CURRICULUM VITAE. University of Pennsylvania, Philadelphia, PA., M.D. Degree, May 1982.
|
|
|
- Tyler Dennis
- 5 years ago
- Views:
Transcription
1 CURRICULUM VITAE JONATHAN OREN HARRIS, M.D. Marcus Neuroscience Institute Boca Raton Regional Hospital 800 Meadows Road Boca Raton, FL June 2017 Present EDUCATION: University of Miami Affiliated Hospitals, Miami, FL. Internal Medicine ( ) Board Certified. Neurology residency ( ) Board Certified. University of Pennsylvania, Philadelphia, PA., M.D. Degree, May University of Chicago, Chicago, IL, BA Degree, June Post baccalaureate premed courses, POST-GRADUATE TRAINING: National Institute of Health, Bethesda, MD. Senior Staff Fellow, Neuroimmunology, ( ). PREVIOUS EMPLOYMENT Neurologic Consultants, P.A., Pompano Beach, FL Neurologist in Private Practice Oct 1990 May 2017 RESEARCH EXPERIENCE: PHARMACEUTICAL RESEARCH: See page four. Cerebral Vascular Research Center, University of Miami, Miami, FL. ( ) Immunosuppression in experimental stroke. Laboratory of CNS Studies, NINCDS, NIH, Bethesda, MD. Epidemiology of slow virus infections of the CNS, Gerontological Research Institute, Philadelphia, PA. Environmental influences on behavior of demented old people, CURRENT APPOINTMENTS Affiliate Associate Professor of Medicine Charles E. Schmidt College of Medicine, Florida Atlantic University, Boca Raton, FL Clinical Assistant Professor of Neurology - Nova Southeastern University Professional Advisory Committee - South Florida Chapter National MS Society President Broward Stroke Council 1
2 HOSPITAL AFFILIATION: Boca Raton Regional Hospital 800 Meadows Road Boca Raton, FL PERSONAL: Date of birth: Available on request Marital status: Married, five children MEDICAL LICENSE: Florida: Available on request PLATFORM PRESENTATIONS Harris JO, Patronas NJ, Frank JA, et al., "The natural history of multiple sclerosis lesions in relapsing-remitting patients between clinical relapse by serial Gd-DTPA", Society of Magnetic Resonance in Medicine, 9th Annual Scientific Meeting, New York, NY, POSTERS Harris JO, Berger JR, Gregorios JB, Norenberg M, "The spectrum of cerebrovascular disease in the acquired Immunodeficiency syndrome", 113th Annual meeting of the American Neurological Association, Philadelphia, PA, Harris JO, Dietrich WD, Busto R, Ginsberg M, "Busulfan immunosuppression in experimental thrombotic stroke." 14th International Joint Conference on Stroke and Cerebral Circulation, San Antonio, TX, Harris JO, Jacobson S, Mcfarlin DE, "Cutaneous anergy in chronic progressive myelopathy associated with Human T-Lymphotropic Virus Type I in a Caucasian male and his asymptomatic seropositive wife", 114th Annual meeting of the American Neurological Association, New Orleans, LA, Harris JO, Frank JA, Patronas NJ, et al., "Serial Gadolinium enhanced MRI scanning in patients with early relapsing-remitting multiple sclerosis", 115th Annual meeting of the American Neurological Association, Atlanta, GA, PRESENTATIONS: Recent presentations available on request PUBLICATIONS: Berger JR, Harris JO, Gregorios J, Norenberg M, Cerebrovascular disease in AIDS: a case-control study. AIDS, 4(3): Mar Bernoulli C, Masters CL, Gajdusek DC, Gibbs CJ, Harris JO, "Early clinical features of Creutzfeldt - Jakob Disease (Subacute spongiform encephalopathy) In: 2
3 Prusiner SB and Hadlow WJ, (eds.) Slow transmissible diseases of the nervous system, Academic Press: New York, 1979, Volume 1, pages Burke A, Harris JO, Younkin D, et al., "Focal diaschisis and bilateral homologous hyperemia after acute stroke", Neurology 32(4): A196, Harris JO, "Proposal to increase the number of physicians providing care to elderly Americans", Joint hearing before the Subcommittee on Health and Long-term Care and the Subcommittee on Human Services of the select Committee on Aging. House of Representatives May 17, Comm. Pub. No U.S. Government Printing Office: Washington D.C., Harris JO, and Berger JR, "Clinical approach to stupor and coma", Chapter 5 in Bradley WG, Daroff RB, Fenichel GM, and Marsden CD, (eds.), Neurology in clinical practice, Butterworth Publishers: Stoneham, MA., Harris JO, Frank JA, Patronas N, McFarlin DE, McFarland HF, Serial gadoliniumenhanced magnetic resonance imaging scans in patients with early relapsing-remitting multiple sclerosis: implications for clinical trials and natural history. Ann Neurol, 29(5): Harris JO, Marquez J, Swerdloff MA, Magana IA, Listeria brain abscess in the acquired immunodeficiency syndrome [letter] Arch Neurol, 46(3): Masters CL, Harris JO, Gajdusek DC, et al., "Creutzfeldt - Jakob Disease: Patterns of worldwide occurrence and the significance of familial and sporadic clustering", Ann Neurol 5(2): , Masters CL, Harris JO, Gajdusek DC, et al., "Creutzfeldt - Jakob Disease: Patterns of worldwide occurrence" In: Pruisner SB and Hadlow WJ, (eds.) Slow transmissible diseases of the nervous system, Academic Press: New York, 1979, Volume 1, pages McFarland HF, Frank JA, Albert PS, Smith ME, Martin R, Harris JO, Patronas N, Maloni H McFarlin DE, Using gadolinium-enhanced magnetic resonance imaging lesions to monitor disease activity in multiple sclerosis. Ann Neurol, 32(6): Smith ME, Katz DA, Harris JO, Frank JA, Kufta CV, McFarlin DE, Systemic histiocytosis presenting as multiple sclerosis. Ann Neurol, 33(5): RECENT HONORS Gold Coast Magazine Top Doctors South Florida Super Doctors The Leading Physicians of the World 2011 Miami Metro Magazine Top Doctors
4 PHARMACEUTICAL STUDY DRUG EXPERIENCE: Principal Investigator Alzheimer s Disease Alzheimer s Disease - - Protocol : A 26-Week, Randomized, Double-Blind, Placebo-Controlled, Parallel-Group, Multicenter Study of Milameline (CI-979/RU35926) in Patients with Probable Alzheimer s Disease (to be read in conjunction with Protocol ) Alzheimer s Disease - - Protocol : A nonrandomized, Open-Label Extension, Multicenter Study of Milameline (CI-979/RU35926) in Patients with Probable Alzheimer s Disease. Alzheimer s Disease - - Protocol : A 16-Week, Randomized, Double-Blind, Placebo-Controlled, Parallel-Group, Dose-Response Multicenter Study of Tacrine (CI- 970) Once-A-Day Formulation (Tacrine GITS) with a 16-Month Open-Label Extension in Patients with Dementia of the Alzheimer s Type. Alzheimer s Disease - - Protocol : Metrifonate Investigational Nationwide Trial (M.I.N.T.) Alzheimer s Disease - - Protocol CV-2619/PNFP-008: A Randomized, Double-Blind, 12-Month Safety and Efficacy Study of Idebenone (CV-2619) 360 mg tid or Placebo Added to Treatment with Donepezil HCL 10 mg qd in Patients with Probable Alzheimer s Disease. Alzheimer s Disease - - Protocol CV-2619/PNFP-005 A Randomized, Double-Blind, Placebo-Controlled, 12-Month Safety and Efficacy Trial of Idebenone (CV-2619) 360 mg tid in Patients with Probable Alzheimer s Disease. Alzheimer s Disease - - Protocol MEM-MD-10: A randomized, double-blind, placebocontrolled evaluation of the safety and efficacy of Memantine in patients with mild to moderate Dementia of the Alzheimer s type. Alzheimer s Disease - - Protocol MEM-MD-11 A Long Term Extension Study Evaluating the Safety and Tolerability of BID and QD Administration of Memantine In Patients with Mild to Moderate Dementia of the Alzheimer s Type. Alzheimer s Disease - - Protocol GAL-INT-10: A placebo controlled evaluation of Galantamine in the treatment of Alzheimer s Disease: Safety and efficacy of a controlled release formulation. 4
5 Alzheimer s Disease - - Protocol GAL-INT-21: An open label extension trial to assess the long term safety of a controlled released formulation of Galantamine HBR in the treatment of Alzheimer s Dementia. Alzheimer s Disease - - Protocol # : A Phase II Double Blind, Randomized, Dose Ranging, Placebo Controlled, Multicenter, Safety and Efficacy Evaluation of Three Doses of Ns 2330 in Patients with Mild To Moderate Dementia of the Alzheimer s Type. Alzheimer s Disease- - Protocol # ONO-2506POU010: A double-blind, phase II, safety and efficacy evaluation of ONO-2506PO in patients with mild to moderate Alzheimer s Disease. Alzheimer s Disease - - Protocol CENA713D2340: A 48-Week, Multicenter, Randomized, Double Blind, Parallel-Group Evaluation of the Comparative Efficacy, Safety, and Tolerability of Exelon 10 and 15 cm2 Patch in patients with Alzheimer s Disease Showing Cognitive Decline during an initial Open-Label Treatment Phase. Alzheimer s disease - - Protocol ELN : A Phase III, Multicenter, Randomized, Double-Blind, Placebo-Controlled, Parallel Group, Efficacy and Safety Trial of Bapineuzumab )AAB-001, ELN115727) in Patients with Mild to Moderate Alzheimer s disease who are Apolipoprotein E e4 Non-Carriers. Alzheimer s Disease - - Protocol ELN : A Phase III, Multicenter, Randomized, Double-Blind, Placebo-Controlled, Parallel Group, Efficacy and Safety Trial of Bapineuzumab (AAB-001, ELN115727) in Patients with Mild to Moderate Alzheimer s Disease who are Apolipoprotein E e4 Carriers. Alzheimer s Disease - - Protocol H6L-MC-LFAN: Effect of y-secretase Inhibition on the Progression of Alzheimer s Disease: LY versus Placebo Alzheimer s Disease - - Protocol CENA713DUS44: A 24 Week, Prospective, Randomized, Parallel-Group, Double-Blind, Multi-Center Study Comparing the Effects of Rivastigmine Patch 15 cm2 vs. Rivastigmine Patch 5 cm2 on ACTivities of Daily Living and CognitION in Patients with Severe Dementia of the Alzheimer s Type (ACTION). Alzheimer s Disease - - Protocol CENA713DUS44E1: A 24 Week, Open-Lavel Extension to Study CENA713DSU44: A 24 Week, Prospective, Randomized, Parallel- Group, Double-blind, Multi-Center Study Comparing the Effects of Rivastigmine Patch 15 cm2 vs. Rivastigmine patch 5 cm2 on ACTivities of Daily Living and CognitiON in Patients with Severe Dementia of the Alzheimer s Type (ACTION). 5
