Questions and answers about the laboratory diagnosis of Clostridium difficile infection (CDI)
|
|
|
- Martha Lamb
- 5 years ago
- Views:
Transcription
1 Questions and answers about the laboratory diagnosis of Clostridium difficile infection (CDI) The NHS Centre for Evidence based Purchasing (CEP) has published the results of an evaluation of the performance of commercial kits for the detection of C. difficile toxins ( EP/CEP08054.pdf). The evaluation represents the largest single study of C. difficile toxin detection kits. None of the nine assays tested had a particularly high sensitivity. Also, the poor positive predictive values of the kits, especially in the context of widespread testing, raises doubts about their appropriateness when used as single tests for the laboratory detection of C. difficile toxins. The results of the CEP evaluation are consistent with those of a recently published review of (smaller) studies that have examined the performance of C. difficile toxin detection kits (Planche et al., Lancet Infect Dis 2008;8:777-84). The following Questions and Answers are in response to the results of the CEP evaluation. How does the diagnosis of CDI affect how patients are cared for? The minority of patients (about 10-20%) with hospital acquired diarrhoea have CDI. There are no robust clinical features that rule in or rule out a diagnosis of CDI. Patients with diarrhoea should be isolated to prevent transmission of norovirus, C. difficile or other transmissible pathogens. Patients should also be considered for treatment of CDI before the results of tests are available, particularly if symptoms/signs indicate severe infection. How is C. difficile detected in the laboratory? The most common tests (EIAs) detect the C. difficile toxin not the bacterium; cytotoxin testing also detects C. difficile toxins, but does this using an in-vitro cell sheet assay as the detection method. Alternative approaches include culture (which is relatively slow and requires supplementary testing to confirm that an isolate is a toxin producer) and detection of an antigen (e.g. the enzyme glutamate dehydrogenase) or a gene (e.g. the toxin B gene). When is it not appropriate to submit faecal samples for the laboratory detection of C. difficile? Only diarrhoeal samples (i.e. unformed stools) should be tested for the presence of C. difficile toxins. If there is a clear explanation (not related to infection) for the onset of diarrhoea (for example, laxative use to overcome constipation) then it is not helpful to submit faecal samples for the detection of C. difficile/toxins. If diarrhoea continues or the patient has other signs or symptoms consistent with CDI then it is appropriate to submit faecal samples for the laboratory detection of C. difficile toxin. It is not recommended to routinely test faecal samples from children aged less than 2 years. (Please note that in Scotland there is mandatory surveillance of CDI in all patients greater than 15 years of age) It should be noted that occasional patients with severe CDI may have little of no diarrhoea.
2 2 What are the implications of an incorrect laboratory test? A false negative result may result in a patient being thought to be free of CDI. This patient may then receive sub-optimum care, and may be not be appropriately isolated, thus increasing the risk of transmission of C. difficile. The consequences of false positives are perhaps less well appreciated. These patients may subsequently have necessary antibiotics curtailed or changed, and receive unnecessary treatment for CDI. Such patients might be nursed in wards or bays together with genuine cases, putting them at increased risk of acquiring true CDI. How has the present uncertainty about the diagnosis of CDI arisen? Almost all laboratory tests for the detection of a bacterium or its products can sometimes give false positive or false negative results. However, recently there has been a large rise in the number of C. difficile tests being performed. Such widespread testing increases the chance of false positive results (as the great majority of patients tested for CDI do not have this infection). Are some toxin detection kits better than others? It is apparent from the available published data on the performance of commercial toxin detection kits, that there may be significant differences in the sensitivity and specificity of these kits. If using these assays, laboratories should ensure that they are using one of the better performing kits. In the CEP evaluation no single assay was clearly superior in terms of both sensitivity and specificity. However, five assays (Remel Xpect, Techlab Tox A/B Quik Chek, Premier Toxin A + B, Vidas C. difficile Toxin A & B and Techlab Toxin A/B II) appear to be superior to the other four. However, even equally good kits can produce very different results. More sensitive tests tend to be less specific and vice versa. Are toxin detection kits sufficiently accurate to be used as single tests for the detection of C. difficile toxins? The currently available kits for detection of C. difficile toxins have variable performance. Recent studies show that the currently available kits may miss 10-20% of CDI cases, and will falsely identify cases as positive when they are not. The poor positive predictive values of toxin detection kits, especially in the context of widespread testing, and the possibility of missing true positives mean there are limitations to using these as single tests for the laboratory diagnosis of CDI. How should the results from toxin detection kits be interpreted? In general the kits are better at ruling out the presence of C. difficile toxins (i.e. they have a high negative predictive value). However, the kits generally have sub-optimal positive predictive values, and thus positive results may sometimes actually be false positives. Some tests miss more positive results than others; for example, approximately 1 in 5 cases of CDI. In general, more sensitive tests that identify a high proportion of the true cases also tend to produce more false positive results. The converse is also true; so, tests with high positive predictive values tend to miss cases of CDI. This means that caution should be applied to the interpretation of toxin detection results generated using currently available kits, particularly if the clinical details are not consistent with the test result.
3 3 Are there particular situations when a test has a low positive predictive value and will yield more incorrect results? In settings where the disease is rare (i.e. low prevalence) tests with a low positive predictive value will more often yield false positive results. In the community setting where CDI is less common particular care should be taken in interpreting positive results on samples from patients who have not recently been hospitalised. When CDI is uncommon or unlikely then caution should be exercised in interpreting test results, and consideration should be given to altering the testing strategy. What is the best single test for the detection of C. difficile toxins? Well-performed cell-culture cytotoxicity assays are still regarded as the gold standard for diagnostic testing. However, these tests have a disadvantage in terms of speed, which may require the use of another rapid assay as an initial screen, particularly in outbreak or endemic settings. Is the cytotoxin test a practicable alternative for routine microbiology laboratories? Cytotoxin testing used to be the most commonly performed test for the laboratory diagnosis of CDI. As laboratories have moved away from carrying out cell culture based methods, this has partly hastened the switch away from cytotoxicity testing, which requires the maintenance of a cell line. While this approach could still be technically feasible for some laboratories, there are other issues to be considered including the significant additional time delay before a result is available. There is also the possibility of sending faecal samples to another laboratory, as these may be stored at 4 o C for several days before re-testing. Should laboratories that are currently using EIA kits alter their practice? Currently available kits for the detection of toxins are generally better at excluding the presence of toxins. However, caution is required in the interpretation of toxin-positive results to ensure that these are consistent with the clinical presentation. The issuing of interpretive comments with reports may aid clinicians in interpreting results. The use of a confirmatory test, as part of a diagnostic algorithm, will increase the accuracy of toxin-positive results. Should laboratories change to using two step protocols (one test followed by another test) for the diagnosis of CDI? The use of a confirmatory or second test, as part of a diagnostic algorithm, will increase the accuracy of toxin-positive results. However, the choice of first and second test is unclear at present. If the first test lacks sensitivity and only positives are checked by a second test then the overall testing strategy will still miss true positives. The use of the gold standard cell cytotoxicity assay is the most accurate test, although there may be practical considerations in the introduction of this assay. Provisional positive results could be issued whilst awaiting confirmation by a cytotoxicity assay. There is also the possibility of performing confirmatory cytotoxicity assays in another laboratory as faecal samples may be stored at 4 o C for several days before re-testing. Can storage and/or transportation of faecal specimens at sub-optimal temperatures change the outcome of the toxin detection assays?
