Two-Year Outcomes With a Novel, Double- Lumen, Saline-Filled Breast Implant
|
|
|
- Stuart Perkins
- 5 years ago
- Views:
Transcription
1 Breast Surgery Two-Year Outcomes With a Novel, Double- Lumen, Saline-Filled Breast Implant Larry S. Nichter, MD; and Robert S. Hamas, MD Aesthetic Surgery Journal 32(7) The American Society for Aesthetic Plastic Surgery, Inc. Reprints and permission: journalspermissions.nav DOI: / X Abstract Background: A double-lumen, saline-filled breast implant with a baffle structure (IDEAL IMPLANT Saline-Filled Breast Implant; Ideal Implant Incorporated, Irving, Texas) was developed to overcome the limitations of single-lumen saline implants by controlling saline movement and providing internal support to the implant edge and upper pole. Objectives: The authors report 2-year data from a 10-year US clinical trial evaluating the safety and effectiveness of this investigational implant. Methods: Women seeking primary breast augmentation or replacement of existing augmentation implants were enrolled between February 2009 and February 2010 at 35 private practice sites, where the women underwent surgery to receive the new technology implant. Data collection included incidence and grade of capsular contracture (CC) and wrinkling as well as patient- and surgeon-reported satisfaction measures. All clinical data were reported as Kaplan-Meier risk rates of first occurrence, per patient, in each cohort. Results: Two-year follow-up visits were completed by 472 of 502 enrolled women (94.0%), 378 of whom had undergone primary breast augmentation and 94 of whom had received replacement augmentation. Patient-reported satisfaction with the outcome was 94.3% for primary augmentations and 92.3% for replacement augmentations; surgeon-reported satisfaction was also high (96.5% and 93.4%, respectively). Baker Grade III and IV CC rates were 3.8% (primary) and 8.2% (replacement), whereas moderate-to-severe wrinkling was 3.8% (primary) and 12.0% (replacement). Deflations occurred in 4.8% of primary augmentations and 3.3% of replacement augmentations. No deflations were caused by a shell fold flaw. Conclusions: Two-year data from 472 women indicate that this double-lumen saline implant containing a baffle structure has a low rate of wrinkling and a lower rate of CC at 2 years than was reported for current single-lumen saline implants at 1 year. Level of Evidence: 2 Keywords breast implant, breast augmentation, breast surgery, saline, baffles, wrinkling, capsular contracture, clinical trial, double-lumen Although saline-filled implants are a safe, effective alternative to silicone gel-filled implants, 1 the currently available saline-filled implants are essentially balloons filled with freely-moving fluid, which may result in an unnatural feel or suboptimal aesthetic result. 2-5 These implants, which have only a single lumen, tend to be less forgiving than silicone gel implants in terms of palpability, visibility, and rippling. 5,6 Scalloping or wrinkling, for example, is a wellknown problem. 5-7 For a more natural result, the only currently available alternative to saline-filled implants are silicone gel-filled implants, which some women will not accept. This may be a significant part of the reason that 31% of women in the United States chose saline-filled implants for breast augmentation in This data suggests that many women considering breast augmentation feel more comfortable with implants containing saline instead of silicone gel. Clearly, there is a need for a salinefilled implant that offers a more natural result, without the wrinkling, bouncing, or globular shape commonly attributed to current saline implants. 2-5 The IDEAL IMPLANT Saline-Filled Breast Implant (Ideal Implant Incorporated, Irving, Texas) is an investigational device with a baffle structure, 2 lumens, and a series of shells of increasing size nested together (Figure 1). It comprises an Dr Nichter is Voluntary Clinical Professor of Plastic Surgery at University of Southern California and University of California, Irvine and Chairman of the Department of Plastic Surgery at Hoag Memorial Hospital, Newport Beach, California. Dr Hamas is a plastic surgeon in private practice in Dallas, Texas, and is also Founder, President, and Medical Director of Ideal Implant Incorporated, Irving, Texas. Corresponding Author: Dr Robert S. Hamas, 8345 Walnut Hill Lane, Suite 120, Dallas, TX, 75231, USA. rsh@yourlooks.com
2 862 Aesthetic Surgery Journal 32(7) Figure 1. Schematic cut-away view of the IDEAL IMPLANT Saline-Filled Breast Implant (Ideal Implant Incorporated, Irving, Texas). Smaller sizes have 1 baffle shell, medium sizes have 2 baffle shells, and larger sizes have 3 baffle shells. Courtesy of Ideal Implant Incorporated. inner shell defining the inner lumen, filled through a valve in the posterior patch; an outer shell defining the outer lumen between these 2 shells, filled through a valve in the front; and 1 to 3 additional shells (depending on implant volume) in the outer lumen, perforated and free-floating, to act as a baffle. This baffle structure is designed to control movement of the saline filler so there is no bouncing, to support the upper pole so it does not collapse when the implant is upright, and to support the edges of the implant so scalloping and wrinkling are minimized. Current saline and silicone gel implants viewed on a convex surface that simulates the typical chest wall convexity behind the breast demonstrate differences in recumbent implant shape, contour to a convex surface, and edge position. When the implant edge is high, they tend to look more globular than when the edge is lower (Figure 2 and Figure 3A). In addition, when the edge is higher, pressure simulating soft tissue can move the edge further toward the underlying convex surface and cause wrinkling. For these reasons, the investigational double-lumen, salinefilled implant was designed so that the edge lies lower and closer to the chest wall, which potentially provides a better contour compared with current single-lumen saline implants (Figure 3). For all implant sizes, the inner lumen fill volume in the investigative device is a constant percentage of the inner shell mandrel volume and is not adjustable. The outer lumen is adjustable, and the minimum and maximum fill volumes are each constant percentages of the outer shell mandrel volume. The increment of outer lumen fill volume is a constant percentage of the outer shell mandrel volume, so although the increment of added saline increases with the implant size, all implant sizes look the same when filled to the minimum or to the maximum. This contrasts with some current saline implants that have increments of added saline unrelated to implant size; for example, 30 cc added to a 150-cc implant equals 20%, whereas 30 cc added to a 300-cc implant equals 10%, so these 2 implants look different when filled to the maximum volume. The IDEAL IMPLANT is manufactured in 14 sizes, from 210 cc to 675 cc. The size labels are based on the minimum total implant volume, which includes the volume of the empty implant and the minimum volume of the saline inside: Implant Size Label = Minimum Total Implant Volume = Empty Implant Volume + Inner Lumen Fill Volume + Minimum Outer Lumen Fill Volume. The size labels of silicone gel implants are also based on the total implant volume, which includes the volume of the empty implant and the volume of the silicone gel inside. In contrast, the size labels of current saline implants are based on the recommended minimum fill volume; the volume of the empty implant is not included. For example, a current saline implant with a size label of 350 cc actually has a total implant volume = 350 cc fill Figure 2. Silicone gel implants on a convex surface simulating the chest wall. (A) Mentor MemoryGel implant, moderate plus profile (Mentor Worldwide LLC, Santa Barbara, California), 350 cc. (B) Allergan Natrelle implant, Style 15, moderate profile (Allergan, Inc, Irvine, California), 371 cc. The edge of Implant B is lower relative to the convex surface, so it contours better to the simulated chest wall and has a more natural, tapered shape than Implant A, which has a more globular shape. Courtesy of Ideal Implant Incorporated (Irving, Texas).
3 Nichter and Hamas 863 Figure 3. Saline implants on a convex surface simulating the chest wall. (A) Mentor implant, Style 2000, moderate plus profile (Mentor Worldwide LLC, Santa Barbara, California) at minimum fill volume, 350 cc. (B) IDEAL IMPLANT (Ideal Implant Incorporated, Irving, Texas) at minimum fill volume, 405 cc. The edge of Implant B is lower relative to the convex surface, so it contours better to the simulated chest wall and has a more natural, tapered shape than Implant A, which has a more globular shape. Courtesy of Ideal Implant Incorporated. volume + about 20 cc empty implant volume = 370 cc. In this study, 2-year outcomes are reported from an ongoing clinical trial evaluating the safety and effectiveness of the IDEAL IMPLANT Saline-Filled Breast Implant. METHODS Although the IDEAL IMPLANT is made of the same silicone materials as current saline implants, the US Food and Drug Administration (FDA) required complete chemistry, toxicology, and mechanical testing, including cyclic fatigue of the implant, before approving an Investigational Device Exemption (IDE). The trial protocol was approved by an institutional review board (RCRC Independent Review Board, Austin, Texas). The 10-year US clinical trial began in February A total of 45 American Board of Plastic Surgery (ABPS) certified plastic surgeons at 35 private practice sites clustered in 7 metropolitan areas of the United States were the investigators. Between February 2009 and February 2010, 502 women gave informed consent, were enrolled in the trial, and underwent breast surgery to receive the double-lumen implant: 399 had primary breast augmentation and 103 had existing saline- or silicone gel filled augmentation implants replaced. A breast reconstruction cohort was not included in this trial. The implanting surgeons were instructed to use the same preoperative, intraoperative, and postoperative procedures used in their practices for primary breast augmentation or replacement of existing augmentation implants. The study protocol contained no instructions regarding the method of determining implant size, minimum thickness of soft tissue, surgical technique, pocket irrigation, dressings, drains, or postoperative massage. Each patient was examined at the 2-month, 6-month, 1-year, and 2-year follow-up visits. Capsular contracture (CC) was graded using the Baker classification system. The absence or presence of palpable wrinkling was determined with the patient standing with arms at her sides. If present, wrinkling was graded on a 5-point scale: negligible, very mild, mild, moderate, and severe. Patient and surgeon satisfaction with the outcome were separately graded at each follow-up visit from definitely satisfied to definitely dissatisfied on a 5-point scale. Baseline and follow-up clinical data were collected by the investigators on standardized case report forms and tabulated by an independent Clinical Research Organization (CRO) in the format required for submission to the FDA, per patient and per breast. If a score was different for the right and left breasts, the worse score was used for data being reported per patient. All adverse events (AE) were followed until they resolved. A premarket approval (PMA) application for the doublelumen saline implant was submitted to the FDA after the last patient completed her 2-year follow-up visit, per FDA guidance. 9 RESULTS Of the 502 enrolled women, 472 completed all follow-up visits through 2 years, a follow-up rate of 94.0%. Of the 472 women, 378 had primary breast augmentation and 94 had replacement of existing augmentation implants with the investigational implant. Demographic data and surgical information are shown in Table 1. Reasons for replacement of existing implants are shown in Table 2. At 2 years, patient-reported satisfaction with the outcome was 94.3% for primary augmentations and 92.3% for augmentations that replaced existing implants. Surgeon-reported satisfaction was 96.5% for primary augmentations and 93.4% for replacement augmentations. Examples of outcomes are shown in Figures 4 and 5.