6 Alzheimer s Disease - - Protocol H6L-MC-LFBF: Open-label Extension for Alzheimer s Disease Patients Who complete One of Two Semagacestat Phase 3 Double- Blind Studies (H6L-MC-LFAN or H6L-MC-LFBC Alzheimer s Disease - - Protocol ELN : A phase III Extension, Multicenter, Double Blind, Long Term Safety and Tolerability Treatment Trial of Bapineuzumab ( AAB-001, ELN115727) in Subjects with Alzheimer s Disease who participated in Study ELN or in Study ELN Alzheimer s Disease - - Protocol 3133K US: A Phase 3, Multicenter, Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Efficacy and Safety Trial of Bapineuzumab (AAB-001, ELN115727) in Subjects With Mild to Moderate Alzheimer Disease Who Are Apolipoprotein E4 Non-Carriers Alzheimer s Disease - - Protocol BP28248: A Phase II, Multicenter, Randomized, double-blind, Parallel-Group. Placebo-Controlled Study to Investigate the Efficacy And Safety Of RO Added To the Background Therapy Of The Acetylcholinesterase Inhibitors Donepezil Or Rivastigmine In Patients With Moderate Severity Alzheimer s Disease. Alzheimer s Disease - - Protocol BN29552 A Phase III, Multicenter, Randomized, Double-Blind, Placebo Controlled, Parallel-Group, Efficacy And Safety Study Of Crenezumab in Patients With Prodromal To Mild Alzheimer s Disease". Assessment Scale Validity Protocol Number CENA713B2314: A 4-Week, Non-Interventional, Cross-Sectional, Multicenter Study to Assess the Validity of Various Assessment Scales Measuring Cognition, Executive Function, Attention, Behavior, and Activities of Daily Living (ADL) in Patients with Mild to Moderate Parkinson;s Disease Dementia (PDD) or Vascular Dementia (VaD). Cerebrovascular disease Acute Ischemic Stroke - - Protocol IX Cervene (Nalmefene) Modification of Outcome in Patients with Acute Ischemic Stroke: A Double-Blind, Randomized, Placebo-Controlled, Parallel-Group, Dose-Comparison Study by 24 Hour Infusion. Acute Ischemic Stroke - - Protocol IX A Phase III, Multi-Center, Double- Blind, Randomized, Placebo-Controlled, Parallel-Group Study to Evaluate the Safety and Efficacy of a 24-Hour Infusion of Cervene in Patients with Acute Ischemic Stroke. Acute Ischemic Stroke - - Protocol LES02: Assessment of the Efficacy and Safety of Eliprodil in Patients With Acute Ischemic Stroke. 6
7 Acute Ischemic Stroke - - Protocol A Phase II/III Multicenter, Double-Blind, Randomized, Placebo-Controlled, Parallel-Group Study to Evaluate the Efficacy. Safety, Tolerability, and Pharmacokinetics of Two Doses of Intravenous Aptiganel Hydrochloride Verses Placebo In Patients with An Acute Ischemic Stroke. Acute Ischemic Stroke - - Protocol CMZ 009 The Clomethiazole Acute Stroke Study in Ischemic Stroke (Class-I): A Double Blind, Parallel Group, Multinational, Multicenter Study of the Safety of IV Clomethiazole Compared to Placebo in Patients with Acute Ischemic Stroke. Acute Ischemic Stroke - - Protocol CMZ 010 The Clomethiazole Acute Stroke Study in Acute Intracerebral Hemorrhage (Class-H): A Double Blind, Parallel Group, Multinational, Multicenter Study of the Safety of IV Clomethiazole Compared to Placebo in Patients with Acute Intracerebral Hemorrhage. Acute Ischemic Stroke - - Protocol CMZ 011 The Clomethiazole Acute Stroke Study in t-pa Treatment Ischemic Stroke (Class -T): A Double Blind, Parallel Group, Multinational, Multicenter Study of the Safety of IV Clomethiazole Compared to Placebo in Patients Treated with t-pa for Acute Ischemic Stroke. Acute Ischemic Stroke - - Protocol GLYA3002, An International, Multi-Center, Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Study to Assess Over 3 Months the Safety, Efficacy and Pharmacoeconomics of an 800 mg Loading Dose and Five 200 mg Maintenance Doses of GV in the Treatment of Patients with a Clinical Diagnosis of Acute Stroke. Acute Ischemic Stroke - - Protocol CN A Double-Blind, Placebo-Controlled, Safety, Efficacy and Dose Response Trial of Three Intravenous Doses of BMS in Patients with acute Stroke. Antiplatelet Drug - - Protocol SB Lotrafiban: Blockade of the GP IIB/IIIA Receptor to Avoid Vascular Occlusion (BRAVO) Stroke - - Protocol GLY30009: A six-month post-stroke follow-up protocol for subjects who have previously completed clinical trial protocol GLYA3002 (an International, Multi-Center, Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Study to Assess over Three Months the Safety, Efficacy and Pharmacoeconomics of an 800mg Loading Dose and Five 200mg Maintenance Doses of GV in the Treatment of Subjects with a Clinical Diagnosis of Acute Stroke). Acute Ischemic Stroke - -Protocol GLY30011: A Multi-Center, Randomized, Double- Blind, Placebo-Controlled, Parallel-Group Study to Assess over 3 Months the Efficacy, using the Stroke Impact Scale, and Safety of an 800mg Loading Dose and Five 200mg Maintenance Doses of GV in the Treatment of Patients with a Clinical Diagnosis of Acute Stroke. 7
8 Post Stroke Study - - Protocol SPARCL: A Double-Blind, Randomized, Placebo-Controlled Study of Atorvastation as Prevention of Cerebrovascular Events in Patients with a previous transient Ischemic Attack (TIA) or Stroke. Acute Ischemic Stroke - -Protocol ARG 251: Randomized, Placebo-Controlled, Three- Treatment Arm Study To Determine The Safety and Efficacy of Argatroban Injection in Patients With Acute Ischemic Stroke (ARGIS 1) Acute Ischemic Stroke - -Protocol 872-CL-004: ARTIST +: A Multicenter, Stratified, Randomized, Double-Blind, Placebo-Controlled Study to Evaluate Neurologic Function and Disability in Patients with Active Ischemic Stroke Given Tissue Plasminogen Activator Plus YM872 or Tissue Plasminogen Activator Plus Placebo. Post Stroke Study - - MATCH Protocol Number EFC7331: Management of Atherothrombosis With Clopidogrel in High-risk Patients With Transient Ischemic Attack or Ischemic Stroke. Post Stroke Study - - Protocol number EFC4505: Clopidogrel For High Atherothrombotic Risk and Ischemic Stabilization, Management and Avoidance. (Charisma) Acute Ischemic Stroke - - Protocol : A Randomized, Double-Blind, Placebo- Controlled Trial to Evaluate the Efficacy, Safety, Tolerability, and Pharmacokinetic/Pharmacodynamic Effects of a Targeted Exposure of Intravenous Repinotan in Patients with Acute Ischemic Stroke. (mrect) Acute Ischemic Stroke - - protocol Number SA-NXY-0007 titled: SAINT II (Stroke- Acute Ischemic-NXY Treatment) A double-blind, randomized, placebo controlled, parallel group, multicenter, phase IIb/II Study to assess the efficacy and safety of intravenous NXY-059 in acute ischemic stroke. Stroke- - Protocol # ONO-2506: A Randomized, double-blind, placebo controlled, multicenter study of the effects of ONO-2506 intravenous infusion on the amelioration of neurological damage and improvement of stroke assessment of scales in patients with acute ischemic stroke. Stroke- - Protocol # SA-NXY-0012: A double-blind, randomized, placebo-controlled, parallel-group, multi-center, phase IIb study to assess the safety and tolerability of 72 hours intravenous infusion of NXY-059 in adult patients with acute intracerebral hemorrhage. Stroke- - Protocol # 9.159: PRoFESS- Prevention Regimen for Effectively avoiding Second Strokes: A Double-blind and placebo controlled study of Aggrenox vs. Clopidogrel, with and without Micardis. 8
