8/31/ ) Intermittent androgen deprivation in androgen-sensitive PCa. 1) Alpharadin (Ra223) in CRPC with bone metastases
|
|
|
- Jonathan Ward
- 5 years ago
- Views:
Transcription
1 Bruce J. Roth, M.D. Clinical Trials: Medivation, Oncogenix 1) Alpharadin (Ra223) in CRPC with bone metastases 2) Enzalutamide (MDV-31) in CRPC and prior docetaxel 3) Abiraterone in chemo-naïve CRPC 4) Intermittent androgen deprivation in androgen-sensitive PCa 1
2 Parker C, et al. LBA4512 Differ from beta particles in terms of energy, tissue range, linear-energy transfer, and number of DNA hits needed to kill a cell Deliver and intense and highly localized radiation dose (range 2-1 cell diameters) Double-stranded DNA breaks (does not require cycling cells) Less irradiation of healthy bone marrow 2
3 Calcium-mimetic, forms complexes with hydroxyapatite, incorporated into bony matrix Preferential uptake in areas of new bone formation, targeting tumor cells in close proximity to areaas of new bone growth surrounding metastases Path length of only 4 1 m Half-life of 11.4 days 3
4 Patients Symptomatic CRPC > 2 bone mets No visceral mets Post-docetaxel or doxetaxel unfit Stratification Total ALP < 22 v > 22 Bisphosponate Y/N Prior docetaxel Y/N R A N D O M I Z E 2:1 Treatment 6 injections q 4 wks Radium-223 (5 kbq/kg) + Best standard of care Placebo + Best standard of care N = 922 Primary overall survival Secondary Time to first SRE Time to alk phos progression Total alk phos response Total alk phos normalization Time to PSA progression QOL Safety 4
5 Parameter Radium-223 n = 614 Placebo n = 37 Age, y Mean Race, n (%) Caucasian 575 (94) 29 (95) Baseline ECOG score, n (%) 1 2 Extent of disease, n (%) < 6 metastases 6 2 metastases > 2 metastases/superscan 536 (87) 76 (12) 1 (16) 262 (43) 249 (41) 265 (86) 4 (13) 38 (12) 147 (48) 121 (4) WHO ladder, cancer pain index 2, n (%) 345 (56) 168 (55) % Placebo, n = 37 Median OS: 11.3 months HR = % CI,.581,.832 P =.7 Radium-223, n = 614 Median OS: 14.9 months Month Radium Placebo
6 % Prior docetaxel use Placebo, n = 174 Median: 11.3 months HR =.71 95% CI,.565,.891 P =.37 Radium-223, n = 352 Median: 14.4 months % NO prior docetaxel use HR = % CI,.562,.987 P =.3932 Placebo, n = 133 Median: 11.5 months Radium-223, n = 262 Median: 16.1 months Month Radium Placebo Month Radium Placebo Current bisphosphonate use NO current bisphosphonate use 1 1 HR =.699 HR = % CI,.525, 9 95% CI,.587, P = P = % 5 % 5 4 Radium-223, n = 25 Median: 15.3 months 4 Radium-223, n = 364 Median: 14.5 months 3 3 Placebo, n = Median: 11.5 months Placebo, n = Median: 11. months Month Radium Placebo Month Radium Placebo
7 Total ALP < 22 U/L HR = % CI,.635, P = Radium-223, n = 348 % 5 Median: 17. months % Placebo, n = 169 Median: 15.8 months Total ALP 22 U/L Placebo, n = 138 Median: 8.1 months HR = % CI,.486,.788 P =.9 Radium-223, n = 266 Median: 11.4 months Month Radium Placebo Month Radium Placebo HR =.64 95% CI,.52,.78 P <.1 % Placebo, n = 37 Median: 6.7 months Radium-223, n = 614 Median: 12.2 months Month Radium Placebo *Provisional data 7
8 All Grades Grades 3 or 4 Patients with AEs n, (%) Radium-223 n = 6 Placebo n = 31 Radium-223 n = 6 Placebo n= 31 Hematologic Anemia 187 (31) 92 (31) 77 (13) 39 (13) Neutropenia 3 (5) 3 (1) 13 (2) 2 (1) Thrombocytopenia 69 (12) 17 (6) 38 (6) 6 (2) Non-Hematologic Bone pain 3 (5) 187 (62) 125 (21) 77 (26) Diarrhea 151 (25) 45 (15) 9 (2) 5 (2) Nausea 213 (36) 14 (35) 1 (2) 5 (2) Vomiting 111 (19) 41 (14) 1 (2) 7 (2) Constipation 18 (18) 64 (21) 6 (1) 4 (1) Radium-223 compared with placebo in CRPC patients with bone metastases: Significantly prolonged median OS by 3.6 months (HR =.695; P =.7) 3.5% reduction in risk of death Significantly prolonged median time to first SRE by 5.5 months (HR =.64; P <.1) Further follow-up in all randomized patients continues to show highly favorable safety profile Radium-223, a first-in-class alpha-emitter, may provide a new standard of care for the treatment of CRPC patients with bone metastases 8
9 What standard of care is this replacing? samarium? cabazitaxel? bisphosphonates? denosumab? Let s wait for additional info Peer-reviewed publication ODAC deliberation Package insert That being said, this is a first-in class compound with unexpectedly good results De Bono J, et al. LBA
10 ANTIANDROGENS Chen Y, et al. Lancet Oncol 1: , 29 Adrenal/Paracrine Synthesis AG Keto Abiraterone 1
11 Adrenal/Paracrine Synthesis Chemotherapy Naïve (N=65) Post Chemotherapy (N=75) 62% (4/65) >5% Decline 51% (38/75) >5% Decline 11
12 Patient Population: 1199 patients with progressive CRPC Failed docetaxel chemotherapy R A N D O M I Z E D 2:1 Enzalutamide 16 mg daily n = 8 Placebo n = 399 Primary Endpoint: Overall Survival 2-sided alpha.5; 9% power to detect 24% reduction in mortality (HR=.76) Solitary planned interim analysis at 52 events DSMC called for halting/unblinding the study HR =.631 (.529,.752) P <.1 37% reduction in risk of death Enzalutamide: 18.4 months (95% CI: 17.3, NYR) Placebo: 13.6 months (95% CI: 11.3, 15.8) Enzalutamide Placebo
13 PSA response > 5%: E - 54%; P - 2% (p <.1) > 9%: E - 25%; P - 1% (p <.1) PSA PFS E 8.3 mths; P 3. mths (HR.248, p <.1) RECIST Response CR + PR: E 28.9%; P 3.8% (p <.1) Radiographic PFS E 8.3 mths; P 2.9 mths (HR.44, p <.1) Time to First SRE E 16.7 mths; P 13.3 mths (HR.621, p <.1) QOL Responses (FACT-P) (1-pt increase in score) E 43.2%; P 18.3% (p <.1) 13
14 All Grades Grade 3 Events Enzalutamide (n = 8) Placebo (n = 399) Enzalutamide (n = 8) Placebo (n = 399) Fatigue 33.6% 29.1% 6.3% 7.3% Cardiac Disorders 6.1% 7.5%.9% 2.% Myocardial Infarction.3%.5%.3%.5% LFT Abnormalities 1.% 1.5%.4%.8% Seizure.6%.%.6%.% CASE Time on Study 2 months 1 months 2 months 5 months 1 months On study drug? Yes Yes Yes Off trial drug for 26 days Yes Seizure type Focal onset Generalized Complex partial status Focal onset Unknown, fall not witnessed Recurrence No No No No No Multiple Alcohol IV Lidocaine Potential Large 5 x 4 Atrophy and CNS excess; inadvertentl cm temporal leukoariosis metastases: started on confounding y given just lobe brain on MRI brain; Eye, haloperidol factors before metastases nil else meninges, 7 days seizure* *4 mgs IV lidocaine (lignocaine); patient also on Na+ channel modulator: propafenone (flecainide cerebellar. like antidysrhythmic). prior 14
15 Enzalutamide, a once a day oral Androgen Receptor Signaling Inhibitor, is well tolerated and prolongs survival in men with CRPC by almost 5 months. Enzalutamide improved secondary measures of antitumor activity including health-related quality of life, response, time to SRE and time to disease progression. The androgen receptor remains a valid therapeutic target for treating CRPC following chemotherapy. FDA approved 8/31/212 Pre-chemotherapy phase III trial has completed accrual results likely available in 213 STRIVE phase III enzalutamide versus bicalutamide as second-line hormonal therapy (both non-metastatic and metastatic) just opening to accrual 15
16 Ryan, CJ et al LBA 4518 Testicle Adrenal Intracrine Androgen synthesis inhibition 16
17 C17α-hydroxylase C17/2 lyase Ketoconazole Abiraterone Tak-7 Abiraterone 1 mg daily Prednisone 1 mg daily Randomization 2:1 Placebo daily Prednisone 1 mg daily Primary Endpoint: Secondary Endpoints: 25% survival increase (12 15 mos) TT PSA progression, PFS, PSA response rate 17
