Document 3 Summary of Data and Guidance for Medical Professionals
|
|
|
- Samuel Holland
- 5 years ago
- Views:
Transcription
1 Document 3 Summary of Data and Guidance for Medical Professionals Anvirzel Anvirzel is a novel antitumor compound extracted from the plant Nerium oleander, which belongs to the family Apocynaceae. Anvirzel consists of several compounds, including complex polysaccharides, proteins and individual sugars. It contains non-water soluble compounds, and two of these have been specifically identified by molecular weight and fragmentation characterization as Oleandrin and Oleandrigenin. These two compounds of Anvirzel are cytotoxic. 1.1 Indications and Usage Potential indications include the treatment of cancer and Hepatitis C. Anvirzel has completed a Phase I Clinical Trial at the Cleveland Clinic, Cleveland, Ohio. 1.2 Contraindications Patients should not receive Anvirzel if they: Have a history of digitalis toxicity of any kind; Are receiving concurrent digoxin or digitoxin; Have acute myocardial infarction or severe pulmonary disease; Patients should not co-administer the following drugs with digoxin / Anvirzel. Verapamil, aminodarone, propafenone, indomethacin, itraconazole, alprazolam, sotalol, quinidine or Furosemide Increase the risk of digitalis toxicity. Flecainide Increases serum digoxin concentration, mechanism not known. Clarithromycin Decreases the conversion of digoxin to dihydrodigoxin by the gut flora.
2 1.3 Warnings Even though there have been no reports from the Clinical Trials of material adverse side effects other than the effects described under Precautions, the following information is included for the benefit of medical professionals. This Guidance has been written to include and address the possible side effects that could be associated with the administration of a cardiac glycoside. 1.4 Precautions General The drug Anvirzel has been shown to be safe in animals and patients. The Phase I Clinical Trials Conclusion is Anvirzel can be safely administered at doses up to 1.2 ml/m2/d. No dose limiting toxicities were found. When therapeutic doses of oleander extract are administered to patients, the following side effects may occur and the attending physician should take the necessary steps to reduce these side effects Allergic Reactions It may be possible to have itching and exfoliation of the epidermis. These symptoms can be alleviated if adjuvant therapies such as antihistamines or cortisones are provided. If adjuvant therapy provided no benefit, then the treatment with Anvirzel should be abandoned Cardiac Toxicity Anvirzel contains very small sub-therapeutic amount of cardiac glycosides such as oleandrin. These glycosides exert similar actions like digoxin, an approved drug to treat cardiac problems. 1 ml of Anvirzel contains approximately between 20 and 100 ug/ml of oleandrin. Anvirzel has a cross reactivity to digoxin immunoassays and has a false-positive digoxin concentration between 10 and 40 ug/ml. The concentration of oleandrin and the cross-reactive concentration of digoxin is very low as compared to the recommended adult therapeutic dose of digoxin, ug/day. Even though the cardiac glycosides are present at a very low concentration in Anvirzel as compared to the recommended doses of digoxin, it is important to monitor the patients for possible cardiac toxicity. Cardiac glycosides toxicity develops more frequently and lasts longer in patients with renal impairment because of the decreased excretion of cardiac glycosides. Therefore, the patients with renal impairment have to be monitored for possible cardiac glycosides toxicity more frequently by measuring the serum oleandrin and false positive digoxin levels than in patients with normal renal function.
3 In patients with hypokalemia, toxicity may occur because potassium depletion sensitizes the myocardium to cardiac glycosides. Therefore, it is desirable to maintain normal serum potassium levels in patients being treated with Anvirzel. Hypokalemia may result from diuretic, amphotericin B or corticosteroid therapy and from dialysis or mechanical suction of gastrointestinal secretions. It may also accompany malnutrition, diarrhea, prolonged vomiting, old age and long-standing heart failure. In general, rapid changes in serum potassium or other electrolytes should be avoided. Calcium, particularly when administered rapidly by the intravenous route, may produce serious arrhythmia in patients taking Anvirzel. Hypercalcemia from any cause predisposes the patient to digitalis toxicity. These interactions are related to the fact that calcium affects contractility and excitability of the heart in a manner similar to cardiac glycosides. Hypomagnesemia may predispose to digitalis toxicity. If low magnesium levels are detected in a patient on Anvirzel, replacement therapy should be instituted. Quinidine, verapamil, amiodarone, propafenone, indomethacin, itraconazole, and alprazolam cause a rise in serum digoxin and oleandrin concentration, with the implication that digitalis intoxication may result. This rise appears to be proportional to the dose. The effect is mediated by a reduction in the cardiac glycosides clearance and, in the case of quinidine, decreased volume of distribution as well. Patients with acute myocardial infarction or severe pulmonary disease may be unusually sensitive to cardiac glycosides-induced disturbances of rhythm. These patients may not be suitable for Anvirzel therapy. In patients with Wolff-Parkinson-White Syndrome and atrial fibrillation, cardiac glycosides can enhance transmission of impulses through the accessory pathway. This effect may result in extremely rapid ventricular rates and even ventricular fibrillation. Anvirzel may produce false positive S-T changes in the electrocardiogram during exercise testing Miscellaneous Effects In most cases, pain in the breast glands, similar to that occurring during adolescence has been noted, along with an increase in sexual activity. During the regular treatment period, the leukocyte count has been observed to rise above normal values, sometimes up to 12,000-24, Drug Interactions Concomitant use of Anvirzel and sympathomimetics may increase the risk of cardiac arrhythmia, because both enhance ectopic pacemaker activity. Succinylcholine may cause a sudden extrusion of potassium from muscle cells, and may thereby cause arrhythmia in patients on Anvirzel. Patients who are on beta-adrenergic blockers or calcium channel blockers may be susceptible to complete heart block if they take Anvirzel.
4 Furthermore, caution should be exercised when combining Anvirzel with any drug that may cause a significant deterioration in renal function, since this may impair the excretion of cardiac glycosides. 1.6 Pregnant Use Animal reproduction studies have not been conducted with Anvirzel. It is not known whether Anvirzel can cause fetal harm when administered to a pregnant woman or whether Anvirzel can affect reproductive capacity. 1.7 Pediatric Use Safety and effectiveness in children have not been studied. 1.8 Nursing Mothers Mothers should refrain from breast-feeding during the treatment to avoid potential injury to their children. 1.9 Overdosage Symptoms The Phase I study demonstrated that dosages up to 3 times the recommended dosage didn t produce cardiologic alterations in the patient without cardiac lesions and who was not using digitalis. Unifocal or multiform ventricular premature contractions, especially in bigeminal or trigeminal patterns, are the most common arrhythmia associated with cardiac glycosides toxicity in adults with heart disease. Ventricular tachycardia may result from digitalis toxicity. Atrioventricular (AV) dissociation, accelerated junctional (nodal) rhythm and atria] tachycardia with block are also common arrhythmia caused by cardiac glycoside overdose. Excessive slowing of the pulse is a clinical sign of Anvirzel overdose. Note: The electrocardiogram is fundamental in determining the presence and nature of these cardiac disturbances. Cardiac glycosides may also induce other changes in the ECG, e.g., PR prolongation, ST depressions, which represent cardiac glycosides effect and may or may not be associated with digitalis toxicity Gastrointestinal Anorexia, nausea, vomiting and less commonly diarrhea are common early symptoms of overdose. However, uncontrolled heart failure may also produce such symptoms. Digitalis toxicity very rarely may cause abdominal pain and hemorrhagic necrosis of the intestines.