9 Stroke - - Protocol NTI-ASP-0502: A Randomized, Double-Blind, Placebo-Controlled Study of Viprinex (Ancrod Injection) in Subjects Beginning Treatment within 6 Hours of the Onset of Acute, Ischemic Stroke. Post Stroke Apathy - - Protocol HPI-001 A Randomized, Controlled Comparison of Nefiracetam with Placebo In The Treatment Of Patients With Post-Stroke Apathy Acute Ischemic Stroke - - Protocol No. NTI-AST-0502 Study of Acute Viprinex for Emergency Stroke: A Randomized, Double-Blind, Placebo-Controlled Study of Ancrod (Viprinex) in Subjects Beginning Treatment within 6 Hours of the Onset of Acute, Ischemic Stroke. Acute Ischemic Stroke - - Protocol NTS-INT06-007: NeuroThera Effectiveness and Safety Trial-2 (NEST-2) A Double Blind, randomized, controlled, parallel group, multicenter, pivotal study to assess the safety and effectiveness of the treatment of acute ischemic stroke with the NeuroThera Laser System within 24 hours from stroke onset. Acute Ischemic Stroke - - Protocol Albumin in Acute Stroke (ALIAS) Trial: A Phase III Randomized Multicenter Clinical Trial of High-Dose Human Albumin Therapy for Neuroprotection in Acute Ischemic Stroke. Post Stroke - - Protocol A Multicenter, Randomized, Double-Blind, Placebo-Controlled Study to Evaluate the Safety and Efficacy of SCH in Addition to Standard of care in Subjects With a History of Atherosclerotic Disease: Thrombin Receptor Antagonist in Secondary Prevention of Atherothrombotic Ischemic Events (TRA 2P TIMI 50). Post Stroke - - Protocol DALF-PS-1003: A Phase 2b Study of Dalfampridine 10 mg Extended Release Tablet in Subjects with Chronic Deficits After Ischemic Stroke. Acute Ischemic Stroke - - Protocol ASBI 801: A Multicenter Observational Study toate the Simplified-Stroke Rehabilitation Assessment of Movement (S_STREAM) Scale in Subjects Obtained Between 24 and 48 Hours of a Nonhemorrhagic Ischemic Stroke. Acute Ischemic Stroke - - Protocol A : A Phase 2 Multicenter, Randomized, Double-Blind, Placebo Controlled Study Of The Safety And Efficacy Of PF In Subjects With Ischemic Stroke. Acute Ischemic Stroke - - Protocol D5134C00001: A Randomized Double-Blind, Multinational Study to Prevent Major Vascular Events with Ticagrelor Compared to Aspirin (ASA) in Patients with Acute Ischaemic Stroke or TIA (SOCRATES- Acute Stroke Or Transient IsChemic Attack Treated with Aspirin or Ticagrelor and Patient OutcomES). Post Stroke - - Protocol DALF-PS 1016 A Double-Blind, Placebo-Controlled, Parallel- Group Study to Evaluate the Efficacy and Safety of Two Dose Strengths of 9
10 Dalfampridine Extended Release Tablets for Treatment of Stable Walking Deficits in Post-Ischemic Stroke (MILESTONE). Post Stroke - - Protocol DALF-PS-1029 An Extension Study to Evaluate the Long-Term Safety, Tolerability and Efficacy of Dalfampridine Extended-Release Tablets for the Treatment of Chronic Post-Ischemic Stroke Walking Deficits in Subjects Who Participated in the DALF-PS-1016 Study (MILESTONE). Post Stroke - - Protocol A Phase II Safety And Efficacy Study Of ALZT-OP1A As Adjuvant Treatment In Subjects With Post-Ischemic Stroke Cognitive Impairment (PSCI). Post Stroke - - Protocol Randomized, double-blind, Evaluation in secondary Stroke Prevention comparing the Efficacy and safety of the oral Thrombin inhibitor dabigatran etexilate (110 mg or 150 mg, oral b.i.d.) versus acetylsalicylie acid (100 mg oral q.d.) in patients with Embolic Stroke of Undetermined Source (RESPECT ESUS). Acute Stroke - - Protocol B A Phase 1B Multicenter Open Label Study to Evaluate the Safety And Tolerability And Determine the Maximum Tolerated Dose Of PF In Subjects With Intracerebral Hemorrhage (ICH) Migraine Migraine - - Protocol VML251/96/07: A Double-Blind, Placebo-Controlled, Parallel- Group Study to Assess the Efficacy and Safety of Up To Two Doses of VML251 in the Acute Treatment of Migraine. Migraine - - Protocol CAPSS-223; Phase IV: A Comparison of the Efficacy and Safety of ULTRACET (Tramadol HCL/Acetaminophen versus Placebo for the Acute Treatment of Migraine Headache Pain. Mild Cognitive Impairment Mild Cognitive Impairment - - Protocol GAL-INT-18: A randomized double blind placebo-controlled trial to evaluate the efficacy and safety of Galantamine in subjects with Mild Cognitive Impairment clinically at risk for development of clinically probable Alzheimer s Disease Mild Cognitive Impairment - - Protocol GAL-MCI-301: An Open-Label Extension Study to Assess the Long-Term Safety and Tolerability of Galantamine HBr in the Treatment of Mild Cognitive Impairment. Mild Cognitive Impairment- - Protocol # E2020-A : A one year, multicenter,randomized double-blind, placebo-controlled evaluation of the efficacy and safety of donepezil hydrochloride {E2020} in subjects with mild cognitive impairment. 10
11 Mild Cognitive Impairment (MCI) - - Protocol No. E2020-A Open Label Extension Study of the E2020-A Protocol Multiple Sclerosis Multiple Sclerosis - - Protocol EPOC: A 6-month, Randomized, Active Comparator, Open-label, Multi-Center Study to Evaluate Patient OutComes, Safety and Tolerability of Fingolimod 0.5 mg/day in Patients with Relapsing Forms of Multiple Sclerosis who are candidates for MS therapy change from Previous Disease Modifying Therapy (EPOC) Multiple Sclerosis - - Protocol CFTY720DUS09: A 12-Month, Prospective, Randomized, Active-Controlled, Open-label Study to Evaluate The Patient Retention Of Fingolimod vs. Approved First-Line Disease Modifying Therapies In Adults Who Are In Early Stages Of Treatment For Relapsing Remitting Multiple Sclerosis (PREFERMS). Multiple Sclerosis - - Protocol CFTY720D2404: the Multi-National Gilenya Pregnancy Exposure Registry in Multiple Sclerosis. Pain Low Back Pain - - Protocol : An 8-Week, Randomized, Double-Blind, Placebo-Controlled, Monotherapy Trial of Pregabalin for Treatment of Chronic Low Back Pain Low Back Pain - - Protocol : Pregabalin Open-Label, Extension Safety Trial in Patients With Chronic Pain. Parkinson s Disease Parkinson s Disease - - Protocol NR154401/M35016:m A Noncomparative Open-Label Study to Evaluate TASMAR Dosage Regimen in Nonfluctuating Parkinson s Disease Patients Treated with Sinemet, with Follow-up Treatment of Tasmar. Parkinson s Disease - - OPRC 352-E-00 A Prospective, Randomized, Parallel-Group, Double-Blind, Placebo-Controlled, Multi-Center Study to Evaluate the Short-Term Efficacy and Safety of Entacapone Administered Together with Levodopa in Subjects with Parkinson s Disease Without Motor Fluctuations. Parkinson s Disease - - Protocol RP54274X-320: A Phase III Multicenter, Double- Blind, Parallel-Group, Placebo-Controlled Study of the Effect of Riluzole 50mg bid or 100 mg bid on the Progression of Parkinson s Disease for Two Years in 1050 Patients. Parkinson s Disease - - Protocol RP54274X-321: A Phase III Multicenter, Double Blind, Parallel-Group, Placebo Controlled Study of the Effect of Riluzole 50mg bid or 11
12 100mg bid on the Progression of Parkinson s Disease in Patients Treated With L-Dopa or Dopamine Agonist Parkinson s Disease - - Protocol CCOM 998 US01: A Multicenter, Double-Blind, Placebo-Controlled Study to Assess the Tolerability and Effect of Entacapone on the Quality of Life in Parkinson s Disease Patients Treated with Levodopa/Carbidopa Experiencing End-of-Dose Wearing-Off. Parkinson s Disease - - Protocol PNU 95666E: A Double-Blind, Placebo-Controlled, Dose-Response Study of Tolerability, Safety, and Efficacy in Patients with Early Parkinson s Disease. Parkinson s Disease - - Protocol PNU-95666E: Open-label, Long Term, Flexible Dose Study of Safety, Tolerability, and Therapeutic Response in Patients with Parkinson s Disease. Parkinson s Disease - - Protocol 666E-CNS-0075: A Phase III, Double-blind, Placebo- Controlled, Randomized Study Comparing the Efficacy, Safety, and Tolerability of Sumanirole versus Placebo or Ropinirole in Patients with Early Parkinson s Disease. Parkinson s Disease - -Protocol 666E-CNS : Pharmacogenomics Blood Sampling Protocol for Sumanirole. Parkinson s Disease - - Protocol DA2APD : A Phase III, Multi-center, Randomized, Double-blind, Placebo-controlled, Fixed Dose Response Study Comparing the Efficacy and Safety of Sumanirole Versus Placebo in Patients With Early Parkinson s Disease. Parkinson s Disease - - Protocol CELC200AUS11: A prospective, multi-center, randomized, open-label study with blinded raters to evaluate the effects of immediate versus delayed switch to Stalevo on motor function and quality of life in patients with Parkinson s disease with end-of-dose wearing off. Parkinson s Disease - - Protocol No. CELC200A A Prospective, Multi-Center, Randomized, Open-Label Study With Blinded Raters To Evaluate The Effects of Immediate Versus Delayed Switch To Stalevo On Motor Function And Quality Of Life In Patients With Parkinson s Disease With End-Of-Dose Wearing Off. Parkinson s Disease - - Protocol No. S The Rembrandt Study A Randomized, Double-blind, Placebo Controlled Parallel-group fixed and Flexible SLV308 Dose Arm Study to Assess Efficacy and Safety of SLV308 Monotherapy in the Treatment of Patients with Early Stage Parkinson s Disease. Parkinson s Disease - - S Open-Label, 6-12 Months Safety and Efficacy Study of Levodopa Carbidopa Intestinal Gel in Levodopa-Responsove Subjects with Advanced Parkinson s Disease and Severe Motor-Fluctuations. 12
