Venous Thromboembolism (VTE) Prophylaxis Guideline
|
|
|
- Heather Cooper
- 5 years ago
- Views:
Transcription
1 Guideline Venous Thromboembolism (VTE) Prophylaxis Guideline 1. Purpose This guideline outlines the requirement for risk assessment, prescribing, administration and monitoring of venous thromboembolism (VTE) prophylaxis for adult women admitted to the Women s Hospital. Venous thromboembolism (VTE) is a major cause of morbidity and mortality and is one of the most preventable causes of death 1,2. Hospitalised women are 100 times more likely to develop VTE than those in the community. Pregnancy is a risk factor for VTE3,4 and is higher post-partum, following caesarean section, preterm or stillbirth or women with a very high body mass index (BMI) 5. Major gynaecological surgery has a significant risk of both asymptomatic and symptomatic VTE. There is a substantial level of evidence supporting the use of pharmacological VTE prophylaxis, together with adequate hydration, early mobilisation and graduated compression stockings (GCS) in hospitalised women with risk factors. Low molecular weight heparin for VTE prophylaxis in pregnant women is preferred 1,5,7. Aspirin is not first-line therapy for prophylaxis Definitions Pharmacological: Medicines used for thromboprophylaxis. This may include the use of low molecular weight heparin, unfractionated heparin, warfarin and/or aspirin Mechanical prophylaxis: Stockings, pneumatic compression, early mobilisation and surveillance BMI: body mass index HRT: hormonal replacement therapy IPC: intermittent pneumatic calf compression GCS: graduated compression stocking LMWH: low molecular weight heparin PAC: pre-admission clinic ONC-PAC: Oncology pre-admission clinic. 3. Responsibilities The decision to administer VTE prophylaxis is a team responsibility i.e. both surgeons and anaesthetists have an equal responsibility to consider VTE prophylaxis. Medical staff responsibilities: VTE risk assessment and documentation (should include consideration of age, renal function, comorbidities, other medicines and appropriate indications/contraindications). VTE prophylaxis prescribed when indicated: o Caesarean section VTE prophylaxis to be prescribed by anaesthetists o Gynaecology/Oncology VTE prophylaxis to be prescribed by surgical registrar o Antenatal and vaginal birth - Obstetric medical team has overall responsibility for ensuring the VTE risk assessment is performed and documented and VTE prophylaxis is prescribed where indicated. Ongoing risk assessment and safety monitoring for VTE prophylaxis. Nursing/midwifery staff responsibilities: Correct administration of prescribed VTE prophylaxis and application of mechanical prophylaxis Notify the prescriber when the condition of the patient changes. 4. Guideline There are 4 steps to ensuring all women at risk receive appropriate VTE prophylaxis: (also see Appendix 1) Step 1: Assess the patient for VTE risk. Step 2: Assess for contraindications to VTE prophylaxis. Uncontrolled document when printed Updated/Formatted: (19/02/2015) Approved by QSC on 16/02/2015 Page 1 of 6
2 Guideline Venous Thromboembolism (VTE) Prophylaxis Guideline Reasons for contraindication include active bleeding or a high risk of bleeding. These women require compression stockings and/or sequential compression devices. Assess patient for CONTRAINDICATIONS to mechanical prophylaxis, e.g. Local trauma to the lower limb. Step 3: Prescribe VTE prophylaxis. Prophylaxis should generally be started within 24 hours of admission or after surgery and should be continued until discharge or post-discharge where appropriate. Step 4: Review the patient. At discharge, review the patient for the need of extended VTE prophylaxis. 5. Evaluation, monitoring and reporting of compliance to this guideline Compliance to this guideline will be monitored, evaluated and reported through incidents, and/ or audit. 6. References 1. Wickham N, Gallus AS, Walters BNJ, Wilson A, the NVTEPGAC. Prevention of venous thromboembolism in patients admitted to Australian hospitals: summary of National Health and Medical Research Council clinical practice guideline. Internal medicine journal. 2012;42(6): Bates SM, Jaeschke R, Stevens SM, Goodacre S, Wells PS, Stevenson MD, et al. Diagnosis of DVT: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest Feb;141(2 Suppl):e351S-418S. 3. McColl MD, Ramsay JE, Tait RC, Walker ID, McCall F, Conkie JA, et al. Risk factors for pregnancy associated venous thromboembolism. Thromb Haemost Oct;78(4): Simpson EL, Lawrenson RA, Nightingale AL, Farmer RD. Venous thromboembolism in pregnancy and the puerperium: incidence and additional risk factors from a London perinatal database. BJOG Jan;108(1): Sultan AA, Tata LJ, West J, Fiaschi L, Fleming KM, Nelson-Piercy C, et al. Risk factors for first venous thromboembolism around pregnancy: a population-based cohort study from the United Kingdom. Blood May 9;121(19): Gould MK, Garcia DA, Wren SM, Karanicolas PJ, Arcelus JI, Heit JA, et al. Prevention of VTE in nonorthopedic surgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest Feb;141(2 Suppl):e227S-77S. 7. Bates SM, Greer IA, Middeldorp S, Veenstra DL, Prabulos AM, Vandvik PO. VTE, thrombophilia, antithrombotic therapy, and pregnancy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest Feb;141(2 Suppl):e691S-736S. 8. Koller M, Schoch U, Buchmann P, Largiader F, von Felten A, Frick PG. Low molecular weight heparin (KABI 2165) as thromboprophylaxis in elective visceral surgery. A randomized, double-blind study versus unfractionated heparin. Thrombosis and haemostasis Dec 15;56(3): Comparison of a low molecular weight heparin and unfractionated heparin for the prevention of deep vein thrombosis in patients undergoing abdominal surgery. The European Fraxiparin Study (EFS) Group. Br J Surg Nov;75(11): Kakkar VV, Cohen AT, Edmonson RA, Phillips MJ, Cooper DJ, Das SK, et al. Low molecular weight versus standard heparin for prevention of venous thromboembolism after major abdominal surgery. The Thromboprophylaxis Collaborative Group. Lancet Jan 30;341(8840): Boneu B. An international multicentre study: Clivarin in the prevention of venous thromboembolism in patients undergoing general surgery. Report of the International Clivarin Assessment Group. Blood coagulation & fibrinolysis : an international journal in haemostasis and thrombosis Dec;4 Suppl 1:S Horlocker TT, Wedel DJ, Rowlingson JC, Enneking FK, Kopp SL, Benzon HT, et al. Regional anesthesia in the patient receiving antithrombotic or thrombolytic therapy: American Society of Regional Anesthesia and Pain Medicine Evidence-Based Guidelines (Third Edition). Regional anesthesia and pain medicine Uncontrolled document when printed Updated/Formatted: (19/02/2015) Approved by QSC on 16/02/2015 Page 2 of 6
3 Guideline Venous Thromboembolism (VTE) Prophylaxis Guideline Jan-Feb;35(1): Macklon NS, Greer IA. Venous thromboembolic disease in obstetrics and gynaecology: the Scottish experience. Scott Med J Jun;41(3): Legislation/Regulations related to this guideline Not applicable. 8. Appendices Appendix 1 Risk Stratification and Recommended VTE Prophylaxis. PGP Disclaimer Statement The Royal Women's Hospital Clinical Guidelines present statements of 'Best Practice' based on thorough evaluation of evidence and are intended for health professionals only. For practitioners outside the Women s this material is made available in good faith as a resource for use by health professionals to draw on in developing their own protocols, guided by published medical evidence. In doing so, practitioners should themselves be familiar with the literature and make their own interpretations of it. Whilst appreciable care has been taken in the preparation of clinical guidelines which appear on this web page, the Royal Women's Hospital provides these as a service only and does not warrant the accuracy of these guidelines. Any representation implied or expressed concerning the efficacy, appropriateness or suitability of any treatment or product is expressly negated In view of the possibility of human error and / or advances in medical knowledge, the Royal Women's Hospital cannot and does not warrant that the information contained in the guidelines is in every respect accurate or complete. Accordingly, the Royal Women's Hospital will not be held responsible or liable for any errors or omissions that may be found in any of the information at this site. You are encouraged to consult other sources in order to confirm the information contained in any of the guidelines and, in the event that medical treatment is required, to take professional, expert advice from a legally qualified and appropriately experienced medical practitioner. NOTE: Care should be taken when printing any clinical guideline from this site. Updates to these guidelines will take place as necessary. It is therefore advised that regular visits to this site will be needed to access the most current version of these guidelines. Uncontrolled document when printed Updated/Formatted: (19/02/2015) Approved by QSC on 16/02/2015 Page 3 of 6