5 CNS Visual disturbances (blurred or yellow vision), headache, weakness, dizziness, apathy and psychosis can occur Others Anvirzel has certain polysaccharides known to exert immune stimulating properties. Overdose of the drug may result in seizures, anaphylactic shocks, breathing difficulties and decreased urine volume. If any of these symptoms occur, the patient should call the attending physician Treatment of Arrhythmia Produced by Overdosage Anvirzel should be discontinued until all signs of toxicity are gone. Discontinuation may be all that is necessary if toxic manifestations are not severe Dosage and Administration The product should be reconstituted by aseptically adding 10 ml of distilled or purified water, into the vial and gently swirling the contents until all visible particles have dissolved. The freeze-dried Anvirzel powder should be stored at room temperature between 15 and 30 C. Any unused, reconstituted Anvirzel solution should be stored between 2 and 8 C and protected from light. Reconstituted product should be used within 20 days How Supplied Anvirzel is a sterile, freeze-dried powder to be reconstituted before using. Anvirzel is supplied in 10 ml butyl-rubber stoppered vials. Each vial contains 150 mg of Nerium oleander extract, 500 mg mannitol, 50 mg sodium ascorbate, 2 mg ascorbic acid, 10 mg methylparaben sodium, and 1 mg propylparaben sodium. Each vial with 150 mg of oleander extract contains approximately ug oleandrin and ug oleandrigenin; therefore, there is approximately ug oleandrin and ug oleandrigenin per ml of reconstituted solution.
Chapter 26. Media Directory. Dysrhythmias. Diagnosis/Treatment of Dysrhythmias. Frequency in Population Difficult to Predict
 Chapter 26 Drugs for Dysrythmias Slide 33 Slide 35 Media Directory Propranolol Animation Amiodarone Animation Upper Saddle River, New Jersey 07458 All rights reserved. Dysrhythmias Abnormalities of electrical
Chapter 26 Drugs for Dysrythmias Slide 33 Slide 35 Media Directory Propranolol Animation Amiodarone Animation Upper Saddle River, New Jersey 07458 All rights reserved. Dysrhythmias Abnormalities of electrical
PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker
 PACKAGE INSERT Pr PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker ACTIONS AND CLINICAL PHARMACOLOGY Phentolamine produces an alpha-adrenergic
PACKAGE INSERT Pr PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker ACTIONS AND CLINICAL PHARMACOLOGY Phentolamine produces an alpha-adrenergic
SODIUM POLYSTYRENE SULFONATE Suspension, USP
 SODIUM POLYSTYRENE SULFONATE Suspension, USP DESCRIPTION Sodium Polystyrene Sulfonate Suspension, USP can be administered orally or in an enema. It is a cherryflavored suspension containing 15 grams of
SODIUM POLYSTYRENE SULFONATE Suspension, USP DESCRIPTION Sodium Polystyrene Sulfonate Suspension, USP can be administered orally or in an enema. It is a cherryflavored suspension containing 15 grams of
Heart Failure (HF) Treatment
 Heart Failure (HF) Treatment Heart Failure (HF) Complex, progressive disorder. The heart is unable to pump sufficient blood to meet the needs of the body. Its cardinal symptoms are dyspnea, fatigue, and
Heart Failure (HF) Treatment Heart Failure (HF) Complex, progressive disorder. The heart is unable to pump sufficient blood to meet the needs of the body. Its cardinal symptoms are dyspnea, fatigue, and
Antiarrhythmic Drugs 1/31/2018 1
 Antiarrhythmic Drugs 1/31/2018 1 Normal conduction pathway: 1- SA node generates action potential and delivers it to the atria and the AV node 2- The AV node delivers the impulse to purkinje fibers Other
Antiarrhythmic Drugs 1/31/2018 1 Normal conduction pathway: 1- SA node generates action potential and delivers it to the atria and the AV node 2- The AV node delivers the impulse to purkinje fibers Other
PRODUCT MONOGRAPH. DIGOXIN INJECTION, C.S.D. Digoxin Injection, C.S.D., 0.25 mg/ml
 PRODUCT MONOGRAPH Pr DIGOXIN INJECTION, C.S.D. Digoxin Injection, C.S.D., 0.25 mg/ml Pr Pediatric DIGOXIN INJECTION, C.S.D. Digoxin Injection, C.S.D., 0.05 mg/ml Cardiotonic Glycoside Sandoz Canada Inc.
PRODUCT MONOGRAPH Pr DIGOXIN INJECTION, C.S.D. Digoxin Injection, C.S.D., 0.25 mg/ml Pr Pediatric DIGOXIN INJECTION, C.S.D. Digoxin Injection, C.S.D., 0.05 mg/ml Cardiotonic Glycoside Sandoz Canada Inc.
Antiarrhythmic Drugs
 Antiarrhythmic Drugs DR ATIF ALQUBBANY A S S I S T A N T P R O F E S S O R O F M E D I C I N E / C A R D I O L O G Y C O N S U L T A N T C A R D I O L O G Y & I N T E R V E N T I O N A L E P A C H D /
Antiarrhythmic Drugs DR ATIF ALQUBBANY A S S I S T A N T P R O F E S S O R O F M E D I C I N E / C A R D I O L O G Y C O N S U L T A N T C A R D I O L O G Y & I N T E R V E N T I O N A L E P A C H D /
Heart Failure. Dr. Alia Shatanawi
 Heart Failure Dr. Alia Shatanawi Left systolic dysfunction secondary to coronary artery disease is the most common cause, account to 70% of all cases. Heart Failure Heart is unable to pump sufficient blood
Heart Failure Dr. Alia Shatanawi Left systolic dysfunction secondary to coronary artery disease is the most common cause, account to 70% of all cases. Heart Failure Heart is unable to pump sufficient blood
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP
 PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
PRODUCT MONOGRAPH. Digoxin Tablets C.S.D mg, mg, 0.25 mg. Cardiotonic Glycoside
 PRODUCT MONOGRAPH Pr APO-DIGOXIN Digoxin Tablets C.S.D. 0.0625 mg, 0.125 mg, 0.25 mg Cardiotonic Glycoside APOTEX INC. DATE OF PREPARATION: 150 Signet Drive June 05, 2006 Weston, Ontario M9L 1T9 Control
PRODUCT MONOGRAPH Pr APO-DIGOXIN Digoxin Tablets C.S.D. 0.0625 mg, 0.125 mg, 0.25 mg Cardiotonic Glycoside APOTEX INC. DATE OF PREPARATION: 150 Signet Drive June 05, 2006 Weston, Ontario M9L 1T9 Control
Clinical Toxicology Toxicity of Digitalis Glycosides 5 th Year (Lab 3)
 Clinical Toxicology Toxicity of Digitalis Glycosides 5 th Year (Lab 3) Lecturer: Rua Abbas Al-Hamdy Department of Pharmacology & Toxicology University of Al-Mustansiriyah 2017-2018 Introduction: Digitalis
Clinical Toxicology Toxicity of Digitalis Glycosides 5 th Year (Lab 3) Lecturer: Rua Abbas Al-Hamdy Department of Pharmacology & Toxicology University of Al-Mustansiriyah 2017-2018 Introduction: Digitalis
PRODUCT MONOGRAPH. Digoxin Tablets, C.S.D mg, mg and 0.25 mg. Cardiotonic Glycoside
 PRODUCT MONOGRAPH Pr LANOXIN Digoxin Tablets, C.S.D. 0.0625 mg, 0.125 mg and 0.25 mg Cardiotonic Glycoside PHARMASCIENCE INC. Date of Revision: 6111 Royalmount Ave., Suite 100 January 6, 2016 Montreal,
PRODUCT MONOGRAPH Pr LANOXIN Digoxin Tablets, C.S.D. 0.0625 mg, 0.125 mg and 0.25 mg Cardiotonic Glycoside PHARMASCIENCE INC. Date of Revision: 6111 Royalmount Ave., Suite 100 January 6, 2016 Montreal,
Instruct patient and caregivers: Need for constant monitoring Potential complications of drug therapy
 Assessment Prior to administration: Assess patient for chest pain, dysrhythmias, and vital signs (initially and throughout therapy) Obtain complete medical history, including allergies, especially heart
Assessment Prior to administration: Assess patient for chest pain, dysrhythmias, and vital signs (initially and throughout therapy) Obtain complete medical history, including allergies, especially heart
PRODUCT MONOGRAPH. Digoxin Tablets, C.S.D mg, mg and 0.25 mg. Digoxin Oral Solution, C.S.D mg/ml. Cardiotonic Glycoside
 PRODUCT MONOGRAPH Pr TOLOXIN Digoxin Tablets, C.S.D. 0.0625 mg, 0.125 mg and 0.25 mg Digoxin Oral Solution, C.S.D. 0.05 mg/ml Cardiotonic Glycoside PENDOPHARM, Division of Pharmascience Inc. 6111 Royalmount
PRODUCT MONOGRAPH Pr TOLOXIN Digoxin Tablets, C.S.D. 0.0625 mg, 0.125 mg and 0.25 mg Digoxin Oral Solution, C.S.D. 0.05 mg/ml Cardiotonic Glycoside PENDOPHARM, Division of Pharmascience Inc. 6111 Royalmount
Arrhythmias. Simple-dysfunction cause abnormalities in impulse formation and conduction in the myocardium.