13 Parkinson s Disease - - S Open-Label Continuation Treatment Study with Levidopa-Carbidopa Intestinal Gel in Subjects with Advanced Parkinson s Disease and Severe Motor-Fluctuations Who Have Exhibited a Persistent and Positive Effect to Treatment in Previous Studies. Peripheral Neuropathy Diabetic Peripheral Neuropathy - - Protocol K0718g A Phase III Multicenter, Double- Blind, Placebo Controlled, Parallel-Group Study of the Efficacy and Safety of Recombinant Human Nerve Growth Factor (rhngf) in Subjects with Diabetic Neuropathy. Diabetic Peripheral Neuropathy - - Protocol K0807g: A Phase III Open-Label Extension Study of the Long-Term Safety of Recombinant Human Nerve Growth Factor (rhngf) in the Treatment of Subjects with Diabetic Peripheral Neuropathy. Diabetic Peripheral Neuropathy - - Protocol MEM-MD-06A : A Double-Blind comparison of Memantine and placebo in the treatment of chronic pain in patients with Diabetic Neuropathy. Diabetic Peripheral Neuropathy - - Protocol MEM-MD-06B: An open-label extension of Memantine treatment in patients with painful Diabetic Neuropathy. Diabetic Peripheral Neuropathy - - Protocol NPP30004: A Multicenter, Double- Blind, Randomized Study to Evaluate the Safety and Efficacy of Lamotrigine 200 mg/day, 300 mg/day, and 400 mg/day Compared With Placebo in Subjects With painful Diabetic Neuropathy. Diabetic Peripheral Neuropathy - - Protocol Number NPP30006: An Open-Label Study to Evaluate the Safety of Lamotrigine in Subjects with Painful diabetic Neuropathy. Diabetic Neuropathy- - Protocol # SP742: A multi-center, randomized, double-blind, placebo-controlled trial to assess the efficacy and safety of 200, 400, and 600mg/day SPM 927 in subjects with painful distal diabetic neuropathy. Peripheral Neuropathy- - Protocol # NPP30010: A multicenter, randomized, doubleblind, placebo-controlled, parallel group to evaluate the safety and efficacy of lamotrigine in subjects with neuropathic pain and inadequate pain relief with gabapentin, tricyclic antidepressants or non-narcotic analgesics. Herpatic Neuropathy - - Protocol A1A20004: A Multicenter Randomized, Double-Blind, Placebo-Controlled, Parallel Group Study to Evaluate the Efficacy and 13
14 Tolerability of a 14 Day Treatment Course of CW mg Compared to Placebo in Subjects with Peripheral Neuropathic Pain. Postherpetic Neuropathy- -Protocol # CXA 20009: A multi-center, randomized, doubleblind, placebo controlled, parallel group study to assess the safety and efficacy of GW mg and 50mg, administered once daily for 21 days to subjects with postherpetic neuralgia. Postherpetic Neuralgia - - Protocol No A Phase 3 Multicenter, Randomized, Double-Blind, Placebo-Controlled Study of the Safety and Efficacy of Gabapentin Extended Release (G-ER) Tablets in the Treatment of Patients with Postherpetic Neuralgia. Diabetic Peripheral Neuropathy - - Protocol R PAI-3015: A Randomized- Withdrawal Phase III Study Evaluating the Safety and Efficacy of CG5503 Extended- Release (ER) in Subjects with Painful Diabetic Peripheral Neuropathy. Vascular Dementia Vascular Dementia - - Protocol GAL-INT-26: A randomized 26 week, double blind placebo controlled trial to evaluate the safety and efficacy of Galantamine in the treatment of Dementia secondary to Cerebrovascular Disease. Vascular Dementia - - Protocol GAL-INT-22: An Open-Label Extension to Assess the Safety of Galantamine HBr in the treatment of Vascular Dementia. Sub-Investigator: Acute Ischemic Stroke - - Protocol LUB-INT-9: Lubeluzole: Effects of Intravenous Therapy in Subjects With Acute Ischemic Stroke, A Placebo-Controlled, Double-Blind, Randomized, Multicenter, Trial. Painful Diabetic Peripheral Neuropathy Protocol Prosaptide TX14-004: A Four- Week, Double-Blind, Randomized, Placebo-Controlled, Multicenter, Phase II Study to Evaluate the Safety, Tolerability, and Efficacy of Three Subcutaneous Doses of Prosaptide (TX14(A) when Administered Daily for 28 days on Diabetic Painful Neuropathy in Patients with Type 1 and Type 2 Diabetes Mellitus. Painful Diabetic Peripheral Neuropathy - - Protocol PRI/TOP INT 30:(TOPMAT-NP 003) A Double-Blind, Placebo-Controlled, Parallel Group, Dose-Response Study to Evaluate the Efficacy and Safety of Topiramate Versus Placebo in the Treatment of Pain Associated with Diabetic Peripheral Polyneuropathy. Migraine - - Protocol PRI/TOP-INT-48 (TOPMAT-MIGR-002): A randomized, double-blind, placebo-controlled, parallel group, dose-response study to evaluate the efficacy and safety of Topiramate in the prophylaxis of migraine. 14
Gregory Aram Sahagian, M.D.
 The Neurology Center of Southern California 3907 Waring Road, Suite 2, 3 and 4, Oceanside, California, 92056 320 Santa Fe Drive, Suite 108, Encinitas, California, 92024 332 Santa Fe Drive, Suite 150. Encinitas,
The Neurology Center of Southern California 3907 Waring Road, Suite 2, 3 and 4, Oceanside, California, 92056 320 Santa Fe Drive, Suite 108, Encinitas, California, 92024 332 Santa Fe Drive, Suite 150. Encinitas,
Irene Jennifer Oh, M.D.
 The Neurology Center of Southern California 6010 Hidden Valley Road, Suite 200, Carlsbad, California 92011 9850 Genesee Ave, Suite 470, La Jolla, CA, 92037 1955 Citracado Pkwy, Suite 102, Escondido, CA
The Neurology Center of Southern California 6010 Hidden Valley Road, Suite 200, Carlsbad, California 92011 9850 Genesee Ave, Suite 470, La Jolla, CA, 92037 1955 Citracado Pkwy, Suite 102, Escondido, CA
Mark Norman Sadoff, M.D.
 6010 Hidden Valley Road, Suite 200, Carlsbad, California 92011 9850 Genesee Ave, Suite 470, La Jolla, CA, 92037 1955 Citracado Pkwy, Suite 102, Escondido, CA 92029 15721 Pomerado Road, Poway, CA 92064
6010 Hidden Valley Road, Suite 200, Carlsbad, California 92011 9850 Genesee Ave, Suite 470, La Jolla, CA, 92037 1955 Citracado Pkwy, Suite 102, Escondido, CA 92029 15721 Pomerado Road, Poway, CA 92064
Jack David Schim, M.D.
 The Neurology Center of Southern California 3907 Waring Road, Suite 2 and 3, Oceanside, California, 92056 320 Santa Fe Drive, Suite 108, Encinitas, California, 92024 332 Santa Fe Drive, Suite 150, Encinitas,
The Neurology Center of Southern California 3907 Waring Road, Suite 2 and 3, Oceanside, California, 92056 320 Santa Fe Drive, Suite 108, Encinitas, California, 92024 332 Santa Fe Drive, Suite 150, Encinitas,
Thomas John Chippendale, M.D., Ph.D.
 The Neurology Center of Southern California 3907 Waring Road, Suite 2, 3 and 4, Oceanside, California, 92056 320 Santa Fe Drive, Suite 108, Encinitas, California, 92024 332 Santa Fe Drive, Suite 150, Encinitas,
The Neurology Center of Southern California 3907 Waring Road, Suite 2, 3 and 4, Oceanside, California, 92056 320 Santa Fe Drive, Suite 108, Encinitas, California, 92024 332 Santa Fe Drive, Suite 150, Encinitas,
Curriculum Vitae, Nirav S. Patel, M.D. Nirav S. Patel, M.D. Collaborative Neuroscience Network, LLC.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
MITCHELL JAY RUBIN, M.D. FAAN (cell) Neurology Expert Witness Stroke tpa Emergency Neurology Telemedicine
 Education University of Pennsylvania Philadelphia, Pennsylvania B. A. Summa Cum Laude 1976 Phi Beta Kappa 1977 Pi Gamma Mu (National Social Science Honor Society) 9/1977-5/1981 University of Pennsylvania
Education University of Pennsylvania Philadelphia, Pennsylvania B. A. Summa Cum Laude 1976 Phi Beta Kappa 1977 Pi Gamma Mu (National Social Science Honor Society) 9/1977-5/1981 University of Pennsylvania
Curriculum Vitae, Nirav S. Patel, M.D. Nirav S. Patel, M.D. Collaborative Neuroscience Network, LLC Redondo Avenue, Suites 415 & 500
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
RHONDA Q. FREEMAN, PH.D. SUNRISE MEDICAL GROUP 4925 Sheridan St. Suite 200 Hollywood, FL (954)
 RHONDA Q. FREEMAN, PH.D. SUNRISE MEDICAL GROUP 4925 Sheridan St. Suite 200 Hollywood, FL 33021 (954) 981-3850 LICENSE: FL #PY 6252 SPECIALTY: EDUCATION: Bachelor of Science (Psychology) June 1992 Master
RHONDA Q. FREEMAN, PH.D. SUNRISE MEDICAL GROUP 4925 Sheridan St. Suite 200 Hollywood, FL 33021 (954) 981-3850 LICENSE: FL #PY 6252 SPECIALTY: EDUCATION: Bachelor of Science (Psychology) June 1992 Master
Curriculum Vitae Department of Neurology University of Colorado Medical Center Denver, Colorado
 Robert W. Pratt, M.D. Robert.Pratt2@dhha.org Curriculum Vitae 303-602-5013 CURRENT APPOINTMENTS 2016-present 2017-present Assistant Professor Department of Neurology University of Colorado Medical Center
Robert W. Pratt, M.D. Robert.Pratt2@dhha.org Curriculum Vitae 303-602-5013 CURRENT APPOINTMENTS 2016-present 2017-present Assistant Professor Department of Neurology University of Colorado Medical Center
CURRICULUM VITAE. University of Columbia Missouri Department of Psychiatry and Neurology
 CURRICULUM VITAE Rubén Bravo Valverde, M.D. Forensic and Geriatric Psychiatrist Centro Especializado de Psiquiatría 1995 Road 2 Suite 1209-1210 Bayamón, Puerto Rico 00959-1201 Telephones: (787) 786-0575/780-3752
CURRICULUM VITAE Rubén Bravo Valverde, M.D. Forensic and Geriatric Psychiatrist Centro Especializado de Psiquiatría 1995 Road 2 Suite 1209-1210 Bayamón, Puerto Rico 00959-1201 Telephones: (787) 786-0575/780-3752
PACIFIC RESEARCH NETWORK_ 3003 Fourth Avenue, San Diego, CA * (619) Ph. (619) Fax
 PACIFIC RESEARCH NETWORK 3003 Fourth Avenue, San Diego, CA 92103 * (619) 294-4302 Ph. (619) 294-4867 Fax JOHN J. DORIA, M.D. EDUCATION 1958 Medical: Universidad Nationale de Mexico, Mexico, Degree: M.