4 Appendix 1 Risk Stratification and Recommended VTE Prophylaxis Step 1: Assess the patient for VTE prophylaxis Every woman on admission to the Women s hospital must have a formal assessment for VTE prophylaxis which will require review if the clinical status changes. The VTE Risk Assessment section on the Medicines Chart must be completed. VTE prophylaxis, when indicated, must be prescribed by the medical staff responsible for the care of the patient. The prescription for VTE prophylaxis must include compression stockings when indicated. The risk of VTE and the appropriateness of prophylaxis must be reviewed during the patient s hospital stay to ensure changes to the patient s clinical condition (e.g. sepsis, bleeding, surgical intervention) are not compromised by prophylaxis treatment. Risk factors for VTE that require VTE prophylaxis consideration: 1. Pregnancy Assisted birth (Rotational forceps) Parity of 3 or more Caesarean birth Labour 24 hours Pregnancy related medical illness (such as pre-eclampsia) Stillbirth 2. Current cancer 3. Blood dyscrasia Antiphospholipid syndrome/antibody Personal history of VTE Family History or Thrombophilia 4. Lower body pathophysiology 5. Patient factors (3 or more) Major pelvic or abdominal surgery Gross varicose veins Paralysis of lower limbs Age > 35 years BMI 30 Current infection Continuous travel > 3 hours (air travel) or prolonged immobility Excessive blood loss Dehydration Pre-existing major medical illness (e.g. heart/lung disease, inflammatory bowel disease) Central venous catheter in situ Oral contraceptive or HRT Smoker Step 2: Assess the patient for contraindications Contraindications to pharmacological VTE prophylaxis: On current therapeutic anticoagulation Active bleeding Individual risks of bleeding as assessed by treating doctor or surgeon Severe coagulation disorders or haemorrhaging of major organ Uncontrolled document when printed Updated/Formatted: (19/02/2015) Approved by QSC on 16/02/2015 Page 4 of 6
5 Appendix 1 Risk Stratification and Recommended VTE Prophylaxis Adverse reaction to low molecular weight heparin e.g. rash, HITTs Hepatic failure or renal failure. Contraindications to mechanical prophylaxis: Severe peripheral vascular disease Severe peripheral neuropathy Severe lower limb oedema Extreme leg deformity Inflammatory conditions of the lower leg e.g.: ulcer Morbid obesity (where correct fitting of GCS cannot be achieved). For antiplatelet and anticoagulant cessation prior to surgery, refer to the guideline Perioperative Management of Antiplatelet and Anticoagulation in Elective Surgery. Step 3: Prescribe VTE prophylaxis The decision about timing of the dose is guided by the responsible surgical unit and/or anaesthetist if the patient had regional anaesthetic. The low molecular weight heparin of choice for VTE prophylaxis at the Women s is dalteparin, however other low molecular weight heparins are also available. Dose of dalteparin for VTE prophylaxis In general, the dose of VTE prophylaxis depends on the patient s body weight, age and renal function. In women with normal renal function and < 75 years of age, the recommended dose is according to the table below: Weight (kg) Dose of dalteparin (in patients with normal renal function < 40kg 2500 units 40 to 100kg 2500 to 5000 units > 100 kg 7500 units Dosing frequency (subcutaneous injection) In women with renal impairment or 75 years, the dose of VTE prophylaxis may need to be reduced. Consult the haematology unit for further advice. Administration of dalteparin for VTE prophylaxis The dosing frequency is usually every 24 hours. The timing of the dose is guided by the type of procedure as well as the post-operative bleeding risk. daily Unless specified by the surgical treating unit, the following is recommended 13. Epidural: Epidural catheters should be removed at least 12 hours after a dose of VTE prophylaxis Following epidural catheter removal, subsequent doses of VTE prophylaxis should be administered no earlier than 6 hours after removal and subsequent doses should be administered daily. Regional anaesthesia: Regional anaesthesia should be administered 12 hours AFTER the last dose of VTE prophylaxis VTE prophylaxis should be administered no earlier than 6 hours after regional anaesthesia and subsequent doses should be administered daily. Mechanical VTE prophylaxis Adequate hydration and early mobilisation reduces the risk of VTE 6. There is little evidence that mechanical VTE prophylaxis provides added benefit to pharmacological prophylaxis, but intermittent pneumatic compression (IPC) reduces VTE risk in comparison to no prophylaxis 7. Uncontrolled document when printed Updated/Formatted: (19/02/2015) Approved by QSC on 16/02/2015 Page 5 of 6
6 Appendix 1 Risk Stratification and Recommended VTE Prophylaxis Mechanical VTE prophylaxis (such as graduated compression stockings (GCS) or IPC) is recommended for women who have contraindications to pharmacological VTE prophylaxis or as an adjunctive therapy in women with a high risk of VTE. Mechanical VTE prophylaxis must be prescribed or initiated on the medicines chart. GCS must be measured and fitted for each woman and should be worn continuously Ideally IPC should begin at induction of anaesthesia and continue post-operatively unless contraindicated. Step 4: Review the patient Duration of dalteparin for VTE prophylaxis The decision regarding the duration of prophylaxis should be made for each patient individually. Generally, VTE prophylaxis is recommended to be continued until the patient is fully mobile or for 7-10 days following gynaecology/oncology surgery and 5-7 days following caesarean birth. High-risk patients: Obstetric patients consider VTE prophylaxis for up to 6 weeks after birth 2,8. Women undergoing major abdominal/pelvic surgery for cancer with other risk factors may require extended duration of VTE prophylaxis up to 4 weeks from discharge 2,14. For women who developed ovarian hyper-stimulation syndrome while undergoing assisted reproduction, consider VTE prophylaxis for up to 3 months post resolution of the syndrome 2. Uncontrolled document when printed Updated/Formatted: (19/02/2015) Approved by QSC on 16/02/2015 Page 6 of 6
Venous Thromboembolism Prophylaxis
 Approved by: Venous Thromboembolism Prophylaxis Vice President and Chief Medical Officer; and Vice President and Chief Operating Officer Corporate Policy & Procedures Manual Number: Date Approved January
Approved by: Venous Thromboembolism Prophylaxis Vice President and Chief Medical Officer; and Vice President and Chief Operating Officer Corporate Policy & Procedures Manual Number: Date Approved January
Objectives. Venous Thromboembolism (VTE) Prophylaxis. Case VTE WHY DO IT? Question: Who Is At Risk?
 Objectives Venous Thromboembolism (VTE) Prophylaxis Rishi Garg, MD Department of Medicine Identify patients at risk for VTE Options for VTE prophylaxis Current Recommendations (based on The Seventh ACCP
Objectives Venous Thromboembolism (VTE) Prophylaxis Rishi Garg, MD Department of Medicine Identify patients at risk for VTE Options for VTE prophylaxis Current Recommendations (based on The Seventh ACCP
NOTE: The first appearance of terms in bold in the body of this document (except titles) are defined terms please refer to the Definitions section.
