Circulating Tumor Cells in Solid Tumor in Metastatic and Localized Stages
|
|
|
- Kristopher McDowell
- 6 years ago
- Views:
Transcription
1 Circulating Tumor Cells in Solid Tumor in and Stages LUISA M. MAESTRO 1, JAVIER SASTRE 2, SARA B. RAFAEL 1, SILVIA B. VEGANZONES 1, MARTA VIDAURRETA 1, MIGUEL MARTÍN 2, CARLOS OLIVIER 3, VIRGINIA B. DE LA ORDEN 1, JOSE ANGEL GARCIA-SAENZ 2, ROSARIO ALFONSO 2, MANUEL ARROYO 1 and EDUARDO DIAZ-RUBIO 2 Departments of 1 Clinical Analysis, 2 Medical Oncology, and 3 Urology, Hospital Clínico San Carlos de Madrid, Martín Lagos s/n , Madrid, Spain Abstract. The aim of this study was the detection of circulating tumor cells (CTC) in three tumor types of epithelial origin. Patients and Methods: Four hundred and thirty-eight patients with breast cancer (56.2% localized and 43.8% metastatic), 195 with colorectal tumors (84.1% localized and 15.9% metastatic) and 50 with prostate cancer (52% localized and 48% metastatic) took part in this study. CTC quantification was performed using the CellSpotter Analyzer (Veridex LLC). Results: 31.5% of patients with cancer had 2 CTCs/7.5 ml but none of the healthy volunteers were above this level (p<0.001). Among patients with metastatic disease, 62.3% of them had 2 CTCs/7.5 ml but only 14.0% of those with localized disease were above this level (p<0.001). The presence of CTCs were correlated to stage in the three studied tumor types and no differences in the number of cells were found between them. Conclusion: The presence of more than 2 CTCs/7.5 ml is a frequent event in metastatic cases. In particular, patients with localized disease who have more than 2 CTCs/7.5 ml should be carefully studied to determine the possible prognostic and predictive value of this finding. Cancer metastasis is the result of several interacting processes at the end of which cancer cells survive in distant organs. Penetration of primary tumor cells into blood and lymph vessels is central to the whole phenomenon of metastasis and is a common step in all cancer types. The presence of circulating tumor cells (CTCs) in patients with tumors may be a surrogate marker of metastatic disease. Nevertheless, it should be taken into account that the interaction of a cancer cell with host defences and the microenviroment at distant tissues may result Correspondence to: Dra. M.L. Maestro, Servicio de Análisis Clínicos, Hospital Clínico San Carlos, C/Martín Lagos s/n , Madrid, España. Fax: , mmaestro.hcsc@ salud.madrid.org Key Words: Circulating tumor cells, solid tumor, metastatic, localized, stages. in its death and thus the process of metastasis is not successfully completed. The metastatic potential of primary tumors differs among different tumor types and even in a specific tumor not all cells are able to successfully disseminate. The presence of CTCs might be more linked with genotypic characteristics of the tumor cell rather than a sign of tumor burden, as the study of Braun et al. suggested (1). The detection of these cells in peripheral blood could have clinical usefulness in three aspects: i) as evidence of early dissemination, and then as a risk factor of clinical recurrence in tumors apparently limited to the primary organ; ii) as a relevant risk factor for metastatic progression and worse prognosis; and iii) as a predictive marker of response to treatment (2, 3). The determination of CTCs can be performed by means of different techniques which, in most cases, include the enrichment of tumor cells with immunomagnetic particles coated with antibodies specific for epithelial antigens. The identification of these isolated cells can be performed by immunohistochemistry or by the analysis of the genetic expression of cytokeratins and tumor antigens (4). The CellSearch detection system represents the first automated and standardized technology that has been approved by the US Food and Drug Administration (FDA) for the detection of CTCs in metastatic breast cancer (5). Subsequently this system was also approved by the FDA for CTC detection in patients with prostate and colon tumors. This technique is reproducible and allows the use of standard protocols for sample preparation and staining and for the interpretation of results. Allard et al. evaluated the accuracy, reproducibility, and linearity of this new detection system and they concluded that this technique has a good reproducibility profile (6). Riethdorf et al. performed a multicentric study to validate the CellSearch technology in patients with metastatic breast cancer and they confirmed its reliability for the assessment of therapeutic effectiveness (7). A prospective study on the incidence according to the clinical stage and prognostic significance of CTC detection in three different tumor types is being carrying out at our /2009 $
2 Table I. Number of circulating tumor cells per 7.5 ml of blood of patients with breast, colon and prostate tumors, and of healthy volunteers. No. of individuals (%) No. of Mean SD Median P 25 P 75 Range < patients Controls Healthy (100) Patients Tumor (68.6) 75 (10.9) 34 (4.9) 48 (7.0) 58 (8.6) Tumor Breast (65.8) 44 (10.0) 23 (5.2) 36 (8.2) 47 (10.8) Colon (77.4) 24 (12.3) 11 (5.6) 8 (4.1) 1 (0.6) Prostate (58.0) 7 (14.0) 0 (0) 4 (8.0) 10 (20.0) Stage (86.0) 42 (9.6) 12 (2.8) 6 (1.4) 1 (0.2) (37.6) 33 (13.4) 22 (8.9) 42 (17.0) 57 (23.1) P 25, 25th percentile; P 75, 75th percentile. centre. Here, we present preliminary results concerning similarities and differences in behaviour with respect to CTC detection among three cancer types with a high tendency to develop metastases, even in early clinical stages. Patients and Methods Patients. In this study, 789 blood samples were evaluated from 683 patients with breast, colon or prostate cancer, and 106 healthy volunteers. The inclusion criteria for healthy volunteers were: no evidence of disease at the moment of blood extraction, age under 35 years and no personal history of tumor pathology. In cancer patients, blood samples were collected before systemic therapy in those cases with metastatic disease and at least three weeks after the surgical operation and before receiving adjuvant treatment (if indicated) for those with only local disease. In all cases, pathologic diagnoses were made. The project was favourably validated by the Ethics and Clinical Investigation Committee of our hospital. Tumors with M0 classification according to the TNM Staging System (localized primary tumor with or without lymph node metastasis and with no distant metastasis) were considered as localized and M1 (tumor with distant metastasis) as metastatic. A total of 438 patients diagnosed with breast cancer (56.2% in localized stages and 43.8% metastatic), 195 with colorectal tumors (84.1% localized and 15.9% metastatic) and 50 with prostate cancer (52% localized and 48% metastatic) entered into the study. Overall, 36.1% of the selected patients had metastases (247/683). Circulating tumor cell detection. Blood collection was carried out in a 10 ml tube with EDTA and cellular preservative (CellSave tubes; Immunicon Corporation, Pennsylvania, USA) and samples were stored for a maximum of 72 hours at room temperature before their processing. Blood (7.5 ml) was then added to a ferrofluid of immunomagnetic particles coated with antibodies of an antiepithelial adhesion molecule (EpCAM). The isolated cells were fluorescently stained with the nucleic acid dye 4,6-diaminodino-2- phenylindole (DAPI) for the cellular nucleus, and labelled with monoclonal antibodies specific for leukocytes (CD45) or epithelial cells (cytokeratin 8, 18 and 19-phycoerythrin)(CK-PE). They were analyzed with a CellSpotter Analyzer (Veridex LLC; NJ, USA). Cellular images obtained with this technisque were reviewed and matched as CTCs manually. To be classified as CTCs, cells had to fulfil the following criteria: round to oval morphology, greater than 4 μm diameter, cytoplasm positively stained with CK-PE and negatively for CD45, as well as more than 50% of the DAPI being stained nucleus inside of the cytoplasm. The results were expressed as the number of CTCs per 7.5 ml of blood. The samples were processed in a blinded manner and result interpretation was independently accomplished by five specialists specifically trained and with no information about the clinical status of patients. Every sample was reviewed at least by two of the specialists to avoid interreader variations. Statistical analysis. The association between qualitative variables was evaluated using the Chi-square test and, when more than 25% of the expected frequencies fell bellow 5, Fisher s exact test was applied instead. A receiver operating characteristic (ROC) curve of the CTC value was constructed to determine the cut-off point to discriminate between localized and metastatic cases. The sensitivity and specificity as well as the area under the curve with its 95% confidence interval (CI) are presented. The stratified analysis was performed according to tumor type. In every test the null hypothesis was rejected with a type I error of 5%. The analysis was performed using SPSS (Statistical Product and Service Solutions, Illinois, USA) for Windows v.11.5 software. Results In the healthy volunteer group, 100% of the individuals showed fewer than 2 CTCs/7.5ml, 18 out of 106 had 1 CTC (17%) (Table I). The number and percentage of samples with <2, 2-4, 5-9, and 50 CTCs/7.5 ml are shown in Table I. According to our results, and considering the CTC values observed in healthy individuals, we established the cut-off point at 2 CTCs/7.5ml to set the limit of pathological detection. This criterion is in agreement with the cut-off chosen by Allard et al. (6). All the statistical analyses performed here used this cut-off point (Table II). Overall, 31.5% of patients with cancer had more than 2 CTCs/7.5ml, which is significantly higher compared with healthy volunteers (p<0.001). According to disease stage (localized 4840
