Use of a novel valve stent for transcatheter pulmonary valve replacement: An animal study
|
|
|
- Milton Hodge
- 5 years ago
- Views:
Transcription
1 Use of a novel valve stent for transcatheter pulmonary valve replacement: An animal study Gang-Jun Zong, MD, PhD, a Yuan Bai, MD, a Hai-Bin Jiang, MD, a Wei-Ping Li, MD, PhD, b Hong Wu, MD, PhD, a Xian-Xian Zhao, MD, PhD, a and Yong-Wen Qin, MD, PhD a Objective: The goal of this study was to evaluate valvular functionality after transcatheter pulmonary valve replacement in sheep using a novel pulmonary valve stent. Methods: Fresh porcine pericardium cross-linked with 0.6% glutaraldehyde was treated with L-glutamine to eliminate glutaraldehyde toxicity and sutured onto a valve ring before mounting on a nitinol stent to construct the pulmonary valve stent. Percutaneous femoral vein transcatheter pulmonary valve replacement was performed with the newly constructed valve stent. Pulmonary valve stents were implanted in 10 healthy sheep (6 males and 4 females) weighing an average of kg. Color Doppler echocardiography, 64-row computed tomography, and direct catheter examination were used to assess valvular function. Results: Implantation was successful in 8 sheep. Shortly after surgery, all artificial valve stents exhibited normal open and close functionality and no stenosis or insufficiency. Heart rate was slightly elevated at this time, while all other hemodynamic parameters were normal. Six-month follow-up revealed no evidence of valve stent dislocation and normal valvular and cardiac functionality. There was no evidence of stent fracture. Repeated valve stent implantation was well tolerated as indicated by good valvular functionality 2 months postdelivery. Conclusion: The novel pulmonary valve stent described herein can be delivered via percutaneous femoral vein transcatheter implantation and is highly efficacious at 6 months postdelivery. Furthermore, repeated valve stent replacement was successful. From the earliest transcatheter valve replacement experimental study performed by Andersen and colleagues in 1992, 1 to Bonhoeffer and colleagues pilot study of percutaneous pulmonary valve replacement in a right ventricle to pulmonary artery prosthetic conduit with valve dysfunction in 2000, 2 pulmonary valve replacement technology has steadily advanced. 3-5 Clinically, this technique appears to offer significant benefits over standard surgical procedures for patients with congenital heart diseases requiring additional surgical intervention. 6,7 Not only is the process efficacious and less traumatic, but the duration of hospital stay is shortened. 6 Despite the fact that the use of percutaneous pulmonary valve transplantation has been readily adopted, various problems associated with stent implantation per se have been reported. These include stent fracture, valve dislocation, and restenosis In addition, as outlined by Attmann and colleagues, 13 currently available stents are not suitable for use in all prospective patients due to anatomic variability. Hence there is a definite need for continued development From the Departments of Cardiology a and Ultrasonography, b Changhai Hospital, Second Military Medical University, Shanghai, China. This research was supported by the National High Technology Research and Development Program of China. G.-J. Zong and Y. Bai contributed equally to this study. Received for publication July 31, 2008; revisions received Nov 18, 2008; accepted for publication Dec 25, Address for reprints: Yong-Wen Qin, MD, PhD, Department of Cardiology, Changhai Hospital, Second Military Medical University, Shanghai, , China ( ywqinsmmu@yahoo.cn). J Thorac Cardiovasc Surg 2009;137: /$36.00 Copyright Ó 2009 by The American Association for Thoracic Surgery doi: /j.jtcvs and refinement with regards to the application of this relatively recent technique. Here we report on the design and use of a novel valve stent device for transcatheter pulmonary valve replacement. This study was performed using an ovine experimental model. Immediate postoperative functional analysis was assessed using color echocardiography, and a comprehensive follow-up evaluation was performed at 6 months using color echocardiography, 64-row computed tomography (CT), and direct catheter examination. METHODS Valve Stent Construction The valve stent was constructed using an artificial valve and a conduitshapedstent. Theartificial stent was constructedfrom a valveleafletand avalve ring. The valve ring was made of supermalleable nitinol wire (Shanghai Malleable Alloy Company, Shanghai, China) with a radial diameter of 0.25 mm, bent into 3 crescent shapes, and connected by a nitinol holder (see Figure 1, A). Porcine pericardium was used for valve leaflet creation following removal of surface fat tissue, agitated digestion with 0.01% trypsin for 8 hours, subsequent cross-linking with 0.6% glutaraldehyde for 36 hours, and, finally, repeated washing with 2% L-glutamine (to eliminate glutaraldehyde toxicity). Pericardium tissue was trimmed into 3 identically shaped valve leaflets according to the valve model profile and sutured onto the valve ring using 7 0 polypropylene thread (Johnson and Johnson, New Brunswick City, NJ). The attached side was denoted as the attachment margin and the nonattached side, the margo liber. The completed artificial valve is depicted in Figure 1, B. The conduit-shaped stent was constructed with supermalleable nitinol wire with a diameter of 0.18 mm and was comprised of 3 parts: the head, neck, and body. The head portion was proximal to the heart end, slightly protruded, and trumpet shaped. The neck portion was thinner and connected the head portion with the body portion. The body portion was straight and tubular shaped. The artificial valve was fixed inside the conduit-shaped stent, with the arc tips facing toward the top of the head portion and the sides attached to the The Journal of Thoracic and Cardiovascular Surgery c Volume 137, Number
2 Zong et al Abbreviations and Acronyms CT ¼ computed tomography DSA ¼ digital subtraction angiography neck portion, both of which were sutured with 5 0 polypropylene thread (Figure 1, C shows the completed valve stent). The valve stent was preserved in 60% ethanol, sterilized with 75% ethanol for 12 hours, and washed 3 times with saline prior to placement. Figure 2, A and B, shows the valved stent crimped by hand and then loaded into a 14-French sheath, respectively. Animals Animal studies were approved by the local hospital ethics committee. All animals received care in compliance with the Guide for the Care and Use of Laboratory Animals ( Ten healthy sheep (6 males and 4 females), 12 to 16 months old and weighing an average of kg, were used. Preoperative electrocardiogram, chest film, and color echocardiographic examinations revealed no abnormalities. No cardiac murmurs were detected. Animals were fasted and water deprived for 8 hours before surgery. Anesthesia was initiated by intramuscular injection of ketamine (10 mg/kg body weight), followed by intravenous injection of 10 ml of 2.5% pentobarbital sodium. Hair was removed from the precordial region and the inner sides of the extremities. Valve Stent Implantation and Postoperative Treatment After sterilizing the skin in the inguinal region, a 6-French leak-proof sheath was introduced percutaneously via the left femoral vein. An incision was made through the right inguinal skin and subcutaneous tissue, and the right femoral vein was subsequently exposed and punctured with a 7-French leak-proof sheath. Heparin (1.5 mg/kg) was introduced via the sheath. A 6-French pigtail catheter (Cordis, Johnson and Johnson, New Brunswick City, NJ) was introduced through the right femoral vein sheath, and imaging of the right ventricle and pulmonary valve was performed using digital subtraction angiography (DSA) (Siemens, Munich, Germany; Figure 3, A). Pressure values for the right ventricle and main pulmonary artery were also determined. The pulmonary valve was identified, and its position was marked according to imaging results. After measuring the radius of the pulmonary valve, a valve stent of appropriate size was selected and loaded into a 14-French sheath. A stent size larger than the measured annulus was selected to ensure the avoidance of paravalvular leaks and in consideration of the growth of the young sheep. The delivery route was established by introducing a steel guide wire into the distal segment of the left or right pulmonary valve via the pigtail catheter (see Figure 3, B). After the pigtail catheter was removed, a 14-French delivery sheath with an expandable stub was deployed into the main pulmonary artery via the guide wire (Shanghai Shape- Memory Alloys Material Limited Company, Shanghai, China; Figure 3, C). The expandable stub and steel guide wire were then removed, and the 14- French sheath carrying the valve stent was connected with the delivery sheath. The stent was inserted into the delivery sheath and the neck advanced (under the DSA) to the previously marked position of the pulmonary valve (Figure 3, D). After confirming that the stent had been delivered to the optimal position, the delivery sheath was retracted to release the stent (Figure 3, E). The delivery sheath was subsequently removed and the right femoral vein sutured. Imaging of the artificial pulmonary valve and the right ventricle was performed by deploying a pigtail catheter via the left femoral vein (Figure 3, F). Pressure values were recorded for the right ventricle and the main pulmonary artery. The catheter and right femoral vein sheath were removed. Iohexol, up to 50 ml, was used as the contrast dye in the procedure. All sheep received postoperative intramuscular injections of penicillin (800,000 U) and subcutaneous injections of heparin (2500 IU) for 3 days. Oral aspirin (3 mg/kg body weight) was given for 60 days. Evaluation of Short-Term Efficacy of Valve Stent Implantation Immediately after implantation, chest color Doppler ultrasonography was performed to assess right ventricular function and the hemodynamic function of the artificial valve. Evaluation of Valve Stent Implantation After 6 Months All survivingsheep underwent chest color Doppler ultrasonography, 64-row enhanced CT, and DSA 6 months after stent implantation. The right ventricle and artificial valve were examined to assess functionality and hemodynamics. One randomly selected sheep was killed by femoral artery bleeding. The heart was collected, the right ventricle incised, and the position of the valve stent noted. The stent was examined using electron microscopy. FIGURE 1. Pulmonary valve stent construction. A, The valve ring was made of supermalleable nitinol wire bent into 3 arc shapes. B, Three valve leaflets were constructed by trimming porcine pericardium. The valve leaflets were sutured onto the valve ring. One side was denoted the attachment region and the other, margo libra. C, The conduit-shaped stent comprised of the head, neck, and body. The head portion was proximal to the heart end, slightly protruded, and trumpet shaped. The neck portion was thinner and connected the head portion with the body portion. The body portion was a straight tubular shape. The artificial valve was placed within the tubular stent The Journal of Thoracic and Cardiovascular Surgery c June 2009
