Neuropathic pain, caused by a lesion or disease affecting the somatosensory
|
|
|
- Lambert Strickland
- 5 years ago
- Views:
Transcription
1 original article The antiallodynic action of pregabalin may depend on the suppression of spinal neuronal hyperexcitability in rats with spared nerve injury Lei Ding MD 1 *, Jie Cai MSc 2 *, Xiang-Yang Guo MD 1, Xiu-Li Meng MD 1, Guo-Gang Xing PhD 2 L Ding, J Cai, X-Y Guo, X-L Meng, G-G Xing. The antiallodynic action of pregabalin may depend on the suppression of spinal neuronal hyperexcitability in rats with spared nerve injury. Pain Res Manag 2014;19(4): BaCkGRounD: Pregabalin (PGB) is a novel antiepileptic drug and is also used as a first-line medication for the treatment of neuropathic pain. However, the mechanisms of its analgesic effects remain largely unknown. objectives: To elucidate the mechanisms underlying the antiallodynic action of PGB in rats with neuropathic pain. MeThoDs: In a rat model of neuropathic pain induced by spared nerve injury, mechanical allodynia, as a behavioural sign of neuropathic pain, was assessed by measuring 50% paw withdrawal threshold with von Frey filaments. Activities of dorsal horn wide dynamic range (WDR) neurons were examined by extracellular electrophysiological recording in vivo. ResuLTs: Spinal administration of PGB exerted a significant antiallodynic effect and a prominent inhibitory effect on the hypersensitivity of dorsal horn WDR neurons in rats with spared nerve injury. ConCLusion: The antiallodynic action of PGB is likely dependent on the suppression of WDR neuron hyperexcitability in rats with neuropathic pain. key Words: Electrophysiology; Neuropathic pain; Pregabalin; Spinal dorsal horn; WDR neurons Neuropathic pain, caused by a lesion or disease affecting the somatosensory system (1), is a common problem and may affect approximately 8% of the general population (2). Persistent allodynia, hyperalgesia and spontaneous pain are common characteristics of neuropathic pain, which can impair the quality of life of patients (3,4). The management of neuropathic pain is challenging. Conventional analgesics, such as nonsteroidal anti-inflammatory drugs and opioids, have limited efficacy and serious side effects in the treatment of this condition (5). Recently, the anticonvulsant pregabalin (PGB) and its close analogue gabapentin (GBP) have emerged as alternative treatments for neuropathic pain (3,6,7). With a superior pharmacokinetic profile to its analogue GBP (8), PGB has quickly become a first-line medication for the treatment of neuropathic pain (9). However, the mechanism of its analgesic action remains largely unknown. PGB (Lyrica; Pfizer, USA), also known as S-(+)-3-(aminomethyl)- 5-methylhexanoic acid or S(+)-3-isobutyl-gamma aminobutyric acid (isobutylgaba), is a potent ligand for the alpha-2-delta (α2δ) subunit of the voltage-gated calcium channels (10-12). Its potent binding L action antiallodynique de la prégabaline peut dépendre de la suppression de l hyperexcitabilité des neurones spinaux chez des rats ayant une lésion avec épargne nerveuse historique : La prégabaline (PGB) est un nouvel antiépileptique, qui est également utilisé comme médicament de première ligne de la douleur neuropathique. Cependant, on ne sait pas grand-chose des mécanismes de ses effets analgésiques. objectifs : Déterminer les mécanismes sous-tendant l action antiallodynique de la PGB chez des rats ayant une douleur neuropathique. MÉThoDoLoGie : Chez un modèle de rat ayant une douleur neuropathique induite par une lésion avec épargne nerveuse, les chercheurs ont évalué l allodynie mécanique comme signe comportemental de douleur neuropathique en mesurant le seuil de retrait de la patte à 50 % à l application des filaments de von Frey. Ils ont examiné les activités des neurones de la corne dorsale à la plage dynamique étendue (WDR) au moyen d un enregistrement électrophysiologique extracellulaire in vivo. RÉsuLTaTs : L administration spinale de PGB exerçait un effet antiallodynamique important et un effet inhibiteur significatif sur l hypersensibilité des neurones de la corne dorsale à WDR de rats ayant une lésion avec épargne nerveuse. ConCLusion : L action antiallodynique de la PGB dépend probablement de la suppression de l hyperexcitabilité des neurones à WDR chez des rats neuropathiques. at this site attenuates calcium influx to presynaptic nerve endings (13), reducing the release of several neurotransmitters involved in nociceptive transmission, such as glutamate (14-16), norepinephrine (17), substance P and calcitonin gene-related peptide (18), thereby attenuating spinal neuronal hyperexcitability (19). Accordingly, PGB exhibits potent analgesic activity in a range of animal models (7,14,20-24), as well as in preclinical and clinical observations (25-31). Although the α2δ subunit of the presynaptic calcium channels may be the primary target for mediating the analgesic actions of PGB (11-13,32), additional mechanisms underlying the analgesic actions of PGB may also be involved. Recent animal studies suggest that PGB may impair the development or maintenance of spinal central sensitization underlying the hyperalgesic state (19,33). PGB suppresses spinal neuronal hyperexcitability and visceral hypersensitivity in the absence of peripheral pathophysiology in a rat model of opioid-induced hyperalgesia (19). Based on this background, we hypothesized that the antiallodynic action of PGB depends on the suppression of spinal neuronal hyperexcitability in rats with *These authors contributed equally to this work 1 Department of Anesthesiology, Peking University Third Hospital; 2 Neuroscience Research Institute and Department of Neurobiology, Peking University; Key Laboratory for Neuroscience of the Ministry of Education and the Ministry of Public Health, Beijing, People s Republic of China Correspondence: Dr Guo-Gang Xing, Neuroscience Research Institute, Peking University, 38 Xue-Yuan Road, Beijing , People s Republic of China. Telephone , fax , ggxing@bjmu.edu.cn; Dr Xiu-Li Meng, Department of Anesthesiology, Peking University Third Hospital, Beijing , People s Republic of China. meng_xiuli@163.com Pain Res Manag Vol 19 No 4 July/August Pulsus Group Inc. All rights reserved 205
2 Ding et al neuropathic pain. The antiallodynic effects of PGB and the effects of PGB on hypersensitivity of dorsal horn wide dynamic range (WDR) neurons in the spinal cord were, therefore, examined in a rat model of neuropathic pain induced by spared nerve injury (SNI). We demonstrate that spinal administration of PGB significantly alleviates the mechanical allodynia and inhibits the hypersensitivity of dorsal horn WDR neurons in SNI rats. MeThoDs animals Male Sprague-Dawley rats, weighing 180 g to 200 g at the beginning of the experiment, were provided by the Department of Experimental Animal Sciences, Peking University Health Science Center (Beijing, China). The rats were housed in separated cages with free access to food and water. The room temperature was maintained at 24±1 C under a natural light-dark cycle. All animal experimental procedures were conducted in accordance with the guidelines of the International Association for the Study of Pain (34) and were approved by the Animal Care and Use Committee of Peking University. sni Under general anesthesia with chloral hydrate (0.3 g/kg administered intraperitoneally [ip]), the SNI procedure comprised an axotomy and ligation of the left tibial and common peroneal nerves, leaving the sural nerve intact. The common peroneal and the tibial nerves were tightly ligated with 4-0 silk sutures and sectioned distal to the ligation, removing 2 mm to 4 mm of the distal nerve stump. Considerable care was taken to avoid contact with or stretching of the intact sural nerve, as previously described by Decosterd and Woolf (35). In control animals, sham surgery involved exposure of the sciatic nerve and its branches without generating any lesions. Ten days after surgery, behavioural testing, immunohistochemical staining or electrophysiological recording were performed. Any rats exhibiting motor deficiency or lack of tactile allodynia after SNI surgery were excluded from the present study. Behavioural studies implantation of intrathecal catheter: Under chloral hydrate (0.3 g/kg ip) anesthesia, implantation of intrathecal cannula was performed following the method described in the authors previous studies (36,37). Briefly, a PE-10 polyethylene catheter was implanted between the L5 and L6 vertebrae to reach the lumber enlargement of the spinal cord. The outer section of the catheter was plugged and fixed to the skin on closure of the wound. All surgical procedures were performed under sterile conditions. Rats showing neurological deficits following the catheter implantation were euthanized. Drugs or vehicle were intrathecally injected via the implanted catheter in a 20 μl volume of solution followed by 10 μl of normal saline (NS) for flushing. Each injection lasted for at least 5 min. After an injection, the needle remained in situ for 2 min before being withdrawn. assessment of mechanical allodynia: Mechanical allodynia, as a behavioural sign of neuropathic pain, was assessed by measuring 50% paw withdrawal threshold (PWT) as described in the authors previous studies (38,39). The 50% PWT in response to a series of von Frey filaments (Stoelting, USA) was determined using the up-and-down method (40). Briefly, the rat was placed on a metal mesh floor covered with an inverted clear plastic cage (18 cm 8 cm 8 cm) and allowed a 20 min period for habituation. Eight von Frey filaments with approximately equal logarithmic incremental (0.224 g) bending forces were chosen (0.41 g, 0.70 g, 1.20 g, 2.00 g, 3.63 g, 5.50 g, 8.50 g and g). Each trial began with a von Frey force of 2.00 g delivered perpendicularly to the plantar surface of the left hind paw for approximately 2 s to 3 s. An abrupt withdrawal of the foot during stimulation or immediately after the removal of the filament was recorded as a positive response. Whenever there was a positive or negative response, the subsequent weaker or stronger