4 864 Aesthetic Surgery Journal 32(7) Table 1. Patient Age and Surgical Information, per Breast, by Cohort Patient age, y Primary Augmentation (n = 798) Replacement Augmentation (n = 206) Mean Range Incision, % Inframammary Periareolar Axillary Abdominoplasty Table 2. Reasons for Replacement of Each Type of Implant a Existing Saline Implant (n = 156), % Existing Silicone Gel Implant (n = 50), % Capsule contracture Scalloping/wrinkling Deflation (saline) 8.3 NA Rupture (silicone gel) NA 26.0 Dissatisfaction with size Other Abbreviation: NA, not applicable. a Patients may have had different reasons for each breast or more than one reason per breast, so they may be counted in more than one category. Position, % Submuscular Subglandular Concomitant procedures, % Mastopexy Capsule procedure NA 60.7 Type of implant replaced, % Saline NA 75.7 Silicone gel NA 24.3 Abbreviation: NA, not applicable. Table 3 lists adverse events as Kaplan-Meier risk rates of first occurrence, per patient, for the IDEAL IMPLANT at 1 year compared with the 1-year Large Simple Trial (LST) data used in the PMA applications for the current saline implants manufactured by Allergan, Inc (Irvine, California) and Mentor Worldwide LLC (Santa Barbara, California). 10,11 In the LST studies, Allergan had a 1-year follow-up rate of 62%, and Mentor had a 1-year follow-up rate of 47%. Table 4 shows 2-year adverse events for the investigational implants as Kaplan-Meier risk rates of first occurrence, per patient. The 2-year Baker Grade III and IV CC rates for the investigational implants were lower than the rates reported at 1 year for current single-lumen saline implants. 10,11 Based on explant analysis of clinical trial implant deflations, none were due to shell failure caused by a shell foldflaw, the usual cause of shell failure for both single-lumen saline and silicone gel implants. One deflation was due to shell failure from iatrogenic puncture by a surgical needle, confirmed by scanning electron microscopy. One deflation was indeterminate because the implant was inadvertently damaged by the surgeon after explantation. All other deflations (n = 23) were due to early manufacturing defects, including inadequate vulcanization bonds of the posterior valve to the patch, inadequate vulcanization bonds of the patch to the shell, or valve damage during assembly. Changes in manufacturing for the commercial implant have been instituted and validated by the study sponsor to provide adequate vulcanization bonds of the valve-patch and shellpatch joints and to prevent valve damage during assembly. DISCUSSION Over the years, many novel designs have been proposed for an improved saline breast implant that would overcome the limitations of current single-lumen saline implants and provide a good alternative to silicone gel implants (Table 5). Adding a baffle to control movement of the saline is not a new concept. However, earlier baffle designs were either complex to manufacture or flawed in that the baffle material could dislocate and concentrate in one area of the implant when the patient changed position. The investigational saline-filled implant discussed in this study was designed to address these earlier design problems: the baffle structure is created by perforating implant shells for ease of manufacturing, and the placement of the baffle structure in the outer lumen, closely fitted between the inner and outer shells, keeps the structure in place when the patient changes position. Although not the subject of the current study, the effect of the baffle structure on saline movement and on upper pole and edge support can also be demonstrated in vitro. The investigational implant behaves like it has the viscosity of a silicone gel implant and does not bounce when dropped onto a table, whereas current singlelumen saline implants bounce. Likewise, when held upright on the outstretched hand, the investigational implant maintains upper pole fullness and the edge does not wrinkle, whereas current single-lumen saline implants collapse in the upper pole and wrinkle at the edge. The clinical trial protocol used in this study closely follows those used for FDA approval of current saline-filled breast implants marketed by Allergan, Inc and Mentor Worldwide LLC. However, in those trials, the investigators reported wrinkling only when they thought it was an AE and completed an AE case report form. Therefore, for
5 Nichter and Hamas 865 Figure 4. (A, C) This nulliparous 19-year-old woman presented for breast enlargement. (B, D) One year after submuscular breast augmentation with the double-lumen, saline-filled investigational implants (IDEAL IMPLANT; Ideal Implant Incorporated, Irving, Texas), with an implant volume of 305 cc. example, some investigators in those trials may not have reported wrinkling when it was palpable, only when it was visible. By comparison, wrinkling may have been overreported in this trial because the investigator was required to examine for the absence or presence of palpable wrinkling at every follow-up visit and, if present, to score it from negligible to severe on a 5-point scale. Similarly, patient and surgeon satisfaction in this trial may have been underreported because the surgeons and patients were required to assess satisfaction with the outcome, which can include such nonimplant items as the quality of the incisional scar, sensory changes, or development of a wound infection. In the Allergan saline implant trial, only patients satisfaction with their breast implants was assessed, 10 and in the Mentor saline implant trial, only patients satisfaction with the general appearance of their breasts was assessed as measured by the Breast Evaluation Questionnaire (BEQ). 11 One of the most significant and unexpected differences between the investigational implant and current singlelumen saline implants was the low rate of CC. Compared with the published 1-year LST data for primary breast augmentation shown in Table 3, the investigational implant at 1 year had a 1.6 (compared with Mentor data) to 2.6 (compared with Allergan data) times lower rate of Baker III and IV CC. For replacement augmentation, the investigational implant had a 3.0 (Allergan) to 3.6 (Mentor) times lower rate of Baker III and IV CC, even though 39.1% of existing saline implants and 58.0% of existing silicone gel implants in this trial were replaced because of bilateral or unilateral CC. As shown in Table 4, the 2-year CC rates for the investigational implant remain lower than the 1-year rates for current single-lumen saline implants. The reason for this low incidence of CC is unknown. The 2-year clinical data from this study show that this double-lumen saline breast implant may provide a good alternative for women who do not want a silicone gel implant. In the authors opinion, the baffle structure of the double-lumen implant discussed in this study successfully controls movement of the saline, preventing bouncing and providing a more natural result compared with current single-lumen saline implants. In addition, 2-year data from this study show that the incidence of scalloping or wrinkling is lower with the investigational saline-filled implant compared with currently approved single-lumen saline implants. The double-lumen implant is designed with a lower edge to conform better to the chest wall. It is possible that the lower rate of CC reported in this study
6 866 Aesthetic Surgery Journal 32(7) Figure 5. (A, C) This 31-year-old woman presented for restoration of breast volume following delivery and nursing of 2 children. (B, D) One year following submuscular breast augmentation with the double-lumen, saline-filled investigational implants (IDEAL IMPLANT; Ideal Implant Incorporated, Irving, Texas), with an implant volume of 325 cc. Table 3. One-Year Adverse Events as Kaplan-Meier Risk Rates of First Occurrence, per Patient, for the Double-Lumen IDEAL IMPLANT a Compared With Allergan and Mentor Saline Implants b Primary Augmentation, % Replacement Augmentation, % Adverse Event IDEAL Allergan Mentor IDEAL Allergan Mentor Capsular contracture (Baker III and IV) Infection No data Removal with or without replacement Deflation a Ideal Implant Incorporated (Irving, Texas). b The Allergan implants are Styles 68, 163, 168, 363, and 468, smooth and textured, round and shaped (Allergan, Inc, Irvine, California, formerly McGhan Medical, Santa Barbara, California), and the Mentor implants are Styles 1600, 2600, 2700, 2900, 5000, 5000PT, 1400, 2400, 2500, 6000, and 6000PT, smooth and textured, round and contoured (Mentor Worldwide LLC, Santa Barbara, California). Data listed for these implants are taken from their respective premarket approval data. 10,11 may be related in some way to the lower edge position and better conformity to the chest wall compared with current single-lumen saline implants. Longer follow-up is needed to confirm that these are persistent findings. Although these 2-year outcomes are favorable, the clinical trial for cosmetic breast augmentation and replacement of existing augmentation implants is ongoing and will continue to provide data on this new implant for 10 years. However, because the implant is saline-filled, a PMA application was submitted to the FDA after the last patient completed her 2-year followup visit. 9
