Migraine treatment in developmental age: guidelines update
|
|
|
- Noel Stevens
- 6 years ago
- Views:
Transcription
1 J Headache Pain (2010) 11: DOI /s TUTORIAL Migraine treatment in developmental age: guidelines update Laura Papetti Alberto Spalice Francesco Nicita Maria Chiara Paolino Rosa Castaldo Paola Iannetti Maria Pia Villa Pasquale Parisi Received: 5 January 2010 / Accepted: 28 February 2010 / Published online: 27 March 2010 Ó Springer-Verlag 2010 Abstract There is a serious lack of controlled studies on the pharmacological treatment of primary migraine in the developmental age; there is, consequently, an urgent need for new, evidence-based approaches to this long-neglected field of research. Moreover, previous studies have stated that the placebo response is greater in pediatric patients than in adults and that a reduction in the attack frequency in the absence of any pharmacological treatment is observed more frequently in pediatric migraine patients than in adults. Besides these preliminary considerations, the shorter duration of migraine attacks and other characteristic semeiological features of the clinical picture in children are such that the design of randomized controlled trial (RCT) is more problematic in the developmental age than in the adult. Bearing in mind all these weak points, the aim of this review was to summarize and update recent guidelines for the treatment of primary migraine in children and adolescents. The most recent guidelines are those published by the Italian Society for the study of Headache, the French Society for the study of Migraine and Headache, and the American Academy of Neurology. We have incorporated into these guidelines the results from the few, recent RCTs, clinical controlled trials, open-label studies, meta-analyses and reviews that have been published since 2004; owing to the lack of strong evidence in this field of L. Papetti A. Spalice F. Nicita P. Iannetti Child Neurology, Paediatric Department, I Faculty of Medicine, Sapienza University, c/o Policlinico Umberto I, Rome, Italy M. C. Paolino R. Castaldo M. P. Villa P. Parisi (&) Child Neurology, Headache Paediatric Center, Paediatric Sleep Disorders Center, Chair of Paediatrics, II Faculty of Medicine, Sapienza University, c/o Sant Andrea Hospital, Via di Grottarossa, Rome, Italy pasquale.parisi@uniroma1.it; parpas@iol.it research, we have sometimes even mentioned pilot noncontrolled studies, case series and expert opinions. Lastly, evidence was classified and the recommendations were categorized according to different levels. Keywords Pediatric migraine Prophylaxis Pharmacological treatment Guidelines Evidence-based medicine Acute treatment Migraine attack Introduction Migraine is a frequent condition in children, with a prevalence ranging from 3% in younger school-age children to approximately 20% in older adolescents [1]. The current classification system for primary migraine (Table 1) was published by the International Headache Society in 2004 and is known as the International Classification of Headache Disorders (ICHD-II) [2]. The ICHD-II for the diagnosis of pediatric migraine has been shown to have a sensitivity of 84% [3]. One of the limitations related to the use of ICHD-II criteria in the diagnosis of pediatric migraine is the difficulty children encounter in describing the features of headache and any associated symptoms. Moreover, as the features of headache may change from preschool age to adolescence [4], some authors have suggested modifying the criteria for headache diagnosis in childhood [5]. Despite the high frequency of migraine and the severity of attacks in the pediatric population, there is a serious lack of controlled data available in the literature on migraine therapy. Few randomized placebo-controlled trials on acute-phase or preventive drugs have yielded successful results in pediatric headache patients. Moreover, the high
2 268 J Headache Pain (2010) 11: Table 1 International headache society classification of migraine (ICHD-II) (1) Migraine without aura (2) Migraine with aura a. Typical aura with migraine headache b. Typical aura with non-migraine headache c. Typical aura without headache d. Familial hemiplegic migraine e. Sporadic hemiplegic migraine f. Basilar-type migraine (3) Childhood periodic syndromes that are commonly precursors of migraine a. Cyclic vomiting b. Abdominal migraine c. Benign paroxysmal vertigo of childhood (4) Retinal migraine (5) Complications of migraine (6) Probable migraine placebo response in some trials makes it difficult to prove the efficacy of the drug being studied [6]. The most recent practice guidelines for the therapy of pediatric migraine were published by the Italian Society for the study of Headache (SISC, 2003), the French Society for the study of Migraine and Headache (2004) and the American Academy of Neurology (AAN, 2004) [7 9]. The aims of this review were to update these guidelines [7 9], incorporating the results from randomized controlled trials (RCTs), clinical controlled trials (CCTs), open-label (OL) studies, meta-analyses and reviews published on this hot topic since Owing to the lack of strong evidence in this field of research, in certain cases we also mentioned pilot non-controlled studies, case series and expert opinions. Lastly, findings regarding pharmacological therapies were reviewed and the recommendations were categorized into different levels (A C) (Table 2) [10]. Literature search strategy In December 2009, an extensive search was undertaken to identify currently available headache treatment guidelines Table 2 Levels of evidence for the treatment of migraine Level A: two or more clinically controlled, randomized studies carried out according to good clinical practice (GCP), versus placebo or versus active treatment of proven efficacy Level B: one clinically controlled, randomized study carried out according to GCP or more than one well-designed clinical case control study or cohort study Level C: favorable judgment of two-third of the Ad Hoc Committee members, historical controls, non-randomized studies, case reports (acute phase and prophylaxis). Moreover, we identified any RCTs, CCTs, OL studies, meta-analyses, reviews and articles that were either published or in press in the last 6 years (from 2004 to 2009) using the keywords migraine and treatment or therapy (293 papers), migraine and prophylaxis (66 papers) and migraine and guidelines (4 papers). Any relevant papers published in the same period that were not selected by these keywords were also taken into account. PubMed was used as the source for our search, which was not subjected to any language restriction, and any citations found in relevant studies were investigated. Our search was limited to the pediatric age group (0 18 years), although any articles that included adults but contained a high proportion of persons under the age of 18 years were considered. The only headache exclusion criteria adopted were the words symptomatic or secondary. After the search and selection of the data was completed, two reviewers independently extracted the data, while a third reviewer solved any discrepancies. The search identified 42 studies. One reviewer scanned these to eliminate any irrelevant studies. Three reviewers scanned the remaining 25 studies and the 30 relevant citations identified within these studies. Pediatric migraine treatment: general approach Appropriate treatment for children with migraine requires an individually tailored strategy, based on both pharmacological and non-pharmacological measures, defined according to the degree of disability caused by the headache [11]. The choice of therapy must be based on the symptoms, response to treatment and headache impact, which reflects an individual patient s frequency, duration, intensity, functional disability, quality of life, comorbidity and pain tolerance [12]. The impact of migraine can be assessed in adults by using the migraine disability assessment (MIDAS) questionnaire [13]. However, since the MIDAS is not suitable for children because their lifestyles differ from those of adults, the pediatric migraine disability assessment (Ped- MIDAS) was developed. The range of the scoring system, which is based on the patient s disability, is wider than that of the MIDAS because children are more likely to omit activities. PedMIDAS can be used clinically to identify the impact of migraine in individual pediatric patients and assess their response to treatment [14]. The MIDAS questionnaire can also be used to stratify adult patients into groups with different treatment needs according to the degree of headache-related disability. Stratified care was developed as an alternative to the stepcare approach, which is instead preferred in the treatment of pediatric migraine, and places patients on non-specific
3 J Headache Pain (2010) 11: medication that is gradually increased until effective relief is achieved [15]. The pharmacological treatment of pediatric migraine includes: acute or episodic medications and prophylactic or preventive agents [12]. Acute treatments of migraine The aim of acute treatment should be a rapid response, resulting in a prompt return to normal activity and no relapse. To date, the only treatments that have been approved for migraine attacks in adolescents are almotriptan, by the US Food and Drugs Administration (FDA), and nasal sumatriptan and zolmitriptan, by the European Medicines Agency (EMEA) [19]. Analgesics Ibuprofen is effective and should be considered for the acute treatment of migraine in children (Level A) [7 9]. Acetaminophen is probably effective and should be considered for the acute treatment of migraine in children (Level B) [7 9]. Ibuprofen (10 mg/kg) and acetaminophen (15 mg/kg) were more effective than placebo; the effect yielded by ibuprofen at 2 h was better than that of acetaminophen (68 vs. 54% of children relieved) and lasted longer. Neither drug had significant side effects [11, 17]. Recently Evers et al. [18] found that ibuprofen mg was more effective than placebo in providing pain relief after 2 h (28% for placebo vs. 69% for ibuprofen; P \ 0.05). The efficacy and safety of other non-steroid antiinflammatory drugs (NSAIDs) (e.g., acetylsalicylic acid, diclofenac, naproxen, mefenamic acid) in the treatment of migraine in children and adolescents have not yet been assessed [11]. Triptans The family of triptans has improved the treatment of acute migraine in adults and several studies have demonstrated the safety of such drugs in children [1]. In 2009, the FDA approved almotriptan for the treatment of adolescent migraine, while nasal sumatriptan and zolmitriptan were approved for the acute treatment