3 Maestro et al: Circulating Tumor Cells in Patients with Solid Tumor Table II. Analysis of the differences in the prevalence of CTC according to different thresholds. <2 (%) 2 (%) p-value <5 (%) 5 (%) p-value <10 (%) 10 (%) p-value <50 (%) 50 (%) p-value Tumor type Breast 214 (87.0) 32 (13.0) < (95.1) 12 (11.3) < (98.0) 5 (2.0) < (99.6) 1 (0.4) < (38.5) 118 (61.5) 98 (51.0) 94 (49.0) 114 (59.4) 78 (40.6) 146 (76.0) 46 (24.0) Colon 139 (84.8) 25 (15.2) < (95.7) 7 (4.3) < (98.8) 2 (1.2) < (100) 0 (0) (38.7) 19 (61.3) 18 (58.1) 13 (41.9) 24 (77.4) 7 (22.6) 30 (96.8) 1 (0.5) Prostate 22 (84.6) 4 (15.4) < (100) 0 (0) < (100) 0 (0) < (100) 0 (0) < (29.2) 17 (70.8) 10 (41.7) 14 (58.3) 10 (41.7) 14 (58.3) 14 (58.3) 10 (41.7) Tumor stage Breast 214 (87.0) 32 (13.0) (95.1) 12 (4.9) (98.0) 5 (2.0) (99.6) 1 (0.4) Colon 139 (84.8) 25 (15.2) 157 (95.7) 7 (4.3) 162 (98.8) 2 (1.2) 164 (100) 0 (0) Breast 74 (38.5) 118 (61.5) (51.0) 94 (49.0) (59.4) 78 (40.6) (76.0) 46 (24.0) Colon 12 (38.7) 19 (61.3) 18 (58.1) 13 (41.9) 24 (77.4) 7 (22.6) 30 (96.8) 1 (3.2) Breast 214 (87.0) 32 (13.0) (95.1) 12 (4.9) (98.0) 5 (2.0) (99.6) 1 (0.4) Prostate 22 (84.6) 4 (15.4) 26 (100) 0 (0) 26 (100) 0 (0) 26 (100) 0 (0) Breast 74 (38.5) 118 (61.5) (51.0) 94 (49.0) (59.4) 78 (40.6) (76.0) 46 (24.0) 0.08 Prostate 7 (29.2) 17 (70.8) 10 (41.7) 14 (58.3) 10 (41.7) 14 (58.3) 14 (58.3) 10 (41.7) Colon 139 (84.8) 25 (15.2) (95.7) 7 (4.3) (98.8) 2 (1.2) (100) 0 (0) Prostate 22 (84.6) 4 (15.4) 26 (100) 0 (0) 26 (100) 0 (0) 26 (100) 0 (0) Colon 12 (38.7) 19 (61.3) (58.1) 13 (41.9) (77.4) 7 (22.6) (96.8) 1 (3.2) Prostate 7 (29.2) 17 (70.8) 10 (41.7) 14 (58.3) 10 (41.7) 14 (58.3) 14 (58.3) 10 (41.7) vs. metastatic), CTCs were present in 62.3% of patients with metastatic disease (regardless of tumor type), whereas only in 14% of patients with localized disease were they present (p<0.001). With regard to the analysis carried out for each tumor type, 61.5% of patients with metastatic breast tumors had 2 CTCs/7.5 ml and 13.0% of the localized cases (p<0.001). In colorectal carcinomas, 61.3% of patients with metastasis had 2 CTCs/7.5 ml, but only 15.2% in localized stages (p<0.001). In prostate tumors, the difference between the patients with metastatic and localized stages was also statistically significant (70.8% vs. 15.4%) (p<0.001) (Table II). The analysis of CTCs among the differences tumor types and according to the tumor stage revealed no statistically significant differences between breast, colon and prostate tumors, neither in localized nor in metastatic stages (Table II). Significant differences between CTCs in patients with breast and those with colon metastatic tumors were observed when considering 50 CTCs as the cut-off point (24.0% vs. 3.2%). The same occurred for metastatic colon and prostate tumors with 10 CTCs (22.6% vs. 58.3%) and 50 CTCs (3.2% vs. 41.7%) (Table II). ROC curves were constructed to determine the cut-off value which better discriminated between localized and metastatic stages. ROC curves confirmed the cut-off point for the three types of tumors to be 2 CTCs/7.5ml. The area under the curve for breast cancer cases was 0.76 (95% CI= ), for colorectal cancer was 0.79 (95% CI= ) and for prostate tumors was 0.84 (95% CI= ). Discussion The detection of CTCs has been pointed out as a potential useful prognostic factor and of predictive value for tumor recurrence and response to treatment. For this reason, several attempts have been made to find a reliable method to measure them. The latest methods for CTC detection have included cytometry with immunocytochemical labelling and immunomagnetic enrichment coupled to DNA and RNA technologies. The only automated and highly reproducible system available so far is the one used in this study and combines immunomagnetic enrichment and immunofluorescent labelling. According to the criteria applied in our 4841
4 technology, a CTC is a round to oval cell, immunomagnetically selected with antibody anti-epcam with nucleus (DAPI stained) and a cytoplasm positive for antibody anti-ck 8, 18 and 19 and negative for anti-cd45. These markers have proven to be valid for the detection of epithelial cells arising from different tumor types in peripheral blood (6, 7-10). Our results of CTCs detection in healthy volunteers confirm those reported by Allard et al. (6). A threshold of 2 CTCs/7.5 ml discriminates between cancer patients and healthy people. Nevertheless, it does not mean that this is the appropriate cutoff when used as prognostic factor or for monitoring response to treatment. In fact, a threshold of 6 CTCs and 3 CTCs have been established in metastatic breast and colon cancer respectively when used as a predictive factor for response to chemotherapy (9, 11). The presence of CTCs correlated with stage: with 62.3% of patients with metastatic tumors having 2 CTCs/7.5 ml in contrast to those with the localized tumors at 14.0%. At the moment, we do not have enough surveillance of our patients to determine if the presence of CTCs in nonmetastatic stages is a surrogate marker of metastatic disease, but in the near future, CTC determination may be of help to select people at high risk of recurrence. Adjuvant treatment with hormonal therapy or chemotherapy is sometimes not indicated in early stages of these tumors because of the low expected rate of recurrence. CTC determination could be a simple test to select patient candidates for adjuvant treatment if the prognostic value for recurrence can be demonstrated. Allard et al. found in their study of metastatic tumors that the presence and the number of CTC varied depending on the tumor type. They suggested that these differences can be explained by differences in tumor vascularization, sites of metastasis and tumor aggressiveness (6). However, we did not observe any significant differences in the CTC quantification among breast, colon and prostate tumors, when stratifying by stage and using a threshold of 2 CTCs. The percentage of patients with CTC in localized and metastatic stages was similar in the three studied tumor types (localized stages of breast 13%, colon 15.2% and prostate 15.4% and metastatic stages of breast 61.5%, colon 61.3% and prostate 70.8%). It should be taken into account that all three tumor types tend to develop hematogenous metastasis frequently, even in apparently early stages. It would be interesting to evaluate the presence of CTCs in patients with tumors with low risk of distant dissemination, such as of the head and neck, or endometrial carcinoma. The differences between localized and metastatic stages were still significant when thresholds of 5, 10 or 50 CTCs/7.5 ml was considered. There is one exception, in that for patients with colon tumors, the 50 CTCs limit could not be evaluated because only one patient had more than 50 CTCs/7.5 ml. We found that the cut-off point which better discriminates between localized and metastatic stages was 2 CTCs/7.5ml and this threshold was applied in the analyses for the three types of tumors. The mean number of CTCs (±SD) observed in localized tumors was 1 (±5) /7.5 ml and the maximum was 86 CTCs. In some patients at metastatic stages, no CTCs were detected (93/247). We excluded patients under systemic therapy to avoid the effect of therapy on CTCs. Another possibility to take into account is that cancer cells might not continuously pass into blood vessels. To test this hypothesis, we carried out an assay to examine the CTC release from the metastatic sites in patients with metastatic breast cancer, and we observed no circadian pattern but a constant flow throughout the day (11). In some cases, a preferential pattern of dissemination throughout the lymphatic vessels may explain the absence of CTCs from the bloodstream. Several studies have recently been reported which used the CellSearch system in patients with metastatic breast cancer. Overall, the percentage of patients with >5CTCs/7.5 ml ranged from 24 to 45% (6, 7, 12, 13). Different studies have shown that CTCs in patients with metastatic breast cancer are related to diseasefree survival (DFS) and with overall survival (OS), as well as with prediction of therapeutic response (14). No studies in patients with localized tumors have been carried out to date. Regarding our population of patients with colorectal carcinoma, 61.3% of those with metastatic tumors had 2 CTCs/7.5 ml. Allard et al. studied 196 patients with metastatic colorectal carcinoma and 30% had 2 CTCs/7.5 ml. This percentage is significantly lower than that observed in our population (6). In a recent study of 456 metastatic colorectal cancer patients, Meropol et al. demonstrated that the number of CTCs evaluated using the CellSearch system is an independent prognostic factor for OS and DFS (9). We are the only group with published results of CTC in localized stages of colorectal cancer using the CellSearch system. In a previous study, we found a correlation between the number of CTCs and the tumor stage in this tumor type (8). In our experience, 70.8% of patients with metastatic prostate tumors had 2 CTCs/7.5 ml and 41.7% 50 CTCs/7.5 ml. Shaffer et al. quantified CTCs using the CellSearch system in 59 patients with metastatic prostate cancer: 84.7% presented 2 CTCs/7.5 ml and 30.5% 50 CTCs/7.5 ml (10). Allard et al. studied 123 patients with metastatic prostate tumors: 57% had 2 CTCs/7.5 ml and 14% 50 CTCs/7.5 ml (6). In conclusion, CTC detection by the CellSearch system is an easy and highly reproducible method that deserves more evaluation in epithelial tumors. A cut-off of 2 CTCs can be considered as pathological, although the threshold for prognostic significance may differ among the different tumor types. The number of CTCs detected was significantly higher in the metastatic stage compared with earlier stages, and no differences were found among epithelial tumors with high affinity for hematogenous dissemination such as breast, colon and prostate adenocarcinomas (15). A longer followup is necessary in our series to assess the prognostic value of the presence of CTCs and the predictive value for response to systemic therapy. 4842
