A Guide for Choosing the Right CRO in Immuno- Oncology Studies. Mitigating Risk in the Clinic.
|
|
|
- Kristina Small
- 6 years ago
- Views:
Transcription
1 A Guide for Choosing the Right CRO in Immuno- Oncology Studies Mitigating Risk in the Clinic
2 Introduction Immunotherapy trials in oncology have shown tremendous promise and may represent the most promising cancer treatment approach in the past 50 years. New scientific breakthroughs always come with a learning curve. It s no different with cancer immunotherapy research. To mitigate risk, it s critical for sponsors and investigators and everyone else working on your immunotherapy trial to understand how the immune system responds to cancer treatments. To avoid misinterpreting your data or missing trial deadlines, ensure that your sites and vendors have experience in immunotherapy studies and can help you avoid the numerous potential pitfalls that could cause you to prematurely end your trial when the immunotherapy treatment is actually working. This guide provides eight recommendations to keep your immuno-oncology study on track and was developed from our hands-on experience managing complex cancer immunotherapy studies since About Medelis Medelis is a specialty oncology CRO focused on preclinical and phase I through phase IV oncology studies in North America and Europe. Founded in 2003, Medelis provides customers with oncology expertise from drug development thoughtleadership to a highly skilled clinical operations team, handling complex oncology trials in all indications, including cutting-edge immunotherapy treatments.
3 Immunotherapy in Oncology an Added Level of Complexity Rapid advances in cancer immunotherapy treatments have fueled a tremendous investment in this promising area of research. While sponsors and investigators rush to get immunotherapy treatments (such as checkpoint inhibitors, adoptive cell transfer, therapeutic antibodies and immune system modulators) into the clinic, it s important not to overlook the operational considerations associated with cancer immunotherapy studies. As we all know, cancer studies are more complex than studies in most other therapeutic areas. And furthermore, cancer immunotherapy studies are more complex than most typical cancer studies. This complexity requires an additional level of detailed planning to properly address the nuances of an immunotherapy study. Failure to do so could cause you to prematurely end a study even though the therapy is actually working or end up with extended timelines, bad data and cost overruns.
4 [ 1 ] Site Selection The First Critical Operational Decision Proper site selection is critical to the success of any oncology study. It s even more critical in the rapidly expanding area of cancer immunotherapy. To get the right data within your timelines, you need: 1. The investigators who have access to the patients your study requires 2. The right mix of sites (academic, government and community-based) that are able to get patients enrolled within your timeline These variables directly affect your study cost and data quality. In addition, if your study is phase I, you will also have to evaluate their dose-escalation experience. 3. Sites that are skilled and experienced in handling the type of treatment being administered 4. Sites that understand how to deal with the differences in how the immune system reacts to these types of treatments
5 [ 2 ] Investigational Product Handling Several of the immunotherapy studies we ve managed recently have required us to follow the NIH BioSafety Level 2 (BSL-2) Guidelines for the handling of therapies utilizing microorganisms associated with human diseases. Biosafety levels, or BSLs, are designations to classify the level of danger of a biological agent in an enclosed laboratory facility. The designations and risks associated are as follows: Level 1 The lowest level, this applies to agents that pose minimal threat to lab workers and the environment. Level 2 This includes work with agents that are associated with human disease pathogenic or infectious organisms posing a moderate health hazard. Level 3 Indigenous or exotic agents that cause serious or lethal disease via aerosol transmission. Level 4 Extremely dangerous agents that pose risk of life-threatening diseases for which there are no vaccines or cures. If your treatment is BSL-2, it will affect your site selection. Not all sites have the processes or infrastructure to handle these types of agents, which require the use of surgical gowns, gloves, goggles, safety glasses and possibly a Class II biological safety cabinet. Additionally, the local investigational biosafety committee (IBC) must approve the site s level of preparation to handle these types of agents. This was a challenge for Medelis during our first cancer immunotherapy studies (back in 2008) because at that time, not all sites had an IBC. Thus, we had to work with the sites to help them establish one. As Level 2 immunotherapy agents become more prominent, institutions are becoming better prepared, but many sites still require assistance with infrastructure issues.
6 [ 3 ] Biosample Collection A key goal of cancer immunotherapy studies is to collect and utilize specimens from patients for immuno monitoring, and for developing and credentialing biomarkers that will assess the pharmacodynamic effects or that can serve as predictors of response. Thus, it s critical to plan for the logistics of sample collection, processing and shipping during trial planning. Consider: Containers Do you require specialized containers? Transport What requirements should be in place for the couriers? Most immunotherapy treatments require temperature control during transport and storage. Customs If you re running a global study, what are the implications of samples passing through customs? This may impact the countries you select. In the Aduro Biotech GVAX Pancreas Prime study, we handled this by working closely with the drug distributor to coordinate just-in-time delivery with the vaccine and dry nitrogen shippers. We also worked closely with the site to ensure that there were no patient treatment delays. [ 4 ] Product Storage Many of the new immunotherapy treatments require specialized storage procedures. Review the site s pharmacy procedures for monitoring the temperature of stored products, along with how they report deviations. For many therapies, an ultra-low temperature freezer is required, and in recent years, vendors have started producing this type of freezer in sizes that fit in a typical site s pharmacy.
7 [ 5 ] Monitoring iraes and SAEs in Oncology Immunotherapy Studies The evaluation and handling of SAEs in cancer immunotherapy studies are critical to the outcome of the study. The majority of the events will be autoimmune-related, since the body will start attacking itself. Many patients enrolled in these studies have advanced cancers and have failed most treatments, so it s common for them to have weakened immune systems. Therapies targeting checkpoint receptors have shown to increase toxicities that can create a unique set of adverse events called immune-related adverse events (iraes). Research is showing that combinations of different checkpoint inhibitors improve the body s ability to eradicate cancer, but at the cost of an increase in frequency and severity of iraes. Patients undergoing immunotherapy treatments may experience reactions like dermatitis (from rashes to toxic epidermal necrolysis), pancreatitis, immunitis, iridocyclitis, hepatotoxicity, hypophysitis, neuropathies, nephritis and hepatitis. Some patients with underlying autoimmune disorders can experience exacerbations of underlying autoimmune conditions when on therapies blocking CTLA-4 and PD-1. These reactions often start out mild but can become severe and life-threatening if not recognized. Since multiple studies have shown that there is an association between iraes and clinical benefit, it s important to have a comprehensive approach for evaluating and treating SAEs. [ 6 ] SAE and irae Management For every oncology immunotherapy study we recommend the following: Training for Early Recognition Early recognition and AE management is critical to allow the patient to continue on the study treatment per the protocol. This can be addressed during site training by educating the study coordinator research nurses about potential immune-related AE symptoms. Combination Therapy Evaluation Each therapy s relationship with the AE must be assessed. Recent studies have shown that algorithms designed for specific therapies can help clinicians manage and treat the most common iraes associated with the treatment. These can often predict the irae frequency and severity based on dosage and frequency of the treatment.
