The undersigned submit this petition under 42 U.S.C. 263b of the Mammogram Quality Standards Advisory Act of 1992 as amended and renewed in the
|
|
|
- Nathan Sherman
- 5 years ago
- Views:
Transcription
1 The undersigned submit this petition under 42 U.S.C. 263b of the Mammogram Quality Standards Advisory Act of 1992 as amended and renewed in the Mammogram Quality Standards Reauthorization Act of 1998 and 2004 to request the Commissioner of Food and Drugs: 1
2 Action Requested 1. The Plaintiffs Hallie Leighton and the Institute for Health Quality and Ethics hereby request the U.S. Food and Drug Administration to: 1. immediately and fully implement and enforce the patient notification amendment of the Mammogram Quality Standards Act of 1992, as amended in the Mammogram Quality standards Reauthorization Act of 1998 and 2004; 2. immediately terminate the unlawful practice of providing false and misleading information to women regarding their mammogram results; 3. immediately include all relevant and material information obtained from the mammogram in the report that is provided to patients as is statutorily required. Statement of Grounds 2. The Mammogram Quality Standards Act of 1992, as amended in the Mammogram Quality Standards Reauthorization Act of 1998 and 2004, was enacted by Congress to ensure that women have access to consistent and high quality breast cancer screening for the early detection of cancer. Early detection of breast cancer has a significant impact treatment success, long term chances of survival, and on the financial and personal costs of treatment. When a cancer is detected at less less than a centimeter, its impact can be minimized, and survival rates approach 100%. However, a woman s prognosis for survival decreases rapidly if the cancer begins to spread to lymph nodes and to other parts of the body. 3. Pursuant to the patient notification amendment of 42 U.S.C. 263b, a mammogram facility is required to promptly provide a patient the results of her mammogram in language that is easily understood by a layperson ( whether or not such a physician is available or there is no such physician, 2
3 a summary of the written report shall be sent directly to the patient in terms easily understood by a lay person. ) 4. The FDA s failure to adequately implement this statute has violated the rights of Ms. Leighton and other women similarly situated by denying them information necessary to make informed decisions regarding their health. 5. Ms. Leighton is one of a class of women who have radiographically dense breast tissue, which obscures the existence of cancers on a mammogram. Approximately 2/3 of women under the age of 50 and 1/4 of women over the age of 50 have breast tissue which contains enough dense tissue to obscure the existence of cancer on a mammogram. Both dense tissue and cancers appear white on a mammogram. 6. The American College of Radiology has used a four point scale, (BI-RADS (4 th ed.) scale for breast density) which categorizes a woman s breast density along a continuum as: Category 1 The breast is almost entirely fat (<25% glandular) Category 2 There are scattered fibroglandular densities (approximately 25-50% glandular) Category 3 The breast tissue is heterogeneously dense, which could obscure detection of small masses (approximately 51% - 75% glandular) Category 4 The breast tissue is extremely dense. This may lower the sensitivity of mammography (>76% glandular). 7. This information on breast tissue density has been utilized since before the patient notification requirements in 42 U.S.C. 263b were added to the MQSA Reauthorization of 1998, and it is provided to the patient s physician, but not to the patient on her mammogram results. 8. 2/3 of women under the age of 50 and ¼ of women over 50 have breast tissue that is heterogeneously dense (Category 3) or extremely dense (Category 4). For these women, the mammogram is not very effective due to the fact that both the dense tissue and cancer appear white 3
4 on the mammogram. For decades, peer reviewed scientific studies have confirmed that dense tissue masks the existence of cancers. For this significant number of women with dense breast tissue, a digital mammogram is unable to detect over 40% of cancers, and a film mammogram is significantly less effective, detecting as few as 27% of cancers. In addition to the fact that cancers may be masked by this dense tissue, density itself increases a woman s risk for breast cancer by 4-6 times. 9. The FDA has knowingly permitted the use of normal to report the results of mammograms to women in this population when no cancer is detected. The use of normal in lay language implies that no cancer is present with a reasonable degree of certainty. Most reasonable people would not categorize certainty which is slightly greater than 50% as a reasonable degree of certainty. A 27% chance of detecting a cancer is undeniably not a reasonable degree of certainty. The false and misleading application of normal in this instance is a serious violation of the public trust and a deterrent to those women who would consider using readily available supplemental screening. Furthermore, it is a direct violation of the federal statute which was designed to provide women important information regarding their own health. 10. Due to the failure of the FDA to adequately implement and enforce the reporting provision of the MQSA over a period of more than 10 years, millions of women: 1. have been provided false and misleading information regarding the results of their mammograms; 2. have been denied access to important information regarding their own bodies and their own health; 3. have been denied the right to make decisions about their own health with informed consent. 4
5 11. The purpose of this Citizen s Petition is to request that the FDA immediately terminate this practice of providing false and misleading information to patients and from withholding information which is necessary for patients to make informed decisions regarding their health care options. 12. The clear intent of the patient notification amendment was to ensure that women received full and accurate reports regarding their mammograms. It was enacted in response to inconsistent reporting and discussions between physicians and patients. The statute was enacted to leave no question that women were entitled to the information from their mammograms so that they could proactively discuss breast health issues with their physicians and make informed medical decisions. 13. The FDA clearly understood the intent of the patient notification requirement. On the FDA website, the organization provides the answers to Frequently Asked Questions: 1. Q: Must a facility provide the patient with a written lay summary even if the results are incomplete and additional imaging is needed? A: Yes. Even if the facility verbally transmits the results to the patient, the facility must provide, within 30 days of the examination, a written lay summary indicating that additional imaging is required. 2. Q: A facility performs non-mammographic breast imaging studies on the same day as the mammographic examination. Must the facility provide the patient a separate mammography lay summary, or can the facility provide a combined lay summary that represents the overall results and recommendations for all the breast imaging studies performed that day? A: The facility has the option of providing either a separate mammography lay summary or a combined lay summary. FDA recommends that, where feasible, the facility provide a combined lay summary, so as to give the patient the most complete and useful information. 14. Even in the absence of the MQSA patient notification statute, withholding material health information is a serious violation of medical ethics. Opinion of the American Medical 5
6 Association ethical guidelines states that withholding medical information from patients without their knowledge or consent is ethically unacceptable. 15. Withholding this material medical information is clearly a violation of well-established medical ethical standards. 16. Providing false and misleading information in the mammogram report is an egregious violation of the public trust which has resulted in irreparable and irreversible harm for thousands of women million women each year obtain a mammogram in the hopes of detecting breast cancer early, when it is most treatable. None of these women receive critical health information about their breast density directly from the physician who interprets their mammogram (with the exception of women in Connecticut and Texas, where legislation has been enacted to address the failure of the FDA to adequately enforce the MQSA). However, 40% of those women - 16 million women - who have dense tissue which obscures the existence of cancers receive a false and misleading mammogram report. 18. False negatives. Thousands of the women who receive these false and misleading reports actually do have an invasive breast cancer which is not detected by the mammogram. Like millions of other women, they receive a standard form letter which states We are happy to report that the results of your mammogram are normal. 19. We have estimated that of the tens of thousands of women who receive false negative mammogram reports each year, as many of 10,000 will die due to an undiagnosed cancer. (While 40 million women obtain mammograms, and 40,000 women die each year due to breast cancer, our calculations indicate that up to 25% of the deaths, (10,000 women) may be attributable to this practice of withholding information and providing false and misleading results.) 20. Petitioner Leighton received a negative mammogram report in 2006, and again in April She proactively sought breast cancer screening because all of her close relatives had been diagnosed with 6
