Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment
|
|
|
- Lily Holland
- 5 years ago
- Views:
Transcription
1 Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: September 21, 2017 Next Review Date: September 2018 Related policies: Policy contains: Macular degeneration. Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin). Vascular endothelial growth factor (VEGF) CP# Vision therapy for visual system disorders ABOUT THIS POLICY: Prestige Health Choice has developed clinical policies to assist with making coverage determinations. Prestige Health Choice s clinical policies are based on guidelines from established industry sources, such as the Centers for Medicare & Medicaid Services (CMS), state regulatory agencies, the American Medical Association (AMA), medical specialty professional societies, and peer-reviewed professional literature. These clinical policies along with other sources, such as plan benefits and state and federal laws and regulatory requirements, including any state- or plan-specific definition of medically necessary, and the specific facts of the particular situation are considered by Prestige Health Choice when making coverage determinations. In the event of conflict between this clinical policy and plan benefits and/or state or federal laws and/or regulatory requirements, the plan benefits and/or state and federal laws and/or regulatory requirements shall control. Prestige Health Choice s clinical policies are for informational purposes only and not intended as medical advice or to direct treatment. Physicians and other health care providers are solely responsible for the treatment decisions for their patients. Prestige Health Choice s clinical policies are reflective of evidence-based medicine at the time of review. As medical science evolves, Prestige Health Choice will update its clinical policies as necessary. Prestige Health Choice s clinical policies are not guarantees of payment. Coverage policy Prestige Health Choice considers the use of ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) to be clinically proven and, therefore, medically necessary when the following criteria are met: Members with a diagnosis of neovascular wet age-related macular degeneration (AMD) - when abnormal blood vessels grow under the retina and macula - with prematurely classic subfoveal choroidal neovascularization (CNV) lesions, where the area of classic CNV occupies at least 50 percent of the entire lesion at the initial visit, as determined by a fluorescein angiogram OR Members present with a subfoveal occult with no classic CNV associated with AMD, when lesions are small (under four disk areas) at initial treatment of within three months prior to 1
2 initial treatment Members present with minimally classic CNV where the area of classic CNV occupies <50 percent of the entire lesion associated with AMD Members present with lesions that have shown evidence of progression within three months prior to initial treatment, as documented by visual acuity (at least five letters on an eye examination chart), lesion growth at least one disk area, or the appearance of blood associated with the lesions Limitations: Coverage determinations are subject to benefit limitations and exclusions as delineated by the state Medicaid authority. The Florida Medicaid website can be accessed at All other uses of OPDT with Visudyne (verteporfin) for the treatment of any other indication are not medically necessary. Alternative covered services: U.S. Food and Drug Administration (FDA)-approved pharmaceuticals, such as Lucentis as approved by the plan. Ongoing monitoring of condition by Ophthalmologist. While no macular degeneration treatment currently approved for use in the United States is likely to completely restore vision lost to this eye disease, some drugs such as Lucentis may be able to slow or prevent additional vision loss or even improve remaining vision to some extent. Background AMD is a common cause of blindness among people over the age of 50 in the western world. Neovascular AMD results when new blood vessels grow across the posterior of the eye, a process known as CNV. These blood vessels often leak blood and serum, causing a blister to form in the retina and eventually damage the macular area of the retina and interfere with central vision. If untreated, the disease results in the distortion of straight lines and, eventually, the loss of central vision. It can be detected in the early, intermediate, and late stage (NEI, 2015). A 2014 analysis of 129,664 individuals age 45 to 85 estimated the prevalence of AMD to be 8.69 percent worldwide, with most cases being early stage. Europeans had a much greater prevalence than did Africans (12.3 versus 7.4 percent). Due to the aging of the population, the projected number of people worldwide with AMD is expected to rise 47 percent, from 196 million to 288 million, from 2020 to 2040 (Wong, 2014). Several risk factors for AMD have been identified, in addition to age and race. Smoking 2
3 doubles AMD risk, and persons with a family history of the disease are at higher risk (NEI, 2015). AMD is detected through a dilated eye exam, which can include a visual acuity test, dilated eye exam, Amsler grid viewing, fluorescein angiogram, and optical coherence tomography (NEI, 2015). There are two types of AMD: atrophic (dry) AMD and exudative (wet) AMD. Atrophic AMD evolves slowly and is the most common form of AMD. This condition is characterized by small yellow lipid debris deposits beneath the retina. It is often a precursor of exudative AMD. The exudative form is distinguished from the atrophic form by serous or hemorrhagic detachment of the retinal pigment epithelium and the development of CNV. The three lesion types associated with exudative AMD are classic, occult and minimally classic. In addition to OPDT, available treatment options for AMD include thermal laser photocoagulation, corticosteroids, and vascular endothelial growth factor (VEGF) antagonists or angiostatics. The safety and effectiveness of each treatment depends on the form and location of the neovascularization. Initially, photocoagulation with a thermal laser was the only viable treatment for patients with AMD. However, this treatment is only beneficial for a small subset of patients with relatively small, welldemarcated lesions and can cause damage to viable neurosensory retinal tissue overlying the treated CNV. This may cause loss of part of the visual field. Beginning in about the year 2000, OPDT with verteporfin (Visudyne, CIBA Vision Corporation, Duluth, GA), was introduced as a treatment for the neovascular form of AMD. CNV is characterized as classic if there is a well-demarcated area of hyperfluorescence early in the fluorescein angiogram, with increased fluorescence caused by pooling of the dye in the late phases of the study. The lesion is characterized as occult if early frames show poorly demarcated areas of hyperfluorescence during fluorescein angiography, with persistent and increased staining in the late phases of the study. This form of CNV is more often associated with subretinal blood, fluid and exudates than the classic form. Lesions can also be mixed when there are both classic and occult neovascular patterns recurrent on the fluorescein angiogram, and recurrent, which occurs in patients with a previous history of leakage or treatment. AMD tends to occur in one eye at a time; however, approximately 50 percent of patients who have neovascular AMD in one eye will develop this condition in their second eye within five years. The progression of this disease varies from a few months to three years. Verteporfin, a benzoporphyrin derivative, is the first treatment to reduce moderate to severe vision loss in macular degeneration. It involves the focus and delivery of laser energy to disease tissue, helping close choroidal neovascular and other active proliferating vessels, while not harming normal retinal tissue (Leung, 2013). Verteporfin is administered by intravenous injection for 10 minutes, followed five minutes later by low-intensity nonthermal light for 83 seconds (Leung, 2013). This drug was first approved by the U.S. Food and Drug Administration in April, 2000 (VPTSG, 2001).. Treatment to combat vascular endothelial growth factor (VEGF) has recently become the first line 3
