5 HT receptor agonists (triptans) and Pediatric Migraine Critically Appraised Topic (CAT)
|
|
|
- Ophelia O’Connor’
- 6 years ago
- Views:
Transcription
1 5 HT receptor agonists (triptans) and Pediatric Migraine Critically Appraised Topic (CAT) PICOT Question: In children and adolescents does use of 5 HT receptor agonists (triptans) versus no treatment treat decrease migraine pain? Clinical bottom line based on literature appraisal below: The 5-HT receptor agonists ( triptans ) have been extensively studied in the treatment of pediatric migraine headache (Ahonen, Hämäläinen, Rantala, Hopp, 2004; Hämäläinen, Hoppu & Santavuori, 1997; Lewis, Hershey & Wasiewski, 2007; Rothner, Wasiewski, Winner, Lewis & Stankowski, 2006; Winner, Adelman, Aurora, Lener & Ames, 2006; Winner, Lewis, Visser, Jiang, Ahrens & Evans, 2002 Winner, Rothner, Wooten, Webster & Ames, 2006). Safety in the pediatric population has been well defined (Ahonen, Hämäläinen, Rantala, Hopp, 2004; Hämäläinen, Hoppu & Santavuori, 1997; Lewis, Hershey & Wasiewski, 2007; Rothner, Wasiewski, Winner, Lewis & Stankowski, 2006; Winner, Adelman, Aurora, Lener & Ames, 2006; Winner, Lewis, Visser, Jiang, Ahrens & Evans, 2002 Winner, Rothner, Wooten, Webster & Ames, 2006). Currently, the only triptan available at Children s Mercy Hospital is sumatriptan (Imitrex). The recommended dose and route of administration for the acute care setting is 0.06 mg/kg (maximum 6 mg) subcutaneously (SQ). Dosing of sumatriptan may be repeated for a total of two (2) doses in 24 hours two hours must elapse between repeat doses of triptans, including sumatriptan (Gunn & Nechyba, 2002). There should be no more than two triptan doses in a 24 hour period, and two different triptans should not be utilized in the same patient. Limitations to triptan use include the following: uncontrolled hypertension, chest pain, basilar migraine, or use of monoamine-oxidase inhibitors (MAOI s). Triptans should not be used concomitantly (or within 24 hours) with dihydroergotamine (DHE) (Gunn & Nechyba, 2002). [GRADE = Weak recommendation / Moderate-quality evidence] Search strategy implemented: (("Infusions, Intravenous"[Mesh] OR "Prochlorperazine"[Mesh] OR "Metoclopramide"[Mesh] OR "Ondansetron"[Mesh] OR "Promethazine"[Mesh] OR "Naproxen"[Mesh] OR "Ketorolac Tromethamine"[Mesh] OR "Diphenhydramine"[Mesh] OR "Valproic Acid"[Mesh] OR "Dihydroergotamine"[Mesh] OR "Methylprednisolone Hemisuccinate"[Mesh] OR "Magnesium"[Mesh] OR "Magnesium Sulfate"[Mesh] OR "Acetaminophen"[Mesh] OR "Ibuprofen"[Mesh] OR "Sumatriptan"[Mesh] OR "Adrenergic beta-antagonists"[mesh] OR "Antidepressive Agents, Tricyclic"[Mesh] OR "petadolex"[all Fields]) AND ("Migraine Disorders"[Mesh] OR "Headache"[Mesh] OR "Headache Disorders"[Mesh])) AND ("Adolescent"[Mesh] OR "Child"[Mesh] OR "Infant"[Mesh] OR "Child, Preschool"[Mesh] OR "pediatric"[all Fields] OR "paediatric"[all Fields]) AND ("humans"[mesh Terms] AND English[lang] AND (systematic[sb] OR Meta-Analysis[ptyp] OR Practice Guideline[ptyp] OR Randomized Controlled Trial[ptyp] NOT Case Reports[ptyp] OR Comparative Study[ptyp] OR Controlled Clinical Trial[ptyp])). Prochlorperazine articles were reviewed for this CAT. Search outcome: 15 papers were found and reviewed. Six studies treated children and adolescents. The remainder treated adults with migraine. The pediatric studies include one systematic review and 5 therapy studies. Synthesis of relevant studies: Author, date, country, and industry of Patient Group funding Lewis, D.,W., Adolescents Hershey, A.D., & years old for the Wasiewski, W.W. duration of the study (2007). On behalf of the adolescent HA were treated in migraine steering the moderate to committee. severe stage, Oxford / GRADE* Oxford: 1b Research design Significant results Limitations It was a multicenter, randomized, doubleblind, placebocontrolled, 2 way, 2 attack, crossover study, with a single blind placebo 171 subjects were treated, 148 with zolmitriptan and 127 with placebo 96 subjects completed sequence A 103 subjects completed sequence B At one hour: Higher HA response rate for the Subjects can taste the study drug, but this is common in all nasal triptan studies This report is not the intent to treat group, 1
2 Pediatrics 120, Mean age 14.2 y (+ 1.5) Rothner, A.D., Wasiewski,W., Winner, P., Lewis, D. & Stankowski, J.(2006). Zolmitriptan oral tablet in migraine Power was met Adolescents years were open to be in this study Mean age ITT population years (SD 1.7) Oxford 2b challenge for each migraine attack. zolmitriptan (5mg) nasal spray or placebo N= 248 who were randomized into each of two arms. Two migraine attacks were treated. Prior to receiving study drug, subjects were treated with placebo. If HA did not abate in 15 minutes, the study drug they were randomized to, was administered. Sequential randomization was used. 171 subjects were reported on. 80 in the placebo first, zolmitriptan second group 91 in the zolmitriptan first, placebo second group Randomized, doubleblind, placebocontrolled 10 mg, 5 mg or 2.5 mg of zolmitriptan zolmitriptan group (OR 1.827, 95% CI: P<0.05) Response in favor of zolmitriptan at 15 min (OR 2.020, 95% CI p< 0.05) 1.5 hours- NS 2 hours NS 2 hours sustained- (OR 2.243; 95% CI: p< 0.01) Pain free rates were higher with zolmitriptan at all times significant at 1 hour -27.7% vs 10.2% 1.5 hours % vs. 15.7% 2 hours % vs % All significant p<0.01 Early onset based on 1 point decrease in pain intensity 15 minutes-- 50% vs. 35.4%, p< minutes % vs %. p<0.05 Resolution of migraine symptoms at 30 minutes favored zolmitriptan Photophobia % vs. 19.6% p< 0.05 Phonophobia % vs. 21.4% p< 0.05 Return to normal activities favored zolmitriptan at all time points, significant at 45 minutes 48.0% vs. 33.9%, p< hour 55.1% vs. 41.3% p< 0.05 No difference in the worst case scenario (ITT analysis) Analysis was done in step wise fashion. If a significant result was found at the 10 mg. dose, the 5 mg dose was tested. If a significant result was found at the 5 mg dose, the 2.5 mg dose was tested. only those who finished both arms. No CIs Hawthorne effect Adult HA criteria Not enough points on the pain scale 2
3 treatments: High placebo responses in adolescents. Headache, 46, mg group N=179 5 mg groups, N= mg groups, N= 171 Placebo group, N= 175 tablet. Self treated moderate to severe migraine attacks There was no difference between treatment and placebo groups at One hour, 30 minutes. The placebo group had slightly better response rate at 2 hours. Winner, P., Rothner, D., Wooten, J., Webster, C. & Ames, M. (2006). Sumatriptan nasal spray in adolescent migraineurs: A randomized, double-blind, placebo-controlled, acute study. Headache, 46, Power was met The population studied was adolescents years of age Mean age 14 y (range y) 738 subjects were recruited, 245 in the placebo group, 255 in the sumatriptan 5 mg group and 238 in the sumatriptan 20 mg group. Power was met. Oxford: 2 b Randomized, placebo controlled, double-blind Randomized (1:1:1) in blocks of six. Self treated a single moderate to severe migraine attack. Sumatriptan NS at 5, and 20 mg vs. placebo Primary outcomes: no significant difference was found between HA relief rates at one hour (61% vs. 52%). Sustained relief rates from 1 to 24 hours post dose were not significant. The proportion of subjects reporting pain free was significantly higher in the sumatriptan NS 20 mg group at 2 hours post dose (44% vs. 30%) p < 0.001, however not at one hour. Secondary outcomes HA relief at time points through 2 hours with sumatriptan NS 20 mg than with placebo within 2 hours post dose (76% vs.65% p = 0.007). The probability of achieving pain free results was significantly greater with sumatriptan NS 20 mg (cumulative percentages of subjects: 45% vs. 34% respectively p = 0.014). Pain free rate was significantly higher in the sumatriptan NS 20 mg group at 2 hours post does (41%vs. 28%, p = 0.003). Photophobia was significantly lower in the sumatriptan NS 20 mg group at 2 hours postdose (31% vs. 36% p = 0.019). Photophobia plus phonophobia was lower in the sumatriptan NS 20 mg (21% vs 29% p = 0.044). Use of rescue medication- was not significantly different. HA recurrence( return of moderate or severe pain 1-24 hours after HA relief at one hour) was not significantly CIs are not reported The data on the secondary endpoints suggest benefit, not primary end points. Again high Hawthorne effect plays a role. 3
