Defining the Optimal Activated Clotting Time During Percutaneous Coronary Intervention
|
|
|
- Lesley Henry
- 5 years ago
- Views:
Transcription
1 Defining the Optimal Activated Clotting Time During Percutaneous Coronary Intervention Aggregate Results From 6 Randomized, Controlled Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; A. Michael Lincoff, MD; David J. Moliterno, MD; Sorin J. Brener, MD; Katherine E. Wolski, MS; Eric J. Topol, MD Background Unfractionated heparin has been the primary anticoagulant therapy for percutaneous coronary intervention for 20 years. Despite the availability of rapid point of care testing, little clinical data defining the optimal level of anticoagulation are available. Furthermore, recent reports have advocated the use of low-dose heparin regimens in the absence of large-scale, well-conducted studies to support this practice. Methods and Results We pooled the data from 6 randomized, controlled trials of novel adjunctive antithrombotic regimens for percutaneous coronary interventions in which unfractionated heparin constituted the control arm. Patients were divided into 25-s intervals of activated clotting times (ACTs), from 275sto 476 s. In a total of 5216 patients, the incidence of death, myocardial infarction, or any revascularization and major or minor bleeding at 7 days were calculated for each group and compared. An ACT in the range of 350 to 375 s provided the lowest composite ischemic event rate of 6.6%, or a 34% relative risk reduction in 7-day ischemic events compared with rates observed between 171 and 295 s by quartile analysis (P 0.001). Conclusions Contrary to recent reports, the optimal suppression of ischemic events with unfractionated heparin therapy in patients undergoing percutaneous coronary intervention demands treatment to ACT levels that are substantially higher than currently appreciated. These data define a goal for heparin dosing within coronary interventions and establish a benchmark of optimal unfractionated heparin therapy against which future trials of novel antithrombotic regimens in percutaneous interventions can be compared. (Circulation. 2001;103: ) Key Words: heparin clinical trials pharmacology coagulation Since the development of percutaneous coronary intervention (PCI), intravenous unfractionated heparin has remained the primary antithrombotic therapy for the prevention of periprocedural ischemic complications. 1 Adequate dosing with unfractionated heparin effectively suppresses the thrombin generation associated with balloon-induced vascular injury. 2 Despite the continued evolution of antithrombotic therapies, unfractionated heparin remains an attractive option given its relatively low cost, the availability of a rapid point of care test for dose individualization (the activated clotting time [ACT]), and a known antagonist that allows the prompt reversal of antithrombin activity. Therefore, considering the wealth of experience associated with the clinical use of this agent, an optimal level of anticoagulation should be attainable in most patients undergoing PCI. However, defining this optimal level of ACT has been hampered by the lack of large-scale, randomized data. Current recommendations of ACT levels of 300 to 350 s are empiric 3 and based on relatively small studies. 4 6 Furthermore, in clinical practice, periprocedural heparin dosing varies markedly, with a recent emerging trend toward the use of lower heparin doses, despite the support of only a few small observational series. 7,8 Therefore, we performed an analysis combining individual patient data from 6 randomized, controlled trials to determine the optimum range of ACT for the suppression of periprocedural ischemic events after PCI. Methods Trial Selection, Heparin Treatments, and ACT Measures Trial selection was confined to studies of novel pharmacotherapies in the setting of PCI using aspirin and unfractionated heparin as the comparator arm. Routine ACT monitoring before device activation, the maximum ACT during the procedure, and 7-day follow-up documenting death, myocardial infarction (MI), urgent revascularization, and major or minor bleeding were also required for inclusion in our combined data set. Therefore, 6 randomized, controlled trials of patients undergoing PCI were available. These trials are summarized in Table ACT values were drawn from the placebo arms of these studies, and a comparative analysis was also performed in the abciximab-treated patients from the Evaluation of c7e3 for the Prevention of Ischemic Complications (EPIC) trial, Evaluation in Received August 2, 2000; revision received October 13, 2000; accepted October 16, From the Department of Cardiology, Cleveland Clinic Foundation, Cleveland, Ohio. Correspondence to Eric J. Topol, MD, Department of Cardiology, Cleveland Clinic Foundation, Desk F25, 9500 Euclid Ave, Cleveland, Ohio topole@ccf.org 2001 American Heart Association, Inc. Circulation is available at 961
2 962 Circulation February 20, 2001 TABLE 1. Summary of Trials EPIC 9 (n 2099) EPILOG 10 (n 2792) EPISTENT 13 (n 2399) IMPACT II 11 (n 4010) RAPPORT 13 (n 438) Hirudin Angioplasty Study 12 (n 4098) Intervention PTCA PTCA (stent) Stent PTCA PTCA PTCA Randomized therapy Abciximab g kg 1 hr 1 Abciximab g kg 1 hr 1 Abciximab g kg 1 hr 1 Eptifibatide 135 g/kg 0.5 or 0.75 g kg 1 min Abciximab g kg 1 h 1 Bivalirudin 1.0 mg/kg 2.5 mg/kg over 4 hours Placebo arm ( U) (175 U/kg) Maximum heparin dose U U U U U 295 U/kg Targeted ACT, s Minimum ACT at device implementation, s Maximum ACT, s Population High-risk PTCA All patients All patients High-risk PTCA Acute MI Unstable angina EPIC indicates Evaluation of c7e3 for the Prevention of Ischemic Complications; EPILOG, Evaluation in PTCA to Improve Long-Term Outcome with abciximab Glycoprotein IIb/IIIa blockade; EPISTENT, Evaluation of IIb/IIIa Platelet Inhibitor for Stenting; IMPACT II, Integrilin to Minimize Platelet Aggregation and Coronary Thrombosis II; and RAPPORT, Reopro and Primary PTCA Organization and Randomized Trial. PTCA to Improve Long-term Outcome with abciximab Glycoprotein IIb/IIIa blockade (EPILOG) trial, Evaluation of IIb/IIIa Platelet Inhibitor for Stenting (EPISTENT) trial, and ReoPro and Primary PTCA Organization and Randomized Trial (RAPPORT). Each protocol targeted an ACT level 300 s in the control arms. The device used to assay the ACT was specified in all but 904 patients. The Hemochron assay (International Technidyne Corporation) and the HemoTec assay (HemoTec Inc) were used in 95% and 5% of patients, respectively, in whom the device used was recorded. Among the trials, mean minimum ACT levels at device activation were similar, whereas the mean maximum ACTs varied more substantially. The minimum ACT at device activation was analyzed with regard to ischemic events at 7 days because this value was considered to reflect the degree of antithrombotic therapy at vascular injury most closely. Conversely, the maximum ACT at any time during the procedure was assessed relative to bleeding events, because this value reflects the greatest risk of bleeding to which the patient was exposed. Statistical Methods The minimum ACTs at or around the time of device activation were collected and divided into the following groups: 275 s, 275 to 300 s, 301 to 325 s, 326 to 350 s, 351 to 375 s, 376 to 400 s, 401 to 425 s, 426 to 475 s, and 476 s. The incidence of periprocedural ischemic events within each group was calculated. The maximum ACT during the procedure was also recorded and similarly divided, and the incidence of bleeding events was calculated for each range. Subgroup analyses were also performed for patients with diabetes and acute coronary syndromes and those receiving stents. The end point definitions for death, MI, and urgent revascularization were as defined in the individual protocols, and they were similar among the trials. The definition of bleeding end points for the Hirulog Angioplasty Study 12 varied substantially from the other 5 studies and, therefore, it was omitted from the bleeding analysis. Baseline characteristics are expressed as percentages for discrete variables or as mean SD for continuous variables. Event rates for each ACT interval were calculated as the percentage of patients experiencing an event within the specific ACT interval. Examination of event rates as quartiles of minimum ACT at device activation was also undertaken to minimize the effect of small sample sizes across some ranges of ACT. The 2 statistic was used to compare the lowest and highest event rates in the arbitrarily determined ACT ranges and to compare event rates among the quartiles. P 0.05 was considered significant. The Lowess smoothing function was used to generate graphic representations of the relationships between increasing ACT and outcomes. Results Population Characteristics In total, 6146 patients received unfractionated heparin alone and 5216 patients (85%) had ACT data available at or around the time of device activation. Maximum ACT results were available for 5444 patients (89%); however, with the Hirulog Angioplasty Study excluded, 3485 patients (64%) were used in the bleeding analysis. The demographic and procedural characteristics of all patients are presented in Table 2. Importantly, patients with acute coronary syndromes represented a substantial proportion of the population studied TABLE 2. Demographics and Procedural Characteristics Characteristic All Studies Pooled Total No. of patients 6146 Age, y Current smoker 1802 (29.5) Diabetes 1369 (22.3) Acute coronary syndrome 3699 (60.2) Hypertension 3399 (55.4) History of CHF 365 (6.0) Prior CABG 746 (12.1) Prior PCI 1489 (24.3) Prior MI 2832 (46.1) Procedure 60 min 1549 (26.6) Initial heparin dose, U Total heparin dose, U Mean ACT at device activation, s Maximum ACT, s Any stent 936 (15.3) 1 Vessel disease 4020 (65.4) 2 Vessel disease 1360 (22.1) 3 Vessel disease 606 (9.9) Values are mean SD or n (%).