18 1195 pts (abiraterone 797: placebo 398); median f/u 12.8 mths: data unblinded at interim analysis OS: abiraterone 14.8 mths: placebo 1.9 mths (HR.65 (95% CI ; P<.1) TT PSA progression: abi 1.2 v placebo 6.6 mths PFS: abi 5.6 v placebo 3.6 mths PSA RR: abi 29% v placebo 6% (P<.1) debono JS, et al: NEJM 364: , 211 Patients Progressive chemonaïve mcrpc patients (Planned N = 188) Asymptomatic or mildly symptomatic R A N D O M I Z E D 1:1 AA 1 mg daily Prednisone 5 mg BID (Actual n = 546) Placebo daily Prednisone 5 mg BID (Actual n = 542) Efficacy end points Co-Primary: rpfs by central review OS Secondary: Time to opiate use (cancer-related pain) Time to initiation of chemotherapy Time to ECOG-PS deterioration TTPP Phase 3 multicenter, randomized, double-blind, placebo-controlled study conducted at 151 sites in 12 countries; USA, Europe, Australia, Canada Stratification by ECOG performance status vs 1 18
19 Progressive disease (PD) by bone scan: Adapted from Consensus Criteria 1 Blinded central radiologist review < 12 weeks after randomization 2 new bone lesions plus 2 additional at confirmation ( 2+2 ) 12 weeks after randomization 2 new bone lesions with subsequent confirmation PD (soft tissue lesions) by CT or MRI by modified RECIST criteria Death from any cause 1. Scher H, J Clin Oncol. 28;26: AA + P (median, mos): NR PL + P (median, mos): 8.3 HR (95% CI):.43 ( ) Progression-Free (%) P value: <.1 2 AA + P PL + P Time to Progression or Death (Months) AA PL Data cutoff 12/2/21. NR, not reached; PL, placebo. 19
20 1 8 Survival (%) AA + P PL + P AA + P (median, mos): NR PL + P (median, mos): 27.2 HR (95% CI):.75 ( ) P value: Time to Death (Months) 33 AA PL Data cutoff 12/2/211. Pre-specified significance level by O Brien-Fleming Boundary = PRESENTED AT AA + P (n = 546) Placebo + P (n = 542) RR (95% CI) P Value PSA decline 5% 62% 24% NA <.1 N=22 N=218 RECIST: Defined objective response 36% 16% (1.591, 3.247) <.1 Complete response Partial response Stable disease Progressive disease 11% 25% 61% 2% 4% 12% 69% 15% 2
21 AA + P Median (months) Placebo + P Median (months) HR (95% CI) P Value Time to opiate use (cancer related pain) NR (.57,.83).1 Time to chemotherapy initiation (.49,.69) <.1 Time to ECOG PS deterioration (.71,.94).53 Time to PSA progression (.42,.57) <.1 Note: All secondary end points remain significant after adjusting for multiplicity testing Data cut off 12/2/211. Patient Reported Outcomes favored AA +P vs Placebo +P Full data to be reported In patients with asymptomatic and mildly symptomatic, chemotherapy-naïve mcrpc, treatment with abiraterone acetate plus prednisone: Delays disease progression Increases survival Extends time with minimal or no symptoms No new important safety signals Granted accelerated review for expanded indication by FDA 21
22 Hussain M, et al 4 Continuous androgen deprivation is the standard approach to advanced disease Preclinical data suggested that intermittent therapy might prolong the time to development of castrate resistant state Smaller clinical trials have confirmed a better QOL for patients receiving intermittent therapy, but have been underpowered to conclude anything regarding overall survival 22
23 S9346 (INT-162): Objectives Primary - Determine if survival with IAD is Not Inferior to survival with CAD. - QOL*: To compare 3 treatment-specific symptoms (Impotence, Libido, Energy/Vitality) and physical and emotional functioning between arms Secondary: -More general QOL measures -PSA dynamics between arms, and correlations with other endpoints *Moinpour et-al, Abstract # 4571 describes results for QOL Eligibility Newly diagnosed metastatic prostate cancer PSA > 5 ng/ml PS -2 Stratification PS (-1 v 2) Extent of disease Minimal (spine, pelvis +/- nodal disease) Extensive (ribs, long bones +/- visceral disease) Prior hormonal tx neoadjuvant therapy v finsteride v neither 23
24 Study Design STEP 1 Induction Registration Newly diagnosed metastatic prostate cancer & a PSA 5 ng/ml Induction AD = Goserelin + Bicalutamide X 7 months STEP 2 Randomly Assign If PSA 4 ng/ml on months 6&7 (PSA normalization criteria) Continuous AD Intermittent AD Discontinue AD, monthly PSAs. Resume AD based on pre-specified criteria Therapy was reinitiated when PSA increased to 2 ng/ml (or returned to baseline for patients who had pre-registration baseline value < 2 ng/ml) or for symptoms. If the PSA after another 7 months induction course met the PSA normalization criterion then the patients started another observation period. If the PSA at either months 6 or 7th of an induction course was greater than 4 ng/ml then the patients received continuous therapy until progression. 24
25 Statistical Methods Primary outcome: Survival post-randomization Hypothesis: IAD is NOT inferior to CAD Design specifications: Survival with IAD is not inferior if the 95% confidence interval for the hazard ratio (IAD vs. CAD) excludes 1.2, α=.5, power=9%, adjusting for stratification factors in proportional hazards model. Assumptions: post-randomization median survival for CAD = 3 years: Sample size: 15 eligible, randomized patients accrual: 6.25 yrs. + 2 additional yrs. of follow-up. Activated: 5/15/1995 Closed: 9/1/28 Step 1: Induction Registrations: 34 pts (9 ineligible) Step 2: Randomization to CAD vs. IAD: 1535 eligible pts (projected 5% randomized) IAD 77 eligible patients CAD 765 eligible patients 25
26 Overall Survival: Intermittent Therapy is Inferior Compared to Continuous Therapy 1% 8% Continuous therapy Intermittent therapy At Risk Death Median in Years yr Survival 42% 38% 6% HR: % CI (.95, 1.24) 4% 2% % At risk Years from Randomization Intermittent Continuous Overall Survival for Patients with Extensive Disease by Treatment Arm 1% 8% Continuous therapy Intermittent therapy At Risk Death Median in Years Year Survival 33% 33% 6% HR:.96 95% CI (.8, 1.16) 4% 2% % At risk Years from Randomization Intermittent Continuous
27 Overall Survival for Patients with Minimal Disease by Treatment Arm 1% 8% 6% Continuous therapy Intermittent therapy At Risk Death Median in Years yr. Survival 5% 42% HR: 1.23, 95% CI (1.2, 1.49) p=.34, 4% 2% % At risk Years from Randomization Intermittent Continuous In this international phase III trial in patients with metastatic hormone sensitive prostate cancer : 1. IAD was inferior to CAD based on our pre-specified definition of survival comparability [HR: 1.9, 95% CI (.95, 1.24)]. Therefore, CAD continues to be the standard of care. 2. In a secondary analysis: IAD was not-inferior to CAD in patients with extensive disease. [HR:.96 95% CI (.8, 1.16)]. IAD was inferior in patients with minimal disease & CAD was statistically significantly superior [HR: 1.23, 95% CI (1.2, 1.49), p=.34]. These observations suggest inherent biological differences and warrant further mechanistic evaluation. 27
28 Huge debate at the meeting regarding the definitions of minimal v extensive metastatic disease Even bigger debate regarding the statistical implications of a therapy being not noninferior If the results of a trial validate your preexisting bias, then you are willing to overlook some statistical aberrations If the results are contrary to your bias, then you attack the statistics unmercifully 28
29 Intermittent androgen deprivation is absolutely superior in terms of QOL But, the assumption that survival is equivalent with this approach is not supported by this trial A patient may still choose IAD based on QOL issues, but the survival differences observed in this study need to be discussed with the patient 29
Until 2004, CRPC was consistently a rapidly lethal disease.