4 4 C. difficile toxin is labile, and degrades at room temperature. False-negative results occur if specimens are not tested promptly or kept refrigerated until testing can be performed. What is the role of membrane-based immunoassay kits? There is no evidence the performance of this kind of assay is any better than that of other assays. These assays are quick to perform, and lend themselves to ad hoc testing without the need to batch specimens. This makes them suitable for situations where a result is required rapidly, or sample volumes are low. However in comparison with well-based assays, their specificity tends to be higher, with a concomitant relative decrease in sensitivity. Are alternative methods to detect C. difficile, such as assays for GDH or PCR for toxin B gene more accurate than toxin assays? Currently available alternative test methods, such as GDH or toxin B PCR tests, may have a role as part of a diagnostic algorithm for the diagnosis of CDI. However, from the information currently available, as standalone tests, their performance is not significantly more accurate than some of the toxin assays. How can we compare the accuracy of different assays for the laboratory diagnosis of CDI? There are now a number of studies which have compared, either directly or by meta-analysis of published studies, the accuracy of many of the currently available tests. However, information on some of the newer tests that do not directly detect C. difficile toxins is less widely available. Should laboratories redefine cut-off levels that define positive results for C. difficile toxin detection kits? Manufacturers instructions should always be followed. We do not currently recommend redefining cut off values, without research using extensive comparisons to reference standards. Should multiple repeat samples be submitted for testing No. If a negative test for the presence of C. difficile toxins has been obtained, it is appropriate to consider one repeat test if there remains a high clinical suspicion of CDI. Otherwise, submission of multiple samples for testing for the presence of C. difficile toxins should be avoided as, with tests that have a sub-optimal positive predictive value, this increases the chance of obtaining false positive results. Are further tests required to demonstrate clearance of the organism? No. Tests of cure (i.e. clearance testing) are not clinically helpful as toxin excretion may continue even when symptoms have stopped.
5 5 Does exclusion of formed stools for testing not cause the sensitivity of toxin testing to go down? No. There is evidence that demonstrates that restricting of testing to diarrhoeal (unformed) stools does not cause the diagnosis of CDI to be missed. Should toxin testing be restricted to patients with known risk factors for CDI? No. It is apparent that currently circulating strains of C. difficile cause CDI in individuals without conventional risk factors, including patients in the community. Can results of toxin testing be discounted in the setting of norovirus infection? No. Suspected or confirmed norovirus infection is not a reason for not testing for the presence of C. difficile toxins. However, potential interactions between the two organisms are poorly understood. How do the results of the CEP evaluation affect the accuracy of C. difficile surveillance data? Local surveillance data based upon a standardised approach to testing, using consistent methodology, reported in a timely fashion, and acted upon appropriately is likely to reflect genuine changes in the level of CDI, and is ultimately the most important tool in controlling the infection at the local level. At a national surveillance level, amalgamation of data based upon different methodologies will inevitably introduce some unwanted noise into the system, but this approach can still provide valuable data on whether CDI is being reduced. Changes to the method being used for the laboratory diagnosis of CDI, including the numbers of samples analysed, should be recorded and audited locally so that the implications for surveillance data can be understood. What key questions need to be answered concerning the optimum laboratory diagnosis of CDI? There is a clear need for research into the optimisation of the laboratory diagnosis of CDI. It is likely that a two stage algorithm will be a simple and practical way to improve diagnosis. However, there needs to be research into how this is best achieved. Guidance on the optimum way of carrying out cytotoxin testing is also required. There may be scope for redefining cut-off values in diagnostic kits, but this requires large numbers of samples to ensure a robust outcome and is not recommended as part of routine practice. Further research is needed concerning the concordance of laboratory test results with CDI symptoms and outcome. Adapted by HPS from the CDI Diagnosis Working Group M H Wilcox J Coia T Planche 25. March 2009
C. Difficile Testing Protocol
 C. Difficile Testing Protocol Caroline Donovan, RN, BSN, ONC- Infection Control Practitioner Abegail Pangan, RN, MSN, CIC- Infection Control Practitioner U.S. NEWS & WORLD REPORT 2017 2018 RANKINGS Acute
C. Difficile Testing Protocol Caroline Donovan, RN, BSN, ONC- Infection Control Practitioner Abegail Pangan, RN, MSN, CIC- Infection Control Practitioner U.S. NEWS & WORLD REPORT 2017 2018 RANKINGS Acute
Protocol for the Scottish Surveillance Programme for Clostridium difficile infection.
 National Services Scotland Protocol for the Scottish Surveillance Programme for Clostridium difficile infection. User manual. Version 4.0 Revised January 2017 Health Protection Scotland is a division of
National Services Scotland Protocol for the Scottish Surveillance Programme for Clostridium difficile infection. User manual. Version 4.0 Revised January 2017 Health Protection Scotland is a division of
on November 8, 2018 by guest
 JCM Accepts, published online ahead of print on December 00 J. Clin. Microbiol. doi:./jcm.01-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
JCM Accepts, published online ahead of print on December 00 J. Clin. Microbiol. doi:./jcm.01-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
DUKE ANTIMICROBIAL STEWARDSHIP OUTREACH NETWORK (DASON) Antimicrobial Stewardship News. Volume 3, Number 6, June 2015
 DUKE ANTIMICROBIAL STEWARDSHIP OUTREACH NETWORK (DASON) Antimicrobial Stewardship News Volume 3, Number 6, June 2015 Diagnostic Testing for Clostridium difficile Infection Background Clostridium difficile
DUKE ANTIMICROBIAL STEWARDSHIP OUTREACH NETWORK (DASON) Antimicrobial Stewardship News Volume 3, Number 6, June 2015 Diagnostic Testing for Clostridium difficile Infection Background Clostridium difficile
EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE
 EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click
EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click
Clostridium difficile infection (CDI) Week 52 (Ending 30/12/2017)
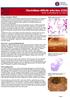 Clostridium difficile infection (CDI) Week 52 (Ending 30/12/2017) What is Clostridium difficile? Clostridium difficile is a Gram-positive anaerobic spore forming bacillus. It is ubiquitous in nature and
Clostridium difficile infection (CDI) Week 52 (Ending 30/12/2017) What is Clostridium difficile? Clostridium difficile is a Gram-positive anaerobic spore forming bacillus. It is ubiquitous in nature and
C. DIFF QUIK CHEK COMPLETE. Get the complete diagnostic picture with just one test. TAP HERE TO SEE THE NEXT PAGE
 C. DIFF QUIK CHEK COMPLETE Get the complete diagnostic picture with just one test. TAP HERE TO SEE THE PAGE CLINICIAN Would actionable C. difficile test results in less than 30 minutes improve patient
C. DIFF QUIK CHEK COMPLETE Get the complete diagnostic picture with just one test. TAP HERE TO SEE THE PAGE CLINICIAN Would actionable C. difficile test results in less than 30 minutes improve patient
Guidance for obtaining faecal specimens from patients with diarrhoea (Background information)
 Guidance for obtaining faecal specimens from patients with diarrhoea (Background information) Version 1.0 Date of Issue: January 2009 Review Date: January 2010 Page 1 of 11 Contents 1. Introduction...
Guidance for obtaining faecal specimens from patients with diarrhoea (Background information) Version 1.0 Date of Issue: January 2009 Review Date: January 2010 Page 1 of 11 Contents 1. Introduction...
