CLINICAL REPORT Methotrexate Treatment for Pityriasis Rubra Pilaris: A Case Series and Literature Review
|
|
|
- Eustacia Gilbert
- 5 years ago
- Views:
Transcription
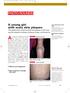
1 CLINICAL REPORT 501 Methotrexate Treatment for Pityriasis Rubra Pilaris: A Case Series and Literature Review Lukas KOCH 1, Clemens SCHÖFFL 1, Werner ABERER 1 and Cesare MASSONE 2 1 Department of Dermatology, Medical University of Graz, Austria, and 2 Dermatology Unit, Galliera Hospital, Genova, Italy Treatment recommendations for pityriasis rubra pilaris (PRP) are based solely on case reports and small case series, as to-date no randomized controlled trials are available. We present here a case series of 3 patients and a literature review of 28 studies treating a total of 116 patients, with the aim of providing data regarding efficacy and safety of methotrexate in the treatment of PRP. Methotrexate was effective in our patients; the review showed an overall response rate of 65.5% with complete clearing in 23.3% and excellent improvement in 17.2%, respectively. After excluding studies with other concurrent systemic therapies or low reliability, the overall response rate increased to 90.9%, with complete clearing in 40.9% and excellent improvement in 31.8%, respectively. Sixteen adverse reactions, of which 11 were mild, were observed in 15 patients (12.9%). In conclusion, the available literature supports good response rates and safety of methotrexate in PRP. Key words: pityriasis rubra pilaris; methotrexate; treatment; response; review. Accepted Jan 15, 2018; Epub ahead of print Jan 16, 2018 Acta Derm Venereol 2018; 98: Corr: Cesare Massone, Dermatology Unit Ospedali Galliera, Via Volta 6, IT-16128, Genova, Italy. cesare.massone@galliera.it Treatment recommendations for pityriasis rubra pilaris (PRP) are based solely on case reports or small case series, as to-date no randomized controlled trials assessing different treatment efficacies are available. We report here our experience with methotrexate (MTX) in the treatment of PRP and provide a systematic review of the literature regarding the efficacy and tolerability of treatment with MTX. CASE REPORTS Case 1 A 55-year-old man consulted our outpatient clinic in August 2014 with a 2-month history of skin changes, which had started on the scalp and spread consecutively to his shoulders and trunk. Prior treatment with systemic corticosteroids had not improved the disease. Apart from an umbilical hernia the patient was otherwise healthy and did not take any medication regularly. Skin examination revealed scaling erythematous to orange confluent plaques with islands of unaffected skin on his trunk, buttocks, neck, face and scalp (Fig. 1A, B). Furthermore, the patient had palmoplantar keratoderma with fissures; the affected body surface area (BSA) was approximately 50%. Histopathological examination of a skin biopsy was compatible with a clinical diagnosis of PRP and after unremarkable pre-treatment screening (Table SI 1 ), MTX treatment was initiated, 20 mg subcutaneously once weekly, followed by 5 mg oral folic acid substitution every following day. At 1-month follow-up there was considerable improvement in the erythema on the trunk, but scaling and erythema on the arms and palmar keratoderma persisted. The dosage of MTX was increased to 25 mg once weekly. At 4-month follow-up the patient showed further improvement on the trunk, but only little improvement of palmoplantar keratoderma, which later completely disappeared after 9 months of MTX treatment, when only a few erythematous non-scaling patches persisted on the trunk. MTX was then consecutively reduced each month, down to 7.5 mg/week in February 2016, when the skin had almost completely cleared. MTX was ceased in June 2016: the total MTX dosage applied was mg, with no adverse drug reactions during the whole treatment period. A 26-month follow-up in October 2016 revealed an almost complete remission of PRP with minimal nonscaling tender plaques on the ventral trunk affecting less than 1% of the BSA (Fig. 1C, D). Case 2 A 50-year-old man with no comorbidities or long-term medication had developed facial erythema with scaling in June 2015, which then had spread to the trunk and arms after 2 weeks. He was hospitalized in our inpatient clinic in July, with a diffuse-scaling erythema covering approximately 50% of the BSA with multiple islands of sparing. The palms showed orange-red discoloration with mild keratoderma. A skin biopsy was compatible with the diagnosis of PRP and after unremarkable pretreatment screening MTX was initiated, 10 mg/week subcutaneously, followed by 5 mg oral folic acid every following day. The MTX dose was increased by 2.5 mg/ 1 This is an open access article under the CC BY-NC license. Journal Compilation 2018 Acta Dermato-Venereologica. doi: / Acta Derm Venereol 2018; 98:
2 502 L. Koch et al. Fig. 1. Pityriasis rubra pilaris. Case 1: skin condition (A, B) before and (C, D) after treatment with methotrexate (MTX). week up to 20 mg weekly with regular unremarkable laboratory controls. At 6-week follow-up the skin worsened showing erythroderma and MTX was increased to 25 mg/week. Three weeks later erythema started to improve and an extensive scaling appeared. The erythema slowly, but progressively, reduced with a marked improvement 4 months after therapy initiation. MTX was continued at 25 mg/week until April With a total dose of mg and less than 5% of BSA affected, weekly MTX doses were then reduced by 2.5 mg each month to a maintaining dose of 12.5 mg/week and a sustained response with < 5% BSA involved. At the last follow-up, in October 2016, the patient had a total MTX dose of mg with stable disease. Apart from an intermittent elevation of liver function tests in April 2016 (GOT 70 U/l (normal 35), GPT 149 U/l (normal 45) and GGT 56 U/l normal 55)), which lowered significantly after dose reduction the patient did not have any laboratory or clinical side-effects during treatment. Case 3 An 83-year-old man had developed erythroderma in January With the initial diagnosis of psoriasis he received 311 nm ultraviolet B (UVB) phototherapy and adalimumab for one month in an external outpatient clinic with worsening of the disease. Besides untreated hypercholesterolemia and atrial fibrillation he did not have any further comorbidity. The patient attended our department in February 2014 showing erythroderma with islands of sparing, palmoplantar hyperkeratosis with fissures and onychodystrophy. A punch biopsy revealed psoriasiform dermatitis compatible with PRP (previous treatment has probably influenced the morphological features). Acitretin, 30 mg/ day, was started and increased up to 50 mg/day within 2 months with only minimal skin improvement. In July 2014 erythroderma worsened and acitretin was switched to MTX 10 mg/week, subcutaneously, followed by 5 mg oral folic acid the every following day. At 2-month follow-up the skin already improved and MTX was increased to 12.5 mg/week. Remarkably, at 3-month follow-up only individual plaques remained on the trunk and both arms. Therapy was maintained, but the dose was not increased further due to good therapy response on low doses and a heterozygous C677T mutation of the MTHFR gene, as well as an intermittent slight elevation of GGT (81 U/l, normal 55 U/l) with GOT and GPT always within normal ranges. Transient elastography of the liver did not show any signs of liver parenchyma impairment. At 6-month follow-up, with a total dose of mg MTX, remission was sustained with less than 1% BSA involved. Unfortunately, follow-up was lost thereafter due to a severe trauma with intracranial bleeding and persistent brain damage. MTX and skin condition were not mentioned in the subsequent medical reports (MTX was probably discontinued at the time of trauma) and the patient died several months later at the age of 85 years due to nosocomial pneumonia. LITERATURE REVIEW A systematic search of MEDLINE was conducted for case reports and case series published from 1964 until August 2017 using the key words pityriasis rubra pilaris, methotrexate and treatment. Studies treating patients of any age and any type of PRP were included. Studies without a clear statement regarding MTX treat ment response were excluded. Reports combining MTX with other systemic drugs, such as retinoids, were included. Together with our case series a total of 28 publications treating 116 patients were included. Methotrexate treatment efficacy was divided into 6 effect groups: total clearance (for complete clearance),
3 Methotrexate treatment for pityriasis rubra pilaris 503 excellent improvement ( almost or 95% clear as well as dramatic, excellent, favourable, significant, marked or considerable improvement, respectively), partial improvement ( partial improvement, improved as well as partial, good or satisfactory effects, respectively), slight improvement ( slight, sluggish or mixed response, respectively), any improvement ( improvement without information regarding the extent of improvement) and no improvement. Reliability of case reports and studies was categorized into high if data on both, MTX dosage and treatment duration were available or low if data on MTX dosage or therapy duration were missing. Table SII 1 presents the clinical data, MTX dosage, treat ment efficacy, adverse drug reactions and reliability of the reviewed studies, separated into positive and negative reports, as well as studies with other concurrent systemic therapies. MTX treatment efficacy is summarized in Table I, separated into overall response and studies with MTX single treatment and high reliability. DISCUSSION MTX is a potent folate analogue that interferes with nucleic acid synthesis and cellular proliferation due to inhibition of dihydrofolate reductase. While the exact mechanism of action in inflammatory skin conditions, such as psoriasis or PRP, remain unclear, it presumably depends mainly on 2 different effects. On the one hand, MTX acts as an antiproliferative agent, as it depresses the mitotic rate of keratinocytes via induction of apoptosis of proliferating keratinocytes