7 Nichter and Hamas 867 Table 4. Two-Year Adverse Events as Kaplan-Meier Risk Rates of First Occurrence, per Patient, for the Double-Lumen IDEAL IMPLANT a Adverse Event Capsule contracture (Baker III and IV) CONCLUSIONS Primary Augmentation (n = 399), % (95% CI) Replacement Augmentation (n = 103), % (95% CI) 3.8 (2.3, 6.3) 8.2 (4.2, 15.8) Infection 0.8 (0.2, 2.3) 1.0 (0.1, 7.0) Wrinkling (moderate to severe) Deflation (early manufacturing defects) Abbreviation: CI, confidence interval. a Ideal Implant Incorporated (Irving, Texas). 3.8 (2.3, 6.3) 12.0 (7.0, 20.2) 4.8 (3.0, 7.6) 3.3 (1.1, 10.0) Table 5. US Patents Related to Improvements in the Design of Saline Implants Inventor Patent No. Date Description L. Bartholdson 4,507,810 April 2, 1985 Filled with interconnected cells J. Bark 5,171,269 December 15, 1992 R. Peterson 5,246,454 September 21, 1993 Filled with fibrous material Filled with small saline pouches J. Fisher 5,496,367 May 5, 1996 Internal baffle structures R. Hamas 5,496,370 May 5, 1996 Double-lumen with baffle material J. Henley 5,534,023 July 9, 1996 Filled with chain of gas-filled beads T. Knapp 5,824,081 October 20, 1996 Filled with foam material D. Carlisle 5,658,330 August 19, 1997 Filled with foam insert R. Hamas 6.802,861 October 12, 2004 Double-lumen with nested shells B. Purkait 7,625,405 December 1, 2009 Filled with silicone tubing Clinical data at 2 years from 472 women show that this double-lumen, saline-filled implant with a baffle structure has a low rate of wrinkling and a lower rate of CC at 2 years than rates reported in the PMA applications for current single-lumen, saline-filled implants at 1 year. There were no deflations due to any fold-flaw of the shell, and satisfaction with the outcome was very high for both patients and surgeons. This double-lumen saline implant may provide women with an alternative to current saline or silicone gel implants. Disclosures Dr Nichter is a paid clinical trial investigator and Dr Hamas is Founder, President, Medical Director, and stockholder of Ideal Implant Incorporated (Irving, Texas), the manufacturer of the products discussed in this study. Funding The clinical trial was entirely funded by the study sponsor (Ideal Implant Incorporated, Irving, Texas). REFERENCES 1. Cunningham BL, Loketh AL, Gutowski KA. Saline-filled breast implant safety and efficacy: a multicenter retrospective review. Plast Reconstr Surg. 2000;105(6): Friedman RM, Gyimesi I, Robinson JB, Rohrich RJ. Saline made viscous with polyethylene glycol: a new alternate breast implant filler material. Plast Reconstr Surg. 1996;98(7): Tebbets JB. Patient acceptance of adequately filled breast implants using the tilt test. Plast Reconstr Surg. 2000;106(1): Spear SL, Jespersen MR. Breast implants saline or silicone. Aesthetic Surg J. 2010;30(4): Fisher J. Breast augmentation using silicone gel-filled implants. Aesthetic Surg J. 2007;27(5): Spear SL, Bulan EJ, Venturi ML. Breast augmentation update discussion. Plast Reconstr Surg. 2006;118(7) (suppl):197s-198s. 7. Walker PS, Walls B, Murphy DK. Natrelle saline-filled breast implants: a prospective 10-year study. Aesthetic Surg J. 2009;29(1): American Society for Aesthetic Plastic Surgery statistics Stats.pdf. Accessed May 19, Food and Drug Administration. Guidance for Industry: Saline, Silicone Gel, and Alternative Breast Implants. November 17, MedicalDevices/DeviceRegulationandGuidance/GuidanceDocuments/ucm pdf. Accessed October 28, Natrelle Biocell textured and Natrelle smooth salinefilled breast implants directions for use. Santa Barbara, CA: Allergan, Inc; m712-04_saline_dfu.pdf. Accessed October 28, Saline-filled and Spectrum breast implants directions for use. Irving, TX: Mentor Corporation; Accessed October 28, 2011.
Scientific Forum. The Comparative Dimensions of Round and Anatomical Saline-filled Breast Implants
 The Comparative Dimensions of Round and Anatomical Saline-filled Breast Implants Robert S. Hamas, MD Background: Anatomical saline-filled breast implants have been portrayed as having a more natural shape
The Comparative Dimensions of Round and Anatomical Saline-filled Breast Implants Robert S. Hamas, MD Background: Anatomical saline-filled breast implants have been portrayed as having a more natural shape
Tackling challenging revision breast augmentation cases
 the BREAST Careful preoperative consultations can reduce the need for revision breast surgery. Second Time Around Tackling challenging revision breast augmentation cases By Adam D. Schaffner, MD, FACS
the BREAST Careful preoperative consultations can reduce the need for revision breast surgery. Second Time Around Tackling challenging revision breast augmentation cases By Adam D. Schaffner, MD, FACS
Since the first breast implants were introduced COSMETIC
 COSMETIC Sientra Portfolio of Silimed Brand Shaped Implants with High-Strength Silicone Gel: A 5-Year Primary Augmentation Clinical Study Experience and a Postapproval Experience Results from a Single-Surgeon
COSMETIC Sientra Portfolio of Silimed Brand Shaped Implants with High-Strength Silicone Gel: A 5-Year Primary Augmentation Clinical Study Experience and a Postapproval Experience Results from a Single-Surgeon
Primary Breast Augmentation Today: A Survey of Current Breast Augmentation Practice Patterns
 Breast Surgery Special Topic Primary Breast Augmentation Today: A Survey of Current Breast Augmentation Practice Patterns Edward M. Reece, MD, MS; Ashkan Ghavami, MD; Ronald E. Hoxworth, MD; Sergio A.
Breast Surgery Special Topic Primary Breast Augmentation Today: A Survey of Current Breast Augmentation Practice Patterns Edward M. Reece, MD, MS; Ashkan Ghavami, MD; Ronald E. Hoxworth, MD; Sergio A.
Guide to Breast Augmentation: Everything You Need to Know
 Northwestern Specialists in Plastic Surgery Dr. Neil Fine, MD, FACS Dr. Clark Schierle, MD, PhD, FACS Contents 3 Introduction 4 Implant Shell 5 Implant Fill 6 Ideal Implant 7 Implant Shape 8 Implant Placement
Northwestern Specialists in Plastic Surgery Dr. Neil Fine, MD, FACS Dr. Clark Schierle, MD, PhD, FACS Contents 3 Introduction 4 Implant Shell 5 Implant Fill 6 Ideal Implant 7 Implant Shape 8 Implant Placement
Capsular Contracture Rate in a Low-Risk Population After Primary Augmentation Mammaplasty
 Capsular Contracture Rate in a Low-Risk Population After Primary Augmentation Mammaplasty Andrew L. Blount, Matthew D. Martin, Kyle D. Lineberry, Nicolas Kettaneh, David R. Alfonso Breast Surgery Capsular
Capsular Contracture Rate in a Low-Risk Population After Primary Augmentation Mammaplasty Andrew L. Blount, Matthew D. Martin, Kyle D. Lineberry, Nicolas Kettaneh, David R. Alfonso Breast Surgery Capsular
Assessing the Augmented Breast: A Blinded Study Comparing Round and Anatomical Form-Stable Implants
 Breast Surgery Assessing the Augmented Breast: A Blinded Study Comparing Round and Anatomical Form-Stable Implants Aesthetic Surgery Journal 2015, Vol 35(3) 273 278 2015 The American Society for Aesthetic
Breast Surgery Assessing the Augmented Breast: A Blinded Study Comparing Round and Anatomical Form-Stable Implants Aesthetic Surgery Journal 2015, Vol 35(3) 273 278 2015 The American Society for Aesthetic
Motiva Implant Matrix Silicone Breast Implants Summary of Clinical Data 5-Year Follow Up
 Motiva Implant Matrix Silicone Breast Implants Summary of Clinical Data 5-Year Follow Up October 21 - February 216 Motiva Implant Matrix Silicone Breast Implants Prospective Clinical Evaluation: 5-Year
Motiva Implant Matrix Silicone Breast Implants Summary of Clinical Data 5-Year Follow Up October 21 - February 216 Motiva Implant Matrix Silicone Breast Implants Prospective Clinical Evaluation: 5-Year
F ORUM. Is One-Stage Breast Augmentation With Mastopexy Safe and Effective? A Review of 186 Primary Cases
 Is One-Stage Breast Augmentation With Mastopexy Safe and Effective? A Review of 186 Primary Cases W. Grant Stevens, MD; David A. Stoker, MD; Mark E. Freeman, MD; Suzanne M. Quardt, MD; Elliot M. Hirsch,
Is One-Stage Breast Augmentation With Mastopexy Safe and Effective? A Review of 186 Primary Cases W. Grant Stevens, MD; David A. Stoker, MD; Mark E. Freeman, MD; Suzanne M. Quardt, MD; Elliot M. Hirsch,
BREAST AUGMENTATION PATIENT PLANNER SHAPE THAT HOLDS. SATISFACTION THAT LASTS. Real NATRELLE Patient: NIKKI
 BREAST AUGMENTATION PATIENT PLANNER SHAPE THAT HOLDS. SATISFACTION THAT LASTS. Real NATRELLE Patient: NIKKI TABLE OF CONTENTS Page Pre-breast augmentation surgery...3 The surgery...9 Post-breast augmentation
BREAST AUGMENTATION PATIENT PLANNER SHAPE THAT HOLDS. SATISFACTION THAT LASTS. Real NATRELLE Patient: NIKKI TABLE OF CONTENTS Page Pre-breast augmentation surgery...3 The surgery...9 Post-breast augmentation
Ideal Implant Incorporated
 Ideal Implant Incorporated IDEAL IMPLANT STRUCTURED BREAST IMPLANTS Instructions for Use October 2015 Caution: Federal law restricts this device to sale by or on the order of a licensed physician. IDEAL
Ideal Implant Incorporated IDEAL IMPLANT STRUCTURED BREAST IMPLANTS Instructions for Use October 2015 Caution: Federal law restricts this device to sale by or on the order of a licensed physician. IDEAL
Sientra High-Strength Cohesive Textured Round Implant Technique: Roundtable Discussion
 Supplement Article Special Topic Sientra High-Strength Cohesive Textured Round Implant Technique: Roundtable Discussion W. Grant Stevens, MD; M. Bradley Calobrace, MD; Robert Cohen, MD; Michael A. Fiorillo,
Supplement Article Special Topic Sientra High-Strength Cohesive Textured Round Implant Technique: Roundtable Discussion W. Grant Stevens, MD; M. Bradley Calobrace, MD; Robert Cohen, MD; Michael A. Fiorillo,
Clinical Experience With a Fourth-Generation Textured Silicone Gel Breast Implant: A Review of 1012 Mentor MemoryGel Breast Implants
 Clinical Experience With a Fourth-Generation Textured Silicone Gel Breast Implant: A Review of 1012 Mentor MemoryGel Breast Implants W. Grant Stevens, MD; Salvatore J. Pacella, MD; Andrew J. L. Gear, MD;
Clinical Experience With a Fourth-Generation Textured Silicone Gel Breast Implant: A Review of 1012 Mentor MemoryGel Breast Implants W. Grant Stevens, MD; Salvatore J. Pacella, MD; Andrew J. L. Gear, MD;
Pocket Conversion Made Easy: A Simple Technique Using Alloderm to Convert Subglandular Breast Implants to the Dual-Plane Position
 Breast Surgery Pocket Conversion Made Easy: A Simple Technique Using Alloderm to Convert Subglandular Breast Implants to the Dual-Plane Position M. Mark Mofid, MD; and Navin K. Singh, MD Background: The
Breast Surgery Pocket Conversion Made Easy: A Simple Technique Using Alloderm to Convert Subglandular Breast Implants to the Dual-Plane Position M. Mark Mofid, MD; and Navin K. Singh, MD Background: The
Why Do Patients Seek Revisionary Breast Surgery?