3 Chew et al Defining Optimal ACT During PCI 963 ischemic events at 7 days was associated with achieving an ACT level 350s(P 0.001; Figure 2). Maximum ACT and Bleeding Events The maximum ACT was correlated with the incidence of major and minor bleeding (n 3485) across the same ranges of the ACT. The lowest level of bleeding (8.6%) was observed in the range of 325 to 350 s, and it progressively increased to 12.4% at 350 to 375 s. A substantial increase in bleeding events was observed when ACT values exceeded 400 s (Figure 1B). Figure 1. A, Relationship between minimum ACT at or around time of device activation and death, MI, or urgent revascularization (Revasc) at 7 days. B, Relationship between maximum ACT and major or minor bleeding events at 7 days. Lowess smoothing estimate and 95% confidence intervals are indicated by solid and dotted lines, respectively. Percentages represent actual event rates observed. (60%). In addition, 15% of all patients received 1 stent, and a procedural duration of 1 hour was observed in 27% of cases. ACT at Device Activation and Ischemic Events The relationship between increasing ACT and ischemic events (n 5216) followed a U-shaped curve. A progressive reduction in the risk of death, MI, and urgent revascularization was observed with increasing ACT levels, as evidenced by a reduction in the composite ischemic end point from 11.1% at an ACT of 275 to 300 s to a nadir of 6.6% with an ACT of 350 to 375 s ( 2 statistic between highest event rate and lowest event rate, P 0.026). Beyond this level of ACT, the risk of ischemic events rises, increasing to 9.3% at ACT levels 475 s (Figure 1). The optimal level of ACT, when grouped by short ( 1 hour) and long ( 1 hour) procedures, remained similar to the overall analysis. Analysis by quartiles also demonstrates a progressive reduction in ischemic event rates with increasing ACT levels. An ACT level at a device activation of 350 s provided a composite ischemic event rate of 7.7%; the rate was 11.6% when the ACT level was in the range of 171 to 295 s. Thus, a 34% relative risk reduction in adverse postprocedural Subgroups Analysis of diabetics included 1135 patients with available ACT data (83%). Correlations between the minimum ACT at device activation and ischemic events at 7 days demonstrated a similar U-shaped relationship. In comparison with the nondiabetic population and the overall population, a more pronounced reduction in ischemic events was evident between ACT levels of 350 to 375 s compared with those of 275 to 300 s in diabetic patients (Figure 3). Assessment of outcomes in the diabetic population when stratified by the same ACT ranges defined by the overall population demonstrated a 38% reduction in 7-day composite ischemic events in these ACT ranges (ACT of 171 to 295 s, 12.5%; ACT 350 s, 7.7%; P 0.062). The relationship between ACT and 7-day ischemic outcomes among patients receiving coronary stents is derived primarily from the EPISTENT 13 study, because stent use was restricted to bailout indications in the other studies. A total of 766 patients are included in this analysis (82%). Again, a decline in adverse ischemic events was observed with increasing levels of ACT, with lowest rates observed in the range of 350 to 375 s. In the 3203 patients enrolled with a diagnosis of unstable angina, non Q-wave MI, evolving MI, or postinfarction angina and who had available ACT data (87%), the relationship between minimum ACT at device activation and ischemic events at 7 days resembled the overall analysis. Concurrent Abciximab With concurrent abciximab administration (n 4362), analysis of the 4 abciximab trials demonstrates lower ischemic event rates across the entire range of ACT values. However, in the 3458 patients with available minimum ACT data, the U-shaped relationship is no longer evident, with a plateau in the ischemic event rate of 4.5% at 7 days across a broad range of values (275 to 375 s; Figure 4A). Data for maximum ACT were available for a total of 3876 patients. The correlation between the maximum ACT and major and minor bleeding events demonstrated a slightly increased rate of bleeding with abciximab compared with heparin alone across the ranges of 275 to 375 s, with a substantial increase associated with ACTs beyond this level (Figure 4B). Discussion This analysis of individual patient data derived from the placebo arms of 6 randomized glycoprotein IIb/IIIa trials represents the largest study correlating the level of unfrac-
4 964 Circulation February 20, 2001 Figure 2. Analysis of ACT and composite ischemic event rates at 7 days as assessed in quartiles. Revasc indicates revascularization. tionated heparin therapy and early postprocedural outcome. From these large-scale PCI trials, an optimal range of ACT for unfractionated heparin therapy of between 350 and 375 s is evident; this range is substantially higher than previously appreciated. Considering that more than two thirds of the patients within this analysis presented with an episode of coronary instability, this degree of anticoagulation seems to confer a low ischemic event rate of 6.6%, as defined by the composite end point of death, MI, or urgent revascularization at 7 days. This analysis runs contrary to the widely held clinical impression suggesting that lower dosing with unfractionated heparin is associated with equivalent clinical outcomes. Recent reports have advocated the use of very-low-dose unfractionated heparin (2500 to 5000 U) among patients undergoing PCI. In a single-center observational study of 1375 patients receiving 5000 U of heparin before PCI, Koch and coworkers 7 observed a relatively low rate of death, MI, or repeat revascularization of 5.4% at 48 hours. However, in addition to the shorter duration of follow-up, patients with acute coronary syndromes and those patients undergoing stenting were excluded from the analysis, and diabetics Figure 3. Relationship between minimum ACT at or around time of device activation and death, MI, or urgent revascularization (Revasc) at 7 days in diabetic patients. Lowess smoothing estimate and 95% confidence intervals are indicated by solid and dotted lines, respectively. Figure 4. A, Relationship between minimum ACT at device activation and death, MI, or urgent revascularization (Revasc) at 7 days in patients receiving concurrent abciximab. B, Relationship between maximum ACT and major or minor bleeding events at 7 days in abciximab-treated patients. Lowess smoothing estimate and 95% confidence intervals are indicated by solid and dotted lines, respectively. constituted only 10% of the study population. Similarly, Kaluski et al 8 reported a 3.3% event rate for death, MI, and urgent revascularization with 2500 U of heparin before PCTA, but postprocedural creatine kinase was not routinely assayed, which likely underestimated the incidence of periprocedural MI. 8 Vainer and colleagues 15 performed a small trial of 404 patients randomized to either 5000 or U of heparin before PCI. In the low-dose heparin group, only 22% of patients attained an ACT 275 s. This group had a composite event rate for death, MI, repeat revascularization, or acute occlusion of 13.2%; the rate for the high-dose group was 8% (P NS). This lack of statistical significance has been interpreted as support for low-dose heparin regimens, 8 although an inadequate sample size is a more likely explanation. This current analysis refutes the notion that low-dose heparin therapy provides an efficacy similar to that of higher doses in PCI. Thus, whereas a previous trial attempting to demonstrate superior safety and efficacy with weightindividualized heparin dosing over a fixed-dosing regimen has been inconclusive, 16 these data identify a therapeutic goal for unfractionated heparin therapy in individuals undergoing PCI. Furthermore, by increasing the therapeutic goal to 350
5 Chew et al Defining Optimal ACT During PCI 965 s, we observed a 34% relative risk reduction in ischemic events at 7 days. However, an increase in bleeding events is evident at this optimal range of ACT. Previous authors have also attempted to define a threshold beyond which additional heparin provided no further incremental benefit. In the setting of a 186-patient, single-center, case-control study drawn from a registry of 1290 patients, Narins and coworkers 4 were unable to determine a safe threshold for the ACT, observing an inverse relationship between ACT and ischemic events that remained linear through the observed ACT values. Likewise, Bittl et al 17 reported a similar relationship in the 2039 heparin-treated patients from the randomized trial of bivalirudin in PCI. In contrast, the present analysis demonstrates a U-shaped curve for ischemic risk, with ACT levels greater than 400 s associated with a greater rate of ischemic events, as observed in patients with acute MI undergoing thrombolytic therapy. 18 The excess thrombotic risk observed at higher ACTs likely represents the clinical confirmation of the platelet activation observed with high doses of unfractionated heparin within in vitro models and ex vivo studies Interestingly, from this analysis, concurrent glycoprotein IIb/IIIa therapy seems to ameliorate the ischemic risk associated with high ACT levels, reinforcing the thrombotic pathophysiology of these events; this, again, is consistent with heparin-induced platelet activation. A similar analysis of bivalirudin therapy observed no increase in ischemic events at high levels of anticoagulation, 17 possibly reflecting an absence of platelet activation observed with direct thrombin inhibitors. 