4 Ahonen, K., Hämäläinen, M.L,, Rantala H., & Hoppu K. (2004). Nasal sumatriptan is effective in treatment of migraine attacks in children: A randomized trial. Neurology, 62, Winner, P., Lewis, D.L., Visser, H., Jiang, K., Ahrens, S., & Evans, J.K. (2002), Rizatriptan 5 mg for the acute treatment of migraine in adolescents: A randomized, doubleblind, placebocontrolled study. Children and adolescents between 8 and 17 years of age N= 83 used both treatments N= 94 used at least one treatment (ITT) Mean age of children studied was 14 years (range, years) Power was met Adolescents between 12 and 17 y with an average of at least 1 but < 8 migraine headaches (based on the International Headache Society criteria) per month over the previous six months whose migraines lasted on average at least 4 Oxford : 2b Oxford: 2b Double blind, randomized, placebo-controlled, two-way crossover design Two moderate to severe migraine attacks were treated at home with study drugs. 10 mg for body weight kg and 20 mg of body weight > 40 kg A five point HA scale was used 5 severe, 4 to 3 moderate, 2 mild, 1 no pain Randomized, double-blind, placebo-controlled study. Treatment Rizatriptan 5 mg vs placebo different. Tolerability evaluation. AE reporting major AE was taste disturbance. No serious AE was reported. Data is reported as a proportion of subjects experiencing an outcome. p values are reported, CIs are not. ITT 94 subjects 177 treatments (90 treatment 87 placebo) Primary end point (HA relief at least 2 points lower in 2 hours time) was improved in the sumatriptan group. 67% vs. 38% p< Secondary endpoints (at 30 minutes post treatment relief from sumatriptan) was slightly superior to placebo 31% Vs 16% p = 0.03 at one hour post treatment relief was better in the sumatriptan than the placebo 53% vs 29% p =0.003 no significance was found between treatment and placebo and total pain free response at 1 hour, or 2 hours post treatment. Rescue meds were taken more after treatment with placebo than sumatriptan 51% vs 32 p = 0.03 AE occurred in 44% of patients, 39% after sumatriptan and 8% after placebo. No serious AE. Most common was bad taste. AE s placebo patients has a significantly higher incidence of nausea and somnolence then treatment group. Pain-free and Pain-relief: Difference was not significant between tx and placebo group. Associated symptoms at baseline: More placebo pts reported nausea than the tx group. Nausea was significantly higher in the placebo group at 1, 1.5, and 4 hours. This reporting might be related to the higher numbers of pts. No CIs The report of taste difference was considered to have unblinded in some instances. Therefore secondary analysis of the first period was done, which had stronger results. It is reported here: Treatment with sumatriptan was 50% effective vs. 21% placebo (p=0.004) at one hour Treatment with sumatriptan was 74% in the treatment group vs. 33% in the placebo group. [< 0.001) Taste of the study drug distinguished the treatment- unblinded the study to the subject. All groups were fairly balanced with the exception of the baseline headache severity groups. The treatment group participants identified 63% with a moderate baseline headache and 37% having a severe headache compared to the placebo group 4
5 Headache, 42, Hämäläinen, M.L., Hoppu, K., & Santavuori P. (1997). Sumatriptan for migraine attacks in children: A randomized placebo controlled study. Neurology, 48, Winner, P., Adelman, J., Aurora, S., Lener, M.E. & Ames, M. (2006). Efficacy and tolerability of sumatriptan injection for the treatment of morning migraine: Two multicenter, prospective, randomized, doubleblind, controlled studies in adults. hours. Treatment group n=149 Placebo group n=147 Mean age 14 Power was met Children > 8 years old.mean age12.3 years (range, ) years old N= 23 children Power calc not done yoa with a > 1 yr hx of migraine with or without aura as defined by the International Headache Society; experience 1-6 migraines/month during each of the 3 previous months, had awakened with moderate to severe migraine pain at Oxford: 2 b Oxford:1 b Double blind randomized placebo controlled two way crossover sumatriptan 50 mg for a body surface area of 0.75 to 1.5 m 2, and 100 mg of a body surface > 1.5 m 2 Randomized, double-blind, parallel-group studies Dose: Sumatriptan injection 6 mg or matching inactive placebo; pts self administered medication within an hour of waking with a moderate severe migraine; reporting nausea in the placebo group at baseline. Functional disability: At 1.5 and 2 hours the tx group reported a significant (p =.03 and.03 respectively) ability to function normally Additional medication required was not significant. Recurrence 11% of tx group and 18% of placebo group had a recurrence; no analysis was performed. They showed no difference in pain resolution if the subjects took sumatriptan or placebo, as measured by 50% pain resolution at 2 hours after treatment. 7/23 showed 50% pain reduction after drug and 5/23 show pain reduction after placebo. 95% CI for the difference -21 to38% p = ns. HA resolution. 5/23 had complete resolution of pain after drug and 2/23 had resolution after placebo. 95% confidence interval for the difference -9 to35 p =ns. Reduction in pain intensity. Pain intensity difference was not different. Neither treatment order nor period had significant effects on pain intensity difference (start to 2 hours after treatment) and sum of pain intensity difference. Tx grps proved to be more effective 48% & 57% vs. control gps 18% & 19% (p <.001); with the onset of pain-free response 30 minutes and 20 minutes in the tx groups ( p<.01). Tx groups headache relief after 2 hours 72% and 77% vs control grps 41% & 32% (p <.001); Efficacy of tx occurred by 20 minutes after tx and 10 minutes after tx in the tx grps (p <.01). Migraine associated symptoms were significantly lower in tx grps than in control grps at 2 hours (p <.001). which had 53% with a moderate baseline headache and 44% with a severe headache. Did not truly randomize; the subjects self randomize. Didn t define the terms escape and rescue meds. Unknown pharmacokinetics for sumatriptan in children. Only used the tablet form. Did not report on lost subjects. IM medication is selfadministered. Multiple exclusion criteria (uncontrolled hypertension, cardiovascular or cerebrovascular disease, basilar or hemiplegic migraine, current use of migraine controlling medication containing ergotamine, ergot derivative, or 5
6 Clinical Therapeutics, 28, Scholpp, J., Schellenberg, R., Moeckesch, B. & Banik, N. (2004). Early treatment of a migraine attack while pain is still mild increased the efficacy of sumatriptan. Cephalagia, 24, Supported by GlaxoSmithKline. Kelly, A.M., Ardagh, M., Curry, C., D Antonio, J., & Zebic, S. (1997). Intervenous chlorpromazine versus intramuscular sumatriptan for acute migraine. J Accident and Emergency Medicine,14, least once in the 3 previous months. Study 1 N=297, Tx grp, n=145, Control, n=152; Study 2, N=287, Tx grp, n=148, Control, n=139. Adults Early ( first sign of pain and not later than one hours after onset of pain) or late (wait until pain was at least moderate or severe (at least grade 2 or 3) treatment with 100 mg tablet of sumatriptan Adults with migraine subjects (mean age) 35 years Control 32 years Oxford: 1B Oxford: 1b pts could administer a second dose or use a rescue medication 2 hours after the initial dose if they experienced inadequate pain relief Prospective controlled and open label study Randomized Sumatriptan 100 mg oral early Control Sumatriptan 100 mg oral late Prospective, randomized unblinded crossover trial chlorpromazine mg IV Control: sumatriptan 6 mg IM Pain-free response sustained in 32% & 34% of patients in tx group than in control grps 14% & 15% (p <.001). Normal function before tx 0%-1% in tx grps.; 2 hrs after intervention tx grp reported 49% & 61%, control grp 22% & 21% (p <.001). Tx grp pts used a lower percentage of second dose or rescue meds within 24 hour time period (46% & 46%) vs. control grp (63% & 65%). Primary outcome- the percentage of patients who were completely pain free at 2 hours after taking study medication was significantly greater (p = 0.43) in the early intervention group, (n = 59/89, 71.1%, 95% CI %) when compared to the late intervention group (n = 38/70, 54.3%, 95% CI %). No difference between mg of chlorpromazine IV versus 6 mg of sumatriptan IM. methysergide and use of monoamine oxidase inhibitor) which may limit generalization to entire population. Study was done by the company who makes the drug. In the early treatment group AEs were reported as drug effect, where they may have been migraine effect, whereas the late group may have had the same AEs and attributed them to the HA. Open label, so the subjects knew they were getting drug- Hawthorne Effect may have increased reported efficacy. Randomization was not clear. The major difference was the IV that had to be inserted to administer the chlorpromazine. Although both groups received 1L of fluid, it appears the chlorpromazine group received more fluid, although it is not accounted for in the results. A power calculation was 6