5 Maestro et al: Circulating Tumor Cells in Patients with Solid Tumor Acknowledgements Financial support for this research was provided by FISS PI , Sanofi-Aventis, and Fundación Mutua Madrileña Automovilística. References 1 Braun S, Hepp F, Kentenich C, Janni W, Pantel K, Riethmüller G, Willgeroth F and Sommer HL: Monoclonal antibody therapy with edrecolomab in breast cancer patients: monitoring of elimination of disseminated cytokeratin-positive tumor cells in bone marrow. Clin Cancer Res 5: , Braun S and Naume B: Circulating and disseminated tumor cells. J Clin Oncol 23: , Braun S and Marth C: Circulating tumor cells in metastatic breast cancer toward individualized treatment? N Engl J Med 351: , Allen-Mersh TG, McCullough TK, Patel H, Wharton RQ, Glover C and Jonas SK: Role of circulating tumour cells in predicting recurrence after excision of primary colorectal carcinoma. Br J Surg 94: , Cristofanilli M, Hayes DF, Budd T, Ellis MJ, Stopeck A, Reuben JM, Doyle GV, Matera J, Allard WJ, Miller MC, Fritsche HA, Hortobagy GN and Terstappen LW: Circulating tumor cells: a novel prognostic factor for newly diagnosed metastatic breast cancer. J Clin Oncol 23: , Allard WJ, Matera J, Miller MC, Repollet M, Connelly MC, Rao C, Tibbe AG, Uhr JW and Terstappen LW: Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin Cancer Res 10: , Riethdorf S, Fritsche H, Müller V, Rau T, Schindlbeck C, Rack B, Janni W, Coith C, Beck K, Jänicke F, Jackson S, Gornet T, Cristofanilli M and Pantel K: Detection of circulating tumor cells in peripheral blood of patients with metastatic breast cancer: a validation study of the CellSearch System. Clin Cancer Res 13: , Sastre J, Maestro ML, Puente J, Veganzones S, Alfonso R, Rafael S, Garcia-Saenz JA, Vidaurreta M, Martin M, Arroyo M, Sanz-Casla MT and Díaz-Rubio E: Circulating tumor cells in colorectal cancer: correlation with clinical and pathological variables. Ann Oncol 19: , Meropol NJ, Cohen SJ, Iannotti N, Saidman BH, Sabbath KD, Miller MC, Doyle GV, Tissing H, Terstappen LW and Punt CJ: Circulating tumor cells (CTC) predict progression free (PFS) and overall survival (OS) in patients with metastatic colorectal cancer. Proc Am Soc Clin Oncol 25: 18S (abstract 4010), Schaffer DR, Leversha MA, Danila DC, Lin O, Gonzalez- Espinoza R, Gu B, Anand A, Smith K, Maslak P, Doyle GV, Terstappen LW, Lilja H, Heller G, Fleisher M and Scher HI: Circulating tumor cell analysis in patients with progressive castration-resistant prostate cancer. Clin Cancer Res 13: , Hayes DF, Cristofanilli M, Budd GT, Matthew JE, Stopeck A, Miller MC, Matera J, Allard WJ, Doyle GW and Terstappen LW: Circulating tumor cells at each follow-up time point during therapy of metastatic breast cancer patients predict progressionfree and overall survival. Clin Cancer Res 12: , Garcia-Saenz JA, Martín M, Maestro M, Vidaurreta M, Veganzones S, Villalobos L, Rodríguez-Lajusticia L, Rafael S, Sanz-Casla MT, Casado A, Sastre J, Arroyo M and Díaz-Rubio E: Circulating tumor cells lack circadian-rhythm in hospitalized metastasic breast cancer patients. Clin Transl Oncol 8: , Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Matera J, Miller MC, Reuben JM, Doyle GV, Allard WJ, Terstappern LW and Hayes DF: Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med 351: , Budd GT, Cristofanilli M, Ellis MJ, Stopeck A, Borden E, Miller MG, Matera J, Repollet M, Doyle M, Terstappern LW and Hayes DF: Circulating tumor cells versus imaging-predicting overall survival in metastatic breast cancer. Clin Cancer Res 12: , Weitz J, Kienle P, Magener A, Koch M, Schrödel A, Willeke F, Autschbach F, Lacroix J, Lehnert T, Herfarth C and von Knebel Doeberitz M: Detection of disseminated colorectal cancer cells in lymph nodes, blood and bone marrow. Clin Cancer Res 5: , Received June 3, 2009 Revised September 25, 2009 Accepted September 28,
In most industrialized countries, primary lung cancer is. Circulating Tumor Cells in Pulmonary Venous Blood of Primary Lung Cancer Patients
 Circulating Tumor Cells in Pulmonary Venous Blood of Primary Lung Cancer Patients Yoshitomo Okumura, MD, Fumihiro Tanaka, MD, PhD, Kazue Yoneda, Masaki Hashimoto, MD, Teruhisa Takuwa, MD, Nobuyuki Kondo,
Circulating Tumor Cells in Pulmonary Venous Blood of Primary Lung Cancer Patients Yoshitomo Okumura, MD, Fumihiro Tanaka, MD, PhD, Kazue Yoneda, Masaki Hashimoto, MD, Teruhisa Takuwa, MD, Nobuyuki Kondo,
External Quality Assurance of Circulating Tumor Cell Enumeration Using the CellSearch V R. System: A Feasibility Study
 Cytometry Part B (Clinical Cytometry) 80B:112 118 (2011) Original Article External Quality Assurance of Circulating Tumor Cell Enumeration Using the CellSearch V R System: A Feasibility Study Jaco Kraan,
Cytometry Part B (Clinical Cytometry) 80B:112 118 (2011) Original Article External Quality Assurance of Circulating Tumor Cell Enumeration Using the CellSearch V R System: A Feasibility Study Jaco Kraan,
Review Article Significance of Circulating Tumor Cells Detected by thecellsearchsysteminpatientswithmetastaticbreast Colorectal and Prostate Cancer
 Oncology Volume, Article ID 67, 8 pages doi:.55//67 Review Article Significance of Circulating Tumor Cells Detected by thecellsearchsysteminpatientswithmetastaticbreast Colorectal and Prostate Cancer M.
Oncology Volume, Article ID 67, 8 pages doi:.55//67 Review Article Significance of Circulating Tumor Cells Detected by thecellsearchsysteminpatientswithmetastaticbreast Colorectal and Prostate Cancer M.
Circulating Tumor Cells in non- Metastatic Triple Negative Breast Cancer
 Circulating Tumor Cells in non- Metastatic Triple Negative Breast Cancer Carolyn Hall, Ph.D. Department of Surgical Oncology The University of Texas MD Anderson Cancer Center Triple Negative Breast Cancer
Circulating Tumor Cells in non- Metastatic Triple Negative Breast Cancer Carolyn Hall, Ph.D. Department of Surgical Oncology The University of Texas MD Anderson Cancer Center Triple Negative Breast Cancer
Page: 1 of 10. Detection of Circulating Tumor Cells in the Management of Patients with Cancer
 Last Review Status/Date: September 2015 Page: 1 of 10 Management of Patients with Cancer Description The prognosis of cancer patients is often determined by the occurrence of metastatic disease. Studies
Last Review Status/Date: September 2015 Page: 1 of 10 Management of Patients with Cancer Description The prognosis of cancer patients is often determined by the occurrence of metastatic disease. Studies
Page: 1 of 10. Detection of Circulating Tumor Cells in the Management of Patients with Cancer
 Last Review Status/Date: September 2014 Page: 1 of 10 Management of Patients with Cancer Description The prognosis of cancer patients is often determined by the occurrence of metastatic disease. Studies
Last Review Status/Date: September 2014 Page: 1 of 10 Management of Patients with Cancer Description The prognosis of cancer patients is often determined by the occurrence of metastatic disease. Studies
Regence. Medical Policy Manual. Topic: Detection of Circulating Tumor Cells in the Management of Patients with Cancer. Date of Origin: July 5, 2005
 Regence Medical Policy Manual Topic: Detection of Circulating Tumor Cells in the Management of Patients with Cancer Date of Origin: July 5, 2005 Section: Laboratory Last Reviewed Date: June 2013 Policy
Regence Medical Policy Manual Topic: Detection of Circulating Tumor Cells in the Management of Patients with Cancer Date of Origin: July 5, 2005 Section: Laboratory Last Reviewed Date: June 2013 Policy
Links in PDF documents are not guaranteed to work. To follow a web link, please use the MCD Website. Jurisdiction Oregon. Retirement Date N/A
 Local Coverage Determination (LCD): Circulating Tumor Cell Marker Assays (L35096) Links in PDF documents are not guaranteed to work. To follow a web link, please use the MCD Website. Contractor Information
Local Coverage Determination (LCD): Circulating Tumor Cell Marker Assays (L35096) Links in PDF documents are not guaranteed to work. To follow a web link, please use the MCD Website. Contractor Information
Sequential immunochemotherapy and edrecolomab in the adjuvant therapy of breast cancer: reduction of 17-1A-positive disseminated tumour cells
 Original article Annals of Oncology 13: 1044 1048, 2002 DOI: 10.1093/annonc/mdf184 Sequential immunochemotherapy and edrecolomab in the adjuvant therapy of breast cancer: reduction of 17-1A-positive disseminated
Original article Annals of Oncology 13: 1044 1048, 2002 DOI: 10.1093/annonc/mdf184 Sequential immunochemotherapy and edrecolomab in the adjuvant therapy of breast cancer: reduction of 17-1A-positive disseminated
Medical Policy Manual. Topic: Detection of Circulating Tumor Cells in the Management of Patients with Cancer. Date of Origin: July 5, 2005
 Medical Policy Manual Topic: Detection of Circulating Tumor Cells in the Management of Patients with Cancer Date of Origin: July 5, 2005 Section: Laboratory Last Reviewed Date: May 2014 Policy No: 46 Effective
Medical Policy Manual Topic: Detection of Circulating Tumor Cells in the Management of Patients with Cancer Date of Origin: July 5, 2005 Section: Laboratory Last Reviewed Date: May 2014 Policy No: 46 Effective
New Approach for Interpreting Changes in Circulating Tumour Cells (CTC) for Evaluation of Treatment Effect in Metastatic Breast Cancer
 Translational Oncology Volume 7 Number December 204 pp. 94 70 94 www.transonc.com New Approach for Interpreting Changes in Circulating Tumour Cells (CTC) for Evaluation of Treatment Effect in Metastatic
Translational Oncology Volume 7 Number December 204 pp. 94 70 94 www.transonc.com New Approach for Interpreting Changes in Circulating Tumour Cells (CTC) for Evaluation of Treatment Effect in Metastatic
Downloaded from:
 Ligthart, ST; Coumans, FA; Attard, G; Cassidy, AM; de Bono, JS; Terstappen, LW (2011) Unbiased and automated identification of a circulating tumour cell definition that associates with overall survival.