 Breast Surgery Why Do Patients Seek Revisionary Breast Surgery? Navanjun S. Grewal, MD; and Jack Fisher, MD In 2011, according to the American Society for Aesthetic Plastic Surgery (ASAPS), 316 848 American
Breast Surgery Why Do Patients Seek Revisionary Breast Surgery? Navanjun S. Grewal, MD; and Jack Fisher, MD In 2011, according to the American Society for Aesthetic Plastic Surgery (ASAPS), 316 848 American
Breast Augmentation - Silicone Implants
 Breast Augmentation - Silicone Implants Breast augmentation, or augmentation mammoplasty, is one of the most common plastic surgery procedures performed today. Over time, factors such as age, genetics,
Breast Augmentation - Silicone Implants Breast augmentation, or augmentation mammoplasty, is one of the most common plastic surgery procedures performed today. Over time, factors such as age, genetics,
BREAST AUGMENTATION. everything you ever wanted to know about. Cosmetic breast specialist Dr Michael Miroshnik uses. breasts.
 everything you ever wanted to know about BREAST AUGMENTATION Actual patient of Dr Miroshnik ACCORDING TO SYDNEY PLASTIC SURGEON DR MICHAEL MIROSHNIK, ADVANCES IN SURGICAL TECHNIQUE AND IMPLANT TECHNOLOGY
everything you ever wanted to know about BREAST AUGMENTATION Actual patient of Dr Miroshnik ACCORDING TO SYDNEY PLASTIC SURGEON DR MICHAEL MIROSHNIK, ADVANCES IN SURGICAL TECHNIQUE AND IMPLANT TECHNOLOGY
MENTORPROMISE AND MENTORPROMISE ENHANCED PROTECTION PLAN
 MENTORPROMISE AND MENTORPROMISE ENHANCED PROTECTION PLAN FOR MEMORYGEL BREAST IMPLANTS AND MEMORYSHAPE BREAST IMPLANTS This document describes the Mentor Worldwide LLC ( Mentor ) Product Replacement Policy
MENTORPROMISE AND MENTORPROMISE ENHANCED PROTECTION PLAN FOR MEMORYGEL BREAST IMPLANTS AND MEMORYSHAPE BREAST IMPLANTS This document describes the Mentor Worldwide LLC ( Mentor ) Product Replacement Policy
Breast Augmentation - Saline Implants
 Breast Augmentation - Saline Implants Breast augmentation, or augmentation mammoplasty, is one of the most common plastic surgery procedures performed today. Over time, factors such as age, genetics, pregnancy,
Breast Augmentation - Saline Implants Breast augmentation, or augmentation mammoplasty, is one of the most common plastic surgery procedures performed today. Over time, factors such as age, genetics, pregnancy,
The Modern Polyurethane-Coated Implant in Breast Augmentation: Long-Term Clinical Experience
 The Modern Polyurethane-Coated Implant in Breast Augmentation: Long-Term Clinical Experience Stefano Pompei, MD, Dora Evangelidou, MD, MRM, Floriana Arelli, MD, Gianluigi Ferrante, MD, MSc Breast Surgery
The Modern Polyurethane-Coated Implant in Breast Augmentation: Long-Term Clinical Experience Stefano Pompei, MD, Dora Evangelidou, MD, MRM, Floriana Arelli, MD, Gianluigi Ferrante, MD, MSc Breast Surgery
Effects of Singulair (Montelukast) Treatment for Capsular Contracture
 Breast Surgery Effects of Singulair (Montelukast) Treatment for Capsular Contracture Catherine K. Huang, MD; and Neal Handel, MD Aesthetic Surgery Journal 30(3) 404 410 2010 The American Society for Aesthetic
Breast Surgery Effects of Singulair (Montelukast) Treatment for Capsular Contracture Catherine K. Huang, MD; and Neal Handel, MD Aesthetic Surgery Journal 30(3) 404 410 2010 The American Society for Aesthetic
A New Breast Shape Classification
 A New Breast Shape Classification plasticsurgerypractice.com/2011/10/a-new-breast-shape-classification Class 1: Appears natural with no superior pole fullness. Class 2: Appears natural with mild superior
A New Breast Shape Classification plasticsurgerypractice.com/2011/10/a-new-breast-shape-classification Class 1: Appears natural with no superior pole fullness. Class 2: Appears natural with mild superior
Strattice Reconstructive Tissue Matrix used in the repair of rippling
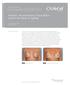 Clinical case study Strattice Tissue Matrix Strattice Reconstructive Tissue Matrix used in the repair of rippling Steven Teitelbaum, MD* Santa Monica, CA Case summary A 48-year-old woman with a history
Clinical case study Strattice Tissue Matrix Strattice Reconstructive Tissue Matrix used in the repair of rippling Steven Teitelbaum, MD* Santa Monica, CA Case summary A 48-year-old woman with a history
Reducing Capsular Contracture in Breast Augmentation: What s the Evidence? Karol A Gutowski, MD, FACS Instructional Course
 Reducing Capsular Contracture in Breast Augmentation: What s the Evidence? Karol A Gutowski, MD, FACS Instructional Course Disclosures RTI Surgical (ADM Maker) - Advisor Suneva Medical - Instructor Angiotech/Surgical
Reducing Capsular Contracture in Breast Augmentation: What s the Evidence? Karol A Gutowski, MD, FACS Instructional Course Disclosures RTI Surgical (ADM Maker) - Advisor Suneva Medical - Instructor Angiotech/Surgical
MENTOR PROMISE AND MENTOR PROMISE ENHANCED PROTECTION PLAN
 MENTOR PROMISE AND MENTOR PROMISE ENHANCED PROTECTION PLAN FOR MEMORYGEL BREAST IMPLANTS, MEMORYGEL XTRA BREAST IMPLANTS AND MEMORYSHAPE BREAST IMPLANTS This document describes the Mentor Worldwide LLC
MENTOR PROMISE AND MENTOR PROMISE ENHANCED PROTECTION PLAN FOR MEMORYGEL BREAST IMPLANTS, MEMORYGEL XTRA BREAST IMPLANTS AND MEMORYSHAPE BREAST IMPLANTS This document describes the Mentor Worldwide LLC
Breast Augmentation MOC: Preventing Capsular Contracture Karol A Gutowski, MD, FACS
 Breast Augmentation MOC: Preventing Capsular Contracture Karol A Gutowski, MD, FACS Private Practice Clinical Associate Professor University of Illinois, Chicago Disclosures Merz Trainer, Advisory Board
Breast Augmentation MOC: Preventing Capsular Contracture Karol A Gutowski, MD, FACS Private Practice Clinical Associate Professor University of Illinois, Chicago Disclosures Merz Trainer, Advisory Board
Advances and Innovations in Breast Reconstruction and Brest Surgery Presented by PCMC plastic surgeons
 Advances and Innovations in Breast Reconstruction and Brest Surgery Presented by PCMC plastic surgeons Options for reconstruction after mastectomy Implants Autologous tissue = from your own body: skin
Advances and Innovations in Breast Reconstruction and Brest Surgery Presented by PCMC plastic surgeons Options for reconstruction after mastectomy Implants Autologous tissue = from your own body: skin
For over 20 years, Mentor has been recognized worldwide as a leading manufacturer of the highest quality breast implants.
 Breast Augmentation For over 20 years, Mentor has been recognized worldwide as a leading manufacturer of the highest quality breast implants. We re here to help you make a confident, informed decision.