of adolescent migraine by the EMEA [19]. Sumatriptan Sumatriptan nasal spray (NS) is effective and should be considered for the acute treatment of migraine in adolescents (Level A) [7 9]. Callenbach et al. [20] published a review of all controlled studies that evaluated the efficacy and safety of sumatriptan NS in the pediatric migraine population. Their review included seven trials, most of which included children aged years as their study subjects. Intranasal sumatriptan was effective in relieving headache after both 1 and 2 h in all the studies, the response rates being comparable to those found in studies on adults. Four of the studies detected a significant difference in relief between sumatriptan and placebo (Table 3). Moreover, sumatriptan NS revealed a positive effect on vomiting and photophobia/ phonophobia. The main side effect was bad taste. Effective doses are 10 mg for children weighing \40 kg and 20 mg for children weighing [40 kg [11, 17, 21]. The most recent RCT was conducted by Winner et al. on 738 adolescent subjects (mean age 14 years) treated with sumatriptan NS (5 and 20 mg). Sumatriptan NS 20 mg provided significantly greater headache relief than placebo at 30 min (42 vs. 33%, respectively; P = 0.046) and 2 h (68 vs. 58%; P = 0.025) post-dose, but did not reach statistical significance at either 1 h (61 vs. 52%; P = 0.087) or for sustained headache relief from 1 to 24 h (P = 0.061). Significant differences (P \ 0.05) in favor of sumatriptan NS 20 mg over placebo were observed for several secondary efficacy endpoints, including sustained relief from 2 to 24 h [21]. No data are available to support or refute the use of any oral or subcutaneous sumatriptan preparations in children or adolescents (Level U) [7 9] because such preparations have yielded less positive data owing to their prominent side and placebo effects [11, 17]. Zolmitriptan Studies in which oral zolmitriptan was compared with placebo have yielded contrasting results [11, 21, 22]. Since 2004, two large studies evaluating the efficacy and tolerability of oral zolmitriptan in pediatric migraine have been published [22, 23]. The first compared zolmitriptan at three different doses (2.5, 5, and 10 mg) with placebo in 640 outpatients aged years. Two-hour headache response rates were 54% (10 mg), 53% (5 mg) and 57% (2.5 mg) for zolmitriptan, and 58% for placebo. Two-hour pain-free rates were 25% (10 mg), 19% (5 mg), and 23% (2.5 mg) for zolmitriptan, and 20% for placebo. Zolmitriptan was well tolerated, with a tolerability profile similar to that observed in adults. The authors hypothesized that the similar efficacy observed in zolmitriptan and placebo may be the result of the high placebo response rate and the shorter duration of headache pain in adolescents if compared with adults [22]. Evers et al. conducted a double-blind, placebo-controlled, cross-over study in which patients received placebo, zolmitriptan 2.5 mg, and ibuprofen mg, each being
4 270 J Headache Pain (2010) 11: Table 3 Symptomatic drugs evaluated in placebo-controlled and open clinical trials Drug Evidence level Study design Ages (years) n Primary end point: pain relief (hours) Responders (%) Placebo responses (%) P value Author/Pubdate/Ref. Ibuprofen 10 mg/kg A rdbpc \0.05 Hamalainen et al., 1997 [10, 16] 7.5 mg/kg DBPC Lewis et al., 2002 [10, 16] mg DBPCCO \0.05 Evers et al., 2006 [21] Acetaminophen 15 mg/kg B rdbpc \0.05 Hamalainen et al., 1997 [10, 16] Sumatriptan nasal 20 mg A rdbpc Ueberall et al., 1999 [18] mg rdbpc \0.05 Winner et al., 2000 [18] mg rdbpcco Ahonen et al., 2004 [18] 20 mg rdbpc ns Winner et al., 2006 [19] Sumatriptan oral mg C rdbpcco ns Hamalainen et al., 1997 [10, 16] Sumatriptan subcutaneous 3 6 mg C OL MacDonald, 1994 [10, 16] 0.06 mg/kg OL Linder, 1996 [10, 16] Zolmitriptan oral mg C OL Linder et al., 2000 [10, 16] 2.5 mg DBPCCO P \ 0.05 Evers et al., 2006 [21] mg rdbpc ns Rothner et al., 2006 [20] Zolmitriptan NS 5 mg B SB-DBPC P \ 0.05 Lewis et al., 2007 [22] Rizatriptan oral 5 mg C rdbpc ns Winner et al., 2002 [10, 16] 5 mg rdbpc ns Visser et al., 2004 [23] 5 mg OL Visser et al., 2004 [23] 5 10 mg rdbpc P = Ahonen et al., 2006 [24] Almotriptan oral mg B OL Charles et al., 2006 [25] rdbpc P \ Linder et al., 2008 [26] 25 mg Eletriptan oral 40 mg C DBPC ns Winner et al., 2007 [27] Naratriptan oral C DBPC ns Rothner et al., 1997 [10, 16] DBPC double-blind placebo-controlled, DBPCCO double-blind placebo-controlled crossover, rdbpc randomized double-blind placebo controlled, HA headache, OL open-label, RR retrospective review used to treat one of three consecutive migraine attacks. Pain relief rates after 2 h were 28% for placebo, 62% for zolmitriptan, and 69% for ibuprofen (placebo vs. zolmitriptan P \ 0.05; placebo vs. ibuprofen P \ 0.05). Both drugs were well tolerated, only mild side effects being reported [23]. The 2-h headache response rates for zolmitriptan were similar to those observed in the previous study, while the placebo rate was lower. A possible reason for the low placebo rate might be that children and adolescents with severe migraine were enrolled in the second study. If compared with the other triptan trials in which the patients characteristics were similar, the duration and frequency of attacks in the study by Evers et al. [23] were higher, resembling those observed in adulthood migraine.
5 J Headache Pain (2010) 11: Lewis et al. conducted a novel, placebo-challenge study to evaluate the efficacy and tolerability of zolmitriptan NS in adolescent migraine. A total of 248 adolescents was studied in a randomized, double-blind, placebo-controlled, two-attack, cross-over trial with a single-blind placebo challenge. Seventy-seven patients responded to the placebo challenge and did not thus continue the study. Of the remaining 171 patients, zolmitriptan produced significantly higher headache response rates than placebo 1 h post-dose (58.1 vs. 43.3%; P \ 0.02), with an onset of action occurring as early as 15 min. Zolmitriptan also produced a significant pain-free response at 1 h (27.7 vs. 10.2% in the placebo group, P \ 0.001), proving to be significantly superior to placebo in improving pain intensity, pain-free rates, sustained resolution of headache and resolution of associated migraine symptoms. Treatment with zolmitriptan NS was well tolerated [24]. Rizatriptan Two recent RCTs compared the efficacy of oral rizatriptan with placebo [25, 26]. Visser et al. evaluated the short- and long-term efficacy and tolerability of rizatriptan 5 mg in adolescents with migraine. They conducted an rdbpc single-attack study followed by a randomized, 1-year, OL extension, as well as a randomized 1-year OL study. In the single-attack study, the proportion of patients in whom pain was relieved at 2 h was not significantly different in the rizatriptan 5 mg (68.2%) and placebo (68.8%) groups. In the multiple attack studies, pain relief at 2 h was achieved in a significantly higher number of attacks treated with a rizatriptan 5-mg tablet (77%) or with a rizatriptan 5-mg wafer (77%) than by means of standard care (64%). Rizatriptan 5 mg was well tolerated in adolescents during both short- and longterm use [25]. The second study investigated the efficacy of oral rizatriptan (5 and 10 mg) and the consistency of the response over two treated migraine attacks in children and adolescents over 6 years of age. Two doses of rizatriptan and a matching placebo were administered at home during three attacks. Headache relief at 2 h was achieved twice as often after each rizatriptan treatment (first 74%; second 73%) as after placebo (36%) (P = 0.001). The efficacy of rizatriptan was constant over the two treated attacks, while the findings were similar regardless of the dose administered (5 and 10 mg). No serious adverse effects were observed. The efficacy and the tolerance of rizatriptan in children below 12 years of age did not differ from those observed in adolescents [26]. Almotriptan Two studies investigated the efficacy and safety of almotriptan in adolescents with migraine [27, 28]. The first is a small OL placebo-controlled trial. Fifteen patients aged years with a history of migraine were treated with almotriptan at doses ranging from 6.25 to 12.5 mg. Almotriptan was effective in 13 patients, with no significant adverse effects being reported [27]. The FDA approval of almotriptan for the acute treatment of migraine headache in adolescents was based on data from a recent randomized placebo-controlled trial. Linder et al. assessed the efficacy and safety of almotriptan 6.25, 12.5, and 25 mg versus placebo for acute migraine treatment in adolescents (866 patients aged years). The 2-h painrelief rates were significantly higher with almotriptan 6.25 mg (71.8%), 12.5 mg (72.9%), and 25 mg (66.7%) than with placebo (55.3%; P = 0.001, P \ and P = 0.028, respectively). Age group sub-analysis demonstrated significantly greater 2-h pain-relief rates for patients aged years, a significantly lower incidence of photophobia and phonophobia 2 h after almotriptan 12.5 mg administration in patients aged years than in the placebo group, and a significantly lower incidence of photophobia following almotriptan 12.5 mg administration in patients aged years than in the placebo group [28]. Eletriptan A multicenter, double-blind, parallel-group, placebo-controlled trial compared 40 mg of oral eletriptan with placebo for the treatment of migraine in adolescent patients aged years. Drug efficacy was evaluated in patients 2 h post-dose. There was no significant difference in the 2 h headache response between eletriptan 40 mg and placebo (57 vs. 57%), nor was any significant improvement observed in any of the outcomes 1 or 2 h post-dose. By contrast, eletriptan 40 mg proved to be significantly more effective in reducing headache recurrence than placebo within 24 h (11 vs. 25%, P = 0.028), with post hoc analyses showing statistically significant differences for sustained headache response rates (52 vs. 39%; P = 0.04) and sustained painfree response rates (22 vs. 10%; P = 0.013) [29]. Antiemetics There is one recent randomized-controlled study on pediatric migraine therapy in the emergency room that compared the efficacy of prochlorperazine with that of ketorolac. In that study, prochlorperazine was found to be
