Research in Real Life. Real Life Effectiveness of Aclidinium Bromide. Version Date: 26 September 2013
|
|
|
- Douglas Oliver
- 5 years ago
- Views:
Transcription
1 Research in Real Life Real Life Effectiveness of Aclidinium Bromide Version Date: 26 September 2013 Project for Almirall Real-life effectiveness evaluation of the long-acting muscarinic antagonist aclidinium bromide (Eklira ) for the management of COPD in a routine UK primary care population Evaluating the real-life effectiveness (with optional cost effectiveness) of aclidinium bromide and the characteristics of patients prescribed aclidinium therapy for COPD following the launch of Eklira. PROTOCOL FOR STUDY 1
2 1 STUDY DESCRIPTION 1.1 STUDY DESIGN Aim of project: To evaluate the real-life effectiveness of the antimuscarine bronchodilator aclidinium bromide (Eklira ) in three combined studies following the launch of Eklira in the UK. Study 1: Characterising patients prescribed aclidinium bromide from tiotropium, and determining how many patients are satisfied with their change to aclidinium therapy. This will determine the acceptability of aclidinium in clinical practice. Study 2: (A) Examine the effects of changing COPD therapy to aclidinium bromide from LAMA therapy other than aclidinium bromide. (B: optional) Examine the effects of changing patient therapy based on patterns of use in study 1 (e.g. changing to aclidinium bromide ± LABA from ICS/LABA). This study is a before and after comparison of changes in patient outcomes in patients changing their COPD therapy to aclidinium bromide from LAMAs other than aclidinium bromide or other therapies to examine the clinical effects of the therapy change. The study will be powered for the change from baseline therapy. Study 3: Matched cohort study comparing COPD outcomes for patients changing to aclidinium bromide from tiotropium with patients remaining on their tiotropium therapy. This study will provide a matched comparison of the two therapies and the relative effectiveness of aclidinium bromide. This protocol covers the procedures required for study 1. Study 1 This is a retrospective effectiveness study consisting of a baseline and outcome period. The baseline period is a minimum of one year before the date of the patient s first aclidinium bromide prescription during which all patients were treated with tiotropium. The outcome period is defined as six months following the first aclidinium bromide prescription. 2
3 6-month therapy success evaluation and patient characterisation Index Prescription Date: Date of first prescription One-year baseline period Patients taking tiotropium Six-month outcome period Patients who change therapy to aclidinium bromide During the six month outcome period, the acceptability of change will be evaluated, defined below. 1.2 STUDY PERIOD The full study will run for three years post UK aclidinium bromide launch. Study 1 is aimed to be completed within the first year. 1.3 STUDY POPULATION INCLUSION CRITERIA The analysis includes patients who changed their therapy from tiotropium to aclidinium bromide following its launch in the UK. Patients should also meet the following inclusion criteria: Aged 40 years at index prescription date COPD diagnosis (diagnostic code and 2 prescriptions for COPD therapy in year prior to IPD) Evidence of COPD treatment: 1 prescription for tiotropium during the baseline period Ongoing COPD treatment: 1 prescription for long acting muscarinic antagonist therapy in the outcome period EXCLUSION CRITERIA As this study is exploring the demographics of patients who change their therapy to aclidinium bromide, no patients will be excluded at this stage. 1.4 STUDY OUTCOMES The following outcome will be evaluated for all patients: 3
4 1.4.1 PRIMARY EFFECTIVENESS OUTCOME The primary effectiveness outcome for this study is the acceptability of change from tiotropium to aclidinium during the six-month outcome period. This is defined as the percentage of the patients changed to aclidinium bromide (received a prescription for aclidinium at date of first prescription) who did not receive 1 prescription for tiotropium during the outcome period. As some patients will change back to their previous tiotropium because of a resistance to change rather than as a reflection of dissatisfaction with their new therapy an acceptability rate of <70% is felt to be potentially indicative of dissatisfaction with the aclinidium therapy. Potential reasons for patients who change back may be explored if required by the study sponsor PATIENT CHARACTERISATION To provide real-world data on the utilisation of aclidinium bromide in clinical practice, the patients prescribed aclidinium bromide will be characterised according to the following in the year prior to aclidinium bromide initiation. As the study aims to characterise the patients who are prescribed aclidinium, all patients will be characterised regardless of whether or not they change back to tiotropium: age sex height weight BMI % predicted FEV1 FEV/FVC ratio smoking status co-morbidities including asthma, rhinitis COPD exacerbations lower respiratory tract infections prior LAMA therapy + doses COPD severity (risk and symptoms) defined by GOLD group (A, B, C, D) lung function defined by GOLD stage (1, 2, 3, 4) 2 DATA AND ANALYTICAL METHODS 2.1 DATA DATA SOURCE Data will be sourced from one database for this study: OPTIMUM PATIENT CARE RESEARCH DATABASE The Optimum Patient Care Research Database (OPCRD) comprises anonymised data extracted from practices in order to perform reviews of their chronic respiratory services. Two types of anonymised patient data are typically collected: 4
5 (1) Routine clinical data OPC software interfaces with primary care practice management systems and extracts disease coding and prescribing information; and (2) Questionnaires Patients identified as recipients of the respiratory service under review are invited to complete validated disease assessment questionnaires to better understand their current health status (and/or possible reasons for sub-optimal status); Anonymised questionnaires are assigned a unique code to aid matching routine data to questionnaire results. The OPCRD has been approved by Trent Multi Centre Research Ethics Committee for clinical research use. The anonymised, longitudinal patient data offers a high-quality data source for use in clinical, epidemiological and pharmaceutical research. It enables research to be carried out across a broad-range of respiratory areas. 2.2 DEFINITIONS BODY MASS INDEX (BMI) The Body Mass Index is a representative measure of body weight based on the weight and height of the subject. It is defined as the weight (in kg) divided by the square of the height (in m) and is measured in kg/m CHARLSON COMORBIDITY INDEX (CCI) The Charlson Comorbidity Index was developed in the US in 1987 as a method of classifying prognostic comorbidity in longitudinal studies i. It predicts the one-year mortality for a patient who may have a range of comorbid conditions such as heart disease, AIDS or cancer. Each condition is assigned a weight depending on the risk of dying associated with the condition; scores are then summed to give a total score predicting mortality. The weights were revised and up-dated (for example, mortality due to HIV has fallen) by Dr. Foster Intelligence (DFI) in their HSMR Methodology documentation ii and calibrated using UK data (due to differences in coding practice and hospital patient population characteristics from the US), using ICD-10 codes. As a result: DFI have expanded the coding definition of some conditions; Only secondary diagnoses (DIAG02-DIAG14) are now considered; There is greater variation in weights between conditions and the Charlson Index (the sum of the weights) can be treated as a continuous variable (limited to the range 0-50) for the purposes of risk adjustment. The weights, codes and conditions used in this study are summarised in Error! Reference source not found...condition Condition Name ICD 10 codes New weight 1 Acute myocardial infarction I21, I22, I23, I252, I Cerebral vascular accident G450, G451, G452, G454, G458, 11 G459, G46, I60-I69 3 Congestive heart failure I Connective tissue disorder M05, M060, M063, M069, M32, 4 5