 TITLE VENOUS THROMBOEMBOLISM PROPHYLAXIS SCOPE Provincial Acute and Sub-Acute Care Facilities APPROVAL AUTHORITY Alberta Health Services Executive Committee SPONSOR Vice President, Quality and Chief Medical
TITLE VENOUS THROMBOEMBOLISM PROPHYLAXIS SCOPE Provincial Acute and Sub-Acute Care Facilities APPROVAL AUTHORITY Alberta Health Services Executive Committee SPONSOR Vice President, Quality and Chief Medical
Referral to the Women s Alcohol and Drug Service (WADS) Procedure
 1. Purpose The following document describes criteria for the referral to Women s Alcohol and Drug Service (WADS) and how a referral is taken and processed. This includes the referral for clinical care
1. Purpose The following document describes criteria for the referral to Women s Alcohol and Drug Service (WADS) and how a referral is taken and processed. This includes the referral for clinical care
Anticoagulation for prevention of venous thromboembolism
 Anticoagulation for prevention of venous thromboembolism Original article by: Michael Tam Note: updated in June 2009 with the eighth edition (from the seventh) evidence-based clinical practice guidelines
Anticoagulation for prevention of venous thromboembolism Original article by: Michael Tam Note: updated in June 2009 with the eighth edition (from the seventh) evidence-based clinical practice guidelines
THROMBOPROPHYLAXIS: NON-ORTHOPEDIC SURGERY
 THROMBOPROPHYLAXIS: NON-ORTHOPEDIC SURGERY OBJECTIVE: To outline a practical approach for the prevention of venous thromboembolism (VTE) in patients undergoing non-orthopedic surgery. BACKGROUND: VTE is
THROMBOPROPHYLAXIS: NON-ORTHOPEDIC SURGERY OBJECTIVE: To outline a practical approach for the prevention of venous thromboembolism (VTE) in patients undergoing non-orthopedic surgery. BACKGROUND: VTE is
INDICATIONS FOR THROMBO-PROPHYLAXIS AND WHEN TO STOP ANTICOAGULATION BEFORE ELECTIVE SURGERY
 INDICATIONS FOR THROMBO-PROPHYLAXIS AND WHEN TO STOP ANTICOAGULATION BEFORE ELECTIVE SURGERY N.E. Pearce INTRODUCTION Preventable death Cause of morbidity and mortality Risk factors Pulmonary embolism
INDICATIONS FOR THROMBO-PROPHYLAXIS AND WHEN TO STOP ANTICOAGULATION BEFORE ELECTIVE SURGERY N.E. Pearce INTRODUCTION Preventable death Cause of morbidity and mortality Risk factors Pulmonary embolism
Getting Started Kit VENOUS THROMBOEMBOLISM PREVENTION. Section 2: Evidence-Based Appropriate VTE Prophylaxis
 Reducing Harm Improving Healthcare Protecting Canadians VENOUS THROMBOEMBOLISM PREVENTION Getting Started Kit Section 2: Evidence-Based Appropriate VTE Prophylaxis January 2017 www.patientsafetyinstitute.ca
Reducing Harm Improving Healthcare Protecting Canadians VENOUS THROMBOEMBOLISM PREVENTION Getting Started Kit Section 2: Evidence-Based Appropriate VTE Prophylaxis January 2017 www.patientsafetyinstitute.ca
1. SCOPE of GUIDELINE:
 Page 1 of 35 CLINICAL PRACTICE GUIDELINE: Venous Thromboembolism (VTE) Prevention Guideline: Thromboprophylaxis AUTHORIZATION: VP, Medicine Date Approved: May 17, 2012 Date Revised: Vancouver Coastal Health
Page 1 of 35 CLINICAL PRACTICE GUIDELINE: Venous Thromboembolism (VTE) Prevention Guideline: Thromboprophylaxis AUTHORIZATION: VP, Medicine Date Approved: May 17, 2012 Date Revised: Vancouver Coastal Health
VTE Management in Surgical Patients: Optimizing Prophylaxis Strategies
 VTE Management in Surgical Patients: Optimizing Prophylaxis Strategies VTE in Surgical Patients: Recognizing the Patients at Risk Pathogenesis of thrombosis: Virchow s triad and VTE Risk Hypercoagulability
VTE Management in Surgical Patients: Optimizing Prophylaxis Strategies VTE in Surgical Patients: Recognizing the Patients at Risk Pathogenesis of thrombosis: Virchow s triad and VTE Risk Hypercoagulability
NICE Guidance: Venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients admitted to hospital 1
 The College of Emergency Medicine Patron: HRH The Princess Royal Churchill House Tel +44 (0)207 404 1999 35 Red Lion Square Fax +44 (0)207 067 1267 London WC1R 4SG www.collemergencymed.ac.uk CLINICAL EFFECTIVENESS
The College of Emergency Medicine Patron: HRH The Princess Royal Churchill House Tel +44 (0)207 404 1999 35 Red Lion Square Fax +44 (0)207 067 1267 London WC1R 4SG www.collemergencymed.ac.uk CLINICAL EFFECTIVENESS
*Corresponding Author:
 Audit of venous thromboembolism prophylaxis administered to general surgical patients undergoing elective and emergency operations at National Hospital, Sri Lanka *Migara Seneviratne 1, Asanka Hemachandra
Audit of venous thromboembolism prophylaxis administered to general surgical patients undergoing elective and emergency operations at National Hospital, Sri Lanka *Migara Seneviratne 1, Asanka Hemachandra
Reducing the risk of venous thrombo-embolism (VTE) in hospital and after discharge
 Reducing the risk of venous thrombo-embolism (VTE) in hospital and after discharge What is a venous thromboembolism (VTE)? This is a medical term that describes a blood clot that develops in a deep vein
Reducing the risk of venous thrombo-embolism (VTE) in hospital and after discharge What is a venous thromboembolism (VTE)? This is a medical term that describes a blood clot that develops in a deep vein
These are guidelines only and can be deviated from if it is thought to be in the patient s best interest.
 Clinical Guideline Venothromboembolism prophylaxis: Trauma and Orthopaedics Venous thromboembolism (VTE) is a recognised complication associated with inactivity and surgical procedures. Therefore, all
Clinical Guideline Venothromboembolism prophylaxis: Trauma and Orthopaedics Venous thromboembolism (VTE) is a recognised complication associated with inactivity and surgical procedures. Therefore, all
Misunderstandings of Venous thromboembolism prophylaxis
 Misunderstandings of Venous thromboembolism prophylaxis Veerendra Chadachan Senior Consultant Dept of General Medicine (Vascular Medicine and Hypertension) Tan Tock Seng Hospital, Singapore Case scenario
Misunderstandings of Venous thromboembolism prophylaxis Veerendra Chadachan Senior Consultant Dept of General Medicine (Vascular Medicine and Hypertension) Tan Tock Seng Hospital, Singapore Case scenario
Venous thromboembolism - reducing the risk
 Venous thromboembolism - reducing the risk Reducing the risk of venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients admitted to hospital NICE guideline Draft for consultation,
Venous thromboembolism - reducing the risk Reducing the risk of venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients admitted to hospital NICE guideline Draft for consultation,
Venous thromboembolism: reducing the risk
 Issue date: January 2010 Venous thromboembolism: reducing the risk Reducing the risk of venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients admitted to hospital This guideline
Issue date: January 2010 Venous thromboembolism: reducing the risk Reducing the risk of venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients admitted to hospital This guideline
Venothromboembolism prophylaxis: Trauma and Orthopaedics Clinical guideline, V2
 Clinical Guideline Venothromboembolism prophylaxis: Trauma and Orthopaedics 11/11/11 TEMPORARY GUIDANCE There is no prophylactic tinzaparin available in the Trust currently. Please substitute enoxaparin
Clinical Guideline Venothromboembolism prophylaxis: Trauma and Orthopaedics 11/11/11 TEMPORARY GUIDANCE There is no prophylactic tinzaparin available in the Trust currently. Please substitute enoxaparin
DVT - initial management NSCCG
 Background information Information resources for patients and carers Updates to this care map Synonyms Below knee DVT and bleeding risks Patient with confirmed DVT Scan confirms superficial thrombophlebitis
Background information Information resources for patients and carers Updates to this care map Synonyms Below knee DVT and bleeding risks Patient with confirmed DVT Scan confirms superficial thrombophlebitis