8 [ 7 ] Specialized Report Forms Adverse event report forms must be designed in a way that addresses the possibility for multiple attributions. For example, if there is more than one drug being given, the case report form (CRF) needs to ask about the AE s relationship to each individual drug, rather than the study treatment as a whole. [ 8 ] CRF Guidelines Case report guidelines must include specific instructions for the site on how to record the AE. The research has made it clear that the preparation for, handling of, and analysis of specific types of SAEs and iraes are critical for immuno-oncology studies to accurately measure the success of the treatment, to reduce trial delays that extend timelines and increase costs, and most importantly, to ensure the safety of the patients on protocol. Experience Is the Key Following these eight guidelines can help you overcome the numerous obstacles that many sponsors encounter in the clinic with their first immuno-oncology study. Connect with us if you d like assistance with site selection or with the details of your upcoming immunotherapy study. immunotherapy@medelis.com To learn more about our expanded capabilities, visit us at
A BSL-1 lab houses activities that require only standard microbial practices. These include:
 BSL-1 A BSL-1 lab houses activities that require only standard microbial practices. These include: Work should be performed on an open lab bench or table Spills must be decontaminated immediately Infectious
BSL-1 A BSL-1 lab houses activities that require only standard microbial practices. These include: Work should be performed on an open lab bench or table Spills must be decontaminated immediately Infectious
BIOSAFETY AND INFECTION CONTROL ISSUES TIPS FOR THE PHYSICIAN DR E.O. SHOBOWALE CONSULTANT MEDICAL MICROBIOLOGIST PATHCARE NIGERIA
 BIOSAFETY AND INFECTION CONTROL ISSUES TIPS FOR THE PHYSICIAN DR E.O. SHOBOWALE CONSULTANT MEDICAL MICROBIOLOGIST PATHCARE NIGERIA OUTLINE PRE-TEST INTRODUCTION BIOSAFETY CONCEPTS INFECTION CONTROL ISSUES
BIOSAFETY AND INFECTION CONTROL ISSUES TIPS FOR THE PHYSICIAN DR E.O. SHOBOWALE CONSULTANT MEDICAL MICROBIOLOGIST PATHCARE NIGERIA OUTLINE PRE-TEST INTRODUCTION BIOSAFETY CONCEPTS INFECTION CONTROL ISSUES
PPD S EXPERT HEMATOLOGY AND ONCOLOGY TEAM
 HEMATOLOGY ONCOLOGY PPD S EXPERT HEMATOLOGY AND ONCOLOGY TEAM COMMITTED TO ADVANCING DRUG DEVELOPMENT IN ONCOLOGY $ MARKETPLACE COMPLEXITIES increasingly competitive marketplace and rising cost pressures
HEMATOLOGY ONCOLOGY PPD S EXPERT HEMATOLOGY AND ONCOLOGY TEAM COMMITTED TO ADVANCING DRUG DEVELOPMENT IN ONCOLOGY $ MARKETPLACE COMPLEXITIES increasingly competitive marketplace and rising cost pressures
Biosafety Basics. Patrick Stockton, Director, Office of Biosafety Biosafety Community Liaison Committee June 26, 2017
 Biosafety Basics Patrick Stockton, Director, Office of Biosafety Biosafety Community Liaison Committee June 26, 2017 What is Biosafety? The discipline that addresses how to provide guidance and assistance
Biosafety Basics Patrick Stockton, Director, Office of Biosafety Biosafety Community Liaison Committee June 26, 2017 What is Biosafety? The discipline that addresses how to provide guidance and assistance
The role of National Influenza Centres (NICs) during Interpandemic, Pandemic Alert and Pandemic Periods
 The role of National Influenza Centres (NICs) during Interpandemic, Pandemic Alert and Pandemic Periods INTRODUCTION National Influenza Centres (NICs) are the backbone of the WHO Global Influenza Surveillance
The role of National Influenza Centres (NICs) during Interpandemic, Pandemic Alert and Pandemic Periods INTRODUCTION National Influenza Centres (NICs) are the backbone of the WHO Global Influenza Surveillance
Laboratory Orientation and Safety
 Laboratory Orientation and Safety Laboratory Safety Prevention of physical injuries and laboratory acquired infections with zoonotic disease agents Laboratory safety is every employee s responsibility!
Laboratory Orientation and Safety Laboratory Safety Prevention of physical injuries and laboratory acquired infections with zoonotic disease agents Laboratory safety is every employee s responsibility!
Immunotherapies. Supporting health professionals to safely care for patients receiving immunotherapy in the cancer setting.
 Immunotherapies Supporting health professionals to safely care for patients receiving immunotherapy in the cancer setting. INDEX Select a topic below for more information Principles of immunotherapy Classes
Immunotherapies Supporting health professionals to safely care for patients receiving immunotherapy in the cancer setting. INDEX Select a topic below for more information Principles of immunotherapy Classes
Bloodborne Pathogens For School Employees
 Bloodborne Pathogens For School Employees Waynesboro Public Schools Bloodborne Pathogens Training and Annual Review Created on May 5, 2010 Reviewed/Revised April 6, 2017 Introduction In an educational
Bloodborne Pathogens For School Employees Waynesboro Public Schools Bloodborne Pathogens Training and Annual Review Created on May 5, 2010 Reviewed/Revised April 6, 2017 Introduction In an educational
BIOMARKER DRIVEN EARLY PHASE ONCOLOGY CLINICAL TRIALS; CHALLENGES IN THE REAL WORLD SID KATUGAMPOLA CENTRE FOR DRUG DEVELOPMENT CANCER RESEARCH UK
 BIOMARKER DRIVEN EARLY PHASE ONCOLOGY CLINICAL TRIALS; CHALLENGES IN THE REAL WORLD SID KATUGAMPOLA CENTRE FOR DRUG DEVELOPMENT CANCER RESEARCH UK Cancer; Some Sobering Thoughts Getting cancer is one of
BIOMARKER DRIVEN EARLY PHASE ONCOLOGY CLINICAL TRIALS; CHALLENGES IN THE REAL WORLD SID KATUGAMPOLA CENTRE FOR DRUG DEVELOPMENT CANCER RESEARCH UK Cancer; Some Sobering Thoughts Getting cancer is one of
Immuno-Oncology: Perspectives on Current Therapies & Future Developments
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/medical-industry-feature/immuno-oncology-perspectives-currenttherapies-future-developments/9502/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/medical-industry-feature/immuno-oncology-perspectives-currenttherapies-future-developments/9502/
Biosafety & Biosecurity in the Laboratory. Based on the OIE Terrestrial Manual for Diagnostic Tests and Vaccines
 Biosafety & Biosecurity in the Laboratory Based on the OIE Terrestrial Manual for Diagnostic Tests and Vaccines Keeping people and animals safe Keeping samples safe Definitions Biosafety: The measures
Biosafety & Biosecurity in the Laboratory Based on the OIE Terrestrial Manual for Diagnostic Tests and Vaccines Keeping people and animals safe Keeping samples safe Definitions Biosafety: The measures
Adverse events in immunotherapy. Ramy Ibrahim, MD Clinical VP Immuno-Oncology Global Medicine Development AstraZeneca
 Adverse events in immunotherapy Ramy Ibrahim, MD Clinical VP Immuno-Oncology Global Medicine Development AstraZeneca Unique features of immune-related Adverse Events iraes Non-specific Inflammation of
Adverse events in immunotherapy Ramy Ibrahim, MD Clinical VP Immuno-Oncology Global Medicine Development AstraZeneca Unique features of immune-related Adverse Events iraes Non-specific Inflammation of
Bloodborne Pathogens Training. July 26, 2012
 Bloodborne Pathogens Training July 26, 2012 Introduction As sure as the sun comes up every day, children end up with scraped knees, cuts, and bruises. Students of all ages hurt themselves on the playground,
Bloodborne Pathogens Training July 26, 2012 Introduction As sure as the sun comes up every day, children end up with scraped knees, cuts, and bruises. Students of all ages hurt themselves on the playground,
Bloodborne Pathogens Training For School Personnel
 Bloodborne Pathogens Training For School Personnel OSHA Defined: Occupational Safety and Health Administration Published a standard to reduce or eliminate health risk, resulting in: Annual training of