19 The impact of concurrent abciximab on the ACT has been previously defined, 22 and this synergy has implications for the optimal dosing of heparin in abciximab-treated patients. This meta-analysis confirms the dictum of targeting lower ACT levels when heparin and abciximab are combined, demonstrating a true inverse relationship between increasing ACT and ischemic events. Overall ischemic event rates are, as expected, reduced in comparison with heparin-only treated patients for all levels of ACT, with a plateau across a broad range (250 to 350 s). Because the ability to inhibit thrombin generation and prolong the ACT varies among glycoprotein IIb/IIIa inhibitors, a similar relationship between heparin, ACT levels, and eptifibatide or tirofiban cannot necessarily be expected; this requires formal definition. The association between diabetes and adverse outcomes after PCI is well established. Abnormalities in baseline platelet function and coagulation may partially account for the increased risk. Complementing the previous observation that diabetics undergoing PCI benefit from higher heparin doses despite concurrent abciximab use, 23 this analysis demonstrates a more prominent reduction in adverse ischemic events associated with achieving ACT levels of 350 s in the absence of concurrent abciximab therapy. By quartile analysis, achieving this higher ACT level provided a 38% relative risk reduction in 7-day composite ischemic events, although this reduction did not reach statistical significance due to the smaller sample size. Beyond daily clinical practice, this meta-analysis has implications for the design of future trials investigating pharmacotherapies in PCI. Many recent studies have used heparin doses designed to achieve ACT levels that are substantially lower than those advocated by this analysis. Therefore, whether the incremental benefit over standard unfractionated heparin therapy defined by these studies would hold true with optimal heparin dosing remains uncertain. Although not the case for adjunctive abciximab therapy in the nondiabetic population, 10 more aggressive heparin dosing may provide a greater suppression of ischemic events within the placebo arm, reducing the relative and absolute benefit of the investigational therapy. In future PCI trials in which aspirin, unfractionated heparin, and thienopyridines are used as the placebo-comparator arm, targeting heparin therapy to ACT levels in the range of 350 to 375 s would be required to define the true relative benefit of any novel therapy. Limitations ACT data were incomplete and, therefore, the potential for unanticipated selection biases remains. However, with such a large population, omission of data are likely to be randomly distributed across the observed ACT values. Consistent with this, analysis of demographic and procedural variables among patients without ACT data seemed similar to those in patients in whom data were available. Similarly, despite the exclusion of the Hirulog Angioplasty Study 12 from bleeding outcome analysis, these results were also previously shown to correlate positively with maximum ACT but not initial ACT. 17 In addition, the specific device used to measure ACT is known to impact ACT values. For 85% of the patients, the device used to determine the ACT was recorded. When patients with ACT values recorded from the Hemochron device (95%) were analyzed separately, the range of optimal ACT remained unchanged, whereas the magnitude of difference between an ACT of 275 to 300 s and of 350 to 375 s increased marginally. As expected, an analysis of the 279 patients with ACT values recorded on the HemoTec assay suggested optimal efficacy in the range of 300 to 325 s, which is consistent with the previously defined 28% lower readings with this device. 24 Finally, among the trials, the relationship between the ACT and ischemic events at 7 days demonstrates some heterogeneity, likely reflecting minor differences in patient populations and procedure-specific factors. Although these issues may contribute some degree of uncertainty in the absolute magnitude of benefit attainable with optimal heparin dosing, overall trends in benefit are similar among the trials. Operator-specific variations in procedural outcomes are inherent limitations of all large-scale studies involving technical procedures, both within and among trials. This limitation is somewhat mitigated by the randomized nature of each study and the availability of patient-specific data for incorporation into this analysis. Conclusions With the escalation in the cost of medical therapies, the optimization of current approaches remains as important as innovation. This meta-analysis of patient-specific data identifies an optimal level of ACT for patients undergoing PCI in the range of 350 to 375 s, a level of anticoagulation that runs
6 966 Circulation February 20, 2001 contrary to recent clinical impressions. Furthermore, in subgroups at greater risk of thrombotic events, a steeper gradient of benefit between lower and higher levels of ACT is evident. However, this analysis also highlights the narrow therapeutic range of unfractionated heparin therapy in PCI, demonstrating the increased bleeding risk incurred when targeting maximal efficacy. Combined with the observed nonlinear relationship between the ACT level and ischemic risk, these data not only define an optimal range for individual patient therapy, but they underscore the limitations of unfractionated heparin. This analysis, therefore, delineates the benchmark of both risk and benefit with unfractionated heparin therapy and sets a standard for the development of new anticoagulant strategies. References 1. Gruntzig AR, Senning A, Siegenthaler WE. Nonoperative dilatation of coronary-artery stenosis: percutaneous transluminal coronary angioplasty. N Engl J Med. 1979;301: Ragosta M, Karve M, Brezynski D, et al. Effectiveness of heparin in preventing thrombin generation and thrombin activity in patients undergoing coronary intervention. Am Heart J. 1999;137: Popma JJ, Weitz J, Bittl JA, et al. Antithrombotic therapy in patients undergoing coronary angioplasty. Chest. 1998;114:728S 741S. 4. Narins CR, Hillegass WB Jr, Nelson CL, et al. Relation between activated clotting time during angioplasty and abrupt closure. Circulation. 1996; 93: Ferguson JJ, Barasch E, Wilson JM, et al. The relation of clinical outcome to dissection and thrombus formation during coronary angioplasty: Heparin Registry Investigators. J Invasive Cardiol. 1995;7: McGarry TF Jr, Gottlieb RS, Morganroth J, et al. The relationship of anticoagulation level and complications after successful percutaneous transluminal coronary angioplasty. Am Heart J. 1992;123: Koch KT, Piek JJ, de Winter RJ, et al. Safety of low dose heparin in elective coronary angioplasty. Heart. 1997;77: Kaluski E, Krakover R, Cotter G, et al. Minimal heparinization in coronary angioplasty-how much heparin is really warranted? Am J Cardiol. 2000;85: The EPIC Investigators. Use of a monoclonal antibody directed against the platelet glycoprotein IIb/IIIa receptor in high-risk coronary angioplasty: the EPIC Investigation. N Engl J Med. 1994;330: EPILOG Investigators. Platelet glycoprotein IIb/IIIa receptor blockade and low-dose heparin during percutaneous coronary revascularization. N Engl J Med. 1997;336: IMPACT II Investigators. Randomised placebo-controlled trial of effect of eptifibatide on complications of percutaneous coronary intervention: IMPACT-II: Integrilin to Minimise Platelet Aggregation and Coronary Thrombosis-II. Lancet. 1997;349: Bittl JA, Strony J, Brinker JA, et al. Treatment with bivalirudin (Hirulog) as compared with heparin during coronary angioplasty for unstable or postinfarction angina: Hirulog Angioplasty Study Investigators. N Engl J Med. 1995;333: Lincoff AM, Califf RM, Moliterno DJ, et al. Complementary clinical benefits of coronary-artery stenting and blockade of platelet glycoprotein IIb/IIIa receptors: Evaluation of Platelet IIb/IIIa Inhibition in Stenting Investigators. N Engl J Med. 1999;341: Brener SJ, Barr LA, Burchenal JE, et al. Randomized, placebo-controlled trial of platelet glycoprotein IIb/IIIa blockade with primary angioplasty for acute myocardial infarction: ReoPro and Primary PTCA Organization and Randomized Trial (RAPPORT) Investigators. Circulation. 1998;98: Vainer J, Fleisch M, Gunnes P, et al. Low-dose heparin for routine coronary angioplasty and stenting. Am J Cardiol. 1996;78: Boccara A, Benamer H, Juliard JM, et al. A randomized trial of a fixed high dose vs a weight-adjusted low dose of intravenous heparin during coronary angioplasty. Eur Heart J. 1997;18: Bittl JA, Ahmed WH. Relation between abrupt vessel closure and the anticoagulant response to heparin or bivalirudin during coronary angioplasty. Am J Cardiol. 1998;82:50P 56P. 18. Granger CB, Hirsch J, Califf RM, et al. Activated partial thromboplastin time and outcome after thrombolytic therapy for acute myocardial infarction: results from the GUSTO-I trial. Circulation. 1996;93: Xiao Z, Theroux P. Platelet activation with unfractionated heparin at therapeutic concentrations and comparisons with a low-molecular-weight heparin and with a direct thrombin inhibitor. Circulation. 1998;97: Mascelli MA, Kleiman NS, Marciniak SJ Jr, et al. Therapeutic heparin concentrations augment platelet reactivity: implications for the pharmacologic assessment of the glycoprotein IIb/IIIa antagonist abciximab. Am Heart J. 2000;139: Knight CJ, Panesar M, Wilson DJ, et al. Increased platelet responsiveness following coronary stenting. Heparin as a possible aetiological factor in stent thrombosis. Eur Heart J. 1998;19: Moliterno DJ, Califf RM, Aguirre FV, et al. Effect of platelet glycoprotein IIb/IIIa integrin blockade on activated clotting time during percutaneous transluminal coronary angioplasty or directional atherectomy (the EPIC trial): Evaluation of c7e3 Fab in the Prevention of Ischemic Complications trial. Am J Cardiol. 1995;75: Kleiman NS, Lincoff AM, Kereiakes DJ, et al. Diabetes mellitus, glycoprotein IIb/IIIa blockade, and heparin: evidence for a complex interaction in a multicenter trial: EPILOG Investigators. Circulation. 1998;97: Avendano A, Ferguson JJ. Comparison of Hemochron and HemoTec activated coagulation time target values during percutaneous transluminal coronary angioplasty. J Am Coll Cardiol. 1994;23:
Platelet glycoprotein IIb/IIIa inhibition in acute coronary syndromes
 European Heart Journal (00) 3, 1441 1448 doi:10.1053/euhj.00.3160, available online at http://www.idealibrary.com on Platelet glycoprotein IIb/IIIa inhibition in acute coronary syndromes Gradient of benefit
European Heart Journal (00) 3, 1441 1448 doi:10.1053/euhj.00.3160, available online at http://www.idealibrary.com on Platelet glycoprotein IIb/IIIa inhibition in acute coronary syndromes Gradient of benefit
THE ECONOMICS OF ADJUNCTIVE THERAPIES IN CORONARY ANGIOPLASTY: DRUGS, DEVICES, OR BOTH?