Prevention and management of deep venous thrombosis (DVT) John Fletcher Wound Care Association of New South Wales
 Prevention and management of deep venous thrombosis (DVT) John Fletcher Wound Care Association of New South Wales Merimbula, 6 th November 2010 University of Sydney Department of Surgery Westmead Hospital
Prevention and management of deep venous thrombosis (DVT) John Fletcher Wound Care Association of New South Wales Merimbula, 6 th November 2010 University of Sydney Department of Surgery Westmead Hospital
Referral to the Women s Alcohol and Drug Service (WADS) Procedure
 Procedure Referral to the Women s Alcohol and Drug Service (WADS) Procedure. Purpose The following document describes criteria for the referral to Women s Alcohol and Drug Service (WADS) and how a referral
Procedure Referral to the Women s Alcohol and Drug Service (WADS) Procedure. Purpose The following document describes criteria for the referral to Women s Alcohol and Drug Service (WADS) and how a referral
AN AUDIT: THROMBOPROPHYLAXIS FOR TOTAL HIP REPLACEMENT PATIENTS AT NORTHWICK PARK AND CENTRAL MIDDLESEX HOSPITALS
 The West London Medical Journal 2010 Vol 2 No 4 pp 19-24 AN AUDIT: THROMBOPROPHYLAXIS FOR TOTAL HIP REPLACEMENT PATIENTS AT NORTHWICK PARK AND CENTRAL MIDDLESEX HOSPITALS Soneji ND Agni NR Acharya MN Anjari
The West London Medical Journal 2010 Vol 2 No 4 pp 19-24 AN AUDIT: THROMBOPROPHYLAXIS FOR TOTAL HIP REPLACEMENT PATIENTS AT NORTHWICK PARK AND CENTRAL MIDDLESEX HOSPITALS Soneji ND Agni NR Acharya MN Anjari
Magnesium Sulphate - Management of Hypertensive Disorders of Pregnancy
 1. Purpose Magnesium sulphate is the anticonvulsant of choice for pre-eclampsia prophylaxis and treatment. This clinical guideline outlines the indications, contraindications, administration and monitoring
1. Purpose Magnesium sulphate is the anticonvulsant of choice for pre-eclampsia prophylaxis and treatment. This clinical guideline outlines the indications, contraindications, administration and monitoring
General. Recommendations. Guideline Title. Bibliographic Source(s) Guideline Status. Major Recommendations
 General Guideline Title Prevention of deep vein thrombosis and pulmonary embolism. Bibliographic Source(s) American College of Obstetricians and Gynecologists (ACOG). Prevention of deep vein thrombosis
General Guideline Title Prevention of deep vein thrombosis and pulmonary embolism. Bibliographic Source(s) American College of Obstetricians and Gynecologists (ACOG). Prevention of deep vein thrombosis
DVT Pathophysiology and Prophylaxis in Medically Hospitalized Patients. David Liff MD Oklahoma Heart Institute Vascular Center
 DVT Pathophysiology and Prophylaxis in Medically Hospitalized Patients David Liff MD Oklahoma Heart Institute Vascular Center Overview Pathophysiology of DVT Epidemiology and risk factors for DVT in the
DVT Pathophysiology and Prophylaxis in Medically Hospitalized Patients David Liff MD Oklahoma Heart Institute Vascular Center Overview Pathophysiology of DVT Epidemiology and risk factors for DVT in the
How to prevent blood clots whilst in hospital and after your return home
 How to prevent blood clots whilst in hospital and after your return home Patient information WHAT What IS is DEEP deep VEIN vein THROMBOSIS? thrombosis? Deep Vein Thrombosis DVT is a blood clot within
How to prevent blood clots whilst in hospital and after your return home Patient information WHAT What IS is DEEP deep VEIN vein THROMBOSIS? thrombosis? Deep Vein Thrombosis DVT is a blood clot within
Venous Thromboembolism. Prevention
 Venous Thromboembolism Prevention August 2010 Venous Thromboembloism Prevention 1 1 Expected Practice Assess all patients upon admission to the ICU for risk factors of venous thromboembolism (VTE) and
Venous Thromboembolism Prevention August 2010 Venous Thromboembloism Prevention 1 1 Expected Practice Assess all patients upon admission to the ICU for risk factors of venous thromboembolism (VTE) and
Prevention of Venous Thromboembolism
 Prevention of Venous Thromboembolism Surgical Care Improvement Project Dale W. Bratzler, DO, MPH President and CEO Dale W. Bratzler, DO, MPH Oklahoma Foundation for Medical Quality QIOSC Medical Director
Prevention of Venous Thromboembolism Surgical Care Improvement Project Dale W. Bratzler, DO, MPH President and CEO Dale W. Bratzler, DO, MPH Oklahoma Foundation for Medical Quality QIOSC Medical Director
Venous Thromboembolism Prophylaxis - Why Should We Care? Harry Gibbs FRACP FCSANZ Vascular Physician The Alfred Hospital
 Venous Thromboembolism Prophylaxis - Why Should We Care? Harry Gibbs FRACP FCSANZ Vascular Physician The Alfred Hospital VTE is common and dangerous 5 VTE is Common VTE Incidence: 1.5 / 1000 per year
Venous Thromboembolism Prophylaxis - Why Should We Care? Harry Gibbs FRACP FCSANZ Vascular Physician The Alfred Hospital VTE is common and dangerous 5 VTE is Common VTE Incidence: 1.5 / 1000 per year
Thalassaemia and Abnormal Haemoglobins in Pregnancy
 1. Purpose Thalassaemias and abnormal haemoglobins are detected in approximately 4% of patients of reproductive age attending the Women's. In almost half of these cases, the abnormality is not evident
1. Purpose Thalassaemias and abnormal haemoglobins are detected in approximately 4% of patients of reproductive age attending the Women's. In almost half of these cases, the abnormality is not evident
Health Care Worker (Pregnant) - Infectious Diseases Risks and Exposure
 1. Purpose The purpose of this guideline is to provide accurate information on the risks to pregnant Health Care Workers (HCWs) in the event of an exposure to a transmissible infectious disease at the
1. Purpose The purpose of this guideline is to provide accurate information on the risks to pregnant Health Care Workers (HCWs) in the event of an exposure to a transmissible infectious disease at the
Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital
 Please Note: This policy is currently under review and is still fit for purpose. Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital This procedural document supersedes
Please Note: This policy is currently under review and is still fit for purpose. Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital This procedural document supersedes
Bath, Philip M.W. and England, Timothy J. (2009) Thighlength compression stockings and DVT after stroke. Lancet. ISSN (In Press)
 Bath, Philip M.W. and England, Timothy J. (2009) Thighlength compression stockings and DVT after stroke. Lancet. ISSN 0140-6736 (In Press) Access from the University of Nottingham repository: http://eprints.nottingham.ac.uk/1087/1/lancet_clots_1_20090522_4.pdf
Bath, Philip M.W. and England, Timothy J. (2009) Thighlength compression stockings and DVT after stroke. Lancet. ISSN 0140-6736 (In Press) Access from the University of Nottingham repository: http://eprints.nottingham.ac.uk/1087/1/lancet_clots_1_20090522_4.pdf
DVT PROPHYLAXIS IN HOSPITALIZED MEDICAL PATIENTS SAURABH MAJI SR (PULMONARY,MEDICINE)
 DVT PROPHYLAXIS IN HOSPITALIZED MEDICAL PATIENTS SAURABH MAJI SR (PULMONARY,MEDICINE) Introduction VTE (DVT/PE) is an important complication in hospitalized patients Hospitalization for acute medical illness
DVT PROPHYLAXIS IN HOSPITALIZED MEDICAL PATIENTS SAURABH MAJI SR (PULMONARY,MEDICINE) Introduction VTE (DVT/PE) is an important complication in hospitalized patients Hospitalization for acute medical illness
Deep Vein Thrombosis
 Deep Vein Thrombosis from NHS (UK) guidelines Introduction Deep vein thrombosis (DVT) is a blood clot in one of the deep veins in the body. Blood clots that develop in a vein are also known as venous thrombosis.
Deep Vein Thrombosis from NHS (UK) guidelines Introduction Deep vein thrombosis (DVT) is a blood clot in one of the deep veins in the body. Blood clots that develop in a vein are also known as venous thrombosis.
