Departments of Biostatistics, Faculty of Health, Mazandaran University of Medical Sciences, Sari, Iran 4
|
|
|
- Sherman Rice
- 5 years ago
- Views:
Transcription
1 Clin Exp Emerg Med 2017;4(3): Single-dose intravenous sodium valproate (Depakine) versus dexamethasone for the treatment of acute migraine headache: a doubleblind randomized clinical trial Narges Karimi 1, Mahdiye Tavakoli 2, Jamshid Yazdani Charati 3, Mastoureh Shamsizade 4 1 Department of Neurology, Faculty of Medicine, Immunogenetics Research Center, Clinical Research Development Unit of Bou Ali Sina Hospital, Mazandaran University of Medical Sciences, Sari, Iran 2 Faculty of Medicine, Mazandaran University of Medical Sciences, Sari, Iran 3 Departments of Biostatistics, Faculty of Health, Mazandaran University of Medical Sciences, Sari, Iran 4 Bou Ali Sina Hospital, Mazandaran University of Medical Sciences, Sari, Iran eissn: Received: 19 December 2016 Revised: 26 January 2017 Accepted: 31 January 2017 Correspondence to: Narges Karimi Department of Neurology, Bou Ali Hospital, Pasdaran Boulevard, Sari city, Mazandaran province, Iran N.karimi@mazums.ac.ir Original Article Objective Migraine headache is a chronic and disabling condition in adults. Some studies have investigated the efficacy of sodium valproate in the treatment of acute migraine, but the effectiveness and tolerability of intravenous valproate as abortive therapy remains unclear. This study aimed to evaluate the effects of sodium valproate and dexamethasone in the treatment of acute migraine. Methods We conducted a double-blind randomized clinical trial including 90 patients aged 18 to 65 years with acute migraine headache but no aura. Patients were randomized to receive intravenous dexamethasone (8 mg) or sodium valproate (400 mg) diluted into 4 ml of normal saline. The primary outcome measure was pain relief after 0.5, 1, 3, or 6 hours after administration. The secondary outcome criteria were the associated symptom recovery, rate of headache recurrence after 24 hours, and medication side effects. Pearson s chi square and the t-test were employed in the data analysis. Results Of the 90 patients, 80 were investigated. The percentage of headache improvement at 0.5 hours after treatment was 55% and 67.5% in the sodium valproate and dexamethasone groups, respectively. Before-treatment and 0.5 hour after treatment pain severity visual analog scale scores were 9.05±0.90 and 3.8±3.09 in the sodium valproate group and 8.92±0.79 and 3.10±2.73 in the dexamethasone group, respectively. There were no significant intergroup differences. Conclusion This randomized clinical trial showed that the intravenous injection of sodium valproate 400 mg has similar effects to those of dexamethasone for improving acute migraine headache. Keywords Migraine disorders; Dexamethasone; Valproic acid; Acute; Therapeutics How to cite this article: Karimi N, Tavakoli M, Charati JY, Shamsizade M. Single-dose intravenous sodium valproate (Depakine) versus dexamethasone for the treatment of acute migraine headache: a double-blind randomized clinical trial. Clin Exp Emerg Med 2017;4(3): This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License ( creativecommons.org/licenses/by-nc/4.0/). 138 Copyright 2017 The Korean Society of Emergency Medicine
2 Narges Karimi, et al. What is already known Sodium valproate is a prophylactic antiepileptic agent. The effect of sodium valproate as abortive therapy remains unclear, and the US Food and Drug Administration just recently approved the drug for this indication. Studies have reported the different effects of intravenous sodium valproate for the treatment of acute migraine. What is new in the current study In this study, we compared the effect of single-dose intravenous sodium valproate with that of dexamethasone for treating acute migraine. Intravenous sodium valproate was as effective as dexamethasone at relieving pain in patients with a migraine headache. INTRODUCTION Migraine headache is a chronic and disabling primary headache in adults that constitutes 1% to 2% of all emergency department admissions. 1 The exact prevalence of migraine is unknown and varies throughout the world. In Iran, the prevalence of migraine is reportedly 18.11% in adults in Tehran and 6% in Yazad city, while the lowest prevalence (1.7%) was reported in primary-school children in Shiraz. 2-4 This disorder presents with attacks of moderate to severe throbbing headaches unilaterally associated with nausea and/or vomiting, photophobia, and phonophobia. 5 Medications used to treat acute attacks include triptans, ergotamine, nonsteroidal anti-inflammatory drugs, intravenous fluids, antiemetic, opioids, neuroleptics, and dexamethasone Studies have shown different effects of dexamethasone in the treatment of migraine headaches. 11,12 Some studies reported that dexamethasone is significantly effective for treating acute migraine and reducing the risk of early headache recurrence due to its anti-inflammatory effects without dangerous side effects, even if administrated as a high single dose Some studies have investigated the efficacy of sodium valproate with different doses in the treatment of acute migraine, and its efficacy has been compared with other drugs such as sumatriptan, ketorolac, metoclopramide, or dexamethasone These studies and other surveys have reported different results In different regions of Iran, two studies had compared intravenous sodium valproate and dexamethasone for the treatment of acute migraine with different samples and dosages. 23,24 Sodium valproate is an antiepileptic agent that is used prophylactically and more recently for the treatment of acute migraine. Sodium valproate increases γ-amino butyric acid levels in the central nervous system and diminishes firing rates of serotonergic cells. This drug directly affects the neuronal cell membrane and limits the repetitive firing rate of the sodium and calcium current. This effect occurs rapidly and can explain the influence of sodium valproate in abortive therapy The influence of sodium valproate as abortive therapy remains unclear, and US Food and Drug Administration only recently approved its use for abortive therapy; additionally, studies have reported the different effects of intravenous sodium valproate for the treatment of acute migraine. 29,34,35 This study aimed to compare the effects of intravenous sodium valproate and other common drugs (dexamethasone) used in the treatment of acute migraine. METHODS Trial design This double-blinded randomized clinical trial (registration no. IRCT N1) included adult patients (18 to 65 years old) with migraine headache without aura who fulfilled the international classification of headache disorders, 3rd edition (beta version), diagnostic criteria for migraine. 5 Settings and participants This study was performed in the clinical research development unit of a university referral emergency department in Sari city, Mazandaran province, Iran, between October 2014 and June The inclusion criteria included age 18 to 65 years, history of migraine type headache without aura for at least 1 year prior to the study, moderate to severe headache persisting for less than 6 days with a visual analog scale (VAS) score > 7, normal vital signs, and normal neurological exam findings. Patients with abnormal vital signs, renal failure, hepatic disease, heart disease, active peptic ulcer disease, or diabetes mellitus were excluded from the study. Participants with allergies to dexamethasone and sodium valproate or who were pregnant or breastfeeding were excluded from the study. Patients who had taken nonsteroidal anti-inflammatory drugs during the previous 24 hours or valproate during the previous 2 weeks were not included in the study. Clin Exp Emerg Med 2017;4(3):
3 Sodium valproate versus dexamethasone Sample size, randomization, and blinding According to the results of previous clinical trials 36,37 with 80% power and a confidence interval of 95%, a total sample size of 38 was calculated in each group. Participation in the study was discontinued when a total of 80 patients were enrolled. A CONSORT (Consolidated Standards of Reporting Trials) flow diagram illustrates the flow of participants enrolling into the study (Fig. 1). Block randomization was used for recruiting participants. Randomization was accomplished using a computerized random number table. The drugs were placed in envelopes randomly labeled with a serial number from 1,000 to 2,000 using the RANDBETWEEN function in Excel (Microsoft, Redmond, WA, USA). Drugs were prepared in the same syringe and injected by a nurse who was not part of the study. Both the investigator (emergency physician and neurologist) and patient remained blinded to the medication delivered until the code was determined at the end of the study. The patients were selected by a general physician after physical examination by a neurologist and meeting the eligibility criteria and recruited for the study. Written informed consent was obtained from all patients and the study proposal was approved by the Regional Ethics Committee of Mazandaran University of Medical Sciences (code no ). Prior to the drug administration, all participants filled out a questionnaire that included demographic information, headache duration and severity, and associated symptoms (photophobia, phonophobia, nausea, and vomiting). Intervention and outcomes After patient randomization into two groups, dexamethasone 8 mg was intravenously administered to the control group and sodium valproate 400 mg (Depakine; Sanofi Aventis, Paris, France) diluted into 4 ml of normal saline was intravenously administered to the intervention group. Pain severity and other associated symptoms such as nausea or vomiting, photophobia, and phonophobia, and the presence of adverse effects were assessed at baseline and at 0.5, 1, 3, and 6 hours and as well the rate of recurrent headache at 24 hours after receiving the drugs. The primary outcome criteria were pain relief (no or mild pain and a VAS score 3) after 0.5, 1, 3, and 6 hours after receiving the drug. Secondary outcome criteria were associated symptom recovery over 24 hours, headache recurrence rate after 24 hours, and medication side effects. Assessed for eligibility (n=90) Excluded (n=10) Not meeting inclusion criteria (n=7) Declined to part (n=3) Randomized (n=80) Allocation Allocated to dexamethasone group (n=40) Received allocated intervention (n=40) Did not receive allocated intervention (n=0) Allocated to valproate sodium (n=40) Received allocated intervention (n=40) Did not receive allocated intervention (n=0) Follow-Up Lost to follow-up (n=0) Discontinued intervention (n=0) Lost to follow-up (n=0) Discontinued intervention (n=0) Analysis Analysed (n=40) Excluded from analysis (n=0) Analysed (n=40) Excluded from analysis (n=0) Fig. 1. CONSORT (Consolidated Standards of Reporting Trials) flow diagram. Ten patients were excluded because they did not meet inclusion or exclusion criteria. In general, 80 participants were analyzed