6 M332, M34, M353 5 Dementia F00, F01, F02, F03, F Diabetes E101, E105, E106, E108, E109, E111, 3 E115, E116, E118, E119, E131, E131, E136, E138, E139, E141, E145, E146, E148, E149 7 Liver disease K702, K703, K717, K73, K Peptic ulcer K25, K26, K27, K Peripheral vascular disease I71, I739, I790, R02, Z958, Z Pulmonary disease J40-J47, J60-J Cancer C00-C76, C80-C Diabetes complications E102, E103, E104, E107, E112, E113, -1 E114, E117, E132, E133, E134, E137, E142, E143, E144, E Paraplegia G041, G81, G820, G821, G Renal disease I12, I13, N01, N03, N052-N056, 10 N072-N074, N18, N19, N25 15 Metastatic cancer C77, C78, C Severe liver disease K721, K729, K766, K HIV B20, B21, B22, B23, B24 2 Table 2: Co-morbid conditions and scores used in the Charlson Co-morbidity Index (CCI) 3 THE ANALYSIS PLAN 3.1 GENERAL Exploratory data analysis will be carried out for all baseline variables and presented in the appendix SOFTWARE All analysis will be carried out using IBM SPSS Statistics version 20 iii, SAS version 9.3 iv and Microsoft Office EXCEL SIGNIFICANCE TESTING Statistically significant results will be defined as p < 0.05 and trends as 0.05 p < EXPLORATORY DATA ANALYSIS SUMMARY STATISTICS Summary statistics will be produced for all baseline variables. For variables measured on the interval or ratio scale, these will include: Sample size (n) Percentage non-missing Mean Variance / Standard Deviation Range (Minimum / Maximum) Median 6
7 Inter-quartile Range (25 th and 75 th percentiles) For categorical variables, the summary statistics will include: Sample size (n) Count and Percentage by category (distribution) PLOTS Plots will be produced for all baseline variables. For variables measured on the interval or ratio scale, these will include: Frequency plots Box and whisker plots Frequency plots will illustrate the distribution of the variable and whether categorisation may be necessary (for example, if heavily skewed). Box plots will illustrate the location and spread of the variable and identify potential outliers. For categorical variables, bar charts will be produced to illustrate distributions DATA PREPARATION The data will be prepared for analysis by: Investigating potential outliers; Indentifying and creating new variables as necessary: o Transformations of skewed data (for example, log transformations); o Categorisation of heavily skewed data; Investigating missing data (type of and reason for missingness). 3.3 STATISTICAL ANALYSIS PRIMARY EFFECTIVENESS OUTCOME The percentage of patients achieving the primary outcome will be calculated PATIENT CHARACTERISATION Summary statistics will be produced for all baseline characteristics 3.4 STATISTICAL TESTS Table summarises the statistical tests that may be used in the analysis as appropriate. Test Chi-square (χ 2 ) test Use Tests for the association between 2 categorical variables (data presented 7
8 Mann Whitney U test t-test in contingency tables). Nonparametric test to compare the distribution of a variable measured on the ratio / interval scale across two groups when the variable is not normally distributed. Parametric test to compare the mean of a variable measured on the ratio / interval scale between two groups when the variable is normally distributed. Table 3: Summary of Statistical Tests 8
9 3.5 RESEARCH TEAM & STEERING COMMITTEE Chief Investigator: Professor David Price, Professor of Primary Care Respiratory Medicine and Director of Research in Real Life Research Team: Research in Real Life Chief Executive: Catherine Hutton, Research in Real Life Researcher: Daina Lim, Research in Real Life Project Coordinator: Victoria Carter, Research in Real Life Senior Statistician: Annie Burden: Research in Real Life Data Analyst: Julie von Ziegenweidt, Research in Real Life Steering Committee: TBC Study Sponsors: Almirall i Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: Development and Validation. J Chronic Dis 1987;40: ii Doctor Foster Health HSMR mortality indicators. Available online at: iii IBM SPSS Statistics Statistics family. Available online at: iv SAS Institute Inc Statistical Analysis with SAS/STAT Software. Available online at: 9
Research in Real Life
 Research in Real Life Study 1: Exploratory study - identifying the benefits of pmdi versus Diskus for delivering fluticasone/salmeterol combination therapy in patients with chronic obstructive pulmonary
Research in Real Life Study 1: Exploratory study - identifying the benefits of pmdi versus Diskus for delivering fluticasone/salmeterol combination therapy in patients with chronic obstructive pulmonary
Research in Real Life Evaluation of patients with COPD and CKD who would benefit from aclidinium bromide treatment. Version Date: 15 th October 2013
 Research in Real Life Evaluation of patients with COPD and CKD who would benefit from aclidinium bromide treatment. Version Date: 15 th October 2013 Draft protocol Characterising patient groups with chronic
Research in Real Life Evaluation of patients with COPD and CKD who would benefit from aclidinium bromide treatment. Version Date: 15 th October 2013 Draft protocol Characterising patient groups with chronic
Research in real life ltd study protocol for Teva Pharmaceutical Industries Ltd.
 Research in real life ltd study protocol for Teva Pharmaceutical Industries Ltd. The use of a spacer in the delivery of large (Fluticasone Propionate) and small particle FP and Qvar Spacer vs Non-Spacer
Research in real life ltd study protocol for Teva Pharmaceutical Industries Ltd. The use of a spacer in the delivery of large (Fluticasone Propionate) and small particle FP and Qvar Spacer vs Non-Spacer
Research in Real Life Ltd Study Protocol for Chiesi Ltd
 Characterising patients and examining real-life outcomes for UK patients with COPD initiating Research in Real Life Ltd Study Protocol for Chiesi Ltd REACH II: Characterising patients and examining real-life
Characterising patients and examining real-life outcomes for UK patients with COPD initiating Research in Real Life Ltd Study Protocol for Chiesi Ltd REACH II: Characterising patients and examining real-life
Impact of lengthening script duration of inhaled corticosteroid (ICS) therapy on clinical outcomes in asthma
 Study protocol Study protocol Impact of lengthening script duration of inhaled corticosteroid (ICS) therapy on clinical outcomes in asthma A historical, observational study to assess whether increasing
Study protocol Study protocol Impact of lengthening script duration of inhaled corticosteroid (ICS) therapy on clinical outcomes in asthma A historical, observational study to assess whether increasing
Online supplementary material
 Online supplementary material Add-on long-acting β2-agonist (LABA) in a separate inhaler as asthma step-up therapy versus increased dose of inhaled corticosteroid (ICS) or ICS/LABA combination inhaler
Online supplementary material Add-on long-acting β2-agonist (LABA) in a separate inhaler as asthma step-up therapy versus increased dose of inhaled corticosteroid (ICS) or ICS/LABA combination inhaler
Inhaled Corticosteroid vs. Add-On Long-Acting Beta-Agonist for Step-Up Therapy in Asthma
 Online Data Supplement Inhaled Corticosteroid vs. Add-On Long-Acting Beta-Agonist for Step-Up Therapy in Asthma Elliot Israel, Nicolas Roche, Richard J. Martin, Gene Colice, Paul M. Dorinsky, Dirkje S.
Online Data Supplement Inhaled Corticosteroid vs. Add-On Long-Acting Beta-Agonist for Step-Up Therapy in Asthma Elliot Israel, Nicolas Roche, Richard J. Martin, Gene Colice, Paul M. Dorinsky, Dirkje S.