 Until 2004, CRPC was consistently a rapidly lethal disease. the entry in systemic disease is declared on a an isolated PSA recurrence after local treatment so!!! The management of CRPC and MCRPC is different
Until 2004, CRPC was consistently a rapidly lethal disease. the entry in systemic disease is declared on a an isolated PSA recurrence after local treatment so!!! The management of CRPC and MCRPC is different
Group Sequential Design: Uses and Abuses
 Group Sequential Design: Uses and Abuses Susan Halabi Department of Biostatistics and Bioinformatics, Duke University October 23, 2015 susan.halabi@duke.edu What Does Interim Data Say? 2 Group Sequential
Group Sequential Design: Uses and Abuses Susan Halabi Department of Biostatistics and Bioinformatics, Duke University October 23, 2015 susan.halabi@duke.edu What Does Interim Data Say? 2 Group Sequential
ASCO 2012 Genitourinary tumors
 ASCO 2012 Genitourinary tumors Post ASCO Bern 14-06-2012 Dr. med. Richard Cathomas leitender Arzt Onkologie, KSGR, Chur Renal cell cancer Changes in first line treatment? Prostate cancer 3 positive phase
ASCO 2012 Genitourinary tumors Post ASCO Bern 14-06-2012 Dr. med. Richard Cathomas leitender Arzt Onkologie, KSGR, Chur Renal cell cancer Changes in first line treatment? Prostate cancer 3 positive phase
SESSIONE PLATINUM SERIES (Best Papers Poster o Abstract on Prostate Cancer) In Oncologia
 SESSIONE PLATINUM SERIES (Best Papers Poster o Abstract on Prostate Cancer) In Oncologia Divisione di Oncologia Medica Unità Tumori Genitourinari SESSIONE PLATINUM SERIES (Best Papers Poster o Abstract
SESSIONE PLATINUM SERIES (Best Papers Poster o Abstract on Prostate Cancer) In Oncologia Divisione di Oncologia Medica Unità Tumori Genitourinari SESSIONE PLATINUM SERIES (Best Papers Poster o Abstract
SYSTEMIC THERAPIES FOR CRPC: Chemotherapy and Radium-223
 SYSTEMIC THERAPIES FOR CRPC: Chemotherapy and Radium-223 ELENA CASTRO Spanish National Cancer Research Centre Prostate Preceptorship. Lugano 4-5 October 2018 Disclosures Participation in advisory boards:
SYSTEMIC THERAPIES FOR CRPC: Chemotherapy and Radium-223 ELENA CASTRO Spanish National Cancer Research Centre Prostate Preceptorship. Lugano 4-5 October 2018 Disclosures Participation in advisory boards:
Index Patients 3& 4. Guideline Statements 10/11/2014. Enzalutamide Reduced the Risk of Death
 //4 Prolonged Radiographic Progression-Free Survival Reduced the Risk of Death Overall ITT Population Estimated median rpfs, months (9% CI): : NYR (.8 NYR); placebo:.9 (.7.4) rpfs (%) ( Enza 9 8 7 4 8
//4 Prolonged Radiographic Progression-Free Survival Reduced the Risk of Death Overall ITT Population Estimated median rpfs, months (9% CI): : NYR (.8 NYR); placebo:.9 (.7.4) rpfs (%) ( Enza 9 8 7 4 8
Novel treatment for castration-resistant prostate cancer
 Novel treatment for castration-resistant prostate cancer Cora N. Sternberg, MD, FACP Chair, Department of Medical Oncology San Camillo and Forlanini Hospitals Rome, Italy Treatment options for patients
Novel treatment for castration-resistant prostate cancer Cora N. Sternberg, MD, FACP Chair, Department of Medical Oncology San Camillo and Forlanini Hospitals Rome, Italy Treatment options for patients
Management of castrate resistant disease: after first line hormone therapy fails
 Management of castrate resistant disease: after first line hormone therapy fails Rob Jones Consultant in Medical Oncology Beatson Cancer Centre Glasgow Relevant Disclosure I have received research support
Management of castrate resistant disease: after first line hormone therapy fails Rob Jones Consultant in Medical Oncology Beatson Cancer Centre Glasgow Relevant Disclosure I have received research support
Management of Incurable Prostate Cancer in 2014
 Management of Incurable Prostate Cancer in 2014 Julie N. Graff, MD, MCR Portland VA Medical Center Assistant Professor of Medicine Knight Cancer Institute, OHSU 2014: Cancer Estimates Stage at Diagnosis
Management of Incurable Prostate Cancer in 2014 Julie N. Graff, MD, MCR Portland VA Medical Center Assistant Professor of Medicine Knight Cancer Institute, OHSU 2014: Cancer Estimates Stage at Diagnosis
Strategic decisions for systemic treatment. metastatic castration resistant prostate cancer (mcrpc)
 Strategic decisions for systemic treatment metastatic castration resistant prostate cancer (mcrpc) SAMO Luzern 14.09.2012 Richard Cathomas Onkologie Kantonsspital Graubünden richard.cathomas@ksgr.ch mcrpc
Strategic decisions for systemic treatment metastatic castration resistant prostate cancer (mcrpc) SAMO Luzern 14.09.2012 Richard Cathomas Onkologie Kantonsspital Graubünden richard.cathomas@ksgr.ch mcrpc
Management of castrate resistant disease; after first line hormone therapy fails
 Management of castrate resistant disease; after first line hormone therapy fails Dr. Syed A Hussain Clinical Senior Lecturer and Consultant in Medical Oncology University of Liverpool and Clatterbridge
Management of castrate resistant disease; after first line hormone therapy fails Dr. Syed A Hussain Clinical Senior Lecturer and Consultant in Medical Oncology University of Liverpool and Clatterbridge
Isotopes and Palliative Radiotherapy for bone metastases
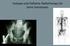
 Isotopes and Palliative Radiotherapy for bone metastases Rationale for Bone-seeking Isotope Therapies in Prostate Cancer > 90% of patients with advanced prostate cancer have bone metastases which can be
Isotopes and Palliative Radiotherapy for bone metastases Rationale for Bone-seeking Isotope Therapies in Prostate Cancer > 90% of patients with advanced prostate cancer have bone metastases which can be
Advanced Prostate Cancer. November Jose W. Avitia, M.D
 Advanced Prostate Cancer November 4 2017 Jose W. Avitia, M.D In 2017 161,000 new cases of prostate cancer diagnosed in US, mostly with elevated PSA 5-10% will present with metastatic disease In 2017: 26,000
Advanced Prostate Cancer November 4 2017 Jose W. Avitia, M.D In 2017 161,000 new cases of prostate cancer diagnosed in US, mostly with elevated PSA 5-10% will present with metastatic disease In 2017: 26,000
Isotopes and Palliative Radiotherapy for bone metastases
 Isotopes and Palliative Radiotherapy for bone metastases Rationale for Bone-seeking Isotope Therapies in Prostate Cancer > 90% of patients with advanced prostate cancer have bone metastases which can be
Isotopes and Palliative Radiotherapy for bone metastases Rationale for Bone-seeking Isotope Therapies in Prostate Cancer > 90% of patients with advanced prostate cancer have bone metastases which can be
Challenging Cases. With Q&A Panel
 Challenging Cases With Q&A Panel Case Studies Index Patient #1 Jeffrey Wieder, MD Case # 1 72 year old healthy male with mild HTN Early 2011: Preop bone scan and pelvic CT = no mets Radical prostatectomy
Challenging Cases With Q&A Panel Case Studies Index Patient #1 Jeffrey Wieder, MD Case # 1 72 year old healthy male with mild HTN Early 2011: Preop bone scan and pelvic CT = no mets Radical prostatectomy
Management of Prostate Cancer
 Management of Prostate Cancer An ESMO Perspective Alan Horwich Conflicts of Interest Disclosure Alan Horwich I have no personal conflicts of interest relating to prostate cancer. European Incidence and
Management of Prostate Cancer An ESMO Perspective Alan Horwich Conflicts of Interest Disclosure Alan Horwich I have no personal conflicts of interest relating to prostate cancer. European Incidence and
Perspective on endocrine and chemotherapy agents. Cora N. Sternberg Department of Medical Oncology San Camillo & Forlanini Hospitals Rome, Italy
 Perspective on endocrine and chemotherapy agents Cora N. Sternberg Department of Medical Oncology San Camillo & Forlanini Hospitals Rome, Italy Disclosures Dr. Sternberg has received research funding for
Perspective on endocrine and chemotherapy agents Cora N. Sternberg Department of Medical Oncology San Camillo & Forlanini Hospitals Rome, Italy Disclosures Dr. Sternberg has received research funding for
LONDON CANCER NEW DRUGS GROUP RAPID REVIEW
 LONDON CANCER NEW DRUGS GROUP RAPID REVIEW Abiraterone for the treatment of metastatic castration-resistant prostate cancer that has progressed on or after a docetaxel-based chemotherapy regimen Disease
LONDON CANCER NEW DRUGS GROUP RAPID REVIEW Abiraterone for the treatment of metastatic castration-resistant prostate cancer that has progressed on or after a docetaxel-based chemotherapy regimen Disease
What will change for men with advanced prostate cancer in the next 24 months? ESO Observatory: Perspective on endocrine and chemotherapy agents