through oxidative stress (1). This could explain why PRP responds to MTX in a much slower manner than psoriasis does (2), because although it has been shown that the epidermal turnover time in PRP is higher than in normal skin, it is considerably slower than in psoriasis (3). On the other hand, MTX has anti-inflammatory effects, especially via reduction of proinflammatory cytokines such as interleukin 1 (IL1) and tumour necrosis factor alpha (TNFα) (4, 5). Furthermore, as shown for rheumatoid arthritis, an increased gene expression of the anti-inflammatory Th2 cytokines IL4 and IL10, a decreased gene expression of the proinflammatory Th1 cytokines IL2 and interferon Table I. Methotrexate (MTX) treatment response according to the literature review Treatment response Total n = 116 Response 65.5% 90.9% No response 34.5% 9.1% Total clearance 23.3% 40.9% Excellent improvement 17.2% 31.8% Partial improvement 6.9% 11.4% Slight improvement 3.4% 6.8% Any improvement 14.7% No improvement 34.5% 9.1% MTX single + reliable n = 44 Overall response rates (total) and response after excluding studies with systemic double-treatment or low reliability (MTX single + reliable). gamma (IFNγ), or the induction of apoptosis in cells involved in the immune/inflammatory reaction could play a therapeutic role (4). Treatment efficacies are difficult to assess in PRP due to potential spontaneous clearing in up to 80% of cases after 3 years reported by some authors (6). Thus, some of the therapeutic successes reported could be related to spontaneous clearing rather than MTX treatment. Nevertheless, the use of MTX in PRP is strongly supported by the remarkable chronical correlation between MTX initiation or MTX reinstitution after therapy discontinuation and disease improvement observed in numerous studies (2, 7 12). MTX was effective in our 3 patients with PRP, inducing almost complete remission in all patients, and it was well tolerated with no major adverse drug reactions. Combining our data with the available literature, an overall response rate after MTX therapy could be observed in 65.5% of patients, with complete clearance or excellent improvement in 23.3% and 17.2%, respectively. Furthermore, after exclusion of publications with other concomitant systemic therapy or low reliability, the overall response rate increased considerably, to 90.9%, with a complete clearance or excellent improvement in 40.9% and 31.8%, respectively. This remarkable difference can mainly be explained by a dropout of the 2 largest negative studies after excluding studies with low reliability. Significant therapy response could be observed after 3 12 weeks in several studies (2, 7 12). However, the majority of patients required therapy for several months (8, 12 21). In fact, in most cases MTX needs several weeks to become effective. MTX is a prodrug, which to become active needs a polyglutamation process, therefore it takes as long as 27.5 weeks to reach a steady state (22). Moreover, differences in absorption after oral vs. subcutaneous administration may explain why some patients respond better than others. In contrast to these positive data, one publication mentioned that only 17 out of 44 patients showed any response to MTX treatment (6). In addition, another study treating 5 patients (23), as well as several case reports did not observe any benefits of MTX treatment (24 29). Unfortunately some of these reports, in particular the 2 larger studies (6, 23), did not mention any MTX doses or treatment durations, hence they had to be classified in the low-reliability group. The reason for MTX failure in the other studies (24 29) could mainly be explained by the relatively short treatment periods of 2 weeks (24), 6 weeks (25), 1 2 months (26) and 3 months (27). The majority of the patients investigated had adultonset PRP; only 14 patients were children or had childhood-onset disease. Although, with overall response rates of 57.1%, the benefit of MTX treatment was lower in childhood-onset compared with adult-onset disease, MTX was also effective in at least every second patient with childhood-onset. Acta Derm Venereol 2018
4 504 L. Koch et al. Ectropion is a characteristic and common finding in PRP. Interestingly, a case report by Durairaj & Horsley (14) observed dramatic resolution of a cicatricial ectropion after treatment with low-dose MTX. There is insufficient data available to recommend whether MTX should be prescribed orally or parenterally for treatment of PRP. Some authors showed an advantage of oral compared with parental drug administration in 2 patients (7). However, others reported a case in which 25 mg orally once weekly for 6 months did not lead to any improvement, but after 3 injections of 50 mg/week intravenously, the skin was 95% clear (12). A study treating patients with rheumatoid arthritis suggests greater efficacy with equal tolerability of subcutaneous MTX compared with oral application (30). This increase in efficacy could be explained by a plateau effect of systemic drug exposure after oral application of 15 mg MTX, which was not observed in subcutaneous administration (31). Although we cannot transfer these findings to PRP patients with certainty, we prefer to administer MTX subcutaneously once weekly due to this mentioned plateau effect and further disadvantages of oral application, such as potentially increased gastrointestinal adverse reactions, varying drug levels because of fluctuating or insufficient gastrointestinal resorption of MTX, or a potentially erroneous daily instead of weekly oral dosing. Concerning MTX doses recommended in PRP, there is no official statement available, as the use is still off-label. Although individual publications had used doses of up to 70 mg/week, most of the studies had administered doses between 15 and 25 mg. Reports with doses higher than 25 mg/week did not show any trend to a better outcome, but the risk of side-effects theoretically increases with higher doses. Combining the available data we recommend doses from 15 to 25 mg per week, similar to the dose for use in psoriasis (32), with individual dose adaption for every patient. After clinical improvement, the dose should be tapered to the lowest effective dose for maintenance therapy and then, as soon as the disease allows, gradually tapered off and ceased. Sixteen adverse drug reactions could be observed in 15 out of 116 patients (12.9% of the study population) (2, 7 10, 12, 15, 19, 20, 26). The majority (n = 11) were mild side-effects including leucopaenia (n = 6), elevated liver function parameters (n = 3) and oral ulcers (n = 2), one of the latter 2 patients also had gastrointestinal side-effects. All of these adverse drug reactions resolved completely after short-term discontinuation of therapy, and although relapse of the disease occurred in some patients when MTX was temporarily suspended, reinstitution of the drug usually resulted in rapid clinical improvement (2, 7, 10, 12). Nevertheless, in 5 patients (4.3% of the study population) MTX therapy was withdrawn completely due to more severe adverse drug reactions, including one case of pneumocystis carinii pneumonia, 2 reports of severe upper gastrointestinal symptoms and 2 reports of gastrointestinal intolerance (9, 19, 20, 26). Three patients died due to diseases unrelated to PRP or therapy. To minimize adverse drug reactions, regular clinical and laboratory controls during MTX treatment are mandatory, as publish ed previously (32). Our approach is listed in Table SI 1. Regarding therapy alternatives, oral retinoids often serve as first-line therapy in PRP. Treatment is usually well tolerated and therapy response is satisfactory, with improvement in up to 76.2% or 96% of patients (13, 33). However, in the latter study only 6 out of 45 patients experienced complete or near complete remission post-treatment (34). Furthermore, in our study population, 15 out of 116 patients had unsuccessful retinoid pre-treatment. For severe and difficult to treat PRP a combination of MTX and retinoids could be an option, although it has to be stated, that according to the Food and Drug Administration (FDA) approved prescribing information, e.g. for acitretin, this combination therapy is contraindicated. The reason is an increased hepatotoxic risk, which resulted in a case of toxic hepatitis in a patient with psoriasis (35). However, a review addressing this issue concluded that the evidence is not strong enough to support a general contraindication regarding the concomitant use of methotrexate and retinoids (36), although consequent laboratory monitoring is mandatory. Out of 116 patients included in this review, 14 patients in 3 studies had concomitant MTX and retinoid therapy with overall response rates of 85.7%, complete clearance in 50% and excellent improvement in 35.7%, with no hepato toxic adverse drug reactions mentioned (20, 37, 38). Another treatment option for therapy-resistant PRP is the use of biologicals such as TNFα antagonists, ustekinumab or secukinumab as well as the use of the phosphodiesterase 4 inhibitor apremilast, with promising results published recently (18, 29, 30, 39 44). Nevertheless, at present these drugs remain second-line in most countries due to availability and costs. This study had a number of major limitations. Potential publication bias due to lower publication rates of negative results and the above-mentioned potential spontaneous clearing of the disease could have led to an overestimation of response rates. In addition, although patients requiring combination therapy, who probably experience the most severe forms of PRP, were included, some cases could have been omitted because MTX treatment effects were not mentioned in all studies with multiple therapies. Furthermore, there is a possibility that we have missed publications that are not listed in MEDLINE or did not meet our search criteria. In conclusion, the available literature confirms positive response rates and good tolerability of MTX in the treatment of PRP. Therefore, MTX might be considered as a first-line treatment option for PRP, considering also the cost of biological therapies.