GUIDELINE FOR THE MANAGEMENT OF ANTIBIOTIC- ASSOCIATED DIARRHOEA IN ADULTS
 GUIDELINE FOR THE MANAGEMENT OF ANTIBIOTIC- ASSOCIATED DIARRHOEA IN ADULTS Version 3.0 Date ratified May 2008 Review date May 2010 Ratified by NUH Antibiotic Guidelines Committee NUH Drugs and Therapeutics
GUIDELINE FOR THE MANAGEMENT OF ANTIBIOTIC- ASSOCIATED DIARRHOEA IN ADULTS Version 3.0 Date ratified May 2008 Review date May 2010 Ratified by NUH Antibiotic Guidelines Committee NUH Drugs and Therapeutics
JMSCR Vol 05 Issue 07 Page July 2017
 www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i7.15 Rapid Diagnosis of Toxigenic Clostridium
www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i7.15 Rapid Diagnosis of Toxigenic Clostridium
Clostridium difficile Infection (CDI) Guideline Update:
 Clostridium difficile Infection (CDI) Guideline Update: Understanding the Data Behind the Recommendations Erik R. Dubberke, MD, MSPH A Webinar for HealthTrust Members Professor of Medicine September 24,
Clostridium difficile Infection (CDI) Guideline Update: Understanding the Data Behind the Recommendations Erik R. Dubberke, MD, MSPH A Webinar for HealthTrust Members Professor of Medicine September 24,
Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister
 Li StarFish S.r.l. Via Cavour, 35-20063 Cernusco S/N (MI), Italy Tel. +39-02-92150794 - Fax. +39-02-92157285 info@listarfish.it -www.listarfish.it Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister One
Li StarFish S.r.l. Via Cavour, 35-20063 Cernusco S/N (MI), Italy Tel. +39-02-92150794 - Fax. +39-02-92157285 info@listarfish.it -www.listarfish.it Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister One
L. Clifford McDonald, MD. Senior Advisor for Science and Integrity September 16, 2015
 Controversies and Current Issues in Diagnosis, Surveillance, and Treatment of Clostridium difficile infeciton L. Clifford McDonald, MD Senior Advisor for Science and Integrity September 16, 2015 Division
Controversies and Current Issues in Diagnosis, Surveillance, and Treatment of Clostridium difficile infeciton L. Clifford McDonald, MD Senior Advisor for Science and Integrity September 16, 2015 Division
Clostridium difficile infection surveillance: Applying the case definition
 Clostridium difficile infection surveillance: Applying the case definition PICNet Conference March 3 rd 2016 Presented by: Tara Leigh Donovan, MSc Managing Consultant (Former Epidemiologist) 1 Disclaimer
Clostridium difficile infection surveillance: Applying the case definition PICNet Conference March 3 rd 2016 Presented by: Tara Leigh Donovan, MSc Managing Consultant (Former Epidemiologist) 1 Disclaimer
Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate
 Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate Objectives Summarize the changing epidemiology and demographics of patients at risk for Clostridium
Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate Objectives Summarize the changing epidemiology and demographics of patients at risk for Clostridium
Clostridium difficile Diagnostic and Clinical Challenges
 Papers in Press. Published December 11, 2015 as doi:10.1373/clinchem.2015.243717 The latest version is at http://hwmaint.clinchem.org/cgi/doi/10.1373/clinchem.2015.243717 Clinical Chemistry 62:2 000 000
Papers in Press. Published December 11, 2015 as doi:10.1373/clinchem.2015.243717 The latest version is at http://hwmaint.clinchem.org/cgi/doi/10.1373/clinchem.2015.243717 Clinical Chemistry 62:2 000 000
Overcoming barriers to effective recognition and diagnosis of Clostridium difficile infection
 REVIEW 10.1111/1469-0691.12057 Overcoming barriers to effective recognition and diagnosis of Clostridium difficile infection M. H. Wilcox Department of Microbiology, Old Medical School, Leeds Teaching
REVIEW 10.1111/1469-0691.12057 Overcoming barriers to effective recognition and diagnosis of Clostridium difficile infection M. H. Wilcox Department of Microbiology, Old Medical School, Leeds Teaching
"QUIK CHEK"[tw])) NOT ((therapy[ti] OR treatment[ti] OR treat*[ti]) NOT (diagnosis[ti] OR
![QUIK CHEK[tw])) NOT ((therapy[ti] OR treatment[ti] OR treat*[ti]) NOT (diagnosis[ti] OR QUIK CHEK[tw])) NOT ((therapy[ti] OR treatment[ti] OR treat*[ti]) NOT (diagnosis[ti] OR](/thumbs/81/84207658.jpg) Supporting information Appendix 1: Search strategy Pubmed (("Clostridium Infections/diagnosis"[majr] AND "Clostridium difficile"[majr]) OR (((("Clostridium difficile"[majr] OR "Clostridium Difficile"[ti])
Supporting information Appendix 1: Search strategy Pubmed (("Clostridium Infections/diagnosis"[majr] AND "Clostridium difficile"[majr]) OR (((("Clostridium difficile"[majr] OR "Clostridium Difficile"[ti])
Clostridium difficile Infection (CDI) Management Guideline
 Clostridium difficile Infection (CDI) Management Guideline Do not test all patients with loose or watery stools for CDI o CDI is responsible for
Clostridium difficile Infection (CDI) Management Guideline Do not test all patients with loose or watery stools for CDI o CDI is responsible for
Clostridium difficile Testing Algorithms Using Glutamate Dehydrogenase Antigen and C.
 JCM Accepts, published online ahead of print on 18 January 2012 J. Clin. Microbiol. doi:10.1128/jcm.05620-11 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 3 Clostridium difficile
JCM Accepts, published online ahead of print on 18 January 2012 J. Clin. Microbiol. doi:10.1128/jcm.05620-11 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 3 Clostridium difficile
Clostridium difficile Infection: Diagnosis and Management
 Clostridium difficile Infection: Diagnosis and Management Brian Viviano D.O. Case study 42 year old female with history of essential hypertension and COPD presents to ED complaining of 24 hours of intractable,
Clostridium difficile Infection: Diagnosis and Management Brian Viviano D.O. Case study 42 year old female with history of essential hypertension and COPD presents to ED complaining of 24 hours of intractable,
SC P. on POOP. Susan E. Sharp, Ph.D., DABMM Director of Microbiology Portland, OR
 The Straight SC P on POOP Susan E. Sharp, Ph.D., DABMM Director of Microbiology Portland, OR 97230 susan.e.sharp@kp.org But FIRST - - Quiz time! But FIRST - - Quiz time! Here s the QUIZ 4 Here s the QUIZ
The Straight SC P on POOP Susan E. Sharp, Ph.D., DABMM Director of Microbiology Portland, OR 97230 susan.e.sharp@kp.org But FIRST - - Quiz time! But FIRST - - Quiz time! Here s the QUIZ 4 Here s the QUIZ
Pros and Cons of Alternative Diagnostic Testing Strategies for C. difficile Infection
 Pros and Cons of Alternative Diagnostic Testing Strategies for C. difficile Infection Christopher R. Polage, MD, MAS Associate Professor of Pathology and Infectious Diseases UC Davis Disclosures Test materials
Pros and Cons of Alternative Diagnostic Testing Strategies for C. difficile Infection Christopher R. Polage, MD, MAS Associate Professor of Pathology and Infectious Diseases UC Davis Disclosures Test materials
A Pharmacist Perspective
 Leveraging Technology to Reduce CDI A Pharmacist Perspective Ed Eiland, Pharm.D., MBA, BCPS (AQ-ID) Clinical Practice and Business Supervisor Huntsville Hospital System Huntsville Hospital 881 licensed
Leveraging Technology to Reduce CDI A Pharmacist Perspective Ed Eiland, Pharm.D., MBA, BCPS (AQ-ID) Clinical Practice and Business Supervisor Huntsville Hospital System Huntsville Hospital 881 licensed
2.1 HIV antibody tests
 Over 100 different kinds of HIV test are currently available worldwide. More are likely to become available in the future. The choice of test depends on factors including laboratory requirements, how easy
Over 100 different kinds of HIV test are currently available worldwide. More are likely to become available in the future. The choice of test depends on factors including laboratory requirements, how easy
MICROBIOLOGICAL TESTING IN PICU
 MICROBIOLOGICAL TESTING IN PICU This is a guideline for the taking of microbiological samples in PICU to diagnose or exclude infection. The diagnosis of infection requires: Ruling out non-infectious causes
MICROBIOLOGICAL TESTING IN PICU This is a guideline for the taking of microbiological samples in PICU to diagnose or exclude infection. The diagnosis of infection requires: Ruling out non-infectious causes
Reference assays for Clostridium difficile infection: one or two gold standards?