 Perspective on endocrine and chemotherapy agents Cora N. Sternberg Department of Medical Oncology San Camillo & Forlanini Hospitals Rome, Italy Disclosures Dr.Sternberg has received research funding for
Perspective on endocrine and chemotherapy agents Cora N. Sternberg Department of Medical Oncology San Camillo & Forlanini Hospitals Rome, Italy Disclosures Dr.Sternberg has received research funding for
Incorporating New Agents into the Treatment Paradigm for Prostate Cancer
 Incorporating New Agents into the Treatment Paradigm for Prostate Cancer Dr. Celestia S. Higano FACP, Professor, Medicine and Urology, Uni. of Washington Member, Fred Hutchinson Cancer Research Center
Incorporating New Agents into the Treatment Paradigm for Prostate Cancer Dr. Celestia S. Higano FACP, Professor, Medicine and Urology, Uni. of Washington Member, Fred Hutchinson Cancer Research Center
Prostate Cancer 2009 MDV Anti-Angiogenesis. Anti-androgen Radiotherapy Surgery Androgen Deprivation Therapy. Docetaxel/Epothilone
 Prostate Cancer 2009 Anti-Angiogenesis MDV 3100 Anti-androgen Radiotherapy Surgery Androgen Deprivation Therapy Docetaxel/Epothilone Abiraterone DC therapy Bisphosphonates Denosumab Secondary Hormonal
Prostate Cancer 2009 Anti-Angiogenesis MDV 3100 Anti-androgen Radiotherapy Surgery Androgen Deprivation Therapy Docetaxel/Epothilone Abiraterone DC therapy Bisphosphonates Denosumab Secondary Hormonal
Advanced Prostate Cancer
 Advanced Prostate Cancer January 13, 2017 Sindu Kanjeekal MD FRCPC Medical Oncology and Hematology Regional Systemic Quality Lead Erie St Clair Adjunct Professor Schulich School of Medicine and University
Advanced Prostate Cancer January 13, 2017 Sindu Kanjeekal MD FRCPC Medical Oncology and Hematology Regional Systemic Quality Lead Erie St Clair Adjunct Professor Schulich School of Medicine and University
PLAATS VAN DE CHEMOTHERAPIE IN DE BEHANDELING VAN EEN PROSTAATCARCINOOM: EEN UPDATE. Daan De Maeseneer, Medisch Oncoloog
 PLAATS VAN DE CHEMOTHERAPIE IN DE BEHANDELING VAN EEN PROSTAATCARCINOOM: EEN UPDATE Daan De Maeseneer, Medisch Oncoloog 1 Overview DEAT PSA/Tumor Burden METASTASES INITIAL DIAGNOSIS & THERAPY ADT CRP SREs/
PLAATS VAN DE CHEMOTHERAPIE IN DE BEHANDELING VAN EEN PROSTAATCARCINOOM: EEN UPDATE Daan De Maeseneer, Medisch Oncoloog 1 Overview DEAT PSA/Tumor Burden METASTASES INITIAL DIAGNOSIS & THERAPY ADT CRP SREs/
Roberto Sabbatini Azienda Ospedaliero Universitaria Policlinico di Modena
 Il Trattamento della Malattia CRPC metastatica Terapie Radiometaboliche Roberto Sabbatini Azienda Ospedaliero Universitaria Policlinico di Modena AIOM: Gestione ottimale del Paziente con Carcinoma della
Il Trattamento della Malattia CRPC metastatica Terapie Radiometaboliche Roberto Sabbatini Azienda Ospedaliero Universitaria Policlinico di Modena AIOM: Gestione ottimale del Paziente con Carcinoma della
Philip Kantoff, MD Dana-Farber Cancer Institute
 CHEMOTHERAPY FOR MCRPC Philip Kantoff, MD Dana-Farber Cancer Institute Harvard Medical School 1 Disclosure of Financial Relationships With Any Commercial Interest Name Nature of Financial Commercial Interests
CHEMOTHERAPY FOR MCRPC Philip Kantoff, MD Dana-Farber Cancer Institute Harvard Medical School 1 Disclosure of Financial Relationships With Any Commercial Interest Name Nature of Financial Commercial Interests
Androgens and prostate cancer: insights from abiraterone acetate and other novel agents
 Androgens and prostate cancer: insights from abiraterone acetate and other novel agents Ian Davis Ludwig Institute for Cancer Research Austin Health, Melbourne, Australia Supported in part by an Australian
Androgens and prostate cancer: insights from abiraterone acetate and other novel agents Ian Davis Ludwig Institute for Cancer Research Austin Health, Melbourne, Australia Supported in part by an Australian
Secondary Hormonal therapies in mcrpc
 Secondary Hormonal therapies in mcrpc Ravindran Kanesvaran Consultant,Division of Medical Oncology National Cancer Centre Singapore 1 Disclosures Research Support/P.I. Sanofi Consultant Major Stockholder
Secondary Hormonal therapies in mcrpc Ravindran Kanesvaran Consultant,Division of Medical Oncology National Cancer Centre Singapore 1 Disclosures Research Support/P.I. Sanofi Consultant Major Stockholder
2014 Treatment Paradigms in mcrpc Docetaxel in hormone sensitive PC
 Ronald de Wit Erasmus MC Cancer Institute The Netherlands 2014 Treatment Paradigms in mcrpc Docetaxel in hormone sensitive PC Disclosures Sanofi ; research grant support, consultancy and speaker fees Astellas;
Ronald de Wit Erasmus MC Cancer Institute The Netherlands 2014 Treatment Paradigms in mcrpc Docetaxel in hormone sensitive PC Disclosures Sanofi ; research grant support, consultancy and speaker fees Astellas;
Management of castration resistant prostate cancer after first line hormonal therapy fails
 Management of castration resistant prostate cancer after first line hormonal therapy fails Simon Crabb Senior Lecturer in Medical Oncology University of Southampton WHAT ARE THE AIMS OF TREATMENT? Cure?
Management of castration resistant prostate cancer after first line hormonal therapy fails Simon Crabb Senior Lecturer in Medical Oncology University of Southampton WHAT ARE THE AIMS OF TREATMENT? Cure?
Advanced Prostate Cancer
 Advanced Prostate Cancer SAMO Masterclass 4 th March 2016 Aurelius Omlin Conflicts of interest Advisory Rolle: Astra Zeneca, Astellas, Bayer, Janssen, Pfizer, Sanofi Aventis Research support: TEVA, Janssen
Advanced Prostate Cancer SAMO Masterclass 4 th March 2016 Aurelius Omlin Conflicts of interest Advisory Rolle: Astra Zeneca, Astellas, Bayer, Janssen, Pfizer, Sanofi Aventis Research support: TEVA, Janssen
mcrpc 2014 TRA EVOLUZIONE E RIVOLUZIONE: COME ORIENTARSI NEL LABIRINTO DELLE TERAPIE
 mcrpc 2014 TRA EVOLUZIONE E RIVOLUZIONE: COME ORIENTARSI NEL LABIRINTO DELLE TERAPIE IL CARCINOMA PROSTATICO, UNA MALATTIA ETEROGENEA? RAZIONALE E RISULTATI DEL TRATTAMENTO CHEMIOTERAPICO ASSOCIATO ALL
mcrpc 2014 TRA EVOLUZIONE E RIVOLUZIONE: COME ORIENTARSI NEL LABIRINTO DELLE TERAPIE IL CARCINOMA PROSTATICO, UNA MALATTIA ETEROGENEA? RAZIONALE E RISULTATI DEL TRATTAMENTO CHEMIOTERAPICO ASSOCIATO ALL
 www.drpaulmainwaring.com Figure 1 Androgen action Harris W P et al. (2009) Nat Clin Pract Urol doi:10.1038/ncpuro1296 Figure 2 Mechanisms of castration resistance in prostate cancer Harris W P et al. (2009)
www.drpaulmainwaring.com Figure 1 Androgen action Harris W P et al. (2009) Nat Clin Pract Urol doi:10.1038/ncpuro1296 Figure 2 Mechanisms of castration resistance in prostate cancer Harris W P et al. (2009)
ACTUALIZACIONES EN TRATAMIENTOS DIRIGIDOS AL HUESO. COMBINACIÓN CON OTRAS ESTRATEGIAS TERAPÉUTICAS.
 ACTUALIZACIONES EN TRATAMIENTOS DIRIGIDOS AL HUESO. COMBINACIÓN CON OTRAS ESTRATEGIAS TERAPÉUTICAS. ÁLVARO PINTO Servicio de Oncología Médica Hospital Universitario La Paz IdiPAZ, Madrid INTRODUCTION High
ACTUALIZACIONES EN TRATAMIENTOS DIRIGIDOS AL HUESO. COMBINACIÓN CON OTRAS ESTRATEGIAS TERAPÉUTICAS. ÁLVARO PINTO Servicio de Oncología Médica Hospital Universitario La Paz IdiPAZ, Madrid INTRODUCTION High
Prostate cancer update: Dr Robert Huddart Cancer Clinic London
 Prostate cancer update: 2013 Dr Robert Huddart Cancer Clinic London Recent developments Improved imaging New radiotherapy technologies Radiotherapy for advanced disease Intermittent hormone therapy New
Prostate cancer update: 2013 Dr Robert Huddart Cancer Clinic London Recent developments Improved imaging New radiotherapy technologies Radiotherapy for advanced disease Intermittent hormone therapy New
Second line hormone therapies. Dr Lisa Pickering Consultant Medical Oncologist ESMO Preceptorship Singapore 2017
 Second line hormone therapies Dr Lisa Pickering Consultant Medical Oncologist ESMO Preceptorship Singapore 2017 Disclosures Institutional Research Support/P.I. Employee Consultant Major Stockholder Speakers
Second line hormone therapies Dr Lisa Pickering Consultant Medical Oncologist ESMO Preceptorship Singapore 2017 Disclosures Institutional Research Support/P.I. Employee Consultant Major Stockholder Speakers
Evolution or revolution in the treatment of prostate cancer
 Evolution or revolution in the treatment of prostate cancer de Johann Sebastian de Bono, MB, ChB, FRCP, MSc, PhD Professor of Experimental Cancer Medicine Department of Medicine/ Drug Development Unit
Evolution or revolution in the treatment of prostate cancer de Johann Sebastian de Bono, MB, ChB, FRCP, MSc, PhD Professor of Experimental Cancer Medicine Department of Medicine/ Drug Development Unit
When exogenous testosterone therapy is. adverse responses can be induced.