Bloodborne Pathogens Training For School Personnel OSHA Defined: Occupational Safety and Health Administration Published a standard to reduce or eliminate health risk, resulting in: Annual training of
HOW TO MAXIMIZE PATIENT RECRUITMENT IN ONCOLOGY TRIALS A BIOPHARMA DIVE PLAYBOOK
 HOW TO MAXIMIZE PATIENT RECRUITMENT IN ONCOLOGY TRIALS A BIOPHARMA DIVE PLAYBOOK Over the last several decades, patient recruitment for clinical trials has remained a major barrier to rapid execution of
HOW TO MAXIMIZE PATIENT RECRUITMENT IN ONCOLOGY TRIALS A BIOPHARMA DIVE PLAYBOOK Over the last several decades, patient recruitment for clinical trials has remained a major barrier to rapid execution of
CANCER 1.7 M 609,000 26% 15.5 M 73% JUST THE FACTS. More Than 1,100 Cancer Treatments in Clinical Testing Offer Hope to Patients
 CANCER MEDICINES IN DEVELOPMENT 2018 REPORT JUST THE FACTS MORE THAN 1.7 M ESTIMATED NEW CASES OF CANCER IN 2018 IN THE UNITED STATES MORE THAN 609,000 U.S. CANCER DEATHS ARE EXPECTED IN 2018 SINCE PEAKING
CANCER MEDICINES IN DEVELOPMENT 2018 REPORT JUST THE FACTS MORE THAN 1.7 M ESTIMATED NEW CASES OF CANCER IN 2018 IN THE UNITED STATES MORE THAN 609,000 U.S. CANCER DEATHS ARE EXPECTED IN 2018 SINCE PEAKING
Testimony of the Association for Professionals in Infection Control and Epidemiology (APIC) and
 Testimony of the Association for Professionals in Infection Control and Epidemiology (APIC) and The Society for Healthcare Epidemiology of America (SHEA) to the U.S. House of Representatives Appropriations
Testimony of the Association for Professionals in Infection Control and Epidemiology (APIC) and The Society for Healthcare Epidemiology of America (SHEA) to the U.S. House of Representatives Appropriations
Facility and Management
 Training Course of Microbial Resources Information Management and Utilization for Developing Countries BSL-3 Laboratory Facility and Management Dr. Yiping Zhu Sep. 14 2016 Pathogens and BSL-3 laboratory
Training Course of Microbial Resources Information Management and Utilization for Developing Countries BSL-3 Laboratory Facility and Management Dr. Yiping Zhu Sep. 14 2016 Pathogens and BSL-3 laboratory
ICLIO Webinar: Immuno-Oncology: From a Community Radiologist Perspective Michael J. DeLeo III, MD
 ICLIO Webinar: Immuno-Oncology: From a Community Radiologist Perspective Michael J. DeLeo III, MD Foundation Medical Partners Southern New Hampshire Health System December 1, 2016 accc-iclio.org Overview
ICLIO Webinar: Immuno-Oncology: From a Community Radiologist Perspective Michael J. DeLeo III, MD Foundation Medical Partners Southern New Hampshire Health System December 1, 2016 accc-iclio.org Overview
Occupational exposure to bloodborne pathogens
 Occupational exposure to bloodborne pathogens Dr. Sadeghniiat Professor of Tehran University of Medical Sciences Director of Iranian Occupational Medicine Association Head of Imam Khomeini complex hospital
Occupational exposure to bloodborne pathogens Dr. Sadeghniiat Professor of Tehran University of Medical Sciences Director of Iranian Occupational Medicine Association Head of Imam Khomeini complex hospital
Bloodborne Pathogens LVHN s Annual Safety Course
 Slide 1 Bloodborne Pathogens LVHN s Annual Safety Course Nucleus Medical Media (2010). Blood Carries Nutrients. Smart Imagebase. Lehigh Valley Health Network cares about your safety especially about protecting
Slide 1 Bloodborne Pathogens LVHN s Annual Safety Course Nucleus Medical Media (2010). Blood Carries Nutrients. Smart Imagebase. Lehigh Valley Health Network cares about your safety especially about protecting
Current strategies, initiatives and challenges to mitigate biorisk: Indonesia s experience
 Current strategies, initiatives and challenges to mitigate biorisk: Indonesia s experience Head of Delegation HERAWATI SUDOYO MD, PHD Eijkman Institute for Molecular Biology Ministry for Research and Technology
Current strategies, initiatives and challenges to mitigate biorisk: Indonesia s experience Head of Delegation HERAWATI SUDOYO MD, PHD Eijkman Institute for Molecular Biology Ministry for Research and Technology
MONTANA STATE UNIVERSITY-BOZEMAN COLLEGE OF NURSING POLICY # D-3
 MONTANA STATE UNIVERSITY-BOZEMAN COLLEGE OF NURSING POLICY # D-3 TITLE: POLICY: COMMUNICABLE DISEASE PREVENTION POLICIES Nursing students are required to adhere to College of Nursing policies on health
MONTANA STATE UNIVERSITY-BOZEMAN COLLEGE OF NURSING POLICY # D-3 TITLE: POLICY: COMMUNICABLE DISEASE PREVENTION POLICIES Nursing students are required to adhere to College of Nursing policies on health
4. Laboratory Diagnosis of Viral Haemorrhagic Fever Viruses
 4. Laboratory Diagnosis of Viral Haemorrhagic Fever Viruses 4.1 Introduction Early infection with VHF is characterised by a short non-specific viral prodrome with an extensive differential diagnosis. The
4. Laboratory Diagnosis of Viral Haemorrhagic Fever Viruses 4.1 Introduction Early infection with VHF is characterised by a short non-specific viral prodrome with an extensive differential diagnosis. The
Translating Duke Health. Accelerating discovery and its translation
 Translating Duke Health Accelerating discovery and its translation Translating Duke Health Biomedical discovery is accelerating innovative and revolutionary advancements in our understanding of human biology,
Translating Duke Health Accelerating discovery and its translation Translating Duke Health Biomedical discovery is accelerating innovative and revolutionary advancements in our understanding of human biology,
A. Background for Trainer: B. What OSHA Requires: Bloodborne Pathogens. Lesson Plan 6080a
 Lesson Plan 6080a This training session outline is designed to follow the accompanying booklet, OSHA s Bloodborne Pathogens Standard. The booklet reviews what employees who are potentially exposed to the
Lesson Plan 6080a This training session outline is designed to follow the accompanying booklet, OSHA s Bloodborne Pathogens Standard. The booklet reviews what employees who are potentially exposed to the
Highlights from AACR 2015: The Emerging Potential of Immunotherapeutic Approaches in Non-Small Cell Lung Cancer
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Biosafety in the Animal Setting. Tina Bogac Institutional Biosafety Officer
 Biosafety in the Animal Setting Tina Bogac Institutional Biosafety Officer May 27, 2009 Biohazards and Biosafety Risk groups/risk assessment Biosafety/animal biosafety levels Essential work practices Biosafety
Biosafety in the Animal Setting Tina Bogac Institutional Biosafety Officer May 27, 2009 Biohazards and Biosafety Risk groups/risk assessment Biosafety/animal biosafety levels Essential work practices Biosafety
BLOODBORNE PATHOGEN EXPOSURE CONTROL. University of Southern Maine Environmental Health and Safety
 BLOODBORNE PATHOGEN EXPOSURE CONTROL University of Southern Maine Environmental Health and Safety 1 Purpose This presentation provides employees and students with important information regarding potential
BLOODBORNE PATHOGEN EXPOSURE CONTROL University of Southern Maine Environmental Health and Safety 1 Purpose This presentation provides employees and students with important information regarding potential
Bloodborne Pathogens. General
 Bloodborne Pathogens General Session Objectives Identify bloodborne pathogens (BBPs) Understand how diseases are transmitted Determine your risk of exposure Protect yourself from exposure through prevention
Bloodborne Pathogens General Session Objectives Identify bloodborne pathogens (BBPs) Understand how diseases are transmitted Determine your risk of exposure Protect yourself from exposure through prevention
Adverse effects of Immunotherapy. Asha Nayak M.D
 Adverse effects of Immunotherapy Asha Nayak M.D None Financial Disclosures Objectives Understand intensity of the AEs. Understanding unique side-effects. Develop effective monitoring and management guidelines.