 1 Centre for Infection, Division of Cellular and Molecular Medicine, St George s University of London, Cranmer Terrace, London, UK 2 Microbiology, Leeds Teaching Hospitals and University of Leeds, Old
1 Centre for Infection, Division of Cellular and Molecular Medicine, St George s University of London, Cranmer Terrace, London, UK 2 Microbiology, Leeds Teaching Hospitals and University of Leeds, Old
Procedure for the Prevention Control and Management of Clostridium difficile Infection in Care Settings in Shetland
 Procedure for the Prevention Control and Management of Clostridium difficile Infection in Care Settings in Shetland Adapted from: Model Infection Control Policies (Transmission Based), HPS ICT September
Procedure for the Prevention Control and Management of Clostridium difficile Infection in Care Settings in Shetland Adapted from: Model Infection Control Policies (Transmission Based), HPS ICT September
Infection Prevention & Control Core Skills Level 2
 Infection Prevention & Control Core Skills Level 2 Learning outcomes Risk assessment of patients Critical examination of the situation MRSA, CDT & CPE Ongoing challenges future-proofing infection control
Infection Prevention & Control Core Skills Level 2 Learning outcomes Risk assessment of patients Critical examination of the situation MRSA, CDT & CPE Ongoing challenges future-proofing infection control
Lifting the lid on a difficile problem part 2 (Clinical) Evidence Based Practice. Problem in evolution (1) Problem in evolution (1) Interventions (2)
 Lifting the lid on a difficile problem part (Clinical) Dr Philip T Mannion Consultant Microbiologist, Rhyl Evidence Based Practice Antibiotic prescribing guidance Isolation policy Hand hygiene (soap and
Lifting the lid on a difficile problem part (Clinical) Dr Philip T Mannion Consultant Microbiologist, Rhyl Evidence Based Practice Antibiotic prescribing guidance Isolation policy Hand hygiene (soap and
DETECTION OF TOXIGENIC CLOSTRIDIUM DIFFICILE
 CLINICAL GUIDELINES For use with the UnitedHealthcare Laboratory Benefit Management Program, administered by BeaconLBS DETECTION OF TOXIGENIC CLOSTRIDIUM DIFFICILE Policy Number: PDS 021 Effective Date:
CLINICAL GUIDELINES For use with the UnitedHealthcare Laboratory Benefit Management Program, administered by BeaconLBS DETECTION OF TOXIGENIC CLOSTRIDIUM DIFFICILE Policy Number: PDS 021 Effective Date:
Jen KOK*, Qinning WANG, Lee C THOMAS, Gwendolyn L GILBERT. Centre for Infectious Diseases and Microbiology Laboratory Services, Institute of
 JCM Accepts, published online ahead of print on 17 August 2011 J. Clin. Microbiol. doi:10.1128/jcm.00752-11 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All
JCM Accepts, published online ahead of print on 17 August 2011 J. Clin. Microbiol. doi:10.1128/jcm.00752-11 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All
Articles. Funding Department of Health and Health Protection Agency, UK.
 Differences in outcome according to Clostridium difficile testing method: a prospective multicentre diagnostic validation study of C difficile infection Timothy D Planche, Kerrie A Davies, Pietro G Coen,
Differences in outcome according to Clostridium difficile testing method: a prospective multicentre diagnostic validation study of C difficile infection Timothy D Planche, Kerrie A Davies, Pietro G Coen,
ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Clostridium difficile Infections
 ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Clostridium difficile Infections Christina M. Surawicz, MD 1, Lawrence J. Brandt, MD 2, David G. Binion, MD 3, Ashwin N. Ananthakrishnan,
ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Clostridium difficile Infections Christina M. Surawicz, MD 1, Lawrence J. Brandt, MD 2, David G. Binion, MD 3, Ashwin N. Ananthakrishnan,
Guidance on the use of molecular testing for Neisseria gonorrhoeae in Diagnostic Laboratories 2011
 Guidance on the use of molecular testing for Neisseria gonorrhoeae in Diagnostic Laboratories 2011 Molecular testing for gonorrhoea working group (see Appendix) Prepared by Kirstine Eastick March 2012-1
Guidance on the use of molecular testing for Neisseria gonorrhoeae in Diagnostic Laboratories 2011 Molecular testing for gonorrhoea working group (see Appendix) Prepared by Kirstine Eastick March 2012-1
Original Article. Infection Control & Hospital Epidemiology (2019), 1 5 doi: /ice
 Infection Control & Hospital Epidemiology (2019), 1 5 doi:10.1017/ice.2018.347 Original Article Healthcare provider diagnostic testing practices for identification of Clostridioides (Clostridium) difficile
Infection Control & Hospital Epidemiology (2019), 1 5 doi:10.1017/ice.2018.347 Original Article Healthcare provider diagnostic testing practices for identification of Clostridioides (Clostridium) difficile
Introduction ORIGINAL ARTICLE 1
 ORIGINAL ARTICLE 1 Laboratory diagnosis of Clostridium difficile infection. An evaluation of tests for faecal toxin, glutamate dehydrogenase, lactoferrin and toxigenic culture in the diagnostic laboratory
ORIGINAL ARTICLE 1 Laboratory diagnosis of Clostridium difficile infection. An evaluation of tests for faecal toxin, glutamate dehydrogenase, lactoferrin and toxigenic culture in the diagnostic laboratory
Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review
 Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review October 18, 2010 James Kahn and Carolyn Kenney, MSIV Overview Burden of disease associated
Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review October 18, 2010 James Kahn and Carolyn Kenney, MSIV Overview Burden of disease associated
6/14/2012. Welcome! PRESENTATION OUTLINE CLOSTRIDIUM DIFFICILE PREVENTION. Teaming Up to Prevent Infections! 1) Impact. 2) Testing Recommendations
 CLOSTRIDIUM DIFFICILE PREVENTION Beth Goodall, RN, BSN Board Certified in Infection Prevention and Control DCH Health System Epidemiology Director Welcome! Teaming Up to Prevent Infections! CLOSTRIDIUM
CLOSTRIDIUM DIFFICILE PREVENTION Beth Goodall, RN, BSN Board Certified in Infection Prevention and Control DCH Health System Epidemiology Director Welcome! Teaming Up to Prevent Infections! CLOSTRIDIUM
New guidelines for the management of norovirus outbreaks in acute and community health and social care settings
 Volume 5 Number 47 Published on: 25 November 2011 Current News New guidelines for the management of norovirus outbreaks in acute and community health and social care settings Surveillance of haemolytic
Volume 5 Number 47 Published on: 25 November 2011 Current News New guidelines for the management of norovirus outbreaks in acute and community health and social care settings Surveillance of haemolytic
Clinical. Clostridium Difficile: Standard Operating Procedure. Document Control Summary. Contents
 Clinical Clostridium Difficile: Standard Operating Procedure Document Control Summary Status: Version: Author/Title: Owner/Title: Approved by: Ratified: Related Trust Strategy and/or Strategic Aims Implementation
Clinical Clostridium Difficile: Standard Operating Procedure Document Control Summary Status: Version: Author/Title: Owner/Title: Approved by: Ratified: Related Trust Strategy and/or Strategic Aims Implementation
March 3, To: Hospitals, Long Term Care Facilities, and Local Health Departments