6 272 J Headache Pain (2010) 11: more effective than ketorolac as regards the primary outcome, which was a decrease in the intensity of the migraine by 50% or complete relief within 1 h: 28/33 (85%) versus 16/29 (55%), respectively [30]. The drugs and acute-phase treatment outcome in pediatric migraine patients are shown in Table 3. Prophylactic treatment of pediatric migraine Prophylactic pharmacological treatment may be considered when headache frequency exceeds three or four episodes per month and/or the attacks are so severe and prolonged that they interfere with school or normal activities. The goals of prophylactic therapy include: reducing attack frequency, severity and duration, improving responsiveness to treatment of acute attacks, improving function and quality of life, and reducing disability [31]. Calcium channel blockers Flunarizine The Italian, French and AAN practice parameters on the management of pediatric migraine (2004) and the Cochrane review (2003) suggest that the calcium channel blocker, flunarizine, is likely to be effective as preventive therapy and may be considered for this purpose (Level b) [9, 32]. Although it is not available in the United States, flunarizine has yielded good results in several controlled trials and has a proven efficacy (5 mg bedtime dose) in reducing headache frequency and headache duration. The main side effects are daytime sedation and weight gain [10, 33]. Antidepressants Amitriptyline Although the efficacy of amitriptyline in pediatric migraine has never been assessed in RCTs, it remains one of the most widely used agents. Starting doses of 5 10 mg at bedtime may gradually be increased to 1 mg/(kg day). Cardiac abnormalities and depression represent a contraindication for the use of this drug [1, 11, 31]. Antiepileptic drugs Topiramate, valproate, levetiracetam and gabapentin may play increasingly important roles in pediatric migraine. In the light of current views on the pathophysiology of migraine, particularly as regards the primary neuronal initiation and propagation through cortical excitation, and subsequent spreading depression, anticonvulsants may play an intriguing, though not yet fully defined, role [33]. Topiramate Recent studies have shown that topiramate is effective in reducing headache frequency and disability in adolescents (Table 4) [34 39]. Since 2004, three double-blind placebo controlled trials [34 37], one pooled analysis of RCTs [38] and one retrospective review have been published [39]. Winner et al. [35] conducted a placebo-controlled trial in 157 children with migraine aged from 6 to 15 years who were randomized in a 2:1 ratio to receive either topiramate [2 mg/(kg day)] or placebo. The primary outcome was the change in the mean number of migraine days per month. Topiramate treatment was associated with a mean reduction of 2.6 migraine days per month as compared with 2.0 in the placebo group. The difference between the two groups marginally missed the significance level (P = 0.06). Response to topiramate (i.e., [50% reduction in migraine frequency) was 55 versus 47% in the placebo group. The most common adverse effects were upper respiration tract infection, anorexia, weight decrease, gastroenteritis, paresthesia and somnolence [35]. Winner et al. [38] performed a second study in which data from subjects aged years who had participated in three previous RCTs with a similar design were pooled. All three previous studies had compared topiramate at a dose of 100 or 200 mg/day with placebo. The primary outcome measure was the median percentage reduction in the monthly migraine frequency. Topiramate at a dose of 100 and 200 mg/day was associated with a statistically significant reduction in migraine frequency when compared with placebo (respectively 63 and 65 vs. 16% for placebo, P = 0.02 for topiramate 100 mg/day and P = 0.04 for topiramate 200 mg/day). Topiramate at a dose of 50 mg/day was less effective (46% reduction, P = 0.07) [38]. Lewis et al. evaluated the efficacy and safety of topiramate (50 or 100 mg/day) for migraine prevention in 85 adolescents (12 17 years of age). The primary efficacy measure was the reduction in the number of monthly migraine attacks. Topiramate at a dose of 100 mg/day, though not 50 mg/day, resulted in a statistically significant reduction in the monthly migraine attack rate if compared with the placebo group (median 72.2 vs. 44.4%). Topiramate at a dose of 100 mg/day also resulted in a statistically significant reduction in the number of monthly migraine days if compared with the placebo group (83 vs. 45% for placebo) [37]. A third double-blind placebo-controlled trial was conducted in 44 children with migraine. Twenty-two patients
7 J Headache Pain (2010) 11: Table 4 continued Drug Evidence level Study design Ages (years) n Drug response rate (%) Placebo response rate (%) P value (primary endpoint) Reference Flunarizine 5 mg A DBPC P \ Sorge et al., 1985 [1, 10] (freq and durat) 5 mg OL Guidetti et al., 1987 [1, 10] 5 mg DBPC CO Nimodipine P \ (freq) P \ 0.01 (durat) Sorge et al., 1988 [1, 10] mg C DBPCCO ns (freq) Battistella et al., 1990 [1, 10] Propanolol mg C DBCO P \ (freq) Ludvigsson et al., 1974 [1, 10] 80 mg DBPC ns Forsythe et al [1, 10] 3 mg/kg DBPC ns ns ns Olness et al., 1987 [1, 10] Timolol C DBPC ns Noronha et al [1, 10] CO Clonidine lg C DBPC \ ns Sillanpaa, 1977 [1, 10] mg DBPC ns ns ns Sills et al., 1982 [1, 10] Cyproheptadine 4 mg C RR Lewis et al., 2004 [32] Amitriptyline 1 mg/kg C OL Hershey et al., 2000 [1, 10] 10 mg RR Lewis et al., 2004 [32] Trazodone 1 mg/(kg day) C DBPC ns Battistella et al., 1993 [1, 10] Pizotifen C DBPC ns Gilles et al., 1986 [1, 10] CO Topiramate mg A OL P \ (freq) Hershey et al., 2002 [1, 10] 2 3 mg/kg rdbpc ns Winner et al., 2005 [35] 50, 100, 200 mg DBPC P = 0.02 (100 mg), P = 0.04 (200 mg) (freq) Winner et al., 2006 [38] 100 mg DBPC P = 0.02 (freq) Lakshmi et al., 2007 [36] 100 mg rdbpc P \ (freq) Lewis et al., 2009 [37] mg RR P \ (freq) Cruz et al., 2009 [39] Valproate mg/ B OL P \ 0.05 (freq) Caruso et al., 2000 [1, 10] (kg day) 500 1,000 mg/ OL P = (freq) Serdaroglu et al., 2002 [1, 10] day 250 1,125 mg/ OL P \ 0.05 (freq) Pekalnis et al., 2001 [1, 10] day mg/ rdbpc P \ 0.05 (freq) Ashrafi et al., 2005 [40] (kg day) 250, 500, rdbpc ns Apostol et al., 2008 [41] 1,000 mg 500 1,000 mg OL [75% (freq) Apostol et al., 2009 [42] Levetiracetam B OL P \ (freq) Miller et al., 2004 [1, 10] 250 1,500 mg OL P \ (freq) Pekalnis et al., 2007 [44]
8 274 J Headache Pain (2010) 11: Table 4 Prophylactic drugs evaluated in placebo-controlled and open clinical trials Drug Evidence level Study design Ages (years) n Drug response rate (%) Placebo response rate (%) P value (primary endpoint) Reference Gabapentin 15 mg/kg C RR P \ (freq) Belman et al., 2001 [1, 10] Zonisamide 5.8 mg/kg C OL Pakalnis et al., 2006 [45] DBPC double-blind placebo-controlled, DBPCCO double-blind placebo-controlled crossover, rdbpc randomized double-blind placebo controlled, OL open-label, RR retrospective review, freq attack frequency, durat. attack duration received topiramate (final dose of 100 mg) while 22 received placebos. There was a statistically significant decrease in the mean monthly migraine frequency in the topiramate group as compared with the placebo group (11.9 vs. 5.9 days, P = 0.025). Ninety-five percent of the subjects in the topiramate group were classified as responders (i.e., [50% reduction in migraine frequency) as compared with 52% in the placebo group (P = 0.002). Moreover, topiramate was associated with a lower degree of functional disability than placebo. The authors reported a statistically significant difference between the two groups in both the decrease in the PedMIDAS score (P = 0.003) and in school absenteeism (P = 0.002) [36]. Cruz et al. retrospectively reviewed the records of 37 patients (mean age 14 years) with migraine. An excellent or good response (i.e., [50% migraine reduction) with topiramate was attained in 28 patients (76%). Ten (27%) patients exhibited adverse effects [39]. Valproic acid Open-label and retrospective studies [1, 10] have suggested that valproic acid may be effective in the prevention of migraine in children and adolescents. Ashrafi et al. compared the effect of sodium valproate [10 40 mg/(kg day)] in pediatric migraine prophylaxis with that of propranolol in an RCT that included 120 children. Both drugs significantly reduced the mean migraine frequency if compared with the pre-randomization period by 5.1 migraine days/month. Response (i.e., [50% reduction in migraine frequency) was observed in 72% of the valproate group and in 69% of the propranolol group. Furthermore, both drugs were found to effectively reduce the severity and duration of headache and to improve the response to rescue medications (P value \0.01). There was no significant difference between the two groups in any of the aforementioned therapeutic effects (P value \0.05) [40]. To compare the efficacy, tolerability and safety of three different doses of divalproex sodium extended-release (DVPX ER) with placebo, Apostol et al. conducted a randomized double-blind placebo-controlled trial. The study included 299 adolescents aged years. The participants were randomized in a 1:1:1:1 ratio to receive DVPX ER 250, 500, or 1,000 mg once daily or placebo. The median reduction in the migraine rate was slight and similar in all four groups in the study (1.7 migraine days in the placebo, 250 and 1,000 mg divalproex groups, and 1.4 in the 500 mg group). Response (i.e., [50% reduction in migraine frequency) was 46% in the placebo group as compared with 41, 36 and 51% in the DVPX ER 250 mg, 500 or 1,000 mg groups, respectively. DVPX ER was not superior to placebo in the migraine-related quality of life measures either, as assessed by the PedMIDAS [41]. A fourth OL study was conducted on 241 adolescents aged years with migraine. The DVPX ER dose range was 250 1,000 mg daily. Efficacy was based on the number of migraine headache days reported in the subjects headache diaries over sequential 4-week periods for the duration of the trial. DVPX ER treatment was associated with a 75% decrease (from 4.0 to 1.0) in the median number of headache days over a 4-week period between the first and the fourth months of the study [42]. Levetiracetam Since 2004, two OL studies have been conducted to evaluate the efficacy and safety of levetiracetam in pediatric migraine [43, 44]. The first study is a retrospective study in which levetiracetam was assessed in 19 patients (mean age 12 years) at doses of mg t.i.d. The mean frequency of headache attacks, which was 6.3/month before treatment, fell to 1.7/month after treatment (P \ ). The migraine attacks disappeared in 52% of the patients during the treatment, though 10.5% discontinued the treatment owing to side effects [43]. The second OL study included 20 pediatric patients (6 17 years). Levetiracetam dosages ranged from 20 to 40 mg/(kg day). The primary outcome measure was response (i.e., C50% reduction in monthly headache frequency). Eighteen of the 20 patients responded positively to levetiracetam, with a reduction of over 50% reduction in