4 Narges Karimi, et al. Measuring headache severity Headache severity was assessed on the basis of VAS score and pain intensity. The VAS scores range from 0 (no pain) to 10 (worst pain). The patients also recorded pain intensity on a four-point categorical scale (0= no pain, 1= mild pain, 2= moderate pain, and 3=severe pain). 38 Pain monitoring was continued using the VAS score and pain intensity by the neurologist throughout the management. The goal of treatment was no or mild pain. Statistical analysis Patients demographic data and the variables mentioned earlier were entered into the IBM SPSS Statistics ver. 20 (IBM Corp., Armonk, NY, USA). Pearson s chi-square test and the t-test were used to determine the relationship between demographic information and drugs, while the independent t-test and chi-square test were used to evaluate post-treatment pain severity in the two groups. Kaplan-Meier survival analysis and log-rank test were utilized to estimate time to headache relief and treatment success between the two groups within the first 6 hours. Treatment success was defined as decreased pain severity with a VAS score 3 as well as no or mild pain. The statistical significance level was set at P<0.05. RESULTS Ninety patients with acute migraine headache were screened for inclusion in this study. Seven patients did not meet the inclusion Table 1. Patient demographics Characteristics Dexamethasone group Sodium valproate group Age 33.4± ±9.5 Male 6 (15.0) 9 (22.5) Female 34 (85.0) 31 (77.5) Level of education Illiterate Elementary Under diploma Diploma Collage education Duration of disease (yr) >5 Time from onset (hr) Family history of migraine Yes No 0 7 (17.5) 5 (12.5) 18 (45.0) 10 (25.0) 6 (15.0) 11 (27.5) 23 (57.5) 2 (5.0) 16 (50.0) 17 (42.5) 5 (12.5) 31 (77.5) 9 (22.5) Values are presented as mean± standard deviation or number (%). 2 (5.0) 6 (15.0) 12 (30.0) 13 (32.5) 7 (17.5) 1 (2.5) 17(42.5) 22 (55) 0 14 (35.0) 19 (47.5) 7 (17.5) 32 (80.0) 8 (20.0) criteria and were excluded. Three participants did not fill out the consent form. A total of 80 patients were enrolled and analyzed. All of these patients continued follow up. There were 34 women and 6 men (mean age, 33.9 ±9.5 years) in the control group (dexamethasone) and 31 women and 9 men (mean age, 33.38± 9.15 years) in the intervention group (sodium valproate). The patients demographic information is shown in Table 1. There were no significant intergroup differences in demographic features and the two groups were similar. All participants mentioned photophobia and phonophobia but only one patient did not mention phonohobia in the valproate group. Seven patients (17.5%) in the dexamethasone group and five patients (12.5%) in the sodium valproate group did not report vomiting. There was no intergroup difference in associated symptoms. The mean VAS pain severity scores prior to drug administration in the dexamethasone and valproate groups were 8.92 ±0.79 (95% confidence interval, 8.68 to 9.18) and 9.05± 0.9 (95% confidence interval, 8.76 to 9.34), respectively. Thirty minutes after the therapeutic intervention, the pain severity was significantly reduced in the dexamethasone (3.10± 2.73) and sodium valproate (3.8±3.09) groups, but there was no significant intergroup difference (Table 2). In terms of headache relief efficacy, the differences between values 0.5, 1, 3, and 6 hours after treatment and those before treatment were statistically significant (P<0.001) (Table 3). Table 4 shows the reduction in pain intensity after versus before treatment according to the 4-point scale. At 0.5 hours after administration, the dexamethasone and valproate groups showed a successful reduction in pain severity in 22 (67.5%) and 27 (55%) of the patients, respectively, but there were no significant intergroup differences (P=0.17). There were also no statistically significant intergroup differences in age, sex, disease duration or improvement of headache. The Kaplan-Meier curve demonstrated the time to pain relief onset and success rates in both groups (Fig. 2). Any patient with a VAS score 3 after treatment was considered a treatment success. The log-rank test demonstrated no inter- Table 2. Visual analog scale pain scores before versus after treatment by group Sodium valproate group Dexamethasone group P-value Before treatment 9.05± ± After treatment 0.5 hr 3.85± ± hr 2.67± ± hr 2.02± ± hr 1.35± ± Values are presented as mean±standard deviation. Clin Exp Emerg Med 2017;4(3):
5 Sodium valproate versus dexamethasone Table 3. Effect of each drug on headache relief by visual analog scale score After treatment Sodium valproate group P-value a) Dexamethasone group P-value a) 0.5 hr 5.25± ± hr 6.37± ± hr 7.02± ± hr 7.73± ± Values are presented as mean± standard deviation. a) Paired t-test. Table 4. Significant improvement of pain after treatment based on four-point scale a) After treatment Sodium valproate group Dexamethasone group 95% CI P-value 0.5 hr 22 (55.0) 27 (67.5) hr 30 (75.0) 36 (90.0) hr 33 (82.5) 37 (92.5) hr 36 (90.0) 39 (97.5) Values are presented as number (%). CI, confidence interval. a) No or mild pain. Cum survival Survival functions A B A-censored B-censored was the same in both groups (7.5%). Migraine associated symptoms including photophobia, phonophobia, and nausea or vomiting were significantly improved in the two groups. No adverse effects were noted in the dexamethasone group, while only one patient in the valproate group had an adverse effect consisting of anxiety, unrest, and shortness of breath that improved 2 hours after treatment. DISCUSSION Time (hr) Fig. 2. Kaplan-Meier curve of efficacy by group. A, sodium valproate group; B, dexamethasone group. group difference in therapeutic effect (χ 2 value [1, 80]=2.018; P=0.155). Overall, 36 patients (90%) in the valproate group and 39 patients (97.5%) in the dexamethasone group showed pain improvement up to 6 hours after drug administration. The patients remained under observation in the emergency department for 24 hours. Six patients (15%) from the sodium valproate group and two patients (5%) from the dexamethasone group required rescue therapy 6 hours after treatment. Four patients (10%) in the valproate group and one patient (2.5%) in the dexamethasone group showed no improvement 6 hours after administration. The frequency of recurrent headache at 24 hours after treatment This randomized clinical trial demonstrated that dexamethasone and sodium valproate effectively treated acute migraine headache. More patients in the dexamethasone than sodium valproate group recovered, but the difference was not statistically significant. In other words, the drugs demonstrated the same efficacy for treating acute migraine headache. Similar studies performed in Iran during 2013 and 2015 showed that the two drugs had the same effects on headache relief. 23,24 In the present study, the headache relief rates in the sodium valproate and dexamethasone groups were 55% and 67.5% in the first 0.5 hour, respectively. In other studies, the percentage of pain relief after 1 hour in patients receiving sodium valproate was reported as 25% 39 and 53.3%. 28 There was no significant association between the two groups in terms of demographic factors and recovery rate in the first 0.5 hour. Foroughipour et al. 24 reported the duration of headache relief in the sodium valproate and dexamethasone groups as 292 and 270 minutes in 26% and 33% of patients, respectively. In that study, the sample size was small and the patients received thera