filament was applied, respectively. This procedure was continued for six stimuli 206 after the first change in response had been observed. The 50% PWT was calculated using the following formula: 50% PWT = 10 (Xf+kδ) where X f is the value of the final von Frey filament used (in log units), k is a value measured from the pattern of positive/negative responses and δ=0.224, which is the average interval (in log units) between the von Frey filaments (41). If an animal responded to the lowest von Frey filament, a value of 0.25 g was assigned. If an animal did not respond to the highest von Frey filament, the value was recorded as 15.0 g. In rats, mechanical allodynia is assessed by measuring the 50% PWT in response to von Frey filaments, and the rat is considered to be allodynic when the 50% PWT is <4.0 g (ie, withdrawal in response to non-noxious tactile stimulus) (36). The antiallodynic effects of PGB were presented as 50% PWT in grams or as percentages of the maximum possible effect (MPE) following the formula described by Obata et al (42): MPE = (postdrug PWT value predrug PWT value) 100/(15 g cut-off value predrug PWT value) 100% PGB, at a dose of 100 μg in a 20 μl volume of solution, or an equal volume of vehicle (NS) was intrathecally administrated to rats on day 10 after SNI surgery (once per day for three days). The inclined-plate testing was performed before drug injection and on day 3 following drug injection, respectively, to assess the effect of PGB on motor function. assessment of locomotor function: Inclined-plate testing was used for the assessment of locomotor function. The rat was placed crosswise to the long axis of an inclined plate. The initial angle of the inclined plate was 50. The angle was then adjusted in 5 increments. The maximum angle of the plate on which the rat maintained its body position for 5 s without falling was determined, according to the method reported by Rivlin and Tator (43). electrophysiological studies surgery: Rats were initially anesthetized using urethane (1.2 g/kg to 1.5 g/kg ip). The trachea was cannulated to allow mechanical ventilation with room air. A catheter was inserted into the right jugular vein for continuous infusion of Tyrode s solution (NaCl 137 mmol/l, KCl 2.7 mmol/l, CaCl mmol/l, MgCl mmol/l, NaHCO mmol/l, NaH 2 PO mmol/l, D-[+]-glucose 6.5 mmol/l; ph 7.4) at a rate of 1.0 ml/h to 1.5 ml/h. The rectal temperature was maintained at 36.5 C to 37.5 C via a feedback-controlled heating pad placed under the body. A pair of bipolar silver hook electrodes was placed under the sciatic nerve immediately proximal to the trifurcation for electrical stimulation. The vertebral column was rigidly fixed in the frame with two clamps. The lumbar enlargement of the spinal cord was exposed by laminectomy at the vertebrae T13 and L1, and the dura covering lumbosacral spinal segments was carefully removed. A small well was built with 3% agar on the dorsal spinal cord at the recording segment to allow application of drugs or vehicles (39,44). The exposed spinal tissue was covered with warm (37 C) saline solution. After surgery, the animal was artificially ventilated with a small animal ventilator and continuous anesthesia was maintained with urethane (0.10 g/kg/h to 0.15 g/kg/h) during the entire experiment. The depth of anesthesia was monitored by examination of pupillary size and reflexes. The physiological condition of the animal was monitored by recording the electrocardiogram (330 beats/min to 460 beats/min), end-expiratory CO 2 (3.5% to 4.5%) and rectal temperature (36.5 C to 37.5 C), and was maintained within the ranges indicated (39). extracellular recording: Single-unit extracellular recordings were made from the lumbar dorsal horn neurons within 1200 μm of the dorsal surface of the spinal cord with 2 MΩ to 5 MΩ parylene-coated tungsten microelectrodes (Friedrick Haer & Co, USA) because the WDR neurons are located in the deep dorsal horn, mainly in laminae IV and V of the dorsal horn in the spinal cord. The Pain Res Manag Vol 19 No 4 July/August 2014
3 Antiallodynic action of PGB via WDR neurons microelectrode was inserted perpendicularly into the dorsal horn from a point approximately midway between the midline and the medial edge of the dorsal root entry zone. During electrode advancement, electrical pulses (0.5 Hz, 0.3 ms pulse width, 0.4 ma) were applied to the ipsilateral sciatic nerve as search stimuli to identify a neuron with no spontaneous firing. Once a single unit was identified, the receptive field and response characteristics were determined using a range of mechanical stimuli of varying intensities, including brushing or touching the skin with a cotton brush, light pressure with a probe and pinching a fold of skin with toothed forceps. A neuron responding to innocuous tactile stimuli, light pressure and noxious pinch in a graded manner was identified as a WDR neuron and was selected for further investigation (44). The recorded signals were amplified using an alternating current preamplifier, filtered with a passing bandwidth of 500 Hz to 1000 Hz, displayed on an oscilloscope and fed to a Pentium computer via a CED 1401 interface for offline analysis using the Spike2 software (Cambridge Electronic Design, United Kingdom). Spikes appearing 45 ms to 300 ms after stimulus were defined as C-fibre responses (ie, responses in the WDR neurons evoked by C-fibre activation) (44). Single-cell recording was ensured on the basis of amplitude and shape of the action potentials. In the following electrophysiological studies, only one cell was studied in each animal, and each animal received PGB or vehicle only once. Measurement of drug effects: The electrophysiological experiment was designed to investigate effects of spinal administration of PGB on the activities of WDR neurons in SNI rats. In this experiment, a train of 10 stimuli (0.5 Hz, 0.5 ms pulse width, with a pulse current of 2 C-fibre response threshold), which was used as test stimulus, was applied repeatedly to the sciatic nerve at 5 min intervals, and poststimulus histograms from the responses of WDR neurons were generated using the Spike2 software. After six stable control responses were recorded, PGB (at a dose of 100 μg in a 20 μl volume of solution) or an equal volume of vehicle was applied topically to the dorsal surface of the spinal cord, and the postdrug responses evoked by the same test stimulus as above were measured at 5 min intervals for up to 120 min. In the present study, only C-fibre responses of WDR neurons, which are usually related to nociception (44,45), were examined and analyzed. All of the C-fibre responses were expressed as percentages of the mean response value of six predrug consecutive trains of test stimuli. Chemical preparation and application PGB (S-[+]-3-[aminomethyl]-5-methylhexanoic acid) (Pfizer, USA), was dissolved in sterile 0.9% saline solution at a final concentration of 5 μg/μl and administered at 100 μg per application in behavioural or electrophysiological studies. statistical analysis Statistical analyses were performed using GraphPad Prism 5 (GraphPad Software, USA) for Windows (Microsoft Corporation, USA). All data were expressed as mean ± SEM. A two-tailed Student s t test was used to compare mean values between two groups. One-way ANOVA followed by the Dunnett multiple-comparison test or two-way ANOVA followed by the Bonferroni post hoc test were used for multiple comparison; P<0.05 was considered to be statistically significant. Area under the time-course curve (AUC) during the analysis time was used to assess the sum of the effects of PGB, as previously described (39,46). ResuLTs effects of intrathecal PGB on the mechanical allodynia and the locomotor function in sni rats As shown in Figures 1A and 1B, SNI that comprised an axotomy and ligation of the left tibial and common peroneal nerves produced a significant mechanical allodynia in rats, as assessed by 50% PWT in response to the mechanical stimulation with von Frey filaments. In contrast to naive and sham controls, the ipsilateral PWT in SNI rats decreased significantly from day 7 (14.62±0.1 g naive, 13.93±0.3 g sham, 3.69±0.2 g SNI; P<0.001, two-way ANOVA, n=10 per group) to day 14 (14.31±0.2 g naive, 13.90±0.3 g sham, 2.34±0.3 g SNI; P<0.001, two-way ANOVA, n=10 per group) after SNI surgery (Figure 1A). The AUC of PWT (zero to 14 days of the analysis time) was significantly decreased from ±1.0 (naive) and ±1.9 (sham) to 85.17±1.8 (SNI) (P<0.001, one-way ANOVA, n=10 per group, Figure 1B). Based on the rat model of neuropathic pain induced by SNI, the antiallodynic action of PGB in SNI rats with neuropathic pain was first confirmed. The reduction of the 50% PWT in SNI rats was found to be markedly reversed by intrathecal administration of PGB. As shown in Figure 1C, the decreased ipsilateral PWT in SNI rats was prominently restored to 13.47±0.9 g on day 1 after intrathecal application of PGB (ie, on day 11 after SNI), in contrast to NS application (3.30±0.4 g) (P<0.001, two-way ANOVA, n=10 per group). This reversed effect of PGB on PWT lasted for >3 days until experiment termination; eg, on day 3 after intrathecal application of PGB (ie, on day 13 after SNI), the ipsilateral PWT in SNI rats was maintained at 13.84±0.7 g in contrast to NS application (3.02±0.4 g) (P<0.001, two-way ANOVA, n=10 per group). The AUC of the PWT (10 to 13 days of the analysis time) was significantly different between the PGB (35.51±2.0) and NS (14.44±0.7) groups (P<0.001, two-tailed unpaired t test, n=10 per group, Figure 1D). Similarly, the MPE of PGB on PWT was significantly increased to 87.98±6.7% on day 1 after PGB administration (ie, on day 11 after SNI) compared with that in NS control (2.37±2.9%) (P<0.001, two-way ANOVA, n=10 per group). On day 3 after PGB administration (ie, on day 13 after SNI), the MPE of PGB on PWT was still maintained at 90.79±4.8% compared with that in the NS control group (3.88±1.6%) (P<0.001, two-way ANOVA, n=10 per group; Figure 1E). To exclude the possibility that the antiallodynic action of PGB was due to the impairment of rat motor function, the