 THE ECONOMICS OF ADJUNCTIVE THERAPIES IN CORONARY ANGIOPLASTY: DRUGS, DEVICES, OR BOTH? Paul I. Oh, 1 Eric A. Cohen, 2 Nicole Mittmann, 3, 4 Soo Jin Seung 4 1 Division of Clinical Pharmacology, Sunnybrook
THE ECONOMICS OF ADJUNCTIVE THERAPIES IN CORONARY ANGIOPLASTY: DRUGS, DEVICES, OR BOTH? Paul I. Oh, 1 Eric A. Cohen, 2 Nicole Mittmann, 3, 4 Soo Jin Seung 4 1 Division of Clinical Pharmacology, Sunnybrook
Despite the development of new intravenous anticoagulants
 Vascular Medicine Relationship Between Activated Clotting Time and Ischemic or Hemorrhagic Complications Analysis of 4 Recent Randomized Clinical Trials of Percutaneous Coronary Intervention Sorin J. Brener,
Vascular Medicine Relationship Between Activated Clotting Time and Ischemic or Hemorrhagic Complications Analysis of 4 Recent Randomized Clinical Trials of Percutaneous Coronary Intervention Sorin J. Brener,
Report on New Patented Drug - Angiomax
 Report on New Patented Drug - Angiomax March 2006 Under its transparency initiative, the PMPRB publishes the results of the reviews of new patented drugs by Board Staff, for purposes of applying the PMPRB
Report on New Patented Drug - Angiomax March 2006 Under its transparency initiative, the PMPRB publishes the results of the reviews of new patented drugs by Board Staff, for purposes of applying the PMPRB
Anti-thrombotic management in interventional cardiology
 Chapter 8 Anti-thrombotic management in interventional cardiology James Tcheng, Steve Kindsvater Introduction Since the dawn of the interventional age, the need for anti-thrombotic therapy during percutaneous
Chapter 8 Anti-thrombotic management in interventional cardiology James Tcheng, Steve Kindsvater Introduction Since the dawn of the interventional age, the need for anti-thrombotic therapy during percutaneous
Interventional Cardiology
 Clinical Investigations Interventional Cardiology Benefit of bolus-only platelet glycoprotein IIb/IIIa inhibition during percutaneous coronary intervention: Insights from the very early outcomes in the
Clinical Investigations Interventional Cardiology Benefit of bolus-only platelet glycoprotein IIb/IIIa inhibition during percutaneous coronary intervention: Insights from the very early outcomes in the
Health technology Abciximab use in high-risk patients undergoing percutaneous transluminal coronary angioplasty.
 Costs and effects in therapy for acute coronary syndromes: the case of abciximab in highrisk patients undergoing percutaneous transluminal coronary angioplasty in the EPIC study van Hout B A, Bowman L,
Costs and effects in therapy for acute coronary syndromes: the case of abciximab in highrisk patients undergoing percutaneous transluminal coronary angioplasty in the EPIC study van Hout B A, Bowman L,
Scottish Medicines Consortium
 Scottish Medicines Consortium bivalirudin 250mg for injection or infusion (Angiox ) (156/05) Nycomed UK Ltd No. 4 February, 2005 The Scottish Medicines Consortium has completed its assessment of the above
Scottish Medicines Consortium bivalirudin 250mg for injection or infusion (Angiox ) (156/05) Nycomed UK Ltd No. 4 February, 2005 The Scottish Medicines Consortium has completed its assessment of the above
QUT Digital Repository:
 QUT Digital Repository: http://eprints.qut.edu.au/ This is the author s version of this journal article. Published as: Doggrell, Sheila (2010) New drugs for the treatment of coronary artery syndromes.
QUT Digital Repository: http://eprints.qut.edu.au/ This is the author s version of this journal article. Published as: Doggrell, Sheila (2010) New drugs for the treatment of coronary artery syndromes.
Collectively, the efficacy of the intravenous glycoprotein
 Increased Mortality With Oral Platelet Glycoprotein IIb/IIIa Antagonists A Meta-Analysis of Phase III Multicenter Randomized Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; Shelly Sapp, MS; Eric J. Topol,
Increased Mortality With Oral Platelet Glycoprotein IIb/IIIa Antagonists A Meta-Analysis of Phase III Multicenter Randomized Trials Derek P. Chew, MBBS; Deepak L. Bhatt, MD; Shelly Sapp, MS; Eric J. Topol,
Journal of the American College of Cardiology Vol. 44, No. 9, by the American College of Cardiology Foundation ISSN /04/$30.
 Journal of the American College of Cardiology Vol. 44, No. 9, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2004.05.085
Journal of the American College of Cardiology Vol. 44, No. 9, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2004.05.085
A randomized trial of a fixed high dose vs a weight-adjusted low dose of intravenous heparin during coronary angioplasty
 European Heart Journal (1997) 18, 631-635 A randomized trial of a fixed high dose vs a weight-adjusted low dose of intravenous heparin during coronary angioplasty A. Boccara, H. Benamer, J.-M. Juliard,
European Heart Journal (1997) 18, 631-635 A randomized trial of a fixed high dose vs a weight-adjusted low dose of intravenous heparin during coronary angioplasty A. Boccara, H. Benamer, J.-M. Juliard,
Percutaneous coronary intervention (PCI), in
 ECONOMIC IMPLICATIONS OF THE USE OF DIRECT THROMBIN INHIBITORS IN PERCUTANEOUS CORONARY INTERVENTION David J. Cohen, MD, MSc * ABSTRACT More than 1.2 million percutaneous coronary intervention (PCI) procedures
ECONOMIC IMPLICATIONS OF THE USE OF DIRECT THROMBIN INHIBITORS IN PERCUTANEOUS CORONARY INTERVENTION David J. Cohen, MD, MSc * ABSTRACT More than 1.2 million percutaneous coronary intervention (PCI) procedures
The use of percutaneous coronary intervention (PCI) as
 Antithrombotic Therapy During Percutaneous Coronary Intervention The Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy Jeffrey J. Popma, MD; Peter Berger, MD; E. Magnus Ohman, MD, FCCP;
Antithrombotic Therapy During Percutaneous Coronary Intervention The Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy Jeffrey J. Popma, MD; Peter Berger, MD; E. Magnus Ohman, MD, FCCP;
Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement
 Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement Pharmaceutical Care Project Outcomes Literature Evaluation
Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement Pharmaceutical Care Project Outcomes Literature Evaluation
Inter-regional differences and outcome in unstable angina
 European Heart Journal (2000) 21, 1433 1439 doi:10.1053/euhj.1999.1983, available online at http://www.idealibrary.com on Inter-regional differences and outcome in unstable angina Analysis of the International
European Heart Journal (2000) 21, 1433 1439 doi:10.1053/euhj.1999.1983, available online at http://www.idealibrary.com on Inter-regional differences and outcome in unstable angina Analysis of the International
Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015
 Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015 Objectives Review the pharmacology and pharmacokinetic
Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015 Objectives Review the pharmacology and pharmacokinetic
Bolus-Only Platelet Glycoprotein IIb/IIIa Inhibition During Percutaneous Coronary Intervention
 Original Contribution Bolus-Only Platelet Glycoprotein IIb/IIIa Inhibition During Percutaneous Coronary Intervention Jonathan D. Marmur, MD, Shyam Poludasu, MD, Ajay Agarwal, MD, Pompeiu Vladutiu, MD,
Original Contribution Bolus-Only Platelet Glycoprotein IIb/IIIa Inhibition During Percutaneous Coronary Intervention Jonathan D. Marmur, MD, Shyam Poludasu, MD, Ajay Agarwal, MD, Pompeiu Vladutiu, MD,
Horizon Scanning Centre November 2012
 Horizon Scanning Centre November 2012 Cangrelor to reduce platelet aggregation and thrombosis in patients undergoing percutaneous coronary intervention99 SUMMARY NIHR HSC ID: 2424 This briefing is based
Horizon Scanning Centre November 2012 Cangrelor to reduce platelet aggregation and thrombosis in patients undergoing percutaneous coronary intervention99 SUMMARY NIHR HSC ID: 2424 This briefing is based
International Journal of Collaborative Research on Internal Medicine & Public Health (IJCRIMPH)
 Same-day discharge after percutaneous coronary intervention in light of the society for cardiovascular angiography and intervention's proposed guidelines: A single-center experience Yazan Khouri, Sachin
Same-day discharge after percutaneous coronary intervention in light of the society for cardiovascular angiography and intervention's proposed guidelines: A single-center experience Yazan Khouri, Sachin
NOVEL ANTI-THROMBOTIC THERAPIES FOR ACUTE CORONARY SYNDROME: DIRECT THROMBIN INHIBITORS
 Judd E. Hollander, MD Professor, Clinical Research Director, Department of Emergency Medicine University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania OBJECTIVES: 1. Discuss the concept
Judd E. Hollander, MD Professor, Clinical Research Director, Department of Emergency Medicine University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania OBJECTIVES: 1. Discuss the concept
Clinical Outcomes After Detection of Elevated Cardiac Enzymes in Patients Undergoing Percutaneous Intervention
 88 JACC Vol. 33, No. 1 Clinical Outcomes After Detection of Elevated Cardiac Enzymes in Patients Undergoing Percutaneous Intervention BARBARA E. TARDIFF, MD, ROBERT M. CALIFF, MD, FACC, JAMES E. TCHENG,
88 JACC Vol. 33, No. 1 Clinical Outcomes After Detection of Elevated Cardiac Enzymes in Patients Undergoing Percutaneous Intervention BARBARA E. TARDIFF, MD, ROBERT M. CALIFF, MD, FACC, JAMES E. TCHENG,