 Theoretical tips It has been reasoned that discontinuation of ADT in nonorchiectomized patients may have detrimental effect on patients with CRPC as discontinuation of ADT can result in renewed release
Theoretical tips It has been reasoned that discontinuation of ADT in nonorchiectomized patients may have detrimental effect on patients with CRPC as discontinuation of ADT can result in renewed release
SIMPOSIO. Radioterapia stereotassica e nuovi farmaci nel tumore e della prostata metastatico
 SIMPOSIO Radioterapia stereotassica e nuovi farmaci nel tumore e della prostata metastatico Definition of Oligometastatic PCa 1-3 synchronous metastases (bone and/or lymph nodes) 2-5 synchronous metastases
SIMPOSIO Radioterapia stereotassica e nuovi farmaci nel tumore e della prostata metastatico Definition of Oligometastatic PCa 1-3 synchronous metastases (bone and/or lymph nodes) 2-5 synchronous metastases
Early Chemotherapy for Metastatic Prostate Cancer
 Early Chemotherapy for Metastatic Prostate Cancer Daniel P. Petrylak, MD Professor of Medicine and Urology Smilow Cancer Center Yale University Medical Center Disclosure Consultant: Sanofi Aventis, Celgene,
Early Chemotherapy for Metastatic Prostate Cancer Daniel P. Petrylak, MD Professor of Medicine and Urology Smilow Cancer Center Yale University Medical Center Disclosure Consultant: Sanofi Aventis, Celgene,
Sequencing Strategies in Metastatic Castration Resistant Prostate Cancer (MCRPC)
 Sequencing Strategies in Metastatic Castration Resistant Prostate Cancer (MCRPC) Amit Bahl Consultant Oncologist Bristol Cancer Institute Clinical Director Spire Specialist Care Centre UK Disclosures Advisory
Sequencing Strategies in Metastatic Castration Resistant Prostate Cancer (MCRPC) Amit Bahl Consultant Oncologist Bristol Cancer Institute Clinical Director Spire Specialist Care Centre UK Disclosures Advisory
MÉTASTASES OSSEUSES ET RADIUM 223
 MÉTASTASES OSSEUSES ET RADIUM 223 Marie-Laure Amram Service d oncologie Hôpitaux Universitaires de Genève Forome du 21.05.2015 Radium-22:3:mécanisme d action Mécanisme d action Mécanisme d action Radium-223
MÉTASTASES OSSEUSES ET RADIUM 223 Marie-Laure Amram Service d oncologie Hôpitaux Universitaires de Genève Forome du 21.05.2015 Radium-22:3:mécanisme d action Mécanisme d action Mécanisme d action Radium-223
Prostate Cancer. Dr. Andres Wiernik 2017
 Prostate Cancer Dr. Andres Wiernik 2017 Objectives YES!!! 1. Epidemiology 2. Biology or Natural History of Prostate Cancer 3. Treatment NO!!! 1. Prostate Cancer Screening - controversies Which is the most
Prostate Cancer Dr. Andres Wiernik 2017 Objectives YES!!! 1. Epidemiology 2. Biology or Natural History of Prostate Cancer 3. Treatment NO!!! 1. Prostate Cancer Screening - controversies Which is the most
Alpha-emitting Radionuclides: Ra-223
 Alpha-emitting Radionuclides: Ra-223 prof. dr. K. Goffin Nuclear Medicine and Molecular Imaging Department of Imaging & Pathology UZ Leuven KU Leuven Belgium International Course on Theranostics and Molecular
Alpha-emitting Radionuclides: Ra-223 prof. dr. K. Goffin Nuclear Medicine and Molecular Imaging Department of Imaging & Pathology UZ Leuven KU Leuven Belgium International Course on Theranostics and Molecular
Updates in Prostate Cancer Treatment 2018
 Updates in Prostate Cancer Treatment 2018 Mountain States Cancer Conference Elaine T. Lam, MD November 3, 2018 Learning Objectives Understand the difference between hormone sensitive and castration resistant
Updates in Prostate Cancer Treatment 2018 Mountain States Cancer Conference Elaine T. Lam, MD November 3, 2018 Learning Objectives Understand the difference between hormone sensitive and castration resistant
Chemohormonal Therapy For Prostate Cancer. What is old, is new again!
 Chemohormonal Therapy For Prostate Cancer What is old, is new again! Mount Tremblant January 20, 2017 Kala S. Sridhar MD, MSc, FRCPC Medical Oncologist, Princess Margaret Hospital Head, GU Medical Oncology
Chemohormonal Therapy For Prostate Cancer What is old, is new again! Mount Tremblant January 20, 2017 Kala S. Sridhar MD, MSc, FRCPC Medical Oncologist, Princess Margaret Hospital Head, GU Medical Oncology
Optimizing Outcomes in Advanced Prostate Cancer
 Optimizing Outcomes in Advanced Prostate Cancer Module 3: Focus on Recent CRPC Guidelines and Advanced Hormone-Sensitive Disease Sébastien J. Hotte, MD, MSc (HRM), FRCPC Medical Oncologist and Head, Phase
Optimizing Outcomes in Advanced Prostate Cancer Module 3: Focus on Recent CRPC Guidelines and Advanced Hormone-Sensitive Disease Sébastien J. Hotte, MD, MSc (HRM), FRCPC Medical Oncologist and Head, Phase
Michiel H.F. Poorthuis*, Robin W.M. Vernooij*, R. Jeroen A. van Moorselaar and Theo M. de Reijke
 First-line non-cytotoxic therapy in chemotherapynaive patients with metastatic castration-resistant prostate cancer: a systematic review of 10 randomised clinical trials Michiel H.F. Poorthuis*, Robin
First-line non-cytotoxic therapy in chemotherapynaive patients with metastatic castration-resistant prostate cancer: a systematic review of 10 randomised clinical trials Michiel H.F. Poorthuis*, Robin
Please consider the following information on ZYTIGA (abiraterone acetate). ZYTIGA - Compendia Communication - NCCN LATITUDE and STAMPEDE June 2017
 Page 1 of 2 Janssen Scientific Affairs, LLC 1125 Trenton-Harbourton Road PO Box 200 Titusville, NJ 08560 800.526.7736 tel 609.730.3138 fax June 08, 2017 Joan McClure 275 Commerce Drive #300 Fort Washington,
Page 1 of 2 Janssen Scientific Affairs, LLC 1125 Trenton-Harbourton Road PO Box 200 Titusville, NJ 08560 800.526.7736 tel 609.730.3138 fax June 08, 2017 Joan McClure 275 Commerce Drive #300 Fort Washington,
SOGUG meeting New drugs after docetaxel chemotherapy in patient with mcrpc
 SOGUG meeting New drugs after docetaxel chemotherapy in patient with mcrpc Stéphane OUDARD, MD, PhD Head of the Oncology department Georges Pompidou Hospital, Paris France University Rene Descartes, Paris
SOGUG meeting New drugs after docetaxel chemotherapy in patient with mcrpc Stéphane OUDARD, MD, PhD Head of the Oncology department Georges Pompidou Hospital, Paris France University Rene Descartes, Paris
Modern Screening and Treatment of Advanced Prostate Cancer John Tuckey
 Modern Screening and Treatment of Advanced Prostate Cancer John Tuckey Commonest male cancer - 2939 per year Third male cancer death 670 per year More die with it than of it but More people die of prostate
Modern Screening and Treatment of Advanced Prostate Cancer John Tuckey Commonest male cancer - 2939 per year Third male cancer death 670 per year More die with it than of it but More people die of prostate
Navigating Prostate Cancer Therapy. Nevin Murray MD Clinical Professor of Medicine, UBC Medical Oncologist, BCCA
 Navigating Prostate Cancer Therapy Nevin Murray MD Clinical Professor of Medicine, UBC Medical Oncologist, BCCA Disclosures In compliance with accreditation, we require the following disclosures to the
Navigating Prostate Cancer Therapy Nevin Murray MD Clinical Professor of Medicine, UBC Medical Oncologist, BCCA Disclosures In compliance with accreditation, we require the following disclosures to the
METASTATIC PROSTATE CANCER MANAGEMENT K I R U B E L T E F E R A M. D. T R I H E A LT H C A N C E R I N S T I T U T E 0 1 / 3 1 /
 METASTATIC PROSTATE CANCER MANAGEMENT K I R U B E L T E F E R A M. D. T R I H E A LT H C A N C E R I N S T I T U T E 0 1 / 3 1 / 2 0 1 8 Prostate Cancer- Statistics Most common cancer in men after a skin
METASTATIC PROSTATE CANCER MANAGEMENT K I R U B E L T E F E R A M. D. T R I H E A LT H C A N C E R I N S T I T U T E 0 1 / 3 1 / 2 0 1 8 Prostate Cancer- Statistics Most common cancer in men after a skin
Have we optimized the use of Androgen Receptor pathway targeted drugs in Castrate-Resistant Prostate Cancer?