4 treatment for AMD (Leung, 2013). This process addresses the secretion of high levels of this protein; and injections are made several times a month to block further growth of the protein (NEI, 2015). Searches Prestige Health Choice searched PubMed and the databases of: UK National Health Services Centre for Reviews and Dissemination. Agency for Healthcare Research and Quality s National Guideline Clearinghouse and other evidence-based practice centers. The Centers for Medicare & Medicaid Services (CMS). We conducted searches on August 14, Search terms were: macular degeneration, photodynamic therapy and Visudyne (verteporfin). We included: Systematic reviews, which pool results from multiple studies to achieve larger sample sizes and greater precision of effect estimation than in smaller primary studies. Systematic reviews use predetermined transparent methods to minimize bias, effectively treating the review as a scientific endeavor, and are thus rated highest in evidence-grading hierarchies. Guidelines based on systematic reviews. Economic analyses, such as cost-effectiveness, and benefit or utility studies (but not simple cost studies), reporting both costs and outcomes sometimes referred to as efficiency studies which also rank near the top of evidence hierarchies. Findings The American Academy of Ophthalmology (AAO) guideline states that verteporfin is still an approved option for AMD, even though VEG-F is still the preferred therapy. Data do not support combination therapy of the two (AAO, 2015). This consensus matches that of the European Society of Retina Specialists (Schmidt-Erfurth, 2014). A Hayes review of the three major clinical trials that led to FDA approval of verteporfin found it was well tolerated, as fewer than two percent of subjects withdrew. Subjects given verteporfin in the two trials with >50 percent classic neovascular lesions had a significantly less vision loss (higher proportion of patients with fewer than 15 letters lost within a year) compared to placebo. No statistically significant difference between the two group was evident in patients where classic CNV was present in <50 percent of the area of the entire lesion contrary to what the third study found (Hayes, 2007). A United Kingdom review of 8323 eyes in 7748 patients treated with verteporfin for AMD attempted to understand if administration and results are similar to those in the large clinical trials leading up to approval of this drug, along with effectiveness. Deterioration of visual acuity in practice in patients 4
5 eligible for trials were similar to those observed in trials. The rate of patients treated beyond one year in practice was less than one-half of that recorded in trials. Adverse reactions were reported for 1.4 percent of first visits, fewer than those in the trials. Estimated cost per life-year was similar to the highest estimate in the trials (Reeves, 2012). A Cochrane review of three trials (n=1022) compared verteporfin therapy to controls (five percent dextrose in water). Participants received five treatments over two years. After treatment ended, the risk of losing at least three lines of visual acuity was 23 percent (significantly) less in the intervention group, and 38 percent (significantly) less risk of losing at least six lines. Acute severe visual acuity decrease occurs in about two percent of patients (Wormald, 2007). A Cochrane review of five trials included one that found treatment with the VEGF drug ranibizumab resulted in fewer subjects with loss of at least 15 letters compared with verteporfin. In addition, the combination of the two therapies was more effective compared to verteporfin alone (Vedula, 2008). A systematic review of verteporfin in photodynamic therapy for AMD by lesion subtype showed a strong response in patients with 100 percent classic lesions. However, the treatment showed no benefit in 100 percent occult lesions with no classic component, enabling the European Medicines Evaluation Agency rescinded its approval for verteporfin as a treatment for this type of lesion (Cruess, 2009). One concern about any therapy for AMD is long-term recurrence. One study of 68 eyes found a 52.7 percent recurrence after three years, which was within the percent range of a systematic review of 48 studies (Wong, 2015). In recent years, more trials on AMD treatment have addressed VEGF therapy, which generally produces outcomes superior to verteporfin, as VEGF became the more-used therapy (in Japan, and likely other developed nations), starting in 2009 (Kume, 2016). A review of patients in a phase III clinical trial determined that after 12 months, ranibizumab therapy exceeded outcomes for verteporfin photodynamic therapy for AMD, specifically the proportion losing < 15 letters, proportion gaining >15 letters, and average change from baseline visual acuity (Kaiser, 2007). A systematic review of 10 Randomized Controlled Trials found that verteporfin therapy for AMD produced better outcomes (measured in visual gain or loss) compared to controls, but not compared to the VEGF drug ranibizumab (Virgili, 2011). Photodynamic therapy has been combined with VEGF treatment. A Cochrane review of 12 Randomized Controlled Trials (n=5496) comparing VEGF (using any of three drugs) with photodynamic therapy or sham treatment found that more subjects in each type of VEGF treatment resulted in more with an increase of at least 15 letters, and more with a vision of 20/200 or better (Solomon, 2014). Even a study comparing VEGF with and without photodynamic therapy concluded that monotherapy yields improved visual acuity after one year of treatment, in terms of the percent who gained at least 15 letters of visual acuity (Tong, 2016). 5