7 not done. Boureau, F., Chazot, G., Emile, J., Bertin, L., & d Allens, H. (1995). Comparison of subcutaneous sumatriptan with usual acute treatments for migraine. European Neurology 35, Major, P.W., Grubisa, H.S.I. & Thie, N.M.R. (2003). Triptans for treatment of acute pediatric migraine: a systematic literature review. Pediatric Neurology, 29, Lampl, C., Huber, G., Haas, S., Ritteberger, E. & Diener, H.C. (2008). Difference in triptan effect in patients with migraine and early allodynia. Adults y Mean age 42 (range 20-65) N= 217 evaluable patients treating a total of 3181 attacks. Treatment groups were similar Studies who included children 6-18 years old with migraine not responsive to other interventions Four studies were found for effectiveness and four studies were found for safety Adults years N= 36 Six subjects per group (mean age): Zolmitriptan 1.5 mg (37.6 ) Eletriptan 80 mg (33.3) Oxford: Oxford- 2b Oxford: 1a- Oxford:2 B low quality RCT Randomized, crossover Control: usual migraine treatment Intervention: Subcutaneous sumatriptan 6 mg auto-inject device versus Only randomized, placebo controlled, double blinded studies were included to study effectiveness Studies published in English and French were searched. Does not state which bibliographic databases were used, if personal contact was made with experts, if unpublished data was used, Reports were chosen by abstract. Discussion by the three authors was how decisions were made. Studies were not blinded. Randomized 6 subjects in each of 6 groups Treatment of 3 consecutive HA with Zolmitriptan 1.5 mg The treatment group had significantly Shorter duration of HA (p < 0.001) Less use of rescue drugs not different Less nausea p < Higher patient rating (p < 0.001) Improvement in quality of life score (p < for global, psychological and social, p = for functional. Iatrogenic disturbance was not significant. Only nasal sumatriptan was better for children and adolescents with migraine VAS score was significantly improved for subject on zolmitriptan nasal spray 5 mg (p = 0.0.5) versus 5 other triptans VAS score was reduced to < 3 in one hour for zolmitriptan nasal spray (regression coefficient G, 2.30: standard error, 0.99; Wald, 5.36; d.f =1; p = 0.21 Exp B = 10; 95% Not blinded Study drug compared to many different migraine drugs Wash out period not discussed Statistics could be stronger Could not combine results, since different drugs were used in each study No p values given Randomization is poor Power calculation was not performed They talk about time after onset of HA that medicines are taken, however there is no mention in the study 7
8 Cephalagia,2, Tfelt-Hansen, P., Bach, F.W., Daugaard, D., Tsiropoulos, I., & Riddersholm, B. (2006). Treatment with sumatriptan 50 mg in the mild phase of migraine attacks in patients with infrequent attacks: A randomized doubleblind, placebocontrolled study. Journal of Headache Pain, 7, Dodick, D., Brandes, J., Elkind, A., Mathew, N. & Rodichok, L (2005). Speed of onset, efficacy and tolerability of zolmitriptan nasal spray in the acute treatment of migraine. CNS Drugs 19, 2, Naratriptan 2.5 mg (36.6) Frovatriptan 2.5 mg (33.3) Zolmitriptan nasal spray 5 mg (32.8) Eletriptan 40 mg (31.2) Adults N= receiving placebo; Mean age receiving treatment; Mean age=38 Adults aged N=2122 screened 935 in zolmitriptan group Age distribution > years 43.7% and >40-69 years 44.4 % placebo group n= 933 age distribution years 44.4% and % Oxford: 1b Oxford:1 b Eletriptan 80 mg Naratriptan 2.5 mg Frovatriptan 2.5 mg Zolmitriptan nasal spray 5 mg Eletriptan 40 mg Control: each other Double blind, placebo controlled, parallel group and randomized 50 mg sumatriptan Control: Placebo Multi-center randomized,doubl e blind parallel group Zolmtriptan5 mg nasal spray Control: placebo to treat up to two migraine attacks within 15 minutes of HA pain becoming moderate/severe confidence interval exp G ) 38% of patients in the intent to treat population were pain free after 2 hours. (95 CI: 25%-53%) Results are presented as percentage of subjects with pain relief using a VAS. Zolmitriptan nasal spray produced a signifiacant p < HA response at 2 hours compared with placebo (66.2% vs. 35.0%), (p < 0.001). All prior time points were significantly better in the zolmitriptan group than the placebo group. Secondary outcomes- Zolmitriptan had significantly higher pain free assessment from the earliest assessment (15 minutes) onward (p < ). design giving the directions relating to time after onset of symptoms that the study med should be taken. Confidence intervals were not used on the primary outcomes. No report if the 12 who dropped out just prior to randomization were different than those who stuck with the study. 33% subjects waited until the severity of pain was at least moderate before treatment. Did not exactly follow protocol. Missing values from the HA diaries were problematic; subjects were not used to filling out HA diaries and follow the protocol. Taste of the zolmitriptan potentially un blinded the treatment subjects. 8
9 Burstein, R., Collins, B. & Jakubowski, M. (2004). Defeating migraine pain with triptans: A race against the development of cutaneous allodynia. Annals of Neurology,55, Adults age years old Mean age 36.2 range, patients were evaluated and 31 patients participated. Oxford- 2b cohort study 24 hour wash out period. Cohort study patients who failed to reach a pain free state after late sumatriptan treatment were asked for a fourth visit 4 hours into another attack.. They were treated with rizatriptan 10 mg or melting zolmtriptan 5 mg tablet Time to onset of HA response was greater in the zolmitriptan group (p < ). Zolmtriptan group used less rescue medication (p < 0.001) and less remedication (0.0001) than the placebo group. Significance was achieved for the resolution of nausea and phonophobia from 15 minutes onward and photophobia 30 minutes onward. Global impressions- subjects preferred the zolmitriptan significantly greater than placebo (p < 0.001). Recurrence and time to recurrence were greater in the zolmitriptan group, but not significantly. Subjects were prepared to use zolmitriptan nasal spray as treatment for HA after this trial. AEs were similar in both groups. The results are presented as presence or absence of allodynia and VAS scores for early and late treatment of HA. Patients without allodynia were more likely to be pain free than patients with allodynia. Early triptan treatment successfully yielded a pain free state in 15 of 17 nonallodynic attacks and only 1 of 12 in allodynic attacks (p < 0.001). Late triptan treatment successfully yielded a pain free state in 10 of 10 nonallodynic attacks and 4 of 22 allodynic attacks (p <0.001). In allodynic attacks, triptans decreased throbbing, but did not significantly decrease allodynia. The prospect of achieving complete pain relief with triptans was strongly associated with the absence of allodynia. Within 2 hours of treatment the painfree state was achieved in 93% of attacks without allodynia and only 15% Patients knew they were getting drug potential Hawthorne Effect 9
10 Meredith, J.T., Wait S. & Brewer, K.L. (2003). A prospective double blind study of nasal sumatriptan versus IV ketorolac in migraine. American Journal of Emergency Medicine, 21, Adults received (mean age 33 years (range, 18-54)) IV ketorolac 16 received (mean age349range,19-56)) nasal sumatriptan Convenience sample Oxford:2b RCT no CIs given Prospective double blind randomized comparison study Sumatriptan nasal spray 20 mg or Control: Ketorolac 30 mg IV of attacks with allodynia. Sumatriptan significantly decreased migraine pain one hour after treatment. p < Reduction in VAS from mm before treatment to mm after treatment, power = 80-90% at p < Ketorolac significantly decreased migraine pain one hour after treatment from mm to mm after ketorolac. Both were significant at p < Pain reduction was greater in patients who received IV ketorolac than in those receiving nasal sumatriptan p < 0.05, 95% power at p < Convenience sample No follow up for recurrence of HA, medication side effects outside the ED, only one hour follow up, other studies have had 2 hour follow up. Literature synthesized by: Nancy H Allen MS RD LD CNSC J. Bartlett, RN, MSN, MBA/HCM Clinical Bottom Line developed by Migraine CPG team. Date created: 5/09 References: Ahonen, K., Hämäläinen, M.L,, Rantala H., & Hoppu K. (2004). Nasal sumatriptan is effective in treatment of migraine attacks in children: A randomized trial. Neurology, 62, Burstein, R., Collins, B. & Jakubowski, M. (2004). Defeating migraine pain with triptans: A race against the development of cutaneous allodynia. Annals of Neurology,55, Boureau, F., Chazot, G., Emile, J., Bertin, L., & d Allens, H. (1995). Comparison of subcutaneous sumatriptan with usual acute treatments for migraine. European Neurology 35, Dodick, D., Brandes, J., Elkind, A., Mathew, N. & Rodichok, L (2005). Speed of onset, efficacy and tolerability of zolmitriptan nasal spray in the acute treatment of migraine. CNS Drugs 19, 2, Gunn, V.L, & Nechyba, C. (2002). The Harriet Lane handbook (16 th ed). St. Louis, MO: Mosby. Hämäläinen, M.L., Hoppu, K., & Santavuori P. (1997). Sumatriptan for migraine attacks in children: A randomized placebo controlled study. Neurology, 48, Kelly, A.M., Ardagh, M., Curry, C., D Antonio, J., & Zebic, S. (1997). Intervenous chlorpromazine versus intramuscular sumatriptan for acute migraine. J Accident and Emergency Medicine,14, Lampl, C., Huber, G., Haas, S., Ritteberger, E. & Diener, H.C. (2008). Difference in triptan effect in patients with migraine and early allodynia. Cephalagia,2, Lewis, D.,W., Hershey, A.D., & Wasiewski, W.W. (2007). On behalf of the adolescent migraine steering committee. Pediatrics 120, Major, P.W., Grubisa, H.S.I. & Thie, N.M.R. (2003). Triptans for treatment of acute pediatric migraine: a systematic literature review. Pediatric Neurology, 29,