5 Methotrexate treatment for pityriasis rubra pilaris 505 The authors have no conflicts of interest to declare. REFERENCES 1. Elango T, Dayalan H, Gnanaraj P, Malligarjunan H, Subramanian S. Impact of methotrexate on oxidative stress and apoptosis markers in psoriatic patients. Clin Exp Med 2014; 14: Brown J, Perry HO. Pityriasis rubra pilaris. Treatment with folic acid antagonists. Arch Dermatol 1966; 94: Porter D, Shuster S. Epidermal renewal and amino acids in psoriasis and pityriasis rubra pilaris. Arch Dermatol 1968; 98: Cutolo M, Sulli A, Pizzorni C, Seriolo B, Straub RH. Antiinflammatory mechanisms of methotrexate in rheumatoid arthritis. Ann Rheum Dis 2001; 60: Czarnecka-Operacz M, Sadowska-Przytocka A. The possibilities and principles of methotrexate treatment of psoriasis the updated knowledge. Postepy Dermatol Alergol 2014; 31: Griffiths WA. Pityriasis rubra pilaris. Clin Exp Dermatol 1980; 5: Knowles WR, Chernosky ME. Pityriasis rubra pilaris. Prolonged treatment with methotrexate. Arch Dermatol 1970; 102: Chapalain V, Beylot-Barry M, Doutre M, Beylot C. Treatment of pityriasis rubra pilaris: a retrospective study of 14 patients. J Dermatolog Treat 1999; 10: Haustein UF, Schulze P, Sonnichsen N. Long-term treatment of the erythrodermic form of pityriasis rubra pilaris using methotrexate. Dermatol Monatsschr 1972; 158: Parish LC, Woo TH. Pityriasis rubra pilaris in Korea. Treatment with methotrexate. Dermatologica 1969; 139: Anderson FE. Pityriasis rubra pilaris treated with methotrexate. Aust J Dermatol 1966; 8: Hanke CW, Steck WD. Childhood-onset pityriasis rubra pilaris treated with methotrexate administered intravenously. Cleveland Clinic Q 1983; 50: Dicken CH. Treatment of classic pityriasis rubra pilaris. J Am Acad Dermatol 1994; 31: Durairaj VD, Horsley MB. Resolution of pityriasis rubra pilaris-induced cicatricial ectropion with systemic low-dose methotrexate. Am J Ophthalmol 2007; 143: Marchetti MA, Greer KE. Pityriasis rubra pilaris treated with methotrexate resolving with an erythema gyratum repenslike appearance. J Am Acad Dermatol 2013; 69: e van Dooren-Greebe RJ, van de Kerkhof PC. Extensive extraspinal hyperostoses after long-term oral retinoid treatment in a patient with pityriasis rubra pilaris. J Am Acad Dermatol 1995; 32: Borrie P. Pityriasis rubra pilaris treated with methotrexate. Br J Dermatol 1966; 79: Manoharan S, White S, Gumparthy K. Successful treatment of type I adult-onset pityriasis rubra pilaris with infliximab. Australas J Dermatol 2006; 47: Duncan KO, Imaeda S, Milstone LM. Pneumocystis carinii pneumonia complicating methotrexate treatment of pityriasis rubra pilaris. J Am Acad Dermatol 1998; 39: Clayton BD, Jorizzo JL, Hitchcock MG, Fleischer AB, Jr, Williford PM, Feldman SR, et al. Adult pityriasis rubra pilaris: a 10-year case series. J Am Acad Dermatol 1997; 36: Conaghan PG, Sommer S, McGonagle D, Veale D, Waldmann H, Hale G, et al. The relationship between pityriasis rubra pilaris and inflammatory arthritis: case report and response of the arthritis to anti-tumor necrosis factor immunotherapy. Arthritis Rheum 1999; 42: Dalrymple JM, Stamp LK, O Donnell JL, Chapman PT, Zhang M, Barclay ML. Pharmacokinetics of oral methotrexate in patients with rheumatoid arthritis. Arthritis Rheum 2008; 58: Allison DS, El-Azhary RA, Calobrisi SD, Dicken CH. Pityriasis rubra pilaris in children. J Am Acad Dermatol 2002; 47: Lamar LM, Gaethe G. Pityriasis rubra pilaris. Arch Dermatol 1964; 89: Kaulen-Becker L. Pityriasis rubra pilaris. Z Haut Geschlechtskr 1972; 47: Schmitt L, Inhoff O, Dippel E. Oral alitretinoin for the treatment of recalcitrant pityriasis rubra pilaris. Case Rep Dermatol 2011; 3: Lim JT, Tham SN. Pityriasis rubra pilaris in Singapore. Clin Exp Dermatol 1991; 16: Ruzzetti M, Saraceno R, Carboni I, Papoutsaki M, Chimenti S. Type III juvenile pityriasis rubra pilaris: a successful treatment with infliximab. J Eur Acad Dermatol Venereol 2008; 22: Barth D, Harth W, Treudler R, Simon JC. Successful treatment of pityriasis rubra pilaris (type 1) under combination of infliximab and methotrexate. J Dtsch Dermatol Ges 2009; 7: Braun J, Kastner P, Flaxenberg P, Wahrisch J, Hanke P, Demary W, et al. Comparison of the clinical efficacy and safety of subcutaneous versus oral administration of methotrexate in patients with active rheumatoid arthritis: results of a sixmonth, multicenter, randomized, double-blind, controlled, phase IV trial. Arthritis Rheum 2008; 58: Schiff MH, Jaffe JS, Freundlich B. Head-to-head, randomised, crossover study of oral versus subcutaneous methotrexate in patients with rheumatoid arthritis: drug-exposure limitations of oral methotrexate at doses >/=15 mg may be overcome with subcutaneous administration. Ann Rheum Dis 2014; 73: Kalb RE, Strober B, Weinstein G, Lebwohl M. Methotrexate and psoriasis: 2009 National Psoriasis Foundation Consensus Conference. J Am Acad Dermatol 2009; 60: Goldsmith LA, Weinrich AE, Shupack J. Pityriasis rubra pilaris response to 13-cis-retinoic acid (isotretinoin). J Am Acad Dermatol 1982; 6: Albert MR, Mackool BT. Pityriasis rubra pilaris. Int J Dermatol 1999; 38: Beck HI, Foged EK. Toxic hepatitis due to combination therapy with methotrexate and etretinate