7 breast cancer and she is Ashkenazi Jewish. Both of these factors indicate a higher risk for breast cancer. 21. Petitioner Leighton did not seek any of the readily available supplemental screening options because she was unaware that her breast tissue was dense, and she was unaware that the screening mammogram alone was not effective for her. 22. Petitioner Leighton was diagnosed with Stage IV breast cancer in April of 2010 at the age of 39. By 2010, the tumor had grown to 6 centimeters, had spread to her lymph nodes, and had metastasized to her spine. Stage IV is the most invasive stage of cancer, with the lowest rate of survival. In 2010, the National Breast Cancer foundation quoted the breast cancer five year survival rates as 98% for Stage I; 88% for Stage II; 56% for Stage IIIA; 49% for Stage IIIB, and 16% for Stage IV. 23. Had Petitioner Leighton known that a mammogram was not an effective screening tool for her, or that she could dramatically improve her chances of detecting cancer by supplementing the mammogram with a readily available ultrasound, she would have requested this from her physician. Had she known that there were other screening methods that were more reliable, she could have chosen one of these instead. 24. Tens of thousands of women every year who diligently obtain mammograms have been unnecessarily subjected to more harmful and less successful treatments for later stage breast cancer because this critical information is withheld from them. 25. Because the FDA has failed to adequately implement and enforce the MQSA patient notification statute, women have been forced to propose legislation at the state level to force radiologists to include information about breast density and the decreasing effectiveness of mammograms for the millions of women with dense breast tissue. A sample report from Connecticut has been included in this Petition, along with the false and misleading mammogram report that Plaintiff Leighton received. 7
8 26. Several organizations have lobbied extensively against legislation proposed at the state level which would address the FDA s shortfall in complying with the patient notification statute of the MQSA. None of the purported rationales for withholding material health information, or for providing false and misleading information are sound, ethical, or based in actual science. 27. There is no sound or ethical reason for withholding material health information from tens of millions of patients. Millions of women are being denied material health information in violation of the MQSA and the ethical standards of all known medical organizations. Tens of thousands suffer needlessly each year after relying on a false negative mammogram result, and an estimated 10,000 die each year as a result of their reliance on a false negative mammogram result. 28. Providing breast tissue density notification in the MQSA mandated patient letter, together with a standard warning regarding the lack of mammographic sensitivity for women with dense breast tissue, and the recommendation that women with dense breast tissue consult further with their physician(s) regarding the possible use of supplemental imaging, will further eliminate the confusion that will be the probable result of enactment of different legislation in each of the 50 states. 29. We are requesting that the FDA immediately terminate this practice and achieve compliance with the patient notification statute of the MQSA. Environmental Impact Statement 30. No environmental impact analysis is required in support of this Petition, and the granting of this Petition would not have any environmental impact. Economic Impact Statement 31. No economic impact statement is necessary. When the patient notification amendment was enacted in 1998, the FDA assessed the economic impact in the cost of additional postage and other costs 8
9 associated with mailing a notification to all mammogram patients. Each mammogram patient already receives a notification letter. 9
10 Certification 32. The undersigned certify that, to the best of their knowledge and belief, this Petition includes all information and views on which the Petition relies, and it includes representative data and information known to Petitioner which are unfavorable to the Petition. Petitioners Thomas W. Nerney Executive Director Institute for Health Quality and Ethics 75 Sprague Hill Road Chepachet, RI (401) Hallie Leighton c/o Institute for Health Quality and Ethics 75 Sprague Hill Road Chepachet, RI
11 11
12 12
13 13
14 14
Guidance for States considering legislation
 ACOG STATE LEGISLATIVE TOOLKIT: Breast Density and Screening Mandates Guidance for States considering legislation These legislative mandates are problematic: No reliable, standardized method for assessing
ACOG STATE LEGISLATIVE TOOLKIT: Breast Density and Screening Mandates Guidance for States considering legislation These legislative mandates are problematic: No reliable, standardized method for assessing
Henda s Law. Supplemental screening for women with dense breast tissue and increased risk
 . Henda s Law Supplemental screening for women with dense breast tissue and increased risk The 2011 Texas Legislature passed House Bill 2102 which is effective 1st September 2011. The law is informally
. Henda s Law Supplemental screening for women with dense breast tissue and increased risk The 2011 Texas Legislature passed House Bill 2102 which is effective 1st September 2011. The law is informally
Challenges to Delivery of High Quality Mammography
 Challenges to Delivery of High Quality Mammography Overview of Current Challenges Barbara Monsees, Washington University Geographic Access, Equity and Impact on Quality Tracy Onega, Dartmouth Medical School
Challenges to Delivery of High Quality Mammography Overview of Current Challenges Barbara Monsees, Washington University Geographic Access, Equity and Impact on Quality Tracy Onega, Dartmouth Medical School
IN THE COUNCIL OF THE DISTRICT OF COLUMBIA
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 A BILL 22-960 IN THE COUNCIL OF THE DISTRICT OF COLUMBIA To require health care facilities to notify patients of mammogram results
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 A BILL 22-960 IN THE COUNCIL OF THE DISTRICT OF COLUMBIA To require health care facilities to notify patients of mammogram results
Supplemental Screening for Women with Dense Breast Tissue. Public Meeting December 13, 2013
 Supplemental Screening for Women with Dense Breast Tissue Public Meeting December 13, 2013 Agenda Meeting Convened 10am-10:15am Presentation of the Evidence and Voting Questions, Q&A 10:15am 11:15am Discussion
Supplemental Screening for Women with Dense Breast Tissue Public Meeting December 13, 2013 Agenda Meeting Convened 10am-10:15am Presentation of the Evidence and Voting Questions, Q&A 10:15am 11:15am Discussion
Literature Review: California Senate Bill 1034 Health Care: Mammograms AT A GLANCE
 Literature Review: California Senate Bill 1034 Health Care: Mammograms Summary to the 2018-2019 California State Legislature, April 16, 2018 AT A GLANCE On February 26, 2018, the Senate Committee on Health
Literature Review: California Senate Bill 1034 Health Care: Mammograms Summary to the 2018-2019 California State Legislature, April 16, 2018 AT A GLANCE On February 26, 2018, the Senate Committee on Health
Introduction to Breast Density
 Introduction to Breast Density A/Prof Wendy Ingman Breast Biology & Cancer Unit Ethical and Legal Considerations in Breast Density Workshop 2018 In this presentation My interest in breast density What
Introduction to Breast Density A/Prof Wendy Ingman Breast Biology & Cancer Unit Ethical and Legal Considerations in Breast Density Workshop 2018 In this presentation My interest in breast density What
now a part of Electronic Mammography Exchange: Improving Patient Callback Rates
 now a part of Electronic Mammography Exchange: Improving Patient Callback Rates Overview This case study explores the impact of a mammography-specific electronic exchange network on patient callback rates
now a part of Electronic Mammography Exchange: Improving Patient Callback Rates Overview This case study explores the impact of a mammography-specific electronic exchange network on patient callback rates
Tissue Breast Density