effect of PGB on rat locomotor function was examined using the inclinedplate test. As shown in Figure 1F, no obvious motor dysfunction was observed after spinal administration of PGB in rats with neuropathic pain (69.20±0.6 versus 69.13±0.5, pre-pgb versus post-pgb, P>0.05, two-tailed unpaired t test, n=10 per group). Collectively, these results suggest that spinal administration of PGB has a significant antiallodynic effect in rats with neuropathic pain. effects of spinal administration of PGB on C-fibre responses of dorsal horn WDR neurons in sni rats To further determine whether the antiallodynic action of PGB was dependent on the suppression of spinal neuronal hyperexcitability in rats with neuropathic pain, the C-fibre responses of dorsal horn WDR neurons that are usually related to nociception (44,45) in SNI rats were first examined. The C-fibre responses of WDR neurons increased significantly in SNI rats compared with those in sham rats (Figure 2). As shown in Figure 2C, the total spike count of the electrically evoked C-fibre responses in 10 stimuli was markedly increased in SNI rats in contrast to sham rats (P<0.001, two-way ANOVA, n=9 per group). Similarly, the AUC (0 min to 25 min) of the C-fibre response was also increased significantly in SNI rats (4525±626.1) compared with that in sham rats (1412±168.9) (P<0.001, SNI versus sham, two-tailed unpaired t test, n=9 per group, Figure 2D). Two representative examples illustrating the C-fibre responses of dorsal horn WDR neurons in SNI and sham rats are shown in Figure 2A and 2B. These results suggest that SNI produces hypersensitivity of dorsal horn WDR neurons that is highly related to the pathogenesis of neuropathic pain. Next, the effects of spinal administration of PGB on the C-fibre responses of dorsal horn WDR neurons was examined in SNI rats. As shown in Figure 3, PGB (at 100 μg) had a marked inhibitory effect on the hypersensitivity of dorsal horn WDR neurons in SNI rats. The inhibitory effect of PGB on C-fibre responses of WDR neurons began Pain Res Manag Vol 19 No 4 July/August
4 Ding et al Figure 1) Effects of intrathecal administration of pregabalin (PGB) on mechanical allodynia and locomotor function in a rat model of neuropathic pain induced by spared nerve injury (SNI). a and B Effects of SNI on the mechanical allodynia, as measured by 50% paw withdrawal threshold (PWT) (a) and area under the time-course curve (AUC) of PWT (B). Note that SNI produced a significant decrease in both 50% PWT and AUC of the 50% PWT, in contrast to naive and sham controls. ***P<0.001, ### P<0.001 compared with naive and sham, respectively; one-way ANOVA followed by the Dunnett multiple comparison test or two-way ANOVA followed by Bonferroni post hoc test; n=10 per group. C to e Effects of intrathecal administration of PGB on 50% PWT (C) and the AUC of PWT (D) as well as on the maximum possible effect (MPE) (e) in rats with neuropathic pain. PGB was administered intrathecally on day (d) 10 postsurgery at 100 μg/day, once per day for three days. The arrows indicate the injection of PGB or normal saline (NS) on day 10, day 11 and day 12 postsurgery. Note that PGB had a significant reversal effect on the decreased 50% PWT in SNI rats, in contrast to NS and control (C). ***P<0.001, ### P<0.001, compared with NS and predrug, respectively; two-way ANOVA followed by Bonferroni post hoc test; n=10 per group. Similarly, the AUC (10 to 13 day) of PGB on 50% PWT (D) and the MPE of PGB (e) was markedly increased, in contrast to NS control. ***P<0.001 compared with NS; one-way ANOVA followed by the Dunnett multiple comparison test or two-way ANOVA followed by Bonferroni post hoc test; n=10 per group. F Effects of PGB on the locomotor function of rats, measured by the angle of the inclined plate at which the rat begin to fall. Note that spinal administration of PGB had no significant impairment on the locomotor function of rats (P>0.05, two-tailed unpaired t test, predrug versus postdrug; n=10 per group) at 20 min post-pgb (70.71±7.4% of baseline), peaked at 65 min (49.47±8.8% of baseline) and lasted >120 min (59.43±11.6% of baseline) until experiment termination. Conversely, spinal administration of NS had no significant effect on the C-fibre responses of WDR neurons in SNI rats (P<0.05 to 0.001, two-way ANOVA, n=9 per group, Figure 3C). The AUC of the C-fibre responses ( 25 min to 120 min of the analysis time) was also decreased significantly in 208 Figure 2) Alterations of the C-fibre responses of dorsal horn wide dynamic range (WDR) neurons in spared nerve injury (SNI) rats. a and B Panels illustrate the poststimulus histogram of the electrically evoked neuronal responses in a dorsal horn WDR neuron in SNI and sham rats, respectively. The insets show the original recordings of the first electrically evoked neuronal responses. C The total spike count of the electrically evoked C-fibre responses in 10 stimuli. ***P<0.001 compared with sham control; twoway ANOVA followed by Bonferroni post hoc test; n=9 per group. D The area under the time-course curve (AUC) (0 min to 25 min) of the C-fibre response. ***P<0.001, SNI versus sham, two-tailed unpaired t test; n=9 per group the PGB group (10200±780.2) compared with the NS group (15670±570.6) (P<0.001, PGB versus NS, two-tailed unpaired t test, n=9 per group, Figure 3D). Two representative examples illustrating the C-fibre responses of dorsal horn WDR neurons before and after PGB or NS administration in SNI rats are shown in Figures 3A and 3B. These data suggest that the antiallodynic action of PGB is likely dependent on the suppression of spinal neuronal hyperexcitability in rats with neuropathic pain. DisCussion In the present study, we found that spinal administration of PGB had a significant antiallodynic effect in a rat model of neuropathic pain induced by SNI, which was associated with a marked inhibitory effect on the hypersensitivity of dorsal horn WDR neurons in SNI rats. These data indicate a novel mechanism underlying the antiallodynic action of PGB that is likely dependent on the suppression of WDR neuron hyperexcitability in neuropathic rats. Consistent with previous reports that PGB had a potent analgesic activity in rodent models of neuropathic pain (7,14,23), the present study provides evidence showing that intrathecal administration of PGB has a significant reversal effect on the reduction of ipsilateral PWT in response to von Frey filaments in SNI rats, confirming the antiallodynic effect of PGB on neuropathic pain. In the present study, PGB was intrathecally administrated on day 10 postsurgery when stable mechanical allodynia (represented as the reduction of PWT) emerged; therefore, the reversal effect of PGB on the SNI-induced reduction of PWT may be considered to represent the antiallodynic action of PGB on neuropathic pain. Together with the data obtained from the inclined-plate test, which showed that intrathecal injection of PGB had no significant impairment to the locomotor function in SNI rats, we suggest that PGB exerts its antiallodynic action in neuropathic rats without causing motor dysfunction. PGB is a lipophilic analogue of γ-aminobutyric acid (GABA), but it neither acts similar to GABA nor binds to GABA receptors (47,48). The α2δ subunit of the plasma membrane voltage-gated calcium channel is regarded as a major site of PGB action (10-12,49,50). Pain Res Manag Vol 19 No 4 July/August 2014
5 Antiallodynic action of PGB via WDR neurons Its potent binding at this site reduces calcium influx to presynaptic nerve endings (13), which reduces the release of several neurotransmitters involved in nociceptive transmission and will attenuate spinal neuronal hyperexcitability (19). To determine whether the antiallodynic action of PGB was dependent on the suppression of spinal neuronal hyperexcitability in rats with neuropathic pain, we first examined the C-fibre responses of dorsal horn WDR neurons in SNI rats. In the present study, we focused on WDR neurons, particularly C-fibre responses of WDR neurons, because they are polymodal neurons that receive multisynaptic input from A-beta, A-delta and C-fibre primary afferent nociceptors and respond to A-delta and C-fibre-mediated noxious thermal and mechanical stimuli, which are usually related to nociception (44,45). We found that the C-fibre responses of WDR neurons increased significantly in SNI rats, in contrast to sham rats, implying that SNI produces hypersensitivity of dorsal horn WDR neurons that is highly related to the pathogenesis of neuropathic pain (51-53). With regard to the action of PGB, we found that spinal administration of PGB had a marked inhibitory effect on the hypersensitivity of dorsal horn WDR neurons in SNI rats, suggesting that the antiallodynic action of PGB is likely dependent on the suppression of spinal neuronal hyperexcitability in rats with neuropathic pain. Supporting this notion, Wallin et al (33) found that PGB or GBP not only potentiated the antiallodynic effects of spinal cord stimulation (SCS) in rats that do not respond to SCS, but also enhanced the SCS-induced suppression of the hyperexcitability of dorsal horn WDR neurons in neuropathic rats. In morphine-treated rats, Bannister et al (19) showed that PGB attenuated the opioid-induced visceral hypersensitivity and reduced the hyperexcitability of WDR neurons to noxious mechanical and thermal stimuli. In rats with bee venom-induced inflammation, You et al (54) demonstrated that PGB selectively inhibited C-fibremediated, but not Aδ-fibre-mediated, spinal nociceptive-specific neuronal late responses including central neuronal plastic changes, ie, wind-up and after-discharges. In addition, in a rat model of craniofacial neuropathic pain, Cao et al (20) reported that PGB effectively attenuated the mechanical hypersensitivity and nociceptive neuronal hyperexcitability recorded in the medullary dorsal horn. Because we did not test responses of WDR neurons and the effects of PGB on responses to natural stimuli in the present study, we could not exclude the possibility that the mechanical allodynia is mediated by increased activity