Real-life effectiveness evaluation of budesonide/formoterol (BF) Spiromax for the management of asthma and COPD
 Study protocol Study protocol Real-life effectiveness evaluation of budesonide/formoterol (BF) Spiromax for the management of asthma and COPD Four complimentary post-marketing retrospective, observational
Study protocol Study protocol Real-life effectiveness evaluation of budesonide/formoterol (BF) Spiromax for the management of asthma and COPD Four complimentary post-marketing retrospective, observational
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A.
 aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
Why is co-morbidity important for cancer patients? Di Riley Associate Director Clinical Outcomes Programme
 Why is co-morbidity important for cancer patients? Di Riley Associate Director Clinical Outcomes Programme Co-morbidity in cancer Definition:- Co-morbidity is a disease or illness affecting a cancer patient
Why is co-morbidity important for cancer patients? Di Riley Associate Director Clinical Outcomes Programme Co-morbidity in cancer Definition:- Co-morbidity is a disease or illness affecting a cancer patient
Comparison of Medicare Fee-for-Service Beneficiaries Treated in Ambulatory Surgical Centers and Hospital Outpatient Departments
 Comparison of Medicare Fee-for-Service Beneficiaries Treated in Ambulatory Surgical Centers and Hospital Outpatient Departments Prepared for: American Hospital Association April 4, 2019 Berna Demiralp,
Comparison of Medicare Fee-for-Service Beneficiaries Treated in Ambulatory Surgical Centers and Hospital Outpatient Departments Prepared for: American Hospital Association April 4, 2019 Berna Demiralp,
Why is co-morbidity important for cancer patients? Michael Chapman Research Programme Manager
 Why is co-morbidity important for cancer patients? Michael Chapman Research Programme Manager Co-morbidity in cancer Definition:- Co-morbidity is a disease or illness affecting a cancer patient in addition
Why is co-morbidity important for cancer patients? Michael Chapman Research Programme Manager Co-morbidity in cancer Definition:- Co-morbidity is a disease or illness affecting a cancer patient in addition
Decline In lung-function Among Patients with chronic obstructive Lung disease On maintenance therapy (DIAPLO)
 Version V1.4 Decline In lung-function Among Patients with chronic obstructive Lung disease On maintenance therapy (DIAPLO) An observational study evaluating the benefits of early intervention with maintenance
Version V1.4 Decline In lung-function Among Patients with chronic obstructive Lung disease On maintenance therapy (DIAPLO) An observational study evaluating the benefits of early intervention with maintenance
Surveillance report Published: 6 April 2016 nice.org.uk. NICE All rights reserved.
 Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year,
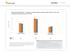 ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
glycopyrronium 44 micrograms hard capsules of inhalation powder (Seebri Breezhaler ) SMC No. (829/12) Novartis Pharmaceuticals Ltd.
 glycopyrronium 44 micrograms hard capsules of inhalation powder (Seebri Breezhaler ) SMC No. (829/12) Novartis Pharmaceuticals Ltd. 07 December 2012 The Scottish Medicines Consortium (SMC) has completed
glycopyrronium 44 micrograms hard capsules of inhalation powder (Seebri Breezhaler ) SMC No. (829/12) Novartis Pharmaceuticals Ltd. 07 December 2012 The Scottish Medicines Consortium (SMC) has completed
Outcomes: Initially, our primary definitions of pneumonia was severe pneumonia, where the subject was hospitalized
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Supplementary materials
 Supplementary materials Table S1 Patient comorbidities by diagnosis Total Asthma n (%) 5348 (148.4) COPD n (%) 4563 (143.4) ACOS n (%) 469 (197.9) No other comorbidities 95 (26.4) 614 (19.3) 27 (11.4)
Supplementary materials Table S1 Patient comorbidities by diagnosis Total Asthma n (%) 5348 (148.4) COPD n (%) 4563 (143.4) ACOS n (%) 469 (197.9) No other comorbidities 95 (26.4) 614 (19.3) 27 (11.4)
Medical Directive. Activation Date: April 24, 2013 Review due by: December 1, Medical Director: Date: December 1, 2017
 Medical Directive Pre and Post Bronchodilator Spirometry Testing and Treatment Initiation Assigned Number: Activation Date: April 24, 2013 Review due by: December 1, 2019 23 Approval Signature & Date Medical
Medical Directive Pre and Post Bronchodilator Spirometry Testing and Treatment Initiation Assigned Number: Activation Date: April 24, 2013 Review due by: December 1, 2019 23 Approval Signature & Date Medical
AWMSG SECRETARIAT ASSESSMENT REPORT. Aclidinium bromide (Eklira Genuair ) 322 micrograms inhalation powder. Reference number: 938 FULL SUBMISSION
 AWMSG SECRETARIAT ASSESSMENT REPORT Aclidinium bromide (Eklira Genuair ) 322 micrograms inhalation powder Reference number: 938 FULL SUBMISSION This report has been prepared by the All Wales Therapeutics
AWMSG SECRETARIAT ASSESSMENT REPORT Aclidinium bromide (Eklira Genuair ) 322 micrograms inhalation powder Reference number: 938 FULL SUBMISSION This report has been prepared by the All Wales Therapeutics
REACH II: Stage 2. Final Report
 Study protocol: R02813 REACH II stage 2 31 st August 2017 Final Report REACH II: Stage 2 Examining real-life outcomes for UK patients with COPD after initiating Fostair pmdi according to its licensed indication
Study protocol: R02813 REACH II stage 2 31 st August 2017 Final Report REACH II: Stage 2 Examining real-life outcomes for UK patients with COPD after initiating Fostair pmdi according to its licensed indication
Risk of Fractures Following Cataract Surgery in Medicare Beneficiaries
 Risk of Fractures Following Cataract Surgery in Medicare Beneficiaries Victoria L. Tseng, MD, Fei Yu, PhD, Flora Lum, MD, Anne L. Coleman, MD, PhD JAMA. 2012;308(5):493-501 Background Visual impairment
Risk of Fractures Following Cataract Surgery in Medicare Beneficiaries Victoria L. Tseng, MD, Fei Yu, PhD, Flora Lum, MD, Anne L. Coleman, MD, PhD JAMA. 2012;308(5):493-501 Background Visual impairment
Biostatistics for Med Students. Lecture 1
 Biostatistics for Med Students Lecture 1 John J. Chen, Ph.D. Professor & Director of Biostatistics Core UH JABSOM JABSOM MD7 February 14, 2018 Lecture note: http://biostat.jabsom.hawaii.edu/education/training.html
Biostatistics for Med Students Lecture 1 John J. Chen, Ph.D. Professor & Director of Biostatistics Core UH JABSOM JABSOM MD7 February 14, 2018 Lecture note: http://biostat.jabsom.hawaii.edu/education/training.html
SGRQ Questionnaire assessing respiratory disease-specific quality of life. Questionnaire assessing general quality of life
 SUPPLEMENTARY MATERIAL e-table 1: Outcomes studied in present analysis. Outcome Abbreviation Definition Nature of data, direction indicating adverse effect (continuous only) Clinical outcomes- subjective
SUPPLEMENTARY MATERIAL e-table 1: Outcomes studied in present analysis. Outcome Abbreviation Definition Nature of data, direction indicating adverse effect (continuous only) Clinical outcomes- subjective
NHS Dumfries & Galloway Triple therapy in COPD patients over 16 years
 Title of Project: NHS Dumfries & Galloway Triple therapy in COPD patients over 16 years 1 Reason for the review Respiratory prescribing is long term and can be costly. Appropriate choice and use of inhaled
Title of Project: NHS Dumfries & Galloway Triple therapy in COPD patients over 16 years 1 Reason for the review Respiratory prescribing is long term and can be costly. Appropriate choice and use of inhaled
REACH II: Stage 2. Study protocol
 Study protocol REACH II: Stage 2 Examining real-life outcomes for UK patients with COPD initiating on Fostair pmdi according to its licensed indication compared to other licensed FDC ICS/LABA therapies
Study protocol REACH II: Stage 2 Examining real-life outcomes for UK patients with COPD initiating on Fostair pmdi according to its licensed indication compared to other licensed FDC ICS/LABA therapies
CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) TREATMENT GUIDELINES
 CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) TREATMENT GUIDELINES Document Description Document Type Service Application Version Guidelines All healthcare professionals(hcps) caring for patients with asthma
CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) TREATMENT GUIDELINES Document Description Document Type Service Application Version Guidelines All healthcare professionals(hcps) caring for patients with asthma
Clinical and radiographic predictors of GOLD-Unclassified smokers in COPDGene
 Clinical and radiographic predictors of GOLD-Unclassified smokers in COPDGene Emily S. Wan, John E. Hokanson, James R. Murphy, Elizabeth A. Regan, Barry J. Make, David A. Lynch, James D. Crapo, Edwin K.