 March 3, 2010 To: Hospitals, Long Term Care Facilities, and Local Health Departments From: NYSDOH Bureau of Healthcare Associated Infections HEALTH ADVISORY: GUIDANCE FOR PREVENTION AND CONTROL OF HEALTHCARE
March 3, 2010 To: Hospitals, Long Term Care Facilities, and Local Health Departments From: NYSDOH Bureau of Healthcare Associated Infections HEALTH ADVISORY: GUIDANCE FOR PREVENTION AND CONTROL OF HEALTHCARE
ECHO-Antibiotic Stewardship Program
 ECHO-Antibiotic Stewardship Program More Interesting Recent Literature Updates April 20, 2017 Charles Krasner, M.D. University of NV, Reno School of Medicine Sierra NV Veterans Affairs Medical Center Antibiotic
ECHO-Antibiotic Stewardship Program More Interesting Recent Literature Updates April 20, 2017 Charles Krasner, M.D. University of NV, Reno School of Medicine Sierra NV Veterans Affairs Medical Center Antibiotic
Impact of clinical symptoms on interpretation of diagnostic assays for Clostridium difficile infections
 Washington University School of Medicine Digital Commons@Becker Open Access Publications 2011 Impact of clinical symptoms on interpretation of diagnostic assays for Clostridium difficile infections Erik
Washington University School of Medicine Digital Commons@Becker Open Access Publications 2011 Impact of clinical symptoms on interpretation of diagnostic assays for Clostridium difficile infections Erik
EVALUATION OF THE COBAS LIAT (ROCHE ) AND THE LIAISON MDX (DIASORIN )
 EVALUATION OF THE COBAS LIAT (ROCHE ) AND THE LIAISON MDX (DIASORIN ) Delefortrie Quentin Intern in Clinical Pathology Corata, 20-21 September 2018 Presentation Plan Evaluation of the cobas Liat Influenza
EVALUATION OF THE COBAS LIAT (ROCHE ) AND THE LIAISON MDX (DIASORIN ) Delefortrie Quentin Intern in Clinical Pathology Corata, 20-21 September 2018 Presentation Plan Evaluation of the cobas Liat Influenza
HEALTHCARE ASSOCIATED INFECTION PREVENTION AND CONTROL REPORT
 Borders NHS Board Meeting Date: 7 ch Approved by: Author(s): Nicky Berry, Director of Nursing, Midwifery and Acute Services Natalie Mallin, Infection Control Administrator Sam Whiting, Infection Control
Borders NHS Board Meeting Date: 7 ch Approved by: Author(s): Nicky Berry, Director of Nursing, Midwifery and Acute Services Natalie Mallin, Infection Control Administrator Sam Whiting, Infection Control
Can a toxin gene NAAT be used to predict toxin EIA and the severity of Clostridium difficile infection?
 Garvey et al. Antimicrobial Resistance and Infection Control (2017) 6:127 DOI 10.1186/s13756-017-0283-z RESEARCH Can a toxin gene NAAT be used to predict toxin EIA and the severity of Clostridium difficile
Garvey et al. Antimicrobial Resistance and Infection Control (2017) 6:127 DOI 10.1186/s13756-017-0283-z RESEARCH Can a toxin gene NAAT be used to predict toxin EIA and the severity of Clostridium difficile
9/18/2018. Clostridium Difficile: Updates on Diagnosis and Treatment. Clostridium difficile Infection (CDI) Clostridium difficile Infection (CDI)
 Clostridium Difficile: Updates on Diagnosis and Treatment Elizabeth Hudson, DO, MPH 9/25/18 Antibiotic-associated diarrhea and colitis were well established soon after widespread use of antibiotics In
Clostridium Difficile: Updates on Diagnosis and Treatment Elizabeth Hudson, DO, MPH 9/25/18 Antibiotic-associated diarrhea and colitis were well established soon after widespread use of antibiotics In
MEETING THE STANDARDS: FDA MANDATORY RAPID INFLUENZA DETECTION TEST (RIDTs) RECLASSIFICATION
 MEETING THE STANDARDS: FDA MANDATORY RAPID INFLUENZA DETECTION TEST (RIDTs) RECLASSIFICATION NOVEMBER 6, 2017 SALLY A. HOJVAT M.Sc., Ph.D. Retired as Director of FDA Division of Microbiology Devices, CDRH
MEETING THE STANDARDS: FDA MANDATORY RAPID INFLUENZA DETECTION TEST (RIDTs) RECLASSIFICATION NOVEMBER 6, 2017 SALLY A. HOJVAT M.Sc., Ph.D. Retired as Director of FDA Division of Microbiology Devices, CDRH
Bacterial Enteric Infections Detected by Culture-Independent Diagnostic Tests FoodNet, United States,
 Bacterial Enteric Infections Detected by Culture-Independent Diagnostic Tests FoodNet, United States, 2012 2014 Martha Iwamoto, MD 1, Jennifer Y. Huang, MPH 1, Alicia B. Cronquist, MPH 2, Carlota Medus,
Bacterial Enteric Infections Detected by Culture-Independent Diagnostic Tests FoodNet, United States, 2012 2014 Martha Iwamoto, MD 1, Jennifer Y. Huang, MPH 1, Alicia B. Cronquist, MPH 2, Carlota Medus,
3/23/2012. Impact of Laboratory Testing on Detection and Treatment of Healthcare Associated Infection: The Case of CDI
 Impact of Laboratory Testing on Detection and Treatment of Healthcare Associated Infection: The Case of CDI L. Clifford McDonald, MD Lance R. Peterson, MD March 27, 2012 1 Potential COI L. Clifford McDonald,
Impact of Laboratory Testing on Detection and Treatment of Healthcare Associated Infection: The Case of CDI L. Clifford McDonald, MD Lance R. Peterson, MD March 27, 2012 1 Potential COI L. Clifford McDonald,
Annex C: - CDI What s the diff? 4 th Annual Outbreak Management Workshop September 19, 2013 Naideen Bailey & Grace Volkening
 Annex C: - CDI What s the diff? 4 th Annual Outbreak Management Workshop September 19, 2013 Naideen Bailey & Grace Volkening There s an updated Annex C Annex C is an extension to the PIDAC Infection Prevention
Annex C: - CDI What s the diff? 4 th Annual Outbreak Management Workshop September 19, 2013 Naideen Bailey & Grace Volkening There s an updated Annex C Annex C is an extension to the PIDAC Infection Prevention
The disease formerly called Clostridium difficile associated
 Improving Patient Care Annals of Internal Medicine Does My Patient Have Clostridium difficile Infection? Lance R. Peterson, MD, and Ari Robicsek, MD Clostridium difficile infection (CDI) seems to be changing
Improving Patient Care Annals of Internal Medicine Does My Patient Have Clostridium difficile Infection? Lance R. Peterson, MD, and Ari Robicsek, MD Clostridium difficile infection (CDI) seems to be changing
Guidelines on the Prevention, Detection, And Management of Clostridium difficile Infection
 Guidelines on the Prevention, Detection, And Management of Clostridium difficile Infection 1 CLINICAL GUIDELINES CHECKLIST Name of guidelines Guidelines on the Prevention, Detection, and Management of
Guidelines on the Prevention, Detection, And Management of Clostridium difficile Infection 1 CLINICAL GUIDELINES CHECKLIST Name of guidelines Guidelines on the Prevention, Detection, and Management of
Historical Perspective
 The Diagnosis of C. difficile Infections: Does Molecular Make it More or Less difficile? Stephen M. Brecher Ph.D. VA Boston Health Care System BU School of Medicine The opinions expressed in this presentation
The Diagnosis of C. difficile Infections: Does Molecular Make it More or Less difficile? Stephen M. Brecher Ph.D. VA Boston Health Care System BU School of Medicine The opinions expressed in this presentation
Rapid and Sensitive Loop-Mediated Isothermal Amplification (LAMP) Test for. Gold Standard
 JCM Accepts, published online ahead of print on 24 November 2010 J. Clin. Microbiol. doi:10.1128/jcm.01824-10 Copyright 2010, American Society for Microbiology and/or the Listed Authors/Institutions. All
JCM Accepts, published online ahead of print on 24 November 2010 J. Clin. Microbiol. doi:10.1128/jcm.01824-10 Copyright 2010, American Society for Microbiology and/or the Listed Authors/Institutions. All
Monitoring Recent HIV Infection in Ireland, 2017