11 Meredith, J.T., Wait S. & Brewer, K.L. (2003). A prospective double blind study of nasal sumatriptan versus IV ketorolac in migraine. American Journal of Emergency Medicine, 21, Rothner, A.D., Wasiewski,W., Winner, P., Lewis, D. & Stankowski, J.(2006). Zolmitriptan oral tablet in migraine treatments: High placebo responses in adolescents. Headache, 46, Scholpp, J., Schellenberg, R., Moeckesch, B. & Banik, N. (2004). Early treatment of a migraine attack while pain is still mild increased the efficacy of sumatriptan. Cephalagia, 24, Supported by GlaxoSmithKline. Tfelt-Hansen, P., Bach, F.W., Daugaard, D., Tsiropoulos, I., & Riddersholm, B. (2006). Treatment with sumatriptan 50 mg in the mild phase of migraine attacks in patients with infrequent attacks: A randomized double-blind, placebo-controlled study. Journal of Headache Pain, 7, Winner, P., Adelman, J., Aurora, S., Lener, M.E. & Ames, M. (2006). Efficacy and tolerability of sumatriptan injection for the treatment of morning migraine: Two multicenter, prospective, randomized, double-blind, controlled studies in adults. Clinical Therapeutics, 28, Winner, P., Lewis, D.L., Visser, H., Jiang, K., Ahrens, S., & Evans, J.K. (2002), Rizatriptan 5 mg for the acute treatment of migraine in adolescents: A randomized, double-blind, placebo-controlled study. Headache, 42, Winner, P., Rothner, D., Wooten, J., Webster, C. & Ames, M. (2006). Sumatriptan nasal spray in adolescent migraineurs: A randomized, double-blind, placebo-controlled, acute study. Headache, 46,
Disclosures. Triptans for Kids 5/16/13
 5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
Specific Care Question : Question Originator: Plain Language Summary from The Office of Evidence Based Practice: Conditional Recommendation
 Ketorolac for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine is ketorolac an effective treatment? Question Originator: Migraine Therapy
Ketorolac for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine is ketorolac an effective treatment? Question Originator: Migraine Therapy
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Adult & Pediatric Patients. Stanford Health Care, Division Pain Medicine
 Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
...SELECTED ABSTRACTS...
 The following abstracts, from medical journals containing literature on migraine management, were selected for their relevance to this Special Report supplement. Two Sumatriptan Studies Two double-blind
The following abstracts, from medical journals containing literature on migraine management, were selected for their relevance to this Special Report supplement. Two Sumatriptan Studies Two double-blind
Dose: Metoclopramide -0.1 mg/kg (max 10 mg) IV, over 15 minutes
 Metoclopramide for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine, is metoclopramide an effective treatment? Question Originator: Migraine
Metoclopramide for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine, is metoclopramide an effective treatment? Question Originator: Migraine
medications. This was an openlabel study consisting of patients with migraines who historically failed to respond to oral triptan
 J Headache Pain (2007) 8:13 18 DOI 10.1007/s10194-007-0354-7 ORIGINAL Seymour Diamond Fred G. Freitag Alexander Feoktistov George Nissan Sumatriptan 6 mg subcutaneous as an effective migraine treatment
J Headache Pain (2007) 8:13 18 DOI 10.1007/s10194-007-0354-7 ORIGINAL Seymour Diamond Fred G. Freitag Alexander Feoktistov George Nissan Sumatriptan 6 mg subcutaneous as an effective migraine treatment
ARTICLE. Migraine Therapeutics in Adolescents. A Systematic Analysis and Historic Perspectives of Triptan Trials in Adolescents
 COMPARATIVE EFFECTIVENESS RESEARCH ARTICLE Migraine Therapeutics in Adolescents A Systematic Analysis and Historic Perspectives of Triptan Trials in Adolescents Haihao Sun, MD, PhD; Eric Bastings, MD;
COMPARATIVE EFFECTIVENESS RESEARCH ARTICLE Migraine Therapeutics in Adolescents A Systematic Analysis and Historic Perspectives of Triptan Trials in Adolescents Haihao Sun, MD, PhD; Eric Bastings, MD;
ISSN doi: /head VC 2015 American Headache Society Published by Wiley Periodicals, Inc.
 ISSN 0017-8748 Headache doi: 10.1111/head.12746 VC 2015 American Headache Society Published by Wiley Periodicals, Inc. Review Article Acute Treatment Therapies for Pediatric Migraine: A Qualitative Systematic
ISSN 0017-8748 Headache doi: 10.1111/head.12746 VC 2015 American Headache Society Published by Wiley Periodicals, Inc. Review Article Acute Treatment Therapies for Pediatric Migraine: A Qualitative Systematic
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Pharmacological treatment of attacks in juvenile migraine
 J Headache Pain (2004) 5:S62 S66 DOI 10.1007/s10194-004-0110-9 Beatrice Gallai Giovanni Mazzotta Paola Sarchielli Pharmacological treatment of attacks in juvenile migraine Received: Accepted in revised
J Headache Pain (2004) 5:S62 S66 DOI 10.1007/s10194-004-0110-9 Beatrice Gallai Giovanni Mazzotta Paola Sarchielli Pharmacological treatment of attacks in juvenile migraine Received: Accepted in revised
Drug Class Review on Triptans
 Drug Class Review on UPDATED FINAL REPORT #1 December 2003 Mark Helfand, MD, MPH Kim Peterson, MS Oregon Evidence-based Practice Center Oregon Health & Science University Table of Contents Introduction
Drug Class Review on UPDATED FINAL REPORT #1 December 2003 Mark Helfand, MD, MPH Kim Peterson, MS Oregon Evidence-based Practice Center Oregon Health & Science University Table of Contents Introduction
Early treatment of a migraine attack while pain is still mild increases the efficacy of sumatriptan
 Blackwell Science, LtdOxford, UKCHACephalalgia1468-2982Blackwell Science, 20042411925933Original ArticleEarly treatment of migraine with sumatriptanj Scholpp et al. Early treatment of a migraine attack
Blackwell Science, LtdOxford, UKCHACephalalgia1468-2982Blackwell Science, 20042411925933Original ArticleEarly treatment of migraine with sumatriptanj Scholpp et al. Early treatment of a migraine attack
Migraine is an important clinical problem in
 REVIEW ARTICLE Symptomatic Treatment of Migraine in Children: A Systematic Review of Medication Trials Léonie Damen, PhD*; Jacques K. J. Bruijn, MD ; Arianne P. Verhagen, PhD*; Marjolein Y. Berger, MD
REVIEW ARTICLE Symptomatic Treatment of Migraine in Children: A Systematic Review of Medication Trials Léonie Damen, PhD*; Jacques K. J. Bruijn, MD ; Arianne P. Verhagen, PhD*; Marjolein Y. Berger, MD
Drug Class Review on the Triptans
 Drug Class Review on the Final Report March 7, 2003 Expires October 31, 2003 Mark Helfand, MD, MPH Kim Peterson, MS Produced by Oregon Health & Science University 3181 SW Sam Jackson Park Road Mailcode:
Drug Class Review on the Final Report March 7, 2003 Expires October 31, 2003 Mark Helfand, MD, MPH Kim Peterson, MS Produced by Oregon Health & Science University 3181 SW Sam Jackson Park Road Mailcode:
MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache
 MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache Measure Description Percentage of patients age 12 years and older with a diagnosis of migraine who were prescribed a guideline recommended
MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache Measure Description Percentage of patients age 12 years and older with a diagnosis of migraine who were prescribed a guideline recommended
Pharmacy Medical Necessity Guidelines: Migraine Medications
 Pharmacy Medical Necessity Guidelines: Effective: September 18, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Pharmacy Medical Necessity Guidelines: Effective: September 18, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
ADVANCES IN MIGRAINE MANAGEMENT
 ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
Zolmitriptan nasal spray provides fast relief of migraine symptoms and is preferred by patients: a Swedish study of preference in clinical practice
 J Headache Pain (2004) 5:237 242 DOI 10.1007/s10194-004-0132-3 ORIGINAL Carl G.H. Dahlöf Mattias Linde Erika Kerekes Zolmitriptan nasal spray provides fast relief of migraine symptoms and is preferred