 Have we optimized the use of Androgen Receptor pathway targeted drugs in Castrate-Resistant Prostate Cancer? Karim Fizazi, MD, PhD Institut Gustave Roussy Villejuif, France Disclosure Participation to
Have we optimized the use of Androgen Receptor pathway targeted drugs in Castrate-Resistant Prostate Cancer? Karim Fizazi, MD, PhD Institut Gustave Roussy Villejuif, France Disclosure Participation to
Cancer de la prostate métastatique: prise en charge précoce
 Cancer de la prostate métastatique: prise en charge précoce Stéphane Oudard, MD, PhD Georges Pompidou Hospital, Oncology Department, Paris, France stephane.oudard@egp.aphp.fr SAGB.CAB.14.08.0382c 3/02/2016
Cancer de la prostate métastatique: prise en charge précoce Stéphane Oudard, MD, PhD Georges Pompidou Hospital, Oncology Department, Paris, France stephane.oudard@egp.aphp.fr SAGB.CAB.14.08.0382c 3/02/2016
- La Terapia Farmacologica -
 XXV Congresso Nazionale AIRO Simposio AIRO-AIMN: Trattamento delle Metastasi Ossee nel Paziente con Tumore della Prostata "Ormonorefrattario": - La Terapia Farmacologica - Sergio Bracarda, Medical Oncology
XXV Congresso Nazionale AIRO Simposio AIRO-AIMN: Trattamento delle Metastasi Ossee nel Paziente con Tumore della Prostata "Ormonorefrattario": - La Terapia Farmacologica - Sergio Bracarda, Medical Oncology
January Abiraterone pre-docetaxel for patients with asymptomatic or minimally symptomatic metastatic castration resistant prostate cancer
 LONDON CANCER NEW DRUGS GROUP RAPID REVIEW Abiraterone pre-docetaxel for asymptomatic/minimally symptomatic metastatic castration resistant prostate cancer Abiraterone pre-docetaxel for patients with asymptomatic
LONDON CANCER NEW DRUGS GROUP RAPID REVIEW Abiraterone pre-docetaxel for asymptomatic/minimally symptomatic metastatic castration resistant prostate cancer Abiraterone pre-docetaxel for patients with asymptomatic
Elderly men with prostate cancer + ADT
 Elderly men with prostate cancer + ADT Background and Rationale ADT and Osteoporosis Proportion of Patients With Fractures 1-5 Yrs After Cancer Diagnosis 21 18 +6.8%; P
Elderly men with prostate cancer + ADT Background and Rationale ADT and Osteoporosis Proportion of Patients With Fractures 1-5 Yrs After Cancer Diagnosis 21 18 +6.8%; P
Radiopharmaceuticals for treating CRPC patients with metastatic bone disease 2014/6/27
 Radiopharmaceuticals for treating CRPC patients with metastatic bone disease 和信醫院黃玉儀 2014/6/27 Bone metastases in prostate cancer The most common site of metastasis in prostate cancer In >90% patients
Radiopharmaceuticals for treating CRPC patients with metastatic bone disease 和信醫院黃玉儀 2014/6/27 Bone metastases in prostate cancer The most common site of metastasis in prostate cancer In >90% patients
Session 4 Chemotherapy for castration refractory prostate cancer First and second- line chemotherapy
 Session 4 Chemotherapy for castration refractory prostate cancer First and second- line chemotherapy October- 2015 ESMO 2004 October- 2015 Fyraftensmøde 2 2010 October- 2015 Fyraftensmøde 3 SWOG 9916 OS
Session 4 Chemotherapy for castration refractory prostate cancer First and second- line chemotherapy October- 2015 ESMO 2004 October- 2015 Fyraftensmøde 2 2010 October- 2015 Fyraftensmøde 3 SWOG 9916 OS
Joelle Hamilton, M.D.
 Joelle Hamilton, M.D. www.urologycentersalabama.com Case Presentation: CRPC, Rising PSA 70 yo healthy, fit, active man post RALP 8 years prior with rising PSA Rising PSA from 0.02 nadir to 3.4 thus ADT
Joelle Hamilton, M.D. www.urologycentersalabama.com Case Presentation: CRPC, Rising PSA 70 yo healthy, fit, active man post RALP 8 years prior with rising PSA Rising PSA from 0.02 nadir to 3.4 thus ADT
Current role of chemotherapy in hormone-naïve patients Elena Castro
 Current role of chemotherapy in hormone-naïve patients Elena Castro Spanish National Cancer Research Centre Lugano, 17 October 2017 Siegel, Ca Cancer J Clin,2017 Buzzoni, Eur Urol, 2015 -Aprox 15-20% of
Current role of chemotherapy in hormone-naïve patients Elena Castro Spanish National Cancer Research Centre Lugano, 17 October 2017 Siegel, Ca Cancer J Clin,2017 Buzzoni, Eur Urol, 2015 -Aprox 15-20% of
Summary of Phase 3 IMPACT Trial Results Presented at AUA Meeting Webcast Conference Call April 28, Nasdaq: DNDN
 Summary of Phase 3 IMPACT Trial Results Presented at AUA Meeting Webcast Conference Call April 28, 2009 Nasdaq: DNDN PROVENGE sipuleucel-t is an autologous active cellular immunotherapy that activates
Summary of Phase 3 IMPACT Trial Results Presented at AUA Meeting Webcast Conference Call April 28, 2009 Nasdaq: DNDN PROVENGE sipuleucel-t is an autologous active cellular immunotherapy that activates
New Treatment Options for Prostate Cancer
 New Treatment Options for Prostate Cancer Moderator: Jeremy P. Goldberg, President, JPG Healthcare LLC Panelists: Philip Kantoff, MD, Director, Lank Center for Genitourinary Oncology, Dana- Farber Cancer
New Treatment Options for Prostate Cancer Moderator: Jeremy P. Goldberg, President, JPG Healthcare LLC Panelists: Philip Kantoff, MD, Director, Lank Center for Genitourinary Oncology, Dana- Farber Cancer
Advanced Prostate Cancer. SAMO Masterclass 17 th of March 2017 PD Dr. med. Aurelius Omlin
 Advanced Prostate Cancer SAMO Masterclass 17 th of March 2017 PD Dr. med. Aurelius Omlin aurelius.omlin@kssg.ch Conflicts of Interest Research Support: TEVA, Janssen Advisory Rolle: Astra Zeneca, Astellas,
Advanced Prostate Cancer SAMO Masterclass 17 th of March 2017 PD Dr. med. Aurelius Omlin aurelius.omlin@kssg.ch Conflicts of Interest Research Support: TEVA, Janssen Advisory Rolle: Astra Zeneca, Astellas,
Management of castrate resistant disease: after first line hormone therapy fails
 Management of castrate resistant disease: after first line hormone therapy fails Rob Jones Consultant in Medical Oncology Beatson Cancer Centre Glasgow Rhona McMenemin Consultant in Clinical Oncology The
Management of castrate resistant disease: after first line hormone therapy fails Rob Jones Consultant in Medical Oncology Beatson Cancer Centre Glasgow Rhona McMenemin Consultant in Clinical Oncology The
Bone-targeted therapies for prostate cancer in Institut Gustave Roussy Villejuif, France
 Bone-targeted therapies for prostate cancer in 2012 Pr Karim Fizazi, MD, PhD Institut Gustave Roussy Villejuif, France Disclosure Participation to advisory boards or speaker for: Amgen, Astellas-Medivation,
Bone-targeted therapies for prostate cancer in 2012 Pr Karim Fizazi, MD, PhD Institut Gustave Roussy Villejuif, France Disclosure Participation to advisory boards or speaker for: Amgen, Astellas-Medivation,
INTERGRATING NON- HORMONAL THERAPIES INTO PROSTATE CANCER
 INTERGRATING NON- HORMONAL THERAPIES INTO PROSTATE CANCER Daniel George, MD Professor of Medicine and Surgery Director of Genitourinary Oncology Program Duke Cancer Institute 1 Disclosures Consultant:
INTERGRATING NON- HORMONAL THERAPIES INTO PROSTATE CANCER Daniel George, MD Professor of Medicine and Surgery Director of Genitourinary Oncology Program Duke Cancer Institute 1 Disclosures Consultant:
When exogenous testosterone therapy is. adverse responses can be induced.
