PRESENTATION SEPTEMBER 15, 2015 COPYRIGHT 2015 XENOPORT, INC. ALL RIGHTS RESERVED.
 XP23829 PHASE 2 PSORIASIS TRIAL PRELIMINARY TOPLINE DATA PRESENTATION SEPTEMBER 15, 2015 COPYRIGHT 2015 XENOPORT, INC. ALL RIGHTS RESERVED. SAFE HARBOR DISCLAIMER These slides and the accompanying oral
XP23829 PHASE 2 PSORIASIS TRIAL PRELIMINARY TOPLINE DATA PRESENTATION SEPTEMBER 15, 2015 COPYRIGHT 2015 XENOPORT, INC. ALL RIGHTS RESERVED. SAFE HARBOR DISCLAIMER These slides and the accompanying oral
Microbiome GI Disorders
 Microbiome GI Disorders Prof. Ram Dickman Neurogastroenterology Unit Rabin Medical Center Israel 1 Key Points Our gut microbiota Were to find them? Symbiosis or Why do we need them? Dysbiosis or when things
Microbiome GI Disorders Prof. Ram Dickman Neurogastroenterology Unit Rabin Medical Center Israel 1 Key Points Our gut microbiota Were to find them? Symbiosis or Why do we need them? Dysbiosis or when things
AGM Presentation For the year to 30 September February 2016
 AGM Presentation For the year to 30 September 2015 25 February 2016 11 Forward Looking Statements This presentation includes forward-looking statements. These forward-looking statements involve known and
AGM Presentation For the year to 30 September 2015 25 February 2016 11 Forward Looking Statements This presentation includes forward-looking statements. These forward-looking statements involve known and
Two-stage study designed to evaluate tolerability and efficacy of pracinostat combined with azacitidine in patients with high and very high risk MDS
 Helsinn Group and MEI Pharma Announce First Patient Dosed in Phase 2 Dose-Optimization Study of Pracinostat and Azacitidine in Myelodysplastic Syndrome Two-stage study designed to evaluate tolerability
Helsinn Group and MEI Pharma Announce First Patient Dosed in Phase 2 Dose-Optimization Study of Pracinostat and Azacitidine in Myelodysplastic Syndrome Two-stage study designed to evaluate tolerability
Position of Biologics in IBD Circa 2006: Top Down vs. Step Up Therapy
 Position of Biologics in IBD Circa 2006: Top Down vs. Step Up Therapy Stephen B. Hanauer, MD University of Chicago Potential Conflicts: Centocor/Schering, Abbott, UCB, Elan, Berlex, PDL Goals of Treatment
Position of Biologics in IBD Circa 2006: Top Down vs. Step Up Therapy Stephen B. Hanauer, MD University of Chicago Potential Conflicts: Centocor/Schering, Abbott, UCB, Elan, Berlex, PDL Goals of Treatment
A Biopharmaceutical Company Focused on Controlled Immunotherapies and Point-of-Care Solutions
 A Biopharmaceutical Company Focused on Controlled Immunotherapies and Point-of-Care Solutions Second Quarter 2017 Company Update and Financial Results July 31, 2017 2 Forward-Looking Statements This presentation
A Biopharmaceutical Company Focused on Controlled Immunotherapies and Point-of-Care Solutions Second Quarter 2017 Company Update and Financial Results July 31, 2017 2 Forward-Looking Statements This presentation
-2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine. -June 2008: Recurrence of rectal blood loss and urgency
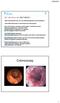 SD, male 40 yrs. old. (680718M467.) -2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine -June 2008: Recurrence of rectal blood loss and urgency Total colonoscopy: ulcerative rectitis,
SD, male 40 yrs. old. (680718M467.) -2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine -June 2008: Recurrence of rectal blood loss and urgency Total colonoscopy: ulcerative rectitis,