Breast Augmentation For over 20 years, Mentor has been recognized worldwide as a leading manufacturer of the highest quality breast implants. We re here to help you make a confident, informed decision.
Implant selection in the setting of prepectoral breast reconstruction
 Review Article Implant selection in the setting of prepectoral breast reconstruction Allen Gabriel, G. Patrick Maxwell Department of Plastic Surgery, Loma Linda University Medical Center, Loma Linda, CA,
Review Article Implant selection in the setting of prepectoral breast reconstruction Allen Gabriel, G. Patrick Maxwell Department of Plastic Surgery, Loma Linda University Medical Center, Loma Linda, CA,
A long term review of augmentation mastopexy in muscle splitting biplane
 Topic: Aesthetic Surgery of the Breast A long term review of augmentation mastopexy in muscle splitting biplane Umar Daraz Khan Aesthetic Plastic Surgeon, Reshape House, West Malling, Kent ME19 6QR, UK.
Topic: Aesthetic Surgery of the Breast A long term review of augmentation mastopexy in muscle splitting biplane Umar Daraz Khan Aesthetic Plastic Surgeon, Reshape House, West Malling, Kent ME19 6QR, UK.
BREAST AUGMENTATION TECHNIQUES
 BREAST AUGMENTATION TECHNIQUES Breast Augmentation Top Surgical Procedure in 2015 (Worldwide) Surgical Procedure : Breast Augmentation Rank : 1 Total : 1,488,992 Percent of Total Surgical Procedures :
BREAST AUGMENTATION TECHNIQUES Breast Augmentation Top Surgical Procedure in 2015 (Worldwide) Surgical Procedure : Breast Augmentation Rank : 1 Total : 1,488,992 Percent of Total Surgical Procedures :
Pre-pectoral Breast Reconstruction in Nipple Sparing Mastectomy
 September 2017 Issue 9 Pre-pectoral Breast Reconstruction in Nipple Sparing Mastectomy Aldona J. Spiegel, MD Director and Founder of the Center for Breast Restoration at the Institute for Reconstructive
September 2017 Issue 9 Pre-pectoral Breast Reconstruction in Nipple Sparing Mastectomy Aldona J. Spiegel, MD Director and Founder of the Center for Breast Restoration at the Institute for Reconstructive
Thank You for the Invitation Japan Society of Plastic Reconstructive Surgery Dr. Professor Nozaki
 Thank You for the Invitation Japan Society of Plastic Reconstructive Surgery Dr. Professor Nozaki Choices In Implant Placement Over Versus Under, Trends and Techniques Brian M. Kinney, MD, FACS, MSME Past
Thank You for the Invitation Japan Society of Plastic Reconstructive Surgery Dr. Professor Nozaki Choices In Implant Placement Over Versus Under, Trends and Techniques Brian M. Kinney, MD, FACS, MSME Past
Considering Breast Enhancement
 Consent Form While every effort has been made by Allergan to ensure the accuracy of the information contained in this booklet, Allergan accepts no responsibility and/or liability for errors or omissions.
Consent Form While every effort has been made by Allergan to ensure the accuracy of the information contained in this booklet, Allergan accepts no responsibility and/or liability for errors or omissions.
Mommy Makeover
 Mommy Makeover Many women experience significant physical changes following pregnancy and breast-feeding, many of which can be persistent and difficult to correct with diet and exercise alone. Changes
Mommy Makeover Many women experience significant physical changes following pregnancy and breast-feeding, many of which can be persistent and difficult to correct with diet and exercise alone. Changes
Controversy regarding the safety of silicone gelfilled
 Featured Operative Technique The Neopectoral Pocket in Revisionary reast Surgery G. Patrick Maxwell, MD; and Allen Gabriel, MD ontroversy regarding the safety of silicone gelfilled breast implants, which
Featured Operative Technique The Neopectoral Pocket in Revisionary reast Surgery G. Patrick Maxwell, MD; and Allen Gabriel, MD ontroversy regarding the safety of silicone gelfilled breast implants, which
SALINE-FILLED & SPECTRUM MAMMARY PROSTHESES
 Product Insert Data Sheet SALINE-FILLED & SPECTRUM MAMMARY PROSTHESES DIRECTIONS FOR USE 102876-001 Rev. A Effective January 2005 CAUTION: Federal (USA) law restricts this device to sale by or on the order
Product Insert Data Sheet SALINE-FILLED & SPECTRUM MAMMARY PROSTHESES DIRECTIONS FOR USE 102876-001 Rev. A Effective January 2005 CAUTION: Federal (USA) law restricts this device to sale by or on the order
NATRELLE 410 HIGHLY COHESIVE ANATOMICALLY SHAPED SILICONE-FILLED BREAST IMPLANTS
 NATRELLE 410 HIGHLY COHESIVE ANATOMICALLY SHAPED SILICONE-FILLED BREAST IMPLANTS Breast Augmentation and Reconstruction Patients Should Consider Introduction Allergan has prepared this brochure to provide
NATRELLE 410 HIGHLY COHESIVE ANATOMICALLY SHAPED SILICONE-FILLED BREAST IMPLANTS Breast Augmentation and Reconstruction Patients Should Consider Introduction Allergan has prepared this brochure to provide
Original Article A retrospective study of primary breast augmentation: recovery period, complications and patient satisfaction
 Int J Clin Exp Med 2015;8(10):18737-18743 www.ijcem.com /ISSN:1940-5901/IJCEM0005598 Original Article A retrospective study of primary breast augmentation: recovery period, complications and patient satisfaction
Int J Clin Exp Med 2015;8(10):18737-18743 www.ijcem.com /ISSN:1940-5901/IJCEM0005598 Original Article A retrospective study of primary breast augmentation: recovery period, complications and patient satisfaction
Sientra High-Strength Cohesive Shaped Technique: Roundtable Discussion
 Supplement Article Special Topic Sientra High-Strength Cohesive Shaped Technique: Roundtable Discussion Michael R. Schwartz, MD; Peter J. Capizzi, MD; Kiya Movassaghi, MD, DMD; and Mia Talmor, MD Abstract
Supplement Article Special Topic Sientra High-Strength Cohesive Shaped Technique: Roundtable Discussion Michael R. Schwartz, MD; Peter J. Capizzi, MD; Kiya Movassaghi, MD, DMD; and Mia Talmor, MD Abstract
Vertical mammaplasty has been developed
 BREAST Y-Scar Vertical Mammaplasty David A. Hidalgo, M.D. New York, N.Y. Background: Vertical mammaplasty is an effective alternative to inverted-t methods. Among other benefits, it results in a significantly
BREAST Y-Scar Vertical Mammaplasty David A. Hidalgo, M.D. New York, N.Y. Background: Vertical mammaplasty is an effective alternative to inverted-t methods. Among other benefits, it results in a significantly
UNDERSTANDiNG THE BREAST AUGMENTATION PROCEDURE
 UNDERSTANDiNG THE BREAST AUGMENTATION PROCEDURE Having fuller more voluptuous breasts is a very important part of feeling feminine and more confident for women. The breasts give the female body more proportion,
UNDERSTANDiNG THE BREAST AUGMENTATION PROCEDURE Having fuller more voluptuous breasts is a very important part of feeling feminine and more confident for women. The breasts give the female body more proportion,
AESTHETIC SURGERY OF THE BREAST: MASTOPEXY, AUGMENTATION & REDUCTION
 CHAPTER 18 AESTHETIC SURGERY OF THE BREAST: MASTOPEXY, AUGMENTATION & REDUCTION Ali A. Qureshi, MD and Smita R. Ramanadham, MD Aesthetic surgery of the breast aims to either correct ptosis with a mastopexy,
CHAPTER 18 AESTHETIC SURGERY OF THE BREAST: MASTOPEXY, AUGMENTATION & REDUCTION Ali A. Qureshi, MD and Smita R. Ramanadham, MD Aesthetic surgery of the breast aims to either correct ptosis with a mastopexy,
inding the fit that s right for you. Your Surgery Planner For Augmentation Surgery with NATRELLE Silicone-Filled Breast Implants
 inding the fit that s right for you. Your Surgery Planner For Augmentation Surgery with NATRELLE Silicone-Filled Breast Implants L Place Your Device Identification Card(s) Here R ACCEPTANCE OF RISK AND
inding the fit that s right for you. Your Surgery Planner For Augmentation Surgery with NATRELLE Silicone-Filled Breast Implants L Place Your Device Identification Card(s) Here R ACCEPTANCE OF RISK AND
Breast Augmentation and Mastopexy Using a Pectoral Muscle Loop
 Aesth Plast Surg (2011) 35:333 340 DOI 10.1007/s00266-010-9612-9 ORIGINAL ARTICLE Breast Augmentation and Mastopexy Using a Pectoral Muscle Loop André Auersvald Luiz Augusto Auersvald Received: 28 April
Aesth Plast Surg (2011) 35:333 340 DOI 10.1007/s00266-010-9612-9 ORIGINAL ARTICLE Breast Augmentation and Mastopexy Using a Pectoral Muscle Loop André Auersvald Luiz Augusto Auersvald Received: 28 April
A decision-making method for breast augmentation based on 25 years of practice
 Communication Introduction reast augmentation is the most commonly performed procedure in the field of aesthetic plastic surgery [1,2]. Women considering breast enhancement are mostly interested in shape
Communication Introduction reast augmentation is the most commonly performed procedure in the field of aesthetic plastic surgery [1,2]. Women considering breast enhancement are mostly interested in shape
Important Information. about Mentor MemoryGel Silicone Gel-Filled Breast Implants
 Important Information for Augmentation Patients about Mentor MemoryGel Silicone Gel-Filled Breast Implants 1 Important Information for Augmentation Patients about Mentor MemoryGel Silicone gel-filled
Important Information for Augmentation Patients about Mentor MemoryGel Silicone Gel-Filled Breast Implants 1 Important Information for Augmentation Patients about Mentor MemoryGel Silicone gel-filled
Breast Reconstruction Postmastectomy. Using DermaMatrix Acellular Dermis in breast reconstruction with tissue expander.
