Psoriasis. Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement
|
|
|
- Cynthia Baldwin
- 6 years ago
- Views:
Transcription
1 Psoriasis Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement
2 Copyright 2017 by Sea Courses Inc. All rights reserved. No part of this document may be reproduced, copied, stored, or transmitted in any form or by any means graphic, electronic, or mechanical, including photocopying, recording, or information storage and retrieval systems without prior written permission of Sea Courses Inc. except where permitted by law. Sea Courses is not responsible for any speaker or participant s statements, materials, acts or omissions.
3 Learning Objectives Discuss Psoriasis Pathogenesis New treatment approaches
4 Overview and Epidemiology of Psoriasis Chronic, immune-mediated inflammatory disease of the skin Affects 2% of people worldwide An estimated 1 million Canadians have psoriasis Nearly one quarter of psoriasis patients have moderate-to-severe disease
5 Overview and Epidemiology of Psoriasis Genetics important 35-90% have family history Can appear at any age but usually 2 peaks y.o y.o.
6 Infections Triggering Factors Bacterial infections (e.g. Streptococcal pharyngitis) HIV more severe psoriasis Drugs Lithium, IFN, beta-blockers, antimalarials Rapid taper of corticosteroids can induce pustular psoriasis Stress Physical trauma (Koebner phenomenon) Cigarette smoking
7 Comorbidities Observed in Psoriasis Patients Cardiovascular/Metabolic comorbidities CVDs or risk factors increased 1 Myocardial infarction (MI; severe psoriasis ~7-fold) 2 Heart failure (~2-fold) 3 Hypertension (~2-fold) 3 Obesity (~2-fold) 3,4 Metabolic syndrome (~2-fold) 5 Increased CV mortality among inpatients (~1.5-fold) 6 Diabetes (~1.5-fold) 3
8 Chronic Plaque Psoriasis Guttate Psoriasis Pustular Psoriasis Inverse Psoriasis Nail psoriasis Sebopsoriasis Erythrodermic psoriasis Psoriatic Arthritis Clinical Variants
9 Chronic Plaque Psoriasis The most common type of psoriasis (90% of patients) Symmetric distribution of red, scaly plaques on elbows, knees, scalp, hands and feet genital involvement in up to 30% of cases Sharp demarcation line between the plaques and unaffected skin Scale is very thick and characteristic
10
11
12
13
14
15
16
17
18
19 PASI = Psoriasis Area and Severity Index (0-72)
20 DLQI = Dermatology Life Quality Index (0-30)
21 Therapeutic Options in Psoriasis Topical treatments Topical corticosteroids Vitamin D analogues Topical retinoids Coal Tar Topical calcineurins Phototherapy UVB, nuvb PUVA Excimer Laser (308 nm) Traditional systemic treatment Acitretin Methotrexate Cyclosporine Biologics Etanercept Infliximab Adalimumab Ustekinumab Secukinumab Ixekizumab Brodalumab Guselkumab (Apremilast)
22 Topicals Treatments Corticosteroids Most widely used for psoriasis Usually high potency (e.g. clobetasol ungt, clobex spray) (except face and flexural) S/E: atrophy, striae, telangiectasia, hypertichosis, contact dermatitis, HPA axis supression Pulse-dosing prevents tachyphylaxis (3-4 weeks at a time) Vitamin D3 analogues Modulate keratinocyte growth and differentiation and inhibit T lymphocyte activity 2 common preparations: Calcipotriol (Dovonex, Silkis) Calcipotriol + betamethasone diproprionate (Dovobet, Enstilar) Well-tolerated and clinically effective Contraindicated if abnormality in bone or calcium metabolism, renal insufficiency, or pregnancy
23 Other Topicals Treatments Topical Retinoids Normalize epidermal differentiation, potent antiproliferative effect High incidence of irritation at sites of application Not used much these days Coal Tar Liquor carbonis detergens (LCD) is a distilled product Sometimes used for scalp psoriasis (lotions or shampoos) Unpleasant smell Contraindicated in pregnant or lactating women Calcineurin Inhibitors Sometimes used for facial and flexural psoriasis E.g. tacrolimus (protopic 0.1% ungt)
24 Phototherapy Best for guttate or sebopsoriasis PUVA>nUVB>UVB Most offices use nuvb (311 nm) S/E with UVB Short-term: erythema, xerosis Long-term: photoaging and carcinogenesis (nuvb is less carcinogenic) S/E with PUVA Short-term: redness, swelling, occasional blister Long-term: cataracts (need eye protection), dark lentigenes, photocarcinogenesis
25 Oral Methotrexate Indications Proposed mechanism of action Dosing Efficacy Chronic plaque psoriasis, pustular psoriasis, erythrodermic psoriasis, psoriatic arthritis, severe nail psoriasis or poor response to topicals and phototherapy Inhibits DNA synthesis and cell replication; also has specific T-cell suppressive activities 7.5 to 25 mg per week (po or IM); usually combine with folic acid (5 mg po daily) > 60% PASI 75 response (16 weeks) S/E Monitoring required Minor: N/V, Abdominal pain, oral erosions Major: myelosuppression, hepatotoxicity, pneumonitis, pulmonary fibrosis, fetal death or abnormalities Pregnancy X Baseline: CBCD, Liver Function tests (AST, ALT), Creatinine, Hep B and C serology, HIV (if risk), CXR if hx of pulmonary problems, Pregnancy test F/U: Repeat CBCD, LFT, Cr q2 weeks for the first month, then monthly +/- Liver biopsy at cumulitive dose of 1.5 g 1