Adverse effects of Immunotherapy Asha Nayak M.D None Financial Disclosures Objectives Understand intensity of the AEs. Understanding unique side-effects. Develop effective monitoring and management guidelines.
RESEARCH WITH HIGHLY PATHOGENIC AVIAN INFLUENZA H5N1
 Page 1 of 8 RESEARCH WITH HIGHLY PATHOGENIC AVIAN INFLUENZA H5N1 The University of Pittsburgh has developed guidelines to establish a system of education and safeguards to ensure compliance with both the
Page 1 of 8 RESEARCH WITH HIGHLY PATHOGENIC AVIAN INFLUENZA H5N1 The University of Pittsburgh has developed guidelines to establish a system of education and safeguards to ensure compliance with both the
Title: Supplementary guidelines on handling of
 Revised 1 st July 2006 Page 1 of 7 Supplementary Guideline on Handling of Clinical Specimens in the Laboratory (Revised July 2006) The following guidelines are supplementary to existing standard laboratory
Revised 1 st July 2006 Page 1 of 7 Supplementary Guideline on Handling of Clinical Specimens in the Laboratory (Revised July 2006) The following guidelines are supplementary to existing standard laboratory
Immunotherapy: Toxicity Management. Dr. Megan Lyle Medical Oncologist Liz Plummer Cancer Care Centre Cairns Hospital
 Immunotherapy: Toxicity Management Dr. Megan Lyle Medical Oncologist Liz Plummer Cancer Care Centre Cairns Hospital Disclosures Honoraria and travel support from BMS, MSD, Novartis Advisory board for MSD
Immunotherapy: Toxicity Management Dr. Megan Lyle Medical Oncologist Liz Plummer Cancer Care Centre Cairns Hospital Disclosures Honoraria and travel support from BMS, MSD, Novartis Advisory board for MSD
INTERVIEW / GEORGE COUKOS. / κεντρικό πρόσωπο
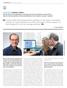 10 INTERVIEW / GEORGE COUKOS MD, PhD, Director of the Department of Oncology at the University Hospital of Lausanne (CHUV), Director of the Lausanne Branch of the Ludwig Institute for Cancer Research,
10 INTERVIEW / GEORGE COUKOS MD, PhD, Director of the Department of Oncology at the University Hospital of Lausanne (CHUV), Director of the Lausanne Branch of the Ludwig Institute for Cancer Research,
Wrentham Public Schools ANNUAL BLOODBORNE PATHOGENS TRAINING
 Wrentham Public Schools ANNUAL BLOODBORNE PATHOGENS TRAINING Are the two diseases specifically addressed by the OSHA Bloodborne Pathogen Standard (Be aware of Hepatitis C as it is becoming more and more
Wrentham Public Schools ANNUAL BLOODBORNE PATHOGENS TRAINING Are the two diseases specifically addressed by the OSHA Bloodborne Pathogen Standard (Be aware of Hepatitis C as it is becoming more and more
INTRODUCTION TO THE OSHA BLOODBORNE PATHOGENS STANDARD
 INTRODUCTION TO THE OSHA BLOODBORNE PATHOGENS STANDARD Background On December 6, 1991 the Occupational Safety and Health Administration (OSHA) published the "Occupational Exposure to Bloodborne Pathogens"
INTRODUCTION TO THE OSHA BLOODBORNE PATHOGENS STANDARD Background On December 6, 1991 the Occupational Safety and Health Administration (OSHA) published the "Occupational Exposure to Bloodborne Pathogens"
Immunotherapy in Lung Cancer
 Immunotherapy in Lung Cancer Jamie Poust Pharm. D., BCOP Oncology Pharmacist University of Colorado Hospital Objectives Describe the recent advances in immunotherapy for patients with lung cancer Outline
Immunotherapy in Lung Cancer Jamie Poust Pharm. D., BCOP Oncology Pharmacist University of Colorado Hospital Objectives Describe the recent advances in immunotherapy for patients with lung cancer Outline
BLOODBORNE PATHOGENS: OR: DO WE REALLY HAVE TO DO THIS AGAIN????
 BLOODBORNE PATHOGENS: OR: DO WE REALLY HAVE TO DO THIS AGAIN???? YES THIS YEAR AND EVERY YEAR HEREAFTER REMEMBER: A REVIEW IS NEVER A BAD IDEA YOUR JOBS PUT YOU AT RISK THE DAY YOU KNOW EVERYTHING AND
BLOODBORNE PATHOGENS: OR: DO WE REALLY HAVE TO DO THIS AGAIN???? YES THIS YEAR AND EVERY YEAR HEREAFTER REMEMBER: A REVIEW IS NEVER A BAD IDEA YOUR JOBS PUT YOU AT RISK THE DAY YOU KNOW EVERYTHING AND
Bloodborne Pathogens Presentation. Itasca County Public Health
 Bloodborne Pathogens Presentation Itasca County Public Health 1 Could You Contract a Disease at Work? Administering first aid? Cleaning the restrooms? Using an item covered with dried blood? A co-worker
Bloodborne Pathogens Presentation Itasca County Public Health 1 Could You Contract a Disease at Work? Administering first aid? Cleaning the restrooms? Using an item covered with dried blood? A co-worker
SMALLPOX QUESTIONS AND ANSWERS: The Disease and the Vaccine
 SMALLPOX QUESTIONS AND ANSWERS: The Disease and the Vaccine In General What should I know about smallpox? Smallpox is an acute, contagious, and sometimes fatal disease caused by the variola virus (an orthopoxvirus),
SMALLPOX QUESTIONS AND ANSWERS: The Disease and the Vaccine In General What should I know about smallpox? Smallpox is an acute, contagious, and sometimes fatal disease caused by the variola virus (an orthopoxvirus),
Overview of Immunotherapy Related Adverse Event (irae) Management
 Overview of Immunotherapy Related Adverse Event (irae) Management DR. ROLAND LEUNG MEDICAL ONCOLOGY DEPARTMENT OF MEDICINE THE UNIVERSITY OF HONG KONG QUEEN MARY HOSPITAL Outline Immune system overview
Overview of Immunotherapy Related Adverse Event (irae) Management DR. ROLAND LEUNG MEDICAL ONCOLOGY DEPARTMENT OF MEDICINE THE UNIVERSITY OF HONG KONG QUEEN MARY HOSPITAL Outline Immune system overview
Enhancing Public Health Disease Surveillance, Detection, Diagnosis and Containment Capability in Nigeria.
