SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary
|
|
|
- Marian Greer
- 5 years ago
- Views:
Transcription
1 July 1, 2015 Bulletin #154 ISSN SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary New Exception Drug Status (EDS) Listings Effective July 1, 2015 according to the following criteria: aripiprazole, 300mg, 400 mg, long acting injection (Abilify Maintena - OTS) For treatment of patients exhibiting a compliance problem with an oral antipsychotic and in whom the administration of a conventional injectable extended action antipsychotic is ineffective or poorly tolerated. eplerenone, 25mg, 50mg tablet (Inspra-PFI) For treatment of patients with New York Heart Association (NYHA) class II chronic heart failure with left ventricular systolic dysfunction (with ejection fraction 35%), as an adjunct to standard therapy Note: patients must be on optimal therapy with an angiotensin-converting enzyme (ACE) inhibitor, an angiotensin-receptor blocker (ARB), or both and a beta-blocker (unless contraindicated) at the recommended dose or maximal tolerated dose. indacaterol/glycopyrronium, inhalation powder capsule, 100mcg/50mcg (Ultibro Breezhaler-NVR) For treatment of airflow obstruction in patients with moderate to severe COPD, as defined by spirometry, who have had an inadequate response to a long-acting beta-2 agonist (LABA), OR a long-acting muscarinic antagonist (LAMA). lurasidone hydrochloride, 40mg, 80mg, 120mg tablet (Latuda SNV) For manifestations of schizophrenia. riociguat, tablet, 0.5mg, 1.0mg, 1.5mg, 2.0mg, 2.5mg (Adempas BAY) For treatment of patients 18 years of age or older with chronic thromboembolic pulmonary hypertension (CTEPH) with World Health Organization (WHO) Functional Class 2 or 3 pulmonary hypertension, with;
2 - 2 - a) inoperable chronic thromboembolic pulmonary hypertension (CTEPH), World Health Organization (WHO) Group 4, OR b) persistent or recurrent CTEPH after surgical treatment. Note: must be prescribed by clinicians experienced in the diagnosis and treatment of CTEPH. romiplostim, solution for injection, 250 mcg/0.5 ml, 500mcg/mL, (Nplate-AMG) For the treatment of refractory chronic idiopathic thrombocytopenic purpura ( ITP ) with bleeding complications in patients who meet the following conditions: a) have undergone a splenectomy 1 ; and b) have tried and are unresponsive to other treatment modalities 2. Dosage: To a maximum of 10 mcg/kg once weekly. Renewal of requests for romiplostim will be assessed on a case-by-case basis. Note: After one year of continuous treatment, therapeutic options should be reassessed. 1. Where surgery is contraindicated, the requesting physician must provide a rationale for why a splenectomy cannot be considered, and where possible, include both a preoperative/surgical evaluation of the patient s risks and a consideration of risks of laparoscopic and open surgical interventions if these are available. The requesting physician s rationale must be evaluated by an independent physician. 2. Patients must be refractory to two of the following first line treatment modalities: Corticosteroids IV anti-d Intravenous immune globulin (IVIG) In addition, patients must be refractory to two of the following second-line treatment modalities: Azathioprine Cyclosporine Cyclophosphamide Mycophenolate Rituximab Danazol Dapsone stiripental, capsules and powder for suspension, 250 mg and 500 mg (Diacomit BCX) For use in combination with clobazam and valproate as adjunctive therapy of refractory generalized tonic-clonic seizures in patients with severe myoclonic epilepsy in infancy (Dravet syndrome), whose seizures are not adequately controlled with clobazam and valproate alone. Note: The patient must be under the care of a neurologist or a pediatrician.
3 - 3 - umeclidinium bromide/vilanterol trifenatate, powder for inhalation 62.5/25mcg (Anoro Ellipta-GSK) For treatment of airflow obstruction in patients with moderate to severe COPD, as defined by spirometry, who have had an inadequate response to a long-acting beta-2 agonist (LABA), OR a long-acting muscarinic antagonist (LAMA). New Exception Drug Status (EDS) Listings Effective July 1, 2015 with online adjudication according to the following criteria: aflibercept 40mg/mL injection (Eylea-BAY) Treatment of wamd For the treatment of neovascular (wet) age-related macular degeneration (AMD) if all of the following circumstances apply to the eye to be treated: (i) The best corrected visual acuity (BCVA) is between 6/12 and 6/96 (ii) The lesion size is less than or equal to 12 disc areas in greatest linear dimension (iii)there is evidence of recent (< 3 months) presumed disease progression (blood vessel growth, as indicated by fluorescein angiography, optical coherence tomography (OCT) or recent visual acuity changes); and (iv) Injection will be by a qualified ophthalmologist with experience in intravitreal injections Coverage will not be provided for patients: (a) With permanent structural damage to the central fovea or no active disease (as defined in the Royal College of Ophthalmology guidelines); and (b) Receiving concurrent verteporfin PDT treatment. The interval between the doses should be no shorter than one month. Treatment with aflibercept should be continued only in people who maintain adequate response to therapy. Aflibercept should be permanently discontinued if any one of the following occurs: (a) Reduction in BCVA in the treated eye to less than 15 letters (absolute) on 2 consecutive visits in the treated eye, attributed to AMD in the absence of other pathology. (b) Reduction in BCVA of 30 letters or more compared to either baseline and/or best recorded level since baseline, as this may indicate either poor treatment effect or adverse event or both. (c) There is evidence of deterioration of the lesion morphology despite optimum treatment over three consecutive visits. Treatment of DME For the treatment of visual impairment due to Diabetic Macular Edema (DME) for patients meeting all of the following: (i.) Diffuse DME involving the central fovea with central fovea thickness of 300 microns or greater on optical coherence tomography (OCT) and vision less than 20/32.