26 Oral Cyclosporine Indications Proposed mechanism of action Dosing Severe Psoriasis Recalcitrant, treatment-resistant psoriasis Calcineurin inhibitor that interferes with early events in T-cell activation, preventing the production of cytokines (IL-2) and activated T cells 2.5 to 5 mg/kg/day Efficacy > 60% PASI 75 response (16 weeks) S/E Monitoring required Self-limiting: paresthesia, tremor, headache, nausea Other: hypertension, renal dysfunction, hypertrichosis, gingival hyperplasia Labs: hyperkalemia, hyperlipidemia, hyperuricemia, hypomangesemia, Pregnancy C Baseline: CBCD, Liver Function tests (AST, ALT), Creatinine, Lipids, Lytes, Uric acid, Mg, Pregnancy test, Blood pressure F/U: repeat q2 weeks for the first month, then monthly 1
27 Oral Acitretin Indications Severe Psoriasis Erythrodermic or pustular psoriasis Recalcitrant, treatment-resistant psoriasis Proposed mechanism of action Binds to retinoic acid receptor and may contribute by normalizing keratinization and proliferation of epidermis Dosing mg daily Efficacy > 60% PASI 75 response S/E Monitoring required Common: mucocutaneous dryness, elevation of triglycerides, sometimes alopecia Rare: skeletal hyperostosis (DISH), transaminitis, fetal abnormalities, Pregnancy X Avoid use of alcohol (re-esterification to etretinate) Baseline: CBCD, Liver Function tests (AST, ALT), Creatinine, Lipids, Pregnancy test F/U: repeat q2 weeks for the first month, then monthly 1
28 Oral Apremilast (Otezla) Indications Moderate to Severe Psoriasis Psoriatic Arthritis Proposed mechanism of action Apremilast inhibits PDE4, which results in increased intracellular camp levels in inflammatory cells Dosing 30 mg twice daily Efficacy > 30% PASI 75 response S/E Diarrhea, Nausea, Vomiting, Abdominal Pain Headache, URTI Depression Monitoring required None 1
29 Structure and Function of Biologics Etanercept (Enbrel) Infliximab (Remicade) Adalimumab (Humira) Ustekinumab (Stelara) Secukinumab (Cosentyx) Ixekizumab (Taltz) Guselkumab (Tremfya) Human fusion protein Chimeric monoclona l antibody Fully human monoclonal antibody Fully human monoclonal antibody Fully human monoclonal antibody Humanized monoclonal antibody Human monoclonal antibody Blocks TNF-α activity Blocks TNF-α activity Blocks TNF-α activity Blocks IL-12 and IL-23 Inhibits IL- 17A interaction with receptor Inhibits IL- 17A interaction with receptor Inhibits IL-23 (binds p19 subunit) SQ IV infusion SQ SQ SQ SQ SQ 50mg BIWx 12 weeks, then 25mg BIW 5mg/kg infusion at week 0, 2, 6 and q 8 weeks 80mg 1 st week, 40 mg 2 nd week, then 40 mg eow 45 or 90 mg at week 0, 4 and then q 12 weeks 300 mg SC at weeks 0, 1, 2, 3, and 4, then monthly 160 mg at week 0, then 80 mg at weeks 2, 4, 6, 8,10, 12 then q 4 weeks 100mg at week 0, 4 and then q8 weeks
30 Canadian Psoriasis Expert Panel Consensus Biologic Agents Etanercept, Infliximab, Adalimumab, Ustekinumab, Secukinumab, Ixekizumab, Guselkumab
31 PEARLs Psoriasis Novel biologic therapies Systemic disease No longer just a skin condition
Psoriasis management. A/Prof Amanda Oakley Dermatologist, Waikato
 Psoriasis management A/Prof Amanda Oakley Dermatologist, Waikato AbbVie Breakfast Session, 14 June 2014 Disclosure This breakfast session is sponsored by Abbvie Autoimmune skin disorders Psoriasis Eczema
Psoriasis management A/Prof Amanda Oakley Dermatologist, Waikato AbbVie Breakfast Session, 14 June 2014 Disclosure This breakfast session is sponsored by Abbvie Autoimmune skin disorders Psoriasis Eczema
Psoriasis. What is Psoriasis? What causes psoriasis? Medical Topics Psoriasis
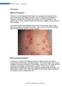 1 Psoriasis What is Psoriasis? Psoriasis is a long standing inflammatory non-contagious skin disease which waxes and wanes with triggering factors. There is a genetic predisposition in psoriasis. Internationally,
1 Psoriasis What is Psoriasis? Psoriasis is a long standing inflammatory non-contagious skin disease which waxes and wanes with triggering factors. There is a genetic predisposition in psoriasis. Internationally,
Psoriasis. and Biologic Treatments. Choose the best treatment to regain your quality of life. Edition 4
 Psoriasis and Biologic Treatments Choose the best treatment to regain your quality of life. Edition 4 PSORIASIS, What is this disease? Psoriasis is a common disease, that affects up to 4% of the population.
Psoriasis and Biologic Treatments Choose the best treatment to regain your quality of life. Edition 4 PSORIASIS, What is this disease? Psoriasis is a common disease, that affects up to 4% of the population.
PSORIASIS PSORIASIS. Anatomic sites FACTS ABOUT PROSIASIS PSORIASIS 11/16/2017 STIGMATA OF PSORIASIS
 PSORIASIS Dr. Sami Alsuwaidan President, Saudi Society of Dermatology & Dermatologic Surgery Associate Professor and Consultant Department of Dermatology, College of Medicine King Saud University PSORIASIS
PSORIASIS Dr. Sami Alsuwaidan President, Saudi Society of Dermatology & Dermatologic Surgery Associate Professor and Consultant Department of Dermatology, College of Medicine King Saud University PSORIASIS
Psoriasis. and Biologic Treatments. Choose the best treatment to regain your quality of life.
 Psoriasis and Biologic Treatments Choose the best treatment to regain your quality of life. PSORIASIS, What is this disease? Psoriasis is a common disease, that affects up to 4% of the population. It usually
Psoriasis and Biologic Treatments Choose the best treatment to regain your quality of life. PSORIASIS, What is this disease? Psoriasis is a common disease, that affects up to 4% of the population. It usually
KEY MESSAGES. Psoriasis patients are more prone to cardiovascular diseases, stroke, lymphoma and non-melanoma skin cancers, and increased mortality.