 Enhancing Public Health Disease Surveillance, Detection, Diagnosis and Containment Capability in Nigeria. A paper presented to the Meeting of Experts of States Parties to the Biological Weapons Convention,
Enhancing Public Health Disease Surveillance, Detection, Diagnosis and Containment Capability in Nigeria. A paper presented to the Meeting of Experts of States Parties to the Biological Weapons Convention,
Zambia infection prevention guidelines & application in health care facilities
 Zambia infection prevention guidelines & application in health care facilities MANOFF GROUP INC. Answell Chipukuma BCC Specialist 17/04/2008 1 Introduction to Infection Prevention Key Concepts you will
Zambia infection prevention guidelines & application in health care facilities MANOFF GROUP INC. Answell Chipukuma BCC Specialist 17/04/2008 1 Introduction to Infection Prevention Key Concepts you will
Ebola Prevention Vaccine Evaluation in Sierra Leone
 Ebola Prevention Vaccine Evaluation in Sierra Leone Dr. SAS Kargbo Sierra Leone Ministry of Health and Sanitation Sierra Leone CDC Collaboration World Health Organization, Geneva 9 January 2015 Principal
Ebola Prevention Vaccine Evaluation in Sierra Leone Dr. SAS Kargbo Sierra Leone Ministry of Health and Sanitation Sierra Leone CDC Collaboration World Health Organization, Geneva 9 January 2015 Principal
Bloodborne Pathogens Training (OHS_BIO500) Course Material
 Introduction (OHS_BIO500) Course Material Welcome to the Bloodborne Pathogens (BBP) Training Course (OHS_BIO500). UAB Campus Employees whose job duties put them at increased risk for exposure to bloodborne
Introduction (OHS_BIO500) Course Material Welcome to the Bloodborne Pathogens (BBP) Training Course (OHS_BIO500). UAB Campus Employees whose job duties put them at increased risk for exposure to bloodborne
Bloodborne Pathogens and Exposure Control
 Bloodborne Pathogens and Exposure Control 2016 Information in the Exposure Control Plan The Bloodborne Pathogen Exposure Control Plan was developed to communicate information to you about: - Your risk
Bloodborne Pathogens and Exposure Control 2016 Information in the Exposure Control Plan The Bloodborne Pathogen Exposure Control Plan was developed to communicate information to you about: - Your risk
LABORATORY ALERT: Ebola Virus Disease (EVD)
 County of Santa Clara Public Health Department Public Health Laboratory 2220 Moorpark Avenue, 2 nd Floor San José, CA 95128 408.885.4272 DATE: August 21, 2014 LABORATORY ALERT: Ebola Virus Disease (EVD)
County of Santa Clara Public Health Department Public Health Laboratory 2220 Moorpark Avenue, 2 nd Floor San José, CA 95128 408.885.4272 DATE: August 21, 2014 LABORATORY ALERT: Ebola Virus Disease (EVD)
Bloodborne Pathogens. Montclair Kimberley Academy 1
 Bloodborne Pathogens Montclair Kimberley Academy 1 Introduction! Approximately 5.6 million workers in health care and other facilities are at risk of exposure to bloodborne pathogens such as human immunodeficiency
Bloodborne Pathogens Montclair Kimberley Academy 1 Introduction! Approximately 5.6 million workers in health care and other facilities are at risk of exposure to bloodborne pathogens such as human immunodeficiency
Human Umbilical Cord Blood CD34 + /133+ Progenitor Cell Care Manual
 Human Umbilical Cord Blood CD34 + /133+ Progenitor Cell Care Manual INSTRUCTION MANUAL ZBM0065.04 SHIPPING CONDITIONS Human Umbilical Cord Blood CD34+/133+ Progenitor Cells, cryopreserved Cryopreserved
Human Umbilical Cord Blood CD34 + /133+ Progenitor Cell Care Manual INSTRUCTION MANUAL ZBM0065.04 SHIPPING CONDITIONS Human Umbilical Cord Blood CD34+/133+ Progenitor Cells, cryopreserved Cryopreserved
DISCOVERING AND DEVELOPING NEW IMMUNE ACTIVATING MECHANISMS AND ANTIBODIES TO IMPROVE CANCER SURVIVAL
 DISCOVERING AND DEVELOPING NEW IMMUNE ACTIVATING MECHANISMS AND ANTIBODIES TO IMPROVE CANCER SURVIVAL Annual General Meeting 24 April 2018 Björn Frendéus, Acting CEO & CSO 1 IMMUNOTHERAPY HAS TRANSFORMED
DISCOVERING AND DEVELOPING NEW IMMUNE ACTIVATING MECHANISMS AND ANTIBODIES TO IMPROVE CANCER SURVIVAL Annual General Meeting 24 April 2018 Björn Frendéus, Acting CEO & CSO 1 IMMUNOTHERAPY HAS TRANSFORMED
Welcome to the UConn Health s Annual Bloodborne Pathogen Training Module
 Welcome to the UConn Health s Annual Module The Occupational Safety and Health Administration (OSHA) requires that any individual potentially exposed to human blood and/or body fluids in a clinical or
Welcome to the UConn Health s Annual Module The Occupational Safety and Health Administration (OSHA) requires that any individual potentially exposed to human blood and/or body fluids in a clinical or
Evolution of Early Phase Trials: Clinical Trial Design in the Modern Era
 Evolution of Early Phase Trials: Clinical Trial Design in the Modern Era Shivaani Kummar, MD, FACP Professor of Medicine (Oncology) Director, Phase I Clinical Research Program Co-Director, Translational
Evolution of Early Phase Trials: Clinical Trial Design in the Modern Era Shivaani Kummar, MD, FACP Professor of Medicine (Oncology) Director, Phase I Clinical Research Program Co-Director, Translational
CAPACITY BUILDING in DETECTION, DIAGNOSIS and CONTAINMENT of INFECTIOUS DISEASE: INDONESIA S EXPERIENCE, CHALLENGES and NEEDS
 CAPACITY BUILDING in DETECTION, DIAGNOSIS and CONTAINMENT of INFECTIOUS DISEASE: INDONESIA S EXPERIENCE, CHALLENGES and NEEDS Herawati Sudoyo (herawati@eijkman.go.id) Eijkman Institute for Molecular Biology,
CAPACITY BUILDING in DETECTION, DIAGNOSIS and CONTAINMENT of INFECTIOUS DISEASE: INDONESIA S EXPERIENCE, CHALLENGES and NEEDS Herawati Sudoyo (herawati@eijkman.go.id) Eijkman Institute for Molecular Biology,
Applying technical requirements of ISO/IEC 17025:2005 in the Zoonotic diseases testing laboratory. Discussion on the challenges. M.