4 (ii.) Patients with focal macular edema for which laser photocoagulation is indicated should be treated with laser, except in situations where focal laser therapy treatment can not be safely performed due to the proximity of microaneurysms to the fovea. (iii.) A haemoglobin A1c of less than 11%. (iv.) (v.) (vi.) (vii.) Note: Treatment should be discontinued if there is no improvement of retinal thickness on OCT or if there is no improvement in visual acuity after five consecutive treatments. The interval between two doses should not be shorter than one month. Patients responding to treatment should be monitored at regular intervals up to monthly for visual acuity AND retinal thickness. Injection will be by a qualified ophthalmologist with experience in intravitreal injections. Fluorescein Angiography (FA) should be considered prior to initiation of treatment to assess perfusion and characterize the leakage, and should also be considered if the patient is not responding to treatment as expected. Treatment of CRVO For the treatment of visual impairment due to clinically significant macular edema secondary to central retinal vein occlusion (CRVO) for patients meeting all of the following: (i.) (ii.) (iii.) (iv.) (v.) Diffuse CRVO with macular thickness of 300 microns or greater on Optical Coherence Tomography (OCT) and a vision of 20/40 or less. The interval between two doses should not be shorter than one month. Patients should be monitored at regular intervals up to monthly for retinal thickness and visual acuity. Treatment should be discontinued if there is no improvement after 6 months of initial treatment; and Injection will be by a qualified ophthalmologist with experience in administering intravitreal injections. Note: Fluorescein Angiography (FA) should be considered prior to initiation of treatment to assess perfusion and characterize the leakage, and should also be considered if the patient is not responding to treatment as expected. dolutegravir, tablet, 50 mg (Tivicay VII) For management of HIV disease in patients 12 years of age and older. This drug, as with other antivirals in the treatment of HIV, should be used under the direction of an infectious disease specialist. Note: Tivicay is not recommended for patients weighing less than 40 kgs. When prescribed by, or on the advice of an Infectious Disease specialist familiar with HIV treatment, for post-exposure prophylaxis (PEP).
5 - 5 - Alternate regimen for patients in whom compliance may be an issue. This regimen may improve adherence and has reduced potential for drug interactions. mirabegron extended release tablet, 25 mg, 50 mg (Myrbetriq-APC) For treatment of overactive bladder (OAB) for patients intolerant to, or with an inadequate response to oxybutynin. Note: Should not be used in combination with other pharmacologic treatments for OAB. Additional Formulations of a Current Exception Drug Status (EDS) Listing effective July 1, 2015 according to the following criteria: dalteparin sodium, syringe, 3,500 IU (0.28mL (Fragmin-PFI) (a) For treatment of venous thromboembolism for up to 10 days. (b) For prophylaxis following total knee arthroplasty for up to 35 days. (c) For major orthopedic trauma for up to10 days (treatment duration may be reassessed). (d) For long-term outpatient prophylaxis in patients who are pregnant. (e) For long-term outpatient prophylaxis in patients who have a contraindication to, are intolerant to, or have failed, warfarin therapy. (f) For long-term outpatient prophylaxis in patients who have lupus anticoagulant syndrome. (g) Prophylaxis in patients undergoing total hip replacement or following hip fracture surgery for up to 35 days following the procedure. Revised Exception Drug Status Criteria; (Prescribers No Longer Required to Submit a Medication List; See Bold Italicized Portion): donepezil HCl, tablet, 5mg, 10mg (Aricept-PFI, and generics) (a) A diagnosis of probable Alzheimer's disease as per DSM-V criteria. All other criteria remains unchanged. rivastigmine, capsule, 1.5mg, 3mg, 4.5mg, 6mg (Exelon-NVR, and generics); oral solution, 2mg/mL ((Exelon-NVR) (a) A diagnosis of probable Alzheimer's disease as per DSM-V criteria. All other criteria remains unchanged. galantamine hydrobromide, extended release capsule, 8mg, 16mg, 24mg (Reminyl ER-JAN, and generics) (a) A diagnosis of probable Alzheimer's disease as per DSM-V criteria. All other criteria remains unchanged. New Exception Drug Status Criteria (in addition to existing criteria): norfloxacin, tablet, 400mg (Apo-Norflox-APX) (Novo-Norfloxacin-NOP) (pms-norfloxacin-pms)
6 - 6 - (e) For secondary prophylaxis in patients who have had an episode of spontaneous bacterial peritonitis and are intolerant or unresponsive to sulfamethoxazole/trimethoprim (f) For primary prophylaxis for patients with cirrhosis considered high risk for spontaneous bacterial peritonitis who are intolerant to sulfamethoxazole/trimethoprim. Note: High risk is defined as cirrhosis with ascities with an ascitic protein concentration less than 15g/L ranibizumab, 10 mg/ml solution for intravitreal injection (Lucentis-NVR) For treatment of visual impairment due to choroidal neovascularization secondary to pathologic myopia. Must be administered by a qualified ophthalmologist with experience in intravitreal injections. Drugs Reviewed and Not Approved for Listing in the Saskatchewan Formulary: eltrombopag, tablet, 25mg, 50mg (Revolade-GSK) for thrombocytopenia associated with chronic hepatitis C infection pasireotide diaspartate, 0.3 mg/ml, 0.6 mg/ml and 0.9 mg/ml solution for injection (Signifor-NVR) for Cushing s disease lomitapide, capsules, 5 mg, 10 mg, 20 mg (Juxtapid -APL) for homozygous familial hypercholesterolemia Saskatchewan Ministry of Health Drug Plan and Extended Benefits Branch 2 nd Floor, 3475 Albert Street Regina, Saskatchewan S4S 6X6 (306) This Bulletin is not to be reproduced or republished except with the approval of the Saskatchewan Ministry of Health. Inquiries should be directed to the address or telephone numbers shown at left.