6 Narges Karimi, et al. peutic dosages higher than those used in our study. Unfortunately, the therapeutic dosage of sodium valproate for acute migraine headache has yet to be determined. In several studies, the therapeutic dosage of valproate was variable (400 to 1,200 mg) and the drug was diluted in normal saline (50 to 200 ml) In this study, both drugs were administered in a single dose diluted in a small amount of normal saline (4 ml) to avoid a confounding effect of normal saline, which is able to reduce headache via hydration. 10,40 Limdi et al. 41 reported the safety of a rapid infusion of undiluted sodium valproate in the treatment of epilepsy. The results of the present study demonstrated that the administration of a low dose of dexamethasone (8 mg) and sodium valproate (400 mg) diluted in 4 ml of normal saline can significantly decrease headache pain. Bakhshayesh et al. 28 reported that sodium valproate was more effective than metoclopramide and sumatriptan at providing headache relief during the 2 hours post-treatment, whereas Rahimdel et al. 25 reported that intravenous sodium valproate and subcutaneous sumatriptan have similar abilities to control acute migraine attacks. Edwards et al. 33 reported equal effectiveness in headache improvement after treatment with sodium valproate compared with dihydroergotamine and metoclopramide. Tanen et al. 34 compared the efficacy of intravenous sodium valproate versus prochlorperazine and demonstrated that the former was less effective at decreasing pain or nausea; however, patients were followed for the first hour and also received rescue therapy, which is a confounding variable. In this study, the rates of headache recurrence after 24 hours were very low in both groups. Foroughipour et al. 24 demonstrated that relapse of headache occurred in 68.4% of the sodium valproate group and 66.7% of the dexamethasone group within 72 hours after injection. In addition, Ghaderibarmi et al. 26 demonstrated that sodium valproate is more effective than sumatriptan at reducing pain and preventing headache recurrence. One study reported that intravenous dexamethasone was less effective at preventing acute migraine headache relapse, while another study revealed a reduced incidence of severe recurrent headache after dexamethasone infusion. 30,42 The number of cases in both studies was lower than that in ours, but both followed up their patients for 72 hours. In this study, the patients were assessed for 24 hours. It seems that a longer follow-up is required to evaluate recurrence. In our research, sodium valproate and dexamethasone were equally effective at improving migraine-associated symptoms. All of our patients reported improved pain at the same post-infusion duration. This finding is consistent with those of other studies, 25,26 but Foroughipour et al. 24 reported that dexamethasone was more effective than sodium valproate at treating the associated symptoms. The improvement of associated symptoms appears to be dependent on their severity and duration prior to treatment. Further investigations of this issue are required. In the current study, only one patient in the sodium valproate group reported a mild side effect. Moreover, other studies reported no serious adverse effect after sodium valproate or dexamethasone treatment The safety and tolerability of intravenous sodium valproate and dexamethasone for treating acute migraine attacks have been demonstrated in several investigations. 16,27,35,39,42 To the best of our knowledge, the study by Shahien et al. 43 is the only one to report serious side effects of intravenous sodium valproate, which can be attributed to the high dose used (900 to 1,200 mg). In conclusion, the rapid intravenous injection of a single dose of sodium valproate 400 mg had similar efficacy to that of dexamethasone at improving acute migraine headache without causing serious side effects. Moreover, these medications significantly improved the patients migraine-associated symptoms. CONFLICT OF INTEREST No potential conflict of interest relevant to this article was reported. ACKNOWLEDGMENTS We would like to appreciate the Vice Chancellor of Research and Technology of Mazandaran University of Medical Sciences for approval and financial support of this trial. The authors would like to thank the emergency physicians at Boo Ali Sina Hospital and also all of the patients that participated in this study. This study was Mahdiye Tavakoli s doctoral dissertation towards graduation in general medicine. REFERENCES 1. Talabi S, Masoumi B, Azizkhani R, Esmailian M. Metoclopramide versus sumatriptan for treatment of migraine headache: a randomized clinical trial. J Res Med Sci 2013;18: Shahbeigi S, Fereshtehnejad SM, Mohammadi N, et al. Epidemiology of headaches in Tehran urban area: a populationbased cross-sectional study in district 8, year Neurol Sci 2013;34: Momayyezi M, Fallahzadeh H, Momayyezi M. Prevalence of migraine and tension-type headache in Yazd, Iran. Zahedan J Res Med Sci 2015;17:e966. Clin Exp Emerg Med 2017;4(3):
7 Sodium valproate versus dexamethasone 4. Ayatollahi SM, Khosravi A. Prevalence of migraine and tension-type headache in primary-school children in Shiraz. East Mediterr Health J 2006;12: Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia 2013; 33: Holland S, Silberstein SD, Freitag F, et al. Evidence-based guideline update: NSAIDs and other complementary treatments for episodic migraine prevention in adults. Report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology 2012;78: Silberstein SD. Practice parameter: evidence-based guidelines for migraine headache (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2000;55: Tfelt-Hansen P, Block G, Dahlof C, et al. Guidelines for controlled trials of drugs in migraine: second edition. Cephalalgia 2000;20: Rizzoli PB. Acute and preventive treatment of migraine. Continuum (Minneap Minn) 2012;18: Gupta S, Oosthuizen R, Pulfrey S. Treatment of acute migraine in the emergency department. Can Fam Physician 2014;60: Rowe BH, Colman I, Edmonds ML, Blitz S, Walker A, Wiens S. Randomized controlled trial of intravenous dexamethasone to prevent relapse in acute migraine headache. Headache 2008; 48: Fiesseler FW, Shih R, Szucs P, et al. Steroids for migraine headaches: a randomized double-blind, two-armed, placebocontrolled trial. J Emerg Med 2011;40: Soleimanpour H, Ghafouri RR, Taheraghdam A, et al. Effectiveness of intravenous dexamethasone versus propofol for pain relief in the migraine headache: a prospective double blind randomized clinical trial. BMC Neurol 2012;12: Gilmore B, Michael M. Treatment of acute migraine headache. Am Fam Physician 2011;83: Singh A, Alter HJ, Zaia B. Does the addition of dexamethasone to standard therapy for acute migraine headache decrease the incidence of recurrent headache for patients treated in the emergency department? A meta-analysis and systematic review of the literature. Acad Emerg Med 2008;15: Bigal M, Sheftell F, Tepper S, Tepper D, Ho TW, Rapoport A. A randomized double-blind study comparing rizatriptan, dexamethasone, and the combination of both in the acute treatment of menstrually related migraine. Headache 2008;48: Ropper AH, Samuels MA, Klein JP. Adams and Victor s principles of neurology. 10th ed. New York, NY: MC Graw Hill; Daroff RB, Fenichel GM, Jankovic J, Mazziotta JC. Neurology in clinical practice. 6th ed. New York, NY: Elsevier; Colman I, Friedman BW, Brown MD, et al. Parenteral dexamethasone for acute severe migraine headache: meta-analysis of randomised controlled trials for preventing recurrence. BMJ 2008;336: Baden EY, Hunter CJ. Intravenous dexamethasone to prevent the recurrence of benign headache after discharge from the emergency department: a randomized, double-blind, placebocontrolled clinical trial. CJEM 2006;8: Hardman JG, Limbird LE. Goodman and Gilman s the pharmacological basis of therapeutics. New York, NY: MC Graw Hill; Friedman BW, Garber L, Yoon A, et al. Randomized trial of IV valproate vs metoclopramide vs ketorolac for acute migraine. Neurology 2014;82: Mazaheri S, Poorolajal J, Hosseinzadeh A, Fazlian MM. Effect of intravenous sodium valproate vs dexamethasone on acute migraine headache: a double blind randomized clinical trial. PLoS One 2015;10:e Foroughipour M, Ghandehari K, Khazaei M, Ahmadi F, Shariatinezhad K, Ghandehari K. Randomized clinical trial of intravenous valproate (orifil) and dexamethasone in patients with migraine disorder. Iran J Med Sci 2013;38: Rahimdel A, Mellat A, Zeinali A, Jafari E, Ayatollahi P. Comparison between intravenous sodium valproate and subcutaneous sumatriptan for treatment of acute migraine attacks: doubleblind randomized clinical trial. Iran J Med Sci 2014; 39: Ghaderibarmi F, Tavakkoli N, Togha M. Intravenous valproate versus subcutaneous sumatriptan in acute migraine attack. Acta Med Iran 2015;53: Friedman BW, Greenwald P, Bania TC, et al. Randomized trial of IV dexamethasone for acute migraine in the emergency department. Neurology 2007;69: Bakhshayesh B, Seyed Saadat SM, Rezania K, Hatamian H, Hossieninezhad M. A randomized open-label study of sodium valproate vs sumatriptan and metoclopramide for prolonged migraine headache. Am J Emerg Med 2013;31: Frazee LA, Foraker KC. Use of intravenous valproic acid for acute migraine. Ann Pharmacother 2008;42: Donaldson D, Sundermann R, Jackson R, Bastani A. Intravenous dexamethasone vs placebo as adjunctive therapy to reduce the recurrence rate of acute migraine headaches: a multicenter, double-blinded, placebo-controlled randomized 144