 Arrhythmias Simple-dysfunction cause abnormalities in impulse formation and conduction in the myocardium. However, in clinic it present as a complex family of disorders that show variety of symptoms, for
Arrhythmias Simple-dysfunction cause abnormalities in impulse formation and conduction in the myocardium. However, in clinic it present as a complex family of disorders that show variety of symptoms, for
PHARMACOLOGY OF ARRHYTHMIAS
 PHARMACOLOGY OF ARRHYTHMIAS Course: Integrated Therapeutics 1 Lecturer: Dr. E. Konorev Date: November 27, 2012 Materials on: Exam #5 Required reading: Katzung, Chapter 14 1 CARDIAC ARRHYTHMIAS Abnormalities
PHARMACOLOGY OF ARRHYTHMIAS Course: Integrated Therapeutics 1 Lecturer: Dr. E. Konorev Date: November 27, 2012 Materials on: Exam #5 Required reading: Katzung, Chapter 14 1 CARDIAC ARRHYTHMIAS Abnormalities
Mr. Eknath Kole M.S. Pharm (NIPER Mohali)
 M.S. Pharm (NIPER Mohali) Drug Class Actions Therapeutic Uses Pharmacokinetics Adverse Effects Other Quinidine IA -Binds to open and inactivated Na+ -Decreases the slope of Phase 4 spontaneous depolarization
M.S. Pharm (NIPER Mohali) Drug Class Actions Therapeutic Uses Pharmacokinetics Adverse Effects Other Quinidine IA -Binds to open and inactivated Na+ -Decreases the slope of Phase 4 spontaneous depolarization
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DigiFab, 40 mg/vial digoxin immune Fab, Powder for solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each glass vial
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DigiFab, 40 mg/vial digoxin immune Fab, Powder for solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each glass vial
PACKAGE LEAFLET: INFORMATION FOR THE USER. Variquel 1 mg powder and solvent for solution for injection Terlipressin acetate
 PACKAGE LEAFLET: INFORMATION FOR THE USER Variquel 1 mg powder and solvent for solution for injection Terlipressin acetate Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER Variquel 1 mg powder and solvent for solution for injection Terlipressin acetate Read all of this leaflet carefully before you start using this medicine because
PRODUCT INFORMATION RESONIUM A. Na m
 PRODUCT INFORMATION RESONIUM A NAME OF THE MEDICINE Non-proprietary Name Sodium polystyrene sulfonate Chemical Structure CH - 2 CH SO 3 Na + n CAS Number 28210-41-5 [9003-59-2] CH 2 CH SO - 3 m DESCRIPTION
PRODUCT INFORMATION RESONIUM A NAME OF THE MEDICINE Non-proprietary Name Sodium polystyrene sulfonate Chemical Structure CH - 2 CH SO 3 Na + n CAS Number 28210-41-5 [9003-59-2] CH 2 CH SO - 3 m DESCRIPTION
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT DigiFab (referred to as DIGIFAB) 40 mg/vial digoxin immune Fab, Powder for solution for infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT DigiFab (referred to as DIGIFAB) 40 mg/vial digoxin immune Fab, Powder for solution for infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
CORDARONE Sanofi-Aventis
 CORDARONE Sanofi-Aventis Composition Amiodarone 200 mg; tablets. Also AMPOULES 150 mg/3 ml. Indications Tablets Prevention of recurrence of the following: life-threatening ventricular tachycardias in which
CORDARONE Sanofi-Aventis Composition Amiodarone 200 mg; tablets. Also AMPOULES 150 mg/3 ml. Indications Tablets Prevention of recurrence of the following: life-threatening ventricular tachycardias in which
BRICANYL INJECTION. terbutaline sulfate PRODUCT INFORMATION
 BRICANYL INJECTION terbutaline sulfate PRODUCT INFORMATION NAME OF THE MEDICINE Terbutaline sulfate, 2-(tert-butylamino)-1-(3,5-dihydroxyphenyl) ethanol sulfate, a sympathomimetic bronchodilator with a
BRICANYL INJECTION terbutaline sulfate PRODUCT INFORMATION NAME OF THE MEDICINE Terbutaline sulfate, 2-(tert-butylamino)-1-(3,5-dihydroxyphenyl) ethanol sulfate, a sympathomimetic bronchodilator with a
Metformin Hydrochloride
 Metformin Hydrochloride 500 mg, 850 mg, 500 mg LA and 750 mg LA Tablet Description Informet is a preparation of metformin hydrochloride that belongs to a biguanide class of oral antidiabetic drugs. Metformin
Metformin Hydrochloride 500 mg, 850 mg, 500 mg LA and 750 mg LA Tablet Description Informet is a preparation of metformin hydrochloride that belongs to a biguanide class of oral antidiabetic drugs. Metformin
Composition Each ml of Ventol solution for inhalation contains 5 mg Salbutamol (as sulphate).
 VENTOL Composition Each ml of Ventol solution for inhalation contains 5 mg Salbutamol (as sulphate). Respiratory Solution Action Salbutamol is a short-acting, relatively selective beta2-adrenoceptor agonist.
VENTOL Composition Each ml of Ventol solution for inhalation contains 5 mg Salbutamol (as sulphate). Respiratory Solution Action Salbutamol is a short-acting, relatively selective beta2-adrenoceptor agonist.
Arrhythmias. 1. beat too slowly (sinus bradycardia). Like in heart block
 Arrhythmias It is a simple-dysfunction caused by abnormalities in impulse formation and conduction in the myocardium. The heart is designed in such a way that allows it to generate from the SA node electrical
Arrhythmias It is a simple-dysfunction caused by abnormalities in impulse formation and conduction in the myocardium. The heart is designed in such a way that allows it to generate from the SA node electrical
Package leaflet: Information for the patient. VEPROL Film coated tablets 40 mg or 80 mg (Verapamil hydrochloride)
 Package leaflet: Information for the patient VEPROL Film coated tablets 40 mg or 80 mg (Verapamil hydrochloride) Read this leaflet carefully before you start taking this medicine. - Keep this leaflet.
Package leaflet: Information for the patient VEPROL Film coated tablets 40 mg or 80 mg (Verapamil hydrochloride) Read this leaflet carefully before you start taking this medicine. - Keep this leaflet.