 Theoretical tips It has been reasoned that discontinuation of ADT in non orchiectomized patients may have detrimental effect on patients with CRPC as discontinuation of ADT can result in renewed release
Theoretical tips It has been reasoned that discontinuation of ADT in non orchiectomized patients may have detrimental effect on patients with CRPC as discontinuation of ADT can result in renewed release
ASCO 2011 Genitourinary Cancer
 ASCO 2011 Genitourinary Cancer Expanding Options for Chronic Diseases? Walter Stadler, MD, FACP University of Chicago Disclosures (All Non-University &/or Financial Dealings with Potential, Real, or Perceived
ASCO 2011 Genitourinary Cancer Expanding Options for Chronic Diseases? Walter Stadler, MD, FACP University of Chicago Disclosures (All Non-University &/or Financial Dealings with Potential, Real, or Perceived
Review of the Stampede Results. Charles Ryan MD University of California San Francisco
 Review of the Stampede Results Charles Ryan MD University of California San Francisco Se#ng and hypothesis Se
Review of the Stampede Results Charles Ryan MD University of California San Francisco Se#ng and hypothesis Se
Λουκάς Κοντοβίνης, παθολόγος - ογκολόγος.
 Λουκάς Κοντοβίνης, παθολόγος - ογκολόγος l.kontovinis@oncomedicare.com Nuclear Medicine Review 2011, 14, 2: 96 104 N Engl J Med 2004;350:1655-64 Nuclear Medicine Review 2011, 14, 2: 96 104 N Engl J Med
Λουκάς Κοντοβίνης, παθολόγος - ογκολόγος l.kontovinis@oncomedicare.com Nuclear Medicine Review 2011, 14, 2: 96 104 N Engl J Med 2004;350:1655-64 Nuclear Medicine Review 2011, 14, 2: 96 104 N Engl J Med
Advanced Prostate Cancer. Searching for Optimal Therapy Sequence and Assessing Emerging Treatment Options
 Advanced Prostate Cancer Searching for Optimal Therapy Sequence and Assessing Emerging Treatment Options Disclaimer This slide deck in its original and unaltered format is for educational purposes and
Advanced Prostate Cancer Searching for Optimal Therapy Sequence and Assessing Emerging Treatment Options Disclaimer This slide deck in its original and unaltered format is for educational purposes and
Hormonal Manipulations in CRPC. NW Clarke Professor of Urological Oncology Manchester UK
 Hormonal Manipulations in CRPC NW Clarke Professor of Urological Oncology Manchester UK Standard Treatment of CRPC Pre 2004 (and in 2013?) PSA progression 99m Tc BS negative CT scan large lymph node component
Hormonal Manipulations in CRPC NW Clarke Professor of Urological Oncology Manchester UK Standard Treatment of CRPC Pre 2004 (and in 2013?) PSA progression 99m Tc BS negative CT scan large lymph node component
The management and treatment options for secondary bone disease. Dr Jason Lester Clinical Oncologist Velindre Cancer Centre
 The management and treatment options for secondary bone disease Dr Jason Lester Clinical Oncologist Velindre Cancer Centre Aims Overview of bone metastases management in castrate-refractory prostate cancer
The management and treatment options for secondary bone disease Dr Jason Lester Clinical Oncologist Velindre Cancer Centre Aims Overview of bone metastases management in castrate-refractory prostate cancer
Convegno Nazionale AIOM Giovani 2016: News in Oncology. Daniele Alesini. Istituto Nazionale dei Tumori Regina Elena
 Convegno Nazionale AIOM Giovani 2016: News in Oncology Daniele Alesini Istituto Nazionale dei Tumori Regina Elena Something Old Something New Something Borrowed Something Blue DOCETAXEL: BACK AND FORTH
Convegno Nazionale AIOM Giovani 2016: News in Oncology Daniele Alesini Istituto Nazionale dei Tumori Regina Elena Something Old Something New Something Borrowed Something Blue DOCETAXEL: BACK AND FORTH
Hormone sensitive prostate cancer To add abiraterone or docetaxel? Dr Lisa Pickering
 > Hormone sensitive prostate cancer To add abiraterone or docetaxel? Dr Lisa Pickering Disclosures Institutional Research Support/P.I. Employee Consultant Major Stockholder Speakers Bureau Honoraria Scientific
> Hormone sensitive prostate cancer To add abiraterone or docetaxel? Dr Lisa Pickering Disclosures Institutional Research Support/P.I. Employee Consultant Major Stockholder Speakers Bureau Honoraria Scientific
Management of mcrpc: Hormonal therapy and treatment sequence for CRPC
 Management of mcrpc: Hormonal therapy and treatment sequence for CRPC Professor Bertrand Tombal, MD, PhD Cliniques universitaires Saint-Luc Université catholique de Louvain Brussels, Belgium Credentials
Management of mcrpc: Hormonal therapy and treatment sequence for CRPC Professor Bertrand Tombal, MD, PhD Cliniques universitaires Saint-Luc Université catholique de Louvain Brussels, Belgium Credentials
X, Y and Z of Prostate Cancer
 X, Y and Z of Prostate Cancer Dr Tony Michele Medical Oncologist Prostate cancer Epidemiology Current EUA (et al) guidelines on Advanced Prostate Cancer Current clinical management in specific scenarios
X, Y and Z of Prostate Cancer Dr Tony Michele Medical Oncologist Prostate cancer Epidemiology Current EUA (et al) guidelines on Advanced Prostate Cancer Current clinical management in specific scenarios
Initial Hormone Therapy
 Initial Hormone Therapy Alan Horwich Institute of Cancer Research and Royal Marsden Hospital, London, UK Alan.Horwich@icr.ac.uk MANAGEMENT OF PROSTATE CANCER Treatment windows Subclinical Localised PSA
Initial Hormone Therapy Alan Horwich Institute of Cancer Research and Royal Marsden Hospital, London, UK Alan.Horwich@icr.ac.uk MANAGEMENT OF PROSTATE CANCER Treatment windows Subclinical Localised PSA
Bone targeting: bisphosphonates, RANK-ligands and radioisotopes. Dr Lisa Pickering Consultant Medical Oncologist ESMO Preceptorship Singapore 2017
 Bone targeting: bisphosphonates, RANK-ligands and radioisotopes Dr Lisa Pickering Consultant Medical Oncologist ESMO Preceptorship Singapore 2017 Disclosures Institutional Research Support/P.I. Employee
Bone targeting: bisphosphonates, RANK-ligands and radioisotopes Dr Lisa Pickering Consultant Medical Oncologist ESMO Preceptorship Singapore 2017 Disclosures Institutional Research Support/P.I. Employee
pan-canadian Oncology Drug Review Final Clinical Guidance Report Abiraterone Acetate (Zytiga) for Metastatic Castration-Resistant Prostate Cancer
 pan-canadian Oncology Drug Review Final Clinical Guidance Report Abiraterone Acetate (Zytiga) for Metastatic Castration-Resistant Prostate Cancer October 22, 2013 DISCLAIMER Not a Substitute for Professional
pan-canadian Oncology Drug Review Final Clinical Guidance Report Abiraterone Acetate (Zytiga) for Metastatic Castration-Resistant Prostate Cancer October 22, 2013 DISCLAIMER Not a Substitute for Professional
Initial Hormone Therapy
 Initial Hormone Therapy Alan Horwich Institute of Cancer Research and Royal Marsden Hospital, London, UK Alan.Horwich@icr.ac.uk MANAGEMENT OF PROSTATE CANCER Treatment windows Subclinical Localised PSA
Initial Hormone Therapy Alan Horwich Institute of Cancer Research and Royal Marsden Hospital, London, UK Alan.Horwich@icr.ac.uk MANAGEMENT OF PROSTATE CANCER Treatment windows Subclinical Localised PSA
GU Guidelines Update Meeting: M0 Castrate Resistant Prostate Cancer. Dr. Simon Yu Nov 18, 2017
 GU Guidelines Update Meeting: M0 Castrate Resistant Prostate Cancer Dr. Simon Yu Nov 18, 2017 Faculty/Presenter Disclosure Faculty: Dr. Simon Yu Relationships with commercial interests: Grants/Research
GU Guidelines Update Meeting: M0 Castrate Resistant Prostate Cancer Dr. Simon Yu Nov 18, 2017 Faculty/Presenter Disclosure Faculty: Dr. Simon Yu Relationships with commercial interests: Grants/Research
SUMMARY. 3. Emerging understanding of mechanisms of resistance to current treatments
 SUMMARY 1. Discuss the active agents in prostate cancer currently available in Australia 2. Celebrate the growing role for Prostate Medical Oncologists in Multi Disc Teams active treaments overall survival
SUMMARY 1. Discuss the active agents in prostate cancer currently available in Australia 2. Celebrate the growing role for Prostate Medical Oncologists in Multi Disc Teams active treaments overall survival
