Composition: Each Tablet contains. Pharmacokinetic properties:
|
|
|
- Oliver Hawkins
- 5 years ago
- Views:
Transcription
1 Composition: Each Tablet contains Torsemide 5/10/20/40/100mg Pharmacokinetic properties: Torsemide is well absorbed from the gastrointestinal tract. Peak serum concentrations are achieved within 1 hour of oral doses. Torsemide is metabolised by the cytochrome P450 isoenzyme CYP2C9, which shows genetic polymorphism. Metabolism takes place in the liver and inactive metabolites are excreted in the urine. The elimination half-life of Torsemide is about 3.5 hours. Torsemide is extensively bound to plasma proteins. In patients with heart failure both hepatic and renal clearance are reduced. In patients with renal impairment, the renal clearance is reduced but total plasma clearance is not significantly altered. Mechanism of Action Torasemide inhibits the Na + /K + /2Cl - -carrier system (via interference of the chloride binding site) in the lumen of the thick ascending portion of the loop of Henle, resulting in a decrease in reabsorption of sodium and chloride. This results in an increase in the rate of delivery of tubular fluid and electrolytes to the distal sites of hydrogen and potassium ion secretion, while plasma volume contraction increases aldosterone production. The increased delivery and high aldosterone levels promote sodium reabsorption at the distal tubules, and By increasing the delivery of sodium to the distal renal tubule, torasemide indirectly increases potassium excretion via the sodium-potassium exchange mechanism. Torasemide's effects in other segments of the nephron have not been demonstrated. Thus torasemide increases the urinary excretion of sodium, chloride, and water, but it does not significantly alter glomerular filtration rate, renal plasma flow, or acid-base balance. Other pharmacodynamic properties: Torasemide (INN) or torsemide (USAN) is a novel loop diuretic belonging to pyridine sulphonyl urea. It differs from other thiazide diuretics in that a double ring system is incorporated into its structure. Like thiazides, loop diuretics must be secreted into the tubular fluid by proximal tubule cells. In the thick ascending loop Na+ and Cl- reabsorption is accomplished by a Na + /K + /2Cl - symporter. The thick ascending limb has a high reabsorptive
2 capacity and is responsible for reabsorbing 25% of the filtered load of Na +. The loop diuretics act by blocking this symporter. Because of the large absorptive capacity and the amount of Na + delivered to the ascending limb, loop diuretics have a profound diuretic action. In addition, more distal nephron segments do not have the reabsorptive capacity to compensate for this increased load. The osmotic gradient for water reabsorption is also reduced resulting in an increase in the amount of water excreted. Torasemide's effects as a antihypertensive are due to its diuretic actions. By reducing extracellular and plasma fluid volume, blood pressure is reduced temporarily, and cardiac output also decreases. Indication: Meltor is indicated for the treatment of edema associated with congestive heart failure, renal disease, or hepatic disease and CKD. Meltor is indicated for the treatment of hypertension alone or in combination with other antihypertensive agents Contraindication: The use of Meltor is contraindicated in patients with: Known hypersensitivity to torsemide or to sulfonylureas Anuria (production of <50 ml urine/day) Drug Interaction: Nonsteroidal Anti-inflammatory Drugs Because torsemide and salicylates compete for secretion by renal tubules, patients receiving high doses of salicylates may experience salicylate toxicity when torsemide is concomitantly administered. Concomitant use of nonsteroidal anti-inflammatory drugs (NSAIDs) and torsemide has been associated with the development of acute renal failure. The antihypertensive and diuretic effects of torsemide can be reduced by NSAIDs. Partial inhibition of the natriuretic effect of torsemide by concomitant administration of indomethacin has been demonstrated for torsemide under conditions of dietary sodium restriction (50 meq/day) but not in the presence of normal sodium intake (150 meq/day). Cytochrome P450 2C9 Inhibitors and Inducers Torsemide is a substrate of CYP2C9. Concomitant use of CYP2C9 inhibitors (e.g., amiodarone, fluconazole, miconazole, oxandrolone) can decrease torsemide clearance and increase torsemide plasma concentrations. Concomitant use of CYP2C9 inducers (e.g., rifampin) increase torsemide clearance and decrease plasma torsemide concentrations. Monitor diuretic effect and blood pressure when used in combination with CYP2C9 inhibitor or inducer. Adjust torsemide dose if necessary.
3 Because of its inhibition of CYP2C9 metabolism, torsemide may affect the efficacy and safety of sensitive CYP2C9 substrates, such as celecoxib, or of substrates with a narrow therapeutic range, such as warfarin or phenytoin. Monitor patients and adjust dosages if necessary. Cholestyramine Concomitant use of torsemide and cholestyramine has not been studied in humans but, in a study in animals, coadministration of cholestyramine decreased the absorption of orally administered torsemide. If torsemide and cholestyramine should be coadministered, administer torsemide at least one hour before or 4 to 6 h after cholestyramine administration. Organic Anion Drugs Coadministration of organic anion drugs (e.g., probenecid) that undergo significant renal tubular secretion have the potential to reduce secretion of torsemide into the proximal tubule and thereby decreases the diuretic activity of torsemide. Monitor diuretic effect and blood pressure during coadministration. Lithium Like other diuretics, torsemide reduces the renal clearance of lithium, inducing a high risk of lithium toxicity. Monitor lithium levels periodically when torsemide is coadministered. Ototoxic Drugs Loop diuretics increase the ototoxic potential of other ototoxic drugs, including aminoglycoside antibiotics and ethacrynic acid. This effect has been reported with concomitant use of torsemide and gentamycin. Avoid concomitant use of torsemide and aminoglycoside antibiotics, if possible. Renin-angiotensin Inhibitors Coadministration of torsemide with ACE inhibitors or angiotensin receptor blockers can increase the risk of hypotension and renal impairment. Radiocontrast Agents Torsemide can increase the risk of renal toxicity related to administration of radiocontrast agents. Corticosteroids and ACTH Concomitant use with torsemide may increase risk of hypokalemia. Adverse effects: Dizziness, headache, nausea, weakness, vomiting, hyperglycemia, excessive urination, hyperuricemia, hypokalemia, excessive thirst, hypovolemia, impotence, esophageal hemorrhage, pancreatitis, leucopenia, thrombocytopenia and dyspepsia. Overdosage