Ligthart, ST; Coumans, FA; Attard, G; Cassidy, AM; de Bono, JS; Terstappen, LW (2011) Unbiased and automated identification of a circulating tumour cell definition that associates with overall survival.
Circulating tumor cells as biomarker for hormonal treatment in breast and prostate cancer. Michal Mego
 National Cancer Institute, Slovakia Translational Research Unit Circulating tumor cells as biomarker for hormonal treatment in breast and prostate cancer Michal Mego 2 nd Department of Oncology, Faculty
National Cancer Institute, Slovakia Translational Research Unit Circulating tumor cells as biomarker for hormonal treatment in breast and prostate cancer Michal Mego 2 nd Department of Oncology, Faculty
Circulating Tumor Cell Kit (Epithelial)
 7900001 16 Test Kit Circulating Tumor Cell Kit (Epithelial) I INTENDED USE For in vitro diagnostic use. The CELLSEARCH Circulating Tumor Cell Kit is intended for the enumeration of circulating tumor cells
7900001 16 Test Kit Circulating Tumor Cell Kit (Epithelial) I INTENDED USE For in vitro diagnostic use. The CELLSEARCH Circulating Tumor Cell Kit is intended for the enumeration of circulating tumor cells
RESEARCH ARTICLE. Detection of Circulating Tumor Cells in Breast Cancer Patients: Prognostic Predictive Role
 DOI:http://dx.doi.org/10.7314/APJCP.2013.14.3.1601 RESEARCH ARTICLE Detection of Circulating Tumor Cells in Breast Cancer Patients: Prognostic Predictive Role Ibrahim Turker¹*, Ummugul Uyeturk¹, Ozlem
DOI:http://dx.doi.org/10.7314/APJCP.2013.14.3.1601 RESEARCH ARTICLE Detection of Circulating Tumor Cells in Breast Cancer Patients: Prognostic Predictive Role Ibrahim Turker¹*, Ummugul Uyeturk¹, Ozlem
Lung cancer is the leading cancer of all cancer-related death
 ORIGINAL ARTICLE Preliminary Investigation of the Clinical Significance of Detecting Circulating Tumor Cells Enriched from Lung Cancer Patients Chi Wu, MD,* Huaijie Hao, MS, Longyun Li, MD,* Xiaoyun Zhou,
ORIGINAL ARTICLE Preliminary Investigation of the Clinical Significance of Detecting Circulating Tumor Cells Enriched from Lung Cancer Patients Chi Wu, MD,* Huaijie Hao, MS, Longyun Li, MD,* Xiaoyun Zhou,
A Novel CTC-Detecting Technique Using TelomeScan and Its Clinical Applications
 A Novel CTC-Detecting Technique Using TelomeScan and Its Clinical Applications Yasuo Urata CEO and President Oncolys BioPharma Inc. February 16, 2013 Telomere Length is a Limiting Factor for Cell Replication
A Novel CTC-Detecting Technique Using TelomeScan and Its Clinical Applications Yasuo Urata CEO and President Oncolys BioPharma Inc. February 16, 2013 Telomere Length is a Limiting Factor for Cell Replication
Circulating Tumor Cells, Disease Progression, and Survival in Metastatic Breast Cancer
 original article Circulating Tumor Cells, Disease Progression, and Survival in Metastatic Breast Cancer Massimo Cristofanilli, M.D., G. Thomas Budd, M.D., Matthew J. Ellis, M.B., Ph.D., Alison Stopeck,
original article Circulating Tumor Cells, Disease Progression, and Survival in Metastatic Breast Cancer Massimo Cristofanilli, M.D., G. Thomas Budd, M.D., Matthew J. Ellis, M.B., Ph.D., Alison Stopeck,
Liquid Biopsy: Implications for Cancer Staging & Therapy
 Prof. Klaus Pantel, MD, PhD Institut für Tumorbiologie Liquid Biopsy: Implications for Cancer Staging & Therapy Tumor cell dissemination and cancer dormancy Primary tumor Local relapse Cancer cells disseminate
Prof. Klaus Pantel, MD, PhD Institut für Tumorbiologie Liquid Biopsy: Implications for Cancer Staging & Therapy Tumor cell dissemination and cancer dormancy Primary tumor Local relapse Cancer cells disseminate
The CellCollector TM technology
 The CellCollector TM technology In vivo isolation of circulating tumor cells by the CellCollector TM system Dr. Klaus Lücke (CEO) Unmet medical need in oncology: Access to tumor cells not only at the time
The CellCollector TM technology In vivo isolation of circulating tumor cells by the CellCollector TM system Dr. Klaus Lücke (CEO) Unmet medical need in oncology: Access to tumor cells not only at the time
WHAT SHOULD WE DO WITH TUMOUR BUDDING IN EARLY COLORECTAL CANCER?
 CANCER STAGING TNM and prognosis in CRC WHAT SHOULD WE DO WITH TUMOUR BUDDING IN EARLY COLORECTAL CANCER? Alessandro Lugli, MD Institute of Pathology University of Bern Switzerland Maastricht, June 19
CANCER STAGING TNM and prognosis in CRC WHAT SHOULD WE DO WITH TUMOUR BUDDING IN EARLY COLORECTAL CANCER? Alessandro Lugli, MD Institute of Pathology University of Bern Switzerland Maastricht, June 19
Quantification of Breast Cancer Cells in Peripheral Blood Samples by Real-Time RT-PCR
 Quantification of Breast Cancer Cells in Peripheral Blood Samples by Real-Time RT-PCR MICHAEL ZEBISCH 1, ALEXANDRA C. KÖLBL 1, CHRISTIAN SCHINDLBECK 2, JULIA NEUGEBAUER 1, SABINE HEUBLEIN 1, MATTHIAS ILMER
Quantification of Breast Cancer Cells in Peripheral Blood Samples by Real-Time RT-PCR MICHAEL ZEBISCH 1, ALEXANDRA C. KÖLBL 1, CHRISTIAN SCHINDLBECK 2, JULIA NEUGEBAUER 1, SABINE HEUBLEIN 1, MATTHIAS ILMER
IMMUNOMAGNETIC QUANTIFICATION OF CIRCULATING TUMORAL CELLS IN PATIENTS WITH PROSTATE CÁNCER: CLINICAL AND PATHOLOGICAL CORRELATION
 Oncologic Urology Arch. Esp. Urol. 21; 63 (1): 23-31 IMMUNOMAGNETIC QUANTIFICATION OF CIRCULATING TUMORAL CELLS IN PATIENTS WITH PROSTATE CÁNCER: CLINICAL AND PATHOLOGICAL CORRELATION L. Resel Folkersman,
Oncologic Urology Arch. Esp. Urol. 21; 63 (1): 23-31 IMMUNOMAGNETIC QUANTIFICATION OF CIRCULATING TUMORAL CELLS IN PATIENTS WITH PROSTATE CÁNCER: CLINICAL AND PATHOLOGICAL CORRELATION L. Resel Folkersman,
In vitro detection of circulating tumor cells compared by the CytoTrack and CellSearch methods
 Tumor Biol. (2015) 36:4597 4601 DOI 10.1007/s13277-015-3105-z RESEARCH ARTICLE In vitro detection of circulating tumor cells compared by the CytoTrack and methods Thore Hillig & Peer Horn & Ann-Britt Nygaard
Tumor Biol. (2015) 36:4597 4601 DOI 10.1007/s13277-015-3105-z RESEARCH ARTICLE In vitro detection of circulating tumor cells compared by the CytoTrack and methods Thore Hillig & Peer Horn & Ann-Britt Nygaard
WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE CANCER PATIENTS
 WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE CANCER PATIENTS There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE CANCER PATIENTS There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
Title: Circulating Tumor Cells: A Tool Whose Time Has Come of Age
 Author's response to reviews Title: Circulating Tumor Cells: A Tool Whose Time Has Come of Age Authors: Ramona F Swaby (ramona.swaby@fccc.edu) Massimo Cristofanilli (Massimo.Cristofanilli@fccc.edu) Version:
Author's response to reviews Title: Circulating Tumor Cells: A Tool Whose Time Has Come of Age Authors: Ramona F Swaby (ramona.swaby@fccc.edu) Massimo Cristofanilli (Massimo.Cristofanilli@fccc.edu) Version:
Circulating tumor cells before and during follow-up after breast cancer surgery
 INTERNATIONAL JOURNAL OF ONCOLOGY 46: 407-413, 2015 Circulating tumor cells before and during follow-up after breast cancer surgery Guus van Dalum 1, Gert Jan van der Stam 1, Arjan G.J. Tibbe 1, Bas Franken
INTERNATIONAL JOURNAL OF ONCOLOGY 46: 407-413, 2015 Circulating tumor cells before and during follow-up after breast cancer surgery Guus van Dalum 1, Gert Jan van der Stam 1, Arjan G.J. Tibbe 1, Bas Franken
Monitoring apoptosis and Bcl-2 on circulating tumor cells in patients with metastatic breast cancer 5
 MOLECULAR ONCOLOGY 7 (2013) 680e692 available at www.sciencedirect.com www.elsevier.com/locate/molonc Monitoring apoptosis and Bcl-2 on circulating tumor cells in patients with metastatic breast cancer
MOLECULAR ONCOLOGY 7 (2013) 680e692 available at www.sciencedirect.com www.elsevier.com/locate/molonc Monitoring apoptosis and Bcl-2 on circulating tumor cells in patients with metastatic breast cancer
CTC in clinical studies: Latest reports on GI cancers
 CTC in clinical studies: Latest reports on GI cancers François-Clément Bidard, MD PhD GI cancers are characterized by Multimodal treatment strategies Treatments are adapted to tumor burden & prognosis