3 FIGURE 2. Placement of valved stent into sheath. A, The valved stent was crimped by hand to load into a 14-French sheath. B, The valved stent completely crimped into the sheath. Efficacy of Repeated Valve Stent Implantation Six months after initial implantation, 2 randomly selected surviving sheep had a repeated valve stent implantation via percutaneous DSA at the same location as the initial implantation. The placement process was the same as the initial implantation. Two months after surgery, follow-up color Doppler ultrasonography was performed and valvular function and location examined. Statistical Analysis Means, standard deviations, and minimum and maximums values are given for continuous variables. Changes in heart rate, mean arterial pressure, right ventricular pressure, pulmonary artery pressure, right ventricular volume, right ventricular ejection fraction, transvalvular gradients, and effective orifice areas over time (before implantation, 12 hours after implantation, and 6 months after implantation) were compared using repeated measurements. Moreover, Wilcoxon signed-rank test was performed to compare the difference between the 2 time points postoperation due to nonnormally data. All statistical assessments were 2-sided and evaluated at the 0.05 level of significant difference. Statistical analyses were performed using SPSS 15.0 statistical software (SPSS Inc, Chicago, Ill). RESULTS Operative Parameters Valve stent implantation was not possible in 2 of the 10 sheep. One of these animals had abnormally small FIGURE 3. Valve stent implantation process (representative images). A, Imaging of the pulmonary valve was performed to measure the pulmonary valve radius and location. B, Steel guide wires were deployed into the left or right pulmonary artery regions to facilitate delivery. C, A 14-French delivery sheath was inserted into the pulmonary artery. D, The neck portion of the stent was delivered to the previously noted location of the pulmonary valve. E, The delivery sheath was retracted and the stent released. F, Image of the artificial pulmonary valve revealing good placement and no regurgitation. The Journal of Thoracic and Cardiovascular Surgery c Volume 137, Number
4 Zong et al femoral vein, and the other died due to excessive anesthesia. Valve stent implantation was successful in the 8 other sheep. The mean diameter of the pulmonary annulus was mm and mm as revealed by Doppler ultrasound and angiography, respectively. The mean stent diameter was (range: mm). Mean surgery duration was minutes (range: minutes). The mean x-ray exposure time was minutes (range: minutes; Table 1). Short-Term Efficacy Evaluation of Valve Stent Implantation In each case, the valve stent was immediately observable under color Doppler ultrasonography following implantation. Artificial valve stents exhibited normal open and close functionality with no evidence of stenosis or insufficiency (data not shown). Heart rate was significantly increased after stent implantation compared with before implantation (P ¼.042, Figure 4, A). There were no other differences detected before and immediately after implantation for any of the other heart function markers examined (Figure 4, B H). Efficacy of Valve Stent Implantation After 6 Months All sheep exhibited normal open and close valvular functionality after 6 months, with no evidence of stenosis or insufficiency apparent (Figure 5, A D). Sixty-four-row CT revealed accurate placement of the devices. Animals had a weight gain of 8.2 to 13.5 kg. All heart function markers were normal (ie, not different to preimplantation values; Figure 4, A H). In all animals, no stent fracture, valve deformation or dislocation problems, or paravalvular leakage was noted. Histologic dissection revealed a complete stent with no evidence of fracture. Electron microscopy revealed that TABLE 1. Surgical information of implantations performed on experimental ewes Pulmonary annulus US (mm) Pulmonary annulus angiography (mm) Diameter of stent (mm) Operation time (min) Radiograph exposure time (min) ID Mean SD Minimum Maximum SD, Standard deviation; US, ultrasound. the stent was completely covered with endothelial cells (see Figure 6, A and B). Repeated Valve Stent Implantation Ultrasonography revealed normal valvular open and close function 2 months after repeated transplantation in both sheep. One sheep exhibited mild pulmonary regurgitation (Figure 7, A). Regurgitation was not apparent in the other animal, cardiac function was normal, and there was no evidence of valve stenosis (Figure 7, B). DISCUSSION Surgical reconstruction of the right ventricular outflow tract, including valve implantation and pulmonary valve replacement or repair, is clinically efficacious. 14 However, biologic valve deterioration often necessitates repeated cardiopulmonary bypass surgery. 15 To circumvent the risks associated with multiple cardiopulmonary bypass surgeries, percutaneous valve replacement has been developed in recent years, 16 providing patients with an alternative option. In this study, we performed percutaneous femoral vein implantation of the sheep pulmonary valve with a newly designed pulmonary valve stent. The objective of our study was to evaluate whether transcatheter implantation of the novel pulmonary valve stent was achievable and to observe implantation efficacy after 6 months. Repeated pulmonary valve stent implantation was also performed. Implantation of the device was found to be highly successful and efficacious at 6 months postsurgery. Repeated implantation was well tolerated. The conduit-shaped stent developed in this study required only 1 nitinol wire to be constructed without a connector, exhibited good flexibility, and was capable of adapting to the vessel after implantation. Electron microscope examination at 6 months postimplantation revealed that the stent was fully covered by endothelial cells, evidencing complete adherence. Because the intersecting region of the woven stent allows vascular contraction and mild diastolic movement, the likelihood of stent fracture should be reduced. The fact that no cases of stent fracture were observed after 6 months supports this assertion. The trumpet-shaped head portion of the stent consolidated placement at the pulmonary orifice, and the adjoining, slightly thinner neck portion facilitated increased overall stent flexibility and prevented stent deformation when the trumpet-shaped opening was pressured. This design is capable of eliminating valve deformation and dislocation problems associated with the use straight tubular stents, arising from compression at the pulmonary orifice. The body portion was conduit-shaped, allowing for greater fixation due to the increased stent diameter. Because the stent shape was well matched with the peripheral valve configuration, the pulmonary valve was compressed between the stent and the right ventricle wall, thus preventing perivalvular leakage. This was substantiated through imaging via color Doppler ultrasonography The Journal of Thoracic and Cardiovascular Surgery c June 2009
5 FIGURE 4. Heart function and hemodynamic markers before, immediately after, and 6 months after implantation. Heart rate (A), mean arterial pressure (B), right ventricle pressure (C), pulmonary artery pressure (D), right ventricular volumes (E), right ventricle ejection fraction (F), transvalvular gradients (G), and effective orifice areas (H) were assessed. *P<.05 between the indicated means. In this study, the artificial valve was sutured onto a nitinol valve ring, as opposed to direct suture into the stent. This made suturing of the valve into the conduit-shaped stent easier and firmer and resulted in the artificial valve being less susceptible to deformation. This also guaranteed that the artificial valve remained on a flat surface, which better prevents valve insufficiency and dislocation from occurring. We found that valvular and hemodynamic functions were normal 6 months postimplantation. Furthermore, there was no evidence of increased thrombotic development on valve ring structure following conventional anticoagulation therapy. With regards to the materials used for artificial pulmonary valve construction, previous studies have typically reported use of the bovine jugular vein valve 17,18 and The Journal of Thoracic and Cardiovascular Surgery c Volume 137, Number