P.E.I. Drug Programs. Formulary Update. Issue June 09, 2010
 P.E.I. Drug Programs Formulary Update Issue 10-01 June 09, 2010 st Effective July 1, 2010, the following medications will be added to the P.E.I. Drug Formulary. New medications for the treatment of ankylosing
P.E.I. Drug Programs Formulary Update Issue 10-01 June 09, 2010 st Effective July 1, 2010, the following medications will be added to the P.E.I. Drug Formulary. New medications for the treatment of ankylosing
Updates to the Alberta Drug Benefit List. Effective August 1, 2017
 Updates to the Alberta Drug Benefit List Effective August 1, 2017 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780) 498-8370
Updates to the Alberta Drug Benefit List Effective August 1, 2017 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780) 498-8370
SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary
 July 1, 2017 Bulletin #166 ISSN 1923-0761 SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary Recommended as a full Formulary benefit: adapalene/benzoyl peroxide, topical
July 1, 2017 Bulletin #166 ISSN 1923-0761 SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary Recommended as a full Formulary benefit: adapalene/benzoyl peroxide, topical
NB Drug Plans Formulary Update
 Bulletin # 914 October 14, 2015 NB Drug Plans Formulary Update This update to the New Brunswick Drug Plans Formulary is effective October 14, 2015. Included in this bulletin: Special Authorization Benefit
Bulletin # 914 October 14, 2015 NB Drug Plans Formulary Update This update to the New Brunswick Drug Plans Formulary is effective October 14, 2015. Included in this bulletin: Special Authorization Benefit
New Exception Status Benefits
 SEPTEMBER 2015 Nova Scotia Formulary Updates New Exception Status Benefits Holkira Pak (ombitasvir/paritaprevir/ ritonavir and dasabuvir) Moderiba (ribavirin) Abilify Maintena (aripiprazole) Invokana (canagliflozin)
SEPTEMBER 2015 Nova Scotia Formulary Updates New Exception Status Benefits Holkira Pak (ombitasvir/paritaprevir/ ritonavir and dasabuvir) Moderiba (ribavirin) Abilify Maintena (aripiprazole) Invokana (canagliflozin)
SASKATCHEWAN FORMULARY BULLETIN. Update to the 60th Edition of the Saskatchewan Formulary
 Saskatchewan Ministry of Health July 1, 2010 Drug Plan and Extended Benefits Branch Bulletin #124 ISSN 1923-077X SASKATCHEWAN FORMULARY BULLETIN Update to the 60th Edition of the Saskatchewan Formulary
Saskatchewan Ministry of Health July 1, 2010 Drug Plan and Extended Benefits Branch Bulletin #124 ISSN 1923-077X SASKATCHEWAN FORMULARY BULLETIN Update to the 60th Edition of the Saskatchewan Formulary
SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary
 April 1, 2018 Bulletin #169 ISSN 1923-0761 SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary Recommended as a full Formulary benefit: benztropine mesylate, tablet,
April 1, 2018 Bulletin #169 ISSN 1923-0761 SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary Recommended as a full Formulary benefit: benztropine mesylate, tablet,
Ophthalmic VEGF Inhibitors. Eylea (aflibercept), Macugen (pegaptanib) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 2Q17 April May
 BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
Ophthalmology Macular Pathways
 Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England. Retinal Vein Occlusion (RVO) with Macular oedema (MO)
 Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England (Royal Victoria Infirmary, Sunderland Eye Infirmary, James Cook University Hospital, Darlington Memorial Hospital, University Hospital
Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England (Royal Victoria Infirmary, Sunderland Eye Infirmary, James Cook University Hospital, Darlington Memorial Hospital, University Hospital
SASKATCHEWAN FORMULARY COMMITTEE UPDATE BULLETIN TO THE 55th EDITION OF THE SASKATCHEWAN FORMULARY
 Saskatchewan Health Drug Plan and Extended Benefits Branch April 2006 Bulletin #106 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE UPDATE BULLETIN TO THE 55th EDITION OF THE SASKATCHEWAN FORMULARY All
Saskatchewan Health Drug Plan and Extended Benefits Branch April 2006 Bulletin #106 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE UPDATE BULLETIN TO THE 55th EDITION OF THE SASKATCHEWAN FORMULARY All
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Health Technology Appraisal. Aflibercept for treating diabetic macular oedema.