 Tissue Breast Density Reporting breast density within the letter to the patient is now mandated by VA law. Therefore, this website has been established by Peninsula Radiological Associates (PRA), the radiologists
Tissue Breast Density Reporting breast density within the letter to the patient is now mandated by VA law. Therefore, this website has been established by Peninsula Radiological Associates (PRA), the radiologists
BREAST DENSITY WHAT IS IT? WHY IS IT IMPORTANT? & What IOWA SF250 Means to Patients and Providers
 BREAST DENSITY WHAT IS IT? WHY IS IT IMPORTANT? & What IOWA SF250 Means to Patients and Providers Arnold Honick, MD Radiology Consultants of Iowa, PLC ahonick@rciowa.com BREAST DENSITY LEGISLATION Nancy
BREAST DENSITY WHAT IS IT? WHY IS IT IMPORTANT? & What IOWA SF250 Means to Patients and Providers Arnold Honick, MD Radiology Consultants of Iowa, PLC ahonick@rciowa.com BREAST DENSITY LEGISLATION Nancy
Breast Density It's the Law
 Last year Iowa became the 30th state in the last 12 years to require that density information be added to the written mammogram report to the patient. This report is sent directly from the interpreting
Last year Iowa became the 30th state in the last 12 years to require that density information be added to the written mammogram report to the patient. This report is sent directly from the interpreting
The Radiology Aspects
 REQUIREMENTS FOR INTERNATIONAL ACCREDITATION OF BREAST CENTERS/UNITS The Radiology Aspects Miri Sklair-Levy, Israel RADIOLOGY GUIDELINES FOR QUALITY ASSURANCE IN BREAST CANCER SCREENING AND DIAGNOSIS Radiologists
REQUIREMENTS FOR INTERNATIONAL ACCREDITATION OF BREAST CENTERS/UNITS The Radiology Aspects Miri Sklair-Levy, Israel RADIOLOGY GUIDELINES FOR QUALITY ASSURANCE IN BREAST CANCER SCREENING AND DIAGNOSIS Radiologists
10.2 Summary of the Votes and Considerations for Policy
 CEPAC Voting and Policy Implications Summary Supplemental Screening for Women with Dense Breast Tissue December 13, 2013 The last CEPAC meeting addressed the comparative clinical effectiveness and value
CEPAC Voting and Policy Implications Summary Supplemental Screening for Women with Dense Breast Tissue December 13, 2013 The last CEPAC meeting addressed the comparative clinical effectiveness and value
Breast Density. Update 2018: Implications for Clinical Practice
 Breast Density Update 2018: Implications for Clinical Practice Matthew A. Stein, MD Assistant professor Breast Imaging Department of Radiology and Imaging Sciences University of Utah Health Disclosures
Breast Density Update 2018: Implications for Clinical Practice Matthew A. Stein, MD Assistant professor Breast Imaging Department of Radiology and Imaging Sciences University of Utah Health Disclosures
Session 2: The Role of Specialist Radiology Technologists
 Session 2: The Role of Specialist Radiology Technologists Louise M. Henderson, MSPH PhD Assistant Professor, Department of Radiology The University of North Carolina, Chapel Hill Overview Role of the technologist
Session 2: The Role of Specialist Radiology Technologists Louise M. Henderson, MSPH PhD Assistant Professor, Department of Radiology The University of North Carolina, Chapel Hill Overview Role of the technologist
Dense Breasts, Get Educated
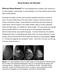 Dense Breasts, Get Educated What are Dense Breasts? The normal appearances to breasts, both visually and on mammography, varies greatly. On mammography, one of the important ways breasts differ is breast
Dense Breasts, Get Educated What are Dense Breasts? The normal appearances to breasts, both visually and on mammography, varies greatly. On mammography, one of the important ways breasts differ is breast
HISTORY OF MQSA AND ACR
 HISTORY OF MQSA AND ACR DEBORAH THAMES R.T. (R)(M)(QM) WHY MQSA? In the United States, there was a lack of standards in mammography imaging. Reporting Imaging Type of imaging screening/diagnostic Equipment
HISTORY OF MQSA AND ACR DEBORAH THAMES R.T. (R)(M)(QM) WHY MQSA? In the United States, there was a lack of standards in mammography imaging. Reporting Imaging Type of imaging screening/diagnostic Equipment
Disclosures. Breast Cancer. Breast Imaging Modalities. Breast Cancer Screening. Breast Cancer 6/4/2014
 : Information for the Primary Care Physician Disclosures No financial relationships with commercial entities producing health care products/services. Roxsann Roberts, MD Section Chief, MRI Erlanger/EmCare
: Information for the Primary Care Physician Disclosures No financial relationships with commercial entities producing health care products/services. Roxsann Roberts, MD Section Chief, MRI Erlanger/EmCare
Breast Density: Significance and Notification. Carol H. Lee Memorial Sloan-Kettering Cancer Center New York, NY
 Breast Density: Significance and Notification Carol H. Lee Memorial Sloan-Kettering Cancer Center New York, NY Significance of Breast Density Association with increased risk for breast cancer Decreased
Breast Density: Significance and Notification Carol H. Lee Memorial Sloan-Kettering Cancer Center New York, NY Significance of Breast Density Association with increased risk for breast cancer Decreased
Breast density. Understand what it is and why it matters
 Breast density Understand what it is and why it matters What is breast density? Breasts are composed of fibrous, glandular and fatty tissues. Breast density is a term that refers to the proportion of these
Breast density Understand what it is and why it matters What is breast density? Breasts are composed of fibrous, glandular and fatty tissues. Breast density is a term that refers to the proportion of these
Breast Imaging! Ravi Adhikary, MD!
 Breast Imaging! Ravi Adhikary, MD! ACS Estimated Cancers Statistics 2014! Breast! New Cases in Women! 232,670 (+67,570 in situ)! Deaths in Women! 40,000! Colon! 48,380! 24,040! Cervical! 12,360! 4,020!
Breast Imaging! Ravi Adhikary, MD! ACS Estimated Cancers Statistics 2014! Breast! New Cases in Women! 232,670 (+67,570 in situ)! Deaths in Women! 40,000! Colon! 48,380! 24,040! Cervical! 12,360! 4,020!
Breast density: imaging, risks and recommendations
 Breast density: imaging, risks and recommendations Maureen Baxter, MD Radiologist Director of Ruth J. Spear Breast Center Providence St. Vincent Medical Center Alison Conlin, MD/MPH Medical Oncologist
Breast density: imaging, risks and recommendations Maureen Baxter, MD Radiologist Director of Ruth J. Spear Breast Center Providence St. Vincent Medical Center Alison Conlin, MD/MPH Medical Oncologist
UNITED STATES DEPARTMENT OF HEALTH AND HUMAN SERVICES FOOD AND DRUG ADMINISTRATION
 UNITED STATES DEPARTMENT OF HEALTH AND HUMAN SERVICES FOOD AND DRUG ADMINISTRATION Citizen Petition to: Margaret A. Hamburg, M.D, Commissioner of Food and Drugs Docket No. For Review of Standard of Identity
UNITED STATES DEPARTMENT OF HEALTH AND HUMAN SERVICES FOOD AND DRUG ADMINISTRATION Citizen Petition to: Margaret A. Hamburg, M.D, Commissioner of Food and Drugs Docket No. For Review of Standard of Identity
GENERAL COMMENTS. The Task Force Process Should be Fully Open, Balanced and Transparent
 December 9, 2013 Submitted Electronically United States Preventive Services Task Force c/o Dr. Robert Cosby Agency for Healthcare Research and Quality 540 Gaither Road Rockville, MD 20850 RE: USPSTF Draft
December 9, 2013 Submitted Electronically United States Preventive Services Task Force c/o Dr. Robert Cosby Agency for Healthcare Research and Quality 540 Gaither Road Rockville, MD 20850 RE: USPSTF Draft
Breast density in quantifying breast cancer risk 9 December 2016
 Breast density in quantifying breast cancer risk 9 December 2016 cancer risk factors is still being debated. University of Malaya researchers, Prof. Kwan Hoong NG and his team published two papers on this
Breast density in quantifying breast cancer risk 9 December 2016 cancer risk factors is still being debated. University of Malaya researchers, Prof. Kwan Hoong NG and his team published two papers on this
Amammography report is a key component of the breast
 Review Article Writing a Mammography Report Amammography report is a key component of the breast cancer diagnostic process. Although mammographic findings were not clearly differentiated between benign
Review Article Writing a Mammography Report Amammography report is a key component of the breast cancer diagnostic process. Although mammographic findings were not clearly differentiated between benign
Frequently Asked Questions about Breast Density, Breast Cancer Risk, and the Breast Density Notification Law in California: A Consensus Document
 RSNA, 2013 Appendix E1 Frequently Asked Questions about Breast Density, Breast Cancer Risk, and the Breast Density Notification Law in California: A Consensus Document 1. I have been getting more questions
RSNA, 2013 Appendix E1 Frequently Asked Questions about Breast Density, Breast Cancer Risk, and the Breast Density Notification Law in California: A Consensus Document 1. I have been getting more questions
Mammography. What is Mammography? What are some common uses of the procedure?