6 Zong et al FIGURE 5. Pulmonary artery valve stent 6 months after implantation: color ultrasonography and computed tomography examination findings from a representative animal. A, Artificial valve opening (white arrow). B, Artificial valve closing (white arrow). C, Color Doppler ultrasonography revealed no patterns of blood passing or regurgitation. D, Right ventricular ejection fraction. polytetrafluoroethylene valve. 19 The use of porcine pericardium as a surgical biologic valve has a long history. We used porcine pericardium as the material for valve construction in this study, applying surgical procedures to establish the pericardium as a valve sutured onto a valvular ring. Porcine pericardium not only was easily obtained, but also had larger valve diameter. The design of the artificial valve was inspirited by biologic valves in vivo. The function of valves was determined by the design of valve ring. In combination with the trumpetshaped stent that provides a strong anchor, the valved stent is functional as well. The woven design of the stent increases compliance to avoid damage from contraction of the heart. The design of the valve ring may assist in preventing postimplantation deformity of the valve. The trumpet shape and use of a slightly oversized stent ensure solid anchoring of the stent and also avoid paravalvular leakage. The surgical implantation technique used in this study was similar to widely used techniques for intracoronary stent implantation but different from implantation techniques involving the fixation of the valve stent on the tip of a delivery sheath. Given that the stent-fixed delivery sheath is hard and less flexible, damage to the heart may occur during the curved passage from the inferior vena cava to the main FIGURE 6. Six-month postoperative electron microscopy images of a stent. A, Nitinol wire covered by endothelial cells (magnification 1003). B, The stent was completely covered by neatly arranged endothelial cells (magnification 15003) The Journal of Thoracic and Cardiovascular Surgery c June 2009
7 FIGURE 7. Color Doppler ultrasonography examinations were performed 2 months after the second valve stent implantation. A, Spectral of the pulmonary valve revealed mild regurgitation. B, Motion mode ultrasonography revealed normal cardiac function. pulmonary artery. In this study, we first introduced the delivery sheath into the pulmonary artery and then pushed the device through the sheath and into the pulmonary artery. This allowed for safer passage, with no significant increase in x- ray exposure time. The life expectancy of biologic valves is an issue of significant concern. Once a valve deteriorates, replacement implantation becomes necessary. Hence it is critical that valve stents are amenable to repeated implantation. The valve stent described in the current study exhibited good vascular compliance upon secondary push delivery implantation and readily overlapped with the initial stent. No significant complications were detected following repeated implantation. This study had several limitations. First, the valve stent could not be completely retrieved once released. Thus, further modifications to the delivery system are needed to provide flexibility in cases of suboptimal placement and/or stent size incompatibility. Such modifications will increase the safety and reliability of this procedure. Second, the animal cohort size was small and the follow-up time short. Last, the use of a single sheep for histologic evaluation and the lack of quantitative assessment of calcification are also limitations. Additional studies, with larger cohorts and longer follow-up durations, are needed to provide sufficient reference data for potential clinical application. References 1. Andersen HR, Knudsen LL, Hasenkam JM. Transluminal implantation of artificial heart valves. Description of a new expandable aortic valve and initial results with implantationbycathetertechnique inclosedchestpigs. Eur Heart J. 1992;13: Bonhoeffer P, Boudjemline Y, Saliba Z, Merckx J, Aggoun Y, Bonnet D, et al. Percutaneous replacement of pulmonary valve in a right-ventricle to pulmonary-artery prosthetic conduit with valve dysfunction. Lancet. 2000;356: Feinstein JA, Kim N, Reddy VM, Perry SB. Percutaneous pulmonary valve placement in a 10-month-old patient using a hand crafted stent-mounted porcine valve. Catheter Cardiovasc Interv. 2006;67: Garay F, Webb J, Hijazi ZM. Percutaneous replacement of pulmonary valve using the Edwards-Cribier percutaneous heart valve: first report in a human patient. Catheter Cardiovasc Interv. 2006;67: Coats L, Bonhoeffer P. New percutaneous treatments for valve disease. Heart. 2007;93: Nordmeyer J, Coats L, Bonhoeffer P. Current experience with percutaneous pulmonary valve implantation. Semin Thorac Cardiovasc Surg. 2006;18: Lurz P, Coats L, Khambadkone S, Nordmeyer J, Boudjemline Y, Schievano S, et al. Percutaneous pulmonary valve implantation: impact of evolving technology and learning curve on clinical outcome. Circulation. 2008;117: Khambadkone S, Coats L, Taylor A, Boudjemline Y, Derrick G, Tsang V, et al. Percutaneous pulmonary valve implantation in humans: results in 59 consecutive patients. Circulation. 2005;112: Khambadkone S, Nordmeyer J, Bonhoeffer P. Percutaneous implantation of the pulmonary and aortic valves: indications and limitations. J Cardiovasc Med (Hagerstown). 2007;8: Nordmeyer J, Lee TY, Bonhoeffer P. Percutaneous pulmonary valve implantation: a 5-year projection. Am Heart Hosp J. 2006;4: Schievano S, Petrini L, Migliavacca F, Coats L, Nordmeyer J, Lurz P, et al. Finite element analysis of stent deployment: understanding stent fracture in percutaneous pulmonary valve implantation. J Interv Cardiol. 2007;20: Kostolny M, Tsang V, Nordmeyer J, Van Doorn C, Frigiola A, Khambadkone S, et al. Rescue surgery following percutaneous pulmonary valve implantation. Eur J Cardiothorac Surg. 2008;33: Attmann T, Quaden R, Jahnke T, Muller-Hulsbeck S, Boening A, Cremer J, et al. Percutaneous pulmonary valve replacement: 3-month evaluation of self-expanding valved stents. Ann Thorac Surg. 2006;82: Wells WJ, Arroyo H Jr, Bremner RM, Wood J, Starnes VA. Homograft conduit failure in infants is not due to somatic outgrowth. J Thorac Cardiovasc Surg. 2002;124: Bouzas B, Kilner PJ, Gatzoulis MA. Pulmonary regurgitation: not a benign lesion. Eur Heart J. 2005;26: Bonhoeffer P, Boudjemline Y, Qureshi SA, Le Bidois J, Iserin L, Acar P, et al. Percutaneous insertion of the pulmonary valve. J Am Coll Cardiol. 2002;39: Bonhoeffer P, Boudjemline Y, Saliba Z, Hausse AO, Aggoun Y, Bonnet D, et al. Transcatheter implantation of a bovine valve in pulmonary position: a lamb study. Circulation. 2000;102: Attmann T, Quaden R, Freistedt A, König C, Cremer J, Lutter G. Percutaneous heart valve replacement: histology and calcification characteristics of biological valved stents in juvenile sheep. Cardiovasc Pathol. 2007;16: Attmann T, Steinseifer U, Cremer J, Lutter G. Percutaneous valve replacement: a novel low-profile polyurethane valved stent. Eur J Cardiothorac Surg. 2006; 30:379. The Journal of Thoracic and Cardiovascular Surgery c Volume 137, Number
Eighteen-month outcome of pulmonary valve stent implantation by direct right ventricle puncture: An animal study
 Eighteen-month outcome of pulmonary valve stent implantation by direct right ventricle puncture: An animal study Gang-Jun Zong, MD, PhD, a Yuan Bai, MD, b Ming-Biao Gu, MD, b Hai-Bin Jiang, MD, b Hong
Eighteen-month outcome of pulmonary valve stent implantation by direct right ventricle puncture: An animal study Gang-Jun Zong, MD, PhD, a Yuan Bai, MD, b Ming-Biao Gu, MD, b Hai-Bin Jiang, MD, b Hong
Percutaneous reimplantation of a pulmonary valved stent in sheep: A potential treatment for bioprosthetic valve degeneration
 Percutaneous reimplantation of a pulmonary valved stent in sheep: A potential treatment for bioprosthetic valve degeneration Yuan Bai, MD, a Gang-Jun Zong, MD, c Hai-Bing Jiang, MD, a Wei-Ping Li, MD,
Percutaneous reimplantation of a pulmonary valved stent in sheep: A potential treatment for bioprosthetic valve degeneration Yuan Bai, MD, a Gang-Jun Zong, MD, c Hai-Bing Jiang, MD, a Wei-Ping Li, MD,
2 Brigham and Women s Hospital, Boston, MA.
 Chapter 6: History of Transcatheter Aortic Valve Replacement (TAVR) Bryan Piccirillo, MD 1 ; Pinak B. Shah, MD, FACC 2 1 Brigham and Women s Heart and Vascular Center 2 Brigham and Women s Hospital, Boston,
Chapter 6: History of Transcatheter Aortic Valve Replacement (TAVR) Bryan Piccirillo, MD 1 ; Pinak B. Shah, MD, FACC 2 1 Brigham and Women s Heart and Vascular Center 2 Brigham and Women s Hospital, Boston,
The need for right ventricular outflow tract reconstruction
 Polytetrafluoroethylene Bicuspid Pulmonary Valve Implantation James A. Quintessenza, MD The need for right ventricular outflow tract reconstruction and pulmonary valve replacement is increasing for many
Polytetrafluoroethylene Bicuspid Pulmonary Valve Implantation James A. Quintessenza, MD The need for right ventricular outflow tract reconstruction and pulmonary valve replacement is increasing for many
Interventional procedures guidance Published: 26 September 2014 nice.org.uk/guidance/ipg504
 Transcatheter valve-in-valve e implantation for aortic bioprosthetic valve dysfunction Interventional procedures guidance Published: 26 September 2014 nice.org.uk/guidance/ipg504 Your responsibility This
Transcatheter valve-in-valve e implantation for aortic bioprosthetic valve dysfunction Interventional procedures guidance Published: 26 September 2014 nice.org.uk/guidance/ipg504 Your responsibility This
Original Policy Date
 MP 7.01.111 Transcatheter Pulmonary Valve Implantation Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to
MP 7.01.111 Transcatheter Pulmonary Valve Implantation Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to
Transcatheter Pulmonary Valve Implantation
 Transcatheter Pulmonary Valve Implantation Policy Number: 7.01.131 Last Review: 2/2014 Origination: 2/2012 Next Review: 2/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Transcatheter Pulmonary Valve Implantation Policy Number: 7.01.131 Last Review: 2/2014 Origination: 2/2012 Next Review: 2/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Portico (St. Jude Medical Inc, St.
