Preclinical Evaluation of the Immunogenicity and Safety of an Inactivated Enterovirus 71 Candidate Vaccine
|
|
|
- Trevor Harrison
- 5 years ago
- Views:
Transcription
1 Preclinical Evaluation of the Immunogenicity and Safety of an Inactivated Enterovirus 71 Candidate Vaccine Shi-Hsia Hwa 1 *, Yock Ann Lee 1, Joseph N. Brewoo 2, Charalambos D. Partidos 2, Jorge E. Osorio 2,3, Joseph D. Santangelo 1 1 Inviragen (Singapore) Pte. Ltd., Singapore, Singapore, 2 Inviragen Inc., Madison, Wisconsin, United States of America, 3 Department of Pathobiological Sciences, School of Veterinary Medicine, University of Wisconsin, Madison, Madison, Wisconsin, United States of America Abstract Human enterovirus 71 (EV71) is a significant cause of morbidity and mortality from Hand, Foot and Mouth Disease (HFMD) and neurological complications, particularly in young children in the Asia-Pacific region. There are no vaccines or antiviral therapies currently available for prevention or treatment of HFMD caused by EV71. Therefore, the development of therapeutic and preventive strategies against HFMD is of growing importance. We report the immunogenic and safety profile of inactivated, purified EV71 preparations formulated with aluminum hydroxide adjuvant in preclinical studies in mice and rabbits. In mice, the candidate vaccine formulations elicited high neutralizing antibody responses. A toxicology study of the vaccine formulations planned for human use performed in rabbits showed no vaccine-related pathological changes and all animals remained healthy. Based on these preclinical studies, Phase 1 clinical testing of the EV71 inactivated vaccine was initiated. Citation: Hwa S-H, Lee YA, Brewoo JN, Partidos CD, Osorio JE, et al. (213) Preclinical Evaluation of the Immunogenicity and Safety of an Inactivated Enterovirus 71 Candidate Vaccine. PLoS Negl Trop Dis 7(11): e2538. doi:1.1371/journal.pntd.2538 Editor: David Joseph Diemert, The George Washington University Medical Center, United States of America Received October 26, 212; Accepted September 3, 213; Published November 7, 213 Copyright: ß 213 Hwa et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Funding: This work was supported by a RISC grant from the Singapore government. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Competing Interests: Authors SHH, YAL, JNB, CDP, JEO, and JDS are employees of Inviragen. This does not alter our adherence to all PLOS policies on sharing data and materials. * hwashihsia@gmail.com Introduction Hand, Foot, and Mouth Disease (HFMD) is caused by several members of the human enterovirus A (HEV-A) group. It is generally a self-limiting infection affecting mostly children and is characterized by ulcers and vesicles on the hands, feet and oral cavity. However, a more severe form of disease may occur with neurological symptoms such as meningitis, encephalitis, polio-like paralysis, and brain stem encephalitis leading to pulmonary edema and death [1 3]. Since the mid-199s, HFMD infections caused by human enterovirus 71 (EV71) have resulted in significant morbidity and mortality, particularly in the Asia-Pacific region [4,5]. China, Viet Nam, and Singapore reported increased activity in January May 212 compared to the same period in 211, including 22 deaths to date in Viet Nam [6]. In addition, HFMD outbreaks disrupt education and economic activities due to school and childcare center closures in efforts to control disease transmission [7]. Human enterovirus A belongs to the Picornaviridae family of nonenveloped, positive-sense RNA viruses, which also includes polioviruses and rhinoviruses. Members of the HEV-A group which cause HFMD include EV71 and Coxsackievirus A16 (CAV16) [8]. HFMD outbreaks due to EV71 infection have the greatest propensity to cause severe neurological disease. Experimental infection of cynomolgus macaques showed that strains isolated across several decades were all neurotropic, as well as showing a broader tissue tropism than polioviruses [9]. Enterovirus 71 has four capsid proteins (VP1 VP4) and seven nonstructural proteins. In addition to protecting the viral RNA, the capsid proteins recognize receptors on the surface of host cells and contribute to the antigenic profile of the virus [8]. Known human cell surface receptors for EV71 are the scavenger receptor B2 (SCARB2), and the P-selectin glycoprotein ligand 1 (PSGL-1) [1,11]. Although the classical method of typing enteroviruses by serum neutralization defines EV71 as a single serotype [12], current molecular typing methods reveal that several genogroups have been circulating in the Asia-Pacific region at least since the 199s [13]. EV71 isolates were previously classified into genogroups A, B, and C and sub-genogroups based on VP1 nucleotide sequences alone [14]; nucleotide sequence identity of the VP1 gene is.92% within genogroups, whereas nucleotide sequence identity between the genogroups is 78 83% [4]. However, whole-genome sequencing resulted in the reclassification of subgenogroup B5 under B4 and addition of genogroup D; the authors suggested that the 3D polymerase sequence together with VP1 better represented whole genomes [15]. Recombination between genogroups and with other Human enterovirus A serotypes also occurs [16]. At present there is no specific antiviral therapy or vaccine available against EV71. Intravenous immunoglobulin has been used in severe HFMD cases, with some therapeutic benefit suggested by the outcomes but as yet unproven by clinical trials [3,17]. Preventive and control measures during EV71 outbreaks are limited to surveillance, closure of educational and childcare facilities, and isolation of patients. Candidate vaccines under development include formaldehyde-inactivated whole-virus vaccine [18,19], VP1 subunit vaccines [2,21], a peptide-based PLOS Neglected Tropical Diseases 1 November 213 Volume 7 Issue 11 e2538
2 Author Summary Enterovirus 71 (EV71) is one of the major viruses causing Hand, Foot, and Mouth Disease, a highly contagious illness which primarily affects young children in the Asia-Pacific region and can sometimes be fatal. No vaccines or antivirals for Hand, Foot, and Mouth Disease are available at this time. We developed an experimental vaccine using inactivated, purified EV71 with an adjuvant to amplify the immune response. When this vaccine was tested in mice and rabbits, they produced large amounts of antibodies that could neutralize the virus. We were reasonably certain that the vaccine would be safe because rabbits given repeated high doses did not develop pathological lesions or clinical symptoms. Based on these results, we proceeded to test the safety of the vaccine in human adults. synthetic vaccine [22], a recombinant bacterial-vectored VP1- based vaccine [23], a plasmid DNA vaccine expressing VP1 [24], virus-like particles of EV71 [25], and a live attenuated vaccine [26]. Common findings include the necessity for an adjuvant [27], and the use of whole virus particles as opposed to recombinant proteins or short peptides alone [28]. Human clinical trials of inactivated virus vaccines producing high neutralizing antibody titers have been conducted in China [29,3], Taiwan [31], and Singapore (Inviragen, manuscript in progress). In the following studies, we describe the development of an inactivated EV71 candidate vaccine in animal models and evaluation of its immunogenicity and safety in preparation for human clinical trials. Materials and Methods Ethics statement Animal studies were performed at the following institutions and approved by the respective animal care and use committees: Alhydrogel adjuvant study at University of Wisconsin-Madison (approved by UW-Madison Institutional Animal Care and Use Committee, under USDA Animal Welfare Act); determination of optimum immunogenic dose at Shanghai Genomics (under Chinese regulatory guidelines); rabbit GLP (Good Laboratory Practice) toxicology study at Jai Research Foundation (approved by JRF Institutional Animal Ethics Committee, under Guidelines for Laboratory Animals Facility, CPCSEA, India). Viruses and cell culture The genogroup B EV71 strain, MS/7423/87 (GenBank accession number U ) was selected to prepare the candidate vaccine on the basis of amino acid sequence similarity to highly immunogenic strains as well as high yield in Vero cell culture. The candidate vaccine strain was grown in Vero cells (WHO Reference Cell Bank 1-87) in 1-tier cell factories as described below, using Dulbecco s Modified Eagle s Medium (DMEM; Sigma-Aldrich, United States) without serum. Small scale preparations for neutralization assays were grown in Vero clone E6 cells (ATCC CRL-1586) in tissue culture flasks (BD Biosciences, United States) using Minimum Essential Medium (MEM; Sigma-Aldrich, United States) with 2% fetal bovine serum (FBS; Biological Industries, Israel). Inactivated EV71 virus Confluent monolayers of Vero cells in 1-tier cell factories (Nalge Nunc International, United States) were infected with the EV71 vaccine strain at a multiplicity of infection (MOI) of.1 to.1. When cytopathic effect (CPE) was complete, the medium was harvested, clarified by filtration, followed by treatment with benzonase (Merck Chemicals, Germany) to remove host cell nucleic acid at 2 U/mL, 37uC for 24 hours. The clarified harvest was treated with freshly prepared binary ethyleneimine (BEI) at 1.5% v/v, 37uC for 6 hours to inactivate EV71 infectivity [32], followed by the addition of 15 mm sodium thiosulfate to hydrolyze unreacted ethylenimine. Inactivation of viral infectivity was confirmed at this stage by blind-passaging the material twice on Vero cells. Tissue culture medium components and low molecular weight proteins were partially removed by concentration and diafiltration against phosphate-buffered saline (Invitrogen, United States) containing.2% Tween 8 (Merck Chemicals, Germany) (PBST). The inactivated virus preparation was further purified by chromatography using ion-exchange and size-exclusion columns (details of the chromatography process are proprietary), concentrated using a 3 kda molecular weight cutoff membrane, and finally sterilized by filtration through a.2 mm membrane. Purified inactivated EV71 antigen was stored at 28uC in suspension in PBST. Characterization of purified inactivated EV71 antigen The presence and estimated purity of EV71 antigen in samples were evaluated by SDS-PAGE and Western blotting. Briefly, samples were heated to 95uC in lithium dodecyl sulfate buffer (Invitrogen) with beta-mercaptoethanol (Sigma-Aldrich) and fractionated on duplicate 4 12% PAGE gels (Invitrogen) in MES-SDS buffer. For purity estimation, one gel was stained with colloidal blue (Invitrogen) followed by image intensity calculations using Quantity One software (Bio-Rad). For detection of EV71 antigens, proteins were transferred to a PVDF membrane using the iblot semi-dry blotting system (Invitrogen) and stained for the presence of VP2 and VP using the monoclonal antibody 422-8D-4C-4D (Merck Millipore, catalogue number MAB979), followed by an HRP-conjugated anti-mouse IgG secondary antibody (DAKO) and DAB substrate (Sigma-Aldrich) to visualise bands. The physical form of the EV71 antigen present was examined by transmission electron microscopy using a phosphotungstic acid negative stain on carbon formvar grids (Electron Microscopy Services). Immunization procedures In the two following mouse studies, male BALB/c mice between 4 6 weeks old were used. Volumes of 1 ml per dose were administered by intramuscular (IM) injection in the hind leg as two injections of 5 ml in the same leg, due the limitations on the volume of a single injection in mice. Immunogenicity of EV71 antigen formulated with or without aluminum hydroxide: Groups of mice (n = 8) were injected on Days and 28 with the following doses of purified inactivated EV71 antigen (.12 mg,.6 mg, or 3. mg) in PBST only or with aluminum hydroxide (Alhydrogel 85, Brenntag Biosector, Denmark) at.5 mg (aluminium content) per dose. Two groups of control animals (n = 8) were injected with PBST or Alhydrogel (.5 mg per dose) using the same immunization protocol as above. Blood samples were collected on day, 28, 42, 56, 91, and 12; sera were stored at 22uC until testing for neutralizing activity. Determination of the optimum immunogenic dose: Groups of mice (n = 1) were immunized on days and 28 with the following doses of purified inactivated EV71:.12 mg,.6 mg, 3 mg, 9 mg, and 15 mg, formulated with Alhydrogel (.5 mg in a volume of 1 ml per dose). A control group received Alhydrogel alone (.5 mg per dose in a volume of 1 ml per dose). Blood samples PLOS Neglected Tropical Diseases 2 November 213 Volume 7 Issue 11 e2538