4 There is no human experience with overdoses of MELTOR, but the signs and symptoms of overdosage can be anticipated to be those of excessive pharmacologic effect: dehydration, hypovolemia, hypotension, hyponatremia, hypokalemia, hypochloremic alkalosis, and hemoconcentration. Treatment of overdosage should consist of fluid and electrolyte replacement. Warnings and Precautions: Hepatic Disease With Cirrhosis and Ascites MELTOR should be used with caution in patients with hepatic disease with cirrhosis and ascites, since sudden alterations of fluid and electrolyte balance may precipitate hepatic coma. In these patients, diuresis with MELTOR (or any other diuretic) is best initiated in the hospital. To prevent hypokalemia and metabolic alkalosis, an aldosterone antagonist or potassium-sparing drug should be used concomitantly with MELTOR. Ototoxicity Tinnitus and hearing loss (usually reversible) have been observed after rapid intravenous injection of other loop diuretics and have also been observed after oral MELTOR. It is not certain that these events were attributable to MELTOR. Ototoxicity has also been seen in animal studies when very high plasma levels of torsemide were induced. Volume and Electrolyte Depletion Patients receiving diuretics should be observed for clinical evidence of electrolyte imbalance, hypovolemia, or prerenal azotemia. Symptoms of these disturbances may include one or more of the following: dryness of the mouth, thirst, weakness, lethargy, drowsiness, restlessness, muscle pains or cramps, muscular fatigue, hypotension, oliguria, tachycardia, nausea, and vomiting. Excessive diuresis may cause dehydration, bloodvolume reduction, and possibly thrombosis and embolism, especially in elderly patients. In patients who develop fluid and electrolyte imbalances, hypovolemia, or prerenal azotemia, the observed laboratory changes may include hyper- or hyponatremia, hyperor hypochloremia, hyper- or hypokalemia, acid-base abnormalities, and increased blood urea nitrogen (BUN). If any of these occur, MELTOR should be discontinued until the situation is corrected; MELTOR may be restarted at a lower dose. In controlled studies in the United States, MELTOR was administered to hypertensive patients at doses of 5 mg or 10 mg daily. After 6 weeks at these doses, the mean decrease in serum potassium was approximately 0.1 meq/l. The percentage of patients who had a serum potassium level below 3.5 meq/l at any time during the studies was essentially the same in patients who received MELTOR (1.5%) as in those who received placebo (3%). In patients followed for 1 year, there was no further change in mean serum potassium levels. In patients with congestive heart failure, hepatic cirrhosis, or renal disease treated with MELTOR at doses higher than those studied in United
5 States antihypertensive trials, hypokalemia was observed with greater frequency, in a doserelated manner. In patients with cardiovascular disease, especially those receiving digitalis glycosides, diuretic-induced hypokalemia may be a risk factor for the development of arrhythmias. The risk of hypokalemia is greatest in patients with cirrhosis of the liver, in patients experiencing a brisk diuresis, in patients who are receiving inadequate oral intake of electrolytes, and in patients receiving concomitant therapy with corticosteroids or ACTH. Periodic monitoring of serum potassium and other electrolytes is advised in patients treated with MELTOR. Laboratory Values Potassium: See WARNINGS. Calcium: Single doses of MELTOR increased the urinary excretion of calcium by normal subjects, but serum calcium levels were slightly increased in 4- to 6-week hypertension trials. In a long-term study of patients with congestive heart failure, the average 1-year change in serum calcium was a decrease of 0.10 mg/dl (0.02 mmol/l). Among 426 patients treated with MELTOR for an average of 11 months, hypocalcemia was not reported as an adverse event. Magnesium: Single doses of MELTOR caused healthy volunteers to increase their urinary excretion of magnesium, but serum magnesium levels were slightly increased in 4- to 6- week hypertension trials. In long-term hypertension studies, the average 1-year change in serum magnesium was an increase of 0.03 mg/dl (0.01 mmol/l). Among 426 patients treated with MELTOR for an average of 11 months, one case of hypomagnesemia (1.3 mg/dl [0.53 mmol/l]) was reported as an adverse event. In a long-term clinical study of MELTOR in patients with congestive heart failure, the estimated annual change in serum magnesium was an increase of 0.2 mg/dl (0.08 mmol/l), but these data are confounded by the fact that many of these patients received magnesium supplements. In a 4-week study in which magnesium supplementation was not given, the rate of occurrence of serum magnesium levels below 1.7 mg/dl (0.70 mmol/l) was 6% and 9% in the groups receiving 5 mg and 10 mg of MELTOR, respectively. Blood Urea Nitrogen (BUN), Creatinine and Uric Acid: MELTOR produces small dose-related increases in each of these laboratory values. In hypertensive patients who received 10 mg of MELTOR daily for 6 weeks, the mean increase in blood urea nitrogen was 1.8 mg/dl (0.6 mmol/l), the mean increase in serum creatinine was 0.05 mg/dl (4 mmol/l), and the mean increase in serum uric acid was 1.2 mg/dl (70 mmol/l). Little further change occurred with long-term treatment, and all changes reversed when treatment was discontinued. Symptomatic gout has been reported in patients receiving MELTOR, but its incidence has been similar to that seen in patients receiving placebo.
6 Glucose: Hypertensive patients who received 10 mg of daily MELTOR experienced a mean increase in serum glucose concentration of 5.5 mg/dl (0.3 mmol/l) after 6 weeks of therapy, with a further increase of 1.8 mg/dl (0.1 mmol/l) during the subsequent year. In 205 long-term studies in diabetics, mean fasting glucose values were not significantly changed from baseline. Cases of hyperglycemia have been reported but are uncommon. Serum Lipids: In the controlled short-term hypertension studies in the United States, daily doses of 5 mg, 10 mg, and 20 mg of MELTOR were associated with increases in total plasma cholesterol of 4, 4, and 8 mg/dl (0.10 to 0.20 mmol/l), respectively. The changes subsided during chronic therapy. In the same short-term hypertension studies, daily doses of 5 mg, 10 mg and 20 mg of MELTOR were associated with mean increases in plasma triglycerides of 16, 13 and 71 mg/dl (0.15 to 0.80 mmol/l), respectively. In long-term studies of 5 mg to 20 mg of MELTOR daily, no clinically significant differences from baseline lipid values were observed after 1 year of therapy. Other: In long-term studies in hypertensive patients, MELTOR has been associated with small mean decreases in hemoglobin, hematocrit, and erythrocyte count and small mean increases in white blood cell count, platelet count, and serum alkaline phosphatase. Although statistically significant, all of these changes were medically inconsequential. No significant trends have been observed in any liver enzyme tests other than alkaline phosphatase. Use in special population: 1. Pediatric: Safety and effectiveness in pediatric patients have not been established. 2. Geriatric: Appropriate studies performed to date have not demonstrated geriatricspecific problems that would limit the usefulness of torsemide in the elderly. However, elderly patients are more likely to have age-related liver, kidney, or heart problems, which may require caution and an adjustment in the dose for elderly patients 3. Liver impairment: Meltor should be used with caution in patients with hepatic disease 4. Renal failure: Use with caution. 5. Pregnancy and lactation: Pregnancy Category B. Meltor should be used during pregnancy only if clearly needed. Caution should be exercised when MELTOR is administered to a nursing woman Dosage: As directed by physician. Presentation: Meltor is available in a pack of 10 tablets.