8 Narges Karimi, et al. clinical trial. Am J Emerg Med 2008;26: Norton J. Use of intravenous valproate sodium in status migraine. Headache 2000;40: Stillman MJ, Zajac D, Rybicki LA. Treatment of primary headache disorders with intravenous valproate: initial outpatient experience. Headache 2004;44: Edwards KR, Norton J, Behnke M. Comparison of intravenous valproate versus intramuscular dihydroergotamine and metoclopramide for acute treatment of migraine headache. Headache 2001;41: Tanen DA, Miller S, French T, Riffenburgh RH. Intravenous sodium valproate versus prochlorperazine for the emergency department treatment of acute migraine headaches: a prospective, randomized, double-blind trial. Ann Emerg Med 2003;41: Waberzinek G, Markova J, Mastik J. Safety and efficacy of intravenous sodium valproate in the treatment of acute migraine. Neuro Endocrinol Lett 2007;28: Foroughipour M, Petramfar P, Saeidi M, Akbarnezhad MA, Ebrahimzadeh S. Efficacy of intravenous Dexamethasone in treatment of acute migraine headache: a randomized clinical trial running title. Iran J Otorhinolaryngol 2008;19: Mathew NT, Kailasam J, Meadors L, Chernyschev O, Gentry P. Intravenous valproate sodium (depacon) aborts migraine rapidly: a preliminary report. Headache 2000;40: Sjaastad O, Fredriksen TA, Petersen HC, Bakketeig LS. Grading of headache intensity: a proposal. J Headache Pain 2002;3: Leniger T, Pageler L, Stude P, Diener HC, Limmroth V. Comparison of intravenous valproate with intravenous lysine-acetylsalicylic acid in acute migraine attacks. Headache 2005;45: Blau JN. Water deprivation: a new migraine precipitant. Headache 2005;45: Limdi NA, Knowlton RK, Cofield SS, et al. Safety of rapid intravenous loading of valproate. Epilepsia 2007;48: Innes GD, Macphail I, Dillon EC, Metcalfe C, Gao M. Dexamethasone prevents relapse after emergency department treatment of acute migraine: a randomized clinical trial. CJEM 1999;1: Shahien R, Saleh SA, Bowirrat A. Intravenous sodium valproate aborts migraine headaches rapidly. Acta Neurol Scand 2011;123: Clin Exp Emerg Med 2017;4(3):
What A Headache! Theresa Biesiada March 8, 2012
 What A Headache! Theresa Biesiada March 8, 2012 Objectives Describe the EM relevance of headaches and migraines Discuss the rationale for steroid therapy Review the evidence Conclusions My inspiration
What A Headache! Theresa Biesiada March 8, 2012 Objectives Describe the EM relevance of headaches and migraines Discuss the rationale for steroid therapy Review the evidence Conclusions My inspiration
ORIGINAL ARTICLE INTRODUCTION
 ORIGINAL ARTICLE The Efficacy of Propofol vs. Subcutaneous Sumatriptan for Treatment of Acute Migraine Headaches in the Emergency Department: A Double-Blinded Clinical Trial Hossein Moshtaghion, MD*; Najmeh
ORIGINAL ARTICLE The Efficacy of Propofol vs. Subcutaneous Sumatriptan for Treatment of Acute Migraine Headaches in the Emergency Department: A Double-Blinded Clinical Trial Hossein Moshtaghion, MD*; Najmeh
Inpatient Treatment of Status Migraine With Dihydroergotamine in Children and Adolescents
 Headache 2008 the Authors Journal compilation 2008 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2008.01293.x Published by Wiley Periodicals, Inc. Brief Communication Inpatient Treatment
Headache 2008 the Authors Journal compilation 2008 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2008.01293.x Published by Wiley Periodicals, Inc. Brief Communication Inpatient Treatment
Original Article. Keywords Migraine Sodium valproate Sumatriptan. Introduction
 IJMS Vol 39, No 2, Supplement March 2014 Original Article Comparison between Intravenous Sodium Valproate and Subcutaneous Sumatriptan for Treatment of Acute Migraine Attacks; Double- Blind Randomized
IJMS Vol 39, No 2, Supplement March 2014 Original Article Comparison between Intravenous Sodium Valproate and Subcutaneous Sumatriptan for Treatment of Acute Migraine Attacks; Double- Blind Randomized
Comparison of Intravenous Metoclopramide and Acetaminophen in Primary Headaches: a Randomized Controlled Trial
 1 Emergency (2015); 3 (*): ***-*** ORIGINAL RESEARCH Comparison of Intravenous Metoclopramide and Acetaminophen in Primary Headaches: a Randomized Controlled Trial Gholamreza Faridaalaee 1, Seyed Hesam
1 Emergency (2015); 3 (*): ***-*** ORIGINAL RESEARCH Comparison of Intravenous Metoclopramide and Acetaminophen in Primary Headaches: a Randomized Controlled Trial Gholamreza Faridaalaee 1, Seyed Hesam
Dose: Metoclopramide -0.1 mg/kg (max 10 mg) IV, over 15 minutes
 Metoclopramide for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine, is metoclopramide an effective treatment? Question Originator: Migraine
Metoclopramide for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine, is metoclopramide an effective treatment? Question Originator: Migraine
The ALTO Program: A Strategy to Reduce Opioid use in the Emergency Department
 The ALTO Program: A Strategy to Reduce Opioid use in the Emergency Department The ALTO Program: A Strategy to Reduce Opioid use in the Emergency Department Steven F. Nerenberg, Pharm.D. Clinical Assistant
The ALTO Program: A Strategy to Reduce Opioid use in the Emergency Department The ALTO Program: A Strategy to Reduce Opioid use in the Emergency Department Steven F. Nerenberg, Pharm.D. Clinical Assistant
Specific Care Question : Question Originator: Plain Language Summary from The Office of Evidence Based Practice: Conditional Recommendation
 Ketorolac for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine is ketorolac an effective treatment? Question Originator: Migraine Therapy
Ketorolac for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine is ketorolac an effective treatment? Question Originator: Migraine Therapy
Migraine is is one of the most common chronic neurological
 Review Article Rescue Treatment for Migraine Headache in Emergency Department Part 2: Role of Antiepileptic, Magnesium, Corticosteroids, and Discharge Care Abstract Migraine is a common chronic neurological
Review Article Rescue Treatment for Migraine Headache in Emergency Department Part 2: Role of Antiepileptic, Magnesium, Corticosteroids, and Discharge Care Abstract Migraine is a common chronic neurological
Abstract The aim of this prospective,
 J Headache Pain (2005) 6:143 148 DOI 10.1007/s10194-005-0169-y ORIGINAL Zulfi Engindeniz Celaleddin Demircan Necdet Karli Erol Armagan Mehtap Bulut Tayfun Aydin Mehmet Zarifoglu Intramuscular tramadol
J Headache Pain (2005) 6:143 148 DOI 10.1007/s10194-005-0169-y ORIGINAL Zulfi Engindeniz Celaleddin Demircan Necdet Karli Erol Armagan Mehtap Bulut Tayfun Aydin Mehmet Zarifoglu Intramuscular tramadol
Migraine Types and Triggering Factors in Children
 original ARTICLE Migraine Types and Triggering Factors in Children How to Cite this Article: Nejad Biglari H, Karimzadeh P, Mohammadi Kord-kheyli M, Hashemi SM. Migraine Types and Triggering Factors in
original ARTICLE Migraine Types and Triggering Factors in Children How to Cite this Article: Nejad Biglari H, Karimzadeh P, Mohammadi Kord-kheyli M, Hashemi SM. Migraine Types and Triggering Factors in
A. At least five attacks fulfilling criteria in B, C, D, and E. B. Attack lasts 4 to 72 hours with or without treatment.
 Soleimanpour et al. BMC Neurology 2012, 12:114 RESEARCH ARTICLE Open Access Effectiveness of intravenous Dexamethasone versus Propofol for pain relief in the migraine headache: A prospective double blind
Soleimanpour et al. BMC Neurology 2012, 12:114 RESEARCH ARTICLE Open Access Effectiveness of intravenous Dexamethasone versus Propofol for pain relief in the migraine headache: A prospective double blind
Prednisone vs. placebo in withdrawal therapy following medication overuse headache
 doi:10.1111/j.1468-2982.2007.01488.x Prednisone vs. placebo in withdrawal therapy following medication overuse headache L Pageler 1,2, Z Katsarava 2, HC Diener 2 & V Limmroth 1,2 1 Department of Neurology,
doi:10.1111/j.1468-2982.2007.01488.x Prednisone vs. placebo in withdrawal therapy following medication overuse headache L Pageler 1,2, Z Katsarava 2, HC Diener 2 & V Limmroth 1,2 1 Department of Neurology,
Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial
 J Headache Pain (2007) 8:175 179 DOI 10.1007/s10194-007-0386-7 ORIGINAL Usha Kant Misra Jayantee Kalita Rama Kant Yadav Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial Received:
J Headache Pain (2007) 8:175 179 DOI 10.1007/s10194-007-0386-7 ORIGINAL Usha Kant Misra Jayantee Kalita Rama Kant Yadav Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial Received:
Treatment of Headache in the ED
 Treatment of Headache in the ED Benjamin W. Friedman, MD, MS, FAAEM Associate professor of Emergency Medicine Albert Einstein College of Medicine Montefiore Medical Center Disclosure Topics of Discussion
Treatment of Headache in the ED Benjamin W. Friedman, MD, MS, FAAEM Associate professor of Emergency Medicine Albert Einstein College of Medicine Montefiore Medical Center Disclosure Topics of Discussion
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 CGRP Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
CGRP Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 CGRP Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
CGRP Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
Chronic Migraine in Primary Care. December 11 th, 2017 Werner J. Becker University of Calgary
 Chronic Migraine in Primary Care December 11 th, 2017 Werner J. Becker University of Calgary Disclosures Faculty: Werner J. Becker Relationships with commercial interests: Grants/Research Support: Clinical
Chronic Migraine in Primary Care December 11 th, 2017 Werner J. Becker University of Calgary Disclosures Faculty: Werner J. Becker Relationships with commercial interests: Grants/Research Support: Clinical
Despite the widespread use of triptans ... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability
 ... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability Randal L. Von Seggern, PharmD, BCPS Abstract Objective: This article summarizes preclinical and clinical data for
... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability Randal L. Von Seggern, PharmD, BCPS Abstract Objective: This article summarizes preclinical and clinical data for
Zolmitriptan is effective and well tolerated in Japanese patients with migraine: a dose response study
 is effective and well tolerated in Japanese patients with migraine: a dose response study F Sakai 1, M Iwata 2, K Tashiro 3, Y Itoyama 4, S Tsuji 5, Y Fukuuchi 6, G Sobue 7, K Nakashima 8 & M Morimatsu
is effective and well tolerated in Japanese patients with migraine: a dose response study F Sakai 1, M Iwata 2, K Tashiro 3, Y Itoyama 4, S Tsuji 5, Y Fukuuchi 6, G Sobue 7, K Nakashima 8 & M Morimatsu
...SELECTED ABSTRACTS...