in psoriasis. Dermatologica 1983; 167: Searles AD, Lee AD, Feldman SR. Is concomitant use of methotrexate and oral retinoids dangerous? A review of the data regarding this combination. J Am Acad Dermatol 2011; 64: Eastham AB, Femia AN, Qureshi A, Vleugels RA. Treatment options for pityriasis rubra pilaris including biologic agents: a retrospective analysis from an academic medical center. JAMA Dermatol 2014; 150: Bonomo RA, Korman N, Nagashima-Whalen L, Briggs J, Graham R, Salata RA. Pityriasis rubra pilaris: an unusual cutaneous complication of AIDS. Am J Med Sci 1997; 314: Gemmeke A, Schonlebe J, Koch A, Wollina U. Pityriasis rubra pilaris a retrospective single center analysis over eight years. J Dtsch Dermatol Ges 2010; 8: Muller H, Gattringer C, Zelger B, Hopfl R, Eisendle K. Infliximab monotherapy as first-line treatment for adult-onset pityriasis rubra pilaris: case report and review of the literature on biologic therapy. J Am Acad Dermatol 2008; 59: S Garcovich S, Di Giampetruzzi AR, Antonelli G, Garcovich A, Didona B. Treatment of refractory adult-onset pityriasis rubra pilaris with TNF-alpha antagonists: a case series. J Eur Acad Dermatol Venereol 2010; 24: Schuster D, Pfister-Wartha A, Bruckner-Tuderman L, Schempp CM. Successful treatment of refractory pityriasis rubra pilaris with secukinumab. JAMA Dermatol 2016; 152: Feldmeyer L, Mylonas A, Demaria O, Mennella A, Yawalkar N, Laffitte E, et al. Interleukin 23-helper T cell 17 axis as a treatment target for pityriasis rubra pilaris. JAMA Dermatol 2017; 153: Krase IZ, Cavanaugh K, Curiel-Lewandrowski C. Treatment of refractory pityriasis rubra pilaris with novel phosphodiesterase 4 (PDE4) inhibitor apremilast. JAMA Dermatol 2016; 152: Acta Derm Venereol 2018
A systematic review of the literature on the treatment of pityriasis rubra pilaris type 1 with TNF-antagonists
 DOI: 10.1111/j.1468-3083.2012.04456.x JEADV SHORT REPORT A systematic review of the literature on the treatment of pityriasis rubra pilaris type 1 with TNF-antagonists G. Petrof,* N. Almaani, C.B. Archer,
DOI: 10.1111/j.1468-3083.2012.04456.x JEADV SHORT REPORT A systematic review of the literature on the treatment of pityriasis rubra pilaris type 1 with TNF-antagonists G. Petrof,* N. Almaani, C.B. Archer,
U059 Hidradeni-s Suppura-va and Pityriasis Rubra Pilaris: Updates on Treatment
 U059 Hidradeni-s Suppura-va and Pityriasis Rubra Pilaris: Updates on Treatment DISCLOSURES I do not have any relevant relationships with industry. Scott Worswick UCLA Dermatology Director of Inpatient
U059 Hidradeni-s Suppura-va and Pityriasis Rubra Pilaris: Updates on Treatment DISCLOSURES I do not have any relevant relationships with industry. Scott Worswick UCLA Dermatology Director of Inpatient
Combination Nonbiologic Therapy in Psoriasis. Sushil Tahiliani, MBBS, MD
 Combination Nonbiologic Therapy in Psoriasis Sushil Tahiliani, MBBS, MD Agenda Rationale Preferred and less preferred combination Morphology-specific preferred combinations Doses used in combinations Potential
Combination Nonbiologic Therapy in Psoriasis Sushil Tahiliani, MBBS, MD Agenda Rationale Preferred and less preferred combination Morphology-specific preferred combinations Doses used in combinations Potential
chemotherapeutic agents in
 Use of biologics and chemotherapeutic agents in cutaneous emergencies: Focus on lifethreatening forms of psoriasis Alice Bendix Gottlieb MD, PhD Professor of Dermatology New York Medical College Metropolitan
Use of biologics and chemotherapeutic agents in cutaneous emergencies: Focus on lifethreatening forms of psoriasis Alice Bendix Gottlieb MD, PhD Professor of Dermatology New York Medical College Metropolitan
Long-term ustekinumab treatment for refractory type I pityriasis rubra pilaris
 DOI: http://dx.doi.org/10.331/jdcr.2013.1127 Long-term ustekinumab treatment for refractory type I pityriasis rubra pilaris Alessandro Di Stefani, Marco Galluzzo, Marina Talamonti, Andrea Chiricozzi, Antonio
DOI: http://dx.doi.org/10.331/jdcr.2013.1127 Long-term ustekinumab treatment for refractory type I pityriasis rubra pilaris Alessandro Di Stefani, Marco Galluzzo, Marina Talamonti, Andrea Chiricozzi, Antonio
The Natural History of Psoriasis and Treatment Goals
 The Natural History of Psoriasis and Treatment Goals Psoriasis Epidemiology Prevalence Affects 2 3% of adult population (>7 million in US) Caucasians: 25% 2.5% African Americans: 1.3% (more likely to have
The Natural History of Psoriasis and Treatment Goals Psoriasis Epidemiology Prevalence Affects 2 3% of adult population (>7 million in US) Caucasians: 25% 2.5% African Americans: 1.3% (more likely to have
Michael P. Heffernan, M.D San Luis Dermatology & Laser Clinic Director, US Probity Medical Research
 Michael P. Heffernan, M.D San Luis Dermatology & Laser Clinic Director, US Probity Medical Research mpheffernanmd@gmail.com DISCLOSURES Consultant, Speaker, Investigator: Abbvie, Amgen, Brickell Biotech,
Michael P. Heffernan, M.D San Luis Dermatology & Laser Clinic Director, US Probity Medical Research mpheffernanmd@gmail.com DISCLOSURES Consultant, Speaker, Investigator: Abbvie, Amgen, Brickell Biotech,
Update on systemic therapies and emerging treatments How do I choose a systemic agent?