7 Storage and handling: Store below 30⁰C in a dry place. Protect from light.
MELTOR (Mouth Melting Torsemide) 5mg, 10mg, 20mg, 40mg and 100mg Tablets
 MELTOR (Mouth Melting Torsemide) 5mg, 10mg, 20mg, 40mg and 100mg Tablets DESCRIPTION MELTOR (torsemide) is a diuretic of the pyridine-sulfonylurea class. Its chemical name is 1-isopropyl-3-[(4-m-toluidino-3-pyridyl)
MELTOR (Mouth Melting Torsemide) 5mg, 10mg, 20mg, 40mg and 100mg Tablets DESCRIPTION MELTOR (torsemide) is a diuretic of the pyridine-sulfonylurea class. Its chemical name is 1-isopropyl-3-[(4-m-toluidino-3-pyridyl)
DIURETICS. Assoc. Prof. Bilgen Başgut
 DIURETICS Assoc. Prof. Bilgen Başgut Classification of Diuretics The best way to classify diuretics is to look for their Site of action in the nephron A. Diuretics that inhibit transport in the Proximal
DIURETICS Assoc. Prof. Bilgen Başgut Classification of Diuretics The best way to classify diuretics is to look for their Site of action in the nephron A. Diuretics that inhibit transport in the Proximal
DYTOR PLUS Tablets (Torsemide + Spironolactone)
 Published on: 10 Jul 2014 DYTOR PLUS Tablets (Torsemide + Spironolactone) Black Box Warning Spironolactone has been shown to be a tumorigen in chronic toxicity studies in rats. Spironolactone should be
Published on: 10 Jul 2014 DYTOR PLUS Tablets (Torsemide + Spironolactone) Black Box Warning Spironolactone has been shown to be a tumorigen in chronic toxicity studies in rats. Spironolactone should be
BURINEX PRODUCT INFORMATION NAME OF THE MEDICINE DESCRIPTION PHARMACOLOGY. Pharmacodynamics. (bumetanide)
 NAME OF THE MEDICINE BURINEX PRODUCT INFORMATION (bumetanide) Burinex 1 mg tablets contain bumetanide i.e. 3-n-(butylamino)-4-phenoxy-5-sulphamoyl-benzoic acid. The structural formula of bumetanide is:
NAME OF THE MEDICINE BURINEX PRODUCT INFORMATION (bumetanide) Burinex 1 mg tablets contain bumetanide i.e. 3-n-(butylamino)-4-phenoxy-5-sulphamoyl-benzoic acid. The structural formula of bumetanide is:
PRINCIPLES OF DIURETIC ACTIONS:
 DIURETIC: A drug that increases excretion of solutes Increased urine volume is secondary All clinically useful diuretics act by blocking Na + reabsorption Has the highest EC to IC ratio = always more sodium
DIURETIC: A drug that increases excretion of solutes Increased urine volume is secondary All clinically useful diuretics act by blocking Na + reabsorption Has the highest EC to IC ratio = always more sodium
Pharmacology I [PHL 313] Diuretics. Dr. Mohammad Nazam Ansari
![Pharmacology I [PHL 313] Diuretics. Dr. Mohammad Nazam Ansari Pharmacology I [PHL 313] Diuretics. Dr. Mohammad Nazam Ansari](/thumbs/90/104323364.jpg) Pharmacology I [PHL 313] Diuretics Dr. Mohammad Nazam Ansari Renal Pharmacology Kidneys: Each adult kidney weighs 125-170g in males and 115-155g in females, represent 0.5% of total body weight, but receive
Pharmacology I [PHL 313] Diuretics Dr. Mohammad Nazam Ansari Renal Pharmacology Kidneys: Each adult kidney weighs 125-170g in males and 115-155g in females, represent 0.5% of total body weight, but receive
Chapter 21. Diuretic Agents. Mosby items and derived items 2008, 2002 by Mosby, Inc., an affiliate of Elsevier Inc.
 Chapter 21 Diuretic Agents Renal Structure and Function Kidneys at level of umbilicus Each weighs 160 to 175 g and is 10 to 12 cm long Most blood flow per gram of weight in body 22% of cardiac output (CO)
Chapter 21 Diuretic Agents Renal Structure and Function Kidneys at level of umbilicus Each weighs 160 to 175 g and is 10 to 12 cm long Most blood flow per gram of weight in body 22% of cardiac output (CO)
Composition: Each tablet contain. Pharmacokinetic properties:
 Composition: Each tablet contain Metolazone 2.5/5mg Pharmacokinetic properties: Metolazone is slowly and incompletely absorbed from the gastrointestinal tract. An average of about 65% of a dose has been
Composition: Each tablet contain Metolazone 2.5/5mg Pharmacokinetic properties: Metolazone is slowly and incompletely absorbed from the gastrointestinal tract. An average of about 65% of a dose has been
Spironolactone has not been demonstrated to elevate serum uric acid, to precipitate gout or to alter carbohydrate metabolism.
 SPIRONE Composition Each tablet contains Spironolactone 100 mg. Tablets Action Spironolactone is a specific pharmacologic antagonist of aldosterone, acting primarily through competitive binding of receptors
SPIRONE Composition Each tablet contains Spironolactone 100 mg. Tablets Action Spironolactone is a specific pharmacologic antagonist of aldosterone, acting primarily through competitive binding of receptors
PACKAGE LEAFLET: Information for the patient FUROSEMID Tablets - 40 mg Solution for injection - 20 mg / 2 ml (Furosemide)
 PACKAGE LEAFLET: Information for the patient FUROSEMID Tablets - 40 mg Solution for injection - 20 mg / 2 ml (Furosemide) Read this leaflet carefully before you start taking this medicine. - Keep this
PACKAGE LEAFLET: Information for the patient FUROSEMID Tablets - 40 mg Solution for injection - 20 mg / 2 ml (Furosemide) Read this leaflet carefully before you start taking this medicine. - Keep this
PRODUCT INFORMATION INSIG
 PRODUCT INFORMATION INSIG NAME OF THE MEDICINE INSIG (Indapamide) DESCRIPTION Chemical name: Indapamide hemihydrate in 4-chloro-N(2-methyl-1-indolinyl)-3-sulfamoyl benzamide hemihydrate. Indapamide is
PRODUCT INFORMATION INSIG NAME OF THE MEDICINE INSIG (Indapamide) DESCRIPTION Chemical name: Indapamide hemihydrate in 4-chloro-N(2-methyl-1-indolinyl)-3-sulfamoyl benzamide hemihydrate. Indapamide is
Diuretic Agents Part-1. Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia
 Diuretic Agents Part-1 Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Kidneys eliminates waste products and regulates the volume, electrolyte and ph of
Diuretic Agents Part-1 Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Kidneys eliminates waste products and regulates the volume, electrolyte and ph of
PHARMACEUTICAL INFORMATION AZILSARTAN
 AZEARLY Tablets Each Tablet Contains Azilsartan 20/40/80 mg PHARMACEUTICAL INFORMATION AZILSARTAN Generic name: Azilsartan Chemical name: 2-Ethoxy-1-{[2'-(5-oxo-2,5-dihydro-1,2,4-oxadiazol-3-yl)-4-biphenylyl]methyl}-
AZEARLY Tablets Each Tablet Contains Azilsartan 20/40/80 mg PHARMACEUTICAL INFORMATION AZILSARTAN Generic name: Azilsartan Chemical name: 2-Ethoxy-1-{[2'-(5-oxo-2,5-dihydro-1,2,4-oxadiazol-3-yl)-4-biphenylyl]methyl}-
Renal Pharmacology. Diuretics: Carbonic Anhydrase Inhibitors Thiazides Loop Diuretics Potassium-sparing Diuretics BIMM118
 Diuretics: Carbonic Anhydrase Inhibitors Thiazides Loop Diuretics Potassium-sparing Diuretics Renal Pharmacology Kidneys: Represent 0.5% of total body weight, but receive ~25% of the total arterial blood
Diuretics: Carbonic Anhydrase Inhibitors Thiazides Loop Diuretics Potassium-sparing Diuretics Renal Pharmacology Kidneys: Represent 0.5% of total body weight, but receive ~25% of the total arterial blood
Furosemide: Properties, Alternatives, and the Medication Approval Process. Heather Brown EMS 209-Advanced Pharmacology Don Knox