9 J Headache Pain (2010) 11: the monthly headache frequency (90% response rate) and a significant difference after treatment. The PedMIDAS revealed a significant decrease in the disability scores following levetiracetam treatment. The drug was well tolerated by the participants; the only adverse effects reported being irritability, aggressiveness and mild memory problems (15%) [44]. Zonisamide A retrospective chart review was conducted to study the efficacy of zonisamide in pediatric migraine prophylaxis. Twelve patients were identified (mean age 13.5 years). Eight patients responded positively to zonisamide, with a reduction of over 50% in the number of headaches if compared with the pretreatment values. Zonisamide was well tolerated, with only two patients reporting adverse effects that consisted in weight loss and behavioral changes [45]. According to our search, no RCTs, CCTs or OL studies designed to assess the efficacy and safety of antidepressants (amitriptyline and trazodone), b-blockers (propranolol and timolol) and other antihypertensive agents, such as clonidine, have been conducted on adolescent and childhood migraine since 2004 (Table 4). The evidence levels of all the aforementioned prophylactic drugs are shown in Table 4. Conclusions Few advances have been made in the treatment of pediatric migraine since As regards the acute phase treatment, prospects are unfortunately not very promising; the only drugs that are effective and safe in children being ibuprofen (Level A), acetaminophen (Level B), and sumatriptan NS (Level A). Zolmitriptan NS, which has recently been investigated, proved to be more effective than placebo (Level B). Almotriptan was approved by the FDA for the acute treatment of adolescent migraine even more recently (Table 3). As regards prophylactic treatment, topiramate has recently displayed a good level of efficacy, safety and tolerance (Level A), while flunarizine (Level A) and valproate (Level B) were already included in the previously published guidelines [7 9]. Further researches are warranted to shed more light on the underlying mechanisms of migraine and provide new treatment options designed to raise the efficacy and safety levels of currently available drugs. Moreover, additional RCTs and more controlled data are needed to help clinicians choose the most appropriate drugs for the treatment of this common clinical problem. In this regard, new and innovative study designs are required to minimize the high placebo response observed in the pediatric population. Acknowledgments This Tutorial partly results from the lesson of the Author (PP) at the Master in Headache Medicine, Sapienza University of Rome, during the academic year ( uniroma1.it/headache). This Academic Educational activity is done under the supervision of Lifting The Burden WHO Global Campaign against Headache Working Group for Specialist Education (SEWG) ( Conflict of interest References None. 1. Lewis D (2009) Pediatric migraine. Neurol Clin 27: Olesen J, Steiner TJ (2004) The international classification of headache disorders. Cephalalgia 24: Hershey AD, Winner P, Kabbouche MA et al (2005) Use of the ICHD-II criteria in the diagnosis of pediatric migraine. Headache 45: Pakalnis A (2007) Current therapies in childhood and adolescent migraine. J Child Neurol 22: Winner P (2008) Pediatric headache. Curr Opin Neurol 21: Evers S, Marziniak M, Frese A et al (2009) Placebo efficacy in childhood and adolescence migraine: an analysis of double-blind and placebo-controlled studies. Cephalalgia 29(4): Società Italiana per lo Studio delle Cefalee (SISC) (2003) Linee guida per la diagnosi e per la terapia della cefalea giovanile. Anno V-S1 8. Géraud G, Lantéri-Minet M, Lucas C et al (2004) French guidelines for the diagnosis and management of migraine in adults and children. French Society for the Study of Migraine Headache (SFEMC). Clin Ther 26(8): Lewis D, Ashwal S, Hershey A et al (2004) Practice parameter: pharmacological treatment of migraine headache in children and adolescence. Neurology 63: Ad Hoc Committee (2001) Diagnostic, therapeutic guidelines for migraine, cluster headache. J Headache Pain 2(3): Balottin U, Termine C (2007) Recommendations for the management of migraine in paediatric patients. Expert Opin Pharmacother 8(6): Gunner KB, Smith HD, Ferguson LE (2008) Practice guideline for diagnosis and management of migraine headaches in children and adolescents: part two. J Pediatr Health Care 22(1): Edmeads J, Láinez JM, Brandes JL et al (2001) Potential of the migraine disability assessment (MIDAS) questionnaire as a public health initiative and in clinical practice. Neurology 56(6 Suppl 1):S29 S Hershey AD, Powers SW, Vockell AL et al (2001) PedMIDAS: development of a questionnaire to assess disability of migraines in children. Neurology 57(11): Lipton RB, Silberstein SD (2001) The role of headache-related disability in migraine management: implications for headache treatment guidelines. Neurology 56(6 Suppl 1):S35 S Lewis DW, Winner P (2006) The pharmacological treatment options for pediatric migraine: an evidence-based appraisal. NeuroRx 3(2): Silver S, Gano D, Gerretsen P (2008) Acute treatment of paediatric migraine: a meta-analysis of efficacy. J Paediatr Child Health 44(1 2):3 9
10 276 J Headache Pain (2010) 11: Evers S, Rahmann A, Kraemer C et al (2006) Treatment of childhood migraine attacks with oral zolmitriptan and ibuprofen. Neurology 67(3): Hershey AD (2010) Current approaches to the diagnosis and management of pediatric migraine. Lancet Neurol 9: Callenbach PM, Pels LP, Mulder PG et al (2007) Sumatriptan nasal spray in the acute treatment of migraine in adolescents and children. Eur J Paediatr Neurol 11(6): Winner P, Rothner AD, Wooten JD (2006) Sumatriptan nasal spray in adolescent migraineurs: a randomized, double-blind, placebo-controlled, acute study. Headache 46(2): Rothner AD, Wasiewski W, Winner P et al (2006) Zolmitriptan oral tablet in migraine treatment: high placebo response in adolescents. Headache 46: Evers S, Rahmann A, Kraemer C et al (2006) Treatment of childhood migraine attacks with oral zolmitriptan and ibuprofen. Neurology 67: Lewis DW, Winner P, Hershey AD et al (2007) Adolescent migraine steering committee. Efficacy of zolmitriptan nasal spray in adolescent migraine. Pediatrics 120: Visser WH, Winner P, Strohmaier K et al (2004) Rizatriptan 5 mg for the acute treatment of migraine in adolescents: results from a double-blind, single-attack study and two open-label, multiple-attack studies. Headache 44(9): Ahonen K, Hamalainem ML, Eerola M (2006) A randomized trial of rizatriptan in migraine attacks in children. Neurology 67: Charles JA (2006) Almotriptan in the acute treatment of migraine in patients years old: an open-label pilot study of efficacy and safety. J Headache Pain 7(2): Linder SL, Mathew NT, Cady RK et al (2008) Efficacy and tolerability of almotriptan in adolescents: a randomized, doubleblind, placebo controlled trial. Headache 48(9): Winner P, Linder S, Lipton RB et al (2007) Eletriptan for the acute treatment of migraine in adolescents: results of a doubleblind, placebo-controlled trial. Headache 47(4): Brousseau DC, Duffy SJ, Anderson AC, Linakis JG (2004) Treatment of pediatric migraine headaches: a randomized, double-blind trial of prochlorperazine versus ketorolac. Ann Emerg Med 43: Eiland LS, Jekins LS, Durham SH et al (2007) Pediatric migraine: pharmacologic agents for prophylaxis. Pediatrics 41: Lewis D, Diamond S, Scott D (2004) Prophylactic treatment of pediatric migraine. Headache 44: Parisi P (2009) Who s still afraid of the link between headache and epilepsy? Some reactions to and reflections on the article by Marte Helene Bjørk and co-workers. J Headache Pain 10(5): Cuvellier JC, Riquet A, Vallée L (2008) Antiepileptic drugs in pediatric migraine. Arch Pediatr 15(11): Winner P, Pearlman EM, Linder SL et al (2005) Topiramate pediatric migraine study investigators. Topiramate for migraine prevention in children: a randomized, double-blind, placebocontrolled trial. Headache 45: Lakshmi CV, Singhi P, Malhi P et al (2007) Topiramate in the prophylaxis of pediatric migraine: a double-blind placebo-controlled trial. Child Neurol 22: Lewis D, Winner P, Saper J et al (2009) Randomized, doubleblind, placebo-controlled study to evaluate the efficacy and safety of topiramate for migraine prevention in pediatric subjects 12 to 17 years of age. Pediatrics : Winner P, Gendolla A, Stayer C et al (2006) Topiramate for migraine prevention in adolescents: a pooled analysis of efficacy and safety. Headache 46: Cruz MJ, Valencia I, Legido A et al (2009) Efficacy and tolerability of topiramate in pediatric migraine. Pediatr Neurol 41(3): Ashrafi MR, Shabanian R, Zamani GR et al (2005) Sodium valproate versus propranolol in paediatric migraine prophylaxis. Eur J Paediatr Neurol 9: Apostol G, Cady RK, Laforet GA, Robieson WZ et al (2008) Divalproex extended-release in adolescent migraine prophylaxis: results of a randomized, double-blind, placebo-controlled study. Headache 48(7): Apostol G, Lewis DW, Laforet GA et al (2009) Divalproex sodium extended-release for the prophylaxis of migraine headache in adolescents: results of a stand-alone, long-term open-label safety study. Headache 49(1): Miller GS (2004) Efficacy and safety of levetiracetam in pediatric migraine. Headache 44: Pakalnis A, Kring D, Meier L (2007) Levetiracetam prophylaxis in pediatric migraine an open-label study. Headache 47(3): Pakalnis A, Kring D (2006) Zonisamide prophylaxis in refractory pediatric headache. Headache 46(5):
MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache
 MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache Measure Description Percentage of patients age 12 years and older with a diagnosis of migraine who were prescribed a guideline recommended
MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache Measure Description Percentage of patients age 12 years and older with a diagnosis of migraine who were prescribed a guideline recommended
Disclosures. Triptans for Kids 5/16/13
 5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
Pharmacological treatment of attacks in juvenile migraine
 J Headache Pain (2004) 5:S62 S66 DOI 10.1007/s10194-004-0110-9 Beatrice Gallai Giovanni Mazzotta Paola Sarchielli Pharmacological treatment of attacks in juvenile migraine Received: Accepted in revised
J Headache Pain (2004) 5:S62 S66 DOI 10.1007/s10194-004-0110-9 Beatrice Gallai Giovanni Mazzotta Paola Sarchielli Pharmacological treatment of attacks in juvenile migraine Received: Accepted in revised
I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation.