 Applying technical requirements of ISO/IEC 17025:2005 in the Zoonotic diseases testing laboratory. Discussion on the challenges M.A Motaung Introduction Laboratory facilities are designated as basic Biosafety
Applying technical requirements of ISO/IEC 17025:2005 in the Zoonotic diseases testing laboratory. Discussion on the challenges M.A Motaung Introduction Laboratory facilities are designated as basic Biosafety
Centers for Disease Control and Prevention (CDC) FY 2009 Budget Request Summary
 Centers for Disease Control and Prevention (CDC) FY 2009 Budget Request Summary The President s FY 2009 Budget Request for the Centers for Disease Control and Prevention (CDC) discretionary funding is
Centers for Disease Control and Prevention (CDC) FY 2009 Budget Request Summary The President s FY 2009 Budget Request for the Centers for Disease Control and Prevention (CDC) discretionary funding is
Submitted to the House Energy and Commerce Committee. Federal Efforts to Combat the Opioid Crisis
 STATEMENT FOR THE RECORD Submitted to the House Energy and Commerce Committee Federal Efforts to Combat the Opioid Crisis October 25, 2017 America s Health Insurance Plans 601 Pennsylvania Avenue, NW Suite
STATEMENT FOR THE RECORD Submitted to the House Energy and Commerce Committee Federal Efforts to Combat the Opioid Crisis October 25, 2017 America s Health Insurance Plans 601 Pennsylvania Avenue, NW Suite
Center for Vaccine Biology and Immunology. Cell Sorting Facility Laboratory Safety Manual. Updated: January 25, 2012
 Center for Vaccine Biology and Immunology Cell Sorting Facility Laboratory Safety Manual Updated: January 25, 2012 I. Standard Laboratory Rules The standard BSL-2 precautions are outlined in the University
Center for Vaccine Biology and Immunology Cell Sorting Facility Laboratory Safety Manual Updated: January 25, 2012 I. Standard Laboratory Rules The standard BSL-2 precautions are outlined in the University
Training Objectives. Provide a basic understanding of:
 Training Objectives Provide a basic understanding of: 1. Bloodborne pathogens (BBP) 2. Common modes of transmission of BBP 3. Methods to prevent transmission of BBP 4. Information to help school staff
Training Objectives Provide a basic understanding of: 1. Bloodborne pathogens (BBP) 2. Common modes of transmission of BBP 3. Methods to prevent transmission of BBP 4. Information to help school staff
your bioassay is in good hands: transfer from a CRO perspective MIKE MERGES March 4, 2013
 your bioassay is in good hands: transfer from a CRO perspective MIKE MERGES March 4, 2013 Contact Information Michael Merges, M.S. Director Analytical Development Solutions Development & Clinical Services
your bioassay is in good hands: transfer from a CRO perspective MIKE MERGES March 4, 2013 Contact Information Michael Merges, M.S. Director Analytical Development Solutions Development & Clinical Services
Bloodborne Pathogens
 Bloodborne Pathogens Session Objectives Identify bloodborne pathogens (BBPs) Understand how diseases are transmitted Risk of exposure Protecting yourself from exposure through prevention Responding appropriately
Bloodborne Pathogens Session Objectives Identify bloodborne pathogens (BBPs) Understand how diseases are transmitted Risk of exposure Protecting yourself from exposure through prevention Responding appropriately
BIOSAFETY IN MICROBIOLOGICAL
 BIOSAFETY BIOSAFETY IN MICROBIOLOGICAL AND BIOMEDICAL LABORATORIES BMBL is the Bible for Biosafety matters Published by US Dept. of Health and Human Services and CDC and NIH Does not have the force of
BIOSAFETY BIOSAFETY IN MICROBIOLOGICAL AND BIOMEDICAL LABORATORIES BMBL is the Bible for Biosafety matters Published by US Dept. of Health and Human Services and CDC and NIH Does not have the force of
GUIDE TO INFECTION CONTROL IN THE HOSPITAL. Emergency Department and Receiving Areas CHAPTER 24: Author P. Suri, MD R. Gopaul, MD
 GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER 24: Emergency Department and Receiving Areas Author P. Suri, MD R. Gopaul, MD Chapter Editor Gonzalo Bearman MD, MPH, FACP, FSHEA, FIDSA Topic Outline
GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER 24: Emergency Department and Receiving Areas Author P. Suri, MD R. Gopaul, MD Chapter Editor Gonzalo Bearman MD, MPH, FACP, FSHEA, FIDSA Topic Outline
9/22/2016. Introduction / Goals. What is Cancer? Pharmacologic Strategies to Treat Cancer. Immune System Modulation
 Immunomodulatory Therapies in Cancer Treatment Bill O Hara, PharmD, BCPS, BCOP Advanced Practice Pharmacist, Oncology/BMT Thomas Jefferson University Hospital Introduction / Goals What is Cancer? How can
Immunomodulatory Therapies in Cancer Treatment Bill O Hara, PharmD, BCPS, BCOP Advanced Practice Pharmacist, Oncology/BMT Thomas Jefferson University Hospital Introduction / Goals What is Cancer? How can
JEFFERIES 2018 LONDON HEALTHCARE CONFERENCE NOVEMBER 15, 2018
 1 JEFFERIES 2018 LONDON HEALTHCARE CONFERENCE NOVEMBER 15, 2018 FORWARD-LOOKING STATEMENTS This presentation includes forward-looking statements that involve risks, uncertainties and other factors, many
1 JEFFERIES 2018 LONDON HEALTHCARE CONFERENCE NOVEMBER 15, 2018 FORWARD-LOOKING STATEMENTS This presentation includes forward-looking statements that involve risks, uncertainties and other factors, many
Guidance Document Infectious Substances
 Guidance Document Infectious Substances Note: 1. The following Guidance Document was developed by the ICAO DGP. The original ICAO document reflects references to the ICAO Technical Instructions these have
Guidance Document Infectious Substances Note: 1. The following Guidance Document was developed by the ICAO DGP. The original ICAO document reflects references to the ICAO Technical Instructions these have
SAM HOUSTON STATE UNIVERSITY ENVIRONMENTAL HEALTH, SAFETY & RISK MANAGMENT
 BLOODBORNE PATHOGENS PROGRAM I. PURPOSE The SHSU Bloodborne Pathogens program ensures SHSU compliance with Occupational Safety and Health (OSHA) Standard, 29 CFR 1910.1030, Blood Borne Pathogens. II. SCOPE
BLOODBORNE PATHOGENS PROGRAM I. PURPOSE The SHSU Bloodborne Pathogens program ensures SHSU compliance with Occupational Safety and Health (OSHA) Standard, 29 CFR 1910.1030, Blood Borne Pathogens. II. SCOPE
Bloodborne Pathogens. Penn State University Environmental Health & Safety
 Bloodborne Pathogens Penn State University Environmental Health & Safety Diseases of Concern Hepatitis B (Serum Hepatitis) Hepatitis C (non-a non-b Hepatitis) HIV (Human Immunodeficiency Virus aka the
Bloodborne Pathogens Penn State University Environmental Health & Safety Diseases of Concern Hepatitis B (Serum Hepatitis) Hepatitis C (non-a non-b Hepatitis) HIV (Human Immunodeficiency Virus aka the
Indonesia s challenges and needs in improving national capabilities for disease surveillance, detection and diagnosis and public health systems:
 Indonesia s challenges and needs in improving national capabilities for disease surveillance, detection and diagnosis and public health systems: National capabilities in public health systems Indonesia
Indonesia s challenges and needs in improving national capabilities for disease surveillance, detection and diagnosis and public health systems: National capabilities in public health systems Indonesia
Bloodborne Pathogens Key Topic for 2015 Gloves The First Line of Defense
