Suxamethonium Chloride Injection BP PRODUCT INFORMATION
|
|
|
- Gyles Barton
- 5 years ago
- Views:
Transcription
1 Suxamethonium Chloride Injection BP Product Information 1 (8) Suxamethonium Chloride Injection BP PRODUCT INFORMATION NAME OF DRUG The Australian Approved Name is suxamethonium chloride. The CAS number for suxamethonium chloride dihydrate is The chemical structure is: o (H 3 C) 3 + N o o N + (CH 3 ) 3 2 Cl - o DESCRIPTION The chemical name for suxamethonium chloride is 2, 2 -succinyldioxybis(ethyltrimethylammonium) dichloride dihydrate. It is a white or almost white, crystalline powder. Suxamethonium Chloride Injection is a sterile solution of suxamethonium chloride suitable for IV injection. Each ml of solution contains 50 mg suxamethonium chloride in Water for Injections BP. The ph of the solution is The presentation of Suxamethonium Chloride Injection is intended for single use only. Any solution remaining from an opened container should be discarded. PHARMACOLOGY Suxamethonium is an ultra short-acting depolarising-type neuromuscular blocking agent. Suxamethonium combines with the cholinergic receptors of the motor end plate to produce depolarisation. Neuromuscular transmission is inhibited so long as an adequate concentration of suxamethonium remains at the receptor site. Suxamethonium has no direct action on smooth muscle structures, including the uterus. Suxamethonium may produce slowing of heart rate via vagal stimulation. When suxamethonium is administered over a prolonged period the characteristics of the neuromuscular block may change from the characteristic depolarising type to one resembling a nondepolarising block.
2 Suxamethonium Chloride Injection BP Product Information 2 (8) Pharmacokinetics Absorption: Suxamethonium has a rapid onset and a short duration of action. Following intravenous (IV) administration of a single therapeutic dose in healthy adults, complete muscle relaxation occurs within 1/2 to 1 minute, persists for about 2-3 minutes, and gradually dissipates within 10 minutes. Following intramuscular (IM) administration the onset of action occurs in about 2-3 minutes, with a duration ranging from minutes. The duration of action is prolonged in patients with low plasma pseudocholinesterase concentration. Distribution: Suxamethonium crosses the placenta, generally in small amounts. Elimination: Plasma pseudocholinesterases hydrolyse suxamethonium to succinylmonocholine (relatively inactive) and choline. Approximately 10% of drug is excreted unchanged in the urine. Patients with impaired renal function may occasionally experience prolonged apnoea due to accumulation of succinylmonocholine. INDICATIONS For the production of skeletal muscle relaxation in anaesthesia. Suited for procedures requiring only brief relaxation such as endotracheal intubation, endoscopic examinations, orthopaedic manipulations, short surgical procedures and electro-convulsive therapy. CONTRAINDICATIONS Patients with a personal or familial history of malignant hyperthermia, genetically determined disorders of pseudocholinesterase, myopathies associated with elevated creatinine phosphokinase (CPK) values, Duchenne's muscular dystrophy, known hypersensitivity to suxamethonium, severe hyperkalaemia, acute narrow-angle glaucoma, and the presence of penetrating eye injuries (suxamethonium may cause a slight, transient increase in intraocular pressure). It is also contraindicated in patients after the acute phase of injury following major burns, or multiple trauma, *renal impairment with a raised plasma-potassium concentration, or in those with extensive muscle degeneration such as recent paraplegia and severe long-lasting sepsis because such patients may become severely hyperkalaemic when given suxamethonium, resulting in cardiac arrhythmia or arrest.
3 Suxamethonium Chloride Injection BP Product Information 3 (8) PRECAUTIONS Suxamethonium should only be administered under strict supervision of an anaesthetist familiar with its actions, characteristics and hazards who is skilled in the management of artificial respiration and only when facilities are instantly available for endotracheal intubation and for providing adequate ventilation of the patient, including the administration of oxygen under positive pressure. Be prepared to assist or control respiration. Suxamethonium has no effect on consciousness, pain threshold or cerebration. It should therefore only be used with adequate anaesthesia. Malignant Hyperthermia The abrupt onset of malignant hyperthermia, a very rare hypermetabolic process of skeletal muscle, may be triggered by suxamethonium. Early premonitory signs include muscle rigidity, tachycardia, tachypnoea unresponsive to increased depth of anaesthesia, evidence of increased oxygen requirement and carbon dioxide production, rising temperature and metabolic acidosis. On evidence of these symptoms the anaesthetic and suxamethonium should be discontinued and supportive measures implemented including administration of oxygen, sodium bicarbonate, lowering of temperature, restoration of fluids and electrolyte balance, maintenance of adequate urinary output and administration of IV dantrolene according to a standard protocol. Hyperkalaemia Administration of suxamethonium causes an immediate rise in serum potassium. This rise is normally small but may be prolonged and exaggerated in patients taking beta-blockers. Great caution should also be observed in patients with pre-existing hyperkalaemia or electrolyte imbalance, uraemia, hemiplegia, paraplegia, extensive burns, massive trauma, diffuse intracranial lesions (head injury, encephalitis, ruptured cerebral aneurysm), tetanus, acute anterior horn cell disease, extensive denervation of skeletal muscle due to disease or injury of the CNS, or who have degenerative neuromuscular disease and in severe long-lasting sepsis. Such patients may become severely hyperkalaemic when given suxamethonium, resulting in cardiac arrhythmia or arrest (see CONTRAINDICATIONS). With burns or trauma the period of greatest risk is from about days after the injury, but may be prolonged further if there is delayed healing or persistent infection. These patients may still react abnormally to suxamethonium 2 years after the injury. In neuromuscular disease the greatest risk period is usually from 3 weeks to 6 months after onset, but severe hyperkalaemia may occur after 24 to 48 hours or later than 6 months. Patients with severe sepsis for more than a week should be considered at risk of hyperkalaemia and suxamethonium should not be given until the infection has cleared.
4 Suxamethonium Chloride Injection BP Product Information 4 (8) Hyperkalaemia Rhabdomyolysis There is a risk of cardiac arrest from hyperkalaemia due to rhabdomyolysis, particularly in male patients with muscular dystrophy. Low plasma pseudocholinesterase Recovery from suxamethonium may occasionally be delayed possibly due to a low serum pseudocholinesterase level, this may occur in patients suffering from severe liver disease, cancer, malnutrition, severe dehydration, collagen diseases, severe anaemia, myxoedema, burns, pregnancy and the puerperium, severe infections, myocardial infarction, renal impairment and abnormal body temperature. Also exposure to neurotoxic insecticides or weed killers, anti-malarial or anti-cancer drugs, monoamine oxidase (MAO) inhibitors, contraceptive pill, pancuronium, chlorpromazine, ecothiopate or neostigmine may result in low levels of pseudocholinesterase. Suxamethonium should be administered with extreme caution and in reduced doses in such patients. If low pseudocholinesterase concentration is suspected slow administration of a small test dose of suxamethonium (5-10 mg as a 0.1% solution) should be considered. Antidysrhythmic drugs Suxamethonium should be administered with great caution in patients receiving quinidine and those who have been digitalised or who may have digitalis toxicity. In these circumstances the rise in serum potassium due to suxamethonium may possibly cause arrhythmias. Delayed recovery When recovery from suxamethonium is delayed, assisted respiration sufficient for full oxygenation, yet avoiding excessive elimination of carbon dioxide, should be maintained until paralysis ceases. This should be combined with light narcosis, e.g. nitrous oxide/oxygen mixture. Neostigmine should not be given when prolonged apnoea follows a single dose of suxamethonium. Neostigmine and other anticholinesterase drugs may have the effect of intensifying the depolarisation block caused by suxamethonium. Nondepolarising blockade If suxamethonium is given repeatedly or over a prolonged period the depolarising block may change to one with characteristics of a nondepolarising block. This may be associated with prolonged respiratory depression and apnoea. Following a positive diagnosis of a nondepolarising blockade the administration of neostigmine preceded by atropine may be considered.