 I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation. In 1962, Bille published landmark epidemiologic survey of headache among 9,000 school
I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation. In 1962, Bille published landmark epidemiologic survey of headache among 9,000 school
Is Topiramate Effective in Preventing Pediatric Migraines?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 Is Topiramate Effective in Preventing
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 Is Topiramate Effective in Preventing
Management of headache
 Management of headache TJ Steiner Imperial College London Based on European principles of management of common headache disorders TJ Steiner, K Paemeleire, R Jensen, D Valade, L Savi, MJA Lainez, H-C Diener,
Management of headache TJ Steiner Imperial College London Based on European principles of management of common headache disorders TJ Steiner, K Paemeleire, R Jensen, D Valade, L Savi, MJA Lainez, H-C Diener,
How do we treat migraine? New SIGN Guidelines
 How do we treat migraine? New SIGN Guidelines Managing your migraine Migraine Trust, Edinburgh 2018 Callum Duncan Consultant Neurologist Aberdeen Royal Infirmary Chair SIGN Guideline 155 Premonitory Mood
How do we treat migraine? New SIGN Guidelines Managing your migraine Migraine Trust, Edinburgh 2018 Callum Duncan Consultant Neurologist Aberdeen Royal Infirmary Chair SIGN Guideline 155 Premonitory Mood
ARTICLE. Migraine Therapeutics in Adolescents. A Systematic Analysis and Historic Perspectives of Triptan Trials in Adolescents
 COMPARATIVE EFFECTIVENESS RESEARCH ARTICLE Migraine Therapeutics in Adolescents A Systematic Analysis and Historic Perspectives of Triptan Trials in Adolescents Haihao Sun, MD, PhD; Eric Bastings, MD;
COMPARATIVE EFFECTIVENESS RESEARCH ARTICLE Migraine Therapeutics in Adolescents A Systematic Analysis and Historic Perspectives of Triptan Trials in Adolescents Haihao Sun, MD, PhD; Eric Bastings, MD;
Faculty Disclosures. Learning Objectives. Acute Treatment Strategies
 WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
Neurologic Disorders: Headache in Children
 1 of 12 17/06/2014 7:57 PM Print Close Neurologic Disorders: Headache in Children Sharon Whiting, MBBS, FRCPC Date of revision: February 2014 Headaches occur commonly in children and adolescents. They
1 of 12 17/06/2014 7:57 PM Print Close Neurologic Disorders: Headache in Children Sharon Whiting, MBBS, FRCPC Date of revision: February 2014 Headaches occur commonly in children and adolescents. They
Adult & Pediatric Patients. Stanford Health Care, Division Pain Medicine
 Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
Cinnarizine versus Topiramate in Prophylaxis of Migraines among Children and Adolescents: A Randomized, Double-Blind Clinical Trial
 original ARTICLE Cinnarizine versus Topiramate in Prophylaxis of Migraines among Children and Adolescents: A Randomized, Double-Blind Clinical Trial How to Cite This Article: Ashrafi MR, Najafi Z, Shafiei
original ARTICLE Cinnarizine versus Topiramate in Prophylaxis of Migraines among Children and Adolescents: A Randomized, Double-Blind Clinical Trial How to Cite This Article: Ashrafi MR, Najafi Z, Shafiei
MEASURE #3: PREVENTIVE MIGRAINE MEDICATION PRESCRIBED Headache
 MEASURE #3: PREVENTIVE MIGRAINE MEDICATION PRESCRIBED Headache Measure Description Percentage of patients age 18 years old and older diagnosed with migraine headache whose migraine frequency is 4 migraine
MEASURE #3: PREVENTIVE MIGRAINE MEDICATION PRESCRIBED Headache Measure Description Percentage of patients age 18 years old and older diagnosed with migraine headache whose migraine frequency is 4 migraine
10/17/2017 CHRONIC MIGRAINES BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES PATIENT CASE EPIDEMIOLOGY EPIDEMIOLOGY
 BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017
BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017
ADVANCES IN MIGRAINE MANAGEMENT
 ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
Çiçek Wöber-Bingöl HEADACHE UNIT FOR CHILDREN AND ADOLESCENCE
 Headache in children and adolescents Çiçek Wöber-Bingöl HEADACHE UNIT FOR CHILDREN AND ADOLESCENCE Dept. of Psychiatry of Childhood and Adolescence Medical University of Vienna, Vienna, Austria Impact
Headache in children and adolescents Çiçek Wöber-Bingöl HEADACHE UNIT FOR CHILDREN AND ADOLESCENCE Dept. of Psychiatry of Childhood and Adolescence Medical University of Vienna, Vienna, Austria Impact
Value of postmarketing surveillance studies in achieving a complete picture of antimigraine agents: using almotriptan as an example
 J Headache Pain (2006) 7:27 33 DOI 10.1007/s10194-006-0266-6 ORIGINAL Julio Pascual Hans-Christoph Diener Hélène Massiou Value of postmarketing surveillance studies in achieving a complete picture of antimigraine
J Headache Pain (2006) 7:27 33 DOI 10.1007/s10194-006-0266-6 ORIGINAL Julio Pascual Hans-Christoph Diener Hélène Massiou Value of postmarketing surveillance studies in achieving a complete picture of antimigraine
Inpatient Treatment of Status Migraine With Dihydroergotamine in Children and Adolescents
 Headache 2008 the Authors Journal compilation 2008 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2008.01293.x Published by Wiley Periodicals, Inc. Brief Communication Inpatient Treatment
Headache 2008 the Authors Journal compilation 2008 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2008.01293.x Published by Wiley Periodicals, Inc. Brief Communication Inpatient Treatment
HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in
 HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in Anesthesia and Neurology Harvard Medical School Limited time
HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in Anesthesia and Neurology Harvard Medical School Limited time
ISSN doi: /head VC 2015 American Headache Society Published by Wiley Periodicals, Inc.
 ISSN 0017-8748 Headache doi: 10.1111/head.12746 VC 2015 American Headache Society Published by Wiley Periodicals, Inc. Review Article Acute Treatment Therapies for Pediatric Migraine: A Qualitative Systematic
ISSN 0017-8748 Headache doi: 10.1111/head.12746 VC 2015 American Headache Society Published by Wiley Periodicals, Inc. Review Article Acute Treatment Therapies for Pediatric Migraine: A Qualitative Systematic
Zonisamide for migraine prophylaxis in refractory patients
 Thomas Jefferson University Jefferson Digital Commons Department of Neurology Faculty Papers Department of Neurology March 2006 Zonisamide for migraine prophylaxis in refractory patients Avi Ashkenazi
Thomas Jefferson University Jefferson Digital Commons Department of Neurology Faculty Papers Department of Neurology March 2006 Zonisamide for migraine prophylaxis in refractory patients Avi Ashkenazi
A case of a patient with chronic headache. Focus on Migraine. None related to the presentation Grants to conduct clinical trials from: Speaker bureau:
 Chronic Daily Headache Bassel F. Shneker, MD, MBA Associate Professor Vice Chair, OSU Neurology The Ohio State University Wexner Medical Center Financial Disclosures None related to the presentation Grants
Chronic Daily Headache Bassel F. Shneker, MD, MBA Associate Professor Vice Chair, OSU Neurology The Ohio State University Wexner Medical Center Financial Disclosures None related to the presentation Grants
Update on Diagnosis and Management of Migraines
 Update on Diagnosis and Management of Migraines Joel J. Heidelbaugh, MD, FAAFP, FACG Clinical Professor Departments of Family Medicine and Urology University of Michigan Learning Objectives To distinguish
Update on Diagnosis and Management of Migraines Joel J. Heidelbaugh, MD, FAAFP, FACG Clinical Professor Departments of Family Medicine and Urology University of Michigan Learning Objectives To distinguish
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 CGRP Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
CGRP Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
...SELECTED ABSTRACTS...
 The following abstracts, from medical journals containing literature on migraine management, were selected for their relevance to this Special Report supplement. Two Sumatriptan Studies Two double-blind
The following abstracts, from medical journals containing literature on migraine management, were selected for their relevance to this Special Report supplement. Two Sumatriptan Studies Two double-blind
References
 References 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. Bigal ME, Lipton RB. The prognosis of migraine. Curr Opin Neurol 2008; 21:301 308. Olesen J, Headache Classification Subcommittee.
References 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. Bigal ME, Lipton RB. The prognosis of migraine. Curr Opin Neurol 2008; 21:301 308. Olesen J, Headache Classification Subcommittee.
Triptans Quantity Limit Program Summary
 Triptans Quantity Limit Program Summary FDA APPROVED INDICATIONS AND DOSAGE 1-13,14,23,24 Agents Amerge (naratriptan) 1, 2.5 tablets Axert (almotriptan) 6.25, 12.5 tablets migraine attacks with/without
Triptans Quantity Limit Program Summary FDA APPROVED INDICATIONS AND DOSAGE 1-13,14,23,24 Agents Amerge (naratriptan) 1, 2.5 tablets Axert (almotriptan) 6.25, 12.5 tablets migraine attacks with/without
Pharmacy Medical Necessity Guidelines: Migraine Medications
 Pharmacy Medical Necessity Guidelines: Effective: September 18, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Pharmacy Medical Necessity Guidelines: Effective: September 18, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Ishaq Abu Arafeh Consultant Paediatrician Royal Hospital for Children, Glasgow Forth Valley Royal Hospital, Larbert
 Ishaq Abu Arafeh Consultant Paediatrician Royal Hospital for Children, Glasgow Forth Valley Royal Hospital, Larbert Childhood headache: Is it really difficult to manage? It shouldn t be... But it can be...
Ishaq Abu Arafeh Consultant Paediatrician Royal Hospital for Children, Glasgow Forth Valley Royal Hospital, Larbert Childhood headache: Is it really difficult to manage? It shouldn t be... But it can be...