4/14/15 HTEC 91. Topics for Today. Guess That Rhythm. Premature Ventricular Contractions (PVCs) Ventricular Rhythms
 4/14/15 Topics for Today HTEC 91 Medical Office Diagnostic Tests Week 5 Ventricular Rhythms PVCs: Premature Ventricular Contractions VT: Ventricular Tachycardia VF: Ventricular Fibrillation Asystole Study
4/14/15 Topics for Today HTEC 91 Medical Office Diagnostic Tests Week 5 Ventricular Rhythms PVCs: Premature Ventricular Contractions VT: Ventricular Tachycardia VF: Ventricular Fibrillation Asystole Study
Rhythm Control: Is There a Role for the PCP? Blake Norris, MD, FACC BHHI Primary Care Symposium February 28, 2014
 Rhythm Control: Is There a Role for the PCP? Blake Norris, MD, FACC BHHI Primary Care Symposium February 28, 2014 Financial disclosures Consultant Medtronic 3 reasons to evaluate and treat arrhythmias
Rhythm Control: Is There a Role for the PCP? Blake Norris, MD, FACC BHHI Primary Care Symposium February 28, 2014 Financial disclosures Consultant Medtronic 3 reasons to evaluate and treat arrhythmias
Dysrhythmias. Dysrythmias & Anti-Dysrhythmics. EKG Parameters. Dysrhythmias. Components of an ECG Wave. Dysrhythmias
 Dysrhythmias Dysrythmias & Anti-Dysrhythmics Rhythm bad in the heart: Whitewater rafting Electrical impulses coordinate heart Reduction in Cardiac Output PEA Asystole Components of an ECG Wave EKG Parameters
Dysrhythmias Dysrythmias & Anti-Dysrhythmics Rhythm bad in the heart: Whitewater rafting Electrical impulses coordinate heart Reduction in Cardiac Output PEA Asystole Components of an ECG Wave EKG Parameters
Drugs Used in Heart Failure. Assistant Prof. Dr. Najlaa Saadi PhD pharmacology Faculty of Pharmacy University of Philadelphia
 Drugs Used in Heart Failure Assistant Prof. Dr. Najlaa Saadi PhD pharmacology Faculty of Pharmacy University of Philadelphia Heart Failure Heart failure (HF), occurs when cardiac output is inadequate to
Drugs Used in Heart Failure Assistant Prof. Dr. Najlaa Saadi PhD pharmacology Faculty of Pharmacy University of Philadelphia Heart Failure Heart failure (HF), occurs when cardiac output is inadequate to
Atrial Fibrillation 10/2/2018. Depolarization & ECG. Atrial Fibrillation. Hemodynamic Consequences
 Depolarization & ECG Atrial Fibrillation How to make ORDER out of CHAOS Julia Shih, VMD, DACVIM (Cardiology) October 27, 2018 Depolarization & ECG Depolarization & ECG Atrial Fibrillation Hemodynamic Consequences
Depolarization & ECG Atrial Fibrillation How to make ORDER out of CHAOS Julia Shih, VMD, DACVIM (Cardiology) October 27, 2018 Depolarization & ECG Depolarization & ECG Atrial Fibrillation Hemodynamic Consequences
Angina pectoris due to coronary atherosclerosis : Atenolol is indicated for the long term management of patients with angina pectoris.
 Lonet Tablet Description Lonet contains Atenolol, a synthetic β1 selective (cardioselective) adrenoreceptor blocking agent without membrane stabilising or intrinsic sympathomimetic (partial agonist) activity.
Lonet Tablet Description Lonet contains Atenolol, a synthetic β1 selective (cardioselective) adrenoreceptor blocking agent without membrane stabilising or intrinsic sympathomimetic (partial agonist) activity.
Pediatrics ECG Monitoring. Pediatric Intensive Care Unit Emergency Division
 Pediatrics ECG Monitoring Pediatric Intensive Care Unit Emergency Division 1 Conditions Leading to Pediatric Cardiology Consultation 12.7% of annual consultation Is arrhythmias problems Geggel. Pediatrics.
Pediatrics ECG Monitoring Pediatric Intensive Care Unit Emergency Division 1 Conditions Leading to Pediatric Cardiology Consultation 12.7% of annual consultation Is arrhythmias problems Geggel. Pediatrics.
Core Safety Profile. Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg. Date of FAR:
 Core Safety Profile Active substance: Bisoprolol Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg P - RMS: FI/H/PSUR/0002/002 Date of FAR: 13.12.2011
Core Safety Profile Active substance: Bisoprolol Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg P - RMS: FI/H/PSUR/0002/002 Date of FAR: 13.12.2011
50% Concentrated Injection
 NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
B. 14 Antidysrhythmic drugs. a. Classify antidysrhythmics by their electrophysiological actions. Vaughan-Williams classification
 B. 14 Antidysrhythmic drugs a. Classify antidysrhythmics by their electrophysiological actions. Vaughan-Williams classification I II III IV membrane stabilizers all ERP, ERP/APD, all except c APD classified
B. 14 Antidysrhythmic drugs a. Classify antidysrhythmics by their electrophysiological actions. Vaughan-Williams classification I II III IV membrane stabilizers all ERP, ERP/APD, all except c APD classified
PRODUCT MONOGRAPH. 2% Lidocaine Hydrochloride Injection USP 20 mg/ml
 PRODUCT MONOGRAPH 2% Lidocaine Hydrochloride Injection USP 20 mg/ml 0.4 % Lidocaine Hydrochloride and 5% Dextrose Injection USP Lidocaine Hydrochloride and Dextrose Injection USP Lidocaine Hydrochloride
PRODUCT MONOGRAPH 2% Lidocaine Hydrochloride Injection USP 20 mg/ml 0.4 % Lidocaine Hydrochloride and 5% Dextrose Injection USP Lidocaine Hydrochloride and Dextrose Injection USP Lidocaine Hydrochloride
NEW ZEALAND DATA SHEET ACUPAN TM. 3. PHARMACEUTICAL FORM White, round, biconvex, film-coated tablets (7 mm diameter) engraved APN on one face.