PACIFIC RESEARCH NETWORK 3003 Fourth Avenue, San Diego, CA 92103 * (619) 294-4302 Ph. (619) 294-4867 Fax JOHN J. DORIA, M.D. EDUCATION 1958 Medical: Universidad Nationale de Mexico, Mexico, Degree: M.
Amy Edmondson Mulroy, MD
 Curriculum Vitae Amy Edmondson Mulroy, MD 7940 Floyd Curl Drive, Suite 700 7940 Floyd Curl Drive, Suite 1020 San Antonio, Texas 78229 San Antonio, Texas 78229 EDUCATION 1997 General Adult Psychiatry Internship
Curriculum Vitae Amy Edmondson Mulroy, MD 7940 Floyd Curl Drive, Suite 700 7940 Floyd Curl Drive, Suite 1020 San Antonio, Texas 78229 San Antonio, Texas 78229 EDUCATION 1997 General Adult Psychiatry Internship
09/84-06/88 BA, Chemistry Robert D. Clark Honors College, University of Oregon, Eugene, OR
 CONTACT INFORMATION: Drissana T Tran, M.D. Oregon Center for Clinical Investigations, Inc. 702 Church Street NE Salem, OR 97301 Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916)
CONTACT INFORMATION: Drissana T Tran, M.D. Oregon Center for Clinical Investigations, Inc. 702 Church Street NE Salem, OR 97301 Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916)
CURRICULUM VITAE Intern in Medicine, Beth Israel Hospital, Harvard Medical School, Boston, MA
 Bruce D. Kohrman, M.D. 12305 SW 69 th Place Miami, Florida 33156 Telephone: (305) 903-8678 Fax: (305) 661-1672 CURRICULUM VITAE EDUCATION 1977 - B.S. Stanford University, Stanford, CA 1982 - M.D. Case
Bruce D. Kohrman, M.D. 12305 SW 69 th Place Miami, Florida 33156 Telephone: (305) 903-8678 Fax: (305) 661-1672 CURRICULUM VITAE EDUCATION 1977 - B.S. Stanford University, Stanford, CA 1982 - M.D. Case
CURRICULUM VITAE. Andrea Marraffino, Ph.D.
 CURRICULUM VITAE CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Site Liaison Tel. (866) 669-0234 Fax (208) 575-3169 Email: clinicaltrials@btheodore.com AFFILIATIONS: Florida Clinical
CURRICULUM VITAE CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Site Liaison Tel. (866) 669-0234 Fax (208) 575-3169 Email: clinicaltrials@btheodore.com AFFILIATIONS: Florida Clinical
Curriculum Vitae for: GIL LICHTSHEIN, M.D. Gil Lichtshein, M.D., P.A West Camino Real, Suite 220 Boca Raton, FL 33433
 Curriculum Vitae for: GIL LICHTSHEIN, M.D. Gil Lichtshein, M.D., P.A. 7200 West Camino Real, Suite 220 Boca Raton, FL 33433 University of Miami Life Science and Technology Park 1951 NW 7 th Ave, Suite
Curriculum Vitae for: GIL LICHTSHEIN, M.D. Gil Lichtshein, M.D., P.A. 7200 West Camino Real, Suite 220 Boca Raton, FL 33433 University of Miami Life Science and Technology Park 1951 NW 7 th Ave, Suite
Curriculum Vitae, Steven H. Reynolds, D.O. Steven H. Reynolds, D.O. Collaborative Neuroscience Network, LLC.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
Curriculum Vitae, Omid Omidvar, M.D. Omid Omidvar, M.D. Collaborative Neuroscience Network, LLC Redondo Avenue, Suites 415 & 500
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
Curriculum Vitae, Edward H. Ortiz, M.D.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: FutureSearch Trials
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: FutureSearch Trials
Dr Julia Hopyan Stroke Neurologist Sunnybrook Health Sciences Centre
 Dr Julia Hopyan Stroke Neurologist Sunnybrook Health Sciences Centre Objectives To learn what s new in stroke care 2010-11 1) Acute stroke management Carotid artery stenting versus surgery for symptomatic
Dr Julia Hopyan Stroke Neurologist Sunnybrook Health Sciences Centre Objectives To learn what s new in stroke care 2010-11 1) Acute stroke management Carotid artery stenting versus surgery for symptomatic
Fax uci.edu. University Major Degree &Year
 Lama Al-Khoury, MD Clinical Assistant Professor Neurology Department Stroke Center University of California Irvine (UCI) Medical Center 101 The City Drive South Building 53, Room 225 Orange, California
Lama Al-Khoury, MD Clinical Assistant Professor Neurology Department Stroke Center University of California Irvine (UCI) Medical Center 101 The City Drive South Building 53, Room 225 Orange, California
MARILYN MOLLMAN RYMER, M.D.
 CURRICULUM VITAE MARILYN MOLLMAN RYMER, M.D. License No.: MO R8A55 KS 04-18937 EDUCATION AND TRAINING: Northwestern University Evanston, Illinois Bachelor of Arts Phi Beta Kappa Washington University School
CURRICULUM VITAE MARILYN MOLLMAN RYMER, M.D. License No.: MO R8A55 KS 04-18937 EDUCATION AND TRAINING: Northwestern University Evanston, Illinois Bachelor of Arts Phi Beta Kappa Washington University School
Curriculum Vitae, Omid Omidvar, M.D. Omid Omidvar, M.D. Collaborative Neuroscience Network, LLC Redondo Avenue, Suite 500 Long Beach, CA 90806
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
Diagnosis and Treatment of Alzhiemer s Disease
 Diagnosis and Treatment of Alzhiemer s Disease Roy Yaari, MD, MAS Director, Memory Disorders Clinic, Banner Alzheimer s Institute 602-839-6900 Outline Introduction Alzheimer s disease (AD)Guidelines -revised
Diagnosis and Treatment of Alzhiemer s Disease Roy Yaari, MD, MAS Director, Memory Disorders Clinic, Banner Alzheimer s Institute 602-839-6900 Outline Introduction Alzheimer s disease (AD)Guidelines -revised
SHANKER N. DIXIT, MD
 Neurology Center of Las Vegas 2480 Professional Ct. Las Vegas, NV 89128 Phone: (702) 405-7100 EDUCATION SHANKER N. DIXIT, MD 1995 Medical: AIIMS, New Delhi, India, DM (Neurology) 1991 Medical: Kasturba
Neurology Center of Las Vegas 2480 Professional Ct. Las Vegas, NV 89128 Phone: (702) 405-7100 EDUCATION SHANKER N. DIXIT, MD 1995 Medical: AIIMS, New Delhi, India, DM (Neurology) 1991 Medical: Kasturba
Director, Movement Disorders Research Banner Good Samaritan Medical Center, Phoenix, Arizona. June 2003
 Curriculum Vitae JOHAN E. S. SAMANTA, M.D. Movement Disorders Neurologist Diplomate ofthe ABPN 2610 N 3 rd St, Suite A Phoenix, AZ 85004 Phone: 602-277-2228 Fax: 602-265-9494 Tax ID: 86-1025375 CURRENT
Curriculum Vitae JOHAN E. S. SAMANTA, M.D. Movement Disorders Neurologist Diplomate ofthe ABPN 2610 N 3 rd St, Suite A Phoenix, AZ 85004 Phone: 602-277-2228 Fax: 602-265-9494 Tax ID: 86-1025375 CURRENT
Curriculum Vitae, Michael J. Downing, M.D.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com PROFESSIONAL AFFILIATIONS: Michael
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com PROFESSIONAL AFFILIATIONS: Michael
Current Clinical Trials for Stroke Survivors in NJ and Philadelphia Areas
 Current Clinical Trials for Survivors in NJ and Philadelphia Areas For more information go to https://clinicaltrials.gov/ and search for the title in search box Condition / Disease 1. Spatial Neglect and
Current Clinical Trials for Survivors in NJ and Philadelphia Areas For more information go to https://clinicaltrials.gov/ and search for the title in search box Condition / Disease 1. Spatial Neglect and
Curriculum Vitae, Conrad O. Beckles, M.S., L.M.H.C., Ph.D.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Conrado Oscar Beckles,
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Conrado Oscar Beckles,
Dementia is not normal aging!