Clinical and radiographic predictors of GOLD-Unclassified smokers in COPDGene Emily S. Wan, John E. Hokanson, James R. Murphy, Elizabeth A. Regan, Barry J. Make, David A. Lynch, James D. Crapo, Edwin K.
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Calverley P M A, Anzueto A R, Carter K, et
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Calverley P M A, Anzueto A R, Carter K, et
Length of stay and cost impact of hospital acquired diagnoses: multiplicative model for SESLHD
 University of Wollongong Research Online Australian Health Services Research Institute Faculty of Business 2014 Length of stay and cost impact of hospital acquired diagnoses: multiplicative model for SESLHD
University of Wollongong Research Online Australian Health Services Research Institute Faculty of Business 2014 Length of stay and cost impact of hospital acquired diagnoses: multiplicative model for SESLHD
Appendix Identification of Study Cohorts
 Appendix Identification of Study Cohorts Because the models were run with the 2010 SAS Packs from Centers for Medicare and Medicaid Services (CMS)/Yale, the eligibility criteria described in "2010 Measures
Appendix Identification of Study Cohorts Because the models were run with the 2010 SAS Packs from Centers for Medicare and Medicaid Services (CMS)/Yale, the eligibility criteria described in "2010 Measures
University of Bristol - Explore Bristol Research
 Hunt, L., Ben-Shlomo, Y., Whitehouse, M., Porter, M., & Blom, A. (2017). The Main Cause of Death Following Primary Total Hip and Knee Replacement for Osteoarthritis: A Cohort Study of 26,766 Deaths Following
Hunt, L., Ben-Shlomo, Y., Whitehouse, M., Porter, M., & Blom, A. (2017). The Main Cause of Death Following Primary Total Hip and Knee Replacement for Osteoarthritis: A Cohort Study of 26,766 Deaths Following
Disclosures. Chronic Obstructive Pulmonary Disease. Chronic Obstructive Pulmonary Disease (COPD)
 Chronic Obstructive Pulmonary Disease May 14, 2016 Orlando, FL COPD (Chronic obstructive pulmonary disease) is a major cause of mortality and morbidity in the United States. Alarmingly, COPD recently became
Chronic Obstructive Pulmonary Disease May 14, 2016 Orlando, FL COPD (Chronic obstructive pulmonary disease) is a major cause of mortality and morbidity in the United States. Alarmingly, COPD recently became
Data Fusion: Integrating patientreported survey data and EHR data for health outcomes research
 Data Fusion: Integrating patientreported survey data and EHR data for health outcomes research Lulu K. Lee, PhD Director, Health Outcomes Research Our Development Journey Research Goals Data Sources and
Data Fusion: Integrating patientreported survey data and EHR data for health outcomes research Lulu K. Lee, PhD Director, Health Outcomes Research Our Development Journey Research Goals Data Sources and
RESEARCH PROTOCOL PROPOSAL
 RESEARCH PROTOCOL PROPOSAL UNCOVERING AND NOTING LONG-TERM OUTCOMES IN COPD AND ASTHMA TO ENHANCE KNOWLEDGE - UNLOCK UNLOCK Study: The prevalence of comorbidities in COPD patients and their impact on the
RESEARCH PROTOCOL PROPOSAL UNCOVERING AND NOTING LONG-TERM OUTCOMES IN COPD AND ASTHMA TO ENHANCE KNOWLEDGE - UNLOCK UNLOCK Study: The prevalence of comorbidities in COPD patients and their impact on the
April 10 th, Bond Street, Toronto ON, M5B 1W8
 Comprehensive Research Plan: Inhaled long-acting muscarinic antagonists (LAMAs; long-acting anticholinergics) for the treatment of chronic obstructive pulmonary disease (COPD) April 10 th, 2014 30 Bond
Comprehensive Research Plan: Inhaled long-acting muscarinic antagonists (LAMAs; long-acting anticholinergics) for the treatment of chronic obstructive pulmonary disease (COPD) April 10 th, 2014 30 Bond
The distribution of COPD in UK general practice using the new GOLD classification
 ORIGINAL ARTICLE COPD The distribution of COPD in UK general practice using the new GOLD classification John Haughney 1, Kevin Gruffydd-Jones 2, June Roberts 3, Amanda J. Lee 4, Alison Hardwell 5 and Lorcan
ORIGINAL ARTICLE COPD The distribution of COPD in UK general practice using the new GOLD classification John Haughney 1, Kevin Gruffydd-Jones 2, June Roberts 3, Amanda J. Lee 4, Alison Hardwell 5 and Lorcan
This is the publisher s version. This version is defined in the NISO recommended practice RP
 Journal Article Version This is the publisher s version. This version is defined in the NISO recommended practice RP-8-2008 http://www.niso.org/publications/rp/ Suggested Reference Chong, J., Karner, C.,
Journal Article Version This is the publisher s version. This version is defined in the NISO recommended practice RP-8-2008 http://www.niso.org/publications/rp/ Suggested Reference Chong, J., Karner, C.,
Aclidinium bromide/ formoterol Benefit assessment according to 35a Social Code Book V 1
 IQWiG Reports Commission No. A15-06 Aclidinium bromide/ formoterol Benefit assessment according to 35a Social Code Book V 1 Extract 1 Translation of Sections 2.1 to 2.6 of the dossier assessment Aclidiniumbromid/Formoterol
IQWiG Reports Commission No. A15-06 Aclidinium bromide/ formoterol Benefit assessment according to 35a Social Code Book V 1 Extract 1 Translation of Sections 2.1 to 2.6 of the dossier assessment Aclidiniumbromid/Formoterol
Understandable Statistics
 Understandable Statistics correlated to the Advanced Placement Program Course Description for Statistics Prepared for Alabama CC2 6/2003 2003 Understandable Statistics 2003 correlated to the Advanced Placement
Understandable Statistics correlated to the Advanced Placement Program Course Description for Statistics Prepared for Alabama CC2 6/2003 2003 Understandable Statistics 2003 correlated to the Advanced Placement
Wirral COPD Prescribing Guidelines
 Wirral COPD Prescribing Guidelines (To be read in conjunction with the Wirral COPD Supplementary Information) STEP 1: Assess symptoms COPD Assessment Test (CAT) [Link for CAT-test Online] is a patient-completed
Wirral COPD Prescribing Guidelines (To be read in conjunction with the Wirral COPD Supplementary Information) STEP 1: Assess symptoms COPD Assessment Test (CAT) [Link for CAT-test Online] is a patient-completed
Chronic obstructive pulmonary disease
 0 Chronic obstructive pulmonary disease Implementing NICE guidance June 2010 NICE clinical guideline 101 What this presentation covers Background Scope Key priorities for implementation Discussion Find
0 Chronic obstructive pulmonary disease Implementing NICE guidance June 2010 NICE clinical guideline 101 What this presentation covers Background Scope Key priorities for implementation Discussion Find
SABA: VENTOLIN EVOHALER (SALBUTAMOL) SAMA: ATROVENT IPRATROPIUM. Offer LAMA (discontinue SAMA) OR LABA
 COPD GUIDELINES DIAGNOSIS >35 years of age Symptoms of cough, breathlessness, sputum, wheeze, Risk factor (SMOKING) Spirometry (post bronchodilator) FEV1/FVC = 0.7 ENCOURAGE PATIENTS TO BRING INHALERS