 KEY MESSAGES Psoriasis is a genetically determined, systemic immune-mediated chronic inflammatory disease that affects primarily the skin and joints. Psoriasis Vulgaris is characterised by well-demarcated
KEY MESSAGES Psoriasis is a genetically determined, systemic immune-mediated chronic inflammatory disease that affects primarily the skin and joints. Psoriasis Vulgaris is characterised by well-demarcated
It is estimated that about 26,000 new cases of
 Focus on CME at Dalhousie University Set On Soothing Psoriasis A. H. Murray, MD, FRCP(C) Presented at the 76th Annual Dalhousie Refresher Course It is estimated that about 26,000 new cases of psoriasis
Focus on CME at Dalhousie University Set On Soothing Psoriasis A. H. Murray, MD, FRCP(C) Presented at the 76th Annual Dalhousie Refresher Course It is estimated that about 26,000 new cases of psoriasis
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Psoriasis: the management of psoriasis 1.1 Short title Psoriasis 2 The remit The Department of Health has asked NICE: 'to produce
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Psoriasis: the management of psoriasis 1.1 Short title Psoriasis 2 The remit The Department of Health has asked NICE: 'to produce
Psoriasis: Causes, Symptoms, And Treatment
 Psoriasis: Causes, Symptoms, And Treatment We all know that a healthy immune system is good. But, do you know that an overactive immune system can cause certain conditions like Psoriasis? Read on to find
Psoriasis: Causes, Symptoms, And Treatment We all know that a healthy immune system is good. But, do you know that an overactive immune system can cause certain conditions like Psoriasis? Read on to find
The role of the practice nurse in managing psoriasis in primary care
 The role of the practice nurse in managing psoriasis in primary care Item type Authors Publisher Journal Article Buckley, David Nursing in General Practice Nursing in general practice Downloaded 16-Sep-2016
The role of the practice nurse in managing psoriasis in primary care Item type Authors Publisher Journal Article Buckley, David Nursing in General Practice Nursing in general practice Downloaded 16-Sep-2016
Psoriasis. Overview. Epidemiology. Epidemiology 08/08/2015. Dr Nigel Burrows Consultant Dermatologist Addenbrooke s Hospital
 Overview Psoriasis 1. Epidemiology of psoriasis 2. Histology Dr Nigel Burrows Consultant Dermatologist Addenbrooke s Hospital Aug 2015 3. Types of psoriasis 4. Assessing severity 5. Treatments Topical
Overview Psoriasis 1. Epidemiology of psoriasis 2. Histology Dr Nigel Burrows Consultant Dermatologist Addenbrooke s Hospital Aug 2015 3. Types of psoriasis 4. Assessing severity 5. Treatments Topical
Predicting the Response to Phototherapy for Psoriasis Patients
 A*STAR-NHG-NTU Skin Research Grant Joint Workshop 17 October 2015 Predicting the Response to Phototherapy for Psoriasis Patients Is it possible? Dr Eugene Tan Consultant Dermatologist National Skin Centre
A*STAR-NHG-NTU Skin Research Grant Joint Workshop 17 October 2015 Predicting the Response to Phototherapy for Psoriasis Patients Is it possible? Dr Eugene Tan Consultant Dermatologist National Skin Centre
Risankizumab (by subcutaneous injection) for moderate to severe chronic plaque psoriasis
 NIHR Innovation Observatory Evidence Briefing: November 2017 Risankizumab (by subcutaneous injection) for moderate to severe chronic plaque psoriasis NIHRIO (HSRIC) ID: 9708 NICE ID: 9191 LAY SUMMARY Plaque
NIHR Innovation Observatory Evidence Briefing: November 2017 Risankizumab (by subcutaneous injection) for moderate to severe chronic plaque psoriasis NIHRIO (HSRIC) ID: 9708 NICE ID: 9191 LAY SUMMARY Plaque
Psoriasis. Jessica Kaffenberger, M.D. Assistant Professor of Dermatology Division of Dermatology The Ohio State University Wexner Medical Center
 Psoriasis Jessica Kaffenberger, M.D. Assistant Professor of Dermatology Division of Dermatology The Ohio State University Wexner Medical Center Learning objectives Recognize the different types of psoriasis
Psoriasis Jessica Kaffenberger, M.D. Assistant Professor of Dermatology Division of Dermatology The Ohio State University Wexner Medical Center Learning objectives Recognize the different types of psoriasis
Psoriasis. Causes of Psoriasis
 Psoriasis Psoriasis is a common, chronic, relapsing/remitting, immune-mediated systemic disease characterized by skin lesions including red, scaly patches, papules, and plaques, which usually itch. The
Psoriasis Psoriasis is a common, chronic, relapsing/remitting, immune-mediated systemic disease characterized by skin lesions including red, scaly patches, papules, and plaques, which usually itch. The
Background AN UPDATED LOOK AT TREATMENTS FOR PLAQUE PSORIASIS JULY 2018 PLAQUE PSORIASIS TARGETED IMMUNOMODULATORS AS A TREATMENT OPTION
 JULY 2018 Background PLAQUE PSORIASIS Plaque psoriasis is a common disease affecting 3% of the US population that causes itchy, red, scaly, raised lesions on the skin, most commonly on the elbows, knees,
JULY 2018 Background PLAQUE PSORIASIS Plaque psoriasis is a common disease affecting 3% of the US population that causes itchy, red, scaly, raised lesions on the skin, most commonly on the elbows, knees,
Psoriasis in Jordan: a single center experience
 Psoriasis in Jordan: a single center experience Shefaa Almashagbeh MD *, Deifallah Alsharari MD *, Hayat Khasawneh MD *, Diana Aljammal * MD, Hamzeh Al-housamieh MD* ABSTRACT Objectives: To evaluate the
Psoriasis in Jordan: a single center experience Shefaa Almashagbeh MD *, Deifallah Alsharari MD *, Hayat Khasawneh MD *, Diana Aljammal * MD, Hamzeh Al-housamieh MD* ABSTRACT Objectives: To evaluate the
Update on systemic therapies and emerging treatments How do I choose a systemic agent?