 The Future of Alzheimer s Disease Treatment Adam L. Boxer, MD, PhD Director, Alzheimer s Disease Clinical Trials Program Memory and Aging Center Assistant Professor of Neurology University of California,
The Future of Alzheimer s Disease Treatment Adam L. Boxer, MD, PhD Director, Alzheimer s Disease Clinical Trials Program Memory and Aging Center Assistant Professor of Neurology University of California,
Howard A. Hassman, D.O. Hassman Research Institute, LLC 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009
 CONTACT INFORMATION: Howard A. Hassman, D.O. Hassman Research Institute, LLC 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
CONTACT INFORMATION: Howard A. Hassman, D.O. Hassman Research Institute, LLC 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
Voice: (802)
 March 2009 CURRICULUM VITAE Alexandra S. Potter Ph.D. Clinical Neuroscience Research Unit Department of Psychiatry University of Vermont College of Medicine 1 South Prospect Street Burlington, VT 05401
March 2009 CURRICULUM VITAE Alexandra S. Potter Ph.D. Clinical Neuroscience Research Unit Department of Psychiatry University of Vermont College of Medicine 1 South Prospect Street Burlington, VT 05401
European Commission Grants Approval for Mavenclad (Cladribine Tablets)
 Your Contact Friederike Segeberg +49 6151 72-6328 Investor Relations +49 6151 72-3321 August 25, 2017 European Commission Grants Approval for Mavenclad (Cladribine Tablets) First oral short-course treatment
Your Contact Friederike Segeberg +49 6151 72-6328 Investor Relations +49 6151 72-3321 August 25, 2017 European Commission Grants Approval for Mavenclad (Cladribine Tablets) First oral short-course treatment
CURRICULUM VITAE APRIL 2012
 CURRICULUM VITAE APRIL 2012 A. Name: Robert Miles Miska B. Address: (Office) Rocky Mountain Neurological Associates Phone: (801) 408-5700 370 East 9th Avenue, Suite 106 Fax: (801) 408-5704 Salt Lake City,
CURRICULUM VITAE APRIL 2012 A. Name: Robert Miles Miska B. Address: (Office) Rocky Mountain Neurological Associates Phone: (801) 408-5700 370 East 9th Avenue, Suite 106 Fax: (801) 408-5704 Salt Lake City,
Anesthesiology University of North Carolina, Chapel Hill, NC (Residency)
 Edward M. Tavel, Jr. M.D. Pain Specialists of Charleston Clinical Trials of South Carolina 2695 Elms Plantation Blvd Suite A and Suite D Charleston, SC 29406 Clinic: 843-818-1181 Research: 843-725-5067
Edward M. Tavel, Jr. M.D. Pain Specialists of Charleston Clinical Trials of South Carolina 2695 Elms Plantation Blvd Suite A and Suite D Charleston, SC 29406 Clinic: 843-818-1181 Research: 843-725-5067
Sponsor. Generic drug name. Trial indication(s) Protocol number. Protocol title. Phase of Drug Development. Study Start/End Dates
 Sponsor Novartis Generic drug name SAB378 [Naphthalen-1-yl-(4-pentyloxynaphthalen-1-yl)methanone] Trial indication(s) Postherpetic neuralgia (PHN) Protocol number CSAB378A2201 Protocol title A multicenter,
Sponsor Novartis Generic drug name SAB378 [Naphthalen-1-yl-(4-pentyloxynaphthalen-1-yl)methanone] Trial indication(s) Postherpetic neuralgia (PHN) Protocol number CSAB378A2201 Protocol title A multicenter,
Clinical Policy: Safinamide (Xadago) Reference Number: CP.CPA.308 Effective Date: Last Review Date: Line of Business: Commercial
 Clinical Policy: Safinamide (Xadago) Reference Number: CP.CPA.308 Effective Date: 05.16.17 Last Review Date: 08.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy
Clinical Policy: Safinamide (Xadago) Reference Number: CP.CPA.308 Effective Date: 05.16.17 Last Review Date: 08.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy
University of North Carolina, Chapel Hill, NC (Chief Resident)
 Edward M. Tavel, Jr. M.D. Pain Specialists of Charleston 2695 Elms Plantation Blvd Suite A and Suite D Charleston, SC 29406 Clinic: 843-818-1181 Research: 843-725-5067 Fax: 843-818-1145 Education 1981-1985
Edward M. Tavel, Jr. M.D. Pain Specialists of Charleston 2695 Elms Plantation Blvd Suite A and Suite D Charleston, SC 29406 Clinic: 843-818-1181 Research: 843-725-5067 Fax: 843-818-1145 Education 1981-1985
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 3Q17 July August
 BRAND NAME Xadago GENERIC NAME Safinamide MANUFACTURER Newron Pharmaceuticals SpA holds license; granted approval. US WorldMeds, LLC exclusive licensee and distributor in the U.S. DATE OF APPROVAL March
BRAND NAME Xadago GENERIC NAME Safinamide MANUFACTURER Newron Pharmaceuticals SpA holds license; granted approval. US WorldMeds, LLC exclusive licensee and distributor in the U.S. DATE OF APPROVAL March
CURRICULUM VITAE TELEPHONE NUMBER/FAX NUMBER: (732) / (732)
 CURRICULUM VITAE NAME: PRESENT TITLE: Didier Demesmin, MD CEO and Medical Director, University Pain Medicine Center OFFICE ADDRESS: 59 Veronica Avenue, Somerset, NJ 08873 TELEPHONE NUMBER/FAX NUMBER: (732)
CURRICULUM VITAE NAME: PRESENT TITLE: Didier Demesmin, MD CEO and Medical Director, University Pain Medicine Center OFFICE ADDRESS: 59 Veronica Avenue, Somerset, NJ 08873 TELEPHONE NUMBER/FAX NUMBER: (732)
Edward M. Tavel, Jr. M.D. Pain Specialists of Charleston 2695 Elms Plantation Blvd Suite A and Suite D Charleston, SC 29406
 Edward M. Tavel, Jr. M.D. Pain Specialists of Charleston 2695 Elms Plantation Blvd Suite A and Suite D Charleston, SC 29406 Clinic: 843-818-1181 Research: 843-725-5067 Fax: 843-818-1145 Education 1981-1985
Edward M. Tavel, Jr. M.D. Pain Specialists of Charleston 2695 Elms Plantation Blvd Suite A and Suite D Charleston, SC 29406 Clinic: 843-818-1181 Research: 843-725-5067 Fax: 843-818-1145 Education 1981-1985
5/2/2016. Outpatient Stroke Management Sheila Smith MD May 5, 2016
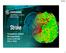 Outpatient Stroke Management Sheila Smith MD May 5, 2016 1 Management of Outpatient Stroke Objectives Review blood pressure management post stroke Review antithrombotic therapy Review statin therapy Discuss
Outpatient Stroke Management Sheila Smith MD May 5, 2016 1 Management of Outpatient Stroke Objectives Review blood pressure management post stroke Review antithrombotic therapy Review statin therapy Discuss
Curriculum Vitae, Joseph J. Savon, M.D.
 CONTACT INFORMATION: Joseph J. Savon, M.D. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
CONTACT INFORMATION: Joseph J. Savon, M.D. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
Drugs Affecting the Central Nervous System
 Asst Prof Inam S Arif isamalhaj@yahoo.com Drugs Affecting the Central Nervous System Ass Efferent neurons in ANS Neurodegenerative Diseases Parkinson s Disease Multiple Sclerosis Alzheimer s Disease
Asst Prof Inam S Arif isamalhaj@yahoo.com Drugs Affecting the Central Nervous System Ass Efferent neurons in ANS Neurodegenerative Diseases Parkinson s Disease Multiple Sclerosis Alzheimer s Disease
Curriculum Vitae, Lara Goenjian Shirikjian, D.O. Lara Goenjian Shirikjian, D.O. Collaborative Neuroscience Network, LLC
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
MEDICAL POLICY SUBJECT: KETAMINE INFUSION THERAPY FOR THE TREATMENT OF CHRONIC PAIN SYNDROMES POLICY NUMBER: CATEGORY: Technology Assessment
 Clinical criteria used to make utilization review decisions are based on credible scientific evidence published in peer reviewed medical literature generally recognized by the medical community. Guidelines
Clinical criteria used to make utilization review decisions are based on credible scientific evidence published in peer reviewed medical literature generally recognized by the medical community. Guidelines
September 26 28, 2013 Westin Tampa Harbour Island. Co-sponsored by
 September 26 28, 2013 Westin Tampa Harbour Island Co-sponsored by From Brains at Risk to Cognitive Dysfunction: The Role of Vascular Pathology Ralph Sacco, MD, MS, FAHA, FAAN Miller School of Medicine
September 26 28, 2013 Westin Tampa Harbour Island Co-sponsored by From Brains at Risk to Cognitive Dysfunction: The Role of Vascular Pathology Ralph Sacco, MD, MS, FAHA, FAAN Miller School of Medicine
Membership Information and Application
 Membership Information and Application Mission Statement The mission of the American Society of Neurorehabilitation (ASNR) is to promote the medical and social wellbeing of persons with disabling neurological
Membership Information and Application Mission Statement The mission of the American Society of Neurorehabilitation (ASNR) is to promote the medical and social wellbeing of persons with disabling neurological
2nd Annual Cerebrovascular and Stroke Symposium
 presents the 2nd Annual Cerebrovascular and Stroke Symposium Friday, May 25, 2018 7:15 a.m. to 6:00 p.m. Boca Raton Regional Hospital Dawson Auditorium Program Director: Jonathan O. Harris, M.D. Co-Chair:
presents the 2nd Annual Cerebrovascular and Stroke Symposium Friday, May 25, 2018 7:15 a.m. to 6:00 p.m. Boca Raton Regional Hospital Dawson Auditorium Program Director: Jonathan O. Harris, M.D. Co-Chair:
Recombinant Factor VIIa for Intracerebral Hemorrhage
 Recombinant Factor VIIa for Intracerebral Hemorrhage January 24, 2006 Justin Lee Pharmacy Resident University Health Network Outline 1. Introduction to patient case 2. Overview of intracerebral hemorrhage
Recombinant Factor VIIa for Intracerebral Hemorrhage January 24, 2006 Justin Lee Pharmacy Resident University Health Network Outline 1. Introduction to patient case 2. Overview of intracerebral hemorrhage
Curriculum Vitae, Carmen Zegarra, M.D. Carmen Zegarra, M.D. FutureSearch Clinical Trials, L.P Parkcrest Drive, Suite 300 Austin, TX 78731
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Site Liaison Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Carmen Zegarra, M.D. FutureSearch
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Site Liaison Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Carmen Zegarra, M.D. FutureSearch
Antiplatelet Therapy in Primary CVD Prevention and Stable Coronary Artery Disease. Καρακώστας Γεώργιος Διευθυντής Καρδιολογικής Κλινικής, Γ.Ν.