J Headache Pain (2004) 5:237 242 DOI 10.1007/s10194-004-0132-3 ORIGINAL Carl G.H. Dahlöf Mattias Linde Erika Kerekes Zolmitriptan nasal spray provides fast relief of migraine symptoms and is preferred
A Review of the Pharmacokinetics, Pharmacodynamics and Effi cacy of Zolmitriptan in the Acute Abortive Treatment of Migraine
 A Review of the Pharmacokinetics, Pharmacodynamics and Effi cacy of Zolmitriptan in the Acute Abortive Treatment of Migraine A.A. Kalanuria 1 and B.L. Peterlin 1,2 REVIEW 1 Drexel University College of
A Review of the Pharmacokinetics, Pharmacodynamics and Effi cacy of Zolmitriptan in the Acute Abortive Treatment of Migraine A.A. Kalanuria 1 and B.L. Peterlin 1,2 REVIEW 1 Drexel University College of
Drug Class Review on Triptans
 Drug Class Review on Triptans Final Report Update 3 November 2005 Original Report Date: March 2003 Update 1 Report Date: December 2003 Update 2 Report Date: September 2004 A literature scan of this topic
Drug Class Review on Triptans Final Report Update 3 November 2005 Original Report Date: March 2003 Update 1 Report Date: December 2003 Update 2 Report Date: September 2004 A literature scan of this topic
Zolmitriptan is effective and well tolerated in Japanese patients with migraine: a dose response study
 is effective and well tolerated in Japanese patients with migraine: a dose response study F Sakai 1, M Iwata 2, K Tashiro 3, Y Itoyama 4, S Tsuji 5, Y Fukuuchi 6, G Sobue 7, K Nakashima 8 & M Morimatsu
is effective and well tolerated in Japanese patients with migraine: a dose response study F Sakai 1, M Iwata 2, K Tashiro 3, Y Itoyama 4, S Tsuji 5, Y Fukuuchi 6, G Sobue 7, K Nakashima 8 & M Morimatsu
Lasmiditan (200 mg and 100 mg) Compared to Placebo for Acute Treatment of Migraine
 (200 mg and 100 mg) Compared to for Acute Treatment of Migraine Bernice Kuca, M.S. 1 ; Linda A. Wietecha, B.S.N., M.S. 2 ; Paul H. Berg, M.S. 2 ; Sheena K. Aurora, M.D. 2 1 CoLucid Pharmaceuticals, Inc.,
(200 mg and 100 mg) Compared to for Acute Treatment of Migraine Bernice Kuca, M.S. 1 ; Linda A. Wietecha, B.S.N., M.S. 2 ; Paul H. Berg, M.S. 2 ; Sheena K. Aurora, M.D. 2 1 CoLucid Pharmaceuticals, Inc.,
Faculty Disclosures. Learning Objectives. Acute Treatment Strategies
 WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
Anti-Migraine Agents
 DRUG POLICY BENEFIT APPLICATION Anti-Migraine Agents Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations or exceptions
DRUG POLICY BENEFIT APPLICATION Anti-Migraine Agents Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations or exceptions
Dex single dose Pred- 5 day course Mean Difference Mean Difference Mean 3.5. Weight 100.0% IV, Fixed, 95% CI [-1.97, 0.37] 100.
![Dex single dose Pred- 5 day course Mean Difference Mean Difference Mean 3.5. Weight 100.0% IV, Fixed, 95% CI [-1.97, 0.37] 100. Dex single dose Pred- 5 day course Mean Difference Mean Difference Mean 3.5. Weight 100.0% IV, Fixed, 95% CI [-1.97, 0.37] 100.](/thumbs/90/101418001.jpg) Question : In the child with asthma exacerbation in the ED should prednisolone/prednisone vs. dexamethasone be used to prevent hospitalization, decrease time in the ED or improve pulmonary function? Forest
Question : In the child with asthma exacerbation in the ED should prednisolone/prednisone vs. dexamethasone be used to prevent hospitalization, decrease time in the ED or improve pulmonary function? Forest
A new questionnaire for assessment of adverse events associated with triptans: methods of assessment influence the results. Preliminary results
 J Headache Pain (2004) 5:S112 S116 DOI 10.1007/s10194-004-0123-4 Michele Feleppa Fred D. Sheftell Luciana Ciannella Amedeo D Alessio Giancarlo Apice Nino N. Capobianco Donato M.T. Saracino Walter Di Iorio
J Headache Pain (2004) 5:S112 S116 DOI 10.1007/s10194-004-0123-4 Michele Feleppa Fred D. Sheftell Luciana Ciannella Amedeo D Alessio Giancarlo Apice Nino N. Capobianco Donato M.T. Saracino Walter Di Iorio
Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial
 J Headache Pain (2007) 8:175 179 DOI 10.1007/s10194-007-0386-7 ORIGINAL Usha Kant Misra Jayantee Kalita Rama Kant Yadav Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial Received:
J Headache Pain (2007) 8:175 179 DOI 10.1007/s10194-007-0386-7 ORIGINAL Usha Kant Misra Jayantee Kalita Rama Kant Yadav Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial Received:
Literature Scan: Triptans
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
OVERVIEW OF COMMENTS RECEIVED ON DRAFT GUIDELINE ON CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS FOR THE TREATMENT OF MIGRAINE
 European Medicines Agency London, 24 January 2007 Doc. Ref. EMEA/CHMP/EWP/21477/2007 OVERVIEW OF COMMENTS RECEIVED ON DRAFT GUIDELINE ON CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS FOR THE TREATMENT OF
European Medicines Agency London, 24 January 2007 Doc. Ref. EMEA/CHMP/EWP/21477/2007 OVERVIEW OF COMMENTS RECEIVED ON DRAFT GUIDELINE ON CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS FOR THE TREATMENT OF
Drug Class Review Triptans
 Drug Class Review Triptans April 2013 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness and harms of different
Drug Class Review Triptans April 2013 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness and harms of different
ONZETRA XSAIL (sumatriptan) nasal powder
 ONZETRA XSAIL (sumatriptan) nasal powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
ONZETRA XSAIL (sumatriptan) nasal powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
Treatment satisfaction with zolmitriptan nasal spray for migraine in a real life setting: results from phase two of the REALIZE study
 J Headache Pain (2005) 6:405 411 DOI 10.1007/s10194-005-0237-3 ORIGINAL Marek Gawel Jürgen Aschoff Arne May Bruce R. Charlesworth on behalf of the REALIZE Study Group Treatment satisfaction with zolmitriptan
J Headache Pain (2005) 6:405 411 DOI 10.1007/s10194-005-0237-3 ORIGINAL Marek Gawel Jürgen Aschoff Arne May Bruce R. Charlesworth on behalf of the REALIZE Study Group Treatment satisfaction with zolmitriptan
Drug Therapy Guidelines
 Drug Therapy Guidelines Applicable Medical Benefit Effective: 5/1/18 Pharmacy- Formulary 1 x Next Review: 3/19 Pharmacy- Formulary 2 x Date of Origin: 8/29/06 Triptans: almotriptan, Amerge, Axert, Frova,
Drug Therapy Guidelines Applicable Medical Benefit Effective: 5/1/18 Pharmacy- Formulary 1 x Next Review: 3/19 Pharmacy- Formulary 2 x Date of Origin: 8/29/06 Triptans: almotriptan, Amerge, Axert, Frova,
UTILIZATION MANAGEMENT CRITERIA
 SUMATRIPTAN (ALSUMA, IMITREX, SUMAVEL DOSEPRO, ZECUITY ) UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: BRAND (generic) NAME: Serotonin 5-HT1 receptor agonists Imitrex (sumatriptan), Alsuma (sumatriptan),
SUMATRIPTAN (ALSUMA, IMITREX, SUMAVEL DOSEPRO, ZECUITY ) UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: BRAND (generic) NAME: Serotonin 5-HT1 receptor agonists Imitrex (sumatriptan), Alsuma (sumatriptan),
SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection
 SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
Patients preference for triptans and other medications as a tool for assessing the efficacy of acute treatments for migraine
 J Headache Pain (2005) 6:112 120 DOI 10.1007/s10194-005-0164-3 REVIEW Andrew J. Dowson Stewart J. Tepper Carl Dahlöf Patients preference for triptans and other medications as a tool for assessing the efficacy
J Headache Pain (2005) 6:112 120 DOI 10.1007/s10194-005-0164-3 REVIEW Andrew J. Dowson Stewart J. Tepper Carl Dahlöf Patients preference for triptans and other medications as a tool for assessing the efficacy
Migranal Nasal Spray. Migranal Nasal Spray (dihydroergotamine) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: June 22, 2017 Migranal Nasal Spray
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: June 22, 2017 Migranal Nasal Spray
The use of combination therapies in the acute management of migraine
 REVIEW The use of combination therapies in the acute management of migraine Abouch Valenty Krymchantowski Headache Center of Rio, Rio de Janeiro, Brazil; Outpatient Headache Unit of the Instituto de Neurologia
REVIEW The use of combination therapies in the acute management of migraine Abouch Valenty Krymchantowski Headache Center of Rio, Rio de Janeiro, Brazil; Outpatient Headache Unit of the Instituto de Neurologia
Migraine treatment in developmental age: guidelines update
 J Headache Pain (2010) 11:267 276 DOI 10.1007/s10194-010-0205-4 TUTORIAL Migraine treatment in developmental age: guidelines update Laura Papetti Alberto Spalice Francesco Nicita Maria Chiara Paolino Rosa
J Headache Pain (2010) 11:267 276 DOI 10.1007/s10194-010-0205-4 TUTORIAL Migraine treatment in developmental age: guidelines update Laura Papetti Alberto Spalice Francesco Nicita Maria Chiara Paolino Rosa
I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation.