COPD GUIDELINES DIAGNOSIS >35 years of age Symptoms of cough, breathlessness, sputum, wheeze, Risk factor (SMOKING) Spirometry (post bronchodilator) FEV1/FVC = 0.7 ENCOURAGE PATIENTS TO BRING INHALERS
Table of Contents. Plots. Essential Statistics for Nursing Research 1/12/2017
 Essential Statistics for Nursing Research Kristen Carlin, MPH Seattle Nursing Research Workshop January 30, 2017 Table of Contents Plots Descriptive statistics Sample size/power Correlations Hypothesis
Essential Statistics for Nursing Research Kristen Carlin, MPH Seattle Nursing Research Workshop January 30, 2017 Table of Contents Plots Descriptive statistics Sample size/power Correlations Hypothesis
Supplementary Online Content
 Supplementary Online Content Shurraw S, Hemmelgarn B, Lin M, et al. Association between glycemic control and adverse outcomes in people with diabetes mellitus and chronic kidney disease: a population-based
Supplementary Online Content Shurraw S, Hemmelgarn B, Lin M, et al. Association between glycemic control and adverse outcomes in people with diabetes mellitus and chronic kidney disease: a population-based
Clinical Policy: Roflumilast (Daliresp) Reference Number: CP.PMN.46 Effective Date: Last Review Date: 08.18
 Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11.01.11 Last Review Date: 08.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11.01.11 Last Review Date: 08.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Budesonide SteriNebs 0.25 mg/0.50 mg (0.25 mg) (0.50 mg)
 TITLE Subtitle Protocol version identifier Active substance Medicinal product National Drug Code (NDC) Marketing authorization holder Marketing category and application number Research questions and objectives
TITLE Subtitle Protocol version identifier Active substance Medicinal product National Drug Code (NDC) Marketing authorization holder Marketing category and application number Research questions and objectives
The Prognostic Importance of Comorbidity for Mortality in Patients With Stable Coronary Artery Disease
 Journal of the American College of Cardiology Vol. 43, No. 4, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2003.10.031
Journal of the American College of Cardiology Vol. 43, No. 4, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2003.10.031
Almirall grants Invida commercial rights for Aclidinium in Australia and New ZealandAlmirall????? (Invida)??????????? Aclidinium?????
 Almirall grants Invida commercial rights for Aclidinium in Australia and New ZealandAlmirall????? (Invida)??????????? Aclidinium????? Almirall licenses Aclidinium to Invida in Australia and New Zealand
Almirall grants Invida commercial rights for Aclidinium in Australia and New ZealandAlmirall????? (Invida)??????????? Aclidinium????? Almirall licenses Aclidinium to Invida in Australia and New Zealand
umeclidinium, 55 micrograms, powder for inhalation (Incruse ) SMC No. (1004/14) GlaxoSmithKline
 umeclidinium, 55 micrograms, powder for inhalation (Incruse ) SMC No. (1004/14) GlaxoSmithKline 07 November 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
umeclidinium, 55 micrograms, powder for inhalation (Incruse ) SMC No. (1004/14) GlaxoSmithKline 07 November 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
Summary of the risk management plan (RMP) for Laventair (umeclidinium bromide and vilanterol)
 EMA/258177/2014 Summary of the risk management plan (RMP) for Laventair (umeclidinium bromide and vilanterol) This is a summary of the risk management plan (RMP) for Laventair, which details the measures
EMA/258177/2014 Summary of the risk management plan (RMP) for Laventair (umeclidinium bromide and vilanterol) This is a summary of the risk management plan (RMP) for Laventair, which details the measures
National COPD Audit Programme
 National COPD Audit Programme Planning for every breath National Chronic Obstructive Pulmonary Disease (COPD) Audit Programme: Primary care audit () 2015 17 Data analysis and methodology Section 4: Providing
National COPD Audit Programme Planning for every breath National Chronic Obstructive Pulmonary Disease (COPD) Audit Programme: Primary care audit () 2015 17 Data analysis and methodology Section 4: Providing
Summary of the risk management plan (RMP) for Duaklir Genuair (aclidinium / formoterol fumarate dihydrate)
 EMA/605453/2014 Summary of the risk management plan (RMP) for Duaklir Genuair (aclidinium / formoterol fumarate dihydrate) This is a summary of the risk management plan (RMP) for Duaklir Genuair, which
EMA/605453/2014 Summary of the risk management plan (RMP) for Duaklir Genuair (aclidinium / formoterol fumarate dihydrate) This is a summary of the risk management plan (RMP) for Duaklir Genuair, which
Managing COPD Learning Collaborative
 Managing COPD Learning Collaborative November 2-4, 2011 San Antonio, TX Sponsored by AMGA and Boehringer Ingelheim Pharmaceuticals, Inc. Geisinger Health System Carolyn C. Houk, MD FACP Paul Simonelli,
Managing COPD Learning Collaborative November 2-4, 2011 San Antonio, TX Sponsored by AMGA and Boehringer Ingelheim Pharmaceuticals, Inc. Geisinger Health System Carolyn C. Houk, MD FACP Paul Simonelli,
This is a cross-sectional analysis of the National Health and Nutrition Examination
 SUPPLEMENTAL METHODS Study Design and Setting This is a cross-sectional analysis of the National Health and Nutrition Examination Survey (NHANES) data 2007-2008, 2009-2010, and 2011-2012. The NHANES is
SUPPLEMENTAL METHODS Study Design and Setting This is a cross-sectional analysis of the National Health and Nutrition Examination Survey (NHANES) data 2007-2008, 2009-2010, and 2011-2012. The NHANES is
Real World Patients: The Intersection of Real World Evidence and Episode of Care Analytics
 PharmaSUG 2018 - Paper RW-05 Real World Patients: The Intersection of Real World Evidence and Episode of Care Analytics David Olaleye and Youngjin Park, SAS Institute Inc. ABSTRACT SAS Institute recently
PharmaSUG 2018 - Paper RW-05 Real World Patients: The Intersection of Real World Evidence and Episode of Care Analytics David Olaleye and Youngjin Park, SAS Institute Inc. ABSTRACT SAS Institute recently
 PATCH Analysis Plan v1.2.doc Prophylactic Antibiotics for the Treatment of Cellulitis at Home: PATCH Analysis Plan for PATCH I and PATCH II Authors: Angela Crook, Andrew Nunn, James Mason and Kim Thomas,
PATCH Analysis Plan v1.2.doc Prophylactic Antibiotics for the Treatment of Cellulitis at Home: PATCH Analysis Plan for PATCH I and PATCH II Authors: Angela Crook, Andrew Nunn, James Mason and Kim Thomas,
Medicines Optimisation Prescribing Audit: Long acting muscarinic antagonist (LAMA ) review