of WDR neurons evoked by low-threshold (A-beta) mechanoreceptors rather than C-fibre nociceptors. Substantial evidence has shown that the spinal neuronal hyperexcitability contributes to the central sensitization and the pain hypersensitivity in neuropathic rats (51,52,55-58); therefore, inhibition of the hyperexcitability of dorsal horn WDR neurons may account for the antiallodynic action of PGB for the treatment of neuropathic pain in rats. Indeed, emerging evidence suggests that PGB may impair the development or maintenance of spinal central sensitization underlying the hyperalgesic state (19,33), and suppress spinal neuronal hyperexcitability and visceral hypersensitivity in a rat model of opioidinduced hyperalgesia (19). ConCLusion The present study demonstrated that PGB significantly alleviates the mechanical allodynia and inhibits the hypersensitivity of dorsal horn WDR neurons in SNI rats. These data suggest that the antiallodynic ReFeRenCes 1. Jensen TS, Baron R, Haanpaa M, et al. A new definition of neuropathic pain. Pain 2011;152: Torrance N, Smith BH, Bennett MI, Lee AJ. The epidemiology of chronic pain of predominantly neuropathic origin. Results from a general population survey. J Pain 2006;7: Vranken JH. Elucidation of pathophysiology and treatment of neuropathic pain. Cent Nerv Syst Agents Med Chem 2012;12: Smith BH, Torrance N. Epidemiology of neuropathic pain and its impact on quality of life. Curr Pain Headache Rep 2012;16: Figure 3) Effects of spinal administration of pregabalin (PGB) on the C-fibre responses of dorsal horn wide dynamic range (WDR) neurons in spared nerve injury (SNI) rats. a and B Panels illustrate the poststimulus histogram of the electrically evoked neuronal responses in a dorsal horn WDR neuron before (a1, normal saline [NS]; b1, PGB) and after (a2, NS; b2, PGB) drug application. The insets show the original recordings of the first electrically evoked neuronal responses corresponding to each time point, respectively. C Analysis of the C-fibre-evoked discharges before and after drug (PGB or NS) application in SNI rats. Note that spinal administration of PGB (at 100 μg) had a significant inhibitory effect on the C-fibreevoked responses, in contrast to the NS control. *P<0.05; **P<0.01; ***P<0.001, compared with NS control; two-way ANOVA followed by Bonferroni post hoc test, n=7 to 9 per group). D The area under the timecourse curve (AUC) ( 25 min to 120 min) of the C-fibre response. ***P<0.001, PGB versus NS; two-tailed unpaired t test, n=7 to 9 per group action of PGB is likely dependent on the suppression of WDR neuron hyperexcitability in neuropathic rats. acknowledgements: The present work was supported by grants from the National Natural Science Foundation of China ( , , and ), and the 973 Program of the Ministry of Science and Technology of China (2013CB531905). 5. Hurley RW, Adams MC, Benzon HT. Neuropathic pain: Treatment guidelines and updates. Curr Opin Anaesthesiol 2013 August 29 (Epub ahead of print). 6. Blommel ML, Blommel AL. Pregabalin: An antiepileptic agent useful for neuropathic pain. Am J Health Syst Pharm 2007;64: Salat K, Librowski T, Nawiesniak B, Gluch-Lutwin M. Evaluation of analgesic, antioxidant, cytotoxic and metabolic effects of pregabalin for the use in neuropathic pain. Neurol Res 2013;35: Pain Res Manag Vol 19 No 4 July/August
6 Ding et al 8. Guay DR. Pregabalin in neuropathic pain: A more pharmaceutically elegant gabapentin? Am J Geriatr Pharmacother 2005;3: Brix FN, Hein SS, Staehelin JT. Management of painful neuropathies. Handb Clin Neurol 2013;115: Bian F, Li Z, Offord J, et al. Calcium channel alpha2-delta type 1 subunit is the major binding protein for pregabalin in neocortex, hippocampus, amygdala, and spinal cord: An ex vivo autoradiographic study in alpha2-delta type 1 genetically modified mice. Brain Res 2006;1075: Field MJ, Cox PJ, Stott E, et al. Identification of the alpha2-delta-1 subunit of voltage-dependent calcium channels as a molecular target for pain mediating the analgesic actions of pregabalin. Proc Natl Acad Sci U S A 2006;103: Taylor CP. Mechanisms of analgesia by gabapentin and pregabalin calcium channel alpha2-delta [Cavalpha2-delta] ligands. Pain 2009;142: Joshi I, Taylor CP. Pregabalin action at a model synapse: Binding to presynaptic calcium channel alpha2-delta subunit reduces neurotransmission in mice. Eur J Pharmacol 2006;553: Kumar N, Laferriere A, Yu JS, Leavitt A, Coderre TJ. Evidence that pregabalin reduces neuropathic pain by inhibiting the spinal release of glutamate. J Neurochem 2010;113: Kumar N, Cherkas PS, Varathan V, et al. Systemic pregabalin attenuates facial hypersensitivity and noxious stimulus-evoked release of glutamate in medullary dorsal horn in a rodent model of trigeminal neuropathic pain. Neurochem Int 2013;62: Quintero JE, Dooley DJ, Pomerleau F, Huettl P, Gerhardt GA. Amperometric measurement of glutamate release modulation by gabapentin and pregabalin in rat neocortical slices: Role of voltagesensitive Ca2+ alpha2delta-1 subunit. J Pharmacol Exp Ther 2011;338: Dooley DJ, Donovan CM, Pugsley TA. Stimulus-dependent modulation of [(3)H]norepinephrine release from rat neocortical slices by gabapentin and pregabalin. J Pharmacol Exp Ther 2000;295: Fehrenbacher JC, Taylor CP, Vasko MR. Pregabalin and gabapentin reduce release of substance P and CGRP from rat spinal tissues only after inflammation or activation of protein kinase C. Pain 2003;105: Bannister K, Sikandar S, Bauer CS, Dolphin AC, Porreca F, Dickenson AH. Pregabalin suppresses spinal neuronal hyperexcitability and visceral hypersensitivity in the absence of peripheral pathophysiology. Anesthesiology 2011;115: Cao Y, Wang H, Chiang CY, Dostrovsky JO, Sessle BJ. Pregabalin suppresses nociceptive behavior and central sensitization in a rat trigeminal neuropathic pain model. J Pain 2013;14: Gustafsson H, Sandin J. Oral pregabalin reverses cold allodynia in two distinct models of peripheral neuropathic pain. Eur J Pharmacol 2009;605: Han DW, Kweon TD, Lee JS, Lee YW. Antiallodynic effect of pregabalin in rat models of sympathetically maintained and sympathetic independent neuropathic pain. Yonsei Med J 2007;48: Park HJ, Joo HS, Chang HW, et al. Attenuation of neuropathyinduced allodynia following intraplantar injection of pregabalin. Can J Anaesth 2010;57: Tanabe M, Takasu K, Takeuchi Y, Ono H. Pain relief by gabapentin and pregabalin via supraspinal mechanisms after peripheral nerve injury. J Neurosci Res 2008;86: Baron R, Brunnmuller U, Brasser M, May M, Binder A. Efficacy and safety of pregabalin in patients with diabetic peripheral neuropathy or postherpetic neuralgia: Open-label, non-comparative, flexible-dose study. Eur J Pain 2008;12: Boyle J, Eriksson ME, Gribble L, et al. Randomized, placebocontrolled comparison of amitriptyline, duloxetine, and pregabalin in patients with chronic diabetic peripheral neuropathic pain: Impact on pain, polysomnographic sleep, daytime functioning, and quality of life. Diabetes Care 2012;35: Cardenas DD, Nieshoff EC, Suda K, et al. A randomized trial of pregabalin in patients with neuropathic pain due to spinal cord injury. Neurology 2013;80: Jenkins TM, Smart TS, Hackman F, Cooke C, Tan KK. Efficient assessment of efficacy in post-traumatic peripheral neuropathic pain patients: Pregabalin in a randomized, placebo-controlled, crossover study. J Pain Res 2012;5: Saldana MT, Perez C, Navarro A, Masramon X, Rejas J. Pain alleviation and patient-reported health outcomes following switching to pregabalin in individuals with gabapentin-refractory neuropathic pain in routine medical practice. Clin Drug Investig 2012;32: Stacey BR, Swift JN. Pregabalin for neuropathic pain based on recent clinical trials. Curr Pain Headache Rep 2006;10: Vinik A, Emir B, Cheung R, Whalen E. Relationship between pain relief and improvements in patient function/quality of life in patients with painful diabetic peripheral neuropathy or postherpetic neuralgia treated with pregabalin. Clin Ther 2013;35: Bauer CS, Rahman W, Tran-van-Minh A, Lujan R, Dickenson AH, Dolphin AC. The anti-allodynic alpha(2)delta ligand pregabalin inhibits the trafficking of the calcium channel alpha(2)delta-1 subunit to presynaptic terminals in vivo. Biochem Soc Trans 2010;38: Wallin J, Cui JG, Yakhnitsa V, Schechtmann G, Meyerson BA, Linderoth B. Gabapentin and pregabalin suppress tactile allodynia and potentiate spinal cord stimulation in a model of neuropathy. Eur J Pain 2002;6: Zimmermann M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 1983;16: Decosterd I, Woolf CJ. Spared nerve injury: An animal model of persistent peripheral neuropathic pain. Pain 2000;87: Liu M, Yang H, Fang D, et al. Upregulation of P2X3 receptors by neuronal calcium sensor protein VILIP-1 in dorsal root ganglions contributes to the bone cancer pain in rats. Pain 2013;154: Zheng Q, Fang D, Cai J, Wan Y, Han JS, Xing GG. Enhanced excitability of small dorsal root ganglion neurons in rats with bone cancer pain. Mol Pain 2012;8: Geng SJ, Liao FF, Dang WH, et al. Contribution of the spinal cord BDNF to the development of neuropathic pain by activation of the NR2B-containing NMDA receptors in rats with spinal nerve ligation. Exp Neurol 2010;222: Qu XX, Cai J, Li MJ, et al. Role of the spinal cord NR2Bcontaining NMDA receptors in the development of neuropathic pain. Exp Neurol 2009;215: Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL. Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 1994;53: Dixon WJ. Efficient analysis of experimental observations. Annu Rev Pharmacol Toxicol 1980;20: Obata H, Saito S, Sasaki M, Ishizaki K, Goto F. Antiallodynic effect of intrathecally administered 5-HT(2) agonists in rats with nerve ligation. Pain 2001;90: Rivlin AS, Tator CH. Objective clinical assessment of motor function after experimental spinal cord injury in the rat. J Neurosurg 1977;47: Liu FY, Qu XX, Ding X, et al. Decrease in the descending inhibitory 5-HT system in rats with spinal nerve ligation. Brain Res 2010;1330: Lu Y, Perl ER. Selective action of noradrenaline and serotonin on neurones of the spinal superficial dorsal horn in the rat. J Physiol 2007;582: Zheng Q, Fang D, Liu M, et al. Suppression of KCNQ/M (Kv7) potassium channels in dorsal root ganglion neurons contributes to the development of bone cancer pain in a rat model. Pain 2013;154: Errante LD, Petroff OA. Acute effects of gabapentin and pregabalin on rat forebrain cellular GABA, glutamate, and glutamine concentrations. Seizure 2003;12: Cheng JK, Lee SZ, Yang JR, et al. Does gabapentin act as an agonist at native GABA(B) receptors? J Biomed Sci 2004;11: Fink K, Dooley DJ, Meder WP, et al. Inhibition of neuronal Ca(2+) influx by gabapentin and pregabalin in the human neocortex. Neuropharmacology 2002;42: Taylor CP, Angelotti T, Fauman E. Pharmacology and mechanism of action of pregabalin: The calcium channel alpha2-delta (alpha2- delta) subunit as a target for antiepileptic drug discovery. Epilepsy Res 2007;73: Chu KL, Faltynek CR, Jarvis MF, McGaraughty S. Increased WDR spontaneous activity and receptive field size in rats following a neuropathic or inflammatory injury: Implications for mechanical sensitivity. Neurosci Lett 2004;372: Pain Res Manag Vol 19 No 4 July/August 2014