 I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation. In 1962, Bille published landmark epidemiologic survey of headache among 9,000 school
I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation. In 1962, Bille published landmark epidemiologic survey of headache among 9,000 school
Despite the widespread use of triptans ... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability
 ... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability Randal L. Von Seggern, PharmD, BCPS Abstract Objective: This article summarizes preclinical and clinical data for
... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability Randal L. Von Seggern, PharmD, BCPS Abstract Objective: This article summarizes preclinical and clinical data for
Financial Support: This study was co-funded by OptiNose US, Inc., Yardley, PA, USA, and Avanir Pharmaceuticals, Inc., Aliso Viejo, CA, USA.
 Headache 2015 The Authors. Headache: The Journal of Head and Face Pain published by Wiley Periodicals, Inc. on behalf of American Headache Society ISSN 0017-8748 doi: 10.1111/head.12583 Published by Wiley
Headache 2015 The Authors. Headache: The Journal of Head and Face Pain published by Wiley Periodicals, Inc. on behalf of American Headache Society ISSN 0017-8748 doi: 10.1111/head.12583 Published by Wiley
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D.
 Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
Acute treatment of migraine in children and adolescents
 Acute treatment of migraine in children and adolescents Cristiano Termine, MD a,b Matteo Ferri, MD a,b Umberto Balottin, MD b,c a Child Neuropsychiatry Unit, Department of Clinical and Biological Sciences,
Acute treatment of migraine in children and adolescents Cristiano Termine, MD a,b Matteo Ferri, MD a,b Umberto Balottin, MD b,c a Child Neuropsychiatry Unit, Department of Clinical and Biological Sciences,
THE WOMAN WHO COULD NOT DECIDE WHICH MEDICATION TO TAKE
 Rapoport Ch 05.qxd 10/15/08 1:06 PM Page 25 CHAPTER 5 THE WOMAN WHO COULD NOT DECIDE WHICH MEDICATION TO TAKE ALLAN PURDY, MD, FRCPC FRED SHEFTELL, MD ALAN RAPOPORT, MD STEWART J. TEPPER, MD Case History
Rapoport Ch 05.qxd 10/15/08 1:06 PM Page 25 CHAPTER 5 THE WOMAN WHO COULD NOT DECIDE WHICH MEDICATION TO TAKE ALLAN PURDY, MD, FRCPC FRED SHEFTELL, MD ALAN RAPOPORT, MD STEWART J. TEPPER, MD Case History
Triptans: Nonresponse, Recurrence, and Serious AEs for Many Patients
 Efficacy, Safety, and Tolerability of Rimegepant 75 mg, an Oral CGRP Receptor Antagonist, for the Acute Treatment of Migraine: Results from a Phase 3, Double-Blind, Randomized, Placebo-Controlled Trial,
Efficacy, Safety, and Tolerability of Rimegepant 75 mg, an Oral CGRP Receptor Antagonist, for the Acute Treatment of Migraine: Results from a Phase 3, Double-Blind, Randomized, Placebo-Controlled Trial,
Maxalt. Maxalt / Maxalt-MLT (rizatriptan) Description. Section: Prescription Drugs Effective Date: April 1, 2016
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Maxalt Page: 1 of 5 Last Review Date: March 18, 2016 Maxalt Description Maxalt / Maxalt-MLT (rizatriptan)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Maxalt Page: 1 of 5 Last Review Date: March 18, 2016 Maxalt Description Maxalt / Maxalt-MLT (rizatriptan)
Regulatory Status FDA approved indication: Migranal Nasal Spray is indicated for the acute treatment of migraine headaches with or without aura (1).
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: November 30, 2018 Migranal Nasal Spray
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: November 30, 2018 Migranal Nasal Spray
Optimizing triptan therapy in clinical practice
 Review Optimizing triptan therapy in clinical practice Carlo Lisotto* & Giorgio Zanchin Practice Points The prevalence of migraine is 12 16% of the general population and is two- to threetimes higher in
Review Optimizing triptan therapy in clinical practice Carlo Lisotto* & Giorgio Zanchin Practice Points The prevalence of migraine is 12 16% of the general population and is two- to threetimes higher in
MIGRAINE UPDATE. Objectives & Disclosures. Learn techniques used to diagnose headaches. Become familiar with medications used for headache treatment.
 MIGRAINE UPDATE Karen L. Bremer, MD November 16, 2018 Objectives & Disclosures Learn techniques used to diagnose headaches. Become familiar with medications used for headache treatment. Disclosure: I am
MIGRAINE UPDATE Karen L. Bremer, MD November 16, 2018 Objectives & Disclosures Learn techniques used to diagnose headaches. Become familiar with medications used for headache treatment. Disclosure: I am
Abortive Agents. Available Strengths. Formulary Limits. Tablet: 5mg, 10mg ODT: 5mg, 10 mg 25mg, 50mg, 100mg. 5mg/act, 20mg/act
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Migraine Therapy P&T DATE: 9/12/2017 CLASS: Neurological Disorders REVIEW HISTORY 12/16, 9/15, 2/15, 2/10, 5/07 LOB: MCL
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Migraine Therapy P&T DATE: 9/12/2017 CLASS: Neurological Disorders REVIEW HISTORY 12/16, 9/15, 2/15, 2/10, 5/07 LOB: MCL
How do we treat migraine? New SIGN Guidelines
 How do we treat migraine? New SIGN Guidelines Managing your migraine Migraine Trust, Edinburgh 2018 Callum Duncan Consultant Neurologist Aberdeen Royal Infirmary Chair SIGN Guideline 155 Premonitory Mood
How do we treat migraine? New SIGN Guidelines Managing your migraine Migraine Trust, Edinburgh 2018 Callum Duncan Consultant Neurologist Aberdeen Royal Infirmary Chair SIGN Guideline 155 Premonitory Mood
Çiçek Wöber-Bingöl HEADACHE UNIT FOR CHILDREN AND ADOLESCENCE
 Headache in children and adolescents Çiçek Wöber-Bingöl HEADACHE UNIT FOR CHILDREN AND ADOLESCENCE Dept. of Psychiatry of Childhood and Adolescence Medical University of Vienna, Vienna, Austria Impact
Headache in children and adolescents Çiçek Wöber-Bingöl HEADACHE UNIT FOR CHILDREN AND ADOLESCENCE Dept. of Psychiatry of Childhood and Adolescence Medical University of Vienna, Vienna, Austria Impact
Update on Diagnosis and Management of Migraines
 Update on Diagnosis and Management of Migraines Joel J. Heidelbaugh, MD, FAAFP, FACG Clinical Professor Departments of Family Medicine and Urology University of Michigan Learning Objectives To distinguish
Update on Diagnosis and Management of Migraines Joel J. Heidelbaugh, MD, FAAFP, FACG Clinical Professor Departments of Family Medicine and Urology University of Michigan Learning Objectives To distinguish
Treatment of Headache in the ED
 Treatment of Headache in the ED Benjamin W. Friedman, MD, MS, FAAEM Associate professor of Emergency Medicine Albert Einstein College of Medicine Montefiore Medical Center Disclosure Topics of Discussion
Treatment of Headache in the ED Benjamin W. Friedman, MD, MS, FAAEM Associate professor of Emergency Medicine Albert Einstein College of Medicine Montefiore Medical Center Disclosure Topics of Discussion
Neurologic Disorders: Headache in Children
 1 of 12 17/06/2014 7:57 PM Print Close Neurologic Disorders: Headache in Children Sharon Whiting, MBBS, FRCPC Date of revision: February 2014 Headaches occur commonly in children and adolescents. They
1 of 12 17/06/2014 7:57 PM Print Close Neurologic Disorders: Headache in Children Sharon Whiting, MBBS, FRCPC Date of revision: February 2014 Headaches occur commonly in children and adolescents. They
Clinical Learning Days November 10, 2017
 Migraine Clinical Learning Days November 10, 2017 Alyssa Lettich. MD Neurosciences Institute/Neurosciences Clinical Program Medical Director Headache Disclosures: none Learning Objectives: At the conclusion
Migraine Clinical Learning Days November 10, 2017 Alyssa Lettich. MD Neurosciences Institute/Neurosciences Clinical Program Medical Director Headache Disclosures: none Learning Objectives: At the conclusion
The Importance of Non-Oral Therapies for Acute Migraine: Addressing Patient Needs
 1 The Importance of Non-Oral Therapies for Acute Migraine: Addressing Patient Needs David W. Dodick, MD Professor Department of Neurology Mayo Clinic Phoenix, Arizona 2 Presentation Objectives Describe
1 The Importance of Non-Oral Therapies for Acute Migraine: Addressing Patient Needs David W. Dodick, MD Professor Department of Neurology Mayo Clinic Phoenix, Arizona 2 Presentation Objectives Describe
Headache Questionnaire
 Date: All Headache Patients We would appreciate your cooperation in filling out this form. In our evaluation of headache, your history is typically our most valuable tool for diagnosis and subsequent treatment.