CTC in clinical studies: Latest reports on GI cancers François-Clément Bidard, MD PhD GI cancers are characterized by Multimodal treatment strategies Treatments are adapted to tumor burden & prognosis
Detection of Circulating Urothelial Cancer Cells in the Blood Using the CellSearch System
 1439 Detection of Circulating Urothelial Cancer Cells in the Blood Using the CellSearch System Michio Naoe, MD, PhD 1 Yoshio Ogawa, MD, PhD 1 Jun Morita, MD 1 Kei Omori, MD 1 Kumiko Takeshita, PhD 1 Takeshi
1439 Detection of Circulating Urothelial Cancer Cells in the Blood Using the CellSearch System Michio Naoe, MD, PhD 1 Yoshio Ogawa, MD, PhD 1 Jun Morita, MD 1 Kei Omori, MD 1 Kumiko Takeshita, PhD 1 Takeshi
Prognostic Role and HER2 Expression of Circulating Tumor Cells in Peripheral Blood of Patients Prior to Radical Cystectomy: A Prospective Study
 EUROPEAN UROLOGY 61 (2012) 810 817 available at www.sciencedirect.com journal homepage: www.europeanurology.com Bladder Cancer Prognostic Role and HER2 Expression of Circulating Tumor Cells in Peripheral
EUROPEAN UROLOGY 61 (2012) 810 817 available at www.sciencedirect.com journal homepage: www.europeanurology.com Bladder Cancer Prognostic Role and HER2 Expression of Circulating Tumor Cells in Peripheral
Dynamics of circulating tumor cells in early breast cancer under neoadjuvant therapy
 EXPERIMENTAL AND THERAPEUTIC MEDICINE 4: 43-48, 2012 Dynamics of circulating tumor cells in early breast cancer under neoadjuvant therapy MARÍA JOSÉ SERRANO 1, PEDRO SÁNCHEZ ROVIRA 2, I. MARTÍNEZ-ZUBIAURRE
EXPERIMENTAL AND THERAPEUTIC MEDICINE 4: 43-48, 2012 Dynamics of circulating tumor cells in early breast cancer under neoadjuvant therapy MARÍA JOSÉ SERRANO 1, PEDRO SÁNCHEZ ROVIRA 2, I. MARTÍNEZ-ZUBIAURRE
Pooled Analysis of the Prognostic Relevance of Circulating Tumor Cells in Primary Breast Cancer
 Biology of Human Tumors Pooled Analysis of the Prognostic Relevance of Circulating Tumor Cells in Primary Breast Cancer Wolfgang J. Janni 1, Brigitte Rack 2, Leon W.M.M. Terstappen 3, Jean-Yves Pierga
Biology of Human Tumors Pooled Analysis of the Prognostic Relevance of Circulating Tumor Cells in Primary Breast Cancer Wolfgang J. Janni 1, Brigitte Rack 2, Leon W.M.M. Terstappen 3, Jean-Yves Pierga
BY Mrs. K.SHAILAJA., M. PHARM., LECTURER DEPT OF PHARMACY PRACTICE, SRM COLLEGE OF PHARMACY
 BY Mrs. K.SHAILAJA., M. PHARM., LECTURER DEPT OF PHARMACY PRACTICE, SRM COLLEGE OF PHARMACY Cancer is a group of more than 100 different diseases that are characterized by uncontrolled cellular growth,
BY Mrs. K.SHAILAJA., M. PHARM., LECTURER DEPT OF PHARMACY PRACTICE, SRM COLLEGE OF PHARMACY Cancer is a group of more than 100 different diseases that are characterized by uncontrolled cellular growth,
Circulating Human Prostate Cancer Cells from an Orthotopic Mouse Model Rapidly Captured by Immunomagnetic Beads and Imaged by GFP Expression
 Circulating Human Prostate Cancer Cells from an Orthotopic Mouse Model Rapidly Captured by Immunomagnetic Beads and Imaged by GFP Expression KATARINA KOLOSTOVA 1, DANIELA PINTEROVA 1, ROBERT M. HOFFMAN
Circulating Human Prostate Cancer Cells from an Orthotopic Mouse Model Rapidly Captured by Immunomagnetic Beads and Imaged by GFP Expression KATARINA KOLOSTOVA 1, DANIELA PINTEROVA 1, ROBERT M. HOFFMAN
Summary... 2 GENITOURINARY TUMOURS - PROSTATE... 3
 ESMO 2016 Congress 7-11 October, 2016 Copenhagen, Denmark Table of Contents Summary... 2 GENITOURINARY TUMOURS - PROSTATE... 3 Custirsen provides no additional survival benefit to cabazitaxel/prednisone
ESMO 2016 Congress 7-11 October, 2016 Copenhagen, Denmark Table of Contents Summary... 2 GENITOURINARY TUMOURS - PROSTATE... 3 Custirsen provides no additional survival benefit to cabazitaxel/prednisone
RD-100i OSNA the new generation of sentinel lymph node analysis in breast cancer
 RD-1i OSNA the new generation of sentinel lymph node analysis in breast cancer RD-1i OSNA the new generation of sentinel lymph node analysis in breast cancer Sentinel node biopsy has rapidly emerged as
RD-1i OSNA the new generation of sentinel lymph node analysis in breast cancer RD-1i OSNA the new generation of sentinel lymph node analysis in breast cancer Sentinel node biopsy has rapidly emerged as
Liquid Biopsy. Jesus Garcia-Foncillas MD PhD. Director
 Liquid Biopsy Jesus Garcia-Foncillas MD PhD Director Main issues about liquid biopsies New paradigm: Precision Medicine Heterogeneity & Dynamics Surrogate mirror for the tumor CTCs in colon cancer ctdna:
Liquid Biopsy Jesus Garcia-Foncillas MD PhD Director Main issues about liquid biopsies New paradigm: Precision Medicine Heterogeneity & Dynamics Surrogate mirror for the tumor CTCs in colon cancer ctdna:
Claudin-4 Expression in Triple Negative Breast Cancer: Correlation with Androgen Receptors and Ki-67 Expression
 Claudin-4 Expression in Triple Negative Breast Cancer: Correlation with Androgen Receptors and Ki-67 Expression Mona A. Abd-Elazeem, Marwa A. Abd- Elazeem Pathology department, Faculty of Medicine, Tanta
Claudin-4 Expression in Triple Negative Breast Cancer: Correlation with Androgen Receptors and Ki-67 Expression Mona A. Abd-Elazeem, Marwa A. Abd- Elazeem Pathology department, Faculty of Medicine, Tanta
19(9), , 2017 doi: /neuonc/nox066 Advance Access date 21 June 2017
 1248 Neuro-Oncology 19(9), 1248 1254, 2017 doi:10.1093/neuonc/nox066 Advance Access date 21 June 2017 Cerebrospinal fluid circulating tumor cells: a novel tool to diagnose leptomeningeal metastases from
1248 Neuro-Oncology 19(9), 1248 1254, 2017 doi:10.1093/neuonc/nox066 Advance Access date 21 June 2017 Cerebrospinal fluid circulating tumor cells: a novel tool to diagnose leptomeningeal metastases from
The concept of circulating tumor cells (CTCs) was not a
 Original Article Folate Receptor Positive Circulating Tumor Cell Detected by LT-PCR Based Method as a Diagnostic Biomarker for Non Small-Cell Lung Cancer Xiaoxia Chen, MD,* Fei Zhou,* Xuefei Li, PhD, Guohua
Original Article Folate Receptor Positive Circulating Tumor Cell Detected by LT-PCR Based Method as a Diagnostic Biomarker for Non Small-Cell Lung Cancer Xiaoxia Chen, MD,* Fei Zhou,* Xuefei Li, PhD, Guohua
1. Introduction. Correspondence should be addressed to François-Clément Bidard; Received 28 March 2013; Accepted 19 April 2013
 International Breast Cancer Volume 203, Article ID 30470, 5 pages http://dx.doi.org/0.55/203/30470 Clinical Study Time-Dependent Prognostic Impact of Circulating Tumor Cells Detection in Non-Metastatic
International Breast Cancer Volume 203, Article ID 30470, 5 pages http://dx.doi.org/0.55/203/30470 Clinical Study Time-Dependent Prognostic Impact of Circulating Tumor Cells Detection in Non-Metastatic
Detection of the Circulating Tumor Cells in Cancer Patients
 Detection of the Circulating Tumor Cells in Cancer Patients Athanasios Armakolas; Zacharoula Panteleakou; Adrianos Nezos; Aikaterini Tsouma; Maria Skondra; Peter Lembessis; Nikolaos Pissimissis; Michael
Detection of the Circulating Tumor Cells in Cancer Patients Athanasios Armakolas; Zacharoula Panteleakou; Adrianos Nezos; Aikaterini Tsouma; Maria Skondra; Peter Lembessis; Nikolaos Pissimissis; Michael
RD-100i OSNA the new generation of sentinel lymph node analysis in breast cancer
 RD-1i OSNA the new generation of sentinel lymph node analysis in breast cancer www.sysmex-europe.com RD-1i OSNA the new generation of sentinel lymph node analysis in breast cancer Sentinel node biopsy
RD-1i OSNA the new generation of sentinel lymph node analysis in breast cancer www.sysmex-europe.com RD-1i OSNA the new generation of sentinel lymph node analysis in breast cancer Sentinel node biopsy
Implications of Progesterone Receptor Status for the Biology and Prognosis of Breast Cancers
 日大医誌 75 (1): 10 15 (2016) 10 Original Article Implications of Progesterone Receptor Status for the Biology and Prognosis of Breast Cancers Naotaka Uchida 1), Yasuki Matsui 1), Takeshi Notsu 1) and Manabu
日大医誌 75 (1): 10 15 (2016) 10 Original Article Implications of Progesterone Receptor Status for the Biology and Prognosis of Breast Cancers Naotaka Uchida 1), Yasuki Matsui 1), Takeshi Notsu 1) and Manabu
maintrac What's the future in precision diagnostics? From screening to stem cells and back!