 Review Article Portico Transcatheter Heart Valve Apostolos Tzikas 1,2, Michael Chrissoheris 2, Antonios Halapas 2, Konstantinos Spargias 2 1 Interbalkan European Medical Centre, Thessaloniki, 2 Hygeia
Review Article Portico Transcatheter Heart Valve Apostolos Tzikas 1,2, Michael Chrissoheris 2, Antonios Halapas 2, Konstantinos Spargias 2 1 Interbalkan European Medical Centre, Thessaloniki, 2 Hygeia
TAVR and Cardiac Surgeons
 TAVR and Cardiac Surgeons TAVR and Cardiac Surgeons Ragheb Hasan Consultant and Clinical Lead Cardiothoracic Surgeon Manchester Royal Infirmary, Oxford Road, Manchester UK Aortic Stenosis Is A Growing
TAVR and Cardiac Surgeons TAVR and Cardiac Surgeons Ragheb Hasan Consultant and Clinical Lead Cardiothoracic Surgeon Manchester Royal Infirmary, Oxford Road, Manchester UK Aortic Stenosis Is A Growing
Adult Echocardiography Examination Content Outline
 Adult Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 2 3 4 5 Anatomy and Physiology Pathology Clinical Care and Safety Measurement Techniques, Maneuvers,
Adult Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 2 3 4 5 Anatomy and Physiology Pathology Clinical Care and Safety Measurement Techniques, Maneuvers,
Transcatheter Pulmonary Valve Replacement Update on progress and outcomes
 Transcatheter Pulmonary Valve Replacement Update on progress and outcomes Barry Love, MD Director, Congenital Cardiac Catheterization Laboratory Mount Sinai Medical Center New York, New York Transcatheter
Transcatheter Pulmonary Valve Replacement Update on progress and outcomes Barry Love, MD Director, Congenital Cardiac Catheterization Laboratory Mount Sinai Medical Center New York, New York Transcatheter
Transcatheter Pulmonary Valve Implantation
 Transcatheter Pulmonary Valve Implantation Policy Number: Original Effective Date: MM.06.022 09/01/2013 Lines of Business: Current Effective Date: HMO; PPO; QUEST Integration 01/23/2015 Section: Surgery
Transcatheter Pulmonary Valve Implantation Policy Number: Original Effective Date: MM.06.022 09/01/2013 Lines of Business: Current Effective Date: HMO; PPO; QUEST Integration 01/23/2015 Section: Surgery
Aortic Valve Implantation
 Transapical Transcatheter Aortic Valve Implantation Early results reveal successful implantation rates in patients with aortic stenosis. BY JOHN C. ALEXANDER, MD, AND SUBHASIS CHATTERJEE, MD Medical progress
Transapical Transcatheter Aortic Valve Implantation Early results reveal successful implantation rates in patients with aortic stenosis. BY JOHN C. ALEXANDER, MD, AND SUBHASIS CHATTERJEE, MD Medical progress
Protocol. Transcatheter Pulmonary Valve Implantation
 Protocol Transcatheter Pulmonary Valve Implantation (701131) Medical Benefit Effective Date: 10/01/13 Next Review Date: 01/19 Preauthorization No Review Dates: 07/12, 07/13, 07/14, 07/15, 01/16, 01/17,
Protocol Transcatheter Pulmonary Valve Implantation (701131) Medical Benefit Effective Date: 10/01/13 Next Review Date: 01/19 Preauthorization No Review Dates: 07/12, 07/13, 07/14, 07/15, 01/16, 01/17,
Conflict of Interests
 Introduction to Interventional Echocardiography Roberto M Lang, MD Tomtec Conflict of Interests Research Grants Philips Medical Imaging Research Grants Speakers bureau Advisory bureau 1 Structural Heart
Introduction to Interventional Echocardiography Roberto M Lang, MD Tomtec Conflict of Interests Research Grants Philips Medical Imaging Research Grants Speakers bureau Advisory bureau 1 Structural Heart
My Choice For Percutaneous Mitral Valve Replacement. Jose Luis Navia, MD.
 My Choice For Percutaneous Mitral Valve Replacement Jose Luis Navia, MD. Disclosure Edwards Lifescienses St. Jude Medical MAQUET NaviGate Consultant, Investigator Consultant, Investigator Consultant, Investigator
My Choice For Percutaneous Mitral Valve Replacement Jose Luis Navia, MD. Disclosure Edwards Lifescienses St. Jude Medical MAQUET NaviGate Consultant, Investigator Consultant, Investigator Consultant, Investigator
Interventions in Congenital & Structural Heart Disease: Who Drives New Techniques and Devices?
 Interventions in Congenital & Structural Heart Disease: Who Drives New Techniques and Devices? Lee Benson MD Professor Pediatrics (Cardiology) Director, Cardiac Diagnostic & Interventional Unit The Hospital
Interventions in Congenital & Structural Heart Disease: Who Drives New Techniques and Devices? Lee Benson MD Professor Pediatrics (Cardiology) Director, Cardiac Diagnostic & Interventional Unit The Hospital
TAVI- Is Stroke Risk the Achilles Heel of Percutaneous Aortic Valve Repair?
 TAVI- Is Stroke Risk the Achilles Heel of Percutaneous Aortic Valve Repair? Elaine E. Tseng, MD and Marlene Grenon, MD Department of Surgery Divisions of Adult Cardiothoracic and Vascular and Endovascular
TAVI- Is Stroke Risk the Achilles Heel of Percutaneous Aortic Valve Repair? Elaine E. Tseng, MD and Marlene Grenon, MD Department of Surgery Divisions of Adult Cardiothoracic and Vascular and Endovascular
CIPG Transcatheter Aortic Valve Replacement- When Is Less, More?
 CIPG 2013 Transcatheter Aortic Valve Replacement- When Is Less, More? James D. Rossen, M.D. Professor of Medicine and Neurosurgery Director, Cardiac Catheterization Laboratory and Interventional Cardiology
CIPG 2013 Transcatheter Aortic Valve Replacement- When Is Less, More? James D. Rossen, M.D. Professor of Medicine and Neurosurgery Director, Cardiac Catheterization Laboratory and Interventional Cardiology
Transcatheter PV replacement: initial experience using selfexpandable
 Transcatheter PV replacement: initial experience using selfexpandable valves. Alejandro Peirone MD, FSCAI Private and Children`s Hospital of Córdoba, Argentina. October 13th; Buenos Aires. Lecture title:
Transcatheter PV replacement: initial experience using selfexpandable valves. Alejandro Peirone MD, FSCAI Private and Children`s Hospital of Córdoba, Argentina. October 13th; Buenos Aires. Lecture title:
Transcatheter Aortic Valve Replacement (TAVR)
 UW MEDICINE PATIENT EDUCATION Transcatheter Aortic Valve Replacement (TAVR) Treatment for aortic stenosis This handout explains when your doctor may advise TAVR to treat aortic stenosis. It includes the
UW MEDICINE PATIENT EDUCATION Transcatheter Aortic Valve Replacement (TAVR) Treatment for aortic stenosis This handout explains when your doctor may advise TAVR to treat aortic stenosis. It includes the
TRANSCATHETER REPLACEMENT OF THE PULMONARY VALVE (PPVI)
 TRANSCATHETER REPLACEMENT OF THE PULMONARY VALVE (PPVI) BASIL D. THANOPOULOS MD, PhD Director Interventional Cardiology of CHD Euroclinic ATHENS - GREECE TRANSCATHETER REPLACEMENT OF THE PULMONARY VALVE
TRANSCATHETER REPLACEMENT OF THE PULMONARY VALVE (PPVI) BASIL D. THANOPOULOS MD, PhD Director Interventional Cardiology of CHD Euroclinic ATHENS - GREECE TRANSCATHETER REPLACEMENT OF THE PULMONARY VALVE
Transcatheter Pulmonary Valve Implantation. Description
 Subject: Transcatheter Pulmonary Valve Implantation Page: 1 of 12 Last Review Status/Date: March 2015 Transcatheter Pulmonary Valve Implantation Description Transcatheter pulmonary valve implantation (TPVI)
Subject: Transcatheter Pulmonary Valve Implantation Page: 1 of 12 Last Review Status/Date: March 2015 Transcatheter Pulmonary Valve Implantation Description Transcatheter pulmonary valve implantation (TPVI)
Pulmonary Valve Replacement
 Pulmonary Valve Replacement with Fascia Lata J. C. R. Lincoln, F.R.C.S., M. Geens, M.D., M. Schottenfeld, M.D., and D. N. Ross, F.R.C.S. ABSTRACT The purpose of this paper is to describe a technique of
Pulmonary Valve Replacement with Fascia Lata J. C. R. Lincoln, F.R.C.S., M. Geens, M.D., M. Schottenfeld, M.D., and D. N. Ross, F.R.C.S. ABSTRACT The purpose of this paper is to describe a technique of
Management of Difficult Aortic Root, Old and New solutions
 Management of Difficult Aortic Root, Old and New solutions Hani K. Najm MD, Msc, FRCSC,, FACC, FESC Chairman, Pediatric and Congenital Heart Surgery Cleveland Clinic Conflict of Interest None Difficult
Management of Difficult Aortic Root, Old and New solutions Hani K. Najm MD, Msc, FRCSC,, FACC, FESC Chairman, Pediatric and Congenital Heart Surgery Cleveland Clinic Conflict of Interest None Difficult
Transapical mitral valved stent implantation: A survival series in swine
 Evolving Technology/Basic Science Lozonschi et al Transapical mitral valved stent implantation: A survival series in swine Lucian Lozonschi, MD, a Rene Bombien, MD, b Satoru Osaki, MD, a Jian Hu, MD, a
Evolving Technology/Basic Science Lozonschi et al Transapical mitral valved stent implantation: A survival series in swine Lucian Lozonschi, MD, a Rene Bombien, MD, b Satoru Osaki, MD, a Jian Hu, MD, a
Transcatheter Mitral Valve Replacement. Lutter G, Lozonschi L
 Transcatheter Mitral Valve Replacement Lutter G, Lozonschi L Department of Cardiovascular Surgery University Hospital, Campus Kiel, Germany and Division of Cardiothoracic Surgery, University of Wisconsin,
Transcatheter Mitral Valve Replacement Lutter G, Lozonschi L Department of Cardiovascular Surgery University Hospital, Campus Kiel, Germany and Division of Cardiothoracic Surgery, University of Wisconsin,
Pulmonary Valve Regurgitation
 Pulmonary Valve Regurgitation Ziyad M. Hijazi, MD, FSCAI, FACC James A. Hunter,MD, University Chair Professor Of Pediatrics & Internal Medicine Rush Center For Congenital & Structural Heart Disease Rush
Pulmonary Valve Regurgitation Ziyad M. Hijazi, MD, FSCAI, FACC James A. Hunter,MD, University Chair Professor Of Pediatrics & Internal Medicine Rush Center For Congenital & Structural Heart Disease Rush
I will not discuss off label use or investigational use in my presentation.