3 were collected on day, 28, and 56; sera were stored at 22uC until testing for neutralizing activity. The toxicology study was conducted according to Good Laboratory Practice (GLP) at the Jai Research Foundation, India. New Zealand White rabbits were used as the test species with females to males at a 1:1 ratio in each group. Low and high dose vaccines were formulated as planned for a Phase I human clinical trial, containing.6 mg and 3. mg EV71 antigen respectively, with.5 mg Alhydrogel in a.5 ml volume. Twenty rabbits received each vaccine formulation. Control groups received normal saline (PBS, n = 12) or.5 mg Alhydrogel alone (vaccine placebo, n = 16). Test articles were administered by IM injection in the left thigh. All animals received a booster injection of the same dose and route on day 28; the animals in the high dose group received a second booster on day 42. Animals were observed for morbidity and mortality at least twice daily during the treatment and observation periods, in addition to weekly clinical examinations. Parameters monitored were: Injection site reactogenicity (Draize numerical scoring system), rectal temperature, body mass, food consumption (grams/rabbit/week), ophthalmological examination, and respiratory rate. Half the animals of each sex in each group were sacrificed 2 days after the last immunization, either on Day 3 (low dose, saline placebo, and Alhydrogel placebo groups) or Day 44 (high dose group); the remaining animals were sacrificed on Day 56. Blood samples were collected on Days, 3, 28, 42 (high dose group only), and 56 and used for clinical chemistry, hematology, and EV71 neutralizing antibody assays. Gross pathology examinations were performed on all sacrificed animals. A complete necropsy and histopathology examinations were performed on organs from the animals sacrificed on Day 3 (saline placebo, Alhydrogel placebo, and vaccine low dose groups) or Day 44 (high dose group). Determination of neutralizing antibody responses Vero cells were seeded into 96-well microtiter plates at 1 4 cells per well in 1 ml growth medium (MEM+1% FBS). Individual serum samples were heat-inactivated at 56uC for 3 min. Twofold serial dilutions of serum samples in assay medium (MEM+2% FBS) were mixed with equal volumes of an EV71 (vaccine strain) suspension at 2 units of 5% tissue culture infectious dose (TCID 5 ) per ml and incubated at 37uC for 1.5 hours. 1 ml of each serum-virus mixture was added to three wells (final virus titre 1 TCID 5 per well). Each well was scored for CPE at 5 days post-infection. The end-point neutralizing titer was defined as the highest serum dilution in which at least two of the three replicates were negative for CPE. Seroconversion was defined as a reciprocal neutralizing titer $128 [33]. samples in Figure 1, right), was.8% in all batches using this process, typically 85 9% (calculations not shown). Typical yields from the process outlined above were <3 mg of protein per liter of harvest. Electron microscopy revealed a mixture of filled and empty icosahedral capsids (Figure 2), consistent with the observation of VP in Western blotting. A particle concentration estimate performed by mixing the purified inactivated EV71 with a polystyrene bead standard (micrograph not shown) was approximately 3E+1 particles/ml. Immunogenicity of EV71 antigen formulated with or without aluminum hydroxide In a preliminary experiment we compared the immunogenicity of EV71 antigen inactivated by heat (56uC for 6 hours) or chemically (BEI) in mice. The BEI-inactivated EV71 virus exhibited superior immunogenicity, inducing,6.4-fold higher neutralizing titres at 28 days post-immunization; the difference decreased to,1.5-fold higher at 56 days post-immunization (data not shown). Subsequently, we tested the immunogenicity of BEI-inactivated, purified EV71 preparations in mice at different concentrations with or without adjuvant. The presence of aluminum hydroxide produced higher seroconversion rates at 28 days after the first dose (Table 1). Neutralizing antibody titers in groups receiving antigen with adjuvant, compared to groups receiving antigen alone ranged from 11-to-23-fold higher at day 28 (after 1 dose), 4-to 6-fold higher at day 42 (after 2 doses) and 2.4-to 9-fold higher at day 56 (two weeks after 2nd dose) (Figure 3). The differences were significant at all dose levels (P,.5). Follow-up to 91 and 12 days showed that high titers were sustained and did not significantly decrease from Day 56 (Figure 3). Determination of the optimum immunogenic dose Following a single dose, only the 3 mg and the 15 mg dose levels elicited a high neutralizing antibody response by day 28 (Figure 4). Seroconversion rates for all groups were incomplete, ranging from 1% to 8% depending on the dose (Table 2). However, a Statistics Data from animal studies were compiled in Microsoft Excel and analyzed using Excel or Prism 5 (GraphPad, Inc). Student s t-test was used on log-transformed reciprocal neutralizing titers to compare immunogenicity between groups. P values #.5 were considered significant. Results Characterization of purified inactivated EV71 material Western blotting with a monoclonal antibody specific for EV71 VP2 and VP proteins confirmed the presence of EV71 antigen in purified inactivated material (samples from a typical batch shown in Figure 1, left). The estimated purity of EV71, based on image intensity measurements of colloidal blue-stained gels (same Figure 1. SDS-PAGE and Western blotting of purified inactivated EV71 antigen. Left: Western blot of inactivated harvest (H), concentrated inactivated harvest (C), and concentrated purified inactivated material (P). VP is found at 38 kda and VP2 at 28 kda. Right: Colloidal blue stain of the same samples. Molecular weight marker (M) is SeeBlue Plus2 (Invitrogen). doi:1.1371/journal.pntd.2538.g1 PLOS Neglected Tropical Diseases 3 November 213 Volume 7 Issue 11 e2538
4 Preclinical Evaluation of an EV71 Vaccine Figure 2. Transmission electron micrograph of purified inactivated EV71. Purified inactivated EV71 material is shown on a carbon formvar grid with phosphotungstic acid negative staining. Examples of full particles are indicated with arrows; examples of empty particles are indicated with arrowheads. The scale bar is 5 nm. doi:1.1371/journal.pntd.2538.g2 booster immunization induced significantly (P#.5) higher levels of neutralizing antibodies in all groups of animals (Figure 4). Twenty-eight days after the boost, only the lowest dose of.12 mg was significantly less immunogenic than the highest dose of 15 mg; doses from.6 mg and higher produced equivalent titers. In addition, 1% seroconversion was observed following the booster dose in animals receiving vaccine doses of 3 mg or higher (Table 2). findings were compiled by the CRO and reported to Inviragen. No morbidity or mortality occurred in any animals, and no skin reactions (i.e. no erythema or edema) at the site of injection were found in any animals. Hematology and clinical chemistry parameters remained within the normal range for NZW rabbits. Animals in the EV71 vaccine-treated groups exhibited no differences from the control groups in group averages of body mass, body mass change during the study period, or food consumption. At day 3, males treated with Alhydrogel placebo had higher thymus weights compared to the saline placebo group. At day 56, EV71 vaccine-treated females had higher adrenal, ovary/oviduct, and uterus weights compared to the Alhydrogel placebo group. However, no lesions of pathological Safety of the EV71 candidate inactivated vaccine A toxicological study using this animal model to test low and high dose vaccine formulations was conducted at a GLP contract research organization (CRO). Clinical and histological Table 1. Seroconversion rates in BALB/c mice immunized with purified inactivated EV71. Group Treatment a Day 28 Day 42 Day 56 % seroconverted (n) % seroconverted (n) % seroconverted (n) 1 PBST control 2.12 mg EV (1/8) 3.6 mg EV (1/8) 87.5 (7/8) 4 3 mg EV (3/8) 5 Alum control 6.12 mg EV71+Alum 37.5 (3/8) 7.6 mg EV71+Alum 8 3 mg EV71+Alum a PBST = Phosphate Buffered Saline +.2% (v/v) Tween 8, buffer base for purified inactivated EV71. Mice were immunized with inactivated virus with or without Alhydrogel at 28, 42 and 56 days post primary immunization. Seroconversion is defined as a neutralizing antibody titre of $128. doi:1.1371/journal.pntd.2538.t1 PLOS Neglected Tropical Diseases 4 November 213 Volume 7 Issue 11 e2538
5 Figure 3. Kinetics of serum neutralizing antibody responses to EV71 with or without adjuvant. Groups of BALB/c mice (n = 8) were immunized with purified inactivated EV71 with or without Alhydrogel, PBST-only control, or Alhydrogel control. Symbols represent mean of reciprocal neutralizing antibody titers from groups of 8 animals 6 standard deviation. Y-axis is plotted on a logarithmic scale. Dotted line shows titer of 128 (7 log 2 ). doi:1.1371/journal.pntd.2538.g3 significance were found on the reproductive or other internal organs. Muscular degeneration at the site of injection, with or without mononuclear cell/eosinophil infiltration, were observed in a sub-population of animals in all groups receiving formulations containing aluminum hydroxide including the Alhydrogel placebo group, but not in animals which received the saline placebo (Table 3). The lesions had healed or were markedly reduced by day 56, i.e. 28 days after the last immunization for the Alhydrogel placebo and low dose groups, and 14 days after the last immunization for the high dose group. Discussion Prompted by the successful control of poliovirus by formalininactivated vaccines, we produced a purified, inactivated, aluminum hydroxide-adjuvanted EV71 candidate vaccine. The inactivation method used leaves no detectable residual inactivant and completely eliminates viral infectivity within,24 hours while preserving antigenicity (data not shown), thus is more efficient than formalin inactivation, which takes several days at 37uC [34]. BEI also better preserves the immunogenicity of the viral particles compared to heat inactivation, possibly because heating EV71 to $5uC triggers a conformational change in the particles [35,36]. The purification process using ion-exchange and size-exclusion chromatography does not separate empty procapsids from RNAcontaining particles, or immature virions with VP from mature virions with cleaved VP2/VP4, therefore the purified inactivated antigen preparations contain a mixed population of particles. However, empty particles from other EV71 candidate vaccine strains are known to be immunogenic albeit inducing somewhat lower neutralizing titres than full particles, the differences in immunogenicity being lesser at higher doses [34]. We then evaluated immunogenicity of several vaccine formulations using this antigen preparation in mice, followed by a Figure 4. Dose titration of EV71 antigen. Groups of BALB/c mice (n = 1) were immunized with purified inactivated EV71 with Alhydrogel, or Alhydrogel control. Symbols represent mean of reciprocal neutralizing antibody titers from groups of 1 animals 6 standard deviation. Y-axis is plotted on a logarithmic scale. Dotted line shows titer of 128 (7 log 2 ). doi:1.1371/journal.pntd.2538.g4 repeated-dose toxicology study in rabbits. The route of administration, dose levels, dosing schedule, and selection of parameters to be monitored in the study were designed according to industry standard recommendations for evaluation of vaccine safety [37]; furthermore, the high dose group received an additional dose beyond the planned human dosing schedule. No clinical or hematological abnormalities, or pathological signs on internal organs were found. Injection site lesions were localised, microscopic, and transient, and consistent with aluminium salt-induced lesions reported by other investigators [38]. These lesions occurred in the Alhydrogel placebo group as well as the low and high dose Table 2. Seroconversion rates following varying doses of purified inactivated EV71. Group Dose Day 28 Day 56 % seroconverted (n) % seroconverted (n) 1 Alum control (/1) (/1) 2.12 mg EV71+Alum 1 (1/1) 5 (5/1) 3.6 mg EV71+Alum 2 (2/1) 9 (9/1) 4 3mg EV71+Alum 7 (7/1) 1 (1/1) 5 9mg EV71+Alum 4 (4/1) 1 (1/1) 6 15 mg EV71+Alum 8 (8/1) 1 (1/1) BALB/c mice were immunized with purified inactivated EV71 virus with Alhydrogel, at 28 days and 56 days post primary immunization. Seroconversion is defined as a neutralizing antibody titre of $128. doi:1.1371/journal.pntd.2538.t2 PLOS Neglected Tropical Diseases 5 November 213 Volume 7 Issue 11 e2538