 Update on systemic therapies and emerging treatments How do I choose a systemic agent? Amy S. Paller, M.D. Walter J. Hamlin Professor and Chair of Dermatology Professor of Pediatrics Northwestern University
Update on systemic therapies and emerging treatments How do I choose a systemic agent? Amy S. Paller, M.D. Walter J. Hamlin Professor and Chair of Dermatology Professor of Pediatrics Northwestern University
A Patient s Guide to. Treatments for Psoriatic Arthritis
 A Patient s Guide to Treatments for Psoriatic Arthritis Who should read this guide? This guide is aimed at people with psoriatic arthritis (abbreviated as PsA), or those who care for a person with this
A Patient s Guide to Treatments for Psoriatic Arthritis Who should read this guide? This guide is aimed at people with psoriatic arthritis (abbreviated as PsA), or those who care for a person with this
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Proposed Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Dimethyl fumarate for treating moderate to severe Draft scope (pre-referral) Draft remit/appraisal objective To appraise
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Dimethyl fumarate for treating moderate to severe Draft scope (pre-referral) Draft remit/appraisal objective To appraise
Topical Calcipotriol Algorithm
 Topical Calcipotriol Algorithm Is this patient an adult previously diagnosed with psoriasis by a doctor? Do the skin patches look the same as those diagnosed as psoriasis? Is this psoriasis covering an
Topical Calcipotriol Algorithm Is this patient an adult previously diagnosed with psoriasis by a doctor? Do the skin patches look the same as those diagnosed as psoriasis? Is this psoriasis covering an
Horizon Scanning Centre March Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798
 Horizon Scanning Centre March 2015 Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798 This briefing is based on information available at the time of research and a limited
Horizon Scanning Centre March 2015 Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798 This briefing is based on information available at the time of research and a limited
Current treatment options in the management of psoriasis
 Drug review Current treatment options in the management of psoriasis Eleanor Higgins MB BCh, BAO, MRCPI and Trevor Markham MD, BAO, MRCPI SPL is a common chronic condition and treatment choice primarily
Drug review Current treatment options in the management of psoriasis Eleanor Higgins MB BCh, BAO, MRCPI and Trevor Markham MD, BAO, MRCPI SPL is a common chronic condition and treatment choice primarily
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Otezla (apremilast) Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Otezla (apremilast) Prime Therapeutics will review Prior Authorization requests Prior
Otezla (apremilast) Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Otezla (apremilast) Prime Therapeutics will review Prior Authorization requests Prior
Difference Between Seborrheic Dermatitis and Psoriasis
 Difference Between Seborrheic Dermatitis and Psoriasis www.differencebetween.com Key Difference Seborrheic Dermatitis vs Psoriasis Dermatological conditions are perhaps the most worrisome diseases in the
Difference Between Seborrheic Dermatitis and Psoriasis www.differencebetween.com Key Difference Seborrheic Dermatitis vs Psoriasis Dermatological conditions are perhaps the most worrisome diseases in the
Biologics for Autoimmune Diseases
 Biologics for Autoimmune Diseases Goal(s): Restrict use of biologics to OHP funded conditions and according to OHP guidelines for use. Promote use that is consistent with national clinical practice guidelines
Biologics for Autoimmune Diseases Goal(s): Restrict use of biologics to OHP funded conditions and according to OHP guidelines for use. Promote use that is consistent with national clinical practice guidelines
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 2104-4 Program Prior Authorization/Medical Necessity Medication Taltz (ixekizumab) P&T Approval Date 8/2016, 5/2017, 2/2018 Effective
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 2104-4 Program Prior Authorization/Medical Necessity Medication Taltz (ixekizumab) P&T Approval Date 8/2016, 5/2017, 2/2018 Effective
DERMATOLOGY. The Changing Landscape of Psoriasis Treatment ABSTRACT
 WHITE PAPER PRESENTED BY PREMIER RESEARCH The Changing Landscape of Psoriasis Treatment ABSTRACT Over the past two decades, biologic therapies have revolutionized the treatment of psoriasis, with more
WHITE PAPER PRESENTED BY PREMIER RESEARCH The Changing Landscape of Psoriasis Treatment ABSTRACT Over the past two decades, biologic therapies have revolutionized the treatment of psoriasis, with more
TREATMENT OPTIONS FOR PSORIASIS. Sandra Hanlon Dermatology Senior Charge Nurse NHS Ayrshire and Arran 07/03/17
 TREATMENT OPTIONS FOR PSORIASIS Sandra Hanlon Dermatology Senior Charge Nurse NHS Ayrshire and Arran 07/03/17 PSORIASIS A chronic, non-infectious inflammatory skin condition that has no cure Characterised
TREATMENT OPTIONS FOR PSORIASIS Sandra Hanlon Dermatology Senior Charge Nurse NHS Ayrshire and Arran 07/03/17 PSORIASIS A chronic, non-infectious inflammatory skin condition that has no cure Characterised
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Psoriatic Arthritis- Secondary Care
 Psoriatic Arthritis- Secondary Care Our Psoriatic Arthritis: First Line Treatments information sheet gives information on the treatments that can be prescribed by a GP, or that might be prescribed if the
Psoriatic Arthritis- Secondary Care Our Psoriatic Arthritis: First Line Treatments information sheet gives information on the treatments that can be prescribed by a GP, or that might be prescribed if the
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Outcome High Priority
 Quality ID #410: Psoriasis: Clinical Response to Systemic Medications National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes Meaningful Measure Area: Management of Chronic
Quality ID #410: Psoriasis: Clinical Response to Systemic Medications National Quality Strategy Domain: Person and Caregiver-Centered Experience and Outcomes Meaningful Measure Area: Management of Chronic
ACTEMRA (tocilizumab)
 Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 1. Active Polyarticular Juvenile Idiopathic Arthritis (PJIA) b. Patient has an intolerance or has experienced
Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 1. Active Polyarticular Juvenile Idiopathic Arthritis (PJIA) b. Patient has an intolerance or has experienced
Psoriasis is a chronic, inflammatory, Prescribing in children
 Psoriasis in children: current approaches to management Laura Proudfoot BSc, MRCP, Elisabeth Higgins MA, FRCP and Judy Davids RGN Our series Prescribing in children gives practical advice for successful
Psoriasis in children: current approaches to management Laura Proudfoot BSc, MRCP, Elisabeth Higgins MA, FRCP and Judy Davids RGN Our series Prescribing in children gives practical advice for successful
CIMZIA (certolizumab pegol)
 Pre - PA Allowance None Prior-Approval Requirements Age Diagnoses 18 years of age or older Patient must have ONE of the following: 1. Moderate to severe Crohn s Disease (CD) a. Inadequate response, intolerance
Pre - PA Allowance None Prior-Approval Requirements Age Diagnoses 18 years of age or older Patient must have ONE of the following: 1. Moderate to severe Crohn s Disease (CD) a. Inadequate response, intolerance
Stelara. Stelara (ustekinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 9 Last Review Date: September 20, 2018 Stelara Description Stelara
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 9 Last Review Date: September 20, 2018 Stelara Description Stelara
50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate).