Otamixaban for non-st-segment elevation acute coronary syndrome
 Otamixaban for non-st-segment elevation acute coronary syndrome September 2011 This technology summary is based on information available at the time of research and a limited literature search. It is not
Otamixaban for non-st-segment elevation acute coronary syndrome September 2011 This technology summary is based on information available at the time of research and a limited literature search. It is not
Heart Online First, published on December 30, 2005 as /hrt
 Heart Online First, published on December 30, 2005 as 10.1136/hrt.2005.078758 Attributable risk of clinical endpoints Clinical endpoint definitions following percutaneous coronary intervention and their
Heart Online First, published on December 30, 2005 as 10.1136/hrt.2005.078758 Attributable risk of clinical endpoints Clinical endpoint definitions following percutaneous coronary intervention and their
Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy
 Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy Franz-Josef Neumann Herz-Zentrum Bad Krozingen Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy
Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy Franz-Josef Neumann Herz-Zentrum Bad Krozingen Antiplatelet and Antithrombotic Therapies in PCI Defining the Optimal Strategy
Cost-efficacy in interventional cardiology
 European Heart Journal (21) 22, 1476 1484 doi:1.153/euhj.2.43, available online at http://www.idealibrary.com on Cost-efficacy in interventional cardiology Results from the EPISTENT study J. E. F. Zwart-van
European Heart Journal (21) 22, 1476 1484 doi:1.153/euhj.2.43, available online at http://www.idealibrary.com on Cost-efficacy in interventional cardiology Results from the EPISTENT study J. E. F. Zwart-van
Prevention of Coronary Stent Thrombosis and Restenosis
 Prevention of Coronary Stent Thrombosis and Restenosis Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine, Seoul, Korea 9/12/03 Coronary
Prevention of Coronary Stent Thrombosis and Restenosis Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine, Seoul, Korea 9/12/03 Coronary
ACUTE CORONARY SYNDROMES
 CLINICAL CARDIOLOGY Current Role of Platelet Glycoprotein IIb/IIIa Inhibitors in Acute Coronary Syndromes Deepak L. Bhatt, MD Eric J. Topol, MD ACUTE CORONARY SYNDROMES (ACSs) span the pathological continuum
CLINICAL CARDIOLOGY Current Role of Platelet Glycoprotein IIb/IIIa Inhibitors in Acute Coronary Syndromes Deepak L. Bhatt, MD Eric J. Topol, MD ACUTE CORONARY SYNDROMES (ACSs) span the pathological continuum
A Pooled Analysis of Five Randomized Clinical Trials
 Lack of Benefit From Intravenous latelet Glycoprotein IIb/IIIa Receptor Inhibition as Adjunctive Treatment for ercutaneous Interventions of Aortocoronary Bypass Grafts A ooled Analysis of Five Randomized
Lack of Benefit From Intravenous latelet Glycoprotein IIb/IIIa Receptor Inhibition as Adjunctive Treatment for ercutaneous Interventions of Aortocoronary Bypass Grafts A ooled Analysis of Five Randomized
Scottish Medicines Consortium
 Scottish Medicines Consortium bivalirudin, 250mg powder for concentrate for solution for injection or infusion (Angiox ) No. (516/08) The Medicines Company UK Ltd 07 November 2008 The Scottish Medicines
Scottish Medicines Consortium bivalirudin, 250mg powder for concentrate for solution for injection or infusion (Angiox ) No. (516/08) The Medicines Company UK Ltd 07 November 2008 The Scottish Medicines
Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network
 Torino 6 Joint meeting with Mayo Clinic Great Innovation in Cardiology 14-15 Ottobre 2010 Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network Diego Ardissino Ischemic vs
Torino 6 Joint meeting with Mayo Clinic Great Innovation in Cardiology 14-15 Ottobre 2010 Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network Diego Ardissino Ischemic vs
bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company
 bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company 06 August 2010 The Scottish Medicines Consortium (SMC) has completed its
bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company 06 August 2010 The Scottish Medicines Consortium (SMC) has completed its
Direct Thrombin Inhibitors for PCI Pharmacology: Role of Bivalirudin in High-Risk PCI
 Direct Thrombin Inhibitors for PCI Pharmacology: Role of Bivalirudin in High-Risk PCI Charles A. Simonton MD, FACC, FSCAI Sanger Clinic Medical Director Clinical Innovation and Research Carolinas Heart
Direct Thrombin Inhibitors for PCI Pharmacology: Role of Bivalirudin in High-Risk PCI Charles A. Simonton MD, FACC, FSCAI Sanger Clinic Medical Director Clinical Innovation and Research Carolinas Heart
Guideline for STEMI. Reperfusion at a PCI-Capable Hospital
 MANSOURA. 2015 Guideline for STEMI Reperfusion at a PCI-Capable Hospital Mahmoud Yossof MANSOURA 2015 Reperfusion Therapy for Patients with STEMI *Patients with cardiogenic shock or severe heart failure
MANSOURA. 2015 Guideline for STEMI Reperfusion at a PCI-Capable Hospital Mahmoud Yossof MANSOURA 2015 Reperfusion Therapy for Patients with STEMI *Patients with cardiogenic shock or severe heart failure
Role of platelet glycoprotein IIb/IIIa inhibitors in rescue percutaneous coronary interventions
 Role of platelet glycoprotein IIb/IIIa inhibitors in rescue percutaneous coronary interventions Anna Sonia Petronio, Marco De Carlo, Roberta Rossini, Giovanni Amoroso, Ugo Limbruno, Nicola Ciabatti, Caterina
Role of platelet glycoprotein IIb/IIIa inhibitors in rescue percutaneous coronary interventions Anna Sonia Petronio, Marco De Carlo, Roberta Rossini, Giovanni Amoroso, Ugo Limbruno, Nicola Ciabatti, Caterina
Contemporary Percutaneous Coronary Intervention Versus Balloon Angioplasty for Multivessel Coronary Artery Disease
 Contemporary Percutaneous Coronary Intervention Versus Balloon Angioplasty for Multivessel Coronary Artery Disease A Comparison of the National Heart, Lung and Blood Institute Dynamic Registry and the
Contemporary Percutaneous Coronary Intervention Versus Balloon Angioplasty for Multivessel Coronary Artery Disease A Comparison of the National Heart, Lung and Blood Institute Dynamic Registry and the
Update on Antithrombotic Therapy in Acute Coronary Syndrome
 Update on Antithrombotic Therapy in Acute Coronary Syndrome Laura Tsang November 13, 2006 Objectives: By the end of this session, you should understand: The role of antithrombotics in ACS Their mechanisms
Update on Antithrombotic Therapy in Acute Coronary Syndrome Laura Tsang November 13, 2006 Objectives: By the end of this session, you should understand: The role of antithrombotics in ACS Their mechanisms
Clopidogrel has been evaluated in clinical trials that included cardiovascular patients
 REVIEW ARTICLE Comparative Benefits of Clopidogrel and Aspirin in High-Risk Patient Populations Lessons From the CAPRIE and CURE Studies Jack Hirsh, CM, MD, FRCPC, FRACP, FRSC, DSc; Deepak L. Bhatt, MD,
REVIEW ARTICLE Comparative Benefits of Clopidogrel and Aspirin in High-Risk Patient Populations Lessons From the CAPRIE and CURE Studies Jack Hirsh, CM, MD, FRCPC, FRACP, FRSC, DSc; Deepak L. Bhatt, MD,
Point-of-Care Measured Platelet Inhibition Correlates With a Reduced Risk of an Adverse Cardiac Event After Percutaneous Coronary Intervention
 Point-of-Care Measured Platelet Inhibition Correlates With a Reduced Risk of an Adverse Cardiac Event After Percutaneous Coronary Intervention Results of the GOLD (AU-Assessing Ultegra) Multicenter Study
Point-of-Care Measured Platelet Inhibition Correlates With a Reduced Risk of an Adverse Cardiac Event After Percutaneous Coronary Intervention Results of the GOLD (AU-Assessing Ultegra) Multicenter Study
JAMA. 2004;292:
 ORIGINAL CONTRIBUTION Long-term Efficacy of and Provisional Blockade vs Heparin and Planned Blockade During Percutaneous Coronary Revascularization REPLACE-2 Randomized Trial A. Michael Lincoff, MD Neal
ORIGINAL CONTRIBUTION Long-term Efficacy of and Provisional Blockade vs Heparin and Planned Blockade During Percutaneous Coronary Revascularization REPLACE-2 Randomized Trial A. Michael Lincoff, MD Neal
Learning Objectives. Epidemiology of Acute Coronary Syndrome
 Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS
 New Horizons In Atherothrombosis Treatment 2012 순환기춘계학술대회 FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS Division of Cardiology, Jeonbuk National University Medical School Jei Keon Chae,
New Horizons In Atherothrombosis Treatment 2012 순환기춘계학술대회 FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS Division of Cardiology, Jeonbuk National University Medical School Jei Keon Chae,
Chapter 15 Glycoprotein IIb/IIIa Antagonists
 Chapter 15 Glycoprotein IIb/IIIa Antagonists Introduction - GP IIb/IIIa receptors - Pharmacologic approaches - Preparations and dosages Mechanism of Action Clinical Efficacy - UA / NSTEMI - STEMI - PCI
Chapter 15 Glycoprotein IIb/IIIa Antagonists Introduction - GP IIb/IIIa receptors - Pharmacologic approaches - Preparations and dosages Mechanism of Action Clinical Efficacy - UA / NSTEMI - STEMI - PCI
Facilitated Percutaneous Coronary Intervention in Acute Myocardial Infarction. Is it beneficial to patients?