This information is current as of April 28, 2010
 Practice Parameter: Pharmacological treatment of migraine headache in children and adolescents: Report of the American Academy of Neurology Quality Standards Subcommittee and the Practice Committee of
Practice Parameter: Pharmacological treatment of migraine headache in children and adolescents: Report of the American Academy of Neurology Quality Standards Subcommittee and the Practice Committee of
Migraine is an important clinical problem in
 REVIEW ARTICLE Symptomatic Treatment of Migraine in Children: A Systematic Review of Medication Trials Léonie Damen, PhD*; Jacques K. J. Bruijn, MD ; Arianne P. Verhagen, PhD*; Marjolein Y. Berger, MD
REVIEW ARTICLE Symptomatic Treatment of Migraine in Children: A Systematic Review of Medication Trials Léonie Damen, PhD*; Jacques K. J. Bruijn, MD ; Arianne P. Verhagen, PhD*; Marjolein Y. Berger, MD
Acute treatment of migraine in children and adolescents
 Acute treatment of migraine in children and adolescents Cristiano Termine, MD a,b Matteo Ferri, MD a,b Umberto Balottin, MD b,c a Child Neuropsychiatry Unit, Department of Clinical and Biological Sciences,
Acute treatment of migraine in children and adolescents Cristiano Termine, MD a,b Matteo Ferri, MD a,b Umberto Balottin, MD b,c a Child Neuropsychiatry Unit, Department of Clinical and Biological Sciences,
Migraine in Children. Germano Falcao, MD Pediatric Neurology 03/07/2014
 Migraine in Children Germano Falcao, MD Pediatric Neurology 03/07/2014 Headaches in Children One of the most common concerns reported by children 3% for children age 3-7 years 4-11% for children age 7-11
Migraine in Children Germano Falcao, MD Pediatric Neurology 03/07/2014 Headaches in Children One of the most common concerns reported by children 3% for children age 3-7 years 4-11% for children age 7-11
SIGN on the pharmacological management of migraine
 GUIDELINES SIGN on the pharmacological management of migraine STEVE CHAPLIN In February 2018, the Scottish Intercollegiate Guidelines Network (SIGN) published a new guideline on the pharmacological management
GUIDELINES SIGN on the pharmacological management of migraine STEVE CHAPLIN In February 2018, the Scottish Intercollegiate Guidelines Network (SIGN) published a new guideline on the pharmacological management
UTILIZATION MANAGEMENT CRITERIA
 SUMATRIPTAN (ALSUMA, IMITREX, SUMAVEL DOSEPRO, ZECUITY ) UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: BRAND (generic) NAME: Serotonin 5-HT1 receptor agonists Imitrex (sumatriptan), Alsuma (sumatriptan),
SUMATRIPTAN (ALSUMA, IMITREX, SUMAVEL DOSEPRO, ZECUITY ) UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: BRAND (generic) NAME: Serotonin 5-HT1 receptor agonists Imitrex (sumatriptan), Alsuma (sumatriptan),
Specific Care Question : Question Originator: Plain Language Summary from The Office of Evidence Based Practice: Conditional Recommendation
 Ketorolac for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine is ketorolac an effective treatment? Question Originator: Migraine Therapy
Ketorolac for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine is ketorolac an effective treatment? Question Originator: Migraine Therapy
A new questionnaire for assessment of adverse events associated with triptans: methods of assessment influence the results. Preliminary results
 J Headache Pain (2004) 5:S112 S116 DOI 10.1007/s10194-004-0123-4 Michele Feleppa Fred D. Sheftell Luciana Ciannella Amedeo D Alessio Giancarlo Apice Nino N. Capobianco Donato M.T. Saracino Walter Di Iorio
J Headache Pain (2004) 5:S112 S116 DOI 10.1007/s10194-004-0123-4 Michele Feleppa Fred D. Sheftell Luciana Ciannella Amedeo D Alessio Giancarlo Apice Nino N. Capobianco Donato M.T. Saracino Walter Di Iorio
Page: 1 of 6. Aimovig (erenumab-aooe) injection, Ajovy (fremanezumab-vfrm) injection, Emgality (galcanezumab-gnim)
 Page: 1 of 6 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Last Review Date: November 30, 2018 Description Aimovig (erenumab-aooe) injection, Ajovy (fremanezumab-vfrm)
Page: 1 of 6 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Last Review Date: November 30, 2018 Description Aimovig (erenumab-aooe) injection, Ajovy (fremanezumab-vfrm)
Outpatient Headache Care Guideline
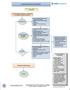 1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
Vestibular Migraine. Information for patients and carers. Department of Neurology and Otolaryngology Aberdeen Royal Infirmary
 Vestibular Migraine Information for patients and carers Department of Neurology and Otolaryngology Aberdeen Royal Infirmary What is vestibular migraine? Migraine is a disabling headache disorder that affects
Vestibular Migraine Information for patients and carers Department of Neurology and Otolaryngology Aberdeen Royal Infirmary What is vestibular migraine? Migraine is a disabling headache disorder that affects
Preventive treatment of migraine. Rebecca Burch, MD Brigham and Women s Faulkner Hospital Harvard Medical School Boston, MA
 Preventive treatment of migraine Rebecca Burch, MD Brigham and Women s Faulkner Hospital Harvard Medical School Boston, MA No disclosures Disclosures Many preventive treatments for migraine are not FDA-approved
Preventive treatment of migraine Rebecca Burch, MD Brigham and Women s Faulkner Hospital Harvard Medical School Boston, MA No disclosures Disclosures Many preventive treatments for migraine are not FDA-approved
Headaches in the Pediatric Emergency Dept
 Headaches in Children February 23, 2011 Jinny Tavee, MD Associate Professor Neuromuscular Center Cleveland Clinic Foundation Cleveland, OH 1 Headaches in the Pediatric Emergency Dept Burton Gutierrez Kan
Headaches in Children February 23, 2011 Jinny Tavee, MD Associate Professor Neuromuscular Center Cleveland Clinic Foundation Cleveland, OH 1 Headaches in the Pediatric Emergency Dept Burton Gutierrez Kan
Drug Class Review on Triptans
 Drug Class Review on UPDATED FINAL REPORT #1 December 2003 Mark Helfand, MD, MPH Kim Peterson, MS Oregon Evidence-based Practice Center Oregon Health & Science University Table of Contents Introduction
Drug Class Review on UPDATED FINAL REPORT #1 December 2003 Mark Helfand, MD, MPH Kim Peterson, MS Oregon Evidence-based Practice Center Oregon Health & Science University Table of Contents Introduction
10/13/17. Christy M. Jackson, MD Director, Dalessio Headache Center Scripps Clinic, La Jolla Clinical Professor, Neurosciences UCSD
 Christy M. Jackson, MD Director, Dalessio Headache Center Scripps Clinic, La Jolla Clinical Professor, Neurosciences UCSD } Depomed Consultant 2014 to present } Avanir Consultant 2014 to present } Amgen
Christy M. Jackson, MD Director, Dalessio Headache Center Scripps Clinic, La Jolla Clinical Professor, Neurosciences UCSD } Depomed Consultant 2014 to present } Avanir Consultant 2014 to present } Amgen
5 HT receptor agonists (triptans) and Pediatric Migraine Critically Appraised Topic (CAT)
 5 HT receptor agonists (triptans) and Pediatric Migraine Critically Appraised Topic (CAT) PICOT Question: In children and adolescents does use of 5 HT receptor agonists (triptans) versus no treatment treat
5 HT receptor agonists (triptans) and Pediatric Migraine Critically Appraised Topic (CAT) PICOT Question: In children and adolescents does use of 5 HT receptor agonists (triptans) versus no treatment treat
Prednisone vs. placebo in withdrawal therapy following medication overuse headache
 doi:10.1111/j.1468-2982.2007.01488.x Prednisone vs. placebo in withdrawal therapy following medication overuse headache L Pageler 1,2, Z Katsarava 2, HC Diener 2 & V Limmroth 1,2 1 Department of Neurology,
doi:10.1111/j.1468-2982.2007.01488.x Prednisone vs. placebo in withdrawal therapy following medication overuse headache L Pageler 1,2, Z Katsarava 2, HC Diener 2 & V Limmroth 1,2 1 Department of Neurology,
COMBINATION THERAPIES PREVENTATIVE THERAPIES BETA BLOCKERS
 ACUTE THEAPIES TIPTANS TICYCLIC ANTIDEPESSANTS When starting acute treatment, healthcare professionals should warn patients about the risk of developing medication-overuse headache. ASPIIN Aspirin (900
ACUTE THEAPIES TIPTANS TICYCLIC ANTIDEPESSANTS When starting acute treatment, healthcare professionals should warn patients about the risk of developing medication-overuse headache. ASPIIN Aspirin (900
Drug Class Review on the Triptans
 Drug Class Review on the Final Report March 7, 2003 Expires October 31, 2003 Mark Helfand, MD, MPH Kim Peterson, MS Produced by Oregon Health & Science University 3181 SW Sam Jackson Park Road Mailcode:
Drug Class Review on the Final Report March 7, 2003 Expires October 31, 2003 Mark Helfand, MD, MPH Kim Peterson, MS Produced by Oregon Health & Science University 3181 SW Sam Jackson Park Road Mailcode:
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D.
 Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
Anti-Migraine Agents
 DRUG POLICY BENEFIT APPLICATION Anti-Migraine Agents Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations or exceptions
DRUG POLICY BENEFIT APPLICATION Anti-Migraine Agents Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations or exceptions
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 CGRP Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
CGRP Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
OVERVIEW OF COMMENTS RECEIVED ON DRAFT GUIDELINE ON CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS FOR THE TREATMENT OF MIGRAINE
 European Medicines Agency London, 24 January 2007 Doc. Ref. EMEA/CHMP/EWP/21477/2007 OVERVIEW OF COMMENTS RECEIVED ON DRAFT GUIDELINE ON CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS FOR THE TREATMENT OF
European Medicines Agency London, 24 January 2007 Doc. Ref. EMEA/CHMP/EWP/21477/2007 OVERVIEW OF COMMENTS RECEIVED ON DRAFT GUIDELINE ON CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS FOR THE TREATMENT OF
Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial
 J Headache Pain (2007) 8:175 179 DOI 10.1007/s10194-007-0386-7 ORIGINAL Usha Kant Misra Jayantee Kalita Rama Kant Yadav Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial Received:
J Headache Pain (2007) 8:175 179 DOI 10.1007/s10194-007-0386-7 ORIGINAL Usha Kant Misra Jayantee Kalita Rama Kant Yadav Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial Received:
Migraine Types and Triggering Factors in Children
 original ARTICLE Migraine Types and Triggering Factors in Children How to Cite this Article: Nejad Biglari H, Karimzadeh P, Mohammadi Kord-kheyli M, Hashemi SM. Migraine Types and Triggering Factors in
original ARTICLE Migraine Types and Triggering Factors in Children How to Cite this Article: Nejad Biglari H, Karimzadeh P, Mohammadi Kord-kheyli M, Hashemi SM. Migraine Types and Triggering Factors in
Despite the widespread use of triptans ... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability
 ... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability Randal L. Von Seggern, PharmD, BCPS Abstract Objective: This article summarizes preclinical and clinical data for
... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability Randal L. Von Seggern, PharmD, BCPS Abstract Objective: This article summarizes preclinical and clinical data for
Clinical Policy: Triptans Reference Number: CP.CPA.217 Effective Date: Last Review Date: Line of Business: Commercial
 Clinical Policy: Reference Number: CP.CPA.217 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Reference Number: CP.CPA.217 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Get ahead of the ACHE: Monoclonal Antibodies in Migraine Prevention
 Get ahead of the ACHE: Monoclonal Antibodies in Migraine Prevention Amanda Janisch, PharmD PGY2 Ambulatory Care Pharmacy Resident MCHS SWMN, Mankato, MN 2018 MFMER slide-1 Disclosures No financial interest
Get ahead of the ACHE: Monoclonal Antibodies in Migraine Prevention Amanda Janisch, PharmD PGY2 Ambulatory Care Pharmacy Resident MCHS SWMN, Mankato, MN 2018 MFMER slide-1 Disclosures No financial interest
Review Article Pediatric Migraine
 International Pediatrics Volume 2009, Article ID 424192, 7 pages doi:10.1155/2009/424192 Review Article Pediatric Migraine Ubaid Hameed Shah and Veena Kalra Apollo Centre for Advanced Pediatrics, Indraprastha
International Pediatrics Volume 2009, Article ID 424192, 7 pages doi:10.1155/2009/424192 Review Article Pediatric Migraine Ubaid Hameed Shah and Veena Kalra Apollo Centre for Advanced Pediatrics, Indraprastha
1/25/2018 ARE CGRP ANTAGONISTS ANY BETTER THAN CURRENT EVIDENCE BASED TREATMENTS? Disclosures: Objectives: Headache Division
 ARE CGRP ANTAGONISTS ANY BETTER THAN CURRENT EVIDENCE BASED TREATMENTS? Lawrence C Newman, MD, FAHS, FAAN Clinical Professor of Neurology Disclosures: Advisory Board: Alder, Allergan, Amgen, Lilly, Supernus,
ARE CGRP ANTAGONISTS ANY BETTER THAN CURRENT EVIDENCE BASED TREATMENTS? Lawrence C Newman, MD, FAHS, FAAN Clinical Professor of Neurology Disclosures: Advisory Board: Alder, Allergan, Amgen, Lilly, Supernus,
TABLE 1. Current Diagnostic Criteria for Migraine Without Aura 2 A. At least 5 attacks fulfilling criteria B-D B. Headache attacks lasting 4-72 hours
 ANSWERS CONCISE TO FREQUENTLY REVIEW ASKED QUESTIONS FOR CLINICIANS ABOUT MIGRAINE Answers to Frequently Asked Questions About Migraine IVAN GARZA, MD, AND JERRY W. SWANSON, MD Migraine is a common primary
ANSWERS CONCISE TO FREQUENTLY REVIEW ASKED QUESTIONS FOR CLINICIANS ABOUT MIGRAINE Answers to Frequently Asked Questions About Migraine IVAN GARZA, MD, AND JERRY W. SWANSON, MD Migraine is a common primary
A one-year prospective costing study of botulinum toxin type A treatment of chronic tension headache
 J Headache Pain (2004) 5:192 196 DOI 10.1007/s10194-004-0100-3 ORIGINAL Fabio Palazzo Francesco S. Mennini Laura Fioravanti Laura Piasini Gabriella Coloprisco Paolo Martelletti A one-year prospective costing
J Headache Pain (2004) 5:192 196 DOI 10.1007/s10194-004-0100-3 ORIGINAL Fabio Palazzo Francesco S. Mennini Laura Fioravanti Laura Piasini Gabriella Coloprisco Paolo Martelletti A one-year prospective costing
What is the Effectiveness of OnabotulinumtoxinA (Botox ) in Reducing the Number of Chronic Migraines (CM) in Patients Years Old?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 What is the Effectiveness of OnabotulinumtoxinA
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 What is the Effectiveness of OnabotulinumtoxinA
Migraine much more than just a headache
 Migraine much more than just a headache Session hosted by Teva UK Limited PUU4 11:15 12:15 UK/NHSS/18/0021b Date of Preparation: August 2018 The views expressed in this presentation are those of the speaker
Migraine much more than just a headache Session hosted by Teva UK Limited PUU4 11:15 12:15 UK/NHSS/18/0021b Date of Preparation: August 2018 The views expressed in this presentation are those of the speaker
Medication For Migraine Chart: Table 1: Acute Treatment when the attack begins
 Medication For Migraine Chart: Table 1: Acute Treatment when the attack begins Page a Analgesics (painkillers) Non-steroidal antiinflammatory drugs (NSAIDs) Prescription required Brand Name Formulation
Medication For Migraine Chart: Table 1: Acute Treatment when the attack begins Page a Analgesics (painkillers) Non-steroidal antiinflammatory drugs (NSAIDs) Prescription required Brand Name Formulation
Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, II. Management of Refractory Headaches
 Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, 2016 II. Management of Refractory Headaches Case presentation 1: A case of intractable daily-persistent headache Keio University School
Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, 2016 II. Management of Refractory Headaches Case presentation 1: A case of intractable daily-persistent headache Keio University School
Effective Health Care Program
 Comparative Effectiveness Review Number 108 Effective Health Care Program Migraine in Children: Preventive Pharmacologic Treatments Executive Summary Introduction The Agency for Healthcare Research and
Comparative Effectiveness Review Number 108 Effective Health Care Program Migraine in Children: Preventive Pharmacologic Treatments Executive Summary Introduction The Agency for Healthcare Research and
Proposed Project Scope. OPTIMAL USE OnabotulinumtoxinA for the Prevention of Chronic Migraine Clinical Evidence, Policies and Practice
 Proposed Project Scope OPTIMAL USE OnabotulinumtoxinA for the Prevention of Chronic Migraine Clinical Evidence, Policies and Practice May 2018 1. BACKGROUND AND RATIONALE Migraine is a common, debilitating
Proposed Project Scope OPTIMAL USE OnabotulinumtoxinA for the Prevention of Chronic Migraine Clinical Evidence, Policies and Practice May 2018 1. BACKGROUND AND RATIONALE Migraine is a common, debilitating
ปวดศ รษะมา 5 ป ก นยาแก ปวดก ย งไม ข น นพ.พาว ฒ เมฆว ช ย โรงพยาบาลนครราชส มา
 ปวดศ รษะมา 5 ป ก นยาแก ปวดก ย งไม ข น นพ.พาว ฒ เมฆว ช ย โรงพยาบาลนครราชส มา 1 CONTENT 1 2 3 Chronic Daily Headache Medical Overused Headache Management Headaches are one of the most common symptoms List
ปวดศ รษะมา 5 ป ก นยาแก ปวดก ย งไม ข น นพ.พาว ฒ เมฆว ช ย โรงพยาบาลนครราชส มา 1 CONTENT 1 2 3 Chronic Daily Headache Medical Overused Headache Management Headaches are one of the most common symptoms List
Drug Therapy Guidelines
 Drug Therapy Guidelines Applicable Medical Benefit Effective: 5/1/18 Pharmacy- Formulary 1 x Next Review: 3/19 Pharmacy- Formulary 2 x Date of Origin: 8/29/06 Triptans: almotriptan, Amerge, Axert, Frova,
Drug Therapy Guidelines Applicable Medical Benefit Effective: 5/1/18 Pharmacy- Formulary 1 x Next Review: 3/19 Pharmacy- Formulary 2 x Date of Origin: 8/29/06 Triptans: almotriptan, Amerge, Axert, Frova,
What is new in the migraine world! Modar Khalil Consultant neurologist Hull Royal Infirmary
 What is new in the migraine world! Modar Khalil Consultant neurologist Hull Royal Infirmary Overview Understanding the burden Commonly used terms Acute therapy What we currently have What we are going
What is new in the migraine world! Modar Khalil Consultant neurologist Hull Royal Infirmary Overview Understanding the burden Commonly used terms Acute therapy What we currently have What we are going
Treatments for migraine
 Treatments for migraine Information for patients and carers Department of Neurology Aberdeen Royal Infirmary Contents Page About this leaflet Abortive medication for migraine Painkillers Antisickness medication
Treatments for migraine Information for patients and carers Department of Neurology Aberdeen Royal Infirmary Contents Page About this leaflet Abortive medication for migraine Painkillers Antisickness medication
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Aimovig) Reference Number: CP.PHAR.128 Effective Date: 07.10.18 Last Review Date: 08.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Aimovig) Reference Number: CP.PHAR.128 Effective Date: 07.10.18 Last Review Date: 08.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
MEASURE #4: Overuse of Barbiturate Containing Medications for Primary Headache Disorders Headache
 MEASURE #4: Overuse of Barbiturate Containing Medications for Primary Headache Disorders Headache Measure Description Percentage of patients age 18 years old and older with a diagnosis of primary headache
MEASURE #4: Overuse of Barbiturate Containing Medications for Primary Headache Disorders Headache Measure Description Percentage of patients age 18 years old and older with a diagnosis of primary headache