 DUPISOR Composition Gel 50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate). Action Calcipotriol is a non-steroidal antipsoriatic agent, derived from vitamin D. Calcipotriol
DUPISOR Composition Gel 50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate). Action Calcipotriol is a non-steroidal antipsoriatic agent, derived from vitamin D. Calcipotriol
4/10/18. Faculty. Disclosures. Confronting Psoriatic Disease: Putting New Tools to Work
 Confronting Psoriatic Disease: Putting New Tools to Work Faculty Brad P. Glick, DO, MPH, FAOCD Glick Skin Institute Skin and Cancer Associates Program Director Dermatology Residency Larkin Hospital - Palm
Confronting Psoriatic Disease: Putting New Tools to Work Faculty Brad P. Glick, DO, MPH, FAOCD Glick Skin Institute Skin and Cancer Associates Program Director Dermatology Residency Larkin Hospital - Palm
Pharmacy Management Drug Policy
 SUBJECT: Inflammatory Conditions Clinical Review Prior Authorization (CRPA) Rx and Medical Drugs POLICY NUMBER: PHARMACY-73 EFFECTIVE DATE: 01/01/2018 LAST REVIEW DATE: 06/11/2018 If the member s subscriber
SUBJECT: Inflammatory Conditions Clinical Review Prior Authorization (CRPA) Rx and Medical Drugs POLICY NUMBER: PHARMACY-73 EFFECTIVE DATE: 01/01/2018 LAST REVIEW DATE: 06/11/2018 If the member s subscriber
The New and Emerging Agents: Dermatology
 Psoriasis Treatment 2013: What is New and on the Horizon? Neil J Korman, MD, PhD Professor of Dermatology University Hospitals Case Medical Center Clinical Director Murdough Family Center for Psoriasis
Psoriasis Treatment 2013: What is New and on the Horizon? Neil J Korman, MD, PhD Professor of Dermatology University Hospitals Case Medical Center Clinical Director Murdough Family Center for Psoriasis
Otezla. Otezla (apremilast) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Otezla Page: 1 of 5 Last Review Date: March 16, 2018 Otezla Description Otezla (apremilast) Background
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Otezla Page: 1 of 5 Last Review Date: March 16, 2018 Otezla Description Otezla (apremilast) Background
chemotherapeutic agents in
 Use of biologics and chemotherapeutic agents in cutaneous emergencies: Focus on lifethreatening forms of psoriasis Alice Bendix Gottlieb MD, PhD Professor of Dermatology New York Medical College Metropolitan
Use of biologics and chemotherapeutic agents in cutaneous emergencies: Focus on lifethreatening forms of psoriasis Alice Bendix Gottlieb MD, PhD Professor of Dermatology New York Medical College Metropolitan
An otherwise healthy 12-year-old
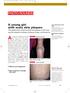 1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
Horizon Scanning Centre March Ixekizumab for moderate to severe chronic plaque psoriasis SUMMARY NIHR HSC ID: 5209
 Horizon Scanning Centre March 2015 Ixekizumab for moderate to severe chronic plaque psoriasis SUMMARY NIHR HSC ID: 5209 This briefing is based on information available at the time of research and a limited
Horizon Scanning Centre March 2015 Ixekizumab for moderate to severe chronic plaque psoriasis SUMMARY NIHR HSC ID: 5209 This briefing is based on information available at the time of research and a limited
Targeted Immunomodulators for the Treatment of Moderate-to- Severe Plaque Psoriasis: Effectiveness and Value
 Targeted Immunomodulators for the Treatment of Moderate-to- Severe Plaque Psoriasis: Effectiveness and Value Condition Update Final Background and Scope January 9, 2018 Background Psoriasis is a common
Targeted Immunomodulators for the Treatment of Moderate-to- Severe Plaque Psoriasis: Effectiveness and Value Condition Update Final Background and Scope January 9, 2018 Background Psoriasis is a common
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 13 May 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
Psoriatic Arthritis- Second Line Treatments
 Psoriatic Arthritis- Second Line Treatments Second line treatments for Psoriatic Arthritis (PsA) are usually prescribed by a Rheumatologist, Dermatologist, or in a combined clinic where both the Dermatologist
Psoriatic Arthritis- Second Line Treatments Second line treatments for Psoriatic Arthritis (PsA) are usually prescribed by a Rheumatologist, Dermatologist, or in a combined clinic where both the Dermatologist
Elements of Successful PBS Applications. Barbara Radulski RN. Copyright
 Elements of Successful PBS Applications Barbara Radulski RN PBS Requirements April 1 2006 THE RULES PBS Requirements 18 years and over Psoriasis x 6 months Failed to achieve an adequate response to 3 systemic
Elements of Successful PBS Applications Barbara Radulski RN PBS Requirements April 1 2006 THE RULES PBS Requirements 18 years and over Psoriasis x 6 months Failed to achieve an adequate response to 3 systemic
Horizon Scanning Centre May Brodalumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 5524
 Horizon Scanning Centre May 2014 Brodalumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 5524 This briefing is based on information available at the time of research and a limited literature
Horizon Scanning Centre May 2014 Brodalumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 5524 This briefing is based on information available at the time of research and a limited literature
Psoriasis: A Therapeutic Update. Presenter: Christine Moussa, PGY-4 Program Director: Stephen Kessler, D.O. Alta Dermatology/LECOM
 Psoriasis: A Therapeutic Update Presenter: Christine Moussa, PGY-4 Program Director: Stephen Kessler, D.O. Alta Dermatology/LECOM Disclosures I have no relevant financial or nonfinancial relationships
Psoriasis: A Therapeutic Update Presenter: Christine Moussa, PGY-4 Program Director: Stephen Kessler, D.O. Alta Dermatology/LECOM Disclosures I have no relevant financial or nonfinancial relationships