 The following abstracts, from medical journals containing literature on migraine management, were selected for their relevance to this Special Report supplement. Two Sumatriptan Studies Two double-blind
The following abstracts, from medical journals containing literature on migraine management, were selected for their relevance to this Special Report supplement. Two Sumatriptan Studies Two double-blind
A new questionnaire for assessment of adverse events associated with triptans: methods of assessment influence the results. Preliminary results
 J Headache Pain (2004) 5:S112 S116 DOI 10.1007/s10194-004-0123-4 Michele Feleppa Fred D. Sheftell Luciana Ciannella Amedeo D Alessio Giancarlo Apice Nino N. Capobianco Donato M.T. Saracino Walter Di Iorio
J Headache Pain (2004) 5:S112 S116 DOI 10.1007/s10194-004-0123-4 Michele Feleppa Fred D. Sheftell Luciana Ciannella Amedeo D Alessio Giancarlo Apice Nino N. Capobianco Donato M.T. Saracino Walter Di Iorio
medications. This was an openlabel study consisting of patients with migraines who historically failed to respond to oral triptan
 J Headache Pain (2007) 8:13 18 DOI 10.1007/s10194-007-0354-7 ORIGINAL Seymour Diamond Fred G. Freitag Alexander Feoktistov George Nissan Sumatriptan 6 mg subcutaneous as an effective migraine treatment
J Headache Pain (2007) 8:13 18 DOI 10.1007/s10194-007-0354-7 ORIGINAL Seymour Diamond Fred G. Freitag Alexander Feoktistov George Nissan Sumatriptan 6 mg subcutaneous as an effective migraine treatment
Preventive Effect of Greater Occipital Nerve Block on Severity and Frequency of Migraine Headache
 Proceeding S.Z.P.G.M.I. Vol: 31(2): pp. 75-79, 2017. Preventive effect of Greater Occipital Nerve Block on Severity and Frequency of Migraine Headache Dr. Syed Mehmood Ali, Dr. Mudassar Aslam, Dr. Dawood
Proceeding S.Z.P.G.M.I. Vol: 31(2): pp. 75-79, 2017. Preventive effect of Greater Occipital Nerve Block on Severity and Frequency of Migraine Headache Dr. Syed Mehmood Ali, Dr. Mudassar Aslam, Dr. Dawood
MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache
 MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache Measure Description Percentage of patients age 12 years and older with a diagnosis of migraine who were prescribed a guideline recommended
MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache Measure Description Percentage of patients age 12 years and older with a diagnosis of migraine who were prescribed a guideline recommended
Adult & Pediatric Patients. Stanford Health Care, Division Pain Medicine
 Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
Overuse of barbiturate and opioid containing medications for primary headache disorders Description
 Measure Title Overuse of barbiturate and opioid containing medications for primary headache disorders Description Percentage of s age 12 years and older with a diagnosis of primary headache who were prescribed
Measure Title Overuse of barbiturate and opioid containing medications for primary headache disorders Description Percentage of s age 12 years and older with a diagnosis of primary headache who were prescribed
Parenteral Dihydroergotamine for Acute Migraine Headache: A Systematic Review of the Literature
 NEUROLOGY/ORIGINAL RESEARCH Parenteral Dihydroergotamine for Acute Migraine Headache: A Systematic Review of the Literature Ian Colman, MSc Michael D. Brown, MD, MSc Grant D. Innes, MD Eric Grafstein,
NEUROLOGY/ORIGINAL RESEARCH Parenteral Dihydroergotamine for Acute Migraine Headache: A Systematic Review of the Literature Ian Colman, MSc Michael D. Brown, MD, MSc Grant D. Innes, MD Eric Grafstein,
Value of postmarketing surveillance studies in achieving a complete picture of antimigraine agents: using almotriptan as an example
 J Headache Pain (2006) 7:27 33 DOI 10.1007/s10194-006-0266-6 ORIGINAL Julio Pascual Hans-Christoph Diener Hélène Massiou Value of postmarketing surveillance studies in achieving a complete picture of antimigraine
J Headache Pain (2006) 7:27 33 DOI 10.1007/s10194-006-0266-6 ORIGINAL Julio Pascual Hans-Christoph Diener Hélène Massiou Value of postmarketing surveillance studies in achieving a complete picture of antimigraine
MEASURE #3: PREVENTIVE MIGRAINE MEDICATION PRESCRIBED Headache
 MEASURE #3: PREVENTIVE MIGRAINE MEDICATION PRESCRIBED Headache Measure Description Percentage of patients age 18 years old and older diagnosed with migraine headache whose migraine frequency is 4 migraine
MEASURE #3: PREVENTIVE MIGRAINE MEDICATION PRESCRIBED Headache Measure Description Percentage of patients age 18 years old and older diagnosed with migraine headache whose migraine frequency is 4 migraine
Disclosures. Triptans for Kids 5/16/13
 5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation.
 I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation. In 1962, Bille published landmark epidemiologic survey of headache among 9,000 school
I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation. In 1962, Bille published landmark epidemiologic survey of headache among 9,000 school
How do we treat migraine? New SIGN Guidelines
 How do we treat migraine? New SIGN Guidelines Managing your migraine Migraine Trust, Edinburgh 2018 Callum Duncan Consultant Neurologist Aberdeen Royal Infirmary Chair SIGN Guideline 155 Premonitory Mood
How do we treat migraine? New SIGN Guidelines Managing your migraine Migraine Trust, Edinburgh 2018 Callum Duncan Consultant Neurologist Aberdeen Royal Infirmary Chair SIGN Guideline 155 Premonitory Mood
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The use of combination therapies in the acute management of migraine
 REVIEW The use of combination therapies in the acute management of migraine Abouch Valenty Krymchantowski Headache Center of Rio, Rio de Janeiro, Brazil; Outpatient Headache Unit of the Instituto de Neurologia
REVIEW The use of combination therapies in the acute management of migraine Abouch Valenty Krymchantowski Headache Center of Rio, Rio de Janeiro, Brazil; Outpatient Headache Unit of the Instituto de Neurologia
Clinical Trial Results Database Page 1
 Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Corporation Generic Drug Name Therapeutic Area of Trial Major Depressive Disorder (MDD) Approved Indication Treatment of major depressive
Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Corporation Generic Drug Name Therapeutic Area of Trial Major Depressive Disorder (MDD) Approved Indication Treatment of major depressive
Treating Headache Recurrence After Emergency Department Discharge: A Randomized Controlled Trial of Naproxen Versus Sumatriptan
 PAIN MANAGEMENT/ORIGINAL RESEARCH Treating Headache Recurrence After Emergency Department Discharge: A Randomized Controlled Trial of Naproxen Versus Sumatriptan Benjamin W. Friedman, MD, MS Clemencia
PAIN MANAGEMENT/ORIGINAL RESEARCH Treating Headache Recurrence After Emergency Department Discharge: A Randomized Controlled Trial of Naproxen Versus Sumatriptan Benjamin W. Friedman, MD, MS Clemencia
Preventive Effect of Greater Occipital Nerve Block on Severity and Frequency of Migraine Headache
 Global Journal of Health Science; Vol. 6, No. 6; 2014 ISSN 1916-9736 E-ISSN 1916-9744 Published by Canadian Center of Science and Education Preventive Effect of Greater Occipital Nerve Block on Severity
Global Journal of Health Science; Vol. 6, No. 6; 2014 ISSN 1916-9736 E-ISSN 1916-9744 Published by Canadian Center of Science and Education Preventive Effect of Greater Occipital Nerve Block on Severity
Zonisamide for migraine prophylaxis in refractory patients
 Thomas Jefferson University Jefferson Digital Commons Department of Neurology Faculty Papers Department of Neurology March 2006 Zonisamide for migraine prophylaxis in refractory patients Avi Ashkenazi
Thomas Jefferson University Jefferson Digital Commons Department of Neurology Faculty Papers Department of Neurology March 2006 Zonisamide for migraine prophylaxis in refractory patients Avi Ashkenazi
Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, II. Management of Refractory Headaches
 Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, 2016 II. Management of Refractory Headaches Case presentation 1: A case of intractable daily-persistent headache Keio University School
Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, 2016 II. Management of Refractory Headaches Case presentation 1: A case of intractable daily-persistent headache Keio University School
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D.
 Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
Migraine much more than just a headache
 Migraine much more than just a headache Session hosted by Teva UK Limited PUU4 11:15 12:15 UK/NHSS/18/0021b Date of Preparation: August 2018 The views expressed in this presentation are those of the speaker
Migraine much more than just a headache Session hosted by Teva UK Limited PUU4 11:15 12:15 UK/NHSS/18/0021b Date of Preparation: August 2018 The views expressed in this presentation are those of the speaker
Faculty Disclosures. Learning Objectives. Acute Treatment Strategies
 WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
Drug Class Review Triptans
 Drug Class Review Triptans April 2013 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness and harms of different
Drug Class Review Triptans April 2013 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness and harms of different
Dexamethasone combined with other antiemetics for prophylaxis after laparoscopic cholecystectomy
 Original Research Article Dexamethasone combined with other antiemetics for prophylaxis after laparoscopic cholecystectomy T. Uma Maheswara Rao * Associate Professor, Department of Surgery, Konaseema Institute
Original Research Article Dexamethasone combined with other antiemetics for prophylaxis after laparoscopic cholecystectomy T. Uma Maheswara Rao * Associate Professor, Department of Surgery, Konaseema Institute
ISSN doi: /head VC 2015 American Headache Society Published by Wiley Periodicals, Inc.
 ISSN 0017-8748 Headache doi: 10.1111/head.12746 VC 2015 American Headache Society Published by Wiley Periodicals, Inc. Review Article Acute Treatment Therapies for Pediatric Migraine: A Qualitative Systematic
ISSN 0017-8748 Headache doi: 10.1111/head.12746 VC 2015 American Headache Society Published by Wiley Periodicals, Inc. Review Article Acute Treatment Therapies for Pediatric Migraine: A Qualitative Systematic
Is Yoga an Effective Treatment for Reducing the Frequency of Episodic Migraine?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2018 Is Yoga an Effective Treatment for Reducing
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2018 Is Yoga an Effective Treatment for Reducing
ADVANCES IN MIGRAINE MANAGEMENT
 ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
A comparison of Ketoprofen and Diclofenac for acute musculoskeletal pain relief: a prospective randomised clinical trial
 Hong Kong Journal of Emergency Medicine A comparison of Ketoprofen and Diclofenac for acute musculoskeletal pain relief: a prospective randomised clinical trial P Ng, CW Kam, HH Yau Objectives: To compare
Hong Kong Journal of Emergency Medicine A comparison of Ketoprofen and Diclofenac for acute musculoskeletal pain relief: a prospective randomised clinical trial P Ng, CW Kam, HH Yau Objectives: To compare
Migraine: Developing Drugs for Acute Treatment Guidance for Industry
 Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
TABLE 1. Current Diagnostic Criteria for Migraine Without Aura 2 A. At least 5 attacks fulfilling criteria B-D B. Headache attacks lasting 4-72 hours
 ANSWERS CONCISE TO FREQUENTLY REVIEW ASKED QUESTIONS FOR CLINICIANS ABOUT MIGRAINE Answers to Frequently Asked Questions About Migraine IVAN GARZA, MD, AND JERRY W. SWANSON, MD Migraine is a common primary
ANSWERS CONCISE TO FREQUENTLY REVIEW ASKED QUESTIONS FOR CLINICIANS ABOUT MIGRAINE Answers to Frequently Asked Questions About Migraine IVAN GARZA, MD, AND JERRY W. SWANSON, MD Migraine is a common primary
Drug Class Review on Triptans
 Drug Class Review on UPDATED FINAL REPORT #1 December 2003 Mark Helfand, MD, MPH Kim Peterson, MS Oregon Evidence-based Practice Center Oregon Health & Science University Table of Contents Introduction
Drug Class Review on UPDATED FINAL REPORT #1 December 2003 Mark Helfand, MD, MPH Kim Peterson, MS Oregon Evidence-based Practice Center Oregon Health & Science University Table of Contents Introduction
Aleksandra Radojičić. Headache Center, Neurology Clinic, Clinical Center of Serbia
 European Headache School Belgrade 2012 MEDICATION OVERUSE HEADACHE Aleksandra Radojičić Headache Center, Neurology Clinic, Clinical Center of Serbia Historical data First cases were described in XVII century
European Headache School Belgrade 2012 MEDICATION OVERUSE HEADACHE Aleksandra Radojičić Headache Center, Neurology Clinic, Clinical Center of Serbia Historical data First cases were described in XVII century
Outpatient Headache Care Guideline
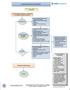 1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
MIGRAINE A MYSTERY HEADACHE
 MIGRAINE A MYSTERY HEADACHE The migraine is a chronic neurological disease that is characterized by moderate to severe episodes of headache that is mostly associated with other central nervous system (CNS)
MIGRAINE A MYSTERY HEADACHE The migraine is a chronic neurological disease that is characterized by moderate to severe episodes of headache that is mostly associated with other central nervous system (CNS)
Jessica Ailani MD FAHS Director, Georgetown Headache Center Associate Professor Neurology Medstar Georgetown University Hospital
 Jessica Ailani MD FAHS Director, Georgetown Headache Center Associate Professor Neurology Medstar Georgetown University Hospital Honorarium from Current Pain and Headache Reports; Section Editor Unusual
Jessica Ailani MD FAHS Director, Georgetown Headache Center Associate Professor Neurology Medstar Georgetown University Hospital Honorarium from Current Pain and Headache Reports; Section Editor Unusual
THE WOMAN WHO COULD NOT DECIDE WHICH MEDICATION TO TAKE
 Rapoport Ch 05.qxd 10/15/08 1:06 PM Page 25 CHAPTER 5 THE WOMAN WHO COULD NOT DECIDE WHICH MEDICATION TO TAKE ALLAN PURDY, MD, FRCPC FRED SHEFTELL, MD ALAN RAPOPORT, MD STEWART J. TEPPER, MD Case History
Rapoport Ch 05.qxd 10/15/08 1:06 PM Page 25 CHAPTER 5 THE WOMAN WHO COULD NOT DECIDE WHICH MEDICATION TO TAKE ALLAN PURDY, MD, FRCPC FRED SHEFTELL, MD ALAN RAPOPORT, MD STEWART J. TEPPER, MD Case History
Migranal Nasal Spray. Migranal Nasal Spray (dihydroergotamine) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: June 22, 2017 Migranal Nasal Spray
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: June 22, 2017 Migranal Nasal Spray
Efficacy of Levetiracetam: A Review of Three Pivotal Clinical Trials
 Epilepsia, 42(Suppl. 4):31 35, 2001 Blackwell Science, Inc. International League Against Epilepsy Efficacy of : A Review of Three Pivotal Clinical Trials Michael Privitera University of Cincinnati Medical
Epilepsia, 42(Suppl. 4):31 35, 2001 Blackwell Science, Inc. International League Against Epilepsy Efficacy of : A Review of Three Pivotal Clinical Trials Michael Privitera University of Cincinnati Medical
ClinialTrials.gov Identifier: Sponsor/company: sanofi-aventis
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinialTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinialTrials.gov
11. HEADACHE 1. Pablo Lapuerta, MD, Steven Asch, MD, MPH, and Kenneth Clark, MD, MPH
 11. HEADACHE 1 Pablo Lapuerta, MD, Steven Asch, MD, MPH, and Kenneth Clark, MD, MPH We identified articles on the evaluation and management of headache by conducting a MEDLINE search of English language