6 Table 3. Muscular lesions at the site of injection in rabbit toxicological study, 2 days after the last immunization. Group Treatment Dose of EV71 (mg) Muscle Degeneration (animals/group) Without Infiltration With Infiltration Total a G1 Saline Placebo /12 /12 /12 G2 Alhydrogel Placebo b 4/16 2/16 6/16 G3 Vaccine Low Dose.6 4/2 6/2 1/2 G4 Vaccine High Dose 3. /2 8/2 8/2 a Values shown under total are the total number of observations of muscle degeneration with and without MNC/eosinophil infiltration. b The Alhydrogel Placebo group received an Alhydrogel-only adjuvant formulation. doi:1.1371/journal.pntd.2538.t3 EV71 vaccine groups, therefore they were considered to be due to the Alhydrogel adjuvant rather than EV71 antigen. The purified inactivated EV71 antigen preparation was shown to elicit neutralizing antibody responses to the homologous virus in mice, which were significantly increased by the addition of aluminum hydroxide adjuvant. Analysis of the neutralizing antibody responses induced by the various antigen concentrations established i) the requirement of two doses for optimum immunogenicity and reliable seroconversion, and ii) the selection of low (.6 mg antigen per dose) and high (3 mg) doses. Currently, the protective anti-ev71 neutralizing antibody titer has not been determined for humans. However, it is interesting to note that the neutralizing antibody titers elicited by the inactivated vaccine in animals were well over the target titer of 128 and were sustained at high levels for over 3 months after the last immunization. This titer was the minimum protective antibody dose in a neonatal mouse passive immunization-challenge model of EV71 infection [33]. Subsequent to the studies described in this paper, a Phase I human clinical trial was conducted in adult volunteers (Inviragen, manuscript in progress). A significant challenge in the development and evaluation of candidate EV71 vaccines has been the lack of a suitable animal model that mimics EV71 pathogenesis in humans. Although nonhuman primates are susceptible to EV71 infection [9,26], cost and animal welfare considerations are barriers to use. Some neonatal mouse models have been established [39 41], but most strains of mice become resistant to EV71 infection by a few days of age. The interferon receptor-deficient mouse strain AG129 has been found by other investigators to be susceptible to EV71 infection [42]. The candidate vaccine described in this paper was later tested in the AG129 model in both passive transfer and vaccination studies, demonstrating its efficacy for protecting against EV71 disease [43]. Adult mice that were immunized with 3 mg EV71 in Alhydrogel (antigen dose equivalent to the planned clinical high dose) were partially protected against morbidity and mortality following a primary vaccination, and fully protected following one booster. We did not extensively test cross-neutralization of heterologous strains of EV71 by animal serum in vitro; however, preliminary data from the Phase I clinical trial of this candidate vaccine showed increases following vaccination of neutralizing titers against the genogroup B2 vaccine strain in addition to genogroup B4, C2, C4, and C5 isolates (Inviragen, manuscript in progress). References 1. Huang CC, Liu CC, Chang YC, Chen CY, Wang ST, et al. (1999) Neurologic complications in children with enterovirus 71 infection. N Engl J Med 341: Chang LY, Huang YC, Lin TY (1998) Fulminant neurogenic pulmonary oedema with hand, foot, and mouth disease. Lancet 352: While neutralizing titers against heterologous strains may vary, there is not a straightforward relationship between antigenicity and the current system of genotyping especially among genogroup B and C strains [18,44], as some neutralizing epitopes appear to be conformational rather than linear [45]. Antisera raised against various strains have been reported to be broadly cross-neutralizing [18,46,47]. Molecular studies also show that isolates across all EV71 genogroups share high predicted amino acid sequence identity including at the immunodominant neutralizing epitope in VP1 [14,28]. The development of an inactivated EV71 vaccine has three potential advantages over live attenuated vaccines: i) unlike live attenuated vaccines, the virus cannot revert to a more pathogenic phenotype; ii) immune responses can be manipulated (e.g. T h 1 vs. T h 2) by the inclusion of appropriate adjuvant; and iii) production will be cost-effective and flexible since the virus can be easily grown to high titers in tissue culture cells at scale. The decision to introduce an EV71 vaccine in developing countries with limited financial resources will depend on the balance of the economic value of EV71 vaccination versus EV71 risk. For instance, a recent study has estimated that in China, based on the current EV71 risk, a vaccine with 7% efficacy would be cost-effective at USD 25 per dose for mass immunization [48]. Encouraging results from a Phase 3 study show that high efficacy is in fact achievable with an inactivated vaccine [3]. Efficacy of an inactivated vaccine against HFMD could be further improved by inclusion of a coxsackievirus A16 (CVA16) strain. CVA16 is another major cause of HFMD and cross-neutralization of CVA16 by anti-ev71 antiserum is low, likely due to sequence divergence at the VP1 neutralizing epitope and different conformation of the virion [28]. Acknowledgments Cell and virus culture and some of the serum neutralization assays were performed by Ping Ping Tong. Electrophoresis and Western blotting of purified inactivated EV71 antigen were performed by Jia Vern Loh. Author Contributions Conceived and designed the experiments: SHH JDS. Performed the experiments: SHH JNB. Analyzed the data: SHH JDS. Contributed reagents/materials/analysis tools: YAL. Wrote the paper: SHH JNB CDP JEO JDS. 3. Ooi MH, Wong SC, Mohan A, Podin Y, Perera D, et al. (29) Identification and validation of clinical predictors for the risk of neurological involvement in children with hand, foot, and mouth disease in Sarawak. BMCInfectDis9:3. PLOS Neglected Tropical Diseases 6 November 213 Volume 7 Issue 11 e2538
7 4. Solomon T, Lewthwaite P, Perera D, Cardosa MJ, McMinn P, et al. (21) Virology, epidemiology, pathogenesis, and control of enterovirus 71. Lancet Infect Dis 1(11): McMinn PC (22) An overview of the evolution of enterovirus 71 and its clinical and public health significance. FEMS Microbiol Rev 26: (212) WPRO Hand, Foot, and Mouth Disease Situation Update, 15 May 212. Western Pacific Regional Office of the World Health Organization. pp (212) Updates on HFMD Situation in Singapore (23 May 212). Hand, Foot & Mouth Disease: Updates: Singapore Ministry of Health. 8. Pallansch MA, Roos R (27) Enteroviruses: polioviruses, coxsackieviruses, and newer enteroviruses. In: Knipe DM, Howley PM, Griffin DE, editors. Fields Virology. Philadelphia, PA Lippincott Williams & Wilkins. pp Nagata N, Shimizu H, Ami Y, Tano Y, Harashima A, et al. (22) Pyramidal and extrapyramidal involvement in experimental infection of cynomolgus monkeys with enterovirus 71. J Med Virol 67: Yamayoshi S, Yamashita Y, Li J, Hanagata N, Minowa T, et al. (29) Scavenger receptor B2 is a cellular receptor for enterovirus 71. Nat Med 15: Nishimura Y, Shimojima M, Tano Y, Miyamura T, Wakita T, et al. (29) Human P-selectin glycoprotein ligand-1 is a functional receptor for enterovirus 71. Nat Med 15: Oberste MS, Maher K, Kilpatrick DR, Flemister MR, Brown BA, et al. (1999) Typing of human enteroviruses by partial sequencing of VP1. J Clin Microbiol 37: Lee MS, Tseng FC, Wang JR, Chi CY, Chong P, et al. (212) Challenges to licensure of enterovirus 71 vaccines. PLoS Negl Trop Dis 6: e Brown BA, Oberste MS, Alexander JP, Jr., Kennett ML, Pallansch MA (1999) Molecular epidemiology and evolution of enterovirus 71 strains isolated from 197 to J Virol 73: Chan YF, Sam IC, Abubakar S (29) Phylogenetic designation of enterovirus 71 genotypes and subgenotypes using complete genome sequences. Infect Genet Evol 1(3): Yoke-Fun C, AbuBakar S (26) Phylogenetic evidence for inter-typic recombination in the emergence of human enterovirus 71 subgenotypes. BMC Microbiol 6: Wang SM, Liu CC, Tseng HW, Wang JR, Huang CC, et al. (1999) Clinical spectrum of enterovirus 71 infection in children in southern Taiwan, with an emphasis on neurological complications. Clin Infect Dis 29: Ong KC, Devi S, Cardosa MJ, Wong KT (21) Formaldehyde-inactivated whole virus vaccine protects a murine model of Enterovirus 71 encephalomyelitis against disease. J Virol 84: Chong P, Hsieh SY, Liu CC, Chou AH, Chang JY, et al. (212) Production of EV71 vaccine candidates. Hum Vaccin Immunother 8: Chen HF, Chang MH, Chiang BL, Jeng ST (26) Oral immunization of mice using transgenic tomato fruit expressing VP1 protein from enterovirus 71. Vaccine 24: Meng T, Kolpe AB, Kiener TK, Chow VT, Kwang J (211) Display of VP1 on the surface of baculovirus and its immunogenicity against heterologous human enterovirus 71 strains in mice. PLoS ONE 6: e Liu JN, Wang W, Duo JY, Hao Y, Ma CM, et al. (21) Combined peptides of human enterovirus 71 protect against virus infection in mice. Vaccine 28: Chiu CH, Chu C, He CC, Lin TY (26) Protection of neonatal mice from lethal enterovirus 71 infection by maternal immunization with attenuated Salmonella enterica serovar Typhimurium expressing VP1 of enterovirus 71. Microbes Infect 8: Tung WS, Bakar SA, Sekawi Z, Rosli R (27) DNA vaccine constructs against enterovirus 71 elicit immune response in mice. Genet Vaccines Ther 5: Chung YC, Ho MS, Wu JC, Chen WJ, Huang JH, et al. (28) Immunization with virus-like particles of enterovirus 71 elicits potent immune responses and protects mice against lethal challenge. Vaccine 26: Arita M, Nagata N, Iwata N, Ami Y, Suzaki Y, et al. (27) An attenuated strain of enterovirus 71 belonging to genotype a showed a broad spectrum of antigenicity with attenuated neurovirulence in cynomolgus monkeys. J Virol 81: Mao Q, Dong C, Li X, Gao Q, Guo Z, et al. (212) Comparative analysis of the immunogenicity and protective effects of inactivated EV71 vaccines in mice. PLoS ONE 7: e Chou AH, Liu CC, Chang JY, Lien SP, Guo MS, et al. (212) Immunological Evaluation and Comparison of Different EV71 Vaccine