 Mammography What is Mammography? Mammography is a specific type of imaging that uses a low-dose x-ray system to examine breasts. A mammography exam, called a mammogram, is used to aid in the early detection
Mammography What is Mammography? Mammography is a specific type of imaging that uses a low-dose x-ray system to examine breasts. A mammography exam, called a mammogram, is used to aid in the early detection
Mammographic Breast Density Classification by a Deep Learning Approach
 Mammographic Breast Density Classification by a Deep Learning Approach Aly Mohamed, PhD Robert Nishikawa, PhD Wendie A. Berg, MD, PhD David Gur, ScD Shandong Wu, PhD Department of Radiology, University
Mammographic Breast Density Classification by a Deep Learning Approach Aly Mohamed, PhD Robert Nishikawa, PhD Wendie A. Berg, MD, PhD David Gur, ScD Shandong Wu, PhD Department of Radiology, University
Barry W. Lovgren PO Box 6744 Gardnerville, NV (775) July 26, 2016
 Barry W. Lovgren PO Box 6744 Gardnerville, NV 89460 (775)265-2659 barrylovgren@yahoo.com July 26, 2016 Nevada Department of Health and Human Services Attn: Richard Whitley, Director 4126 Technology Way,
Barry W. Lovgren PO Box 6744 Gardnerville, NV 89460 (775)265-2659 barrylovgren@yahoo.com July 26, 2016 Nevada Department of Health and Human Services Attn: Richard Whitley, Director 4126 Technology Way,
5/24/16. Current Issues in Breast Cancer Screening. Breast cancer screening guidelines. Outline
 Disclosure information: An Evidence based Approach to Breast Cancer Karla Kerlikowske, MDDis Current Issues in Breast Cancer Screening Grant/Research support from: National Cancer Institute - and - Karla
Disclosure information: An Evidence based Approach to Breast Cancer Karla Kerlikowske, MDDis Current Issues in Breast Cancer Screening Grant/Research support from: National Cancer Institute - and - Karla
Screening for Breast Cancer
 Understanding Task Force Recommendations Screening for Breast Cancer U.S. Preventive Services Task Force (Task Force) has issued a final recommendation statement on Screening for Breast Cancer. se final
Understanding Task Force Recommendations Screening for Breast Cancer U.S. Preventive Services Task Force (Task Force) has issued a final recommendation statement on Screening for Breast Cancer. se final
Breast Ultrasound. Radiology Department. Patient information leaflet
 Breast Ultrasound Radiology Department Patient information leaflet This leaflet provides you with information about our breast ultrasound service. It explains what is involved and who you will meet. What
Breast Ultrasound Radiology Department Patient information leaflet This leaflet provides you with information about our breast ultrasound service. It explains what is involved and who you will meet. What
Policy / Drug and Alcohol-Free Workshops
 Policy 4118.235/4218.235 Drug and Alcohol-Free Workshops DATE: February 13, 2017 PREVIOUS ITEM: None ENCLOSURES: CABE s Suggested Policy 4118.235/4218.235 CABE s July 15, 2016 Policy Update REASON: To
Policy 4118.235/4218.235 Drug and Alcohol-Free Workshops DATE: February 13, 2017 PREVIOUS ITEM: None ENCLOSURES: CABE s Suggested Policy 4118.235/4218.235 CABE s July 15, 2016 Policy Update REASON: To
AN ACT. (S. B. 2016) (Conference) (No ) (Approved September 13, 2012)
 (S. B. 2016) (Conference) (No. 224-2012) (Approved September 13, 2012) AN ACT To amend Sections 1, 2, 3, 4, and 5 of Act No. 56-2006, better known as the Asthmatic School Children Treatment Act, in order
(S. B. 2016) (Conference) (No. 224-2012) (Approved September 13, 2012) AN ACT To amend Sections 1, 2, 3, 4, and 5 of Act No. 56-2006, better known as the Asthmatic School Children Treatment Act, in order
Breast Tomosynthesis. What is breast tomosynthesis?
 Scan for mobile link. Breast Tomosynthesis Breast tomosynthesis is an advanced form of mammography, a specific type of breast imaging that uses low-dose x-rays to detect cancer early when it is most treatable.
Scan for mobile link. Breast Tomosynthesis Breast tomosynthesis is an advanced form of mammography, a specific type of breast imaging that uses low-dose x-rays to detect cancer early when it is most treatable.
QUALITY REVIEW PROGRAM REVIEW OF FORENSIC ACCOUNTING ENGAGEMENT QUESTIONNAIRE
 QUALITY REVIEW PROGRAM REVIEW OF FORENSIC ACCOUNTING ENGAGEMENT QUESTIONNAIRE 2 Quality Review Program Review of forensic accounting engagement questionnaire Review Code(s) Reviewer Review Date INTRODUCTION
QUALITY REVIEW PROGRAM REVIEW OF FORENSIC ACCOUNTING ENGAGEMENT QUESTIONNAIRE 2 Quality Review Program Review of forensic accounting engagement questionnaire Review Code(s) Reviewer Review Date INTRODUCTION
Current issues and controversies in breast imaging. Kate Brown, South GP CME 2015
 Current issues and controversies in breast imaging Kate Brown, South GP CME 2015 JUDICIOUS USE OF RESOURCES IN REFERRALS FOR BREAST IMAGING THE DILEMMA How do target referrals for breast imaging? Want
Current issues and controversies in breast imaging Kate Brown, South GP CME 2015 JUDICIOUS USE OF RESOURCES IN REFERRALS FOR BREAST IMAGING THE DILEMMA How do target referrals for breast imaging? Want
Breast Density and Screening
 Breast Density and Screening Alberta Breast Cancer Screening Program Version: Jan 2019 What is this Booklet About? This is a guide to help you understand breast density and how it may affect you. Having
Breast Density and Screening Alberta Breast Cancer Screening Program Version: Jan 2019 What is this Booklet About? This is a guide to help you understand breast density and how it may affect you. Having
VERIFICATION OF VOLUNTARY DONATION FOR HUMAN EMBRYONIC STEM CELL RESEARCH
 VERIFICATION OF VOLUNTARY DONATION FOR HUMAN EMBRYONIC STEM CELL RESEARCH Connecticut General Statutes (C.G.S.) 19a-32d(d)(3) (as amended by P.A. No. 08-80) requires documentation to be provided to the
VERIFICATION OF VOLUNTARY DONATION FOR HUMAN EMBRYONIC STEM CELL RESEARCH Connecticut General Statutes (C.G.S.) 19a-32d(d)(3) (as amended by P.A. No. 08-80) requires documentation to be provided to the
As Introduced. 132nd General Assembly Regular Session S. B. No
 132nd General Assembly Regular Session S. B. No. 121 2017-2018 Senator Eklund Cosponsors: Senators Yuko, Schiavoni A B I L L To amend sections 1751.62, 3923.52, and 3923.54 of the Revised Code to include
132nd General Assembly Regular Session S. B. No. 121 2017-2018 Senator Eklund Cosponsors: Senators Yuko, Schiavoni A B I L L To amend sections 1751.62, 3923.52, and 3923.54 of the Revised Code to include
Breast Cancer Screening and High Risk
 Breast Cancer Screening and High Risk Mary Freyvogel, DO Breast Surgeon Clinical Assistant Professor of Surgery University Hospitals Case Medical Center St. John Medical Center / Elyria Medical Center
Breast Cancer Screening and High Risk Mary Freyvogel, DO Breast Surgeon Clinical Assistant Professor of Surgery University Hospitals Case Medical Center St. John Medical Center / Elyria Medical Center