 1. PRODUCT NAME ACUPAN 30 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains nefopam hydrochloride 30 mg. For a full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM
1. PRODUCT NAME ACUPAN 30 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains nefopam hydrochloride 30 mg. For a full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM
PRESCRIBING ALERT
 www.empr.com PRESCRIBING ALERT Dear Healthcare Professional, At MPR we strive to bring you important drug information in a concise and timely fashion. In keeping with this goal, we are pleased to bring
www.empr.com PRESCRIBING ALERT Dear Healthcare Professional, At MPR we strive to bring you important drug information in a concise and timely fashion. In keeping with this goal, we are pleased to bring
PACKAGE INSERT CALCIUM GLUCONATE INJECTION, USP 10% Electrolyte Replenisher mosmol/ml 680 mosmol/l Ca meq/ml ph
 CALCIUM GLUCONATE INJECTION, USP 10% Electrolyte Replenisher 0.68 mosmol/ml 680 mosmol/l Ca ++ 0.465 meq/ml ph 6.0 8.2 DESCRIPTION Calcium Gluconate Injection, USP is a sterile, nonpyrogenic supersaturated
CALCIUM GLUCONATE INJECTION, USP 10% Electrolyte Replenisher 0.68 mosmol/ml 680 mosmol/l Ca ++ 0.465 meq/ml ph 6.0 8.2 DESCRIPTION Calcium Gluconate Injection, USP is a sterile, nonpyrogenic supersaturated
Chapter 14. Agents used in Cardiac Arrhythmias
 Chapter 14 Agents used in Cardiac Arrhythmias Cardiac arrhythmia Approximately 50% of post-myocardial infarction fatalities result from ventricular tachycarida (VT) or ventricular fibrillation (VF). These
Chapter 14 Agents used in Cardiac Arrhythmias Cardiac arrhythmia Approximately 50% of post-myocardial infarction fatalities result from ventricular tachycarida (VT) or ventricular fibrillation (VF). These
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION
 DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
PACKAGE LEAFLET: INFORMATION FOR THE USER. Active substance: enoximone
 PACKAGE LEAFLET 1 PACKAGE LEAFLET: INFORMATION FOR THE USER Perfan Injection 100 mg/20 ml Concentrate for Solution for Injection Active substance: enoximone Read all of this leaflet carefully before you
PACKAGE LEAFLET 1 PACKAGE LEAFLET: INFORMATION FOR THE USER Perfan Injection 100 mg/20 ml Concentrate for Solution for Injection Active substance: enoximone Read all of this leaflet carefully before you
PRESCRIBING INFORMATION K-10. (Potassium Chloride Oral Solution, 10% USP) POTASSIUM REPLACEMENT THERAPY
 PRESCRIBING INFORMATION K-10 (Potassium Chloride Oral Solution, 10% USP) POTASSIUM REPLACEMENT THERAPY GlaxoSmithKline Inc Date of Revision: 7333 Mississauga Road North Aug 30, 2017 Mississauga, Ontario
PRESCRIBING INFORMATION K-10 (Potassium Chloride Oral Solution, 10% USP) POTASSIUM REPLACEMENT THERAPY GlaxoSmithKline Inc Date of Revision: 7333 Mississauga Road North Aug 30, 2017 Mississauga, Ontario
Package leaflet: Information for the user. Zoledronic Acid Accord 4 mg/5 ml concentrate for solution for infusion Zoledronic acid
 Package leaflet: Information for the user Zoledronic Acid Accord 4 mg/5 ml concentrate for solution for infusion Zoledronic acid Read all of this leaflet carefully before you start taking this medicine
Package leaflet: Information for the user Zoledronic Acid Accord 4 mg/5 ml concentrate for solution for infusion Zoledronic acid Read all of this leaflet carefully before you start taking this medicine
PACKAGE LEAFLET: INFORMATION FOR THE USER. Zoledronic Acid IBIGEN 4 mg powder and solvent for solution for infusion Zoledronic acid
 PACKAGE LEAFLET: INFORMATION FOR THE USER Zoledronic Acid IBIGEN 4 mg powder and solvent for solution for infusion Zoledronic acid Read all of this leaflet carefully before you are given Zoledronic Acid
PACKAGE LEAFLET: INFORMATION FOR THE USER Zoledronic Acid IBIGEN 4 mg powder and solvent for solution for infusion Zoledronic acid Read all of this leaflet carefully before you are given Zoledronic Acid
PRODUCT INFORMATION. Colistin Link. Colistin 150 mg/2 ml (as colistimethate sodium) powder for injection vial
 PRODUCT INFORMATION Colistin Link Colistin 150 mg/2 ml (as colistimethate sodium) powder for injection vial For Intramuscular and Intravenous use. NAME OF THE MEDICINE Colistimethate sodium for injection,
PRODUCT INFORMATION Colistin Link Colistin 150 mg/2 ml (as colistimethate sodium) powder for injection vial For Intramuscular and Intravenous use. NAME OF THE MEDICINE Colistimethate sodium for injection,
Chapter 9. Learning Objectives. Learning Objectives 9/11/2012. Cardiac Arrhythmias. Define electrical therapy
 Chapter 9 Cardiac Arrhythmias Learning Objectives Define electrical therapy Explain why electrical therapy is preferred initial therapy over drug administration for cardiac arrest and some arrhythmias
Chapter 9 Cardiac Arrhythmias Learning Objectives Define electrical therapy Explain why electrical therapy is preferred initial therapy over drug administration for cardiac arrest and some arrhythmias
Core Safety Profile. Pharmaceutical form(s)/strength: Sterile eye drops 1%, 2% Date of FAR:
 Core Safety Profile Active substance: Carteolol Pharmaceutical form(s)/strength: Sterile eye drops 1%, 2% P - RMS: SK/H/PSUR/0002/002 Date of FAR: 16.03.2012 4.1 THERAPEUTIC INDICATIONS Ocular hypertension
Core Safety Profile Active substance: Carteolol Pharmaceutical form(s)/strength: Sterile eye drops 1%, 2% P - RMS: SK/H/PSUR/0002/002 Date of FAR: 16.03.2012 4.1 THERAPEUTIC INDICATIONS Ocular hypertension
Pharmaceutical form(s)/strength: Solution: 5 mg/ml Suspensions: 2.5 and 5 mg/ml P-RMS:
 0BCore Safety Profile Active substance: Betaxolol eyedrops Pharmaceutical form(s)/strength: Solution: 5 mg/ml Suspensions: 2.5 and 5 mg/ml P-RMS: HU/H/PSUR/0010/002 Date of FAR: 20.03.2013 4.2 Posology
0BCore Safety Profile Active substance: Betaxolol eyedrops Pharmaceutical form(s)/strength: Solution: 5 mg/ml Suspensions: 2.5 and 5 mg/ml P-RMS: HU/H/PSUR/0010/002 Date of FAR: 20.03.2013 4.2 Posology
Chapter 16: Arrhythmias and Conduction Disturbances
 Complete the following. Chapter 16: Arrhythmias and Conduction Disturbances 1. Cardiac arrhythmias result from abnormal impulse, abnormal impulse, or both mechanisms together. 2. is the ability of certain
Complete the following. Chapter 16: Arrhythmias and Conduction Disturbances 1. Cardiac arrhythmias result from abnormal impulse, abnormal impulse, or both mechanisms together. 2. is the ability of certain
a lecture series by SWESEMJR
 Electrolyte disturbances Hypokalaemia Decreased extracellular potassium increases excitability in the myocardial cells and consequently the effect of very severe hypokalaemia is ventricular arrhythmia.
Electrolyte disturbances Hypokalaemia Decreased extracellular potassium increases excitability in the myocardial cells and consequently the effect of very severe hypokalaemia is ventricular arrhythmia.
2) Heart Arrhythmias 2 - Dr. Abdullah Sharif
 2) Heart Arrhythmias 2 - Dr. Abdullah Sharif Rhythms from the Sinus Node Sinus Tachycardia: HR > 100 b/m Causes: o Withdrawal of vagal tone & Sympathetic stimulation (exercise, fight or flight) o Fever
2) Heart Arrhythmias 2 - Dr. Abdullah Sharif Rhythms from the Sinus Node Sinus Tachycardia: HR > 100 b/m Causes: o Withdrawal of vagal tone & Sympathetic stimulation (exercise, fight or flight) o Fever
M0BCore Safety Profile
 M0BCore Safety Profile Active substance: Aciclovir Pharmaceutical form(s)/strength: Tablets 200, 400 or 800 mg Dispersible tablets 200, 400 or 800 mg Oral suspensions 200 mg or 400 mg per 5 ml. Freeze
M0BCore Safety Profile Active substance: Aciclovir Pharmaceutical form(s)/strength: Tablets 200, 400 or 800 mg Dispersible tablets 200, 400 or 800 mg Oral suspensions 200 mg or 400 mg per 5 ml. Freeze
Chapter 03: Sinus Mechanisms Test Bank MULTIPLE CHOICE
 Instant download and all chapters Tesst Bank ECGs Made Easy 5th Edition Barbara J Aehlert https://testbanklab.com/download/tesst-bank-ecgs-made-easy-5th-edition-barbara-jaehlert/ Chapter 03: Sinus Mechanisms
Instant download and all chapters Tesst Bank ECGs Made Easy 5th Edition Barbara J Aehlert https://testbanklab.com/download/tesst-bank-ecgs-made-easy-5th-edition-barbara-jaehlert/ Chapter 03: Sinus Mechanisms
Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup.
 Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
PRESCRIBING INFORMATION WITH CONSUMER INFORMATION MYDRIACYL. tropicamide ophthalmic solution, USP. 0.5% and 1% w/v.
 PRESCRIBING INFORMATION WITH CONSUMER INFORMATION Pr MYDRIACYL tropicamide ophthalmic solution, USP 0.5% and 1% w/v Anticholinergic Alcon Canada Inc. 2665 Meadowpine Blvd. Mississauga, ON L5N 8C7 www.alcon.ca
PRESCRIBING INFORMATION WITH CONSUMER INFORMATION Pr MYDRIACYL tropicamide ophthalmic solution, USP 0.5% and 1% w/v Anticholinergic Alcon Canada Inc. 2665 Meadowpine Blvd. Mississauga, ON L5N 8C7 www.alcon.ca
ANTI-ARRHYTHMICS AND WARFARIN. Dr Nithish Jayakumar
 ANTI-ARRHYTHMICS AND WARFARIN Dr Nithish Jayakumar Contents 1. Anti-arrhythmics Pacemaker and myocardial potentials Drug classes mechanisms; s/e; contra-indications Management of common arrhythmias 2.
ANTI-ARRHYTHMICS AND WARFARIN Dr Nithish Jayakumar Contents 1. Anti-arrhythmics Pacemaker and myocardial potentials Drug classes mechanisms; s/e; contra-indications Management of common arrhythmias 2.
DOBUTamine INJECTION, USP R x only
 DOBUTamine INJECTION, USP R x only DESCRIPTION Dobutamine Injection, USP is 1,2-benzenediol, 4-[2-[[3-(4-hydro-xyphenyl)-1- methylpropyl]amino]ethyl]-hydrochloride, (±). It is a synthetic catecholamine.
DOBUTamine INJECTION, USP R x only DESCRIPTION Dobutamine Injection, USP is 1,2-benzenediol, 4-[2-[[3-(4-hydro-xyphenyl)-1- methylpropyl]amino]ethyl]-hydrochloride, (±). It is a synthetic catecholamine.
NORMOSOL -R MULTIPLE ELECTROLYTES INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid Flexible Plastic Container
 NORMOSOL -R MULTIPLE ELECTROLYTES INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid Flexible Plastic Container R x only DESCRIPTION Normosol-R is a sterile, nonpyrogenic isotonic
NORMOSOL -R MULTIPLE ELECTROLYTES INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid Flexible Plastic Container R x only DESCRIPTION Normosol-R is a sterile, nonpyrogenic isotonic
Atrial fibrillation in the ICU
 Atrial fibrillation in the ICU Atrial fibrillation Preexisting or incident (new onset) among nearly one in three critically ill patients Formation of arrhythogenic substrate usually fibrosis (CHF, hypertension,
Atrial fibrillation in the ICU Atrial fibrillation Preexisting or incident (new onset) among nearly one in three critically ill patients Formation of arrhythogenic substrate usually fibrosis (CHF, hypertension,
PACKAGE LEAFLET: INFORMATION FOR THE USER. CALCICLO STEROP 11 meq 10 ml Solution for injection. Dihydrated Calcium Chloride
 PACKAGE LEAFLET: INFORMATION FOR THE USER CALCICLO STEROP 11 meq 10 ml Solution for injection Dihydrated Calcium Chloride Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER CALCICLO STEROP 11 meq 10 ml Solution for injection Dihydrated Calcium Chloride Read all of this leaflet carefully before you start using this medicine because
MAT vs AFIB. Henry Clemo. Fast & Easy ECGs, 2E 2013 The McGraw-Hill Companies, Inc. All rights reserved.
 MAT vs AFIB Henry Clemo 1 Multifocal Atrial Tachycardia (MAT) > 3 P wave morphologies HR > 100 HR < 100 wandering pacemaker I 2 Multifocal Atrial Tachycardia 3 Multifocal Atrial Tachycardia 4 Multifocal
MAT vs AFIB Henry Clemo 1 Multifocal Atrial Tachycardia (MAT) > 3 P wave morphologies HR > 100 HR < 100 wandering pacemaker I 2 Multifocal Atrial Tachycardia 3 Multifocal Atrial Tachycardia 4 Multifocal
ROSOBAC-1GM / ROSOBAC-FORT
 ROSOBAC-1GM / ROSOBAC-FORT ROSOBAC - 1GM. COMPOSITION : Each vial contains Sterile Cefoperazone Sodium IP Eq. to Anhydrous Cefoperazone - Sterile Sulbactam Sodium USP Eq. to Anhydrous Sulbactam - ROSOBAC
ROSOBAC-1GM / ROSOBAC-FORT ROSOBAC - 1GM. COMPOSITION : Each vial contains Sterile Cefoperazone Sodium IP Eq. to Anhydrous Cefoperazone - Sterile Sulbactam Sodium USP Eq. to Anhydrous Sulbactam - ROSOBAC
ALS MODULE 7 Pharmacology
 ALS MODULE 7 Pharmacology Relates to HLT404C Apply Advanced Resuscitation Techniques Introduction There are no studies that addressed the order of drug administration. There is inadequate evidence to define
ALS MODULE 7 Pharmacology Relates to HLT404C Apply Advanced Resuscitation Techniques Introduction There are no studies that addressed the order of drug administration. There is inadequate evidence to define
Comparative Study of Different Digoxin Treatment Regimens in Egyptian Hospitals. For Partial Fulfillment of Master Degree in Pharmaceutical Sciences
 Comparative Study of Different Digoxin Treatment Regimens in Egyptian Hospitals A Thesis presented by Sahar Atef Azmy Al Shabasy, BSc Teaching Assistant, Clinical Pharmacy Department, Faculty of Pharmacy,
Comparative Study of Different Digoxin Treatment Regimens in Egyptian Hospitals A Thesis presented by Sahar Atef Azmy Al Shabasy, BSc Teaching Assistant, Clinical Pharmacy Department, Faculty of Pharmacy,
SOTALAR Tablets (Sotalol)
 Published on: 10 Jul 2014 SOTALAR Tablets (Sotalol) Composition SOTALAR 40 Each tablet contains: Sotalol hydrochloride... 40 mg Dosage Forms Tablet Pharmacology Pharmacodynamics D,l-sotalol is a non-selective
Published on: 10 Jul 2014 SOTALAR Tablets (Sotalol) Composition SOTALAR 40 Each tablet contains: Sotalol hydrochloride... 40 mg Dosage Forms Tablet Pharmacology Pharmacodynamics D,l-sotalol is a non-selective
UNDERSTANDING YOUR ECG: A REVIEW
 UNDERSTANDING YOUR ECG: A REVIEW Health professionals use the electrocardiograph (ECG) rhythm strip to systematically analyse the cardiac rhythm. Before the systematic process of ECG analysis is described
UNDERSTANDING YOUR ECG: A REVIEW Health professionals use the electrocardiograph (ECG) rhythm strip to systematically analyse the cardiac rhythm. Before the systematic process of ECG analysis is described
Antihypertensive drugs SUMMARY Made by: Lama Shatat
 Antihypertensive drugs SUMMARY Made by: Lama Shatat Diuretic Thiazide diuretics The loop diuretics Potassium-sparing Diuretics *Hydrochlorothiazide *Chlorthalidone *Furosemide *Torsemide *Bumetanide Aldosterone
Antihypertensive drugs SUMMARY Made by: Lama Shatat Diuretic Thiazide diuretics The loop diuretics Potassium-sparing Diuretics *Hydrochlorothiazide *Chlorthalidone *Furosemide *Torsemide *Bumetanide Aldosterone
Immodium / loprarmide
 Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
DIGOXIN TOXICITY: A REVIEW
 DIGOXIN TOXICITY: A REVIEW Dr. Laxmi Narayan Goit 1* and Prof. Dr. Yang Shaning 2 1 Department of Cardiology, the first affiliated Hospital of Yangtze University, Jingzhou, Hubei, P.R China. 2 Department
DIGOXIN TOXICITY: A REVIEW Dr. Laxmi Narayan Goit 1* and Prof. Dr. Yang Shaning 2 1 Department of Cardiology, the first affiliated Hospital of Yangtze University, Jingzhou, Hubei, P.R China. 2 Department
PRESCRIBING INFORMATION ADRENALIN TOPICAL SOLUTION. Epinephrine Chloride Topical Solution, manufacturer standard 30 mg / 30 ml (1 mg/ml)
 PRESCRIBING INFORMATION ADRENALIN TOPICAL SOLUTION Epinephrine Chloride Topical Solution, manufacturer standard 30 mg / 30 ml (1 mg/ml) HEMOSTATIC AGENT For Topical Application Only DO NOT INJECT DATE
PRESCRIBING INFORMATION ADRENALIN TOPICAL SOLUTION Epinephrine Chloride Topical Solution, manufacturer standard 30 mg / 30 ml (1 mg/ml) HEMOSTATIC AGENT For Topical Application Only DO NOT INJECT DATE
PACKAGE INSERT USP ANTIBIOTIC
 Pr AMPICILLIN for Injection USP ANTIBIOTIC ACTIONS AND CLINICAL PHARMACOLOGY Ampicillin has a broad spectrum of bactericidal activity against many gram-positive and gramnegative aerobic and anaerobic bacteria.