5 Suxamethonium Chloride Injection BP Product Information 5 (8) Debilitated Patients Use with caution in patients who are hypoxic or those who have cardiovascular, hepatic, pulmonary, metabolic or renal disorders or myasthenia gravis. The action of suxamethonium may be altered in these patients. Its use is not advisable in patients with phaeochromocytoma. As suxamethonium produces muscle contractions before relaxation it should be used with caution in patients with bone fractures. Suxamethonium should be avoided in patients with myotonias, as response is unpredictable. Use in eye surgery Suxamethonium causes a slight transient increase in intraocular pressure immediately after injection and during the fasciculation phase. It should therefore be used cautiously if at all during intraocular surgery and in patients with glaucoma. Use in pregnancy Category A Safety of the use of suxamethonium in pregnancy has not been established with respect to effects on fetal development. Therefore suxamethonium should not be administered to pregnant women unless the potential benefit outweighs the possible hazards. Plasma pseudocholinesterase levels are decreased in pregnancy and several days postpartum by approximately 25%, therefore a high proportion of these patients may be expected to show prolonged apnoea. Suxamethonium crosses the placenta, but generally only in small amounts. Residual neuromuscular blockade may occasionally occur in the neonate after repeated high doses of suxamethonium to the mother during delivery by caesarean section. Interactions with other drugs Co-administration of inhaled anaesthetics (cyclopane, diethylether, halothane and nitrous oxide) may increase the incidence of dysrhythmias (especially bradycardia), apnoea and the occurrence of malignant hyperthermia in susceptible persons. Inhaled anaesthetics have little effect on the usual depolarising neuromuscular blockade of suxamethonium but may enhance the Phase II block (nondepolarising) that may be produced by repeated dosage of suxamethonium. Severe bradycardia and asystole have occurred when suxamethonium is used in anaesthetic regimens with propofol and opioids such as fentanyl. Drugs which may enhance or prolong the effects of suxamethonium include lignocaine, procaine, oxytocin, oral contraceptives, some non-penicillin antibiotics, (streptomycin, neomycin, kanamycin, capreomycin, tobramycin, framycetin, amikacin, gentamicin, colistin and polymyxins), tacrine, beta-adrenergic blockers, trimethaphan, phenelzine, aprotinin, quinidine, promazine, lithium carbonate, phenytoin, carbamazepine, magnesium salts, quinine, chloroquine, cimetidine, terbutaline sulfate, high dose corticosteroids and cytostatic agents such as cyclophosphamide, thiotepa and azathioprine. Diazepam may reduce the duration of neuromuscular blockade produced by suxamethonium.
6 Suxamethonium Chloride Injection BP Product Information 6 (8) Amphotericin B and thiazide diuretics may increase the effects of suxamethonium secondary to induced electrolyte imbalance. Patients with hypokalemia or hypocalcaemia require reduced doses of suxamethonium. Inhibitors of plasma cholinesterases such as neostigmine, pyridostigmine bromide, rivastigmine, donepezil, metoclopramide, physostigmine and phospholine iodide can considerably prolong the depolarising action of suxamethonium. It is recommended that long-acting anticholinesterase inhibitor (ecothiopate) eye drops should be discontinued several months prior to administration of suxamethonium. Administration of suxamethonium prior to or with a nondepolarising muscle relaxant e.g. pancuronium, mivacurium can alter the intensity and/or duration of neuromuscular blockade. Simultaneous administration of suxamethonium and atracurium significantly reduces the duration of suxamethonium. Concomitant digoxin or verapamil, and suxamethonium therapy has been reported to result in cardiac arrhythmias. ADVERSE REACTIONS The following adverse reactions have been reported following administration of suxamethonium: Neuromuscular: post-operative muscle pain, muscle fasciculation, rhabdomyolysis, myoglobinuria, myoglobinaemia, elevated creatine phosphokinase, hypertonia, trismus. Cardiovascular: bradycardia, tachycardia, arrhythmias, cardiac arrest, hypertension, hypotension, tachyphylaxis, ventricular Respiratory: fibrillation as a result of hyperkalaemia. apnoea, prolonged respiratory failure, bronchospasm, increased bronchial secretions, pulmonary oedema in infants. Endocrine, metabolic: malignant hyperthermia, porphyria, hyperkalaemia, excessive salivation. Gastrointestinal: increased intragastric pressure, increased bowel movements, increased gastric secretions, *possible aspiration. Special senses: Other: increased intraocular pressure. Rise in intracranial pressure, renal failure, precipitation or exacerbation of myasthenia gravis Hypersensitivity reactions including circulatory collapse, flushing, rash, urticaria, bronchospasm and shock, which may lead to death.
7 Suxamethonium Chloride Injection BP Product Information 7 (8) DOSAGE AND ADMINISTRATION Dosage is individualised and its administration should be determined after careful assessment of the patient. The dose of suxamethonium is dependent on bodyweight, the degree of muscle relaxation required and the response of individual patients. Suxamethonium causes paralysis of the respiratory muscles, therefore after administration, respiration must be controlled. It should not be administered to a conscious patient. Suxamethonium should not be mixed with any neuromuscular blocking agent, nor with general anaesthetics such as short acting barbiturates, nor any other therapeutic agent in the same syringe. Suxamethonium Chloride Injection contains no antimicrobial agent. It should be used only once and any residue discarded. An initial test dose of 0.1 mg/kg may be given intravenously to determine the patients response. Adult: For short procedures, such as endotracheal intubation the usual adult dose is 0.6 mg/kg (range mg/kg) administered IV over 10 to 30 seconds. This dose produces muscle relaxation in about 60 seconds and has a duration of approximately 4 to 6 minutes. Larger doses produce more prolonged muscle relaxation. For more prolonged surgical procedures in an adult, suxamethonium is commonly given by IV infusion at a rate of mg/minute. When given by intravenous infusion suxamethonium should be diluted to 0.1 to 0.2% (1-2 mg/ml) in 5% dextrose solution or sterile isotonic saline. Children: Neonates and premature infants may be relatively resistant to suxamethonium. The usual paediatric IV dose is 1 to 2 mg/kg. If necessary, additional doses maybe administered in accordance with patients response. Continuous IV infusions of suxamethonium are considered unsafe in neonates and children because of the risk of inducing malignant hyperthermia. Intravenous bolus in children may result in profound bradycardia or on occasion asystole. This tends to be more common after a second dose. Pre-treatment with atropine can reduce the risk of bradycardia. When a suitable vein is inaccessible, suxamethonium may occasionally be given by intramuscular injection. A suggested i.m. dose for adults and children may be up to 2.5 mg/kg but the total dose should not exceed 150 mg. Diluted solutions of suxamethonium must be used within 24 hours of preparation. Discard unused solutions.
8 Suxamethonium Chloride Injection BP Product Information 8 (8) OVERDOSAGE The most serious effects of overdosage are apnoea and prolonged muscle paralysis. It is essential to maintain the airway and adequate ventilation until spontaneous respiration is fully restored. The use of neostigmine to reverse a nondepolarising block is a clinical decision which depends on the subject, the experience, and the judgment of the clinician. If neostigmine is used, its administration should be accompanied by an appropriate dose of atropine. PRESENTATION PolyampDuoFit ampoules 100 mg/2 ml, in packs of 50 STORAGE Store between 2-8 C. REFRIGERATE - DO NOT FREEZE. NAME AND ADDRESS OF THE SPONSOR AstraZeneca Pty Ltd ABN Alma Road, North Ryde NSW 2113 Australia Polyamp DuoFit is a trade mark of the AstraZeneca group of companies Date of TGA approval letter 4 September 1996 Date of Safety Related Notification: 20 Oct 2011
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET 1. PRODUCT NAME SUXAMETHONIUM CHLORIDE INJECTION B.P. 50 mg/ml 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each 2 ml of solution contains 100 mg of suxamethonium chloride BP For
NEW ZEALAND DATA SHEET 1. PRODUCT NAME SUXAMETHONIUM CHLORIDE INJECTION B.P. 50 mg/ml 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each 2 ml of solution contains 100 mg of suxamethonium chloride BP For
NEW ZEALAND DATA SHEET. Injection 2 mg/ml: a clear, colourless, particle-free solution containing 2 mg/ml pancuronium bromide.
 NEW ZEALAND DATA SHEET NAME OF MEDICINE Pancuronium Bromide B.P. Injection 2 mg/ml PRESENTATION Injection 2 mg/ml: a clear, colourless, particle-free solution containing 2 mg/ml pancuronium bromide. INDICATIONS
NEW ZEALAND DATA SHEET NAME OF MEDICINE Pancuronium Bromide B.P. Injection 2 mg/ml PRESENTATION Injection 2 mg/ml: a clear, colourless, particle-free solution containing 2 mg/ml pancuronium bromide. INDICATIONS
NEW ZEALAND DATA SHEET. Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution containing 2.5 mg/ml Neostigmine methylsulphate.