 Furosemide: Properties, Alternatives, and the Medication Approval Process Heather Brown EMS 209-Advanced Pharmacology Don Knox Pre-hospital treatment of critical patients is a key factor in determining
Furosemide: Properties, Alternatives, and the Medication Approval Process Heather Brown EMS 209-Advanced Pharmacology Don Knox Pre-hospital treatment of critical patients is a key factor in determining
PRESCRIBING INFORMATION CHLORTHALIDONE. Chlortalidone Tablets BP. 50 mg. Diuretic - Antihypertensive
 0 PRESCRIBING INFORMATION CHLORTHALIDONE Chlortalidone Tablets BP 50 mg Diuretic - Antihypertensive AA PHARMA INC. DATE OF PREPARATION: 1165 Creditstone Road, Unit # 1 June 24, 2010 Vaughan, Ontario L4K
0 PRESCRIBING INFORMATION CHLORTHALIDONE Chlortalidone Tablets BP 50 mg Diuretic - Antihypertensive AA PHARMA INC. DATE OF PREPARATION: 1165 Creditstone Road, Unit # 1 June 24, 2010 Vaughan, Ontario L4K
LOSARTAN + HYDROCHLOROTHIAZIDE HYZAAR /HYZAAR DS
 PRODUCT CIRCULAR WPC-HYZ-T-092007 THERAPEUTIC CLASS LOSARTAN + HYDROCHLOROTHIAZIDE HYZAAR /HYZAAR DS Film-Coated Tablet Antihypertensive LOSARTAN + HYDROCHLOROTHIAZIDE (HYZAAR) is the first combination
PRODUCT CIRCULAR WPC-HYZ-T-092007 THERAPEUTIC CLASS LOSARTAN + HYDROCHLOROTHIAZIDE HYZAAR /HYZAAR DS Film-Coated Tablet Antihypertensive LOSARTAN + HYDROCHLOROTHIAZIDE (HYZAAR) is the first combination
azilsartan medoxomil
 azilsartan medoxomil edarbi 40mg Tablet 80mg Tablet ANTIHYPERTENSIVE Angiotensin II Receptor Antagonist FORMULATION: Each tablet contains 40mg Azilsartan medoxomil (as potassium) Each tablet contains 80mg
azilsartan medoxomil edarbi 40mg Tablet 80mg Tablet ANTIHYPERTENSIVE Angiotensin II Receptor Antagonist FORMULATION: Each tablet contains 40mg Azilsartan medoxomil (as potassium) Each tablet contains 80mg
Diuretic Use in Neonates
 Neonatal Nursing Education Brief: Diuretic Use in the Neonate http://www.seattlechildrens.org/healthcareprofessionals/education/continuing-medical-nursing-education/neonatalnursing-education-briefs/ Diuretics
Neonatal Nursing Education Brief: Diuretic Use in the Neonate http://www.seattlechildrens.org/healthcareprofessionals/education/continuing-medical-nursing-education/neonatalnursing-education-briefs/ Diuretics
Amlodipine plus Lisinopril Tablets AMLOPRES-L
 Amlodipine plus Lisinopril Tablets AMLOPRES-L COMPOSITION AMLOPRES-L Each uncoated tablet contains: Amlodipine besylate equivalent to Amlodipine 5 mg and Lisinopril USP equivalent to Lisinopril (anhydrous)
Amlodipine plus Lisinopril Tablets AMLOPRES-L COMPOSITION AMLOPRES-L Each uncoated tablet contains: Amlodipine besylate equivalent to Amlodipine 5 mg and Lisinopril USP equivalent to Lisinopril (anhydrous)
Therapeutics of Diuretics
 (Last Updated: 08/22/2018) Created by: Socco, Samantha Therapeutics of Diuretics Thambi, M. (2017). The Clinical Use of Diuretics. Lecture presented at PHAR 503 Lecture in UIC College of Pharmacy, Chicago.
(Last Updated: 08/22/2018) Created by: Socco, Samantha Therapeutics of Diuretics Thambi, M. (2017). The Clinical Use of Diuretics. Lecture presented at PHAR 503 Lecture in UIC College of Pharmacy, Chicago.
New Zealand Data Sheet. Arrow-Bendrofluazide Bendroflumethiazide (also known as Bendrofluazide) Tablets 2.5mg and 5mg
 New Zealand Data Sheet Arrow-Bendrofluazide Bendroflumethiazide (also known as Bendrofluazide) Tablets 2.5mg and 5mg Description Arrow-Bendrofluazide Tablets contain 2.5 and 5 mg of the active ingredient
New Zealand Data Sheet Arrow-Bendrofluazide Bendroflumethiazide (also known as Bendrofluazide) Tablets 2.5mg and 5mg Description Arrow-Bendrofluazide Tablets contain 2.5 and 5 mg of the active ingredient
LESSON ASSIGNMENT. Diuretic and Antidiuretic Agents. After you finish this lesson you should be able to:
 LESSON ASSIGNMENT SUBCOURSE MD0806 LESSON 8 Therapeutics III. Diuretic and Antidiuretic Agents. LESSON ASSIGNMENT Paragraphs 8-1--8-7. LESSON OBJECTIVES After you finish this lesson you should be able
LESSON ASSIGNMENT SUBCOURSE MD0806 LESSON 8 Therapeutics III. Diuretic and Antidiuretic Agents. LESSON ASSIGNMENT Paragraphs 8-1--8-7. LESSON OBJECTIVES After you finish this lesson you should be able
Diuretics (Saluretics)
 Diuretics (Saluretics) Diuretics increase urine excretion mainly by reabsorption of salts and water from kidney tubules These agents are ion transport inhibitors that decrease the reabsorption of Na+ at
Diuretics (Saluretics) Diuretics increase urine excretion mainly by reabsorption of salts and water from kidney tubules These agents are ion transport inhibitors that decrease the reabsorption of Na+ at
SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS PRODUCT SUMMARY 1. NAME OF THE MEDICINAL PRODUCT Sterile Potassium Chloride Concentrate 15%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15% of Potassium Chloride in
SUMMARY OF PRODUCT CHARACTERISTICS PRODUCT SUMMARY 1. NAME OF THE MEDICINAL PRODUCT Sterile Potassium Chloride Concentrate 15%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15% of Potassium Chloride in
Medical Translation: Welcome to our webinar
 r Welcome to our webinar Medical Translation: The Pharmacodynamics and Physiological Effects of Drug Classes Commonly Studied during Clinical Trials with Carmen Cross 13 February 2014 www.ecpdwebinars.co.uk
r Welcome to our webinar Medical Translation: The Pharmacodynamics and Physiological Effects of Drug Classes Commonly Studied during Clinical Trials with Carmen Cross 13 February 2014 www.ecpdwebinars.co.uk
COMPOSITION. A film coated tablet contains. Active ingredient: irbesartan 75 mg, 150 mg or 300 mg. Rotazar (Film coated tablets) Irbesartan
 Rotazar (Film coated tablets) Irbesartan Rotazar 75 mg, 150 mg, 300 mg COMPOSITION A film coated tablet contains Active ingredient: irbesartan 75 mg, 150 mg or 300 mg. Rotazar 75 mg, 150 mg, 300 mg PHARMACOLOGICAL
Rotazar (Film coated tablets) Irbesartan Rotazar 75 mg, 150 mg, 300 mg COMPOSITION A film coated tablet contains Active ingredient: irbesartan 75 mg, 150 mg or 300 mg. Rotazar 75 mg, 150 mg, 300 mg PHARMACOLOGICAL
Each tablet contains:
 Composition: Each tablet contains: Tolvaptan 15/30mg Pharmacokinetic properties: In healthy subjects the pharmacokinetics of tolvaptan after single doses of up to 480 mg and multiple doses up to 300 mg
Composition: Each tablet contains: Tolvaptan 15/30mg Pharmacokinetic properties: In healthy subjects the pharmacokinetics of tolvaptan after single doses of up to 480 mg and multiple doses up to 300 mg
Composition: Each tablet contain. Levocetirizine. Each 5ml contains. Montelukast. Pharmacokinetic properties:
 Composition: Each tablet contain Montelukast Levocetirizine 10mg 5mg Each 5ml contains Montelukast Levocetirizine 4mg 2.5mg Pharmacokinetic properties: Peak plasma concentrations of montelukast are achieved
Composition: Each tablet contain Montelukast Levocetirizine 10mg 5mg Each 5ml contains Montelukast Levocetirizine 4mg 2.5mg Pharmacokinetic properties: Peak plasma concentrations of montelukast are achieved
Diuretic Agents Part-2. Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia
 Diuretic Agents Part-2 Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Potassium-sparing diuretics The Ion transport pathways across the luminal and basolateral
Diuretic Agents Part-2 Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Potassium-sparing diuretics The Ion transport pathways across the luminal and basolateral
INSPRA 25 & 50 mg TABLETS
 INSPRA 25 & 50 mg TABLETS SCHEDULING STATUS: Schedule 4 PROPRIETARY NAMES (and dosage forms): INSPRA 25 (Tablets) INSPRA 50 (Tablets) COMPOSITION: INSPRA 25: INSPRA 50: Each tablet contains 25 mg eplerenone
INSPRA 25 & 50 mg TABLETS SCHEDULING STATUS: Schedule 4 PROPRIETARY NAMES (and dosage forms): INSPRA 25 (Tablets) INSPRA 50 (Tablets) COMPOSITION: INSPRA 25: INSPRA 50: Each tablet contains 25 mg eplerenone
Calcium (Ca 2+ ) mg/dl
 Quick Guide to Laboratory Values Use this handy cheat-sheet to help you monitor laboratory values related to fluid and electrolyte status. Remember, normal values may vary according to techniques used
Quick Guide to Laboratory Values Use this handy cheat-sheet to help you monitor laboratory values related to fluid and electrolyte status. Remember, normal values may vary according to techniques used
Diuretics having the quality of exciting excessive excretion of urine. OED. Inhibitors of Sodium Reabsorption Saluretics not Aquaretics
 Diuretics having the quality of exciting excessive excretion of urine. OED Inhibitors of Sodium Reabsorption Saluretics not Aquaretics 1 Sodium Absorption Na Entry into the Cell down an electrochemical
Diuretics having the quality of exciting excessive excretion of urine. OED Inhibitors of Sodium Reabsorption Saluretics not Aquaretics 1 Sodium Absorption Na Entry into the Cell down an electrochemical
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Colecalciferol Meda 800 IU tablet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains colecalciferol (vitamin D 3 ) 800 IU
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Colecalciferol Meda 800 IU tablet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains colecalciferol (vitamin D 3 ) 800 IU
Human Physiology - Problem Drill 17: The Kidneys and Nephronal Physiology
 Human Physiology - Problem Drill 17: The Kidneys and Nephronal Physiology Question No. 1 of 10 Instructions: (1) Read the problem statement and answer choices carefully, (2) Work the problems on paper
Human Physiology - Problem Drill 17: The Kidneys and Nephronal Physiology Question No. 1 of 10 Instructions: (1) Read the problem statement and answer choices carefully, (2) Work the problems on paper
DIURETICS-4 Dr. Shariq Syed
 DIURETICS-4 Dr. Shariq Syed AIKTC - Knowledge Resources & Relay Center 1 Pop Quiz!! Loop diuretics act on which transporter PKCC NKCC2 AIKTCC I Don t know AIKTC - Knowledge Resources & Relay Center 2 Pop
DIURETICS-4 Dr. Shariq Syed AIKTC - Knowledge Resources & Relay Center 1 Pop Quiz!! Loop diuretics act on which transporter PKCC NKCC2 AIKTCC I Don t know AIKTC - Knowledge Resources & Relay Center 2 Pop
Daiichi Sankyo s U.S. Products Liability Litigation Settlement Program Moves Forward; More than 97 Percent of Eligible Litigants and Claimants Opt In
 Daiichi Sankyo s U.S. Products Liability Litigation Settlement Program Moves Forward; More than 97 Percent of Eligible Litigants and Claimants Opt In Tokyo, Japan and Basking Ridge, NJ June 6, 2018 Daiichi
Daiichi Sankyo s U.S. Products Liability Litigation Settlement Program Moves Forward; More than 97 Percent of Eligible Litigants and Claimants Opt In Tokyo, Japan and Basking Ridge, NJ June 6, 2018 Daiichi
Chapter 19 The Urinary System Fluid and Electrolyte Balance
 Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Nursing Process Focus: Patients Receiving Dextran 40 (Gentran 40)
 Assess for presence/history of hypovolemia, shock, venous thrombosis. Assess vital signs: Hypovolemic shock secondary to surgery, burns, hemorrhage, other serious condition PT and PTT abnormalities Venous
Assess for presence/history of hypovolemia, shock, venous thrombosis. Assess vital signs: Hypovolemic shock secondary to surgery, burns, hemorrhage, other serious condition PT and PTT abnormalities Venous
DIAGNOSIS AND MANAGEMENT OF DIURETIC RESISTANCE. Jules B. Puschett, M.D.
 DIAGNOSIS AND MANAGEMENT OF DIURETIC RESISTANCE Jules B. Puschett, M.D. Diuretic Resistance A clinical circumstance in which patients do not respond to a combination of salt restriction and even large
DIAGNOSIS AND MANAGEMENT OF DIURETIC RESISTANCE Jules B. Puschett, M.D. Diuretic Resistance A clinical circumstance in which patients do not respond to a combination of salt restriction and even large
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION
 DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
WATER, SODIUM AND POTASSIUM
 WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
P-RMS: FR/H/PSUR/0056/001
 Core Safety Profile Active substance: Piretanide Pharmaceutical form(s)/strength: Tablets, 3mg, 6mg Capsules, 6mg Ampoules, 6mg, 12mg, 60mg P-RMS: FR/H/PSUR/0056/001 Date of FAR: 21.12.2009 4.2 Posology
Core Safety Profile Active substance: Piretanide Pharmaceutical form(s)/strength: Tablets, 3mg, 6mg Capsules, 6mg Ampoules, 6mg, 12mg, 60mg P-RMS: FR/H/PSUR/0056/001 Date of FAR: 21.12.2009 4.2 Posology
LACIPIL QUALITATIVE AND QUANTITATIVE COMPOSITION
 LACIPIL lacidipine QUALITATIVE AND QUANTITATIVE COMPOSITION Lacidipine, 2 mg - round shaped white engraved on one face. Lacidipine, 4 mg - oval white with break line on both faces. Lacidipine, 6 mg - oval
LACIPIL lacidipine QUALITATIVE AND QUANTITATIVE COMPOSITION Lacidipine, 2 mg - round shaped white engraved on one face. Lacidipine, 4 mg - oval white with break line on both faces. Lacidipine, 6 mg - oval
Renal Quiz - June 22, 21001
 Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
Renal Quiz - June 22, 21001 1. The molecular weight of calcium is 40 and chloride is 36. How many milligrams of CaCl 2 is required to give 2 meq of calcium? a) 40 b) 72 c) 112 d) 224 2. The extracellular
Chapter 15 Diuretic Agents
 Chapter 15 Diuretic Agents Diuretics Diuretics are agents that increase the rate of urine formation and salt excretion. Diuresis = increased water formation, but the term is also used to indicate increased
Chapter 15 Diuretic Agents Diuretics Diuretics are agents that increase the rate of urine formation and salt excretion. Diuresis = increased water formation, but the term is also used to indicate increased
PRODUCT CIRCULAR. Tablets COZAAR (losartan potassium) I. THERAPEUTIC CLASS II. INDICATIONS III. DOSAGE AND ADMINISTRATION PAK-CZR-T
 PRODUCT CIRCULAR Tablets I. THERAPEUTIC CLASS, the first of a new class of agents for the treatment of hypertension, is an angiotensin II receptor (type AT 1 ) antagonist. also provides a reduction in
PRODUCT CIRCULAR Tablets I. THERAPEUTIC CLASS, the first of a new class of agents for the treatment of hypertension, is an angiotensin II receptor (type AT 1 ) antagonist. also provides a reduction in
ZAROXOLYN 5mg tablets ZAROXOLYN 10 mg tablets Metolazone. PHARMACOTHERAPEUTIC CATEGORY Diuretic with minor diuretic action, non-associated.