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
Non-Insured Health Benefits
 Winter 2017 Non-Insured Health Benefits First Nations and Inuit Health Branch Updates to the Drug Benefit List The Non-Insured Health Benefits (NIHB) Program provides supplementary health benefits, including
Winter 2017 Non-Insured Health Benefits First Nations and Inuit Health Branch Updates to the Drug Benefit List The Non-Insured Health Benefits (NIHB) Program provides supplementary health benefits, including
Treatment pathway for adult patients with immune (idiopathic) thrombocytopenic purpura (ITP)
 Prescribing Clinical Network Surrey (East Surrey CCG, Guildford & Waverley CCG, North West Surrey CCG, Surrey Downs CCG & Surrey Heath CCG) Crawley and Horsham & Mid-Sussex CCG Treatment pathway for adult
Prescribing Clinical Network Surrey (East Surrey CCG, Guildford & Waverley CCG, North West Surrey CCG, Surrey Downs CCG & Surrey Heath CCG) Crawley and Horsham & Mid-Sussex CCG Treatment pathway for adult
Management of Neovascular AMD
 Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
aflibercept 40mg/mL solution for injection (Eylea ) SMC No. (1074/15) Bayer
 aflibercept 40mg/mL solution for injection (Eylea ) SMC No. (1074/15) Bayer 07 August 2015 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS Boards
aflibercept 40mg/mL solution for injection (Eylea ) SMC No. (1074/15) Bayer 07 August 2015 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS Boards
SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary
 April 1, 2018 Bulletin #170 ISSN 1923-0761 SASKATCHEWAN FMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary Related Information for Prescribers: The April 1, 2018 updates to existing
April 1, 2018 Bulletin #170 ISSN 1923-0761 SASKATCHEWAN FMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary Related Information for Prescribers: The April 1, 2018 updates to existing
Clinically Significant Macular Edema (CSME)
 Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Ophthalmologic Policy. Vascular Endothelial Growth Factor (VEGF) Inhibitors
 Ophthalmologic Policy UnitedHealthcare Commercial Drug Policy Vascular Endothelial Growth Factor (VEGF) Inhibitors Policy Number: 2016D0042H Effective Date: October 1, 2016 Table of Contents Page INSTRUCTIONS
Ophthalmologic Policy UnitedHealthcare Commercial Drug Policy Vascular Endothelial Growth Factor (VEGF) Inhibitors Policy Number: 2016D0042H Effective Date: October 1, 2016 Table of Contents Page INSTRUCTIONS
Ontario Drug Benefit Formulary/Comparative Drug Index
 Ontario Drug Benefit Formulary/Comparative Drug Index Edition 42 Summary of Changes - July 2015 Effective July 29, 2015 Ministry of Health and Long-Term Care Table of Contents New Single Source Products...
Ontario Drug Benefit Formulary/Comparative Drug Index Edition 42 Summary of Changes - July 2015 Effective July 29, 2015 Ministry of Health and Long-Term Care Table of Contents New Single Source Products...
There are no published randomized, double-blind trials comparing aflibercept to other therapies in neovascular AMD.
 Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
SASKATCHEWAN FORMULARY COMMITTEE UPDATE BULLETIN 51st Edition
 Saskatchewan Health Drug Plan and Extended Benefits Branch July, 2002 Bulletin #91 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE UPDATE BULLETIN 51st Edition NEW LISTINGS NEW EXCEPTION DRUG STATUS AGENT
Saskatchewan Health Drug Plan and Extended Benefits Branch July, 2002 Bulletin #91 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE UPDATE BULLETIN 51st Edition NEW LISTINGS NEW EXCEPTION DRUG STATUS AGENT
Treatment of Age Related Macular Degeneration (AMD) by Intravitreal Injection
 Information for Patients Manchester Royal Eye Hospital Medical Retina Services Treatment of Age Related Macular Degeneration (AMD) by Intravitreal Injection What is age related macular degeneration (AMD)?
Information for Patients Manchester Royal Eye Hospital Medical Retina Services Treatment of Age Related Macular Degeneration (AMD) by Intravitreal Injection What is age related macular degeneration (AMD)?
The Era of anti- - - VEGF Kirk L. Halvorson, OD
 The Era of anti- - - VEGF Kirk L. Halvorson, OD Introduction: Anti- - - Vascular Endothelial Growth Factor (Anti- - - VEGF) medication is a relatively a new line of medications used in treating a variety
The Era of anti- - - VEGF Kirk L. Halvorson, OD Introduction: Anti- - - Vascular Endothelial Growth Factor (Anti- - - VEGF) medication is a relatively a new line of medications used in treating a variety
Thrombopoietin Receptor Agonists Prior Authorization with Quantity Limit Program Summary
 Thrombopoietin Receptor Agonists Prior Authorization with Quantity Limit Program Summary Thrombopoietin Receptor Agonists Prior Authorization with Quantity Limit OBJECTIVE The intent of the prior authorization
Thrombopoietin Receptor Agonists Prior Authorization with Quantity Limit Program Summary Thrombopoietin Receptor Agonists Prior Authorization with Quantity Limit OBJECTIVE The intent of the prior authorization
Eye injection treatment costs and rebates
 Eye injection treatment costs and rebates This fact sheet provides general information on the bill you receive from your ophthalmologist for eye injections for the management of wet macular degeneration,
Eye injection treatment costs and rebates This fact sheet provides general information on the bill you receive from your ophthalmologist for eye injections for the management of wet macular degeneration,
Vascular Disease Ocular Manifestations of Systemic Hypertension
 Vascular Disease Ocular Manifestations of Systemic Hypertension Maynard L. Pohl, OD, FAAO Pacific Cataract & Laser Institute 10500 NE 8 th Street, Suite 1650 Bellevue, WA 98004 USA 425-462-7664 Cerebrovascular
Vascular Disease Ocular Manifestations of Systemic Hypertension Maynard L. Pohl, OD, FAAO Pacific Cataract & Laser Institute 10500 NE 8 th Street, Suite 1650 Bellevue, WA 98004 USA 425-462-7664 Cerebrovascular
eltrombopag (Promacta )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) DABIGATRAN RECOMMENDED What it is Indications Date decision last revised
 Name: generic (trade) Dabigatran etexilate (Pradaxa ) HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) DABIGATRAN RECOMMENDED What it is Indications Date decision last revised Direct thrombin inhibitor