6 One review of six trials that compared ranibizumab monotherapy to a combination with photodynamic therapy showed no difference between the two groups for 1) central retinal thickness reduction; 2) number of patients with >0 lines gained; 3) tolerance; and 4) adverse events. Monotherapy actually had more patients with three or more lines gained and better visual acuity correction (Si, 2014). A value-based medicine analysis compared laser photocoagulation, intravitreal pegaptanib therapy, and photodynamic therapy (with verteporfin) for treating classic subfoveal choroidal neovascularization. Using subjects from the large Phase III trials, photodynamic therapy/verteporfin had the greatest improvement in quality-adjusted life years (8.1 percent), significantly greater than 5.9 percent for pegaptanib) and 4.4 percent for laser photocoagulation (Brown, 2007). Another economic analysis concluded that verteporfin for AMD is more cost effective than conventional macular laser; pegaptanib is likely more cost effective than verteporfin, and ranibizumab is effective but at an unacceptably high cost per quality-adjusted life year (Hodge, 2010). Policy updates: A total of six guidelines/other and 16 peer-reviewed references were added to this policy in 2017; a total of two guidelines/other and 17 peer-reviewed references were removed. Summary of clinical evidence: Citation Solomon (2014) Content, Methods, Recommendations Key points: Investigation of anti-vegf agents as a treatment for AMD Cochrane review of 12 trials (n=5496), investigating VEGF; verteporfin prevented clinically significant vision loss, but offered no significant chance for improving vision. 3 of 12 studies compared VEGF (ramibizumab) with verteporfin or sham treatment. Fewer subjects with VEGF lost <15 letters after 12 months. Reeves (2012) Key points: Improvement of visual acuity after verteporfin treatment compared to persons eligible for prior study Trial in UK involving 8323 eyes, 7748 patients. Comparison of visual loss in verteporfin photodynamic therapy-treated eyes with those eligible for an earlier trial, using verteporfin. No difference in visual loss between the two groups. Verteporfin group averaged reduction of 9.9 letters after one year, significantly more than the 9.9 in the earlier group. Verteporfin not prime choice for monotherapy for AMD, but has potential as monotherapy in the management of vascular malformations of the retina and choroid and with trials underway in neovascularization due to myopia and polypoidal choroidopathy. Virgili (2011) Key points: 6
7 Citation Analysis of mixed treatments for AMD Kaiser (2007) Content, Methods, Recommendations A mixed meta-analysis of sham treatments vs. interventions; 10 Randomized Controlled Trials (n=3108); 4 of these included verteporfin (n=1124). Visual loss defined as loss or gain of at least 15 lines, measured in monthly increments. Ranibizumab is superior to verteporfin in visual loss. Key points: Visual acuity changes for AMD, ranibizumab vs. verteporfin photodynamic therapy Results of Phase III trial, comparing number of patients losing at least 15 lines, gaining at least 15 lines, and overall visual acuity after 12 months. Ranibizumab superior to verteporfin in all three categories. Results consistent at 24 months. References Professional society guidelines/other: American Academy of Ophthalmology (AAO). -Age-related macular degeneration, AAO Retina/Vitreous PPP Panel. San Francisco CA: AAO, updated January Accessed August 14, American Academy of Ophthalmology (AAO) Retina/Vitreous Panel. Retina Summary Benchmarks for Preferred Practice Pattern Guideline. San Francisco CA: AAO, October Accessed August 14, 2017.Androudi S, Dastridou A, Pharmakakis N, et al. Guidelines for the Management of Wet Age- Related Macular Degeneration: Recommendations from a Panel of Greek Experts. Adv Ther. 2016;33: Leung RS, Blumenkranz MS. Thermal Laser Photocoagulation. Age-Related Macular Degeneration and Related Disease. American Academy of Ophthalmology (AAO). October 22, Accessed August 14, National Eye Institute (NEI). Facts About Age-Related Macular Degeneration. Bethesda MD: NEI, Accessed August 16, Royal College of Ophthalmologists (RCO). Age-Related Macular Degeneration. Guidelines for Management. London: RCO, September Accessed August 17, Schmidt-Erfurth U, Chong V, Loewenstein A, et al. Guidelines for the management of neovascular agerelated macular degeneration by the European Society of Retina Specialists (EURETINA). Br J 7
8 Ophthalmol. 2014;98(9): Peer-reviewed references: Brown GC, Brown MM, Brown HC, Kindermann S, Sharma S. A value-based medicine comparison of interventions for subfoveal neovascular macular degeneration. Ophthalmology. 2007;114(6): Cruess AF, Zlateva G, Pleil AM, Wirostko B. Photodynamic therapy with verteporfin in age-related macular degeneration: a systematic review of efficacy, safety, treatment modifications and pharmacoeconomic properties. Acta Ophthalmol. 2009;87(2): Hayes Inc. Photodynamic therapy for verteporfin (Visudyne) for neovascular age-related macular degeneration. Lansdale PA: Hayes, Inc., Hodge W, Brown A, Kymes S. Pharmacologic management of neovascular age-related macular degeneration: systematic review of economic evidence and primary economic evaluation. Can J Ophthalmol. 2010;45(3): Kaiser PK, Brown DM, Zhang K, et al. Ranibizumab for predominantly classic neovascular age-related macular degeneration: subgroup analysis of first-year ANCHOR results. Am J Ophthalmol. 2007;144(6): Kume A, Oshiro T, Sakurada Y, Kikushima W, Yoneyama S, Kashiwagi K. Treatment patterns and health care costs for age-related macular degeneration in Japan: an analysis of national insurance claims data. Ophthalmology. 2016;123(6): Reeves BC, Harding SP, Langham J. Verteporfin photodynamic therapy for neovascular age-related macular degeneration: cohort study for the UK. Health Technol Assess. 2012;16(6):i-xii, Si JK, Tang K, Bi HS, Guo DD, et al. Combination of ranibizumab with photodynamic therapy vs ranibizumab monotherapy in the treatment of age-related macular degeneration: a systematic review and meta-analysis of randomized controlled trials. Int J Ophthalmol. 2014;7(3): Solomon SD, Lindsley K, Vedula SS, Kryzstolik MG, Hawkins BS. Anti-vascular endothelial growth factor for neovascular age-related macular degeneration. Cochrane Database Syst Rev. 2014;(8):CD Tong Y, Zhao KK, Feng D. Comparison of the efficacy of anti-vegf monotherapy versus PDT and intravitreal anti-vegf combination treatment in AMD: a Meta-analysis and systematic review. Int J Ophthalmol. 2016;9(7): Vedula SS, Krzystolik MG. Antiangiogenic therapy with anti-vascular endothelial growth factor modalities for neovascular age-related macular degeneration. Cochrane Database Syst Rev. 8
9 2008;(2):CD Verteporfin in Photodynamic Therapy Study Group (VPTSG). Photodynamic therapy of subfoveal choroidal neovascularization in pathologic myopia with verteporfin. 1-year results of a randomized clinical trial--vip report no. 1. Ophthalmology. 2001;108(5): Virgili G, Novielli N, Menchini F, Murro V, Giacomelli G. Pharmacological treatments for neovascular age-related macular degeneration: can mixed treatment comparison meta-analysis be useful? Curr Drug Targets. 2011;12(2): Wong WL, Xu X, Xiang L, et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. The Lancet Global Health. 2014;2(2):e106 e116. Wong CW, Cheung CM, Mathur R, et al. Three-year results of polypoidal choroidal vasculopathy treated with photodynamic therapy: retrospective study and systematic review. Retina. 2015;35(8): Wormald R, Evans J, Smeeth L, Henshaw K. Photodynamic therapy for neovascular age-related macular degeneration. Cochrane Database Syst Rev. 2007;(3):CD CMS National Coverage Determination (NCDs): Ocular Photodynamic Therapy (OPT). Effective April 3, Medicare+Coverage+Database.+You+may+only+view+the+page+you+attempted+to+access+via+normal +usage+of+the+medicare+coverage+database.&bc=gaaaabaaaaaaaa%3d%3d&. Accessed August 14, Medicare Statement For patients with age-related macular degeneration (AMD), verteporfin is only covered with a diagnosis of neovascular AMD with predominately classic subfoveal CNV lesions (where the area of classic CNV occupies > 50 percent of the area of the entire lesion) at the initial visit as determined by an FA. Subsequent follow-up visits will require either an optical coherence tomography or an FA to access treatment response. OPT with verteporfin is covered for the above indication and will remain non-covered for all other indications related to AMD (section 80.2 of National Coverage Determination [NCD]). OPT with Verteporfin for use in non-amd conditions is eligible for coverage through individual Medicare Administrative Contractor discretion. Local Coverage Determinations (LCDs): L33705 Ocular Photodynamic Therapy (OPT) with Verteporfin. Effective 10/01/ Accessed August 14,