Goals. Primary Headache Syndromes. One-Year Prevalence of Common Headache Disorders
 Goals One-Year Prevalence of Common Headache Disorders Impact of primary headache syndromes Non pharmacologic Rx of migraine individualized to patient triggers Complementary and alternative Rx of migraine
Goals One-Year Prevalence of Common Headache Disorders Impact of primary headache syndromes Non pharmacologic Rx of migraine individualized to patient triggers Complementary and alternative Rx of migraine
Long-Term Care Updates
 Long-Term Care Updates September 2017 By Lindsay Slowiczek, PharmD Migraines are often considered to be a condition affecting younger or middle-aged patients, during which patients experience episodic,
Long-Term Care Updates September 2017 By Lindsay Slowiczek, PharmD Migraines are often considered to be a condition affecting younger or middle-aged patients, during which patients experience episodic,
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Literature Scan: Triptans
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Migraine Headache Prophylaxis in Adolescents
 I Migraine Headache Prophylaxis in Adolescents It s estimated that up to 28 percent of adolescents between the ages of 15 and 19 (mostly females) are affected by migraine (Lewis, Yonker, Winner, & Sowell,
I Migraine Headache Prophylaxis in Adolescents It s estimated that up to 28 percent of adolescents between the ages of 15 and 19 (mostly females) are affected by migraine (Lewis, Yonker, Winner, & Sowell,
Migraine Management. Jane Melling Headache nurse Mater Misericordiae Hospital
 Migraine Management Jane Melling Headache nurse Mater Misericordiae Hospital Migraine facts Among the most common disorders of the nervous system 3 rd most prevalent medical disorder on the planet (lancet
Migraine Management Jane Melling Headache nurse Mater Misericordiae Hospital Migraine facts Among the most common disorders of the nervous system 3 rd most prevalent medical disorder on the planet (lancet
Overuse of barbiturate and opioid containing medications for primary headache disorders Description
 Measure Title Overuse of barbiturate and opioid containing medications for primary headache disorders Description Percentage of s age 12 years and older with a diagnosis of primary headache who were prescribed
Measure Title Overuse of barbiturate and opioid containing medications for primary headache disorders Description Percentage of s age 12 years and older with a diagnosis of primary headache who were prescribed
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Emgality) Reference Number: CP.PHAR.## Effective Date: 11.13.18 Last Review Date: 02.19 Line of Business: Commercial, TBD HIM*, Medicaid Revision Log See Important Reminder at the end
Clinical Policy: (Emgality) Reference Number: CP.PHAR.## Effective Date: 11.13.18 Last Review Date: 02.19 Line of Business: Commercial, TBD HIM*, Medicaid Revision Log See Important Reminder at the end
Drug Class Review Triptans
 Drug Class Review Triptans April 2013 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness and harms of different
Drug Class Review Triptans April 2013 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness and harms of different
Management options for Migraine. Prof. Dr. Khwaja Nazimuddin Head Dept. of Internal Medicine BIRDEM
 Management options for Migraine Prof. Dr. Khwaja Nazimuddin Head Dept. of Internal Medicine BIRDEM Assessment The Migraine Disability Assessment Score MIDAS Complete loss of work Partial loss of work Off
Management options for Migraine Prof. Dr. Khwaja Nazimuddin Head Dept. of Internal Medicine BIRDEM Assessment The Migraine Disability Assessment Score MIDAS Complete loss of work Partial loss of work Off
ONZETRA XSAIL (sumatriptan) nasal powder
 ONZETRA XSAIL (sumatriptan) nasal powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
ONZETRA XSAIL (sumatriptan) nasal powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
Clinical Policy: Triptans Reference Number: CP.HNMC.217 Effective Date: Last Review Date: Line of Business: Medicaid Medi-Cal
 Clinical Policy: Reference Number: CP.HNMC.217 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: Reference Number: CP.HNMC.217 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this policy for important
Dubai Standards of Care (Migraine)
 Dubai Standards of Care 2018 (Migraine) Preface Migraine is one of the most common problem dealt with in daily practice. In Dubai, the management of migraine is done through various different strategies.
Dubai Standards of Care 2018 (Migraine) Preface Migraine is one of the most common problem dealt with in daily practice. In Dubai, the management of migraine is done through various different strategies.
Case Presentation. Case Presentation. Case Presentation. Truths about Headaches (2017) Most headaches were muscle-tension headaches
 Agenda Case presentation Migraine Morphology Primary and Premonitory Phase Secondary Headache Aura Headache Primer on Pain Medication Overuse Headache Case Presentation RT is a 25 year old woman with daily
Agenda Case presentation Migraine Morphology Primary and Premonitory Phase Secondary Headache Aura Headache Primer on Pain Medication Overuse Headache Case Presentation RT is a 25 year old woman with daily
Specific features of migraine syndrome in children
 J Headache Pain (2006) 7:206 210 DOI 10.1007/s10194-006-0312-4 RAPID COMMUNICATION Marija Knezevic-Pogancev Specific features of migraine syndrome in children Received: 16 April 2006 Accepted in revised
J Headache Pain (2006) 7:206 210 DOI 10.1007/s10194-006-0312-4 RAPID COMMUNICATION Marija Knezevic-Pogancev Specific features of migraine syndrome in children Received: 16 April 2006 Accepted in revised
Abortive Agents. Available Strengths. Formulary Limits. Tablet: 5mg, 10mg ODT: 5mg, 10 mg 25mg, 50mg, 100mg. 5mg/act, 20mg/act
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Migraine Therapy P&T DATE: 9/12/2017 CLASS: Neurological Disorders REVIEW HISTORY 12/16, 9/15, 2/15, 2/10, 5/07 LOB: MCL
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Migraine Therapy P&T DATE: 9/12/2017 CLASS: Neurological Disorders REVIEW HISTORY 12/16, 9/15, 2/15, 2/10, 5/07 LOB: MCL
Chronic Migraine in Primary Care. December 11 th, 2017 Werner J. Becker University of Calgary
 Chronic Migraine in Primary Care December 11 th, 2017 Werner J. Becker University of Calgary Disclosures Faculty: Werner J. Becker Relationships with commercial interests: Grants/Research Support: Clinical
Chronic Migraine in Primary Care December 11 th, 2017 Werner J. Becker University of Calgary Disclosures Faculty: Werner J. Becker Relationships with commercial interests: Grants/Research Support: Clinical
Index. Prim Care Clin Office Pract 31 (2004) Note: Page numbers of article titles are in boldface type.
 Prim Care Clin Office Pract 31 (2004) 441 447 Index Note: Page numbers of article titles are in boldface type. A Abscess, brain, headache in, 388 Acetaminophen for migraine, 406 407 headache from, 369
Prim Care Clin Office Pract 31 (2004) 441 447 Index Note: Page numbers of article titles are in boldface type. A Abscess, brain, headache in, 388 Acetaminophen for migraine, 406 407 headache from, 369
Treatment Of Medication. Overuse Headache
 7 November 2012 BASH GPwSI Meeting Lecture title... Treatment Of Medication Dr... Overuse Headache Dr Marcus Lewis Dr... The National Migraine Centre International Headache Society Diagnostic criteria
7 November 2012 BASH GPwSI Meeting Lecture title... Treatment Of Medication Dr... Overuse Headache Dr Marcus Lewis Dr... The National Migraine Centre International Headache Society Diagnostic criteria
Overview of diagnosis and management of paediatric headache. Part II: therapeutic management
 J Headache Pain (2011) 12:25 34 DOI 10.1007/s10194-010-0256-6 REVIEW ARTICLE Overview of diagnosis and management of paediatric headache. Part II: therapeutic management Cristiano Termine Aynur Özge Fabio
J Headache Pain (2011) 12:25 34 DOI 10.1007/s10194-010-0256-6 REVIEW ARTICLE Overview of diagnosis and management of paediatric headache. Part II: therapeutic management Cristiano Termine Aynur Özge Fabio
The use of combination therapies in the acute management of migraine
 REVIEW The use of combination therapies in the acute management of migraine Abouch Valenty Krymchantowski Headache Center of Rio, Rio de Janeiro, Brazil; Outpatient Headache Unit of the Instituto de Neurologia
REVIEW The use of combination therapies in the acute management of migraine Abouch Valenty Krymchantowski Headache Center of Rio, Rio de Janeiro, Brazil; Outpatient Headache Unit of the Instituto de Neurologia
MIGRAINE UPDATE. Objectives & Disclosures. Learn techniques used to diagnose headaches. Become familiar with medications used for headache treatment.
 MIGRAINE UPDATE Karen L. Bremer, MD November 16, 2018 Objectives & Disclosures Learn techniques used to diagnose headaches. Become familiar with medications used for headache treatment. Disclosure: I am
MIGRAINE UPDATE Karen L. Bremer, MD November 16, 2018 Objectives & Disclosures Learn techniques used to diagnose headaches. Become familiar with medications used for headache treatment. Disclosure: I am
Current Migraine Treatment Therapy. Daniel Kassicieh, DO, FAAN
 Current Migraine Treatment Therapy Daniel Kassicieh, DO, FAAN Migraine a Disease Process Migraines are a chronic disease process similar to many other chronic medical conditions Migraine has a low mortality
Current Migraine Treatment Therapy Daniel Kassicieh, DO, FAAN Migraine a Disease Process Migraines are a chronic disease process similar to many other chronic medical conditions Migraine has a low mortality
Clinical Learning Days November 10, 2017
 Migraine Clinical Learning Days November 10, 2017 Alyssa Lettich. MD Neurosciences Institute/Neurosciences Clinical Program Medical Director Headache Disclosures: none Learning Objectives: At the conclusion
Migraine Clinical Learning Days November 10, 2017 Alyssa Lettich. MD Neurosciences Institute/Neurosciences Clinical Program Medical Director Headache Disclosures: none Learning Objectives: At the conclusion
SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection
 SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
Migranal Nasal Spray. Migranal Nasal Spray (dihydroergotamine) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: June 22, 2017 Migranal Nasal Spray
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: June 22, 2017 Migranal Nasal Spray