Name: generic (trade) Dabigatran etexilate (Pradaxa ) HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) DABIGATRAN RECOMMENDED What it is Indications Date decision last revised Direct thrombin inhibitor
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Romiplostim Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 12/15/2017 Next
Cigna Drug and Biologic Coverage Policy Subject Romiplostim Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 12/15/2017 Next
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Diagnosis and treatment of diabetic retinopathy. Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City
 Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
SASKATCHEWAN FORMULARY COMMITTEE UPDATE BULLETIN TO THE 55th EDITION OF THE SASKATCHEWAN FORMULARY
 Saskatchewan Health Drug Plan and Extended Benefits Branch October 2005 Bulletin #104 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE UPDATE BULLETIN TO THE 55th EDITION OF THE SASKATCHEWAN FORMULARY All
Saskatchewan Health Drug Plan and Extended Benefits Branch October 2005 Bulletin #104 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE UPDATE BULLETIN TO THE 55th EDITION OF THE SASKATCHEWAN FORMULARY All
Scottish Medicines Consortium
 Scottish Medicines Consortium eltrombopag, 25mg and 50mg film-coated tablets (Revolade ) No. (625/10) GlaxoSmithKline UK 09 July 2010 The Scottish Medicines Consortium (SMC) has completed its assessment
Scottish Medicines Consortium eltrombopag, 25mg and 50mg film-coated tablets (Revolade ) No. (625/10) GlaxoSmithKline UK 09 July 2010 The Scottish Medicines Consortium (SMC) has completed its assessment
Clinical Trials Related to Age Related Macular Degeneration
 Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad
Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad
From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014
 From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014 1. This course is designed to review the important ophthalmic literature that was released between October 2013
From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014 1. This course is designed to review the important ophthalmic literature that was released between October 2013
SASKATCHEWAN FORMULARY COMMITTEE BULLETIN UPDATE TO THE 57th EDITION OF THE SASKATCHEWAN FORMULARY
 Saskatchewan Health Drug Plan and Extended Benefits Branch April 2008 Bulletin #114 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE BULLETIN UPDATE TO THE 57th EDITION OF THE SASKATCHEWAN FORMULARY The
Saskatchewan Health Drug Plan and Extended Benefits Branch April 2008 Bulletin #114 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE BULLETIN UPDATE TO THE 57th EDITION OF THE SASKATCHEWAN FORMULARY The
The Trust does not record electronically the number of patients who are treated by laser for a condition.
 Ref: FOI/CAD/ID 3224 06 April 20 Please reply to: FOI Administrator Trust Management Maidstone Hospital Hermitage Lane Maidstone Kent ME 9QQ Email: mtw-tr.foiadmin@nhs.net Freedom of Information Act 2000
Ref: FOI/CAD/ID 3224 06 April 20 Please reply to: FOI Administrator Trust Management Maidstone Hospital Hermitage Lane Maidstone Kent ME 9QQ Email: mtw-tr.foiadmin@nhs.net Freedom of Information Act 2000
THE ROLE OF anti-vegf IN DIABETIC RETINOPATHY AND AGE RELATED MACULAR DEGENERATION
 THE ROLE OF anti-vegf IN DIABETIC RETINOPATHY AND AGE RELATED MACULAR DEGENERATION MOESTIDJAB DEPARTMENT OF OPHTHALMOLOGY SCHOOL OF MEDICINE AIRLANGGA UNIVERSITY DR SOETOMO HOSPITAL SURABAYA INTRODUCTION
THE ROLE OF anti-vegf IN DIABETIC RETINOPATHY AND AGE RELATED MACULAR DEGENERATION MOESTIDJAB DEPARTMENT OF OPHTHALMOLOGY SCHOOL OF MEDICINE AIRLANGGA UNIVERSITY DR SOETOMO HOSPITAL SURABAYA INTRODUCTION
Scottish Medicines Consortium
 Scottish Medicines Consortium romiplostim, 250 microgram vial of powder for solution for subcutaneous injection (Nplate ) No. (553/09) Amgen 08 May 2009 (Issued 4 September 2009) The Scottish Medicines
Scottish Medicines Consortium romiplostim, 250 microgram vial of powder for solution for subcutaneous injection (Nplate ) No. (553/09) Amgen 08 May 2009 (Issued 4 September 2009) The Scottish Medicines
Clinical Policy: Roflumilast (Daliresp) Reference Number: CP.PMN.46 Effective Date: Last Review Date: 08.18
 Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11.01.11 Last Review Date: 08.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11.01.11 Last Review Date: 08.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Macular edema (ME) is the most common
 MANAGEMENT OF RETINAL VEIN OCCLUSIONS * Peter A. Campochiaro, MD ABSTRACT Macular edema (ME) is the most common cause of reduced vision in patients with retinal vein occlusions (RVOs). The primary cause
MANAGEMENT OF RETINAL VEIN OCCLUSIONS * Peter A. Campochiaro, MD ABSTRACT Macular edema (ME) is the most common cause of reduced vision in patients with retinal vein occlusions (RVOs). The primary cause
Clinical Policy: Dalteparin (Fragmin) Reference Number: ERX.SPA.207 Effective Date:
 Clinical Policy: (Fragmin) Reference Number: ERX.SPA.207 Effective Date: 01.11.17 Last Review Date: 02.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Fragmin) Reference Number: ERX.SPA.207 Effective Date: 01.11.17 Last Review Date: 02.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Diabetic Macular Edema Treatment in the 21st Century
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Diabetic Retinopathy A Presentation for the Public
 Diabetic Retinopathy A Presentation for the Public Ray M. Balyeat, MD The Eye Institute Tulsa, Oklahoma The Healthy Eye Light rays enter the eye through the cornea, pupil and lens. These light rays are
Diabetic Retinopathy A Presentation for the Public Ray M. Balyeat, MD The Eye Institute Tulsa, Oklahoma The Healthy Eye Light rays enter the eye through the cornea, pupil and lens. These light rays are
OPHTHALMOLOGIC POLICY: VASCULAR ENDOTHELIAL GROWTH FACTOR (VEGF) INHIBITORS
 UnitedHealthcare Commercial Medical Benefit Drug Policy OPHTHALMOLOGIC POLICY: VASCULAR ENDOTHELIAL GROWTH FACTOR (VEGF) INHIBITORS Policy Number: PHA020 Effective Date: September 1, 2018 Table of Contents
UnitedHealthcare Commercial Medical Benefit Drug Policy OPHTHALMOLOGIC POLICY: VASCULAR ENDOTHELIAL GROWTH FACTOR (VEGF) INHIBITORS Policy Number: PHA020 Effective Date: September 1, 2018 Table of Contents