Candidates. Clin Dev Immunol 212: Li YP, Liang ZL, Gao Q, Huang LR, Mao QY, et al. (212) Safety and immunogenicity of a novel human Enterovirus 71 (EV71) vaccine: a randomized, placebo-controlled, double-blind, Phase I clinical trial. Vaccine 3: Zhu FC, Meng FY, Li JX, Li XL, Mao QY, et al. (213) Efficacy, safety, and immunology of an inactivated alum-adjuvant enterovirus 71 vaccine in children in China: a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 381: Cheng A, Fung CP, Liu CC, Lin YT, Tsai HY, et al. (213) A Phase I, randomized, open-label study to evaluate the safety and immunogenicity of an enterovirus 71 vaccine. Vaccine 31: Bahnemann HG (199) Inactivation of viral antigens for vaccine preparation with particular reference to the application of binary ethylenimine. Vaccine 8: Yu CK, Chen CC, Chen CL, Wang JR, Liu CC, et al. (2) Neutralizing antibody provided protection against enterovirus type 71 lethal challenge in neonatal mice. J Biomed Sci 7: Liu CC, Guo MS, Lin FH, Hsiao KN, Chang KH, et al. (211) Purification and characterization of enterovirus 71 viral particles produced from vero cells grown in a serum-free microcarrier bioreactor system. PLoS One 6: e Shingler KL, Yoder JL, Carnegie MS, Ashley RE, Makhov AM, et al. (213) The Enterovirus 71 A-particle Forms a Gateway to Allow Genome Release: A CryoEM Study of Picornavirus Uncoating. PLoS Pathog 9: e Curry S, Chow M, Hogle JM (1996) The poliovirus 135S particle is infectious. J Virol 7: Verdier F (22) Non-clinical vaccine safety assessment. Toxicology 174: Verdier F, Burnett R, Michelet-Habchi C, Moretto P, Fievet-Groyne F, et al. (25) Aluminium assay and evaluation of the local reaction at several time points after intramuscular administration of aluminium containing vaccines in the Cynomolgus monkey. Vaccine 23: Wang YF, Chou CT, Lei HY, Liu CC, Wang SM, et al. (24) A mouseadapted enterovirus 71 strain causes neurological disease in mice after oral infection. J Virol 78: Wang W, Duo J, Liu J, Ma C, Zhang L, et al. (211) A mouse muscle-adapted enterovirus 71 strain with increased virulence in mice. Microbes Infect 13: Huang SW, Wang YF, Yu CK, Su IJ, Wang JR (212) Mutations in VP2 and VP1 capsid proteins increase infectivity and mouse lethality of enterovirus 71 by virus binding and RNA accumulation enhancement. Virology 422: Khong WX, Yan B, Yeo H, Tan EL, Lee JJ, et al. (211) A non mouse-adapted Enterovirus 71 (EV71) strain exhibits neurotropism causing neurological manifestations in a novel mouse model of EV71 infection. J Virol 86(4): Caine EA, Partidos CD, Santangelo JD, Osorio JE (213) Adaptation of enterovirus 71 to adult interferon deficient mice. PLoS ONE 8: e Huang ML, Chiang PS, Chia MY, Luo ST, Chang LY, et al. (213) Crossreactive neutralizing antibody responses to enterovirus 71 infections in young children: implications for vaccine development. PLoS Negl Trop Dis 7: e Chen Y, Li C, He D, Cheng T, Ge S, et al. (213) Antigenic analysis of divergent genotypes human Enterovirus 71 viruses by a panel of neutralizing monoclonal antibodies: current genotyping of EV71 does not reflect their antigenicity. Vaccine 31: Kung SH, Wang SF, Huang CW, Hsu CC, Liu HF, et al. (27) Genetic and antigenic analyses of enterovirus 71 isolates in Taiwan during Clin Microbiol Infect 13: Mizuta K, Aoki Y, Suto A, Ootani K, Katsushima N, et al. (29) Crossantigenicity among EV71 strains from different genogroups isolated in Yamagata, Japan, between 199 and 27. Vaccine 27: Lee BY, Wateska AR, Bailey RR, Tai JH, Bacon KM, et al. (21) Forecasting the economic value of an Enterovirus 71 (EV71) vaccine. Vaccine 28: PLOS Neglected Tropical Diseases 7 November 213 Volume 7 Issue 11 e2538
DOI: /ICJ poliovirus. [14] (Hand-Foot-Mouth Disease, HFMD) (Herpangina) 71 [26] [17,18,27] 71 picornaviridae
![DOI: /ICJ poliovirus. [14] (Hand-Foot-Mouth Disease, HFMD) (Herpangina) 71 [26] [17,18,27] 71 picornaviridae DOI: /ICJ poliovirus. [14] (Hand-Foot-Mouth Disease, HFMD) (Herpangina) 71 [26] [17,18,27] 71 picornaviridae](/thumbs/93/112612467.jpg) 174 DOI: 10.6526/ICJ.2016.404 71 71 [14] (Hand-Foot-Mouth Disease, HFMD) (Herpangina) 71 71 picornaviridae poliovirus / 1990 71 [26] [17,18,27] 71 175 [31] ICR NOD/SCID AG129 hpsgl-1 hscarb2 (MP4) (MP4)
174 DOI: 10.6526/ICJ.2016.404 71 71 [14] (Hand-Foot-Mouth Disease, HFMD) (Herpangina) 71 71 picornaviridae poliovirus / 1990 71 [26] [17,18,27] 71 175 [31] ICR NOD/SCID AG129 hpsgl-1 hscarb2 (MP4) (MP4)
IgM. (Polioviruses) 71 (EV71) B (Coxsackievirus B) (Virus isolation/ifa, VI-IFA) 7~14 [1,2] (Centers for Disease Control and Prevention, CDC) 1.
![IgM. (Polioviruses) 71 (EV71) B (Coxsackievirus B) (Virus isolation/ifa, VI-IFA) 7~14 [1,2] (Centers for Disease Control and Prevention, CDC) 1. IgM. (Polioviruses) 71 (EV71) B (Coxsackievirus B) (Virus isolation/ifa, VI-IFA) 7~14 [1,2] (Centers for Disease Control and Prevention, CDC) 1.](/thumbs/93/111050187.jpg) 267 DOI: 10.6526/ICJ.2017.603 (Polioviruses) 71 (EV71) B (Coxsackievirus B) [1,2] 1998 EV71 (Centers for Disease Control and Prevention, CDC) 1. (Hand, foot and mouth disease, HFMD) (herpangina) [3,4]
267 DOI: 10.6526/ICJ.2017.603 (Polioviruses) 71 (EV71) B (Coxsackievirus B) [1,2] 1998 EV71 (Centers for Disease Control and Prevention, CDC) 1. (Hand, foot and mouth disease, HFMD) (herpangina) [3,4]
Adaptation of Enterovirus 71 to Adult Interferon Deficient Mice
 Adaptation of Enterovirus 71 to Adult Interferon Deficient Mice Elizabeth A. Caine 1 *, Charalambos D. Partidos 2, Joseph D. Santangelo 3, Jorge E. Osorio 1,2 1 Department of Pathobiological Sciences,
Adaptation of Enterovirus 71 to Adult Interferon Deficient Mice Elizabeth A. Caine 1 *, Charalambos D. Partidos 2, Joseph D. Santangelo 3, Jorge E. Osorio 1,2 1 Department of Pathobiological Sciences,
Sequence analysis for VP4 of enterovirus 71 isolated in Beijing during 2007 to 2008
 16 2009 3 4 1 Journal of Microbes and Infection, March 2009, Vol. 4, No. 1 2007 2008 71 VP4 1, 2, 2, 2, 1, 2, 2, 2, 1, 2 1., 100730; 2., 100020 : 2007 2008 71 ( EV71), 2007 3 EV71( 1, 2 ) 2008 5 EV71(
16 2009 3 4 1 Journal of Microbes and Infection, March 2009, Vol. 4, No. 1 2007 2008 71 VP4 1, 2, 2, 2, 1, 2, 2, 2, 1, 2 1., 100730; 2., 100020 : 2007 2008 71 ( EV71), 2007 3 EV71( 1, 2 ) 2008 5 EV71(
Control and Management of EV 71 Associated HFMD
 Control and Management of EV 71 Associated HFMD Amr Ismail MBBCh, MSc, MD, MACP, FISQua, FABAM, CHPI Consultant Dermatologist Objectives To overview EV 71 associated HFMD outbreaks. To describe prevention
Control and Management of EV 71 Associated HFMD Amr Ismail MBBCh, MSc, MD, MACP, FISQua, FABAM, CHPI Consultant Dermatologist Objectives To overview EV 71 associated HFMD outbreaks. To describe prevention
Enterovirus 71 Outbreak in P. R. China, 2008
 JCM Accepts, published online ahead of print on 13 May 2009 J. Clin. Microbiol. doi:10.1128/jcm.00563-09 Copyright 2009, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JCM Accepts, published online ahead of print on 13 May 2009 J. Clin. Microbiol. doi:10.1128/jcm.00563-09 Copyright 2009, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
Original Article Relationship between serologic response and clinical symptoms in children with enterovirus 71-infected hand-foot-mouth disease
 Int J Clin Exp Pathol 2015;8(9):11608-11614 www.ijcep.com /ISSN:1936-2625/IJCEP0009527 Original Article Relationship between serologic response and clinical symptoms in children with enterovirus 71-infected
Int J Clin Exp Pathol 2015;8(9):11608-11614 www.ijcep.com /ISSN:1936-2625/IJCEP0009527 Original Article Relationship between serologic response and clinical symptoms in children with enterovirus 71-infected
A comparative study of multiple clinical enterovirus 71 isolates and evaluation of cross protection of inactivated vaccine strain FY-23 K-B in vitro
 Yang et al. Virology Journal (2017) 14:206 DOI 10.1186/s12985-017-0872-8 RESEARCH Open Access A comparative study of multiple clinical enterovirus 71 isolates and evaluation of cross protection of inactivated
Yang et al. Virology Journal (2017) 14:206 DOI 10.1186/s12985-017-0872-8 RESEARCH Open Access A comparative study of multiple clinical enterovirus 71 isolates and evaluation of cross protection of inactivated
Prevalence of Antibodies against Enterovirus 71 in Children from Lu'an City in Central China
 Jpn. J. Infect. Dis., 64, 528-532, 2011 Epidemiological Report Prevalence of Antibodies against Enterovirus 71 in Children from Lu'an City in Central China Haiyang Yu 1,2, Mingli Wang 1,2, Hongwei Chang
Jpn. J. Infect. Dis., 64, 528-532, 2011 Epidemiological Report Prevalence of Antibodies against Enterovirus 71 in Children from Lu'an City in Central China Haiyang Yu 1,2, Mingli Wang 1,2, Hongwei Chang
Formalin-Inactivated EV71 Vaccine Candidate Induced Cross-Neutralizing Antibody against Subgenotypes B1, B4, B5 and C4A in Adult Volunteers
 Formalin-Inactivated EV71 Vaccine Candidate Induced Cross-Neutralizing Antibody against Subgenotypes B1, B4, B5 and C4A in Adult Volunteers Ai-Hsiang Chou 1., Chia-Chyi Liu 1., Jui-Yuan Chang 1, Renee
Formalin-Inactivated EV71 Vaccine Candidate Induced Cross-Neutralizing Antibody against Subgenotypes B1, B4, B5 and C4A in Adult Volunteers Ai-Hsiang Chou 1., Chia-Chyi Liu 1., Jui-Yuan Chang 1, Renee
What is the role of animal models in studying protective titres and the need for establishing surrogates/correlates of protection?
 What is the role of animal models in studying protective titres and the need for establishing surrogates/correlates of protection? Alan D.T. Barrett Department of Pathology and Sealy Center for Vaccine
What is the role of animal models in studying protective titres and the need for establishing surrogates/correlates of protection? Alan D.T. Barrett Department of Pathology and Sealy Center for Vaccine
Biomedical Research 2017; 28 (20): ISSN X
 Biomedical Research 2017; 28 (20): 8755-8759 ISSN 0970-938X www.biomedres.info Detection of serum immune factors in children with hand-foot-and-mouth disease (HFMD) and the clinical significance. Lieping
Biomedical Research 2017; 28 (20): 8755-8759 ISSN 0970-938X www.biomedres.info Detection of serum immune factors in children with hand-foot-and-mouth disease (HFMD) and the clinical significance. Lieping
Zheng, BJ; Du, LY; Zhao, GY; Lin, YP; Sui, HY; Chan, C; Ma, S; Guan, Y; Yuen, KY. Citation Hong Kong Medical Journal, 2008, v. 14 suppl. 4, p.
 Title Studies of SARS virus vaccines Author(s) Zheng, BJ; Du, LY; Zhao, GY; Lin, YP; Sui, HY; Chan, C; Ma, S; Guan, Y; Yuen, KY Citation Hong Kong Medical Journal, 2008, v. 14 suppl. 4, p. 39-43 Issued
Title Studies of SARS virus vaccines Author(s) Zheng, BJ; Du, LY; Zhao, GY; Lin, YP; Sui, HY; Chan, C; Ma, S; Guan, Y; Yuen, KY Citation Hong Kong Medical Journal, 2008, v. 14 suppl. 4, p. 39-43 Issued
RISK FACTORS FOR SEVERE HAND, FOOT AND MOUTH DISEASE
 RISK FACTORS FOR SEVERE HAND, FOOT AND MOUTH DISEASE Somchai Owatanapanich 1, Rochana Wutthanarungsan 1, Wipaporn Jaksupa 1 and Usa Thisyakorn 2 1 King Narai Hospital, Lopburi; 2 Faculty of Medicine, Chulalongkorn
RISK FACTORS FOR SEVERE HAND, FOOT AND MOUTH DISEASE Somchai Owatanapanich 1, Rochana Wutthanarungsan 1, Wipaporn Jaksupa 1 and Usa Thisyakorn 2 1 King Narai Hospital, Lopburi; 2 Faculty of Medicine, Chulalongkorn
Pre-clinical Development of a Dengue Vaccine. Jeremy Brett Sanofi Pasteur, Singapore
 Pre-clinical Development of a Dengue Vaccine Jeremy Brett Sanofi Pasteur, Singapore Dengue Vaccine B Guy 1 Talk flow Introduction: What are the challenges of dengue vaccine development? The Virus The host
Pre-clinical Development of a Dengue Vaccine Jeremy Brett Sanofi Pasteur, Singapore Dengue Vaccine B Guy 1 Talk flow Introduction: What are the challenges of dengue vaccine development? The Virus The host
Changes in serum cytokine levels in hand-foot-and-mouth disease.