Clinical Trial Report Synopsis
 Clinical Trial Report Synopsis A phase 2a, proof of concept trial, testing twice daily application of LEO 124249 ointment 30 mg/g in the treatment of mild to moderate inverse psoriasis Design of trial:
Clinical Trial Report Synopsis A phase 2a, proof of concept trial, testing twice daily application of LEO 124249 ointment 30 mg/g in the treatment of mild to moderate inverse psoriasis Design of trial:
Psoriasis: Therapeutic goals
 Psoriasis: Therapeutic goals I want to die 50 45 impetiginization infliximab 600 40 35 30 400 25 20 15 200 10 5 0 22-ene 21-feb 23-mar 22-abr 22- may Efalizumab 6 doses: flare + REBOUND CSA 3 21-jun 21-jul
Psoriasis: Therapeutic goals I want to die 50 45 impetiginization infliximab 600 40 35 30 400 25 20 15 200 10 5 0 22-ene 21-feb 23-mar 22-abr 22- may Efalizumab 6 doses: flare + REBOUND CSA 3 21-jun 21-jul
Psoriasiform Dermatitis in Children: Calling in the Troops
 Psoriasiform Dermatitis in Children: Calling in the Troops Markus Boos, MD PhD Attending Physician, Dermatology Seattle Children s Hospital Assistant Professor of Pediatrics, University of Washington School
Psoriasiform Dermatitis in Children: Calling in the Troops Markus Boos, MD PhD Attending Physician, Dermatology Seattle Children s Hospital Assistant Professor of Pediatrics, University of Washington School
PSORIASIS BEST PRACTICE IN MANAGEMENT
 PSORIASIS BEST PRACTICE IN MANAGEMENT Objectives Discuss pathology of psoriasis Review types of psoriasis Review triggers and factors affecting disease severity Common comorbidity review Review first and
PSORIASIS BEST PRACTICE IN MANAGEMENT Objectives Discuss pathology of psoriasis Review types of psoriasis Review triggers and factors affecting disease severity Common comorbidity review Review first and
INFLIXIMAB Remicade (infliximab), Inflectra (infliximab-dyyb), Ixifi* (infliximabqbtx), Renflexis (infliximab-abda)
 Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 6 years of age or older 1. Moderate to severe Crohn s disease (CD) a. Patient has fistulizing disease
Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 6 years of age or older 1. Moderate to severe Crohn s disease (CD) a. Patient has fistulizing disease
Etanercept: a new option in paediatric plaque psoriasis
 : a new option in paediatric plaque psoriasis Steve Chaplin MSc, MRPharmS, Medical Writer, Dr David Atherton MA, MB, BChir, FRCP, Honorary Consultant in Paediatric Dermatology, Great Ormond Street Hospital
: a new option in paediatric plaque psoriasis Steve Chaplin MSc, MRPharmS, Medical Writer, Dr David Atherton MA, MB, BChir, FRCP, Honorary Consultant in Paediatric Dermatology, Great Ormond Street Hospital
Single Technology Appraisal (STA) Tildrakizumab for treating moderate to severe plaque psoriasis
 Single Technology Appraisal (STA) Tildrakizumab for treating moderate to severe plaque psoriasis Response to consultee and commentator comments on the draft remit and draft scope (pre-referral) Please
Single Technology Appraisal (STA) Tildrakizumab for treating moderate to severe plaque psoriasis Response to consultee and commentator comments on the draft remit and draft scope (pre-referral) Please
Andrei Metelitsa, MD, FRCPC, FAAD Co-Director, Institute for Skin Advancement Clinical Associate Professor, Dermatology University of Calgary, Canada
 Andrei Metelitsa, MD, FRCPC, FAAD Co-Director, Institute for Skin Advancement Clinical Associate Professor, Dermatology University of Calgary, Canada Copyright 2017 by Sea Courses Inc. All rights reserved.
Andrei Metelitsa, MD, FRCPC, FAAD Co-Director, Institute for Skin Advancement Clinical Associate Professor, Dermatology University of Calgary, Canada Copyright 2017 by Sea Courses Inc. All rights reserved.
Clinical Policy: Ustekinumab (Stelara) Reference Number: ERX.SPMN.167
 Clinical Policy: (Stelara) Reference Number: ERX.SPMN.167 Effective Date: 10/16 Last Review Date: 12/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Stelara) Reference Number: ERX.SPMN.167 Effective Date: 10/16 Last Review Date: 12/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
What you need to know about PSORIASIS. Psoriasis
 What you need to know about PSORIASIS Ps Psoriasis The Irish Skin Foundation is a national charity with a mission to improve quality of life for people with skin disorders, promote skin health and the
What you need to know about PSORIASIS Ps Psoriasis The Irish Skin Foundation is a national charity with a mission to improve quality of life for people with skin disorders, promote skin health and the
Pharmacy Accreditation
 EASING THE PATIENT BURDEN OF PSORIASIS AND PSORIATIC ARTHRITIS: THE ROLE OF THE SPECIALTY PHARMACIST Claire Lee, PharmD, CSP, CPHQ Clinical Quality Improvement Supervisor Diplomat Flint, Michigan Pharmacy
EASING THE PATIENT BURDEN OF PSORIASIS AND PSORIATIC ARTHRITIS: THE ROLE OF THE SPECIALTY PHARMACIST Claire Lee, PharmD, CSP, CPHQ Clinical Quality Improvement Supervisor Diplomat Flint, Michigan Pharmacy
Faculty David M. Pariser, MD Professor Eastern Virginia Medical School Norfolk, VA
 Disclaimer This slide deck in its original and unaltered format is for educational purposes and is current as of May 2015. The content and views presented in this educational activity are those of the
Disclaimer This slide deck in its original and unaltered format is for educational purposes and is current as of May 2015. The content and views presented in this educational activity are those of the
1 P a g e. Systemic Juvenile Idiopathic Arthritis (SJIA) (1.3) Patients 2 years of age and older with active systemic juvenile idiopathic arthritis.
 LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
COMMON SKIN CONDITIONS IN PRIMARY CARE. Ibrahim M. Zayneh, MD Dermatology Private Practice, Portsmouth, Ohio
 COMMON SKIN CONDITIONS IN PRIMARY CARE Ibrahim M. Zayneh, MD Dermatology Private Practice, Portsmouth, Ohio DISCLOSURE The Speaker and members of the planning committee do not have a conflict of interest
COMMON SKIN CONDITIONS IN PRIMARY CARE Ibrahim M. Zayneh, MD Dermatology Private Practice, Portsmouth, Ohio DISCLOSURE The Speaker and members of the planning committee do not have a conflict of interest
Psoriasis is an inflammatory condition of the skin that
 Mayo Clin Proc, September 2001, Vol 76 Topical Therapies for Localized Psoriasis 943 Concise Review for Clinicians Concise Review for Clinicians Topical Therapies for Localized Psoriasis PATRICIA M. WITMAN,
Mayo Clin Proc, September 2001, Vol 76 Topical Therapies for Localized Psoriasis 943 Concise Review for Clinicians Concise Review for Clinicians Topical Therapies for Localized Psoriasis PATRICIA M. WITMAN,
TRANSPARENCY COMMITTEE OPINION. 26 April 2006
 TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
Health Related Quality of Life: The Impact of Psoriasis When Designing Tailored Treatment Plans
 Disclaimer This slide deck in its original and unaltered format is for educational purposes and is current as of October 2015. The content and views presented in this educational activity are those of
Disclaimer This slide deck in its original and unaltered format is for educational purposes and is current as of October 2015. The content and views presented in this educational activity are those of
PSORIASIS DRUGS IN EUROPE - MARKET ACCESS DECISIONS IN COMPARISION BASED ON THE PRISMACCESS DATABASE
 PSORIASIS DRUGS IN EUROPE - MARKET ACCESS DECISIONS IN COMPARISION BASED ON THE PRISMACCESS DATABASE Lutz Vollmer 1,3, Daniel Dröschel 1,4, Bruno de Paz 2, Stefan Walzer 1,5 1 MArS Market Access & Pricing
PSORIASIS DRUGS IN EUROPE - MARKET ACCESS DECISIONS IN COMPARISION BASED ON THE PRISMACCESS DATABASE Lutz Vollmer 1,3, Daniel Dröschel 1,4, Bruno de Paz 2, Stefan Walzer 1,5 1 MArS Market Access & Pricing
Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV])
![Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV]) Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV])](/thumbs/89/99382632.jpg) Origination: 09/27/07 Revised: 08/2/17 Annual Review: 11/2/17 Purpose: To provide Phototherapy and Photochemotherapy Treatment (PUVA and UBV) guidelines for the Medical Department staff to reference when
Origination: 09/27/07 Revised: 08/2/17 Annual Review: 11/2/17 Purpose: To provide Phototherapy and Photochemotherapy Treatment (PUVA and UBV) guidelines for the Medical Department staff to reference when
Cosentyx. Cosentyx (secukinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.11 Subject: Cosentyx Page: 1 of 7 Last Review Date: September 20, 2018 Cosentyx Description Cosentyx
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.11 Subject: Cosentyx Page: 1 of 7 Last Review Date: September 20, 2018 Cosentyx Description Cosentyx
Stelara. Stelara (ustekinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 6 Last Review Date: December 8, 2017 Stelara Description Stelara (ustekinumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 6 Last Review Date: December 8, 2017 Stelara Description Stelara (ustekinumab)
NB Drug Plans Formulary Update
 Bulletin # 995 March 27, 2019 NB Drug Plans Formulary Update This update to the New Brunswick Drug Plans Formulary is effective March 27, 2019. Included in this bulletin: Special Authorization Benefit
Bulletin # 995 March 27, 2019 NB Drug Plans Formulary Update This update to the New Brunswick Drug Plans Formulary is effective March 27, 2019. Included in this bulletin: Special Authorization Benefit
Subject: Guselkumab (Tremfya ) Injection
 09-J2000-87 Original Effective Date: 09/15/17 Reviewed: 09/12/18 Revised: 01/01/19 Subject: Guselkumab (Tremfya ) Injection THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
09-J2000-87 Original Effective Date: 09/15/17 Reviewed: 09/12/18 Revised: 01/01/19 Subject: Guselkumab (Tremfya ) Injection THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
ETANERCEPT Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Psoriasis: A Cutaneous or Systemic Disease
 Psoriasis: A Cutaneous or Systemic Disease Ian D.R. Landells, MD, FRCPC Clinical Chief Dermatology, Eastern Health Clinical Assistant Professor Disciplines of Medicine and Paediatrics Faculty of Medicine
Psoriasis: A Cutaneous or Systemic Disease Ian D.R. Landells, MD, FRCPC Clinical Chief Dermatology, Eastern Health Clinical Assistant Professor Disciplines of Medicine and Paediatrics Faculty of Medicine
Regulatory Status FDA-approved indication: Orencia is a selective T cell co-stimulation modulator indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Orencia Page: 1 of 9 Last Review Date: September 20, 2018 Orencia Description Orencia (abatacept)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Orencia Page: 1 of 9 Last Review Date: September 20, 2018 Orencia Description Orencia (abatacept)
STELARA (ustekinumab)
 STELARA (ustekinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
STELARA (ustekinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Dermatology elective for yr. 5. Natta Rajatanavin, MD. Div. of dermatology Dep. Of Medicine, Ramathibodi Hospital Mahidol University 23 rd Feb 2015
 Dermatology elective for yr. 5 Natta Rajatanavin, MD. Div. of dermatology Dep. Of Medicine, Ramathibodi Hospital Mahidol University 23 rd Feb 2015 How to diagnosis and manage eczema and psoriasis. Objectives
Dermatology elective for yr. 5 Natta Rajatanavin, MD. Div. of dermatology Dep. Of Medicine, Ramathibodi Hospital Mahidol University 23 rd Feb 2015 How to diagnosis and manage eczema and psoriasis. Objectives
COST-EFFECTIVENESS OF TREATMENT FOR MODERATE-TO-SEVERE PSORIASIS
 COST-EFFECTIVENESS OF TREATMENT FOR MODERATE-TO-SEVERE PSORIASIS Cheryl S. Hankin PhD 1 ; Steven R. Feldman, MD, PhD 2 ; Daniel Pearce, MD 2 1 BioMedEcon, San Jose, CA, USA; 2 Wake Forest University School
COST-EFFECTIVENESS OF TREATMENT FOR MODERATE-TO-SEVERE PSORIASIS Cheryl S. Hankin PhD 1 ; Steven R. Feldman, MD, PhD 2 ; Daniel Pearce, MD 2 1 BioMedEcon, San Jose, CA, USA; 2 Wake Forest University School
Breakthrough Drugs in Dermatology. Mark Lebwohl, MD
 Breakthrough Drugs in Dermatology Mark Lebwohl, MD Sol and Clara Kest Professor And Chairman Kimberly and Eric J. Waldman Department of Dermatology Icahn School of Medicine at Mount Sinai LIFE CHANGING
Breakthrough Drugs in Dermatology Mark Lebwohl, MD Sol and Clara Kest Professor And Chairman Kimberly and Eric J. Waldman Department of Dermatology Icahn School of Medicine at Mount Sinai LIFE CHANGING
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 1097-7 Program Prior Authorization/Notification Medication *Stelara (ustekinumab) *This program applies to the subcutaneous formulation