2. Does the patient have a diagnosis of chronic idiopathic thrombocytopenic purpura (ITP)?
 Pharmacy Prior Authorization MERC CARE (MEDICAID) Promacta (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE (MEDICAID) Promacta (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization
 Pharmacy Prior Authorization MERC CARE (MEDICAID) Promacta (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE (MEDICAID) Promacta (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Promacta (eltrombopag)
 Promacta (eltrombopag) Policy Number: 5.01.542 Last Review: 5/2018 Origination: 6/2013 Next Review: 5/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Promacta
Promacta (eltrombopag) Policy Number: 5.01.542 Last Review: 5/2018 Origination: 6/2013 Next Review: 5/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Promacta
International Journal of Health Sciences and Research ISSN:
 International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
April May For adults for the treatment of visual impairment due to macular oedema secondary to central retinal vein occlusion.
 April May 2014 Recommended for use within NHS Scotland April May 2014 aflibercept intravitreal (Eylea ) 954/14 For adults for the treatment of visual impairment due to macular oedema secondary to central
April May 2014 Recommended for use within NHS Scotland April May 2014 aflibercept intravitreal (Eylea ) 954/14 For adults for the treatment of visual impairment due to macular oedema secondary to central
QUICK REFERENCE Clinical Practice Guideline on the Evaluation and Management of Immune Thrombocytopenia (ITP)
 QUICK REFERENCE 2011 Clinical Practice Guideline on the Evaluation and Management of Immune Thrombocytopenia (ITP) Presented by the American Society of Hematology, adapted from: The American Society of
QUICK REFERENCE 2011 Clinical Practice Guideline on the Evaluation and Management of Immune Thrombocytopenia (ITP) Presented by the American Society of Hematology, adapted from: The American Society of
Clinical Policy: Aflibercept (Eylea) Reference Number: CP.PHAR.184
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial
 ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
The Foundation WHAT IS THE RETINA?
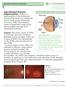 Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187
 Clinical Policy: (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Although photocoagulation and photodynamic PROCEEDINGS PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION *
 PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
Bayer to showcase latest Ophthalmology research at EURETINA 2017
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com 17 th European Society of Retina Specialists (EURETINA) Congress Bayer to showcase
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com 17 th European Society of Retina Specialists (EURETINA) Congress Bayer to showcase
SMC briefing note. The following medicines were accepted for use: The following medicine has not been recommended for use: About SMC.
 Monthly briefings are produced in order to help members of the media and other interested people understand the work and advice of the Scottish Medicines Consortium. The detailed advice for each medicine
Monthly briefings are produced in order to help members of the media and other interested people understand the work and advice of the Scottish Medicines Consortium. The detailed advice for each medicine
BENEFIT CHANGES TO NBPDP
 Bulletin #789 June 15, 2010 BENEFIT CHANGES TO NBPDP This update to the New Brunswick Prescription Drug Program (NBPDP) Formulary is effective June 15, 2010. Included in this bulletin: Regular Benefit
Bulletin #789 June 15, 2010 BENEFIT CHANGES TO NBPDP This update to the New Brunswick Prescription Drug Program (NBPDP) Formulary is effective June 15, 2010. Included in this bulletin: Regular Benefit
Is OCT-A Needed As An Investigative Tool During The Management Of Diabetic Macular Edema
 Is OCT-A Needed As An Investigative Tool During The Management Of Diabetic Macular Edema Ayman M Khattab MD, FRCS Professor of Ophthalmology Cairo University Diabetic Macular Edema (DME) Diabetic macular
Is OCT-A Needed As An Investigative Tool During The Management Of Diabetic Macular Edema Ayman M Khattab MD, FRCS Professor of Ophthalmology Cairo University Diabetic Macular Edema (DME) Diabetic macular
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care
 The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Acute & Emergency Care November 2016 Association of Health Professions in Ophthalmology General
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Acute & Emergency Care November 2016 Association of Health Professions in Ophthalmology General
Pharmacy Prior Authorization
 Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Promacta (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Promacta (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
SASKATCHEWAN FORMULARY COMMITTEE BULLETIN TO THE 58th EDITION OF THE SASKATCHEWAN FORMULARY
 Saskatchewan Ministry of Health Drug Plan and Extended Benefits Branch July 2008 Bulletin #115 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE BULLETIN TO THE 58th EDITION OF THE SASKATCHEWAN FORMULARY
Saskatchewan Ministry of Health Drug Plan and Extended Benefits Branch July 2008 Bulletin #115 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE BULLETIN TO THE 58th EDITION OF THE SASKATCHEWAN FORMULARY
Clinical Policy: Eltrombopag (Promacta) Reference Number: ERX.SPA.71 Effective Date:
 Clinical Policy: (Promacta) Reference Number: ERX.SPA.71 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Promacta) Reference Number: ERX.SPA.71 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Opinion 4 December 2013