 Biomedical Research 2015; 26 (3): 594-598 ISSN 0970-938X www.biomedres.info Changes in serum cytokine levels in hand-foot-and-mouth disease. Tong-Zeng Li, Tai-Yi Jiang, Lian-Chun Liang 3 rd Department
Biomedical Research 2015; 26 (3): 594-598 ISSN 0970-938X www.biomedres.info Changes in serum cytokine levels in hand-foot-and-mouth disease. Tong-Zeng Li, Tai-Yi Jiang, Lian-Chun Liang 3 rd Department
Biotechnology-Based Vaccines. Dr. Aws Alshamsan Department of Pharmaceutics Office: AA87 Tel:
 Biotechnology-Based Vaccines Dr. Aws Alshamsan Department of Pharmaceutics Office: AA87 Tel: 4677363 aalshamsan@ksu.edu.sa Objectives of this lecture By the end of this lecture you will be able to: 1.
Biotechnology-Based Vaccines Dr. Aws Alshamsan Department of Pharmaceutics Office: AA87 Tel: 4677363 aalshamsan@ksu.edu.sa Objectives of this lecture By the end of this lecture you will be able to: 1.
Broad protection with an inactivated vaccine against primary-isolated lethal enterovirus 71 infection in newborn mice
 Chang et al. BMC Microbiology (2015) 15:139 DOI 10.1186/s12866-015-0474-9 RESEARCH ARTICLE Open Access Broad protection with an inactivated vaccine against primary-isolated lethal enterovirus 71 infection
Chang et al. BMC Microbiology (2015) 15:139 DOI 10.1186/s12866-015-0474-9 RESEARCH ARTICLE Open Access Broad protection with an inactivated vaccine against primary-isolated lethal enterovirus 71 infection
Clinical Trials of Pandemic Vaccines: Key Issues. John Treanor University of Rochester Rochester, NY
 Clinical Trials of Pandemic Vaccines: Key Issues John Treanor University of Rochester Rochester, NY Inactivated vaccine approach Proven technology Used successfully in 1957 and 1968 Abundant efficacy data
Clinical Trials of Pandemic Vaccines: Key Issues John Treanor University of Rochester Rochester, NY Inactivated vaccine approach Proven technology Used successfully in 1957 and 1968 Abundant efficacy data
Vaccine 31 (2013) Contents lists available at SciVerse ScienceDirect. Vaccine. jou rn al h om epa ge:
 Vaccine 31 (2013) 2215 2221 Contents lists available at SciVerse ScienceDirect Vaccine jou rn al h om epa ge: www.elsevier.com/locate/vaccine Active immunization with a Coxsackievirus A16 experimental
Vaccine 31 (2013) 2215 2221 Contents lists available at SciVerse ScienceDirect Vaccine jou rn al h om epa ge: www.elsevier.com/locate/vaccine Active immunization with a Coxsackievirus A16 experimental
Enterovirus 71 infection and vaccines
 Review article CLINICAL EXPERIMENTAL VACCINE RESEARCH Enterovirus 71 infection and vaccines Clin Exp Vaccine Res 2017;6:4-14 pissn 2287-3651 eissn 2287-366X Eun-Je Yi 1, Yun-Ju Shin 1, Jeong-Hwan Kim 1,
Review article CLINICAL EXPERIMENTAL VACCINE RESEARCH Enterovirus 71 infection and vaccines Clin Exp Vaccine Res 2017;6:4-14 pissn 2287-3651 eissn 2287-366X Eun-Je Yi 1, Yun-Ju Shin 1, Jeong-Hwan Kim 1,
Evidence for Other Non-Poliovirus Enteroviruses Multiplies in L20B Cells. ACCEPTED LUIS SARMIENTO*, PEDRO MÁS, ROSA PALOMERA, LUIS MORIER, MAGILÉ
 CVI Accepts, published online ahead of print on 14 March 2007 Clin. Vaccine Immunol. doi:10.1128/cvi.00017-06 Copyright 2007, American Society for Microbiology and/or the Listed Authors/Institutions. All
CVI Accepts, published online ahead of print on 14 March 2007 Clin. Vaccine Immunol. doi:10.1128/cvi.00017-06 Copyright 2007, American Society for Microbiology and/or the Listed Authors/Institutions. All
Genetic Diversity of Coxsackievirus A16 Associated with Hand, Foot, and Mouth Disease Epidemics in Japan from 1983 to 2003
 JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2007, p. 112 120 Vol. 45, No. 1 0095-1137/07/$08.00 0 doi:10.1128/jcm.00718-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Genetic Diversity
JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2007, p. 112 120 Vol. 45, No. 1 0095-1137/07/$08.00 0 doi:10.1128/jcm.00718-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Genetic Diversity
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates
 HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
Change of Major Genotype of Enterovirus 71 in Outbreaks of Hand-Foot-and-Mouth Disease in Taiwan between 1998 and 2000
 JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2002, p. 10 15 Vol. 40, No. 1 0095-1137/02/$04.00 0 DOI: 10.1128/JCM.40.1.10 15.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Change
JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2002, p. 10 15 Vol. 40, No. 1 0095-1137/02/$04.00 0 DOI: 10.1128/JCM.40.1.10 15.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Change
Supporting Information
 Supporting Information Valkenburg et al. 10.1073/pnas.1403684111 SI Materials and Methods ELISA and Microneutralization. Sera were treated with Receptor Destroying Enzyme II (RDE II, Accurate) before ELISA
Supporting Information Valkenburg et al. 10.1073/pnas.1403684111 SI Materials and Methods ELISA and Microneutralization. Sera were treated with Receptor Destroying Enzyme II (RDE II, Accurate) before ELISA
E E Hepatitis E SARS 29, Lancet. E A B Enterically-Transmitted Non-A, Hepatitis E. Virus HEV nm. 1.35g/cm s ALT AST HEV HEV
 7850 2004 Hepatitis E Tian-Cheng LI Naokazu TAKEDA Tatsuo MIYAMURA SARS 8 Lancet E E E Hepatitis E VirusHEV E E HEV HEV E 1955 29,000 E E A A B Enterically-Transmitted Non-A, Non-B Hepatitis 1983 Balayan
7850 2004 Hepatitis E Tian-Cheng LI Naokazu TAKEDA Tatsuo MIYAMURA SARS 8 Lancet E E E Hepatitis E VirusHEV E E HEV HEV E 1955 29,000 E E A A B Enterically-Transmitted Non-A, Non-B Hepatitis 1983 Balayan
Chang Gung University co-commissioned final report. Research Ttitle: Antiviral mechanism study for 254 UVC robot system
 co-commissioned final report Research Ttitle: Antiviral mechanism study for 254 UVC robot system Project/Research Number:: Execution duration: 2014.10.16-2015.01.16 Principle Investigator: Professor Shin-Ru
co-commissioned final report Research Ttitle: Antiviral mechanism study for 254 UVC robot system Project/Research Number:: Execution duration: 2014.10.16-2015.01.16 Principle Investigator: Professor Shin-Ru
G. W. WOOD J. C. MUSKETT and D. H. THORNTON MAFF, Central Veterinary Laboratory, New Haw, Weybridge, Surrey, U.K.
 J. Comp. Path. 1986 vol. 96 OBSERVATIONS ON THE ABILITY OF AVIAN REOVIRUS VACCINMATION OF HENS TO PROTECT THEIR PROGENY AGAINST THE EFFECTS OF CHALLENGE WITH HOMOLOGOUS AND HETEROLOGOUS STRAINS By G. W.
J. Comp. Path. 1986 vol. 96 OBSERVATIONS ON THE ABILITY OF AVIAN REOVIRUS VACCINMATION OF HENS TO PROTECT THEIR PROGENY AGAINST THE EFFECTS OF CHALLENGE WITH HOMOLOGOUS AND HETEROLOGOUS STRAINS By G. W.
Foot and Mouth Disease Vaccine Research and Development in India
 Foot and Mouth Disease Vaccine Research and Development in India R.Venkataramanan Indian Veterinary Research Institute, Hebbal, Bangalore 560 024 Foot and Mouth Disease in India Present Status Large Susceptible
Foot and Mouth Disease Vaccine Research and Development in India R.Venkataramanan Indian Veterinary Research Institute, Hebbal, Bangalore 560 024 Foot and Mouth Disease in India Present Status Large Susceptible
Is a multivalent hand, foot, and mouth disease vaccine feasible?
 Human Vaccines & Immunotherapeutics ISSN: 2164-5515 (Print) 2164-554X (Online) Journal homepage: https://www.tandfonline.com/loi/khvi20 Is a multivalent hand, foot, and mouth disease vaccine feasible?
Human Vaccines & Immunotherapeutics ISSN: 2164-5515 (Print) 2164-554X (Online) Journal homepage: https://www.tandfonline.com/loi/khvi20 Is a multivalent hand, foot, and mouth disease vaccine feasible?
SURVEILLANCE TECHNICAL
 CHAPTER 5 SURVEILLANCE TECHNICAL ASPECTS 55 Protect - detect - protect Polio eradication strategies can be summed up as protect and detect protect children against polio by vaccinating them, and detect
CHAPTER 5 SURVEILLANCE TECHNICAL ASPECTS 55 Protect - detect - protect Polio eradication strategies can be summed up as protect and detect protect children against polio by vaccinating them, and detect
Enterovirus 71 infection in children with hand, foot, and mouth disease in Shanghai, China: epidemiology, clinical feature and diagnosis
 Wang et al. Virology Journal (2015) 12:83 DOI 10.1186/s12985-015-0308-2 RESEARCH Enterovirus 71 infection in children with hand, foot, and mouth disease in Shanghai, China: epidemiology, clinical feature
Wang et al. Virology Journal (2015) 12:83 DOI 10.1186/s12985-015-0308-2 RESEARCH Enterovirus 71 infection in children with hand, foot, and mouth disease in Shanghai, China: epidemiology, clinical feature
Introduction. In the past 15 years, several technological advancements have open new perspectives and applications in the field of vaccinology.