DESCRIPTION: Percentage of final reports for screening mammograms that are classified as probably benign
 Measure #146 (NQF 0508): Radiology: Inappropriate Use of Probably Benign Assessment Category in Screening Mammograms National Quality Strategy Domain: Efficiency and Cost Reduction 2016 PQRS OPTIONS F
Measure #146 (NQF 0508): Radiology: Inappropriate Use of Probably Benign Assessment Category in Screening Mammograms National Quality Strategy Domain: Efficiency and Cost Reduction 2016 PQRS OPTIONS F
GAO MAMMOGRAPHY. Current Nationwide Capacity Is Adequate, but Access Problems May Exist in Certain Locations. Report to Congressional Requesters
 GAO United States Government Accountability Office Report to Congressional Requesters July 2006 MAMMOGRAPHY Current Nationwide Capacity Is Adequate, but Access Problems May Exist in Certain Locations GAO-06-724
GAO United States Government Accountability Office Report to Congressional Requesters July 2006 MAMMOGRAPHY Current Nationwide Capacity Is Adequate, but Access Problems May Exist in Certain Locations GAO-06-724
The FDA Food Safety Modernization Act: The Key New Requirements
 June 15, 2011 The FDA Food Safety Modernization Act: The Key New Requirements Executive Summary INSTITUTE FOR FOOD LAWS & REGULATIONS Michigan State University 140 G.M. Trout Building East Lansing, MI
June 15, 2011 The FDA Food Safety Modernization Act: The Key New Requirements Executive Summary INSTITUTE FOR FOOD LAWS & REGULATIONS Michigan State University 140 G.M. Trout Building East Lansing, MI
Authorized By: Elizabeth Connolly, Acting Commissioner, Department of Human
 HUMAN SERVICES 48 NJR 7(2) July 18, 2016 BUREAU OF GUARDIANSHIP SERVICES Decision-Making for the Terminally Ill Proposed Readoption with Amendments: N.J.A.C. 10:48B Authorized By: Elizabeth Connolly, Acting
HUMAN SERVICES 48 NJR 7(2) July 18, 2016 BUREAU OF GUARDIANSHIP SERVICES Decision-Making for the Terminally Ill Proposed Readoption with Amendments: N.J.A.C. 10:48B Authorized By: Elizabeth Connolly, Acting
Clinical Policy: Digital Breast Tomosynthesis Reference Number: CP.MP.90
 Clinical Policy: Reference Number: CP.MP.90 Effective Date: 01/18 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.90 Effective Date: 01/18 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
5.I.1. GENERAL PRACTITIONER ANNOUNCEMENT OF CREDENTIALS IN NON-SPECIALTY INTEREST AREAS
 Report of the Council on Ethics, Bylaws and Judicial Affairs on Advisory Opinion 5.I.1. GENERAL PRACTITIONER ANNOUNCEMENT OF CREDENTIALS IN NON-SPECIALTY INTEREST AREAS Ethical Advertising under ADA Code:
Report of the Council on Ethics, Bylaws and Judicial Affairs on Advisory Opinion 5.I.1. GENERAL PRACTITIONER ANNOUNCEMENT OF CREDENTIALS IN NON-SPECIALTY INTEREST AREAS Ethical Advertising under ADA Code:
Kish chakrabarti, Ph.D. Senior Physicist CDRH/FDA
 Facility Certification Extension Requirements, Quality Assurance and Medical Physicists role for Hologic Selenia Dimensions Digital Breast Tomosynthesis (DBT) System Kish chakrabarti, Ph.D. Senior Physicist
Facility Certification Extension Requirements, Quality Assurance and Medical Physicists role for Hologic Selenia Dimensions Digital Breast Tomosynthesis (DBT) System Kish chakrabarti, Ph.D. Senior Physicist
DESCRIPTION: Percentage of final reports for screening mammograms that are classified as probably benign
 Quality ID #146 (NQF 0508): Radiology: Inappropriate Use of Probably Benign Assessment Category in Screening Mammograms National Quality Strategy Domain: Efficiency and Cost Reduction 2018 OPTIONS F INDIVIDUAL
Quality ID #146 (NQF 0508): Radiology: Inappropriate Use of Probably Benign Assessment Category in Screening Mammograms National Quality Strategy Domain: Efficiency and Cost Reduction 2018 OPTIONS F INDIVIDUAL
April 30, By Electronic Mail
 April 30, 2018 By Electronic Mail Dr. Scott Gottlieb, Commissioner Office of the Commissioner Food and Drug Administration 10903 New Hampshire Avenue Silver Spring, MD 20993 CommissionerFDA@fda.hhs.gov
April 30, 2018 By Electronic Mail Dr. Scott Gottlieb, Commissioner Office of the Commissioner Food and Drug Administration 10903 New Hampshire Avenue Silver Spring, MD 20993 CommissionerFDA@fda.hhs.gov
The Comparative Clinical Effectiveness and Value of Supplemental Screening Tests Following Negative Mammography in Women with Dense Breast Tissue
 The New England Comparative Effectiveness Public Advisory Council Public Meeting December 13, 2013 The Comparative Clinical Effectiveness and Value of Supplemental Screening Tests Following Negative Mammography
The New England Comparative Effectiveness Public Advisory Council Public Meeting December 13, 2013 The Comparative Clinical Effectiveness and Value of Supplemental Screening Tests Following Negative Mammography
Advances in Breast Cancer Diagnosis and Treatment. Heidi Memmel, MD FACS Surgical Director of Caldwell Breast Center September 26, 2015
 Advances in Breast Cancer Diagnosis and Treatment Heidi Memmel, MD FACS Surgical Director of Caldwell Breast Center September 26, 2015 Advances in Breast Cancer Diagnosis and Treatment Recommendations
Advances in Breast Cancer Diagnosis and Treatment Heidi Memmel, MD FACS Surgical Director of Caldwell Breast Center September 26, 2015 Advances in Breast Cancer Diagnosis and Treatment Recommendations
Use of Standards in Substantial Equivalence Determinations
 Guidance for Industry and for FDA Staff Use of Standards in Substantial Equivalence Determinations Document issued on: March 12, 2000 U.S. Department Of Health And Human Services Food and Drug Administration
Guidance for Industry and for FDA Staff Use of Standards in Substantial Equivalence Determinations Document issued on: March 12, 2000 U.S. Department Of Health And Human Services Food and Drug Administration
Screening Mammograms: Questions and Answers
 CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Screening Mammograms:
CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Screening Mammograms:
Analysis of Senate Bill 173: Mammograms
 Analysis of Senate Bill 173: Mammograms A Report to the 2011-2012 California Legislature April 7, 2011 CHBRP 11-09 The California Health Benefits Review Program (CHBRP) responds to requests from the State
Analysis of Senate Bill 173: Mammograms A Report to the 2011-2012 California Legislature April 7, 2011 CHBRP 11-09 The California Health Benefits Review Program (CHBRP) responds to requests from the State
NONE. Disclosures. Accreditation Update
 ACR Ultrasound Accreditation: Requirements and Pitfalls Presented to: American Association of Physicists in Medicine Presented by: Jennifer Walter RDMS,RVT, RT(R) ACR Quality & Safety August 03, 2016 Disclosures
ACR Ultrasound Accreditation: Requirements and Pitfalls Presented to: American Association of Physicists in Medicine Presented by: Jennifer Walter RDMS,RVT, RT(R) ACR Quality & Safety August 03, 2016 Disclosures
2018 Farm Bill to Lift Federal Prohibition on Hemp Production, but State Laws May Restrict Certain Activities