 Monitoring Recent HIV Infection in Ireland, 2017 January 2019 Key Facts 1 13% of HIV diagnoses in 2017 (of those tested) were likely to be recent infections (within 4 months), using the Recent Infection
Monitoring Recent HIV Infection in Ireland, 2017 January 2019 Key Facts 1 13% of HIV diagnoses in 2017 (of those tested) were likely to be recent infections (within 4 months), using the Recent Infection
Comprehensive evaluation of chemiluminescent immunoassays for the laboratory diagnosis of Clostridium difficile infection
 Eur J Clin Microbiol Infect Dis (2017) 36:1253 1259 DOI 10.1007/s10096-017-2916-9 ORIGINAL ARTICLE Comprehensive evaluation of chemiluminescent immunoassays for the laboratory diagnosis of Clostridium
Eur J Clin Microbiol Infect Dis (2017) 36:1253 1259 DOI 10.1007/s10096-017-2916-9 ORIGINAL ARTICLE Comprehensive evaluation of chemiluminescent immunoassays for the laboratory diagnosis of Clostridium
The Bristol Stool Scale and Its Relationship to Clostridium difficile Infection
 JCM Accepts, published online ahead of print on 16 July 2014 J. Clin. Microbiol. doi:10.1128/jcm.01303-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 The Bristol Stool Scale
JCM Accepts, published online ahead of print on 16 July 2014 J. Clin. Microbiol. doi:10.1128/jcm.01303-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 The Bristol Stool Scale
Guidance on Prevention and Control of Clostridium difficile Infection (CDI) in health and social care settings in Scotland.
 Guidance on Prevention and Control of Clostridium difficile Infection (CDI) in health and social care settings in Scotland. Scottish Health Protection Network Scottish Guidance No 6 2017 edition. September
Guidance on Prevention and Control of Clostridium difficile Infection (CDI) in health and social care settings in Scotland. Scottish Health Protection Network Scottish Guidance No 6 2017 edition. September
FMD diagnostics: current developments and application in the context of FMD control in endemic countries Wilna Vosloo
 FMD diagnostics: current developments and application in the context of FMD control in endemic countries Wilna Vosloo CSIRO Livestock Industries, Australian Animal Health Laboratory (AAHL), Geelong, Australia
FMD diagnostics: current developments and application in the context of FMD control in endemic countries Wilna Vosloo CSIRO Livestock Industries, Australian Animal Health Laboratory (AAHL), Geelong, Australia
New Molecular Diagnostic Assays for Solana Performance Assessment, Workflow Analysis, and Clinical Utility
 New Molecular Diagnostic Assays for Solana Performance Assessment, Workflow Analysis, and Clinical Utility Gerald A. Capraro, Ph.D., D(ABMM) Director, Clinical Microbiology Carolinas Pathology Group Atrium
New Molecular Diagnostic Assays for Solana Performance Assessment, Workflow Analysis, and Clinical Utility Gerald A. Capraro, Ph.D., D(ABMM) Director, Clinical Microbiology Carolinas Pathology Group Atrium
Bacterial Enteropathogens Faecal PCR and Culture Results
 Bacterial Enteropathogens - 2019 Faecal PCR and Culture Results Because most diarrhoeal illness is self-limiting, microbiological investigation is usually not indicated. It should however be considered
Bacterial Enteropathogens - 2019 Faecal PCR and Culture Results Because most diarrhoeal illness is self-limiting, microbiological investigation is usually not indicated. It should however be considered
Clostridium difficile
 Clostridium difficile Care Homes IPC Study Day Sue Barber Infection Prevention & Control Lead AV & Chiltern CCG s Clostridium difficile A spore forming Bacterium. Difficult to grow in the laboratory hence
Clostridium difficile Care Homes IPC Study Day Sue Barber Infection Prevention & Control Lead AV & Chiltern CCG s Clostridium difficile A spore forming Bacterium. Difficult to grow in the laboratory hence
Test Bulletin. Save time, money, resources, and precious blood by NOT collecting extra specimens just in case
 Test Bulletin October 2018 Save time, money, resources, and precious blood by NOT collecting extra specimens just in case ACL Laboratories has seen an increase in the number of specimens submitted without
Test Bulletin October 2018 Save time, money, resources, and precious blood by NOT collecting extra specimens just in case ACL Laboratories has seen an increase in the number of specimens submitted without
A Physician Perspective. Marguerite A. Neill, MD Alpert Medical School Brown University Culture Independent Diagnostics Forum April 24, 2012
 A Physician Perspective Marguerite A. Neill, MD Alpert Medical School Brown University Culture Independent Diagnostics Forum April 24, 2012 For diagnostics, It s all about context. The Clinical Drivers
A Physician Perspective Marguerite A. Neill, MD Alpert Medical School Brown University Culture Independent Diagnostics Forum April 24, 2012 For diagnostics, It s all about context. The Clinical Drivers
Care homes - Homely remedies
 Bulletin 72 August 2014 Care homes - Homely remedies Care home staff have a recognised duty of care to be able to respond to minor symptoms experienced by residents. A homely remedy is a medicinal product
Bulletin 72 August 2014 Care homes - Homely remedies Care home staff have a recognised duty of care to be able to respond to minor symptoms experienced by residents. A homely remedy is a medicinal product
Proposed Data Collection Submitted for Public Comment and. AGENCY: Centers for Disease Control and Prevention (CDC),
 This document is scheduled to be published in the Federal Register on 11/16/2017 and available online at https://federalregister.gov/d/2017-24804, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
This document is scheduled to be published in the Federal Register on 11/16/2017 and available online at https://federalregister.gov/d/2017-24804, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
Round Robin Highlights of Infection Control Practices
 Round Robin Highlights of Infection Control Practices Arkansas Department of Health Healthcare-Associated Infections Program Staff To Isolate or Not To Isolate: That Is The Question Kelley Garner, MPH,
Round Robin Highlights of Infection Control Practices Arkansas Department of Health Healthcare-Associated Infections Program Staff To Isolate or Not To Isolate: That Is The Question Kelley Garner, MPH,
HEALTHCARE ASSOCIATED INFECTION PREVENTION AND CONTROL REPORT SEPTEMBER 2014
 Borders NHS Board HEALTHCARE ASSOCIATED INFECTION PREVENTION AND CONTROL REPORT SEPTEMBER Aim The purpose of this paper is to update Board members of the current status of Healthcare Associated Infections
Borders NHS Board HEALTHCARE ASSOCIATED INFECTION PREVENTION AND CONTROL REPORT SEPTEMBER Aim The purpose of this paper is to update Board members of the current status of Healthcare Associated Infections
NHS GRAMPIAN. Healthcare Associated Infection (HAI) Bimonthly Report January 2017
 NHS GRAMPIAN Healthcare Associated Infection (HAI) Bimonthly Report uary 2017 1. Actions Recommended The Board is requested to note the content of this summary bimonthly HAI Report, as directed by the
NHS GRAMPIAN Healthcare Associated Infection (HAI) Bimonthly Report uary 2017 1. Actions Recommended The Board is requested to note the content of this summary bimonthly HAI Report, as directed by the
Clostridium difficile Asymptomatic Carriers The Hidden Part of the Iceberg?