10 Commonly submitted codes Below are the most commonly submitted codes for the service(s)/item(s) subject to this policy. This is not an exhaustive list of codes. Providers are expected to consult the appropriate coding manuals and bill accordingly. CPT Code Description Comment Destruction of localized lesion of choroid (e.g., choroidal neovascularization); photodynamic therapy (includes intravenous infusion) Destruction of localized lesion of choroid (eg, choroidal neovascularization); photodynamic therapy, second eye, at single session (List separately in addition to code for primary eye treatment) ICD 10 Code Description Comment B39.4 Histoplasmosis capsulati, unspecified B39.5 Histoplasmosis duboisii B39.9 Histoplasmosis, unspecified H32 H H Chorioretinal disorders in diseases classified elsewhere [Histoplasmosis] Retinal neovascularization H35.30 Macular degeneration, unspecified H H H H H H H44.20 H44.23 Nonexudative age-related macular degeneration Exudative age-related macular degeneration Central serous retinopathy [serous chorioretinopathy] Degenerative myopia HCPCS Level II Code N/A Description Comment 10
Clinical Policy Title: Ocular photodynamic therapy with Visudyne (verteporfin) for macular degeneration treatment
 Clinical Policy Title: Ocular photodynamic therapy with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review Date: August
Clinical Policy Title: Ocular photodynamic therapy with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review Date: August
Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment
 Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review
Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187
 Clinical Policy: (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Neovascular (WET) Macular Degeneration Treatment Reference Number: CP.MP.283
 Clinical Policy: Neovascular (WET) Macular Degeneration Treatment Reference Number: CP.MP.283 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Clinical Policy: Neovascular (WET) Macular Degeneration Treatment Reference Number: CP.MP.283 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
MEDICAL POLICY SUBJECT: PHOTODYNAMIC THERAPY FOR EFFECTIVE DATE: 07/20/00 SUBFOVEAL CHOROIDAL NEOVASCULARIZATION
 MEDICAL POLICY SUBJECT: PHOTODYNAMIC THERAPY FOR PAGE: 1 OF: 8 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: PHOTODYNAMIC THERAPY FOR PAGE: 1 OF: 8 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.20 Intraocular Radiation Therapy for Age-Related Macular Degeneration Photodynamic Therapy for Choroidal
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.20 Intraocular Radiation Therapy for Age-Related Macular Degeneration Photodynamic Therapy for Choroidal
Although photocoagulation and photodynamic PROCEEDINGS PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION *
 PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
Despite our growing knowledge of the
 OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
Photodynamic Therapy for Choroidal Neovascularization
 Photodynamic Therapy for Choroidal Neovascularization Policy Number: 9.03.08 Last Review: 10/2014 Origination: 10/2000 Next Review: 10/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Photodynamic Therapy for Choroidal Neovascularization Policy Number: 9.03.08 Last Review: 10/2014 Origination: 10/2000 Next Review: 10/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Description. Section: Other Effective Date: July 15, Subsection: Vision Original Policy Date: December 7, 2011 Subject: Page: 1 of 23
 Last Review Status/Date: June 2015 Page: 1 of 23 Description Photodynamic therapy (PDT) is a treatment modality designed to selectively occlude ocular choroidal neovascular tissue. The therapy is a 2-step
Last Review Status/Date: June 2015 Page: 1 of 23 Description Photodynamic therapy (PDT) is a treatment modality designed to selectively occlude ocular choroidal neovascular tissue. The therapy is a 2-step
SUMMARY. Heather Casparis, MD,* and Neil M. Bressler, MD MARINA AND ANCHOR
 The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis)
 Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Original Policy Date
 MP 9.03.08 Photocoagulation of Macular Drusen Medical Policy Section Miscellaneous Policies Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return
MP 9.03.08 Photocoagulation of Macular Drusen Medical Policy Section Miscellaneous Policies Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return
CMS Manual System Pub Medicare National Coverage Determinations
 CMS Manual System Pub. 100-03 Medicare National Coverage Determinations Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 9 Date: APRIL 1, 2004 CHANGE
CMS Manual System Pub. 100-03 Medicare National Coverage Determinations Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 9 Date: APRIL 1, 2004 CHANGE
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy Title: Cardiac rehabilitation
 Clinical Policy Title: Cardiac rehabilitation Clinical Policy Number: 04.02.02 Effective Date: September 1, 2013 Initial Review Date: February 19, 2013 Most Recent Review Date: February 6, 2018 Next Review
Clinical Policy Title: Cardiac rehabilitation Clinical Policy Number: 04.02.02 Effective Date: September 1, 2013 Initial Review Date: February 19, 2013 Most Recent Review Date: February 6, 2018 Next Review
The limited number of currently approved
 CLINICAL TRIALS OF VERTEPORFIN AND PEGAPTANIB: WHAT ARE THE RESULTS? * William F. Mieler, MD ABSTRACT Currently available treatment options for the management of choroidal neovascularization (CNV) in age-related
CLINICAL TRIALS OF VERTEPORFIN AND PEGAPTANIB: WHAT ARE THE RESULTS? * William F. Mieler, MD ABSTRACT Currently available treatment options for the management of choroidal neovascularization (CNV) in age-related
Clinical Trials Related to Age Related Macular Degeneration
 Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad
Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad
Ophthalmologic Policy. Vascular Endothelial Growth Factor (VEGF) Inhibitors
 Ophthalmologic Policy UnitedHealthcare Commercial Drug Policy Vascular Endothelial Growth Factor (VEGF) Inhibitors Policy Number: 2016D0042H Effective Date: October 1, 2016 Table of Contents Page INSTRUCTIONS
Ophthalmologic Policy UnitedHealthcare Commercial Drug Policy Vascular Endothelial Growth Factor (VEGF) Inhibitors Policy Number: 2016D0042H Effective Date: October 1, 2016 Table of Contents Page INSTRUCTIONS
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 2Q17 April May
 BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
Clinical Policy: Implantable Miniature Telescope for Age Related Macular Degeneration Reference Number: CP.MP.517
 Clinical Policy: Implantable Miniature Telescope for Age Related Macular Reference Number: CP.MP.517 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Clinical Policy: Implantable Miniature Telescope for Age Related Macular Reference Number: CP.MP.517 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Intraocular Radiation Therapy for Age-Related Macular Degeneration