 maintrac What's the future in precision diagnostics? From screening to stem cells and back! Cancer is a frightening diagnosis: why? Malignant tumours are detectable when they have reached a size of about
maintrac What's the future in precision diagnostics? From screening to stem cells and back! Cancer is a frightening diagnosis: why? Malignant tumours are detectable when they have reached a size of about
RESEARCH ARTICLE. Nigel P Murray 1 *, Vidal Albarran 2, Guillermo Perez 2, Ricardo Villalon 3, Amparo Ruiz 3. Abstract.
 RESEARCH ARTICLE Secondary Circulating Tumor Cells (CTCs) but not Primary CTCs are Associated with the Clinico-Pathological Parameters in Chilean Patients With Colo-Rectal Cancer Nigel P Murray 1 *, Vidal
RESEARCH ARTICLE Secondary Circulating Tumor Cells (CTCs) but not Primary CTCs are Associated with the Clinico-Pathological Parameters in Chilean Patients With Colo-Rectal Cancer Nigel P Murray 1 *, Vidal
DetectionofCirculatingTumorCellsinPeripheralBloodofPatients with Metastatic Breast Cancer: A Validation Study of the CellSearch System
 Imaging, Diagnosis, Prognosis DetectionofCirculatingTumorCellsinPeripheralBloodofPatients with Metastatic Breast Cancer: A Validation Study of the CellSearch System Sabine Riethdorf, 1 Herbert Fritsche,
Imaging, Diagnosis, Prognosis DetectionofCirculatingTumorCellsinPeripheralBloodofPatients with Metastatic Breast Cancer: A Validation Study of the CellSearch System Sabine Riethdorf, 1 Herbert Fritsche,
Early dissemination in prostate cancer
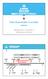 Early dissemination in prostate cancer Miodrag Guzvic, University of Regensburg, Germany Adjuvant Palliative M0 Initiation Diagnosis Surgery Metastasis Death Intervention window to delay or prevent metastasis
Early dissemination in prostate cancer Miodrag Guzvic, University of Regensburg, Germany Adjuvant Palliative M0 Initiation Diagnosis Surgery Metastasis Death Intervention window to delay or prevent metastasis
IJC International Journal of Cancer
 IJC International Journal of Cancer Comparison of assay methods for detection of circulating tumor cells in metastatic breast cancer: AdnaGen AdnaTest BreastCancer Select/Detect TM versus Veridex CellSearch
IJC International Journal of Cancer Comparison of assay methods for detection of circulating tumor cells in metastatic breast cancer: AdnaGen AdnaTest BreastCancer Select/Detect TM versus Veridex CellSearch
A comparison of CellCollector with CellSearch in patients with neuroendocrine
 Page of 5 Accepted Preprint first posted on August 6 as Manuscript ERC-6- A comparison of CellCollector with CellSearch in patients with neuroendocrine tumours 3 4 5 D Mandair, C Vesely,L Ensell, H Lowe,
Page of 5 Accepted Preprint first posted on August 6 as Manuscript ERC-6- A comparison of CellCollector with CellSearch in patients with neuroendocrine tumours 3 4 5 D Mandair, C Vesely,L Ensell, H Lowe,
Taiwan. Taiwan. 807, Taiwan. Taiwan
 Annals of Surgical Oncology 15(8):2120 2128 DOI: 10.1245/s10434-008-9961-7 Persistent Presence of Postoperative Circulating Tumor Cells is a Poor Prognostic Factor for Patients with Stage I III Colorectal
Annals of Surgical Oncology 15(8):2120 2128 DOI: 10.1245/s10434-008-9961-7 Persistent Presence of Postoperative Circulating Tumor Cells is a Poor Prognostic Factor for Patients with Stage I III Colorectal
WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE 1 CANCER PATIENTS
 WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE 1 CANCER PATIENTS There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE 1 CANCER PATIENTS There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
LIQUID BIOPSY
 www.idisantiago.es LIQUID BIOPSY Miguel Abal Investigador I3SNS Oncoloxía Médica Traslacional Instituto de Investigación Sanitaria de Santiago (IDIS) Complexo Hospitalario Universitario de Santiago/SERGAS
www.idisantiago.es LIQUID BIOPSY Miguel Abal Investigador I3SNS Oncoloxía Médica Traslacional Instituto de Investigación Sanitaria de Santiago (IDIS) Complexo Hospitalario Universitario de Santiago/SERGAS
Evaluation and significance of circulating epithelial cells in patients with hormone-refractory prostate cancer
 Urological Oncology CIRCULATING EPITHELIAL CELLS IN PATIENTS WITH HRPC GARCIA et al. The urological oncology section is relatively long this month, and this reflects the many high-quality manuscripts we
Urological Oncology CIRCULATING EPITHELIAL CELLS IN PATIENTS WITH HRPC GARCIA et al. The urological oncology section is relatively long this month, and this reflects the many high-quality manuscripts we
Circulating tumor cells
 Circulating tumor cells counts and characteristics Bianca Mostert Circulating tumor cells counts and characteristics Bianca Mostert The studies described in this thesis were performed at the Department
Circulating tumor cells counts and characteristics Bianca Mostert Circulating tumor cells counts and characteristics Bianca Mostert The studies described in this thesis were performed at the Department
Oncology 101. Cancer Basics
 Oncology 101 Cancer Basics What Will You Learn? What is Cancer and How Does It Develop? Cancer Diagnosis and Staging Cancer Treatment What is Cancer? Cancer is a group of more than 100 different diseases
Oncology 101 Cancer Basics What Will You Learn? What is Cancer and How Does It Develop? Cancer Diagnosis and Staging Cancer Treatment What is Cancer? Cancer is a group of more than 100 different diseases
ORIGINAL ARTICLE. Clinical Node-Negative Thick Melanoma
 ORIGINAL ARTICLE Clinical Node-Negative Thick Melanoma George I. Salti, MD; Ashwin Kansagra, MD; Michael A. Warso, MD; Salve G. Ronan, MD ; Tapas K. Das Gupta, MD, PhD, DSc Background: Patients with T4
ORIGINAL ARTICLE Clinical Node-Negative Thick Melanoma George I. Salti, MD; Ashwin Kansagra, MD; Michael A. Warso, MD; Salve G. Ronan, MD ; Tapas K. Das Gupta, MD, PhD, DSc Background: Patients with T4
Circulating and Disseminated Tumor Cells in the Management of Breast Cancer
 Anatomic Pathology / CTCs and DTCs in Breast Cancer Management Circulating and Disseminated Tumor Cells in the Management of Breast Cancer Jeffrey S. Ross, MD, and Elzbieta A. Slodkowska, MD Key Words:
Anatomic Pathology / CTCs and DTCs in Breast Cancer Management Circulating and Disseminated Tumor Cells in the Management of Breast Cancer Jeffrey S. Ross, MD, and Elzbieta A. Slodkowska, MD Key Words:
Decreased incidence of isolated tumor cells in lymph nodes after laparoscopic resection for colorectal cancer
 Surg Endosc (2011) 25:3652 3657 DOI 10.1007/s00464-011-1773-3 Decreased incidence of isolated tumor cells in lymph nodes after laparoscopic resection for colorectal cancer E. S. van der Zaag C. J. Buskens
Surg Endosc (2011) 25:3652 3657 DOI 10.1007/s00464-011-1773-3 Decreased incidence of isolated tumor cells in lymph nodes after laparoscopic resection for colorectal cancer E. S. van der Zaag C. J. Buskens
Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY
 Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY Methodology The test is based on the reported 50-gene classifier algorithm originally named PAM50 and is performed on the ncounter Dx Analysis System
Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY Methodology The test is based on the reported 50-gene classifier algorithm originally named PAM50 and is performed on the ncounter Dx Analysis System
Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY
 Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY GENE EXPRESSION PROFILING WITH PROSIGNA What is Prosigna? Prosigna Breast Cancer Prognostic Gene Signature Assay is an FDA-approved assay which provides
Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY GENE EXPRESSION PROFILING WITH PROSIGNA What is Prosigna? Prosigna Breast Cancer Prognostic Gene Signature Assay is an FDA-approved assay which provides
Blood-based biomarkers in lung cancer: prognosis and treatment decisions
 Mini-Review Blood-based biomarkers in lung cancer: prognosis and treatment decisions Meng Xu-Welliver 1, David P. Carbone 2 1 Department of Radiation Oncology, 2 Division of Medical Oncology, Department
Mini-Review Blood-based biomarkers in lung cancer: prognosis and treatment decisions Meng Xu-Welliver 1, David P. Carbone 2 1 Department of Radiation Oncology, 2 Division of Medical Oncology, Department
What to do after pcr in different subtypes?