 Clostridium difficile Asymptomatic Carriers The Hidden Part of the Iceberg? Disclosures Merck Canada, BD Diagnostics, AMD Medical, Canadian Institute for Health Research Merck Canada, Pfizer OBJECTIVES
Clostridium difficile Asymptomatic Carriers The Hidden Part of the Iceberg? Disclosures Merck Canada, BD Diagnostics, AMD Medical, Canadian Institute for Health Research Merck Canada, Pfizer OBJECTIVES
Clostridium difficile: An Overview
 Clostridium difficile: An Overview CDI Webinar July 11, 2017 PUBLIC HEALTH DIVISION Acute and Communicable Disease Prevention Section Outline Background Microbiology Burden Pathogenesis Diagnostic testing
Clostridium difficile: An Overview CDI Webinar July 11, 2017 PUBLIC HEALTH DIVISION Acute and Communicable Disease Prevention Section Outline Background Microbiology Burden Pathogenesis Diagnostic testing
Impact of hospital infections on our
 Impact of hospital infections on our ageing population Professor Peter Lambert School of Life and Health Sciences P. A. Lambert Slides are for personal use only. They are NOT for reproduction or publication
Impact of hospital infections on our ageing population Professor Peter Lambert School of Life and Health Sciences P. A. Lambert Slides are for personal use only. They are NOT for reproduction or publication
Case 1. Which of the following would be next appropriate investigation/s regarding the pts diarrhoea?
 Case 1 21 yr old HIV +ve, Cd4-100 HAART naïve Profuse diarrhoea for 3/52. Stool MC&S ve Which of the following would be next appropriate investigation/s regarding the pts diarrhoea? Repeat stool MC&S Stool
Case 1 21 yr old HIV +ve, Cd4-100 HAART naïve Profuse diarrhoea for 3/52. Stool MC&S ve Which of the following would be next appropriate investigation/s regarding the pts diarrhoea? Repeat stool MC&S Stool
Stool bench. Cultures: SARAH
 Stool bench The bacteria found in stool are representative of the bacteria that are present in the digestive system (gastrointestinal tract). Certain bacteria and fungi called normal flora inhabit everyone's
Stool bench The bacteria found in stool are representative of the bacteria that are present in the digestive system (gastrointestinal tract). Certain bacteria and fungi called normal flora inhabit everyone's
Scottish E. coli O157/Shiga toxin-producing E. coli Reference Laboratory
 Scottish E. coli O157/Shiga toxin-producing E. coli Reference Laboratory 1 Authors: SERL68:Version L. Allison 6 Issue date & M. 27/09/2017 Hanson This is an electronic document Authors: L. Allison which
Scottish E. coli O157/Shiga toxin-producing E. coli Reference Laboratory 1 Authors: SERL68:Version L. Allison 6 Issue date & M. 27/09/2017 Hanson This is an electronic document Authors: L. Allison which
HRET HIIN Diagnostic Error Virtual Event
 HRET HIIN Diagnostic Error Virtual Event Promoting Diagnostic Stewardship: Effectively Reducing Diagnostic Errors September 19, 2017 1 Jordan Steiger, MPH Program Manager, HRET WELCOME AND INTRODUCTIONS
HRET HIIN Diagnostic Error Virtual Event Promoting Diagnostic Stewardship: Effectively Reducing Diagnostic Errors September 19, 2017 1 Jordan Steiger, MPH Program Manager, HRET WELCOME AND INTRODUCTIONS
PROCEDURE FOR DEALING WITH MENINGITIS AND MENINGOCOCCAL DISEASE
 PROCEDURE FOR DEALING WITH MENINGITIS AND MENINGOCOCCAL DISEASE 1 INTRODUCTION 1.1 Purpose of Procedure To ensure that the University has clear guidelines on preventing or managing cases of meningitis
PROCEDURE FOR DEALING WITH MENINGITIS AND MENINGOCOCCAL DISEASE 1 INTRODUCTION 1.1 Purpose of Procedure To ensure that the University has clear guidelines on preventing or managing cases of meningitis
T he diagnosis of malaria in clinical laboratories in the UK
 862 ORIGINAL ARTICLE Use of rapid diagnostic tests for diagnosis of malaria in the UK D Chilton, A N J Malik, M Armstrong, M Kettelhut, J Parker-Williams, P L Chiodini... See end of article for authors
862 ORIGINAL ARTICLE Use of rapid diagnostic tests for diagnosis of malaria in the UK D Chilton, A N J Malik, M Armstrong, M Kettelhut, J Parker-Williams, P L Chiodini... See end of article for authors
This is a repository copy of Clostridium difficile infection: advances in epidemiology, diagnosis and transmission.
 This is a repository copy of Clostridium difficile infection: advances in epidemiology, diagnosis and transmission. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/93246/
This is a repository copy of Clostridium difficile infection: advances in epidemiology, diagnosis and transmission. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/93246/
Laboratory Investigation of. Infectious Diarrhoea. Quiz Feedback. bpac nz better medicine
 Laboratory Investigation of Infectious Diarrhoea Quiz Feedback bpac nz better medicine GP Review Panel: Dr Janine Bailey, Motueka Dr Stephen Kuzmich, Wellington Dr Randall Sturm, Auckland Dr Neil Whittaker,
Laboratory Investigation of Infectious Diarrhoea Quiz Feedback bpac nz better medicine GP Review Panel: Dr Janine Bailey, Motueka Dr Stephen Kuzmich, Wellington Dr Randall Sturm, Auckland Dr Neil Whittaker,
Infection control in Aged Residential Care Facilities. Dr Sally Roberts Clinical Advisor for IP&C Service, ADHB
 Infection control in Aged Residential Care Facilities Dr Sally Roberts Clinical Advisor for IP&C Service, ADHB Background Endemic infections Epidemic infections Managing outbreaks Administrative measures
Infection control in Aged Residential Care Facilities Dr Sally Roberts Clinical Advisor for IP&C Service, ADHB Background Endemic infections Epidemic infections Managing outbreaks Administrative measures
Improving Prevention and Control of Infection Quarter 2 Report: April 2009 September 2009
 Improving Prevention and Control of Infection Quarter 2 Report: April 2009 September 2009 1. Introduction This Quarter 2 updates the Health Board on infection prevention and control issues within the BCUHB.
Improving Prevention and Control of Infection Quarter 2 Report: April 2009 September 2009 1. Introduction This Quarter 2 updates the Health Board on infection prevention and control issues within the BCUHB.