 Update on systemic therapies and emerging treatments How do I choose a systemic agent? Amy S. Paller, M.D. Walter J. Hamlin Professor and Chair of Dermatology Professor of Pediatrics Northwestern University
Update on systemic therapies and emerging treatments How do I choose a systemic agent? Amy S. Paller, M.D. Walter J. Hamlin Professor and Chair of Dermatology Professor of Pediatrics Northwestern University
Sarcoidosis Case. Robert P. Baughman Interstitial Lung Disease and Sarcoidosis Clinic University of Cincinnati, USA. WASOG: educational material
 Sarcoidosis Case Robert P. Baughman Interstitial Lung Disease and Sarcoidosis Clinic University of Cincinnati, USA WASOG: educational material Sarcoidosis Case patient is a Caucasian male age 46 was diagnosed
Sarcoidosis Case Robert P. Baughman Interstitial Lung Disease and Sarcoidosis Clinic University of Cincinnati, USA WASOG: educational material Sarcoidosis Case patient is a Caucasian male age 46 was diagnosed
Scottish Medicines Consortium
 Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
Recalcitrant Warty Erythroderma With Severe Pruritus. Gil Yosipovitch Professor & Chair Department of Dermatology & Itch Center Temple University
 Recalcitrant Warty Erythroderma With Severe Pruritus Gil Yosipovitch Professor & Chair Department of Dermatology & Itch Center Temple University DISCLOSURE OF RELEVANT RELATIONSHIPS WITH INDUSTRY Gil Yosipovitch,
Recalcitrant Warty Erythroderma With Severe Pruritus Gil Yosipovitch Professor & Chair Department of Dermatology & Itch Center Temple University DISCLOSURE OF RELEVANT RELATIONSHIPS WITH INDUSTRY Gil Yosipovitch,
Psoriasiform Dermatitis in Children: Calling in the Troops
 Psoriasiform Dermatitis in Children: Calling in the Troops Markus Boos, MD PhD Attending Physician, Dermatology Seattle Children s Hospital Assistant Professor of Pediatrics, University of Washington School
Psoriasiform Dermatitis in Children: Calling in the Troops Markus Boos, MD PhD Attending Physician, Dermatology Seattle Children s Hospital Assistant Professor of Pediatrics, University of Washington School
Original Policy Date
 MP 2.01.07 Psoralens with Ultraviolet A (PUVA) Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed by consensus/12:2013 Return to Medical Policy
MP 2.01.07 Psoralens with Ultraviolet A (PUVA) Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed by consensus/12:2013 Return to Medical Policy
TRANSPARENCY COMMITTEE OPINION. 26 April 2006
 TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
Ustekinumab Treatment of Erythrodermic Psoriasis Occurring after Physical Stress: A Report of Two Cases
 Published online: September 26, 2013 1662 6567/13/0053 0254$38.00/0 This is an Open Access article licensed under the terms of the Creative Commons Attribution-NonCommercial 3.0 Unported license (CC BY-NC)
Published online: September 26, 2013 1662 6567/13/0053 0254$38.00/0 This is an Open Access article licensed under the terms of the Creative Commons Attribution-NonCommercial 3.0 Unported license (CC BY-NC)
ETANERCEPT Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Clinical Policy: Brodalumab (Siliq) Reference Number: CP.PHAR.375 Effective Date: Last Review Date: 05.18
 Clinical Policy: (Siliq) Reference Number: CP.PHAR.375 Effective Date: 06.01.18 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Siliq) Reference Number: CP.PHAR.375 Effective Date: 06.01.18 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Clinical Policy: Ixekizumab (Taltz) Reference Number: ERX.SPA.122 Effective Date:
 Clinical Policy: (Taltz) Reference Number: ERX.SPA.122 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Taltz) Reference Number: ERX.SPA.122 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8
 London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
The role of current biologic therapies in psoriasis
 : An Update on and IL-17 Inhibitors Joanna Dong, BA; Gary Goldenberg, MD PRACTICE POINTS The newest biologics for treatment of moderate to severe plaque psoriasis are and IL-17 inhibitors with unprecedented
: An Update on and IL-17 Inhibitors Joanna Dong, BA; Gary Goldenberg, MD PRACTICE POINTS The newest biologics for treatment of moderate to severe plaque psoriasis are and IL-17 inhibitors with unprecedented
Clinical Policy: Ustekinumab (Stelara) Reference Number: ERX.SPA.01 Effective Date:
 Clinical Policy: (Stelara) Reference Number: ERX.SPA.01 Effective Date: 04.01.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Stelara) Reference Number: ERX.SPA.01 Effective Date: 04.01.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
C. Assess clinical response after the first three months of treatment.
 Government Health Plan (GHP) of Puerto Rico Authorization Criteria Tumor Necrosis Factor Alpha (TNFα) Adalimumab (Humira ) Managed by MCO Section I. Prior Authorization Criteria A. Physician must submit
Government Health Plan (GHP) of Puerto Rico Authorization Criteria Tumor Necrosis Factor Alpha (TNFα) Adalimumab (Humira ) Managed by MCO Section I. Prior Authorization Criteria A. Physician must submit
Table SIII. Evidence table. First author (Ref), year Design Dosage Outcome measures Adalimumab Ryan (12), Date of evaluation
 Table SIII. Evidence table First author (Ref), year Design Dosage Outcome measures Adalimumab Ryan (12), 2009 (n = 39 a ) van Lümig (13), 2010 (n = 30 a ) Warren (14), 2010 (n = 46 a ) Retrosp b Adalimumab
Table SIII. Evidence table First author (Ref), year Design Dosage Outcome measures Adalimumab Ryan (12), 2009 (n = 39 a ) van Lümig (13), 2010 (n = 30 a ) Warren (14), 2010 (n = 46 a ) Retrosp b Adalimumab
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 1097-7 Program Prior Authorization/Notification Medication *Stelara (ustekinumab) *This program applies to the subcutaneous formulation
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 1097-7 Program Prior Authorization/Notification Medication *Stelara (ustekinumab) *This program applies to the subcutaneous formulation
Clinical Policy: Ixekizumab (Taltz) Reference Number: CP.PHAR.257 Effective Date: Last Review Date: 05.18
 Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Elements of Successful PBS Applications. Barbara Radulski RN. Copyright
 Elements of Successful PBS Applications Barbara Radulski RN PBS Requirements April 1 2006 THE RULES PBS Requirements 18 years and over Psoriasis x 6 months Failed to achieve an adequate response to 3 systemic
Elements of Successful PBS Applications Barbara Radulski RN PBS Requirements April 1 2006 THE RULES PBS Requirements 18 years and over Psoriasis x 6 months Failed to achieve an adequate response to 3 systemic
Pityriasis rubra pilaris: Not just skin deep a case series
 Hong Kong J. Dermatol. Venereol. (2016) 24, 129-134 Views and Practice Pityriasis rubra pilaris: Not just skin deep a case series SN Chee and S Lee Case series Pityriasis rubra pilaris (PRP) is an idiopathic
Hong Kong J. Dermatol. Venereol. (2016) 24, 129-134 Views and Practice Pityriasis rubra pilaris: Not just skin deep a case series SN Chee and S Lee Case series Pityriasis rubra pilaris (PRP) is an idiopathic
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 2104-4 Program Prior Authorization/Medical Necessity Medication Taltz (ixekizumab) P&T Approval Date 8/2016, 5/2017, 2/2018 Effective
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 2104-4 Program Prior Authorization/Medical Necessity Medication Taltz (ixekizumab) P&T Approval Date 8/2016, 5/2017, 2/2018 Effective
ADALIMUMAB Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
1. Background: Infliximab is administered parenterally; therefore, it is not covered under retail pharmacy benefits.
 Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Keywords: Psoriasis vulgaris Zinc pyrithione Betamethasone dipropionate
 CLINICAL EFFICACY AND SAFETY OF A COMBINED FORMULATION OF ZINC PYRITHIONE 0.25% AND BETAMETHASONE DIPROPIONATE MICRONIZED 0.05% IN THE TREATMENT OF MILD TO MODERATE PLAQUE PSORIASIS. Abstract Background
CLINICAL EFFICACY AND SAFETY OF A COMBINED FORMULATION OF ZINC PYRITHIONE 0.25% AND BETAMETHASONE DIPROPIONATE MICRONIZED 0.05% IN THE TREATMENT OF MILD TO MODERATE PLAQUE PSORIASIS. Abstract Background
UvA-DARE (Digital Academic Repository) Innovative therapies and new targets in psoriasis de Groot, M. Link to publication
 UvA-DARE (Digital Academic Repository) Innovative therapies and new targets in psoriasis de Groot, M. Link to publication Citation for published version (APA): de Groot, M. (2011). Innovative therapies
UvA-DARE (Digital Academic Repository) Innovative therapies and new targets in psoriasis de Groot, M. Link to publication Citation for published version (APA): de Groot, M. (2011). Innovative therapies
Ustekinumab for severe treatment-resistant psoriasis: a 24-week pilot study in Hong Kong Chinese
 Hong Kong J. Dermatol. Venereol. (2011) 19, 59-64 Original Article Ustekinumab for severe treatment-resistant psoriasis: a 24-week pilot study in Hong Kong Chinese Ustekinumab SKF Loo, KH Lau, KM Ho Introduction:
Hong Kong J. Dermatol. Venereol. (2011) 19, 59-64 Original Article Ustekinumab for severe treatment-resistant psoriasis: a 24-week pilot study in Hong Kong Chinese Ustekinumab SKF Loo, KH Lau, KM Ho Introduction:
A study of treatment modalities in psoriasis in dermatology outpatient department of a tertiary care teaching hospital
 Original article A study of treatment modalities in psoriasis in dermatology outpatient department of a tertiary care teaching hospital 1Y Roja Ramani, 2 Benu Panigrahy, 3 Sailenkumar Mishra, 4 BTPS Singh
Original article A study of treatment modalities in psoriasis in dermatology outpatient department of a tertiary care teaching hospital 1Y Roja Ramani, 2 Benu Panigrahy, 3 Sailenkumar Mishra, 4 BTPS Singh
Methotrexate Injectable Step Therapy Program Summary
 Methotrexate Injectable Step Therapy Program Summary This prior authorization applies to Commercial, SourceRx and Health Insurance Marketplace formularies. OBJECTIVE The intent of the methotrexate injectable
Methotrexate Injectable Step Therapy Program Summary This prior authorization applies to Commercial, SourceRx and Health Insurance Marketplace formularies. OBJECTIVE The intent of the methotrexate injectable
ustekinumab (Stelara )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Clinical Policy: Apremilast (Otezla) Reference Number: CP.PHAR.245 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: Ixekizumab (Taltz) Reference Number: CP.PHAR.257 Effective Date: Last Review Date: 11.18
 Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 11.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 11.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
USTEKINUMAB Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA GUIDELINES FOR USE
 Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA 36187 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of psoriatic arthritis (PsA)
Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA 36187 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of psoriatic arthritis (PsA)
Psoriasis: Therapeutic goals
 Psoriasis: Therapeutic goals I want to die 50 45 impetiginization infliximab 600 40 35 30 400 25 20 15 200 10 5 0 22-ene 21-feb 23-mar 22-abr 22- may Efalizumab 6 doses: flare + REBOUND CSA 3 21-jun 21-jul
Psoriasis: Therapeutic goals I want to die 50 45 impetiginization infliximab 600 40 35 30 400 25 20 15 200 10 5 0 22-ene 21-feb 23-mar 22-abr 22- may Efalizumab 6 doses: flare + REBOUND CSA 3 21-jun 21-jul
An otherwise healthy 12-year-old
 1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
What is Cosentyx (secukinumab)?