 Antiplatelet Therapy in Primary CVD Prevention and Stable Coronary Artery Disease Καρακώστας Γεώργιος Διευθυντής Καρδιολογικής Κλινικής, Γ.Ν.Κιλκίς Primary CVD Prevention A co-ordinated set of actions,
Antiplatelet Therapy in Primary CVD Prevention and Stable Coronary Artery Disease Καρακώστας Γεώργιος Διευθυντής Καρδιολογικής Κλινικής, Γ.Ν.Κιλκίς Primary CVD Prevention A co-ordinated set of actions,
CURRICULUM VITAE. Daniel K. White January, 2012
 CURRICULUM VITAE Daniel K. White January, 2012 ADDRESS Department of Physical Therapy and Athletic Training 635 Commonwealth Ave, 5 th Floor Boston, MA 02115 dwtbn@bu.edu (617) 638 4316 EDUCATION M.Sc
CURRICULUM VITAE Daniel K. White January, 2012 ADDRESS Department of Physical Therapy and Athletic Training 635 Commonwealth Ave, 5 th Floor Boston, MA 02115 dwtbn@bu.edu (617) 638 4316 EDUCATION M.Sc
Pharmacologic Treatment of Parkinson s Disease. Nicholas J. Silvestri, M.D. Assistant Professor of Neurology
 + Pharmacologic Treatment of Parkinson s Disease Nicholas J. Silvestri, M.D. Assistant Professor of Neurology + Overview n Brief review of Parkinson s disease (PD) n Clinical manifestations n Pathophysiology
+ Pharmacologic Treatment of Parkinson s Disease Nicholas J. Silvestri, M.D. Assistant Professor of Neurology + Overview n Brief review of Parkinson s disease (PD) n Clinical manifestations n Pathophysiology
Neuren Pharmaceuticals
 Neuren Pharmaceuticals Annual Shareholders Meeting May 2010 2009: A Watershed Year for Neuren Prioritized focus on NNZ-2566 and Motiva Reduced operating costs Obtained approval and Fast Track designation
Neuren Pharmaceuticals Annual Shareholders Meeting May 2010 2009: A Watershed Year for Neuren Prioritized focus on NNZ-2566 and Motiva Reduced operating costs Obtained approval and Fast Track designation
Howard A. Hassman, D.O. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009
 CONTACT INFORMATION: Howard A. Hassman, D.O. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
CONTACT INFORMATION: Howard A. Hassman, D.O. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
Joshua D. Lenchus, DO, RPh, FACP, SFHM Associate Professor of Medicine and Anesthesiology University of Miami Miller School of Medicine
 Joshua D. Lenchus, DO, RPh, FACP, SFHM Associate Professor of Medicine and Anesthesiology University of Miami Miller School of Medicine Antithrombotics Antiplatelets Aspirin Ticlopidine Prasugrel Dipyridamole
Joshua D. Lenchus, DO, RPh, FACP, SFHM Associate Professor of Medicine and Anesthesiology University of Miami Miller School of Medicine Antithrombotics Antiplatelets Aspirin Ticlopidine Prasugrel Dipyridamole
Updates in Stroke Management. Jessica A Starr, PharmD, FCCP, BCPS Associate Clinical Professor Auburn University Harrison School of Pharmacy
 Updates in Stroke Management Jessica A Starr, PharmD, FCCP, BCPS Associate Clinical Professor Auburn University Harrison School of Pharmacy Disclosure I have no actual or potential conflict of interest
Updates in Stroke Management Jessica A Starr, PharmD, FCCP, BCPS Associate Clinical Professor Auburn University Harrison School of Pharmacy Disclosure I have no actual or potential conflict of interest
Evaluation and Management of Parkinson s Disease in the Older Patient
 Evaluation and Management of Parkinson s Disease in the Older Patient David A. Hinkle, MD, PhD Comprehensive Movement Disorders Clinic Pittsburgh Institute for Neurodegenerative Diseases University of
Evaluation and Management of Parkinson s Disease in the Older Patient David A. Hinkle, MD, PhD Comprehensive Movement Disorders Clinic Pittsburgh Institute for Neurodegenerative Diseases University of
Learning Objectives. Epidemiology of Acute Coronary Syndrome
 Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
Yi-Po Chiu, PT, MHS, PhD, CEEAA 6/11 CURRICULUM VITAE
 CURRICULUM VITAE Yi-Po Chiu, PT, MHS, PhD, CEEAA Assistant Professor Doctor of Physical Therapy Program Department of Nursing and Rehabilitation Sciences College of Health and Human Services Angelo State
CURRICULUM VITAE Yi-Po Chiu, PT, MHS, PhD, CEEAA Assistant Professor Doctor of Physical Therapy Program Department of Nursing and Rehabilitation Sciences College of Health and Human Services Angelo State
Next patient please Dementia Clare Hawley 2018
 Next patient please Dementia Clare Hawley 2018 I have no conflict of interest to declare Dr Clare Hawley Associate Specialist Cardiology Chesterfield Royal Hospital GPwSI Refractory Angina Hon Clinical
Next patient please Dementia Clare Hawley 2018 I have no conflict of interest to declare Dr Clare Hawley Associate Specialist Cardiology Chesterfield Royal Hospital GPwSI Refractory Angina Hon Clinical
Faculty Academic Appointments 7/2011-8/2016 Assistant Professor 7/2012-8/2016 Instructor Neurology Harvard Medical School
 Vera Fridman, M.D. October 25, 2017 Office Address: Place of Birth: Neuromuscular Division Department of Neurology 12631 East 17 th Ave Mail Stop B185 Aurora, CO 80045 Phone: (303) 724-2188 Fax: (303)
Vera Fridman, M.D. October 25, 2017 Office Address: Place of Birth: Neuromuscular Division Department of Neurology 12631 East 17 th Ave Mail Stop B185 Aurora, CO 80045 Phone: (303) 724-2188 Fax: (303)
FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS
 New Horizons In Atherothrombosis Treatment 2012 순환기춘계학술대회 FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS Division of Cardiology, Jeonbuk National University Medical School Jei Keon Chae,
New Horizons In Atherothrombosis Treatment 2012 순환기춘계학술대회 FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS Division of Cardiology, Jeonbuk National University Medical School Jei Keon Chae,
EMERGING TREATMENTS FOR PARKINSON S DISEASE
 EMERGING TREATMENTS FOR PARKINSON S DISEASE Katerina Markopoulou, MD, PhD Director Neurodegenerative Diseases Program Department of Neurology NorthShore University HealthSystem Clinical Assistant Professor
EMERGING TREATMENTS FOR PARKINSON S DISEASE Katerina Markopoulou, MD, PhD Director Neurodegenerative Diseases Program Department of Neurology NorthShore University HealthSystem Clinical Assistant Professor
David Henry Jablonski, M.D North Mills Avenue Orlando, FL 32803
 David Henry Jablonski, M.D. 1812 North Mills Avenue Orlando, FL 32803 EDUCATION: University of Florida College of Medicine, Gainesville, Florida Division of Urology Residency Training Program July 1994-June
David Henry Jablonski, M.D. 1812 North Mills Avenue Orlando, FL 32803 EDUCATION: University of Florida College of Medicine, Gainesville, Florida Division of Urology Residency Training Program July 1994-June
Delirium & Dementia. Nicholas J. Silvestri, MD
 Delirium & Dementia Nicholas J. Silvestri, MD Outline Delirium vs. Dementia Neural pathways relating to consciousness Encephalopathy Stupor Coma Dementia Delirium vs. Dementia Delirium Abrupt onset Lasts
Delirium & Dementia Nicholas J. Silvestri, MD Outline Delirium vs. Dementia Neural pathways relating to consciousness Encephalopathy Stupor Coma Dementia Delirium vs. Dementia Delirium Abrupt onset Lasts
West Virginia University School of Medicine, Morgantown, West Virginia
 MARY CARROLL SHEMO, M.D., D.F.A.P.A. CURRICULUM VITAE Psychiatric Alliance of the Blue Ridge Office: (434) 984-6777 First Floor, Old Ivy Commons, 2496 Old Ivy Road, Suite 400 Fax: (434) 296-1412 22903-4895
MARY CARROLL SHEMO, M.D., D.F.A.P.A. CURRICULUM VITAE Psychiatric Alliance of the Blue Ridge Office: (434) 984-6777 First Floor, Old Ivy Commons, 2496 Old Ivy Road, Suite 400 Fax: (434) 296-1412 22903-4895
SUBURBAN LUNG ASSOCIATES, S.C. Eberle Medical Building, 800 Biesterfield road, Suite 510 Elk Grove Village, IL Sleep Group
 SUBURBAN LUNG ASSOCIATES, S.C. Eberle Medical Building, 800 Biesterfield road, Suite 510 Elk Grove Village, IL 60007 NAME Kuljeet Kaur Gill M.D. Nickname: Kelly Address: Suburban Lung Associates SC 800
SUBURBAN LUNG ASSOCIATES, S.C. Eberle Medical Building, 800 Biesterfield road, Suite 510 Elk Grove Village, IL 60007 NAME Kuljeet Kaur Gill M.D. Nickname: Kelly Address: Suburban Lung Associates SC 800
Literature Scan: Alzheimer s Drugs
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
TRANSPARENCY COMMITTEE OPINION. 18 March 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 18 March 2009 REQUIP LP 2 mg extended-release tablet Box of 21 tablets (CIP: 379 214-8) Box of 28 tablets (CIP: 379
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 18 March 2009 REQUIP LP 2 mg extended-release tablet Box of 21 tablets (CIP: 379 214-8) Box of 28 tablets (CIP: 379
Overview of Parkinson s disease Research at University of Colorado
 Overview of Parkinson s disease Research at University of Colorado Brian D. Berman, MD, MS May 12, 2018 University of Colorado Movement Disorders Center Terminology Basic science pure/fundamental scientific
Overview of Parkinson s disease Research at University of Colorado Brian D. Berman, MD, MS May 12, 2018 University of Colorado Movement Disorders Center Terminology Basic science pure/fundamental scientific
Alzheimer Disease Agents Drug Class Prior Authorization Protocol
 Line of Business: Medi-Cal Effective Date: August 16, 2017 Revision Date: August 16, 2017 Alzheimer Disease Agents Drug Class Prior Authorization Protocol This policy has been developed through review
Line of Business: Medi-Cal Effective Date: August 16, 2017 Revision Date: August 16, 2017 Alzheimer Disease Agents Drug Class Prior Authorization Protocol This policy has been developed through review
Optimal medical therapy in patients with stable CAD
 Optimal medical therapy in patients with stable CAD Robert Storey Professor of Cardiology, University of Sheffield and Academic Director and Honorary Consultant Cardiologist, Cardiology and Cardiothoracic
Optimal medical therapy in patients with stable CAD Robert Storey Professor of Cardiology, University of Sheffield and Academic Director and Honorary Consultant Cardiologist, Cardiology and Cardiothoracic
Pharmacologic Treatment of Parkinson s Disease. Nicholas J. Silvestri, M.D. Associate Professor of Neurology
 + Pharmacologic Treatment of Parkinson s Disease Nicholas J. Silvestri, M.D. Associate Professor of Neurology + Disclosures n NO SIGNIFICANT FINANCIAL, GENERAL, OR OBLIGATION INTERESTS TO REPORT + Learning
+ Pharmacologic Treatment of Parkinson s Disease Nicholas J. Silvestri, M.D. Associate Professor of Neurology + Disclosures n NO SIGNIFICANT FINANCIAL, GENERAL, OR OBLIGATION INTERESTS TO REPORT + Learning
Dementia. Assessing Brain Damage. Mental Status Examination
 Dementia Assessing Brain Damage Mental status examination Information about current behavior and thought including orientation to reality, memory, and ability to follow instructions Neuropsychological
Dementia Assessing Brain Damage Mental status examination Information about current behavior and thought including orientation to reality, memory, and ability to follow instructions Neuropsychological
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Neuropathic pain pharmacological management: the pharmacological management of neuropathic pain in adults in non-specialist
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Neuropathic pain pharmacological management: the pharmacological management of neuropathic pain in adults in non-specialist
Curriculum Vitae, John Douglas Hudson, M.D.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com EDUCATION: John Douglas Hudson, M.D.