 Medical Policy Manual Medicine, Policy No. 134 Intraocular Radiation Therapy for Age-Related Macular Degeneration Next Review: April 2019 Last Review: June 2018 Effective: August 1, 2018 IMPORTANT REMINDER
Medical Policy Manual Medicine, Policy No. 134 Intraocular Radiation Therapy for Age-Related Macular Degeneration Next Review: April 2019 Last Review: June 2018 Effective: August 1, 2018 IMPORTANT REMINDER
Michael P. Blair, MD Retina Consultants, Ltd Libertyville/Des Plaines, Illinois Clinical Associate University of Chicago 17 October 2015
 Michael P. Blair, MD Retina Consultants, Ltd Libertyville/Des Plaines, Illinois Clinical Associate University of Chicago 17 October 2015 So What Parts of the Eye Retina are Affected by VHL Neural tissue
Michael P. Blair, MD Retina Consultants, Ltd Libertyville/Des Plaines, Illinois Clinical Associate University of Chicago 17 October 2015 So What Parts of the Eye Retina are Affected by VHL Neural tissue
Intraocular Radiation Therapy for Age-Related Macular Degeneration
 Intraocular Radiation Therapy for Age-Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2014 Origination: 9/2008 Next Review: 9/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue
Intraocular Radiation Therapy for Age-Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2014 Origination: 9/2008 Next Review: 9/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue
Photodynamic Therapy (PDT) for agerelated
 Page 1 of 5 Photodynamic Therapy (PDT) for agerelated eye conditions Introduction This leaflet gives you information about Photodynamic Therapy (PDT) for the age--related eye conditions macular degeneration
Page 1 of 5 Photodynamic Therapy (PDT) for agerelated eye conditions Introduction This leaflet gives you information about Photodynamic Therapy (PDT) for the age--related eye conditions macular degeneration
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Clinical Policy: Aflibercept (Eylea) Reference Number: CP.PHAR.184
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Fundamentals of Retina Coding
 Fundamentals of Retina Coding Presented by: Joy Woodke, COE, OCS Sunday, April 2, 2017 ASRS Business of Retina Meeting Dallas, TX American Academy of Ophthalmic Executives Financial Disclosure Joy Woodke,
Fundamentals of Retina Coding Presented by: Joy Woodke, COE, OCS Sunday, April 2, 2017 ASRS Business of Retina Meeting Dallas, TX American Academy of Ophthalmic Executives Financial Disclosure Joy Woodke,
Clinical Policy Title: Zoster (shingles) vaccine
 Clinical Policy Title: Zoster (shingles) vaccine Clinical Policy Number: 18.02.10 Effective Date: June 1, 2018 Initial Review Date: April 10, 2018 Most Recent Review Date: May 1, 2018 Next Review Date:
Clinical Policy Title: Zoster (shingles) vaccine Clinical Policy Number: 18.02.10 Effective Date: June 1, 2018 Initial Review Date: April 10, 2018 Most Recent Review Date: May 1, 2018 Next Review Date:
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 6.01.10 Stereotactic Radiosurgery and Stereotactic Body Radiotherapy 8.01.10 Charged-Particle (Proton
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 6.01.10 Stereotactic Radiosurgery and Stereotactic Body Radiotherapy 8.01.10 Charged-Particle (Proton
Clinical Policy Title: Genicular nerve block
 Clinical Policy Title: Genicular nerve block Clinical Policy Number: 14.01.10 Effective Date: October 1, 2017 Initial Review Date: September 21, 2017 Most Recent Review Date: October 19, 2017 Next Review
Clinical Policy Title: Genicular nerve block Clinical Policy Number: 14.01.10 Effective Date: October 1, 2017 Initial Review Date: September 21, 2017 Most Recent Review Date: October 19, 2017 Next Review
ANTIVASCULAR ENDOTHELIAL GROWTH FACTOR
 THE ROLE OF RANIBIZUMAB * Michael L. Klein, MD ABSTRACT The positive 1-year results of ranibizumab trials reported at the 23rd Annual Meeting of the American Society of Retina Specialists have generated
THE ROLE OF RANIBIZUMAB * Michael L. Klein, MD ABSTRACT The positive 1-year results of ranibizumab trials reported at the 23rd Annual Meeting of the American Society of Retina Specialists have generated
Clinical Policy Title: Strep testing
 Clinical Policy Title: Strep testing Clinical Policy Number: 07.01.09 Effective Date: December 1, 2017 Initial Review Date: October 19, 2017 Most Recent Review Date: November 16, 2017 Next Review Date:
Clinical Policy Title: Strep testing Clinical Policy Number: 07.01.09 Effective Date: December 1, 2017 Initial Review Date: October 19, 2017 Most Recent Review Date: November 16, 2017 Next Review Date:
Age-Related Macular Degeneration (AMD)
 Age-Related Macular Degeneration (AMD) What is the Macula? What is Dry AMD (Age-related Macular Degeneration)? Dry AMD is an aging process that causes accumulation of waste product under the macula leading
Age-Related Macular Degeneration (AMD) What is the Macula? What is Dry AMD (Age-related Macular Degeneration)? Dry AMD is an aging process that causes accumulation of waste product under the macula leading
2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY. MEASURE TYPE: Process
 Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY
Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY
There are no published randomized, double-blind trials comparing aflibercept to other therapies in neovascular AMD.
 Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Age-Related. macular degeneration.
 Age-Related Macular Degeneration This pamphlet is designed to help people with age-related macular degeneration and their families better understand the disease. It describes the causes, symptoms, diagnosis,
Age-Related Macular Degeneration This pamphlet is designed to help people with age-related macular degeneration and their families better understand the disease. It describes the causes, symptoms, diagnosis,
VERTEPORFIN IN PHOTODYNAMIC THERAPY STUDY GROUP
 Verteporfin Therapy of Subfoveal Choroidal Neovascularization in Age-related Macular Degeneration: Two-year Results of a Randomized Clinical Trial Including Lesions With Occult With No Classic Choroidal
Verteporfin Therapy of Subfoveal Choroidal Neovascularization in Age-related Macular Degeneration: Two-year Results of a Randomized Clinical Trial Including Lesions With Occult With No Classic Choroidal
Intraocular Radiotherapy for Age- Related Macular Degeneration
 Intraocular Radiotherapy for Age- Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2017 Origination: 9/2008 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Intraocular Radiotherapy for Age- Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2017 Origination: 9/2008 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration
 A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
Since /01/2014 Private practice Ophthalmology, Retinal Medical Specialty and AMD, private office
 Corinne GONZALEZ Ophthalmology (MD, Ph.D ) Retinal Medical Specialist SELARL CABINET DOCTEUR GONZALEZ 27, Boulevard des Minimes 31 200 Toulouse! 05 34 40 77 30 e-mail : cabinet.dr.gonzalez@wanadoo.fr Fax
Corinne GONZALEZ Ophthalmology (MD, Ph.D ) Retinal Medical Specialist SELARL CABINET DOCTEUR GONZALEZ 27, Boulevard des Minimes 31 200 Toulouse! 05 34 40 77 30 e-mail : cabinet.dr.gonzalez@wanadoo.fr Fax
CASE STUDIES CURRENT AND FUTURE TREATMENT OPTIONS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Discussion led by Neil M.