 NEW ZEALAND DATA SHEET NAME OF MEDICINE NEOSTIGMINE METHYLSULPHATE INJECTION B.P. Solution for injection 2.5 mg/ml. PRESENTATIONS Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution
NEW ZEALAND DATA SHEET NAME OF MEDICINE NEOSTIGMINE METHYLSULPHATE INJECTION B.P. Solution for injection 2.5 mg/ml. PRESENTATIONS Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution
Quelicin TM SUCCINYLCHOLINE CHLORIDE INJECTION, USP
 Quelicin TM SUCCINYLCHOLINE CHLORIDE INJECTION, USP A short-acting depolarizing skeletal muscle relaxant. Fliptop Vial Rx only WARNING RISK OF CARDIAC ARREST FROM HYPERKALEMIC RHABDOMYOLYSIS There have
Quelicin TM SUCCINYLCHOLINE CHLORIDE INJECTION, USP A short-acting depolarizing skeletal muscle relaxant. Fliptop Vial Rx only WARNING RISK OF CARDIAC ARREST FROM HYPERKALEMIC RHABDOMYOLYSIS There have
TRACRIUM GlaxoSmithKline
 TRACRIUM GlaxoSmithKline Atracurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: A sterile solution containing 10 mg atracurium besylate per ml, without an antimicrobial preservative, supplied in
TRACRIUM GlaxoSmithKline Atracurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: A sterile solution containing 10 mg atracurium besylate per ml, without an antimicrobial preservative, supplied in
Core Safety Profile. Pharmaceutical form(s)/strength: Solution for Injection CZ/H/PSUR/0005/002 Date of FAR:
 Core Safety Profile Active substance: Rocuronium bromide Pharmaceutical form(s)/strength: Solution for Injection P-RMS: CZ/H/PSUR/0005/002 Date of FAR: 31.05.2012 4.3 Contraindications Hypersensitivity
Core Safety Profile Active substance: Rocuronium bromide Pharmaceutical form(s)/strength: Solution for Injection P-RMS: CZ/H/PSUR/0005/002 Date of FAR: 31.05.2012 4.3 Contraindications Hypersensitivity
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 AUSTRALIAN PRODUCT INFORMATION NEOSTIGMINE INJECTION BP (NEOSTIGMINE METHYLSULFATE) 1 NAME OF THE MEDICINE Neostigmine methylsulfate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml sterile solution of
AUSTRALIAN PRODUCT INFORMATION NEOSTIGMINE INJECTION BP (NEOSTIGMINE METHYLSULFATE) 1 NAME OF THE MEDICINE Neostigmine methylsulfate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml sterile solution of
50% Concentrated Injection
 NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION
 DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
PRODUCT INFORMATION. TRACRIUM INJECTION atracurium besylate DESCRIPTION:
 PRODUCT INFORMATION TRACRIUM INJECTION (atracurium besylate) NAME OF THE DRUG: atracurium besylate DESCRIPTION: TRACRIUM (atracurium besylate) is an intermediate-duration, non-depolarising, skeletal muscle
PRODUCT INFORMATION TRACRIUM INJECTION (atracurium besylate) NAME OF THE DRUG: atracurium besylate DESCRIPTION: TRACRIUM (atracurium besylate) is an intermediate-duration, non-depolarising, skeletal muscle
MUSCLE RELAXANTS. Mr. D.Raju, M.pharm, Lecturer
 MUSCLE RELAXANTS Mr. D.Raju, M.pharm, Lecturer Muscle Relaxants are classified as: I)Peripherally acting A.Neuromuscular blocking agents:- 1) Depolarizing muscle relaxants. 2) Non-depolarizing muscle relaxants
MUSCLE RELAXANTS Mr. D.Raju, M.pharm, Lecturer Muscle Relaxants are classified as: I)Peripherally acting A.Neuromuscular blocking agents:- 1) Depolarizing muscle relaxants. 2) Non-depolarizing muscle relaxants
NEUROMUSCULAR BLOCKING AGENTS
 NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
Core Safety Profile. Pharmaceutical form(s)/strength: 5mg/ml and 25 mg/ml, Solution for injection, IM/IV FI/H/PSUR/0010/002 Date of FAR:
 Core Safety Profile Active substance: Esketamine Pharmaceutical form(s)/strength: 5mg/ml and 25 mg/ml, Solution for injection, IM/IV P-RMS: FI/H/PSUR/0010/002 Date of FAR: 29.05.2012 4.3 Contraindications
Core Safety Profile Active substance: Esketamine Pharmaceutical form(s)/strength: 5mg/ml and 25 mg/ml, Solution for injection, IM/IV P-RMS: FI/H/PSUR/0010/002 Date of FAR: 29.05.2012 4.3 Contraindications
Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml
 New Zealand Datasheet Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml Solution for injection Glycopyrronium bromide 0.5 mg/ml & Neostigmine metilsulfate 2.5 mg/ml Presentation is
New Zealand Datasheet Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml Solution for injection Glycopyrronium bromide 0.5 mg/ml & Neostigmine metilsulfate 2.5 mg/ml Presentation is
MIVACRON GlaxoSmithKline
 MIVACRON GlaxoSmithKline Mivacurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: Sterile solution containing 2 mg mivacurium per ml as mivacurium chloride, without an antimicrobial preservative,
MIVACRON GlaxoSmithKline Mivacurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: Sterile solution containing 2 mg mivacurium per ml as mivacurium chloride, without an antimicrobial preservative,
A Nondepolarizing Neuromuscular Blocking (NMB) Agent
 DOSING GUIDE A Nondepolarizing Neuromuscular Blocking (NMB) Agent Easy to remember dosing for the 0.20 mg/kg adult intubating doses of NIMBEX 1 *: For every 10 kg, give 1 ml of NIMBEX (2 mg/ml concentration)
DOSING GUIDE A Nondepolarizing Neuromuscular Blocking (NMB) Agent Easy to remember dosing for the 0.20 mg/kg adult intubating doses of NIMBEX 1 *: For every 10 kg, give 1 ml of NIMBEX (2 mg/ml concentration)
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory TRACRIUM. Atracurium Besylate Injection IP
 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory TRACRIUM Atracurium Besylate Injection IP QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains Atracurium Besylate
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory TRACRIUM Atracurium Besylate Injection IP QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains Atracurium Besylate
Drugs used in obstetrics
 Drugs used in obstetrics Drugs used in obstetrics Drugs may be used to modify uterine contractions. These include oxytocic drugs used to stimulate uterine contractions both in induction of labour and to
Drugs used in obstetrics Drugs used in obstetrics Drugs may be used to modify uterine contractions. These include oxytocic drugs used to stimulate uterine contractions both in induction of labour and to
1. What Anectine is and what it is used for
 Patient Information Leaflet: Information for the user Anectine 50 mg/ml Solution for Injection or Infusion (suxamethonium chloride) Read all of this leaflet carefully before you start using this medicine
Patient Information Leaflet: Information for the user Anectine 50 mg/ml Solution for Injection or Infusion (suxamethonium chloride) Read all of this leaflet carefully before you start using this medicine
PRODUCT INFORMATION RESONIUM A. Na m
 PRODUCT INFORMATION RESONIUM A NAME OF THE MEDICINE Non-proprietary Name Sodium polystyrene sulfonate Chemical Structure CH - 2 CH SO 3 Na + n CAS Number 28210-41-5 [9003-59-2] CH 2 CH SO - 3 m DESCRIPTION
PRODUCT INFORMATION RESONIUM A NAME OF THE MEDICINE Non-proprietary Name Sodium polystyrene sulfonate Chemical Structure CH - 2 CH SO 3 Na + n CAS Number 28210-41-5 [9003-59-2] CH 2 CH SO - 3 m DESCRIPTION
PRESCRIBING INFORMATION QUELICIN. Neuromuscular Blocking Agent