7 Antiallodynic action of PGB via WDR neurons 52. Sotgiu ML, Biella G. Contribution of central sensitization to the pain-related abnormal activity in neuropathic rats. Somatosens Mot Res 2000;17: Tabo E, Jinks SL, Eisele JH Jr, Carstens E. Behavioral manifestations of neuropathic pain and mechanical allodynia, and changes in spinal dorsal horn neurons, following L4-L6 dorsal root constriction in rats. Pain 1999;80: You HJ, Lei J, Arendt-Nielsen L. Selective inhibitory effects of pregabalin on peripheral C but not A-delta fibers mediated nociception in intact and spinalized rats. Neuroscience 2009;164: Latremoliere A, Woolf CJ. Central sensitization: A generator of pain hypersensitivity by central neural plasticity. J Pain 2009;10: Rygh LJ, Kontinen VK, Suzuki R, Dickenson AH. Different increase in C-fibre evoked responses after nociceptive conditioning stimulation in sham-operated and neuropathic rats. Neurosci Lett 2000;288: Sotgiu ML, Biella G. Differential effects of MK-801, a N-methyl-Daspartate non-competitive antagonist, on the dorsal horn neuron hyperactivity and hyperexcitability in neuropathic rats. Neurosci Lett 2000;283: Yakhnitsa V, Linderoth B, Meyerson BA. Spinal cord stimulation attenuates dorsal horn neuronal hyperexcitability in a rat model of mononeuropathy. Pain 1999;79: Pain Res Manag Vol 19 No 4 July/August
Pharmacology of Pain Transmission and Modulation
 Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA
 Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD
 Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Changes in long2term syna ptic plasticity in the spinal dorsal horn of neuropathic pa in rats
 226 ( ) JOURNAL OF PEKIN G UNIVERSITY( HEAL TH SCIENCES) Vol. 35 No. 3 J une 2003,,, (, 100083) [ ] / ; ; ; / [ ] :, (central sensitization) :Sprague2Dawley 5/ 6 (L5/ L6), C2, C2 L TP : (1), ( 100 Hz,
226 ( ) JOURNAL OF PEKIN G UNIVERSITY( HEAL TH SCIENCES) Vol. 35 No. 3 J une 2003,,, (, 100083) [ ] / ; ; ; / [ ] :, (central sensitization) :Sprague2Dawley 5/ 6 (L5/ L6), C2, C2 L TP : (1), ( 100 Hz,
Seizure: the clinical manifestation of an abnormal and excessive excitation and synchronization of a population of cortical
 Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page
 The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
Spinal Cord Injury Pain. Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018
 Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
NMDA-Receptor Antagonists and Opioid Receptor Interactions as Related to Analgesia and Tolerance
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Pain Pathways. Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH
 Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Evaluation of Gabapentin and S-( )-3-Isobutylgaba in a Rat Model of Postoperative Pain
 0022-3565/97/2823-1242$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 282, No. 3 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
0022-3565/97/2823-1242$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 282, No. 3 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor
 Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
What Does the Mechanism of Spinal Cord Stimulation Tell Us about Complex Regional Pain Syndrome?pme_
 Pain Medicine 2010; 11: 1278 1283 Wiley Periodicals, Inc. What Does the Mechanism of Spinal Cord Stimulation Tell Us about Complex Regional Pain Syndrome?pme_915 1278..1283 Joshua P. Prager, MD, MS Center
Pain Medicine 2010; 11: 1278 1283 Wiley Periodicals, Inc. What Does the Mechanism of Spinal Cord Stimulation Tell Us about Complex Regional Pain Syndrome?pme_915 1278..1283 Joshua P. Prager, MD, MS Center
Decrease in the descending inhibitory 5-HT system in rats with spinal nerve ligation
 BRAIN RESEARCH 1330 (2010) 45 60 available at www.sciencedirect.com www.elsevier.com/locate/brainres Research Report Decrease in the descending inhibitory 5-HT system in rats with spinal nerve ligation
BRAIN RESEARCH 1330 (2010) 45 60 available at www.sciencedirect.com www.elsevier.com/locate/brainres Research Report Decrease in the descending inhibitory 5-HT system in rats with spinal nerve ligation
1604 IEEE TRANSACTIONS ON BIOMEDICAL ENGINEERING, VOL. 62, NO. 6, JUNE 2015
 1604 IEEE TRANSACTIONS ON BIOMEDICAL ENGINEERING, VOL. 62, NO. 6, JUNE 2015 Burst and Tonic Spinal Cord Stimulation Differentially Activate GABAergic Mechanisms to Attenuate Pain in a Rat Model of Cervical
1604 IEEE TRANSACTIONS ON BIOMEDICAL ENGINEERING, VOL. 62, NO. 6, JUNE 2015 Burst and Tonic Spinal Cord Stimulation Differentially Activate GABAergic Mechanisms to Attenuate Pain in a Rat Model of Cervical
Special Issue on Pain and Itch
 Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
Receptors and Neurotransmitters: It Sounds Greek to Me. Agenda. What We Know About Pain 9/7/2012
 Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
 University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
San Francisco Chronicle, June 2001
 PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
Cancer-induced bone pain
 Cancer-induced bone pain Common Prevalent in particular cancers: breast (73%), prostate (68%), thyroid (42%), lung (36%), renal (35%), colon (5%) Correlates with an increased morbidity Reduced performance
Cancer-induced bone pain Common Prevalent in particular cancers: breast (73%), prostate (68%), thyroid (42%), lung (36%), renal (35%), colon (5%) Correlates with an increased morbidity Reduced performance
Supporting Information
 Supporting Information Synthesis and Evaluation of Antiallodynic and Anticonvulsant Activity of Novel Amide and Urea Derivatives of Valproic Acid Analogues Dan Kaufmann, Meir Bialer, $, Jakob Avi Shimshoni,
Supporting Information Synthesis and Evaluation of Antiallodynic and Anticonvulsant Activity of Novel Amide and Urea Derivatives of Valproic Acid Analogues Dan Kaufmann, Meir Bialer, $, Jakob Avi Shimshoni,
Refractory Central Neurogenic Pain in Spinal Cord Injury. Case Presentation
 Refractory Central Neurogenic Pain in Spinal Cord Injury Case Presentation Edwin B. George, MD, PhD Wayne State University John D. Dingell VAMC 2012 Disclosures This continuing education activity is managed
Refractory Central Neurogenic Pain in Spinal Cord Injury Case Presentation Edwin B. George, MD, PhD Wayne State University John D. Dingell VAMC 2012 Disclosures This continuing education activity is managed
Gabapentin Antinociception in Mice with Acute Herpetic Pain Induced by Herpes Simplex Virus Infection
 0022-3565/01/2962-270 275$3.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 296, No. 2 Copyright 2001 by The American Society for Pharmacology and Experimental Therapeutics 3229/880565
0022-3565/01/2962-270 275$3.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 296, No. 2 Copyright 2001 by The American Society for Pharmacology and Experimental Therapeutics 3229/880565
Hong Jiang 1, Dong Fang 1, Ling-Yu Kong 1, Zi-Run Jin 1, Jie Cai 1, Xue-Jing Kang 1, You Wan 2,3* and Guo-Gang Xing 1,2,3*
 Jiang et al. Molecular Brain 2014, 7:72 RESEARCH Open Access Sensitization of neurons in the central nucleus of the amygdala via the decreased GABAergic inhibition contributes to the development of neuropathic
Jiang et al. Molecular Brain 2014, 7:72 RESEARCH Open Access Sensitization of neurons in the central nucleus of the amygdala via the decreased GABAergic inhibition contributes to the development of neuropathic
Basolateral Amygdala Lesion Inhibits the Development of Pain Chronicity in Neuropathic Pain Rats
 Basolateral Amygdala Lesion Inhibits the Development of Pain Chronicity in Neuropathic Pain Rats Zheng Li 1,3., Jing Wang 2., Lin Chen 1,3, Meng Zhang 1,3, You Wan 1,3 * 1 Neuroscience Research Institute,
Basolateral Amygdala Lesion Inhibits the Development of Pain Chronicity in Neuropathic Pain Rats Zheng Li 1,3., Jing Wang 2., Lin Chen 1,3, Meng Zhang 1,3, You Wan 1,3 * 1 Neuroscience Research Institute,
Mechanical sensitization of cutaneous sensory fibers in the spared nerve injury mouse model
 Smith et al. Molecular Pain 2013, 9:61 MOLECULAR PAIN SHORT REPORT Open Access Mechanical sensitization of cutaneous sensory fibers in the spared nerve injury mouse model Amanda K Smith, Crystal L O Hara
Smith et al. Molecular Pain 2013, 9:61 MOLECULAR PAIN SHORT REPORT Open Access Mechanical sensitization of cutaneous sensory fibers in the spared nerve injury mouse model Amanda K Smith, Crystal L O Hara
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
211MDS Pain theories
 211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
Sensory Assessment of Regional Analgesia in Humans
 REVIEW ARTICLE Dennis M. Fisher, M.D., Editor-in-Chief Anesthesiology 2000; 93:1517 30 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Sensory Assessment of Regional
REVIEW ARTICLE Dennis M. Fisher, M.D., Editor-in-Chief Anesthesiology 2000; 93:1517 30 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Sensory Assessment of Regional
Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D.
 Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture the student should be familiar with: 1. The relationship between nociception
Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture the student should be familiar with: 1. The relationship between nociception
Virtually everyone has experienced pain in one
 Transfer of Advances in Sciences into Dental Education Recent Insights into Brainstem Mechanisms Underlying Craniofacial Pain Barry J. Sessle, B.D.S., M.D.S., B.Sc., Ph.D., F.R.S.C., D.Sc. (honorary) Abstract:
Transfer of Advances in Sciences into Dental Education Recent Insights into Brainstem Mechanisms Underlying Craniofacial Pain Barry J. Sessle, B.D.S., M.D.S., B.Sc., Ph.D., F.R.S.C., D.Sc. (honorary) Abstract:
Neuropathic Pain in Palliative Care
 Neuropathic Pain in Palliative Care Neuropathic Pain in Advanced Cancer Affects 40% of patients Multiple concurrent pains are common Often complex pathophysiology with mixed components Nocioceptive Neuropathic
Neuropathic Pain in Palliative Care Neuropathic Pain in Advanced Cancer Affects 40% of patients Multiple concurrent pains are common Often complex pathophysiology with mixed components Nocioceptive Neuropathic
Experimental Neurology
 Experimental Neurology 215 (2009) 298 307 Contents lists available at ScienceDirect Experimental Neurology journal homepage: www.elsevier.com/locate/yexnr Role of the spinal cord NR2B-containing NMDA receptors
Experimental Neurology 215 (2009) 298 307 Contents lists available at ScienceDirect Experimental Neurology journal homepage: www.elsevier.com/locate/yexnr Role of the spinal cord NR2B-containing NMDA receptors
Effects of Palonosetron, a 5-HT3 Receptor Antagonist, on Mechanical Allodynia in a Rat Model of Postoperative Pain
 Original Article Korean J Pain 2013 April; Vol. 26, No. 2: 125-129 pissn 2005-9159 eissn 2093-0569 http://dx.doi.org/10.3344/kjp.2013.26.2.125 Effects of Palonosetron, a 5-HT3 Receptor Antagonist, on Mechanical
Original Article Korean J Pain 2013 April; Vol. 26, No. 2: 125-129 pissn 2005-9159 eissn 2093-0569 http://dx.doi.org/10.3344/kjp.2013.26.2.125 Effects of Palonosetron, a 5-HT3 Receptor Antagonist, on Mechanical
Original Article Upregulation of neuregulin-1 reverses signs of neuropathic pain in rats
 Int J Clin Exp Pathol 2014;7(9):5916-5921 www.ijcep.com /ISSN:1936-2625/IJCEP0001263 Original Article Upregulation of neuregulin-1 reverses signs of neuropathic pain in rats Guojun Wang 1,2*, Dawei Dai
Int J Clin Exp Pathol 2014;7(9):5916-5921 www.ijcep.com /ISSN:1936-2625/IJCEP0001263 Original Article Upregulation of neuregulin-1 reverses signs of neuropathic pain in rats Guojun Wang 1,2*, Dawei Dai
SOMATOSENSORY SYSTEMS AND PAIN
 SOMATOSENSORY SYSTEMS AND PAIN A 21 year old man presented with a stab wound of the right side of the neck (Panel A). Neurological examination revealed right hemiplegia and complete right-sided loss of
SOMATOSENSORY SYSTEMS AND PAIN A 21 year old man presented with a stab wound of the right side of the neck (Panel A). Neurological examination revealed right hemiplegia and complete right-sided loss of
Pathophysiology of Pain
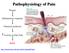 Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
TRANSCUTANEOUS ELECTRICAL STIMULATION
 TRANSCUTANEOUS ELECTRICAL STIMULATION Transcutaneous electrical stimulation (TENS) Transcutaneous electrical stimulation ; An electronic device that produces electrical signals used to stimulate nerve
TRANSCUTANEOUS ELECTRICAL STIMULATION Transcutaneous electrical stimulation (TENS) Transcutaneous electrical stimulation ; An electronic device that produces electrical signals used to stimulate nerve
Pain teaching. Muhammad Laklouk
 Pain teaching Muhammad Laklouk Definition Pain An unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage. Sensory (discriminatiory)
Pain teaching Muhammad Laklouk Definition Pain An unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage. Sensory (discriminatiory)
Supplementary information to. Mesenchymal Stem Cells Reversed Morphine Tolerance and Opioid-induced Hyperalgesia
 Supplementary information to Mesenchymal Stem Cells Reversed Morphine Tolerance and Opioid-induced Hyperalgesia Zhen Hua 1,2 *, LiPing Liu 1 *, Jun Shen 1, Katherine Cheng 1, Aijun Liu 1, Jing Yang 1,
Supplementary information to Mesenchymal Stem Cells Reversed Morphine Tolerance and Opioid-induced Hyperalgesia Zhen Hua 1,2 *, LiPing Liu 1 *, Jun Shen 1, Katherine Cheng 1, Aijun Liu 1, Jing Yang 1,
Pathophysiology of Pain. Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC
 Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
Potential for delta opioid receptor agonists as analgesics in chronic pain therapy
 Potential for delta opioid receptor agonists as analgesics in chronic pain therapy David Kendall & Bengt von Mentzer; PharmNovo AB/UK Alex Conibear & Eamonn Kelly, University of Bristol Junaid Asghar,
Potential for delta opioid receptor agonists as analgesics in chronic pain therapy David Kendall & Bengt von Mentzer; PharmNovo AB/UK Alex Conibear & Eamonn Kelly, University of Bristol Junaid Asghar,
ANAT2010. Concepts of Neuroanatomy (II) S2 2018
 ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
Dose-response relationship analysis of pregabalin doses and their antinociceptive effects in hot-plate test in mice
 Pharmacological Reports 2010, 62, 942 948 ISSN 1734-1140 Copyright 2010 by Institute of Pharmacology Polish Academy of Sciences Short communication Dose-response relationship analysis of pregabalin doses
Pharmacological Reports 2010, 62, 942 948 ISSN 1734-1140 Copyright 2010 by Institute of Pharmacology Polish Academy of Sciences Short communication Dose-response relationship analysis of pregabalin doses
A role for uninjured afferents in neuropathic pain
 Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus. MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive
 PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
The Effect of Intrathecal Gabapentin on Mechanical and Thermal Hyperalgesia in Neuropathic Rats Induced by Spinal Nerve Ligation
 J Korean Med Sci 2002; 17: 225-9 ISSN 1011-8934 Copyright The Korean Academy of Medical Sciences The Effect of Intrathecal Gabapentin on Mechanical and Thermal Hyperalgesia in Neuropathic Rats Induced
J Korean Med Sci 2002; 17: 225-9 ISSN 1011-8934 Copyright The Korean Academy of Medical Sciences The Effect of Intrathecal Gabapentin on Mechanical and Thermal Hyperalgesia in Neuropathic Rats Induced
S E C T I O N I M E C H A N I S M S A N D E P I D E M I O L O G Y
 SECTION I MECHANISMS AND EPIDEMIOLOGY 1 Nociception: basic principles rie suzuki, shafaq sikandar, and anthony h. dickenson University College London Introduction Pain has been a major concern in the clinic
SECTION I MECHANISMS AND EPIDEMIOLOGY 1 Nociception: basic principles rie suzuki, shafaq sikandar, and anthony h. dickenson University College London Introduction Pain has been a major concern in the clinic
CHAPTER 4 PAIN AND ITS MANAGEMENT
 CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
Analgesic effects of the COX-2 inhibitor parecoxib on surgical pain through suppression of spinal ERK signaling
 EXPERIMENTAL AND THERAPEUTIC MEDICINE 6: 275-279, 2013 Analgesic effects of the COX-2 inhibitor parecoxib on surgical pain through suppression of spinal ERK signaling YA-JING GUO 1, XU-DAN SHI 2, DI FU
EXPERIMENTAL AND THERAPEUTIC MEDICINE 6: 275-279, 2013 Analgesic effects of the COX-2 inhibitor parecoxib on surgical pain through suppression of spinal ERK signaling YA-JING GUO 1, XU-DAN SHI 2, DI FU
Update on the Neurophysiology of Pain Transmission and Modulation: Focus on the NMDA-Receptor
 S2 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Update on the Neurophysiology of Pain Transmission
S2 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Update on the Neurophysiology of Pain Transmission
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007)
 Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
International Conference on Biological Sciences and Technology (BST 2016)
 International Conference on Biological Sciences and Technology (BST 2016) Analgesic Contrast of Diverse Administrations of Botulinum Neurotoxin A (BoTN/A) Using Rat Neuropathic Pain Model Qiong-Fang YANG1,3,
International Conference on Biological Sciences and Technology (BST 2016) Analgesic Contrast of Diverse Administrations of Botulinum Neurotoxin A (BoTN/A) Using Rat Neuropathic Pain Model Qiong-Fang YANG1,3,
What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle.