Society of Trauma Nurses TraumaCon 03/22/2018
 Prophylaxis Against Venous Thromboemblism (VTE) in Pediatric Trauma Society of Trauma Nurses TraumaCon 03/22/2018 Arash Mahajerin, MD, MSCr Hematology, CHOC Children s Specialists Orange, CA Disclosure
Prophylaxis Against Venous Thromboemblism (VTE) in Pediatric Trauma Society of Trauma Nurses TraumaCon 03/22/2018 Arash Mahajerin, MD, MSCr Hematology, CHOC Children s Specialists Orange, CA Disclosure
Early Ambulation Reduces the Risk of Venous Thromboembolism After Total Knee Replacement. Marilyn Szekendi, PhD, RN
 Early Ambulation Reduces the Risk of Venous Thromboembolism After Total Knee Replacement Marilyn Szekendi, PhD, RN ANA 7 th Annual Nursing Quality Conference, February 2013 Research Team Banafsheh Sadeghi,
Early Ambulation Reduces the Risk of Venous Thromboembolism After Total Knee Replacement Marilyn Szekendi, PhD, RN ANA 7 th Annual Nursing Quality Conference, February 2013 Research Team Banafsheh Sadeghi,
Clinical Guideline for Anticoagulation in VTE
 Clinical Guideline for Anticoagulation in VTE These clinical guidelines are intended to provide evidence-based recommendations regarding the anticoagulation in patients with DVT and PE. Please note that
Clinical Guideline for Anticoagulation in VTE These clinical guidelines are intended to provide evidence-based recommendations regarding the anticoagulation in patients with DVT and PE. Please note that
SUBJECT: LIMB PNEUMATIC COMPRESSION EFFECTIVE DATE: 06/27/13 DEVICES FOR VENOUS REVISED DATE: 06/26/14 THROMBOEMBOLISM PROPHYLAXIS
 MEDICAL POLICY SUBJECT: LIMB PNEUMATIC COMPRESSION PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical
MEDICAL POLICY SUBJECT: LIMB PNEUMATIC COMPRESSION PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical
Prevention and treatment of venous thromboembolic disease
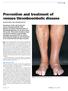 REVIEW Prevention and treatment of venous thromboembolic disease SUSAN McNEILL AND CATHERINE BAGOT Awareness of the risk factors for venous thromboembolic (VTE) disease and timely administration of thromboprophylaxis
REVIEW Prevention and treatment of venous thromboembolic disease SUSAN McNEILL AND CATHERINE BAGOT Awareness of the risk factors for venous thromboembolic (VTE) disease and timely administration of thromboprophylaxis
Medical Patients: A Population at Risk
 Case Vignette A 68-year-old woman with obesity was admitted to the Medical Service with COPD and pneumonia and was treated with oral corticosteroids, bronchodilators, and antibiotics. She responded well
Case Vignette A 68-year-old woman with obesity was admitted to the Medical Service with COPD and pneumonia and was treated with oral corticosteroids, bronchodilators, and antibiotics. She responded well
Comparison of Venothromboembolism Prophylaxis Practices in a Winnipeg Tertiary Care Hospital to Chest Guidelines: A Quality Improvement Project
 Comparison of Venothromboembolism Prophylaxis Practices in a Winnipeg Tertiary Care Hospital to Chest Guidelines: A Quality Improvement Project Dr. Jonathan Laxton, FRCPC, R5 GIM University of Manitoba
Comparison of Venothromboembolism Prophylaxis Practices in a Winnipeg Tertiary Care Hospital to Chest Guidelines: A Quality Improvement Project Dr. Jonathan Laxton, FRCPC, R5 GIM University of Manitoba
Protocol. Postsurgical Outpatient Use of Limb Compression Devices for Venous Thromboembolism Prophylaxis
 Postsurgical Outpatient Use of Limb Compression Devices for (10128) (Formerly Outpatient Use of Limb Pneumatic Compression Devices for ) Medical Benefit Effective Date: 07/01/14 Next Review Date: 03/15
Postsurgical Outpatient Use of Limb Compression Devices for (10128) (Formerly Outpatient Use of Limb Pneumatic Compression Devices for ) Medical Benefit Effective Date: 07/01/14 Next Review Date: 03/15
THROMBOPROPHYLAXIS POLICY FOR ADULT PATIENTS ADMITTED AS INPATIENTS. 362 Version No: 2 Previous Trust / LHB Ref No:
 Reference No: THROMBOPROPHYLAXIS POLICY FOR ADULT PATIENTS ADMITTED AS INPATIENTS 362 Version No: 2 Previous Trust / LHB Ref No: 362 Documents to read alongside this Policy, Procedure etc (delete as necessary)
Reference No: THROMBOPROPHYLAXIS POLICY FOR ADULT PATIENTS ADMITTED AS INPATIENTS 362 Version No: 2 Previous Trust / LHB Ref No: 362 Documents to read alongside this Policy, Procedure etc (delete as necessary)
Postsurgical Home Use of Limb Compression Devices for Venous Thromboembolism Prophylaxis
 Clinical Position Statement Postsurgical Home Use of Limb Compression Devices for Venous Thromboembolism Prophylaxis Effective: October 2017 Next Review: September 2018 CLINICAL POSITION STATEMENT Postsurgical
Clinical Position Statement Postsurgical Home Use of Limb Compression Devices for Venous Thromboembolism Prophylaxis Effective: October 2017 Next Review: September 2018 CLINICAL POSITION STATEMENT Postsurgical
Venous thrombosis is common and often occurs spontaneously, but it also frequently accompanies medical and surgical conditions, both in the community
 Venous Thrombosis Venous Thrombosis It occurs mainly in the deep veins of the leg (deep vein thrombosis, DVT), from which parts of the clot frequently embolize to the lungs (pulmonary embolism, PE). Fewer
Venous Thrombosis Venous Thrombosis It occurs mainly in the deep veins of the leg (deep vein thrombosis, DVT), from which parts of the clot frequently embolize to the lungs (pulmonary embolism, PE). Fewer
CHAPTER 2 VENOUS THROMBOEMBOLISM
 CHAPTER 2 VENOUS THROMBOEMBOLISM Objectives Venous Thromboembolism (VTE) Prevalence Patho-physiology Risk Factors Diagnosis Pulmonary Embolism (PE) Management of DVT/PE Prevention VTE Patho-physiology
CHAPTER 2 VENOUS THROMBOEMBOLISM Objectives Venous Thromboembolism (VTE) Prevalence Patho-physiology Risk Factors Diagnosis Pulmonary Embolism (PE) Management of DVT/PE Prevention VTE Patho-physiology
The Royal College of Emergency Medicine. VTE Risk in Lower Limb Immobilisation in Plaster Cast 2015/2016
 The Royal College of Emergency Medicine Clinical Audits VTE Risk in Lower Limb Immobilisation in Plaster Cast Introduction 2015/2016 EXCELLENCE IN EMERGENCY MEDICINE A significant number of patients attend
The Royal College of Emergency Medicine Clinical Audits VTE Risk in Lower Limb Immobilisation in Plaster Cast Introduction 2015/2016 EXCELLENCE IN EMERGENCY MEDICINE A significant number of patients attend
Guideline Quick View: Venous Thromboembolism
 Guideline Quick View: Venous Thromboembolism The AORN Guideline Quick View is a key component of Guideline Essentials, a suite of online implementation tools designed to help the perioperative team translate
Guideline Quick View: Venous Thromboembolism The AORN Guideline Quick View is a key component of Guideline Essentials, a suite of online implementation tools designed to help the perioperative team translate
NICE guideline Published: 21 March 2018 nice.org.uk/guidance/ng89
 Venous thromboembolism in over 16s: reducing the risk of hospital-acquired deep vein thrombosis or pulmonary embolism NICE guideline Published: 21 March 2018 nice.org.uk/guidance/ng89 NICE 2018. All rights
Venous thromboembolism in over 16s: reducing the risk of hospital-acquired deep vein thrombosis or pulmonary embolism NICE guideline Published: 21 March 2018 nice.org.uk/guidance/ng89 NICE 2018. All rights
Prevention of Venous Thromboembolism
 Prevention of Venous Thromboembolism Reference No: P_CS_05 Version 2.4 Ratified by: LCHS Trust Board Date ratified: 27 th January 2015 Name of originator / author: Medical Director Name of responsible
Prevention of Venous Thromboembolism Reference No: P_CS_05 Version 2.4 Ratified by: LCHS Trust Board Date ratified: 27 th January 2015 Name of originator / author: Medical Director Name of responsible
Page: 1 of 13. Post-Surgical Outpatient Use of Limb Compression Devices for Venous Thromboembolism Prophylaxis