 PRESCRIBING INFORMATION QUELICIN Succinylcholine Chloride Injection USP Neuromuscular Blocking Agent Pfizer Canada Inc. 17300 Trans-Canada Highway Kirkland, Québec H9J 2M5 DATE OF REVISION: June 23, 2017
PRESCRIBING INFORMATION QUELICIN Succinylcholine Chloride Injection USP Neuromuscular Blocking Agent Pfizer Canada Inc. 17300 Trans-Canada Highway Kirkland, Québec H9J 2M5 DATE OF REVISION: June 23, 2017
New Zealand Data Sheet COLISTIN-LINK
 New Zealand Data Sheet COLISTIN-LINK Colistimethate sodium equivalent to colistin 150mg powder for injection For intramuscular and intravenous use. Description Colistimethate sodium for injection, USP
New Zealand Data Sheet COLISTIN-LINK Colistimethate sodium equivalent to colistin 150mg powder for injection For intramuscular and intravenous use. Description Colistimethate sodium for injection, USP
NERVOUS SYSTEM NERVOUS SYSTEM. Somatic nervous system. Brain Spinal Cord Autonomic nervous system. Sympathetic nervous system
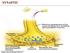 SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
BRICANYL INJECTION. terbutaline sulfate PRODUCT INFORMATION
 BRICANYL INJECTION terbutaline sulfate PRODUCT INFORMATION NAME OF THE MEDICINE Terbutaline sulfate, 2-(tert-butylamino)-1-(3,5-dihydroxyphenyl) ethanol sulfate, a sympathomimetic bronchodilator with a
BRICANYL INJECTION terbutaline sulfate PRODUCT INFORMATION NAME OF THE MEDICINE Terbutaline sulfate, 2-(tert-butylamino)-1-(3,5-dihydroxyphenyl) ethanol sulfate, a sympathomimetic bronchodilator with a
PRODUCT INFORMATION. Colistin Link. Colistin 150 mg/2 ml (as colistimethate sodium) powder for injection vial
 PRODUCT INFORMATION Colistin Link Colistin 150 mg/2 ml (as colistimethate sodium) powder for injection vial For Intramuscular and Intravenous use. NAME OF THE MEDICINE Colistimethate sodium for injection,
PRODUCT INFORMATION Colistin Link Colistin 150 mg/2 ml (as colistimethate sodium) powder for injection vial For Intramuscular and Intravenous use. NAME OF THE MEDICINE Colistimethate sodium for injection,
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT ACURMIL 25 mg/2.5 ml solution for intravenous injection ACURMIL 50 mg/5 ml solution for intravenous injection 2. QUALITATIVE AND QUANTITATIVE
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT ACURMIL 25 mg/2.5 ml solution for intravenous injection ACURMIL 50 mg/5 ml solution for intravenous injection 2. QUALITATIVE AND QUANTITATIVE
PROSTIGMIN (neostigmine bromide)
 PROSTIGMIN (neostigmine bromide) TABLETS DESCRIPTION: Prostigmin (neostigmine bromide), an anticholinesterase agent, is available for oral administration in 15 mg tablets. Each tablet also contains gelatin,
PROSTIGMIN (neostigmine bromide) TABLETS DESCRIPTION: Prostigmin (neostigmine bromide), an anticholinesterase agent, is available for oral administration in 15 mg tablets. Each tablet also contains gelatin,
Ganglion blocking agents
 Ganglion blocking agents -out of date -Specifically act on the nicotinic receptors of both parasymphatetic and sympathetic ganglia - no selectivity toward PG or SG -These drugs are non-depolarizing, competitive
Ganglion blocking agents -out of date -Specifically act on the nicotinic receptors of both parasymphatetic and sympathetic ganglia - no selectivity toward PG or SG -These drugs are non-depolarizing, competitive
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Citanest with Octapressin Dental 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains Prilocaine Hydrochloride 30 mg (54 mg/1.8
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Citanest with Octapressin Dental 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains Prilocaine Hydrochloride 30 mg (54 mg/1.8
PRODUCT INFORMATION CISATRACURIUM PFIZER NAME OF THE MEDICINE DESCRIPTION PHARMACOLOGY. Cisatracurium besylate
 PRODUCT INFORMATION CISATRACURIUM PFIZER Cisatracurium besylate NAME OF THE MEDICINE The chemical name of cisatracurium besylate is (1R,1 R,2R,2 R,)-2,2 -(3,11-dioxo-4,10- dioxatridecamethylene) bis (1,2,3,4-tetrahydro-6,7-dimethoxy-2-methyl-1-
PRODUCT INFORMATION CISATRACURIUM PFIZER Cisatracurium besylate NAME OF THE MEDICINE The chemical name of cisatracurium besylate is (1R,1 R,2R,2 R,)-2,2 -(3,11-dioxo-4,10- dioxatridecamethylene) bis (1,2,3,4-tetrahydro-6,7-dimethoxy-2-methyl-1-
P-RMS: NO/H/PSUR/0009/001
 Core Safety Profile Active substance: Propofol Pharmaceutical form(s)/strength: Emulsion for injection, infusion, 10mg/ml Emulsion for infusion, 20mg/ml P-RMS: NO/H/PSUR/0009/001 Date of FAR: 30.06.2011
Core Safety Profile Active substance: Propofol Pharmaceutical form(s)/strength: Emulsion for injection, infusion, 10mg/ml Emulsion for infusion, 20mg/ml P-RMS: NO/H/PSUR/0009/001 Date of FAR: 30.06.2011
Neuromuscular Junction
 Muscle Relaxants Neuromuscular Junction Cholinergic antagonists Neuromuscular-blocking agents (mostly nicotinic antagonists): interfere with transmission of efferent impulses to skeletal muscles. These
Muscle Relaxants Neuromuscular Junction Cholinergic antagonists Neuromuscular-blocking agents (mostly nicotinic antagonists): interfere with transmission of efferent impulses to skeletal muscles. These
VENTOLIN RESPIRATOR SOLUTION
 VENTOLIN RESPIRATOR SOLUTION Salbutamol QUALITATIVE AND QUANTITATIVE COMPOSITION VENTOLIN Respirator Solution contains 5mg salbutamol, as sulphate, per ml of solution and is supplied in 10 ml bottles.
VENTOLIN RESPIRATOR SOLUTION Salbutamol QUALITATIVE AND QUANTITATIVE COMPOSITION VENTOLIN Respirator Solution contains 5mg salbutamol, as sulphate, per ml of solution and is supplied in 10 ml bottles.
Benztropine and trihexyphenidyl: Centrally acting antimuscarinic agents used for treatment of Parkinson disease & extrapyramidal symptoms.
 Scopolamine: Tertiary amine plant alkaloid. Produces peripheral effects similar to those of atropine. Unlike atropine, scopolamine has greater action on the CNS (observed at therapeutic doses). It has
Scopolamine: Tertiary amine plant alkaloid. Produces peripheral effects similar to those of atropine. Unlike atropine, scopolamine has greater action on the CNS (observed at therapeutic doses). It has
(PP XI) Dr. Samir Matloob
 DRUGS ACTING ON THE CHOLINERGIC SYSTEM AND THE NEUROMUSCULAR BLOCKING DRUGS IV (NICOTINIC ANTAGONISTS) (PP XI) Dr. Samir Matloob Dept. of Pharmacology Baghdad College of Medicine Drugs acting on the cholinergic
DRUGS ACTING ON THE CHOLINERGIC SYSTEM AND THE NEUROMUSCULAR BLOCKING DRUGS IV (NICOTINIC ANTAGONISTS) (PP XI) Dr. Samir Matloob Dept. of Pharmacology Baghdad College of Medicine Drugs acting on the cholinergic
Shaded areas=not MARKETED 24/2/09
 PACKAGE INSERT SCHEDULING STATUS Schedule 6 PROPRIETARY NAME AND DOSAGE FORM RAPIFEN 2 ml IV injection RAPIFEN 10 ml IV injection COMPOSITION Each ml contains alfentanil hydrochloride 0,544 mg (equivalent
PACKAGE INSERT SCHEDULING STATUS Schedule 6 PROPRIETARY NAME AND DOSAGE FORM RAPIFEN 2 ml IV injection RAPIFEN 10 ml IV injection COMPOSITION Each ml contains alfentanil hydrochloride 0,544 mg (equivalent
Please refer to the Summary of Product Characteristics (SPC) for further details on this product.