 What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle. Nociception The detection of tissue damage or impending tissue damage, but There can be tissue damage without
What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle. Nociception The detection of tissue damage or impending tissue damage, but There can be tissue damage without
Materials and Methods
 Anesthesiology 2004; 100:1258 62 2004 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Spinal Adenosine Receptor Activation Reduces Hypersensitivity after Surgery by a Different
Anesthesiology 2004; 100:1258 62 2004 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Spinal Adenosine Receptor Activation Reduces Hypersensitivity after Surgery by a Different
NEUROFIX S MISSION & VISION
 NEUROFIX S MISSION & VISION MISSION Neurofix aims to discover and develop innovative therapies for the treatment of neurological disorders. Our main focus now is Spinal Cord Injury. VISION Spinal Cord
NEUROFIX S MISSION & VISION MISSION Neurofix aims to discover and develop innovative therapies for the treatment of neurological disorders. Our main focus now is Spinal Cord Injury. VISION Spinal Cord
Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the rat dorsal horn
 J Physiol (2003), 548.1, pp. 131 138 DOI: 10.1113/jphysiol.2002.036186 The Physiological Society 2003 www.jphysiol.org Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the
J Physiol (2003), 548.1, pp. 131 138 DOI: 10.1113/jphysiol.2002.036186 The Physiological Society 2003 www.jphysiol.org Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the
Pain Mechanisms. Prof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong. The Somatosensory System
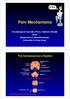 ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
In Vivo Electrophysiological Recording Techniques for the Study of Neuropathic Pain in Rodent Models
 In Vivo Recording Techniques for the Study of Neuropathic Pain in Rodent Models Fei-Yue Zhao, 1 Ross Jeggo, 1 Haifeng Wei, 1 Andrew Whyment, 1 Xin Fang, 1 and David Spanswick 1,2,3 UNIT 11.15 1 NeuroSolutions
In Vivo Recording Techniques for the Study of Neuropathic Pain in Rodent Models Fei-Yue Zhao, 1 Ross Jeggo, 1 Haifeng Wei, 1 Andrew Whyment, 1 Xin Fang, 1 and David Spanswick 1,2,3 UNIT 11.15 1 NeuroSolutions
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Brown EN, Lydic R, Schiff ND, et al. General anesthesia, sleep,
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Brown EN, Lydic R, Schiff ND, et al. General anesthesia, sleep,
Life Sciences 91 (2012) Contents lists available at SciVerse ScienceDirect. Life Sciences. journal homepage:
 Life Sciences 91 (2012) 837 842 Contents lists available at SciVerse ScienceDirect Life Sciences journal homepage: www.elsevier.com/locate/lifescie The effect of intrathecal gabapentin on neuropathic pain
Life Sciences 91 (2012) 837 842 Contents lists available at SciVerse ScienceDirect Life Sciences journal homepage: www.elsevier.com/locate/lifescie The effect of intrathecal gabapentin on neuropathic pain
What it Takes to be a Pain
 What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
CHAPTER 4 PAIN AND ITS MANAGEMENT
 CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
Supplementary Information
 Hyperpolarization-activated cation channels inhibit EPSPs by interactions with M-type K + channels Meena S. George, L.F. Abbott, Steven A. Siegelbaum Supplementary Information Part 1: Supplementary Figures
Hyperpolarization-activated cation channels inhibit EPSPs by interactions with M-type K + channels Meena S. George, L.F. Abbott, Steven A. Siegelbaum Supplementary Information Part 1: Supplementary Figures
The anatomy and physiology of pain
 The anatomy and physiology of pain Charlotte E Steeds Abstract Pain is an unpleasant experience that results from both physical and psychological responses to injury. A complex set of pathways transmits
The anatomy and physiology of pain Charlotte E Steeds Abstract Pain is an unpleasant experience that results from both physical and psychological responses to injury. A complex set of pathways transmits
What is on the Horizon in Drug Therapy for OAB?
 What is on the Horizon in Drug Therapy for OAB? K-E Andersson, MD, PhD Wake Forest Institute for Regenerative Medicine Wake Forest University School of Medicine Winston Salem, North Carolina Disclosures
What is on the Horizon in Drug Therapy for OAB? K-E Andersson, MD, PhD Wake Forest Institute for Regenerative Medicine Wake Forest University School of Medicine Winston Salem, North Carolina Disclosures
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/317/5841/183/dc1 Supporting Online Material for Astrocytes Potentiate Transmitter Release at Single Hippocampal Synapses Gertrudis Perea and Alfonso Araque* *To whom
www.sciencemag.org/cgi/content/full/317/5841/183/dc1 Supporting Online Material for Astrocytes Potentiate Transmitter Release at Single Hippocampal Synapses Gertrudis Perea and Alfonso Araque* *To whom
SYLLABUS SPRING 2011 COURSE: NSC NEUROBIOLOGY OF PAIN
 SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
Acute Pain NETP: SEPTEMBER 2013 COHORT
 Acute Pain NETP: SEPTEMBER 2013 COHORT Pain & Suffering an unpleasant sensory & emotional experience associated with actual or potential tissue damage, or described in terms of such damage International
Acute Pain NETP: SEPTEMBER 2013 COHORT Pain & Suffering an unpleasant sensory & emotional experience associated with actual or potential tissue damage, or described in terms of such damage International
Rapamycin Suppresses Astrocytic and Microglial Activation and Reduced Development of Neuropathic Pain after Spinal Cord Injury in Mice.