 Key Topic for 2015 In 2014, the Occupation Health & Safety (OH&S) course focused on the first line of defense - gloves. The information covers: 1. Determining the different types of gloves for specific
Key Topic for 2015 In 2014, the Occupation Health & Safety (OH&S) course focused on the first line of defense - gloves. The information covers: 1. Determining the different types of gloves for specific
WHO Laboratory Biosafety Manual (LBM) Revision Update. Dr Kazunobu KOJIMA World Health Organization (WHO)
 WHO Laboratory Biosafety Manual (LBM) Revision Update Dr Kazunobu KOJIMA World Health Organization (WHO) Presentation overview Background Key concepts Aiming to achieve Risk (hazard) groups Biosafety levels
WHO Laboratory Biosafety Manual (LBM) Revision Update Dr Kazunobu KOJIMA World Health Organization (WHO) Presentation overview Background Key concepts Aiming to achieve Risk (hazard) groups Biosafety levels
Bloodborne Pathogens Training
 Bloodborne Pathogens Training OSHA S Bloodborne Pathogen Standard 29CFR 1910.1030 Employers must: Develop an Exposure Control Plan (ECP) that details their Bloodborne Pathogens (BBP) Program Provide employees
Bloodborne Pathogens Training OSHA S Bloodborne Pathogen Standard 29CFR 1910.1030 Employers must: Develop an Exposure Control Plan (ECP) that details their Bloodborne Pathogens (BBP) Program Provide employees
How to be Successful in Clinical Research
 How to be Successful in Clinical Research W. Gerald Teague, MD Child Health Research Center Department of Pediatrics A True Elevator Story It is Monday, September 12 7:15 am at the big academic medical
How to be Successful in Clinical Research W. Gerald Teague, MD Child Health Research Center Department of Pediatrics A True Elevator Story It is Monday, September 12 7:15 am at the big academic medical
Setting the Scene in Immuno-oncology
 Applying lessons learnt ICONplc.com/oncology Contents General Aspects of Immuno-oncology 3 Immuno-oncology trials for the last 5 years 3 Lessons Learned from I-O Trials 4 Pseudo-Progression and I-O Therapy
Applying lessons learnt ICONplc.com/oncology Contents General Aspects of Immuno-oncology 3 Immuno-oncology trials for the last 5 years 3 Lessons Learned from I-O Trials 4 Pseudo-Progression and I-O Therapy
BLOODBORNE PATHOGENS IN FIRST RESPONSE ENVIRONMENTS
 1708 BLOODBORNE PATHOGENS IN FIRST RESPONSE ENVIRONMENTS Leader s Guide Marcom Group Ltd. Structure and Organization Information in this program is presented in a definite order so that employees will
1708 BLOODBORNE PATHOGENS IN FIRST RESPONSE ENVIRONMENTS Leader s Guide Marcom Group Ltd. Structure and Organization Information in this program is presented in a definite order so that employees will
OPTIMAL MANAGEMENT OF IMMUNE- RELATED ADVERSE EVENTS ASSOCIATED WITH CHECKPOINT INHIBITORS
 OPTIMAL MANAGEMENT OF IMMUNE- RELATED ADVERSE EVENTS ASSOCIATED WITH CHECKPOINT INHIBITORS Alberto Fusi Charité Comprehensive Cancer Centre Berlin, Germany 1 Immune check point blockade with CTLA-4, anti-pd-1
OPTIMAL MANAGEMENT OF IMMUNE- RELATED ADVERSE EVENTS ASSOCIATED WITH CHECKPOINT INHIBITORS Alberto Fusi Charité Comprehensive Cancer Centre Berlin, Germany 1 Immune check point blockade with CTLA-4, anti-pd-1
BLOODBORNE PATHOGENS TRAINING for Faculty, Staff, Instructors and Students. DePaul University School of Nursing Academic year
 BLOODBORNE PATHOGENS TRAINING for Faculty, Staff, Instructors and Students DePaul University School of Nursing 2017-2018 Academic year WELCOME TO BBP TRAINING Your job is helping others. But sometimes
BLOODBORNE PATHOGENS TRAINING for Faculty, Staff, Instructors and Students DePaul University School of Nursing 2017-2018 Academic year WELCOME TO BBP TRAINING Your job is helping others. But sometimes
Gabriel L. Hamer, PhD DEPARTMENT OF ENTOMOLOGY. Minnie Belle Heep 2475 TAMU College Station, TX
 DEPARTMENT OF ENTOMOLOGY To: Texas Municipalities From: Gabriel L. Hamer, PhD, Clinical Assistant Professor, Department of Entomology Date: 17 April 2013 Subject: Dead bird surveillance for West Nile virus
DEPARTMENT OF ENTOMOLOGY To: Texas Municipalities From: Gabriel L. Hamer, PhD, Clinical Assistant Professor, Department of Entomology Date: 17 April 2013 Subject: Dead bird surveillance for West Nile virus
Safety Data Sheet Product Name: Heparin / PF4 Serum Panel - For In Vitro Diagnostic Use Only
 Section 1 Identification Product Name Catalog Numbers Heparin / PF4 Assayed Antibody Serum Panel 4036027, 2 Member QC Panel 4036028, Multi-Member Qualification Panel (6 member) Company Identification AKERS
Section 1 Identification Product Name Catalog Numbers Heparin / PF4 Assayed Antibody Serum Panel 4036027, 2 Member QC Panel 4036028, Multi-Member Qualification Panel (6 member) Company Identification AKERS
Biosafety Level 2 Standard Practices and Containment Office of Research Safety Affairs
 Biosafety Level 2 Standard Practices and Containment Office of Research Safety Affairs Purpose and Objectives Purpose To promote the safe conduct of research with biohazardous materials known to present
Biosafety Level 2 Standard Practices and Containment Office of Research Safety Affairs Purpose and Objectives Purpose To promote the safe conduct of research with biohazardous materials known to present
SURVEY REPORT CLINICAL TRIALS: PAST, PRESENT AND FUTURE
 SURVEY REPORT CLINICAL TRIALS: PAST, PRESENT AND FUTURE + OVERVIEW Clinical trials have changed dramatically in the last 10 years. Sponsors are increasingly pressured to get drugs to market faster and
SURVEY REPORT CLINICAL TRIALS: PAST, PRESENT AND FUTURE + OVERVIEW Clinical trials have changed dramatically in the last 10 years. Sponsors are increasingly pressured to get drugs to market faster and
Occupational Exposures to Bloodborne Pathogen (BBP) Training
 Occupational Exposures to Bloodborne Pathogen (BBP) Training OSHA 29 CFR 1910.1030 Protects workers exposed to blood or other potentially infectious diseases Who are at Risk? Workers in many different
Occupational Exposures to Bloodborne Pathogen (BBP) Training OSHA 29 CFR 1910.1030 Protects workers exposed to blood or other potentially infectious diseases Who are at Risk? Workers in many different
Drew University Bloodborne Pathogens Exposure Control Plan and Procedures
 PURPOSE To provide a written plan for preventing and/or minimizing exposure to bloodborne pathogens for those Drew University personnel who may be involved in the handling of human blood, blood products,
PURPOSE To provide a written plan for preventing and/or minimizing exposure to bloodborne pathogens for those Drew University personnel who may be involved in the handling of human blood, blood products,
Goal of site data management 12/2/2009. Ultimate goal: reliable and valid clinical trial to improve health
 RC-4 Overview of Clinical Site Operations and Management Unit 4: Overview of Data Quality and Monitoring Principal Investigators Lynda Wilson, PhD, RN Marti Rice, PhD, RN Program Manager TBA Course Instructors:
RC-4 Overview of Clinical Site Operations and Management Unit 4: Overview of Data Quality and Monitoring Principal Investigators Lynda Wilson, PhD, RN Marti Rice, PhD, RN Program Manager TBA Course Instructors:
FACEMASKS. Helping you face infection control. a comprehensive guide for safety and protection