 Last Review Status/Date: March 2014 Page: 1 of 13 Compression Devices for Venous Description Patients undergoing major orthopedic surgery are at increased risk for venous thromboembolism (VTE). Patients
Last Review Status/Date: March 2014 Page: 1 of 13 Compression Devices for Venous Description Patients undergoing major orthopedic surgery are at increased risk for venous thromboembolism (VTE). Patients
New v1.0 Date: December 2015 Patricia Wain - Associate Director Physical Care. Kenny Laing - Deputy Director of Nursing
 Clinical Venous Thromboembolism: Standing Operating Procedure Document Control Summary Status: Version: Author/Title: Owner/Title: New v1.0 Date: December 2015 Patricia Wain - Associate Director Physical
Clinical Venous Thromboembolism: Standing Operating Procedure Document Control Summary Status: Version: Author/Title: Owner/Title: New v1.0 Date: December 2015 Patricia Wain - Associate Director Physical
GUIDELINES FOR THE PREVENTION AND TREATMENT OF VENOUS THROMBOEMBOLISM (VTE) IN MEDICAL AND SURGICAL INPATIENTS ON THE MDGH SITE
 GUIDELINES FOR THE PREVENTION AND TREATMENT OF VENOUS THROMBOEMBOLISM (VTE) IN MEDICAL AND SURGICAL INPATIENTS ON THE MDGH SITE This local guideline has been produced as an aid to risk assessment and prevention
GUIDELINES FOR THE PREVENTION AND TREATMENT OF VENOUS THROMBOEMBOLISM (VTE) IN MEDICAL AND SURGICAL INPATIENTS ON THE MDGH SITE This local guideline has been produced as an aid to risk assessment and prevention
Adam Goldfarb, M.A., D.C., D.E.S.S. Introduction
 Venous Thromboembolism Prophylaxis following Lower Extremity Orthopedic Surgery: A Review of the Biomedical Research Literature and Evidence-Based Policy in the United States. Adam Goldfarb, M.A., D.C.,
Venous Thromboembolism Prophylaxis following Lower Extremity Orthopedic Surgery: A Review of the Biomedical Research Literature and Evidence-Based Policy in the United States. Adam Goldfarb, M.A., D.C.,
Page: 1 of 14. Post-Surgical Outpatient Use of Limb Compression Devices for Venous Thromboembolism Prophylaxis
 Subject: Post-Surgical Outpatient Use of Limb Page: 1 of 14 Last Review Status/Date: March 2015 Post-Surgical Outpatient Use of Limb Compression Devices for Venous Description Patients undergoing major
Subject: Post-Surgical Outpatient Use of Limb Page: 1 of 14 Last Review Status/Date: March 2015 Post-Surgical Outpatient Use of Limb Compression Devices for Venous Description Patients undergoing major
Deep vein thrombosis: diagnosis, prevention and treatment
 Deep vein thrombosis: diagnosis, prevention and treatment Catherine Bagot BSc, MD, MRCP, FRCPath and Campbell Tait BSc, FRCP, FRCPath Deep vein thrombosis can lead to significant morbidity and has well-recognised
Deep vein thrombosis: diagnosis, prevention and treatment Catherine Bagot BSc, MD, MRCP, FRCPath and Campbell Tait BSc, FRCP, FRCPath Deep vein thrombosis can lead to significant morbidity and has well-recognised
Low Molecular Weight Heparin for Prevention and Treatment of Venous Thromboembolic Disorders
 SURGICAL GRAND ROUNDS March 17 th, 2007 Low Molecular Weight Heparin for Prevention and Treatment of Venous Thromboembolic Disorders Guillermo Escobar, M.D. LMWH vs UFH Jayer s sales pitch: FALSE LMW is
SURGICAL GRAND ROUNDS March 17 th, 2007 Low Molecular Weight Heparin for Prevention and Treatment of Venous Thromboembolic Disorders Guillermo Escobar, M.D. LMWH vs UFH Jayer s sales pitch: FALSE LMW is
Dr. Steve Ligertwood Dr. Roderick Tukker Dr. David Wilton
 Dr. Steve Ligertwood Hospitalist Royal Columbian Hospital Regional Department Head-Hospitalist for Fraser Health Authority Project Lead BC Hospitalist VTE Collaborative Clinical Instructor, UBC School
Dr. Steve Ligertwood Hospitalist Royal Columbian Hospital Regional Department Head-Hospitalist for Fraser Health Authority Project Lead BC Hospitalist VTE Collaborative Clinical Instructor, UBC School
EXTENDING VTE PROPHYLAXIS IN ACUTELY ILL MEDICAL PATIENTS
 EXTENDING VTE PROPHYLAXIS IN ACUTELY ILL MEDICAL PATIENTS Samuel Z. Goldhaber, MD Director, VTE Research Group Cardiovascular Division Brigham and Women s Hospital Professor of Medicine Harvard Medical
EXTENDING VTE PROPHYLAXIS IN ACUTELY ILL MEDICAL PATIENTS Samuel Z. Goldhaber, MD Director, VTE Research Group Cardiovascular Division Brigham and Women s Hospital Professor of Medicine Harvard Medical
RCT on GCS/LMWH vs LMWH in VTE prophylaxis
 RCT on GCS/LMWH vs LMWH in VTE prophylaxis GAPS: Graduated compression as an Adjunct to Pharmacoprophylaxis in Surgery A Multi-Centre Randomised Clinical Trial Joseph Shalhoub, Alun H Davies on behalf
RCT on GCS/LMWH vs LMWH in VTE prophylaxis GAPS: Graduated compression as an Adjunct to Pharmacoprophylaxis in Surgery A Multi-Centre Randomised Clinical Trial Joseph Shalhoub, Alun H Davies on behalf
Shared Care Protocol for the Prescription and Supply of Low Molecular Weight Heparins
 Tameside Hospital NHS Foundation Trust and NHS Tameside and Glossop Shared Care Protocol for the Prescription and Supply of Low Molecular Weight Heparins Version 5.2 Version: 5.2 Authorised by: Joint Medicines
Tameside Hospital NHS Foundation Trust and NHS Tameside and Glossop Shared Care Protocol for the Prescription and Supply of Low Molecular Weight Heparins Version 5.2 Version: 5.2 Authorised by: Joint Medicines
Trust Policy. Venous Thromboembolism (VTE) Risk Assessment and Prophylactic Treatment. Dr Allison Tso, Consultant Haematologist Version 3.
 Trust Policy Venous Thromboembolism (VTE) Risk Assessment and Prophylactic Treatment Author(s) Dr Allison Tso, Consultant Haematologist Version 3.1 Version Date March 2013 Implementation/approval Date
Trust Policy Venous Thromboembolism (VTE) Risk Assessment and Prophylactic Treatment Author(s) Dr Allison Tso, Consultant Haematologist Version 3.1 Version Date March 2013 Implementation/approval Date
VTE in the Trauma Population
 VTE in the Trauma Population Erik Peltz, D.O. February 11 th, 2015 * contributions from Eduardo Gonzalez, M.D. University of Colorado T-32 Research Fellow The problem. VTE - Scope of the Problem One of
VTE in the Trauma Population Erik Peltz, D.O. February 11 th, 2015 * contributions from Eduardo Gonzalez, M.D. University of Colorado T-32 Research Fellow The problem. VTE - Scope of the Problem One of
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #23 (NQF 0239): Perioperative Care: Venous Thromboembolism (VTE) Prophylaxis (When Indicated in ALL Patients) National Quality Strategy Domain: Patient Safety 2018 OPTIONS FOR INDIVIDUAL MEASURES:
Quality ID #23 (NQF 0239): Perioperative Care: Venous Thromboembolism (VTE) Prophylaxis (When Indicated in ALL Patients) National Quality Strategy Domain: Patient Safety 2018 OPTIONS FOR INDIVIDUAL MEASURES:
Clinical Policy: Dalteparin (Fragmin) Reference Number: ERX.SPA.207 Effective Date:
 Clinical Policy: (Fragmin) Reference Number: ERX.SPA.207 Effective Date: 01.11.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Fragmin) Reference Number: ERX.SPA.207 Effective Date: 01.11.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
DENOMINATOR: All surgical patients aged 18 years and older undergoing procedures for which VTE prophylaxis is indicated in all patients
 Measure #23 (NQF 0239): Perioperative Care: Venous Thromboembolism (VTE) Prophylaxis (When Indicated in ALL Patients) National Quality Strategy Domain: Patient Safety 2017 OPTIONS FOR INDIVIDUAL MEASURES:
Measure #23 (NQF 0239): Perioperative Care: Venous Thromboembolism (VTE) Prophylaxis (When Indicated in ALL Patients) National Quality Strategy Domain: Patient Safety 2017 OPTIONS FOR INDIVIDUAL MEASURES:
Primary VTE Prophylaxis. Ponlapat Rojnuckarin, MD PhD Chulalongkorn University Bangkok, Thailand
 Primary VTE Prophylaxis Ponlapat Rojnuckarin, MD PhD Chulalongkorn University Bangkok, Thailand A 70-yr-old female before THA BMI 31 kg/m 2 with varicose vein What do you recommend for VTE prevention?