 What is Cosentyx (secukinumab)? Cosentyx is the first of a new class of medicines called interleukin- 17A (IL- 17A) inhibitors to be approved for the treatment of moderate- to- severe plaque psoriasis,
What is Cosentyx (secukinumab)? Cosentyx is the first of a new class of medicines called interleukin- 17A (IL- 17A) inhibitors to be approved for the treatment of moderate- to- severe plaque psoriasis,
HARVARD PILGRIM HEALTH CARE RECOMMENDED MEDICATION REQUEST GUIDELINES HUMIRA PEDIATRIC
 Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 HUMIRA PEDIATRIC GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the patient currently taking Humira? If
Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 HUMIRA PEDIATRIC GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the patient currently taking Humira? If
Rheumatology journal club October 20, 2017 Presented by: Matthew Stoll MD,PhD,PSCS
 Efficacy and safety of abatacept, a T-cell modulator, in a randomised, double-blind, placebo-controlled, phase III study in psoriatic arthritis (Mease et al., 2017) Rheumatology journal club October 20,
Efficacy and safety of abatacept, a T-cell modulator, in a randomised, double-blind, placebo-controlled, phase III study in psoriatic arthritis (Mease et al., 2017) Rheumatology journal club October 20,
Moderately to severely active ulcerative colitis
 Adalimumab in the Treatment of Moderate-to-Severe Ulcerative Colitis: ULTRA 2 Trial Results Sandborn WJ, van Assche G, Reinisch W, et al. Adalimumab induces and maintains clinical remission in patients
Adalimumab in the Treatment of Moderate-to-Severe Ulcerative Colitis: ULTRA 2 Trial Results Sandborn WJ, van Assche G, Reinisch W, et al. Adalimumab induces and maintains clinical remission in patients
Abatacept (Orencia) for active rheumatoid arthritis. August 2009
 Abatacept (Orencia) for active rheumatoid arthritis August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Abatacept (Orencia) for active rheumatoid arthritis August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
1 P a g e. Systemic Juvenile Idiopathic Arthritis (SJIA) (1.3) Patients 2 years of age and older with active systemic juvenile idiopathic arthritis.
 LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
Primary Results Citation 2
 Table S1. Adalimumab clinical trials 1 ClinicalTrials.gov Rheumatoid Arthritis 3 NCT00195663 Breedveld FC, Weisman MH, Kavanaugh AF, et al. The PREMIER study. A multicenter, randomized, double-blind clinical
Table S1. Adalimumab clinical trials 1 ClinicalTrials.gov Rheumatoid Arthritis 3 NCT00195663 Breedveld FC, Weisman MH, Kavanaugh AF, et al. The PREMIER study. A multicenter, randomized, double-blind clinical
Pediatric Use: Safety and effectiveness of Ustekinumab (STELARA ) in pediatric patients have not been evaluated.
 Original Issue Date (Created): January 1, 2010 Most Recent Review Date (Revised): January 28, 2014 Effective Date: April 1, 2014 I. POLICY Preauthorization Requirements for Ustekinumab (STELARA ) Note:
Original Issue Date (Created): January 1, 2010 Most Recent Review Date (Revised): January 28, 2014 Effective Date: April 1, 2014 I. POLICY Preauthorization Requirements for Ustekinumab (STELARA ) Note:
Psoriasis in Jordan: a single center experience
 Psoriasis in Jordan: a single center experience Shefaa Almashagbeh MD *, Deifallah Alsharari MD *, Hayat Khasawneh MD *, Diana Aljammal * MD, Hamzeh Al-housamieh MD* ABSTRACT Objectives: To evaluate the
Psoriasis in Jordan: a single center experience Shefaa Almashagbeh MD *, Deifallah Alsharari MD *, Hayat Khasawneh MD *, Diana Aljammal * MD, Hamzeh Al-housamieh MD* ABSTRACT Objectives: To evaluate the
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC
 Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC Update on the Treatment of Rheumatoid Arthritis Sabrina Fallavollita MDCM McGill University Canadian Society of Internal Medicine
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC Update on the Treatment of Rheumatoid Arthritis Sabrina Fallavollita MDCM McGill University Canadian Society of Internal Medicine
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C
 A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Otezla (apremilast) Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Otezla (apremilast) Prime Therapeutics will review Prior Authorization requests Prior
Otezla (apremilast) Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Otezla (apremilast) Prime Therapeutics will review Prior Authorization requests Prior
Clinical Policy: Etanercept (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid
 Clinical Policy: (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log Description (Enbrel ) is tumor necrosis
Clinical Policy: (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log Description (Enbrel ) is tumor necrosis
Inflectra Frequently Asked Questions
 Inflectra Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Earlier in 2016, Inflectra (infliximab) was added to the Ontario Drug Benefit (ODB) Formulary as a Limited
Inflectra Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Earlier in 2016, Inflectra (infliximab) was added to the Ontario Drug Benefit (ODB) Formulary as a Limited
Efficacy of Concomitant Use of PUVA and Methotrexate in Disease Clearance Time in Plaque Type Psoriasis
 Efficacy of Concomitant Use of PUVA and Methotrexate in Disease Clearance Time in Plaque Type Psoriasis T. Shehzad ( Departments of Dermatology Naval Hospital PNS Shifa, Karachi. ) N. R. Dar ( Departments
Efficacy of Concomitant Use of PUVA and Methotrexate in Disease Clearance Time in Plaque Type Psoriasis T. Shehzad ( Departments of Dermatology Naval Hospital PNS Shifa, Karachi. ) N. R. Dar ( Departments
Clinical Policy: Secukinumab (Cosentyx) Reference Number: ERX.SPA.165 Effective Date:
 Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Medication Prior Authorization Form
 Policy Number: 1055 Policy History Approve Date: 06/01/2018 Effective Date: 06/01/2018 Preauthorization All Plans Benefit plans vary in coverage and some plans may not provide coverage for certain service(s)
Policy Number: 1055 Policy History Approve Date: 06/01/2018 Effective Date: 06/01/2018 Preauthorization All Plans Benefit plans vary in coverage and some plans may not provide coverage for certain service(s)
Certolizumab pegol (Cimzia) for chronic plaque psoriasis in adults
 NIHR Innovation Observatory Evidence Briefing: April 2017 Certolizumab pegol (Cimzia) for chronic plaque psoriasis in adults NIHRIO (HSRIC) ID: 2406 NICE ID: 9112 LAY SUMMARY Plaque psoriasis is the most
NIHR Innovation Observatory Evidence Briefing: April 2017 Certolizumab pegol (Cimzia) for chronic plaque psoriasis in adults NIHRIO (HSRIC) ID: 2406 NICE ID: 9112 LAY SUMMARY Plaque psoriasis is the most
COSENTYX (secukinumab)
 COSENTYX (secukinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
COSENTYX (secukinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Psoriasis. Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement
 Psoriasis Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement Copyright 2017 by Sea Courses Inc. All rights reserved. No part