 The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 4 December 2013 LUCENTIS 10 mg/ml, solution for injection Vial of 0.23 ml (CIP: 34009 378 101 5 9) Applicant: Novartis
The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 4 December 2013 LUCENTIS 10 mg/ml, solution for injection Vial of 0.23 ml (CIP: 34009 378 101 5 9) Applicant: Novartis
SUMMARY. Heather Casparis, MD,* and Neil M. Bressler, MD MARINA AND ANCHOR
 The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
Pharmacy Prior Authorization
 Pharmacy Prior Authorization AETA BETTER HEALTH EW JERSE (MEDICAID) Anticoagulant Injectable (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Pharmacy Prior Authorization AETA BETTER HEALTH EW JERSE (MEDICAID) Anticoagulant Injectable (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Technology appraisal guidance Published: 27 April 2011 nice.org.uk/guidance/ta221
 Romiplostim for the treatment of chronic immune (idiopathic) thrombocytopenic purpura Technology appraisal guidance Published: 27 April 2011 nice.org.uk/guidance/ta221 NICE 2018. All rights reserved. Subject
Romiplostim for the treatment of chronic immune (idiopathic) thrombocytopenic purpura Technology appraisal guidance Published: 27 April 2011 nice.org.uk/guidance/ta221 NICE 2018. All rights reserved. Subject
Diabetic maculopathy 11/ An update on. Miss Vasuki Sivagnanavel
 Miss Vasuki Sivagnanavel Consultant Ophthalmologist An update on Diabetic maculopathy Despite advances in the management of diabetes, diabetic retinopathy is already the commonest cause of blindness among
Miss Vasuki Sivagnanavel Consultant Ophthalmologist An update on Diabetic maculopathy Despite advances in the management of diabetes, diabetic retinopathy is already the commonest cause of blindness among
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care
 The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Medical Retina November 2016 Association of Health Professions in Ophthalmology General basic
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Medical Retina November 2016 Association of Health Professions in Ophthalmology General basic
Eye injection treatment costs and rebates
 Eye injection treatment costs and rebates This fact sheet provides general information on the bill you receive from your ophthalmologist for eye injections for the management of wet macular degeneration,
Eye injection treatment costs and rebates This fact sheet provides general information on the bill you receive from your ophthalmologist for eye injections for the management of wet macular degeneration,
Facts About Diabetic Eye Disease
 Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Area Drug and Therapeutics Committee Prescribing Supplement No 47 July 2011
 Area Drug and Therapeutics Committee Prescribing Supplement No 47 In this issue Drugs reviewed by in May / June 2011 ADTC UPDATES ON DRUGS REVIEWED BY THE SMC The following new drugs have been reviewed
Area Drug and Therapeutics Committee Prescribing Supplement No 47 In this issue Drugs reviewed by in May / June 2011 ADTC UPDATES ON DRUGS REVIEWED BY THE SMC The following new drugs have been reviewed
IQ 532 Micropulse Green Laser treatment for Refractory Chronic Central Serous Retinopathy
 Cronicon OPEN ACCESS EC OPHTHALMOLOGY Case Report IQ 532 Micropulse Green Laser treatment for Refractory Chronic Central Serous Retinopathy Fawwaz Al Mamoori* Medical Retina Department, Eye Specialty Hospital,
Cronicon OPEN ACCESS EC OPHTHALMOLOGY Case Report IQ 532 Micropulse Green Laser treatment for Refractory Chronic Central Serous Retinopathy Fawwaz Al Mamoori* Medical Retina Department, Eye Specialty Hospital,
Clinical Policy: Roflumilast (Daliresp) Reference Number: CP.PMN.46. Line of Business: Medicaid
 Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11/11 Last Review Date: 08/17 Line of Business: Medicaid See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11/11 Last Review Date: 08/17 Line of Business: Medicaid See Important Reminder at the end of this policy for important regulatory
Eylea (aflibercept) Document Number: IC-0026
 Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
An Update on Branch Retinal Vein Occlusion Treatment Studies. Amiee Ho, O.D. Pacific University College of Optometry
 An Update on Branch Retinal Vein Occlusion Treatment Studies Amiee Ho, O.D. Pacific University College of Optometry Course Description This course focuses on current treatment options available for macular
An Update on Branch Retinal Vein Occlusion Treatment Studies Amiee Ho, O.D. Pacific University College of Optometry Course Description This course focuses on current treatment options available for macular
Treatment practice in the
 Strategies for managing neovascular AMD and in routine clinical care BY ROD MCNEIL Treatment practice in the management of neovascular age-related macular degeneration (AMD) and diabetic macular oedema
Strategies for managing neovascular AMD and in routine clinical care BY ROD MCNEIL Treatment practice in the management of neovascular age-related macular degeneration (AMD) and diabetic macular oedema
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Reference Number: CP.PMN.69 Effective Date: 11/15 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Reference Number: CP.PMN.69 Effective Date: 11/15 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important regulatory
Persistent Macular Thickening After Ranibizumab Treatment for Diabetic Macular Edema With Vision Impairment
 9:30 AM Persistent Macular Thickening After Ranibizumab Treatment for Diabetic Macular Edema With Vision Impairment Lee Jampol, MD OBJECTIVE To assess subsequent visual and anatomic outcomes of eyes with