 I will not discuss off label use or investigational use in my presentation. Surgical valves Design and Durability Testing Potential Concerns Real Practice 1952-1962 1963-1966 1967-1969 1969-1977 1977-1984
I will not discuss off label use or investigational use in my presentation. Surgical valves Design and Durability Testing Potential Concerns Real Practice 1952-1962 1963-1966 1967-1969 1969-1977 1977-1984
New insights in minimally invasive valve replacement: description of a cooperative approach for the off-pump replacement of mitral valves
 European Heart Journal (2005) 26, 2013 2017 doi:10.1093/eurheartj/ehi307 Clinical research New insights in minimally invasive valve replacement: description of a cooperative approach for the off-pump replacement
European Heart Journal (2005) 26, 2013 2017 doi:10.1093/eurheartj/ehi307 Clinical research New insights in minimally invasive valve replacement: description of a cooperative approach for the off-pump replacement
IMAGES. in PAEDIATRIC CARDIOLOGY
 IMAGES in PAEDIATRIC CARDIOLOGY Images Paediatr Cardiol. 2005 Jan-Mar; 7(1): 12 17. PMCID: PMC3232568 Stent implantation for coarctation facilitated by the anterograde trans-septal approach N Sreeram and
IMAGES in PAEDIATRIC CARDIOLOGY Images Paediatr Cardiol. 2005 Jan-Mar; 7(1): 12 17. PMCID: PMC3232568 Stent implantation for coarctation facilitated by the anterograde trans-septal approach N Sreeram and
Acute dissections of the descending thoracic aorta (Debakey
 Endovascular Treatment of Acute Descending Thoracic Aortic Dissections Nimesh D. Desai, MD, PhD, and Joseph E. Bavaria, MD Acute dissections of the descending thoracic aorta (Debakey type III or Stanford
Endovascular Treatment of Acute Descending Thoracic Aortic Dissections Nimesh D. Desai, MD, PhD, and Joseph E. Bavaria, MD Acute dissections of the descending thoracic aorta (Debakey type III or Stanford
For Personal Use. Copyright HMP 2013
 12-00415 Case Report J INVASIVE CARDIOL 2013;25(4):E69-E71 A Concert in the Heart. Bilateral Melody Valve Implantation in the Branch Pulmonary Arteries Nicola Maschietto, MD, PhD and Ornella Milanesi,
12-00415 Case Report J INVASIVE CARDIOL 2013;25(4):E69-E71 A Concert in the Heart. Bilateral Melody Valve Implantation in the Branch Pulmonary Arteries Nicola Maschietto, MD, PhD and Ornella Milanesi,
Transcatheter Aortic Valve Replacement: Current and Future Devices: How do They Work, Eligibility, Review of Data
 Transcatheter Aortic Valve Replacement: Current and Future Devices: How do They Work, Eligibility, Review of Data Echo Florida 2013 Jonathan J. Passeri, M.D. Co-Director, Heart Valve Program Director,
Transcatheter Aortic Valve Replacement: Current and Future Devices: How do They Work, Eligibility, Review of Data Echo Florida 2013 Jonathan J. Passeri, M.D. Co-Director, Heart Valve Program Director,
2/28/2010. Speakers s name: Paul Chiam. I have the following potential conflicts of interest to report: NONE. Antegrade transvenous transseptal route
 Transcatheter Aortic Valve Implantation Asian perspective Speakers s name: Paul Chiam Paul TL Chiam MBBS, MRCP, FACC I have the following potential conflicts of interest to report: NONE Consultant National
Transcatheter Aortic Valve Implantation Asian perspective Speakers s name: Paul Chiam Paul TL Chiam MBBS, MRCP, FACC I have the following potential conflicts of interest to report: NONE Consultant National
Percutaneous Therapy for Calcific Mitral Valve Disease
 31 st Annual State of the Art Echocardiography San Diego, CA February 18, 2018 5:00 5:15 PM 15 min Percutaneous Therapy for Calcific Mitral Valve Disease Muhamed Sarić MD, PhD, MPA Director of Noninvasive
31 st Annual State of the Art Echocardiography San Diego, CA February 18, 2018 5:00 5:15 PM 15 min Percutaneous Therapy for Calcific Mitral Valve Disease Muhamed Sarić MD, PhD, MPA Director of Noninvasive
Autologous Pulmonary Valve Replacement of the Diseased Aortic Valve
 Autologous Pulmonary Valve Replacement of the Diseased Aortic Valve By L. GONZALEZ-LAvIN, M.D., M. GEENS. M.D., J. SOMERVILLE, M.D., M.R.C.P., ANm D. N. Ross, M.B., CH.B., F.R.C.S. SUMMARY Living tissue
Autologous Pulmonary Valve Replacement of the Diseased Aortic Valve By L. GONZALEZ-LAvIN, M.D., M. GEENS. M.D., J. SOMERVILLE, M.D., M.R.C.P., ANm D. N. Ross, M.B., CH.B., F.R.C.S. SUMMARY Living tissue
New Cardiovascular Devices and Interventions: Non-Contrast MRI for TAVR Abhishek Chaturvedi Assistant Professor. Cardiothoracic Radiology
 New Cardiovascular Devices and Interventions: Non-Contrast MRI for TAVR Abhishek Chaturvedi Assistant Professor Cardiothoracic Radiology Disclosure I have no disclosure pertinent to this presentation.
New Cardiovascular Devices and Interventions: Non-Contrast MRI for TAVR Abhishek Chaturvedi Assistant Professor Cardiothoracic Radiology Disclosure I have no disclosure pertinent to this presentation.
Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully
 Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Indication, Timing, Assessment and Update on TAVI
 Indication, Timing, Assessment and Update on TAVI Swedish Heart and Vascular Institute Ming Zhang MD PhD Interventional Cardiology Structure Heart Disease Conflict of Interest None Starr- Edwards Mechanical
Indication, Timing, Assessment and Update on TAVI Swedish Heart and Vascular Institute Ming Zhang MD PhD Interventional Cardiology Structure Heart Disease Conflict of Interest None Starr- Edwards Mechanical
Edwards Sapien. Medtronic CoreValve. Inoperable FDA approved High risk: in trials. FDA approved
 Transcatheter Aortic Valve Replacement Symptomatic Aortic Stenosis Asymptomatic Juan Crestanello, MD Interim Director, Division of Cardiac Surgery Associate Professor Division of Cardiac Surgery The Ohio
Transcatheter Aortic Valve Replacement Symptomatic Aortic Stenosis Asymptomatic Juan Crestanello, MD Interim Director, Division of Cardiac Surgery Associate Professor Division of Cardiac Surgery The Ohio
14 Valvular Stenosis
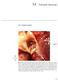 14 Valvular Stenosis 14-1. Valvular Stenosis unicuspid valve FIGUE 14-1. This photograph shows severe valvular stenosis as it occurs in a newborn. There is a unicuspid, horseshoe-shaped leaflet with a
14 Valvular Stenosis 14-1. Valvular Stenosis unicuspid valve FIGUE 14-1. This photograph shows severe valvular stenosis as it occurs in a newborn. There is a unicuspid, horseshoe-shaped leaflet with a
Percutaneous Valve Interventions. Percutaneous Valve Interventions
 Percutaneous Valve Interventions Stanton J. Rowe President, Percutaneous Valve Interventions Edwards is Best Positioned to Capitalize on Percutaneous Valve Opportunities #1 global valve replacement and
Percutaneous Valve Interventions Stanton J. Rowe President, Percutaneous Valve Interventions Edwards is Best Positioned to Capitalize on Percutaneous Valve Opportunities #1 global valve replacement and
The Edge-to-Edge Technique f For Barlow's Disease
 The Edge-to-Edge Technique f For Barlow's Disease Ottavio Alfieri, Michele De Bonis, Elisabetta Lapenna, Francesco Maisano, Lucia Torracca, Giovanni La Canna. Department of Cardiac Surgery, San Raffaele
The Edge-to-Edge Technique f For Barlow's Disease Ottavio Alfieri, Michele De Bonis, Elisabetta Lapenna, Francesco Maisano, Lucia Torracca, Giovanni La Canna. Department of Cardiac Surgery, San Raffaele
First Transfemoral Aortic Valve Implantation In Bulgaria - Crossing The Valve With The Device Is Not Always
 ISPUB.COM The Internet Journal of Cardiology Volume 9 Number 2 First Transfemoral Aortic Valve Implantation In Bulgaria - Crossing The Valve With The Device Is Not T D, J P. Citation T D, J P.. First Transfemoral
ISPUB.COM The Internet Journal of Cardiology Volume 9 Number 2 First Transfemoral Aortic Valve Implantation In Bulgaria - Crossing The Valve With The Device Is Not T D, J P. Citation T D, J P.. First Transfemoral
Cite this article as:
 doi: 10.21037/acs.2018.09.05 Cite this article as: Cheung A. Early experience of TIARA transcatheter mitral valve replacement system.. doi: 10.21037/acs.2018.09.05 This is a PDF file of an edited manuscript
doi: 10.21037/acs.2018.09.05 Cite this article as: Cheung A. Early experience of TIARA transcatheter mitral valve replacement system.. doi: 10.21037/acs.2018.09.05 This is a PDF file of an edited manuscript
Percutaneous Pulmonary Valve Implantation
 Review Print ISSN 1738-5520 On-line ISSN 1738-5555 Korean Circulation Journal Percutaneous Pulmonary Valve Implantation Young-Seok Lee, MD 1 and Hyoung-Doo Lee, MD 2 1 Division of Pediatric Cardiology,
Review Print ISSN 1738-5520 On-line ISSN 1738-5555 Korean Circulation Journal Percutaneous Pulmonary Valve Implantation Young-Seok Lee, MD 1 and Hyoung-Doo Lee, MD 2 1 Division of Pediatric Cardiology,
CP STENT. Large Diameter, Balloon Expandable Stent
 CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
Congenital cardiac defects involving the right
 Percutaneous Pulmonary Valve Implantation for RVOT Defects How recent advances in PPVI can improve the treatment of patients who have undergone previous repair of congenital heart defects. BY WAIL ALKASHKARI,
Percutaneous Pulmonary Valve Implantation for RVOT Defects How recent advances in PPVI can improve the treatment of patients who have undergone previous repair of congenital heart defects. BY WAIL ALKASHKARI,
An anterior aortoventriculoplasty, known as the Konno-
 The Konno-Rastan Procedure for Anterior Aortic Annular Enlargement Mark E. Roeser, MD An anterior aortoventriculoplasty, known as the Konno-Rastan procedure, is a useful tool for the cardiac surgeon. Originally,
The Konno-Rastan Procedure for Anterior Aortic Annular Enlargement Mark E. Roeser, MD An anterior aortoventriculoplasty, known as the Konno-Rastan procedure, is a useful tool for the cardiac surgeon. Originally,
Zenith Alpha T HORACIC ENDOVASCULAR GRAFT
 Device description Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21181-EN-F Modular design The two-piece modular system allows the physician to customize a graft system to fit each
Device description Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21181-EN-F Modular design The two-piece modular system allows the physician to customize a graft system to fit each
Valve Replacement without a Scalpel Transcatheter Aortic Valve Replacement (TAVR) Charles T. Klodell, M.D.
 Valve Replacement without a Scalpel Transcatheter Aortic Valve Replacement (TAVR) Charles T. Klodell, M.D. Professor, Thoracic and Cardiovascular Surgery University of Florida klodell@surgery.ufl.edu Disclosures
Valve Replacement without a Scalpel Transcatheter Aortic Valve Replacement (TAVR) Charles T. Klodell, M.D. Professor, Thoracic and Cardiovascular Surgery University of Florida klodell@surgery.ufl.edu Disclosures
Comments restricted to Sapien and Corevalve 9/12/2016. Disclosures: Core Lab contracts with Edwards Lifesciences, Middlepeak, Medtronic
 Para-ValvularRegurgitation post TAVR: Predict, Prevent, Quantitate, Manage Linda D. Gillam, MD, MPH, FACC, FASE Chair, Department of Cardiovascular Medicine Morristown Medical Center/Atlantic Health System
Para-ValvularRegurgitation post TAVR: Predict, Prevent, Quantitate, Manage Linda D. Gillam, MD, MPH, FACC, FASE Chair, Department of Cardiovascular Medicine Morristown Medical Center/Atlantic Health System
Transapical mitral valve implantation: the Lutter valve
 EXPERT opinion Heart, Lung and Vessels. 2013; 5(4): 201-206 Transapical mitral valve implantation: the Lutter valve 201 * G. Lutter 1, S. * Pokorny 1, D. Frank 2, J. Cremer 1, L. Lozonschi 3 1 Department
EXPERT opinion Heart, Lung and Vessels. 2013; 5(4): 201-206 Transapical mitral valve implantation: the Lutter valve 201 * G. Lutter 1, S. * Pokorny 1, D. Frank 2, J. Cremer 1, L. Lozonschi 3 1 Department
Successful percutaneous treatment of late-onset femoral pseudoaneurysm after transcatheter, aortic valve implantation procedure
 Case Report Page 1 of 5 Successful percutaneous treatment of late-onset femoral pseudoaneurysm after transcatheter, aortic valve implantation procedure Murat Celik, Uygar Cagdas Yuksel Correspondence to:
Case Report Page 1 of 5 Successful percutaneous treatment of late-onset femoral pseudoaneurysm after transcatheter, aortic valve implantation procedure Murat Celik, Uygar Cagdas Yuksel Correspondence to:
Research Article A New Coated Nitinol Occluder for Transcatheter Closure of Ventricular Septal Defects in a Canine Model
 BioMed Research International Volume 2013, Article ID 507919, 7 pages http://dx.doi.org/10.1155/2013/507919 Research Article A New Coated Nitinol Occluder for Transcatheter Closure of Ventricular Septal
BioMed Research International Volume 2013, Article ID 507919, 7 pages http://dx.doi.org/10.1155/2013/507919 Research Article A New Coated Nitinol Occluder for Transcatheter Closure of Ventricular Septal
Nit-Occlud. Coil System for PDA Closure IMPLANTATION POCKET GUIDE. Rx only CV / B. Braun Interventional Systems Inc.
 Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
Alternate Vascular Access for TAVR. Gian Paolo Ussia Campus Bio-medico University, Rome Italy
 Alternate Vascular Access for TAVR Gian Paolo Ussia Campus Bio-medico University, Rome Italy g.ussia@unicampus.it REQUIRED Gian Paolo Ussia I have no relevant financial relationships Transcatheter Valves
Alternate Vascular Access for TAVR Gian Paolo Ussia Campus Bio-medico University, Rome Italy g.ussia@unicampus.it REQUIRED Gian Paolo Ussia I have no relevant financial relationships Transcatheter Valves
The Colibri heart valve: theory and practice in the achievement of a low-profile, pre-mounted, pre-packaged TAVI valve
 INNOVATIONS / NOVEL TECHNOLOGIES The Colibri heart valve: theory and practice in the achievement of a low-profile, pre-mounted, pre-packaged TAVI valve R. David Fish 1 *, MD; David Paniagua 1, MD; Pedro
INNOVATIONS / NOVEL TECHNOLOGIES The Colibri heart valve: theory and practice in the achievement of a low-profile, pre-mounted, pre-packaged TAVI valve R. David Fish 1 *, MD; David Paniagua 1, MD; Pedro
Aortic stenosis (AS) remains the most common
 Sapien Valve: Past, Present, and Future A look at how the Sapien family of valves continues to evolve to treat a range of patients seeking transcatheter aortic valve replacement. BY RAVINDER SINGH RAO,
Sapien Valve: Past, Present, and Future A look at how the Sapien family of valves continues to evolve to treat a range of patients seeking transcatheter aortic valve replacement. BY RAVINDER SINGH RAO,
Versatility and Proven Safety
 Versatility and Proven Safety Indicated for: Temporary or Permanent placement Standard or Over-The-Wire Delivery Jugular, Femoral, Antecubital, and Popliteal Access > Proven Safety Initial Clinical Trial
Versatility and Proven Safety Indicated for: Temporary or Permanent placement Standard or Over-The-Wire Delivery Jugular, Femoral, Antecubital, and Popliteal Access > Proven Safety Initial Clinical Trial
Clinical Needs Assessment at Strong Memorial Hospital
 Clinical Needs Assessment at Strong Memorial Hospital Endoleaks in Endovascular Repair Endoleaks are a common problem during endovascular aortic repair and occur in 29% of all TEVAR (Thoracic Endovascular
Clinical Needs Assessment at Strong Memorial Hospital Endoleaks in Endovascular Repair Endoleaks are a common problem during endovascular aortic repair and occur in 29% of all TEVAR (Thoracic Endovascular
TAVR: Echo Measurements Pre, Post And Intra Procedure
 2017 ASE Florida, Orlando, FL October 10, 2017 8:00 8:25 AM 25 min TAVR: Echo Measurements Pre, Post And Intra Procedure Muhamed Sarić MD, PhD, MPA Director of Noninvasive Cardiology Echo Lab Associate
2017 ASE Florida, Orlando, FL October 10, 2017 8:00 8:25 AM 25 min TAVR: Echo Measurements Pre, Post And Intra Procedure Muhamed Sarić MD, PhD, MPA Director of Noninvasive Cardiology Echo Lab Associate
The Melody R valve and Ensemble R delivery system for transcatheter pulmonary valve replacement
 Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Pharmaceutical Science to Improve the Human Condition: Prix Galien 2012 The Melody R valve and Ensemble R delivery
Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Pharmaceutical Science to Improve the Human Condition: Prix Galien 2012 The Melody R valve and Ensemble R delivery
Coronary Artery from the Wrong Sinus of Valsalva: A Physiologic Repair Strategy
 Coronary Artery from the Wrong Sinus of Valsalva: A Physiologic Repair Strategy Tom R. Karl, MS, MD he most commonly reported coronary artery malformation leading to sudden death in children and young
Coronary Artery from the Wrong Sinus of Valsalva: A Physiologic Repair Strategy Tom R. Karl, MS, MD he most commonly reported coronary artery malformation leading to sudden death in children and young
Peripheral and Cardiology Coder 2018
 Peripheral and Cardiology Coder 2018 Cardiovascular Services and Procedures Prepared and Published By: MedLearn Publishing A Division of MedLearn Media, Inc. 445 Minnesota Street, Suite 514 St. Paul, MN
Peripheral and Cardiology Coder 2018 Cardiovascular Services and Procedures Prepared and Published By: MedLearn Publishing A Division of MedLearn Media, Inc. 445 Minnesota Street, Suite 514 St. Paul, MN
In 1980, Bex and associates 1 first introduced the initial
 Technique of Aortic Translocation for the Management of Transposition of the Great Arteries with a Ventricular Septal Defect and Pulmonary Stenosis Victor O. Morell, MD, and Peter D. Wearden, MD, PhD In
Technique of Aortic Translocation for the Management of Transposition of the Great Arteries with a Ventricular Septal Defect and Pulmonary Stenosis Victor O. Morell, MD, and Peter D. Wearden, MD, PhD In
Index. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Acute coronary syndrome(s), anticoagulant therapy in, 706, 707 antiplatelet therapy in, 702 ß-blockers in, 703 cardiac biomarkers in,
Index Note: Page numbers of article titles are in boldface type. A Acute coronary syndrome(s), anticoagulant therapy in, 706, 707 antiplatelet therapy in, 702 ß-blockers in, 703 cardiac biomarkers in,
Mitral Regurgitation
 UW MEDICINE PATIENT EDUCATION Mitral Regurgitation Causes, symptoms, diagnosis, and treatment This handout describes mitral regurgitation, a disease of the mitral valve. It explains how this disease is
UW MEDICINE PATIENT EDUCATION Mitral Regurgitation Causes, symptoms, diagnosis, and treatment This handout describes mitral regurgitation, a disease of the mitral valve. It explains how this disease is
Finite Element Analysis of Stent Deployment: Understanding Stent Fracture in Percutaneous Pulmonary Valve Implantation
 C 2007, the Authors Journal compilation C 2007, Blackwell Publishing, Inc. DOI: 10.1111/j.1540-8183.2007.00294.x Finite Element Analysis of Stent Deployment: Understanding Stent Fracture in Percutaneous
C 2007, the Authors Journal compilation C 2007, Blackwell Publishing, Inc. DOI: 10.1111/j.1540-8183.2007.00294.x Finite Element Analysis of Stent Deployment: Understanding Stent Fracture in Percutaneous
TSDA ACGME Milestones
 TSDA ACGME Milestones Short MW and Edwards JA. Assessing resident milestones using a CASPE March 2012 Short MW and Edwards JA. Assessing resident milestones using a CASPE March 2012 Short
TSDA ACGME Milestones Short MW and Edwards JA. Assessing resident milestones using a CASPE March 2012 Short MW and Edwards JA. Assessing resident milestones using a CASPE March 2012 Short
Mitral valve infective endocarditis (IE) is the most
 Mitral Valve Replacement for Infective Endocarditis With Annular Abscess: Annular Reconstruction Gregory J. Bittle, MD, Murtaza Y. Dawood, MD, and James S. Gammie, MD Mitral valve infective endocarditis
Mitral Valve Replacement for Infective Endocarditis With Annular Abscess: Annular Reconstruction Gregory J. Bittle, MD, Murtaza Y. Dawood, MD, and James S. Gammie, MD Mitral valve infective endocarditis
Results of Transapical Valves. A.P. Kappetein Dept Cardio-thoracic surgery
 Results of Transapical Valves A.P. Kappetein Dept Cardio-thoracic surgery Rotterda am, The Netherlands 2002 FIM 2003 2005 2006 2010 THV THV Cribier-Edwards Edwards Edwards Sapien Sapien XT Bovine pericardium
Results of Transapical Valves A.P. Kappetein Dept Cardio-thoracic surgery Rotterda am, The Netherlands 2002 FIM 2003 2005 2006 2010 THV THV Cribier-Edwards Edwards Edwards Sapien Sapien XT Bovine pericardium
Diversion of the inferior vena cava following repair of atrial septal defect causing hypoxemia
 Marshall University Marshall Digital Scholar Internal Medicine Faculty Research Spring 5-2004 Diversion of the inferior vena cava following repair of atrial septal defect causing hypoxemia Ellen A. Thompson
Marshall University Marshall Digital Scholar Internal Medicine Faculty Research Spring 5-2004 Diversion of the inferior vena cava following repair of atrial septal defect causing hypoxemia Ellen A. Thompson
Initial Experience Using the Palmaz Corinthian Stent for Right Ventricular Outflow Obstruction in Infants and Small Children
 Catheterization and Cardiovascular Interventions 51:444 449 (2000) Initial Experience Using the Palmaz Corinthian Stent for Right Ventricular Outflow Obstruction in Infants and Small Children Daniel R.
Catheterization and Cardiovascular Interventions 51:444 449 (2000) Initial Experience Using the Palmaz Corinthian Stent for Right Ventricular Outflow Obstruction in Infants and Small Children Daniel R.
IMAGES. in PAEDIATRIC CARDIOLOGY. Abstract
 IMAGES in PAEDIATRIC CARDIOLOGY Images Paediatr Cardiol. 2008 Apr-Jun; 10(2): 11 17. PMCID: PMC3232589 Transcatheter closure of symptomatic aortopulmonary window in an infant F Pillekamp, 1 T Hannes, 1
IMAGES in PAEDIATRIC CARDIOLOGY Images Paediatr Cardiol. 2008 Apr-Jun; 10(2): 11 17. PMCID: PMC3232589 Transcatheter closure of symptomatic aortopulmonary window in an infant F Pillekamp, 1 T Hannes, 1
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Index. interventional.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Ablation, of mitral leaflets, 80 Accucinch Annuloplasty system, for mitral regurgitation, 79, 94 95 Accutrak delivery system, for CoreValve
Index Note: Page numbers of article titles are in boldface type. A Ablation, of mitral leaflets, 80 Accucinch Annuloplasty system, for mitral regurgitation, 79, 94 95 Accutrak delivery system, for CoreValve
E xtracardiac conduits placed between the right ventricle
 1058 INTERVENTIONAL CARDIOLOGY AND SURGERY Implantation of endovascular stents for the obstructive right ventricular outflow tract H Sugiyama, W Williams, L N Benson... See end of article for authors affiliations...
1058 INTERVENTIONAL CARDIOLOGY AND SURGERY Implantation of endovascular stents for the obstructive right ventricular outflow tract H Sugiyama, W Williams, L N Benson... See end of article for authors affiliations...