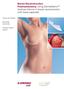 Breast Reconstruction Postmastectomy. Using DermaMatrix Acellular Dermis in breast reconstruction with tissue expander. Strong and flexible Bacterially inactivated Provides implant support Breast Reconstruction
Breast Reconstruction Postmastectomy. Using DermaMatrix Acellular Dermis in breast reconstruction with tissue expander. Strong and flexible Bacterially inactivated Provides implant support Breast Reconstruction
Periareolar Extra-Glandular Breast Augmentation
 Original Article 93 Periareolar Extra-Glandular Breast Augmentation Muhammad Humayun Mohmand 1 *, Muhammad Ahmad 2 1. Cosmetic Plastic Surgeon, La Chirurgie, Islamabad Cosmetic Surgery Centre, Islamabad,
Original Article 93 Periareolar Extra-Glandular Breast Augmentation Muhammad Humayun Mohmand 1 *, Muhammad Ahmad 2 1. Cosmetic Plastic Surgeon, La Chirurgie, Islamabad Cosmetic Surgery Centre, Islamabad,
I BRITISH JOURNAL OF ~ PLASTIC SURGERY [
 British Journal of Plastic Surgery (2000), 53, 479-483 9 2000 The British Association of Plastic Surgeons DOI: 10.1054/bjps.2000.3386 I BRITISH JOURNAL OF ~ PLASTIC SURGERY [ Patient satisfaction with
British Journal of Plastic Surgery (2000), 53, 479-483 9 2000 The British Association of Plastic Surgeons DOI: 10.1054/bjps.2000.3386 I BRITISH JOURNAL OF ~ PLASTIC SURGERY [ Patient satisfaction with
BREAST AUGMENTATION. Your complete guide to breast augmentation and enhancing your silhouette.
 BREAST AUGMENTATION Your complete guide to breast augmentation and enhancing your silhouette. (07) 3257 7950 drsamuelyang.com.au CONTENTS What is a Breast Augmentation? 3 Breast Augmentation Considerations
BREAST AUGMENTATION Your complete guide to breast augmentation and enhancing your silhouette. (07) 3257 7950 drsamuelyang.com.au CONTENTS What is a Breast Augmentation? 3 Breast Augmentation Considerations
Directions for Use (DFU) Natrelle. Saline-Filled Breast Implants. Physician Labeling
 Directions for Use (DFU) Natrelle Saline-Filled Breast Implants Physician Labeling Table of Contents Page Introduction... 1 Directions to the Physician... 1 Device Description... 2 Indications... 3 Contraindications...
Directions for Use (DFU) Natrelle Saline-Filled Breast Implants Physician Labeling Table of Contents Page Introduction... 1 Directions to the Physician... 1 Device Description... 2 Indications... 3 Contraindications...
Directions for Use. NATRELLE Saline-Filled Breast Implants. Smooth & BIOCELL Texture. Directions for Use
 Directions for Use Directions for Use NATRELLE Saline-Filled Breast Implants Smooth & BIOCELL Texture Caution: Federal (USA) law restricts this device to sale by or on the order of a licensed physician.
Directions for Use Directions for Use NATRELLE Saline-Filled Breast Implants Smooth & BIOCELL Texture Caution: Federal (USA) law restricts this device to sale by or on the order of a licensed physician.
Breast Augmentation: IMPLant placement
 Breast Augmentation: IMPLant placement There are few choices when it comes to the placement of the breast implants you select. They are subglandular (above the muscle), submuscular (below the muscle) or
Breast Augmentation: IMPLant placement There are few choices when it comes to the placement of the breast implants you select. They are subglandular (above the muscle), submuscular (below the muscle) or
Subpectoral and Precapsular Implant Repositioning Technique: Correction of Capsular Contracture and Implant Malposition
 Aesth Plast Surg (2011) 35:1126 1132 DOI 10.1007/s00266-011-9714-z INNOVATIVE TECHNIQUES Subpectoral and Precapsular Implant Repositioning Technique: Correction of Capsular Contracture and Implant Malposition
Aesth Plast Surg (2011) 35:1126 1132 DOI 10.1007/s00266-011-9714-z INNOVATIVE TECHNIQUES Subpectoral and Precapsular Implant Repositioning Technique: Correction of Capsular Contracture and Implant Malposition
INFORMED-CONSENT- AUGMENTATION MAMMOPLASTY
 INFORMED-CONSENT- AUGMENTATION MAMMOPLASTY Instructions This is an informed-consent document that has been prepared to help inform you about augmentation mammoplasty, its risks, and alternative treatments.
INFORMED-CONSENT- AUGMENTATION MAMMOPLASTY Instructions This is an informed-consent document that has been prepared to help inform you about augmentation mammoplasty, its risks, and alternative treatments.
Evolution and update on current devices for prosthetic breast reconstruction
 Review Article Evolution and update on current devices for prosthetic breast reconstruction Kristina O Shaughnessy Chief of Plastic Surgery, St. Thomas Midtown Hospital, Nashville, TN, USA Correspondence
Review Article Evolution and update on current devices for prosthetic breast reconstruction Kristina O Shaughnessy Chief of Plastic Surgery, St. Thomas Midtown Hospital, Nashville, TN, USA Correspondence
A breast implant for me? Information on breast enhancement
 A breast implant for me? Information on breast enhancement A breast implant for me? In a woman's life, the shape, size and health of her breast are an important issue. A beautiful breast is good for female
A breast implant for me? Information on breast enhancement A breast implant for me? In a woman's life, the shape, size and health of her breast are an important issue. A beautiful breast is good for female
The use of the Alexis device in breast augmentation to improve outcomes: a comparative randomized case-control survey
 Original Article The use of the Alexis device in breast augmentation to improve outcomes: a comparative randomized case-control survey Luca Andrea Dessy 1, Nefer Fallico 1, Francesco Serratore 1, Diego
Original Article The use of the Alexis device in breast augmentation to improve outcomes: a comparative randomized case-control survey Luca Andrea Dessy 1, Nefer Fallico 1, Francesco Serratore 1, Diego
inding the fit that s right for you. Your Surgery Planner For Breast Augmentation or Reconstruction with NATRELLE Saline-Filled Breast Implants
 inding the fit that s right for you. Your Surgery Planner For Breast Augmentation or Reconstruction with NATRELLE Saline-Filled Breast Implants L Place Your Device Identification Card(s) Here R INTRODUCTION
inding the fit that s right for you. Your Surgery Planner For Breast Augmentation or Reconstruction with NATRELLE Saline-Filled Breast Implants L Place Your Device Identification Card(s) Here R INTRODUCTION
Consumer summary Autologous fat transfer for breast augmentation (The report of the Review Group has been adapted for consumers by E.
 ASERNIP S Australian Safety and Efficacy Register of New Interventional Procedures Surgical Consumer summary Autologous fat transfer for breast augmentation (The report of the Review Group has been adapted
ASERNIP S Australian Safety and Efficacy Register of New Interventional Procedures Surgical Consumer summary Autologous fat transfer for breast augmentation (The report of the Review Group has been adapted
Revisions in Breast Augmentation
 Revisions in Breast Augmentation Editor s note: My thanks to the moderator, Neal Handel, MD (board-certified plastic surgeon and ASAPS member, Los Angeles, CA), and to panelists Barbara B. Hayden, MD (board-certified
Revisions in Breast Augmentation Editor s note: My thanks to the moderator, Neal Handel, MD (board-certified plastic surgeon and ASAPS member, Los Angeles, CA), and to panelists Barbara B. Hayden, MD (board-certified
Frederick J. Duffy, Jr., MD, FACS and Brice W. McKane, MD, FACS BREAST RECONSTRUCTION
 Frederick J. Duffy, Jr., MD, FACS and Brice W. McKane, MD, FACS BREAST RECONSTRUCTION BREAST RECONSTRUCTION: A WOMAN S DECISION Options and Information Our approach to breast reconstruction entails a very
Frederick J. Duffy, Jr., MD, FACS and Brice W. McKane, MD, FACS BREAST RECONSTRUCTION BREAST RECONSTRUCTION: A WOMAN S DECISION Options and Information Our approach to breast reconstruction entails a very
All about the GCA Comfort Guarantee. GCA Comfort Guarantee
 All about the GCA Comfort Guarantee. GCA Comfort Guarantee Beautiful results, complete with peace of mind. GC Aesthetics (GCA) is the company behind Nagor and Eurosilicone, brands built around a shared
All about the GCA Comfort Guarantee. GCA Comfort Guarantee Beautiful results, complete with peace of mind. GC Aesthetics (GCA) is the company behind Nagor and Eurosilicone, brands built around a shared
ASPS Recommended Insurance Coverage Criteria for Third- Party Payers
 ASPS Recommended Insurance Coverage Criteria for Third- Party Payers Breast Implant Associated Anaplastic Large Cell Lymphoma BACKGROUND Anaplastic Large Cell Lymphoma (ALCL) is a rare type of cancer of
ASPS Recommended Insurance Coverage Criteria for Third- Party Payers Breast Implant Associated Anaplastic Large Cell Lymphoma BACKGROUND Anaplastic Large Cell Lymphoma (ALCL) is a rare type of cancer of
Breast implants What types of implants are available Under or On top of muscle placement of breast implants Miss Anita Hazari MBBS, MD, FRCS (Plast)
 Breast implants What types of implants are available Breast implants may be referred to as Breast Augmentation. There are two types of implants that are commonly used in the UK - Silicone and Saline. Both
Breast implants What types of implants are available Breast implants may be referred to as Breast Augmentation. There are two types of implants that are commonly used in the UK - Silicone and Saline. Both
Information about breast augmentation (enlargement) surgery Part 1 of 3
 Information about breast augmentation (enlargement) surgery Part 1 of 3 This leaflet explains breast augmentation surgery. It is important that you read this information carefully and completely. Please
Information about breast augmentation (enlargement) surgery Part 1 of 3 This leaflet explains breast augmentation surgery. It is important that you read this information carefully and completely. Please
IMPORTANT INFORMATION FOR AUGMENTATION PATIENTS ABOUT MENTOR MEMORYGEL SILICONE GEL-FILLED BREAST IMPLANTS August 2006
 1 Canadian IMPORTANT INFORMATION FOR AUGMENTATION PATIENTS ABOUT MENTOR MEMORYGEL SILICONE GEL-FILLED BREAST IMPLANTS August 2006 2 11859-00 1 Important Information for Augmentation Patients About Mentor
1 Canadian IMPORTANT INFORMATION FOR AUGMENTATION PATIENTS ABOUT MENTOR MEMORYGEL SILICONE GEL-FILLED BREAST IMPLANTS August 2006 2 11859-00 1 Important Information for Augmentation Patients About Mentor
Individual Women. Individual Choices. onsidering Breast CEnhancement
 While every effort has been made to ensure the accuracy of the information contained in this booklet, Allergan accepts no responsibility and/or liability for errors or omissions. The information contained
While every effort has been made to ensure the accuracy of the information contained in this booklet, Allergan accepts no responsibility and/or liability for errors or omissions. The information contained
Aesthetic Plasuc Surgery 1993 Springer-Verlag New York Inc.