 CURRENT AND FUTURE TREATMENT OPTIONS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Discussion led by Neil M. Bressler, MD Dr Bressler: The overview articles in this monograph summarize safety, efficacy,
CURRENT AND FUTURE TREATMENT OPTIONS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Discussion led by Neil M. Bressler, MD Dr Bressler: The overview articles in this monograph summarize safety, efficacy,
Stabilization of visual acuity with photodynamic therapy in eyes with chorioretinal anastomoses
 Graefe s Arch Clin Exp Ophthalmol (2004) 242:368 376 CLINICAL INVESTIGATION DOI 10.1007/s00417-003-0844-0 Rufino M. Silva José R. Faria de Abreu António Travassos José G. Cunha-Vaz Stabilization of visual
Graefe s Arch Clin Exp Ophthalmol (2004) 242:368 376 CLINICAL INVESTIGATION DOI 10.1007/s00417-003-0844-0 Rufino M. Silva José R. Faria de Abreu António Travassos José G. Cunha-Vaz Stabilization of visual
Corinne GONZALEZ Ophthalmology (MD, Ph.D ) Retinal Medical Specialist
 Corinne GONZALEZ Ophthalmology (MD, Ph.D ) Retinal Medical Specialist PRESENT PROFESSIONAL POSITION OPHTALMOLOGIST DOCTOR, Private practice Retinal medical Specialist, DMLA in particular Installation year
Corinne GONZALEZ Ophthalmology (MD, Ph.D ) Retinal Medical Specialist PRESENT PROFESSIONAL POSITION OPHTALMOLOGIST DOCTOR, Private practice Retinal medical Specialist, DMLA in particular Installation year
OPHTHALMOLOGIC POLICY: VASCULAR ENDOTHELIAL GROWTH FACTOR (VEGF) INHIBITORS
 UnitedHealthcare Commercial Medical Benefit Drug Policy OPHTHALMOLOGIC POLICY: VASCULAR ENDOTHELIAL GROWTH FACTOR (VEGF) INHIBITORS Policy Number: PHA020 Effective Date: September 1, 2018 Table of Contents
UnitedHealthcare Commercial Medical Benefit Drug Policy OPHTHALMOLOGIC POLICY: VASCULAR ENDOTHELIAL GROWTH FACTOR (VEGF) INHIBITORS Policy Number: PHA020 Effective Date: September 1, 2018 Table of Contents
Photodynamic therapy (PDT) for Central Serous Retinopathy (CSR)/ Polypoidal Choroidal Vasculopathy (PCV)
 Photodynamic therapy (PDT) for Central Serous Retinopathy (CSR)/ Polypoidal Choroidal Vasculopathy (PCV) About photodynamic therapy for Retinal Conditions The macula is the central part of the retina at
Photodynamic therapy (PDT) for Central Serous Retinopathy (CSR)/ Polypoidal Choroidal Vasculopathy (PCV) About photodynamic therapy for Retinal Conditions The macula is the central part of the retina at
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Fluorescein Angiography
 Last revision: October 2011 by Luis Arias Fluorescein Angiography Authors: Luis Arias, MD Hospital Universitari de Bellvitge - University of Barcelona. Spain Jordi Monés, MD Institut de la Màcula i de
Last revision: October 2011 by Luis Arias Fluorescein Angiography Authors: Luis Arias, MD Hospital Universitari de Bellvitge - University of Barcelona. Spain Jordi Monés, MD Institut de la Màcula i de
Description. Section: Other Effective Date: July 15, Subsection: Vision Original Policy Date: September 13, 2011 Subject: Page: 1 of 18
 Last Review Status/Date: June 2014 Page: 1 of 18 Description Angiogenesis inhibitors (e.g., ranibizumab, bevacizumab, pegaptanib, aflibercept) are being evaluated for the treatment of disorders of choroidal
Last Review Status/Date: June 2014 Page: 1 of 18 Description Angiogenesis inhibitors (e.g., ranibizumab, bevacizumab, pegaptanib, aflibercept) are being evaluated for the treatment of disorders of choroidal
The Natural History of Occult Choroidal Neovascularisation Associated With Age-related Macular Degeneration. A Systematic Review
 Review Article 145 The Natural History of Occult Choroidal Neovascularisation Associated With Age-related Macular Degeneration. A Systematic Review Antonio Polito, 1 MD, Miriam Isola, 2 MHS, Paolo Lanzetta,
Review Article 145 The Natural History of Occult Choroidal Neovascularisation Associated With Age-related Macular Degeneration. A Systematic Review Antonio Polito, 1 MD, Miriam Isola, 2 MHS, Paolo Lanzetta,
MONTHLY ADMINISTERED INTRAVITREOUS PHARmacotherapy
 Polypoidal Choroidal Vasculopathy Masquerading as Neovascular Age-Related Macular Degeneration Refractory to Ranibizumab ALEXANDROS N. STANGOS, JAGDEEP SINGH GANDHI, JAYASHREE NAIR-SAHNI, HEINRICH HEIMANN,
Polypoidal Choroidal Vasculopathy Masquerading as Neovascular Age-Related Macular Degeneration Refractory to Ranibizumab ALEXANDROS N. STANGOS, JAGDEEP SINGH GANDHI, JAYASHREE NAIR-SAHNI, HEINRICH HEIMANN,
Management of Neovascular AMD
 Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
Clinical Policy Title: Genetic testing for G1691A polymorphism factor V Leiden
 Clinical Policy Title: Genetic testing for G1691A polymorphism factor V Leiden Clinical Policy Number: 05.01.03 Effective Date: January 1, 2016 Initial Review Date: July 15, 2015 Most Recent Review Date:
Clinical Policy Title: Genetic testing for G1691A polymorphism factor V Leiden Clinical Policy Number: 05.01.03 Effective Date: January 1, 2016 Initial Review Date: July 15, 2015 Most Recent Review Date:
Clinical Policy Title: Abdominal aortic aneurysm screening
 Clinical Policy Title: Abdominal aortic aneurysm screening Clinical Policy Number: 08.01.10 Effective Date: August 1, 2017 Initial Review Date: June 22, 2017 Most Recent Review Date: June 5, 2018 Next
Clinical Policy Title: Abdominal aortic aneurysm screening Clinical Policy Number: 08.01.10 Effective Date: August 1, 2017 Initial Review Date: June 22, 2017 Most Recent Review Date: June 5, 2018 Next
Date approved: 04/18/18. Approved by: Pharmacy and Therapeutics Quality Management Subcommittee Effective Date: Department of Origin: Pharmacy
 Integrated Healthcare Services and Criteria Document: Reference #: PC/V001 Page: 1 of 9 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services,
Integrated Healthcare Services and Criteria Document: Reference #: PC/V001 Page: 1 of 9 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services,
Subgroup Analysis of the MARINA Study of Ranibizumab in Neovascular Age-Related Macular Degeneration
 Subgroup Analysis of the MARINA Study of in Neovascular Age-Related Macular Degeneration David S. Boyer, MD, 1 Andrew N. Antoszyk, MD, 2 Carl C. Awh, MD, 3 Robert B. Bhisitkul, MD, PhD, 4 Howard Shapiro,
Subgroup Analysis of the MARINA Study of in Neovascular Age-Related Macular Degeneration David S. Boyer, MD, 1 Andrew N. Antoszyk, MD, 2 Carl C. Awh, MD, 3 Robert B. Bhisitkul, MD, PhD, 4 Howard Shapiro,
CLINICAL SCIENCES. Verteporfin Therapy for Subfoveal Choroidal Neovascularization in Age-Related Macular Degeneration
 CLINICAL SCIENCES Verteporfin Therapy for Subfoveal Choroidal Neovascularization in Age-Related Macular Degeneration Three-Year Results of an Open-Label Extension of 2 Randomized Clinical Trials TAP Report
CLINICAL SCIENCES Verteporfin Therapy for Subfoveal Choroidal Neovascularization in Age-Related Macular Degeneration Three-Year Results of an Open-Label Extension of 2 Randomized Clinical Trials TAP Report
Eylea (aflibercept) Document Number: IC-0026
 Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
FEEDER VESSEL TREATMENT
 THERAPEUTIC OPTIONS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Discussion led by Donald J. D'Amico, MD Dr D Amico: The overview articles in this monograph highlight findings from clinical trials