 Facilitated Percutaneous Coronary Intervention in Acute Myocardial Infarction Is it beneficial to patients? Seung-Jea Tahk, MD. PhD. Suwon, Korea Facilitated PCI.. background Degree of coronary flow at
Facilitated Percutaneous Coronary Intervention in Acute Myocardial Infarction Is it beneficial to patients? Seung-Jea Tahk, MD. PhD. Suwon, Korea Facilitated PCI.. background Degree of coronary flow at
Columbia University Medical Center Cardiovascular Research Foundation
 STEMI and NSTEMI Pharmacology Confusion: How to Choose and Use Antithrombins (Unfractionated and Low Molecular Heparins, Bivalirudin, Fondaparinux) and Antiplatelet Agents (Aspirin, Clopidogrel and Prasugrel)
STEMI and NSTEMI Pharmacology Confusion: How to Choose and Use Antithrombins (Unfractionated and Low Molecular Heparins, Bivalirudin, Fondaparinux) and Antiplatelet Agents (Aspirin, Clopidogrel and Prasugrel)
Medicine Dr. Omed Lecture 2 Stable and Unstable Angina
 Medicine Dr. Omed Lecture 2 Stable and Unstable Angina Risk stratification in stable angina. High Risk; *post infarct angina, *poor effort tolerance, *ischemia at low workload, *left main or three vessel
Medicine Dr. Omed Lecture 2 Stable and Unstable Angina Risk stratification in stable angina. High Risk; *post infarct angina, *poor effort tolerance, *ischemia at low workload, *left main or three vessel
Journal of the American College of Cardiology Vol. 38, No. 3, by the American College of Cardiology ISSN /01/$20.
 Journal of the American College of Cardiology Vol. 38, No. 3, 2001 2001 by the American College of Cardiology ISSN 0735-1097/01/$20.00 Published by Elsevier Science Inc. PII S0735-1097(01)01423-1 Effect
Journal of the American College of Cardiology Vol. 38, No. 3, 2001 2001 by the American College of Cardiology ISSN 0735-1097/01/$20.00 Published by Elsevier Science Inc. PII S0735-1097(01)01423-1 Effect
Acute coronary syndromes refers to a spectrum of
 Appropriate Use of Glycoprotein IIb/IIIa Blockade for Unstable Angina and Non ST Segment Elevation Myocardial Infarction Albert W. Chan, MD, MS, FRCP(C), and Sorin J. Brener, MD Objective: To review the
Appropriate Use of Glycoprotein IIb/IIIa Blockade for Unstable Angina and Non ST Segment Elevation Myocardial Infarction Albert W. Chan, MD, MS, FRCP(C), and Sorin J. Brener, MD Objective: To review the
Link between effectiveness and cost data Costing was conducted prospectively on the same patient sample as that used in the effectiveness analysis.
 Heparin after percutaneous intervention (HAPI): a prospective multicenter randomized trial of three heparin regimens after successful coronary intervention Rabah M, Mason D, Muller D W, Hundley R, Kugelmass
Heparin after percutaneous intervention (HAPI): a prospective multicenter randomized trial of three heparin regimens after successful coronary intervention Rabah M, Mason D, Muller D W, Hundley R, Kugelmass
Acute Coronary Syndromes
 Overview Acute Coronary Syndromes Rabeea Aboufakher, MD, FACC, FSCAI Section Chief of Cardiology Altru Health System Grand Forks, ND Epidemiology Pathophysiology Clinical features and diagnosis STEMI management
Overview Acute Coronary Syndromes Rabeea Aboufakher, MD, FACC, FSCAI Section Chief of Cardiology Altru Health System Grand Forks, ND Epidemiology Pathophysiology Clinical features and diagnosis STEMI management
Impact of Gender on In-Hospital Percutaneous Coronary Interventional Outcomes in African-Americans
 Original Contribution Impact of Gender on In-Hospital Percutaneous Coronary Interventional Outcomes in African-Americans Shyam Poludasu, MD, Erdal Cavusoglu, MD, Luther T. Clark, MD, Jonathan D. Marmur,
Original Contribution Impact of Gender on In-Hospital Percutaneous Coronary Interventional Outcomes in African-Americans Shyam Poludasu, MD, Erdal Cavusoglu, MD, Luther T. Clark, MD, Jonathan D. Marmur,
Glycoprotein (GP) IIb/IIIa antagonists are potent inhibitors
 Current Perspective Platelet Glycoprotein IIb/IIIa Inhibitors Recognition of a Two-Edged Sword? Martin J. Quinn, MD, PhD; Edward F. Plow, PhD; Eric J. Topol, MD Glycoprotein (GP) IIb/IIIa antagonists are
Current Perspective Platelet Glycoprotein IIb/IIIa Inhibitors Recognition of a Two-Edged Sword? Martin J. Quinn, MD, PhD; Edward F. Plow, PhD; Eric J. Topol, MD Glycoprotein (GP) IIb/IIIa antagonists are
Incremental Prognostic Value of Elevated Baseline C-Reactive Protein Among Established Markers of Risk in Percutaneous Coronary Intervention
 Incremental Prognostic Value of Elevated Baseline C-Reactive Protein Among Established Markers of Risk in Percutaneous Coronary Intervention Derek P. Chew, MBBS; Deepak L. Bhatt, MD; Mark A. Robbins, MD;
Incremental Prognostic Value of Elevated Baseline C-Reactive Protein Among Established Markers of Risk in Percutaneous Coronary Intervention Derek P. Chew, MBBS; Deepak L. Bhatt, MD; Mark A. Robbins, MD;
Impact of Clinical Syndrome Acuity on the Differential Response to 2 Glycoprotein IIb/IIIa Inhibitors in Patients Undergoing Coronary Stenting
 Impact of Clinical Syndrome Acuity on the Differential Response to 2 Glycoprotein IIb/IIIa Inhibitors in Patients Undergoing Coronary Stenting The TARGET Trial* Gregg W. Stone, MD; David J. Moliterno,
Impact of Clinical Syndrome Acuity on the Differential Response to 2 Glycoprotein IIb/IIIa Inhibitors in Patients Undergoing Coronary Stenting The TARGET Trial* Gregg W. Stone, MD; David J. Moliterno,
Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland
 Advances in Antiplatelet Therapy in PCI and ACS Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland Targets for Platelet
Advances in Antiplatelet Therapy in PCI and ACS Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland Targets for Platelet
Role of Clopidogrel in Acute Coronary Syndromes. Hossam Kandil,, MD. Professor of Cardiology Cairo University
 Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Journal of the American College of Cardiology Vol. 48, No. 11, by the American College of Cardiology Foundation ISSN /06/$32.
 Journal of the American College of Cardiology Vol. 48, No. 11, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.12.084
Journal of the American College of Cardiology Vol. 48, No. 11, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.12.084
Manual of Interventional Cardiology
 782 PLATELET GLYCOPROTEIN IIb/IIIa RECEPTOR ANTAGONISTS (Tables 34.15, 34.16) Activation of the platelet glycoprotein (GP) IIb/IIIa receptor complex constitutes the final common pathway for platelet aggregation,
782 PLATELET GLYCOPROTEIN IIb/IIIa RECEPTOR ANTAGONISTS (Tables 34.15, 34.16) Activation of the platelet glycoprotein (GP) IIb/IIIa receptor complex constitutes the final common pathway for platelet aggregation,
An update on the management of UA / NSTEMI. Michael H. Crawford, MD
 An update on the management of UA / NSTEMI Michael H. Crawford, MD New ACC/AHA Guidelines 2007 What s s new in the last 5 years CT imaging advances Ascendancy of troponin and BNP Clarification of ACEI/ARB
An update on the management of UA / NSTEMI Michael H. Crawford, MD New ACC/AHA Guidelines 2007 What s s new in the last 5 years CT imaging advances Ascendancy of troponin and BNP Clarification of ACEI/ARB
Intra-Procedural Stent Thrombosis
 JACC: CARDIOVASCULAR INTERVENTIONS VOL. 6, NO. 1, 2013 2013 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/$36.00 PUBLISHED BY ELSEVIER INC. http://dx.doi.org/10.1016/j.jcin.2012.08.018
JACC: CARDIOVASCULAR INTERVENTIONS VOL. 6, NO. 1, 2013 2013 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/$36.00 PUBLISHED BY ELSEVIER INC. http://dx.doi.org/10.1016/j.jcin.2012.08.018
Percutaneous transluminal coronary
 Heart 2000;83:481 490 CORONARY DISEASE Intervention in coronary artery disease Stephan Windecker, Bernhard Meier Swiss Cardiovascular Center Bern, University Hospital, Bern, Switzerland Correspondence
Heart 2000;83:481 490 CORONARY DISEASE Intervention in coronary artery disease Stephan Windecker, Bernhard Meier Swiss Cardiovascular Center Bern, University Hospital, Bern, Switzerland Correspondence
Clinical Investigation and Reports
 Clinical Investigation and Reports Relationship of Activated Partial Thromboplastin Time to Coronary Events and Bleeding in Patients With Acute Coronary Syndromes Who Receive Heparin Sonia S. Anand, MD,
Clinical Investigation and Reports Relationship of Activated Partial Thromboplastin Time to Coronary Events and Bleeding in Patients With Acute Coronary Syndromes Who Receive Heparin Sonia S. Anand, MD,
METHODS OBJECTIVES BACKGROUND METHODS
 Journal of the American College of Cardiology Vol. 36, No. 3, 2000 2000 by the American College of Cardiology ISSN 0735-1097/00/$20.00 Published by Elsevier Science Inc. PII S0735-1097(00)00814-7 Platelet
Journal of the American College of Cardiology Vol. 36, No. 3, 2000 2000 by the American College of Cardiology ISSN 0735-1097/00/$20.00 Published by Elsevier Science Inc. PII S0735-1097(00)00814-7 Platelet
Journal of the American College of Cardiology Vol. 36, No. 5, by the American College of Cardiology ISSN /00/$20.