PROSTATE CANCER HORMONE THERAPY AND BEYOND. Przemyslaw Twardowski MD Professor of Oncology Department of Urologic Oncology John Wayne Cancer Institute
 PROSTATE CANCER HORMONE THERAPY AND BEYOND Przemyslaw Twardowski MD Professor of Oncology Department of Urologic Oncology John Wayne Cancer Institute Disclosures I am a Consultant for Bayer and Sanofi-Aventis
PROSTATE CANCER HORMONE THERAPY AND BEYOND Przemyslaw Twardowski MD Professor of Oncology Department of Urologic Oncology John Wayne Cancer Institute Disclosures I am a Consultant for Bayer and Sanofi-Aventis
Prostate Cancer: Vision of the Future By: H.R.Jalalian
 1 H. R. Jalalian Hematologist&Oncologist Baqiyatallah University of Medical Sciences 2 State of the art: vision on the future Diagnosis Surgery Radiotherapy Medical Oncology 3 Early Detection PSA sensitivity
1 H. R. Jalalian Hematologist&Oncologist Baqiyatallah University of Medical Sciences 2 State of the art: vision on the future Diagnosis Surgery Radiotherapy Medical Oncology 3 Early Detection PSA sensitivity
American Urological Association (AUA) Guideline
 1 Approved by the AUA Board of Directors April 2014 Authors disclosure of potential conflicts of interest and author/staff contributions appear at the end of the article. 2014 by the American Urological
1 Approved by the AUA Board of Directors April 2014 Authors disclosure of potential conflicts of interest and author/staff contributions appear at the end of the article. 2014 by the American Urological
ESMO SUMMIT AFRICA Practice changing studies in Prostate Cancer in 2016 and 2017 and cost-effectiveness Ronald de Wit
 ESMO SUMMIT AFRICA 2018 Practice changing studies in Prostate Cancer in 2016 and 2017 and cost-effectiveness Ronald de Wit CONFLICT OF INTEREST DISCLOSURE Sub-title Sanofi Roche Merck Lilly 14 years of
ESMO SUMMIT AFRICA 2018 Practice changing studies in Prostate Cancer in 2016 and 2017 and cost-effectiveness Ronald de Wit CONFLICT OF INTEREST DISCLOSURE Sub-title Sanofi Roche Merck Lilly 14 years of
Timing of Androgen Deprivation: The Modern Debate Must be conducted in the following Contexts: 1. Clinical States Model
 Timing and Type of Androgen Deprivation Charles J. Ryan MD Associate Professor of Clinical Medicine UCSF Comprehensive Cancer Center Timing of Androgen Deprivation: The Modern Debate Must be conducted
Timing and Type of Androgen Deprivation Charles J. Ryan MD Associate Professor of Clinical Medicine UCSF Comprehensive Cancer Center Timing of Androgen Deprivation: The Modern Debate Must be conducted
Mapping the Complexity of Androgen Signaling In Prostate Cancer Progression Eleni Efstathiou MD PhD
 Mapping the Complexity of Androgen Signaling In Prostate Cancer Progression Eleni Efstathiou MD PhD The University of Athens Medical School Dept of Clinical Therapeutics Prostate Cancer Evolution Chemotherapy
Mapping the Complexity of Androgen Signaling In Prostate Cancer Progression Eleni Efstathiou MD PhD The University of Athens Medical School Dept of Clinical Therapeutics Prostate Cancer Evolution Chemotherapy
Radium-223 (Alpharadin)
 Radium-223 (Alpharadin) A Novel Targeted Alpha-Emitter for Bone-Metastatic Castrate-Resistant Prostate Cancer OLIVER SARTOR, M.D. North American Principal Investigator, ALSYMPCA trial Laborde Professor
Radium-223 (Alpharadin) A Novel Targeted Alpha-Emitter for Bone-Metastatic Castrate-Resistant Prostate Cancer OLIVER SARTOR, M.D. North American Principal Investigator, ALSYMPCA trial Laborde Professor
Evolving Paradigms in HER2+ MBC: Strategies for Individualizing Therapy with Available Agents
 Evolving Paradigms in HER2+ MBC: Strategies for Individualizing Therapy with Available Agents Kimberly L. Blackwell MD Professor Department of Medicine and Radiation Oncology Duke University Medical Center
Evolving Paradigms in HER2+ MBC: Strategies for Individualizing Therapy with Available Agents Kimberly L. Blackwell MD Professor Department of Medicine and Radiation Oncology Duke University Medical Center
August 2012 Volume 10, Issue 8, Supplement 12
 August 2012 Volume 10, Issue 8, Supplement 12 A SPECIAL MEETING REVIEW EDITION Highlights in Advanced Prostate Cancer From the 2012 American Urological Association Annual Meeting and the 2012 American
August 2012 Volume 10, Issue 8, Supplement 12 A SPECIAL MEETING REVIEW EDITION Highlights in Advanced Prostate Cancer From the 2012 American Urological Association Annual Meeting and the 2012 American
Metastatic prostate carcinoma. Lee Say Bob July 2017
 Metastatic prostate carcinoma Lee Say Bob July 2017 Scenario A 58 year old gentleman presents with PSA 200 ng/ml with hard prostate and bone mets. LUTS but upper tracts are normal with normal RP. history
Metastatic prostate carcinoma Lee Say Bob July 2017 Scenario A 58 year old gentleman presents with PSA 200 ng/ml with hard prostate and bone mets. LUTS but upper tracts are normal with normal RP. history
American Urological Association (AUA) Guideline
 1 Approved by the AUA Board of Directors May 2018 Authors disclosure of potential conflicts of interest and author/staff contributions appear at the end of the article. 2018 by the American Urological
1 Approved by the AUA Board of Directors May 2018 Authors disclosure of potential conflicts of interest and author/staff contributions appear at the end of the article. 2018 by the American Urological
Edith A. Perez, Ahmad Awada, Joyce O Shaughnessy, Hope Rugo, Chris Twelves, Seock-Ah Im, Carol Zhao, Ute Hoch, Alison L. Hannah, Javier Cortes
 BEACON: A Phase 3 Open-label, Randomized, Multicenter Study of Etirinotecan Pegol (EP) versus Treatment of Physician s Choice (TPC) in Patients With Locally Recurrent or Metastatic Breast Cancer Previously
BEACON: A Phase 3 Open-label, Randomized, Multicenter Study of Etirinotecan Pegol (EP) versus Treatment of Physician s Choice (TPC) in Patients With Locally Recurrent or Metastatic Breast Cancer Previously
Published on The YODA Project (
 Principal Investigator First Name: David Last Name: Lorente Degree: MD Primary Affiliation: Medical Oncology Service, Hospital Provincial de Castellón E-mail: lorente.davest@gmail.com Phone number: +34
Principal Investigator First Name: David Last Name: Lorente Degree: MD Primary Affiliation: Medical Oncology Service, Hospital Provincial de Castellón E-mail: lorente.davest@gmail.com Phone number: +34
PSMA Targeted radionuclide therapy in Prostate Cancer
 PSMA Targeted radionuclide therapy in Prostate Cancer Tawatchai Chaiwatanarat Division of Nuclear Medicine Department of Radiology Chulalongkorn University Bangkok, Thailand * cell si ze ~20 µm Radionuclide
PSMA Targeted radionuclide therapy in Prostate Cancer Tawatchai Chaiwatanarat Division of Nuclear Medicine Department of Radiology Chulalongkorn University Bangkok, Thailand * cell si ze ~20 µm Radionuclide
Oligometastasis. Körperstereotaxie bei oligo-metastasiertem Prostatakarzinom wann und wie in Kombination mit Systemtherapie?
 Körperstereotaxie bei oligo-metastasiertem Prostatakarzinom wann und wie in Kombination mit Systemtherapie? Daniel M. Aebersold 09. Dezember 2016 Oligometastasis JCO, 1995 1 Oligometastasis: Chance for
Körperstereotaxie bei oligo-metastasiertem Prostatakarzinom wann und wie in Kombination mit Systemtherapie? Daniel M. Aebersold 09. Dezember 2016 Oligometastasis JCO, 1995 1 Oligometastasis: Chance for
TRIALs of CDK4/6 inhibitor in women with hormone-receptor-positive metastatic breast cancer
 TRIALs of CDK4/6 inhibitor in women with hormone-receptor-positive metastatic breast cancer Marta Bonotto Department of Oncology University Hospital of Udine TRIALs of CDK4/6 inhibitor in women with hormone-receptor-positive
TRIALs of CDK4/6 inhibitor in women with hormone-receptor-positive metastatic breast cancer Marta Bonotto Department of Oncology University Hospital of Udine TRIALs of CDK4/6 inhibitor in women with hormone-receptor-positive