 ZAROXOLYN 5mg tablets ZAROXOLYN 10 mg tablets Metolazone PHARMACOTHERAPEUTIC CATEGORY Diuretic with minor diuretic action, non-associated. THERAPEUTIC INDICATIONS Zaroxolyn is indicated in the treatment
ZAROXOLYN 5mg tablets ZAROXOLYN 10 mg tablets Metolazone PHARMACOTHERAPEUTIC CATEGORY Diuretic with minor diuretic action, non-associated. THERAPEUTIC INDICATIONS Zaroxolyn is indicated in the treatment
Diuretics are drugs that increase the volume of urine excreted. Most diuretic agents are inhibitors of renal ion transporters that decrease the
 Diuretics Diuretics are drugs that increase the volume of urine excreted. Most diuretic agents are inhibitors of renal ion transporters that decrease the reabsorption of Na+ at different sites in the nephron.
Diuretics Diuretics are drugs that increase the volume of urine excreted. Most diuretic agents are inhibitors of renal ion transporters that decrease the reabsorption of Na+ at different sites in the nephron.
Potassium regulation. -Kidney is a major regulator for potassium Homeostasis.
 Potassium regulation. -Kidney is a major regulator for potassium Homeostasis. Normal potassium intake, distribution, and output from the body. Effects of severe hyperkalemia Partial depolarization of cell
Potassium regulation. -Kidney is a major regulator for potassium Homeostasis. Normal potassium intake, distribution, and output from the body. Effects of severe hyperkalemia Partial depolarization of cell
Antihypertensive drugs SUMMARY Made by: Lama Shatat
 Antihypertensive drugs SUMMARY Made by: Lama Shatat Diuretic Thiazide diuretics The loop diuretics Potassium-sparing Diuretics *Hydrochlorothiazide *Chlorthalidone *Furosemide *Torsemide *Bumetanide Aldosterone
Antihypertensive drugs SUMMARY Made by: Lama Shatat Diuretic Thiazide diuretics The loop diuretics Potassium-sparing Diuretics *Hydrochlorothiazide *Chlorthalidone *Furosemide *Torsemide *Bumetanide Aldosterone
Circular, pink, biconvex, film-coated tablets of 6.4mm diameter embossed "n" on one side. Tablets contain 2.5 mg Indapamide hemihydrate.
 NAPAMIDE Indapamide hemihydrate 2.5 mg tablet Presentation Indapamide hemihydrate as: Circular, pink, biconvex, film-coated tablets of 6.4mm diameter embossed "n" on one side. Tablets contain 2.5 mg Indapamide
NAPAMIDE Indapamide hemihydrate 2.5 mg tablet Presentation Indapamide hemihydrate as: Circular, pink, biconvex, film-coated tablets of 6.4mm diameter embossed "n" on one side. Tablets contain 2.5 mg Indapamide
M0BCore Safety Profile
 M0BCore Safety Profile Active substance: Aciclovir Pharmaceutical form(s)/strength: Tablets 200, 400 or 800 mg Dispersible tablets 200, 400 or 800 mg Oral suspensions 200 mg or 400 mg per 5 ml. Freeze
M0BCore Safety Profile Active substance: Aciclovir Pharmaceutical form(s)/strength: Tablets 200, 400 or 800 mg Dispersible tablets 200, 400 or 800 mg Oral suspensions 200 mg or 400 mg per 5 ml. Freeze
Faculty version with model answers
 Faculty version with model answers Urinary Dilution & Concentration Bruce M. Koeppen, M.D., Ph.D. University of Connecticut Health Center 1. Increased urine output (polyuria) can result in a number of
Faculty version with model answers Urinary Dilution & Concentration Bruce M. Koeppen, M.D., Ph.D. University of Connecticut Health Center 1. Increased urine output (polyuria) can result in a number of
Olmesartan Medoxomil/ Hydrochlorothiazide
 Olmesartan Medoxomil/ Hydrochlorothiazide ` OLMETEC PLUS 1.0 PHARMACEUTICAL CATEGORY Antihypertensive Diuretic 2.0 DESCRIPTION Olmesartan medoxomil-hydrochlorothiazide is a fixed-dose combination of the
Olmesartan Medoxomil/ Hydrochlorothiazide ` OLMETEC PLUS 1.0 PHARMACEUTICAL CATEGORY Antihypertensive Diuretic 2.0 DESCRIPTION Olmesartan medoxomil-hydrochlorothiazide is a fixed-dose combination of the
Low Efficacy Diuretics. Potassium sparing diuretics. Carbonic anhydrase inhibitors. Osmotic diuretics. Miscellaneous
 University of Al Qadisiyah College of Pharmacy Dr. Bassim I Mohammad, MBChB, MSc, Ph.D Low Efficacy Diuretics 1. Potassium sparing diuretics 2. Carbonic anhydrase inhibitors 3. Osmotic diuretics 4. Miscellaneous
University of Al Qadisiyah College of Pharmacy Dr. Bassim I Mohammad, MBChB, MSc, Ph.D Low Efficacy Diuretics 1. Potassium sparing diuretics 2. Carbonic anhydrase inhibitors 3. Osmotic diuretics 4. Miscellaneous
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance
 Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Instrumental determination of electrolytes in urine. Amal Alamri
 Instrumental determination of electrolytes in urine Amal Alamri What is the Electrolytes? Electrolytes are positively and negatively chargedions, Found in Within body's cells extracellular fluids, including
Instrumental determination of electrolytes in urine Amal Alamri What is the Electrolytes? Electrolytes are positively and negatively chargedions, Found in Within body's cells extracellular fluids, including
Renal Excretion of Drugs
 Renal Excretion of Drugs 3 1 Objectives : 1 Identify main and minor routes of Excretion including renal elimination and biliary excretion 2 Describe its consequences on duration of drugs. For better understanding:
Renal Excretion of Drugs 3 1 Objectives : 1 Identify main and minor routes of Excretion including renal elimination and biliary excretion 2 Describe its consequences on duration of drugs. For better understanding:
Excretion of Drugs. Prof. Hanan Hagar Pharmacology Unit Medical College
 Excretion of Drugs Prof. Hanan Hagar Pharmacology Unit Medical College Excretion of Drugs By the end of this lecture, students should be able to! Identify main and minor routes of excretion including renal
Excretion of Drugs Prof. Hanan Hagar Pharmacology Unit Medical College Excretion of Drugs By the end of this lecture, students should be able to! Identify main and minor routes of excretion including renal
LESSON ASSIGNMENT. After completing this lesson, you will be able to: 4-1. Identify the general characteristics of diuretics.
 LESSON ASSIGNMENT LESSON 4 Diuretics. LESSON ASSIGNMENT Paragraphs 4-1 through 4-6. LESSON OBJECTIVES After completing this lesson, you will be able to: 4-1. Identify the general characteristics of diuretics.
LESSON ASSIGNMENT LESSON 4 Diuretics. LESSON ASSIGNMENT Paragraphs 4-1 through 4-6. LESSON OBJECTIVES After completing this lesson, you will be able to: 4-1. Identify the general characteristics of diuretics.
50% Concentrated Injection
 NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE
 Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Therapeutic Uses of Diuretics
 DIURETICS Diuretics are drugs that promote the output of urine excreted by the Kidneys. The primary action of most diuretics is the direct inhibition of a + transport at one or more of the four major anatomical
DIURETICS Diuretics are drugs that promote the output of urine excreted by the Kidneys. The primary action of most diuretics is the direct inhibition of a + transport at one or more of the four major anatomical
Potassium secretion. E k = -61 log ([k] inside / [k] outside).