Date: All Headache Patients We would appreciate your cooperation in filling out this form. In our evaluation of headache, your history is typically our most valuable tool for diagnosis and subsequent treatment.
Page: 1 of 5. Sumatriptan Tablets and Nasal Spray (Imitrex) / sumatriptan and naproxen sodium (Treximet tablets)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 0 Subject: Sumatriptan (Imitrex / Treximet) Page: 1 of 5 Last Review Date: September 12, 2014 Sumatriptan
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 0 Subject: Sumatriptan (Imitrex / Treximet) Page: 1 of 5 Last Review Date: September 12, 2014 Sumatriptan
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME / GENERIC DRUG NAME: Advil / Ibuprofen
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME / GENERIC DRUG NAME: Advil / Ibuprofen
Paediatric headaches. Dr Jaycen Cruickshank Director of Clinical Training Ballarat Health Services. Brevity, levity, repetition
 Paediatric headaches Dr Jaycen Cruickshank Director of Clinical Training Ballarat Health Services Brevity, levity, repetition Paediatric)headache?)! Headache!in!children!is!not!that!common.!The!question!is!which!headaches!do!I!
Paediatric headaches Dr Jaycen Cruickshank Director of Clinical Training Ballarat Health Services Brevity, levity, repetition Paediatric)headache?)! Headache!in!children!is!not!that!common.!The!question!is!which!headaches!do!I!
Zomig. Zomig / Zomig-ZMT (zolmitriptan) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.22 Subject: Zomig Page: 1 of 5 Last Review Date: November 30, 2018 Zomig Description Zomig / Zomig-ZMT
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.22 Subject: Zomig Page: 1 of 5 Last Review Date: November 30, 2018 Zomig Description Zomig / Zomig-ZMT
NARAMIG QUALITATIVE AND QUANTITATIVE COMPOSITION
 NARAMIG Naratriptan QUALITATIVE AND QUANTITATIVE COMPOSITION Tablets containing 2.5 mg of naratriptan as naratriptan hydrochloride. PHARMACEUTICAL FORM Film coated tablets. CLINICAL PARTICULARS Indications
NARAMIG Naratriptan QUALITATIVE AND QUANTITATIVE COMPOSITION Tablets containing 2.5 mg of naratriptan as naratriptan hydrochloride. PHARMACEUTICAL FORM Film coated tablets. CLINICAL PARTICULARS Indications
Zomig. Zomig / Zomig-ZMT (zolmitriptan) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.22 Subject: Zomig Page: 1 of 5 Last Review Date: March 16, 2018 Zomig Description Zomig / Zomig-ZMT
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.22 Subject: Zomig Page: 1 of 5 Last Review Date: March 16, 2018 Zomig Description Zomig / Zomig-ZMT
The best defense is a good offense. Optimizing the Acute Treatment of Migraine. Disclosures 11/10/2017
 Optimizing the Acute Treatment of Migraine Brian M. Plato, DO, FAHS Norton Neuroscience Institute Louisville, KY Disclosures Speakers Bureau (personal): Allergan, Depomed, Avanir Research Funding (paid
Optimizing the Acute Treatment of Migraine Brian M. Plato, DO, FAHS Norton Neuroscience Institute Louisville, KY Disclosures Speakers Bureau (personal): Allergan, Depomed, Avanir Research Funding (paid
Value of postmarketing surveillance studies in achieving a complete picture of antimigraine agents: using almotriptan as an example
 J Headache Pain (2006) 7:27 33 DOI 10.1007/s10194-006-0266-6 ORIGINAL Julio Pascual Hans-Christoph Diener Hélène Massiou Value of postmarketing surveillance studies in achieving a complete picture of antimigraine
J Headache Pain (2006) 7:27 33 DOI 10.1007/s10194-006-0266-6 ORIGINAL Julio Pascual Hans-Christoph Diener Hélène Massiou Value of postmarketing surveillance studies in achieving a complete picture of antimigraine
Drug Class Review Triptans
 Drug Class Review Triptans Preliminary Update Scan #3 February 2014 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness
Drug Class Review Triptans Preliminary Update Scan #3 February 2014 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness
Acute Migraine Treatment in Adults
 PL Detail-Document #301104 This PL Detail-Document gives subscribers additional insight related the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER November 2014 Acute Migraine Treatment
PL Detail-Document #301104 This PL Detail-Document gives subscribers additional insight related the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER November 2014 Acute Migraine Treatment
Vestibular Migraine. Information for patients and carers. Department of Neurology and Otolaryngology Aberdeen Royal Infirmary
 Vestibular Migraine Information for patients and carers Department of Neurology and Otolaryngology Aberdeen Royal Infirmary What is vestibular migraine? Migraine is a disabling headache disorder that affects
Vestibular Migraine Information for patients and carers Department of Neurology and Otolaryngology Aberdeen Royal Infirmary What is vestibular migraine? Migraine is a disabling headache disorder that affects
Eletriptan vs sumatriptan: A double-blind, placebo-controlled, multiple migraine attack study
 Eletriptan vs sumatriptan: A double-blind, placebo-controlled, multiple migraine attack study G. Sandrini, M. Färkkilä, G. Burgess, et al. Neurology 2002;59;1210-1217 DOI 10.1212/WNL.59.8.1210 This information
Eletriptan vs sumatriptan: A double-blind, placebo-controlled, multiple migraine attack study G. Sandrini, M. Färkkilä, G. Burgess, et al. Neurology 2002;59;1210-1217 DOI 10.1212/WNL.59.8.1210 This information
Sumatriptan Injection (Imitrex / Alsuma / Sumavel / Zembrace)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.11 Subject: Sumatriptan Injection Page: 1 of 6 Last Review Date: March 16, 2018 Sumatriptan Injection
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.11 Subject: Sumatriptan Injection Page: 1 of 6 Last Review Date: March 16, 2018 Sumatriptan Injection
Sumatriptan Tablets, Nasal Spray (Imitrex), Nasal Powder (Onzetra Xsail), sumatriptan and naproxen sodium (Treximet tablets)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 0 Subject: Sumatriptan Page: 1 of 6 Last Review Date: November 30, 2018 Sumatriptan Description Sumatriptan
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 0 Subject: Sumatriptan Page: 1 of 6 Last Review Date: November 30, 2018 Sumatriptan Description Sumatriptan
UPDATE IN MIGRAINE MANAGEMENT
 UPDATE IN MIGRAINE MANAGEMENT Eric P. Baron, DO Cleveland Clinic Neurological Institute Center for Neurological Restoration Headache and Chronic Pain Medicine barone2@ccf.org @Neuralgroover Disclosures
UPDATE IN MIGRAINE MANAGEMENT Eric P. Baron, DO Cleveland Clinic Neurological Institute Center for Neurological Restoration Headache and Chronic Pain Medicine barone2@ccf.org @Neuralgroover Disclosures
HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in
 HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in Anesthesia and Neurology Harvard Medical School Limited time
HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in Anesthesia and Neurology Harvard Medical School Limited time
What A Headache! Theresa Biesiada March 8, 2012
 What A Headache! Theresa Biesiada March 8, 2012 Objectives Describe the EM relevance of headaches and migraines Discuss the rationale for steroid therapy Review the evidence Conclusions My inspiration
What A Headache! Theresa Biesiada March 8, 2012 Objectives Describe the EM relevance of headaches and migraines Discuss the rationale for steroid therapy Review the evidence Conclusions My inspiration
Migraineurs have specific preferences with regard to migraine therapy. In surveys,
 Patient Treatment Preferences and the 5-HT 1B/1D Agonists Robert E. Ryan, Jr, MD REVIEW ARTICLE Migraineurs have specific preferences with regard to migraine therapy. In surveys, they consistently cite
Patient Treatment Preferences and the 5-HT 1B/1D Agonists Robert E. Ryan, Jr, MD REVIEW ARTICLE Migraineurs have specific preferences with regard to migraine therapy. In surveys, they consistently cite
Sumatriptan Tablets, Nasal Spray (Imitrex), Nasal Powder (Onzetra Xsail), sumatriptan and naproxen sodium (Treximet tablets)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 05.70.10 Subject: Sumatriptan Page: 1 of 6 Last Review Date: March 16, 2018 Sumatriptan Description Sumatriptan
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 05.70.10 Subject: Sumatriptan Page: 1 of 6 Last Review Date: March 16, 2018 Sumatriptan Description Sumatriptan