 Introduction In the past 15 years, several technological advancements have open new perspectives and applications in the field of vaccinology. - Genomics: fasten antigen discovery for complex pathogens
Introduction In the past 15 years, several technological advancements have open new perspectives and applications in the field of vaccinology. - Genomics: fasten antigen discovery for complex pathogens
Gene Vaccine Dr. Sina Soleimani
 Gene Vaccine Dr. Sina Soleimani Human Viral Vaccines Quality Control Laboratory (HVVQC) Titles 1. A short Introduction of Vaccine History 2. First Lineage of Vaccines 3. Second Lineage of Vaccines 3. New
Gene Vaccine Dr. Sina Soleimani Human Viral Vaccines Quality Control Laboratory (HVVQC) Titles 1. A short Introduction of Vaccine History 2. First Lineage of Vaccines 3. Second Lineage of Vaccines 3. New
Long persistence of EV71 specific nucleotides in respiratory and feces samples of the patients with Hand-Foot-Mouth Disease after recovery
 RESEARCH ARTICLE Open Access Research article Long persistence of EV71 specific nucleotides in respiratory and feces samples of the patients with Hand-Foot-Mouth Disease after recovery Jun Han 1, Xue-Jun
RESEARCH ARTICLE Open Access Research article Long persistence of EV71 specific nucleotides in respiratory and feces samples of the patients with Hand-Foot-Mouth Disease after recovery Jun Han 1, Xue-Jun
Received 15 November 2015; received in revised form 6 December 2015; accepted 8 December 2015
 Tropical Biomedicine 33(2): 238 245 (2016) Diagnosis of human enterovirus A71 infection in Malaysia using a commercial IgM-capture enzyme-linked immunosorbent assay and an IgM-colloidal gold immunochromatographic
Tropical Biomedicine 33(2): 238 245 (2016) Diagnosis of human enterovirus A71 infection in Malaysia using a commercial IgM-capture enzyme-linked immunosorbent assay and an IgM-colloidal gold immunochromatographic
Nature Medicine: doi: /nm.4322
 1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. Predicted RNA structure of 3 UTR and sequence alignment of deleted nucleotides. (a) Predicted RNA secondary structure of ZIKV 3 UTR. The stem-loop structure
1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. Predicted RNA structure of 3 UTR and sequence alignment of deleted nucleotides. (a) Predicted RNA secondary structure of ZIKV 3 UTR. The stem-loop structure
A THESIS SUBMITTED FOR THE MASTERS OF SCIENCE IN LIFE SCIENCES
 RATIONAL DESIGN OF LIVE ATTENUATED VACCINE BY SITE-DIRECTED MUTAGENESIS AND DELETION IN THE 5 -NON CODING REGION OF THE ENTEROVIRUS 71 (EV-A71) GENOME ISABEL YEE PINN TSIN B.Sc (Honours) (Biochemistry),
RATIONAL DESIGN OF LIVE ATTENUATED VACCINE BY SITE-DIRECTED MUTAGENESIS AND DELETION IN THE 5 -NON CODING REGION OF THE ENTEROVIRUS 71 (EV-A71) GENOME ISABEL YEE PINN TSIN B.Sc (Honours) (Biochemistry),
Cross-Reactivity to Field Isolates of Canine Influenza Virus by a Killed Canine Influenza Virus (H3N8, Iowa05) Vaccine
 Cross-Reactivity to Field Isolates of Canine Influenza Virus by a Killed Canine Influenza Virus (H3N8, Iowa05) Vaccine Nancee Oien, B.S., M.S. a Sally Mattern, B.S a Jaime Brozowski, B.S., M.S. b Janet
Cross-Reactivity to Field Isolates of Canine Influenza Virus by a Killed Canine Influenza Virus (H3N8, Iowa05) Vaccine Nancee Oien, B.S., M.S. a Sally Mattern, B.S a Jaime Brozowski, B.S., M.S. b Janet
A Novel Recombinant Virus Reagent Products for Efficient Preparation Of Hepatitis B Animal Models
 About FivePlus Beijing FivePlus Molecular Medicine Institute was established in 2005. The company has been dedicating itself to continuous innovation of viral vectors. The meaning of FivePlus is based
About FivePlus Beijing FivePlus Molecular Medicine Institute was established in 2005. The company has been dedicating itself to continuous innovation of viral vectors. The meaning of FivePlus is based
Influenza or flu is a
 Clinical and Research Area Infectious Diseases Influenza Virus Types A and B Influenza or flu is a respiratory illness that is caused by influenza viruses. Influenza viruses type A and type B cause seasonal
Clinical and Research Area Infectious Diseases Influenza Virus Types A and B Influenza or flu is a respiratory illness that is caused by influenza viruses. Influenza viruses type A and type B cause seasonal
Author's response to reviews
 Author's response to reviews Title: Sentinel surveillance for human enterovirus 71 in Sarawak, Malaysia: Lessons from the first 7 years. Authors: Yuwana Podin (ypodin@yahoo.com) Edna LM Gias (amberr74@yahoo.com)
Author's response to reviews Title: Sentinel surveillance for human enterovirus 71 in Sarawak, Malaysia: Lessons from the first 7 years. Authors: Yuwana Podin (ypodin@yahoo.com) Edna LM Gias (amberr74@yahoo.com)
Effect of Complement and Viral Filtration on the
 APPLIED MICROBIOLOGY, JUlY 1968, p. 1076-1080 Copyright @ 1968 American Society for Microbiology Vol. 16, No. 7 Printed in U.S.A. Effect of Complement and Viral Filtration on the Neutralization of Respiratory
APPLIED MICROBIOLOGY, JUlY 1968, p. 1076-1080 Copyright @ 1968 American Society for Microbiology Vol. 16, No. 7 Printed in U.S.A. Effect of Complement and Viral Filtration on the Neutralization of Respiratory
Hand -foot- mouth disease (HFMD), one of the more distinctive rash syndromes, is most
 Continuing Education Column Hand-Foot- Mouth Disease Related to Enterovirus 71 Jong-Hyun Kim, MD Seong -Joon Kim, MD Doo-Sung Cheon, PhD Department of Childhood and Adolescent Medicine, College of Medicine,
Continuing Education Column Hand-Foot- Mouth Disease Related to Enterovirus 71 Jong-Hyun Kim, MD Seong -Joon Kim, MD Doo-Sung Cheon, PhD Department of Childhood and Adolescent Medicine, College of Medicine,
Development of influenza vaccine production by means of chromatographic methods
 Development of influenza vaccine production by means of chromatographic methods WHO meeting on prospects for influenza vaccine technology transfer to vaccine manufacturers of developing countries. 27-28
Development of influenza vaccine production by means of chromatographic methods WHO meeting on prospects for influenza vaccine technology transfer to vaccine manufacturers of developing countries. 27-28
Int.J.Curr.Microbiol.App.Sci (2018) 7(8):
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 7 Number 08 (2018) Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2018.708.471
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 7 Number 08 (2018) Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2018.708.471
ADCC Assay Protocol Vikram Srivastava 1, Zheng Yang 1, Ivan Fan Ngai Hung 2, Jianqing Xu 3, Bojian Zheng 3 and Mei- Yun Zhang 3*
 ADCC Assay Protocol Vikram Srivastava 1, Zheng Yang 1, Ivan Fan Ngai Hung 2, Jianqing Xu 3, Bojian Zheng 3 and Mei- Yun Zhang 3* 1 Department of Microbiology, Li Ka Shing Faculty of Medicine, University
ADCC Assay Protocol Vikram Srivastava 1, Zheng Yang 1, Ivan Fan Ngai Hung 2, Jianqing Xu 3, Bojian Zheng 3 and Mei- Yun Zhang 3* 1 Department of Microbiology, Li Ka Shing Faculty of Medicine, University
DOI: /ICJ (Dengue fever) [2] (Aedes aegypti) (Aedes albopictus) (Dengue. (Flavivirus) 50 nm. ssrna) (capsid, C) ( ) (membrane, prm/m)
![DOI: /ICJ (Dengue fever) [2] (Aedes aegypti) (Aedes albopictus) (Dengue. (Flavivirus) 50 nm. ssrna) (capsid, C) ( ) (membrane, prm/m) DOI: /ICJ (Dengue fever) [2] (Aedes aegypti) (Aedes albopictus) (Dengue. (Flavivirus) 50 nm. ssrna) (capsid, C) ( ) (membrane, prm/m)](/thumbs/91/106127266.jpg) 210 DOI: 10.6526/ICJ.2016.504 T (Dengue fever) [1] 128 39 [2] 4 5 21,000 [3] 3 1981 1987 1995 2014 [4] 2015 ( ) (Aedes aegypti) (Aedes albopictus) (Dengue virus) (Flaviviridae) (Flavivirus) 50 nm 11 kb
210 DOI: 10.6526/ICJ.2016.504 T (Dengue fever) [1] 128 39 [2] 4 5 21,000 [3] 3 1981 1987 1995 2014 [4] 2015 ( ) (Aedes aegypti) (Aedes albopictus) (Dengue virus) (Flaviviridae) (Flavivirus) 50 nm 11 kb
SPECIFIC ANTIINFECTIOUS IMMUNITY. colostral immunity. administration of antibodies VIRULENT VACCINE INACTIVATED VACCINE
 INDUCTION OF IMMUNITY AGAINST INFECTION SPECIFIC ANTIINFECTIOUS IMMUNITY active passive S AND VACCINATION infection vaccination colostral immunity administration of antibodies VIRULENT MODIFIED LIVE INACTIVATED
INDUCTION OF IMMUNITY AGAINST INFECTION SPECIFIC ANTIINFECTIOUS IMMUNITY active passive S AND VACCINATION infection vaccination colostral immunity administration of antibodies VIRULENT MODIFIED LIVE INACTIVATED
The humoral immune responses to IBV proteins.
 The humoral immune responses to IBV proteins. E. Dan Heller and Rosa Meir The Hebrew University of Jerusalem, Israel COST FA1207 meeting WG2 + WG3, Budapest, Jan. 2015 1 IBV encodes four major structural
The humoral immune responses to IBV proteins. E. Dan Heller and Rosa Meir The Hebrew University of Jerusalem, Israel COST FA1207 meeting WG2 + WG3, Budapest, Jan. 2015 1 IBV encodes four major structural
IMMUNOGENICITY OF FORMALDYDE INACTIVATED NEWCASTLE DISEASE VIRUS FIELD ISOLATE IN MATERNAL ANTIBODY FREE CHICKENS
 IMMUNOGENICITY OF FORMALDYDE INACTIVATED NEWCASTLE DISEASE VIRUS FIELD ISOLATE IN MATERNAL ANTIBODY FREE CHICKENS Anak Agung Ayu Mirah Adi 1 *, IGusti Agung Arta Putra 2, Nyoman Mantik Astawa 3, I Made
IMMUNOGENICITY OF FORMALDYDE INACTIVATED NEWCASTLE DISEASE VIRUS FIELD ISOLATE IN MATERNAL ANTIBODY FREE CHICKENS Anak Agung Ayu Mirah Adi 1 *, IGusti Agung Arta Putra 2, Nyoman Mantik Astawa 3, I Made
Test Report. Efficacy of A New JM Nanocomposite Material in Inhibiting Respiratory Syncytial Virus Cellular Infection
 Test Report Efficacy of A New JM Nanocomposite Material in Inhibiting Respiratory Syncytial Virus Cellular Infection Test Reagent New JM Nanocomposite Material Project Commissioner JM Material Technology,
Test Report Efficacy of A New JM Nanocomposite Material in Inhibiting Respiratory Syncytial Virus Cellular Infection Test Reagent New JM Nanocomposite Material Project Commissioner JM Material Technology,
Principles of Vaccination
 Immunology and Vaccine-Preventable Diseases Immunology is a complicated subject, and a detailed discussion of it is beyond the scope of this text. However, an understanding of the basic function of the
Immunology and Vaccine-Preventable Diseases Immunology is a complicated subject, and a detailed discussion of it is beyond the scope of this text. However, an understanding of the basic function of the
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/35908 holds various files of this Leiden University dissertation Author: Soema, Peter Title: Formulation of influenza T cell peptides : in search of a universal
Cover Page The handle http://hdl.handle.net/1887/35908 holds various files of this Leiden University dissertation Author: Soema, Peter Title: Formulation of influenza T cell peptides : in search of a universal
GOVX-B11: A Clade B HIV Vaccine for the Developed World
 GeoVax Labs, Inc. 19 Lake Park Drive Suite 3 Atlanta, GA 3 (678) 384-72 GOVX-B11: A Clade B HIV Vaccine for the Developed World Executive summary: GOVX-B11 is a Clade B HIV vaccine targeted for use in
GeoVax Labs, Inc. 19 Lake Park Drive Suite 3 Atlanta, GA 3 (678) 384-72 GOVX-B11: A Clade B HIV Vaccine for the Developed World Executive summary: GOVX-B11 is a Clade B HIV vaccine targeted for use in
Case Report Pathological examinations of an enterovirus 71 infection: an autopsy case
 Int J Clin Exp Pathol 2014;7(8):5236-5241 www.ijcep.com /ISSN:1936-2625/IJCEP0001082 Case Report Pathological examinations of an enterovirus 71 infection: an autopsy case Lulu Gao 1, Peixin Lin 1, Shuguang
Int J Clin Exp Pathol 2014;7(8):5236-5241 www.ijcep.com /ISSN:1936-2625/IJCEP0001082 Case Report Pathological examinations of an enterovirus 71 infection: an autopsy case Lulu Gao 1, Peixin Lin 1, Shuguang
Development and Application of a Novel Attenuated Live Japanese Encephalitis Vaccine SA
 Development and Application of a Novel Attenuated Live Japanese Encephalitis Vaccine SA14-14-2 Yu Yongxin (National Institute for the Control of Pharmaceutical and Biological Products, Beijing 100050,
Development and Application of a Novel Attenuated Live Japanese Encephalitis Vaccine SA14-14-2 Yu Yongxin (National Institute for the Control of Pharmaceutical and Biological Products, Beijing 100050,
ORIGINAL ARTICLE /j x
 ORIGINAL ARTICLE 1.1111/j.1469-691.24.19.x Chronological evolution of,, and neutralisation antibodies after infection with SARS-associated coronavirus P.-R. Hsueh 1,2, L.-M. Huang 3, P.-J. Chen 2, C.-L.