 Rapamycin Suppresses Astrocytic and Microglial Activation and Reduced Development of Neuropathic Pain after Spinal Cord Injury in Mice. Satoshi Tateda, M.D., Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D.,
Rapamycin Suppresses Astrocytic and Microglial Activation and Reduced Development of Neuropathic Pain after Spinal Cord Injury in Mice. Satoshi Tateda, M.D., Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D.,
GABA B Receptor Agonist Baclofen Has Non-Specific Antinociceptive Effect in the Model of Peripheral Neuropathy in the Rat
 Physiol. Res. 3: 31-3, GABA B Receptor Agonist Baclofen Has Non-Specific Antinociceptive Effect in the Model of Peripheral Neuropathy in the Rat M. FRANĚK, Š. VACULÍN, R. ROKYTA Department of Normal, Pathological
Physiol. Res. 3: 31-3, GABA B Receptor Agonist Baclofen Has Non-Specific Antinociceptive Effect in the Model of Peripheral Neuropathy in the Rat M. FRANĚK, Š. VACULÍN, R. ROKYTA Department of Normal, Pathological
SUPPLEMENTARY INFORMATION. Supplementary Figure 1
 SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
Centrally Mediated Anti-Hyperalgesic and Anti-Allodynic Effect of Tolperisone in Spared Nerve Injury Model of Neuropathic Pain
 Centrally Mediated Anti-Hyperalgesic and Anti-Allodynic Effect of Tolperisone in Spared Nerve Injury Model of Neuropathic Pain Keyur S Patel *, Punam D Sachdeva Department of Pharmacology, A.R.College
Centrally Mediated Anti-Hyperalgesic and Anti-Allodynic Effect of Tolperisone in Spared Nerve Injury Model of Neuropathic Pain Keyur S Patel *, Punam D Sachdeva Department of Pharmacology, A.R.College
Fibromyalgia: Current Trends and Concepts
 Fibromyalgia: Current Trends and Concepts Dr. Brian Kahan Fellow American Academy of Physical Medicine and Rehabilitation Diplomat American Academy of Pain Medicine American College of Rheumatology (ACR)
Fibromyalgia: Current Trends and Concepts Dr. Brian Kahan Fellow American Academy of Physical Medicine and Rehabilitation Diplomat American Academy of Pain Medicine American College of Rheumatology (ACR)
ANAT2010. Concepts of Neuroanatomy (II) S2 2018
 ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
Materials and Methods
 Anesthesiology 2003; 98:203 8 2003 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Intrathecal Lidocaine Reverses Tactile Allodynia Caused by Nerve Injuries and Potentiates
Anesthesiology 2003; 98:203 8 2003 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Intrathecal Lidocaine Reverses Tactile Allodynia Caused by Nerve Injuries and Potentiates
PAIN MANAGEMENT in the CANINE PATIENT
 PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
Experimental Neurology
 Experimental Neurology 222 (2010) 256 266 Contents lists available at ScienceDirect Experimental Neurology journal homepage: www.elsevier.com/locate/yexnr Contribution of the spinal cord BDNF to the development
Experimental Neurology 222 (2010) 256 266 Contents lists available at ScienceDirect Experimental Neurology journal homepage: www.elsevier.com/locate/yexnr Contribution of the spinal cord BDNF to the development
By the end of this lecture the students will be able to:
 UNIT VII: PAIN Objectives: By the end of this lecture the students will be able to: Review the concept of somatosensory pathway. Describe the function of Nociceptors in response to pain information. Describe
UNIT VII: PAIN Objectives: By the end of this lecture the students will be able to: Review the concept of somatosensory pathway. Describe the function of Nociceptors in response to pain information. Describe
Abbott s TRPV1 Antagonist Program
 Abbott s TRPV1 Antagonist Program Connie R. Faltynek, Ph.D. Sr. Director, Neuroscience and Pain Discovery American Pain Society Annual Meeting Basic Science Dinner Symposium May 9, 2008 Early History of
Abbott s TRPV1 Antagonist Program Connie R. Faltynek, Ph.D. Sr. Director, Neuroscience and Pain Discovery American Pain Society Annual Meeting Basic Science Dinner Symposium May 9, 2008 Early History of
(Received 10 April 1956)
 446 J. Physiol. (I956) I33, 446-455 A COMPARISON OF FLEXOR AND EXTENSOR REFLEXES OF MUSCULAR ORIGIN BY M. G. F. FUORTES AND D. H. HUBEL From the Department ofneurophysiology, Walter Reed Army Institute
446 J. Physiol. (I956) I33, 446-455 A COMPARISON OF FLEXOR AND EXTENSOR REFLEXES OF MUSCULAR ORIGIN BY M. G. F. FUORTES AND D. H. HUBEL From the Department ofneurophysiology, Walter Reed Army Institute
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/3/3/e1600955/dc1 Supplementary Materials for Flexible and stretchable nanowire-coated fibers for optoelectronic probing of spinal cord circuits Chi Lu, Seongjun
advances.sciencemag.org/cgi/content/full/3/3/e1600955/dc1 Supplementary Materials for Flexible and stretchable nanowire-coated fibers for optoelectronic probing of spinal cord circuits Chi Lu, Seongjun
Glycine-gated ion channels Converging mechanism and therapeutic potentials
 Glycine-gated ion channels Converging mechanism and therapeutic potentials Li Zhang Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health,
Glycine-gated ion channels Converging mechanism and therapeutic potentials Li Zhang Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health,
PAIN MODULATION. numerical value. adjectives. DR SYED SHAHID HABIB Professor & Consultant Dept. of Physiology College of Medicine & KKUH
 PAIN MODULATION numerical value adjectives DR SYED SHAHID HABIB Professor & Consultant Dept. of Physiology College of Medicine & KKUH OBJECTIVES At the end of this lecture you should be able to describe:
PAIN MODULATION numerical value adjectives DR SYED SHAHID HABIB Professor & Consultant Dept. of Physiology College of Medicine & KKUH OBJECTIVES At the end of this lecture you should be able to describe:
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/325/5941/760/dc1 Supporting Online Material for Spinal Endocannabinoids and CB 1 Receptors Mediate C-Fiber Induced Heterosynaptic Pain Sensitization Alejandro J. Pernía-Andrade,
www.sciencemag.org/cgi/content/full/325/5941/760/dc1 Supporting Online Material for Spinal Endocannabinoids and CB 1 Receptors Mediate C-Fiber Induced Heterosynaptic Pain Sensitization Alejandro J. Pernía-Andrade,
Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007
 Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
About 70% of brachial plexus injuries are of. Neuromodulation in the Management of Pain from Brachial Plexus Injury. Case Report
 Pain Physician 2008; 11:81-85 ISSN 1533-3159 Case Report Neuromodulation in the Management of Pain from Brachial Plexus Injury Silviu Brill, MD, and Itay Goor Aryeh, MD From: Pain Clinic, Department of
Pain Physician 2008; 11:81-85 ISSN 1533-3159 Case Report Neuromodulation in the Management of Pain from Brachial Plexus Injury Silviu Brill, MD, and Itay Goor Aryeh, MD From: Pain Clinic, Department of
Biomechanics of Pain: Dynamics of the Neuromatrix
 Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Overview of Neuropathic pain
 Overview of Neuropathic pain Kongkiat Kulkantrakorn,M.D. Neurology division Thammasat University 1 Contents Overview of pain New concepts and mechanism Treatment options New data in management 2 3 Breaking
Overview of Neuropathic pain Kongkiat Kulkantrakorn,M.D. Neurology division Thammasat University 1 Contents Overview of pain New concepts and mechanism Treatment options New data in management 2 3 Breaking
SUPPLEMENTARY INFORMATION
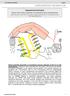 doi: 1.138/nature588 SUPPLEMENTARY INFORMATION Supplemental Information Sensory neuron sodium channel Na v 1.8 is essential for pain at cold temperatures Katharina Zimmermann*, Andreas Leffler*, Alexandru
doi: 1.138/nature588 SUPPLEMENTARY INFORMATION Supplemental Information Sensory neuron sodium channel Na v 1.8 is essential for pain at cold temperatures Katharina Zimmermann*, Andreas Leffler*, Alexandru
Received 26 April 2004; revised 21 August 2004; accepted 20 September 2004 Available online 19 November 2004
 Experimental Neurology 191 (2005) 128 136 www.elsevier.com/locate/yexnr Ectopic discharges from injured nerve fibers are highly correlated with tactile allodynia only in early, but not late, stage in rats
Experimental Neurology 191 (2005) 128 136 www.elsevier.com/locate/yexnr Ectopic discharges from injured nerve fibers are highly correlated with tactile allodynia only in early, but not late, stage in rats
Spinal nociceptive transmission by mechanical stimulation of bone marrow
 Original Article Spinal nociceptive transmission by mechanical stimulation of bone marrow Molecular Pain Volume 12: 1 15! The Author(s) 2016 Reprints and permissions: sagepub.co.uk/journalspermissions.nav
Original Article Spinal nociceptive transmission by mechanical stimulation of bone marrow Molecular Pain Volume 12: 1 15! The Author(s) 2016 Reprints and permissions: sagepub.co.uk/journalspermissions.nav
Peripheral nerve stimulation suppression of C-fiber-evoked flexion reflex in rats. Part 1: Parameters of continuous stimulation
 J Neurosurg 63:612-616, 1985 Peripheral nerve stimulation suppression of C-fiber-evoked flexion reflex in rats Part 1: Parameters of continuous stimulation BENGT H. SJOLUND, M.D., PH.D. Science Branch,
J Neurosurg 63:612-616, 1985 Peripheral nerve stimulation suppression of C-fiber-evoked flexion reflex in rats Part 1: Parameters of continuous stimulation BENGT H. SJOLUND, M.D., PH.D. Science Branch,
Introduction to Neurobiology
 Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
MANAGEMENT OF DIABETIC NEUROPATHY. Chungnam University Hospital Soo-Kyung, Bok, M.D., Ph.D.
 MANAGEMENT OF DIABETIC NEUROPATHY Chungnam University Hospital Soo-Kyung, Bok, M.D., Ph.D. The Diabetic neuropathy cannot be reversed Not to restore function to damaged nerve Slowly progress no initial
MANAGEMENT OF DIABETIC NEUROPATHY Chungnam University Hospital Soo-Kyung, Bok, M.D., Ph.D. The Diabetic neuropathy cannot be reversed Not to restore function to damaged nerve Slowly progress no initial
Intracranial Studies Of Human Epilepsy In A Surgical Setting
 Intracranial Studies Of Human Epilepsy In A Surgical Setting Department of Neurology David Geffen School of Medicine at UCLA Presentation Goals Epilepsy and seizures Basics of the electroencephalogram
Intracranial Studies Of Human Epilepsy In A Surgical Setting Department of Neurology David Geffen School of Medicine at UCLA Presentation Goals Epilepsy and seizures Basics of the electroencephalogram
NORLAND AVENUE PHARMACY PRESCRIPTION COMPOUNDING FOR PAIN MANAGEMENT
 NOVEMBER 2011 NORLAND AVENUE PHARMACY PRESCRIPTION COMPOUNDING N ORLANDA VENUEP HARMACY. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Sciatic Pain
NOVEMBER 2011 NORLAND AVENUE PHARMACY PRESCRIPTION COMPOUNDING N ORLANDA VENUEP HARMACY. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Sciatic Pain
Analgesia Mediated by the TRPM8 Cold Receptor in Chronic Neuropathic Pain
 Current Biology 16, 1591 1605, August 22, 2006 ª2006 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2006.07.061 Analgesia Mediated by the TRPM8 Cold Receptor in Chronic Neuropathic Pain Article Clare
Current Biology 16, 1591 1605, August 22, 2006 ª2006 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2006.07.061 Analgesia Mediated by the TRPM8 Cold Receptor in Chronic Neuropathic Pain Article Clare
Mechanism of Pain Production
 Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