 Medicines Optimisation Prescribing Audit: Long acting muscarinic antagonist (LAMA ) review Aim To identify all patients prescribed long acting muscarinic antagonists (LAMAs) and where clinically appropriate
Medicines Optimisation Prescribing Audit: Long acting muscarinic antagonist (LAMA ) review Aim To identify all patients prescribed long acting muscarinic antagonists (LAMAs) and where clinically appropriate
If you require this document in another format such as Braille, large print, audio or another language please contact the Trusts Communications Team
 MANAGEMENT OF CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) IN ADULTS Summary statement: How does the document support patient care? Staff/stakeholders involved in development: Job titles only Division:
MANAGEMENT OF CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) IN ADULTS Summary statement: How does the document support patient care? Staff/stakeholders involved in development: Job titles only Division:
The U.S. asthma population poses a significant burden
 RESEARCH Clinical and Economic Burden of Blood Eosinophils in Patients With and Without Uncontrolled Asthma Julian Casciano, BS; Jerry Krishnan, MD, PhD; Zenobia Dotiwala, MS; Chenghui Li, PhD; and Shawn
RESEARCH Clinical and Economic Burden of Blood Eosinophils in Patients With and Without Uncontrolled Asthma Julian Casciano, BS; Jerry Krishnan, MD, PhD; Zenobia Dotiwala, MS; Chenghui Li, PhD; and Shawn
MAKING THE NSQIP PARTICIPANT USE DATA FILE (PUF) WORK FOR YOU
 MAKING THE NSQIP PARTICIPANT USE DATA FILE (PUF) WORK FOR YOU Hani Tamim, PhD Clinical Research Institute Department of Internal Medicine American University of Beirut Medical Center Beirut - Lebanon Participant
MAKING THE NSQIP PARTICIPANT USE DATA FILE (PUF) WORK FOR YOU Hani Tamim, PhD Clinical Research Institute Department of Internal Medicine American University of Beirut Medical Center Beirut - Lebanon Participant
RELATIONSHIP BETWEEN RESPIRATORY DISEASES OF SCHOOLCHILDREN AND TOBACCO SMOKE IN HONG KONG AND SRI LANKA
 RELATIONSHIP BETWEEN RESPIRATORY DISEASES OF SCHOOLCHILDREN AND TOBACCO SMOKE IN HONG KONG AND SRI LANKA 1 S.H. LEE and W.T. HUNG Department of Civil and Environmental Engineering, The Hong Kong Polytechnic
RELATIONSHIP BETWEEN RESPIRATORY DISEASES OF SCHOOLCHILDREN AND TOBACCO SMOKE IN HONG KONG AND SRI LANKA 1 S.H. LEE and W.T. HUNG Department of Civil and Environmental Engineering, The Hong Kong Polytechnic
Evaluation of Asthma Management in Middle EAst North Africa Adult population
 STUDY REPORT SUMMARY Evaluation of Asthma Management in Middle EAst North Africa Adult population Descriptive study on the management of asthma in an asthmatic Middle East Africa adult population Background/Rationale:
STUDY REPORT SUMMARY Evaluation of Asthma Management in Middle EAst North Africa Adult population Descriptive study on the management of asthma in an asthmatic Middle East Africa adult population Background/Rationale:
Design - Multicentre prospective cohort study. Setting UK Community Pharmacies within one CCG area within the UK
 Enabling Patient Health Improvements through COPD (EPIC) Medicines Optimisation within Community Pharmacy: a prospective cohort study Abstract Objectives To improve patients ability to manage their own
Enabling Patient Health Improvements through COPD (EPIC) Medicines Optimisation within Community Pharmacy: a prospective cohort study Abstract Objectives To improve patients ability to manage their own
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
DIABETES MORTALITY IN NOVA SCOTIA FROM 1998 TO 2005: A DESCRIPTIVE ANALYSIS USING BOTH UNDERLYING AND MULTIPLE CAUSES OF DEATH
 DIABETES MORTALITY IN NOVA SCOTIA FROM 1998 TO 2005: A DESCRIPTIVE ANALYSIS USING BOTH UNDERLYING AND MULTIPLE CAUSES OF DEATH Alison Zwaagstra Health Information Analyst Network for End of Life Studies
DIABETES MORTALITY IN NOVA SCOTIA FROM 1998 TO 2005: A DESCRIPTIVE ANALYSIS USING BOTH UNDERLYING AND MULTIPLE CAUSES OF DEATH Alison Zwaagstra Health Information Analyst Network for End of Life Studies
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD)
 Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) Zahava Picado, PharmD PGY1 Pharmacy Practice Resident Central Texas Veterans Healthcare System Temple, TX October
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) Zahava Picado, PharmD PGY1 Pharmacy Practice Resident Central Texas Veterans Healthcare System Temple, TX October
Co. Durham & Darlington Respiratory Network COPD Treatment Guide
 Co. Durham & Darlington Respiratory Network COPD Treatment Guide Age > 35, Productive cough, Breathless, Smoking Hx Spirometry (post-bronchodilator) COPD Advice Intensive smoking cessation support Vaccination
Co. Durham & Darlington Respiratory Network COPD Treatment Guide Age > 35, Productive cough, Breathless, Smoking Hx Spirometry (post-bronchodilator) COPD Advice Intensive smoking cessation support Vaccination
Pharmacotherapy for COPD
 10/3/2017 Topics to be covered Pharmacotherapy for chronic treatment Pharmacotherapy for COPD Dr. W C Yu 3rd September 2017 Commonly used drugs Guidelines for their use Inhaled corticosteroids (ICS) in
10/3/2017 Topics to be covered Pharmacotherapy for chronic treatment Pharmacotherapy for COPD Dr. W C Yu 3rd September 2017 Commonly used drugs Guidelines for their use Inhaled corticosteroids (ICS) in
CSRC COPD Initiative Think Tank meeting Washington DC March 6, 2014
 What is the role of outcome studies in pulmonary drug development and what data can be collected during development to sufficiently characterize an agent so that an outcome study not necessary? CSRC COPD
What is the role of outcome studies in pulmonary drug development and what data can be collected during development to sufficiently characterize an agent so that an outcome study not necessary? CSRC COPD
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Jain S, Kamimoto L, Bramley AM, et al. Hospitalized patients
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Jain S, Kamimoto L, Bramley AM, et al. Hospitalized patients
Changing Landscapes in COPD New Zealand Respiratory Conference
 Changing Landscapes in COPD New Zealand Respiratory Conference Dr Robert Young BMedSc MBChB DPhil (Oxon) FRACP FRCP Associate Professor Consultant Physician Changing Landscapes in COPD: Summary 1. Overview
Changing Landscapes in COPD New Zealand Respiratory Conference Dr Robert Young BMedSc MBChB DPhil (Oxon) FRACP FRCP Associate Professor Consultant Physician Changing Landscapes in COPD: Summary 1. Overview
APPENDIX EXHIBITS. Appendix Exhibit A2: Patient Comorbidity Codes Used To Risk- Standardize Hospital Mortality and Readmission Rates page 10
 Ross JS, Bernheim SM, Lin Z, Drye EE, Chen J, Normand ST, et al. Based on key measures, care quality for Medicare enrollees at safety-net and non-safety-net hospitals was almost equal. Health Aff (Millwood).