THERAPEUTIC OPTIONS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Discussion led by Donald J. D'Amico, MD Dr D Amico: The overview articles in this monograph highlight findings from clinical trials
Instudies of vascular endothelial growth factor
 MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
Long-term Management of AMD. Motasem Al-latayfeh, MD Assistant Prof. Ophthalmology Hashemite University Jordan
 Long-term Management of AMD Motasem Al-latayfeh, MD Assistant Prof. Ophthalmology Hashemite University Jordan DEFINITION 1 Age-related macular degeneration (AMD) is a disorder of the macula characterized
Long-term Management of AMD Motasem Al-latayfeh, MD Assistant Prof. Ophthalmology Hashemite University Jordan DEFINITION 1 Age-related macular degeneration (AMD) is a disorder of the macula characterized
Clinical Policy Title: Genetic testing for G1691A polymorphism factor V Leiden
 Clinical Policy Title: Genetic testing for G1691A polymorphism factor V Leiden Clinical Policy Number: 05.01.03 Effective Date: January 1, 2016 Initial Review Date: July 15, 2015 Most Recent Review Date:
Clinical Policy Title: Genetic testing for G1691A polymorphism factor V Leiden Clinical Policy Number: 05.01.03 Effective Date: January 1, 2016 Initial Review Date: July 15, 2015 Most Recent Review Date:
Drug Class Update: Vascular Endothelial Growth Factors
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
VEGF Trap-Eye: New Data Confirm Successes in the Treatment of Age-related Macular Degeneration
 Investor News Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Final Phase 2 Results Presented at Retina Society Meeting VEGF Trap-Eye: New Data Confirm Successes in the Treatment
Investor News Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Final Phase 2 Results Presented at Retina Society Meeting VEGF Trap-Eye: New Data Confirm Successes in the Treatment
Clinical Policy Title: Fluorescence in situ hybridization for cervical cancer screening
 Clinical Policy Title: Fluorescence in situ hybridization for cervical cancer screening Clinical Policy Number: 01.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21, 2015 Most Recent
Clinical Policy Title: Fluorescence in situ hybridization for cervical cancer screening Clinical Policy Number: 01.01.02 Effective Date: April 1, 2015 Initial Review Date: January 21, 2015 Most Recent
Ophthalmology Macular Pathways
 Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
Clinically Significant Macular Edema (CSME)
 Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Retina Conference. Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014
 Retina Conference Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014 Subjective CC/HPI: 64 year old Caucasian female referred by outside ophthalmologist
Retina Conference Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014 Subjective CC/HPI: 64 year old Caucasian female referred by outside ophthalmologist
London Medicines Evaluation Network Review
 London Medicines Evaluation Network Review Evidence for initiating intravitreal bevacizumab for the management of wet age-related macular degeneration (wet-amd) in eyes with vision better than 6/12 November
London Medicines Evaluation Network Review Evidence for initiating intravitreal bevacizumab for the management of wet age-related macular degeneration (wet-amd) in eyes with vision better than 6/12 November
Clinical Policy Title: Abdominal aortic aneurysm screening
 Clinical Policy Title: Abdominal aortic aneurysm screening Clinical Policy Number: 08.01.10 Effective Date: August 1, 2017 Initial Review Date: June 22, 2017 Most Recent Review Date: July 20, 2017 Next
Clinical Policy Title: Abdominal aortic aneurysm screening Clinical Policy Number: 08.01.10 Effective Date: August 1, 2017 Initial Review Date: June 22, 2017 Most Recent Review Date: July 20, 2017 Next
Retinal pigment epithelial tears after intravitreal bevacizumab injection for exudative age-related macular degeneration.
 Thomas Jefferson University Jefferson Digital Commons Wills Eye Institute Papers Wills Eye Institute 4-1-2008 Retinal pigment epithelial tears after intravitreal bevacizumab injection for exudative age-related
Thomas Jefferson University Jefferson Digital Commons Wills Eye Institute Papers Wills Eye Institute 4-1-2008 Retinal pigment epithelial tears after intravitreal bevacizumab injection for exudative age-related
Bayer and Regeneron start additional Phase 3 Study for VEGF Trap-Eye in Wet Age-related Macular Degeneration
 Investor News Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer and Regeneron start additional Phase 3 Study for VEGF Trap-Eye in Wet Age-related Macular Degeneration International
Investor News Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer and Regeneron start additional Phase 3 Study for VEGF Trap-Eye in Wet Age-related Macular Degeneration International
Ranibizumab and pegaptanib for the treatment of agerelated macular degeneration
 Ranibizumab and pegaptanib for the treatment of agerelated Issued: August 2008 last modified: May 2012 guidance.nice.org.uk/ta155 NHS Evidence has accredited the process used by the Centre for Health Technology
Ranibizumab and pegaptanib for the treatment of agerelated Issued: August 2008 last modified: May 2012 guidance.nice.org.uk/ta155 NHS Evidence has accredited the process used by the Centre for Health Technology
Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial
 ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
Clinical Policy Title: Breast cancer index genetic testing
 Clinical Policy Title: Breast cancer index genetic testing Clinical Policy Number: 02.01.22 Effective Date: January 1, 2017 Initial Review Date: October 19, 2016 Most Recent Review Date: October 19, 2016
Clinical Policy Title: Breast cancer index genetic testing Clinical Policy Number: 02.01.22 Effective Date: January 1, 2017 Initial Review Date: October 19, 2016 Most Recent Review Date: October 19, 2016
Clinical Policy Title: Abdominal aortic aneurysm screening
 Clinical Policy Title: Abdominal aortic aneurysm screening Clinical Policy Number: 08.01.10 Effective Date: August 1, 2017 Initial Review Date: June 22, 2017 Most Recent Review Date: June 5, 2018 Next
Clinical Policy Title: Abdominal aortic aneurysm screening Clinical Policy Number: 08.01.10 Effective Date: August 1, 2017 Initial Review Date: June 22, 2017 Most Recent Review Date: June 5, 2018 Next
 measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
CENTRAL SEROUS CHORIORETINOPATHY (CSC) IS
 Association Between the Efficacy of Half-Dose Photodynamic Therapy With Indocyanine Green Angiography and Optical Coherence Tomography Findings in the Treatment of Central Serous Chorioretinopathy MASSIMO
Association Between the Efficacy of Half-Dose Photodynamic Therapy With Indocyanine Green Angiography and Optical Coherence Tomography Findings in the Treatment of Central Serous Chorioretinopathy MASSIMO
Contractor Information. LCD Information. Local Coverage Determination (LCD): Ophthalmic Angiography (Fluorescein and Indocyanine Green) (L34426)
 Local Coverage Determination (LCD): Ophthalmic Angiography (Fluorescein and Indocyanine Green) (L34426) Links in PDF documents are not guaranteed to work. To follow a web link, please use the MCD Website.