 What to do after pcr in different subtypes? Luca Moscetti Breast Unit Università degli Studi di Modena e Reggio Emilia Policlinico di Modena, Italy Aims of neoadjuvant therapy in breast cancer Primary
What to do after pcr in different subtypes? Luca Moscetti Breast Unit Università degli Studi di Modena e Reggio Emilia Policlinico di Modena, Italy Aims of neoadjuvant therapy in breast cancer Primary
High Sensitivity Immunomagnetic CTC Isolation as Compared to Alternative Isolation Methods
 High Sensitivity Immunomagnetic CTC Isolation as Compared to Alternative Isolation Methods 1. Introduction: An overview of CTC isolation methods 2. Challenges for direct comparisons of CTC recovery 3.
High Sensitivity Immunomagnetic CTC Isolation as Compared to Alternative Isolation Methods 1. Introduction: An overview of CTC isolation methods 2. Challenges for direct comparisons of CTC recovery 3.
Keywords: circulating tumour cells; breast cancer; CellSearch System; RT PCR; AdnaTest
 British Journal of Cancer (2010) 102, 276 284 All rights reserved 0007 0920/10 $32.00 www.bjcancer.com Circulating tumour cell detection: a direct comparison between the CellSearch System, the AdnaTest
British Journal of Cancer (2010) 102, 276 284 All rights reserved 0007 0920/10 $32.00 www.bjcancer.com Circulating tumour cell detection: a direct comparison between the CellSearch System, the AdnaTest
Talk to Your Doctor. Fact Sheet
 Talk to Your Doctor Hearing the words you have skin cancer is overwhelming and would leave anyone with a lot of questions. If you have been diagnosed with Stage I or II cutaneous melanoma with no apparent
Talk to Your Doctor Hearing the words you have skin cancer is overwhelming and would leave anyone with a lot of questions. If you have been diagnosed with Stage I or II cutaneous melanoma with no apparent
Preoperative CA 15-3 and CEA serum levels as predictor for breast cancer outcomes
 Annals of Oncology 19: 675 681, 2008 doi:10.1093/annonc/mdm538 Published online 23 November 2007 Preoperative CA 15-3 and CEA serum levels as predictor for breast cancer outcomes B.-W. Park 1,4 *, J.-W.
Annals of Oncology 19: 675 681, 2008 doi:10.1093/annonc/mdm538 Published online 23 November 2007 Preoperative CA 15-3 and CEA serum levels as predictor for breast cancer outcomes B.-W. Park 1,4 *, J.-W.
VALUE AND ROLE OF PSA AS A TUMOUR MARKER OF RESPONSE/RELAPSE
 Session 3 Advanced prostate cancer VALUE AND ROLE OF PSA AS A TUMOUR MARKER OF RESPONSE/RELAPSE 1 PSA is a serine protease and the physiological role is believed to be liquefying the seminal fluid PSA
Session 3 Advanced prostate cancer VALUE AND ROLE OF PSA AS A TUMOUR MARKER OF RESPONSE/RELAPSE 1 PSA is a serine protease and the physiological role is believed to be liquefying the seminal fluid PSA
a new approach to personalized medicine in breast cancer patients American Association for Clinical Chemistry
 Clinical Chemistry 61:1 259 266 (2015) Cancer Diagnostics Circulating Tumor Cell Enumeration with a Combination of Epithelial Cell Adhesion Molecule and Cell-Surface Vimentin Based Methods for Monitoring
Clinical Chemistry 61:1 259 266 (2015) Cancer Diagnostics Circulating Tumor Cell Enumeration with a Combination of Epithelial Cell Adhesion Molecule and Cell-Surface Vimentin Based Methods for Monitoring
Prospective Clinical Study of Circulating Tumor Cells For Colorectal Cancer Screening
 Prospective Clinical Study of Circulating Tumor Cells For Colorectal Cancer Screening Results From a Multi-Year 620-Sample Study CRC: SLOW GROWING, PREVENTABLE POLYP TO CANCER CAN TAKE 5 TO 15 YEARS CANCER
Prospective Clinical Study of Circulating Tumor Cells For Colorectal Cancer Screening Results From a Multi-Year 620-Sample Study CRC: SLOW GROWING, PREVENTABLE POLYP TO CANCER CAN TAKE 5 TO 15 YEARS CANCER
WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE1 CANCER PATIENTS
 WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE1 CANCER PATIENTS There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
WHEN STARTING A NEW LINE OF THERAPY FOR YOUR METASTATIC BREAST, COLORECTAL, AND PROSTATE1 CANCER PATIENTS There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
The Presence and Persistence of Resistant and Stem Cell- Like Tumor Cells as a Major Problem in Ovarian Cancer
 Welcome! The Presence and Persistence of Resistant and Stem Cell- Like Tumor Cells as a Major Problem in Ovarian Cancer Prof. Sabine Kasimir-Bauer Department of Gynecology and Obstetrics University Hospital
Welcome! The Presence and Persistence of Resistant and Stem Cell- Like Tumor Cells as a Major Problem in Ovarian Cancer Prof. Sabine Kasimir-Bauer Department of Gynecology and Obstetrics University Hospital
The clinical relevance of circulating, cell-free and exosomal micrornas as biomarkers for gynecological tumors
 Department of Tumor Biology The clinical relevance of circulating, cell-free and exosomal micrornas as biomarkers for gynecological tumors cfdna Copenhagen April 6-7, 2017 Heidi Schwarzenbach, PhD Tumor
Department of Tumor Biology The clinical relevance of circulating, cell-free and exosomal micrornas as biomarkers for gynecological tumors cfdna Copenhagen April 6-7, 2017 Heidi Schwarzenbach, PhD Tumor
Circulating Stromal Cells in Immunotherapy Utilizing a Total Blood Based Biopsy
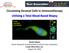 Circulating Stromal Cells in Immunotherapy Utilizing a Total Blood Based Biopsy Cancer associated macrophage CTCs Daniel Adams Senior Research Scientist/Head of Clinical Core Laboratory Creatv MicroTech,
Circulating Stromal Cells in Immunotherapy Utilizing a Total Blood Based Biopsy Cancer associated macrophage CTCs Daniel Adams Senior Research Scientist/Head of Clinical Core Laboratory Creatv MicroTech,
Article Comparison of Two EpCAM-Based Methods for CTC Detection and Molecular Characterization in Advanced Colorectal Cancer
 Article Comparison of Two EpCAM-Based Methods for CTC Detection and Molecular Characterization in Advanced Colorectal Cancer Santiago Cabezas-Camarero 1, Virginia De La Orden 2, Silvia Veganzones-De-Castro
Article Comparison of Two EpCAM-Based Methods for CTC Detection and Molecular Characterization in Advanced Colorectal Cancer Santiago Cabezas-Camarero 1, Virginia De La Orden 2, Silvia Veganzones-De-Castro
Circulating Tumor DNA and Circulating Tumor Cells for Management (Liquid Biopsy) of Solid Tumor Cancers
 NOTE: This policy is not effective until December 1, 2018. To view the current policy, click here. Medical Policy Manual Laboratory, Policy No. 46 Circulating Tumor DNA and Circulating Tumor Cells for
NOTE: This policy is not effective until December 1, 2018. To view the current policy, click here. Medical Policy Manual Laboratory, Policy No. 46 Circulating Tumor DNA and Circulating Tumor Cells for
Can we prevent metastasis?