 Aesth. P ast. Surg. 17:233-237, 1993 Aesthetic Plasuc Surgery 1993 Springer-Verlag New York Inc. Calf Augmentation with New Solid Silicone Implants Gottfried Lemperle M.D. and Karlheinz Kostka, M.D. Frankfurt
Aesth. P ast. Surg. 17:233-237, 1993 Aesthetic Plasuc Surgery 1993 Springer-Verlag New York Inc. Calf Augmentation with New Solid Silicone Implants Gottfried Lemperle M.D. and Karlheinz Kostka, M.D. Frankfurt
Breast Implants & Imaging Considerations Update
 Fleitz Continuing Education Jeana Fleitz, M.E.D., RT(R)(M) The X-Ray Lady 6511 Glenridge Park Place, Suite 6 Louisville, KY 40222 Telephone (502) 425-0651 Fax (502) 327-7921 Website www.x-raylady.com Email
Fleitz Continuing Education Jeana Fleitz, M.E.D., RT(R)(M) The X-Ray Lady 6511 Glenridge Park Place, Suite 6 Louisville, KY 40222 Telephone (502) 425-0651 Fax (502) 327-7921 Website www.x-raylady.com Email
Breast Augmentation: What to Expect
 Breast Augmentation: What to Expect Electing to undergo breast augmentation surgery can be both thrilling and intimidating. Although the operation has many great benefits, many people are often unaware
Breast Augmentation: What to Expect Electing to undergo breast augmentation surgery can be both thrilling and intimidating. Although the operation has many great benefits, many people are often unaware
Secondary Breast Augmentation: Managing Each Case
 Aesth Plast Surg (2010) 34:691 700 DOI 10.1007/s00266-010-9510-1 ORIGINAL ARTICLE Secondary Breast Augmentation: Managing Each Case Javier de Benito Kyrenia Sanchez Received: 21 August 2009 / Accepted:
Aesth Plast Surg (2010) 34:691 700 DOI 10.1007/s00266-010-9510-1 ORIGINAL ARTICLE Secondary Breast Augmentation: Managing Each Case Javier de Benito Kyrenia Sanchez Received: 21 August 2009 / Accepted:
High-Resolution Ultrasound in the Detection of Silicone Gel Breast Implant Shell Failure: Background, In Vitro Studies, and Early Clinical Results
 Breast Surgery Preliminary Report High-Resolution Ultrasound in the Detection of Silicone Gel Breast Implant Shell Failure: Background, In Vitro Studies, and Early Clinical Results Aesthetic Surgery Journal
Breast Surgery Preliminary Report High-Resolution Ultrasound in the Detection of Silicone Gel Breast Implant Shell Failure: Background, In Vitro Studies, and Early Clinical Results Aesthetic Surgery Journal
CONTOUR PROFILE TISSUE EXPANDER WITH BUFFERZONE AREA
 1 ENGLISH Product Insert Data Sheet CONTOUR PROFILE TISSUE EXPANDER WITH BUFFERZONE AREA CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician or properly licensed
1 ENGLISH Product Insert Data Sheet CONTOUR PROFILE TISSUE EXPANDER WITH BUFFERZONE AREA CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician or properly licensed
Tips for using shaped implants in breast augmentation
 Tips for using shaped implants in breast augmentation Sientra would like to thank Dr. Patricia McGuire of St. Louis, MO for her significant contributions to Sientra s educational efforts. Dr. McGuire has
Tips for using shaped implants in breast augmentation Sientra would like to thank Dr. Patricia McGuire of St. Louis, MO for her significant contributions to Sientra s educational efforts. Dr. McGuire has
Statistics from the American Society of Plastic
 SPECIAL TOPIC Textured Silicone Breast Implant Use in Primary Augmentation: Core Data Update and Review Brian M. Derby, M.D. Mark A. Codner, M.D. Atlanta, Ga. Summary: Evolution of silicone breast implant
SPECIAL TOPIC Textured Silicone Breast Implant Use in Primary Augmentation: Core Data Update and Review Brian M. Derby, M.D. Mark A. Codner, M.D. Atlanta, Ga. Summary: Evolution of silicone breast implant
INFORMED-CONSENT-AUGMENTATION MAMMAPLASTY
 INFORMED-CONSENT-AUGMENTATION MAMMAPLASTY 2000 American Society of Plastic Surgeons. Purchasers of the Patient Consultation Resource Book are given a limited license to modify documents contained herein
INFORMED-CONSENT-AUGMENTATION MAMMAPLASTY 2000 American Society of Plastic Surgeons. Purchasers of the Patient Consultation Resource Book are given a limited license to modify documents contained herein
Executive Summary Silicone Gel-Filled Breast Implants General Issues Panel August 30-31, 2011
 Executive Summary Silicone Gel-Filled Breast Implants General Issues Panel August 30-31, 2011 Table of Contents I. Background... 1 a. Purpose of the Panel Meeting...1 b. FDA Update June 22, 2011...5 II.
Executive Summary Silicone Gel-Filled Breast Implants General Issues Panel August 30-31, 2011 Table of Contents I. Background... 1 a. Purpose of the Panel Meeting...1 b. FDA Update June 22, 2011...5 II.
INFORMED- CONSENT- BREAST IMPLANT REMOVAL SURGERY
 INFORMED- CONSENT- BREAST IMPLANT REMOVAL SURGERY INSTRUCTIONS This is an informed- consent document that has been prepared to help your plastic surgeon inform you concerning breast implant removal, its
INFORMED- CONSENT- BREAST IMPLANT REMOVAL SURGERY INSTRUCTIONS This is an informed- consent document that has been prepared to help your plastic surgeon inform you concerning breast implant removal, its
Information For Women AMERICAN SOCIETYOF PLASTIC SURGEONS
 Information For Women AMERICAN SOCIETYOF PLASTIC SURGEONS CONTENTS What are silicone implants?.............................................................4 Risks related to silicone gel-filled implants.................................................5
Information For Women AMERICAN SOCIETYOF PLASTIC SURGEONS CONTENTS What are silicone implants?.............................................................4 Risks related to silicone gel-filled implants.................................................5
Directions for Use. INAMED Style 410 Silicone-Filled Breast Implants. and TM owned by Allergan Inc Allergan Inc. L037-02
 Directions for Use INAMED Style 410 Silicone-Filled Breast Implants w w w a l l e r g a n c o m 110 Cochrane Drive Markham, ON L3R 9S1 8009628728 5540 Ekwill Street Santa Barbara, CA 93111 8006244261 and
Directions for Use INAMED Style 410 Silicone-Filled Breast Implants w w w a l l e r g a n c o m 110 Cochrane Drive Markham, ON L3R 9S1 8009628728 5540 Ekwill Street Santa Barbara, CA 93111 8006244261 and
BREAST RECONSTRUCTION
 BREAST RECONSTRUCTION PATIENT PLANNER SHAPE THAT HOLDS. SATISFACTION THAT LASTS. TABLE OF CONTENTS Page Pre-breast reconstruction surgery...3 About breast reconstruction...6 The surgery...14 Post-breast
BREAST RECONSTRUCTION PATIENT PLANNER SHAPE THAT HOLDS. SATISFACTION THAT LASTS. TABLE OF CONTENTS Page Pre-breast reconstruction surgery...3 About breast reconstruction...6 The surgery...14 Post-breast
Restoring a body. Restoring a life. Mentor Breast Reconstruction Product Portfolio NOT FOR DISTRIBUTION IN USA. APPROVED FOR EMEA REGION.