11. HEADACHE 1 Pablo Lapuerta, MD, Steven Asch, MD, MPH, and Kenneth Clark, MD, MPH We identified articles on the evaluation and management of headache by conducting a MEDLINE search of English language
Triptans: Nonresponse, Recurrence, and Serious AEs for Many Patients
 Efficacy, Safety, and Tolerability of Rimegepant 75 mg, an Oral CGRP Receptor Antagonist, for the Acute Treatment of Migraine: Results from a Phase 3, Double-Blind, Randomized, Placebo-Controlled Trial,
Efficacy, Safety, and Tolerability of Rimegepant 75 mg, an Oral CGRP Receptor Antagonist, for the Acute Treatment of Migraine: Results from a Phase 3, Double-Blind, Randomized, Placebo-Controlled Trial,
Caspian Journal of Neurological Sciences "Caspian J Neurol Sci" Journal Homepage:
 Caspian Journal of Neurological Sciences "Caspian J Neurol Sci" Journal Homepage: http://cjns.gums.ac.ir Research aper: or for Treatment of Medication Overuse Headache: A Randomized, Double- Blind Clinical
Caspian Journal of Neurological Sciences "Caspian J Neurol Sci" Journal Homepage: http://cjns.gums.ac.ir Research aper: or for Treatment of Medication Overuse Headache: A Randomized, Double- Blind Clinical
Management options for Migraine. Prof. Dr. Khwaja Nazimuddin Head Dept. of Internal Medicine BIRDEM
 Management options for Migraine Prof. Dr. Khwaja Nazimuddin Head Dept. of Internal Medicine BIRDEM Assessment The Migraine Disability Assessment Score MIDAS Complete loss of work Partial loss of work Off
Management options for Migraine Prof. Dr. Khwaja Nazimuddin Head Dept. of Internal Medicine BIRDEM Assessment The Migraine Disability Assessment Score MIDAS Complete loss of work Partial loss of work Off
A Prospective, Randomized Trial of Intravenous Prochlorperazine Versus Subcutaneous Sumatriptan in Acute Migraine Therapy in the Emergency Department
 PAIN MANAGEMENT/ORIGINAL RESEARCH A Prospective, Randomized Trial of Intravenous Prochlorperazine Versus Subcutaneous Sumatriptan in Acute Migraine Therapy in the Emergency Department Mark A. Kostic, MD,
PAIN MANAGEMENT/ORIGINAL RESEARCH A Prospective, Randomized Trial of Intravenous Prochlorperazine Versus Subcutaneous Sumatriptan in Acute Migraine Therapy in the Emergency Department Mark A. Kostic, MD,
Migraine Management. Roger Cady, MD Headache Care Center Springfield, MO
 Migraine Management Roger Cady, MD Headache Care Center Springfield, MO Disclosures Objectives The evolution of migraine From benign episodic (benign) headache to potentially a devastating chronic disease
Migraine Management Roger Cady, MD Headache Care Center Springfield, MO Disclosures Objectives The evolution of migraine From benign episodic (benign) headache to potentially a devastating chronic disease
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Aimovig) Reference Number: CP.PHAR.128 Effective Date: 07.10.18 Last Review Date: 08.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Aimovig) Reference Number: CP.PHAR.128 Effective Date: 07.10.18 Last Review Date: 08.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Drug Class Review on the Triptans
 Drug Class Review on the Final Report March 7, 2003 Expires October 31, 2003 Mark Helfand, MD, MPH Kim Peterson, MS Produced by Oregon Health & Science University 3181 SW Sam Jackson Park Road Mailcode:
Drug Class Review on the Final Report March 7, 2003 Expires October 31, 2003 Mark Helfand, MD, MPH Kim Peterson, MS Produced by Oregon Health & Science University 3181 SW Sam Jackson Park Road Mailcode:
Regulatory Status FDA approved indication: Migranal Nasal Spray is indicated for the acute treatment of migraine headaches with or without aura (1).
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: November 30, 2018 Migranal Nasal Spray
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: November 30, 2018 Migranal Nasal Spray
Drug Class Review Triptans
 Drug Class Review Triptans Preliminary Update Scan #3 February 2014 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness
Drug Class Review Triptans Preliminary Update Scan #3 February 2014 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness
The effect of haloperidol administration on relieving cluster headache, probable role of dopaminergic pathway; a double blind clinical trial
 72 Journal of Medical Physiology. 2016; 1(2): 72-77. Original research The effect of haloperidol administration on relieving cluster headache, probable role of dopaminergic pathway; a double blind clinical
72 Journal of Medical Physiology. 2016; 1(2): 72-77. Original research The effect of haloperidol administration on relieving cluster headache, probable role of dopaminergic pathway; a double blind clinical
Nee Kong CHEW, Chong Tin TAN, Heng Thay CHONG, Yau Hoong CHAN, Kuoh Ren CHONG, Hee Hong LAM, Yan Beng NGE, Chek Tung NGO.
 Neurol J Southeast Asia 2001; 6 : 13 17 Ramadan headache Nee Kong CHEW, Chong Tin TAN, Heng Thay CHONG, Yau Hoong CHAN, Kuoh Ren CHONG, Hee Hong LAM, Yan Beng NGE, Chek Tung NGO. Department of Medicine,
Neurol J Southeast Asia 2001; 6 : 13 17 Ramadan headache Nee Kong CHEW, Chong Tin TAN, Heng Thay CHONG, Yau Hoong CHAN, Kuoh Ren CHONG, Hee Hong LAM, Yan Beng NGE, Chek Tung NGO. Department of Medicine,
UTILIZATION MANAGEMENT CRITERIA
 SUMATRIPTAN (ALSUMA, IMITREX, SUMAVEL DOSEPRO, ZECUITY ) UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: BRAND (generic) NAME: Serotonin 5-HT1 receptor agonists Imitrex (sumatriptan), Alsuma (sumatriptan),
SUMATRIPTAN (ALSUMA, IMITREX, SUMAVEL DOSEPRO, ZECUITY ) UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: BRAND (generic) NAME: Serotonin 5-HT1 receptor agonists Imitrex (sumatriptan), Alsuma (sumatriptan),
(For National Authority Use Only) Name of Study Drug: to Part of Dossier:
 2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: Vicodin CR Name of Active Ingredient: Page: Hydrocodone/Acetaminophen
2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: Vicodin CR Name of Active Ingredient: Page: Hydrocodone/Acetaminophen
Ce12halalgia. Effectiveness of oral diclofenac in the acute treatment of common migraine attacks: a double-blind study versus placebo
 Effectiveness of oral diclofenac in the acute treatment of common migraine attacks: a double-blind study versus placebo Helene Massiou, Dominique Serrurier, Odile Lasserre, Marie-Germaine Bousser Ce12halalgia
Effectiveness of oral diclofenac in the acute treatment of common migraine attacks: a double-blind study versus placebo Helene Massiou, Dominique Serrurier, Odile Lasserre, Marie-Germaine Bousser Ce12halalgia
Is Acupuncture an Effective Treatment for Acute Migraine?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2015 Is Acupuncture an Effective Treatment
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2015 Is Acupuncture an Effective Treatment
HEADACHE: Benign or Severe Dr Gobinda Chandra Roy
 HEADACHE: Benign or Severe Dr Gobinda Chandra Roy Associate Professor, Department of Medicine, Shaheed Suhrawardy Medical College and Hospital Outlines 1. Introduction 2. Classification of headache 3.
HEADACHE: Benign or Severe Dr Gobinda Chandra Roy Associate Professor, Department of Medicine, Shaheed Suhrawardy Medical College and Hospital Outlines 1. Introduction 2. Classification of headache 3.