 Can we prevent metastasis? A research example to translate from the bench to the bedside Diane Palmieri, Ph.D. Women s Cancers Section Laboratory of Molecular Pharmacology CCR, NCI Some Basic Truths Most
Can we prevent metastasis? A research example to translate from the bench to the bedside Diane Palmieri, Ph.D. Women s Cancers Section Laboratory of Molecular Pharmacology CCR, NCI Some Basic Truths Most
Staging for Residents, Nurses, and Multidisciplinary Health Care Team
 Staging for Residents, Nurses, and Multidisciplinary Health Care Team Donna M. Gress, RHIT, CTR Validating science. Improving patient care. Learning Objectives Introduce the concept and history of stage
Staging for Residents, Nurses, and Multidisciplinary Health Care Team Donna M. Gress, RHIT, CTR Validating science. Improving patient care. Learning Objectives Introduce the concept and history of stage
The Avatar System TM Yields Biologically Relevant Results
 Application Note The Avatar System TM Yields Biologically Relevant Results Liquid biopsies stand to revolutionize the cancer field, enabling early detection and noninvasive monitoring of tumors. In the
Application Note The Avatar System TM Yields Biologically Relevant Results Liquid biopsies stand to revolutionize the cancer field, enabling early detection and noninvasive monitoring of tumors. In the
Only Estrogen receptor positive is not enough to predict the prognosis of breast cancer
 Young Investigator Award, Global Breast Cancer Conference 2018 Only Estrogen receptor positive is not enough to predict the prognosis of breast cancer ㅑ Running head: Revisiting estrogen positive tumors
Young Investigator Award, Global Breast Cancer Conference 2018 Only Estrogen receptor positive is not enough to predict the prognosis of breast cancer ㅑ Running head: Revisiting estrogen positive tumors
Reflex Testing Guidelines for Immunotherapy in Non-Small Cell Lung Cancer
 Reflex Testing Guidelines for Immunotherapy in Non-Small Cell Lung Cancer Jimmy Ruiz, MD Assistant Professor Thoracic Oncology Program Wake Forest Comprehensive Cancer Center Disclosures I have no actual
Reflex Testing Guidelines for Immunotherapy in Non-Small Cell Lung Cancer Jimmy Ruiz, MD Assistant Professor Thoracic Oncology Program Wake Forest Comprehensive Cancer Center Disclosures I have no actual
Early colorectal cancer Quality and rules for a good pathology report Histoprognostic factors
 Early colorectal cancer Quality and rules for a good pathology report Histoprognostic factors Frédéric Bibeau, MD, PhD Pathology department Biopathology unit Institut du Cancer de Montpellier France Quality
Early colorectal cancer Quality and rules for a good pathology report Histoprognostic factors Frédéric Bibeau, MD, PhD Pathology department Biopathology unit Institut du Cancer de Montpellier France Quality
Nature Methods: doi: /nmeth Supplementary Figure 1
 Supplementary Figure 1 Finite-element analysis of cell cluster dynamics in different cluster trap architectures. (a) Cluster-Chip (b) Filter (c) A structure identical to the Cluster-Chip except that one
Supplementary Figure 1 Finite-element analysis of cell cluster dynamics in different cluster trap architectures. (a) Cluster-Chip (b) Filter (c) A structure identical to the Cluster-Chip except that one
All India Institute of Medical Sciences, New Delhi, INDIA. Department of Pediatric Surgery, Medical Oncology, and Radiology
 All India Institute of Medical Sciences, New Delhi, INDIA Department of Pediatric Surgery, Medical Oncology, and Radiology Clear cell sarcoma of the kidney- rare renal neoplasm second most common renal
All India Institute of Medical Sciences, New Delhi, INDIA Department of Pediatric Surgery, Medical Oncology, and Radiology Clear cell sarcoma of the kidney- rare renal neoplasm second most common renal
Cancers of unknown primary : Knowing the unknown. Prof. Ahmed Hossain Professor of Medicine SSMC
 Cancers of unknown primary : Knowing the unknown Prof. Ahmed Hossain Professor of Medicine SSMC Definition Cancers of unknown primary site (CUPs) Represent a heterogeneous group of metastatic tumours,
Cancers of unknown primary : Knowing the unknown Prof. Ahmed Hossain Professor of Medicine SSMC Definition Cancers of unknown primary site (CUPs) Represent a heterogeneous group of metastatic tumours,
When starting a new line of therapy for your metastatic breast, colorectal, and prostate 1 cancer patients
 When starting a new line of therapy for your metastatic breast, colorectal, and prostate 1 cancer patients There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
When starting a new line of therapy for your metastatic breast, colorectal, and prostate 1 cancer patients There is uncertainty whether their prognosis will improve It can take as long as 3 months to learn
Title: Synuclein Gamma Predicts Poor Clinical Outcome in Colon Cancer with Normal Levels of Carcinoembryonic Antigen
 Author's response to reviews Title: Synuclein Gamma Predicts Poor Clinical Outcome in Colon Cancer with Normal Levels of Carcinoembryonic Antigen Authors: Caiyun Liu (liucaiyun23@yahoo.com.cn) Bin Dong
Author's response to reviews Title: Synuclein Gamma Predicts Poor Clinical Outcome in Colon Cancer with Normal Levels of Carcinoembryonic Antigen Authors: Caiyun Liu (liucaiyun23@yahoo.com.cn) Bin Dong
Biomarker Utility of Circulating Tumor Cells in Metastatic Cutaneous Melanoma
 ORIGINAL ARTICLE See related commentary on pg 146 Biomarker Utility of Circulating Tumor Cells in Metastatic Cutaneous Melanoma Leila Khoja 1, Paul Lorigan 2,3,CongZhou 1, Matthew Lancashire 1, Jessica
ORIGINAL ARTICLE See related commentary on pg 146 Biomarker Utility of Circulating Tumor Cells in Metastatic Cutaneous Melanoma Leila Khoja 1, Paul Lorigan 2,3,CongZhou 1, Matthew Lancashire 1, Jessica
ABSTRACT INTRODUCTION
 /, 2017, Vol. 8, (No. 13), pp: 21639-21649 Clinical significance of detecting circulating tumor cells in colorectal cancer using subtraction enrichment and immunostainingfluorescence in situ hybridization
/, 2017, Vol. 8, (No. 13), pp: 21639-21649 Clinical significance of detecting circulating tumor cells in colorectal cancer using subtraction enrichment and immunostainingfluorescence in situ hybridization
METACELL. PERSONALIZED CANCER THERAPY USING CIRCULATING TUMOR CELLS (CTCs) METACELL LIQUID BIOPSY
 METACELL PERSONALIZED CANCER THERAPY USING CIRCULATING TUMOR CELLS (s) AN EASY WAY TO LIQUID BIOPSY MORE THAN A METASTATIC CELL IN BLOOD A STEP TOWARDS PERSONALIZED CANCER TREATMENT LIQUID BIOPSY REAL-TIME
METACELL PERSONALIZED CANCER THERAPY USING CIRCULATING TUMOR CELLS (s) AN EASY WAY TO LIQUID BIOPSY MORE THAN A METASTATIC CELL IN BLOOD A STEP TOWARDS PERSONALIZED CANCER TREATMENT LIQUID BIOPSY REAL-TIME
Review of NEO Testing Platforms. Lawrence M. Weiss, MD Medical Director, Aliso Viejo
 Review of NEO Testing Platforms Lawrence M. Weiss, MD Medical Director, Aliso Viejo Lawrence Weiss, M.D. Medical Director, Aliso Viejo Dr. Weiss currently serves as NeoGenomics Medical Director, Aliso
Review of NEO Testing Platforms Lawrence M. Weiss, MD Medical Director, Aliso Viejo Lawrence Weiss, M.D. Medical Director, Aliso Viejo Dr. Weiss currently serves as NeoGenomics Medical Director, Aliso
Position Statement on Management of the Axilla in Patients with Invasive Breast Cancer
 - Official Statement - Position Statement on Management of the Axilla in Patients with Invasive Breast Cancer Sentinel lymph node (SLN) biopsy has replaced axillary lymph node dissection (ALND) for the
- Official Statement - Position Statement on Management of the Axilla in Patients with Invasive Breast Cancer Sentinel lymph node (SLN) biopsy has replaced axillary lymph node dissection (ALND) for the
User s Manual Version 1.0
 User s Manual Version 1.0 #639 Longmian Avenue, Jiangning District, Nanjing,211198,P.R.China. http://tcoa.cpu.edu.cn/ Contact us at xiaosheng.wang@cpu.edu.cn for technical issue and questions Catalogue
User s Manual Version 1.0 #639 Longmian Avenue, Jiangning District, Nanjing,211198,P.R.China. http://tcoa.cpu.edu.cn/ Contact us at xiaosheng.wang@cpu.edu.cn for technical issue and questions Catalogue
Update on Colorectal Cancer: Integrating the Host Immune Response 3 rd Emirates Surgical Pathology Conference 15 December 2017 Dubai, UAE
 Update on Colorectal Cancer: Integrating the Host Immune Response 3 rd Emirates Surgical Pathology Conference 15 December 2017 Dubai, UAE Martine McManus, MD, FCAP Pathology & Laboratory Medicine Institute
Update on Colorectal Cancer: Integrating the Host Immune Response 3 rd Emirates Surgical Pathology Conference 15 December 2017 Dubai, UAE Martine McManus, MD, FCAP Pathology & Laboratory Medicine Institute
Use of Archived Tissues in the Development and Validation of Prognostic & Predictive Biomarkers
 Use of Archived Tissues in the Development and Validation of Prognostic & Predictive Biomarkers Richard Simon, D.Sc. Chief, Biometric Research Branch National Cancer Institute http://brb.nci.nih.gov Different
Use of Archived Tissues in the Development and Validation of Prognostic & Predictive Biomarkers Richard Simon, D.Sc. Chief, Biometric Research Branch National Cancer Institute http://brb.nci.nih.gov Different
She counts on your breast cancer expertise at the most vulnerable time of her life.
 HOME She counts on your breast cancer expertise at the most vulnerable time of her life. Empowering the right treatment choice for better patient outcomes. The comprehensive genomic assay experts trust.
HOME She counts on your breast cancer expertise at the most vulnerable time of her life. Empowering the right treatment choice for better patient outcomes. The comprehensive genomic assay experts trust.
Triple Negative Breast Cancer
 Triple Negative Breast Cancer Prof. Dr. Pornchai O-charoenrat Division of Head-Neck & Breast Surgery Department of Surgery Faculty of Medicine Siriraj Hospital Breast Cancer Classification Traditional
Triple Negative Breast Cancer Prof. Dr. Pornchai O-charoenrat Division of Head-Neck & Breast Surgery Department of Surgery Faculty of Medicine Siriraj Hospital Breast Cancer Classification Traditional
Serum Ca 15-3: A Useful Tumor Marker in the Prognostication of Locally Advanced Breast Cancer
 Original Article DOI: 10.21276/awch.1782 Serum Ca 15-3: A Useful Tumor Marker in the Prognostication of Locally Advanced Breast Cancer Mohd Rafey 1, Kafil Akhtar 1 *, Atia Z Rab 2 and Shahid A Siddiqui
Original Article DOI: 10.21276/awch.1782 Serum Ca 15-3: A Useful Tumor Marker in the Prognostication of Locally Advanced Breast Cancer Mohd Rafey 1, Kafil Akhtar 1 *, Atia Z Rab 2 and Shahid A Siddiqui