Chapter 1 The Public Health Role of Clinical Laboratories
 Chapter 1 The Public Health Role of Clinical Laboratories A. Epidemic Diarrhea The two most common types of epidemic diarrhea in developing countries are watery diarrhea caused by Vibrio cholerae serogroup
Chapter 1 The Public Health Role of Clinical Laboratories A. Epidemic Diarrhea The two most common types of epidemic diarrhea in developing countries are watery diarrhea caused by Vibrio cholerae serogroup
SECTION 13.0 SPECIMENS FOR LABORATORY EXAMINATION
 SECTION 13.0 SPECIMENS FOR LABORATORY EXAMINATION Introduction Collection of Specimens for Microbiological Examination: Safe Handling of Specimens Packaging & Transportation of Specimens Specimen Rejection
SECTION 13.0 SPECIMENS FOR LABORATORY EXAMINATION Introduction Collection of Specimens for Microbiological Examination: Safe Handling of Specimens Packaging & Transportation of Specimens Specimen Rejection
Antimicrobial Stewardship in Community Acquired Pneumonia
 Antimicrobial Stewardship in Community Acquired Pneumonia Medicine Review Course 2018 Dr Lee Tau Hong Consultant Department of Infectious Diseases National Centre for Infectious Diseases Scope 1. Diagnosis
Antimicrobial Stewardship in Community Acquired Pneumonia Medicine Review Course 2018 Dr Lee Tau Hong Consultant Department of Infectious Diseases National Centre for Infectious Diseases Scope 1. Diagnosis
Modern approach to Clostridium Difficile Infection
 Modern approach to Clostridium Difficile Infection Pseudomembranous Colitis: Principles for diagnosis and treatment Aggelos Stefos Internist, Infectious diseases Specialist Department of Medicine and Research
Modern approach to Clostridium Difficile Infection Pseudomembranous Colitis: Principles for diagnosis and treatment Aggelos Stefos Internist, Infectious diseases Specialist Department of Medicine and Research
Transmission Based Precautions Literature Reviews. Disinfectants
 Transmission Based Precautions Literature Reviews Disinfectants April 2008 PRINCIPAL RESEARCH QUESTION/OBJECTIVE: Search Strategy Disinfectants What is the evidence to support the use of disinfectants
Transmission Based Precautions Literature Reviews Disinfectants April 2008 PRINCIPAL RESEARCH QUESTION/OBJECTIVE: Search Strategy Disinfectants What is the evidence to support the use of disinfectants
The national surveillance of Clostridium difficile in Denmark
 The national surveillance of Clostridium difficile in Denmark Katharina E. P. Olsen, PhD (PharmD) National Reference Laboratory for Enteropathogenic Bacteria Departement of Microbiology and Infection control
The national surveillance of Clostridium difficile in Denmark Katharina E. P. Olsen, PhD (PharmD) National Reference Laboratory for Enteropathogenic Bacteria Departement of Microbiology and Infection control
Clostridium difficile infection in an endemic setting in the Netherlands
 Eur J Clin Microbiol Infect Dis (2011) 30:587 593 DOI 10.1007/s10096-010-1127-4 ARTICLE Clostridium difficile infection in an endemic setting in the Netherlands M. P. M. Hensgens & A. Goorhuis & C. M.
Eur J Clin Microbiol Infect Dis (2011) 30:587 593 DOI 10.1007/s10096-010-1127-4 ARTICLE Clostridium difficile infection in an endemic setting in the Netherlands M. P. M. Hensgens & A. Goorhuis & C. M.
Laboratory diagnosis of Clostridium difficile infection
 Review Article Page 1 of 5 Laboratory diagnosis of Clostridium difficile infection Katherine Kendrick Mercer University School of Medicine (SOM), Navicent Health, Macon, GA, USA Correspondence to: Katherine
Review Article Page 1 of 5 Laboratory diagnosis of Clostridium difficile infection Katherine Kendrick Mercer University School of Medicine (SOM), Navicent Health, Macon, GA, USA Correspondence to: Katherine
Does Empirical Clostridium difficile Infection (CDI) Therapy Result in False-Negative CDI Diagnostic Test Results?
 MAJOR ARTICLE Does Empirical Clostridium difficile Infection (CDI) Therapy Result in False-Negative CDI Diagnostic Test Results? Venkata C. K. Sunkesula, 1 Sirisha Kundrapu, 1 Christine Muganda, 3 Ajay
MAJOR ARTICLE Does Empirical Clostridium difficile Infection (CDI) Therapy Result in False-Negative CDI Diagnostic Test Results? Venkata C. K. Sunkesula, 1 Sirisha Kundrapu, 1 Christine Muganda, 3 Ajay
ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE
 ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE The diagnosis of CDI should be based on a combination of clinical and laboratory findings. A case definition for the usual
ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE The diagnosis of CDI should be based on a combination of clinical and laboratory findings. A case definition for the usual
In the Name of God. Talat Mokhtari-Azad Director of National Influenza Center
 In the Name of God Overview of influenza laboratory diagnostic technology: advantages and disadvantages of each test available Talat Mokhtari-Azad Director of National Influenza Center Tehran- Iran 1 1)
In the Name of God Overview of influenza laboratory diagnostic technology: advantages and disadvantages of each test available Talat Mokhtari-Azad Director of National Influenza Center Tehran- Iran 1 1)
Pain relief to take home after your surgery
 Pain relief to take home after your surgery Information for patients from the Department of Anaesthesia This factsheet explains: About pain and pain relief at home after surgery. Which pain relief drugs
Pain relief to take home after your surgery Information for patients from the Department of Anaesthesia This factsheet explains: About pain and pain relief at home after surgery. Which pain relief drugs
Ayrshire and Arran NHS Board
 Paper 4 Ayrshire and Arran NHS Board Monday 11 ember Healthcare Associated Infection Reporting Template Report Author: Bob Wilson, Infection Control Manager Sponsoring Director: Professor Hazel Borland,
Paper 4 Ayrshire and Arran NHS Board Monday 11 ember Healthcare Associated Infection Reporting Template Report Author: Bob Wilson, Infection Control Manager Sponsoring Director: Professor Hazel Borland,
TITLE OF PRESENTATION
 TITLE OF PRESENTATION Can C. Subtitle difficile of Infection Presentation Rates Be Used to Judge Prevention Success for Hospitals? David P. Calfee, MD, MS Associate Professor of Medicine and Public Health
TITLE OF PRESENTATION Can C. Subtitle difficile of Infection Presentation Rates Be Used to Judge Prevention Success for Hospitals? David P. Calfee, MD, MS Associate Professor of Medicine and Public Health
Clostridium difficile (C. difficile) and Staphylococcus aureus bacteraemia (MRSA and MSSA) Bi-annual Report. Surveillance: Report:
 Surveillance: Report: Clostridium difficile (C. difficile) and Staphylococcus aureus ( and ) Bi-annual Report Time period: 1 st April to 30 th September 2016 Health Board: Wales Content: Issued by: Pg
Surveillance: Report: Clostridium difficile (C. difficile) and Staphylococcus aureus ( and ) Bi-annual Report Time period: 1 st April to 30 th September 2016 Health Board: Wales Content: Issued by: Pg
Safe Patient Care Keeping our Residents Safe
 Safe Patient Care Keeping our Residents Safe 2016 Diarrhoea & Vomiting Infection Prevention & Control in Residential Care Setting Patricia Coughlan, Infection Prevention Control Nurse, HSE Disability Services
Safe Patient Care Keeping our Residents Safe 2016 Diarrhoea & Vomiting Infection Prevention & Control in Residential Care Setting Patricia Coughlan, Infection Prevention Control Nurse, HSE Disability Services