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com EDUCATION: John Douglas Hudson, M.D.
Medications used to treat Parkinson s disease
 Medications used to treat Parkinson s disease Edwin B. George, M.D., Ph.D. Director of Wayne State University Movement Disorder Clinic University Health Center Neurology Clinic University Health The John
Medications used to treat Parkinson s disease Edwin B. George, M.D., Ph.D. Director of Wayne State University Movement Disorder Clinic University Health Center Neurology Clinic University Health The John
Dementia Medications Acetylcholinesterase Inhibitors (AChEIs) and Glutamate (NMDA) Receptor Antagonist
 Dementia Medications Acetylcholinesterase Inhibitors (AChEIs) and Glutamate (NMDA) Receptor Antagonist Medication Dosage Indication for Use Aricept (donepezil) Exelon (rivastigmine) 5mg 23mg* ODT 5mg Solution
Dementia Medications Acetylcholinesterase Inhibitors (AChEIs) and Glutamate (NMDA) Receptor Antagonist Medication Dosage Indication for Use Aricept (donepezil) Exelon (rivastigmine) 5mg 23mg* ODT 5mg Solution
Pharmacy STROKE. Anne Kinnear Lead Pharmacist NHS Lothian. Educational Solutions for Workforce Development
 STROKE Anne Kinnear Lead Pharmacist NHS Lothian Aim To update pharmacists on Stroke: the disease and its management and explore ways to implement pharmaceutical care for this patient group as part of normal
STROKE Anne Kinnear Lead Pharmacist NHS Lothian Aim To update pharmacists on Stroke: the disease and its management and explore ways to implement pharmaceutical care for this patient group as part of normal
Stroke: What did we learn in the last year?
 Stroke: What did we learn in the last year? J. Claude Hemphill III, MD, MAS Associate Professor of Clinical Neurology and Neurological Surgery University of California, San Francisco Director, Neurocritical
Stroke: What did we learn in the last year? J. Claude Hemphill III, MD, MAS Associate Professor of Clinical Neurology and Neurological Surgery University of California, San Francisco Director, Neurocritical
Results from RE-LY and RELY-ABLE
 Results from RE-LY and RELY-ABLE Assessment of the safety and efficacy of dabigatran etexilate (Pradaxa ) in longterm stroke prevention EXECUTIVE SUMMARY Dabigatran etexilate (Pradaxa ) has shown a consistent
Results from RE-LY and RELY-ABLE Assessment of the safety and efficacy of dabigatran etexilate (Pradaxa ) in longterm stroke prevention EXECUTIVE SUMMARY Dabigatran etexilate (Pradaxa ) has shown a consistent
Is there enough evidence for DAPT after endovascular intervention for PAOD?
 Is there enough evidence for DAPT after endovascular intervention for PAOD? Prof. I. Baumgartner Head Clinical & Interventional Angiology University Hospital Bern Disclosure Speaker name:...i. Baumgartner...
Is there enough evidence for DAPT after endovascular intervention for PAOD? Prof. I. Baumgartner Head Clinical & Interventional Angiology University Hospital Bern Disclosure Speaker name:...i. Baumgartner...
Origins. Update on Parkinson s Disease
 Update on Parkinson s Disease Melinda Burnett, MD Origins First described in 1817 by James Parkinson: Involuntary tremulous motion, with lessened muscular power, in parts not in action and even when supported;
Update on Parkinson s Disease Melinda Burnett, MD Origins First described in 1817 by James Parkinson: Involuntary tremulous motion, with lessened muscular power, in parts not in action and even when supported;
Curriculum Vitae, Glenn Michael Dempsey, M.D.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com EDUCATION: Glenn Michael Dempsey,
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com EDUCATION: Glenn Michael Dempsey,
Chapter 20. Media Directory. Amyotrophic Lateral Sclerosis. Alzheimer s Disease. Huntington s Chorea. Multiple Sclerosis
 Chapter 20 Drugs for Degenerative Diseases of the Nervous System Slide 18 Media Directory Levadopa Animation Upper Saddle River, New Jersey 07458 All rights reserved. Alzheimer s Disease Amyotrophic Lateral
Chapter 20 Drugs for Degenerative Diseases of the Nervous System Slide 18 Media Directory Levadopa Animation Upper Saddle River, New Jersey 07458 All rights reserved. Alzheimer s Disease Amyotrophic Lateral
Gateshead Pain Guidelines for Chronic Conditions
 Gateshead Pain Guidelines for Chronic Conditions Effective Date: 13.2.2013 Review Date: 13.2.2015 Gateshead Pain Guidelines: Contents PAIN GUIDELINES Chronic Non-Malignant Pain 5 Musculoskeletal Pain 6
Gateshead Pain Guidelines for Chronic Conditions Effective Date: 13.2.2013 Review Date: 13.2.2015 Gateshead Pain Guidelines: Contents PAIN GUIDELINES Chronic Non-Malignant Pain 5 Musculoskeletal Pain 6
Biogen, Cambridge, MA, USA; 2 Cytel, Cambridge, MA, USA; 3 Neurimmune, Schlieren-Zurich, and University of Zurich, Switzerland
 Aducanumab 36-Month Data From PRIME: A Randomized, Double-Blind, Placebo-Controlled Phase 1b Study in Patients With Prodromal or Mild Alzheimer s Disease Samantha Budd Haeberlein, PhD 1, Sarah Gheuens,
Aducanumab 36-Month Data From PRIME: A Randomized, Double-Blind, Placebo-Controlled Phase 1b Study in Patients With Prodromal or Mild Alzheimer s Disease Samantha Budd Haeberlein, PhD 1, Sarah Gheuens,
Review of Anticonvulsant Medications: Traditional and Alternative Uses. Andrea Michel, PharmD, CACP
 Review of Anticonvulsant Medications: Traditional and Alternative Uses Andrea Michel, PharmD, CACP Objectives Review epidemiology of epilepsy Classify types of seizures Discuss non-pharmacologic and pharmacologic
Review of Anticonvulsant Medications: Traditional and Alternative Uses Andrea Michel, PharmD, CACP Objectives Review epidemiology of epilepsy Classify types of seizures Discuss non-pharmacologic and pharmacologic
Curriculum Vitae, Glenn Michael Dempsey, M.D.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com EDUCATION: Glenn Michael Dempsey,
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com EDUCATION: Glenn Michael Dempsey,
CEREBRO VASCULAR ACCIDENTS
 CEREBRO VASCULAR S MICHAEL OPONG-KUSI, DO MBA MORTON CLINIC, TULSA, OK, USA 8/9/2012 1 Cerebrovascular Accident Third Leading cause of deaths (USA) 750,000 strokes in USA per year. 150,000 deaths in USA
CEREBRO VASCULAR S MICHAEL OPONG-KUSI, DO MBA MORTON CLINIC, TULSA, OK, USA 8/9/2012 1 Cerebrovascular Accident Third Leading cause of deaths (USA) 750,000 strokes in USA per year. 150,000 deaths in USA
Type of intervention Secondary prevention. Economic study type Cost-effectiveness analysis.
 Aspirin plus extended-release dipyridamole or clopidogrel compared with aspirin monotherapy for the prevention of recurrent ischemic stroke: a cost-effectiveness analysis Shah H, Gondek K Record Status
Aspirin plus extended-release dipyridamole or clopidogrel compared with aspirin monotherapy for the prevention of recurrent ischemic stroke: a cost-effectiveness analysis Shah H, Gondek K Record Status
Outlines! Current Issue in Management. of Dementia! Cholinesterase Inhibitors! Medications that soon to be used in. Papan Thaipisuttikul, M.D.
 Current Issue in Management of Dementia! Papan Thaipisuttikul, M.D. Department of Psychiatry Ramathibodi Hospital! Outlines! Current medications use in dementia management in Thailand. Medications that
Current Issue in Management of Dementia! Papan Thaipisuttikul, M.D. Department of Psychiatry Ramathibodi Hospital! Outlines! Current medications use in dementia management in Thailand. Medications that
Pros and Cons of Individual Agents Based on Large Trial Results: RELY, ROCKET, ARISTOTLE, AVERROES
 Pros and Cons of Individual Agents Based on Large Trial Results: RELY, ROCKET, ARISTOTLE, AVERROES Ralph L. Sacco, MS MD FAAN FAHA Olemberg Family Chair in Neurological Disorders Miller Professor of Neurology,
Pros and Cons of Individual Agents Based on Large Trial Results: RELY, ROCKET, ARISTOTLE, AVERROES Ralph L. Sacco, MS MD FAAN FAHA Olemberg Family Chair in Neurological Disorders Miller Professor of Neurology,
The Use of Amantadine HCL in Clinical Practice: A Study of Old and New Indications
 The Use of Amantadine HCL in Clinical Practice: A Study of Old and New Indications Carlos Singer, MD* Spiridon Papapetropoulos, MD, PhD* Gadith Uzcategui, BA Lydia Vela, MD * Department of Neurology, University
The Use of Amantadine HCL in Clinical Practice: A Study of Old and New Indications Carlos Singer, MD* Spiridon Papapetropoulos, MD, PhD* Gadith Uzcategui, BA Lydia Vela, MD * Department of Neurology, University
Chapter Fifteen. Neurological Disorders
 Chapter Fifteen Neurological Disorders Causes of Neurological Disorders Head Injuries Tumors Seizures Drugs (primary effects, side effects, and withdrawal) Circulation Issues Circulation Issues STROKES!
Chapter Fifteen Neurological Disorders Causes of Neurological Disorders Head Injuries Tumors Seizures Drugs (primary effects, side effects, and withdrawal) Circulation Issues Circulation Issues STROKES!