Early treatment of a migraine attack while pain is still mild increases the efficacy of sumatriptan
 Blackwell Science, LtdOxford, UKCHACephalalgia1468-2982Blackwell Science, 20042411925933Original ArticleEarly treatment of migraine with sumatriptanj Scholpp et al. Early treatment of a migraine attack
Blackwell Science, LtdOxford, UKCHACephalalgia1468-2982Blackwell Science, 20042411925933Original ArticleEarly treatment of migraine with sumatriptanj Scholpp et al. Early treatment of a migraine attack
Çiçek Wöber-Bingöl HEADACHE UNIT FOR CHILDREN AND ADOLESCENCE
 Headache in children and adolescents Çiçek Wöber-Bingöl HEADACHE UNIT FOR CHILDREN AND ADOLESCENCE Dept. of Psychiatry of Childhood and Adolescence Medical University of Vienna, Vienna, Austria Impact
Headache in children and adolescents Çiçek Wöber-Bingöl HEADACHE UNIT FOR CHILDREN AND ADOLESCENCE Dept. of Psychiatry of Childhood and Adolescence Medical University of Vienna, Vienna, Austria Impact
ปวดศ รษะมา 5 ป ก นยาแก ปวดก ย งไม ข น นพ.พาว ฒ เมฆว ช ย โรงพยาบาลนครราชส มา
 ปวดศ รษะมา 5 ป ก นยาแก ปวดก ย งไม ข น นพ.พาว ฒ เมฆว ช ย โรงพยาบาลนครราชส มา 1 CONTENT 1 2 3 Chronic Daily Headache Medical Overused Headache Management Headaches are one of the most common symptoms List
ปวดศ รษะมา 5 ป ก นยาแก ปวดก ย งไม ข น นพ.พาว ฒ เมฆว ช ย โรงพยาบาลนครราชส มา 1 CONTENT 1 2 3 Chronic Daily Headache Medical Overused Headache Management Headaches are one of the most common symptoms List
UNDERSTANDING CHRONIC MIGRAINE. Learn about diagnosis, management, and treatment options for this headache condition
 UNDERSTANDING CHRONIC MIGRAINE Learn about diagnosis, management, and treatment options for this headache condition 1 What We re Going to Cover Today The symptoms and phases of migraine Differences between
UNDERSTANDING CHRONIC MIGRAINE Learn about diagnosis, management, and treatment options for this headache condition 1 What We re Going to Cover Today The symptoms and phases of migraine Differences between
Current Migraine Treatment Therapy. Daniel Kassicieh, DO, FAAN
 Current Migraine Treatment Therapy Daniel Kassicieh, DO, FAAN Migraine a Disease Process Migraines are a chronic disease process similar to many other chronic medical conditions Migraine has a low mortality
Current Migraine Treatment Therapy Daniel Kassicieh, DO, FAAN Migraine a Disease Process Migraines are a chronic disease process similar to many other chronic medical conditions Migraine has a low mortality
Immediate-release Hydrocodone/Acetaminophen M Abbreviated Clinical Study Report R&D/08/1020
 2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: Hydrocodone Bitartrate- Acetaminophen (NORCO ) Name of
2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: Hydrocodone Bitartrate- Acetaminophen (NORCO ) Name of
Cinnarizine versus Topiramate in Prophylaxis of Migraines among Children and Adolescents: A Randomized, Double-Blind Clinical Trial
 original ARTICLE Cinnarizine versus Topiramate in Prophylaxis of Migraines among Children and Adolescents: A Randomized, Double-Blind Clinical Trial How to Cite This Article: Ashrafi MR, Najafi Z, Shafiei
original ARTICLE Cinnarizine versus Topiramate in Prophylaxis of Migraines among Children and Adolescents: A Randomized, Double-Blind Clinical Trial How to Cite This Article: Ashrafi MR, Najafi Z, Shafiei
ACUTE TREATMENT FOR MIGRAINE. Cristina Tassorelli
 The European Headache School 2012 ACUTE TREATMENT FOR MIGRAINE Cristina Tassorelli Headache Science Centre, IRCCS Neurological Institute C. Mondino Foundation - Pavia University Centre for Adaptive Disorders
The European Headache School 2012 ACUTE TREATMENT FOR MIGRAINE Cristina Tassorelli Headache Science Centre, IRCCS Neurological Institute C. Mondino Foundation - Pavia University Centre for Adaptive Disorders
Treatment Of Medication. Overuse Headache
 7 November 2012 BASH GPwSI Meeting Lecture title... Treatment Of Medication Dr... Overuse Headache Dr Marcus Lewis Dr... The National Migraine Centre International Headache Society Diagnostic criteria
7 November 2012 BASH GPwSI Meeting Lecture title... Treatment Of Medication Dr... Overuse Headache Dr Marcus Lewis Dr... The National Migraine Centre International Headache Society Diagnostic criteria
Guidance for Industry Migraine: Developing Drugs for Acute Treatment
 Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Get ahead of the ACHE: Monoclonal Antibodies in Migraine Prevention
 Get ahead of the ACHE: Monoclonal Antibodies in Migraine Prevention Amanda Janisch, PharmD PGY2 Ambulatory Care Pharmacy Resident MCHS SWMN, Mankato, MN 2018 MFMER slide-1 Disclosures No financial interest
Get ahead of the ACHE: Monoclonal Antibodies in Migraine Prevention Amanda Janisch, PharmD PGY2 Ambulatory Care Pharmacy Resident MCHS SWMN, Mankato, MN 2018 MFMER slide-1 Disclosures No financial interest
MEASURE #4: Overuse of Barbiturate Containing Medications for Primary Headache Disorders Headache
 MEASURE #4: Overuse of Barbiturate Containing Medications for Primary Headache Disorders Headache Measure Description Percentage of patients age 18 years old and older with a diagnosis of primary headache
MEASURE #4: Overuse of Barbiturate Containing Medications for Primary Headache Disorders Headache Measure Description Percentage of patients age 18 years old and older with a diagnosis of primary headache
HEADACHE 1 Pablo Lapuerta, M.D., and Steven Asch, M.D., M.P.H.
 - 253-13. HEADACHE 1 Pablo Lapuerta, M.D., and Steven Asch, M.D., M.P.H. We identified articles on the evaluation and management of headache by conducting a MEDLINE search of English language articles
- 253-13. HEADACHE 1 Pablo Lapuerta, M.D., and Steven Asch, M.D., M.P.H. We identified articles on the evaluation and management of headache by conducting a MEDLINE search of English language articles
ORIGINAL INVESTIGATION. Headache Evaluation and Treatment by Primary Care Physicians in an Emergency Department in the Era of Triptans
 Headache Evaluation and Treatment by Primary Care Physicians in an Emergency Department in the Era of Triptans Morris Maizels, MD ORIGINAL INVESTIGATION Background: Despite advances in treatment, patients
Headache Evaluation and Treatment by Primary Care Physicians in an Emergency Department in the Era of Triptans Morris Maizels, MD ORIGINAL INVESTIGATION Background: Despite advances in treatment, patients
Migraine is defined as recurrent attacks of unilateral
 JOURNAL OF CAFFEINE RESEARCH Volume 5, Number 3, 2015 ª Mary Ann Liebert, Inc. DOI: 10.1089/jcr.2015.0004 Intravenous Caffeine for the Treatment of Acute Migraine: APilotStudy Alireza Baratloo, MD, 1 Ahmed
JOURNAL OF CAFFEINE RESEARCH Volume 5, Number 3, 2015 ª Mary Ann Liebert, Inc. DOI: 10.1089/jcr.2015.0004 Intravenous Caffeine for the Treatment of Acute Migraine: APilotStudy Alireza Baratloo, MD, 1 Ahmed
Clinical Characteristics of Chronic Daily Headache Patients Visit to University Hospital
 Clinical Characteristics of Chronic Daily Headache Patients Visit to University Hospital Jin-Kuk Do, M.D., Hee-Jong Oh, M.D., Dong-Kuck Lee, M.D. Department of Neurology, Taegu Hyosung Catholic University
Clinical Characteristics of Chronic Daily Headache Patients Visit to University Hospital Jin-Kuk Do, M.D., Hee-Jong Oh, M.D., Dong-Kuck Lee, M.D. Department of Neurology, Taegu Hyosung Catholic University
10/17/2017 CHRONIC MIGRAINES BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES PATIENT CASE EPIDEMIOLOGY EPIDEMIOLOGY
 BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017
BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017
Study No.:MPX Title: Rationale: Phase: IIB Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Inpatient Treatment of Refractory Headache
 Inpatient Treatment of Refractory Headache MaryAnn Mays, MD Headache Center Center for Neurological Restoration Neurological Institute Cleveland Clinic Disclosures Speakers Bureau for Allergan, Teva, and
Inpatient Treatment of Refractory Headache MaryAnn Mays, MD Headache Center Center for Neurological Restoration Neurological Institute Cleveland Clinic Disclosures Speakers Bureau for Allergan, Teva, and
Atenolol in the prophylaxis of chronic migraine: a 3-month open-label study
 Edvardsson SpringerPlus 2013, 2:479 a SpringerOpen Journal RESEARCH Open Access Atenolol in the prophylaxis of chronic migraine: a 3-month open-label study Bengt Edvardsson Abstract Background: Chronic
Edvardsson SpringerPlus 2013, 2:479 a SpringerOpen Journal RESEARCH Open Access Atenolol in the prophylaxis of chronic migraine: a 3-month open-label study Bengt Edvardsson Abstract Background: Chronic
CONSORT 2010 checklist of information to include when reporting a randomised trial*
 Supplemental Figures for: Ramosetron Versus Ondansetron in Combination with Aprepitant and Dexamethasone for the Prevention of Highly Emetogenic Chemotherapy-induced Nausea and Vomiting: A Multicenter,
Supplemental Figures for: Ramosetron Versus Ondansetron in Combination with Aprepitant and Dexamethasone for the Prevention of Highly Emetogenic Chemotherapy-induced Nausea and Vomiting: A Multicenter,
Rimegepant Pivotal Phase 3 Trial Results - Conference Call March 26, Biohaven
 Rimegepant Pivotal Phase 3 Trial Results - Conference Call March 26, 2018 1 Forward-Looking Statement This presentation contains forward-looking statements, including: statements about our plans to develop
Rimegepant Pivotal Phase 3 Trial Results - Conference Call March 26, 2018 1 Forward-Looking Statement This presentation contains forward-looking statements, including: statements about our plans to develop
The presenting and prescribing patterns of migraine in an Australian emergency department: A descriptive exploratory study
 170 Shao et al Original Article The presenting and prescribing patterns of migraine in an Australian emergency department: A descriptive exploratory study Emily Shao 1, James Hughes 2, Rob Eley 2,3 1 Royal
170 Shao et al Original Article The presenting and prescribing patterns of migraine in an Australian emergency department: A descriptive exploratory study Emily Shao 1, James Hughes 2, Rob Eley 2,3 1 Royal