Local Coverage Determination (LCD): Ophthalmic Angiography (Fluorescein and Indocyanine Green) (L34426) Links in PDF documents are not guaranteed to work. To follow a web link, please use the MCD Website.
The Foundation WHAT IS THE RETINA?
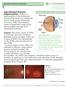 Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
Clinical Policy Title: Ketamine for treatment-resistant depression
 Clinical Policy Title: Ketamine for treatment-resistant depression Clinical Policy Number: 00.02.13 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: January
Clinical Policy Title: Ketamine for treatment-resistant depression Clinical Policy Number: 00.02.13 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: January
Ophthalmic VEGF Inhibitors. Eylea (aflibercept), Macugen (pegaptanib) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
These issues are covered in more detail below.
 26.3.07 Comments from Novartis on the Assessment Report for the Health Technology Appraisal of Pegaptinib and Ranibizumab for the treatment of age-related macular degeneration In general we feel that the
26.3.07 Comments from Novartis on the Assessment Report for the Health Technology Appraisal of Pegaptinib and Ranibizumab for the treatment of age-related macular degeneration In general we feel that the
Current indications for photodynamic therapy in medical retina practice
 réalités Current indications for photodynamic therapy in medical retina practice No. 254 September 2018 OPHTALMOLOGIQUES SUMMARY: In medical retina practice, the use of photodynamic therapy (PDT) revolutionised
réalités Current indications for photodynamic therapy in medical retina practice No. 254 September 2018 OPHTALMOLOGIQUES SUMMARY: In medical retina practice, the use of photodynamic therapy (PDT) revolutionised
Digital Journal of Ophthalmology Digital Journal of Ophthalmology. Abstract
 Original Articles Combined intravitreal ranibizumab and verteporfin photodynamic therapy versus ranibizumab alone for the treatment of age-related macular degeneration Rosalia Giustolisi, MD, Nicoletta
Original Articles Combined intravitreal ranibizumab and verteporfin photodynamic therapy versus ranibizumab alone for the treatment of age-related macular degeneration Rosalia Giustolisi, MD, Nicoletta
Class Update: Vascular Endothelial Growth Factors
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE GUIDANCE EXECUTIVE (GE)
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE GUIDANCE EXECUTIVE (GE) Review of TA68; The clinical and cost effectiveness of photodynamic therapy for age-related macular degeneration TA68 guidance
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE GUIDANCE EXECUTIVE (GE) Review of TA68; The clinical and cost effectiveness of photodynamic therapy for age-related macular degeneration TA68 guidance
Treatment of Age Related Macular Degeneration (AMD) by Intravitreal Injection
 Information for Patients Manchester Royal Eye Hospital Medical Retina Services Treatment of Age Related Macular Degeneration (AMD) by Intravitreal Injection What is age related macular degeneration (AMD)?
Information for Patients Manchester Royal Eye Hospital Medical Retina Services Treatment of Age Related Macular Degeneration (AMD) by Intravitreal Injection What is age related macular degeneration (AMD)?
Evaluation of efficacy of eplerenone in the management of chronic central serous choroidoretinopathy
 Original article: Evaluation of efficacy of eplerenone in the management of chronic central serous choroidoretinopathy Dr. Sushant Madaan* Department of Ophthalmology, NIMS Medical College and Hopsital,Jaipur,
Original article: Evaluation of efficacy of eplerenone in the management of chronic central serous choroidoretinopathy Dr. Sushant Madaan* Department of Ophthalmology, NIMS Medical College and Hopsital,Jaipur,
Vision loss in elderly. Erica Weir, April 2015
 Vision loss in elderly Erica Weir, April 2015 1 Burden Enter nursing homes 3 years earlier Twice the risk of falling 4x the risk of hip fracture Independent risk factor for delirium What are the leading
Vision loss in elderly Erica Weir, April 2015 1 Burden Enter nursing homes 3 years earlier Twice the risk of falling 4x the risk of hip fracture Independent risk factor for delirium What are the leading
NEOVASCULAR AGE-RELATED MACULAR DEGENERation
 Randomized, Double-Masked, -Controlled Trial of Ranibizumab for Neovascular Age-Related Macular Degeneration: PIER Study Year 2 PREMA ABRAHAM, HUIBIN YUE, AND LAURA WILSON PURPOSE: To evaluate efficacy
Randomized, Double-Masked, -Controlled Trial of Ranibizumab for Neovascular Age-Related Macular Degeneration: PIER Study Year 2 PREMA ABRAHAM, HUIBIN YUE, AND LAURA WILSON PURPOSE: To evaluate efficacy
Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular Degeneration
 Ophthalmology Volume 12, Article ID 69641, 8 pages doi:1.1155/12/69641 Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular
Ophthalmology Volume 12, Article ID 69641, 8 pages doi:1.1155/12/69641 Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular
Abicipar pegol for wet age related macular degeneration
 EVIDENCE BRIEFING AUGUST 2018 Abicipar pegol for wet age related macular degeneration NIHRIO ID 7084 NICE ID 9724 Developer/Company Allergan Ltd and Molecular Partners UKPS ID 646978 Licensing and market
EVIDENCE BRIEFING AUGUST 2018 Abicipar pegol for wet age related macular degeneration NIHRIO ID 7084 NICE ID 9724 Developer/Company Allergan Ltd and Molecular Partners UKPS ID 646978 Licensing and market
IQ 532 Micropulse Green Laser treatment for Refractory Chronic Central Serous Retinopathy
 Cronicon OPEN ACCESS EC OPHTHALMOLOGY Case Report IQ 532 Micropulse Green Laser treatment for Refractory Chronic Central Serous Retinopathy Fawwaz Al Mamoori* Medical Retina Department, Eye Specialty Hospital,
Cronicon OPEN ACCESS EC OPHTHALMOLOGY Case Report IQ 532 Micropulse Green Laser treatment for Refractory Chronic Central Serous Retinopathy Fawwaz Al Mamoori* Medical Retina Department, Eye Specialty Hospital,
Vision loss in elderly
 Vision loss in elderly Erica Weir, February 2016 1 Burden Enter nursing homes 3 years earlier Twice the risk of falling 4x the risk of hip fracture Independent risk factor for delirium What are the leading
Vision loss in elderly Erica Weir, February 2016 1 Burden Enter nursing homes 3 years earlier Twice the risk of falling 4x the risk of hip fracture Independent risk factor for delirium What are the leading