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2018 P 1097-7 Program Prior Authorization/Notification Medication *Stelara (ustekinumab) *This program applies to the subcutaneous formulation
USTEKINUMAB Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA GUIDELINES FOR USE
 Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA 36187 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of psoriatic arthritis (PsA)
Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA 36187 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of psoriatic arthritis (PsA)
Horizon Scanning Centre January Apremilast for psoriasis SUMMARY NIHR HSC ID: 2652
 Horizon Scanning Centre January 2013 Apremilast for psoriasis SUMMARY NIHR HSC ID: 2652 This briefing is based on information available at the time of research and a limited literature search. It is not
Horizon Scanning Centre January 2013 Apremilast for psoriasis SUMMARY NIHR HSC ID: 2652 This briefing is based on information available at the time of research and a limited literature search. It is not
Regulatory Status FDA-approved indication: Orencia is a selective T cell costimulation modulator indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.18 Subject: Orencia Page: 1 of 8 Last Review Date: March 16, 2018 Orencia Description Orencia (abatacept)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.18 Subject: Orencia Page: 1 of 8 Last Review Date: March 16, 2018 Orencia Description Orencia (abatacept)
Etanercept for Treatment of Hidradenitis
 Home Search Browse Resources Help What's New About Purpose Etanercept for Treatment of Hidradenitis This study is currently recruiting patients. Sponsors and Collaborators: University of Pennsylvania Amgen
Home Search Browse Resources Help What's New About Purpose Etanercept for Treatment of Hidradenitis This study is currently recruiting patients. Sponsors and Collaborators: University of Pennsylvania Amgen
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
Xeljanz. Xeljanz, Xeljanz XR (tofacitinib) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.24 Subject: Xeljanz Page: 1 of 6 Last Review Date: March 16, 2018 Xeljanz Description Xeljanz, Xeljanz
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.24 Subject: Xeljanz Page: 1 of 6 Last Review Date: March 16, 2018 Xeljanz Description Xeljanz, Xeljanz
Contemporary Management of Moderate to Severe Plaque Psoriasis
 SUPPLEMENT THE AMERICAN JOURNAL OF MANAGED CARE December 2017 Vol. 23 No. 21, Sup. Contemporary Management of Moderate to Severe Plaque Psoriasis HIGHLIGHTS Effect of Plaque Psoriasis on Physical, Cardiovascular,
SUPPLEMENT THE AMERICAN JOURNAL OF MANAGED CARE December 2017 Vol. 23 No. 21, Sup. Contemporary Management of Moderate to Severe Plaque Psoriasis HIGHLIGHTS Effect of Plaque Psoriasis on Physical, Cardiovascular,
COSENTYX (secukinumab)
 COSENTYX (secukinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
COSENTYX (secukinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Clinical Policy: Ustekinumab (Stelara) Reference Number: CP.PHAR.264
 Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08/16 Last Review Date: 05/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08/16 Last Review Date: 05/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Abatacept (Orencia) Reference Number: ERX.SPA.123 Effective Date:
 Clinical Policy: (Orencia) Reference Number: ERX.SPA.123 Effective Date: 10.01.16 Last Review Date: 05.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Orencia) Reference Number: ERX.SPA.123 Effective Date: 10.01.16 Last Review Date: 05.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Actemra. Actemra (tocilizumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.12 Subject: Actemra Page: 1 of 13 Last Review Date: September 20, 2018 Actemra Description Actemra
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.12 Subject: Actemra Page: 1 of 13 Last Review Date: September 20, 2018 Actemra Description Actemra
Review Article The Safety of Systemic Treatments That Can Be Used for Geriatric Psoriasis Patients: A Review
 Dermatology Research and Practice Volume 2012, Article ID 367475, 4 pages doi:10.1155/2012/367475 Review Article The Safety of Systemic Treatments That Can Be Used for Geriatric Psoriasis Patients: A Review
Dermatology Research and Practice Volume 2012, Article ID 367475, 4 pages doi:10.1155/2012/367475 Review Article The Safety of Systemic Treatments That Can Be Used for Geriatric Psoriasis Patients: A Review
Topical treatment of psoriasis in difficult to treat areas: Genital, folds and palmoplantar
 Topical treatment of psoriasis in difficult to treat areas: Genital, folds and palmoplantar Dr. Steven Feldman 1 Genital psoriasis: a questionnaire-based survey on a concealed skin disease in the Netherlands
Topical treatment of psoriasis in difficult to treat areas: Genital, folds and palmoplantar Dr. Steven Feldman 1 Genital psoriasis: a questionnaire-based survey on a concealed skin disease in the Netherlands
Psoriasis the latest recommendations for management: where can primary care make a real difference?
 Dermatology Psoriasis the latest recommendations for management: where can primary care make a real difference? Dr Stephen Kownacki Executive chair, Primary Care Dermatology Society (PCDS) This session
Dermatology Psoriasis the latest recommendations for management: where can primary care make a real difference? Dr Stephen Kownacki Executive chair, Primary Care Dermatology Society (PCDS) This session
Clinical Policy: Ustekinumab (Stelara) Reference Number: ERX.SPA.01 Effective Date:
 Clinical Policy: (Stelara) Reference Number: ERX.SPA.01 Effective Date: 04.01.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Stelara) Reference Number: ERX.SPA.01 Effective Date: 04.01.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Drugs and Applicable Coding: J-code: Enbrel-J1438; Humira-J0135; Remicade-J1745; Inflectra-Q5102; Cimzia-J0718; Simponi-J1602 Renflexis - pending
 Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Combination Nonbiologic Therapy in Psoriasis. Sushil Tahiliani, MBBS, MD
 Combination Nonbiologic Therapy in Psoriasis Sushil Tahiliani, MBBS, MD Agenda Rationale Preferred and less preferred combination Morphology-specific preferred combinations Doses used in combinations Potential
Combination Nonbiologic Therapy in Psoriasis Sushil Tahiliani, MBBS, MD Agenda Rationale Preferred and less preferred combination Morphology-specific preferred combinations Doses used in combinations Potential
REFERENCE CODE GDHC1114CFR PUBLICATION DATE M AY 2013
 REFERENCE CODE GDHC1114CFR PUBLICATION DATE M AY 2013 PSORIASIS - Psoriasis - India Drug Forecast and Market Analysis to Executive Summary Sales for Psoriasis in India 2012 We estimate the 2012 PsO drug
REFERENCE CODE GDHC1114CFR PUBLICATION DATE M AY 2013 PSORIASIS - Psoriasis - India Drug Forecast and Market Analysis to Executive Summary Sales for Psoriasis in India 2012 We estimate the 2012 PsO drug