 Debevoise Update D&P 2018 Farm Bill to Lift Federal Prohibition on Hemp Production, but State Laws May Restrict Certain Activities December 13, 2018 Earlier this week, the U.S. House of Representatives
Debevoise Update D&P 2018 Farm Bill to Lift Federal Prohibition on Hemp Production, but State Laws May Restrict Certain Activities December 13, 2018 Earlier this week, the U.S. House of Representatives
Cancer Screening Coverage Toolkit Colorectal and Breast Cancer
 Cancer Screening Coverage Toolkit Colorectal and Breast Cancer 1. Introduction 2. Colorectal Cancer Screening a. Background b. Talking Points c. Sample Legislation d. Sample Letter of Support e. Sample
Cancer Screening Coverage Toolkit Colorectal and Breast Cancer 1. Introduction 2. Colorectal Cancer Screening a. Background b. Talking Points c. Sample Legislation d. Sample Letter of Support e. Sample
Breast Density. Information for Health Professionals
 Breast Density Information for Health Professionals BreastScreen NSW provides free screening mammography to asymptomatic women aged 50-74 every two years, with the aim of diagnosing breast cancer at an
Breast Density Information for Health Professionals BreastScreen NSW provides free screening mammography to asymptomatic women aged 50-74 every two years, with the aim of diagnosing breast cancer at an
Imaging in breast cancer. Mammography and Ultrasound Donya Farrokh.MD Radiologist Mashhad University of Medical Since
 Imaging in breast cancer Mammography and Ultrasound Donya Farrokh.MD Radiologist Mashhad University of Medical Since A mammogram report is a key component of the breast cancer diagnostic process. A mammogram
Imaging in breast cancer Mammography and Ultrasound Donya Farrokh.MD Radiologist Mashhad University of Medical Since A mammogram report is a key component of the breast cancer diagnostic process. A mammogram
STATE BOARD OF COMMUNITY COLLEGES CURRICULUM PROGRAM APPLICATION (New to the System) Pitt Community College Breast Sonography (Certificate) (C45xxx)
 Attachment PROG 4 STATE BOARD OF COMMUNITY COLLEGES CURRICULUM PROGRAM APPLICATION (New to the System) The State Board of Community Colleges is asked to approve the curriculum program at the listed college
Attachment PROG 4 STATE BOARD OF COMMUNITY COLLEGES CURRICULUM PROGRAM APPLICATION (New to the System) The State Board of Community Colleges is asked to approve the curriculum program at the listed college
Supplemental Screening for Dense Breasts. Reagan Leverett, MD, MS
 Supplemental Screening for Dense Breasts Reagan Leverett, MD, MS Outline Anatomy and Density Risk of dense breasts Theory of Supplemental Screening Options for supplemental screening Tomosynthesis Ultrasound
Supplemental Screening for Dense Breasts Reagan Leverett, MD, MS Outline Anatomy and Density Risk of dense breasts Theory of Supplemental Screening Options for supplemental screening Tomosynthesis Ultrasound
S. Murgo, MD. Chr St-Joseph, Mons Erasme Hospital, Brussels
 S. Murgo, MD Chr St-Joseph, Mons Erasme Hospital, Brussels? Introduction Mammography reports are sometimes ambiguous and indecisive. ACR has developped the BIRADS. BIRADS consists of a lexicon in order
S. Murgo, MD Chr St-Joseph, Mons Erasme Hospital, Brussels? Introduction Mammography reports are sometimes ambiguous and indecisive. ACR has developped the BIRADS. BIRADS consists of a lexicon in order
AN ACT. The General Assembly of the Commonwealth of Pennsylvania hereby enacts as follows:
 Pennsylvania General Assembly http://www.legis.state.pa.us/cfdocs/legis/li/uconscheck.cfm?txttype=htm&yr=2014&sessind=0&smthlwind=0&act=89 07/17/2014 12:54 PM Home / Statutes of Pennsylvania / Unconsolidated
Pennsylvania General Assembly http://www.legis.state.pa.us/cfdocs/legis/li/uconscheck.cfm?txttype=htm&yr=2014&sessind=0&smthlwind=0&act=89 07/17/2014 12:54 PM Home / Statutes of Pennsylvania / Unconsolidated
Public Hearing Before U.S. Food and Drug Administration
 Substances in Food and Labeling; Regulatory Framework for Foods that Companies are Marketing as Functional Foods Public Hearing Before U.S. Food and Drug Administration Ilene Ringel Heller Senior Staff
Substances in Food and Labeling; Regulatory Framework for Foods that Companies are Marketing as Functional Foods Public Hearing Before U.S. Food and Drug Administration Ilene Ringel Heller Senior Staff
Running head: BREAST CANCER SCREENING 1
 Running head: BREAST CANCER SCREENING 1 Breast Cancer Screening Carol A. Lamoureux-Lewallen Briar Cliff University BREAST CANCER SCREENING 2 Breast Cancer Screening Policy Problem The federally appointed
Running head: BREAST CANCER SCREENING 1 Breast Cancer Screening Carol A. Lamoureux-Lewallen Briar Cliff University BREAST CANCER SCREENING 2 Breast Cancer Screening Policy Problem The federally appointed
SPECIAL DISCLAIMER FOR INTERPRETING SERVICES INVOLVING CALLS TO EMERGENCY SERVICE PROVIDERS (911/E911), OR LEGAL, MEDICAL OR MENTAL HEALTH ISSUES
 SPECIAL DISCLAIMER FOR INTERPRETING SERVICES INVOLVING CALLS TO EMERGENCY SERVICE PROVIDERS (911/E911), OR LEGAL, MEDICAL OR MENTAL HEALTH ISSUES Governing Language. The English language version of this
SPECIAL DISCLAIMER FOR INTERPRETING SERVICES INVOLVING CALLS TO EMERGENCY SERVICE PROVIDERS (911/E911), OR LEGAL, MEDICAL OR MENTAL HEALTH ISSUES Governing Language. The English language version of this
CITIZENS PETITION TO FOOD AND DRUG ADMINISTRATION. Division of Dockets Management Food and Drug Administration Room Fishers Lane
 CITIZENS PETITION TO FOOD AND DRUG ADMINISTRATION Division of Dockets Management Food and Drug Administration Room 1061 5630 Fishers Lane Rockville, MD 20852 ) ALLIANCE FOR NATURAL HEALTH USA ) 1350 Connecticut
CITIZENS PETITION TO FOOD AND DRUG ADMINISTRATION Division of Dockets Management Food and Drug Administration Room 1061 5630 Fishers Lane Rockville, MD 20852 ) ALLIANCE FOR NATURAL HEALTH USA ) 1350 Connecticut
North Carolina Department of Health & Human Services Division of Health Service Regulation Radiation Protection Section
 The Radiation Protection Section recently moved to a new department and division. Our physical location is the same, however we are now in the Department of Health and Human services and the Division of
The Radiation Protection Section recently moved to a new department and division. Our physical location is the same, however we are now in the Department of Health and Human services and the Division of
SENATE, No. 359 STATE OF NEW JERSEY. 217th LEGISLATURE PRE-FILED FOR INTRODUCTION IN THE 2016 SESSION
 SENATE, No. STATE OF NEW JERSEY th LEGISLATURE PRE-FILED FOR INTRODUCTION IN THE 0 SESSION Sponsored by: Senator RICHARD J. CODEY District (Essex and Morris) Senator JOSEPH F. VITALE District (Middlesex)
SENATE, No. STATE OF NEW JERSEY th LEGISLATURE PRE-FILED FOR INTRODUCTION IN THE 0 SESSION Sponsored by: Senator RICHARD J. CODEY District (Essex and Morris) Senator JOSEPH F. VITALE District (Middlesex)
RESOLUTION NO A PUBLIC HEARING TO CONSIDER THE ADOPTION OF A LOCAL LAW AMENDING CHAPTER 70 OF THE TOWN CODE ENTITLED "ZONING.