 E D U C A T I O N A L S E R I E S FACEMASKS Helping you face infection control a comprehensive guide for safety and protection Prevention is a lot easier when you re properly equipped with appropriate
E D U C A T I O N A L S E R I E S FACEMASKS Helping you face infection control a comprehensive guide for safety and protection Prevention is a lot easier when you re properly equipped with appropriate
GLOVES. Getting a grip on infection control. a complete guide for every exposure level
 E D U C A T I O N A L S E R I E S GLOVES Getting a grip on infection control a complete guide for every exposure level TIDI understands that choosing what gloves to wear, how to wear them, and knowing
E D U C A T I O N A L S E R I E S GLOVES Getting a grip on infection control a complete guide for every exposure level TIDI understands that choosing what gloves to wear, how to wear them, and knowing
1 st International Reference Panel for HIV-1 RNA Genotypes NIBSC code: 01/466
 1 st International Reference Panel for HIV-1 RNA Genotypes NIBSC code: 01/466 Instructions for Use (8 th September 2004, Version 3) Changes from previous version are highlighted in red, deletions are shown
1 st International Reference Panel for HIV-1 RNA Genotypes NIBSC code: 01/466 Instructions for Use (8 th September 2004, Version 3) Changes from previous version are highlighted in red, deletions are shown
Bloodborne Pathogens 29 CFR
 Bloodborne Pathogens 29 CFR 1910.1030 Revised OSHA Bloodborne Pathogens Compliance Directive (CPL2-2.44D) Could You Contract a Disease at Work? Administering first aid? Cleaning the restrooms? Using a
Bloodborne Pathogens 29 CFR 1910.1030 Revised OSHA Bloodborne Pathogens Compliance Directive (CPL2-2.44D) Could You Contract a Disease at Work? Administering first aid? Cleaning the restrooms? Using a
ULTIMATE GBG 95 UC-0140/1606 BIG UnLock The IMmune cells ATtraction in ER+ breast cancer
 ULTIMATE GBG 95 UC-0140/1606 BIG 16-01 UnLock The IMmune cells ATtraction in ER+ breast cancer A PHASE II TRIAL TESTING DURVALUMAB COMBINED WITH ENDOCRINE THERAPY IN PATIENTS WITH ER+/HER2- BREAST CANCER
ULTIMATE GBG 95 UC-0140/1606 BIG 16-01 UnLock The IMmune cells ATtraction in ER+ breast cancer A PHASE II TRIAL TESTING DURVALUMAB COMBINED WITH ENDOCRINE THERAPY IN PATIENTS WITH ER+/HER2- BREAST CANCER
Bloodborne Pathogens
 Bloodborne Pathogens Eddie Pena Environmental October 29, 2012 and November 20, 2012 Based upon SAISD presentation from SAISD Student Health Services BBP Fac Serv Oct / Nov 2012 1 Why Talk About Bloodborne
Bloodborne Pathogens Eddie Pena Environmental October 29, 2012 and November 20, 2012 Based upon SAISD presentation from SAISD Student Health Services BBP Fac Serv Oct / Nov 2012 1 Why Talk About Bloodborne
Questions and Answers on Ebola
 Updated: August 26, 2014 The 2014 Ebola outbreak is the largest Ebola outbreak in history and the first in West Africa. The current outbreak is affecting four countries in West Africa: Guinea, Liberia,
Updated: August 26, 2014 The 2014 Ebola outbreak is the largest Ebola outbreak in history and the first in West Africa. The current outbreak is affecting four countries in West Africa: Guinea, Liberia,
Blood borne Pathogen
 Blood borne Pathogen Training For Certified Nursing Assistants Meets the Blood borne Pathogens & Infection Control Update (Formerly HIV/AIDS) 1 0 In-service Hour Meets the Blood borne Pathogens & Infection
Blood borne Pathogen Training For Certified Nursing Assistants Meets the Blood borne Pathogens & Infection Control Update (Formerly HIV/AIDS) 1 0 In-service Hour Meets the Blood borne Pathogens & Infection
Guidelines for Wildlife Disease Surveillance: An Overview 1
 Guidelines for Wildlife Disease Surveillance: An Overview 1 Purpose of Wildlife Disease Surveillance Wildlife disease surveillance can be a useful and complementary component of human and animal disease
Guidelines for Wildlife Disease Surveillance: An Overview 1 Purpose of Wildlife Disease Surveillance Wildlife disease surveillance can be a useful and complementary component of human and animal disease
Cancer changes lives, but so do you. How YOU are changing the cancer story
 Cancer changes lives, but so do you. How YOU are changing the cancer story Research and Clinical Trials Report to Donors 2017 You ve given the gift of HOPE Hope is at the heart of cancer research. Hope
Cancer changes lives, but so do you. How YOU are changing the cancer story Research and Clinical Trials Report to Donors 2017 You ve given the gift of HOPE Hope is at the heart of cancer research. Hope
immunotherapy a guide for the patient
 immunotherapy FOR LUNG CANCER a guide for the patient TABLE OF CONTENTS lung cancer basics... 2 immunotherapy... 5 Checkpoint Inhibitors... 6-9 Monoclonal Antibodies... 9 Vaccines... 10 Adoptive T-Cell
immunotherapy FOR LUNG CANCER a guide for the patient TABLE OF CONTENTS lung cancer basics... 2 immunotherapy... 5 Checkpoint Inhibitors... 6-9 Monoclonal Antibodies... 9 Vaccines... 10 Adoptive T-Cell
Occupational Infections: Risks for the Anesthesiologist
 Occupational Infections: Risks for the Anesthesiologist 2017 {Music} Dr. Alan Jay Schwartz: Hello. This is Alan Jay Schwartz, Editor-in-Chief of the American Society of Anesthesiologists 2017 Refresher
Occupational Infections: Risks for the Anesthesiologist 2017 {Music} Dr. Alan Jay Schwartz: Hello. This is Alan Jay Schwartz, Editor-in-Chief of the American Society of Anesthesiologists 2017 Refresher
"BLOODBORNE PATHOGENS IN FIRST RESPONSE ENVIRONMENTS"
 MAJOR PROGRAM POINTS "BLOODBORNE PATHOGENS IN FIRST RESPONSE ENVIRONMENTS" Training for THE OSHA BLOODBORNE PATHOGENS STANDARD Quality Safety and Health Products, for Today... and Tomorrow Outline of Major
MAJOR PROGRAM POINTS "BLOODBORNE PATHOGENS IN FIRST RESPONSE ENVIRONMENTS" Training for THE OSHA BLOODBORNE PATHOGENS STANDARD Quality Safety and Health Products, for Today... and Tomorrow Outline of Major
the Office of Research Application Portal: Materials to submit:
 Title: PAR 17 237: Centers for AIDS Research (P30) Slots: 1 Internal Deadline: LOI: External Deadline: Award Information: Submission Process: May 26, 2018, 5pm PDT 30 days prior to the application due
Title: PAR 17 237: Centers for AIDS Research (P30) Slots: 1 Internal Deadline: LOI: External Deadline: Award Information: Submission Process: May 26, 2018, 5pm PDT 30 days prior to the application due
Guidelines for Sample Collection and Handling of Human Clinical samples for Laboratory Diagnosis of H1N1 Influenza
 Guidelines for Sample Collection and Handling of Human Clinical samples for Laboratory Diagnosis of H1N1 Influenza National Institute Of Communicable Diseases 22- Sham Nath Marg New Delhi -110054 DIRECTORATE
Guidelines for Sample Collection and Handling of Human Clinical samples for Laboratory Diagnosis of H1N1 Influenza National Institute Of Communicable Diseases 22- Sham Nath Marg New Delhi -110054 DIRECTORATE
SU2C TOP SCIENCE ACCOMPLISHMENTS
 SU2C TOP SCIENCE ACCOMPLISHMENTS INTRODUCTION Since our founding in 2008, Stand Up To Cancer has funded 87 team science projects, with budgets ranging from $250,000 for a short research project to over
SU2C TOP SCIENCE ACCOMPLISHMENTS INTRODUCTION Since our founding in 2008, Stand Up To Cancer has funded 87 team science projects, with budgets ranging from $250,000 for a short research project to over