Percutaneous Pulmonary Valve Replacement
 Percutaneous Pulmonary Valve Replacement Indications, current practice, and the ongoing evolution of this technique. By Damien Kenny, MB, MD; Qi-Ling Cao, MD; and Ziyad M. Hijazi, MD, MPH, FSCAI A B Figure
Percutaneous Pulmonary Valve Replacement Indications, current practice, and the ongoing evolution of this technique. By Damien Kenny, MB, MD; Qi-Ling Cao, MD; and Ziyad M. Hijazi, MD, MPH, FSCAI A B Figure
Introduction What Causes Peripheral Vascular Disease? How Do Doctors Treat Peripheral Vascular Disease?... 9
 Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
Transcatheter closure of the patent ductus arteriosus using the new Amplatzer duct occluder: Initial clinical applications in children
 Transcatheter closure of the patent ductus arteriosus using the new Amplatzer duct occluder: Initial clinical applications in children Basil Thanopoulos, MD, PhD, a Nikolaos Eleftherakis, MD, a Konstantinos
Transcatheter closure of the patent ductus arteriosus using the new Amplatzer duct occluder: Initial clinical applications in children Basil Thanopoulos, MD, PhD, a Nikolaos Eleftherakis, MD, a Konstantinos
TAVR: It s a Career, Not Just a Procedure! Jeffrey A Southard, MD Cardiology May 5, 2012
 TAVR: It s a Career, Not Just a Procedure! Jeffrey A Southard, MD Cardiology May 5, 2012 Transcatheter Aortic Valve Replacement T- eam A- pproach to V- alve R- eplacement UCDMC Team Administration Cardiology
TAVR: It s a Career, Not Just a Procedure! Jeffrey A Southard, MD Cardiology May 5, 2012 Transcatheter Aortic Valve Replacement T- eam A- pproach to V- alve R- eplacement UCDMC Team Administration Cardiology
Percutaneous mitral valve repair/replacement. Jan Van der Heyden MD, PhD St.Antonius Hospital Nieuwegein
 Percutaneous mitral valve repair/replacement Jan Van der Heyden MD, PhD St.Antonius Hospital Nieuwegein Mitral Valve anatomy Difference between AoV and MV Aortic Valve Mitral Valve Transcatheter Mitral
Percutaneous mitral valve repair/replacement Jan Van der Heyden MD, PhD St.Antonius Hospital Nieuwegein Mitral Valve anatomy Difference between AoV and MV Aortic Valve Mitral Valve Transcatheter Mitral
Implantation of the Melody Transcatheter Pulmonary Valve in Patients With a Dysfunctional Right Ventricular Outflow Tract Conduit
 Journal of the American College of Cardiology Vol. 54, No. 18, 2009 2009 by the American College of Cardiology Foundation ISSN 0735-1097/09/$36.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2009.06.034
Journal of the American College of Cardiology Vol. 54, No. 18, 2009 2009 by the American College of Cardiology Foundation ISSN 0735-1097/09/$36.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2009.06.034
Progress In Transcatheter Aortic Valve Implantation
 Progress In Transcatheter Aortic Valve Implantation Gerald Yong MBBS (Hons) FRACP FSCAI Interventional Cardiologist Royal Perth Hospital Western Australia 4 th APCASH 8 th Sept 2013 Disclosure Statement
Progress In Transcatheter Aortic Valve Implantation Gerald Yong MBBS (Hons) FRACP FSCAI Interventional Cardiologist Royal Perth Hospital Western Australia 4 th APCASH 8 th Sept 2013 Disclosure Statement
Repair or Replacement
 Surgical intervention post MitraClip Device: Repair or Replacement Saudi Heart Association, February 21-24 Rüdiger Lange, MD, PhD Nicolo Piazza, MD, FRCPC, FESC German Heart Center, Munich, Germany Division
Surgical intervention post MitraClip Device: Repair or Replacement Saudi Heart Association, February 21-24 Rüdiger Lange, MD, PhD Nicolo Piazza, MD, FRCPC, FESC German Heart Center, Munich, Germany Division
Straub Endovascular System &
 Straub Endovascular System & S t r a u b E n d o v a s c u l a r To o l s Straub Endovascular System Effective debulking in occluded arteries and veins Effective debulking in many indications Rotarex
Straub Endovascular System & S t r a u b E n d o v a s c u l a r To o l s Straub Endovascular System Effective debulking in occluded arteries and veins Effective debulking in many indications Rotarex
Introduction 3. What is Peripheral Vascular Disease? 5. What Are Some of the Symptoms of Peripheral Vascular Disease? 6
 Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
TAVI Versus Suturless Valve In Intermediate Risk Patients
 TAVI Versus Suturless Valve In Intermediate Risk Patients Walid Abukhudair FRCSc President of Saudi Society for Cardiac Surgeons Head of Cardiac Surgery in KFAFH Background AS is the most frequent cardiac
TAVI Versus Suturless Valve In Intermediate Risk Patients Walid Abukhudair FRCSc President of Saudi Society for Cardiac Surgeons Head of Cardiac Surgery in KFAFH Background AS is the most frequent cardiac
Trend and Outcomes of Direct Transcatheter Aortic Valve Replacement from a Single-Center Experience
 Cardiol Ther (2018) 7:191 196 https://doi.org/10.1007/s40119-018-0115-0 BRIEF REPORT Trend and Outcomes of Direct Transcatheter Aortic Valve Replacement from a Single-Center Experience Anthony A. Bavry.
Cardiol Ther (2018) 7:191 196 https://doi.org/10.1007/s40119-018-0115-0 BRIEF REPORT Trend and Outcomes of Direct Transcatheter Aortic Valve Replacement from a Single-Center Experience Anthony A. Bavry.
Journal of the American College of Cardiology Vol. 43, No. 4, by the American College of Cardiology Foundation ISSN /04/$30.
 Journal of the American College of Cardiology Vol. 43, No. 4, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2003.11.026
Journal of the American College of Cardiology Vol. 43, No. 4, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2003.11.026
Zenith Renu AAA Converter Graft. Device Description Planning and Sizing Deployment Sequence Patient Follow-Up
 Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
Surgical Management of TOF in Adults. Dr Flora Tsang Associate Consultant Department of Cardiothoracic Surgery Queen Mary Hospital
 Surgical Management of TOF in Adults Dr Flora Tsang Associate Consultant Department of Cardiothoracic Surgery Queen Mary Hospital Tetralogy of Fallot (TOF) in Adults Most common cyanotic congenital heart
Surgical Management of TOF in Adults Dr Flora Tsang Associate Consultant Department of Cardiothoracic Surgery Queen Mary Hospital Tetralogy of Fallot (TOF) in Adults Most common cyanotic congenital heart
Multicentre clinical study evaluating a novel resheatable self-expanding transcatheter aortic valve system
 Multicentre clinical study evaluating a novel resheatable self-expanding transcatheter aortic valve system Preliminary Results: Acute and 1-year Outcomes Ganesh Manoharan, MBBCh, MD, FRCP Consultant Cardiologist
Multicentre clinical study evaluating a novel resheatable self-expanding transcatheter aortic valve system Preliminary Results: Acute and 1-year Outcomes Ganesh Manoharan, MBBCh, MD, FRCP Consultant Cardiologist
TAVI Technology and Procedural Changes
 TCT AP 2013 Seoul, South Korea April, 2013 TAVI Technology and Procedural Changes Eberhard Grube MD, FACC, FSCAI University Hospital, Dept of Medicine II, Bonn, Germany Hospital Alemão Oswaldo Cruz, São
TCT AP 2013 Seoul, South Korea April, 2013 TAVI Technology and Procedural Changes Eberhard Grube MD, FACC, FSCAI University Hospital, Dept of Medicine II, Bonn, Germany Hospital Alemão Oswaldo Cruz, São
Transcatheter Pulmonary Valve Implantation. Description
 Subject: Transcatheter Pulmonary Valve Implantation Page: 1 of 16 Last Review Status/Date: September 2016 Transcatheter Pulmonary Valve Implantation Description Transcatheter pulmonary valve implantation
Subject: Transcatheter Pulmonary Valve Implantation Page: 1 of 16 Last Review Status/Date: September 2016 Transcatheter Pulmonary Valve Implantation Description Transcatheter pulmonary valve implantation
(12) United States Patent
 US0094.80557B2 (12) United States Patent Pellegrini et al. (10) Patent No.: (45) Date of Patent: US 9.480,557 B2 Nov. 1, 2016 (54) (75) (73) (*) (21) (22) (65) (60) (51) (52) (58) STENTS FOR PROSTHETIC
US0094.80557B2 (12) United States Patent Pellegrini et al. (10) Patent No.: (45) Date of Patent: US 9.480,557 B2 Nov. 1, 2016 (54) (75) (73) (*) (21) (22) (65) (60) (51) (52) (58) STENTS FOR PROSTHETIC
Options for my no option Patients Treating Heart Conditions Via a Tiny Catheter
 Options for my no option Patients Treating Heart Conditions Via a Tiny Catheter Nirat Beohar, MD Associate Professor of Medicine Director Cardiac Catheterization Laboratory, Medical Director Structural
Options for my no option Patients Treating Heart Conditions Via a Tiny Catheter Nirat Beohar, MD Associate Professor of Medicine Director Cardiac Catheterization Laboratory, Medical Director Structural
Aortic valve implantation using the femoral and apical access: a single center experience.
 Aortic valve implantation using the femoral and apical access: a single center experience. R. Hoffmann, K. Brehmer, R. Koos, R. Autschbach, N. Marx, G. Dohmen Rainer Hoffmann, University Aachen, Germany
Aortic valve implantation using the femoral and apical access: a single center experience. R. Hoffmann, K. Brehmer, R. Koos, R. Autschbach, N. Marx, G. Dohmen Rainer Hoffmann, University Aachen, Germany