 Journal of the American College of Cardiology Vol. 36, No. 5, 2000 2000 by the American College of Cardiology ISSN 0735-1097/00/$20.00 Published by Elsevier Science Inc. PII S0735-1097(00)00923-2 Facilitation
Journal of the American College of Cardiology Vol. 36, No. 5, 2000 2000 by the American College of Cardiology ISSN 0735-1097/00/$20.00 Published by Elsevier Science Inc. PII S0735-1097(00)00923-2 Facilitation
The Evolving Role of Glycoprotein IIb/IIIa Inhibitors in the Setting of Percutaneous Coronary Intervention
 JACC: CARDIOVASCULAR INTERVENTIONS VOL. 3, NO. 12, 2010 2010 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/$36.00 PUBLISHED BY ELSEVIER INC. DOI: 10.1016/j.jcin.2010.09.015 STATE-OF-THE-ART
JACC: CARDIOVASCULAR INTERVENTIONS VOL. 3, NO. 12, 2010 2010 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/$36.00 PUBLISHED BY ELSEVIER INC. DOI: 10.1016/j.jcin.2010.09.015 STATE-OF-THE-ART
Acute coronary syndromes
 Acute coronary syndromes 1 Acute coronary syndromes Acute coronary syndromes results primarily from diminished myocardial blood flow secondary to an occlusive or partially occlusive coronary artery thrombus.
Acute coronary syndromes 1 Acute coronary syndromes Acute coronary syndromes results primarily from diminished myocardial blood flow secondary to an occlusive or partially occlusive coronary artery thrombus.
Oral Antiplatelet Therapy in PCI/ACS. Dominick J. Angiolillo, MD, PhD, FACC, FESC Director of Cardiovascular Research Assistant Professor of Medicine
 Oral Antiplatelet Therapy in PCI/ACS Dominick J. Angiolillo, MD, PhD, FACC, FESC Director of Cardiovascular Research Assistant Professor of Medicine Basic Concepts Thrombus Formation Two key elements:
Oral Antiplatelet Therapy in PCI/ACS Dominick J. Angiolillo, MD, PhD, FACC, FESC Director of Cardiovascular Research Assistant Professor of Medicine Basic Concepts Thrombus Formation Two key elements:
Preoperative Management of Patients Receiving Antithrombotics
 Preoperative Management of Patients Receiving Antithrombotics Bleeding complications remain an important concern for most surgical procedures. Attempts to minimize the risk of these complications by removing
Preoperative Management of Patients Receiving Antithrombotics Bleeding complications remain an important concern for most surgical procedures. Attempts to minimize the risk of these complications by removing
Index. Note: Page numbers of article titles are in boldface type.
 Cardiol Clin 24 (2006) 299 304 Index Note: Page numbers of article titles are in boldface type. A Abciximab in PCI, 180 182 ACE trial, 182 Actinomycin D-eluting stent, 224 ACUITY trial, 190 Acute myocardial
Cardiol Clin 24 (2006) 299 304 Index Note: Page numbers of article titles are in boldface type. A Abciximab in PCI, 180 182 ACE trial, 182 Actinomycin D-eluting stent, 224 ACUITY trial, 190 Acute myocardial
During the past 15 years, we have learned an enormous amount about the pathogenesis
 REVIEW ARTICLE Unstable Angina Current Concepts of Pathogenesis and Treatment John A. Ambrose, MD; George Dangas, MD During the past 15 years, we have learned an enormous amount about the pathogenesis
REVIEW ARTICLE Unstable Angina Current Concepts of Pathogenesis and Treatment John A. Ambrose, MD; George Dangas, MD During the past 15 years, we have learned an enormous amount about the pathogenesis
Clinical Seminar. Which Diabetic Patient is a Candidate for Percutaneous Coronary Intervention - European Perspective
 Clinical Seminar Which Diabetic Patient is a Candidate for Percutaneous Coronary Intervention - European Perspective Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical
Clinical Seminar Which Diabetic Patient is a Candidate for Percutaneous Coronary Intervention - European Perspective Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical
ATHEROSCLEROTIC HEART DISease
 CLINICAL CARDIOLOGY Platelet Glycoprotein IIb/IIIa Receptor Antagonists in Cardiovascular Disease David A. Vorchheimer, MD Juan Jose Badimon, PhD Valentin Fuster, MD, PhD ATHEROSCLEROTIC HEART DISease
CLINICAL CARDIOLOGY Platelet Glycoprotein IIb/IIIa Receptor Antagonists in Cardiovascular Disease David A. Vorchheimer, MD Juan Jose Badimon, PhD Valentin Fuster, MD, PhD ATHEROSCLEROTIC HEART DISease
Acute ischemic coronary syndromes (AICS)
 ...CONTINUING MEDICAL EDUCATION... CME ARTICLE Overview of Platelet Glycoprotein IIb/IIIa Receptor Antagonist Treatment in the Management of Coronary Interventions and Acute Ischemic Coronary Syndromes
...CONTINUING MEDICAL EDUCATION... CME ARTICLE Overview of Platelet Glycoprotein IIb/IIIa Receptor Antagonist Treatment in the Management of Coronary Interventions and Acute Ischemic Coronary Syndromes
Abciximab provides cost-effective survival advantage in high-volume interventional practice
 Interventional Cardiology Abciximab provides cost-effective survival advantage in high-volume interventional practice Dean J. Kereiakes, M.D. 1, Robert L. Obenchain, Ph.D. 2, Beth L. Barber, Ph.D. 2, Andrew
Interventional Cardiology Abciximab provides cost-effective survival advantage in high-volume interventional practice Dean J. Kereiakes, M.D. 1, Robert L. Obenchain, Ph.D. 2, Beth L. Barber, Ph.D. 2, Andrew
Journal of the American College of Cardiology Vol. 37, No. 4, by the American College of Cardiology ISSN /01/$20.
 Journal of the American College of Cardiology Vol. 37, No. 4, 2001 2001 by the American College of Cardiology ISSN 0735-1097/01/$20.00 Published by Elsevier Science Inc. PII S0735-1097(01)01143-3 Acute
Journal of the American College of Cardiology Vol. 37, No. 4, 2001 2001 by the American College of Cardiology ISSN 0735-1097/01/$20.00 Published by Elsevier Science Inc. PII S0735-1097(01)01143-3 Acute
A Unique, Low Dose of Intravenous Enoxaparin in Elective Percutaneous Coronary Intervention
 Journal of the American College of Cardiology Vol. 40, No. 11, 2002 2002 by the American College of Cardiology Foundation ISSN 0735-1097/02/$22.00 Published by Elsevier Science Inc. PII S0735-1097(02)02531-7
Journal of the American College of Cardiology Vol. 40, No. 11, 2002 2002 by the American College of Cardiology Foundation ISSN 0735-1097/02/$22.00 Published by Elsevier Science Inc. PII S0735-1097(02)02531-7
Journal of the American College of Cardiology Vol. 34, No. 7, by the American College of Cardiology ISSN /99/$20.