Primary VTE Prophylaxis Ponlapat Rojnuckarin, MD PhD Chulalongkorn University Bangkok, Thailand A 70-yr-old female before THA BMI 31 kg/m 2 with varicose vein What do you recommend for VTE prevention?
Coversheet for Network Site Specific Group Agreed Documentation
 Coversheet for Network Site Specific Group Agreed Documentation This sheet is to accompany all documentation agreed by Pan Birmingham Cancer Network Site Specific Groups. This will assist the Network Governance
Coversheet for Network Site Specific Group Agreed Documentation This sheet is to accompany all documentation agreed by Pan Birmingham Cancer Network Site Specific Groups. This will assist the Network Governance
Preventing Blood Clots in Adult Patients
 Manchester Royal Eye Hospital Surgical Services Information for Patients Preventing Blood Clots in Adult Patients This leaflet will give you information on how to reduce the risk of developing blood clots
Manchester Royal Eye Hospital Surgical Services Information for Patients Preventing Blood Clots in Adult Patients This leaflet will give you information on how to reduce the risk of developing blood clots
THROMBOSIS RISK FACTOR ASSESSMENT
 Name: Procedure: Doctor: Date: THROMBOSIS RISK FACTOR ASSESSMENT CHOOSE ALL THAT APPLY EACH RISK FACTOR REPRESENTS 1 POINT Age 41 60 years Minor Surgery Planned History of Prior Major Surgery (< 1 month)
Name: Procedure: Doctor: Date: THROMBOSIS RISK FACTOR ASSESSMENT CHOOSE ALL THAT APPLY EACH RISK FACTOR REPRESENTS 1 POINT Age 41 60 years Minor Surgery Planned History of Prior Major Surgery (< 1 month)
SUBJECT: LIMB PNEUMATIC COMPRESSION EFFECTIVE DATE: 06/27/13 DEVICES FOR VENOUS REVISED DATE: 06/26/14, 09/15/15,09/21/17. THROMBOEMBOLISM PROPHYLAXIS
 MEDICAL POLICY REVISED DATE: 06/26/14, 09/15/15,09/21/17. PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases,
MEDICAL POLICY REVISED DATE: 06/26/14, 09/15/15,09/21/17. PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases,
Corporate Medical Policy
 Corporate Medical Policy Postsurgical Home Use of Limb Compression Devices for Venous File Name: Origination: Last CAP Review: Next CAP Review: Last Review: postsurgical_home_use_of_limb_ compression_devices_for_vte_prophylaxis
Corporate Medical Policy Postsurgical Home Use of Limb Compression Devices for Venous File Name: Origination: Last CAP Review: Next CAP Review: Last Review: postsurgical_home_use_of_limb_ compression_devices_for_vte_prophylaxis
VTE Prevention Guidelines (Venous Thromboembolism) (Venous Thromboembolism)
 VTE Prevention Guidelines (Venous Thromboembolism) (Venous Thromboembolism) When using this document please ensure that the version you are using is the most up to date either by checking on the Trust
VTE Prevention Guidelines (Venous Thromboembolism) (Venous Thromboembolism) When using this document please ensure that the version you are using is the most up to date either by checking on the Trust
Thromboprophylaxis in Adult General Medical Patients - Guidelines for Management
 Thromboprophylaxis in Adult General Medical Patients - Guidelines for Management Adapted from the Worcestershire Acute Hospitals NHS Trust Guideline WAHT-MED-010 Version: Final Ratified by: Provider Quality
Thromboprophylaxis in Adult General Medical Patients - Guidelines for Management Adapted from the Worcestershire Acute Hospitals NHS Trust Guideline WAHT-MED-010 Version: Final Ratified by: Provider Quality
GUIDELINE FOR THROMBOPROPHYLAXIS IN ADULT (18 YEARS AND OLDER) IN GENERAL MEDICAL PATIENTS AND INPATIENTS UNDERGOING SURGERY
 GUIDELINE FOR THROMBOPROPHYLAXIS IN ADULT (18 YEARS AND OLDER) IN GENERAL MEDICAL PATIENTS AND INPATIENTS UNDERGOING SURGERY SEE SEPARATE GUIDELINES FOR THROMBOPROPHYLAXIS IN OBSTETRICS This guidance does
GUIDELINE FOR THROMBOPROPHYLAXIS IN ADULT (18 YEARS AND OLDER) IN GENERAL MEDICAL PATIENTS AND INPATIENTS UNDERGOING SURGERY SEE SEPARATE GUIDELINES FOR THROMBOPROPHYLAXIS IN OBSTETRICS This guidance does
Linas Venclauskas, Juan V. Llau, Jean-Yves Jenny, Per Kjaersgaard-Andersen and Øivind Jans, for the ESA VTE Guidelines Task Force PROOF
 GUIDELINES European guidelines on perioperative venous thromboembolism prophylaxis Day surgery and fast-track surgery Linas Venclauskas, Juan V. Llau, Jean-Yves Jenny, Per Kjaersgaard-Andersen and Øivind
GUIDELINES European guidelines on perioperative venous thromboembolism prophylaxis Day surgery and fast-track surgery Linas Venclauskas, Juan V. Llau, Jean-Yves Jenny, Per Kjaersgaard-Andersen and Øivind
Venous Thromboembolism (VTE) Prevention
 Venous Thromboembolism (VTE) Prevention 7 VTE Risk Assessment: General Patient Population Assess VTE risk at admission, post-op, and transfer See page 2 for VTE risk assessment among Obstetrical (OB) patients
Venous Thromboembolism (VTE) Prevention 7 VTE Risk Assessment: General Patient Population Assess VTE risk at admission, post-op, and transfer See page 2 for VTE risk assessment among Obstetrical (OB) patients
Measurement and Improvement of Quality of Cardiovascular Care DR : DEHESTANI
 Measurement and Improvement of Quality of Cardiovascular Care DR : DEHESTANI Hospitals For hospitals in the United States, measures of cardiovascular care mandated by the Joint Commission have recently
Measurement and Improvement of Quality of Cardiovascular Care DR : DEHESTANI Hospitals For hospitals in the United States, measures of cardiovascular care mandated by the Joint Commission have recently
Hospital Acquired VTE: update on national guidance
 Hospital Acquired VTE: update on national guidance Rebecca Chanda Consultant Pharmacist - Thrombosis and Haemostasis Guy s and St Thomas Hospitals NHS Foundation Trust Chair of the UK Clinical Pharmacy
Hospital Acquired VTE: update on national guidance Rebecca Chanda Consultant Pharmacist - Thrombosis and Haemostasis Guy s and St Thomas Hospitals NHS Foundation Trust Chair of the UK Clinical Pharmacy
PREVENTION AND TREATMENT OF VENOUS THROMBOEMBOLISM
 PREVENTION AND TREATMENT OF VENOUS THROMBOEMBOLISM International Consensus Statement 2013 Guidelines According to Scientific Evidence Developed under the auspices of the: Cardiovascular Disease Educational
PREVENTION AND TREATMENT OF VENOUS THROMBOEMBOLISM International Consensus Statement 2013 Guidelines According to Scientific Evidence Developed under the auspices of the: Cardiovascular Disease Educational
Symptomatic Venous Thromboembolism after Total Hip/Knee Replacement: A Population-based Taiwan Study
 IMPROVING PATIENT SAFETY Preventing & Managing Venous Thromboembolism Session 8 Data Driving Strategies for VTE Prevention and Management 3/30/2012; 15.35-15.55 Symptomatic Venous Thromboembolism after
IMPROVING PATIENT SAFETY Preventing & Managing Venous Thromboembolism Session 8 Data Driving Strategies for VTE Prevention and Management 3/30/2012; 15.35-15.55 Symptomatic Venous Thromboembolism after
CANCER ASSOCIATED THROMBOSIS. Pankaj Handa Department of General Medicine Tan Tock Seng Hospital
 CANCER ASSOCIATED THROMBOSIS Pankaj Handa Department of General Medicine Tan Tock Seng Hospital My Talk Today 1.Introduction 2. Are All Cancer Patients at Risk of VTE? 3. Should All VTE Patients Be Screened
CANCER ASSOCIATED THROMBOSIS Pankaj Handa Department of General Medicine Tan Tock Seng Hospital My Talk Today 1.Introduction 2. Are All Cancer Patients at Risk of VTE? 3. Should All VTE Patients Be Screened
Duration of Anticoagulant Therapy. Linda R. Kelly PharmD, PhC, CACP September 17, 2016
 Duration of Anticoagulant Therapy Linda R. Kelly PharmD, PhC, CACP September 17, 2016 Conflicts of Interest No conflicts of interest to report Objectives At the end of the program participants will be
Duration of Anticoagulant Therapy Linda R. Kelly PharmD, PhC, CACP September 17, 2016 Conflicts of Interest No conflicts of interest to report Objectives At the end of the program participants will be
Prophylaxis for Hospitalized and Non-Hospitalized Medical Patients
 Prophylaxis for Hospitalized and Non-Hospitalized Medical Patients An Educational Slide Set American Society of Hematology 2018 Guidelines for Management of Venous Thromboembolism Slide set authors: Eric
Prophylaxis for Hospitalized and Non-Hospitalized Medical Patients An Educational Slide Set American Society of Hematology 2018 Guidelines for Management of Venous Thromboembolism Slide set authors: Eric
Deep vein thrombosis (DVT) and pulmonary embolism (PE) advice for ophthalmic surgery patients
 Deep vein thrombosis (DVT) and pulmonary embolism (PE) advice for ophthalmic surgery patients What is a deep vein thrombosis (DVT)? A DVT is a blood clot that forms within a vein deep in the leg but can
Deep vein thrombosis (DVT) and pulmonary embolism (PE) advice for ophthalmic surgery patients What is a deep vein thrombosis (DVT)? A DVT is a blood clot that forms within a vein deep in the leg but can
TRANSPARENCY COMMITTEE OPINION. 18 April 2007
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 18 April 2007 ARIXTRA 2.5 mg/0.5 ml, solution for injection in prefilled syringe Pack of 2 (CIP: 359 225-4) Pack of
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 18 April 2007 ARIXTRA 2.5 mg/0.5 ml, solution for injection in prefilled syringe Pack of 2 (CIP: 359 225-4) Pack of
Venous thromboembolism after total knee replacement or total hip replacement: what can be learnt from root-cause analysis?