 [Aspen Logo] Information for the Physician Nimbex cisatracurium Please refer to the Summary of Product Characteristics (SPC) for further details on this product. Trade name of the medicinal product Nimbex
[Aspen Logo] Information for the Physician Nimbex cisatracurium Please refer to the Summary of Product Characteristics (SPC) for further details on this product. Trade name of the medicinal product Nimbex
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET VECURE 1. Product Name VECURE 10 mg, powder for injection. 2. Qualitative and Quantitative Composition VECURE 10 mg, 1 vial contains 10 mg vecuronium bromide. When reconstituted
NEW ZEALAND DATA SHEET VECURE 1. Product Name VECURE 10 mg, powder for injection. 2. Qualitative and Quantitative Composition VECURE 10 mg, 1 vial contains 10 mg vecuronium bromide. When reconstituted
DBL NALOXONE HYDROCHLORIDE INJECTION USP
 Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
PANCURONIUM BROMIDE INJECTION B.P. Pancuronium Bromide
 PANCURONIUM BROMIDE INJECTION B.P. Pancuronium Bromide Consumer Medicine Information What is in this leaflet This leaflet answers some of the common questions people ask about Pancuronium Bromide Injection.
PANCURONIUM BROMIDE INJECTION B.P. Pancuronium Bromide Consumer Medicine Information What is in this leaflet This leaflet answers some of the common questions people ask about Pancuronium Bromide Injection.
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP
 PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
Final Core Safety Profile for propofol 10 mg/ml (1%) and 20 mg/ml (2%) emulsion for injection or infusion
 CSP Drug Substance Propofol Date September 04 2014 Final Core Safety Profile for propofol 10 mg/ml (1%) and 20 mg/ml (2%) emulsion for injection or infusion CORE SAFETY PROFILE 4.3 Contraindications Propofol
CSP Drug Substance Propofol Date September 04 2014 Final Core Safety Profile for propofol 10 mg/ml (1%) and 20 mg/ml (2%) emulsion for injection or infusion CORE SAFETY PROFILE 4.3 Contraindications Propofol
Active ingredients: Metoclopramide Hydrochloride mg Equivalent to metoclopramide hydrochloride anhydrous mg
 Name Primperan 10 mg / 2 ml Metoclopramide hydrochloride anhydrous Solution for I.M. or I.V. injection (Ampoules) Composition Each 2 ml ampoule contains: Active ingredients: Metoclopramide Hydrochloride.
Name Primperan 10 mg / 2 ml Metoclopramide hydrochloride anhydrous Solution for I.M. or I.V. injection (Ampoules) Composition Each 2 ml ampoule contains: Active ingredients: Metoclopramide Hydrochloride.
Calcium Chloride 10% Injection 1g in 10 ml (as calcium chloride dihydrate containing 6.8 mmol calcium ions in 10 ml)
 10% Injection 1g in 10 ml (as calcium chloride dihydrate containing 6.8 mmol calcium ions in 10 ml) NAME OF THE MEDICINE Name: Calcium chloride dihydrate Molecular formula: CaCl2.2H20 Molecular weight:
10% Injection 1g in 10 ml (as calcium chloride dihydrate containing 6.8 mmol calcium ions in 10 ml) NAME OF THE MEDICINE Name: Calcium chloride dihydrate Molecular formula: CaCl2.2H20 Molecular weight:
RMP section VI.2 Elements for Public Summary
 RMP section VI.2 Elements for Public Summary Product: Atropin Stragen, 0,1 mg/ml solution for injection in pre-filled syringe Atropin Stragen, 0,2 mg/ml solution for injection in pre-filled syringe RMP:
RMP section VI.2 Elements for Public Summary Product: Atropin Stragen, 0,1 mg/ml solution for injection in pre-filled syringe Atropin Stragen, 0,2 mg/ml solution for injection in pre-filled syringe RMP:
Composition Each ml of Ventol solution for inhalation contains 5 mg Salbutamol (as sulphate).
 VENTOL Composition Each ml of Ventol solution for inhalation contains 5 mg Salbutamol (as sulphate). Respiratory Solution Action Salbutamol is a short-acting, relatively selective beta2-adrenoceptor agonist.
VENTOL Composition Each ml of Ventol solution for inhalation contains 5 mg Salbutamol (as sulphate). Respiratory Solution Action Salbutamol is a short-acting, relatively selective beta2-adrenoceptor agonist.
Core Safety Profile. Date of FAR:
 Core Safety Profile Active substance: Levobupivicaine Pharmaceutical form(s)/strength: Solution for injection, concentrate for solution for infusion, 2,5 mg/ml, 5 mg/ml, 7,5 mg/ml, 0,625 mg/ml, 1,25 mg/ml
Core Safety Profile Active substance: Levobupivicaine Pharmaceutical form(s)/strength: Solution for injection, concentrate for solution for infusion, 2,5 mg/ml, 5 mg/ml, 7,5 mg/ml, 0,625 mg/ml, 1,25 mg/ml
DRUG GUIDELINE. MAGNESIUM SULFATE (Obstetric)
 DRUG GUIDELINE MAGNESIUM SULFATE (Obstetric) SCOPE (Area): FOR USE IN: Labour Ward, 5N, HDU, ED and Theatre EXCLUSIONS: Paediatrics (seek Paediatrician advice) and other General Wards SCOPE (Staff): Medical,
DRUG GUIDELINE MAGNESIUM SULFATE (Obstetric) SCOPE (Area): FOR USE IN: Labour Ward, 5N, HDU, ED and Theatre EXCLUSIONS: Paediatrics (seek Paediatrician advice) and other General Wards SCOPE (Staff): Medical,
ALS MODULE 7 Pharmacology
 ALS MODULE 7 Pharmacology Relates to HLT404C Apply Advanced Resuscitation Techniques Introduction There are no studies that addressed the order of drug administration. There is inadequate evidence to define
ALS MODULE 7 Pharmacology Relates to HLT404C Apply Advanced Resuscitation Techniques Introduction There are no studies that addressed the order of drug administration. There is inadequate evidence to define
PACKAGE LEAFLET: INFORMATION FOR THE USER. Atracurium Besilate 10 mg/ml Solution for Injection The active substance is atracurium besilate
 PACKAGE LEAFLET: INFORMATION FOR THE USER Atracurium Besilate 10 mg/ml Solution for Injection The active substance is atracurium besilate Read all of this leaflet carefully before you start using this
PACKAGE LEAFLET: INFORMATION FOR THE USER Atracurium Besilate 10 mg/ml Solution for Injection The active substance is atracurium besilate Read all of this leaflet carefully before you start using this
SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS PRODUCT SUMMARY 1. NAME OF THE MEDICINAL PRODUCT Sterile Potassium Chloride Concentrate 15%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15% of Potassium Chloride in
SUMMARY OF PRODUCT CHARACTERISTICS PRODUCT SUMMARY 1. NAME OF THE MEDICINAL PRODUCT Sterile Potassium Chloride Concentrate 15%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15% of Potassium Chloride in
PACKAGE LEAFLET: INFORMATION FOR THE USER. Cisatracurium 2 mg/ml Solution for Injection/Infusion Cisatracurium 5 mg/ml Solution for Injection/Infusion
 PACKAGE LEAFLET: INFORMATION FOR THE USER Cisatracurium 2 mg/ml Solution for Injection/Infusion Cisatracurium 5 mg/ml Solution for Injection/Infusion Cisatracurium Read all of this leaflet carefully before
PACKAGE LEAFLET: INFORMATION FOR THE USER Cisatracurium 2 mg/ml Solution for Injection/Infusion Cisatracurium 5 mg/ml Solution for Injection/Infusion Cisatracurium Read all of this leaflet carefully before
Package leaflet: Information for the user
 Package leaflet: Information for the user Cisatracurium 2 mg/ml Solution for injection/ infusion Cisatracurium 5 mg/ml Solution for injection/ infusion Cisatracurium The name of your medicine are Cisatracurium
Package leaflet: Information for the user Cisatracurium 2 mg/ml Solution for injection/ infusion Cisatracurium 5 mg/ml Solution for injection/ infusion Cisatracurium The name of your medicine are Cisatracurium
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS Page 1 of 9 1. NAME OF THE MEDICINAL PRODUCT Magnesium sulfate-kalceks 500 mg/ml solution for injection, BP * 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml of solution
SUMMARY OF PRODUCT CHARACTERISTICS Page 1 of 9 1. NAME OF THE MEDICINAL PRODUCT Magnesium sulfate-kalceks 500 mg/ml solution for injection, BP * 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml of solution
Chapter 18 Neuromuscular Blocking Agents Study Guide and Application Exercise
 Chapter 18 Neuromuscular Blocking Agents Study Guide and Application Exercise 1. Read chapter 2. Review objectives (p.305) 3. Review key terms and definitions (p.305) Add: Cholinesterase inhibitor Vagal
Chapter 18 Neuromuscular Blocking Agents Study Guide and Application Exercise 1. Read chapter 2. Review objectives (p.305) 3. Review key terms and definitions (p.305) Add: Cholinesterase inhibitor Vagal
New Zealand Data Sheet DBL TM Rocuronium Bromide Injection
 New Zealand Data Sheet DBL TM Rocuronium Bromide Injection NAME OF THE MEDICINE DBL TM Rocuronium Bromide Injection 50 mg/5 ml Vial PRESENTATION Rocuronium bromide is a quaternary aminosteroid and an analogue
New Zealand Data Sheet DBL TM Rocuronium Bromide Injection NAME OF THE MEDICINE DBL TM Rocuronium Bromide Injection 50 mg/5 ml Vial PRESENTATION Rocuronium bromide is a quaternary aminosteroid and an analogue
PRODUCT INFORMATION. NAME OF THE MEDICINE Compound Sodium Lactate (Hartmann's) Solution for Injection
 PRODUCT INFORMATION NAME OF THE MEDICINE Compound Sodium Lactate (Hartmann's) Solution for Injection DESCRIPTION Molecular formulae. Potassium chloride: KCl; sodium chloride: NaCl; calcium chloride dihydrate:
PRODUCT INFORMATION NAME OF THE MEDICINE Compound Sodium Lactate (Hartmann's) Solution for Injection DESCRIPTION Molecular formulae. Potassium chloride: KCl; sodium chloride: NaCl; calcium chloride dihydrate:
Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup.
 Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
FOR REPRESENTATIVE EDUCATION
 Neuromuscular Blockade in the ICU NIMBEX Indication 1 NIMBEX (cisatracurium besylate) is indicated as an adjunct to general anesthesia to facilitate tracheal intubation in adults and in pediatric patients
Neuromuscular Blockade in the ICU NIMBEX Indication 1 NIMBEX (cisatracurium besylate) is indicated as an adjunct to general anesthesia to facilitate tracheal intubation in adults and in pediatric patients
FENTANYL INJECTION PRODUCT INFORMATION
 Fentanyl Injection Product Information 1 (11) FENTANYL INJECTION PRODUCT INFORMATION NAME OF THE MEDICINE Fentanyl citrate is chemically identified as N-(1-phenethyl-4-piperidyl) propionanilide citrate,
Fentanyl Injection Product Information 1 (11) FENTANYL INJECTION PRODUCT INFORMATION NAME OF THE MEDICINE Fentanyl citrate is chemically identified as N-(1-phenethyl-4-piperidyl) propionanilide citrate,
ADULT DRUG REFERENCE Drug Indication Adult Dosage Precautions / Comments
 ADENOSINE Paroxysmal SVT 1 st Dose 6 mg rapid IV 2 nd & 3 rd Doses 12 mg rapid IV push Follow each dose with rapid bolus of 20 ml NS May cause transient heart block or asystole. Side effects include chest
ADENOSINE Paroxysmal SVT 1 st Dose 6 mg rapid IV 2 nd & 3 rd Doses 12 mg rapid IV push Follow each dose with rapid bolus of 20 ml NS May cause transient heart block or asystole. Side effects include chest
DRUG GUIDELINE. HYDRALAZINE (Intravenous severe hypertension in pregnancy)
 DRUG GUIDELINE HYDRALAZINE (Intravenous severe hypertension SCOPE (Area): FOR USE IN: Labour Ward, HDU, Theatre and ED EXCLUSIONS: Paediatrics (seek Paediatrician advice) and other general wards. SCOPE
DRUG GUIDELINE HYDRALAZINE (Intravenous severe hypertension SCOPE (Area): FOR USE IN: Labour Ward, HDU, Theatre and ED EXCLUSIONS: Paediatrics (seek Paediatrician advice) and other general wards. SCOPE
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 AUSTRALIAN PRODUCT INFORMATION ASTRAZENECA FENTANYL INJECTION (FENTANYL CITRATE) 1 NAME OF THE MEDICINE Fentanyl citrate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 2 ml sterile solution of ph 4.0 7.5
AUSTRALIAN PRODUCT INFORMATION ASTRAZENECA FENTANYL INJECTION (FENTANYL CITRATE) 1 NAME OF THE MEDICINE Fentanyl citrate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 2 ml sterile solution of ph 4.0 7.5
PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker
 PACKAGE INSERT Pr PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker ACTIONS AND CLINICAL PHARMACOLOGY Phentolamine produces an alpha-adrenergic
PACKAGE INSERT Pr PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker ACTIONS AND CLINICAL PHARMACOLOGY Phentolamine produces an alpha-adrenergic
Product Information BROWN SNAKE ANTIVENOM AUST R 74897
 Product Information APPROVED NAME BROWN SNAKE ANTIVENOM AUST R 74897 DESCRIPTION BROWN SNAKE ANTIVENOM is prepared from the plasma of horses immunised with the venom of the brown snake (Pseudonaja textilis).
Product Information APPROVED NAME BROWN SNAKE ANTIVENOM AUST R 74897 DESCRIPTION BROWN SNAKE ANTIVENOM is prepared from the plasma of horses immunised with the venom of the brown snake (Pseudonaja textilis).
Pharmacology of the Neuromuscular Junction (NMJ)
 Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
PRODUCT INFORMATION. (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine dihydrogen 2-hydroxypropane-1,2,3-tricarboxylate
![PRODUCT INFORMATION. (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine dihydrogen 2-hydroxypropane-1,2,3-tricarboxylate PRODUCT INFORMATION. (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine dihydrogen 2-hydroxypropane-1,2,3-tricarboxylate](/thumbs/81/82665370.jpg) NORGESIC Orphenadrine citrate and paracetamol PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Chemical name: CAS number: 4682-36-4 Chemical structure: Orphenadrine citrate (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine
NORGESIC Orphenadrine citrate and paracetamol PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Chemical name: CAS number: 4682-36-4 Chemical structure: Orphenadrine citrate (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine
TRAPADOL INJECTION FOR I.V./I.M. USE ONLY
 TRAPADOL INJECTION FOR I.V./I.M. USE ONLY Composition : Each 2ml. contains : Tramadol Hydrochloride I.P. Water for injection I.P. 100mg. q.s. CLINICAL PHARMACOLOGY : Pharmacodynamics Tramadol is a centrally
TRAPADOL INJECTION FOR I.V./I.M. USE ONLY Composition : Each 2ml. contains : Tramadol Hydrochloride I.P. Water for injection I.P. 100mg. q.s. CLINICAL PHARMACOLOGY : Pharmacodynamics Tramadol is a centrally
This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards.
 MIVACRON Injection (mivacurium chloride) This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards. DESCRIPTION MIVACRON (mivacurium
MIVACRON Injection (mivacurium chloride) This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards. DESCRIPTION MIVACRON (mivacurium
SANDOMIGRAN (pizotifen malate)
 SANDOMIGRAN (pizotifen malate) S N CH 3 Pizotifen. COOH CH OH CH 2 COOH MALATE DESCRIPTION Pizotifen is a cycloheptathiophene derivative structurally related to cyproheptadine and the tricyclic antidepressants.
SANDOMIGRAN (pizotifen malate) S N CH 3 Pizotifen. COOH CH OH CH 2 COOH MALATE DESCRIPTION Pizotifen is a cycloheptathiophene derivative structurally related to cyproheptadine and the tricyclic antidepressants.
08-15 NORCURON ORGANON
 08-15 NORCURON ORGANON 1. NAME OF THE MEDICINAL PRODUCT Norcuron 4 mg, powder for solution for injection Norcuron 10 mg, powder for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Norcuron
08-15 NORCURON ORGANON 1. NAME OF THE MEDICINAL PRODUCT Norcuron 4 mg, powder for solution for injection Norcuron 10 mg, powder for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Norcuron
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Rocuronium bromide 10 mg/ml solution for injection/ infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution of Rocuronium
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Rocuronium bromide 10 mg/ml solution for injection/ infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution of Rocuronium
Angina pectoris due to coronary atherosclerosis : Atenolol is indicated for the long term management of patients with angina pectoris.