 Journal of the American College of Cardiology Vol. 34, No. 7, 1999 1999 by the American College of Cardiology ISSN 0735-1097/99/$20.00 Published by Elsevier Science Inc. PII S0735-1097(99)00443-X CLINICAL
Journal of the American College of Cardiology Vol. 34, No. 7, 1999 1999 by the American College of Cardiology ISSN 0735-1097/99/$20.00 Published by Elsevier Science Inc. PII S0735-1097(99)00443-X CLINICAL
Timing of Anti-Platelet Therapy for ACS (EARLY-ACS & ACUITY) Mitchell W. Krucoff, MD, FACC
 Timing of Anti-Platelet Therapy for ACS (EARLY-ACS & ACUITY) Mitchell W. Krucoff, MD, FACC Professor, Medicine/Cardiology Duke University Medical Center Director, Cardiovascular Devices Unit Duke Clinical
Timing of Anti-Platelet Therapy for ACS (EARLY-ACS & ACUITY) Mitchell W. Krucoff, MD, FACC Professor, Medicine/Cardiology Duke University Medical Center Director, Cardiovascular Devices Unit Duke Clinical
COAGULATION, BLEEDING, AND TRANSFUSION IN URGENT AND EMERGENCY CORONARY SURGERY
 COAGULATION, BLEEDING, AND TRANSFUSION IN URGENT AND EMERGENCY CORONARY SURGERY VALTER CASATI, M.D. DIVISION OF CARDIOVASCULAR ANESTHESIA AND INTENSIVE CARE CLINICA S. GAUDENZIO NOVARA (ITALY) ANTIPLATELET
COAGULATION, BLEEDING, AND TRANSFUSION IN URGENT AND EMERGENCY CORONARY SURGERY VALTER CASATI, M.D. DIVISION OF CARDIOVASCULAR ANESTHESIA AND INTENSIVE CARE CLINICA S. GAUDENZIO NOVARA (ITALY) ANTIPLATELET
Acute myocardial infarction. Cardiovascular disorders. main/0202_new 02/03/06. Search date August 2004 Nicholas Danchin and Eric Durand
 main/0202_new 02/03/06 Acute myocardial infarction Search date August 2004 Nicholas Danchin and Eric Durand QUESTIONS Which treatments improve outcomes in acute myocardial infarction?...4 Which treatments
main/0202_new 02/03/06 Acute myocardial infarction Search date August 2004 Nicholas Danchin and Eric Durand QUESTIONS Which treatments improve outcomes in acute myocardial infarction?...4 Which treatments
Mortality Risk Conferred by Small Elevations of Creatine Kinase-MB Isoenzyme After Percutaneous Coronary Intervention
 Journal of the American College of Cardiology Vol. 42, No. 8, 2003 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Inc. doi:10.1016/s0735-1097(03)01044-1
Journal of the American College of Cardiology Vol. 42, No. 8, 2003 2003 by the American College of Cardiology Foundation ISSN 0735-1097/03/$30.00 Published by Elsevier Inc. doi:10.1016/s0735-1097(03)01044-1
Timing of Surgery After Percutaneous Coronary Intervention
 Timing of Surgery After Percutaneous Coronary Intervention Deepak Talreja, MD, FACC Bayview/EVMS/Sentara Outline/Highlights Timing of elective surgery What to do with medications Stopping anti-platelet
Timing of Surgery After Percutaneous Coronary Intervention Deepak Talreja, MD, FACC Bayview/EVMS/Sentara Outline/Highlights Timing of elective surgery What to do with medications Stopping anti-platelet
CLINICIAN INTERVIEW RECOGNIZING ACS AND STRATIFYING RISK IN PRIMARY CARE. An interview with A. Michael Lincoff, MD, and Eric R. Bates, MD, FACC, FAHA
 RECOGNIZING ACS AND STRATIFYING RISK IN PRIMARY CARE An interview with A. Michael Lincoff, MD, and Eric R. Bates, MD, FACC, FAHA Dr Lincoff is an interventional cardiologist and the Vice Chairman for Research
RECOGNIZING ACS AND STRATIFYING RISK IN PRIMARY CARE An interview with A. Michael Lincoff, MD, and Eric R. Bates, MD, FACC, FAHA Dr Lincoff is an interventional cardiologist and the Vice Chairman for Research
Acute coronary syndrome (ACS) is an
 OVERVIEW OF MEDICAL MANAGEMENT OF ACUTE CORONARY SYNDROMES Robert B. Parker, PharmD * Acute coronary syndrome (ACS) is an umbrella term used to describe any group of symptoms of acute myocardial ischemia
OVERVIEW OF MEDICAL MANAGEMENT OF ACUTE CORONARY SYNDROMES Robert B. Parker, PharmD * Acute coronary syndrome (ACS) is an umbrella term used to describe any group of symptoms of acute myocardial ischemia
2007 ACC/AHA GUIDELINES FOR THE MANAGEMENT OF NSTE-ACS: OPTIMAL ANTICOAGULATION AND ANTIPLATELET THERAPY
 2007 ACC/AHA GUIDELINES FOR THE MANAGEMENT OF NSTE-ACS: OPTIMAL ANTICOAGULATION AND ANTIPLATELET THERAPY Charles V. Pollack, Jr., MA, MD, FACEP, FAAEM, FAHA Professor and Chairman, Department of Emergency
2007 ACC/AHA GUIDELINES FOR THE MANAGEMENT OF NSTE-ACS: OPTIMAL ANTICOAGULATION AND ANTIPLATELET THERAPY Charles V. Pollack, Jr., MA, MD, FACEP, FAAEM, FAHA Professor and Chairman, Department of Emergency
Modeling and Risk Prediction in the Current Era of Interventional Cardiology
 Modeling and Risk Prediction in the Current Era of Interventional Cardiology A Report From the National Heart, Lung, and Blood Institute Dynamic Registry David R. Holmes, MD; Faith Selzer, PhD; Janet M.
Modeling and Risk Prediction in the Current Era of Interventional Cardiology A Report From the National Heart, Lung, and Blood Institute Dynamic Registry David R. Holmes, MD; Faith Selzer, PhD; Janet M.
Mortality from ST-segment elevated acute
 Choosing Pharmacotherapy for Primary PCI in Acute Myocardial Infarction A review of the agents and strategies commonly used during percutaneous intervention for ST-segment elevated myocardial infarction.
Choosing Pharmacotherapy for Primary PCI in Acute Myocardial Infarction A review of the agents and strategies commonly used during percutaneous intervention for ST-segment elevated myocardial infarction.
Adults With Diagnosed Diabetes
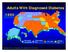 Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
Continuing Medical Education Post-Test
 Continuing Medical Education Post-Test Based on the information presented in this monograph, please choose one correct response for each of the following questions or statements. Record your answers on
Continuing Medical Education Post-Test Based on the information presented in this monograph, please choose one correct response for each of the following questions or statements. Record your answers on
Pathophysiology of ACS
 Pathophysiology of ACS ~ 2.0 MM patients admitted to CCU or telemetry annually 0.6 MM ST-segment elevation MI 1.4 MM Non-ST-segment elevation ACS NSTEMI vs STEMI VANQWISH Boden et al N Engl J Med 1998;338:1785-1792
Pathophysiology of ACS ~ 2.0 MM patients admitted to CCU or telemetry annually 0.6 MM ST-segment elevation MI 1.4 MM Non-ST-segment elevation ACS NSTEMI vs STEMI VANQWISH Boden et al N Engl J Med 1998;338:1785-1792
The restoration of coronary flow after an
 Pharmacological Reperfusion in Acute Myicardial Infarction after ASSENT 3 and GUSTO V [81] DANIEL FERREIRA, MD, FESC Serviço de Cardiologia, Hospital Fernando Fonseca, Amadora, Portugal Rev Port Cardiol
Pharmacological Reperfusion in Acute Myicardial Infarction after ASSENT 3 and GUSTO V [81] DANIEL FERREIRA, MD, FESC Serviço de Cardiologia, Hospital Fernando Fonseca, Amadora, Portugal Rev Port Cardiol
The CIAO (Coronary Interventions Antiplatelet-based Only) Study
 Journal of the American College of Cardiology Vol. 52, No. 16, 2008 2008 by the American College of Cardiology Foundation ISSN 0735-1097/08/$34.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2008.07.026
Journal of the American College of Cardiology Vol. 52, No. 16, 2008 2008 by the American College of Cardiology Foundation ISSN 0735-1097/08/$34.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2008.07.026
Current Advances and Best Practices in Acute STEMI Management A pharmacoinvasive approach
 Current Advances and Best Practices in Acute STEMI Management A pharmacoinvasive approach Frans Van de Werf, MD, PhD University Hospitals, Leuven, Belgium Frans Van de Werf: Disclosures Research grants
Current Advances and Best Practices in Acute STEMI Management A pharmacoinvasive approach Frans Van de Werf, MD, PhD University Hospitals, Leuven, Belgium Frans Van de Werf: Disclosures Research grants
Methods The PARAGON-B trial
 Misreporting of myocardial infarction end points: Results of adjudication by a central clinical events committee in the PARAGON-B trial Kenneth W. Mahaffey, MD, a Matthew T. Roe, MD, a Christopher K. Dyke,
Misreporting of myocardial infarction end points: Results of adjudication by a central clinical events committee in the PARAGON-B trial Kenneth W. Mahaffey, MD, a Matthew T. Roe, MD, a Christopher K. Dyke,
Is the role of bivalirudin established?
 Is the role of bivalirudin established? Rob Henderson Consultant Cardiologist Trent Cardiac Centre Nottingham University Hospitals Conflicts of Interest: None Declarations: Member NICE Unstable Angina
Is the role of bivalirudin established? Rob Henderson Consultant Cardiologist Trent Cardiac Centre Nottingham University Hospitals Conflicts of Interest: None Declarations: Member NICE Unstable Angina
High-Dose Tirofiban Administered as Bolus-Only during Percutaneous Coronary Intervention
 Original Contribution High-Dose Tirofiban Administered as Bolus-Only during Percutaneous Coronary Intervention Jonathan D. Marmur, MD, Shyam Poludasu, MD, Ajay Agarwal, MD, Nagarathna Manjappa MD, Erdal
Original Contribution High-Dose Tirofiban Administered as Bolus-Only during Percutaneous Coronary Intervention Jonathan D. Marmur, MD, Shyam Poludasu, MD, Ajay Agarwal, MD, Nagarathna Manjappa MD, Erdal
The Effects of Aspirin and Clopidogrel Response on Myonecrosis After Percutaneous Coronary Intervention
 JACC: CARDIOVASCULAR INTERVENTIONS VOL. 1, NO. 6, 2008 2008 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/08/$34.00 PUBLISHED BY ELSEVIER INC. DOI: 10.1016/j.jcin.2008.08.017 The Effects
JACC: CARDIOVASCULAR INTERVENTIONS VOL. 1, NO. 6, 2008 2008 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/08/$34.00 PUBLISHED BY ELSEVIER INC. DOI: 10.1016/j.jcin.2008.08.017 The Effects
Treatment of Saphenous Vein Bypass Grafts With Ultrasound Thrombolysis. A Randomized Study (ATLAS)
 Treatment of Saphenous Vein Bypass Grafts With Ultrasound Thrombolysis A Randomized Study (ATLAS) Mandeep Singh, MD; Uri Rosenschein, MD; Kalon K.L. Ho, MD; Peter B. Berger, MD; Richard Kuntz, MD; David
Treatment of Saphenous Vein Bypass Grafts With Ultrasound Thrombolysis A Randomized Study (ATLAS) Mandeep Singh, MD; Uri Rosenschein, MD; Kalon K.L. Ho, MD; Peter B. Berger, MD; Richard Kuntz, MD; David
3/23/2017. Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate Europace Oct;14(10): Epub 2012 Aug 24.
 Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017 Explain the efficacy and safety of triple therapy, in regards to thromboembolic and bleeding risk Summarize the guideline recommendations
Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017 Explain the efficacy and safety of triple therapy, in regards to thromboembolic and bleeding risk Summarize the guideline recommendations