 Restoring a body Restoring a life Mentor Breast Reconstruction Product Portfolio NOT FOR DISTRIBUTION IN USA. APPROVED FOR EMEA REGION. Rebuilding beauty. Breast cancer is a reality in our world today
Restoring a body Restoring a life Mentor Breast Reconstruction Product Portfolio NOT FOR DISTRIBUTION IN USA. APPROVED FOR EMEA REGION. Rebuilding beauty. Breast cancer is a reality in our world today
Allograft Based Breast Reconstruction: Opportunity for a Second Look
 Allograft Based Breast Reconstruction: Opportunity for a Second Look Martin I. Newman, MD, FACS Director of Resident Education and Associate Program Director Department of Plastic and Reconstructive Surgery
Allograft Based Breast Reconstruction: Opportunity for a Second Look Martin I. Newman, MD, FACS Director of Resident Education and Associate Program Director Department of Plastic and Reconstructive Surgery
Directions for Use. NATRELLE Silicone-Filled Breast Implants and NATRELLE INSPIRA Breast Implants
 Directions for Use Directions for Use NATRELLE Silicone-Filled Breast Implants and NATRELLE INSPIRA Breast Implants Smooth & BIOCELL Texture Caution: Federal (USA) law restricts this device to sale by
Directions for Use Directions for Use NATRELLE Silicone-Filled Breast Implants and NATRELLE INSPIRA Breast Implants Smooth & BIOCELL Texture Caution: Federal (USA) law restricts this device to sale by
From ancient times to the present day, the aesthetic female breast has been portrayed. A Classification and Algorithm for Treatment of Breast Ptosis
 lassification and lgorithm for Treatment of reast Ptosis Laurence Kirwan, M ackground: The Regnault classification of breast ptosis is insufficient for determining surgical strategies for different stages
lassification and lgorithm for Treatment of reast Ptosis Laurence Kirwan, M ackground: The Regnault classification of breast ptosis is insufficient for determining surgical strategies for different stages
Interesting Case Series. Breast Augmentation
 Interesting Case Series Breast Augmentation Sachin M. Shridharani, MD, Justin L. Bellamy, BS, Mark M. Mofid, MD, and Navin K. Singh, MD Department of Plastic Surgery, The Johns Hopkins University School
Interesting Case Series Breast Augmentation Sachin M. Shridharani, MD, Justin L. Bellamy, BS, Mark M. Mofid, MD, and Navin K. Singh, MD Department of Plastic Surgery, The Johns Hopkins University School
Augmentation BREAST AUGMENTATION WITH NATRELLE SILICONE-FILLED BREAST IMPLANTS AND NATRELLE INSPIRA BREAST IMPLANTS
 Augmentation BREAST AUGMENTATION WITH NATRELLE SILICONE-FILLED BREAST IMPLANTS AND NATRELLE INSPIRA BREAST IMPLANTS Table of Contents Glossary... 2 1. Considering Silicone Gel-Filled Breast Implant Surgery...11
Augmentation BREAST AUGMENTATION WITH NATRELLE SILICONE-FILLED BREAST IMPLANTS AND NATRELLE INSPIRA BREAST IMPLANTS Table of Contents Glossary... 2 1. Considering Silicone Gel-Filled Breast Implant Surgery...11
Breast Augmentation with NATRELLE Silicone-Filled Breast Implants
 Breast Augmentation with NATRELLE Silicone-Filled Breast Implants AUG Patient Labeling Rev 4/6/09 page 1 TABLE OF CONTENTS Page GLOSSARY... 3 1.0 CONSIDERING SILICONE GEL-FILLED BREAST IMPLANT SURGERY...
Breast Augmentation with NATRELLE Silicone-Filled Breast Implants AUG Patient Labeling Rev 4/6/09 page 1 TABLE OF CONTENTS Page GLOSSARY... 3 1.0 CONSIDERING SILICONE GEL-FILLED BREAST IMPLANT SURGERY...
Increase of Visible Veins After Breast Augmentation. Yuri Andonakis, MD,* and Berend van der Lei, MD, PhD*
 BREAST SURGERY A Retrospective Analysis of 78 Consecutive Breast Augmentation Patients Yuri Andonakis, MD,* and Berend van der Lei, MD, PhD* Abstract: A retrospective study was undertaken to determine
BREAST SURGERY A Retrospective Analysis of 78 Consecutive Breast Augmentation Patients Yuri Andonakis, MD,* and Berend van der Lei, MD, PhD* Abstract: A retrospective study was undertaken to determine
Postreduction Breast Augmentation
 Postreduction Breast Augmentation Original Article Breast David A. Hidalgo, MD Melissa A. Doft, MD Background: Most breast reduction patients are highly satisfied after surgery. However, there is a subset
Postreduction Breast Augmentation Original Article Breast David A. Hidalgo, MD Melissa A. Doft, MD Background: Most breast reduction patients are highly satisfied after surgery. However, there is a subset
Preoperative planning and breast implant selection for volume difference management in asymmetrical breasts
 . Plast Aesthet Res 2017;4:108-15 DOI: 10.20517/2347-9264.2017.36 Original Article Plastic and Aesthetic Research www.parjournal.net Open Access Preoperative planning and breast implant selection for volume
. Plast Aesthet Res 2017;4:108-15 DOI: 10.20517/2347-9264.2017.36 Original Article Plastic and Aesthetic Research www.parjournal.net Open Access Preoperative planning and breast implant selection for volume
CASEY. Paralegal NATRELLE Style 15. YOUR SURGERY PLANNER Breast Augmentation With Natrelle Silicone-Filled Breast Implants
 The Right Time. The Right Fit. CASEY Paralegal NATRELLE Style 15 YOUR SURGERY PLANNER Breast Augmentation With Natrelle Silicone-Filled Breast Implants Dear Patient, Allergan has developed this AUGMENTATION
The Right Time. The Right Fit. CASEY Paralegal NATRELLE Style 15 YOUR SURGERY PLANNER Breast Augmentation With Natrelle Silicone-Filled Breast Implants Dear Patient, Allergan has developed this AUGMENTATION
MENTOR Course in Surgical Excellence:
 MENTOR Course in Surgical Excellence: Managing Complications and Revision Surgery April 23 rd -24 th, 2015 Beurs van Berlage Amsterdam, The Netherlands Managing Complications and Revision Surgery Objective
MENTOR Course in Surgical Excellence: Managing Complications and Revision Surgery April 23 rd -24 th, 2015 Beurs van Berlage Amsterdam, The Netherlands Managing Complications and Revision Surgery Objective
NIPPLE SPARING PRE-PECTORAL BREAST RECONSTRUCTION
 NIPPLE SPARING PRE-PECTORAL BREAST RECONSTRUCTION 42 yo female healthy athlete Right breast mass. Past medical history: none Family history: aunt with Breast cancer Candidates for nipple-sparing mastectomy
NIPPLE SPARING PRE-PECTORAL BREAST RECONSTRUCTION 42 yo female healthy athlete Right breast mass. Past medical history: none Family history: aunt with Breast cancer Candidates for nipple-sparing mastectomy
Based on my discussions with Dr Gutowski, I agree with, and choose to have the following options as part of my breast augmentation:
 INFORMED CONSENT FOR BREAST AUGMENTATION PLEASE REVIEW AND BRING WITH YOU ON THE DAY OF YOUR PROCEDURE PATIENT NAME Based on my discussions with Dr Gutowski, I agree with, and choose to have the following
INFORMED CONSENT FOR BREAST AUGMENTATION PLEASE REVIEW AND BRING WITH YOU ON THE DAY OF YOUR PROCEDURE PATIENT NAME Based on my discussions with Dr Gutowski, I agree with, and choose to have the following
MISS CAROLINE PAYNE. Breast Augmentation
 MISS CAROLINE PAYNE BSc (Hons) MSc FRCS (Eng) FRCS(Plast) Consultant Plastic Reconstructive Surgeon Breast Augmentation What types of implants are available? Breast implant surgery may be referred to as
MISS CAROLINE PAYNE BSc (Hons) MSc FRCS (Eng) FRCS(Plast) Consultant Plastic Reconstructive Surgeon Breast Augmentation What types of implants are available? Breast implant surgery may be referred to as
COPE Library Sample
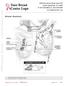 Breast Anatomy LOBULE LOBE ACINI (MILK PRODUCING UNITS) NIPPLE AREOLA COMPLEX ENLARGEMENT OF DUCT AND LOBE LOBULE SUPRACLAVICULAR NODES INFRACLAVICULAR NODES DUCT DUCT ACINI (MILK PRODUCING UNITS) 8420
Breast Anatomy LOBULE LOBE ACINI (MILK PRODUCING UNITS) NIPPLE AREOLA COMPLEX ENLARGEMENT OF DUCT AND LOBE LOBULE SUPRACLAVICULAR NODES INFRACLAVICULAR NODES DUCT DUCT ACINI (MILK PRODUCING UNITS) 8420
For women at hereditary risk for breast carcinoma, risk reduction. Reoperations after Prophylactic Mastectomy with or without Implant Reconstruction
 2152 Reoperations after Prophylactic Mastectomy with or without Implant Reconstruction Sara M. Zion, M.D. 1 Jeffrey M. Slezak, M.S. 2 Thomas A. Sellers, Ph.D. 2 John E. Woods, M.D. 3 Phillip G. Arnold,
2152 Reoperations after Prophylactic Mastectomy with or without Implant Reconstruction Sara M. Zion, M.D. 1 Jeffrey M. Slezak, M.S. 2 Thomas A. Sellers, Ph.D. 2 John E. Woods, M.D. 3 Phillip G. Arnold,