 Lonet Tablet Description Lonet contains Atenolol, a synthetic β1 selective (cardioselective) adrenoreceptor blocking agent without membrane stabilising or intrinsic sympathomimetic (partial agonist) activity.
Lonet Tablet Description Lonet contains Atenolol, a synthetic β1 selective (cardioselective) adrenoreceptor blocking agent without membrane stabilising or intrinsic sympathomimetic (partial agonist) activity.
SUMMARY OF PRODUCT CHARACTERISTICS. 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for injection
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for injection
PRODUCT INFORMATION. 2-(dimethylamino) ethyl (RS)-2-(1-hydroxycyclopentyl)-2- phenylacetate hydrochloride
 PRODUCT INFORMATION MINIMS CYCLOPENTOLATE EYE DROPS NAME OF THE MEDICINE Cyclopentolate hydrochloride Structural formula: Chemical name: Molecular formula: Molecular weight: 327.9 2-(dimethylamino) ethyl
PRODUCT INFORMATION MINIMS CYCLOPENTOLATE EYE DROPS NAME OF THE MEDICINE Cyclopentolate hydrochloride Structural formula: Chemical name: Molecular formula: Molecular weight: 327.9 2-(dimethylamino) ethyl
Magnesium Sulphate - Management of Hypertensive Disorders of Pregnancy
 1. Purpose Magnesium sulphate is the anticonvulsant of choice for pre-eclampsia prophylaxis and treatment. This clinical guideline outlines the indications, contraindications, administration and monitoring
1. Purpose Magnesium sulphate is the anticonvulsant of choice for pre-eclampsia prophylaxis and treatment. This clinical guideline outlines the indications, contraindications, administration and monitoring
Pharmacology of the Neuromuscular Junction (NMJ)
 Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
Pain: 1-2µg/kg q30-60min prn. effects in 10 minutes. Contraindications: Morphine is preferred in. Duration of Action: minutes. renal failure.
 Procedural Sedation / Analgesia / Anaesthesia Chart - Page 1 Diazepam (Valium) Anxiolytic / Sedative Etomidate (Amidate) Hypnotic / Anesthetic Fentanyl Citrate (Sublimaze) Narcotic Analgesic Dose Pediatric:
Procedural Sedation / Analgesia / Anaesthesia Chart - Page 1 Diazepam (Valium) Anxiolytic / Sedative Etomidate (Amidate) Hypnotic / Anesthetic Fentanyl Citrate (Sublimaze) Narcotic Analgesic Dose Pediatric:
למידע נוסף ניתן לעיין בעלון לרופא באתר משרד הבריאות או ליצור קשר טלפוני בטלפון
 חברת פרמה מדיס שמחה לבשר על תחילת שיווקו של תכשיר חדש. שם התכשיר: NEOSTIGMINE - HAMELN 2.5 MG/ML INJECTION צורת מינון: SOLUTION FOR INJECTION דרך מתן: I.M, I.V, S.C הרכב )חומר פעיל(: NEOSTIGMINE METHYLSULFATE
חברת פרמה מדיס שמחה לבשר על תחילת שיווקו של תכשיר חדש. שם התכשיר: NEOSTIGMINE - HAMELN 2.5 MG/ML INJECTION צורת מינון: SOLUTION FOR INJECTION דרך מתן: I.M, I.V, S.C הרכב )חומר פעיל(: NEOSTIGMINE METHYLSULFATE
MESTINON NAME OF THE MEDICINE. Description: Pharmacology: Pyridostigmine Bromide. CAS number:
 MESTINON NAME OF THE MEDICINE Pyridostigmine Bromide. C9H13BrN2O2 CAS number: 101-26-8 Description: Mestinon contains, as active substance, pyridostigmine bromide, chemically described as the dimethylcarbamic
MESTINON NAME OF THE MEDICINE Pyridostigmine Bromide. C9H13BrN2O2 CAS number: 101-26-8 Description: Mestinon contains, as active substance, pyridostigmine bromide, chemically described as the dimethylcarbamic
Neosynephrine. Name of the Medicine
 Name of the Medicine Neosynephrine Phenylephrine hydrochloride 1% injection Neosynephrine Presentation Neosynephrine is a clear, colourless, aqueous solution, free from visible particulates, in sterile
Name of the Medicine Neosynephrine Phenylephrine hydrochloride 1% injection Neosynephrine Presentation Neosynephrine is a clear, colourless, aqueous solution, free from visible particulates, in sterile
ASTHALIN Respirator Solution (Salbutamol sulphate)
 Published on: 28 Jan 2016 ASTHALIN Respirator Solution (Salbutamol sulphate) Composition Each 1 ml contains: Salbutamol Sulphate IP equivalent to Salbutamol IP.. 5 mg Dosage Form Solution for inhalation
Published on: 28 Jan 2016 ASTHALIN Respirator Solution (Salbutamol sulphate) Composition Each 1 ml contains: Salbutamol Sulphate IP equivalent to Salbutamol IP.. 5 mg Dosage Form Solution for inhalation
200 mg/20 ml (10 mg/ml) in single-dose vials (3)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use NIMBEX safely and effectively. See full prescribing information for NIMBEX. NIMBEX (cisatracurium
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use NIMBEX safely and effectively. See full prescribing information for NIMBEX. NIMBEX (cisatracurium
PACKAGE LEAFLET: INFORMATION FOR THE USER. Cisatracurium 2 mg/ml solution for injection/infusion Cisatracurium 5 mg/ml solution for injection/infusion
 PACKAGE LEAFLET: INFORMATION FOR THE USER Cisatracurium 2 mg/ml solution for injection/infusion Cisatracurium 5 mg/ml solution for injection/infusion Cisatracurium Read all of this leaflet carefully before
PACKAGE LEAFLET: INFORMATION FOR THE USER Cisatracurium 2 mg/ml solution for injection/infusion Cisatracurium 5 mg/ml solution for injection/infusion Cisatracurium Read all of this leaflet carefully before
SUMMARY OF PRODUCT CHARACTERISTICS. 1 ml solution contains 75 micrograms of sufentanilcitrate, corresponding to 50 micrograms of sufentanil.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Sufentanil Narcomed, 50 microgram / ml, solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 75
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Sufentanil Narcomed, 50 microgram / ml, solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 75
Chapter 18. Skeletal Muscle Relaxants (Neuromuscular Blocking Agents)
 Chapter 18 Skeletal Muscle Relaxants (Neuromuscular Blocking Agents) Uses of Neuromuscular Blocking Facilitate intubation Surgery Agents Enhance ventilator synchrony Reduce intracranial pressure (ICP)
Chapter 18 Skeletal Muscle Relaxants (Neuromuscular Blocking Agents) Uses of Neuromuscular Blocking Facilitate intubation Surgery Agents Enhance ventilator synchrony Reduce intracranial pressure (ICP)
ASTHALIN Respules (Salbutamol sulphate)
 Published on: 28 Jan 2016 ASTHALIN Respules (Salbutamol sulphate) Composition Each 2.5 ml respule contains: Salbutamol Sulphate IP equivalent to Salbutamol IP. 2.5 mg Normal saline solution.q.s. Dosage
Published on: 28 Jan 2016 ASTHALIN Respules (Salbutamol sulphate) Composition Each 2.5 ml respule contains: Salbutamol Sulphate IP equivalent to Salbutamol IP. 2.5 mg Normal saline solution.q.s. Dosage
The Latest Approaches to Reversal of Neuromuscular Blocking Agents
 The Latest Approaches to Reversal of Neuromuscular Blocking Agents Janay Bailey, Pharm.D. Anesthesiology 2017; 126:173-90 Objectives Pharmacists Determine optimal paralytic choices in knowing if reversal
The Latest Approaches to Reversal of Neuromuscular Blocking Agents Janay Bailey, Pharm.D. Anesthesiology 2017; 126:173-90 Objectives Pharmacists Determine optimal paralytic choices in knowing if reversal
The hydrated salt contains approximately 15% water (3½ moles) and has a molecular weight of 399. The anhydrous salt has a molecular weight of 336.