![Potassium secretion. E k = -61 log ([k] inside / [k] outside). Potassium secretion. E k = -61 log ([k] inside / [k] outside).](/thumbs/80/80478709.jpg) 1 Potassium secretion In this sheet, we will continue talking about ultrafiltration in kidney but with different substance which is K+. Here are some informations that you should know about potassium;
1 Potassium secretion In this sheet, we will continue talking about ultrafiltration in kidney but with different substance which is K+. Here are some informations that you should know about potassium;
Salicylate (Aspirin) Ingestion California Poison Control Background 1. The prevalence of aspirin-containing analgesic products makes
 Salicylate (Aspirin) Ingestion California Poison Control 1-800-876-4766 Background 1. The prevalence of aspirin-containing analgesic products makes these agents, found in virtually every household, common
Salicylate (Aspirin) Ingestion California Poison Control 1-800-876-4766 Background 1. The prevalence of aspirin-containing analgesic products makes these agents, found in virtually every household, common
MESULID 100 REVISED PRODUCT INFORMATION
 MESULID 100 REVISED PRODUCT INFORMATION COMPOSITION Each caplet of MESULID 100 contains: Nimesulide 100 mg. Inactive Ingredients Lactose, microcrystalline cellulose, sodium starch glycolate, hydrogenated
MESULID 100 REVISED PRODUCT INFORMATION COMPOSITION Each caplet of MESULID 100 contains: Nimesulide 100 mg. Inactive Ingredients Lactose, microcrystalline cellulose, sodium starch glycolate, hydrogenated
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS. Page 4
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS Page 4 1. NAME OF THE MEDICINAL PRODUCT CoAprovel 150/12.5 mg tablets. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 150 mg irbesartan and
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS Page 4 1. NAME OF THE MEDICINAL PRODUCT CoAprovel 150/12.5 mg tablets. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 150 mg irbesartan and
NATURAL HISTORY AND SURVIVAL OF PATIENTS WITH ASCITES. PATIENTS WHO DO NOT DEVELOP COMPLICATIONS HAVE MARKEDLY BETTER SURVIVAL THAN THOSE WHO DEVELOP
 PROGNOSIS Mortality rates as high as 18-30% are reported for hyponatremic patients. High mortality rates reflect the severity of underlying conditions and are not influenced by treatment of hyponatremia
PROGNOSIS Mortality rates as high as 18-30% are reported for hyponatremic patients. High mortality rates reflect the severity of underlying conditions and are not influenced by treatment of hyponatremia
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Desunin 4000 IU Tablets Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains colecalciferol (vitamin D 3 ) 4000 IU (equivalent
1 NAME OF THE MEDICINAL PRODUCT Desunin 4000 IU Tablets Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains colecalciferol (vitamin D 3 ) 4000 IU (equivalent
Major intra and extracellular ions Lec: 1
 Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
CHAPTER 27 LECTURE OUTLINE
 CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
Dr.Nahid Osman Ahmed 1
 1 ILOS By the end of the lecture you should be able to Identify : Functions of the kidney and nephrons Signs and symptoms of AKI Risk factors to AKI Treatment alternatives 2 Acute kidney injury (AKI),
1 ILOS By the end of the lecture you should be able to Identify : Functions of the kidney and nephrons Signs and symptoms of AKI Risk factors to AKI Treatment alternatives 2 Acute kidney injury (AKI),
keyword: diuretics Drug monitoring Monitoring diuretics in primary care 2 March 2009 best tests
 www.bpac.org.nz keyword: diuretics Drug monitoring Monitoring diuretics in primary care 2 March 2009 best tests Why do we monitor patients taking diuretics and what do we monitor? Monitoring a person on
www.bpac.org.nz keyword: diuretics Drug monitoring Monitoring diuretics in primary care 2 March 2009 best tests Why do we monitor patients taking diuretics and what do we monitor? Monitoring a person on
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D.
 RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
Dosage in renal impairment Kalcipos-D chewable tablets should not be used in patients with severe renal impairment.
 SUMMARY OF THE PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Kalcipos-D 500 mg/400 IU chewable tablet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION One chewable tablet contains: calcium 500 mg as
SUMMARY OF THE PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Kalcipos-D 500 mg/400 IU chewable tablet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION One chewable tablet contains: calcium 500 mg as
CCRN Review - Renal. CCRN Review - Renal 10/16/2014. CCRN Review Renal. Sodium Critical Value < 120 meq/l > 160 meq/l
 CCRN Review Renal Leanna R. Miller, RN, MN, CCRN-CMC, PCCN-CSC, CEN, CNRN, CMSRN, NP Education Specialist LRM Consulting Nashville, TN Sodium 136-145 Critical Value < 120 meq/l > 160 meq/l Sodium Etiology
CCRN Review Renal Leanna R. Miller, RN, MN, CCRN-CMC, PCCN-CSC, CEN, CNRN, CMSRN, NP Education Specialist LRM Consulting Nashville, TN Sodium 136-145 Critical Value < 120 meq/l > 160 meq/l Sodium Etiology
Metformin Hydrochloride
 Metformin Hydrochloride 500 mg, 850 mg, 500 mg LA and 750 mg LA Tablet Description Informet is a preparation of metformin hydrochloride that belongs to a biguanide class of oral antidiabetic drugs. Metformin
Metformin Hydrochloride 500 mg, 850 mg, 500 mg LA and 750 mg LA Tablet Description Informet is a preparation of metformin hydrochloride that belongs to a biguanide class of oral antidiabetic drugs. Metformin
** TMP mean page 340 in 12 th edition. Questions 1 and 2 Use the following clinical laboratory test results for questions 1 and 2:
 QUESTION Questions 1 and 2 Use the following clinical laboratory test results for questions 1 and 2: Urine flow rate = 1 ml/min Urine inulin concentration = 100 mg/ml Plasma inulin concentration = 2 mg/ml
QUESTION Questions 1 and 2 Use the following clinical laboratory test results for questions 1 and 2: Urine flow rate = 1 ml/min Urine inulin concentration = 100 mg/ml Plasma inulin concentration = 2 mg/ml
HYDRODIURIL (HYDROCHLOROTHIAZIDE)
 7897450 TABLETS HYDRODIURIL (HYDROCHLOROTHIAZIDE) DESCRIPTION HydroDIURIL * (Hydrochlorothiazide) is a diuretic and antihypertensive. It is the 3,4-dihydro derivative of chlorothiazide. Its chemical name
7897450 TABLETS HYDRODIURIL (HYDROCHLOROTHIAZIDE) DESCRIPTION HydroDIURIL * (Hydrochlorothiazide) is a diuretic and antihypertensive. It is the 3,4-dihydro derivative of chlorothiazide. Its chemical name
45788C/Revised: April 2008 FUROSEMIDE INJECTION, USP
 45788C/Revised: April 2008 FUROSEMIDE INJECTION, USP WARNING Furosemide is a potent diuretic which, if given in excessive amounts, can lead to a profound diuresis with water and electrolyte depletion.
45788C/Revised: April 2008 FUROSEMIDE INJECTION, USP WARNING Furosemide is a potent diuretic which, if given in excessive amounts, can lead to a profound diuresis with water and electrolyte depletion.