Pr AMPICILLIN for Injection USP ANTIBIOTIC ACTIONS AND CLINICAL PHARMACOLOGY Ampicillin has a broad spectrum of bactericidal activity against many gram-positive and gramnegative aerobic and anaerobic bacteria.
Glucophage XR is contra-indicated during breast-feeding.
 Name GLUCOPHAGE XR 1000 mg Prolonged release tablets Active ingredient Metformin hydrochloride Composition Each Glucophage XR 1000 mg prolonged release tablet contains as active ingredient 1000 mg metformin
Name GLUCOPHAGE XR 1000 mg Prolonged release tablets Active ingredient Metformin hydrochloride Composition Each Glucophage XR 1000 mg prolonged release tablet contains as active ingredient 1000 mg metformin
Fundamentals of Pharmacology for Veterinary Technicians Chapter 8
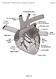 Figure 8-1 Figure 8-2 Figure 8-3 Figure 8-4 Figure 8-5 Figure 8-7 Figure 8-8 Figure 8-9 TABLE 8-1 Blood Flow Through the Heart The right atrium receives blood from all tissues, except the lungs, through
Figure 8-1 Figure 8-2 Figure 8-3 Figure 8-4 Figure 8-5 Figure 8-7 Figure 8-8 Figure 8-9 TABLE 8-1 Blood Flow Through the Heart The right atrium receives blood from all tissues, except the lungs, through
Package Leaflet: Information for the User. Lanoxin mg Tablets digoxin
 Package Leaflet: Information for the User Lanoxin 0.125 mg Tablets digoxin Read all of this leaflet carefully before you start using this medicine because it contains important information for you. - Keep
Package Leaflet: Information for the User Lanoxin 0.125 mg Tablets digoxin Read all of this leaflet carefully before you start using this medicine because it contains important information for you. - Keep
Antiarrhythmic Drugs. Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2018
 Antiarrhythmic Drugs Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2018 2 Ion Permeability Changes Potential Changes Genes and Proteins 3 Cardiac Na+ channels 5 6
Antiarrhythmic Drugs Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2018 2 Ion Permeability Changes Potential Changes Genes and Proteins 3 Cardiac Na+ channels 5 6
Radioimmunoassay of serum digoxin in relation to digoxin intoxication
 British Heart journal, I975, 37, 6I9-623. Radioimmunoassay of serum digoxin in relation to digoxin intoxication R. B. Singh, A. N. Rai, K. P. Dube, D. K. Srivastav, P. N. Somani, and B. C. Katiyar From
British Heart journal, I975, 37, 6I9-623. Radioimmunoassay of serum digoxin in relation to digoxin intoxication R. B. Singh, A. N. Rai, K. P. Dube, D. K. Srivastav, P. N. Somani, and B. C. Katiyar From
Elements for a Public Summary. Overview of disease epidemiology
 VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Gout i Gout has a worldwide distribution. In the United Kingdom from 2000 to 2007, the estimated occurrence of gout is 5.9% in
VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Gout i Gout has a worldwide distribution. In the United Kingdom from 2000 to 2007, the estimated occurrence of gout is 5.9% in
Amiodarone Prescribing and Monitoring: Back to the Future
 Amiodarone Prescribing and Monitoring: Back to the Future Subha L. Varahan, MD, FHRS, CCDS Electrophysiologist Oklahoma Heart Hospital Oklahoma City, OK Friday, February, 8 th, 2019 Iodinated benzofuran
Amiodarone Prescribing and Monitoring: Back to the Future Subha L. Varahan, MD, FHRS, CCDS Electrophysiologist Oklahoma Heart Hospital Oklahoma City, OK Friday, February, 8 th, 2019 Iodinated benzofuran
Alaska Nurse Practitioner Annual Conference 2009
 Alaska Nurse Practitioner Annual Conference 2009 LAURIE RACENET, RN, MSN, ANP, CEPS, CCDS Alaska Heart Institute Member: Boston Scientific Allied Professional Advisory Board Participant in Industry Sponsored
Alaska Nurse Practitioner Annual Conference 2009 LAURIE RACENET, RN, MSN, ANP, CEPS, CCDS Alaska Heart Institute Member: Boston Scientific Allied Professional Advisory Board Participant in Industry Sponsored
EFFECTIVE SHARED CARE AGREEMENT. Dronedarone - For the treatment and management of atrial fibrillation
 EFFECTIVE SHARED CARE AGREEMENT Dronedarone - For the treatment and management of atrial fibrillation Specialist details Patient identifier Name INTRODUCTION This shared care agreement outlines how the
EFFECTIVE SHARED CARE AGREEMENT Dronedarone - For the treatment and management of atrial fibrillation Specialist details Patient identifier Name INTRODUCTION This shared care agreement outlines how the
PATIENT INFORMATION LEAFLET CARVETREND RANGE
 SCHEDULING STATUS: S3 PATIENT INFORMATION LEAFLET PROPRIETARY NAME, STRENGTH AND PHARMACEUTICAL FORM: CARVETREND 6,25 mg tablet. CARVETREND 12,5 mg tablet. CARVETREND 25 mg tablet. Please read this leaflet
SCHEDULING STATUS: S3 PATIENT INFORMATION LEAFLET PROPRIETARY NAME, STRENGTH AND PHARMACEUTICAL FORM: CARVETREND 6,25 mg tablet. CARVETREND 12,5 mg tablet. CARVETREND 25 mg tablet. Please read this leaflet
Flecainide BNM. Please read this leaflet carefully before you start taking Flecainide BNM.