 PRODUCT INFORMATION DANTRIUM powder for injection 20 mg NAME OF THE MEDICINE Dantrolene sodium. Chemically, the drug is hydrated 1-[[[5-(4-nitrophenyl)-2-furanyl]methylene]- amino]-2,4-imidazolidinedione
PRODUCT INFORMATION DANTRIUM powder for injection 20 mg NAME OF THE MEDICINE Dantrolene sodium. Chemically, the drug is hydrated 1-[[[5-(4-nitrophenyl)-2-furanyl]methylene]- amino]-2,4-imidazolidinedione
ROSOBAC-1GM / ROSOBAC-FORT
 ROSOBAC-1GM / ROSOBAC-FORT ROSOBAC - 1GM. COMPOSITION : Each vial contains Sterile Cefoperazone Sodium IP Eq. to Anhydrous Cefoperazone - Sterile Sulbactam Sodium USP Eq. to Anhydrous Sulbactam - ROSOBAC
ROSOBAC-1GM / ROSOBAC-FORT ROSOBAC - 1GM. COMPOSITION : Each vial contains Sterile Cefoperazone Sodium IP Eq. to Anhydrous Cefoperazone - Sterile Sulbactam Sodium USP Eq. to Anhydrous Sulbactam - ROSOBAC
PACKAGE LEAFLET: INFORMATION FOR THE USER. SODIPHOS 22mEq / 10ml Concentrate for solution for infusion. Disodium phosphate dihydrate
 PACKAGE LEAFLET: INFORMATION FOR THE USER SODIPHOS 22mEq / 10ml Concentrate for solution for infusion Disodium phosphate dihydrate Read all of this leaflet carefully before you start using this medicine.
PACKAGE LEAFLET: INFORMATION FOR THE USER SODIPHOS 22mEq / 10ml Concentrate for solution for infusion Disodium phosphate dihydrate Read all of this leaflet carefully before you start using this medicine.
PRODUCT MONOGRAPH. Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium. Non-depolarizing Skeletal Neuromuscular Blocking Agent
 PRODUCT MONOGRAPH Pr CISATRACURIUM BESYLATE INJECTION Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium Non-depolarizing Skeletal Neuromuscular Blocking Agent This drug should be administered
PRODUCT MONOGRAPH Pr CISATRACURIUM BESYLATE INJECTION Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium Non-depolarizing Skeletal Neuromuscular Blocking Agent This drug should be administered
PRODUCT INFORMATION. Bis[(1R,3r,5S)-8-methyl-8-azabicyclo[3.2.1]oct-3-yl (2RS)-3- hydroxy-2-phenylpropanoate] sulfate monohydrate
![PRODUCT INFORMATION. Bis[(1R,3r,5S)-8-methyl-8-azabicyclo[3.2.1]oct-3-yl (2RS)-3- hydroxy-2-phenylpropanoate] sulfate monohydrate PRODUCT INFORMATION. Bis[(1R,3r,5S)-8-methyl-8-azabicyclo[3.2.1]oct-3-yl (2RS)-3- hydroxy-2-phenylpropanoate] sulfate monohydrate](/thumbs/94/119690146.jpg) PRODUCT INFORMATION MINIMS ATROPINE EYE DROPS NAME OF THE MEDICINE Atropine sulphate Structural formula: Chemical name: Molecular formula: Molecular weight: 694.8 CAS number: DESCRIPTION Bis[(1R,3r,5S)-8-methyl-8-azabicyclo[3.2.1]oct-3-yl
PRODUCT INFORMATION MINIMS ATROPINE EYE DROPS NAME OF THE MEDICINE Atropine sulphate Structural formula: Chemical name: Molecular formula: Molecular weight: 694.8 CAS number: DESCRIPTION Bis[(1R,3r,5S)-8-methyl-8-azabicyclo[3.2.1]oct-3-yl
DOSAGE AND ADMINISTRATION
 Pr COLISTIMETHATE FOR INJECTION USP Colistimethate for Injection contains the sodium salt of colistimethate which is a polypeptide antibiotic with an approximate molecular weight of 1 750. The empirical
Pr COLISTIMETHATE FOR INJECTION USP Colistimethate for Injection contains the sodium salt of colistimethate which is a polypeptide antibiotic with an approximate molecular weight of 1 750. The empirical
GenRx SALBUTAMOL INHALATION AMPOULE
 GenRx SALBUTAMOL INHALATION AMPOULE NAME OF THE MEDICINE Salbutamol sulfate. Chemical Name: di[(rs)-2-(1,1-dimethyl)ethylamino-1-[4-hydroxy-3- (hydroxymethyl)phenyl]ethanol] sulfate. Structural Formula:
GenRx SALBUTAMOL INHALATION AMPOULE NAME OF THE MEDICINE Salbutamol sulfate. Chemical Name: di[(rs)-2-(1,1-dimethyl)ethylamino-1-[4-hydroxy-3- (hydroxymethyl)phenyl]ethanol] sulfate. Structural Formula:
MINIMS OXYBUPROCAINE EYE DROPS
 MINIMS OXYBUPROCAINE EYE DROPS Name of Medicine: Oxybuprocaine Hydrochloride Description: Composition: Active. Benoxinate (oxybuprocaine) HCI USP. Inactive. Water Purified and Hydrochloric acid. No preservatives
MINIMS OXYBUPROCAINE EYE DROPS Name of Medicine: Oxybuprocaine Hydrochloride Description: Composition: Active. Benoxinate (oxybuprocaine) HCI USP. Inactive. Water Purified and Hydrochloric acid. No preservatives
COMPOSITION Each bottle contains the active ingredient, sevoflurane, only. No additive or stabilizer is present.
 PROPRIETARY NAME (and dosage form) ULTANE TM liquid COMPOSITION Each bottle contains the active ingredient, sevoflurane, only. No additive or stabilizer is present. PHARMACOLOGICAL CLASSIFICATION N01AB08
PROPRIETARY NAME (and dosage form) ULTANE TM liquid COMPOSITION Each bottle contains the active ingredient, sevoflurane, only. No additive or stabilizer is present. PHARMACOLOGICAL CLASSIFICATION N01AB08
PRODUCT INFOMATION APO-IPRATROPIUM SOLUTION
 PRODUCT INFOMATION APO-IPRATROPIUM SOLUTION NAME OF THE MEDICINE Ipratropium bromide. Chemical Name: (1R,3r,5S,8r)-3-[[(2RS)-3-Hydroxy-2-phenylpropanoyl]oxy]-8-methyl-8-(1- methylethyl)-8-azoniabicyclo[3.2.1]octane
PRODUCT INFOMATION APO-IPRATROPIUM SOLUTION NAME OF THE MEDICINE Ipratropium bromide. Chemical Name: (1R,3r,5S,8r)-3-[[(2RS)-3-Hydroxy-2-phenylpropanoyl]oxy]-8-methyl-8-(1- methylethyl)-8-azoniabicyclo[3.2.1]octane
PAEDIATRIC ANAESTHETIC EMERGENCIES PART I. Dr James Cockcroft, South West School of Anaesthesia. Dr Sarah Rawlinson, Derriford Hospital, Plymouth, UK
 PAEDIATRIC ANAESTHETIC EMERGENCIES PART I Original Article by: Dr Claire Todd, South West School of Anaesthesia Dr James Cockcroft, South West School of Anaesthesia Dr Sarah Rawlinson, Derriford Hospital,
PAEDIATRIC ANAESTHETIC EMERGENCIES PART I Original Article by: Dr Claire Todd, South West School of Anaesthesia Dr James Cockcroft, South West School of Anaesthesia Dr Sarah Rawlinson, Derriford Hospital,
Adult Drug Reference. Dopamine Drip Chart. Pediatric Drug Reference. Pediatric Drug Dosage Charts DRUG REFERENCES
 Adult Drug Reference Dopamine Drip Chart Pediatric Drug Reference Pediatric Drug Dosage Charts DRUG REFERENCES ADULT DRUG REFERENCE Drug Indication Adult Dosage Precautions / Comments ADENOSINE Paroxysmal
Adult Drug Reference Dopamine Drip Chart Pediatric Drug Reference Pediatric Drug Dosage Charts DRUG REFERENCES ADULT DRUG REFERENCE Drug Indication Adult Dosage Precautions / Comments ADENOSINE Paroxysmal