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory OSTOCALCIUM FORTE TABLETS
 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory OSTOCALCIUM FORTE TABLETS Calcium and Phosphorus with Vitamin D3 Tablets QUALITATIVE AND QUANTITATIVE COMPOSITION Each
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory OSTOCALCIUM FORTE TABLETS Calcium and Phosphorus with Vitamin D3 Tablets QUALITATIVE AND QUANTITATIVE COMPOSITION Each
Chapter 23. Composition and Properties of Urine
 Chapter 23 Composition and Properties of Urine Composition and Properties of Urine (1 of 2) urinalysis the examination of the physical and chemical properties of urine appearance - clear, almost colorless
Chapter 23 Composition and Properties of Urine Composition and Properties of Urine (1 of 2) urinalysis the examination of the physical and chemical properties of urine appearance - clear, almost colorless
Article 30 Referral for Calcitugg (and associated names) Chewable tablets (Calcium carbonate)
 ANNEX I LIST OF THE NAMES, PHARMACEUTICAL FORMS, STRENGTHS, ROUTES OF ADMINISTRATION, MARKETING AUTHORISATION HOLDERS, PACKAGING AND PACKAGE SIZES OF THE MEDICINAL PRODUCT IN THE MEMBER STATES 1 Article
ANNEX I LIST OF THE NAMES, PHARMACEUTICAL FORMS, STRENGTHS, ROUTES OF ADMINISTRATION, MARKETING AUTHORISATION HOLDERS, PACKAGING AND PACKAGE SIZES OF THE MEDICINAL PRODUCT IN THE MEMBER STATES 1 Article
SUMMARY OF PRODUCT CHARACTERISTICS. One chewable tablet contains 1250 mg calcium carbonate (equivalent to 500 mg calcium).
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT [XXX] 500 mg chewable tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One chewable tablet contains 1250 mg calcium carbonate (equivalent
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT [XXX] 500 mg chewable tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One chewable tablet contains 1250 mg calcium carbonate (equivalent
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS
 RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (2) Dr. Attila Nagy 2017 TUBULAR FUNCTIONS (Learning objectives 54-57) 1 Tubular Transport About 99% of filtrated water and more than 90% of the filtrated
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (2) Dr. Attila Nagy 2017 TUBULAR FUNCTIONS (Learning objectives 54-57) 1 Tubular Transport About 99% of filtrated water and more than 90% of the filtrated
Osmotic Diuretics and Carbonic anhydrase inhibitors
 Lecture : Osmotic Diuretics and Carbonic anhydrase inhibitors Contents : Explanation Summary Questions 1 DIURETICS Definition: Are drugs that increase renal excretion of sodium and water resulting in increase
Lecture : Osmotic Diuretics and Carbonic anhydrase inhibitors Contents : Explanation Summary Questions 1 DIURETICS Definition: Are drugs that increase renal excretion of sodium and water resulting in increase
Mannitol-induced Metabolic Alkalosis
 Electrolyte & Blood Pressure :, 00 ) Mannitolinduced Metabolic Alkalosis Kyung Pyo Kang, M.D., Sik Lee, M.D., Kyung Hoon Lee, M.D., and Sung Kyew Kang, M.D. Department of Internal Medicine, Research Institute
Electrolyte & Blood Pressure :, 00 ) Mannitolinduced Metabolic Alkalosis Kyung Pyo Kang, M.D., Sik Lee, M.D., Kyung Hoon Lee, M.D., and Sung Kyew Kang, M.D. Department of Internal Medicine, Research Institute
014 Chapter 14 Created: 9:25:14 PM CST
 014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
014 Chapter 14 Created: 9:25:14 PM CST Student: 1. Functions of the kidneys include A. the regulation of body salt and water balance. B. hydrogen ion homeostasis. C. the regulation of blood glucose concentration.
1. remove: waste products: urea, creatinine, and uric acid foreign chemicals: drugs, water soluble vitamins, and food additives, etc.
 Making Water! OR is it really Just Water Just Ask the Nephron!! Author: Patricia L. Ostlund ostlundp@faytechcc.edu (910) 678-9892 Fayetteville Technical Community College Fayetteville, NC 28303 Its just
Making Water! OR is it really Just Water Just Ask the Nephron!! Author: Patricia L. Ostlund ostlundp@faytechcc.edu (910) 678-9892 Fayetteville Technical Community College Fayetteville, NC 28303 Its just
DIURETICS CARBONIC ANHYDRASE INHIBITORS THIAZIDE THIAZIDE-LIKE OSMOTIC DIURETICS LOOP DIURETICS POTASSIUM SPARING DIURETICS
 DIURETICS A diuretic is any substance that promotes diuresis, that is, the increased production of urine. This includes forced diuresis. There are several categories of diuretics. All diuretics increase
DIURETICS A diuretic is any substance that promotes diuresis, that is, the increased production of urine. This includes forced diuresis. There are several categories of diuretics. All diuretics increase
Furosemide Injection, USP (10 mg/ml) Fliptop Vial Ansyr TM Plastic Syringe
 Furosemide Injection, USP (10 mg/ml) Fliptop Vial Ansyr TM Plastic Syringe Rx only WARNING: Furosemide is a potent diuretic which, if given in excessive amounts, can lead to a profound diuresis with water
Furosemide Injection, USP (10 mg/ml) Fliptop Vial Ansyr TM Plastic Syringe Rx only WARNING: Furosemide is a potent diuretic which, if given in excessive amounts, can lead to a profound diuresis with water
Kidney Physiology. Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed
 Kidney Physiology Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed The purpose of tubular secrection To dispose of certain substances that are bound to plasma proteins. To
Kidney Physiology Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed The purpose of tubular secrection To dispose of certain substances that are bound to plasma proteins. To
AROMASIN 25mg (Tablets)
 APPROVED PACKAGE INSERT AROMASIN SCHEDULING STATUS: S4 PROPRIETARY NAME AND DOSAGE FORM: AROMASIN 25mg (Tablets) COMPOSITION: Each sugar-coated tablet contains 25 mg exemestane. Preservative: methyl p-hydroxybenzoate
APPROVED PACKAGE INSERT AROMASIN SCHEDULING STATUS: S4 PROPRIETARY NAME AND DOSAGE FORM: AROMASIN 25mg (Tablets) COMPOSITION: Each sugar-coated tablet contains 25 mg exemestane. Preservative: methyl p-hydroxybenzoate
LOZAR. Composition Each tablet contains Losartan potassium 50 mg.
 LOZAR Composition Each tablet contains Losartan potassium 50 mg. Tablets Action Angiotensin II [formed from angiotensin I in a reaction catalyzed by angiotensin converting enzyme (ACE, kininase II)], is
LOZAR Composition Each tablet contains Losartan potassium 50 mg. Tablets Action Angiotensin II [formed from angiotensin I in a reaction catalyzed by angiotensin converting enzyme (ACE, kininase II)], is
Water, Electrolytes, and Acid-Base Balance
 Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Hypertension Update. Sarah J. Payne, MS, PharmD, BCPS Assistant Professor, Department of Pharmacotherapy UNT System College of Pharmacy
 Hypertension Update Sarah J. Payne, MS, PharmD, BCPS Assistant Professor, Department of Pharmacotherapy UNT System College of Pharmacy Introduction 1/3 of US adults have HTN More prevalent in non-hispanic
Hypertension Update Sarah J. Payne, MS, PharmD, BCPS Assistant Professor, Department of Pharmacotherapy UNT System College of Pharmacy Introduction 1/3 of US adults have HTN More prevalent in non-hispanic
Homeostatic Regulation
 Homeostatic Regulation A hormone is :a Water-soluble hormones: Composed of amino acids and bind a receptor protein on the of the target cell. This starts a signal cascade inside the cell and the signal
Homeostatic Regulation A hormone is :a Water-soluble hormones: Composed of amino acids and bind a receptor protein on the of the target cell. This starts a signal cascade inside the cell and the signal
1. Urinary System, General
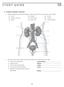 S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