Ross JS, Bernheim SM, Lin Z, Drye EE, Chen J, Normand ST, et al. Based on key measures, care quality for Medicare enrollees at safety-net and non-safety-net hospitals was almost equal. Health Aff (Millwood).
ESM1 for Glucose, blood pressure and cholesterol levels and their relationships to clinical outcomes in type 2 diabetes: a retrospective cohort study
 ESM1 for Glucose, blood pressure and cholesterol levels and their relationships to clinical outcomes in type 2 diabetes: a retrospective cohort study Statistical modelling details We used Cox proportional-hazards
ESM1 for Glucose, blood pressure and cholesterol levels and their relationships to clinical outcomes in type 2 diabetes: a retrospective cohort study Statistical modelling details We used Cox proportional-hazards
Journal of the COPD Foundation
 132 Predictors of Change in SGRQ Score Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Original Research Baseline Severity as Predictor of Change in St George s Respiratory Questionnaire
132 Predictors of Change in SGRQ Score Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Original Research Baseline Severity as Predictor of Change in St George s Respiratory Questionnaire
LAMA Products for the Treatment of COPD
 FINAL REPORT LAMA Products for the Treatment of COPD Pharmacoepidemiology Unit: Censored Final Report Tara Gomes, Andrea Gershon, Matthew Stanbrook, Ximena Camacho, Diana Martins, Samantha Singh, Zhan
FINAL REPORT LAMA Products for the Treatment of COPD Pharmacoepidemiology Unit: Censored Final Report Tara Gomes, Andrea Gershon, Matthew Stanbrook, Ximena Camacho, Diana Martins, Samantha Singh, Zhan
Asthma COPD Overlap (ACO)
 Asthma COPD Overlap (ACO) Dr Thomas Brown Consultant Respiratory Physician Thomas.Brown@porthosp.nhs.uk Dr Hitasha Rupani Consultant Respiratory Physician Hitasha.rupani@porthosp.nhs.uk What is Asthma
Asthma COPD Overlap (ACO) Dr Thomas Brown Consultant Respiratory Physician Thomas.Brown@porthosp.nhs.uk Dr Hitasha Rupani Consultant Respiratory Physician Hitasha.rupani@porthosp.nhs.uk What is Asthma
Respiratory Function Testing Is Safe in Patients With Abdominal Aortic Aneurysms.
 Respiratory Function Testing Is Safe in Patients With Abdominal Aortic Aneurysms. Author Zagami, Debbie, Wilson, Jessica, Bodger, Alanna, Sriram, Krishna Published 2014 Journal Title Vascular and Endovascular
Respiratory Function Testing Is Safe in Patients With Abdominal Aortic Aneurysms. Author Zagami, Debbie, Wilson, Jessica, Bodger, Alanna, Sriram, Krishna Published 2014 Journal Title Vascular and Endovascular
Potential risks of ICS use
 Potential risks of ICS use Randomised controlled trial Observational study Systematic review Pneumonia Tuberculosis Bone fracture Skin thinning/easy bruising Cataract Diabetes No effect on fracture risk
Potential risks of ICS use Randomised controlled trial Observational study Systematic review Pneumonia Tuberculosis Bone fracture Skin thinning/easy bruising Cataract Diabetes No effect on fracture risk
Chronic Obstructive Learning Collaborative Sponsored by AMGA and Boehringer Ingelheim Pharmaceuticals, Inc.
 Chronic Obstructive Learning Collaborative Sponsored by AMGA and Boehringer Ingelheim Pharmaceuticals, Inc. November 2-4, 2011 San Antonio, Texas University of Michigan Faculty Group Practice Improving
Chronic Obstructive Learning Collaborative Sponsored by AMGA and Boehringer Ingelheim Pharmaceuticals, Inc. November 2-4, 2011 San Antonio, Texas University of Michigan Faculty Group Practice Improving
Medical Statistics 1. Basic Concepts Farhad Pishgar. Defining the data. Alive after 6 months?
 Medical Statistics 1 Basic Concepts Farhad Pishgar Defining the data Population and samples Except when a full census is taken, we collect data on a sample from a much larger group called the population.
Medical Statistics 1 Basic Concepts Farhad Pishgar Defining the data Population and samples Except when a full census is taken, we collect data on a sample from a much larger group called the population.
Salbutamol/Albuterol. Salbutamol Sterinebs (2.5 and 5.0mg/2.5ml) FN0032 (2.5mg/2.5ml) FN0031 (5.0mg/2.5ml)
 TITLE Subtitle Protocol version identifier Active substance Medicinal product Product code Marketing authorization holder COMPARATIVE EFFECTIVENESS AND SAFETY OF SALBUTAMOL STERINEBS VS. VENTOLIN NEBULES
TITLE Subtitle Protocol version identifier Active substance Medicinal product Product code Marketing authorization holder COMPARATIVE EFFECTIVENESS AND SAFETY OF SALBUTAMOL STERINEBS VS. VENTOLIN NEBULES
USAID Health Care Improvement Project
 IMPROVEMENT OBJECTIVE: Decrease asthma/copd morbidity and mortalityin affected adults and children through improved quality of asthma/copd case-management (ambulatory & hospital) Cross-cutting ambulatory
IMPROVEMENT OBJECTIVE: Decrease asthma/copd morbidity and mortalityin affected adults and children through improved quality of asthma/copd case-management (ambulatory & hospital) Cross-cutting ambulatory
ERS Investor & Analyst Event. Munich Tuesday 9 th September 2014
 ERS Investor & Analyst Event Munich Tuesday 9 th September 2014 Darrell Baker SVP, Global Head of Respiratory Agenda GSK s Respiratory Portfolio Eosinophils Research in COPD Eosinophils Clinical Experience
ERS Investor & Analyst Event Munich Tuesday 9 th September 2014 Darrell Baker SVP, Global Head of Respiratory Agenda GSK s Respiratory Portfolio Eosinophils Research in COPD Eosinophils Clinical Experience
vz Strategic Transaction with Almirall
 vz Strategic Transaction with Almirall in Respiratory Disease Accelerating AstraZeneca Respiratory Leadership July 30, 204 Long-term value generation and strong strategic fit Stronger inhaled portfolio
vz Strategic Transaction with Almirall in Respiratory Disease Accelerating AstraZeneca Respiratory Leadership July 30, 204 Long-term value generation and strong strategic fit Stronger inhaled portfolio
Disclosure Statement. Epidemiological Data
 EVALUATION OF THE MEDICATION UTILIZATION OF COPD PATIENTS AT THE MIAMI VA HEALTHCARE SYSTEM Simone Edgerton, PharmD. PGY 1 Pharmacy Resident Miami VA Healthcare System Miami, Florida Simone.edgerton2@va.gov
EVALUATION OF THE MEDICATION UTILIZATION OF COPD PATIENTS AT THE MIAMI VA HEALTHCARE SYSTEM Simone Edgerton, PharmD. PGY 1 Pharmacy Resident Miami VA Healthcare System Miami, Florida Simone.edgerton2@va.gov
Defining COPD. Georgina Grantham Community Respiratory Team Leader/ Respiratory Nurse Specialist