ORIGINAL ARTICLE 1.1111/j.1469-691.24.19.x Chronological evolution of,, and neutralisation antibodies after infection with SARS-associated coronavirus P.-R. Hsueh 1,2, L.-M. Huang 3, P.-J. Chen 2, C.-L.
<20 <20 <20 < <20 <20 <20 <20. Mock
 Cross-Lineage Neutralization PRNT 80 Titers Asian Asian West African Indian Ocean Group NHP Strain 181/25 Strain 99659 Strain 37997 Strain LR 142590 80 80 20 40 EILV/CHIKV 150844 640 640 160 320 Mock 150849
Cross-Lineage Neutralization PRNT 80 Titers Asian Asian West African Indian Ocean Group NHP Strain 181/25 Strain 99659 Strain 37997 Strain LR 142590 80 80 20 40 EILV/CHIKV 150844 640 640 160 320 Mock 150849
New Technology in Vaccine Engineering
 Viruses in May Katoomba, August, 2012 New Technology in Vaccine Engineering Anton Middelberg Australian Institute for Bioengineering and Nanotechnology The University of Queensland, Australia Introduction
Viruses in May Katoomba, August, 2012 New Technology in Vaccine Engineering Anton Middelberg Australian Institute for Bioengineering and Nanotechnology The University of Queensland, Australia Introduction
New prospects for vaccination: from polio to dengue and flu
 New prospects for vaccination: from polio to dengue and flu From ancient Chinese variolation to Jenner and cowpox Paul Young Australian Infectious Diseases Research Centre School of Chemistry and Molecular
New prospects for vaccination: from polio to dengue and flu From ancient Chinese variolation to Jenner and cowpox Paul Young Australian Infectious Diseases Research Centre School of Chemistry and Molecular
Canadian Immunization Conference 2018 Dec 4
 Enveloped Virus-Like Particle (evlp) Cytomegalovirus (CMV) Vaccine is immunogenic and safe: preliminary results of a First-in-Humans Canadian Immunization Network (CIRN) Clinical Trials Network (CTN) -
Enveloped Virus-Like Particle (evlp) Cytomegalovirus (CMV) Vaccine is immunogenic and safe: preliminary results of a First-in-Humans Canadian Immunization Network (CIRN) Clinical Trials Network (CTN) -
Development of a Recombinant Subunit Dengue Vaccine. Flavivirus Vaccination Fondation Mérieux December 8, 2010 Beth-Ann Coller
 Development of a Recombinant Subunit Dengue Vaccine Flavivirus Vaccination Fondation Mérieux December 8, 2010 Beth-Ann Coller Recombinant Subunit Dengue Vaccine Recombinant Envelope Protein Vaccine Hawaii
Development of a Recombinant Subunit Dengue Vaccine Flavivirus Vaccination Fondation Mérieux December 8, 2010 Beth-Ann Coller Recombinant Subunit Dengue Vaccine Recombinant Envelope Protein Vaccine Hawaii
Identification of Microbes Lecture: 12
 Diagnostic Microbiology Identification of Microbes Lecture: 12 Electron Microscopy 106 virus particles per ml required for visualization, 50,000-60,000 magnification normally used. Viruses may be detected
Diagnostic Microbiology Identification of Microbes Lecture: 12 Electron Microscopy 106 virus particles per ml required for visualization, 50,000-60,000 magnification normally used. Viruses may be detected
Epidemiology and Control of Hand, Foot and Mouth Disease in Singapore,
 16 Original Article Epidemiology and Control of Hand, Foot and Mouth Disease in Singapore, 21-27 Li Wei Ang, 1 Msc (Statistics), Benjamin KW Koh, 2 MBBS, MPH, Kwai Peng Chan, 3 MBBS, Dip Bact, FRCPA, Lian
16 Original Article Epidemiology and Control of Hand, Foot and Mouth Disease in Singapore, 21-27 Li Wei Ang, 1 Msc (Statistics), Benjamin KW Koh, 2 MBBS, MPH, Kwai Peng Chan, 3 MBBS, Dip Bact, FRCPA, Lian
Engineering Foot-and-Mouth Disease Virus with Improved Properties for the Development of Effective Vaccine Introduction: Materials and methods:
 Engineering Foot-and-Mouth Disease Virus with Improved Properties for the Development of Effective Vaccine Haixue Zheng, Fan Yang, Ye Jin, Jianhong Guo, Jijun He, Kaiqi Lian, Zixiang Zhu, Weijun Cao, Lvlv,
Engineering Foot-and-Mouth Disease Virus with Improved Properties for the Development of Effective Vaccine Haixue Zheng, Fan Yang, Ye Jin, Jianhong Guo, Jijun He, Kaiqi Lian, Zixiang Zhu, Weijun Cao, Lvlv,
An update on the laboratory detection and epidemiology of astrovirus, adenovirus, sapovirus, and enterovirus in gastrointestinal disease
 An update on the laboratory detection and epidemiology of astrovirus, adenovirus, sapovirus, and enterovirus in gastrointestinal disease Christopher McIver, Principal Hospital Scientist, Microbiology Department
An update on the laboratory detection and epidemiology of astrovirus, adenovirus, sapovirus, and enterovirus in gastrointestinal disease Christopher McIver, Principal Hospital Scientist, Microbiology Department
1. Engineering Foot-and-Mouth Disease Viruses with Improved
 Engineering Foot-and-Mouth Disease Virus with Improved Properties for the Development of Effective Vaccine Haixue Zheng, Fan Yang, Ye Jin, Jianhong Guo, Jijun He, Lvlv, Xuepeng Cai, Xiangtao Liu, Hong
Engineering Foot-and-Mouth Disease Virus with Improved Properties for the Development of Effective Vaccine Haixue Zheng, Fan Yang, Ye Jin, Jianhong Guo, Jijun He, Lvlv, Xuepeng Cai, Xiangtao Liu, Hong
Polio vaccines and polio immunization in the pre-eradication era: WHO position paper. Published in WER 4 June, 2010
 Polio vaccines and polio immunization in the pre-eradication era: WHO position paper Published in WER 4 June, 2010 Epidemiology & Background Poliomyelitis (polio) is an acute communicable disease of humans
Polio vaccines and polio immunization in the pre-eradication era: WHO position paper Published in WER 4 June, 2010 Epidemiology & Background Poliomyelitis (polio) is an acute communicable disease of humans
Measles is a chilhood disease caused
 Paediatrica Indonesiana VOLUME 48 May 2008 NUMBER 3 Original Article Antigenic differences between wildtype measles viruses and vaccine viruses in Indonesia Made Setiawan 1, Agus Sjahrurachman 2, Fera
Paediatrica Indonesiana VOLUME 48 May 2008 NUMBER 3 Original Article Antigenic differences between wildtype measles viruses and vaccine viruses in Indonesia Made Setiawan 1, Agus Sjahrurachman 2, Fera
Animal hosts Natural host Laboratory animals Rabbits Mice Rats Hamsters Newborn or suckling rodents Animal models for viral pathogenesis 4 Growth of v
 Principles of Virology Department of Molecular Genetics & Microbiology Univ ersity of Florida, Gainesv ille, FL 1 Outline Virus cultivation Assay of viruses Virus genetics 2 Virus isolation Evidence of
Principles of Virology Department of Molecular Genetics & Microbiology Univ ersity of Florida, Gainesv ille, FL 1 Outline Virus cultivation Assay of viruses Virus genetics 2 Virus isolation Evidence of
Hand Foot and Mouth Disease Due to Enterovirus 71 in Malaysia
 VIROLOGICA SINICA, August 2011, 26 (4): 221-228 DOI 10.1007/s12250-011-3195-8 Wuhan Institute of Virology, CAS and Springer-Verlag Berlin Heidelberg 2011 Hand Foot and Mouth Disease Due to Enterovirus
VIROLOGICA SINICA, August 2011, 26 (4): 221-228 DOI 10.1007/s12250-011-3195-8 Wuhan Institute of Virology, CAS and Springer-Verlag Berlin Heidelberg 2011 Hand Foot and Mouth Disease Due to Enterovirus
Necrotizing myositis causes restrictive hypoventilation in a mouse model for human enterovirus 71 infection
 Xiu et al. Virology Journal 2013, 10:215 RESEARCH Necrotizing myositis causes restrictive hypoventilation in a mouse model for human enterovirus 71 infection Jing-hui Xiu 1, Hao Zhu 1, Yan-feng Xu 2, Jiang-ning
Xiu et al. Virology Journal 2013, 10:215 RESEARCH Necrotizing myositis causes restrictive hypoventilation in a mouse model for human enterovirus 71 infection Jing-hui Xiu 1, Hao Zhu 1, Yan-feng Xu 2, Jiang-ning
Chapter 5. Virus isolation and identification of measles and rubella in cell culture
 Chapter 5. Virus isolation and identification of measles and rubella in cell culture In this chapter: 5.1. Recommended cell line for measles and rubella virus isolation 5.2. Propagation of Vero/hSLAM cells
Chapter 5. Virus isolation and identification of measles and rubella in cell culture In this chapter: 5.1. Recommended cell line for measles and rubella virus isolation 5.2. Propagation of Vero/hSLAM cells
Original Article Cellular immunity status and cytokine assay in enterovirus 71-infected children
 Int J Clin Exp Med 2018;11(9):9362-9370 www.ijcem.com /ISSN:1940-5901/IJCEM0071954 Original Article Cellular immunity status and cytokine assay in enterovirus 71-infected children Zhongwei Yin 1,2, Ting
Int J Clin Exp Med 2018;11(9):9362-9370 www.ijcem.com /ISSN:1940-5901/IJCEM0071954 Original Article Cellular immunity status and cytokine assay in enterovirus 71-infected children Zhongwei Yin 1,2, Ting
Phylogenetic Analysis of Enterovirus 71 Circulating in Beijing, China from 2007 to 2009
 Phylogenetic Analysis of Enterovirus 71 Circulating in Beijing, China from 2007 to 2009 Junping Zhu 1, Zhen Luo 2, Juan Wang 1, Zigang Xu 2, Hui Chen 1, Dongying Fan 1, Na Gao 1, Guoling Ping 1, Zhen Zhou
Phylogenetic Analysis of Enterovirus 71 Circulating in Beijing, China from 2007 to 2009 Junping Zhu 1, Zhen Luo 2, Juan Wang 1, Zigang Xu 2, Hui Chen 1, Dongying Fan 1, Na Gao 1, Guoling Ping 1, Zhen Zhou
Active and Passive Immunization for Avian Influenza Virus Infections
 NIAID Active and Passive Immunization for Avian Influenza Virus Infections Kanta Subbarao, MD, MPH Laboratory of Infectious Diseases NIAID, NIH Immortalizing H5 HA-Specific Memory B Cells Collection of
NIAID Active and Passive Immunization for Avian Influenza Virus Infections Kanta Subbarao, MD, MPH Laboratory of Infectious Diseases NIAID, NIH Immortalizing H5 HA-Specific Memory B Cells Collection of
Determination Of Thermal Stability Of Oral Polio Vaccine (Opv) At Different Temperature Under Laboratory Conditions
 Determination Of Thermal Stability Of Oral Polio Vaccine (Opv) At Different Temperature Under Laboratory Conditions Muhammad T 1, SS Baba 2, LT Zaria 2, AD El-Yuguda 2 And IB Thilza 3, 1 who National Polio
Determination Of Thermal Stability Of Oral Polio Vaccine (Opv) At Different Temperature Under Laboratory Conditions Muhammad T 1, SS Baba 2, LT Zaria 2, AD El-Yuguda 2 And IB Thilza 3, 1 who National Polio
Severity of enterovirus A71 infection in a human SCARB2 knock-in mouse model is dependent on infectious strain and route
 Zhu et al. Emerging Microbes & Infections (2018) 7:205 Emerging Microbes & Infections https://doi.org/10.1038/s41426-018-0201-3 www.nature.com/emi ARTICLE Severity of enterovirus A71 infection in a human
Zhu et al. Emerging Microbes & Infections (2018) 7:205 Emerging Microbes & Infections https://doi.org/10.1038/s41426-018-0201-3 www.nature.com/emi ARTICLE Severity of enterovirus A71 infection in a human
Rabies virus-like particles expressed in HEK293 cells
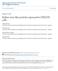 Engineering Conferences International ECI Digital Archives Vaccine Technology IV Proceedings Spring 5-21-2012 Rabies virus-like particles expressed in HEK293 cells Diego Fontana Cell Culture Laboratory
Engineering Conferences International ECI Digital Archives Vaccine Technology IV Proceedings Spring 5-21-2012 Rabies virus-like particles expressed in HEK293 cells Diego Fontana Cell Culture Laboratory
Vincent Racaniello
 Vincent Racaniello vrr1@columbia.edu www.virology.ws Poliomyelitis Polio (grey), myelon (marrow) = Greek itis (inflammation of) = Latin A common, acute viral disease characterized clinically by a brief
Vincent Racaniello vrr1@columbia.edu www.virology.ws Poliomyelitis Polio (grey), myelon (marrow) = Greek itis (inflammation of) = Latin A common, acute viral disease characterized clinically by a brief
Supplementary Figure 1. ALVAC-protein vaccines and macaque immunization. (A) Maximum likelihood
 Supplementary Figure 1. ALVAC-protein vaccines and macaque immunization. (A) Maximum likelihood tree illustrating CRF01_AE gp120 protein sequence relationships between 107 Envs sampled in the RV144 trial
Supplementary Figure 1. ALVAC-protein vaccines and macaque immunization. (A) Maximum likelihood tree illustrating CRF01_AE gp120 protein sequence relationships between 107 Envs sampled in the RV144 trial
The Development of Chronic Hepatitis in Rabbits Experimentally Infected. with HEV Isolate from Rabbit
 The Development of Chronic Hepatitis in Rabbits Experimentally Infected with HEV Isolate from Rabbit Peking University, Beijing, China Ling Wang October 7 2014 Hepatitis E Virus (HEV) Characteristics Faecal-oral
The Development of Chronic Hepatitis in Rabbits Experimentally Infected with HEV Isolate from Rabbit Peking University, Beijing, China Ling Wang October 7 2014 Hepatitis E Virus (HEV) Characteristics Faecal-oral
Chapter 6- An Introduction to Viruses*
 Chapter 6- An Introduction to Viruses* *Lecture notes are to be used as a study guide only and do not represent the comprehensive information you will need to know for the exams. 6.1 Overview of Viruses
Chapter 6- An Introduction to Viruses* *Lecture notes are to be used as a study guide only and do not represent the comprehensive information you will need to know for the exams. 6.1 Overview of Viruses
Coronaviruses cause acute, mild upper respiratory infection (common cold).