Psoriasis Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement Copyright 2017 by Sea Courses Inc. All rights reserved. No part
New Evidence reports on presentations given at EULAR Tocilizumab for the Treatment of Rheumatoid Arthritis and Juvenile Idiopathic Arthritis
 New Evidence reports on presentations given at EULAR 2011 Tocilizumab for the Treatment of Rheumatoid Arthritis and Juvenile Idiopathic Arthritis Report on EULAR 2011 presentations Benefit of continuing
New Evidence reports on presentations given at EULAR 2011 Tocilizumab for the Treatment of Rheumatoid Arthritis and Juvenile Idiopathic Arthritis Report on EULAR 2011 presentations Benefit of continuing
Appendix 1: Frequently Asked Questions
 Appendix 1: Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added to the Ontario Drug Benefit (ODB) Formulary
Appendix 1: Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added to the Ontario Drug Benefit (ODB) Formulary
Clinical Policy: Apremilast (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17
 Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 13 May 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
INFLIXIMAB Remicade (infliximab), Inflectra (infliximab-dyyb), Ixifi* (infliximabqbtx), Renflexis (infliximab-abda)
 Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 6 years of age or older 1. Moderate to severe Crohn s disease (CD) a. Patient has fistulizing disease
Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 6 years of age or older 1. Moderate to severe Crohn s disease (CD) a. Patient has fistulizing disease
Psoriasis is a chronic, inflammatory, Prescribing in children
 Psoriasis in children: current approaches to management Laura Proudfoot BSc, MRCP, Elisabeth Higgins MA, FRCP and Judy Davids RGN Our series Prescribing in children gives practical advice for successful
Psoriasis in children: current approaches to management Laura Proudfoot BSc, MRCP, Elisabeth Higgins MA, FRCP and Judy Davids RGN Our series Prescribing in children gives practical advice for successful
Horizon Scanning Centre March Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798
 Horizon Scanning Centre March 2015 Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798 This briefing is based on information available at the time of research and a limited
Horizon Scanning Centre March 2015 Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798 This briefing is based on information available at the time of research and a limited
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
Technology appraisal guidance Published: 25 November 2015 nice.org.uk/guidance/ta368
 Apremilast for treating moderate to severe ere plaque psoriasis Technology appraisal guidance Published: 25 November 2015 nice.org.uk/guidance/ta368 NICE 2015. All rights reserved. Contents 1 Guidance...
Apremilast for treating moderate to severe ere plaque psoriasis Technology appraisal guidance Published: 25 November 2015 nice.org.uk/guidance/ta368 NICE 2015. All rights reserved. Contents 1 Guidance...
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
It is estimated that about 26,000 new cases of
 Focus on CME at Dalhousie University Set On Soothing Psoriasis A. H. Murray, MD, FRCP(C) Presented at the 76th Annual Dalhousie Refresher Course It is estimated that about 26,000 new cases of psoriasis
Focus on CME at Dalhousie University Set On Soothing Psoriasis A. H. Murray, MD, FRCP(C) Presented at the 76th Annual Dalhousie Refresher Course It is estimated that about 26,000 new cases of psoriasis
Public Assessment Report Scientific discussion. Metoject Metojectpen (methotrexate disodium) SE/H/643/01/DC SE/H/643/02-11/DC
 Public Assessment Report Scientific discussion Metoject Metojectpen (methotrexate disodium) SE/H/643/01/DC SE/H/643/02-11/DC This module reflects the scientific discussion for the approval of Metoject
Public Assessment Report Scientific discussion Metoject Metojectpen (methotrexate disodium) SE/H/643/01/DC SE/H/643/02-11/DC This module reflects the scientific discussion for the approval of Metoject
IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients
 IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients with 4 weeks of treatment in a Phase 2a trial A. B. Kimball 1, J. Krueger 2, T. Sullivan
IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients with 4 weeks of treatment in a Phase 2a trial A. B. Kimball 1, J. Krueger 2, T. Sullivan
Interesting Case Series. Skin Grafting in Pyoderma Gangrenosum
 Interesting Case Series Skin Grafting in Pyoderma Gangrenosum Marco Romanelli, MD, PhD, Agata Janowska, MD, Teresa Oranges, MD, and Valentina Dini, MD, PhD Department of Dermatology, University of Pisa,
Interesting Case Series Skin Grafting in Pyoderma Gangrenosum Marco Romanelli, MD, PhD, Agata Janowska, MD, Teresa Oranges, MD, and Valentina Dini, MD, PhD Department of Dermatology, University of Pisa,
USTEKINUMAB. London New Drugs Group APC/DTC Briefing Document. May 2009
 Page 1 London New Drugs Group APC/DTC Briefing Document USTEKINUMAB Contents Summary 1 Background 4 Clinical efficacy 5 Cost implications 9 Reference list 10 Appendices 11 Produced for the London New Drugs
Page 1 London New Drugs Group APC/DTC Briefing Document USTEKINUMAB Contents Summary 1 Background 4 Clinical efficacy 5 Cost implications 9 Reference list 10 Appendices 11 Produced for the London New Drugs
STELARA DATA SHEET NAME OF THE MEDICINE DESCRIPTION V L C L V H C H 1 C H 2 C H 3. Fab. F(ab)' 2. hinge
 DATA SHEET NAME OF THE MEDICINE Ustekinumab (rmc). CAS Registry Number: 815610-63-0. DESCRIPTION (ustekinumab) is a human IgG1kappa monoclonal antibody with an approximate molecular weight of 148,600 daltons.
DATA SHEET NAME OF THE MEDICINE Ustekinumab (rmc). CAS Registry Number: 815610-63-0. DESCRIPTION (ustekinumab) is a human IgG1kappa monoclonal antibody with an approximate molecular weight of 148,600 daltons.
Beneficial effect of ustekinumab in familial pityriasis rubra pilaris with a
 DR SU M LWIN (Orcid ID : 0000-0002-3325-3675) Received Date : 09-Jan-2017 Revised Date : 03-Mar-2017 Accepted Date : 08-Mar-2017 Article type : Case Report Beneficial effect of ustekinumab in familial
DR SU M LWIN (Orcid ID : 0000-0002-3325-3675) Received Date : 09-Jan-2017 Revised Date : 03-Mar-2017 Accepted Date : 08-Mar-2017 Article type : Case Report Beneficial effect of ustekinumab in familial
Psoriasis. Overview. Epidemiology. Epidemiology 08/08/2015. Dr Nigel Burrows Consultant Dermatologist Addenbrooke s Hospital
 Overview Psoriasis 1. Epidemiology of psoriasis 2. Histology Dr Nigel Burrows Consultant Dermatologist Addenbrooke s Hospital Aug 2015 3. Types of psoriasis 4. Assessing severity 5. Treatments Topical
Overview Psoriasis 1. Epidemiology of psoriasis 2. Histology Dr Nigel Burrows Consultant Dermatologist Addenbrooke s Hospital Aug 2015 3. Types of psoriasis 4. Assessing severity 5. Treatments Topical
Circle Yes or No Y N. [If no, skip to question 7.] 2. Does the patient have a diagnosis of ulcerative colitis? Y N. [If no, skip to question 4.