 Defining COPD Georgina Grantham Community Respiratory Team Leader/ Respiratory Nurse Specialist Defining COPD Chronic Obstructive Pulmonary Disease (COPD) is a common, preventable and treatable disease
Defining COPD Georgina Grantham Community Respiratory Team Leader/ Respiratory Nurse Specialist Defining COPD Chronic Obstructive Pulmonary Disease (COPD) is a common, preventable and treatable disease
«Impact of a pharmaceutical intervention to improve adherence of inhaled medication in asthma and COPD patients»
 «Impact of a pharmaceutical intervention to improve adherence of inhaled medication in asthma and COPD patients» Baseline data from a randomized controlled trial 5 th Swiss Health Services Research Symposium,
«Impact of a pharmaceutical intervention to improve adherence of inhaled medication in asthma and COPD patients» Baseline data from a randomized controlled trial 5 th Swiss Health Services Research Symposium,
Clinical and economic outcomes of an ambulatory urinary tract infection disease management program Armstrong E P
 Clinical and economic outcomes of an ambulatory urinary tract infection disease management program Armstrong E P Record Status This is a critical abstract of an economic evaluation that meets the criteria
Clinical and economic outcomes of an ambulatory urinary tract infection disease management program Armstrong E P Record Status This is a critical abstract of an economic evaluation that meets the criteria
Comparative Study on Three Algorithms of the ICD-10 Charlson Comorbidity Index with Myocardial Infarction Patients
 Journal of Preventive Medicine and Public Health January 2010, Vol. 43, No. 1, 42-49 doi: 10.3961/jpmph.2010.43.1.42 Comparative Study on Three Algorithms of the ICD-10 Charlson Comorbidity Index with
Journal of Preventive Medicine and Public Health January 2010, Vol. 43, No. 1, 42-49 doi: 10.3961/jpmph.2010.43.1.42 Comparative Study on Three Algorithms of the ICD-10 Charlson Comorbidity Index with
roflumilast 500 microgram tablets (Daxas ) SMC No. (635/10) Nycomed Ltd
 roflumilast 500 microgram tablets (Daxas ) SMC No. (635/10) Nycomed Ltd 06 August 2010 (Issued 10 September 2010) The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
roflumilast 500 microgram tablets (Daxas ) SMC No. (635/10) Nycomed Ltd 06 August 2010 (Issued 10 September 2010) The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
TOTAL HIP AND KNEE REPLACEMENTS. FISCAL YEAR 2002 DATA July 1, 2001 through June 30, 2002 TECHNICAL NOTES
 TOTAL HIP AND KNEE REPLACEMENTS FISCAL YEAR 2002 DATA July 1, 2001 through June 30, 2002 TECHNICAL NOTES The Pennsylvania Health Care Cost Containment Council April 2005 Preface This document serves as
TOTAL HIP AND KNEE REPLACEMENTS FISCAL YEAR 2002 DATA July 1, 2001 through June 30, 2002 TECHNICAL NOTES The Pennsylvania Health Care Cost Containment Council April 2005 Preface This document serves as
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
TEMPLATE CASE REPORT FORM. Rapid Response Pharmacovigilance in Palliative Care
 TEMPLATE CASE REPORT FORM Rapid Response Pharmacovigilance in Palliative Care The case report form is to be completed in compliance with PaCCSC Standard Operating Procedures 1 Staff email: Participant
TEMPLATE CASE REPORT FORM Rapid Response Pharmacovigilance in Palliative Care The case report form is to be completed in compliance with PaCCSC Standard Operating Procedures 1 Staff email: Participant
Aclidinium bromide/formoterol Endpoint category. RR Endpoint. [95% CI 2 ] Study. with event
![Aclidinium bromide/formoterol Endpoint category. RR Endpoint. [95% CI 2 ] Study. with event Aclidinium bromide/formoterol Endpoint category. RR Endpoint. [95% CI 2 ] Study. with event](/thumbs/90/102875145.jpg) Resolution by the Federal Joint Committee on an amendment to the Pharmaceutical Directive (AM-RL): Appendix XII Resolutions on the benefit assessment of pharmaceuticals with new active ingredients, in
Resolution by the Federal Joint Committee on an amendment to the Pharmaceutical Directive (AM-RL): Appendix XII Resolutions on the benefit assessment of pharmaceuticals with new active ingredients, in
AWMSG SECRETARIAT ASSESSMENT REPORT. Glycopyrronium bromide (Seebri Breezhaler ) 44 micrograms inhalation powder as hard capsules
 AWMSG SECRETARIAT ASSESSMENT REPORT Glycopyrronium bromide (Seebri Breezhaler ) 44 micrograms inhalation powder as hard capsules Reference number: 1455 FULL SUBMISSION This report has been prepared by
AWMSG SECRETARIAT ASSESSMENT REPORT Glycopyrronium bromide (Seebri Breezhaler ) 44 micrograms inhalation powder as hard capsules Reference number: 1455 FULL SUBMISSION This report has been prepared by
Journal of the COPD Foundation. Journal Club. Chronic Obstructive Pulmonary Diseases:
 85 Journal Club Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Journal Club Ron Balkissoon, MD, MSc, DIH, FRCPC 1 Abbreviations: inhaled corticosteroid, ICS; long acting β 2 -agonist,
85 Journal Club Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Journal Club Ron Balkissoon, MD, MSc, DIH, FRCPC 1 Abbreviations: inhaled corticosteroid, ICS; long acting β 2 -agonist,
A Study of Prescription Pattern in the Management of COPD in a Tertiary Care Hospital.
 DOI: 10.21276/aimdr.2016.2.3.39 Original Article ISSN (O):2395-2822; ISSN (P):2395-2814 A Study of Prescription Pattern in the Management of COPD in a Tertiary Care Hospital. Mazher Maqusood 1, Farhan
DOI: 10.21276/aimdr.2016.2.3.39 Original Article ISSN (O):2395-2822; ISSN (P):2395-2814 A Study of Prescription Pattern in the Management of COPD in a Tertiary Care Hospital. Mazher Maqusood 1, Farhan
Bridges to Excellence Chronic Obstructive Pulmonary Disease Care Recognition Program Guide
 Bridges to Excellence Chronic Obstructive Pulmonary Disease Care Recognition Program Guide Altarum Bridges to Excellence 3520 Green Court, Suite 300 Ann Arbor, MI 48105 bte@altarum.org www.bridgestoexcellence.org
Bridges to Excellence Chronic Obstructive Pulmonary Disease Care Recognition Program Guide Altarum Bridges to Excellence 3520 Green Court, Suite 300 Ann Arbor, MI 48105 bte@altarum.org www.bridgestoexcellence.org
Chronic obstructive pulmonary disease (COPD) is characterized
 RESEARCH Impact of COPD Exacerbation Frequency on Costs for a Managed Care Population Anand A. Dalal, PhD, MBA; Jeetvan Patel, MS; Anna D Souza, BPharm, PhD; Eileen Farrelly, MPH; Saurabh Nagar, MS; and
RESEARCH Impact of COPD Exacerbation Frequency on Costs for a Managed Care Population Anand A. Dalal, PhD, MBA; Jeetvan Patel, MS; Anna D Souza, BPharm, PhD; Eileen Farrelly, MPH; Saurabh Nagar, MS; and