9:30 AM Persistent Macular Thickening After Ranibizumab Treatment for Diabetic Macular Edema With Vision Impairment Lee Jampol, MD OBJECTIVE To assess subsequent visual and anatomic outcomes of eyes with
Date approved: 04/18/18. Approved by: Pharmacy and Therapeutics Quality Management Subcommittee Effective Date: Department of Origin: Pharmacy
 Integrated Healthcare Services and Criteria Document: Reference #: PC/V001 Page: 1 of 9 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services,
Integrated Healthcare Services and Criteria Document: Reference #: PC/V001 Page: 1 of 9 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services,
 measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
Drug Class Update: Vascular Endothelial Growth Factors
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
SASKATCHEWAN FORMULARY COMMITTEE BULLETIN 55th EDITION
 Saskatchewan Health Drug Plan and Extended Benefits Branch July 2005 Bulletin #103 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE BULLETIN 55th EDITION All listings are effective July 1, 2005 NEW FULL
Saskatchewan Health Drug Plan and Extended Benefits Branch July 2005 Bulletin #103 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE BULLETIN 55th EDITION All listings are effective July 1, 2005 NEW FULL
NBPDP Formulary Update
 Bulletin #878 December 19, 2013 NBPDP Formulary Update This update to the New Brunswick Prescription Drug Program (NBPDP) Formulary is effective December 19, 2013. Included in this bulletin: Regular Benefit
Bulletin #878 December 19, 2013 NBPDP Formulary Update This update to the New Brunswick Prescription Drug Program (NBPDP) Formulary is effective December 19, 2013. Included in this bulletin: Regular Benefit
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Promacta) Reference Number: CP.PHAR.180 Effective Date: 03.01.16 Last Review Date: 02.18 Line of Business: Commercial, Health Insurance Marketplace, Medicaid Revision Log See Important
Clinical Policy: (Promacta) Reference Number: CP.PHAR.180 Effective Date: 03.01.16 Last Review Date: 02.18 Line of Business: Commercial, Health Insurance Marketplace, Medicaid Revision Log See Important
Treatment of Retinal Vein Occlusion (RVO)
 Manchester Royal Eye Hospital Medical Retina Services Information for Patients Treatment of Retinal Vein Occlusion (RVO) What is a Retinal Vein Occlusion (RVO)? The retina is the light sensitive layer
Manchester Royal Eye Hospital Medical Retina Services Information for Patients Treatment of Retinal Vein Occlusion (RVO) What is a Retinal Vein Occlusion (RVO)? The retina is the light sensitive layer
The legally binding text is the original French version
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 12 September 2007 INFRACYANINE 25 mg/10 ml, powder and solvent for solution for injection 25 mg vial with one 10 ml
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 12 September 2007 INFRACYANINE 25 mg/10 ml, powder and solvent for solution for injection 25 mg vial with one 10 ml
Photodynamic Therapy (PDT) for agerelated
 Page 1 of 5 Photodynamic Therapy (PDT) for agerelated eye conditions Introduction This leaflet gives you information about Photodynamic Therapy (PDT) for the age--related eye conditions macular degeneration
Page 1 of 5 Photodynamic Therapy (PDT) for agerelated eye conditions Introduction This leaflet gives you information about Photodynamic Therapy (PDT) for the age--related eye conditions macular degeneration
SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary
 April 1, 2017 Bulletin #165 ISSN 1923-0761 SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary Related Information for Prescribers: Only prescribers who have completed
April 1, 2017 Bulletin #165 ISSN 1923-0761 SASKATCHEWAN FORMULARY BULLETIN Update to the 62nd Edition of the Saskatchewan Formulary Related Information for Prescribers: Only prescribers who have completed
EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY
 EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY Diwakar chaudhary *1, 2, Hu shuqiong, Long Yuan and Xiong kun 1 Yangtze University, 1 Nanhuan Road
EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY Diwakar chaudhary *1, 2, Hu shuqiong, Long Yuan and Xiong kun 1 Yangtze University, 1 Nanhuan Road
Avastin. Avastin (bevacizumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.04 Subject: Avastin Page: 1 of 9 Last Review Date: September 15, 2017 Avastin Description Avastin
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.04 Subject: Avastin Page: 1 of 9 Last Review Date: September 15, 2017 Avastin Description Avastin
Appendix to Notification Letter for rituximab and eltrombopag dated 20 February 2014
 Appendix to Notification Letter for rituximab and eltrombopag dated 20 February 2014 The notification letter which contains details of the decision to widen the restriction criteria for rituximab and eltrombopag
Appendix to Notification Letter for rituximab and eltrombopag dated 20 February 2014 The notification letter which contains details of the decision to widen the restriction criteria for rituximab and eltrombopag
Ranibizumab and pegaptanib for the treatment of agerelated macular degeneration
 Ranibizumab and pegaptanib for the treatment of agerelated Issued: August 2008 last modified: May 2012 guidance.nice.org.uk/ta155 NHS Evidence has accredited the process used by the Centre for Health Technology
Ranibizumab and pegaptanib for the treatment of agerelated Issued: August 2008 last modified: May 2012 guidance.nice.org.uk/ta155 NHS Evidence has accredited the process used by the Centre for Health Technology