![Circle Yes or No Y N. [If no, skip to question 7.] 2. Does the patient have a diagnosis of ulcerative colitis? Y N. [If no, skip to question 4. Circle Yes or No Y N. [If no, skip to question 7.] 2. Does the patient have a diagnosis of ulcerative colitis? Y N. [If no, skip to question 4.](/thumbs/85/91758188.jpg) 06/01/2016 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Humira (MI88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
06/01/2016 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Humira (MI88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Ixekizumab (plaque psoriasis)
 IQWiG Reports Commission No. A17-07 Ixekizumab (plaque psoriasis) Benefit assessment according to 35a Social Code Book V 1 Extract 1 Translation of Sections 2.1 to 2.5 of the dossier assessment Ixekizumab
IQWiG Reports Commission No. A17-07 Ixekizumab (plaque psoriasis) Benefit assessment according to 35a Social Code Book V 1 Extract 1 Translation of Sections 2.1 to 2.5 of the dossier assessment Ixekizumab
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Proposed Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Dimethyl fumarate for treating moderate to severe Draft scope (pre-referral) Draft remit/appraisal objective To appraise
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Dimethyl fumarate for treating moderate to severe Draft scope (pre-referral) Draft remit/appraisal objective To appraise
Minami-Hori M, Tsuji H, Takahashi H, Hanada K, Iizuka H
 The Journal of Dermatology (2013.Jan) :. Optic neuritis in a psoriatic arthritis patient treated by infliximab Minami-Hori M, Tsuji H, Takahashi H, Hanada K, Iizuka H 1 Optic neuritis in a psoriatic arthritis
The Journal of Dermatology (2013.Jan) :. Optic neuritis in a psoriatic arthritis patient treated by infliximab Minami-Hori M, Tsuji H, Takahashi H, Hanada K, Iizuka H 1 Optic neuritis in a psoriatic arthritis
THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS
 Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 5 (54) No. 2-2012 THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS Mădălina FRÎNCU 1 Abstract: Biological
Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 5 (54) No. 2-2012 THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS Mădălina FRÎNCU 1 Abstract: Biological
The New and Emerging Agents: Dermatology
 Psoriasis Treatment 2013: What is New and on the Horizon? Neil J Korman, MD, PhD Professor of Dermatology University Hospitals Case Medical Center Clinical Director Murdough Family Center for Psoriasis
Psoriasis Treatment 2013: What is New and on the Horizon? Neil J Korman, MD, PhD Professor of Dermatology University Hospitals Case Medical Center Clinical Director Murdough Family Center for Psoriasis
Drug Utilization Evaluation of Methotrexate in Psoriasis in a Tertiary Care Teaching Hospital
 Original Article Drug Utilization Evaluation of Methotrexate in Psoriasis in a Tertiary Care Teaching Hospital Vijeesh Tholur Subrahmanian 1, Kannan Gopal 2, Murugan Sundaram 3, Purnima Ashok 1, Satya
Original Article Drug Utilization Evaluation of Methotrexate in Psoriasis in a Tertiary Care Teaching Hospital Vijeesh Tholur Subrahmanian 1, Kannan Gopal 2, Murugan Sundaram 3, Purnima Ashok 1, Satya
Clinical Policy: Ustekinumab (Stelara) Reference Number: ERX.SPMN.167
 Clinical Policy: (Stelara) Reference Number: ERX.SPMN.167 Effective Date: 10/16 Last Review Date: 12/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Stelara) Reference Number: ERX.SPMN.167 Effective Date: 10/16 Last Review Date: 12/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Pravit Asawanonda, MD, DSc, and Yaowalak Nateetongrungsak, MD Bangkok, Thailand
 Methotrexate plus narrowband UVB phototherapy versus narrowband UVB phototherapy alone in the treatment of plaque-type psoriasis: A randomized, placebo-controlled study Pravit Asawanonda, MD, DSc, and
Methotrexate plus narrowband UVB phototherapy versus narrowband UVB phototherapy alone in the treatment of plaque-type psoriasis: A randomized, placebo-controlled study Pravit Asawanonda, MD, DSc, and
This PDF is available for free download from a site hosted by Medknow Publications
 Net Study Comparison of clinical efficacy of topical tazarotene.1% cream with topical clobetasol propionate.5% cream in chronic plaque psoriasis: A double-blind, randomized, right-left comparison study
Net Study Comparison of clinical efficacy of topical tazarotene.1% cream with topical clobetasol propionate.5% cream in chronic plaque psoriasis: A double-blind, randomized, right-left comparison study
Break the Liver Biopsy Habit
 Break the Liver Biopsy Habit Dirk Elston MD Professor and Chairman Department of Dermatology and Dermatologic Surgery Medical University of South Carolina No conflict of interest Father of one of my nurses:
Break the Liver Biopsy Habit Dirk Elston MD Professor and Chairman Department of Dermatology and Dermatologic Surgery Medical University of South Carolina No conflict of interest Father of one of my nurses:
Subcutaneous Methotrexate for Rheumatoid Arthritis
 DRUG NEWS Subcutaneous Methotrexate for Rheumatoid Arthritis Summary Subcutaneous administration of methotrexate is recommended in the treatment of rheumatoid arthritis by the Canadian Rheumatology Association
DRUG NEWS Subcutaneous Methotrexate for Rheumatoid Arthritis Summary Subcutaneous administration of methotrexate is recommended in the treatment of rheumatoid arthritis by the Canadian Rheumatology Association
Horizon Scanning Centre January Apremilast for psoriasis SUMMARY NIHR HSC ID: 2652
 Horizon Scanning Centre January 2013 Apremilast for psoriasis SUMMARY NIHR HSC ID: 2652 This briefing is based on information available at the time of research and a limited literature search. It is not
Horizon Scanning Centre January 2013 Apremilast for psoriasis SUMMARY NIHR HSC ID: 2652 This briefing is based on information available at the time of research and a limited literature search. It is not
BJD. Summary. British Journal of Dermatology THERAPEUTICS
 THERAPEUTICS BJD British Journal of Dermatology Efficacy of psoralen plus ultraviolet A therapy vs. biologics in moderate to severe chronic plaque psoriasis: retrospective data analysis of a patient registry
THERAPEUTICS BJD British Journal of Dermatology Efficacy of psoralen plus ultraviolet A therapy vs. biologics in moderate to severe chronic plaque psoriasis: retrospective data analysis of a patient registry
The prevalence of psoriasis among human immunodeficiency
 CLINICAL REVIEW Psoriasis Treatment in HIV-Positive Patients: A Systematic Review of Systemic Immunosuppressive Therapies Mio Nakamura, MD; Michael Abrouk, MD; Benjamin Farahnik, MD; Tian Hao Zhu, MD;
CLINICAL REVIEW Psoriasis Treatment in HIV-Positive Patients: A Systematic Review of Systemic Immunosuppressive Therapies Mio Nakamura, MD; Michael Abrouk, MD; Benjamin Farahnik, MD; Tian Hao Zhu, MD;
Psoriasis. Dr. Pablo de la Cueva Hospital Universitario Infanta Leonor Madrid
 Psoriasis Dr. Pablo de la Cueva Hospital Universitario Infanta Leonor Madrid PSORIASIS Psoriasis News. Topical treatment Calcipotriene/Betamethasone Dipropionate (Cal/BD) foam: In the real-world, Cal/BD
Psoriasis Dr. Pablo de la Cueva Hospital Universitario Infanta Leonor Madrid PSORIASIS Psoriasis News. Topical treatment Calcipotriene/Betamethasone Dipropionate (Cal/BD) foam: In the real-world, Cal/BD
Prior Authorization Conditions for Approval of Humira (adalimumab) Website Form Submit request via: Fax
 Prior Authorization Conditions for Approval of Humira (adalimumab) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Humira (adalimumab) require a prior
Prior Authorization Conditions for Approval of Humira (adalimumab) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Humira (adalimumab) require a prior
Pharmacy Management Drug Policy
 SUBJECT: Cimzia (Certolizumab pegol) - for Ankylosing Spondylitis, Crohn s Disease, Psoriatic Arthritis and Rheumatoid Arthritis POLICY NUMBER: PHARMACY-07 EFFECTIVE DATE: 5/2009 LAST REVIEW DATE: 6/13/2018
SUBJECT: Cimzia (Certolizumab pegol) - for Ankylosing Spondylitis, Crohn s Disease, Psoriatic Arthritis and Rheumatoid Arthritis POLICY NUMBER: PHARMACY-07 EFFECTIVE DATE: 5/2009 LAST REVIEW DATE: 6/13/2018