 1 Consumer Medicine Information Flecainide acetate Tablets 50 mg & 100 mg What is in this leaflet Please read this leaflet carefully before you start taking. This leaflet answers some common questions
1 Consumer Medicine Information Flecainide acetate Tablets 50 mg & 100 mg What is in this leaflet Please read this leaflet carefully before you start taking. This leaflet answers some common questions
Composition: Each Tablet contains. Pharmacokinetic properties:
 Composition: Each Tablet contains Torsemide 5/10/20/40/100mg Pharmacokinetic properties: Torsemide is well absorbed from the gastrointestinal tract. Peak serum concentrations are achieved within 1 hour
Composition: Each Tablet contains Torsemide 5/10/20/40/100mg Pharmacokinetic properties: Torsemide is well absorbed from the gastrointestinal tract. Peak serum concentrations are achieved within 1 hour
Patient Information VERSACLOZ (VER sa kloz) (clozapine) Oral Suspension
 Patient Information VERSACLOZ (VER sa kloz) (clozapine) Oral Suspension Read this Patient Information before you start taking VERSACLOZ and each time you get a refill. There may be new information. This
Patient Information VERSACLOZ (VER sa kloz) (clozapine) Oral Suspension Read this Patient Information before you start taking VERSACLOZ and each time you get a refill. There may be new information. This
INSPRA 25 & 50 mg TABLETS
 INSPRA 25 & 50 mg TABLETS SCHEDULING STATUS: Schedule 4 PROPRIETARY NAMES (and dosage forms): INSPRA 25 (Tablets) INSPRA 50 (Tablets) COMPOSITION: INSPRA 25: INSPRA 50: Each tablet contains 25 mg eplerenone
INSPRA 25 & 50 mg TABLETS SCHEDULING STATUS: Schedule 4 PROPRIETARY NAMES (and dosage forms): INSPRA 25 (Tablets) INSPRA 50 (Tablets) COMPOSITION: INSPRA 25: INSPRA 50: Each tablet contains 25 mg eplerenone
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT TRANSISOFT 8.5 g powder for oral solution in sachet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains 8.5 g of macrogol
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT TRANSISOFT 8.5 g powder for oral solution in sachet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains 8.5 g of macrogol
Drugs Controlling Myocyte Excitability and Conduction at the AV node Singh and Vaughan-Williams Classification
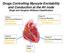 Drugs Controlling Myocyte Excitability and Conduction at the AV node Singh and Vaughan-Williams Classification Class I Na Channel Blockers Flecainide Propafenone Class III K channel Blockers Dofetilide,
Drugs Controlling Myocyte Excitability and Conduction at the AV node Singh and Vaughan-Williams Classification Class I Na Channel Blockers Flecainide Propafenone Class III K channel Blockers Dofetilide,
Sodium Bicarbonate Injection, USP
 Sodium Bicarbonate Injection, USP FOR THE CORRECTION OF METABOLIC ACIDOSIS AND OTHER CONDITIONS REQUIRING SYSTEMIC ALKALINIZATION. Plastic Ansyr II Syringe R x only DESCRIPTION Sodium Bicarbonate Injection,
Sodium Bicarbonate Injection, USP FOR THE CORRECTION OF METABOLIC ACIDOSIS AND OTHER CONDITIONS REQUIRING SYSTEMIC ALKALINIZATION. Plastic Ansyr II Syringe R x only DESCRIPTION Sodium Bicarbonate Injection,
Amlodipine plus Lisinopril Tablets AMLOPRES-L
 Amlodipine plus Lisinopril Tablets AMLOPRES-L COMPOSITION AMLOPRES-L Each uncoated tablet contains: Amlodipine besylate equivalent to Amlodipine 5 mg and Lisinopril USP equivalent to Lisinopril (anhydrous)
Amlodipine plus Lisinopril Tablets AMLOPRES-L COMPOSITION AMLOPRES-L Each uncoated tablet contains: Amlodipine besylate equivalent to Amlodipine 5 mg and Lisinopril USP equivalent to Lisinopril (anhydrous)
Cardiovascular Disorders. Heart Disorders. Diagnostic Tests for CV Function. Bio 375. Pathophysiology
 Cardiovascular Disorders Bio 375 Pathophysiology Heart Disorders Heart disease is ranked as a major cause of death in the U.S. Common heart diseases include: Congenital heart defects Hypertensive heart
Cardiovascular Disorders Bio 375 Pathophysiology Heart Disorders Heart disease is ranked as a major cause of death in the U.S. Common heart diseases include: Congenital heart defects Hypertensive heart
Amiodarone. Traffic light classification- Amber 2 Information sheet for Primary Care Prescribers
 Amiodarone Traffic light classification- Amber 2 Information sheet for Primary Care Prescribers Licensed Indications Tachyarrhythmias associated with Wolff-Parkinson-White syndrome All types of tachyarrhythmias
Amiodarone Traffic light classification- Amber 2 Information sheet for Primary Care Prescribers Licensed Indications Tachyarrhythmias associated with Wolff-Parkinson-White syndrome All types of tachyarrhythmias
Antiarrhythmic Drugs. Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2017
 Antiarrhythmic Drugs Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2017 Types of Cardiac Arrhythmias Abnormalities of Impulse Formation: Rate disturbances. Triggered
Antiarrhythmic Drugs Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2017 Types of Cardiac Arrhythmias Abnormalities of Impulse Formation: Rate disturbances. Triggered
Package leaflet: Information for the user. AmBisome 50 mg Powder for solution for infusion Liposomal amphotericin B
 Package leaflet: Information for the user AmBisome 50 mg Powder for solution for infusion Liposomal amphotericin B Read all of this leaflet carefully before you are given this medicine because it contains
Package leaflet: Information for the user AmBisome 50 mg Powder for solution for infusion Liposomal amphotericin B Read all of this leaflet carefully before you are given this medicine because it contains
Adenosine. poison/drug induced. flushing, chest pain, transient asystole. Precautions: tachycardia. fibrillation, atrial flutter. Indications: or VT
 Adenosine Indications: 1. Narrow complex PSVT 2. Does not convert atrial fibrillation, atrial flutter or VT 1. Side effects include flushing, chest pain, transient asystole 2. May deteriorate widecomplex
Adenosine Indications: 1. Narrow complex PSVT 2. Does not convert atrial fibrillation, atrial flutter or VT 1. Side effects include flushing, chest pain, transient asystole 2. May deteriorate widecomplex
Package Leaflet: Information for the User. Lanoxin Injection. digoxin
 Package Leaflet: Information for the User Lanoxin Injection digoxin Read all of this leaflet carefully before you start using this medicine because it contains important information for you. - Keep this
Package Leaflet: Information for the User Lanoxin Injection digoxin Read all of this leaflet carefully before you start using this medicine because it contains important information for you. - Keep this
Draft Labeling Package Insert Not Actual Size. BRAINTREE LABORATORIES, INC. PhosLo Capsules (Calcium Acetate)
 Draft Labeling Package Insert Not Actual Size BRAINTREE LABORATORIES, INC. PhosLo Capsules (Calcium Acetate) DESCRIPTION: Full Size: Each opaque capsule with a white cap and white body is spin printed
Draft Labeling Package Insert Not Actual Size BRAINTREE LABORATORIES, INC. PhosLo Capsules (Calcium Acetate) DESCRIPTION: Full Size: Each opaque capsule with a white cap and white body is spin printed
Pediatrics. Arrhythmias in Children: Bradycardia and Tachycardia Diagnosis and Treatment. Overview
 Pediatrics Arrhythmias in Children: Bradycardia and Tachycardia Diagnosis and Treatment See online here The most common form of cardiac arrhythmia in children is sinus tachycardia which can be caused by
Pediatrics Arrhythmias in Children: Bradycardia and Tachycardia Diagnosis and Treatment See online here The most common form of cardiac arrhythmia in children is sinus tachycardia which can be caused by