 TRAUMA AND ORTHOPAEDIC SURGERY Ann R Coll Surg Engl 2016; 98: 538 542 doi 10.1308/rcsann.2016.0202 Venous thromboembolism after total knee replacement or total hip replacement: what can be learnt from
TRAUMA AND ORTHOPAEDIC SURGERY Ann R Coll Surg Engl 2016; 98: 538 542 doi 10.1308/rcsann.2016.0202 Venous thromboembolism after total knee replacement or total hip replacement: what can be learnt from
Venous Thromboembolism National Hospital Inpatient Quality Measures
 Venous Thromboembolism National Hospital Inpatient Quality Measures Presentation Overview Review venous thromboembolism as a new mandatory measure set Outline measures with exclusions and documentation
Venous Thromboembolism National Hospital Inpatient Quality Measures Presentation Overview Review venous thromboembolism as a new mandatory measure set Outline measures with exclusions and documentation
Clinical Policy: Dalteparin (Fragmin) Reference Number: ERX.SPA.207 Effective Date:
 Clinical Policy: (Fragmin) Reference Number: ERX.SPA.207 Effective Date: 01.11.17 Last Review Date: 02.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Fragmin) Reference Number: ERX.SPA.207 Effective Date: 01.11.17 Last Review Date: 02.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
CPT only copyright 2014 American Medical Association. All rights reserved. 12/23/2014 Page 66 of 593
 Measure #23 (NQF 0239): Perioperative Care: Venous Thromboembolism (VTE) Prophylaxis (When Indicated in ALL Patients) National Quality Strategy Domain: Patient Safety 2015 PQRS OPTIONS FOR INDIVIDUAL MEASURES:
Measure #23 (NQF 0239): Perioperative Care: Venous Thromboembolism (VTE) Prophylaxis (When Indicated in ALL Patients) National Quality Strategy Domain: Patient Safety 2015 PQRS OPTIONS FOR INDIVIDUAL MEASURES:
IF YOU RECEIVED THIS FACSIMILE IN ERROR, PLEASE CALL IMMEDIATELY. ADMISSION INSTRUCTIONS: Admitted to Dr.
 ADMISSION INSTRUCTIONS: Admitted to Dr. Procedure: Right Hip Left Hip CODE STATUS: Full DNR level of intervention DIET: Dysphagia screen consult SLP High protein, high energy Regular Nutrient dense Boost
ADMISSION INSTRUCTIONS: Admitted to Dr. Procedure: Right Hip Left Hip CODE STATUS: Full DNR level of intervention DIET: Dysphagia screen consult SLP High protein, high energy Regular Nutrient dense Boost
LIMB COMPRESSION DEVICES FOR VENOUS THROMBOEMBOLISM PROPHYLAXIS
 PROPHYLAXIS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs are dependent
PROPHYLAXIS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs are dependent
Venous Thrombo-Embolism (VTE)
 Venous Thrombo-Embolism (VTE) Information for service users and carers RDaSH leading the way with care Older People s Mental Health Services Reducing risk of unwanted blood clots whilst in hospital About
Venous Thrombo-Embolism (VTE) Information for service users and carers RDaSH leading the way with care Older People s Mental Health Services Reducing risk of unwanted blood clots whilst in hospital About
Slide 1. Slide 2. Slide 3. Outline of This Presentation
 Slide 1 Current Approaches to Venous Thromboembolism Prevention in Orthopedic Patients Hujefa Vora, MD Maria Fox, RN June 9, 2017 Slide 2 Slide 3 Outline of This Presentation Pathophysiology of venous
Slide 1 Current Approaches to Venous Thromboembolism Prevention in Orthopedic Patients Hujefa Vora, MD Maria Fox, RN June 9, 2017 Slide 2 Slide 3 Outline of This Presentation Pathophysiology of venous
Perioperative VTE Prophylaxis
 Perioperative VTE Prophylaxis Gregory J. Misky, M.D. Assistant Professor of Medicine University Of Colorado Denver You recommend the following 72 y.o. man admitted for an elective R hip repair. Patient
Perioperative VTE Prophylaxis Gregory J. Misky, M.D. Assistant Professor of Medicine University Of Colorado Denver You recommend the following 72 y.o. man admitted for an elective R hip repair. Patient
Venous Thrombosis in Asia
 Venous Thrombosis in Asia Pantep Angchaisuksiri, M.D. Professor of Medicine, Mahidol University, Thailand Adjunct Associate Professor, University of North Carolina, Chapel Hill, USA Venous Thromboembolism
Venous Thrombosis in Asia Pantep Angchaisuksiri, M.D. Professor of Medicine, Mahidol University, Thailand Adjunct Associate Professor, University of North Carolina, Chapel Hill, USA Venous Thromboembolism
Deep vein thrombosis and its prevention in critically ill adults Attia J, Ray J G, Cook D J, Douketis J, Ginsberg J S, Geerts W H
 Deep vein thrombosis and its prevention in critically ill adults Attia J, Ray J G, Cook D J, Douketis J, Ginsberg J S, Geerts W H Authors' objectives To systematically review the incidence of deep vein
Deep vein thrombosis and its prevention in critically ill adults Attia J, Ray J G, Cook D J, Douketis J, Ginsberg J S, Geerts W H Authors' objectives To systematically review the incidence of deep vein
DEEP VEIN THROMBOSIS (DVT): TREATMENT
 DEEP VEIN THROMBOSIS (DVT): TREATMENT OBJECTIVE: To provide an evidence-based approach to treatment of patients presenting with deep vein thrombosis (DVT). BACKGROUND: An estimated 45,000 patients in Canada
DEEP VEIN THROMBOSIS (DVT): TREATMENT OBJECTIVE: To provide an evidence-based approach to treatment of patients presenting with deep vein thrombosis (DVT). BACKGROUND: An estimated 45,000 patients in Canada