 Supervisor Bosworth offered the following resolution and moved its adoption, which resolution was declared adopted after a poll of the members of this Board: RESOLUTION NO. 561-2018 A PUBLIC HEARING TO
Supervisor Bosworth offered the following resolution and moved its adoption, which resolution was declared adopted after a poll of the members of this Board: RESOLUTION NO. 561-2018 A PUBLIC HEARING TO
Update in Breast Cancer Screening
 Disclosure information: Update in Breast Cancer Screening Karla Kerlikowske, MDDis Update in Breast Cancer Screening Grant/Research support from: National Cancer Institute and Grail - and - Karla Kerlikowske,
Disclosure information: Update in Breast Cancer Screening Karla Kerlikowske, MDDis Update in Breast Cancer Screening Grant/Research support from: National Cancer Institute and Grail - and - Karla Kerlikowske,
FDA s Nutrition Facts Panel and the Labeling of Added Sugars September 8, 2016 Bruce Silverglade Principal, OFW Law
 FDA s Nutrition Facts Panel and the Labeling of Added Sugars September 8, 2016 Bruce Silverglade Principal, OFW Law November 7, 2017 Sweetener Systems Conference bsilverglade@ofwlaw.com (1) 202 518 6316
FDA s Nutrition Facts Panel and the Labeling of Added Sugars September 8, 2016 Bruce Silverglade Principal, OFW Law November 7, 2017 Sweetener Systems Conference bsilverglade@ofwlaw.com (1) 202 518 6316
Policy Title. Control Number HR003. Exception The Scotland County Sheriff s Department is subject to a separate policy.
 Purpose To ensure compliance with federal regulations as outlined under the Drug-Free Workplace Act and by the U.S. Department of Transportation; to identify the conditions by which personnel are subject
Purpose To ensure compliance with federal regulations as outlined under the Drug-Free Workplace Act and by the U.S. Department of Transportation; to identify the conditions by which personnel are subject
Q: Why is breast cancer a big deal?
 I hate breast cancer. As a radiologist who specializes in breast imaging, my career is devoted to the detection and diagnosis of breast cancer. I am passionate about women s health and my goal is to find
I hate breast cancer. As a radiologist who specializes in breast imaging, my career is devoted to the detection and diagnosis of breast cancer. I am passionate about women s health and my goal is to find
perpetuate -- and perhaps even intensify -- that controversy. 1 On July 18th, the Fifth Circuit affirmed FDA s longstanding position that
 Food & Drug July 29, 2008 Fifth Circuit Rules that FDA May Regulate Compounded Drugs as New Drugs Update on Medical Center Pharmacy v. Mukasey For decades, the pharmacy compounding industry has disputed
Food & Drug July 29, 2008 Fifth Circuit Rules that FDA May Regulate Compounded Drugs as New Drugs Update on Medical Center Pharmacy v. Mukasey For decades, the pharmacy compounding industry has disputed
INGHAM COUNTY. Effective January 1, 2016 as amended November 10, 2015
 INGHAM COUNTY REGULATION TO REQUIRE A LICENSE FOR THE RETAIL SALE OF ELECTROINC SMOKING DEVICES, PROHIBIT SALE OF ELECTROINC SMOKING DEVICESTO MINORS, AND TO RESTRICT LOCATION OF ELECTROINC SMOKING DEVICES
INGHAM COUNTY REGULATION TO REQUIRE A LICENSE FOR THE RETAIL SALE OF ELECTROINC SMOKING DEVICES, PROHIBIT SALE OF ELECTROINC SMOKING DEVICESTO MINORS, AND TO RESTRICT LOCATION OF ELECTROINC SMOKING DEVICES
Executive summary. The usefulness of breast cancer screening
 Executive summary Health Council of the Netherlands. Population screening for breast cancer: expectations and developments. The Hague: Health Council of the Netherlands, 2014; publication no. 2014/01 Randomized
Executive summary Health Council of the Netherlands. Population screening for breast cancer: expectations and developments. The Hague: Health Council of the Netherlands, 2014; publication no. 2014/01 Randomized
Stanford Breast in the West: Multi-Modality Breast Imaging Symposium for Radiologists
 Course Overview Stanford Breast in the West: Multi-Modality Breast Imaging Symposium for Radiologists October 18-20, 2018 Monterey Plaza Hotel Monterey, CA A Continuing Medical Education Conference presented
Course Overview Stanford Breast in the West: Multi-Modality Breast Imaging Symposium for Radiologists October 18-20, 2018 Monterey Plaza Hotel Monterey, CA A Continuing Medical Education Conference presented
Breast cancer screening programme: France. Dr. Rosemary Ancelle-Park Department of health Office for chronic diseases
 Breast cancer screening programme: France Dr. Rosemary Ancelle-Park Department of health Office for chronic diseases French health care system General population of 65 Million GP clinics = 32 200 Radiology
Breast cancer screening programme: France Dr. Rosemary Ancelle-Park Department of health Office for chronic diseases French health care system General population of 65 Million GP clinics = 32 200 Radiology
Legislative Counsel s Digest:
 Senate Bill No. 250 Senator Carlton (by request) CHAPTER... AN ACT relating to dentistry and dental hygiene; revising various provisions governing the qualifications, examination and licensure of dentists
Senate Bill No. 250 Senator Carlton (by request) CHAPTER... AN ACT relating to dentistry and dental hygiene; revising various provisions governing the qualifications, examination and licensure of dentists
[ Special Feature ] In-house breast imaging unit at KK Breast Centre. Busting the Fear for. Breast Cancer
![[ Special Feature ] In-house breast imaging unit at KK Breast Centre. Busting the Fear for. Breast Cancer [ Special Feature ] In-house breast imaging unit at KK Breast Centre. Busting the Fear for. Breast Cancer](/thumbs/90/103887616.jpg) In-house breast imaging unit at KK Breast Centre. Busting the Fear for Breast Cancer by Yah Yuen Tan Consultant Surgeon, KK Breast Centre KK Women s and Children s Hospital, Singapore Sze Yiun Teo Consultant
In-house breast imaging unit at KK Breast Centre. Busting the Fear for Breast Cancer by Yah Yuen Tan Consultant Surgeon, KK Breast Centre KK Women s and Children s Hospital, Singapore Sze Yiun Teo Consultant
The early detection programme for breast cancer
 Making a wellinformed decision The early detection programme for breast cancer Why are women offered a mammogram through the quality controlled screening programme? Women between 50 and 69 years of age
Making a wellinformed decision The early detection programme for breast cancer Why are women offered a mammogram through the quality controlled screening programme? Women between 50 and 69 years of age
3/21/11 Tabar et al Lancet 2003;361:
 1 2 3 Tabar et al Lancet 2003;361:1405-1410 4 Tabar et al Lancet 2003;361:1405-1410 Tabar Rad Clin NA 2000;38:625-652, via R. Edward Hendrick, PhD, U. Colorado 5 6 7 8 Screening 40-49 50-59 60-69 Interval
1 2 3 Tabar et al Lancet 2003;361:1405-1410 4 Tabar et al Lancet 2003;361:1405-1410 Tabar Rad Clin NA 2000;38:625-652, via R. Edward Hendrick, PhD, U. Colorado 5 6 7 8 Screening 40-49 50-59 60-69 Interval
ADOPTED REGULATION OF THE STATE BOARD OF HEALTH. LCB File No. R Effective January 1, 2014
 ADOPTED REGULATION OF THE STATE BOARD OF HEALTH LCB File No. R100-13 Effective January 1, 2014 EXPLANATION Matter in italics is new; matter in brackets [omitted material] is material to be omitted. AUTHORITY:
ADOPTED REGULATION OF THE STATE BOARD OF HEALTH LCB File No. R100-13 Effective January 1, 2014 EXPLANATION Matter in italics is new; matter in brackets [omitted material] is material to be omitted. AUTHORITY:
ATHENA WISDOM INITIATIVE: RISK-BASED SCREENING FOR BREAST CANCER
 ATHENA WISDOM INITIATIVE: RISK-BASED SCREENING FOR BREAST CANCER The Screening Debate in the US? ACS vs. USPSTF When to start, when to stop, screening intervals, modality] Little guidance around operationalizing
ATHENA WISDOM INITIATIVE: RISK-BASED SCREENING FOR BREAST CANCER The Screening Debate in the US? ACS vs. USPSTF When to start, when to stop, screening intervals, modality] Little guidance around operationalizing
BREAST CANCER SCREENING IS A CHOICE
 BREAST CANCER SCREENING IS A CHOICE by ELAINE SCHATTNER, MD no financial disclosures (ES) American Association for Cancer Research Typical headlines focus on controversy 2 Data: Breast Cancer Incidence
BREAST CANCER SCREENING IS A CHOICE by ELAINE SCHATTNER, MD no financial disclosures (ES) American Association for Cancer Research Typical headlines focus on controversy 2 Data: Breast Cancer Incidence
LEGAL ASPECTS of MEDICAL MARIJUANA Florida Nurse Practitioner Network Annual Conference September 17, 2018
 LEGAL ASPECTS of MEDICAL MARIJUANA Florida Nurse Practitioner Network Annual Conference September 17, 2018 DISCLAIMER The material presented is general legal information and is not legal advice. Specific
LEGAL ASPECTS of MEDICAL MARIJUANA Florida Nurse Practitioner Network Annual Conference September 17, 2018 DISCLAIMER The material presented is general legal information and is not legal advice. Specific
The Dilemma of Breast Density in Screening
 The Dilemma of Breast Density in Screening Priscilla J. Slanetz MD, MPH, FACR, FSBI Associate Professor of Radiology, Harvard Medical School Beth Israel Deaconess Medical Center, Boston, MA No financial
The Dilemma of Breast Density in Screening Priscilla J. Slanetz MD, MPH, FACR, FSBI Associate Professor of Radiology, Harvard Medical School Beth Israel Deaconess Medical Center, Boston, MA No financial
Current Strategies in the Detection of Breast Cancer. Karla Kerlikowske, M.D. Professor of Medicine & Epidemiology and Biostatistics, UCSF
 Current Strategies in the Detection of Breast Cancer Karla Kerlikowske, M.D. Professor of Medicine & Epidemiology and Biostatistics, UCSF Outline ν Screening Film Mammography ν Film ν Digital ν Screening
Current Strategies in the Detection of Breast Cancer Karla Kerlikowske, M.D. Professor of Medicine & Epidemiology and Biostatistics, UCSF Outline ν Screening Film Mammography ν Film ν Digital ν Screening
WHAT TO EXPECT. Genius 3D MAMMOGRAPHY Exam. The most exciting advancement in mammography in over 30 years
 WHAT TO EXPECT Genius 3D MAMMOGRAPHY Exam The most exciting advancement in mammography in over 30 years 91% of patients agree the quality of care provided by the facility was better with a Genius 3D MAMMOGRAPHY
WHAT TO EXPECT Genius 3D MAMMOGRAPHY Exam The most exciting advancement in mammography in over 30 years 91% of patients agree the quality of care provided by the facility was better with a Genius 3D MAMMOGRAPHY
Z E N I T H M E D I C A L P R O V I D E R N E T W O R K P O L I C Y Title: Provider Appeal of Network Exclusion Policy
 TheZenith's Z E N I T H M E D I C A L P R O V I D E R N E T W O R K P O L I C Y Title: Provider Appeal of Network Exclusion Policy Application: Zenith Insurance Company and Wholly Owned Subsidiaries Policy
TheZenith's Z E N I T H M E D I C A L P R O V I D E R N E T W O R K P O L I C Y Title: Provider Appeal of Network Exclusion Policy Application: Zenith Insurance Company and Wholly Owned Subsidiaries Policy
Patient information leaflet. Royal Surrey County Hospital. NHS Foundation Trust. Having a Mammogram. Radiology Department
 Patient information leaflet Royal Surrey County Hospital NHS Foundation Trust Having a Mammogram Radiology Department This leaflet provides you with information about our mammography service (breast x-ray).
Patient information leaflet Royal Surrey County Hospital NHS Foundation Trust Having a Mammogram Radiology Department This leaflet provides you with information about our mammography service (breast x-ray).
UPMC 1 Delineation of Privileges Request Criteria Summary Sheet
 UPMC 1 Facility: Specialty: UPMC Altoona Radiology KNOWLEDGE TRAINING Successful Completion of an ACGME/AOA, accredited program. The successful completion of an approved (ACGME/AOA) post graduate residency
UPMC 1 Facility: Specialty: UPMC Altoona Radiology KNOWLEDGE TRAINING Successful Completion of an ACGME/AOA, accredited program. The successful completion of an approved (ACGME/AOA) post graduate residency
Patient information leaflet. Royal Surrey County Hospital. NHS Foundation Trust. Having a Mammogram. Radiology Department
 Patient information leaflet Royal Surrey County Hospital NHS Foundation Trust Having a Mammogram Radiology Department This leaflet provides you with information about our mammography service (breast x-ray).
Patient information leaflet Royal Surrey County Hospital NHS Foundation Trust Having a Mammogram Radiology Department This leaflet provides you with information about our mammography service (breast x-ray).
State of Connecticut Department of Education Division of Teaching and Learning Programs and Services Bureau of Special Education
 State of Connecticut Department of Education Division of Teaching and Learning Programs and Services Bureau of Special Education Introduction Steps to Protect a Child s Right to Special Education: Procedural
State of Connecticut Department of Education Division of Teaching and Learning Programs and Services Bureau of Special Education Introduction Steps to Protect a Child s Right to Special Education: Procedural
Screening Mammography as an example of Access to Healthcare A starting point for safe affordable medical treatment. Prof C A Benn
 Screening Mammography as an example of Access to Healthcare A starting point for safe affordable medical treatment Prof C A Benn to screen or not to screen may be To be or not the to be question? re national
Screening Mammography as an example of Access to Healthcare A starting point for safe affordable medical treatment Prof C A Benn to screen or not to screen may be To be or not the to be question? re national
April 7, You have asked me to prepare an opinion letter regarding the legal status of cannabidiol ( CBD ).
 Palmetto Synergistic Research, LLC d/b/a Palmetto Harmony Attn. Janel Ralph 216 Labonte Street, Unit B Conway, SC 29526 April 7, 2016 Re: Cannabidiol opinion letter Dear Ms. Ralph: You have asked me to
Palmetto Synergistic Research, LLC d/b/a Palmetto Harmony Attn. Janel Ralph 216 Labonte Street, Unit B Conway, SC 29526 April 7, 2016 Re: Cannabidiol opinion letter Dear Ms. Ralph: You have asked me to
Learning Objectives. 1. Identify which patients meet criteria for annual lung cancer screening
 Disclosure I, Taylor Rowlett, DO NOT have a financial interest /arrangement or affiliation with one or more organizations that could be perceived as a real or apparent conflict of interest in the context
Disclosure I, Taylor Rowlett, DO NOT have a financial interest /arrangement or affiliation with one or more organizations that could be perceived as a real or apparent conflict of interest in the context