 Coronaviruses David A. J. Tyrrell Steven H. Myint GENERAL CONCEPTS Clinical Presentation Coronaviruses cause acute, mild upper respiratory infection (common cold). Structure Spherical or pleomorphic enveloped
Coronaviruses David A. J. Tyrrell Steven H. Myint GENERAL CONCEPTS Clinical Presentation Coronaviruses cause acute, mild upper respiratory infection (common cold). Structure Spherical or pleomorphic enveloped
The Infectious Cycle. Lecture 2 Biology W3310/4310 Virology Spring You know my methods, Watson --SIR ARTHUR CONAN DOYLE
 The Infectious Cycle Lecture 2 Biology W3310/4310 Virology Spring 2016 You know my methods, Watson --SIR ARTHUR CONAN DOYLE The Infectious Cycle Virologists divide the infectious cycle into steps to facilitate
The Infectious Cycle Lecture 2 Biology W3310/4310 Virology Spring 2016 You know my methods, Watson --SIR ARTHUR CONAN DOYLE The Infectious Cycle Virologists divide the infectious cycle into steps to facilitate
Japanese Encephalitis: Prevention Through Vaccination. Jaime A. Santos
 Japanese Encephalitis: Prevention Through Vaccination Jaime A. Santos Outline Introduction: Epidemiology, Disease and Sequelae Prevention: Role of Vaccination Vaccines against JE; SP Chimeric Vaccine Conclusions
Japanese Encephalitis: Prevention Through Vaccination Jaime A. Santos Outline Introduction: Epidemiology, Disease and Sequelae Prevention: Role of Vaccination Vaccines against JE; SP Chimeric Vaccine Conclusions
Flaviviruses New Challenges, New Vaccines
 Flaviviruses New Challenges, New Vaccines Christian W. Mandl Institute of Virology Medical University of Vienna, AUSTRIA Family Flaviviridae Genus Hepacivirus Genus Pestivirus Genus Flavivirus (>70 members)
Flaviviruses New Challenges, New Vaccines Christian W. Mandl Institute of Virology Medical University of Vienna, AUSTRIA Family Flaviviridae Genus Hepacivirus Genus Pestivirus Genus Flavivirus (>70 members)
A solid-phase competition ELISA for measuring antibody to foot-and-mouth disease virus
 197 Appendix 24 A solid-phase competition ELISA for measuring antibody to foot-and-mouth disease virus N.P. Ferris a, A.N. Bulut b, T. Rendle a, F. Davidson a and D.K.J. Mackay c a b c Institute for Animal
197 Appendix 24 A solid-phase competition ELISA for measuring antibody to foot-and-mouth disease virus N.P. Ferris a, A.N. Bulut b, T. Rendle a, F. Davidson a and D.K.J. Mackay c a b c Institute for Animal
Viral vaccines. Lec. 3 أ.د.فائزة عبد هللا مخلص
 Lec. 3 أ.د.فائزة عبد هللا مخلص Viral vaccines 0bjectives 1-Define active immunity. 2-Describe the methods used for the preparation of attenuated live & killed virus vaccines. 3- Comparison of Characteristics
Lec. 3 أ.د.فائزة عبد هللا مخلص Viral vaccines 0bjectives 1-Define active immunity. 2-Describe the methods used for the preparation of attenuated live & killed virus vaccines. 3- Comparison of Characteristics
Application of Reverse Genetics to Influenza Vaccine Development
 NIAID Application of Reverse Genetics to Influenza Vaccine Development Kanta Subbarao Laboratory of Infectious Diseases NIAID, NIH Licensed Vaccines for Influenza Principle: Induction of a protective
NIAID Application of Reverse Genetics to Influenza Vaccine Development Kanta Subbarao Laboratory of Infectious Diseases NIAID, NIH Licensed Vaccines for Influenza Principle: Induction of a protective
PREVALENCE OF HUMAN ENTEROVIRUS AMONG PATIENTS WITH HAND, FOOT, AND MOUTH DISEASE AND HERPANGINA IN THAILAND, 2013
 Prevalence of enterovirus in Thailand PREVALENCE OF HUMAN ENTEROVIRUS AMONG PATIENTS WITH HAND, FOOT, AND MOUTH DISEASE AND HERPANGINA IN THAILAND, 2013 John Mauleekoonphairoj, Jiratchaya Puenpa, Sumeth
Prevalence of enterovirus in Thailand PREVALENCE OF HUMAN ENTEROVIRUS AMONG PATIENTS WITH HAND, FOOT, AND MOUTH DISEASE AND HERPANGINA IN THAILAND, 2013 John Mauleekoonphairoj, Jiratchaya Puenpa, Sumeth
Development of a Novel Recombinant Influenza Vaccine in Insect Cells
 Development of a Novel Recombinant Influenza Vaccine in Insect Cells Clifton McPherson New Cells for New Vaccines II September 18, 2007 cmcpherson@proteinsciences.com New Cell for New Vaccines II Topics:
Development of a Novel Recombinant Influenza Vaccine in Insect Cells Clifton McPherson New Cells for New Vaccines II September 18, 2007 cmcpherson@proteinsciences.com New Cell for New Vaccines II Topics:
Pathogenesis of Simian Foamy Virus Infection in Natural and Experimental Hosts
 INCTION AD ImmuNrry, Sept. 1975, p. 470-474 Copyright 0 1975 American Society for Microbiology Vol. 12, No. 3 Printed in U.S.A. Pathogenesis of Simian Foamy Virus Infection in Natural and Experimental
INCTION AD ImmuNrry, Sept. 1975, p. 470-474 Copyright 0 1975 American Society for Microbiology Vol. 12, No. 3 Printed in U.S.A. Pathogenesis of Simian Foamy Virus Infection in Natural and Experimental
SIV p27 ANTIGEN CAPTURE ASSAY
 SIV p27 ANTIGEN CAPTURE ASSAY Enzyme Immunoassay for the detection of Simian Immunodeficiency Virus (SIV) p27 in tissue culture media Catalog #5436 and #5450 Version 6; 12/2012 ABL PRODUCTS AND SERVICES
SIV p27 ANTIGEN CAPTURE ASSAY Enzyme Immunoassay for the detection of Simian Immunodeficiency Virus (SIV) p27 in tissue culture media Catalog #5436 and #5450 Version 6; 12/2012 ABL PRODUCTS AND SERVICES
Astrovirus-associated gastroenteritis in children
 Journal of Clinical Pathology, 1978, 31, 939-943 Astrovirus-associated gastroenteritis in children C. R. ASHLEY, E. 0. CAUL, AND W. K. PAVER1 From the Public Health Laboratory, Myrtle Road, Bristol BS2
Journal of Clinical Pathology, 1978, 31, 939-943 Astrovirus-associated gastroenteritis in children C. R. ASHLEY, E. 0. CAUL, AND W. K. PAVER1 From the Public Health Laboratory, Myrtle Road, Bristol BS2
!
 ! 71!" 1 I= 2 Enterovirus!"#$%&'()*+,-./0123 8!"#$%&10!"#$%&'()*+343!!"#$%&'()*+,-./0123456789:;?!"#$%&'()*+,-."/01234567"89:;
! 71!" 1 I= 2 Enterovirus!"#$%&'()*+,-./0123 8!"#$%&10!"#$%&'()*+343!!"#$%&'()*+,-./0123456789:;?!"#$%&'()*+,-."/01234567"89:;
Replication Defective Enterovirus Infections: Implications for Type I Diabetes
 Replication Defective Enterovirus Infections: Implications for Type I Diabetes N. M. Chapman Department of Pathology & Microbiology University of Nebraska Medical Center Enterovirus Genome and 2 Capsid
Replication Defective Enterovirus Infections: Implications for Type I Diabetes N. M. Chapman Department of Pathology & Microbiology University of Nebraska Medical Center Enterovirus Genome and 2 Capsid
THE CYTOPATHOGENIC ACTION OF BLUETONGUE VIRUS ON TISSUE CULTURES AND ITS APPLICATION TO THE DETECTION OF ANTIBODIES IN THE SERUM OF SHEEP.
 Onderstepoort Journal of Veterinary Research, Volume 27, Number 2, October, 1956. The Government Printer. THE CYTOPATHOGENIC ACTION OF BLUETONGUE VIRUS ON TISSUE CULTURES AND ITS APPLICATION TO THE DETECTION
Onderstepoort Journal of Veterinary Research, Volume 27, Number 2, October, 1956. The Government Printer. THE CYTOPATHOGENIC ACTION OF BLUETONGUE VIRUS ON TISSUE CULTURES AND ITS APPLICATION TO THE DETECTION