Sumatriptan Tablets, Nasal Spray (Imitrex), Nasal Powder (Onzetra Xsail), sumatriptan and naproxen sodium (Treximet tablets)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 05.70.10 Subject: Sumatriptan Page: 1 of 5 Last Review Date: December 2, 2016 Sumatriptan Description Sumatriptan
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 05.70.10 Subject: Sumatriptan Page: 1 of 5 Last Review Date: December 2, 2016 Sumatriptan Description Sumatriptan
Migraine is a common and disabling illness, affecting
 original Research Sumatriptan/Naproxen Sodium as Early Intervention for Migraine: Effects on Functional Ability, Productivity, and Satisfaction in 2 Randomized Controlled Trials Frederick R. Taylor, MD,
original Research Sumatriptan/Naproxen Sodium as Early Intervention for Migraine: Effects on Functional Ability, Productivity, and Satisfaction in 2 Randomized Controlled Trials Frederick R. Taylor, MD,
Guidance for Industry Migraine: Developing Drugs for Acute Treatment
 Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
A case of a patient with chronic headache. Focus on Migraine. None related to the presentation Grants to conduct clinical trials from: Speaker bureau:
 Chronic Daily Headache Bassel F. Shneker, MD, MBA Associate Professor Vice Chair, OSU Neurology The Ohio State University Wexner Medical Center Financial Disclosures None related to the presentation Grants
Chronic Daily Headache Bassel F. Shneker, MD, MBA Associate Professor Vice Chair, OSU Neurology The Ohio State University Wexner Medical Center Financial Disclosures None related to the presentation Grants
Children s Mercy Hospitals and Clinics Evidence Based Practice Clinical Practice Guide
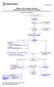 Children s Mercy Hospitals and Clinics Evidence Based Practice Clinical Practice Guide Migraine in the ED/UCC 1 Definition: Acute migraine is a primary headache disorder. It is periodic in nature. Migraine
Children s Mercy Hospitals and Clinics Evidence Based Practice Clinical Practice Guide Migraine in the ED/UCC 1 Definition: Acute migraine is a primary headache disorder. It is periodic in nature. Migraine
Migraine: Developing Drugs for Acute Treatment Guidance for Industry
 Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
Evaluating the triptans
 The American Journal of Medicine (2005) Vol 118, Suppl 1, 28S 35S Evaluating the triptans Ninan T. Mathew, MD, a Elizabeth W. Loder, MD b a From the Houston Headache Clinic, and Department of Neurology,
The American Journal of Medicine (2005) Vol 118, Suppl 1, 28S 35S Evaluating the triptans Ninan T. Mathew, MD, a Elizabeth W. Loder, MD b a From the Houston Headache Clinic, and Department of Neurology,
Migraine Acute treatment
 Migraine Acute treatment Elizabeth Loder, MD, MPH Professor of Neurology, Harvard Medical School Chief, Division of Headache, Department of Neurology, Brigham and Women s Hospital, Boston, MA Disclosures
Migraine Acute treatment Elizabeth Loder, MD, MPH Professor of Neurology, Harvard Medical School Chief, Division of Headache, Department of Neurology, Brigham and Women s Hospital, Boston, MA Disclosures
Quality Design Limitations Inconsistency Indirectness Imprecision studies. Other. considerations Heliox Control (95% Absolute
 Question 7: In the child with asthma exacerbation in the ED, should heliox driven albuterol vs. oxygen or room air driven albuterol be used to prevent hospitalization, decrease time in the ED or improve
Question 7: In the child with asthma exacerbation in the ED, should heliox driven albuterol vs. oxygen or room air driven albuterol be used to prevent hospitalization, decrease time in the ED or improve
10/17/2017 CHRONIC MIGRAINES BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES PATIENT CASE EPIDEMIOLOGY EPIDEMIOLOGY
 BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017
BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017
TABLE 1. Current Diagnostic Criteria for Migraine Without Aura 2 A. At least 5 attacks fulfilling criteria B-D B. Headache attacks lasting 4-72 hours
 ANSWERS CONCISE TO FREQUENTLY REVIEW ASKED QUESTIONS FOR CLINICIANS ABOUT MIGRAINE Answers to Frequently Asked Questions About Migraine IVAN GARZA, MD, AND JERRY W. SWANSON, MD Migraine is a common primary
ANSWERS CONCISE TO FREQUENTLY REVIEW ASKED QUESTIONS FOR CLINICIANS ABOUT MIGRAINE Answers to Frequently Asked Questions About Migraine IVAN GARZA, MD, AND JERRY W. SWANSON, MD Migraine is a common primary
An Economic Evaluation of Triptan Products for Migraine
 Blackwell Science, LtdOxford, UKVHEValue in Health1098-30152005 ISPOR86647655Original ArticleEconomic Evaluation of TriptansPerfetto et al. Volume 8 Number 6 2005 VALUE IN HEALTH An Economic Evaluation
Blackwell Science, LtdOxford, UKVHEValue in Health1098-30152005 ISPOR86647655Original ArticleEconomic Evaluation of TriptansPerfetto et al. Volume 8 Number 6 2005 VALUE IN HEALTH An Economic Evaluation
Treatment of Primary Headache Syndromes
 Presenter Disclosure Information 2:45 3:45pm Treatment of Primary Headache Syndromes SPEAKER Gerald W. Smetana, MD The following relationships exist related to this presentation: Gerald W.Smetana, MD,
Presenter Disclosure Information 2:45 3:45pm Treatment of Primary Headache Syndromes SPEAKER Gerald W. Smetana, MD The following relationships exist related to this presentation: Gerald W.Smetana, MD,
Faculty Disclosure. Karen L. Bremer, MD. Dr. Bremer has listed no financial interest/arrangement that would be considered a conflict of interest.
 Faculty Disclosure Karen L. Bremer, MD Dr. Bremer has listed no financial interest/arrangement that would be considered a conflict of interest. HEADACHE UPDATE Karen L. Bremer, MD November 10, 2017 karen.bremer@creighton.edu
Faculty Disclosure Karen L. Bremer, MD Dr. Bremer has listed no financial interest/arrangement that would be considered a conflict of interest. HEADACHE UPDATE Karen L. Bremer, MD November 10, 2017 karen.bremer@creighton.edu
ACUTE MIGRAINE: OLD AND NEW DRUGS JOHN ROBROCK MD FORT WILLIAM FAMILY HEALTH TEAM
 ACUTE MIGRAINE: OLD AND NEW DRUGS JOHN ROBROCK MD FORT WILLIAM FAMILY HEALTH TEAM Conflict of Interest Declaration: Nothing to Disclose Presenter: John Robrock, MD Title of Presentation: Acute Migraine:
ACUTE MIGRAINE: OLD AND NEW DRUGS JOHN ROBROCK MD FORT WILLIAM FAMILY HEALTH TEAM Conflict of Interest Declaration: Nothing to Disclose Presenter: John Robrock, MD Title of Presentation: Acute Migraine:
Study No.:MPX Title: Rationale: Phase: IIB Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Migraine Management. Roger Cady, MD Headache Care Center Springfield, MO
 Migraine Management Roger Cady, MD Headache Care Center Springfield, MO Disclosures Objectives The evolution of migraine From benign episodic (benign) headache to potentially a devastating chronic disease
Migraine Management Roger Cady, MD Headache Care Center Springfield, MO Disclosures Objectives The evolution of migraine From benign episodic (benign) headache to potentially a devastating chronic disease
Management of headache
 Management of headache TJ Steiner Imperial College London Based on European principles of management of common headache disorders TJ Steiner, K Paemeleire, R Jensen, D Valade, L Savi, MJA Lainez, H-C Diener,
Management of headache TJ Steiner Imperial College London Based on European principles of management of common headache disorders TJ Steiner, K Paemeleire, R Jensen, D Valade, L Savi, MJA Lainez, H-C Diener,
10/31/2017 PRIMARY CARE AND HEADACHE DISCLOSURES WHERE DO THOSE WITH HEADACHE SEEK MEDICAL CARE? Primary Care 67%
 PRIMARY CARE AND HEADACHE Sonja Potrebic MD PhD Regional Headache Specialist Kaiser LAMC 1 WHERE DO THOSE WITH HEADACHE SEEK MEDICAL CARE? Column1 Primary Care 67% Primary Care Headache Specialty Other
PRIMARY CARE AND HEADACHE Sonja Potrebic MD PhD Regional Headache Specialist Kaiser LAMC 1 WHERE DO THOSE WITH HEADACHE SEEK MEDICAL CARE? Column1 Primary Care 67% Primary Care Headache Specialty Other
Model-based quantification of the relationship between age and anti-migraine therapy
 6 Model-based quantification of the relationship between age and anti-migraine therapy HJ Maas, M Danhof, OE Della Pasqua Submitted to BMC. Clin. Pharmacol. Migraine is a neurological disease that affects
6 Model-based quantification of the relationship between age and anti-migraine therapy HJ Maas, M Danhof, OE Della Pasqua Submitted to BMC. Clin. Pharmacol. Migraine is a neurological disease that affects
